Recent Progress on Fluorescent Probes in Heavy Metal Determinations for Food Safety: A Review
Abstract
1. Introduction
2. Common Fluorescence Spectroscopy Detection Methods
| Method | Analytes | LOD | Sample | Ref. |
|---|---|---|---|---|
| ICP-MS | As, Pb, Cd, etc. | 0.0003–2.47 mg·kg−1 | peanuts | [30] |
| HPLC-ICP-MS | As | 1.12 μg·kg−1 | salted foods, sea salt | [31] |
| LA-ICP-MS | Hg | 0.006 µg·g−1 | mushrooms | [32] |
| Se | 0.3 µg·g−1 | |||
| GF-ETV-ICP-MS | Se | 0.5 µg·kg−1 | rice | [33] |
| Cd | 0.16 µg·kg−1 | |||
| FI-SPE-ICP-MS | Cd, Hg, Pb, etc. | 0.8 ng·L−1–0.09 μg·L−1 | rice | [34] |
| SPE-HPLC-AFS | Hg2+, MeHg, etc. | 0.001–0.002 µg·L−1 | water | [35] |
| ICP-AES | Hg | parts-per-trillion (ng·L−1) | cannabinoid-based products | [36] |
| ICP-MS-AES | Ag, As, Pb, etc. | below 3 mg·kg | turmeric | [37] |
| ICP-AES | Cd, As, Cu, etc. | 0.008, 0.017, 0.0006, etc. (μg·L−1) | wheat and flour products | [38] |
| ICP-MS/AAS | Mn, Cr, As, etc. | 0.1–23.2 mg·kg−1 | honey | [39] |
| ICP-AES | Hg, As, Cd, etc. | 1.80 × 10−5–2.17 × 10−3 mg·kg−1 | milk powder | [40] |
| EcHG-AFS | Cd | 0.05 ng·mL−1 | drinking water | [41] |
| CVG-NDAFS | As | 0.051 ng·mL−1 | Chinese herbal foods | [42] |
| Sb | 0.034 ng·mL−1 | |||
| Se | 0.050 ng·mL−1 | |||
| Hg | 0.0058 ng·mL−1 | |||
| SS-ETV-AAS | Cd | 70 pg·g−1 | chocolate | [43] |
| TXRF | Mn, Ni, Rb, etc. | 0.25–0.50 mg·kg−1 | herbal infusion teas | [45] |
| HDXRF | As, Cd, Pb, etc. | 0.072, 0.502, 0.063, etc. (mg·kg−1) | scallops | [46] |
| pXRF | Cr, Ni, As | 20 mg·kg−1 | fertilizers | [47] |
| pXRF-ICP-MS | Pb | 2 mg·kg−1 | spices | [48] |
| NIR-LIBS | Zn, Cu, Pb | 4.3047, 4.9592, 8.3881 (mg·kg−1) | lilies | [50] |
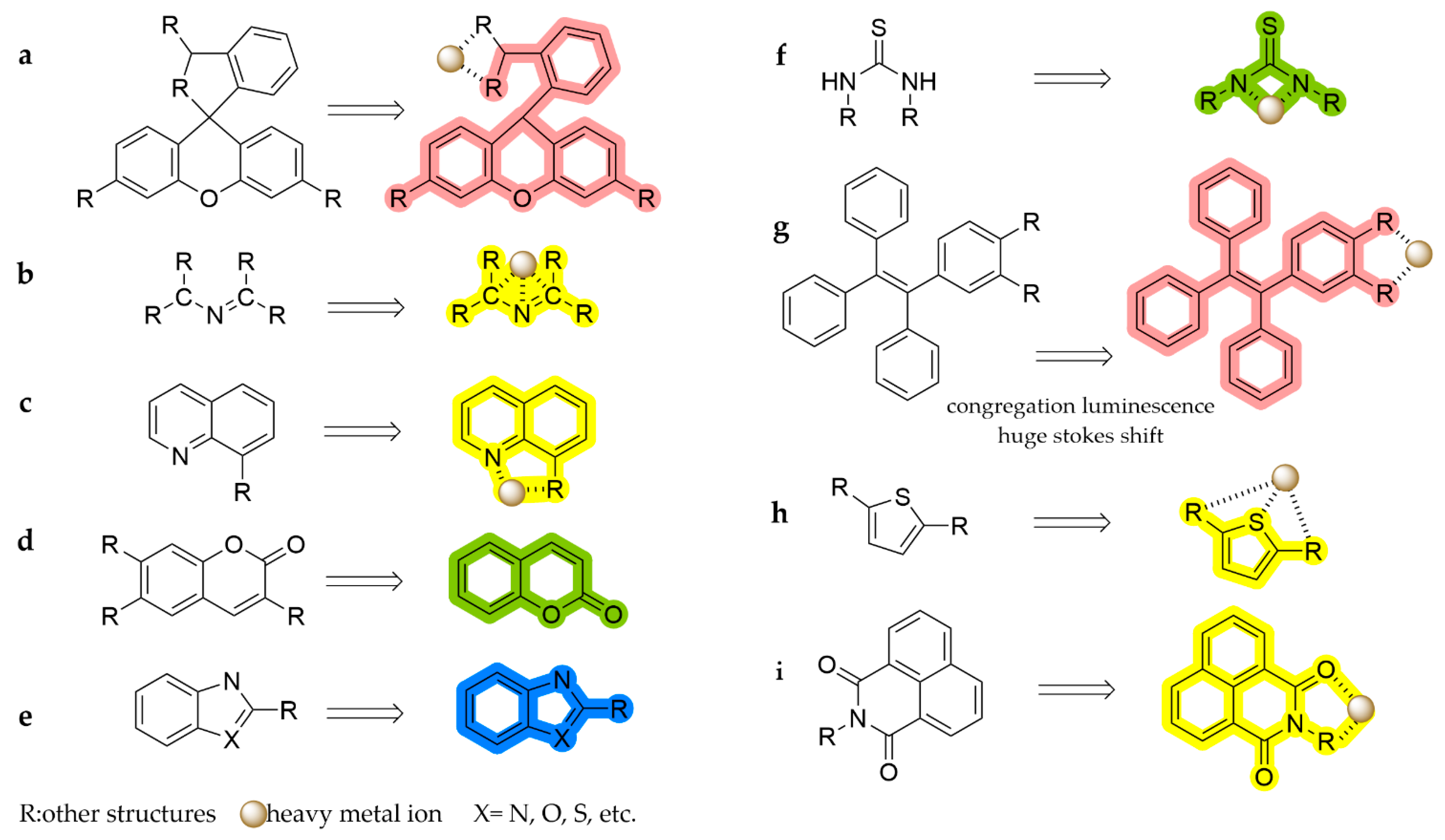
3. Spectroscopic Detection Methods Based on Fluorescent Probes
3.1. Rhodamine-Based Fluorescent Probes
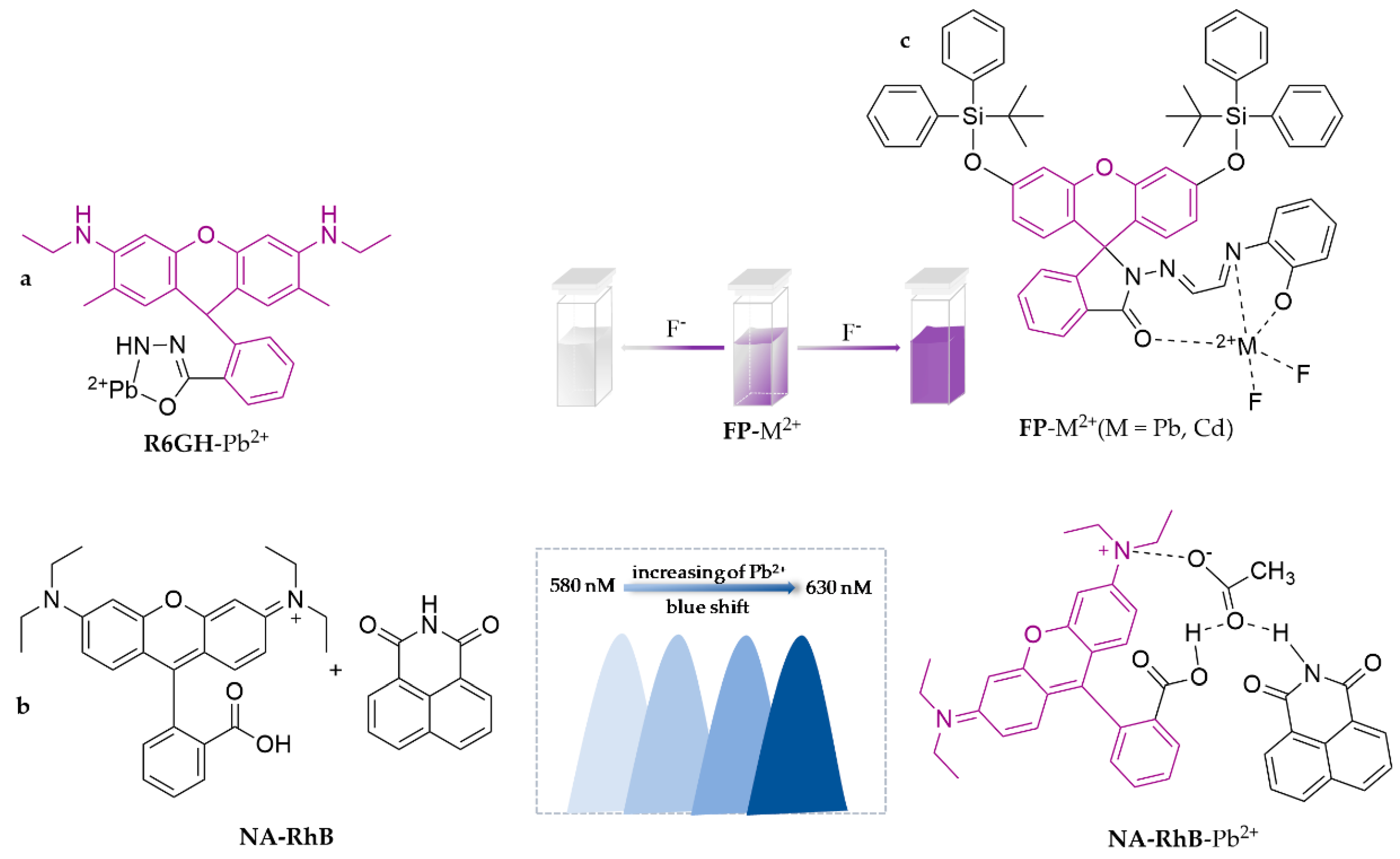
3.1.1. Fluorescent Probes to Detect Hg2+
3.1.2. Fluorescent Probes to Detect Pb2+
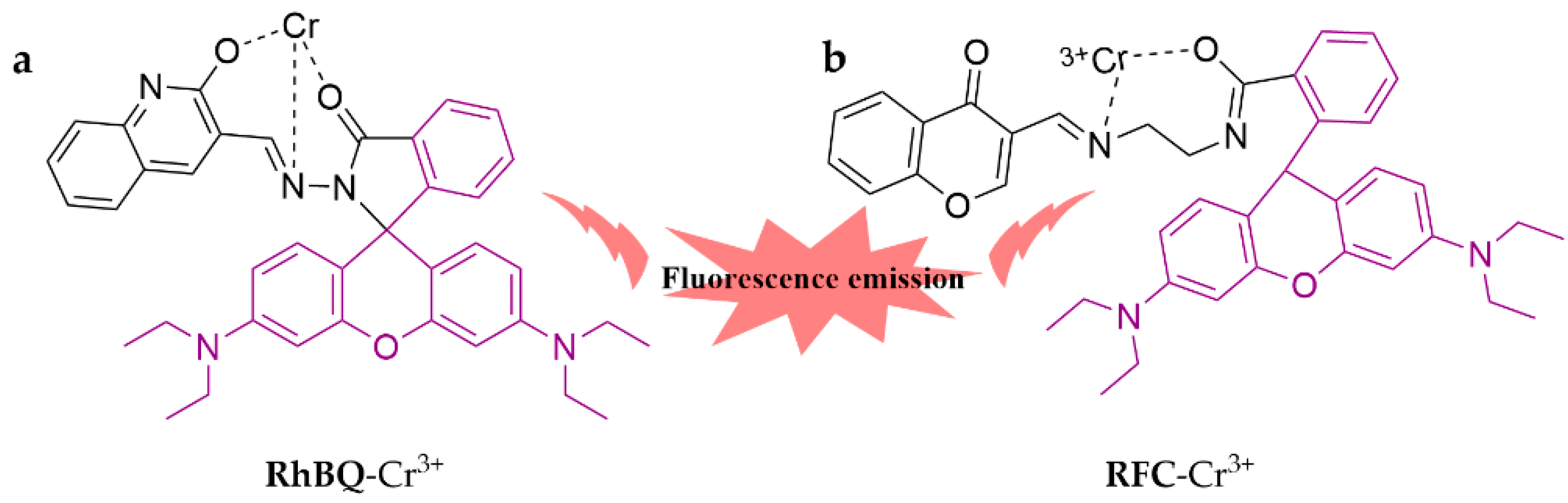
3.1.3. Fluorescent Probes to Detect Cr3+
3.2. Schiff Base-Based Fluorescent Probes
3.2.1. Fluorescent Probes to Detect Cr3+
3.2.2. Fluorescent Probes to Detect Cd3+
3.2.3. Fluorescent Probes to Detect Pb2+
3.3. Quinoline-Based Fluorescent Probes
3.3.1. Fluorescent Probes to Detect Cd3+
3.3.2. Fluorescent Probes to Detect Pb2+ and Hg2+
3.4. Coumarin-Based Fluorescent Probes
3.4.1. Fluorescent Probes to Detect Hg2+
3.4.2. Fluorescent Probes to Detect Cd2+ and Pb2+
3.5. Fluorescent Probes Based on Imidazole, Benzoxazole, Pyrazole, and Other Azoles
3.5.1. Imidazole-Based Fluorescent Probes
3.5.2. Benzoxazole-Based Fluorescent Probes
3.5.3. Pyrazole-Based Fluorescent Probes
3.6. Thiourea-Based Fluorescent Probes
3.7. TPE-Based Fluorescent Probes
3.8. Thiophene-Based Fluorescent Probes
3.9. Naphthoimide-Based Fluorescent Probes
3.10. Other Scaffolds
3.10.1. Fluorescent Probes to Detect Cd2+
3.10.2. Fluorescent Probes to Detect Cr3+
3.10.3. Fluorescent Probes to Detect Hg2+
3.10.4. Fluorescent Probes to Detect Pb2+
4. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Xu, L.; Dai, H.; Skuza, L.; Xu, J.; Shi, J.; Wang, Y.; Shentu, J.; Wei, S. Integrated survey on the heavy metal distribution, sources and risk assessment of soil in a commonly developed industrial area. J. Photochem. Photobiol. A Chem. 2022, 236, 113462. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, B.; Zhou, Z. Pollution assessment and source apportionment of heavy metals in soil from lead—Zinc mining areas of south China. J. Environ. Chem. Eng. 2023, 11, 109320. [Google Scholar] [CrossRef]
- Yang, J.; Li, X.; Xiong, Z.; Wang, M.; Liu, Q. Environmental Pollution Effect Analysis of Lead Compounds in China Based on Life Cycle. Int. J. Environ. Res. Public Health 2020, 17, 2184. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.M.; Zakeel, M.C.M.; Zavahir, J.S.; Marikar, F.; Jahan, I. Heavy Metal Accumulation in Rice and Aquatic Plants Used as Human Food: A General Review. Toxics 2021, 9, 360. [Google Scholar] [CrossRef] [PubMed]
- Briffa, J.; Sinagra, E.; Blundell, R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 2020, 6, e04691. [Google Scholar] [CrossRef]
- Qing, Y.; Li, Y.; Yang, J.; Li, S.; Gu, K.; Bao, Y.; Zhan, Y.; He, K.; Wang, X.; Li, Y. Risk assessment of mercury through dietary exposure in China. Environ. Pollut. 2022, 312, 120026. [Google Scholar] [CrossRef]
- Mielcarek, K.; Nowakowski, P.; Puscion-Jakubik, A.; Gromkowska-Kepka, K.J.; Soroczynska, J.; Markiewicz-Zukowska, R.; Naliwajko, S.K.; Grabia, M.; Bielecka, J.; Zmudzinska, A.; et al. Arsenic, cadmium, lead and mercury content and health risk assessment of consuming freshwater fish with elements of chemometric analysis. Food Chem. 2022, 379, 132167. [Google Scholar] [CrossRef]
- Shen, Y.; Nie, C.; Wei, Y.; Zheng, Z.; Xu, Z.-L.; Xiang, P. FRET-based innovative assays for precise detection of the residual heavy metals in food and agriculture-related matrices. Coord. Chem. Rev. 2022, 469, 214676. [Google Scholar] [CrossRef]
- Balali-Mood, M.; Naseri, K.; Tahergorabi, Z.; Khazdair, M.R.; Sadeghi, M. Toxic Mechanisms of Five Heavy Metals: Mercury, Lead, Chromium, Cadmium, and Arsenic. Front. Pharmacol. 2021, 12, 643972. [Google Scholar] [CrossRef]
- Majdanik, S.; Potocka-Banas, B.; Glowinski, S.; Luzny, S. Suicidal intoxication with mercury chloride. Forensic Toxicol. 2022, 41, 304–308. [Google Scholar] [CrossRef]
- Chen, B.; Dong, S. Mercury Contamination in Fish and Its Effects on the Health of Pregnant Women and Their Fetuses, and Guidance for Fish Consumption-A Narrative Review. Int. J. Environ. Res. Public Health 2022, 19, 15929. [Google Scholar] [CrossRef]
- Wang, Z.; Huang, X.; Li, J.; Liu, N.; Wei, Q. Lead exposure is non-linearly associated with subclinical myocardial injury in the general population without cardiovascular disease. Front. Inpublichealth 2022, 10, 975413. [Google Scholar] [CrossRef]
- Mielke, H.W.; Gonzales, C.R.; Powell, E.T.; Egendorf, S.P. Lead in Air, Soil, and Blood: Pb Poisoning in a Changing World. Int. J. Environ. Res. Public Health 2022, 19, 9500. [Google Scholar] [CrossRef]
- Hemmaphan, S.; Bordeerat, N.K. Genotoxic Effects of Lead and Their Impact on the Expression of DNA Repair Genes. Int. J. Environ. Res. Public Health 2022, 19, 4307. [Google Scholar] [CrossRef]
- He, Z.; Shen, P.; Feng, L.; Hao, H.; He, Y.; Fan, G.; Liu, Z.; Zhu, K.; Wang, Y.; Zhang, N.; et al. Cadmium induces liver dysfunction and ferroptosis through the endoplasmic stress-ferritinophagy axis. Ecotoxicol. Environ. Saf. 2022, 245, 114123. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Xu, Y.; Xu, J.; Zhang, J.; Xi, Y.; Pi, H.; Yang, L.; Yu, Z.; Wu, Q.; Meng, Z.; et al. Cadmium exposure impairs pancreatic beta-cell function and exaggerates diabetes by disrupting lipid metabolism. Environ. Int. 2021, 149, 106406. [Google Scholar] [CrossRef] [PubMed]
- Azarmehr, Z.; Ranji, N.; Khazaei Koohpar, Z.; Habibollahi, H. The effect of N-Acetyl cysteine on the expression of Fxr (Nr1h4), LXRalpha (Nr1h3) and Sirt1 genes, oxidative stress, and apoptosis in the liver of rats exposed to different doses of cadmium. Mol. Biol. Rep. 2021, 48, 2533–2542. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Chen, Y.; Qu, H.; Sun, J.; Wang, T.; Ma, Y.; Yuan, Y.; Bian, J.; Liu, Z. Microplastics Exacerbate Cadmium-Induced Kidney Injury by Enhancing Oxidative Stress, Autophagy, Apoptosis, and Fibrosis. Int. J. Mol. Sci. 2022, 23, 14411. [Google Scholar] [CrossRef] [PubMed]
- McClam, M.; Liu, J.; Fan, Y.; Zhan, T.; Zhang, Q.; Porter, D.E.; Scott, G.I.; Xiao, S. Associations between exposure to single cadmium, lead, mercury and mixtures and women’s infertility and long-term amenorrhea. Medrxiv-Epidemiology 2022, 1–59. [Google Scholar] [CrossRef]
- Dutta, S.; Gorain, B.; Choudhury, H.; Roychoudhury, S.; Sengupta, P. Environmental and occupational exposure of metals and female reproductive health. Environ. Sci. Pollut. Res. 2022, 29, 62067–62092. [Google Scholar] [CrossRef]
- Chakraborty, R.; Renu, K.; Eladl, M.A.; El-Sherbiny, M.; Elsherbini, D.M.A.; Mirza, A.K.; Vellingiri, B.; Iyer, M.; Dey, A.; Valsala Gopalakrishnan, A. Mechanism of chromium-induced toxicity in lungs, liver, and kidney and their ameliorative agents. Biomed. Pharmacother. 2022, 151, 113119. [Google Scholar] [CrossRef]
- Stortini, A.M.; Baldo, M.A.; Moro, G.; Polo, F.; Moretto, L.M. Bio- and Biomimetic Receptors for Electrochemical Sensing of Heavy Metal Ions. Sensors 2020, 20, 6800. [Google Scholar] [CrossRef] [PubMed]
- Bec, K.B.; Grabska, J.; Huck, C.W. Miniaturized NIR Spectroscopy in Food Analysis and Quality Control: Promises, Challenges, and Perspectives. Foods 2022, 11, 1465. [Google Scholar] [CrossRef] [PubMed]
- Dodo, K.; Fujita, K.; Sodeoka, M. Raman Spectroscopy for Chemical Biology Research. J. Am. Chem. Soc. 2022, 144, 19651–19667. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gao, F.; Wang, Y.; Li, H.; Zhang, J.; Sun, Z.; Jiang, Y. Fluorescent Organic Small Molecule Probes for Bioimaging and Detection Applications. Molecules 2022, 27, 8421. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.T.; Huang, Z.Y.; Wu, A.B.; Hu, Y.X.; Wang, N.C.; Zhang, Y.; Shu, W.M.; Yu, W.C. Recent progress in cadmium fluorescent and colorimetric probes. RSC Adv. 2021, 11, 29632–29660. [Google Scholar] [CrossRef]
- Mai, S.; Gonzalez, L. Molecular Photochemistry: Recent Developments in Theory. Angew. Chem. Int. Ed. 2020, 59, 16832–16846. [Google Scholar] [CrossRef] [PubMed]
- Bogomolov, A. Developing Multisensory Approach to the Optical Spectral Analysis. Sensors 2021, 21, 541. [Google Scholar] [CrossRef]
- Wu, D.; Hu, Y.; Cheng, H.; Ye, X. Detection Techniques for Lead Ions in Water: A Review. Molecules 2023, 28, 3601. [Google Scholar] [CrossRef]
- Chen, L.; Ding, M.; Li, Z.; Li, X.; Deng, L. Determination of macro, micro and toxic element concentrations in peanuts from main peanut producing areas of China by ICP-MS: A pilot study on the geographical characterization. RSC Adv. 2022, 12, 16790–16797. [Google Scholar] [CrossRef]
- Hwang, I.M.; Lee, H.M.; Lee, H.W.; Jung, J.H.; Moon, E.W.; Khan, N.; Kim, S.H. Determination of Toxic Elements and Arsenic Species in Salted Foods and Sea Salt by ICP-MS and HPLC-ICP-MS. ACS Omega 2021, 6, 19427–19434. [Google Scholar] [CrossRef] [PubMed]
- Braeuer, S.; Van Helden, T.; Van Acker, T.; Leroux, O.; Van Der Straeten, D.; Verbeken, A.; Borovicka, J.; Vanhaecke, F. Quantitative mapping of mercury and selenium in mushroom fruit bodies with laser ablation-inductively coupled plasma-mass spectrometry. Anal. Bioanal. Chem. 2022, 414, 7517–7530. [Google Scholar] [CrossRef] [PubMed]
- Lan, G.; Li, X.; Jia, H.; Yu, X.; Wang, Z.; Yao, J.; Mao, X. Fast and Sensitive Determination of Cadmium and Selenium in Rice by Direct Sampling Electrothermal Vaporization Inductively Coupled Plasma Mass Spectrometry. Molecules 2022, 27, 8176. [Google Scholar] [CrossRef] [PubMed]
- Londonio, A.; Morzan, E.; Smichowski, P. Simultaneous on-line preconcentration and determination of toxic elements in rice and rice-based products by SPE–ICP–MS: Multiple response optimization. J. Food Compos. Anal. 2022, 107, 104388. [Google Scholar] [CrossRef]
- Chen, D.; Lu, L.; Zhang, H.; Lu, B.; Feng, J.; Zeng, D. Sensitive Mercury Speciation Analysis in Water by High-Performance Liquid Chromatography-Atomic Fluorescence Spectrometry Coupling with Solid-Phase Extraction. Anal. Sci. Sepetember 2021, 37, 1235–1240. [Google Scholar] [CrossRef]
- Catenza, K.F.; Donkor, K.K. Determination of Heavy Metals in Cannabinoid-Based Food Products Using Microwave-Assisted Digestion and ICP-MS. Food Anal. Methods 2022, 15, 2537–2546. [Google Scholar] [CrossRef]
- Zeiner, M.; Soltic, M.; Nemet, I.; Juranovic Cindric, I. Multielement Determination in Turmeric (Curcuma longa L.) Using Different Digestion Methods. Molecules 2022, 27, 8392. [Google Scholar] [CrossRef]
- Pirhadi, M.; Alikord, M.; Tajdar-Oranj, B.; Khaniki, G.J.; Nazmara, S.; Fathabad, A.E.; Ghalhari, M.R.; Sadighara, P. Potential toxic elements (PTEs) concentration in wheat and flour products in Iran: A probabilistic risk assessment. Heliyon 2022, 8, e11803. [Google Scholar] [CrossRef]
- Bereksi-Reguig, D.; Bouchentouf, S.; Allali, H.; Adamczuk, A.; Kowalska, G.; Kowalski, R. Trace Elements and Heavy Metal Contents in West Algerian Natural Honey. J. Anal. Methods Chem. 2022, 2022, 7890856. [Google Scholar] [CrossRef]
- Kiani, A.; Arabameri, M.; Moazzen, M.; Shariatifar, N.; Aeenehvand, S.; Khaniki, G.J.; Abdel-Wahhab, M.; Shahsavari, S. Probabilistic Health Risk Assessment of Trace Elements in Baby Food and Milk Powder Using ICP-OES Method. Biol. Trace Elem. Res. 2022, 200, 2486–2497. [Google Scholar] [CrossRef]
- Liu, S.; Duan, X.; Sun, J. Determination of Cadmium in Water Samples by Electrochemical Hydride Generation Atomic Fluorescence Spectrometry Using Series Graphite Tubes as Electrolytic Cells under Constant Voltage. Anal. Sci. April 2020, 36, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Li, R.; Deng, S. Determination of the Total Content of Arsenic, Antimony, Selenium and Mercury in Chinese Herbal Food by Chemical Vapor Generation-Four-Channel Non-dispersive Atomic Fluorescence Spectrometry. J. Fluoresc. 2020, 30, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Li, X.; Lan, G.; Wang, Z.; Feng, L.; Mao, X. Fast Detection of Cadmium in Chocolate by Solid Sampling Electrothermal Vaporization Atomic Absorption Spectrometry and Its Application on Dietary Exposure Risk Assessment. Molecules 2022, 27, 6197. [Google Scholar] [CrossRef] [PubMed]
- Pushie, M.J.; Sylvain, N.J.; Hou, H.; Hackett, M.J.; Kelly, M.E.; Webb, S.M. X-ray fluorescence microscopy methods for biological tissues. Metallomics 2022, 14, mfac032. [Google Scholar] [CrossRef]
- Winkler, A.; Rauwolf, M.; Sterba, J.H.; Wobrauschek, P.; Streli, C.; Turyanskaya, A. Total reflection X-ray fluorescence analysis of elemental composition of herbal infusions and teas. J. Sci. Food Agric. 2020, 100, 4226–4236. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, S.; Xiao, J.; Hu, Q.; Zhao, L. A Rapid and Multi-Element Method for the Determination of As, Cd, Ni, Pb, Sn, and Zn in Scallops Using High Definition X-Ray Fluorescence (HDXRF) Spectrometry. Food Anal. Methods 2022, 15, 2712–2724. [Google Scholar] [CrossRef]
- Acquah, G.E.; Hernandez-Allica, J.; Thomas, C.L.; Dunham, S.J.; Towett, E.K.; Drake, L.B.; Shepherd, K.D.; McGrath, S.P.; Haefele, S.M. Portable X-ray fluorescence (pXRF) calibration for analysis of nutrient concentrations and trace element contaminants in fertilisers. PLoS ONE 2022, 17, 1–20. [Google Scholar] [CrossRef]
- Lopez, A.M.; Nicolini, C.M.; Aeppli, M.; Luby, S.P.; Fendorf, S.; Forsyth, J.E. Assessing Analytical Methods for the Rapid Detection of Lead Adulteration in the Global Spice Market. Environ. Sci. Technol. 2022, 56, 16996–17006. [Google Scholar] [CrossRef]
- Liu, F.; Ye, L.; Peng, J.; Song, K.; Shen, T.; Zhang, C.; He, Y. Fast Detection of Copper Content in Rice by Laser-Induced Breakdown Spectroscopy with Uni- and Multivariate Analysis. Sensors 2018, 18, 705. [Google Scholar] [CrossRef]
- Zhao, Q.; Yu, Y.; Hao, N.; Miao, P.; Li, X.; Liu, C.; Li, Z. Data fusion of Laser-induced breakdown spectroscopy and Near-infrared spectroscopy to quantitatively detect heavy metals in lily. Microchem. J. 2023, 190, 108670. [Google Scholar] [CrossRef]
- Liu, G.; Li, L.; Huang, X.; Zheng, S.; Xu, D.; Xu, X.; Zhang, Y.; Lin, H. Determination of triazole pesticides in aqueous solution based on magnetic graphene oxide functionalized MOF-199 as solid phase extraction sorbents. Microporous Mesoporous Mater. 2018, 270, 258–264. [Google Scholar] [CrossRef]
- Wagh, S.B.; Maslivetc, V.A.; La Clair, J.J.; Kornienko, A. Lessons in Organic Fluorescent Probe Discovery. Chembiochem 2021, 22, 3109–3139. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Zhang, K.; Li, C.; Li, Y.; Niu, S. A novel fluorescent chemosensor based on a rhodamine 6G derivative for the detection of Pb2+ ion. Sens. Actuators B Chem. 2017, 246, 696–702. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, W.; Li, R.; Zhang, E.; Li, Z.; Tang, L.; Han, B.; Hou, X.; Wang, J.J. A novel rhodamine-based colorimetric and fluorometric probe for simultaneous detection of multi-metal ions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 230, 118050. [Google Scholar] [CrossRef]
- Roy, A.; Das, S.; Sacher, S.; Mandal, S.K.; Roy, P. A rhodamine based biocompatible chemosensor for Al3+, Cr3+ and Fe3+ ions: Extraordinary fluorescence enhancement and a precursor for future chemosensors. Dalton Trans. 2019, 48, 17594–17604. [Google Scholar] [CrossRef]
- Qi, M.; Zhang, Z.; Li, L.; Mu, X.; Wang, Y. A sensitive ratiometric fluorescent chemosensor for visual and wearable mercury (II) recognition in river prawn and water samples. Food Chem. 2023, 408, 135211. [Google Scholar] [CrossRef]
- Mohammad, H.; Saleh Musha Islam, A.; Sasmal, M.; Prodhan, C.; Ali, M. A fluorescein-2-(Pyridin-2-ylmethoxy) benzaldehyde conjugate for fluorogenic turn-ON recognition of Hg2+ in water and living cells with logic gate and memory device applications. Inorganica Chim. Acta 2022, 543, 121165. [Google Scholar] [CrossRef]
- Chepak, A.; Balatskiy, D.; Tutov, M.; Mironenko, A.; Bratskaya, S. Light Harvesting Nanoprobe for Trace Detection of Hg2+ in Water. Molecules 2023, 28, 1633. [Google Scholar] [CrossRef]
- Wu, L.; Wang, X.; Huang, J.; Wei, H.; Kan, C. Switch-type near-infrared fluorescent probes for Hg2+ based on rhodamines. J. Photochem. Photobiol. A Chem. 2022, 429, 113936. [Google Scholar] [CrossRef]
- Xie, X.; Pan, M.; Hong, L.; Liu, K.; Yang, J.; Wang, S.; Wang, S. An “Off-On” Rhodamine 6G Hydrazide-Based Output Platform for Fluorescence and Visual Dual-Mode Detection of Lead(II). J. Agric. Food Chem. 2021, 69, 7209–7217. [Google Scholar] [CrossRef]
- Shen, Y.; Wu, T.; Wang, Y.; Zhang, S.L.; Zhao, X.; Chen, H.Y.; Xu, J.J. Nucleolin-Targeted Ratiometric Fluorescent Carbon Dots with a Remarkably Large Emission Wavelength Shift for Precise Imaging of Cathepsin B in Living Cancer Cells. Anal Chem 2021, 93, 4042–4050. [Google Scholar] [CrossRef]
- Radiul, S.M.; Hazarika, S. Variation of Stokes Shift and Peak Wavelength Shift as a Sensing Probe for Detection of Lead in Water Using Laser Induced Fluorescence Resonance Energy Transfer. J. Fluoresc. 2021, 31, 889–896. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, L.; Yang, J.; Zhang, Y.; Yuan, M.S. An “OR-AND” logic gate based multifunctional colorimetric sensor for the discrimination of Pb2+ and Cd2+. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 232, 1–18. [Google Scholar] [CrossRef]
- Shekhar Samanta, S.; Mandal, U.; Das, B.; Mandal, S.; Upadhyay, P.; Debraj Singh, T.; Misra, A. Rhodamine B-Quinoline based schiff base as fluorescent ‘turn on’ sensor of Al3+, Cr3+, HSO4− and its cytotoxicity and cell imaging application on TPC-1 and HtH-7 cell lines. J. Photochem. Photobiol. A Chem. 2023, 442, 114806. [Google Scholar] [CrossRef]
- Bikash Maity, M.; Talukdar, D.; Dutta, B.; Bairy, G.; Murmu, N.; Das, G.; Sinha, C. Application of a Rhodamine-chromone Schiff base probe for the sensing of Fe3+, Al3+, Cr3+ at low concentration and exploration of the anticancer activity and bio-imaging. Inorganica Chim. Acta 2023, 545, 121276. [Google Scholar] [CrossRef]
- Guo, S.; Liu, G.; Fan, C.; Pu, S. A highly selective fluorescent probe for detection of Cd2+ and HSO3− based on photochromic diarylethene with a triazole-bridged coumarin-quinoline group. RSC Adv. 2018, 8, 22786–22798. [Google Scholar] [CrossRef]
- Punithakumari, G.; Wu, S.P.; Velmathi, S. Highly Selective Detection of Cr3+ Ion with Colorimetric & Fluorescent Response Via Chemodosimetric Approach in Aqueous Medium. J. Fluoresc. 2018, 28, 663–670. [Google Scholar] [CrossRef]
- Minhaz, A.; Anwar, A.; Ahmad, I.; Khattak, R.; Yaseen, M.; Ahmed, F.; Shah, M.R.; Khan, N.A.; Ishaq, M. A flexible Schiff base probe for spectrophotometric detection of chromium (III). Int. J. Environ. Sci. Technol. 2018, 16, 5577–5584. [Google Scholar] [CrossRef]
- Liu, Y.; Li, L.; Zhang, Y.; Sheng, M.; Wang, Y.; Xing, Z.; Yang, L.; Yue, M.; Fu, Y.; Ye, F. A novel functional fluorescent probe based on a pyrene derivative for the detection of multiple pollutants. J. Mol. Liq. 2023, 382, 121888. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, F.; Wei, K.; Kang, M.; Liu, P.; Yang, X.; Pei, M.; Zhang, G. 5-(Thiophene-2-yl)oxazole derived “off-on-off” fluorescence chemosensor for sequential recognition of In3+ and Cr3+ ions. J. Photochem. Photobiol. A Chem. 2023, 437, 114464. [Google Scholar] [CrossRef]
- Khan, S.; Muhammad, M.; Algethami, J.S.; Al-Saidi, H.M.; Almahri, A.; Hassanian, A.A. Synthesis, Characterization and Applications of Schiff Base Chemosensor for Determination of Cr(III) Ions. J. Fluoresc. 2022, 32, 1889–1898. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Zhang, L.; Zhou, X.; Mu, F.; Shi, G. A sensitive fluorescent probe for beta-galactosidase activity detection and application in ovarian tumor imaging. J. Mater. Chem. B 2021, 9, 170–715. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Huang, Q.; Wu, W.; Ren, T.; Zhang, J. Dicyanomethylene-4H-pyran based Schiff base for turn on NIR fluorescence sensing of Fe3+, Al3+ and Cr3+ and its application in molecular logic gate. Opt. Mater. 2022, 130, 112568. [Google Scholar] [CrossRef]
- Musikavanhu, B.; Zhang, Y.; Zhu, D.; Xue, Z.; Yuan, R.; Wang, S.; Zhao, L. Turn-off detection of Cr(III) with chelation enhanced fluorescence quenching effect by a naphthyl hydrazone Shiff base chemosensor. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 281, 121599. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.; Manna, A.K.; Sahu, M.; Rout, K.; Patra, G.K. Simple salicylaldimine-functionalized dipodal bis Schiff base chromogenic and fluorogenic chemosensors for selective and sensitive detection of Al3+ and Cr3+. Inorganica Chim. Acta 2020, 499, 119192. [Google Scholar] [CrossRef]
- Wang, J.H.; Liu, Y.M.; Chao, J.B.; Wang, Y.; Wang, H.; Shuang, S.M. A phenazine-imidazole based ratiometric fluorescent probe for Cd2+ ions and its application in in vivo imaging. Anal. Methods 2022, 14, 1462–1470. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, C.; Xu, D.; Liao, G.; Pu, S. A fluorescent sensor for Zn2+ and Cd2+ based on a diarylethene derivative with an indole-2-methylhydrazone moiety. J. Photochem. Photobiol. A Chem. 2022, 424, 113634. [Google Scholar] [CrossRef]
- Ma, J.; Dong, Y.; Yu, Z.; Wu, Y.; Zhao, Z. A pyridine based Schiff base as a selective and sensitive fluorescent probe for cadmium ions with “turn-on” fluorescence responses. New J. Chem. 2022, 46, 3348–3357. [Google Scholar] [CrossRef]
- Xue, S.; Xie, Z.; Chu, Y.; Shi, W.; Liu, Y.; Zhao, Y. Highly selective and sensitive fluorescent probe possessing AIEE and ICT properties for rapid detection of Pb2+ in aqueous medium and its applications in living cells. Luminescence 2022, 37, 108–117. [Google Scholar] [CrossRef]
- Rahimi, M.; Amini, A.; Behmadi, H. Novel symmetric Schiff-base benzobisthiazole-salicylidene derivative with fluorescence turn-on behavior for detecting Pb2+ ion. J. Photochem. Photobiol. A Chem. 2020, 388, 112190. [Google Scholar] [CrossRef]
- Rout, K.; Manna, A.K.; Sahu, M.; Mondal, J.; Singh, S.K.; Patra, G.K. Triazole-based novel bis Schiff base colorimetric and fluorescent turn-on dual chemosensor for Cu2+ and Pb2+: Application to living cell imaging and molecular logic gates. RSC Adv. 2019, 9, 25919–25931. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Zhang, Z. A quinoline-based ratiometric fluorescent probe for discriminative detection of Zn2+ and Cd2+ with different binding modes, and its Zn2+ complex for relay sensing of pyrophosphate and adenosine triphosphate. Dye. Pigment. 2019, 165, 172–181. [Google Scholar] [CrossRef]
- Xiao, Y.; Ma, J.; Li, D.; Liu, L.; Wang, H. Preparation 4′-Quinolin-2-yl-[2, 2′; 6′, 2”] terpyridine as a ratiometric fluorescent probe for cadmium ions and zinc ions in aqueous. J. Photochem. Photobiol. A Chem. 2020, 399, 112613. [Google Scholar] [CrossRef]
- Mohanasundaram, D.; Bhaskar, R.; Gangatharan Vinoth Kumar, G.; Rajesh, J.; Rajagopal, G. A quinoline based Schiff base as a turn-on fluorescence chemosensor for selective and robust detection of Cd2+ ion in semi-aqueous medium. Microchem. J. 2021, 164, 106030. [Google Scholar] [CrossRef]
- Panda, S.K.; Mishra, S.; Mamidi, P.; Chattopadhyay, S.; Singh, A.K. An efficient PET-based probe for detection and discrimination of Zn2+ and Cd2+ in near-aqueous media and live-cell imaging. J. Photochem. Photobiol. A Chem. 2022, 427, 113816. [Google Scholar] [CrossRef]
- René, W.; Lenoble, V.; Chioukh, M.; Branger, C. A turn-on fluorescent ion-imprinted polymer for selective and reliable optosensing of lead in real water samples. Sens. Actuators B Chem. 2020, 319, 128252. [Google Scholar] [CrossRef]
- Velmurugan, K.; Vickram, R.; Jipsa, C.V.; Karthick, R.; Prabakaran, G.; Suresh, S.; Prabhu, J.; Velraj, G.; Tang, L.; Nandhakumar, R. Quinoline based reversible fluorescent probe for Pb2+; applications in milk, bioimaging and INHIBIT molecular logic gate. Food Chem. 2021, 348, 129098. [Google Scholar] [CrossRef]
- Che, S.; Yin, L.; Chen, M.; Fan, Y.; Xu, A.; Zhou, C.; Fu, H.; She, Y. Real-time monitoring of mercury(II) in water and food samples using a quinoline-based ionic probe. Food Chem. 2023, 407, 135052. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zheng, D.; Xu, Y.; Liu, Q.; Xu, C.; Jiao, Q.; Zhu, H. Naked-eye Detection of Hg2+ in Practical Applications Using a Highly Selective and Sensitive Fluorescent Probe. Anal. Sci. Dec. 2018, 34, 1411–1417. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Shi, W.; Yang, C.; Xu, J.; Liu, H.; Xu, J.; Zhu, B. Highly selective and sensitive fluorescent probe for the rapid detection of mercury ions. RSC Adv. 2019, 9, 10554–10560. [Google Scholar] [CrossRef]
- Isaad, J.; Malek, F.; Achari, A.E. Colorimetric and fluorescent probe based on coumarin/thiophene derivative for sequential detection of mercury(II) and cyanide ions in an aqueous medium. J. Mol. Struct. 2022, 1270, 133838. [Google Scholar] [CrossRef]
- Shaily, S.; Kumar, A.; Ahmed, N. A coumarin–chalcone hybrid used as a selective and sensitive colorimetric and turn-on fluorometric sensor for Cd2+ detection. New J. Chem. 2017, 41, 14746–14753. [Google Scholar] [CrossRef]
- Zehra, S.; Khan, R.A.; Alsalme, A.; Tabassum, S. Coumarin Derived “Turn on” Fluorescent Sensor for Selective Detection of Cadmium (II) Ion: Spectroscopic Studies and Validation of Sensing Mechanism by DFT Calculations. J. Fluoresc. 2019, 29, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Abedi, S.A.A.; Chi, W.; Tan, D.; Shen, T.; Wang, C.; Ang, E.C.X.; Tan, C.H.; Anariba, F.; Liu, X. Restriction of Twisted Intramolecular Charge Transfer Enables the Aggregation-Induced Emission of 1-(N,N-Dialkylamino)-naphthalene Derivatives. J. Phys. Chem. A 2021, 125, 8397–8403. [Google Scholar] [CrossRef]
- Sharma, N.; Gulati, A. Amino Appended Coumarin Composites for Pb2+ Capturing in Aqueous Medium. J. Fluoresc. 2022, 32, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Liu, Y.M.; Chao, J.B.; Wang, H.; Wang, Y.; Shuang, S. A simple but efficient fluorescent sensor for ratiometric sensing of Cd2+ and bio-imaging studies. Sens. Actuators B Chem. 2020, 303, 127216. [Google Scholar] [CrossRef]
- Yang, J.Y.; Han, J.H.; Shang, Z.B.; Wang, Y.; Shuang, S.M. New Schiff base probe for the fluorometric turn-on sensing of Cd2+ ions and bio-imaging application. J. Lumin. 2022, 249, 119017. [Google Scholar] [CrossRef]
- Barot, Y.B.; Anand, V.; Vyas, S.; Mishra, R. Paper-based device for nanomolar detection of Cd2+ using AIEE-active imidazolium ionic liquid functionalized phenothiazine based Schiff-Base. J. Mol. Liq. 2023, 376, 121490. [Google Scholar] [CrossRef]
- Karuppiah, K.; Malini, N.; Chinnamadhaiyan, M.; Yesudhasan, C.; Sepperumal, M.; Rajabathar, J.R.; Karnan, M.; Siva, A.; Selvaraj, M. A novel hydrazone platform for the recognition of Cd2+ and F− ions: Imaging analysis in Zebrafish embryos. J. Mol. Struct. 2023, 1282, 135152. [Google Scholar] [CrossRef]
- Zhang, B.; Suo, Q.; Li, Q.; Hu, J.; Zhu, Y.; Gao, Y.; Wang, Y. Multiresponsive chemosensors based on ferrocenylimidazo[4,5-b]pyridines: Solvent-dependent selective dual sensing of Hg2+ and Pb2+. Tetrahedron 2022, 120, 132878. [Google Scholar] [CrossRef]
- Du, Y.; Zhao, H.; Peng, X.; Zhou, X.; Yang, X.; Li, Y.; Yan, M.; Cui, Y.; Sun, G. A novel phenanthroline[9,10-d] imidazole-based fluorescent sensor for Hg2+ with “turn-on” fluorescence response. J. Photochem. Photobiol. A Chem. 2023, 439, 114604. [Google Scholar] [CrossRef]
- Paderni, D.; Giorgi, L.; Voccia, M.; Formica, M.; Caporaso, L.; Macedi, E.; Fusi, V. A New Benzoxazole-Based Fluorescent Macrocyclic Chemosensor for Optical Detection of Zn2+ and Cd2+. Chemosensors 2022, 10, 188. [Google Scholar] [CrossRef]
- He, X.; Wu, C.; Qian, Y.; Li, Y.; Zhang, L.; Ding, F.; Chen, H.; Shen, J. Highly sensitive and selective light-up fluorescent probe for monitoring gallium and chromium ions in vitro and in vivo. Analyst 2019, 144, 3807–3816. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qi, Y.; Xu, Z.; Song, Y.; Li, H.; Zhang, J.; Tang, J.; Zhou, J. A cyanine-based dual-modal probe for fluorimetric detection of mercury ion and colorimetric sensing of pH. Color. Technol. 2022, 138, 427–439. [Google Scholar] [CrossRef]
- Li, G.; Wang, J.; Li, D.; Liu, S.; Yin, J.; Lai, Z.; Yang, G. A Hg(II)-specific probe for imaging application in living systems and quantitative analysis in environmental/food samples. Chin. Chem. Lett. 2021, 32, 1527–1531. [Google Scholar] [CrossRef]
- Ghorbanian, M.; Asghari, S.; Tajbakhsh, M. A new benzothiazole azo dye colorimetric chemosensor for detecting Pb2+ ion. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 296, 122652. [Google Scholar] [CrossRef]
- Sharma, P.; Bhogal, S.; Lealam, A.; Kumar, S.; Yusuf, M.; Malik, A.K. Experimental and Theoretical Studies of the Pyrazoline Derivative 5-(4-methylphenyl)-3-(5-methylfuran-2-yl)-1-phenyl-4,5-dihydro-1H-Pyrazole and its Application for Selective Detection of Cd2+ ion as Fluorescent Sensor. J. Fluoresc. 2022, 32, 969–981. [Google Scholar] [CrossRef]
- Krishnaveni, K.; Murugesan, S.; Siva, A. Fluorimetric and colorimetric detection of multianalytes Zn2+/Cd2+/F− ions via 5-bromosalicyl hydrazone appended pyrazole receptor; live cell imaging analysis in HeLa cells and zebra fish embryos. Inorg. Chem. Commun. 2021, 132, 108843. [Google Scholar] [CrossRef]
- Huang, M.X.; Lv, C.H.; Huang, Q.D.; Lai, J.P.; Sun, H. A novel and fast responsive turn-on fluorescent probe for the highly selective detection of Cd2+ based on photo-induced electron transfer. RSC Adv. 2019, 9, 36011–36019. [Google Scholar] [CrossRef]
- Rahimi, H.; Hosseinzadeh, R.; Tajbakhsh, M. A new and efficient pyridine-2,6-dicarboxamide-based fluorescent and colorimetric chemosensor for sensitive and selective recognition of Pb2+ and Cu2+. J. Photochem. Photobiol. A Chem. 2021, 407, 113049. [Google Scholar] [CrossRef]
- Zhang, Z.; Yuan, S.; Wang, E. A Dual-Target Fluorescent Probe with Response-Time Dependent Selectivity for Cd2+ and Cu2+. J. Fluoresc. 2018, 28, 1115–1119. [Google Scholar] [CrossRef]
- Samanta, S.S.; Giri, P.K.; Mudi, N.; Mandal, U.; Misra, A. Fluorescence ‘Turn-on’ Dual Sensor for Selective Detection of Cd2+ and H2AsO4− in Water. J. Fluoresc. 2023, 33, 517–526. [Google Scholar] [CrossRef]
- Jiang, C.; Yang, L.; Li, P.; Liu, Y.; Li, S.; Fu, Y.; Ye, F. A simple and rapid fluorescent approach for Pb2+ determination and application in water samples and living cells. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 263, 120168. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, C.; Yang, H.; Wu, X.; Yan, L. Two near-infrared fluorescent probes based on dicyanoisfluorone for rapid monitoring of Zn2+ and Pb2+. Methods Appl. Fluoresc. 2022, 10, 035010. [Google Scholar] [CrossRef] [PubMed]
- Erdemir, E.; Suna, G.; Gunduz, S.; Sahin, M.; Eglence-Bakir, S.; Karakus, E. Rapid, ultrasensitive, highly selective detection of toxic Hg(II) ions in seabass, swordfish and water samples. Food Chem. 2022, 371, 131309. [Google Scholar] [CrossRef]
- Pan, Y.; Guo, Y.; Li, Y.; Tang, L.; Yan, X. A new aggregation-induced emission-based fluorescent probe for effective detection of Hg2+ and its multiple applications. Chin. Chem. Lett. 2023, 2023, 108237. [Google Scholar] [CrossRef]
- Tang, L.; Yu, H.; Zhong, K.; Gao, X.; Li, J. An aggregation-induced emission-based fluorescence turn-on probe for Hg2+ and its application to detect Hg2+ in food samples. RSC Adv. 2019, 9, 23316–23323. [Google Scholar] [CrossRef] [PubMed]
- Ho, F.C.; Huang, K.H.; Cheng, H.W.; Huang, Y.J.; Nhien, P.Q.; Wu, C.H.; Wu, J.I.; Chen, S.Y.; Lin, H.C. FRET processes of bi-fluorophoric sensor material containing tetraphenylethylene donor and optical-switchable merocyanine acceptor for lead ion (Pb2+) detection in semi-aqueous media. Dye. Pigment. 2021, 189, 109238. [Google Scholar] [CrossRef] [PubMed]
- Lan, L.; Niu, Q.; Li, T. A highly selective colorimetric and ratiometric fluorescent probe for instantaneous sensing of Hg2+ in water, soil and seafood and its application on test strips. Anal. Chim. Acta 2018, 1023, 105–114. [Google Scholar] [CrossRef]
- Rajadurai, M.; Reddy, E.R. Tuning the sensitivity towards mercury via cooperative binding to d-fructose: Dual fluorescent chemosensor based on 1,8-naphthyridine-boronic acid derivative. RSC Adv. 2021, 11, 14862–14870. [Google Scholar] [CrossRef]
- Farshbaf, S.; Anzenbacher, P. Fluorimetric sensing of ATP in water by an imidazolium hydrazone based sensor. Chem. Commun. 2019, 00, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Anbu Durai, W.; Ramu, A.; Dhakshinamoorthy, A. A Visual and Ratiometric Chemosensor Using Thiophene Functionalized Hydrazone for the Selective Sensing of Pb2+ and F− Ions. J. Fluoresc. 2021, 31, 465–474. [Google Scholar] [CrossRef]
- Ye, F.; Wu, N.; Li, P.; Liu, Y.L.; Li, S.J.; Fu, Y. A lysosome-targetable fluorescent probe for imaging trivalent cations Fe3+, Al3+ and Cr3+ in living cells. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 222, 117242. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Luo, J.; Wang, Y.; Ding, L.; Liang, Q.; Yang, Z.; Lu, J.; Wu, A. A water-soluble naphthalimide fluorescent probe for Cr2O72− and Fe3+ based on inner filter effect. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 289, 122245. [Google Scholar] [CrossRef]
- Su, M.; Liu, C.; Zhang, Y.; Rong, X.; Wang, X.; Li, X.; Wang, K.; Zhu, H.; Zhu, B. Rational design of a water-soluble TICT-AIEE-active fluorescent probe for mercury ion detection. Anal. Chim. Acta 2022, 1230, 340337. [Google Scholar] [CrossRef] [PubMed]
- Sadia, M.; Khan, J.; Khan, R.; Kamran, A.W.; Zahoor, M.; Ullah, R.; Bari, A.; Ali, E.A. Rapid Detection of Cd2+ Ions in the Aqueous Medium Using a Highly Sensitive and Selective Turn-On Fluorescent Chemosensor. Molecules 2023, 28, 3635. [Google Scholar] [CrossRef]
- Jiang, H.; Chen, L.; Li, Z.; Li, J.; Ma, H.; Ning, L.; Li, N.; Liu, X. A facile AIE fluorescent probe with large Stokes shift for the detection of Cd2+ in real water samples and living cells. J. Lumin. 2022, 243, 118672. [Google Scholar] [CrossRef]
- Petdum, A.; Kaewnok, N.; Panchan, W.; Charoenpanich, A.; Sirirak, J.; Sahasithiwat, S.; Sooksimuang, T.; Wanichacheva, N. Novel rapid “turn on” tetrahydro-[5]helicene-based fluorescence sensor for selective detection of Cd2+ with a remarkable large Stokes shift and its applications in food samples and living cell. J. Photochem. Photobiol. A Chem. 2022, 423, 113578. [Google Scholar] [CrossRef]
- Khan, J.; Sadia, M.; Shah, S.W.A.; Naz, R.; Ali, F. 2,6-bis(E)-4-methylbenzylidine)-cyclohexan-1-one as a Fluorescent-on Sensor for Ultra Selective Detection of Chromium Ion in Aqueous Media. J. Fluoresc. 2021, 31, 1759–1770. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, P.F.; Mishra, D.R. A Sensor for the Detection of Cr (III) and Fe (III) Ions Based on “Turn Off” Mechanism of Fluorescence with Computational Studies. J. Fluoresc. 2022, 32, 215–226. [Google Scholar] [CrossRef]
- Kamaci, M. Poly(Azomethine-urethane)-based Fluorescent Chemosensor for the Detection of Cr3+ Cations in Different Water Samples. J. Fluoresc. 2023, 33, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Suna, G.; Gunduz, S. An Anthracene and Indole-based Fluorescent Probe for the Detection of Chromium(III) Ions in Real Water Samples. J. Fluoresc. 2023, 33, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Chakraborty, S.; Lohar, S.; Ahmmed, E.; Saha, N.C.; Mandal, S.K.; Dhara, K.; Chattopadhyay, P. A Lysosome-Targetable Fluorescence Sensor for Ultrasensitive Detection of Hg2+ in Living Cells and Real Samples. Chem. Res. Toxicol. 2019, 32, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Ma, J.; Liu, L.; Li, D.; Huo, Y.; Liu, H. A novel carbazole-based highly sensitive and selective turn-on fluorescent probe for mercury (II) ions in aqueous THF. J. Photochem. Photobiol. A Chem. 2021, 416, 113322. [Google Scholar] [CrossRef]
- Rasin, P.; Musthafa, M.; Sreekanth, A. An Effective Selenium-Based Fluorescence Chemosensor for Selective Recognition of Hg2+ in Aqueous Medium: Experimental and Theoretical Studies. J. Fluoresc. 2022, 32, 2065–2076. [Google Scholar] [CrossRef]
- Merz, V.; Merz, J.; Kirchner, M.; Lenhart, J.; Marder, T.B.; Krueger, A. Pyrene-Based “Turn-Off” Probe with Broad Detection Range for Cu2+, Pb2+ and Hg2+ Ions. Chemistry 2021, 27, 8118–8126. [Google Scholar] [CrossRef]
- Ding, Y.; Zhu, W.; Xie, Y. Development of Ion Chemosensors Based on Porphyrin Analogues. Chem. Rev. 2017, 117, 2203–2256. [Google Scholar] [CrossRef]
- Qi, D.; Zhang, J.; Zhang, D.; Zhu, M.; Gong, L.; Su, C.; Lu, W.; Bian, Y.; Jiang, J. A phthalocyanine-porphyrin triad for ratiometric fluorescent detection of Lead(II) ions. Dye. Pigment. 2020, 173, 107941. [Google Scholar] [CrossRef]
- Wang, L.; Peng, X.; Fu, H.; Huang, C.; Li, Y.; Liu, Z. Recent advances in the development of electrochemical aptasensors for detection of heavy metals in food. Biosens. Bioelectron. 2020, 147, 111777. [Google Scholar] [CrossRef]
- Fan, Y.; Wang, S.; Zhang, F. Optical Multiplexed Bioassays for Improved Biomedical Diagnostics. Angew. Chem. Int. Ed. 2019, 58, 13208–13219. [Google Scholar] [CrossRef]


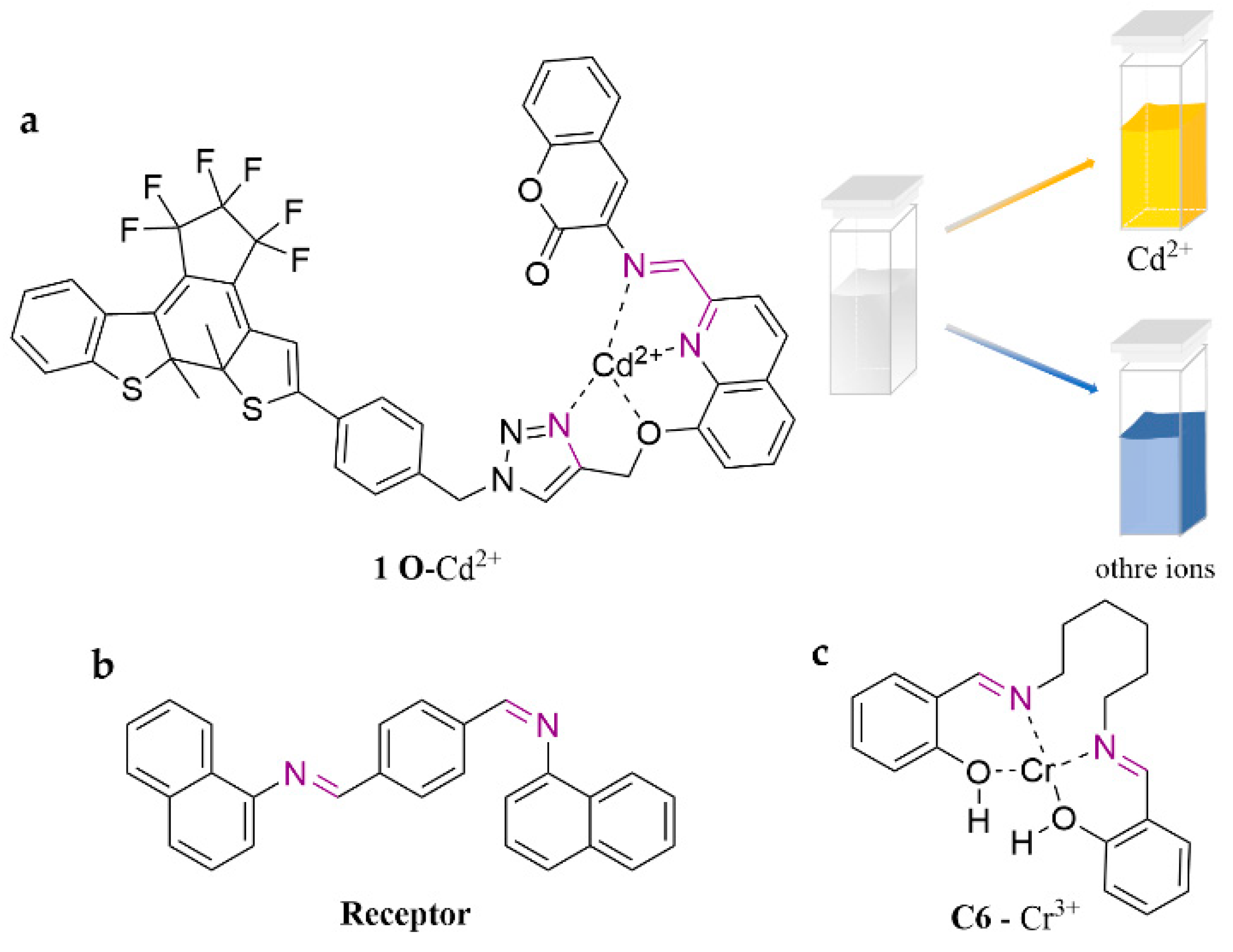
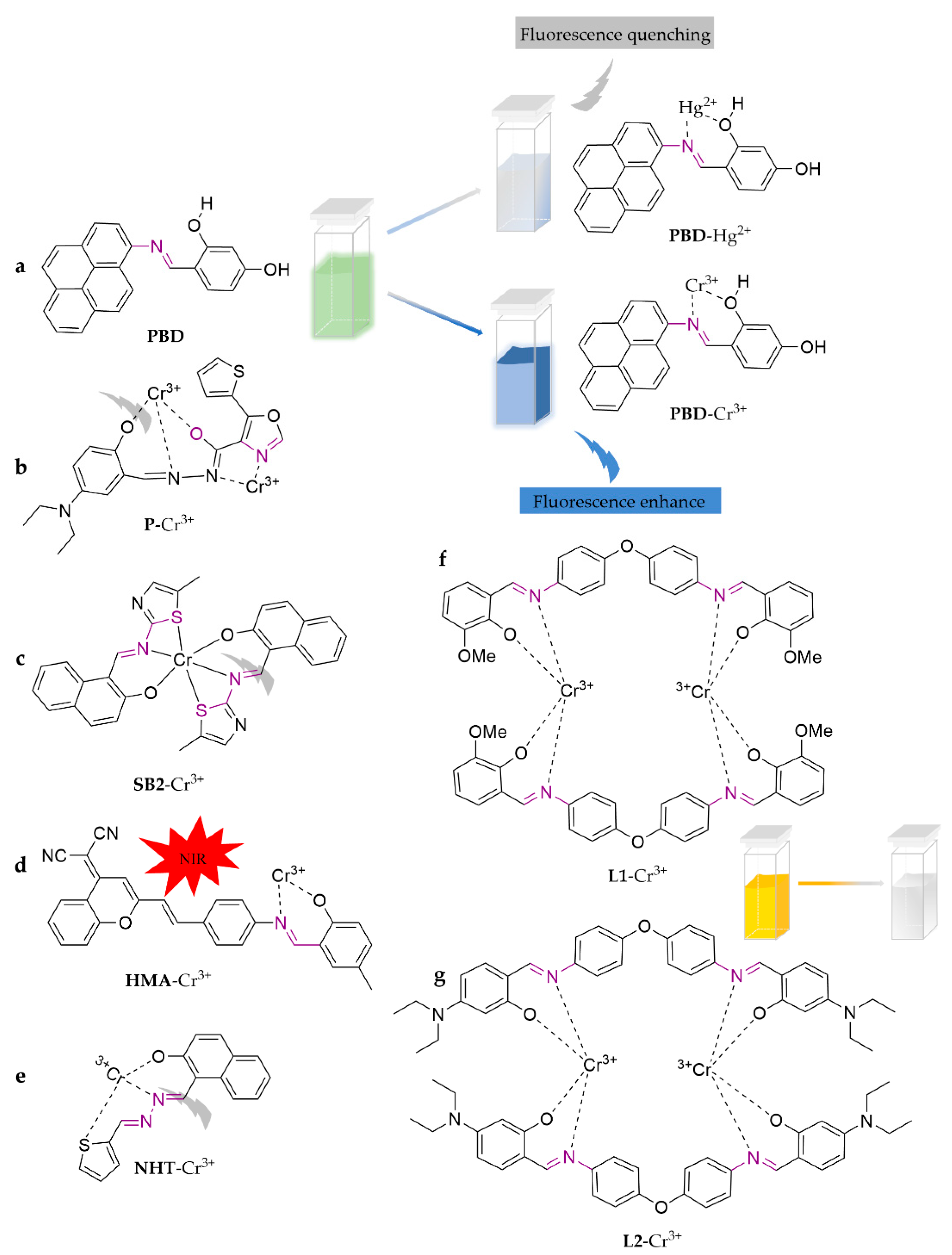
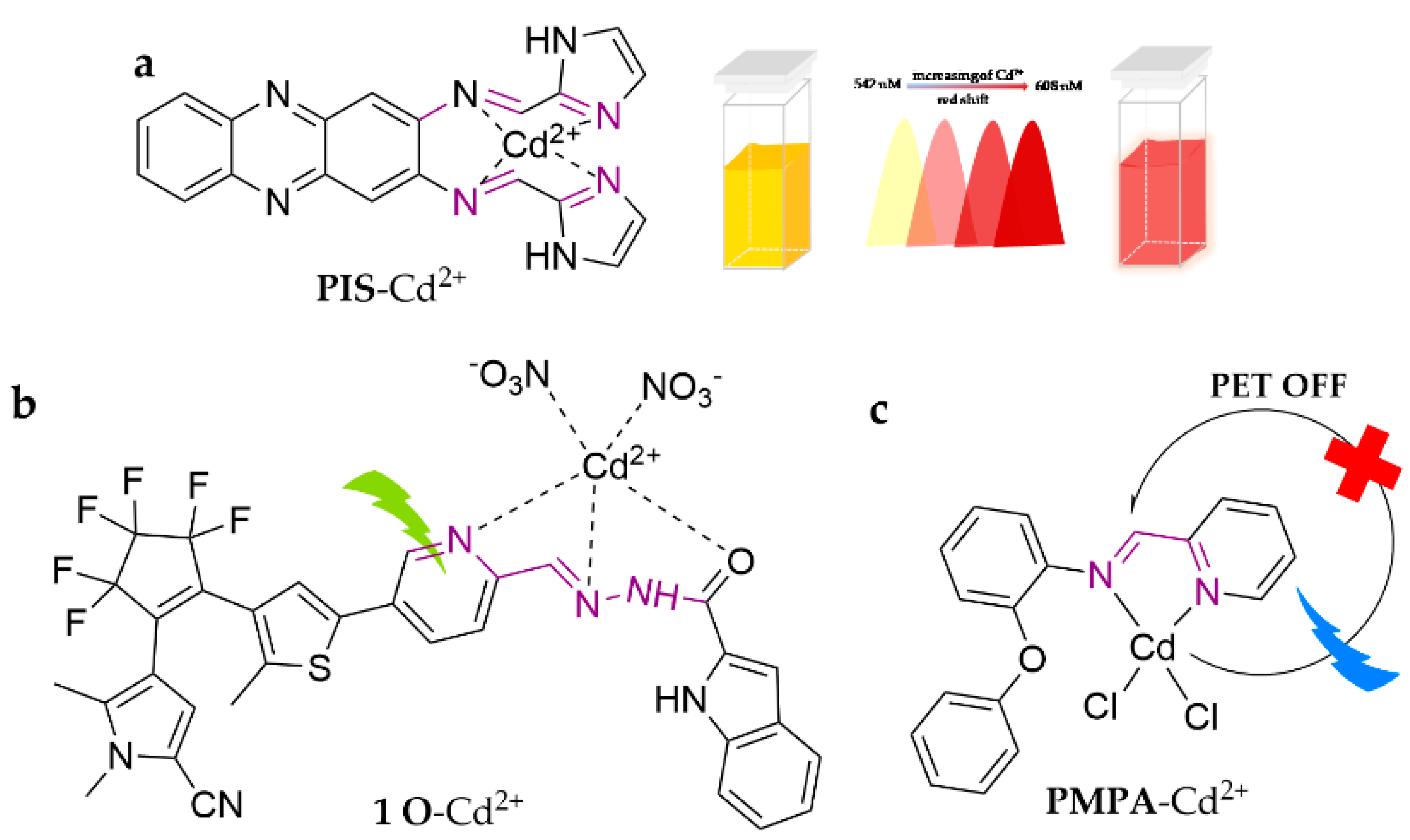


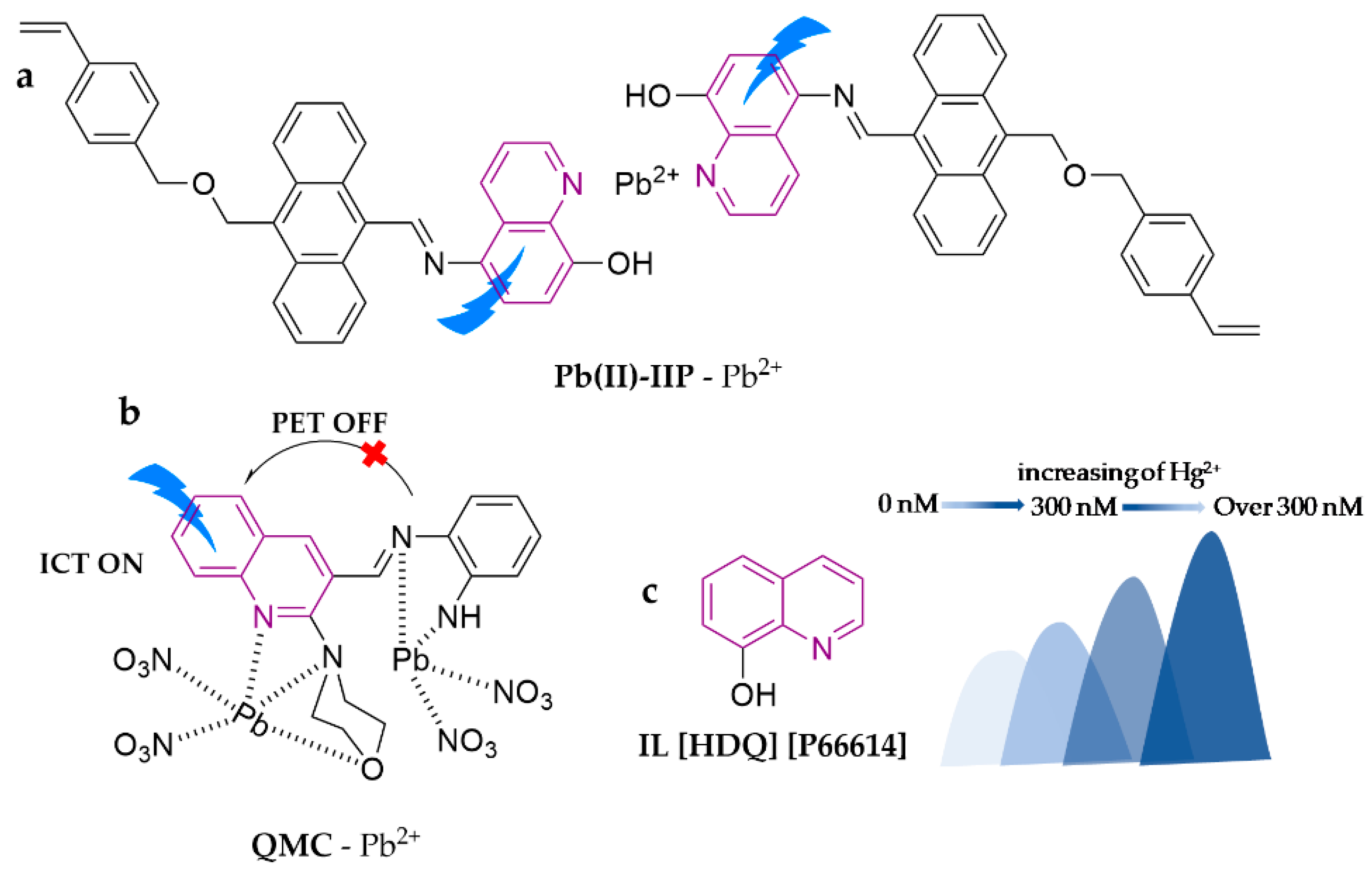
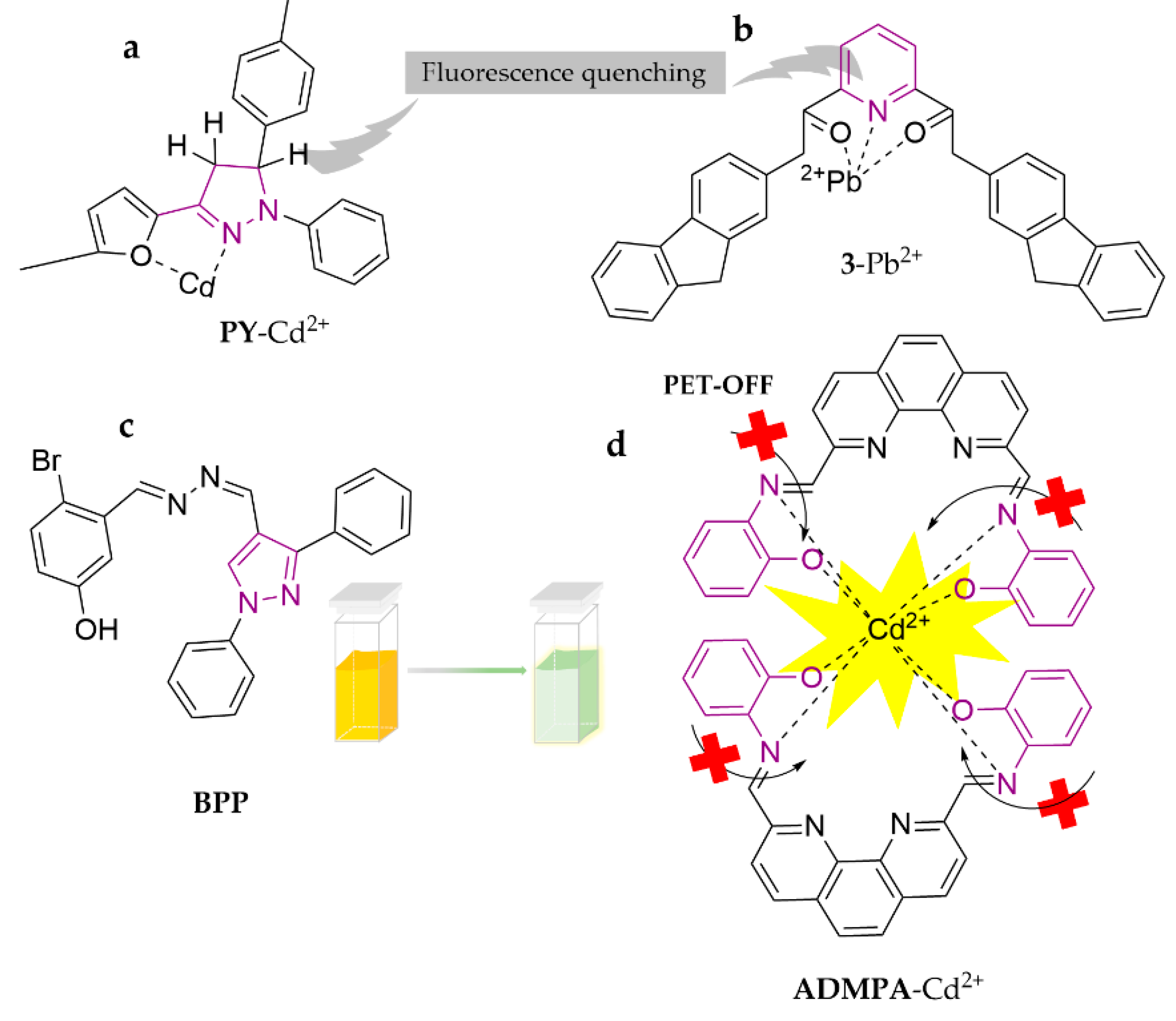
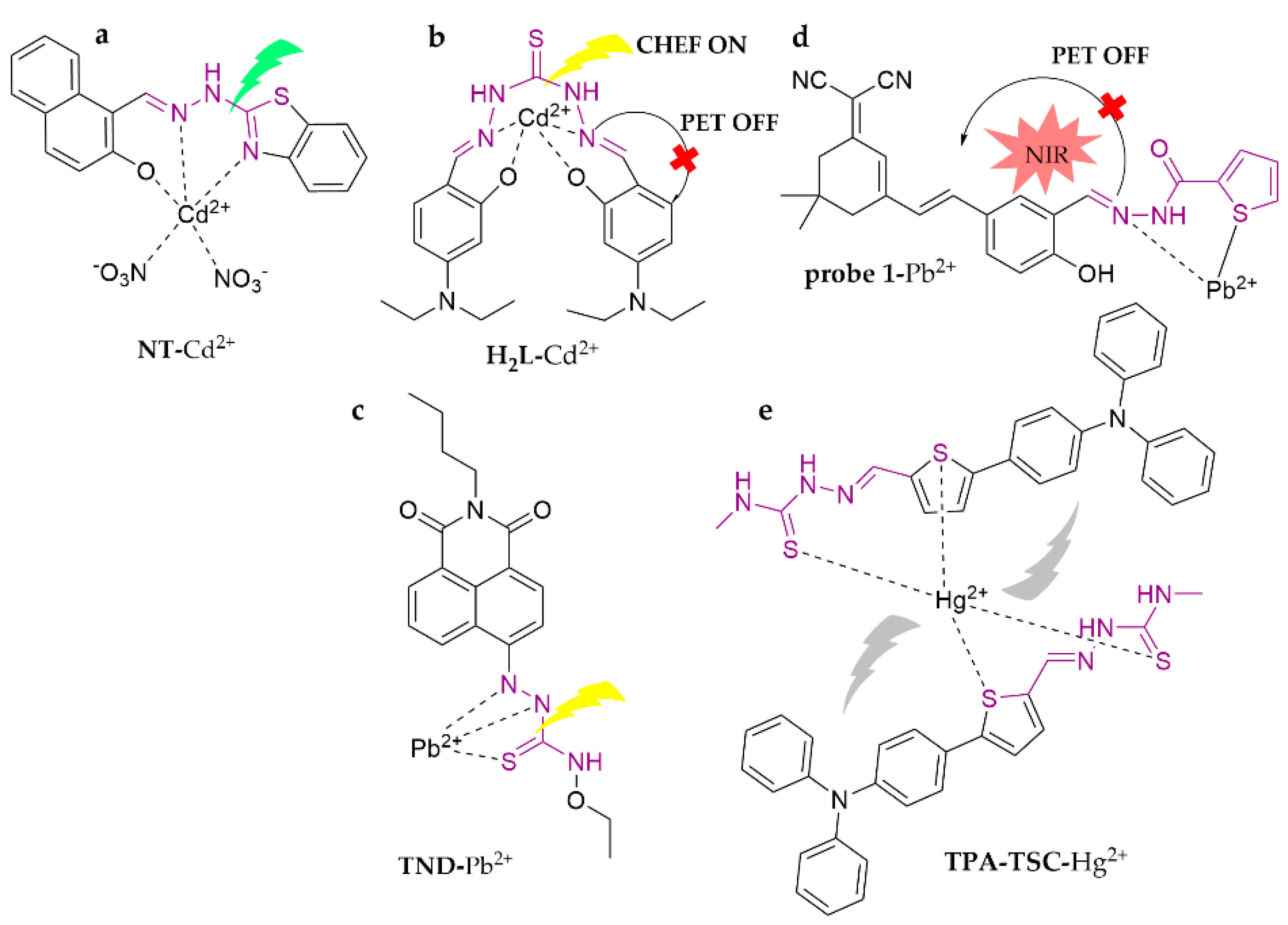
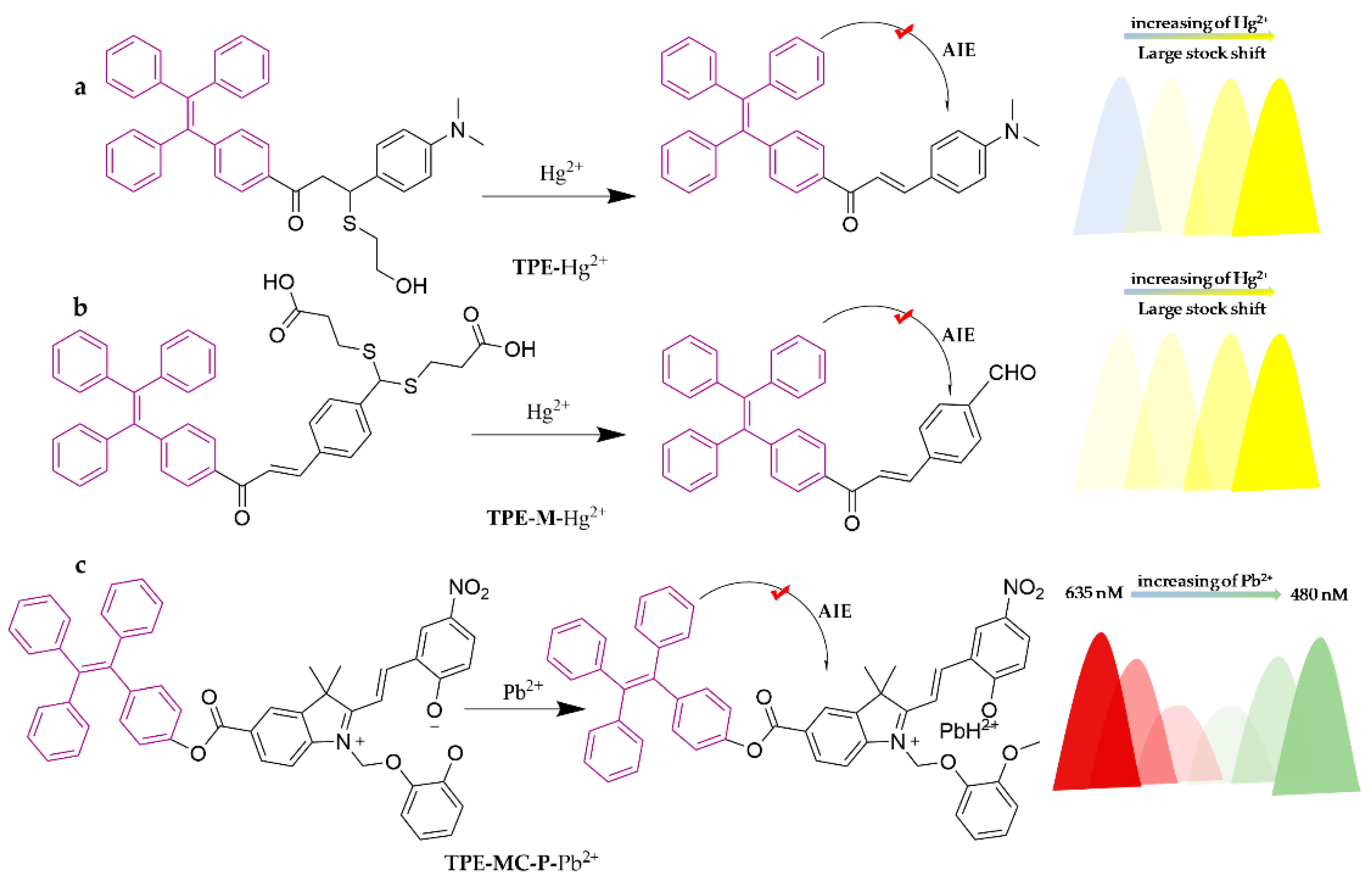

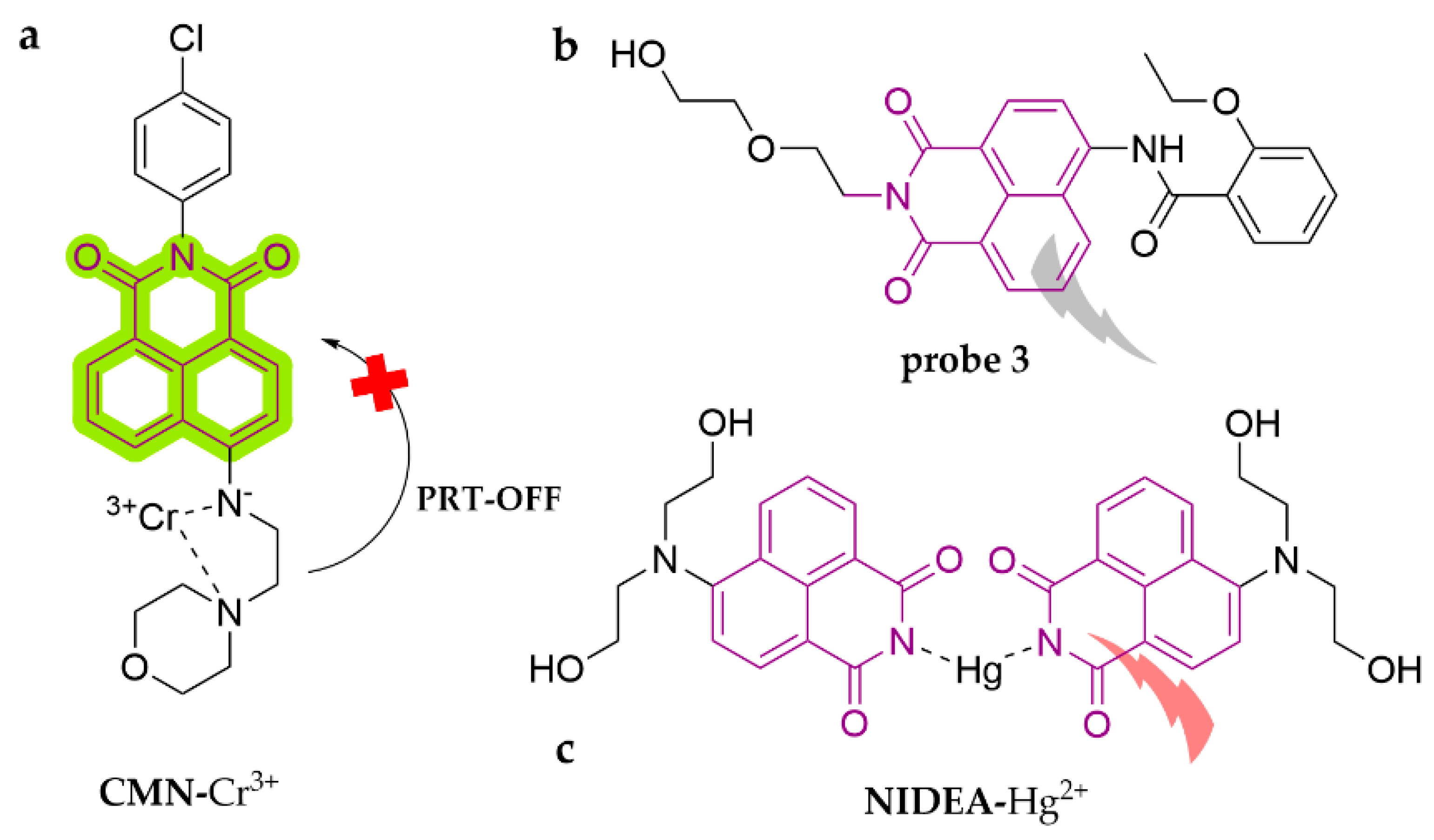

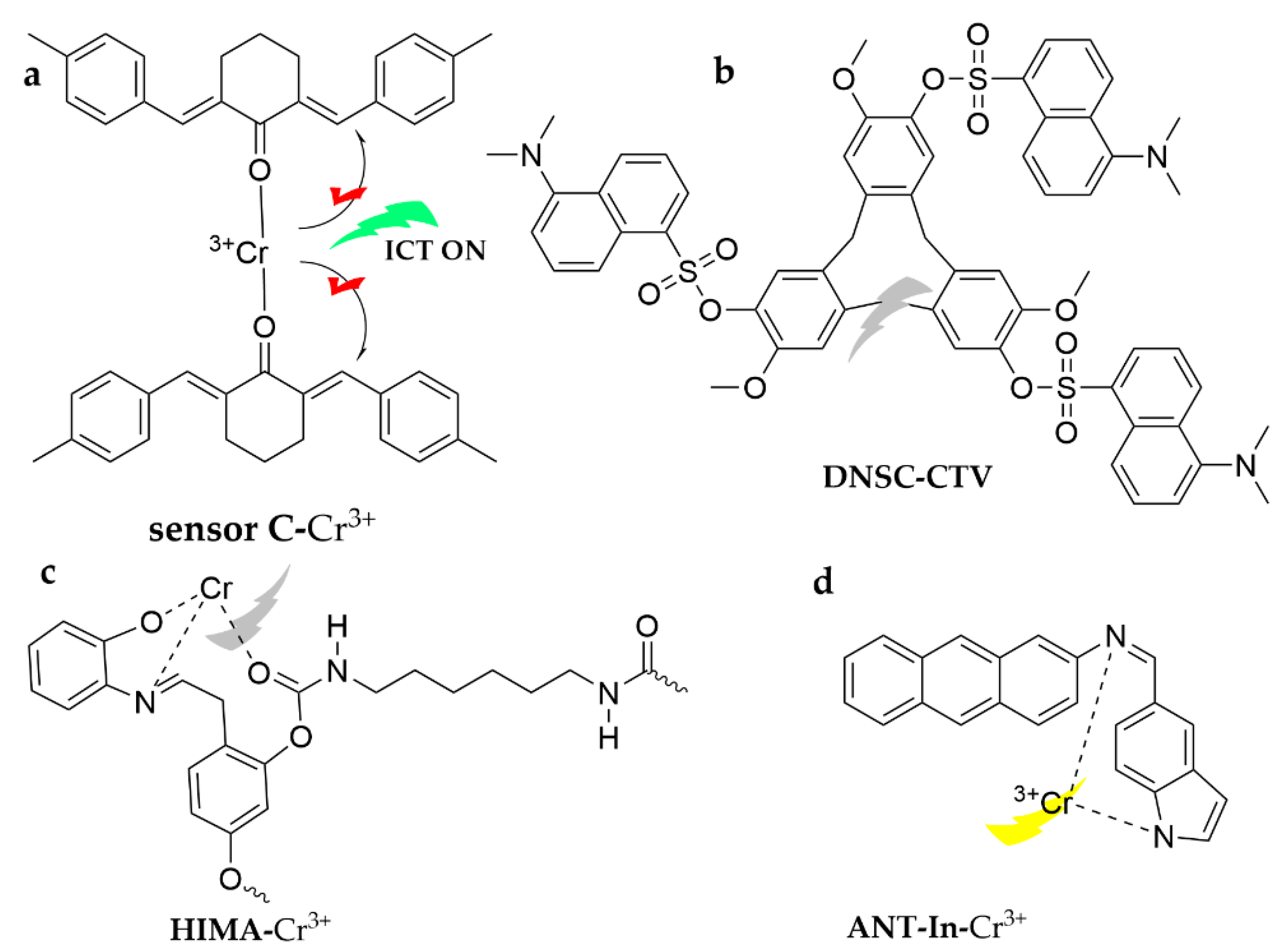

| Probe | Analytes | LOD | Working Solution | Application | Ref. |
|---|---|---|---|---|---|
| R1 | Pb2+ | 2.7 × 10−9 M | DMSO | seafood | [40] |
| REHBA | Pb2+ | 0.73 μM | Tris-HCl (10 mM, pH = 7.0) | water | [41] |
| 3 | Hg2+ | 15.80 nM | MeOH/HEPES (1:9, v/v, pH = 7.4) | cell imaging | [42] |
| HL-CHO | Cr3+ | ||||
| p-RPT | Hg2+ | 1.2 × 10−8 M | THF/H2O (3:2, v/v) | water | [43] |
| d114 | Hg2+ | 8.6 nM | MeOH/H2O (1:1, v/v) | drinking water | [44] |
| FO511 | Hg2+ | 92.7 nM | HEPES (10 mM, pH = 7.2) | cell imaging | [45] |
| RBLY | Hg2+ | 0.34 μM | EtOH/H2O (1:5, v/v) | water | [46] |
| R6GH | Pb2+ | 0.02 μM | THF/H2O (1:1, v/v) | seafood | [47] |
| NA-RhB | Pb2+ | 0.00001 g·L−1 | water | [48] | |
| FP | Pb2+ | 0.42 μM | EtOH/H2O (99:1, v/v) | tap water | [49] |
| Cd2+ | 0.53 μM | ||||
| RhBQ | Cr3+ | 2.12 × 10−8 M | ACN/H2O (9:1, v/v) | cell imaging | [50] |
| RFC | Cr3+ | 0.0052 ppm | MeOH/H2O (99:1, v/v) | cell imaging | [51] |
| 1 O | Cd2+ | ACN | [52] | ||
| Receptor | Cr3+ | 3.92 μM | H2O | zebrafish | [53] |
| C6 | Cr3+ | 13.3 µM | ACN | tap water | [54] |
| PBD | Cr3+ | 0.32 μM | EtOH/H2O (1:1, v/v) | water, soil | [55] |
| Hg2+ | 1.93 μM | ||||
| P | Cr3+ | 9.82 × 10−9 M | DMF/H2O (9:1, v/v) | water | [56] |
| SB2 | Cr3+ | 0.5 µM | MeOH/H2O (3:1, v/v) | soil | [57] |
| HMA | Cr3+ | 5.63 × 10−7 M | DMSO/H2O (9:1, v/v) | water | [58] |
| NHT | Cr3+ | 41 nM | HEPES (0.2 mM, pH = 7.2) | cell imaging | [59] |
| L1 | Cr3+ | 1.12 × 10−7 M | ACN/H2O (1:1, v/v) | water | [60] |
| L2 | Cr3+ | 7.73 × 10−7 M | |||
| 1 O | Cd2+ | 5.74 × 10−7 M | THF | water | [62] |
| PIS | Cd2+ | 2.10 × 10−8 M | ACN/HEPES (10 mM, pH = 7.4) (1:4, v/v) | zebrafish | [61] |
| PMPA | Cd2+ | 0.12 mM | ACN | water | [63] |
| DBTBH | Pb2+ | 4.49 × 10−8 M | THF/Tris-HCl (10 mM, 1 mM KI, pH = 7.4) (1:9, v/v) | water | [64] |
| BSBBT | Pb2+ | 2.23 × 10−6 M | DMSO/H2O (3:7, v/v) | [65] | |
| L | Pb2+ | 9 × 10−7 M | MeOH/Tris (1:1, v/v) | water | [66] |
| probe 1 | Cd2+ | 0.055 μM | ACN | water, bean sprouts | [67] |
| QTPY | Cd2+ | 3.5 × 10−8 M | DMF/H2O (4:6, v/v) | [68] | |
| L | Cd2+ | 14.8 nM | ACN/H2O (8:2, v/v) | water | [69] |
| DDTQ | Cd2+ | 126 nM | H2O | cell imaging | [70] |
| Pb(II)-IIP | Pb2+ | 2.1 μg∙L−1 | H2O | water | [71] |
| QMC | Pb2+ | 13 μM | ACN/H2O (1:1, v/v) | milk, wine | [72] |
| IL [HDQ] [P66614] | Hg2+ | 0.8 nM | EtOH | water, seafood | [73] |
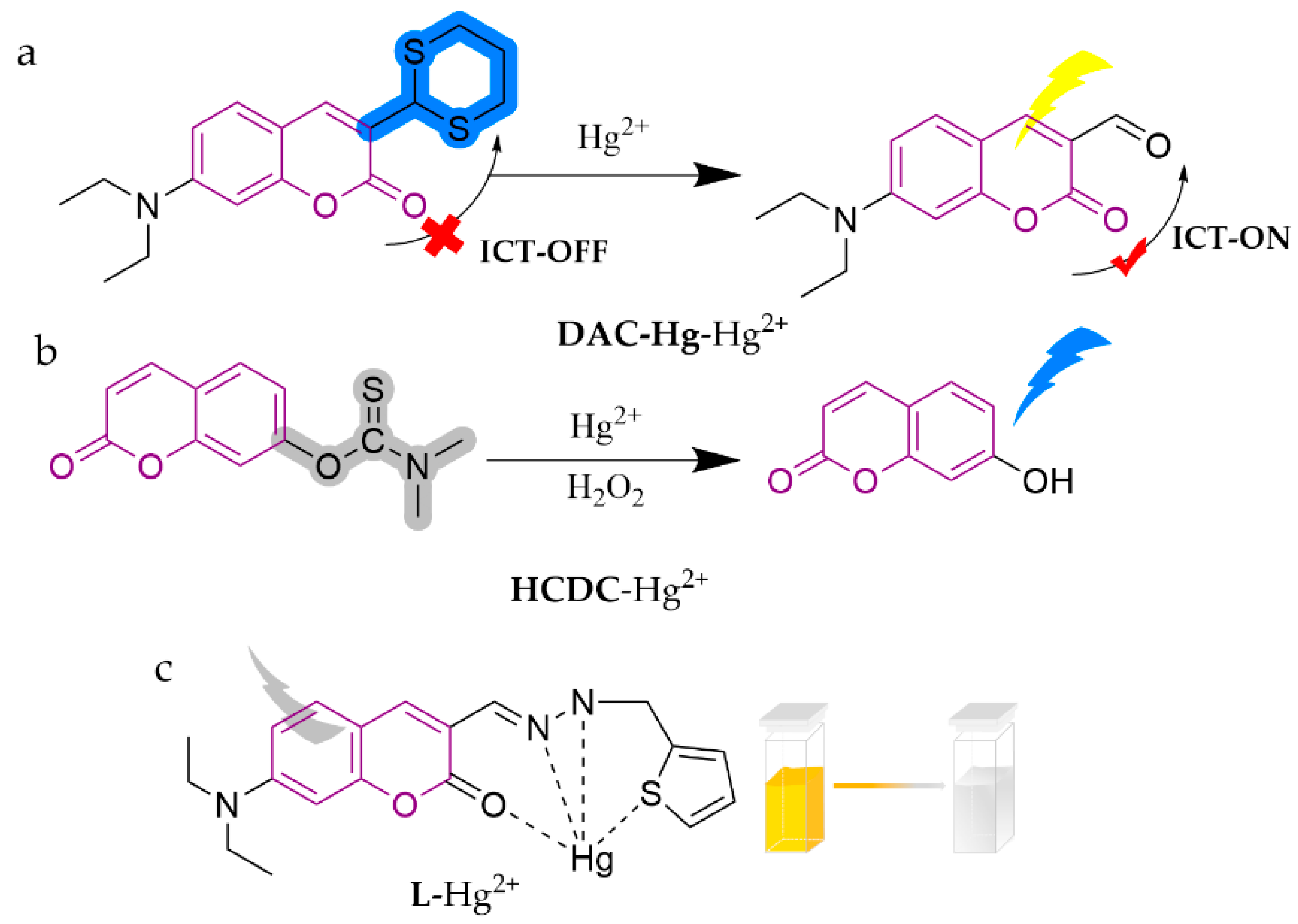
| DAC-Hg | Hg2+ | 5.0 nM | PBS | water, seafood, soil | [74] |
| HCDC | Hg2+ | 0.3 nM | HEPES (5 M, pH = 7.4) | water | [75] |
| L | Hg2+ | 5.15 nM | bis-tris (10−2 M, pH = 7.0, 0.5% DMSO) | water | [76] |
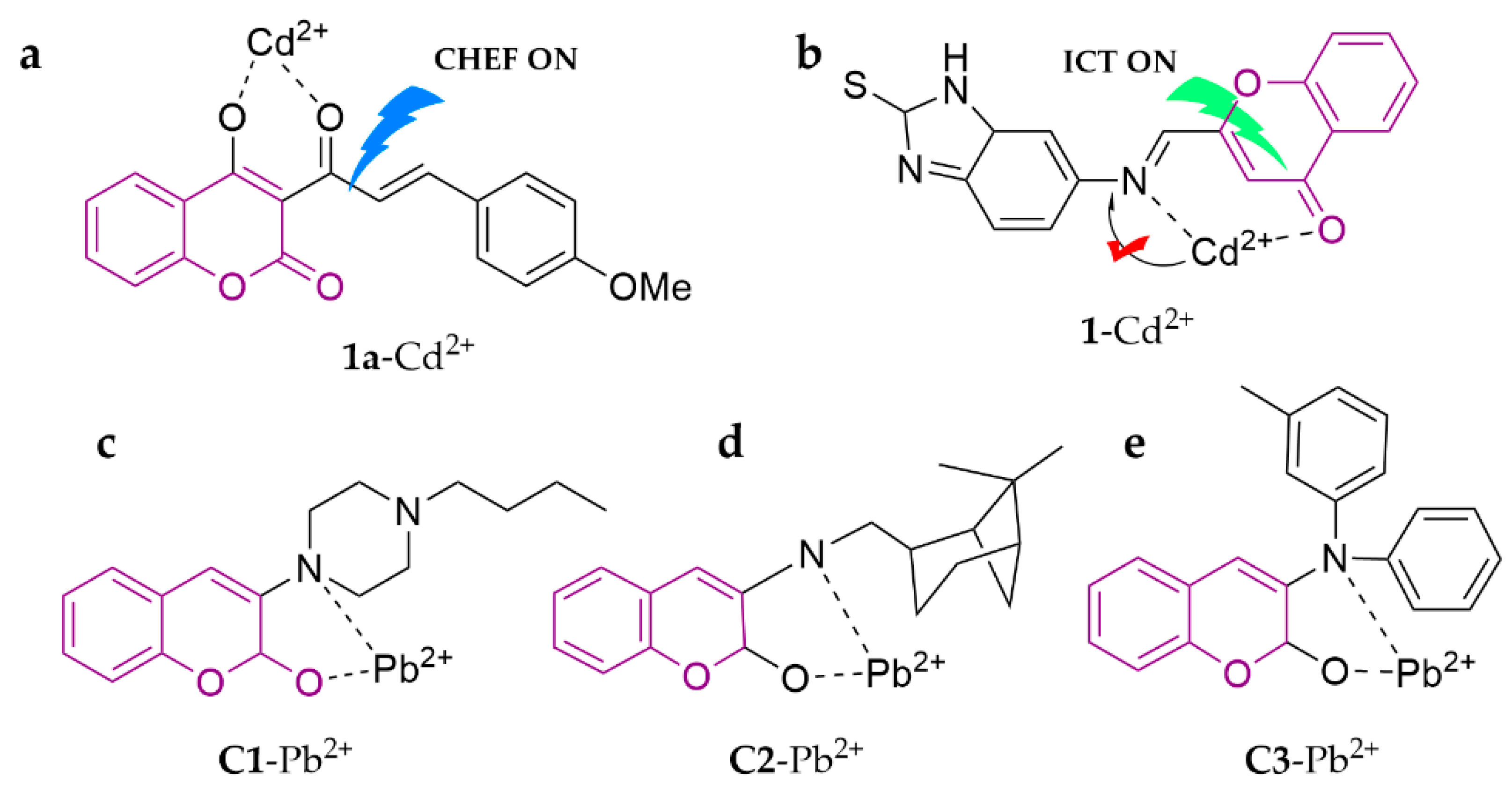
| 1a | Cd2+ | 5.84 × 10−8 M | HEPES (20 mM, ACN/H2O, 3:7, v/v, pH = 7.0) | [77] | |
| 1 | Cd2+ | 0.114 μM | THF/H2O (1:1, v/v) | [78] | |
| C1-C3 | Pb2+ | 0.009–0.0015 | ACN | [79] | |
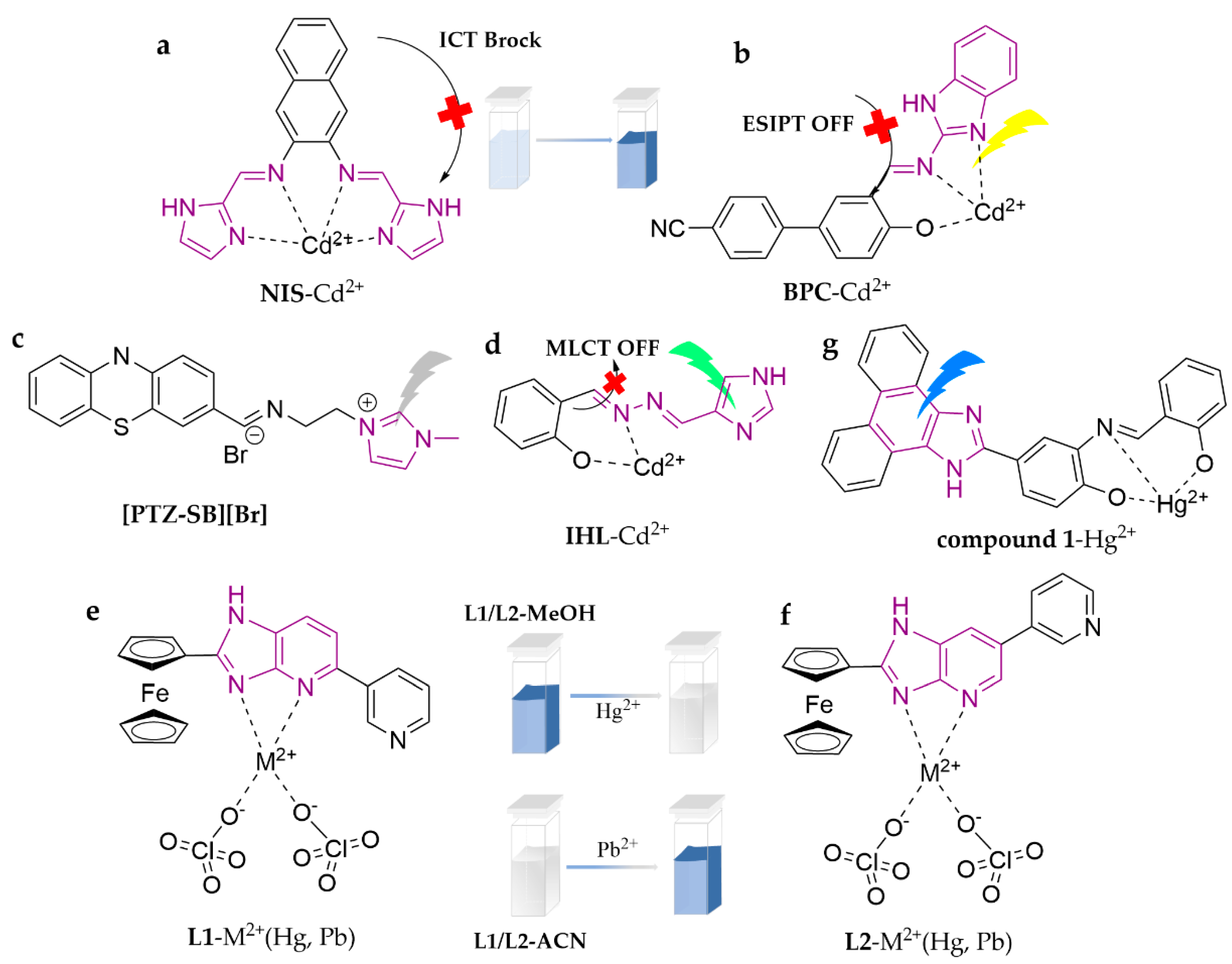
| NIS | Cd2+ | 3.87 × 10−7 M | HEPES (EtOH/H2O = 9:1, v/v, pH = 7.4) | zebrafish | [80] |
| BPC | Cd2+ | 1.05 × 10−8 M | ACN/Tris-HCl (3:2, v/v, pH = 7.4) | water | [81] |
| [PTZ-SB][Br] | Cd2+ | 3.8 × 10−7 M | THF/H2O (1:9, v/v) | water | [82] |
| IHL | Cd2+ | 0.4 × 10−10 M | DMSO/H2O (9:1, v/v) | zebrafish | [83] |
| L1 | Hg2+ | 7.6 × 10−6 M | ACN/H2O (1:1, v/v) | [84] | |
| Pb2+ | 8.5 × 10−6 M | ACN | |||
| L2 | Hg2+ | 6.7 × 10−6 M | ACN/H2O (1:1, v/v) | ||
| Pb2+ | 2.5 × 10−6 M | ACN | |||
| compound 1 | Hg2+ | 45.76 nM | DMF/PBS (1:4, v/v, pH = 7.4) | water | [85] |
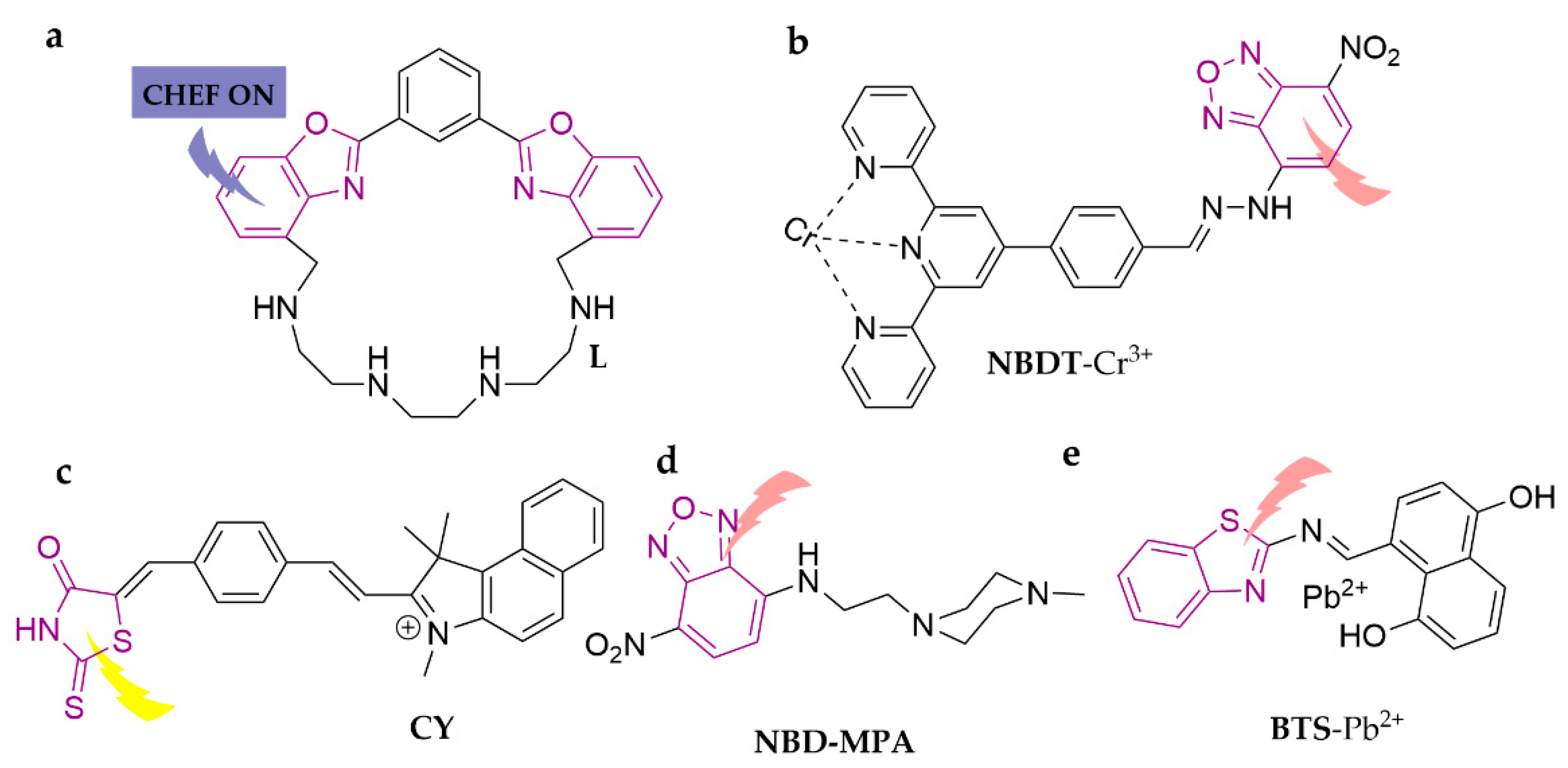
| L | Cd2+ | 0.03 ppm | ACN/H2O (4:1, v/v) | [86] | |
| NBDT | Cr3+ | 0.041 μM | DMSO/H2O (9:2, v/v) | zebrafish | [87] |
| CY | Hg2+ | 1.61 × 10−7 M | DMSO/H2O (7:3, v/v) | water | [88] |
| BTS | Pb2+ | 0.67 µM | DMSO/H2O (1:4, v/v) | water | [89] |
| NBD-MPA | Hg2+ | 9.2 × 10−7 M | ACN/HEPES (1:9, v/v) | water, soil, green tea, seafood | [90] |
| PY | Cd2+ | 0.09 μM | MeOH (pH = 7, PBS) | water | [91] |
| BPP | 0.02 nM | DMSO/H2O (9:1, v/v) | zebrafish | [92] | |
| ADMPA | 29.3 nM | DMF/H2O (3:7, v/v) | water | [93] | |
| 3 | Pb2+ | 2.31 × 10−6 M | ACN | [94] | |
| NT | Cd2+ | ACN | [95] | ||
| H2L | Cd2+ | 2.67 × 10−8 M | MeOH | food | [96] |
| TND | Pb2+ | 4.7 nM | ACN/H2O (1:1, v/v) | water | [97] |
| probe 1 | Pb2+ | 1.65 nM | ACN/EtOH/HEPES (1:1:2, v/v/v, pH = 7.0) | water | [98] |
| TPA-TSC | Hg2+ | 0.14 nM | ACN/PBS (1:1, v/v, pH = 7.4) | sea bass, water, swordfish | [99] |
| TPE-Hg | Hg2+ | 7.548×10−7 M | THF/HEPES (20 mM, pH = 7.3) (1:9, v/v) | green tea, seafood | [100] |
| TPE-M | Hg2+ | 4.16×10−6 M | MeOH/PBS (20 mM, pH = 7.4) (3:7, v/v) | green tea, seafood | [101] |
| TPE-MC-P | Pb2+ | 0.27 μM | THF/H2O (1:9, v/v) | water | [102] |
| 3 TS | Hg2+ | 1.03 × 10−8 M | EtOH/H2O (1:1, v/v) | water, soil, seafood | [103] |
| 1.1 | Hg2+ | MeOH/H2O | high-fructose corn syrup | [104] | |
| NAPABTH | Pb2+ | 1.06 ppm | DMSO | [105] | |
| CMN | Cr3+ | 0.68 μM | MeOH | [106] | |
| probe 3 | Cr6+ | 1.15 μM | ACN/HEPES (10 mM, pH = 7.4) (1:9, v/v) | water | [107] |
| NIDEA | Hg2+ | 46.7 nM | HEPES (10 mM, pH = 7.4) | water | [108] |
| CM 1 | Cd2+ | 19.25 nM | H2O | water | [109] |
| SAF | 1.5 × 10−7 M | ACN/H2O (95:5, v/v) | water | [110] | |
| PM | Cd2+ | 53 nM | H2O/dioxane (1/19, v/v) | water, rice | [111] |
| sensor C | Cr3+ | 2.3 × 10−9 M | ACN | water | [112] |
| DNSC-CTV | ACN | [113] | |||
| HIMA | 7.98 × 10−7 M | DMF/H2O (1:2, v/v) | drinking water | [114] | |
| ANT-In | 0.2 µM | ACN/HEPES (7:3, v/v, pH = 7) | drinking water | [115] | |
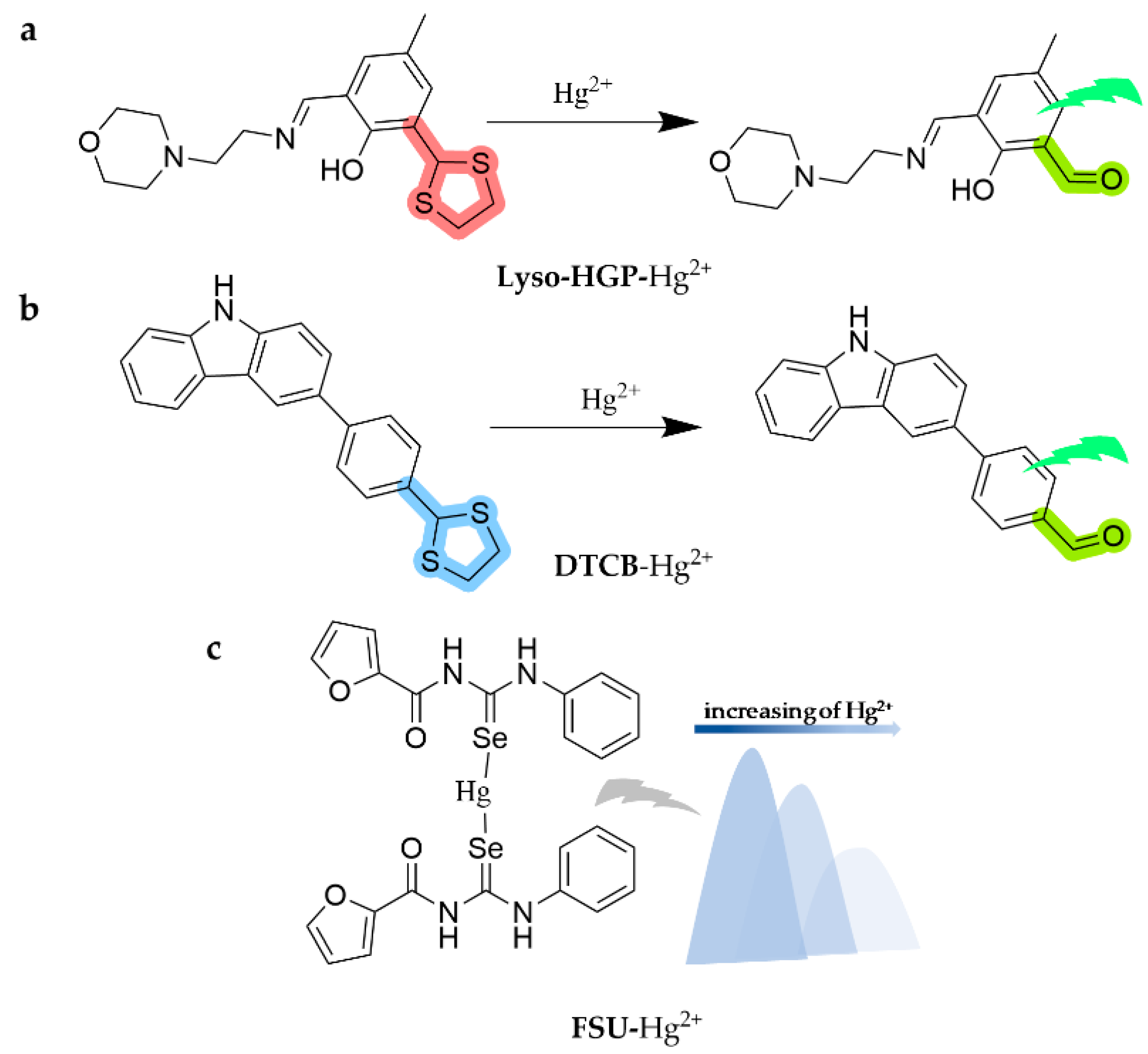
| Lyso-HGP | Hg2+ | 6.82 nM | HEPES (10 mM, pH = 7.0, 1%DMSO) | water | [116] |
| DTCB | 2.05 × 10−7 M | THF/H2O | water | [117] | |
| FSU | 7.35 × 10−7 M | DMSO/H2O (95:5, v/v) | [118] | ||
| sensor 3 | Pb2+ | 6 × 10−7 M | ACN | water | [119] |
| 1 | 4.1 nM | THF/MeOH (4:1, v/v) | [121] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, L.; Yan, F.; Chen, G.; Huang, Y.; Huang, L.; Li, D. Recent Progress on Fluorescent Probes in Heavy Metal Determinations for Food Safety: A Review. Molecules 2023, 28, 5689. https://doi.org/10.3390/molecules28155689
Lai L, Yan F, Chen G, Huang Y, Huang L, Li D. Recent Progress on Fluorescent Probes in Heavy Metal Determinations for Food Safety: A Review. Molecules. 2023; 28(15):5689. https://doi.org/10.3390/molecules28155689
Chicago/Turabian StyleLai, Liqing, Fang Yan, Geng Chen, Yiwen Huang, Luqiang Huang, and Daliang Li. 2023. "Recent Progress on Fluorescent Probes in Heavy Metal Determinations for Food Safety: A Review" Molecules 28, no. 15: 5689. https://doi.org/10.3390/molecules28155689
APA StyleLai, L., Yan, F., Chen, G., Huang, Y., Huang, L., & Li, D. (2023). Recent Progress on Fluorescent Probes in Heavy Metal Determinations for Food Safety: A Review. Molecules, 28(15), 5689. https://doi.org/10.3390/molecules28155689






