Upgrading Pyrolytic Oil via Catalytic Co-Pyrolysis of Beechwood and Polystyrene
Abstract
1. Introduction
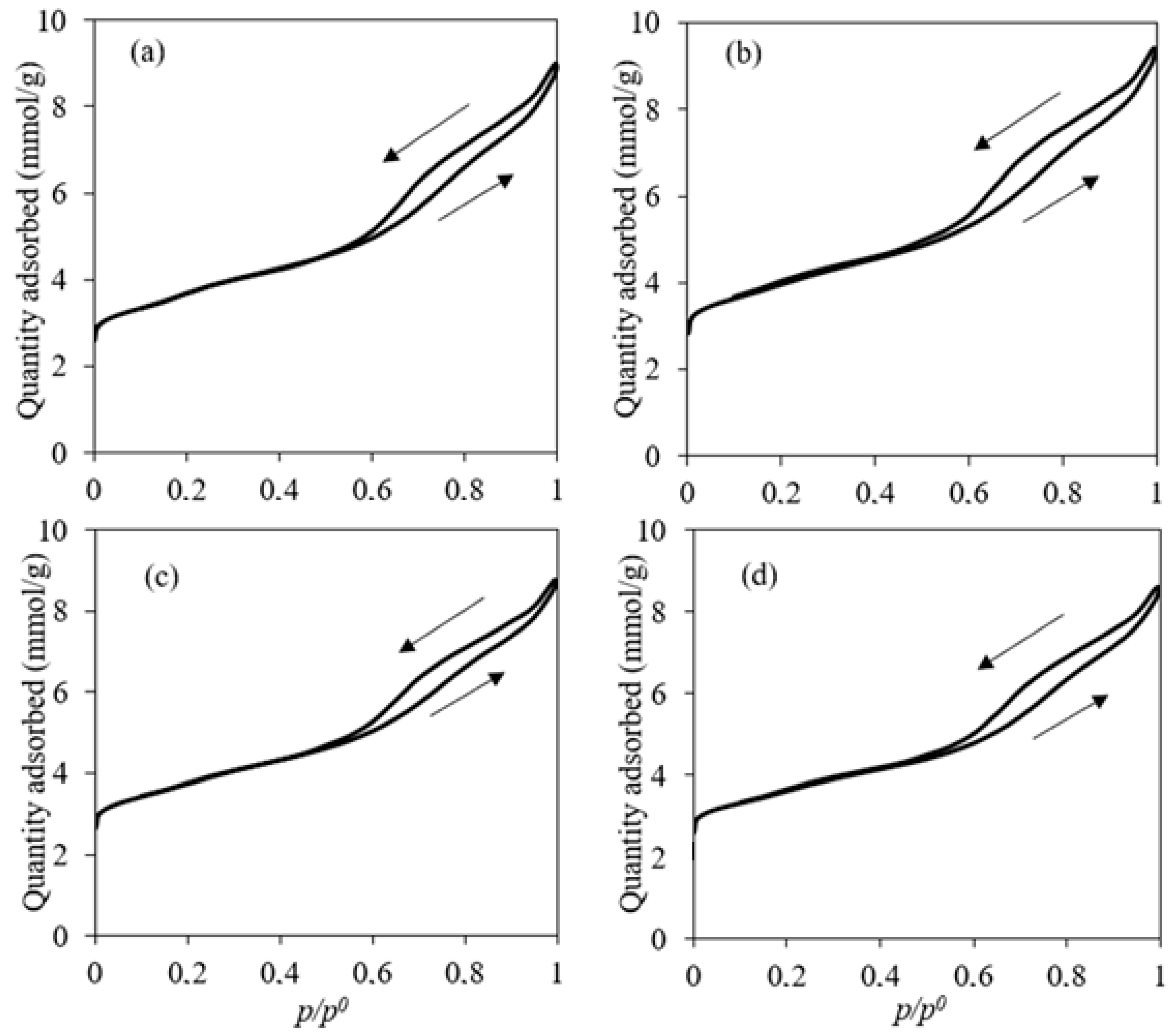
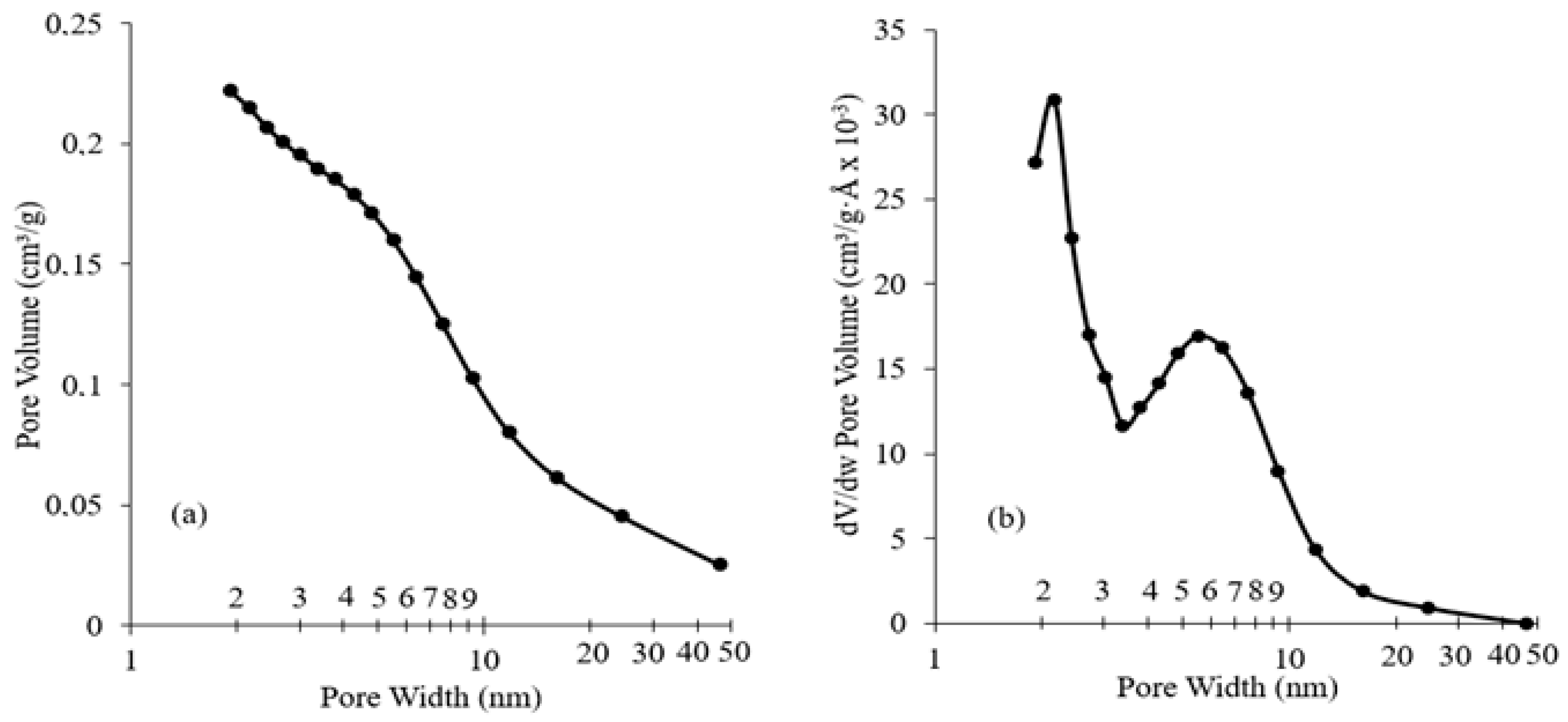
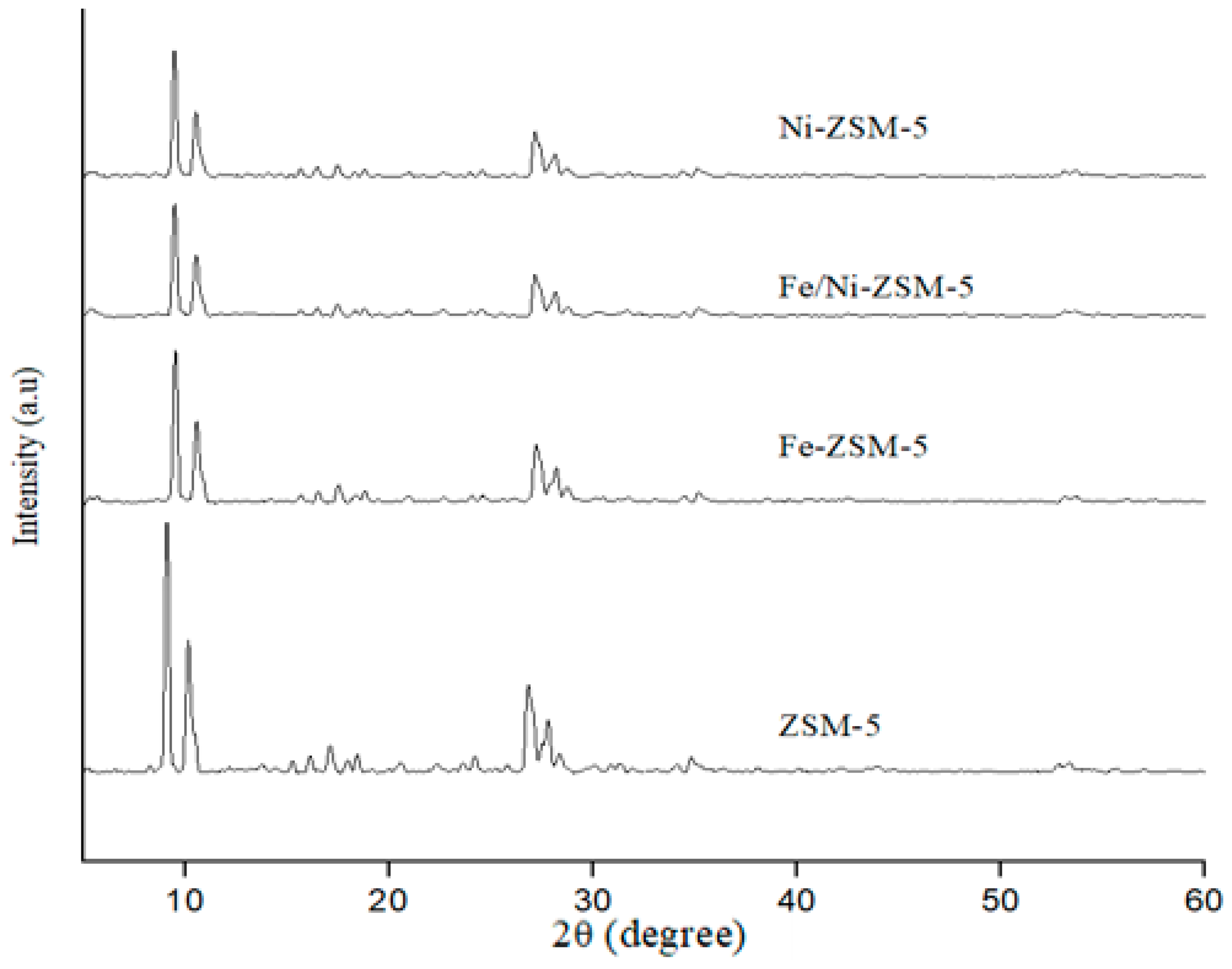
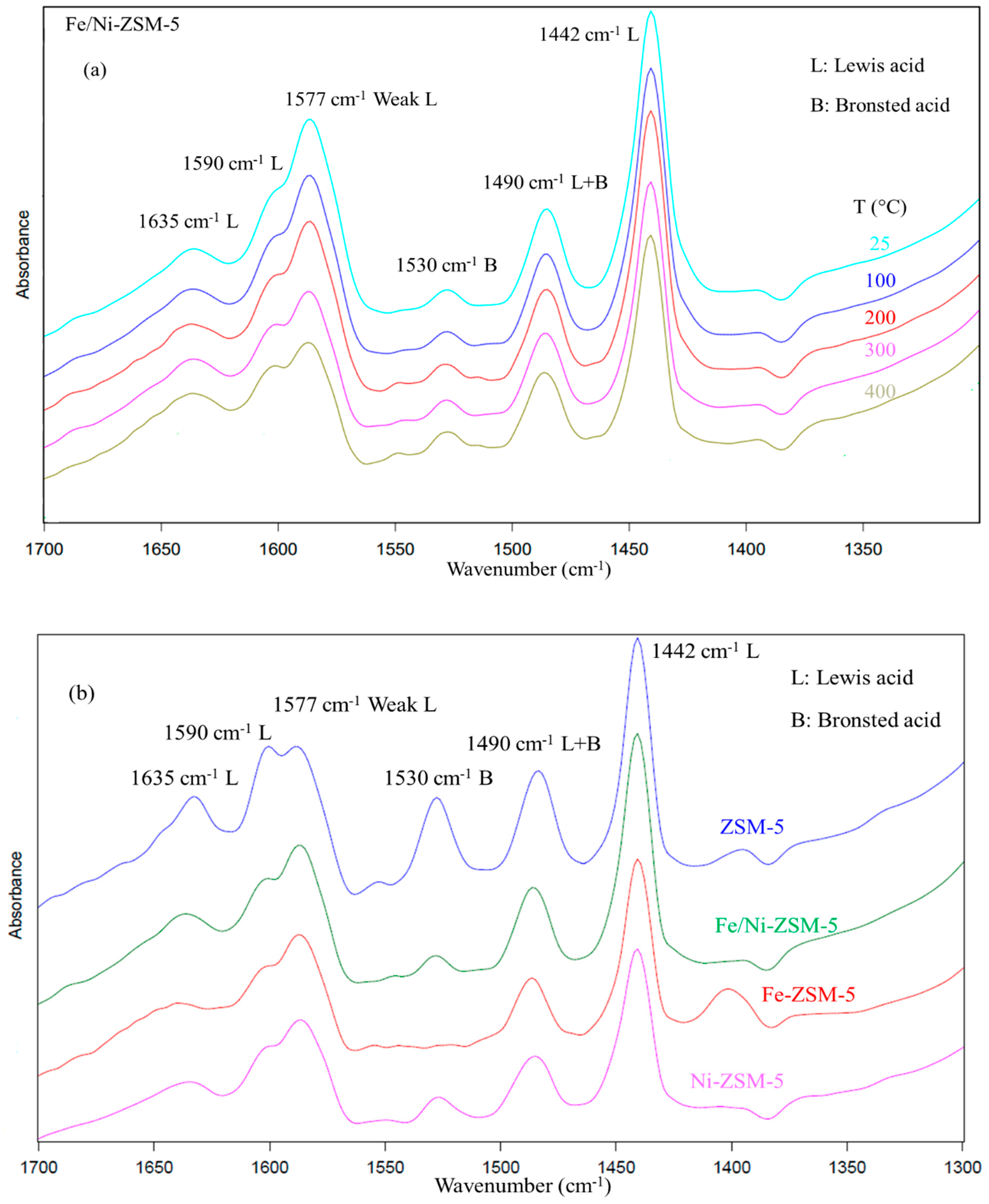
1.1. Catalyst Characterization
1.2. Catalytic Pyrolysis of Individual Biomass and Plastic
1.3. Catalytic Co-Pyrolysis of Biomass and Plastic
1.4. Catalytic Co-Pyrolysis under Fe/Ni-ZSM-5
2. Experimental Section
2.1. Materials
2.2. Catalyst Preparation
2.3. Catalyst Characterization
2.4. Experimental Methods
2.4.1. Pyrolysis Experimental Setup
2.4.2. GC-MS Analysis for Liquid Products
2.4.3. GC-FID Analysis for Liquid Products
2.4.4. GC-FID/TCD Analysis of Non-Condensable Gases
2.4.5. Lower Heating Value (LHV) and Synergism between Biomass and Plastic
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviations | Definition | |
| FID | Flame-ionization detector | |
| TCD | Thermal conductivity detector | |
| GC-MS | Gas chromatography–mass spectroscopy | |
| PS PAH | Polystyrene Polycyclic aromatic hydrocarbon | |
| ECN | Effective carbon number | |
| TGA | Thermogravimetric analysis | |
| n.a. | Not applicable | |
| Symbols | Symbol name | Unit |
| m | Mass | [g] |
| z | Charge number | |
| (m/z) | Mass-to-charge ratio | [g·mol−1] |
| wt.% | Weight percentage | [%] |
| LHV | Lower heating value | [MJ·kg−1] |
| HHV | Higher heating value | [MJ·kg−1] |
| r2 | Pearson’s coefficient of determination | |
| I.D./O.D. | Inner and outer diameter | [m] |
| a.u | Arbitrary unit | |
References
- Zhang, H.; Cheng, F.; Li, Y.; He, C.; Li, H.; Yang, S. Polymeric organophosphate-hafnium unconventional MOFs nanohybrids enable high-efficiency upgrading of biomass feedstocks via cascade catalytic transfer hydrogenation-dehydration. Ind. Crops Prod. 2022, 188, 115606. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, S.; Li, Z.; Zhang, H.; Li, H.; Yang, S. Green synthesis of heterogeneous polymeric bio-based acid decorated with hydrophobic regulator for efficient catalytic production of biodiesel at low temperatures. Fuel 2022, 329, 125467. [Google Scholar] [CrossRef]
- Abnisa, F. Co-pyrolysis of palm shell and polystyrene waste mixtures to synthesis liquid fuel. Fuel 2013, 108, 311–318. [Google Scholar] [CrossRef]
- Czernik, S.; Bridgwater, A.V. Overview of Applications of Biomass Fast Pyrolysis Oil. Energy Fuels 2004, 18, 590–598. [Google Scholar] [CrossRef]
- Zhang, X. Catalytic co-pyrolysis of lignocellulosic biomass with polymers: A critical review. Green Chem. 2016, 18, 4131–4314. [Google Scholar] [CrossRef]
- Roy, P.; Jahromi, H.; Rahman, T.; Baltrusaitis, J.; Hassan, E.B.; Torbert, A.; Adhikari, S. Hydrotreatment of pyrolysis bio-oil with non-edible carinata oil and poultry fat for producing transportation fuels. Fuel Process. Technol. 2023, 245, 107753. [Google Scholar] [CrossRef]
- Yan, W.; Acharjee, T.C.; Coronella, C.J.; Vásquez, V.R. Thermal pretreatment of lignocellulosic biomass. Environ. Prog. Sustain. Energy 2009, 28, 435–440. [Google Scholar] [CrossRef]
- Qi, Z. Review of biomass pyrolysis oil properties and upgrading research. Energy Convers. Manag. 2007, 48, 87–92. [Google Scholar]
- Abnisa, F.; Wan Daud, W.M.A. A review on co-pyrolysis of biomass: An optional technique to obtain a high-grade pyrolysis oil. Energy Convers. Manag. 2014, 87, 71–85. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Liu, C.; Wang, H.; Karim, A.M.; Sun, J.; Wang, Y. Catalytic fast pyrolysis of lignocellulosic biomass. Chem. Soc. Rev. 2014, 43, 7594–7623. [Google Scholar] [CrossRef] [PubMed]
- Ruddy, D.A.; Schaidle, J.A.; Iii, J.R.F.; Wang, J.; Moens, L.; Hensley, J.E. Recent advances in heterogeneous catalysts for bio-oil upgrading via “ex situ catalytic fast pyrolysis”: Catalyst development through the study of model compounds. Green Chem. 2014, 16, 454–490. [Google Scholar] [CrossRef]
- Iliopoulou, E.F.; Stefanidis, S.; Kalogiannis, K.; Psarras, A.C.; Delimitis, A.; Triantafyllidis, K.S.; Lappas, A.A. Pilot-scale validation of Co-ZSM-5 catalyst performance in the catalytic upgrading of biomass pyrolysis vapours. Green Chem. 2014, 16, 662–674. [Google Scholar] [CrossRef]
- Carlson, T.R.; Cheng, Y.-T.; Jae, J.; Huber, G.W. Production of green aromatics and olefins by catalytic fast pyrolysis of wood sawdust. Energy Environ. Sci. 2011, 4, 145–161. [Google Scholar] [CrossRef]
- Zhang, H.; Carlson, T.R.; Xiao, R.; Huber, G.W. Catalytic fast pyrolysis of wood and alcohol mixtures in a fluidized bed reactor. Green Chem. 2012, 14, 98–110. [Google Scholar] [CrossRef]
- Mohabeer, C.; Reyes, L.; Abdelouahed, L.; Marcotte, S.; Taouk, B. Investigating catalytic de-oxygenation of cellulose, xylan and lignin bio-oils using HZSM-5 and Fe-HZSM-5. J. Anal. Appl. Pyrolysis 2019, 137, 118–127. [Google Scholar] [CrossRef]
- Zhang, H.; Nie, J.; Xiao, R.; Jin, B.; Dong, C.; Xiao, G. Catalytic Co-pyrolysis of Biomass and Different Plastics (Polyethylene, Polypropylene, and Polystyrene) To Improve Hydrocarbon Yield in a Fluidized-Bed Reactor. Energy Fuels 2014, 28, 1940–1947. [Google Scholar] [CrossRef]
- Wang, K.; Kim, K.H.; Brown, R.C. Catalytic pyrolysis of individual components of lignocellulosic biomass. Green Chem. 2014, 16, 727–735. [Google Scholar] [CrossRef]
- Choi, S.J.; Park, S.H.; Jeon, J.-K.; Lee, I.G.; Ryu, C.; Suh, D.J.; Park, Y.-K. Catalytic conversion of particle board over microporous catalysts. Renew. Energy 2013, 54, 105–110. [Google Scholar] [CrossRef]
- Wang, Z.; Burra, K.G.; Lei, T.; Gupta, A.K. Co-pyrolysis of waste plastic and solid biomass for synergistic production of biofuels and chemicals-A review. Prog. Energy Combust. Sci. 2021, 84, 100899. [Google Scholar] [CrossRef]
- Zhang, B.; Zhong, Z.; Ding, K.; Song, Z. Production of aromatic hydrocarbons from catalytic co-pyrolysis of biomass and high density polyethylene: Analytical Py–GC/MS study. Fuel 2015, 139, 622–628. [Google Scholar] [CrossRef]
- Xue, Y.; Kelkar, A.; Bai, X. Catalytic co-pyrolysis of biomass and polyethylene in a tandem micropyrolyzer. Fuel 2016, 166, 227–236. [Google Scholar] [CrossRef]
- Vo, T.A.; Tran, Q.K.; Ly, H.V.; Kwon, B.; Hwang, H.T.; Kim, J.; Kim, S.-S. Co-pyrolysis of lignocellulosic biomass and plastics: A comprehensive study on pyrolysis kinetics and characteristics. J. Anal. Appl. Pyrolysis 2022, 163, 105464. [Google Scholar] [CrossRef]
- Engamba Esso, S.B.; Xiong, Z.; Chaiwat, W.; Kamara, M.F.; Longfei, X.; Xu, J.; Ebako, J.; Jiang, L.; Su, S.; Hu, S.; et al. Review on synergistic effects during co-pyrolysis of biomass and plastic waste: Significance of operating conditions and interaction mechanism. Biomass Bioenergy 2022, 159, 106415. [Google Scholar] [CrossRef]
- Anuar Sharuddin, S.D.; Abnisa, F.; Wan Daud, W.M.A.; Aroua, M.K. A review on pyrolysis of plastic wastes. Energy Convers. Manag. 2016, 115, 308–326. [Google Scholar] [CrossRef]
- Artetxe, M.; Lopez, G.; Amutio, M.; Barbarias, I.; Arregi, A.; Aguado, R.; Bilbao, J.; Olazar, M. Styrene recovery from polystyrene by flash pyrolysis in a conical spouted bed reactor. Waste Manag. 2015, 45, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Jaafar, Y.; Abdelouahed, L.; Hage, R.E.; Samrani, A.E.; Taouk, B. Pyrolysis of common plastics and their mixtures to produce valuable petroleum-like products. Polym. Degrad. Stab. 2022, 195, 109770. [Google Scholar] [CrossRef]
- Mullen, C.A.; Boateng, A.A. Production of Aromatic Hydrocarbons via Catalytic Pyrolysis of Biomass over Fe-Modified HZSM-5 Zeolites. ACS Sustain. Chem. Eng. 2015, 3, 1623–1631. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, X.; Chen, L.; Zhao, B.; Yang, S.; Xie, X. Comparision of catalytic fast pyrolysis of biomass to aromatic hydrocarbons over ZSM-5 and Fe/ZSM-5 catalysts. J. Anal. Appl. Pyrolysis 2016, 121, 342–346. [Google Scholar] [CrossRef]
- Matovic, M.D. Biomass Now: Cultivation and Utilization; BoD—Books on Demand: Norderstedt, Germany, 2013. [Google Scholar]
- Yao, W.; Li, J.; Feng, Y.; Wang, W.; Zhang, X.; Chen, Q.; Komarneni, S.; Wang, Y. Thermally stable phosphorus and nickel modified ZSM-5 zeolites for catalytic co-pyrolysis of biomass and plastics. RSC Adv. 2015, 5, 30485–30494. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, Z.; Sun, J.; Guo, W.; Wang, Q. Effects of phosphorus-modified HZSM-5 on distribution of hydrocarbon compounds from wood–plastic composite pyrolysis using Py-GC/MS. J. Anal. Appl. Pyrolysis 2015, 116, 223–230. [Google Scholar] [CrossRef]
- Li, J.; Yu, Y.; Li, X.; Wang, W.; Yu, G.; Deng, S.; Huang, J.; Wang, B.; Wang, Y. Maximizing carbon efficiency of petrochemical production from catalytic co-pyrolysis of biomass and plastics using gallium-containing MFI zeolites. Appl. Catal. B Environ. 2015, 172–173, 154–164. [Google Scholar] [CrossRef]
- Li, X.; Li, J.; Zhou, G.; Feng, Y.; Wang, Y.; Yu, G.; Deng, S.; Huang, J.; Wang, B. Enhancing the production of renewable petrochemicals by co-feeding of biomass with plastics in catalytic fast pyrolysis with ZSM-5 zeolites. Appl. Catal. A Gen. 2014, 481, 173–182. [Google Scholar] [CrossRef]
- Dorado, C.; Mullen, C.A.; Boateng, A.A. H-ZSM5 Catalyzed Co-Pyrolysis of Biomass and Plastics. ACS Sustain. Chem. Eng. 2014, 2, 301–311. [Google Scholar] [CrossRef]
- Hassan, E.B.; Elsayed, I.; Eseyin, A. Production high yields of aromatic hydrocarbons through catalytic fast pyrolysis of torrefied wood and polystyrene. Fuel 2016, 174, 317–324. [Google Scholar] [CrossRef]
- Mohabeer, C.; Reyes, L.; Abdelouahed, L.; Marcotte, S.; Buvat, J.-C.; Tidahy, L.; Abi-Aad, E.; Taouk, B. Production of liquid bio-fuel from catalytic de-oxygenation: Pyrolysis of beech wood and flax shives. J. Fuel Chem. Technol. 2019, 47, 153–166. [Google Scholar] [CrossRef]
- Valle, B.; Gayubo, A.G.; Aguayo, A.T.; Olazar, M.; Bilbao, J. Selective Production of Aromatics by Crude Bio-oil Valorization with a Nickel-Modified HZSM-5 Zeolite Catalyst. Energy Fuels 2010, 24, 2060–2070. [Google Scholar] [CrossRef]
- Ding, Y.-L.; Wang, H.-Q.; Xiang, M.; Yu, P.; Li, R.-Q.; Ke, Q.-P. The Effect of Ni-ZSM-5 Catalysts on Catalytic Pyrolysis and Hydro-Pyrolysis of Biomass. Front. Chem. 2020, 8, 790. [Google Scholar] [CrossRef]
- Klein, A.; Bockhorn, O.; Mayer, K.; Grabner, M. Central European wood species: Characterization using old knowledge. J. Wood Sci. 2016, 62, 194–202. [Google Scholar] [CrossRef]
- Li, X.; Dong, W.; Zhang, J.; Shao, S.; Cai, Y. Preparation of bio-oil derived from catalytic upgrading of biomass vacuum pyrolysis vapor over metal-loaded HZSM-5 zeolites. J. Energy Inst. 2020, 93, 605–613. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Botas, J.A.; Serrano, D.P.; García, A.; Ramos, R. Catalytic conversion of rapeseed oil for the production of raw chemicals, fuels and carbon nanotubes over Ni-modified nanocrystalline and hierarchical ZSM-5. Appl. Catal. B Environ. 2014, 145, 205–215. [Google Scholar] [CrossRef]
- Zhu, L.; Lv, X.; Tong, S.; Zhang, T.; Song, Y.; Wang, Y.; Hao, Z.; Huang, C.; Xia, D. Modification of zeolite by metal and adsorption desulfurization of organic sulfide in natural gas. J. Nat. Gas Sci. Eng. 2019, 69, 102941. [Google Scholar] [CrossRef]
- Topaloǧlu Yazıcı, D.; Bilgiç, C. Determining the surface acidic properties of solid catalysts by amine titration using Hammett indicators and FTIR-pyridine adsorption methods: Determining the surface acidic properties of solid catalysts. Surf. Interface Anal. 2010, 42, 959–962. [Google Scholar] [CrossRef]
- Srivastava, R.; Srinivas, D.; Ratnasamy, P. Sites for CO2 activation over amine-functionalized mesoporous Ti(Al)-SBA-15 catalysts. Microporous Mesoporous Mater. 2006, 90, 314–326. [Google Scholar] [CrossRef]
- Ward, J.W. Thermal decomposition of ammonium Y zeolite. J. Catal. 1970, 18, 348–351. [Google Scholar] [CrossRef]
- Ferretto, L.; Glisenti, A. Surface Acidity and Basicity of a Rutile Powder. Chem. Mater. 2003, 15, 1181–1188. [Google Scholar] [CrossRef]
- Mohabeer, C.; Abdelouahed, L.; Marcotte, S.; Taouk, B. Comparative analysis of pyrolytic liquid products of beech wood, flax shives and woody biomass components. J. Anal. Appl. Pyrolysis 2017, 127, 269–277. [Google Scholar] [CrossRef]
- Gucho, E.M.; Shahzad, K.; Bramer, E.A.; Akhtar, N.A.; Brem, G. Experimental Study on Dry Torrefaction of Beech Wood and Miscanthus. Energies 2015, 8, 3903–3923. [Google Scholar] [CrossRef]
- Zhao, C.; Jiang, E.; Chen, A. Volatile production from pyrolysis of cellulose, hemicellulose and lignin. J. Energy Inst. 2017, 90, 902–913. [Google Scholar] [CrossRef]
- Mihalcik, D.J.; Mullen, C.A.; Boateng, A.A. Screening acidic zeolites for catalytic fast pyrolysis of biomass and its components. J. Anal. Appl. Pyrolysis 2011, 92, 224–232. [Google Scholar] [CrossRef]
- Cheng, Y.-T.; Huber, G.W. Production of targeted aromatics by using Diels–Alder classes of reactions with furans and olefins over ZSM-5. Green Chem. 2012, 14, 3114. [Google Scholar] [CrossRef]
- Dorado, C.; Mullen, C.A.; Boateng, A.A. Origin of carbon in aromatic and olefin products derived from HZSM-5 catalyzed co-pyrolysis of cellulose and plastics via isotopic labeling. Appl. Catal. B Environ. 2015, 162, 338–345. [Google Scholar] [CrossRef]
- Dyer, A.C.; Nahil, M.A.; Williams, P.T. Catalytic co-pyrolysis of biomass and waste plastics as a route to upgraded bio-oil. J. Energy Inst. 2021, 97, 27–36. [Google Scholar] [CrossRef]
- Aitani, A.M. Oil Refining and Products. In Encyclopedia of Energy; Cleveland, C.J., Ed.; Elsevier: New York, NY, USA, 2004; pp. 715–729. [Google Scholar] [CrossRef]
- Kamrin, M.A. Gasoline. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Academic Press: Oxford, UK, 2014; pp. 700–701. [Google Scholar] [CrossRef]
- Scanlon, J.T.; Willis, D.E. Calculation of Flame Ionization Detector Relative Response Factors Using the Effective Carbon Number Concept. J. Chromatogr. Sci. 1985, 23, 333–340. [Google Scholar] [CrossRef]
- Marlair, G.; Cwiklinski, C.; Tewarson, A. An analysis of some practical methods for estimating heats of combustion in fire safety studies. In Interflam 99; ineris-00972167; Ministry of Higher Education and Research: Edimbourg, UK, June 1999. [Google Scholar]

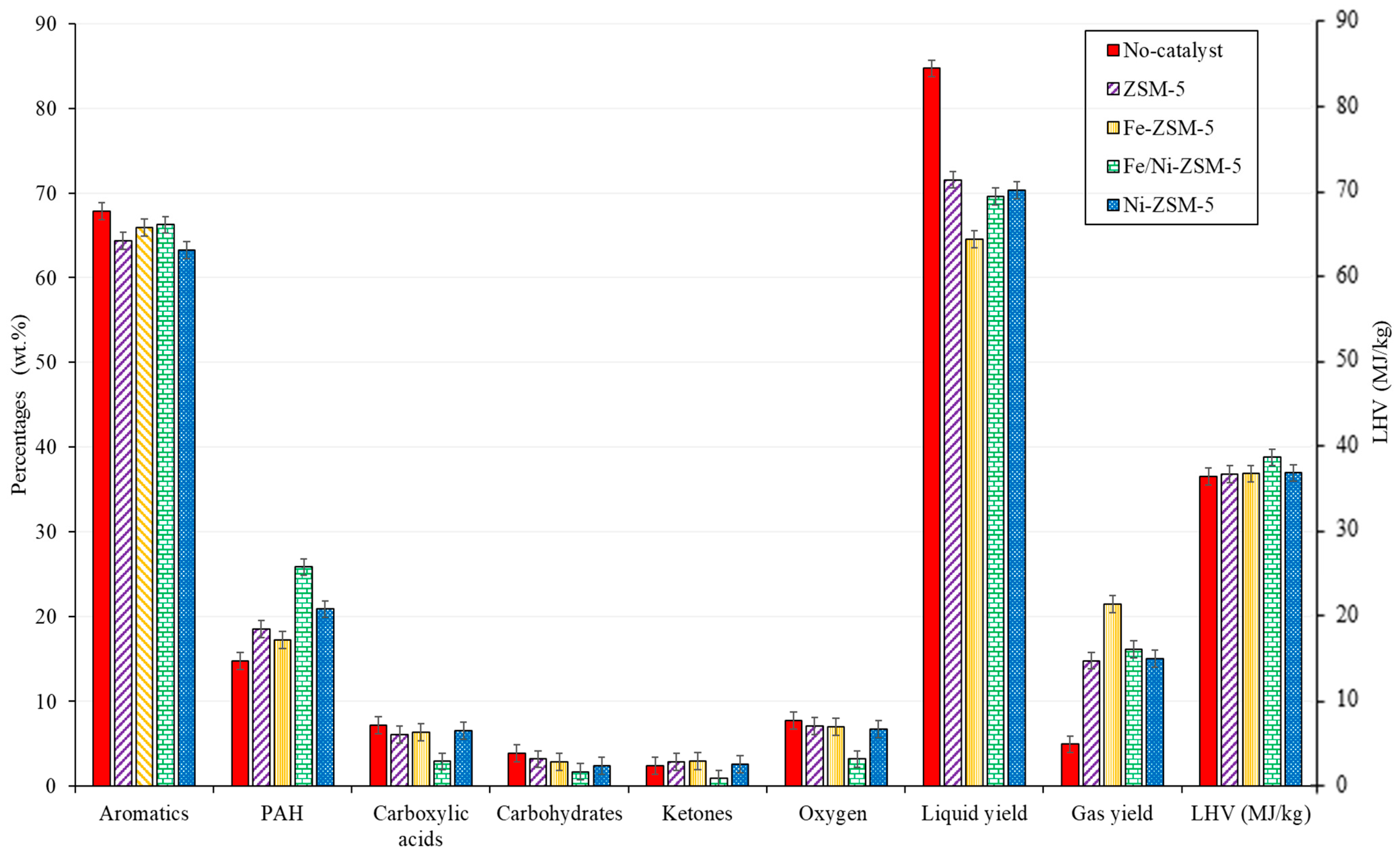
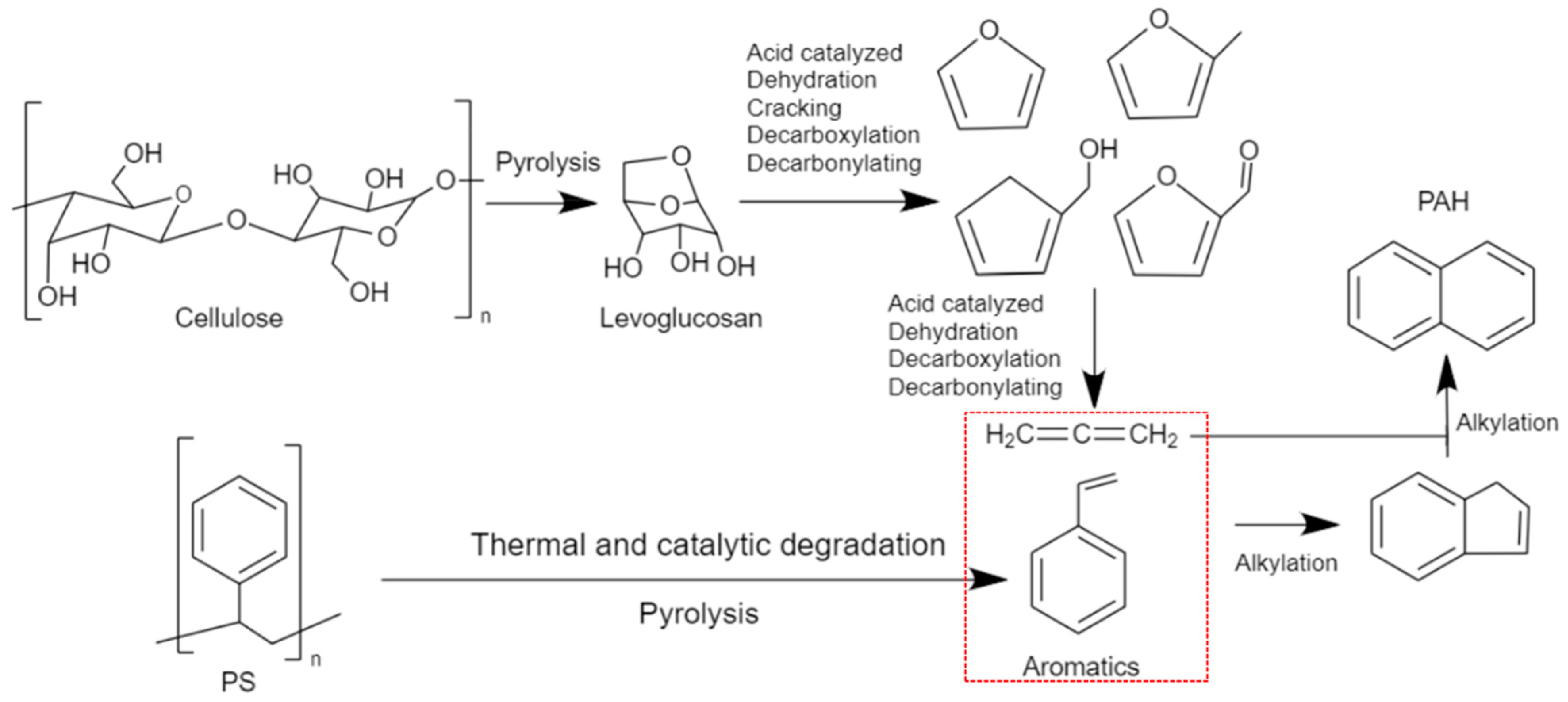



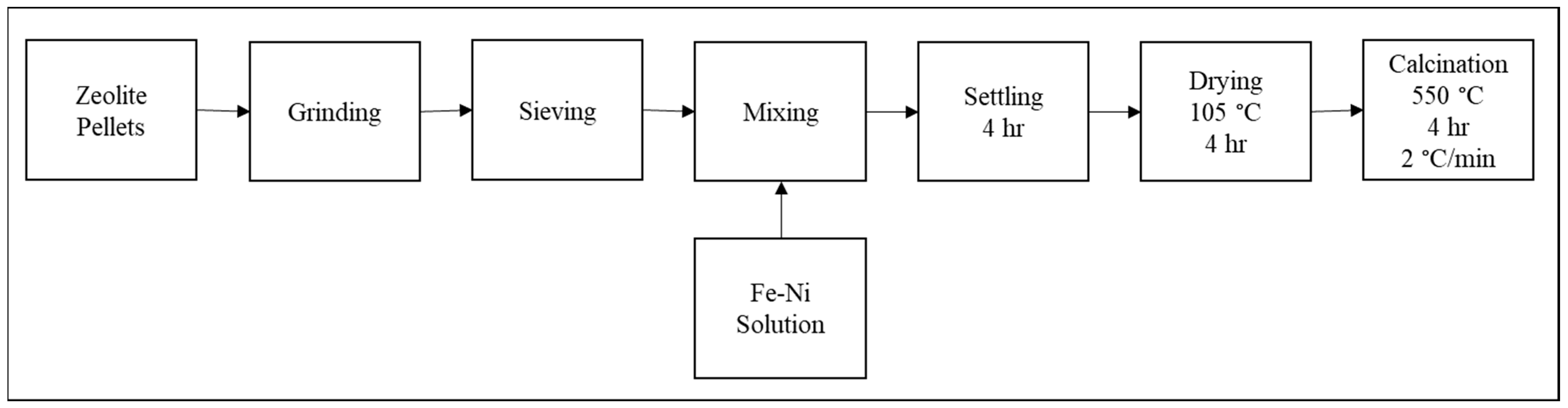
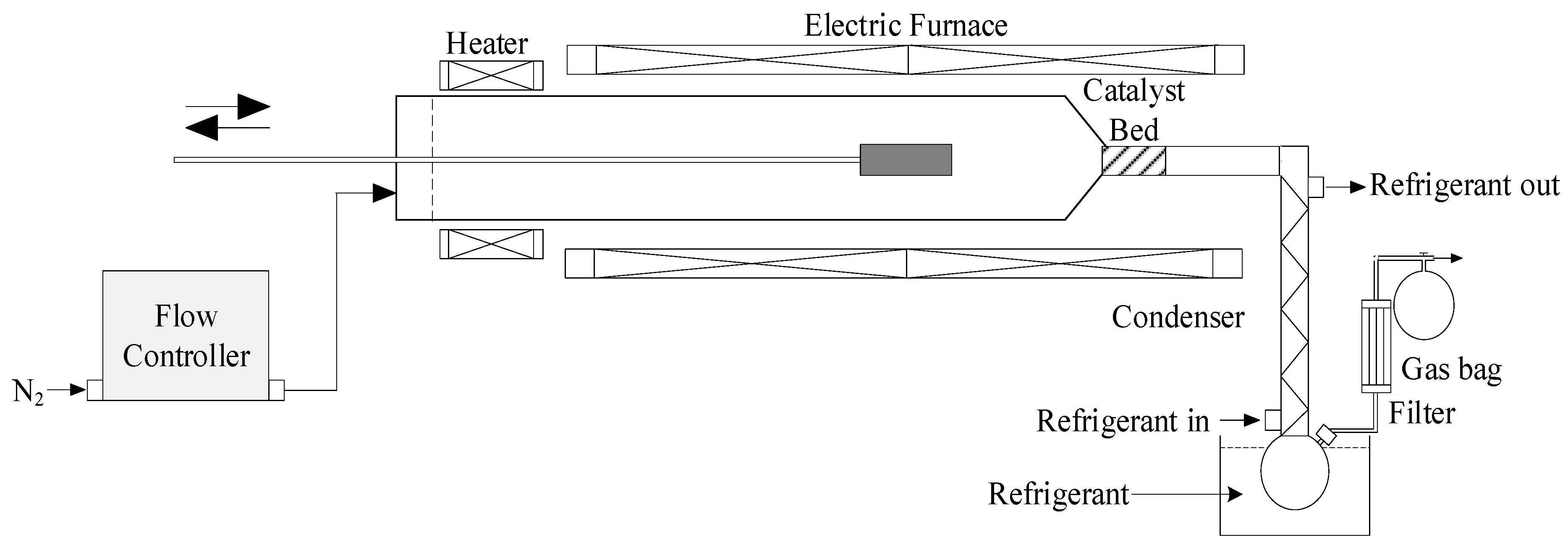
| ZSM-5 | Fe-ZSM-5 | Fe/Ni-ZSM-5 | Ni-ZSM-5 | |
|---|---|---|---|---|
| SiO2/Al2O3 | 38.0 | 38.4 | 38.9 | 38.5 |
| Fe (wt.%) a | 0.04 | 1.46 | 1.32 | 0.04 |
| Ni (wt.%) a | - | - | 1.22 | 1.21 |
| BET surface area (m2/g) b | 282.0 | 299.1 | 284.9 | 274.0 |
| Micropore surface area (m2/g) c | 109.5 | 135.5 | 123.8 | 126.8 |
| External surface area (m2/g) c | 172.5 | 163.6 | 161.1 | 147.3 |
| Specific pore volume (cm3/g) c | 0.24 | 0.24 | 0.23 | 0.22 |
| Micropore volume (cm3/g) c | 0.05 | 0.06 | 0.06 | 0.06 |
| RT (min) * | Chemical Family | Reference Compound |
|---|---|---|
| 6.63 | Furans | Furan |
| 11.15 | Carboxylic acids | Acetic acid |
| 12.13 | Aromatics | Toluene |
| 18.39 | Esters & Ethers | Allyl butyrate |
| 19.52 | Aldehydes | Furfural |
| 21.63 | Ketones | 2-methyl-2-cyclopenten-1-one |
| 31.82 | Phenols | p-cresol |
| 40.76 | PAH | Bibenzyl |
| 41.74 | Guaiacols | 4-methylcatechol |
| 42.74 | Nitrogenates | Benzamide |
| 49.58 | Carbohydrates | Levoglucosan |
| Concentrations (wt.%) | |||||
|---|---|---|---|---|---|
| No Catalyst | ZSM-5 | Fe-ZSM-5 | Fe/Ni-ZSM-5 | Ni-ZSM-5 | |
| Carboxylic acids | 35.3 | 35.7 | 33.5 | 36.1 | 35.3 |
| Ester | 5.8 | 4.5 | 4.4 | 4.7 | 4.4 |
| Ether | 0.7 | 1.0 | 0.9 | 0.9 | 0.9 |
| Nitrogenates | 4.4 | 3.5 | 3.8 | 4.3 | 4.1 |
| Ketones | 15.0 | 14.0 | 14.0 | 13.8 | 14.7 |
| Furans | 9.8 | 8.8 | 8.5 | 8.8 | 9.6 |
| Aldehydes | 2.4 | 0.5 | 0.5 | 0.7 | 0.8 |
| Carbohydrates | 17.3 | 9.5 | 11.7 | 12.5 | 12.4 |
| Phenols | 2.8 | 7.7 | 8.0 | 5.0 | 3.9 |
| Guaiacols | 5.2 | 6.3 | 7.0 | 6.6 | 6.9 |
| Aromatics | - | 2.8 | 2.6 | 2.6 | 2.7 |
| PAH | - | 3.9 | 3.3 | 2.3 | 2.1 |
| Oxygen content | 41.3 | 36.4 | 37.3 | 39.0 | 38.9 |
| LHV (MJ/kg) | 18.4 | 20.4 | 20.3 | 19.4 | 19.4 |
| Liquid yield | 65.0 | 56.2 | 56.0 | 47.9 | 56.3 |
| Char yield | 20.0 | 21.3 | 20.7 | 21.3 | 20.0 |
| Coke | - | 3.0 | 3.3 | 3.3 | 2.3 |
| Gaz yield | 15.0 | 19.5 | 20.0 | 27.5 | 21.4 |
| Concentrations (wt.%) | |||||
|---|---|---|---|---|---|
| No catalyst | ZSM-5 | Fe-ZSM-5 | Fe/Ni-ZSM-5 | Ni-ZSM-5 | |
| Aromatics | 76.7 | 82.2 | 77.7 | 81.7 | 77.8 |
| PAH | 23.3 | 17.8 | 22.3 | 18.3 | 22.2 |
| LHV (MJ/kg) | 40.6 | 40.6 | 40.0 | 40.6 | 40.5 |
| Liquid yield | 99.9 | 96.2 | 96.1 | 96.6 | 96.3 |
| Char yield | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Coke | 0.0 | 3.3 | 3.1 | 3.0 | 3.3 |
| Gaz yield | 0.1 | 0.5 | 0.8 | 0.4 | 0.4 |
| Name | LHV (MJ/kg) | LHV Change (%) | O% (wt.%) | O% Change (%) | Ref. |
|---|---|---|---|---|---|
| BW (No cat.) | 18.4 | n.a | 41.3 | n.a | This work |
| BW (ZSM-5) | 20.4 | +10.9 a | 36.4 | −11.9 a | |
| BW-PS 50-50 (No cat.) | 36.5 | +98.4 | 7.8 | −81.1 | |
| BW-PS 50-50 (Fe/Ni-ZSM-5) | 38.8 | +110.9 | 3.2 | −92.3 | |
| BW-PS 75-25 (No cat.) | 30.0 | +63.0 | 19.6 | −52.5 | |
| BW-PS 75-25 (Fe/Ni-ZSM-5) | 35.8 | +94.6 | 8.8 | −78.7 | |
| PS | 40.6 | n.a | - | n.a | |
| Gasoline | 43.4–46.5 | ~2.7 | [40,41,42] | ||
| Kerosene | 43.0–46.2 | n.a. | |||
| Diesel | 42.8–45.8 | ~1.8 | |||
| Chemical Families | Operating Temperature (°C) ± 5 °C | |||
|---|---|---|---|---|
| 450 | 500 | 550 | 600 | |
| Carboxylic acids | 3.6 | 2.9 | 4.1 | 5.6 |
| Ketones | 1.3 | 0.9 | 1.5 | 2.1 |
| Furans | 0.7 | 0.8 | 1.2 | 1.5 |
| Carbohydrates | 1.3 | 1.7 | 2.7 | 3.0 |
| Aromatics | 68.9 | 66.2 | 68.4 | 74.0 |
| PAH | 22.5 | 25.8 | 20.3 | 12.2 |
| Gas yield | 14.5 | 16.1 | 17.9 | 23.0 |
| Liquid yield | 70.2 | 69.9 | 66.1 | 61.0 |
| Char yield | 12.7 | 10.3 | 10.0 | 9.0 |
| Coke | 2.7 | 3.6 | 6.0 | 7.0 |
| O% | 3.6 | 3.2 | 4.7 | 6.0 |
| LHV * | 38.7 | 38.8 | 38.0 | 37.5 |
| Name | Ultimate Analysis | Proximate Analysis | |||||
|---|---|---|---|---|---|---|---|
| % C | % H | % O * | Moisture | Fixed Carbon | Volatile | Ash | |
| PS | 92.6 | 7.4 | - | - | 0.3 | 99.7 | - |
| BW | 47.4 | 6.1 | 46.5 | 5.7 | 17.5 | 75.9 | 0.9 |
| Reaction | Catalyst | BW-PS Percentage (wt.%) | Temperature (°C) |
|---|---|---|---|
| Biomass pyrolysis | No catalyst | 500 | |
| ZSM-5 | |||
| Fe-ZSM-5 | 100-0 | ||
| Fe/Ni-ZSM-5 | |||
| Ni-ZSM-5 | |||
| Plastic pyrolysis | No catalyst | 500 | |
| ZSM-5 | |||
| Fe-ZSM-5 | 0-100 | ||
| Fe/Ni-ZSM-5 | |||
| Ni-ZSM-5 | |||
| Co-pyrolysis | No catalyst | 25-50-75 | 500 |
| No catalyst | 50 | 450-500-550-600 | |
| ZSM-5 | 50 | 500 | |
| Fe-ZSM-5 | 50 | ||
| Fe/Ni-ZSM-5 | 25-50-75 | ||
| Ni-ZSM-5 | 50 | ||
| Fe/Ni-ZSM-5 | 50 | 450-500-550-600 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaafar, Y.; Ramirez, G.C.A.; Abdelouahed, L.; El Samrani, A.; El Hage, R.; Taouk, B. Upgrading Pyrolytic Oil via Catalytic Co-Pyrolysis of Beechwood and Polystyrene. Molecules 2023, 28, 5758. https://doi.org/10.3390/molecules28155758
Jaafar Y, Ramirez GCA, Abdelouahed L, El Samrani A, El Hage R, Taouk B. Upgrading Pyrolytic Oil via Catalytic Co-Pyrolysis of Beechwood and Polystyrene. Molecules. 2023; 28(15):5758. https://doi.org/10.3390/molecules28155758
Chicago/Turabian StyleJaafar, Yehya, Gian Carlos Arias Ramirez, Lokmane Abdelouahed, Antoine El Samrani, Roland El Hage, and Bechara Taouk. 2023. "Upgrading Pyrolytic Oil via Catalytic Co-Pyrolysis of Beechwood and Polystyrene" Molecules 28, no. 15: 5758. https://doi.org/10.3390/molecules28155758
APA StyleJaafar, Y., Ramirez, G. C. A., Abdelouahed, L., El Samrani, A., El Hage, R., & Taouk, B. (2023). Upgrading Pyrolytic Oil via Catalytic Co-Pyrolysis of Beechwood and Polystyrene. Molecules, 28(15), 5758. https://doi.org/10.3390/molecules28155758








