Columnar Liquid Crystals of Copper(I) Complexes with Ionic Conductivity and Solid State Emission
Abstract
1. Introduction
2. Results and Discussions
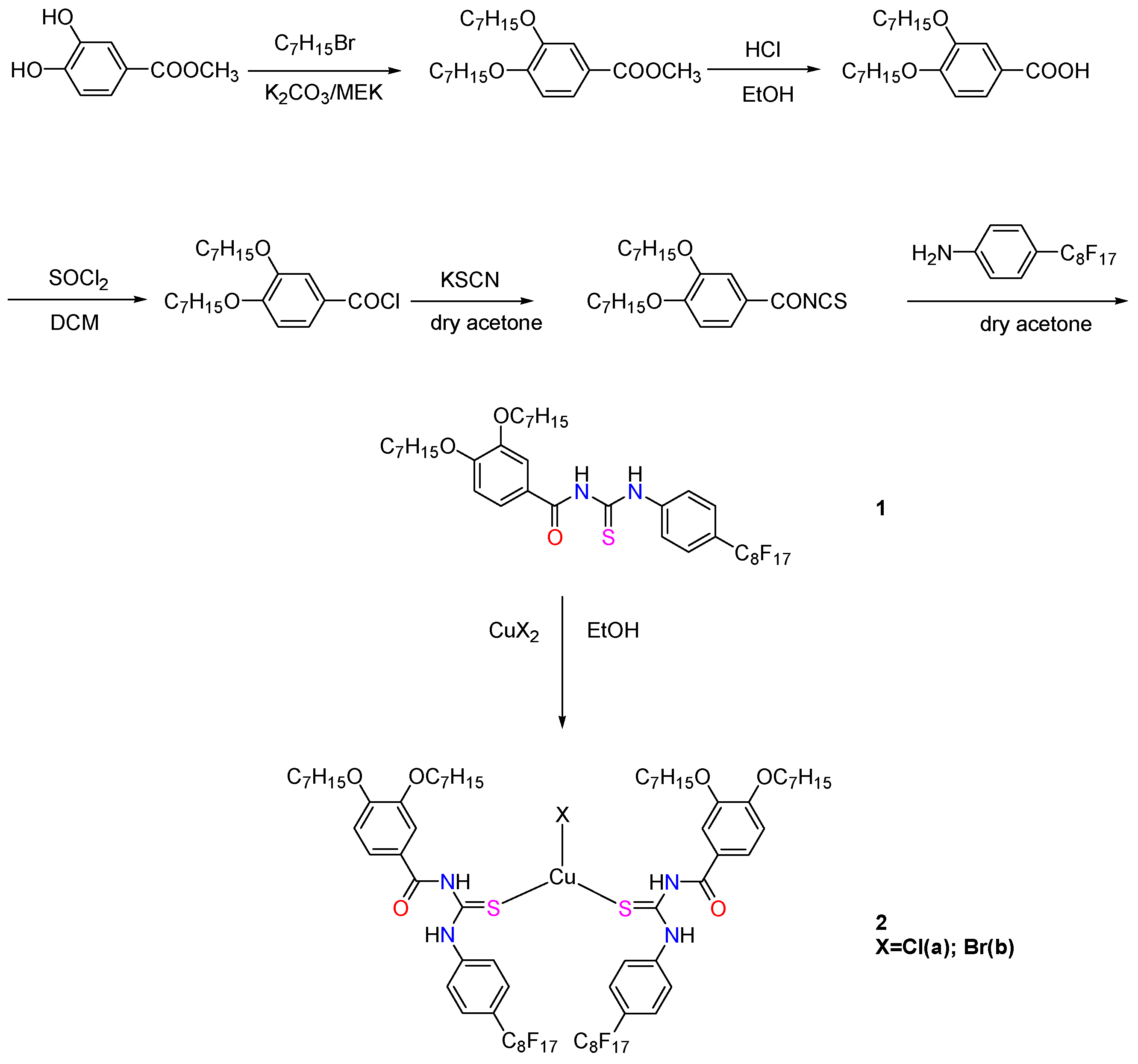
2.1. Synthesis and Structural Characterization
2.2. Characterization of Liquid Crystalline Properties
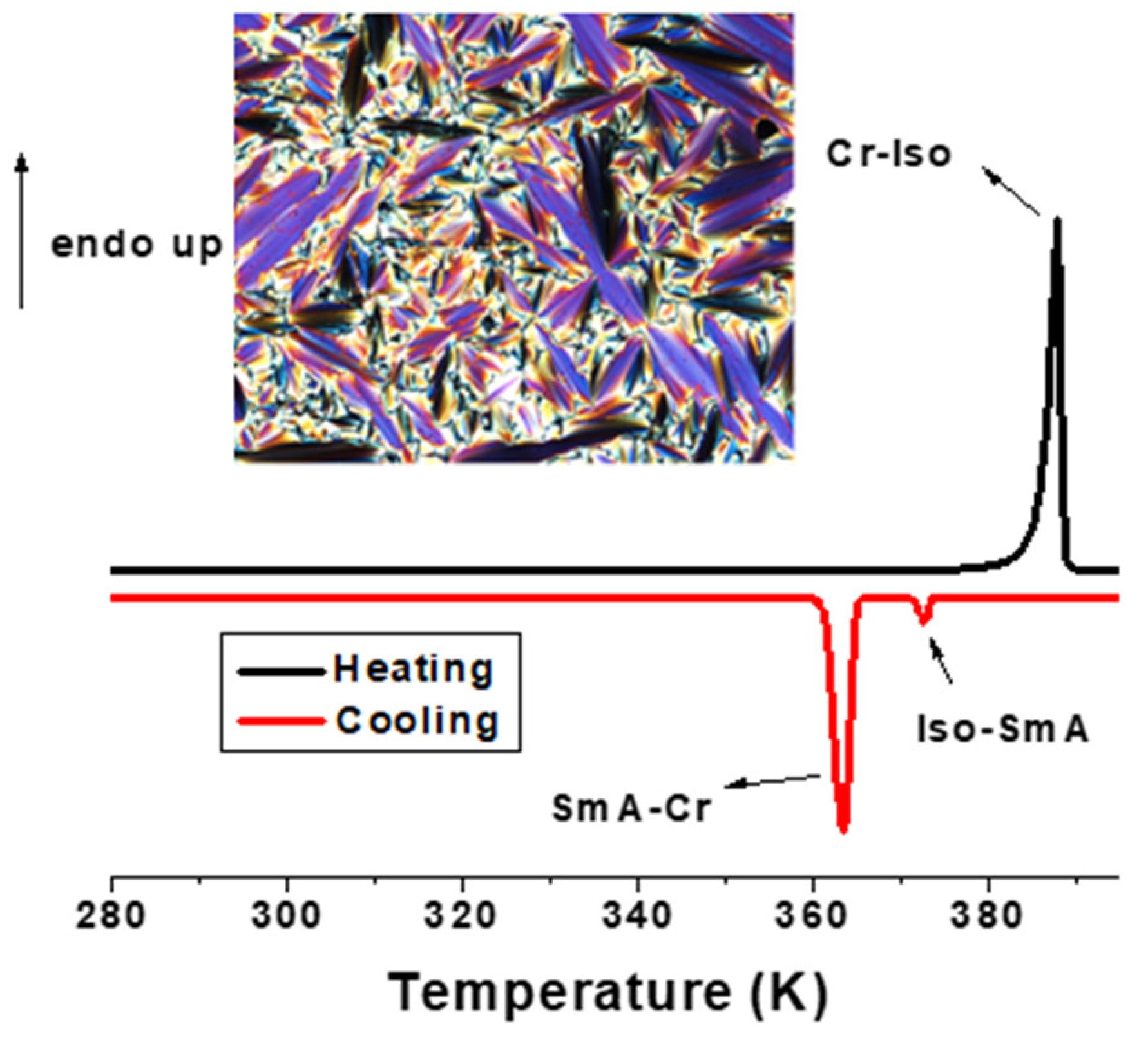
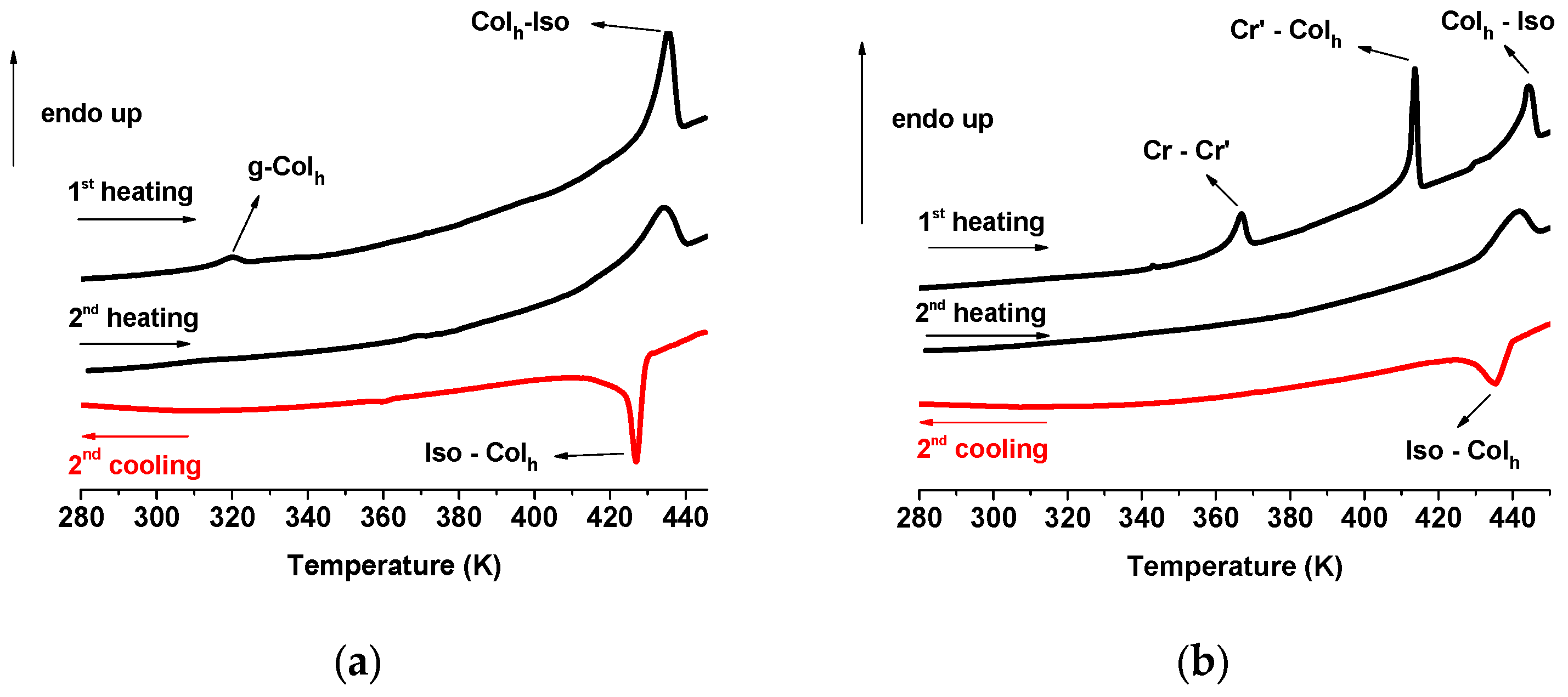
2.2.1. DSC and POM Studies
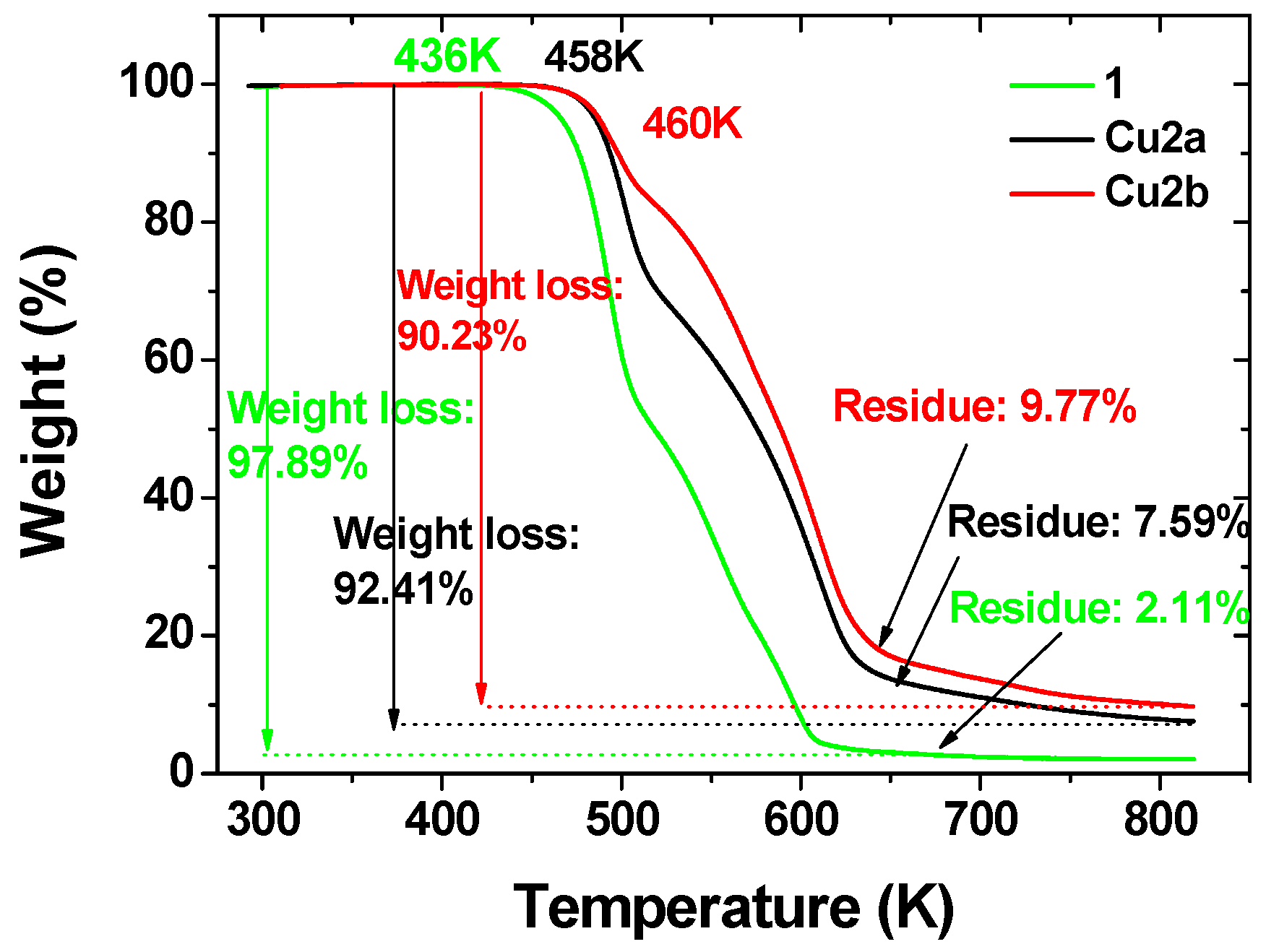
2.2.2. Thermal Decomposition
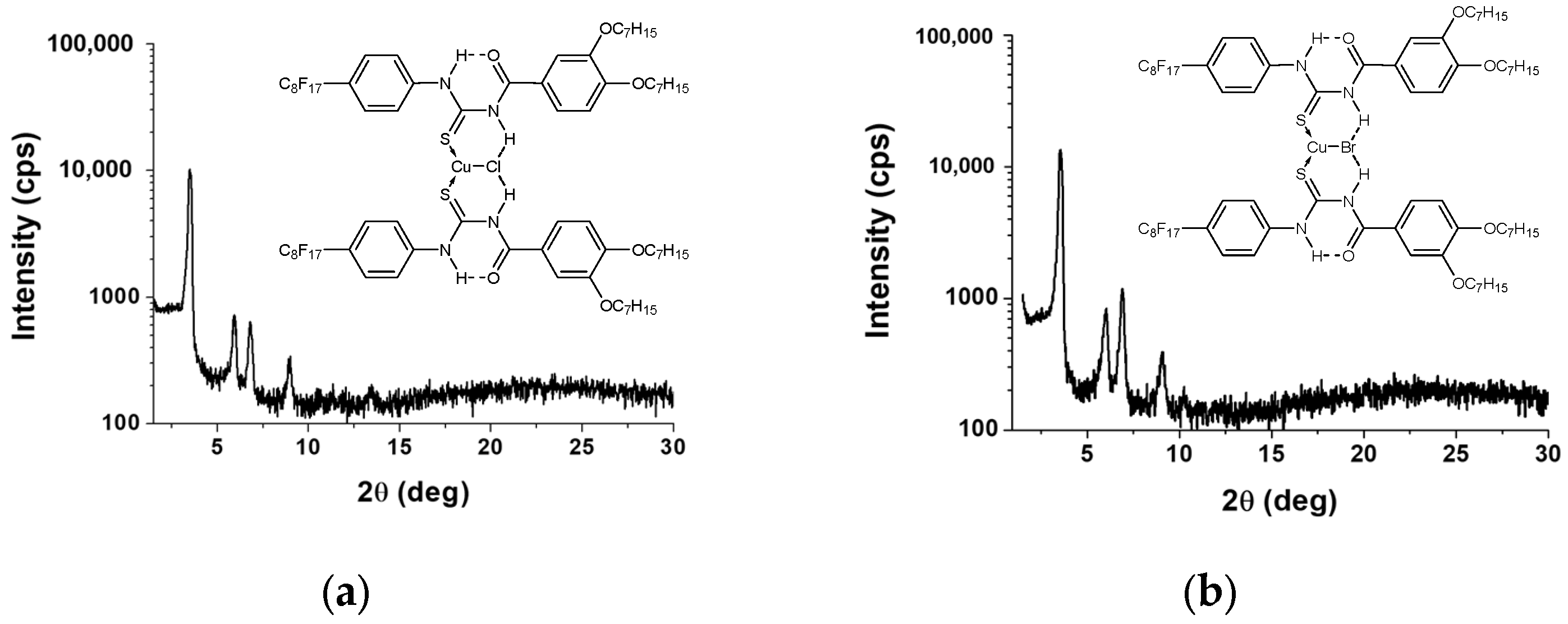
2.2.3. X-ray Diffraction Investigation
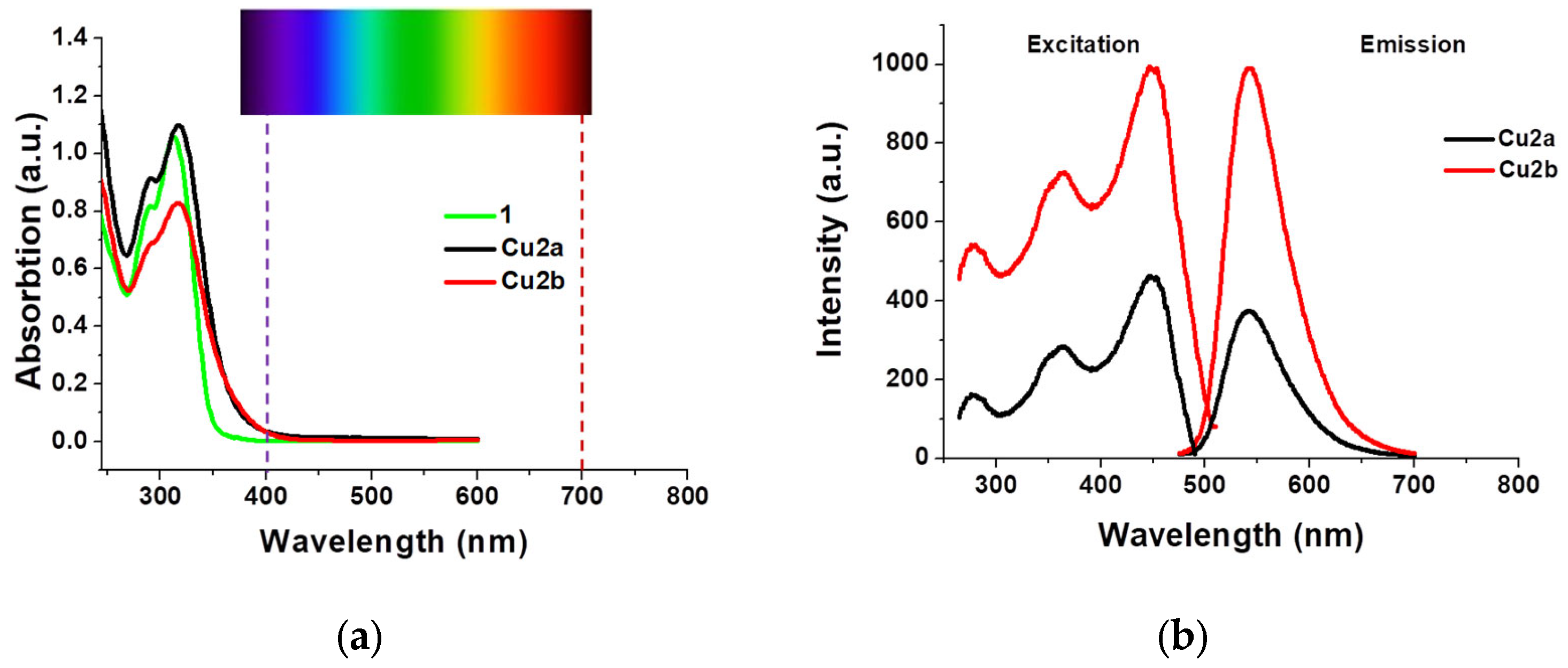
2.3. UV–VIS and Emission Properties
2.4. Dielectric Spectroscopy
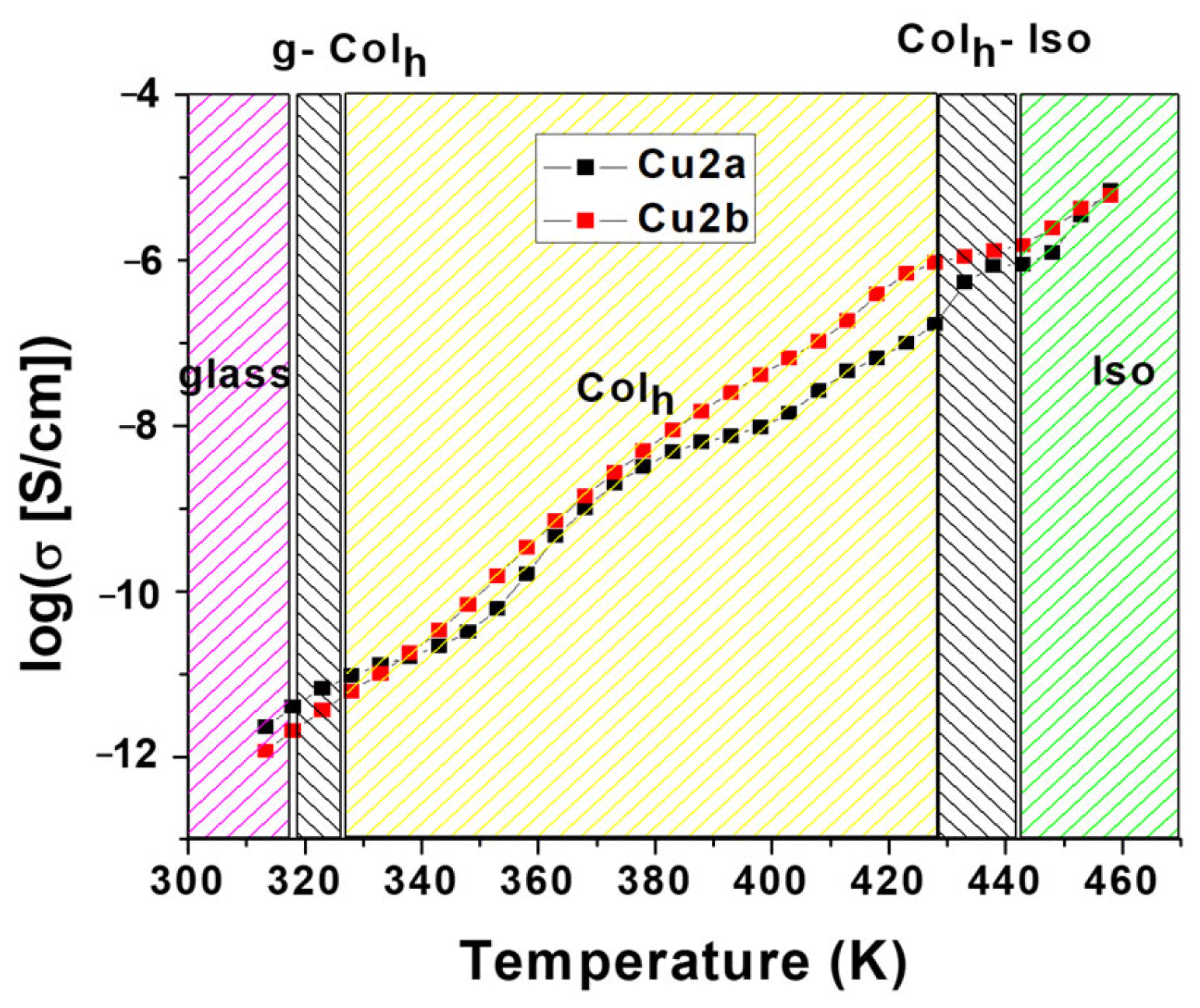
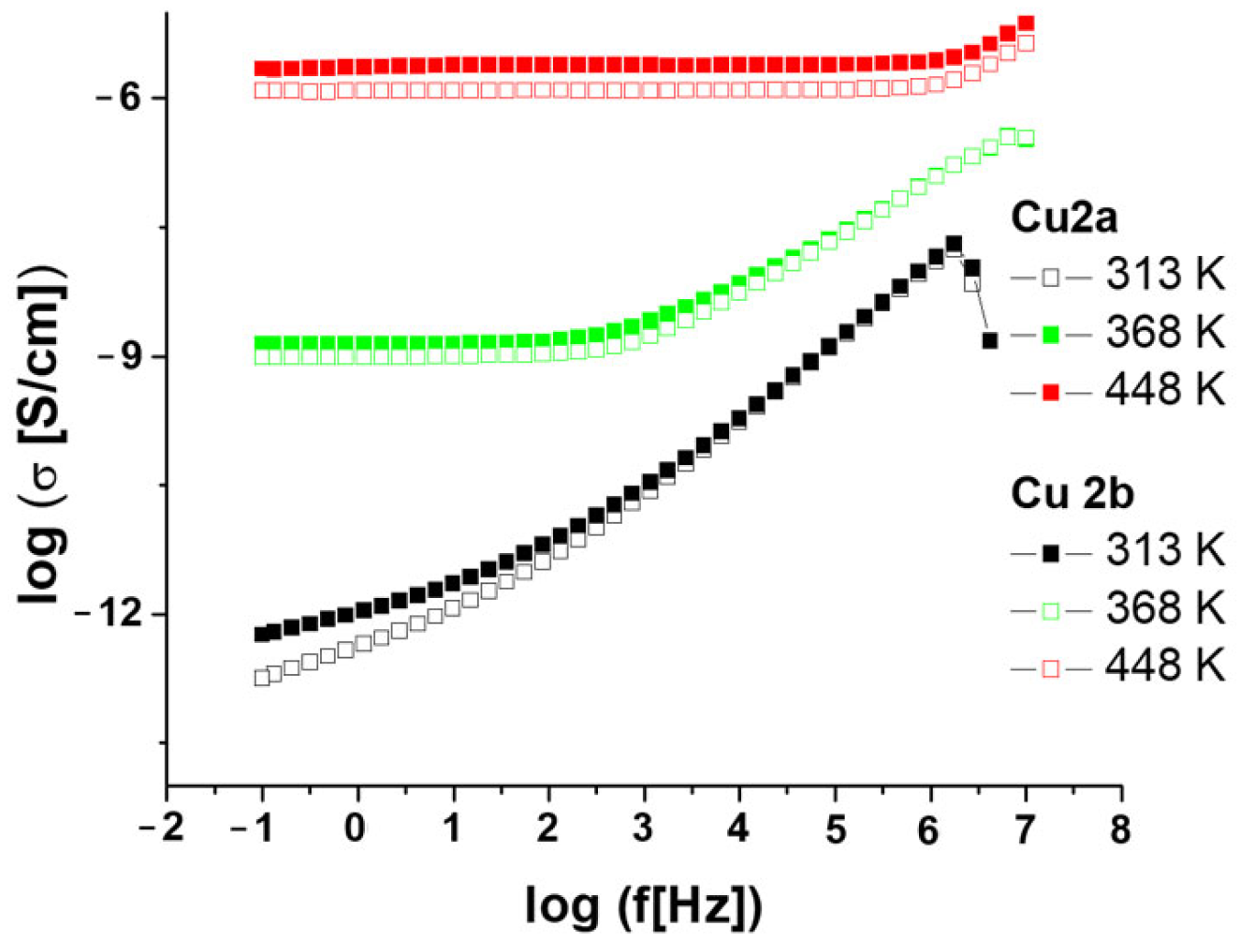
2.4.1. Electrical Conductivity
2.4.2. Dielectric Properties
3. Conclusions
4. Experimental Section
4.1. Characterization Methods
4.2. Preparation of N-(4-Perfluorooctylphenylcarbamothioyl)-3,4-Diheptyloxybenzamide (1)
4.3. Preparation of Copper(I) Complexes (Cu2a and Cu2b)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Ravaro, L.P.; Zanoni, K.P.; de Camargo, A.S. Luminescent Copper(I) complexes as promising materials for the next generation of energy-saving OLED devices. Energy Rep. 2020, 6, 37–45. [Google Scholar] [CrossRef]
- Bergmann, L.; Hedley, G.J.; Baumann, T.; Bräse, S.; Samuel, I.D.W. Direct observation of intersystem crossing in a thermally activated delayed fluorescence copper complex in the solid state. Sci. Adv. 2016, 2, e1500889. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, L. New Emitters for OLEDs: The Coordination- and Photo-Chemistry of Mononuclear Neutral Copper(I) Complexes; Logos Verlag Berlin GmbH: Berlin, Germany, 2016; ISBN 978-3-8325-4183-5. [Google Scholar]
- Housecroft, C.E.; Constable, E.C. TADF: Enabling luminescent copper(i) coordination compounds for light-emitting electrochemical cells. J. Mater. Chem. C 2022, 10, 4456–4482. [Google Scholar] [CrossRef] [PubMed]
- Au, V.K.-M. Organic Light-Emitting Diodes Based on Luminescent Self-Assembled Materials of Copper(I). Energy Fuels 2021, 35, 18982–18999. [Google Scholar] [CrossRef]
- Chen, X.-W.; He, L.-H.; Ju, P.; Chen, J.-L.; Liu, S.-J.; Wen, H.-R. Mechanochromic luminescent materials of bimetallic Cu(i) complexes showing thermally activated delayed fluorescence. J. Mater. Chem. C 2020, 8, 16160–16167. [Google Scholar] [CrossRef]
- Czerwieniec, R.; Leitl, M.J.; Homeier, H.H.; Yersin, H. Cu(I) complexes—Thermally activated delayed fluorescence. Photophysical approach and material design. Coord. Chem. Rev. 2016, 325, 2–28. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, J.; Liu, C.; Yin, G.; Wu, M.; Du, C.; Zhang, B. Three-coordinated mononuclear Cu(I) complexes with crystallization-enhanced thermally activated delayed fluorescence characteristics. Polyhedron 2022, 218, 115761. [Google Scholar] [CrossRef]
- Mara, M.W.; Fransted, K.A.; Chen, L.X. Interplays of excited state structures and dynamics in copper(I) diimine complexes: Implications and perspectives. Coord. Chem. Rev. 2015, 282–283, 2–18. [Google Scholar] [CrossRef]
- Zhang, Y.; Schulz, M.; Wächtler, M.; Karnahl, M.; Dietzek, B. Heteroleptic diimine–diphosphine Cu(I) complexes as an alternative towards noble-metal based photosensitizers: Design strategies, photophysical properties and perspective applications. Coord. Chem. Rev. 2018, 356, 127–146. [Google Scholar] [CrossRef]
- Langer, R.; Yadav, M.; Weinert, B.; Fenske, D.; Fuhr, O. Luminescence in Functionalized Copper Thiolate Clusters—Synthesis and Structural Effects. Eur. J. Inorg. Chem. 2013, 21, 3623–3631. [Google Scholar] [CrossRef]
- Koutsari, A.; Karasmani, F.; Kapetanaki, E.; Zainuddin, D.I.; Hatzidimitriou, A.G.; Angaridis, P.; Aslanidis, P. Luminescent thione/phosphane mixed-ligand copper(I) complexes: The effect of thione on structural properties. Inorg. Chim. Acta 2017, 458, 138–145. [Google Scholar] [CrossRef]
- Favarin, L.R.; Rosa, P.P.; Pizzuti, L.; Machulek, A.; Caires, A.R.; Bezerra, L.; Pinto, L.; Maia, G.; Gatto, C.C.; Back, D.F.; et al. Synthesis and structural characterization of new heteroleptic copper(I) complexes based on mixed phosphine/thiocarbamoyl-pyrazoline ligands. Polyhedron 2017, 121, 185–190. [Google Scholar] [CrossRef]
- Borges, A.P.; Carneiro, Z.A.; Prado, F.S.; Souza, J.R.; Silva, L.H.F.E.; Oliveira, C.G.; Deflon, V.M.; de Albuquerque, S.; Leite, N.B.; Machado, A.E.; et al. Cu(I) complexes with thiosemicarbazides derived from p-toluenesulfohydrazide: Structural, luminescence and biological studies. Polyhedron 2018, 155, 170–179. [Google Scholar] [CrossRef]
- Aslanidis, P.; Cox, P.; Tsaliki, P. Copper(I) halide complexes with 2,2′-bis(diphenylphosphano)-1,1′-binaphthyl (rac-binap) and heterocyclic thiones. Racemic compounds in chiral and achiral crystal space groups. Polyhedron 2008, 27, 3029–3035. [Google Scholar] [CrossRef]
- Tudor, C.A.; Iliş, M.; Secu, M.; Ferbinteanu, M.; Cîrcu, V. Luminescent heteroleptic copper(I) complexes with phosphine and N-benzoyl thiourea ligands: Synthesis, structure and emission properties. Polyhedron 2022, 211, 115542. [Google Scholar] [CrossRef]
- Troyano, J.; Perles, J.; Amo-Ochoa, P.; Martínez, J.I.; Gimeno, M.C.; Fernández-Moreira, V.; Zamora, F.; Delgado, S. Luminescent Thermochromism of 2D Coordination Polymers Based on Copper(I) Halides with 4-Hydroxythiophenol. Chem. A Eur. J. 2016, 22, 18027–18035. [Google Scholar] [CrossRef]
- Troyano, J.; Castillo, Ó.; Amo-Ochoa, P.; Martínez, J.I.; Zamora, F.; Delgado, S. Reversible transformation between Cu(I)-thiophenolate coordination polymers displaying luminescence and electrical properties. CrystEngComm 2019, 21, 3232–3239. [Google Scholar] [CrossRef]
- Sun, Y.; Lemaur, V.; Beltrán, J.I.; Cornil, J.; Huang, J.; Zhu, J.; Wang, Y.; Fröhlich, R.; Wang, H.; Jiang, L.; et al. Neutral Mononuclear Copper(I) Complexes: Synthesis, Crystal Structures, and Photophysical Properties. Inorg. Chem. 2016, 55, 5845–5852. [Google Scholar] [CrossRef]
- Bergmann, L.; Friedrichs, J.; Mydlak, M.; Baumann, T.; Nieger, M.; Bräse, S. Outstanding luminescence from neutral copper(i) complexes with pyridyl-tetrazolate and phosphine ligands. Chem. Commun. 2013, 49, 6501–6503. [Google Scholar] [CrossRef]
- Chan, K.; Cheng, S.; Lo, L.T.; Yiu, S.; Ko, C. Luminescent Charge-Neutral Copper(I) Phenanthroline Complexes with Isocyanoborate Ligand. Eur. J. Inorg. Chem. 2018, 2018, 897–903. [Google Scholar] [CrossRef]
- Espinet, P.; Lequerica, M.C.; Álvarez, J.M.M. Synthesis, Structural Characterization and Mesogenic Behavior of Copper(I)n-Alkylthiolates. Chem. A Eur. J. 1999, 5, 1982–1986. [Google Scholar] [CrossRef]
- Benouazzane, M.; Coco, S.; Espinet, P.; Barberá, J. Supramolecular Organization in Copper(I) Isocyanide Complexes: Copper(I) Liquid Crystals from a Simple Molecular Structure. J. Mater. Chem. 2001, 11, 1740–1744. [Google Scholar] [CrossRef]
- Benouazzane, M.; Coco, S.; Espinet, P.; Barberá, J. Binuclear Mesogenic Copper(I) Isocyanide Complexes with an Unusual Inorganic Core Formed by Two Tetrahedra Sharing an Edge. Inorg. Chem. 2002, 41, 5754–5759. [Google Scholar] [CrossRef] [PubMed]
- Coco, S.; Cordovilla, C.; Donnio, B.; Espinet, P.; García-Casas, M.J.; Guillon, D. Self-Organization of Dendritic Supermolecules, Based on Isocyanide–Gold(I), –Copper(I), –Palladium(II), and –Platinum(II) Complexes, into Micellar Cubic Mesophases. Chem. A Eur. J. 2008, 14, 3544–3552. [Google Scholar] [CrossRef] [PubMed]
- Dembinski, R.; Espinet, P.; Lentijo, S.; Markowicz, M.W.; Martín-Alvarez, J.M.; Rheingold, A.L.; Schmidt, D.J.; Sniady, A. Fluorophobic Effect in Metallomesogens—The Synthesis and Mesomorphism of Ag, Au, Cu, Fe, Pd, and Pt Fluorous Isocyanide Complexes. Eur. J. Inorg. Chem. 2008, 2008, 1565–1572. [Google Scholar] [CrossRef]
- Chico, R.; de Domingo, E.; Domínguez, C.; Donnio, B.; Heinrich, B.; Termine, R.; Golemme, A.; Coco, S.; Espinet, P. High One-Dimensional Charge Mobility in Semiconducting Columnar Mesophases of Isocyano-Triphenylene Metal Complexes. Chem. Mater. 2017, 29, 7587–7595. [Google Scholar] [CrossRef]
- Cretu, C.; Andelescu, A.A.; Candreva, A.; Crispini, A.; Szerb, E.I.; La Deda, M. Bisubstituted-biquinoline Cu(I) complexes: Synthesis, mesomorphism and photophysical studies in solution and condensed states. J. Mater. Chem. C 2018, 6, 10073–10082. [Google Scholar] [CrossRef]
- Douce, L.; El-Ghayoury, A.; Ziessel, R.; Skoulios, A. Columnar mesophases from tetrahedral copper(I) cores and Schiff-base derived polycatenar ligands. Chem. Commun. 1999, 20, 2033–2034. [Google Scholar] [CrossRef]
- El-Ghayoury, A.; Douce, L.; Skoulios, A.; Ziessel, R. Cation-Induced Macroscopic Ordering of Non-Mesomorphic Modules—A New Application for Metallohelicates. Angew. Chem. Int. Ed. 1998, 37, 2205–2208. [Google Scholar] [CrossRef]
- Alkali, M.; Cîrcu, V. Perspectives on the Structural Design and Luminescent Behavior of Liquid Crystalline Materials Based on Copper(I) Complexes. Chemistry 2023, 5, 646–661. [Google Scholar] [CrossRef]
- Giménez, R.; Crespo, O.; Diosdado, B.; Elduque, A. Liquid crystalline copper(i) complexes with bright room temperature phosphorescence. J. Mater. Chem. C 2020, 8, 6552–6557. [Google Scholar] [CrossRef]
- Kishimura, A.; Yamashita, T.; Yamaguchi, K.; Aida, T. Rewritable phosphorescent paper by the control of competing kinetic and thermodynamic self-assembling events. Nat. Mater. 2005, 4, 546–549. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-D.; Lai, C.K. Ionic columnar metallomesogens formed by three-coordinated copper(I) complexes. J. Chem. Soc. Dalton Trans. 2001, 16, 2383–2387. [Google Scholar] [CrossRef]
- Sergeyev, S.; Pisula, W.; Geerts, Y.H. Discotic liquid crystals: A new generation of organic semiconductors. Chem. Soc. Rev. 2007, 36, 1902–1929. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Wei, B.; Liang, T.; Yang, X.; Wu, Y. Anhydrous proton conduction in liquid crystals containing benzimidazole moieties. RSC Adv. 2016, 6, 34038–34042. [Google Scholar] [CrossRef]
- Wei, B.; Tan, S.; Liang, T.; Cao, S.; Wu, Y. Synthesis, structural and electrochemical characterization of benzimidazole compounds exhibiting a smectic C liquid crystal phase. J. Mol. Struct. 2017, 1133, 392–397. [Google Scholar] [CrossRef]
- Basak, D.; Christensen, S.; Surampudi, S.K.; Versek, C.; Toscano, D.T.; Tuominen, M.T.; Hayward, R.C.; Venkataraman, D. Proton conduction in discotic mesogens. Chem. Commun. 2011, 47, 5566–5568. [Google Scholar] [CrossRef]
- Cuerva, C.; Campo, J.A.; Cano, M.; Schmidt, R. Nanostructured discotic Pd(ii) metallomesogens as one-dimensional proton conductors. Dalton Trans. 2017, 46, 96–105. [Google Scholar] [CrossRef]
- Peighambardoust, S.; Rowshanzamir, S.; Amjadi, M. Review of the proton exchange membranes for fuel cell applications. Int. J. Hydrog. Energy 2010, 35, 9349–9384. [Google Scholar] [CrossRef]
- Kato, T.; Yoshio, M.; Ichikawa, T.; Soberats, B.; Ohno, H.; Funahashi, M. Transport of ions and electrons in nanostructured liquid crystals. Nat. Rev. Mater. 2017, 2, 17001. [Google Scholar] [CrossRef]
- Gu, J.; Luo, J.; Yang, J.; Tan, S.; Wang, C.; Wu, Y. Anhydrous proton conduction in protic ionic liquid crystals formed by 1-alkyl-3H-imidazolium hydrogen sulfates. Ionics 2022, 28, 2293–2300. [Google Scholar] [CrossRef]
- Chai, S.; Xu, F.; Zhang, R.; Wang, X.; Zhai, L.; Li, X.; Qian, H.-J.; Wu, L.; Li, H. Hybrid Liquid-Crystalline Electrolytes with High-Temperature-Stable Channels for Anhydrous Proton Conduction. J. Am. Chem. Soc. 2021, 143, 21433–21442. [Google Scholar] [CrossRef] [PubMed]
- Iliş, M.; Bucos, M.; Dumitraşcu, F.; Cîrcu, V. Mesomorphic behaviour of N-benzoyl-N′-aryl thioureas liquid crystalline compounds. J. Mol. Struct. 2011, 987, 1–6. [Google Scholar] [CrossRef]
- Iliş, M.; Micutz, M.; Pasuk, I.; Staicu, T.; Cîrcu, V. Synthesis and liquid crystalline properties of novel fluorinated N-benzoyl thiourea compounds. Effect of perfluoroalkyl chains on the thermal behavior and smectic phases stability. J. Fluor. Chem. 2017, 204, 84–89. [Google Scholar] [CrossRef]
- Staicu, T.; Iliş, M.; Cîrcu, V.; Micutz, M. Influence of hydrocarbon moieties of partially fluorinated N -benzoyl thiourea compounds on their gelation properties. A detailed rheological study of complex viscoelastic behavior of decanol/N -benzoyl thiourea mixtures. J. Mol. Liq. 2018, 255, 297–312. [Google Scholar] [CrossRef]
- Duan, X.-H.; Su, B.-Q.; Xian, L.; Sheng, L.; Tan, R. Synthesis, Crystal Structure and Fluorescent Property of Cis-bis[N,N-diethyl-N′-(p-nitrobenzoyl)thiourea-kS,O] Platinum(II) Complex. J. Chem. Crystallogr. 2022, 1–13. [Google Scholar] [CrossRef]
- Cîrcu, V.; Mocanu, A.S.; Roşu, C.; Manaila-Maximean, D.; Dumitraşcu, F. Thermal behaviour and electro-optical properties of a series of liquid crystals based on palladium complexes with mixed ligands Schiff bases and N-benzoyl thioureas. J. Therm. Anal. Calorim. 2012, 107, 877–886. [Google Scholar] [CrossRef]
- Mühl, P.; Gloe, K.; Dietze, F.; Hoyer, E.; Beyer, L. N-Acyl-thioharnstoffe—Effektive Extraktionsmittel für die Flüssig-Flüssig-Extraktion von Metallionen. Z. Chem. 1986, 26, 81–94. [Google Scholar] [CrossRef]
- Iliş, M.; Micutz, M.; Dumitraşcu, F.; Pasuk, I.; Molard, Y.; Roisnel, T.; Cîrcu, V. Enhancement of smectic C mesophase stability by using branched alkyl chains in the auxiliary ligands of luminescent Pt(II) and Pd(II) complexes. Polyhedron 2014, 69, 31–39. [Google Scholar] [CrossRef]
- Molter, A.; Kathrein, S.; Kircher, B.; Mohr, F. Anti-tumour active gold(I), palladium(II) and ruthenium(II) complexes with thio- and selenoureato ligands: A comparative study. Dalton Trans. 2018, 47, 5055–5064. [Google Scholar] [CrossRef]
- Yeşilkaynak, T. 2-Chloro-N-((5-chloropyridine-2-yl)carbamothioyl)benzamide and its Co(II), Ni(II) and Cu(II) metal complexes. J. Therm. Anal. Calorim. 2016, 124, 1029–1037. [Google Scholar] [CrossRef]
- Keskin, E.; Solmaz, U.; Gumus, I.; Arslan, H. Di- and tetra-nuclear oxorhenium(V) complexes of benzoylthiourea derivative ligands: Synthesis, structural characterization, and catalytic applications. Polyhedron 2022, 219, 115786. [Google Scholar] [CrossRef]
- Sternberg, M.; Rust, J.; Lehmann, C.W.; Mohr, F. Synthesis and Metal Complexes of Thiourea Ligands Containing Carbohydrate-Derived Substituents. Helv. Chim. Acta 2013, 96, 280–288. [Google Scholar] [CrossRef]
- Nkabyo, H.A.; Luckay, R.C.; Koch, K.R. Photo-isomerization of palladium(II) N,N-di-substituted acylthioureas: The role of free ligands and formation of mixed-ligand complexes. Inorg. Chem. Commun. 2020, 119, 108035. [Google Scholar] [CrossRef]
- Kuchar, J.; Rust, J.; Lehmann, C.W.; Mohr, F. Copper(I) Complexes with Anionic Acylthio- or Acylselenourea Ligands and N-Heterocyclic Carbenes or Phosphanes. Eur. J. Inorg. Chem. 2018, 2018, 5215–5222. [Google Scholar] [CrossRef]
- Roşu, C.; Manaila-Maximean, D.; Circu, V.; Molard, Y.; Roisnel, T. Differential negative resistance in the current–voltage characteristics of a new palladium(II) metallomesogen. Liq. Cryst. 2011, 38, 757–765. [Google Scholar] [CrossRef]
- Ilis, M.; Batalu, D.; Pasuk, I.; Cîrcu, V. Cyclometalated palladium(II) metallomesogens with Schiff bases and N -benzoyl thiourea derivatives as co-ligands. J. Mol. Liq. 2017, 233, 45–51. [Google Scholar] [CrossRef]
- Nkabyo, H.A.; Barnard, I.; Koch, K.R.; Luckay, R.C. Recent advances in the coordination and supramolecular chemistry of monopodal and bipodal acylthiourea-based ligands. Coord. Chem. Rev. 2021, 427, 213588. [Google Scholar] [CrossRef]
- Zhao, X.-Y.; Zhu, C.-B.; Li, H.-P.; Yang, Y.; Roesky, H.W. Synthesis and Characterization of Copper(I) Halide Complexes withN-(2, 6-Diisopropylphenyl)-N′-benzoylthiourea: Monomeric, Dimeric, and Cage Structures. Z. Anorg. Allg. Chem. 2014, 640, 1614–1621. [Google Scholar] [CrossRef]
- Wang, D.; Wu, S.; Li, H.; Yang, Y.; Roesky, H.W. Synthesis and Characterization of Copper Complexes with the N -(2,6-Diisopropylphenyl)- N′-acylthiourea Ligands. Eur. J. Inorg. Chem. 2017, 2017, 1406–1413. [Google Scholar] [CrossRef]
- Li, G.; Che, D.-J.; Li, Z.-F.; Zhu, Y.; Zou, D.-P. Versatile coordination patterns in the reaction system of N-benzoyl-N′-(2-pyridyl)thiourea with CuCl2. Their reaction conditions, systematic isolation and crystal structures. New J. Chem. 2002, 26, 1629–1633. [Google Scholar] [CrossRef]
- Singh, D.P.; Pratap, S.; Pandey, S.K.; Butcher, R.J.; Marverti, G. N-(naphthyl)-N′-(methoxy carbonyl)thiocarbamide and its Cu(I) complex: Synthesis, spectroscopic, X-ray, DFT and in vitro cytotoxicity study. J. Coord. Chem. 2015, 68, 261–276. [Google Scholar] [CrossRef]
- Xian, L.; Wei, T.-B.; Zhang, Y.-M. Synthesis and crystal structure of bis(N-o-methylphenyl-N′-ethoxycarbonylthiourea) copper(I) chloride. J. Coord. Chem. 2004, 57, 453–457. [Google Scholar] [CrossRef]
- Peng, L.; Wang, D.; Zhang, D.; Zhang, Z.; Zhou, Q.; Li, H.; Yang, Y. Synthesis and Characterization of Copper(I) Halide Complexes Prepared with Bipodal Diacylthioureas. Eur. J. Inorg. Chem. 2020, 2020, 2521–2529. [Google Scholar] [CrossRef]
- Hou, X.; Wang, F.; Han, L.; Pan, X.; Li, H.; Yang, Y.; Roesky, H.W. Self-Assembly of Discrete Copper(I)-Halide Complexes with Diacylthioureas. Z. Anorg. Allg. Chem. 2018, 644, 142–148. [Google Scholar] [CrossRef]
- Pandey, S.K.; Pratap, S.; Pokharia, S.; Mishra, H.; Marverti, G.; Kaur, M.; Jasinski, J.P. Copper (I) complexes based on novel N, N′-disubstituted thiocarbamides: Synthesis, spectroscopic, in vitro cytotoxicity, DNA damage and G0/G1 cell cycle arrest studies. Inorg. Chim. Acta 2019, 491, 105–117. [Google Scholar] [CrossRef]
- Pandey, S.K.; Singh, D.P.; Pratap, S.; Marverti, G.; Butcher, R. Copper(I) complexes of N-(2/4 methoxy/2-chloro-4-nitro)phenyl-N′ (methoxycarbonyl)thiocarbamides as potential anticancer agents: Synthesis, crystal structure, in vitro cytotoxicity and DNA damage studies. Polyhedron 2019, 170, 431–439. [Google Scholar] [CrossRef]
- Pandey, S.K.; Singh, D.P.; Marverti, G.; Butcher, R.J.; Pratap, S. Monodentate Coordination of N, N′-Disubstituted Thiocarbamide Ligands: Syntheses, Structural Analyses, In Vitro Cytotoxicity and DNA Damage Studies of Cu(I) Complexes. ChemistrySelect 2018, 3, 3675–3679. [Google Scholar] [CrossRef]
- ElHusseiny, A.F.; Eldissouky, A.; Al-Hamza, A.M.; Hassan, H.H. Structure–property relationship studies of copper(I) complexes of nanosized hypodentate ligands and evaluation of their antitumor and antimicrobial activities. J. Coord. Chem. 2015, 68, 241–260. [Google Scholar] [CrossRef]
- Yuan, Y.F.; Wang, J.T.; Gimeno, M.; Laguna, A.; Jones, P.G. Synthesis and characterisation of copper complexes with N-ferrocenoyl-N′-aryl(alkyl)thioureas. Inorg. Chim. Acta 2001, 324, 309–317. [Google Scholar] [CrossRef]
- Krishna, P.M.; Reddy, K.H. Synthesis, single crystal structure and DNA cleavage studies on first 4N-ethyl substituted three coordinate copper(I) complex of thiosemicarbazone. Inorg. Chim. Acta 2009, 362, 4185–4190. [Google Scholar] [CrossRef]
- Beheshti, A.; Nozarian, K.; Mousavifard, E.S.; Abrahams, C.T.; Mayer, P.; Gajda, R.; Woźniak, K.; Motamedi, H. Design and construction of the imidazole-2-thione-based copper(I) complexes by varying the co-anion and synthesis conditions and verifying their antimicrobial activity. J. Solid State Chem. 2021, 294, 121874. [Google Scholar] [CrossRef]
- Iliş, M.; Cîrcu, V. Discotic Liquid Crystals Based on Cu(I) Complexes with Benzoylthiourea Derivatives Containing a Perfluoroalkyl Chain. J. Chem. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Weiqun, Z.; Baolong, L.; Liming, Z.; Jiangang, D.; Yong, Z.; Lude, L.; Xujie, Y. Structural and spectral studies on N-(4-chloro)benzoyl-N′-(4-tolyl)thiourea. J. Mol. Struct. 2004, 690, 145–150. [Google Scholar] [CrossRef]
- Hird, M. Fluorinated liquid crystals—Properties and applications. Chem. Soc. Rev. 2007, 36, 2070–2095. [Google Scholar] [CrossRef]
- Spengler, M.; Dong, R.Y.; Michal, C.A.; Pfletscher, M.; Giese, M. Fluorination of supramolecular liquid crystals—Tuning tool and analytical probe. J. Mater. Chem. C 2017, 5, 2235–2239. [Google Scholar] [CrossRef]
- Marzouk, S.; Khalfallah, A.; Heinrich, B.; Khiari, J.E.; Kriaa, A.; Méry, S. Synthesis and mesomorphic properties of liquid crystals containing a perfluorinated segment via different linkers. J. Fluor. Chem. 2017, 197, 15–23. [Google Scholar] [CrossRef]
- Jang, J.Y.; Park, Y.W. Smectic C Mesogens with Terminal Perfluoroalkyl Chains. Mol. Cryst. Liq. Cryst. 2015, 606, 90–100. [Google Scholar] [CrossRef]
- Jang, J.Y.; Park, Y.W. Synthesis and structural studies of smectic C mesogens with terminal perfluoroalkyl chains. Liq. Cryst. 2013, 40, 511–515. [Google Scholar] [CrossRef]
- Ciastek, S.; Szymańska, K.; Kaszyński, P.; Jasiński, M.; Pociecha, D. Smectic behaviour of methyl 4-alkoxybenzoates with a partially fluorinated alkyl chain. Liq. Cryst. 2018, 45, 11–21. [Google Scholar] [CrossRef]
- Bilgin-Eran, B.; Ocak, H.; Tschierske, C.; Baumeister, U. Synthesis and mesomorphism of fluoroalkylated platinum complexes. Liq. Cryst. 2012, 39, 467–476. [Google Scholar] [CrossRef]
- Piccionello, A.P.; Calabrese, A.; Pibiri, I.; Giacalone, V.; Pace, A.; Buscemi, S. Synthesis of Fluorinated Bent-Core Mesogens (BCMs) Containing the 1,2,4-Oxadiazole Ring. J. Heterocycl. Chem. 2016, 53, 1935–1940. [Google Scholar] [CrossRef]
- Doi, T.; Sakurai, Y.; Tamatani, A.; Takenaka, S.; Kusabayashi, S.; Nishihata, Y.; Terauchi, H. Thermal and X-ray diffraction studies of liquid crystals incorporating a perfluoroalkyl group. J. Mater. Chem. 1991, 1, 169–173. [Google Scholar] [CrossRef]
- Ocak, H.; Bilgin-Eran, B.; Tschierske, C.; Baumeister, U.; Pelzl, G. Effect of fluorocarbon chains on the mesomorphic properties of chiral imines and their complexes with copper(II). J. Mater. Chem. 2009, 19, 6995–7001. [Google Scholar] [CrossRef]
- Aslanidis, P.; Kyritsis, S.; Lalia-Kantouri, M.; Wicher, B.; Gdaniec, M. Copper(I) halide complexes of 2-thiohydantoin and 5,5-diphenyl-2-thiohydantoin. Polyhedron 2012, 48, 140–145. [Google Scholar] [CrossRef]
- Aslanidis, P.; Gaki, V.; Chrissafis, K.; Lalia-Kantouri, M. Luminescence and thermal behavior by simultaneous TG/DTG–DTA coupled with MS of neutral copper (I) complexes with heterocyclic thiones. J. Therm. Anal. Calorim. 2011, 103, 525–531. [Google Scholar] [CrossRef]
- Brammer, L.; Bruton, E.A.; Sherwood, P. Understanding the Behavior of Halogens as Hydrogen Bond Acceptors. Cryst. Growth Des. 2001, 1, 277–290. [Google Scholar] [CrossRef]
- Tschierske, C. Fluorinated liquid crystals: Design of soft nanostructures and increased complexity of self-assembly by perfluorinated segments. Top Curr. Chem. 2012, 318, 1–108. [Google Scholar] [CrossRef]
- Hori, K.; Maeda, M.; Yano, M.; Kunugi, M. The effect of fluorination (2): Dependence of alkyl chain length on the crystal structures of mesogenic alkyl 4-[2-(perfluorohexyl)ethoxy]benzoate. Liq. Cryst. 2011, 38, 287–293. [Google Scholar] [CrossRef]
- Wang, S.; Tian, H.; Ren, C.; Yu, J.; Sun, M. Electronic and optical properties of heterostructures based on transition metal dichalcogenides and graphene-like zinc oxide. Sci. Rep. 2018, 8, 12009. [Google Scholar] [CrossRef]
- Wang, S.; Ukhtary, M.S.; Saito, R. Strain effect on circularly polarized electroluminescence in transition metal dichalcogenides. Phys. Rev. Res. 2020, 2, 033340. [Google Scholar] [CrossRef]
- Takeda, H.; Monma, Y.; Sugiyama, H.; Uekusa, H.; Ishitani, O. Development of Visible-Light Driven Cu(I) Complex Photosensitizers for Photocatalytic CO2 Reduction. Front. Chem. 2019, 7, 418. [Google Scholar] [CrossRef]
- Armaroli, N.; Accorsi, G.; Cardinali, F.; Listorti, A. Photochemistry and Photophysics of Coordination Compounds: Copper. Top Curr. Chem. 2007, 280, 69–115. [Google Scholar] [CrossRef]
- Cariati, E.; Lucenti, E.; Botta, C.; Giovanella, U.; Marinotto, D.; Righetto, S. Cu(I) hybrid inorganic–organic materials with intriguing stimuli responsive and optoelectronic properties. Coord. Chem. Rev. 2016, 306, 566–614. [Google Scholar] [CrossRef]
- Binnemans, K. Luminescence of metallomesogens in the liquid crystal state. J. Mater. Chem. 2009, 19, 448–453. [Google Scholar] [CrossRef]
- Kremer, F.; Schonhals, A. (Eds.) Broadband Dielectric Spectroscopy; Springer-Verlag: Berlin/Heidelberg, Germany, 2003. [Google Scholar] [CrossRef]
- Jonscher, A.K. The ‘universal’ dielectric response. Nature 1977, 267, 673–679. [Google Scholar] [CrossRef]
- Dyre, J.C.; Maass, P.; Roling, B.; Sidebottom, D.L. Fundamental questions relating to ion conduction in disordered solids. Rep. Prog. Phys. 2009, 72, 046501. [Google Scholar] [CrossRef]
- Almond, D.; West, A. Impedance and modulus spectroscopy of “real” dispersive conductors. Solid State Ion. 1983, 11, 57–64. [Google Scholar] [CrossRef]
- Dyre, J.C. On the mechanism of glass ionic conductivity. J. Non-Cryst. Solids 1986, 88, 271–280. [Google Scholar] [CrossRef]
- Debbebi, I.S.; Megdiche-Borchani, S.; Cheikhrouhou-Koubaa, W.; Cheikhrouhou, A. Study of complex impedance spectroscopic properties of La0.7−xDyxSr0.3 MnO3 perovskite oxides. R. Soc. Open Sci. 2018, 5, 172201. [Google Scholar] [CrossRef]
- Dhahri, A.; Dhahri, E.; Hlil, E.K. Electrical conductivity and dielectric behaviour of nanocrystalline La0.6Gd0.1Sr0.3Mn0.75Si0.25O3. RSC Adv. 2018, 8, 9103–9111. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Pal, J.; Kaur, S.; Malhi, P.S.; Singh, M.; Babu, P.; Singh, A. The structural and magnetic properties, non-Debye relaxation and hopping mechanism in PbxNd1−xFeO3 (where x = 0.1, 0.2 and 0.3) solid solutions. J. Asian Ceram. Soc. 2019, 7, 133–140. [Google Scholar] [CrossRef]






| Compound | Transitions (T/K, ∆H/kJmol−1) 1,2 |
|---|---|
| 1 (BTU) | 1st: Cr 386 (45.1) Iso 373 (2.1) SmA 365 (33.2) Cr 2nd: Cr 385 (45.0) Iso 373 (2.0) SmA 364 (32.9) Cr |
| Cu2a | 1st: g 318 Colh 430 (22.9) Iso 429 (23.0) Colh 318 g 2nd: g 318 Colh 429 (22.8) Iso 428 (23.0) Colh 318 g |
| Cu2b | 1st: Cr 363 (7.9) Cr’ 412 (18.0) Colh 441 (16.7) Iso 440 (16.9) Colh 313 g 2nd: g 313 Colh (17.1) 440 Iso 439 (17.2) Colh 313 g |
| Compound | Mesophase | T(K) | Indexation | d-Spacing | d-Spacing | Lattice |
|---|---|---|---|---|---|---|
| exp.(Å) | calc. (Å) | parameter (Å) a | ||||
| Cu2a | Colh | 388 | 100 | 24.94 | 24.94 | a = 28.80 |
| 110 | 14.24 | 14.40 | ||||
| 200 | 12.34 | 12.47 | ||||
| 210 | 9.28 | 9.43 | ||||
| 300 | 8.22 | 8.31 | ||||
| Cu2b | Colh | 388 | 100 | 25.52 | 25.52 | a = 29.48 |
| 110 | 14.67 | 14.73 | ||||
| 200 | 12.62 | 12.76 | ||||
| 210 | 9.52 | 9.64 | ||||
| 300 | 8.42 | 8.51 |
| Compound | Absorption, λmax/nm (ε × 10−3/M−1 cm−1) | Emission | ||
|---|---|---|---|---|
| Solid λem/nm (λexc = 365nm) | τ/μs | Φ (%) | ||
| 1 | 290 (20.4), 313 (26.4) | - | - | - |
| Cu2a | 291 (45.7), 317 (54.9) | 545, 580 (sh) | 11, 172 | 2.5 |
| Cu2b | 290 (sh, 34.4), 316 (41.4) | 546, 580 (sh) | 9, 176 | 8.0 |
| Compound | Conductivity (σ/S·cm−1) | EA (Mesophase) | |||
|---|---|---|---|---|---|
| Glass | Mesophase | Isotropic | |||
| 320 K | 370 K | 400 K | 450 K | (eV) | |
| Cu2a | 5.05 × 10−12 | 1.32 × 10−9 | 1.12 × 10−8 | 1.88 × 10−6 | 1.2 |
| Cu2b | 2.64 × 10−12 | 1.86 × 10−9 | 4.98 × 10−8 | 3.03 × 10−6 | 1.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cîrcu, V.; Ganea, C.P.; Secu, M.; Manaila-Maximean, D.; Marinescu, G.C.; Popescu, R.G.; Pasuk, I. Columnar Liquid Crystals of Copper(I) Complexes with Ionic Conductivity and Solid State Emission. Molecules 2023, 28, 4196. https://doi.org/10.3390/molecules28104196
Cîrcu V, Ganea CP, Secu M, Manaila-Maximean D, Marinescu GC, Popescu RG, Pasuk I. Columnar Liquid Crystals of Copper(I) Complexes with Ionic Conductivity and Solid State Emission. Molecules. 2023; 28(10):4196. https://doi.org/10.3390/molecules28104196
Chicago/Turabian StyleCîrcu, Viorel, Constantin P. Ganea, Mihail Secu, Doina Manaila-Maximean, George Cătălin Marinescu, Roua Gabriela Popescu, and Iuliana Pasuk. 2023. "Columnar Liquid Crystals of Copper(I) Complexes with Ionic Conductivity and Solid State Emission" Molecules 28, no. 10: 4196. https://doi.org/10.3390/molecules28104196
APA StyleCîrcu, V., Ganea, C. P., Secu, M., Manaila-Maximean, D., Marinescu, G. C., Popescu, R. G., & Pasuk, I. (2023). Columnar Liquid Crystals of Copper(I) Complexes with Ionic Conductivity and Solid State Emission. Molecules, 28(10), 4196. https://doi.org/10.3390/molecules28104196









