Terroir Influence on Polyphenol Metabolism from Grape Canes: A Spatial Metabolomic Study at Parcel Scale
Abstract
1. Introduction
2. Results
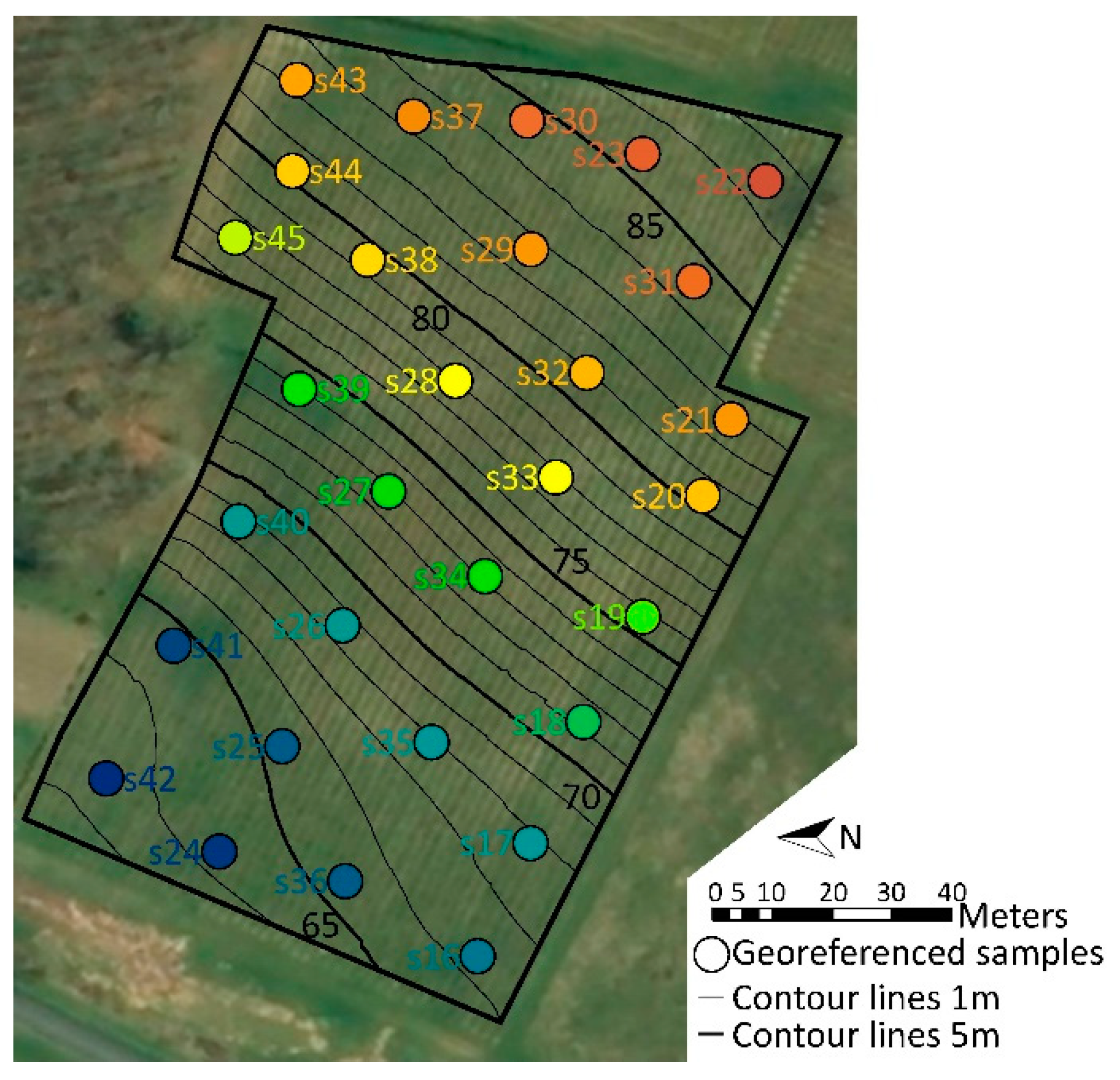
2.1. Soil Properties and Topography Revealed Contrasted Zones in the Vineyard
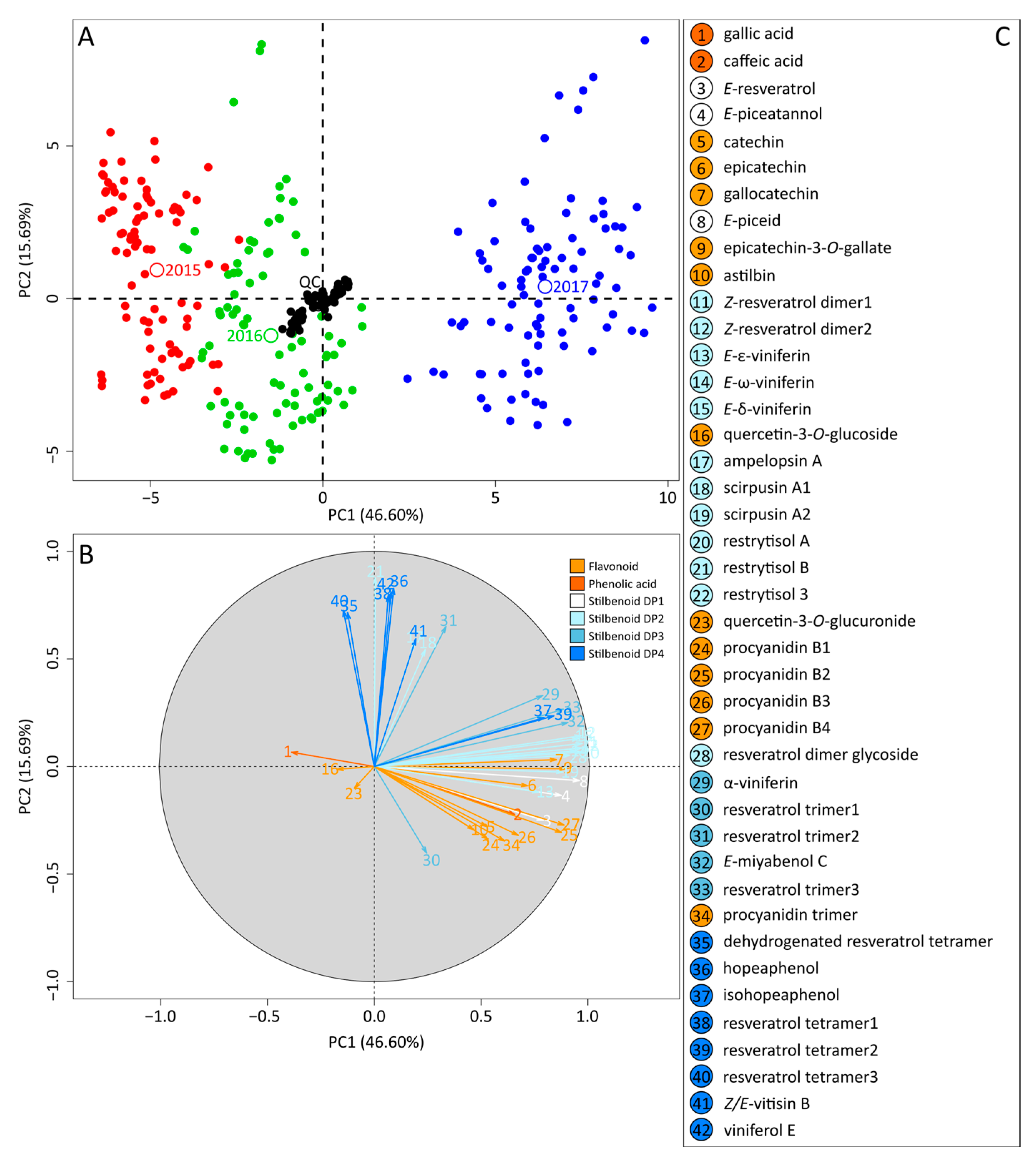
2.2. Metabolomics Study of Grape Canes over the Three Vintages
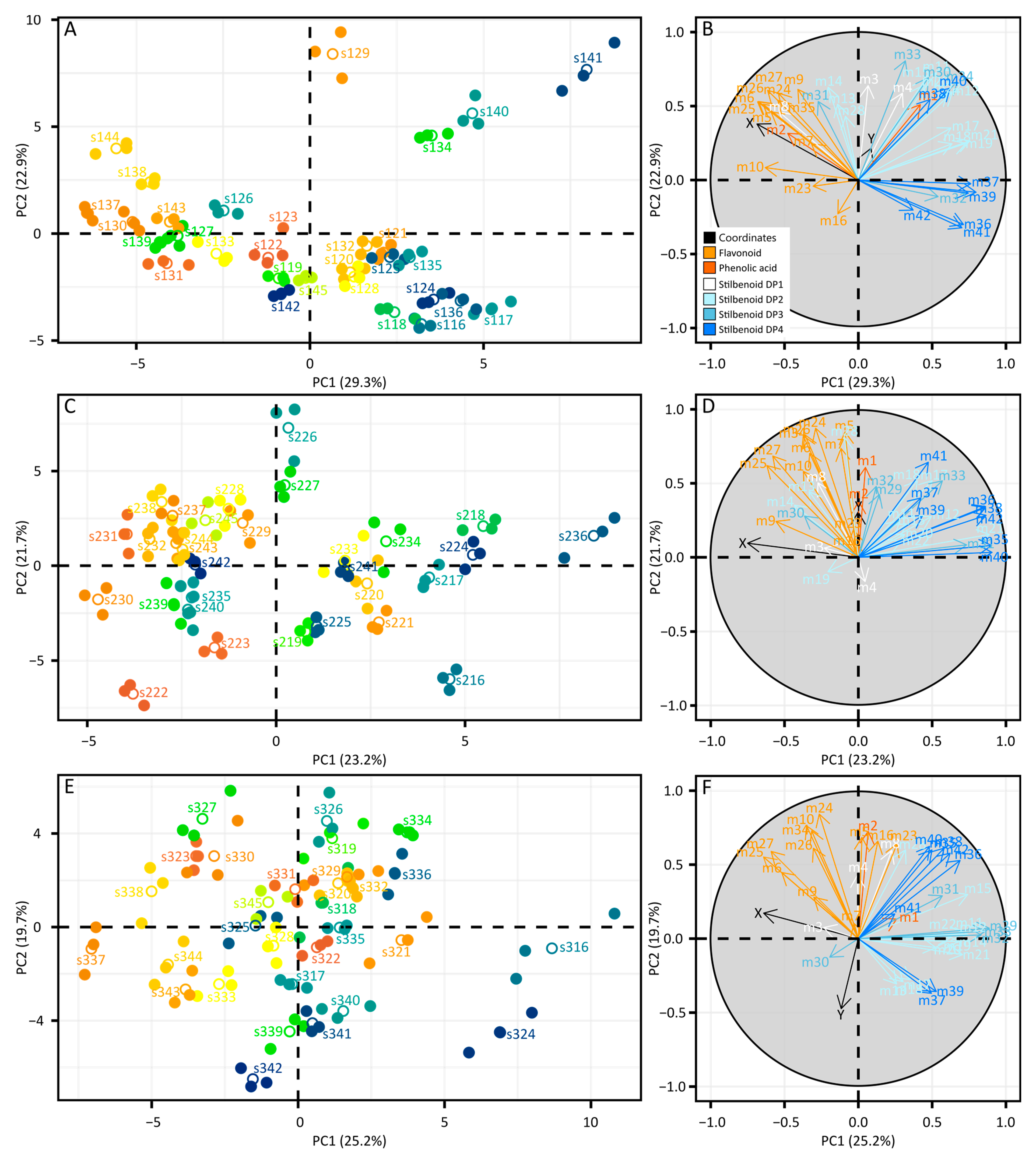
2.3. Intra-Vintage Variations in Metabolomics Data
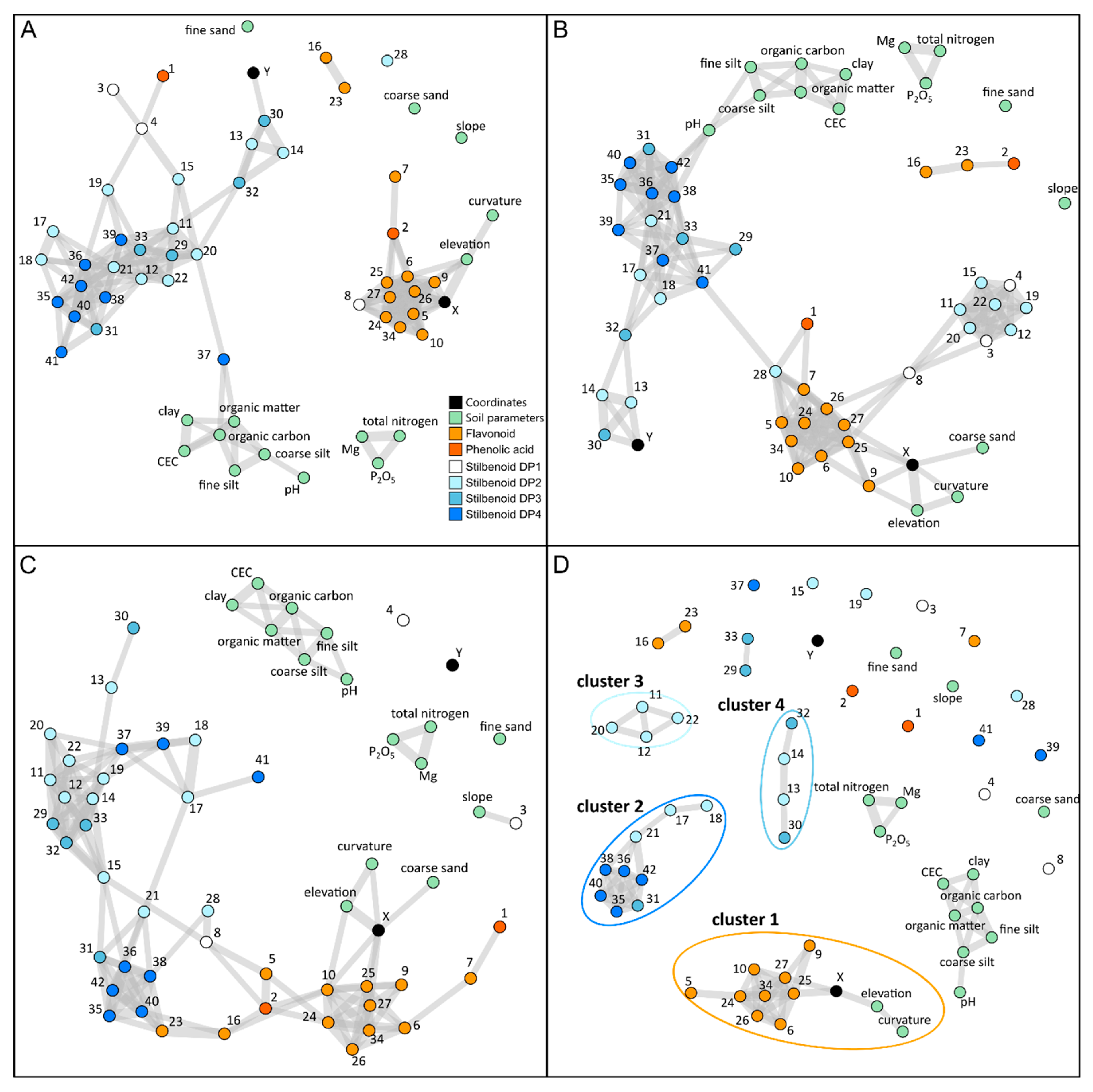
2.4. Correlation-Based Metabolite Networks to Assess the Structuration of Polyphenol Metabolism with Topography and Soil Parameters
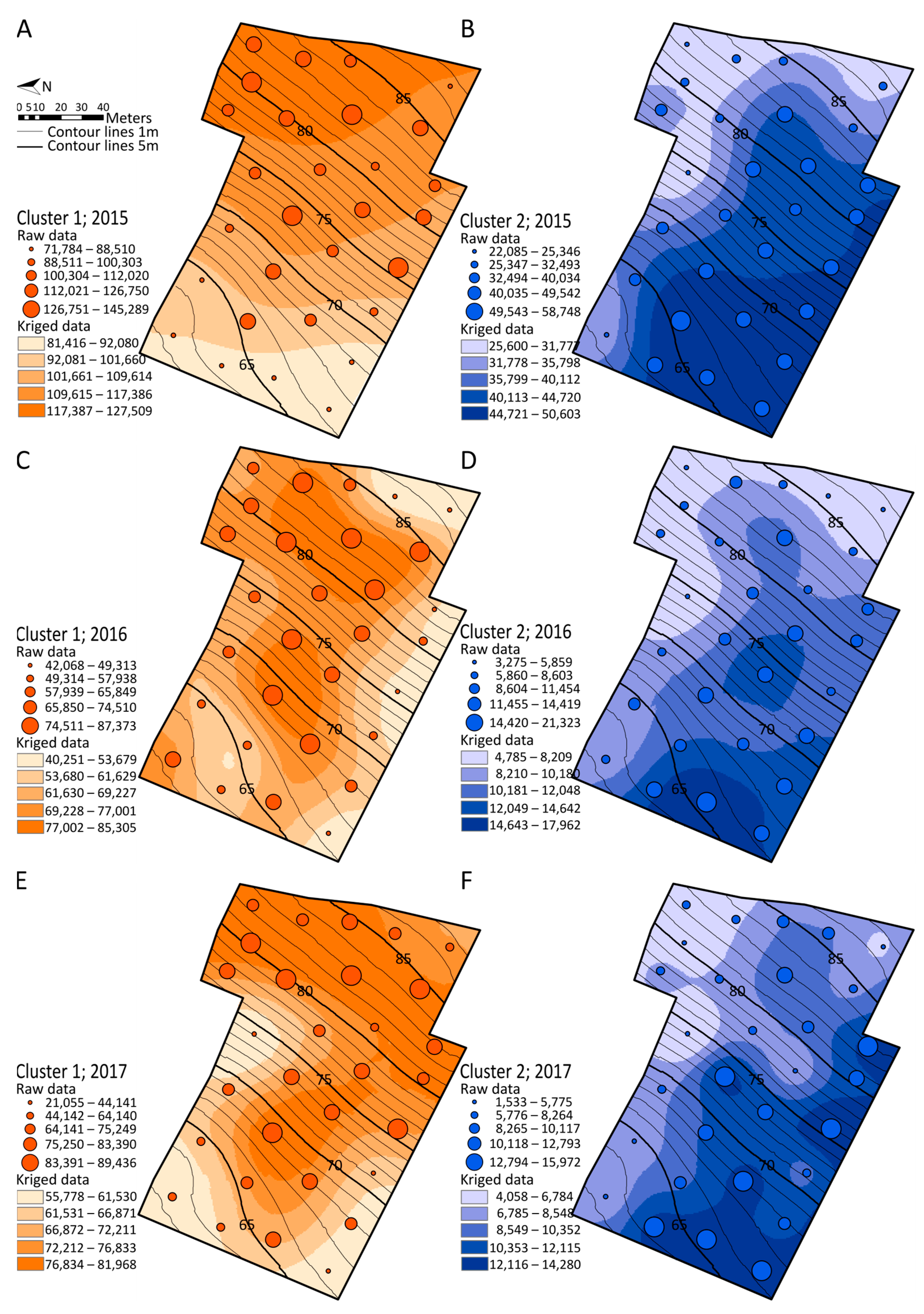
2.5. Spatialization Studies of Metabolomics Clusters
3. Discussion
3.1. Experimental Design to Study Soil- and Topography-Based Terroir Influence
3.2. Soil- and Topography-Based Terroir Signature Emerged behind the Vintage Effect
3.3. Correlation-Based Networks Give an Overview of Metabolomic Data Structuration in Response to Environmental Changes
3.4. Specific Metabolite Clusters Correlated with Topography but Not Soil Composition
3.5. Mapping Metabolomics Data in Agronomic Studies
4. Materials and Methods
4.1. Vineyard Features, Environmental Parameters, and Geo-Referencing
4.2. Soil Material and Particle Size Analysis
4.3. Extraction, Analysis, and Identification of Metabolites
4.4. Metabolomic Data Analysis
4.5. Statistical Analysis
4.6. Spatialization
4.7. Chemicals
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Jeandet, P.; Delaunois, B.; Conreux, A.; Donnez, D.; Nuzzo, V.; Cordelier, S.; Clément, C.; Courot, E. Biosynthesis, Metabolism, Molecular Engineering, and Biological Functions of Stilbene Phytoalexins in Plants. BioFactors 2010, 36, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Billet, K.; Houillé, B.; de Bernonville, T.D.; Besseau, S.; Oudin, A.; Courdavault, V.; Delanoue, G.; Guérin, L.; Clastre, M.; Giglioli-Guivarc’h, N.; et al. Field-Based Metabolomics of Vitis vinifera L. Stems Provides New Insights for Genotype Discrimination and Polyphenol Metabolism Structuring. Front. Plant Sci. 2018, 9, 798. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.W.; Fong, H.H.S.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G.; et al. Cancer Chemopreventive Activity of Resveratrol, a Natural Product Derived from Grapes. Science 1997, 275, 218–220. [Google Scholar] [CrossRef]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol Improves Health and Survival of Mice on a High-Calorie Diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Romain, C.; Gaillet, S.; Carillon, J.; Vidé, J.; Ramos, J.; Izard, J.-C.; Cristol, J.-P.; Rouanet, J.-M. Vineatrol and Cardiovascular Disease: Beneficial Effects of a Vine-Shoot Phenolic Extract in a Hamster Atherosclerosis Model. J. Agric. Food Chem. 2012, 60, 11029–11036. [Google Scholar] [CrossRef]
- Mattivi, F.; Guzzon, R.; Vrhovsek, U.; Stefanini, M.; Velasco, R. Metabolite Profiling of Grape: Flavonols and Anthocyanins. J. Agric. Food Chem. 2006, 54, 7692–7702. [Google Scholar] [CrossRef] [PubMed]
- Chira, K.; Schmauch, G.; Saucier, C.; Fabre, S.; Teissedre, P.-L. Grape Variety Effect on Proanthocyanidin Composition and Sensory Perception of Skin and Seed Tannin Extracts from Bordeaux Wine Grapes (Cabernet Sauvignon and Merlot) for Two Consecutive Vintages (2006 and 2007). J. Agric. Food Chem. 2009, 57, 545–553. [Google Scholar] [CrossRef]
- Pinasseau, L.; Vallverdú-Queralt, A.; Verbaere, A.; Roques, M.; Meudec, E.; Le Cunff, L.; Péros, J.-P.; Ageorges, A.; Sommerer, N.; Boulet, J.-C.; et al. Cultivar Diversity of Grape Skin Polyphenol Composition and Changes in Response to Drought Investigated by LC-MS Based Metabolomics. Front. Plant Sci. 2017, 8, 1826. [Google Scholar] [CrossRef]
- Houillé, B.; Besseau, S.; Delanoue, G.; Oudin, A.; Papon, N.; Clastre, M.; Simkin, A.J.; Guérin, L.; Courdavault, V.; Giglioli-Guivarc’H, N.; et al. Composition and Tissue-Specific Distribution of Stilbenoids in Grape Canes Are Affected by Downy Mildew Pressure in the Vineyard. J. Agric. Food Chem. 2015, 63, 8472–8477. [Google Scholar] [CrossRef]
- Belda, I.; Zarraonaindia, I.; Perisin, M.; Palacios, A.; Acedo, A. From Vineyard Soil to Wine Fermentation: Microbiome Approximations to Explain the “Terroir” Concept. Front. Microbiol. 2017, 8, 821. [Google Scholar] [CrossRef]
- Cozzolino, D. Metabolomics in Grape and Wine: Definition, Current Status and Future Prospects. Food Anal. Methods 2016, 9, 2986–2997. [Google Scholar] [CrossRef]
- Anesi, A.; Stocchero, M.; Dal Santo, S.; Commisso, M.; Zenoni, S.; Ceoldo, S.; Tornielli, G.B.; Siebert, T.E.; Herderich, M.; Pezzotti, M.; et al. Towards a Scientific Interpretation of the Terroir Concept: Plasticity of the Grape Berry Metabolome. BMC Plant Biol. 2015, 15, 191. [Google Scholar] [CrossRef] [PubMed]
- Roullier-Gall, C.; Boutegrabet, L.; Gougeon, R.D.; Schmitt-Kopplin, P. A Grape and Wine Chemodiversity Comparison of Different Appellations in Burgundy: Vintage vs. Terroir Effects. Food Chem. 2014, 152, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Dal Santo, S.; Tornielli, G.B.; Zenoni, S.; Fasoli, M.; Farina, L.; Anesi, A.; Guzzo, F.; Delledonne, M.; Pezzotti, M. The Plasticity of the Grapevine Berry Transcriptome. Genome Biol. 2013, 14, r54. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Thorngate, J.H.; Richardson, P.M.; Mills, D.A. Microbial Biogeography of Wine Grapes Is Conditioned by Cultivar, Vintage, and Climate. Proc. Natl. Acad. Sci. USA 2014, 111, E139–E148. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Konate, M.; Sai, N.; Tesfamicael, K.G.; Cavagnaro, T.; Gilliham, M.; Breen, J.; Metcalfe, A.; Stephen, J.R.; De Bei, R.; et al. Global DNA Methylation Patterns Can Play a Role in Defining Terroir in Grapevine (Vitis vinifera Cv. Shiraz). Front. Plant Sci. 2017, 8, 1860. [Google Scholar] [CrossRef] [PubMed]
- Vaudour, E.; Costantini, E.; Jones, G.V.; Mocali, S. An Overview of the Recent Approaches to Terroir Functional Modelling, Footprinting and Zoning. SOIL 2015, 1, 287–312. [Google Scholar] [CrossRef]
- Toubiana, D.; Fernie, A.R.; Nikoloski, Z.; Fait, A. Network Analysis: Tackling Complex Data to Study Plant Metabolism. Trends Biotechnol. 2013, 31, 29–36. [Google Scholar] [CrossRef]
- US Department of Health and Human Services. Food and Drug Administration Bioanalytical Method Validation: Guidance for Industry; US Department of Health and Human Services: Washington, DC, USA, 2018; Volume 1043, p. 25. Available online: https://www.fda.gov/files/drugs/published/Bioanalytical-Method-Validation-Guidance-for-Industry.pdf (accessed on 4 June 2023).
- De Andrés-De Prado, R.; Yuste-Rojas, M.; Sort, X.; Andrés-Lacueva, C.; Torres, M.; Lamuela-Raventós, R.M. Effect of Soil Type on Wines Produced from Vitisvinifera L. Cv. Grenache in Commercial Vineyards. J. Agric. Food Chem. 2007, 55, 779–786. [Google Scholar] [CrossRef]
- Wong, D.C.J.; Matus, J.T. Constructing Integrated Networks for Identifying New Secondary Metabolic Pathway Regulators in Grapevine: Recent Applications and Future Opportunities. Front. Plant Sci. 2017, 8, 505. [Google Scholar] [CrossRef]
- Vannozzi, A.; Wong, D.C.J.; Höll, J.; Hmmam, I.; Matus, J.T.; Bogs, J.; Ziegler, T.; Dry, I.; Barcaccia, G.; Lucchin, M. Combinatorial Regulation of Stilbene Synthase Genes by WRKY and MYB Transcription Factors in Grapevine (Vitis vinifera L.). Plant Cell Physiol. 2018, 59, 1043–1059. [Google Scholar] [CrossRef] [PubMed]
- Pawlus, A.D.; Waffo-Téguo, P.; Shaver, J.; Mérillon, J.-M. Stilbenoid Chemistry from Wine and the Genus Vitis, a Review. OENO One 2012, 46, 57. [Google Scholar] [CrossRef]
- Chong, J.; Poutaraud, A.; Hugueney, P. Metabolism and Roles of Stilbenes in Plants. Plant Sci. 2009, 177, 143–155. [Google Scholar] [CrossRef]
- Duan, D.; Halter, D.; Baltenweck, R.; Tisch, C.; Tröster, V.; Kortekamp, A.; Hugueney, P.; Nick, P. Genetic Diversity of Stilbene Metabolism in Vitis Sylvestris. J. Exp. Bot. 2015, 66, 3243–3257. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-C.; Pyun, J.W.; Jeong, Y.J.; Park, S.H.; Kim, S.; Kim, Y.-H.; Lee, J.R.; Kim, C.Y.; Jeong, J.C. Overexpression of VlPRX21 and VlPRX35 Genes in Arabidopsis Plants Leads to Bioconversion of Trans-Resveratrol to δ-Viniferin. Plant Physiol. Biochem. 2021, 162, 556–563. [Google Scholar] [CrossRef]
- Corso, M.; Vannozzi, A.; Maza, E.; Vitulo, N.; Meggio, F.; Pitacco, A.; Telatin, A.; D’Angelo, M.; Feltrin, E.; Negri, A.S.; et al. Comprehensive Transcript Profiling of Two Grapevine Rootstock Genotypes Contrasting in Drought Susceptibility Links the Phenylpropanoid Pathway to Enhanced Tolerance. J. Exp. Bot. 2015, 66, 5739–5752. [Google Scholar] [CrossRef]
- Xu, W.; Ma, F.; Li, R.; Zhou, Q.; Yao, W.; Jiao, Y.; Zhang, C.; Zhang, J.; Wang, X.; Xu, Y.; et al. VpSTS29/STS2 Enhances Fungal Tolerance in Grapevine through a Positive Feedback Loop. Plant Cell Environ. 2019, 42, 2979–2998. [Google Scholar] [CrossRef]
- Mania, E.; Petrella, F.; Giovannozzi, M.; Piazzi, M.; Wilson, A.; Guidoni, S. Managing Vineyard Topography and Seasonal Variability to Improve Grape Quality and Vineyard Sustainability. Agronomy 2021, 11, 1142. [Google Scholar] [CrossRef]
- Cortell, J.M.; Halbleib, M.; Gallagher, A.V.; Righetti, T.L.; Kennedy, J.A. Influence of Vine Vigor on Grape (Vitis vinifera L. Cv. Pinot Noir) and Wine Proanthocyanidins. J. Agric. Food Chem. 2007, 53, 5798–5808. [Google Scholar] [CrossRef]
- Castellarin, S.D.; Matthews, M.A.; Di Gaspero, G.; Gambetta, G.A. Water Deficits Accelerate Ripening and Induce Changes in Gene Expression Regulating Flavonoid Biosynthesis in Grape Berries. Planta 2007, 227, 101–112. [Google Scholar] [CrossRef]
- Acevedo-Opazo, C.; Ortega-Farias, S.; Fuentes, S. Effects of Grapevine (Vitis vinifera L.) Water Status on Water Consumption, Vegetative Growth and Grape Quality: An Irrigation Scheduling Application to Achieve Regulated Deficit Irrigation. Agric. Water Manag. 2010, 97, 956–964. [Google Scholar] [CrossRef]
- Brillante, L.; Martínez-Luscher, J.; Yu, R.; Plank, C.M.; Sanchez, L.; Bates, T.L.; Brenneman, C.; Oberholster, A.; Kurtural, S.K. Assessing Spatial Variability of Grape Skin Flavonoids at the Vineyard Scale Based on Plant Water Status Mapping. J. Agric. Food Chem. 2017, 65, 5255–5265. [Google Scholar] [CrossRef] [PubMed]
- Echeverría, G.; Ferrer, M.; Mirás-Avalos, J.M. Effects of Soil Type on Vineyard Performance and Berry Composition in the Río de La Plata Coast. OENO One 2017, 51, 5–17. [Google Scholar] [CrossRef]
- Mazzei, P.; Celano, G.; Palese, A.M.; Lardo, E.; Drosos, M.; Piccolo, A. HRMAS-NMR Metabolomics of Aglianicone Grapes Pulp to Evaluate Terroir and Vintage Effects, and, as Assessed by the Electromagnetic Induction (EMI) Technique, Spatial Variability of Vineyard Soils. Food Chem. 2019, 283, 215–223. [Google Scholar] [CrossRef]
- Kerkech, M.; Hafiane, A.; Canals, R. Vine Disease Detection in UAV Multispectral Images Using Optimized Image Registration and Deep Learning Segmentation Approach. Comput. Electron. Agric. 2020, 174, 105446. [Google Scholar] [CrossRef]
- Chawade, A.; van Ham, J.; Blomquist, H.; Bagge, O.; Alexandersson, E.; Ortiz, R. High-Throughput Field-Phenotyping Tools for Plant Breeding and Precision Agriculture. Agronomy 2019, 9, 258. [Google Scholar] [CrossRef]
- Walvoort, D.J.J.; Brus, D.J.; de Gruijter, J.J. An R Package for Spatial Coverage Sampling and Random Sampling from Compact Geographical Strata by K-Means. Comput. Geosci. 2010, 36, 1261–1267. [Google Scholar] [CrossRef]
- NF X31-107; Qualité du Sol–Détermination de la Distribution Granulométrique des Particules du Sol–Méthode à La Pipette. Association Française de Normalisation: Paris, France, 2003.
- Houillé, B.; Besseau, S.; Courdavault, V.; Oudin, A.; Glévarec, G.; Delanoue, G.; Guérin, L.; Simkin, A.J.; Papon, N.; Clastre, M.; et al. Biosynthetic Origin of E-Resveratrol Accumulation in Grape Canes during Postharvest Storage. J. Agric. Food Chem. 2015, 63, 1631–1638. [Google Scholar] [CrossRef]
- Fiehn, O.; Wohlgemuth, G.; Scholz, M.; Kind, T.; Lee, D.Y.; Lu, Y.; Moon, S.; Nikolau, B. Quality Control for Plant Metabolomics: Reporting MSI-Compliant Studies. Plant J. 2008, 53, 691–704. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Billet, K.; Salvador-Blanes, S.; Dugé De Bernonville, T.; Delanoue, G.; Hinschberger, F.; Oudin, A.; Courdavault, V.; Pichon, O.; Besseau, S.; Leturcq, S.; et al. Terroir Influence on Polyphenol Metabolism from Grape Canes: A Spatial Metabolomic Study at Parcel Scale. Molecules 2023, 28, 4555. https://doi.org/10.3390/molecules28114555
Billet K, Salvador-Blanes S, Dugé De Bernonville T, Delanoue G, Hinschberger F, Oudin A, Courdavault V, Pichon O, Besseau S, Leturcq S, et al. Terroir Influence on Polyphenol Metabolism from Grape Canes: A Spatial Metabolomic Study at Parcel Scale. Molecules. 2023; 28(11):4555. https://doi.org/10.3390/molecules28114555
Chicago/Turabian StyleBillet, Kévin, Sébastien Salvador-Blanes, Thomas Dugé De Bernonville, Guillaume Delanoue, Florent Hinschberger, Audrey Oudin, Vincent Courdavault, Olivier Pichon, Sébastien Besseau, Samuel Leturcq, and et al. 2023. "Terroir Influence on Polyphenol Metabolism from Grape Canes: A Spatial Metabolomic Study at Parcel Scale" Molecules 28, no. 11: 4555. https://doi.org/10.3390/molecules28114555
APA StyleBillet, K., Salvador-Blanes, S., Dugé De Bernonville, T., Delanoue, G., Hinschberger, F., Oudin, A., Courdavault, V., Pichon, O., Besseau, S., Leturcq, S., Giglioli-Guivarc’h, N., & Lanoue, A. (2023). Terroir Influence on Polyphenol Metabolism from Grape Canes: A Spatial Metabolomic Study at Parcel Scale. Molecules, 28(11), 4555. https://doi.org/10.3390/molecules28114555







