Sigma-1 Receptor Activation Improves Oligodendrogenesis and Promotes White-Matter Integrity after Stroke in Mice with Diabetic Mellitus
Abstract
1. Introduction
2. Results
2.1. Sigma-1 Receptor Activation Reverses Cognitive Dysfunction and Improves Long-Term Histological Deficits in Mice with Diabetes and Stroke
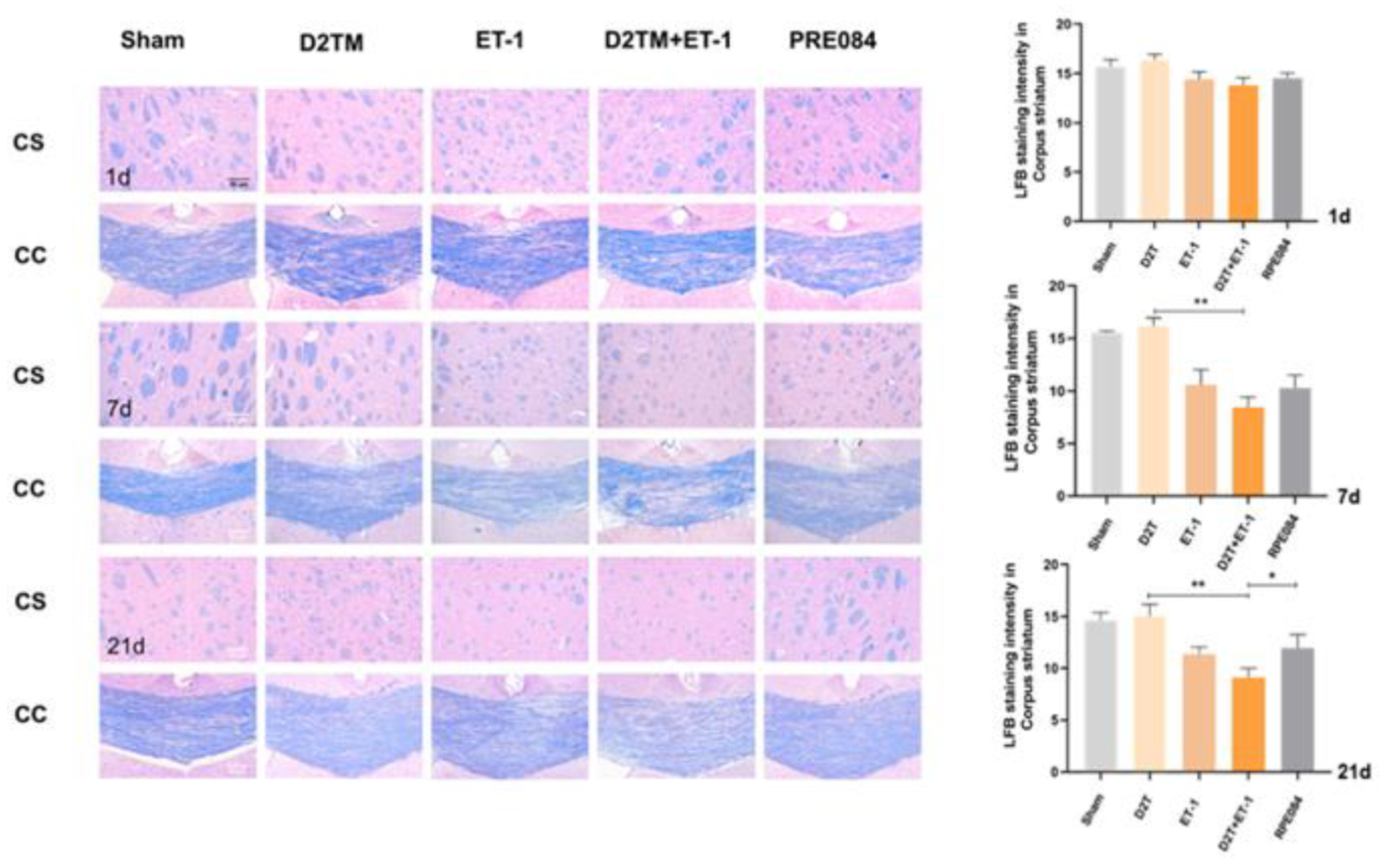
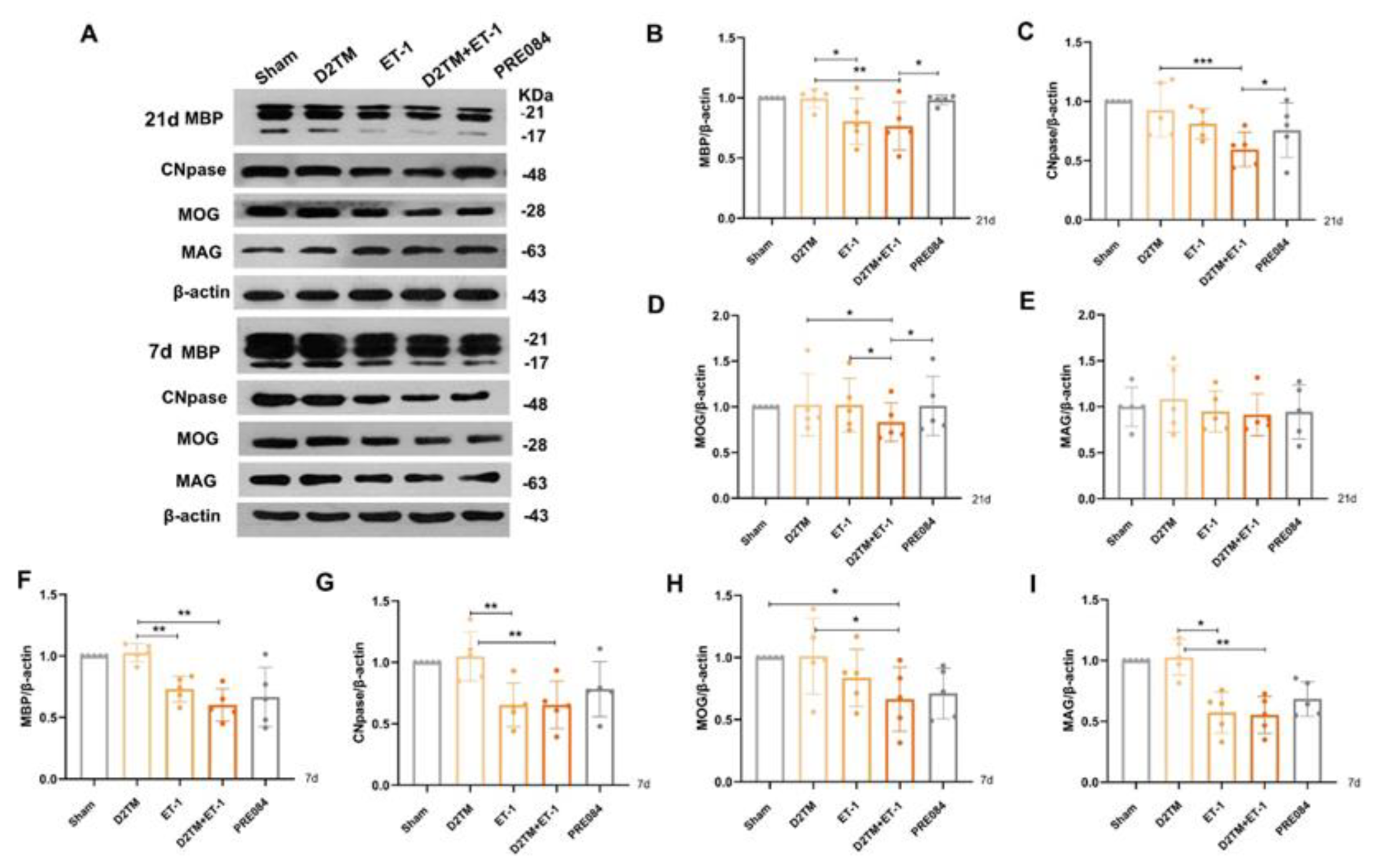
2.2. Sigma-1 Receptor Activation Prevents Levels of Brain Demyelination and White-Matter Damage in Mice with Diabetes and Stroke
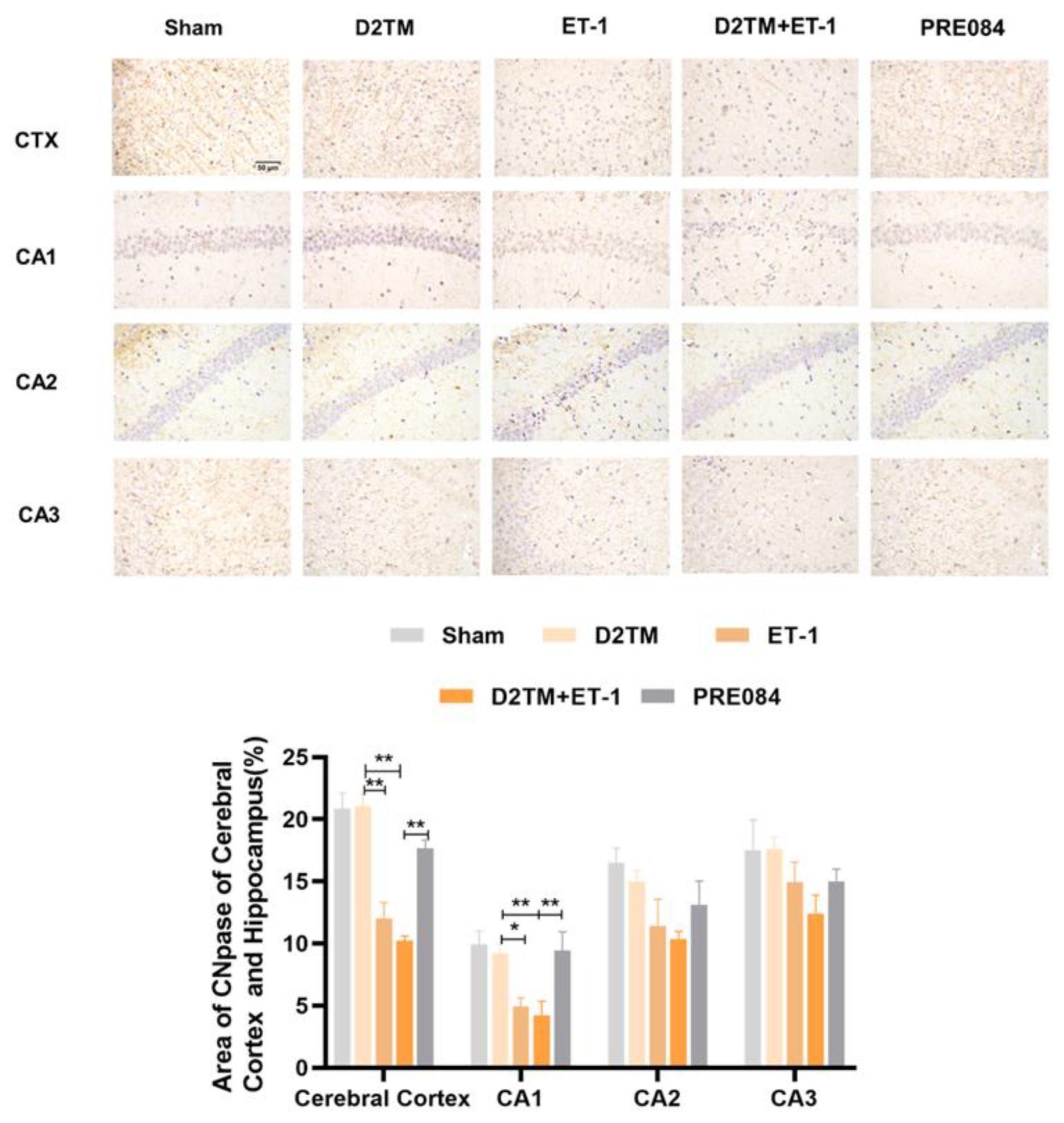
2.3. Sigma-1 Receptor Activation Promotes Mature Oligodendrogenesis and Myelin Regeneration in Mice with Diabetes and Stroke
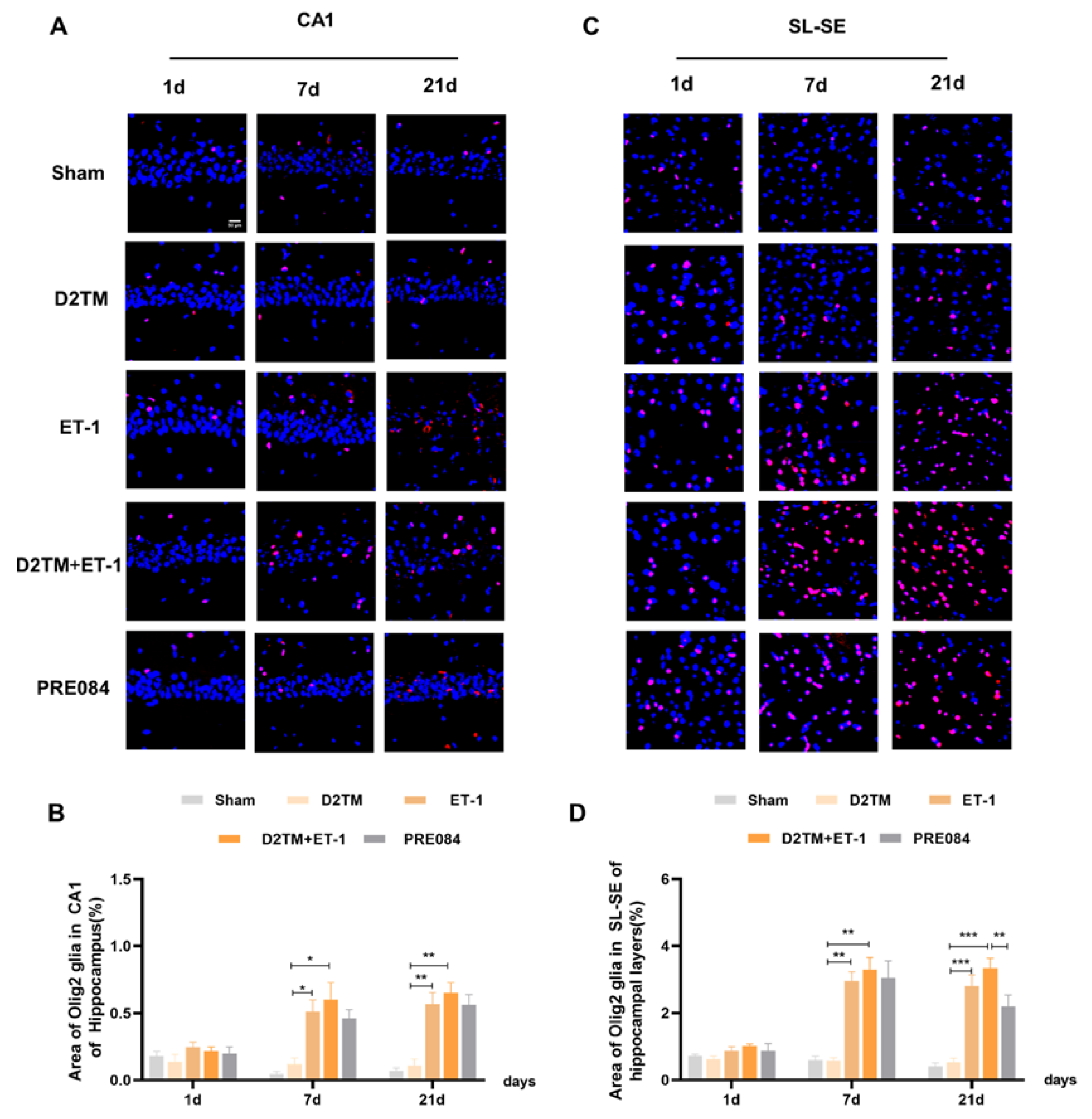
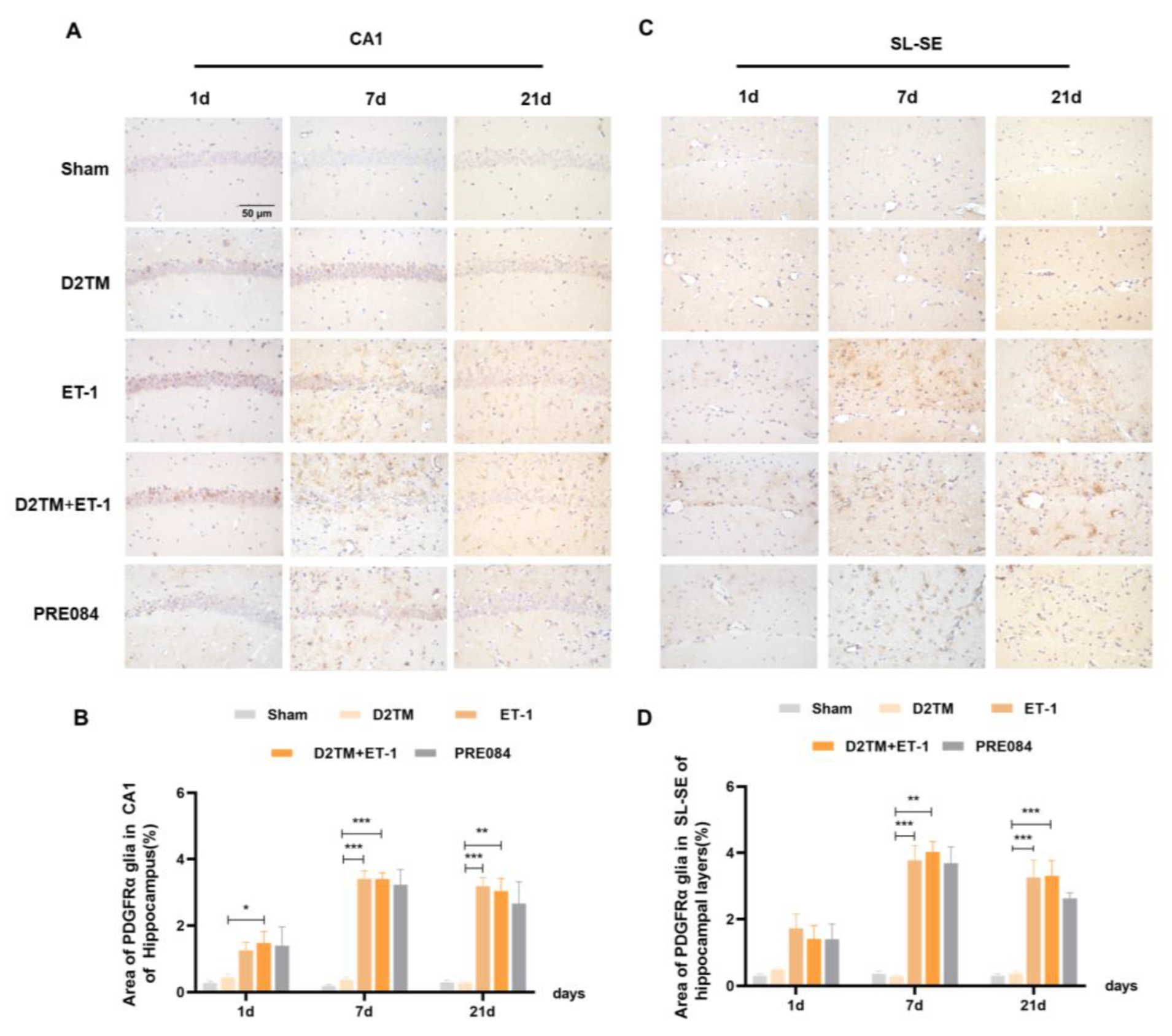
2.4. Sigma-1 Receptor Activation Promotes Oligodendrogenesis Differentiation in Mice with Diabetes and Stroke
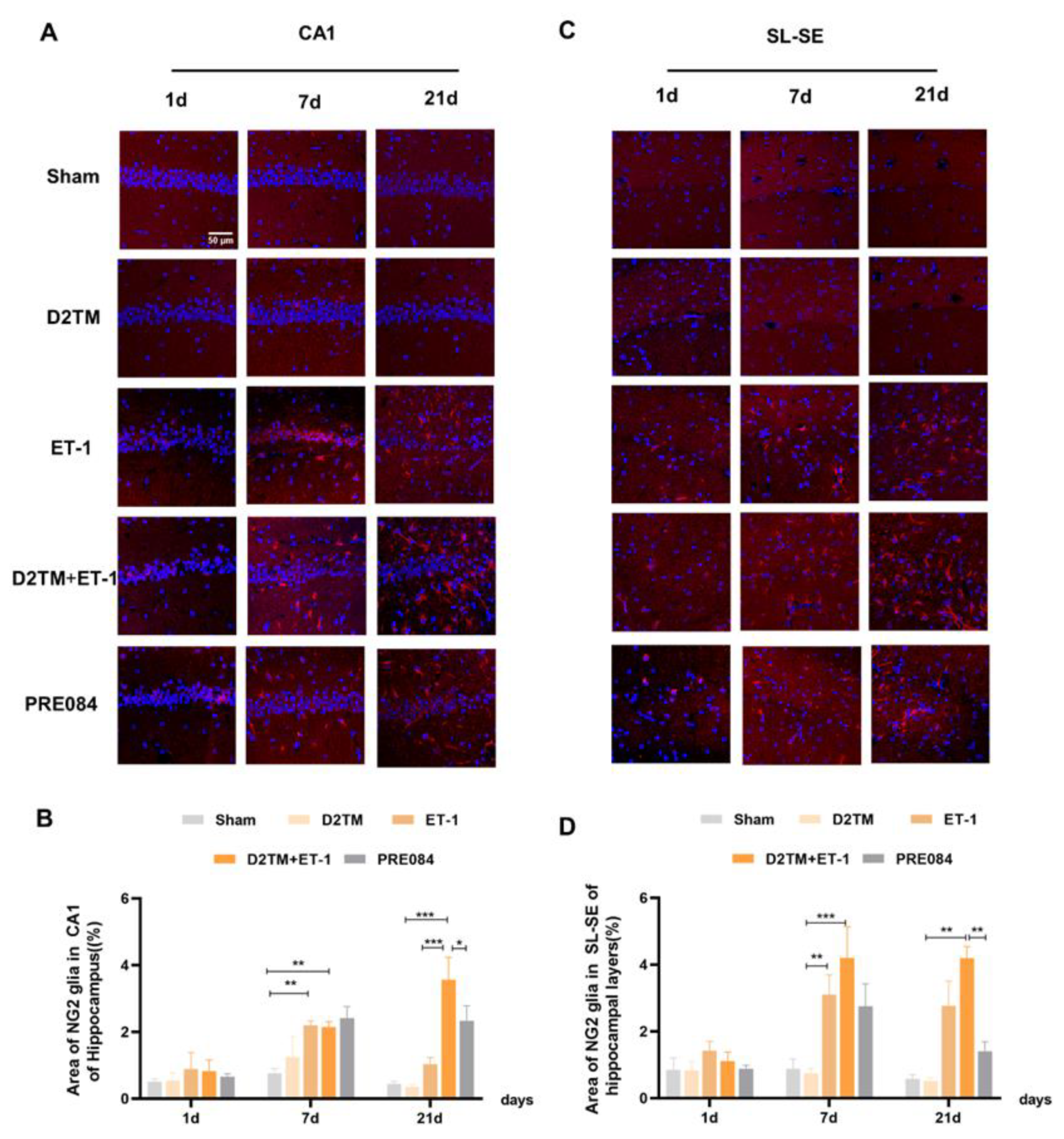
2.5. Sigma-1 Receptor Activation Regulates OPC Proliferation in Mice with Diabetes and Stroke
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Induction of Hyperglycemia
4.3. Bilateral Hippocampal Injection of ET-1
4.4. Drug and Treatment Schedule
4.5. Y-Maze Test
4.6. Novel Object Recognition Test
4.7. Passive Avoidance Test
4.8. LFB Staining and Nissl Staining
4.9. Immunohistochemistry
4.10. Immunofluorescence
4.11. Western Blotting
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Virani, S.S.; Alonso, A.; Aparicio, H.J.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Cheng, S.; Delling, F.N.; et al. Heart disease and stroke statistics—2021 update: A report from the American Heart Association. Circulation 2021, 143, e254–e743. [Google Scholar] [CrossRef]
- Hill, M.D. Stroke and diabetes mellitus. Handb. Clin. Neurol. 2014, 126, 167–174. [Google Scholar]
- Sander, D.; Sander, K.; Poppert, H. Stroke in type 2 diabetes. Br. J. Diabetes Vasc. Dis. 2008, 8, 222–229. [Google Scholar] [CrossRef]
- Luitse, M.J.; Biessels, G.J.; Rutten, G.E.; Kappelle, L.J. Diabetes, hyperglycaemia, and acute ischaemic stroke. Lancet Neurol. 2012, 11, 261–271. [Google Scholar] [CrossRef]
- Putaala, J.; Liebkind, R.; Gordin, D.; Thorn, L.M.; Haapaniemi, E.; Forsblom, C.; Groop, P.H.; Kaste, M.; Tatlisumak, T. Diabetes mellitus and ischemic stroke in the young: Clinical features and long-term prognosis. Neurology 2011, 76, 1831–1837. [Google Scholar] [CrossRef]
- Srinivasan, K.; Sharma, S.S. Augmentation of endoplasmic reticulum stress in cerebral ischemia/reperfusion injury associated with comorbid type 2 diabetes. Neurol. Res. 2011, 33, 858–865. [Google Scholar] [CrossRef]
- Srinivasan, K.; Sharma, S.S. Sodium phenylbutyrate ameliorates focal cerebral ischemic/reperfusion injury associated with comorbid type 2 diabetes by reducing endoplasmic reticulum stress and DNA fragmentation. Behav. Brain Res. 2011, 225, 110–116. [Google Scholar] [CrossRef]
- Pantoni, L.; Garcia, J.H.; Gutierrez, J.A. Cerebral White Matter Is Highly Vulnerable to Ischemia. Stroke 1996, 27, 1641–1646. [Google Scholar] [CrossRef]
- Valeriani, V.; Dewar, D.; McCulloch, J. Quantitative Assessment of Ischemic Pathology in Axons, Oligodendrocytes, and Neurons: Attenuation of Damage after Transient Ischemia. J. Cereb. Blood Flow Metab. 2000, 20, 765–771. [Google Scholar] [CrossRef]
- Baumann, N.; Pham-Dinh, D. Biology of oligodendrocyte and myelin in the mammalian central nervous system. Physiol. Rev. 2001, 81, 871–927. [Google Scholar] [CrossRef]
- Zhang, R.; Chopp, M.; Zhang, Z.G. Oligodendrogenesis after cerebral ischemia. Front. Cell. Neurosci. 2013, 7, 201. [Google Scholar] [CrossRef]
- Goldman, S.A.; Osorio, J. So many progenitors, so little myelin. Nat. Neurosci. 2014, 17, 483–485. [Google Scholar] [CrossRef]
- Hayashi, T.; Su, T.-P. Sigma-1 Receptor Chaperones at the ER-Mitochondrion Interface Regulate Ca2+ Signaling and Cell Survival. Cell 2007, 131, 596–610. [Google Scholar] [CrossRef]
- Alonso, G.; Phan, V.-L.; Guillemain, I.; Saunier, M.; Legrand, A.; Anoal, M.; Maurice, T. Immunocytochemical localization of the sigma1 receptor in the adult rat central nervous system. Neuroscience 2000, 97, 155–170. [Google Scholar] [CrossRef]
- Maurice, T.; Su, T.-P. The pharmacology of sigma-1 receptors. Pharmacol. Ther. 2009, 124, 195–206. [Google Scholar] [CrossRef]
- Palacios, G.; Muro, A.; Verdú, E.; Pumarola, M.; Vela, J.M. Immunohistochemical localization of the sigma1 receptor in Schwann cells of rat sciatic nerve. Brain Res. 2004, 1007, 65–70. [Google Scholar] [CrossRef]
- Lisak, R.P.; Nedelkoska, L.; Benjamins, J.A. Sigma-1 receptor agonists as potential protective therapies in multiple sclerosis. J. Neuroimmunol. 2020, 342, 577188. [Google Scholar] [CrossRef]
- Hayashi, T.; Su, T.P. Sigma-1 receptors at galactosylceramide-enriched lipid microdomains regulate oligoden-drocyte differentiation. Proc. Natl. Acad. Sci. USA 2004, 101, 14949–14954. [Google Scholar] [CrossRef]
- Liu, D.-Y.; Chi, T.-Y.; Ji, X.-F.; Liu, P.; Qi, X.-X.; Zhu, L.; Wang, Z.-Q.; Li, L.; Chen, L.; Zou, L.-B. Sigma-1 receptor activation alleviates blood-brain barrier dysfunction in vascular dementia mice. Exp. Neurol. 2018, 308, 90–99. [Google Scholar] [CrossRef]
- Xu, Q.; Ji, X.F.; Chi, T.Y.; Liu, P.; Jin, G.; Gu, S.L.; Zou, L.B. Sigma 1 receptor activation regulates brain-derived neurotrophic factor through NR2A-CaMKIV-TORC1 pathway to rescue the impairment of learning and memory induced by brain ischaemia/reperfusion. Psychopharmacology 2015, 232, 1779–1791. [Google Scholar] [CrossRef]
- Zhao, X.; Zhu, L.; Liu, D.; Chi, T.; Ji, X.; Liu, P.; Yang, X.; Tian, X.; Zou, L. Sigma-1 receptor protects against endoplasmic reticulum stress-mediated apoptosis in mice with cerebral ische-mia/reperfusion injury. Apoptosis 2019, 24, 157–167. [Google Scholar] [CrossRef]
- Mundugaru, R.; Sivanesan, S.; Popa-Wagner, A.; Udaykumar, P.; Kirubagaran, R.; Guruprasad, K.P.; Vidyadhara, D.J. Pluchea lanceolata protects hippocampal neurons from endothelin-1 induced ischemic injury to ameliorate cog-nitive deficits. J. Chem. Neuroanat. 2018, 94, 75–85. [Google Scholar] [CrossRef]
- Sharkey, J. Perivascular microapplication of endothelin-1: A new model of focal cerebral ischaemia in the rat. J. Cereb. Blood Flow Metab. 1993, 13, 865–871. [Google Scholar] [CrossRef]
- O’Neill, M.J.; Clemens, J.A. Rodent Models of Focal Cerebral Ischemia. Curr. Protoc. Neurosci. 2000, 12, 9.6.1–9.6.32. [Google Scholar] [CrossRef]
- Mecca, A.P.; O’Connor, T.E.; Katovich, M.J.; Sumners, C. Candesartan pretreatment is cerebroprotective in a rat model of endothelin-1-induced middle cerebral artery occlusion. Exp. Physiol. 2009, 94, 937–946. [Google Scholar] [CrossRef]
- Sheng, T.; Zhang, X.; Wang, S.; Zhang, J.; Lu, W.; Dai, Y. Endothelin-1-induced mini-stroke in the dorsal hippocampus or lateral amygdala results in deficits in learning and memory. J. Biomed. Res. 2015, 29, 362–369. [Google Scholar] [CrossRef]
- Faraji, J.; Soltanpour, N.; Moeeini, R.; Roudaki, S.; Soltanpour, N.; Abdollahi, A.A.; Metz, G.A. Topographical disorientation after ischemic mini infarct in the dorsal hippocampus: Whispers in silence. Front. Behav. Neurosci. 2014, 8, 261. [Google Scholar] [CrossRef]
- Muranyi, M.; Fujioka, M.; He, Q.; Han, A.; Yong, G.; Csiszar, K.; Li, P.-A. Diabetes Activates Cell Death Pathway After Transient Focal Cerebral Ischemia. Diabetes 2003, 52, 481–486. [Google Scholar] [CrossRef][Green Version]
- Jing, L.; He, Q.; Zhang, J.-Z.; Li, P.A. Temporal Profile of Astrocytes and Changes of Oligodendrocyte-Based Myelin Following Middle Cerebral Artery Occlusion in Diabetic and Non-diabetic Rats. Int. J. Biol. Sci. 2013, 9, 190–199. [Google Scholar] [CrossRef]
- Tomsick, T.A. Intravenous thrombolysis for acute ischemic stroke. J. Vasc. Interv. Radiol. 2004, 15, S67–S76. [Google Scholar] [CrossRef]
- Olsen, T.S.; Lassen, N.A. A dynamic concept of middle cerebral artery occlusion and cerebral infarction in the acute state based on interpreting severe hyperemia as a sign of embolic migration. Stroke 1984, 15, 458–468. [Google Scholar] [CrossRef]
- Xu, Q.; Ji, X.-F.; Chi, T.-Y.; Liu, P.; Jin, G.; Chen, L.; Zou, L.-B. Sigma-1 receptor in brain ischemia/reperfusion: Possible role in the NR2A-induced pathway to regulate brain-derived neurotrophic factor. J. Neurol. Sci. 2017, 376, 166–175. [Google Scholar] [CrossRef]
- Ma, S.; Wang, J.; Wang, Y.; Dai, X.; Xu, F.; Gao, X.; Johnson, J.; Xu, N.; Leak, R.K.; Hu, X.; et al. Diabetes Mellitus Impairs White Matter Repair and Long-Term Functional Deficits After Cerebral Ischemia. Stroke 2018, 49, 2453–2463. [Google Scholar] [CrossRef]
- Han, L.; Cai, W.; Mao, L.; Liu, J.; Li, P.; Leak, R.K.; Xu, Y.; Hu, X.; Chen, J. Rosiglitazone Promotes White Matter Integrity and Long-Term Functional Recovery After Focal Cerebral Ischemia. Stroke 2015, 46, 2628–2636. [Google Scholar] [CrossRef]
- Zhang, L.; Chopp, M.; Zhang, Y.; Xiong, Y.; Li, C.; Sadry, N.; Rhaleb, I.; Lu, M.; Zhang, Z.G. Diabetes Mellitus Impairs Cognitive Function in Middle-Aged Rats and Neurological Recovery in Middle-Aged Rats After Stroke. Stroke 2016, 47, 2112–2118. [Google Scholar] [CrossRef]
- Liu, H.; Yang, Y.; Xia, Y.; Zhu, W.; Leak, R.K.; Wei, Z.; Wang, J.; Hu, X. Aging of cerebral white matter. Ageing Res. Rev. 2017, 34, 64–76. [Google Scholar] [CrossRef]
- Liu, X.S.; Chopp, M.; Pan, W.L.; Wang, X.L.; Fan, B.Y.; Zhang, Y.; Kassis, H.; Zhang, R.L.; Zhang, X.M.; Zhang, Z.G. MicroRNA-146a Promotes Oligodendrogenesis in Stroke. Mol. Neurobiol. 2017, 54, 227–237. [Google Scholar] [CrossRef]
- McIver, S.; Muccigrosso, M.; Gonzales, E.; Lee, J.; Roberts, M.; Sands, M.; Goldberg, M. Oligodendrocyte degeneration and recovery after focal cerebral ischemia. Neuroscience 2010, 169, 1364–1375. [Google Scholar] [CrossRef]
- Back, S.A.; Han, B.H.; Luo, N.L.; Chricton, C.A.; Xanthoudakis, S.; Tam, J.; Arvin, K.L.; Holtzman, D.M. Selective Vulnerability of Late Oligodendrocyte Progenitors to Hypoxia–Ischemia. J. Neurosci. 2002, 22, 455–463. [Google Scholar] [CrossRef]
- Jin, X.; Riew, T.-R.; Kim, S.; Kim, H.L.; Lee, H.L.K.A.M.-Y. Spatiotemporal Profile and Morphological Changes of NG2 Glia in the CA1 Region of the Rat Hippocampus after Transient Forebrain Ischemia. Exp. Neurobiol. 2020, 29, 50–69. [Google Scholar] [CrossRef]
- Bell, L.A.; Wallis, G.J.; Wilcox, K.S. Reactivity and increased proliferation of NG2 cells following central nervous system infection with Theiler’s murine encephalomyelitis virus. J. Neuroinflamm. 2020, 17, 1–14. [Google Scholar] [CrossRef]
- Motavaf, M.; Piao, X. Oligodendrocyte Development and Implication in Perinatal White Matter Injury. Front. Cell. Neurosci. 2021, 15, 764486. [Google Scholar] [CrossRef]
- Chu, M. Focal cerebral ischemia activates neurovascular restorative dynamics in mouse brain. Front. Biosci. 2012, E4, 1926–1936. [Google Scholar] [CrossRef]
- Matsumoto, H.; Kumon, Y.; Watanabe, H.; Ohnishi, T.; Shudou, M.; Chuai, M.; Imai, Y.; Takahashi, H.; Tanaka, J. Accumulation of Macrophage-Like Cells Expressing NG2 Proteoglycan and Iba1 in Ischemic Core of Rat Brain after Transient Middle Cerebral Artery Occlusion. J. Cereb. Blood Flow Metab. 2008, 28, 149–163. [Google Scholar] [CrossRef]
- Cheng, X.; Wang, H.; Liu, C.; Zhong, S.; Niu, X.; Zhang, X.; Qi, R.; Zhao, S.; Zhang, X.; Qu, H.; et al. Dl-3-n-butylphthalide promotes remyelination process in cerebral white matter in rats subjected to ischemic stroke. Brain Res. 2019, 1717, 167–175. [Google Scholar] [CrossRef]
- Zhang, C.; Deng, J.; Liu, D.; Tuo, X.; Xiao, L.; Lai, B.; Yao, Q.; Liu, J.; Yang, H.; Wang, N. Nuciferine ameliorates hepatic steatosis in high-fat diet/streptozocin-induced diabetic mice through a PPARα/PPARγ coactivator-1α pathway. Br. J. Pharmacol. 2018, 175, 4218–4228. [Google Scholar] [CrossRef]
- Karelina, K.; Liu, Y.; Alzate-Correa, D.; Wheaton, K.L.; Hoyt, K.R.; Arthur, J.S.C.; Obrietan, K. Mitogen and stress-activated kinases 1/2 regulate ischemia-induced hippocampal progenitor cell proliferation and neurogenesis. Neuroscience 2015, 285, 292–302. [Google Scholar] [CrossRef]
- Karelina, K.; Alzate-Correa, D.; Obrietan, K. Ribosomal S6 kinase regulates ischemia-induced progenitor cell proliferation in the adult mouse hippocampus. Exp. Neurol. 2014, 253, 72–81. [Google Scholar] [CrossRef]
- Liu, D.; Yang, L.; Liu, P.; Ji, X.; Qi, X.; Wang, Z.; Chi, T.; Zou, L. Sigma–1 receptor activation alleviates blood–brain barrier disruption post cerebral ischemia stroke by stimulating the GDNF–GFRα1–RET pathway. Exp. Neurol. 2022, 347, 113867. [Google Scholar] [CrossRef]
- Liu, P.; Li, Y.; Yang, W.; Liu, D.; Ji, X.; Chi, T.; Guo, Z.; Li, L.; Zou, L. Prevention of Huntington’s disease-like behavioral deficits in R6/1 mouse by tolfenamic acid is associated with decreases in mutant huntingtin and oxidative stress. Oxidative Med. Cell. Longev. 2019, 2019, 4032428. [Google Scholar] [CrossRef]
- Wu, Q.; Miao, X.; Zhang, J.; Xiang, L.; Li, X.; Bao, X.; Du, S.; Wang, M.; Miao, S.; Fan, Y.; et al. Astrocytic YAP protects the optic nerve and retina in an experimental autoimmune encephalomyelitis model through TGF-β signaling. Theranostics 2021, 11, 8480–8499. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, H.-X.; Shi, S.-T.; Bai, Y.-L.; Zhe, X.; Zhang, S.-J.; Li, Y.-J. Interleukin-11 treatment protected against cerebral ischemia/reperfusion injury. Biomed. Pharmacother. 2019, 115, 108816. [Google Scholar] [CrossRef]
- Kanno, T.; Kurotaki, T.; Yamada, N.; Tomonari, Y.; Sato, J.; Tsuchitani, M.; Kobayashi, Y. Supplemental study on 2′, 3′-Cyclic Nucleotide 3′-Phosphodiesterase (CNPase) activity in developing rat spinal cord lesions induced by hexachlorophene and cuprizone. J. Vet. Med. Sci. 2019, 81, 1368–1372. [Google Scholar] [CrossRef]
- Anan’Ina, T.; Kisel, A.; Kudabaeva, M.; Chernysheva, G.; Smolyakova, V.; Usov, K.; Krutenkova, E.; Plotnikov, M.; Khodanovich, M. Neurodegeneration, Myelin Loss and Glial Response in the Three-Vessel Global Ischemia Model in Rat. Int. J. Mol. Sci. 2020, 21, 6246. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, W.; Yao, Y.; Zhang, H.; Hao, X.; Zhou, L.; Song, Z.; Wei, T.; Chi, T.; Liu, P.; Ji, X.; et al. Sigma-1 Receptor Activation Improves Oligodendrogenesis and Promotes White-Matter Integrity after Stroke in Mice with Diabetic Mellitus. Molecules 2023, 28, 390. https://doi.org/10.3390/molecules28010390
Song W, Yao Y, Zhang H, Hao X, Zhou L, Song Z, Wei T, Chi T, Liu P, Ji X, et al. Sigma-1 Receptor Activation Improves Oligodendrogenesis and Promotes White-Matter Integrity after Stroke in Mice with Diabetic Mellitus. Molecules. 2023; 28(1):390. https://doi.org/10.3390/molecules28010390
Chicago/Turabian StyleSong, Wenjing, Yang Yao, Heling Zhang, Xin Hao, Liping Zhou, Zhixiao Song, Tiantian Wei, Tianyan Chi, Peng Liu, Xuefei Ji, and et al. 2023. "Sigma-1 Receptor Activation Improves Oligodendrogenesis and Promotes White-Matter Integrity after Stroke in Mice with Diabetic Mellitus" Molecules 28, no. 1: 390. https://doi.org/10.3390/molecules28010390
APA StyleSong, W., Yao, Y., Zhang, H., Hao, X., Zhou, L., Song, Z., Wei, T., Chi, T., Liu, P., Ji, X., & Zou, L. (2023). Sigma-1 Receptor Activation Improves Oligodendrogenesis and Promotes White-Matter Integrity after Stroke in Mice with Diabetic Mellitus. Molecules, 28(1), 390. https://doi.org/10.3390/molecules28010390





