Comparative Study of the Synthetic Approaches and Biological Activities of the Bioisosteres of 1,3,4-Oxadiazoles and 1,3,4-Thiadiazoles over the Past Decade
Abstract
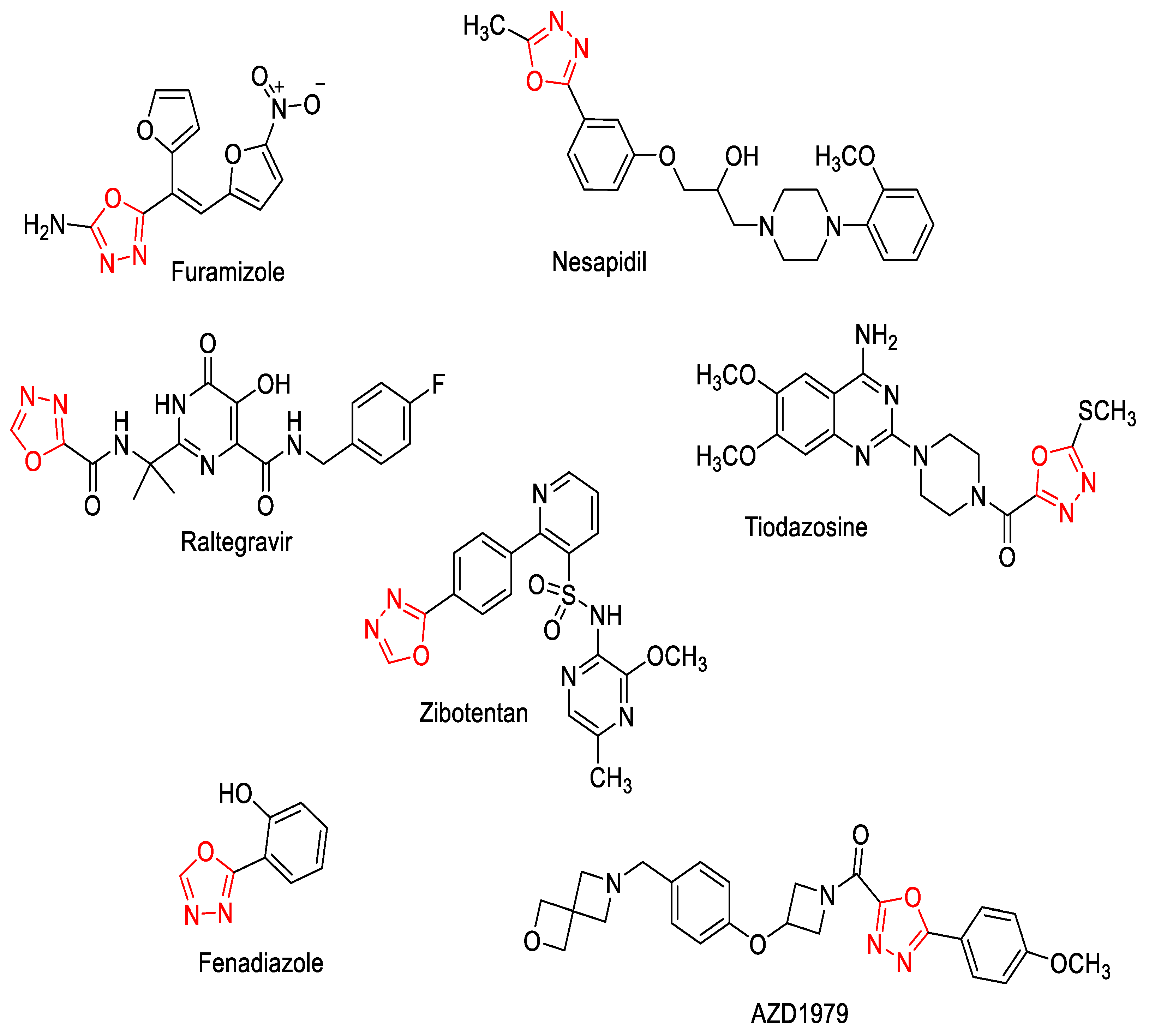
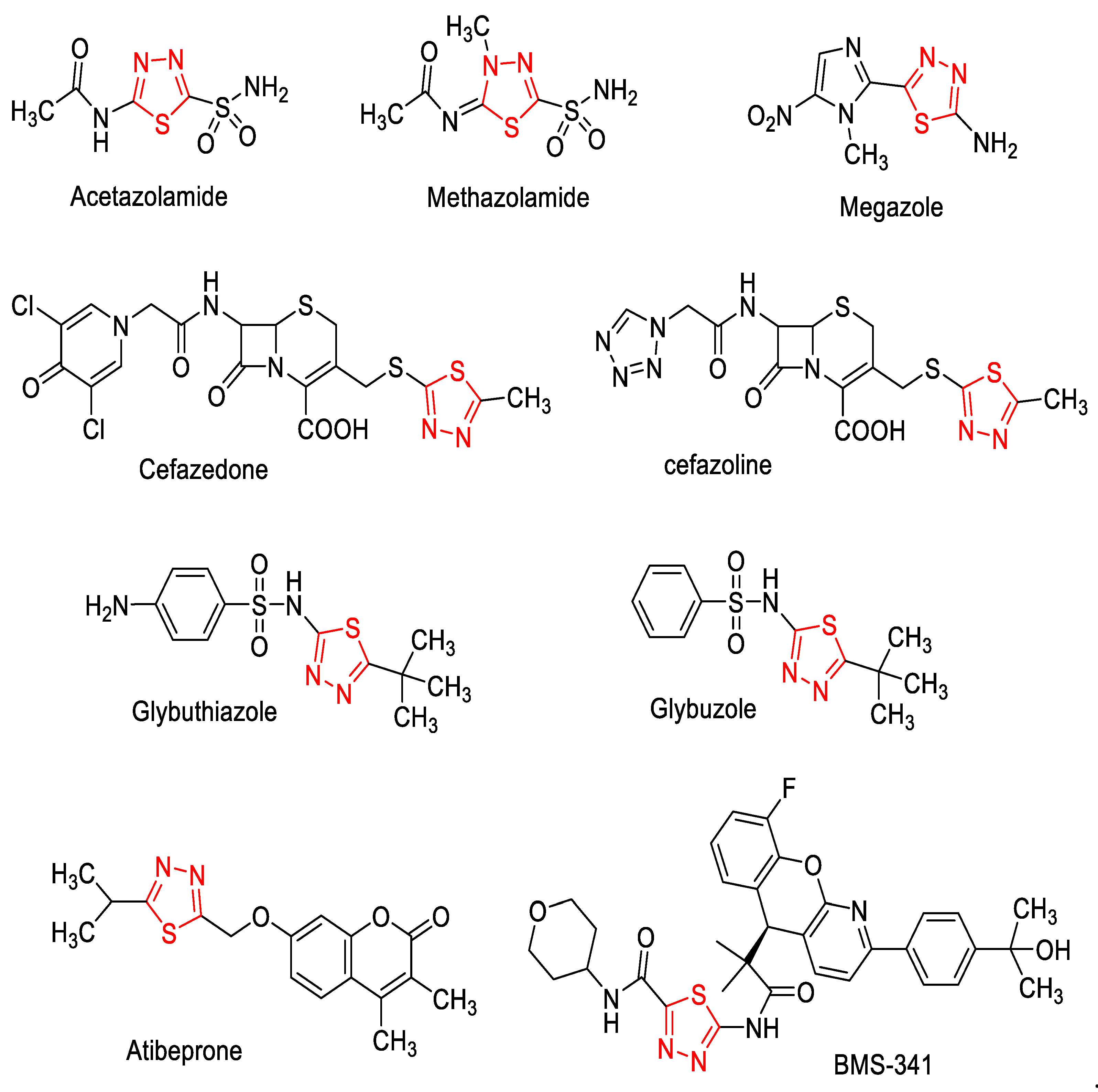
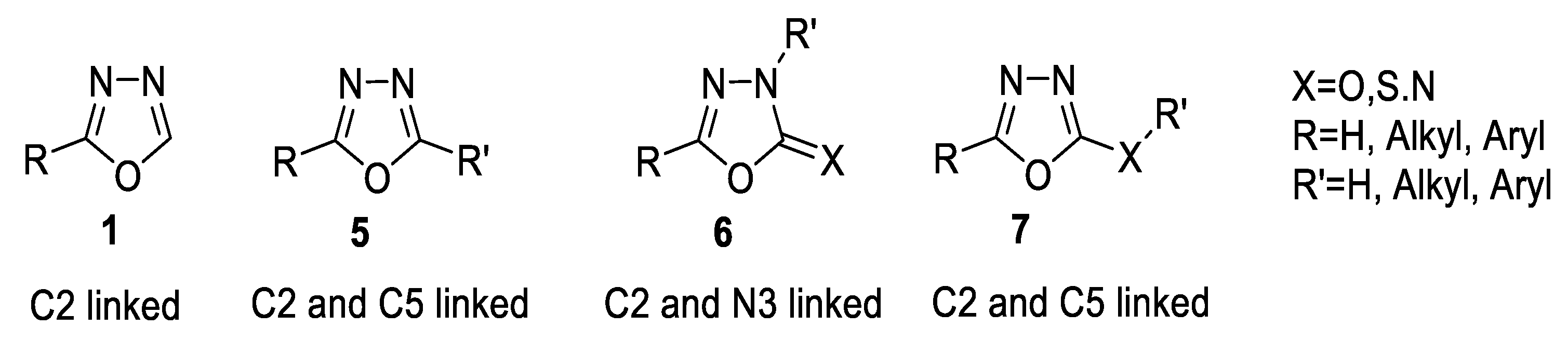
1. Introduction
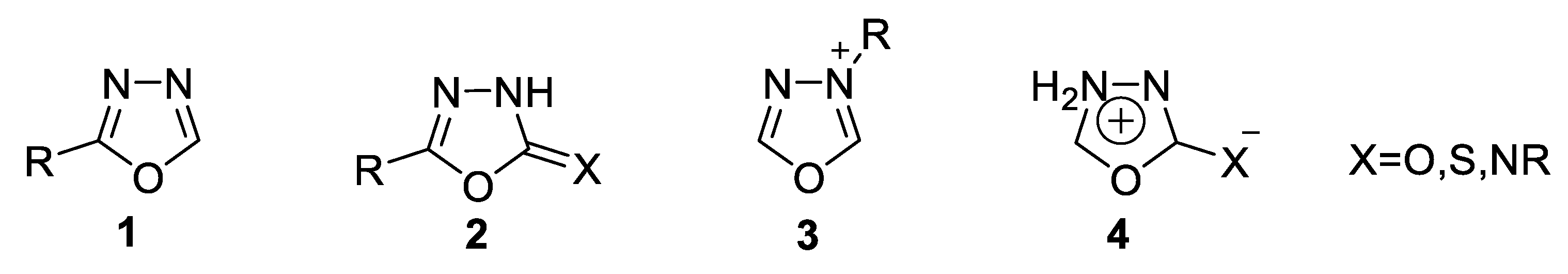
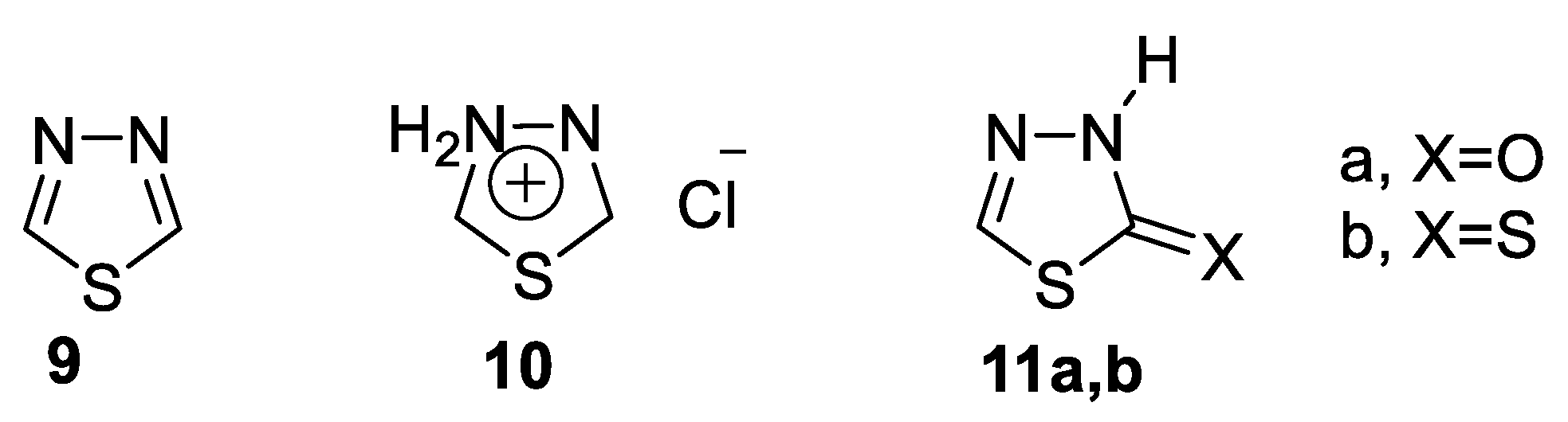
- Aromatic systems that include the neutral thiadiazoles 9 [45].
- Mesoionic systems that are defined as a poly-heteroatomic system containing a five-membered heterocyclic ring associated with conjugated p and π electrons and distinct regions of positive and negative charges 10. Mesoionic systems are dense and highly polarizable, with a net neutral electron charge; these characteristics allow mesoionic compounds to cross cellular membranes and interact with biological targets with distinct affinities [31].
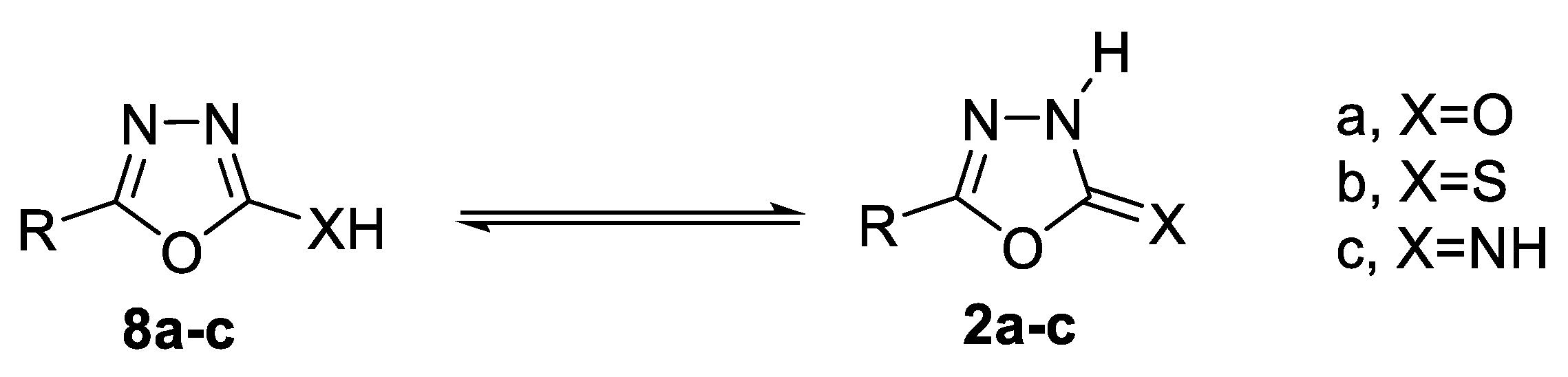
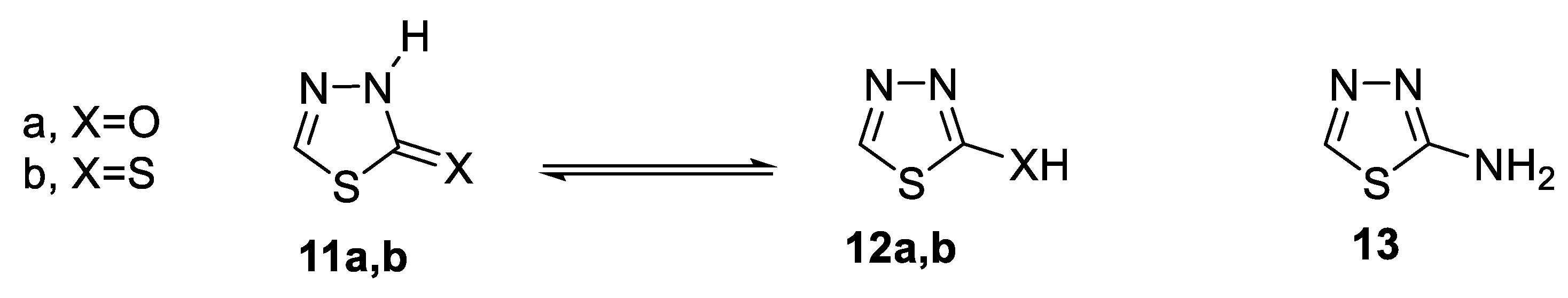
- 1,3,4-thiadiazol-2-one(thione) 11a,b [47].
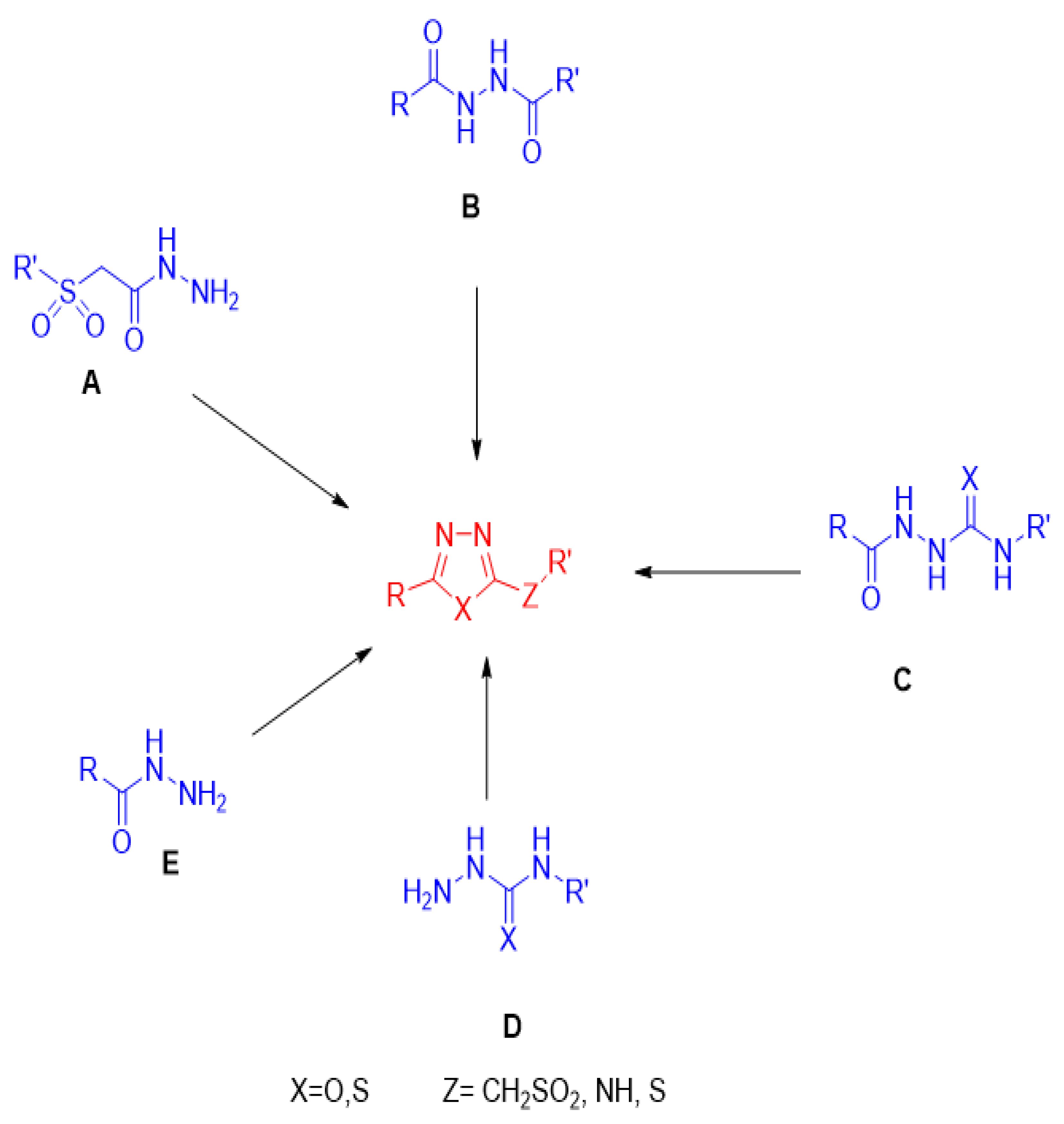
2. Synthetic Approaches for the Preparation of Different Classes of 1,3,4-Oxadiazole and 1,3,4-Thiadiazole Derivatives
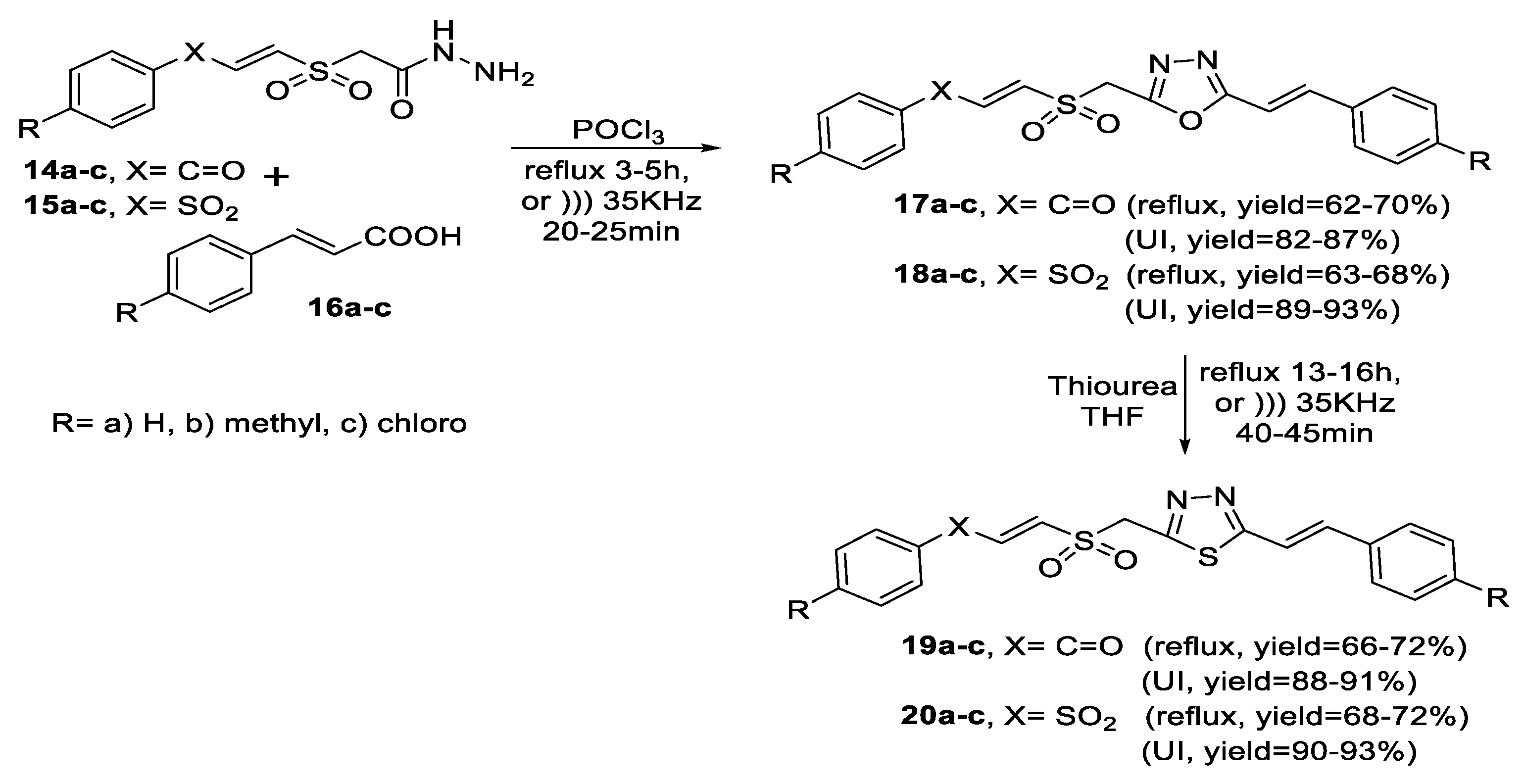
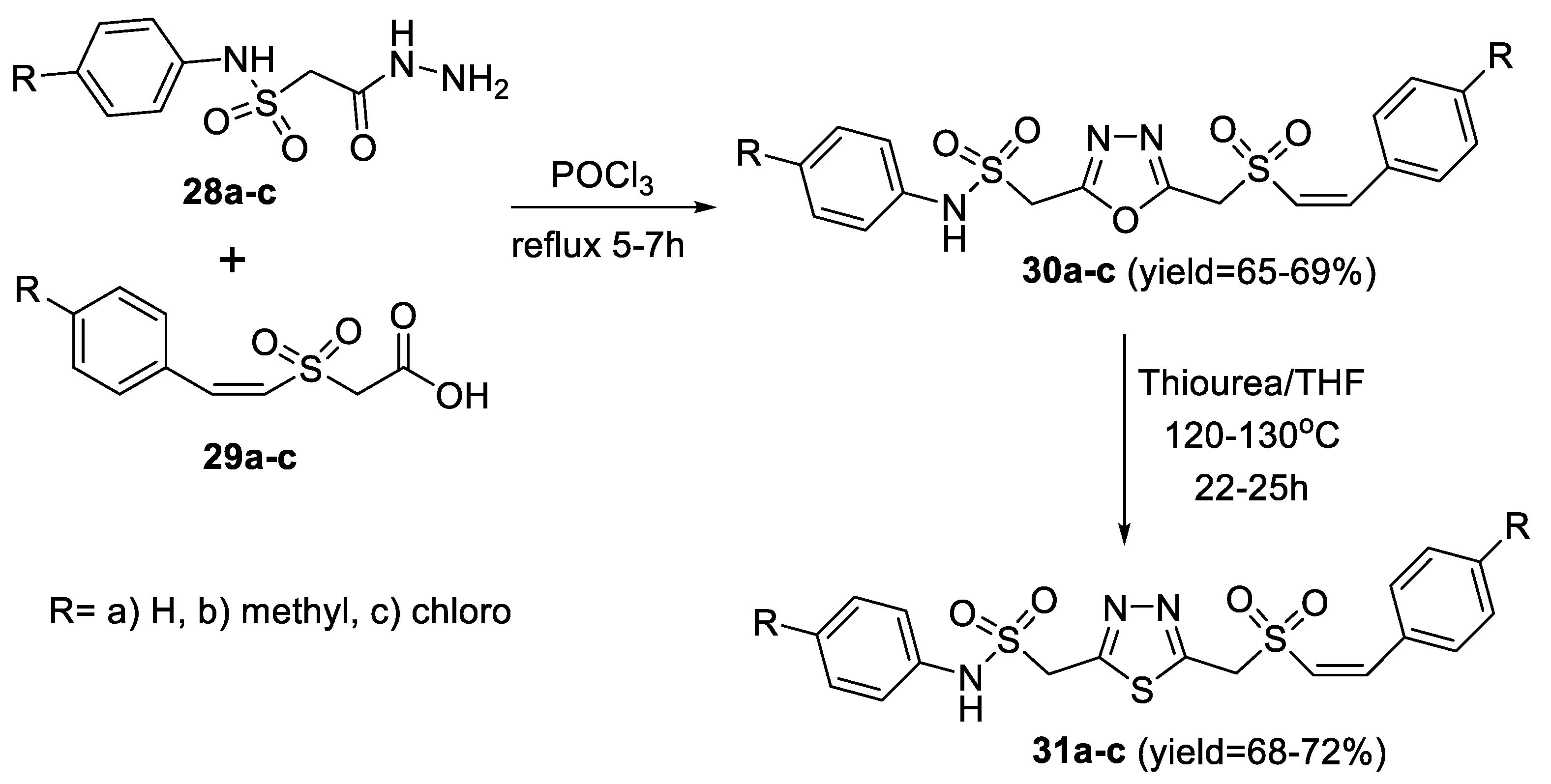
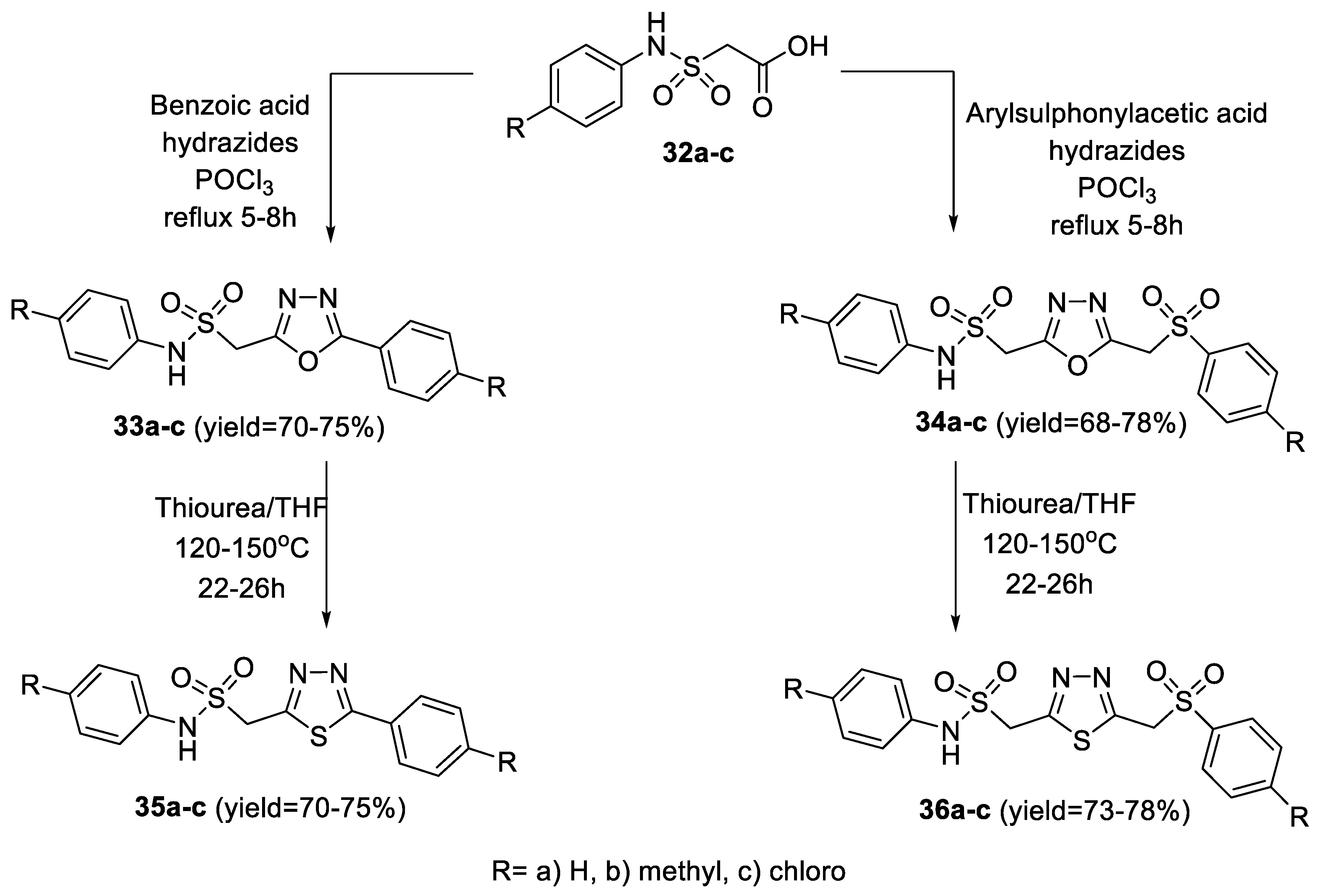
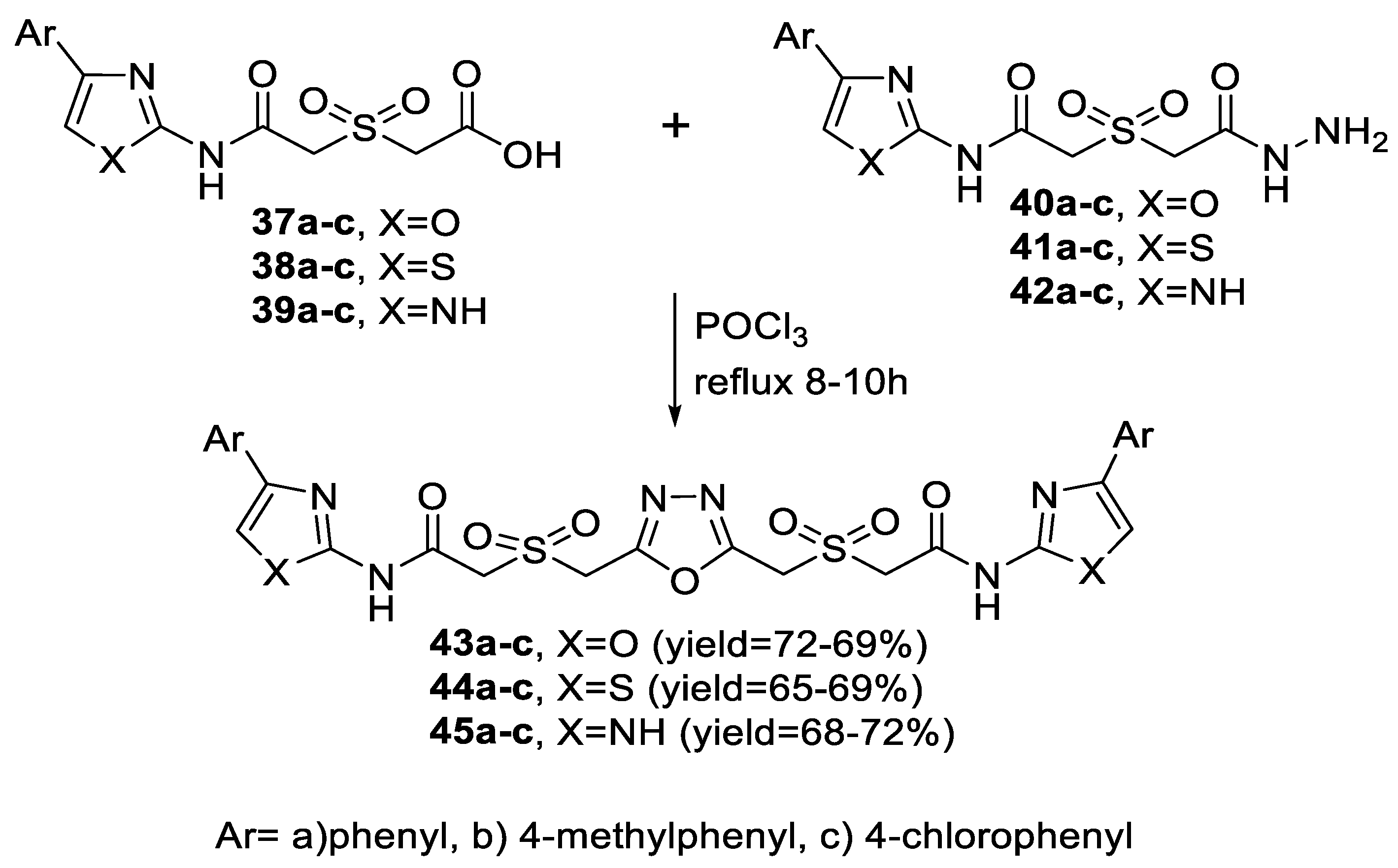
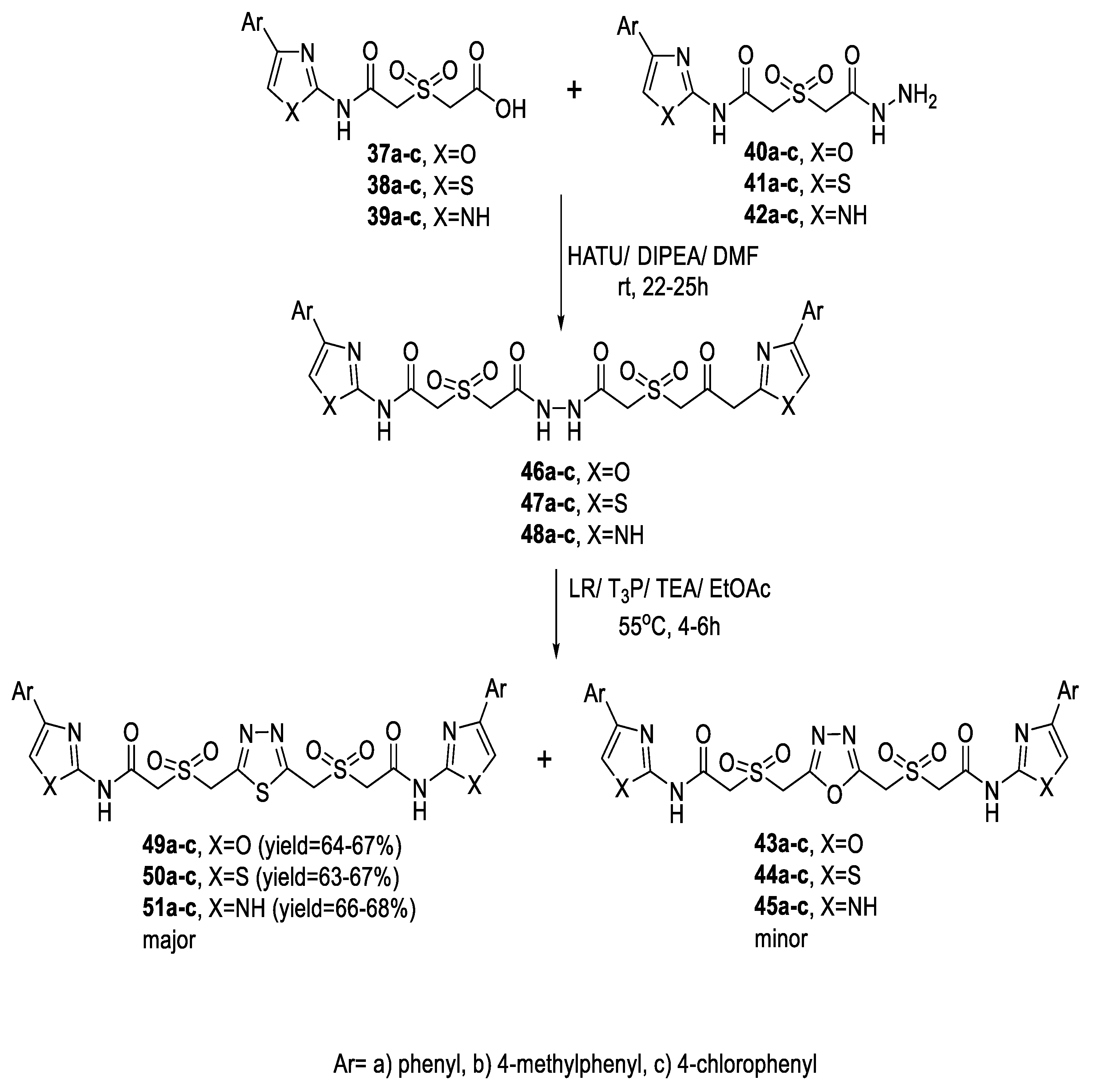
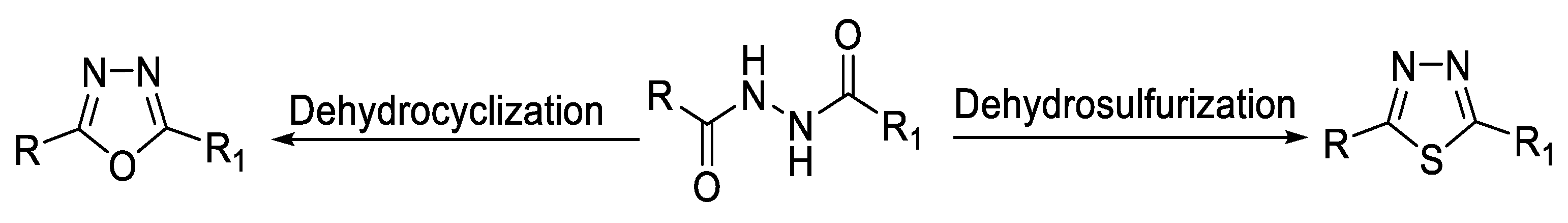
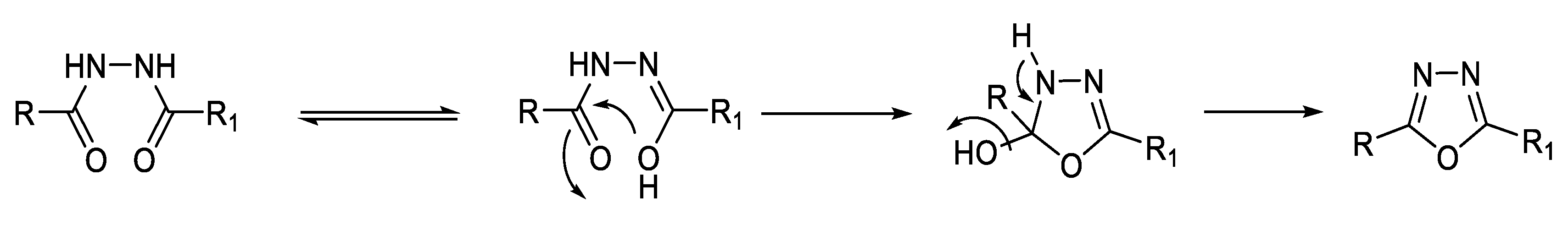
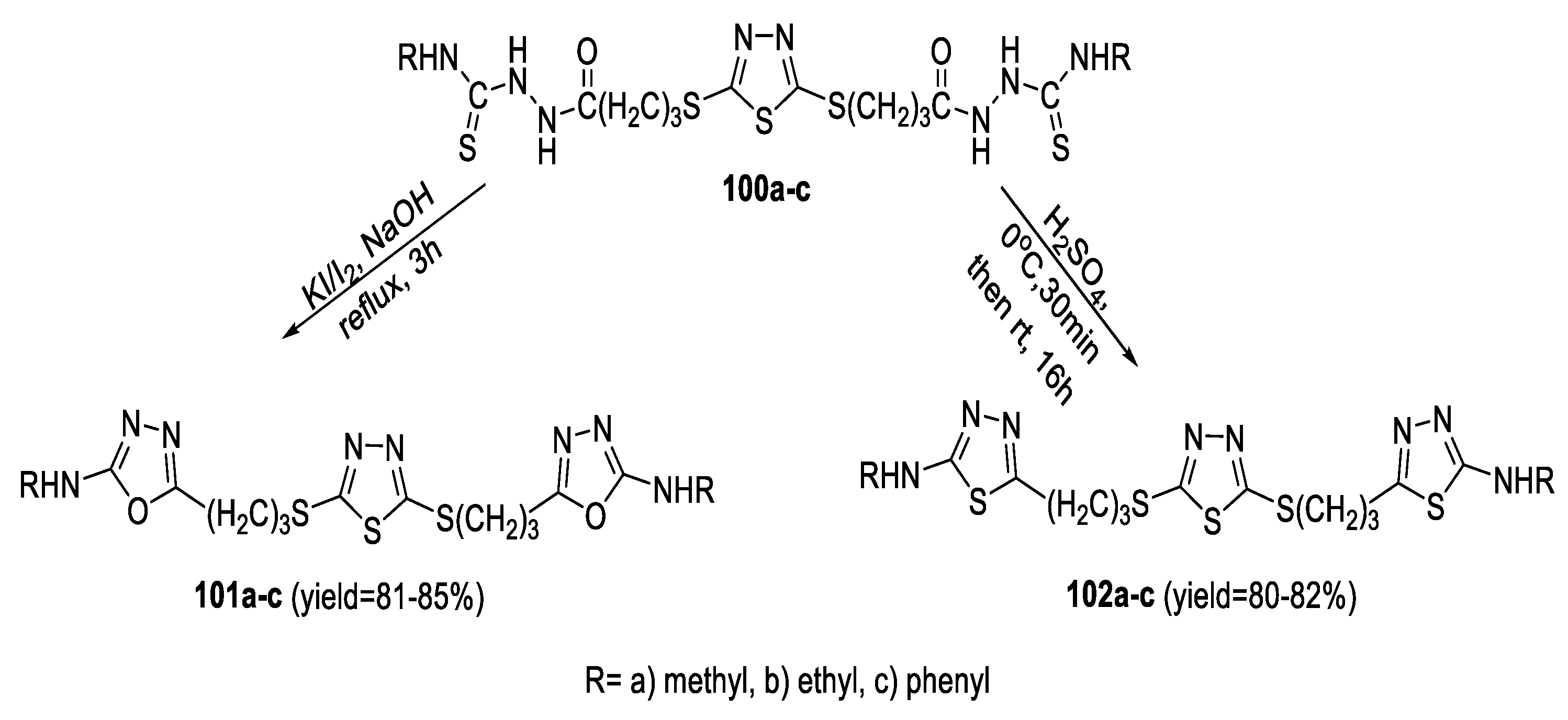
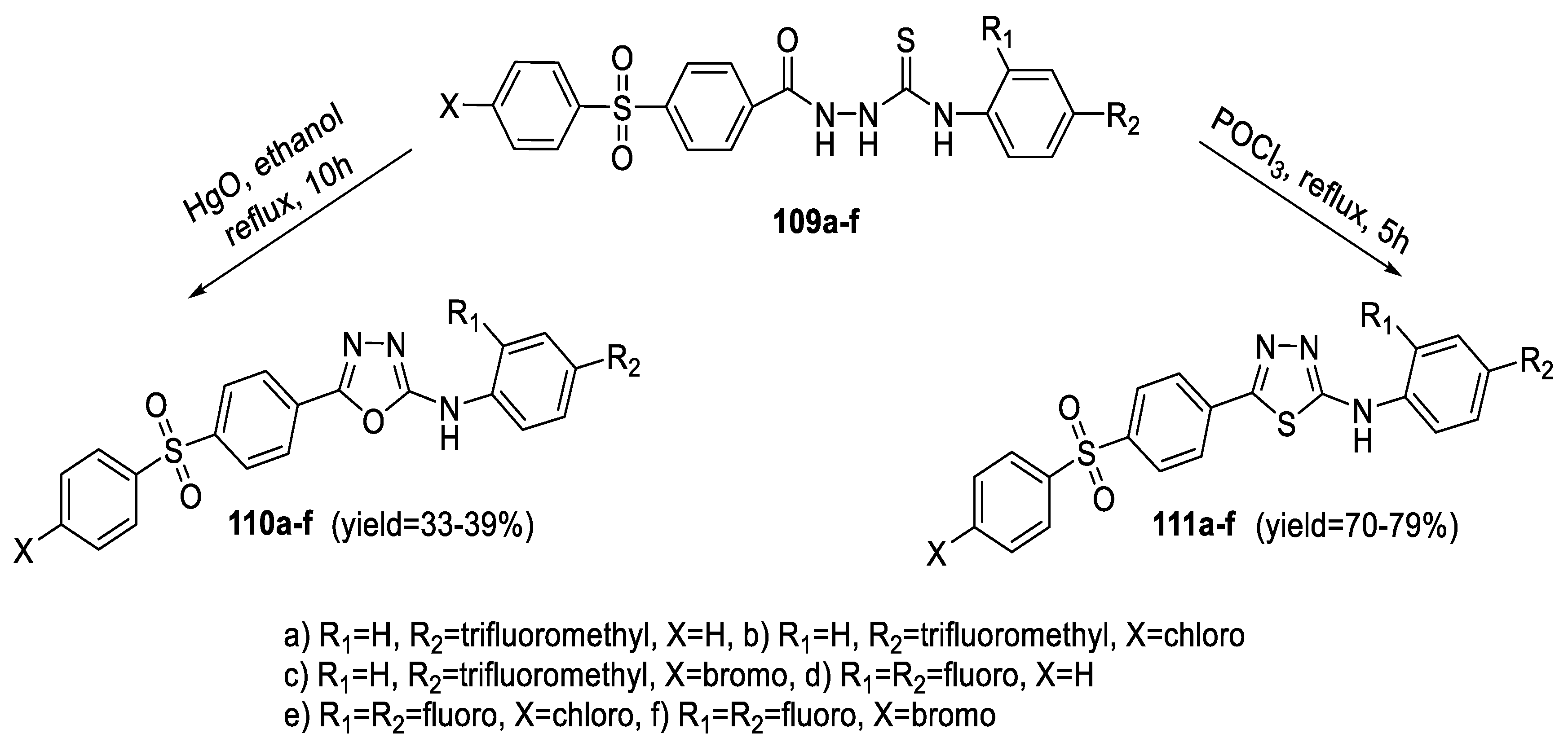
2.1. Synthesis from Sulfonyl Acetic Acid Hydrazide
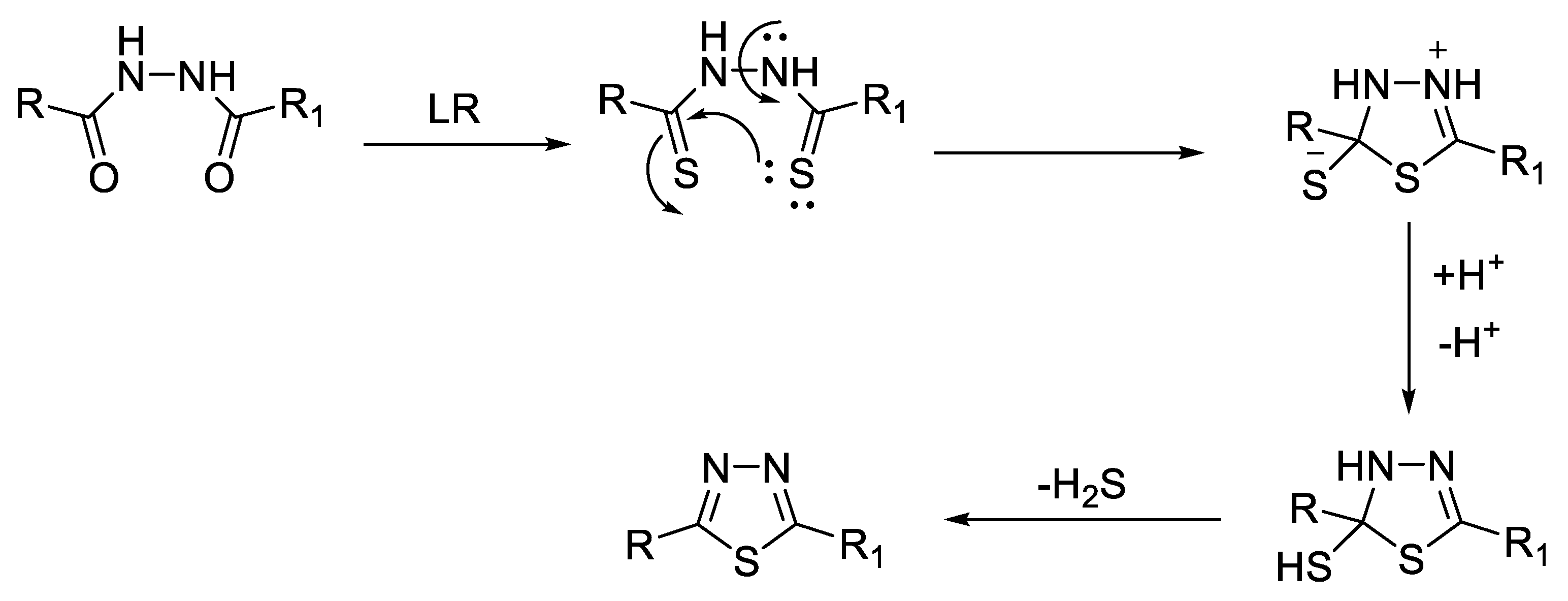
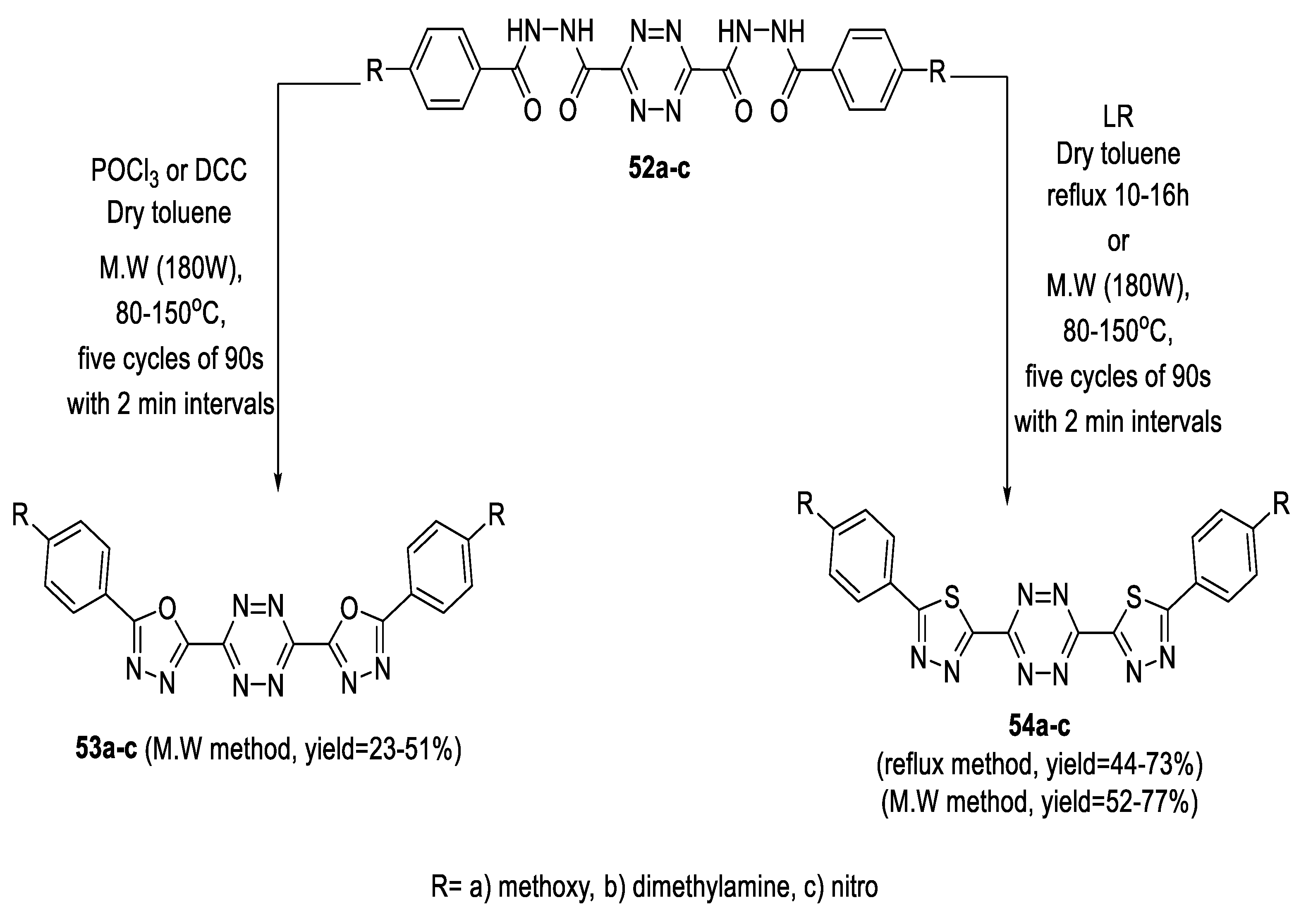
2.2. Synthesis from 1,2-Diacylhydrazines
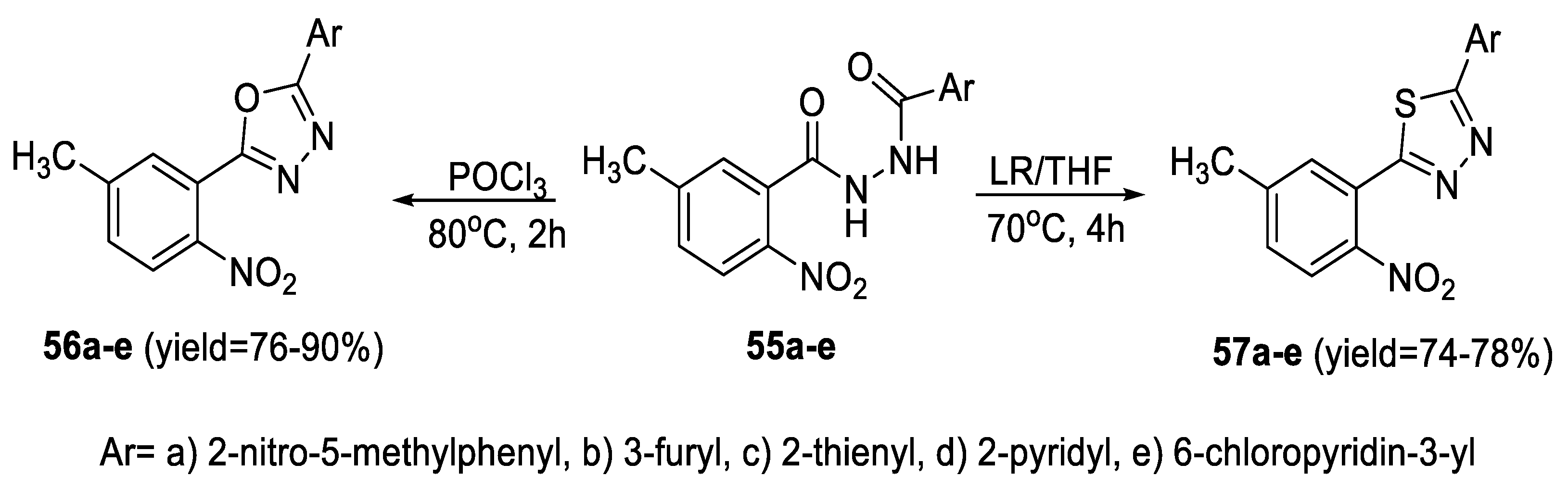
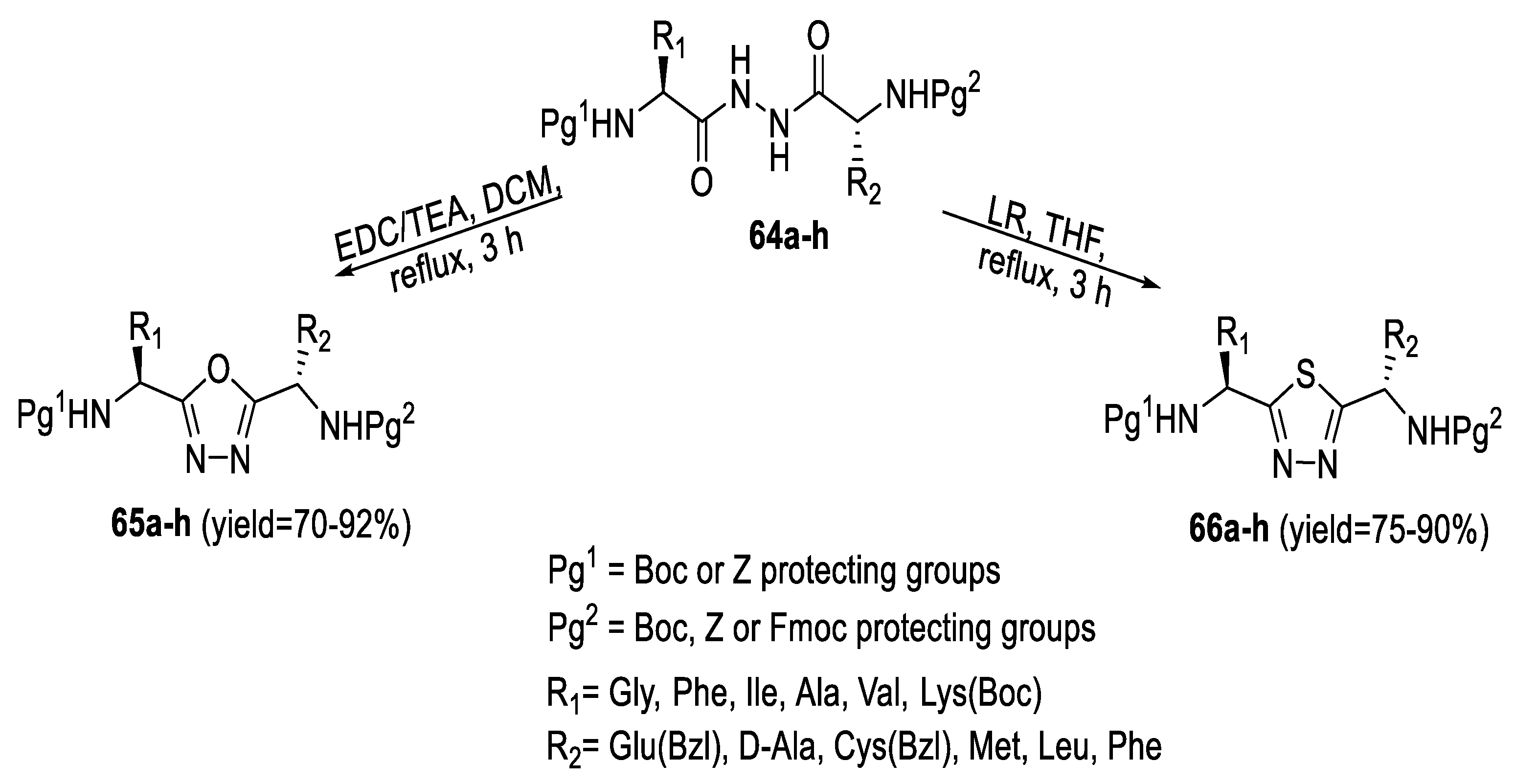
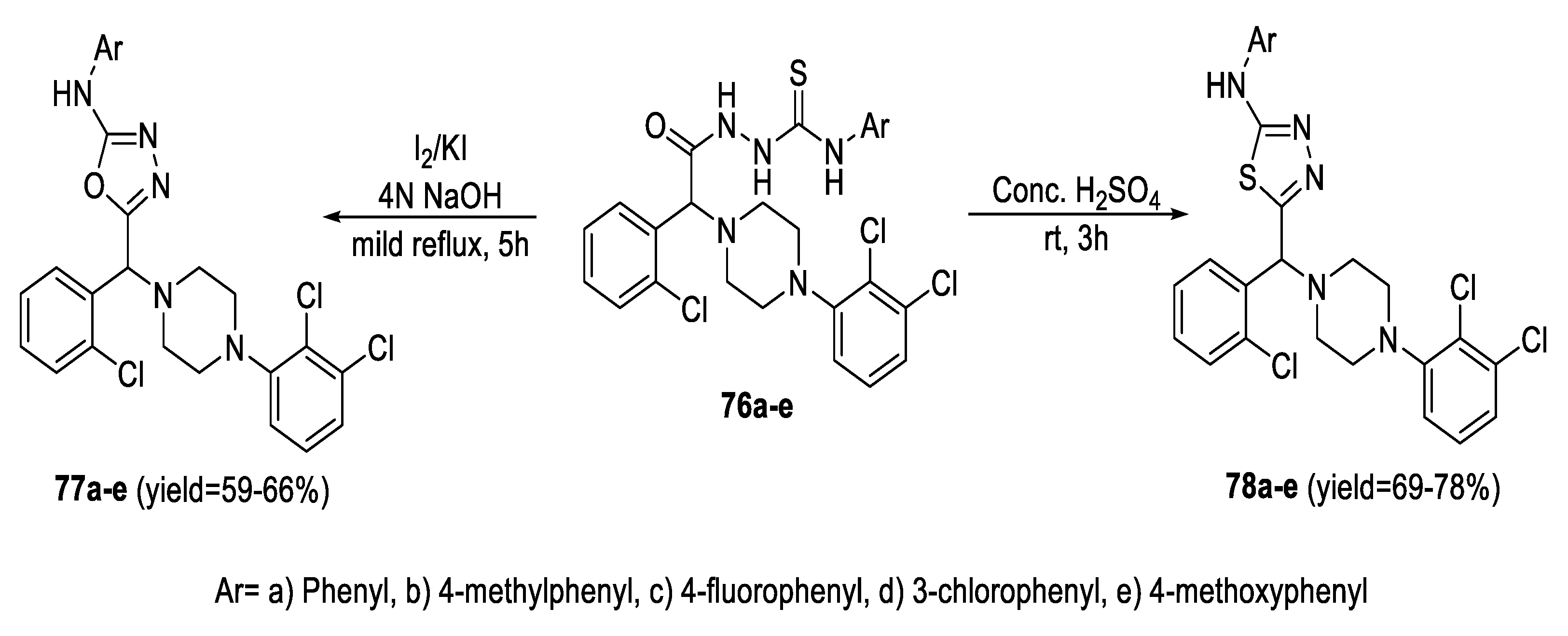
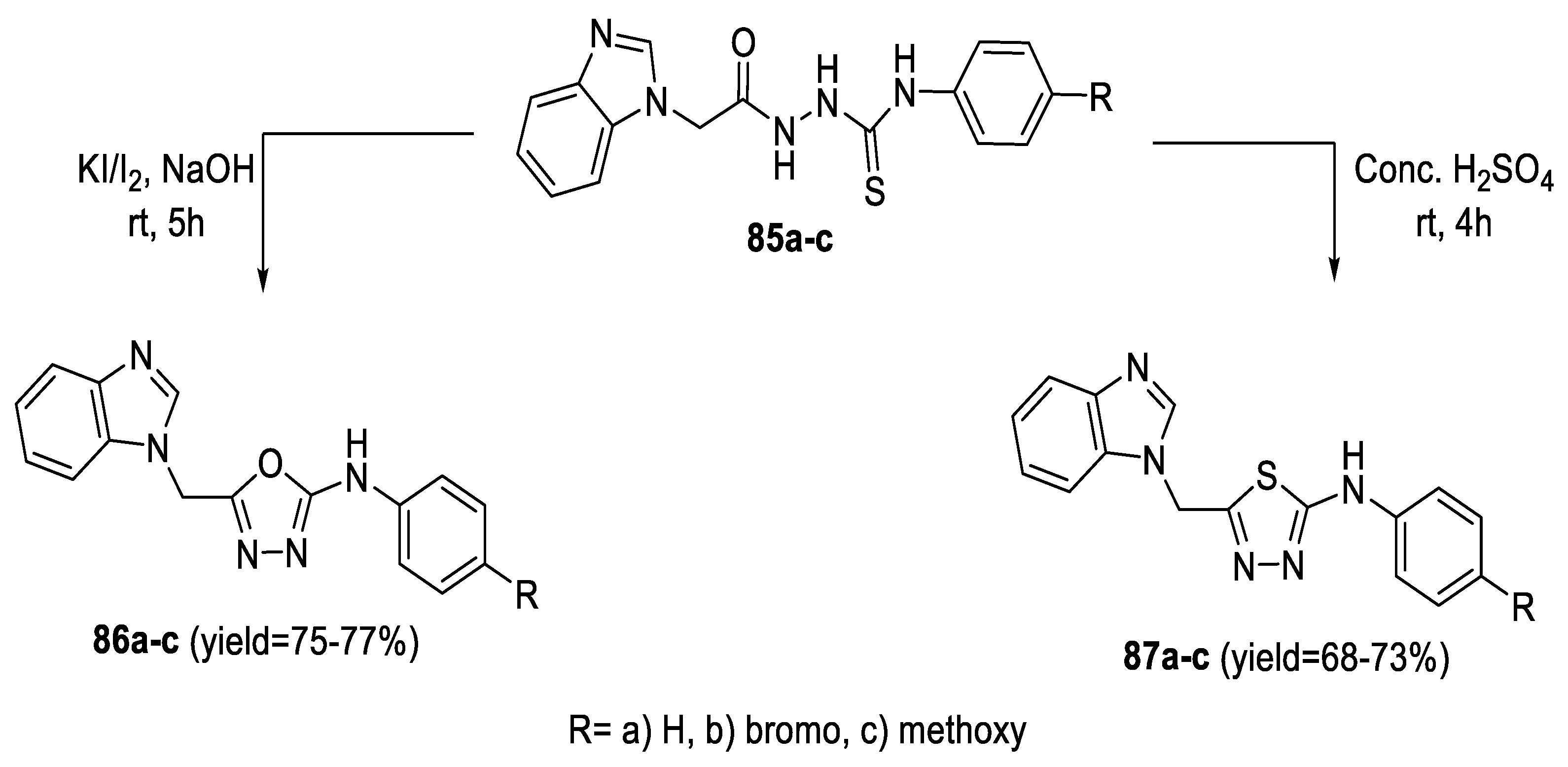
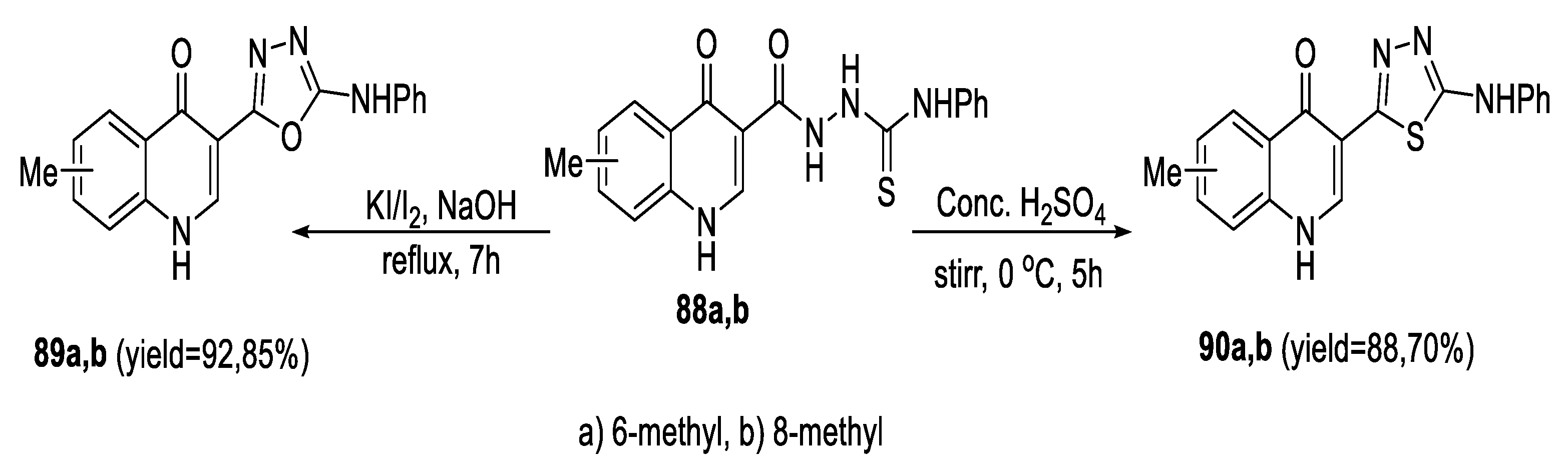
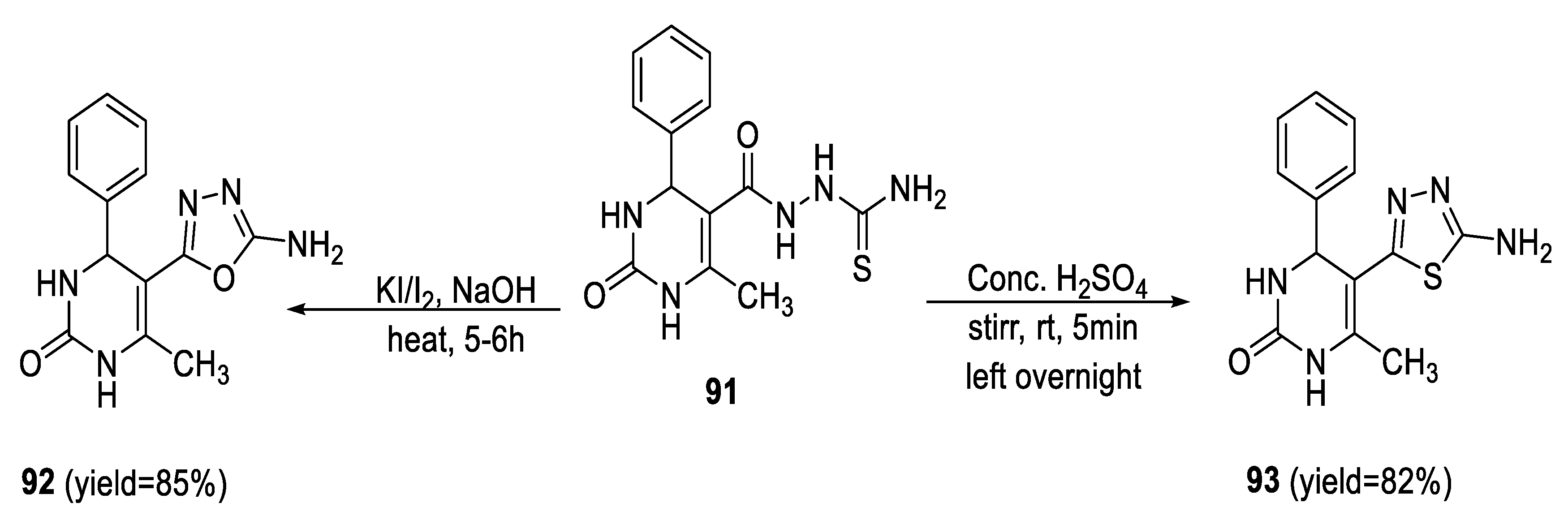
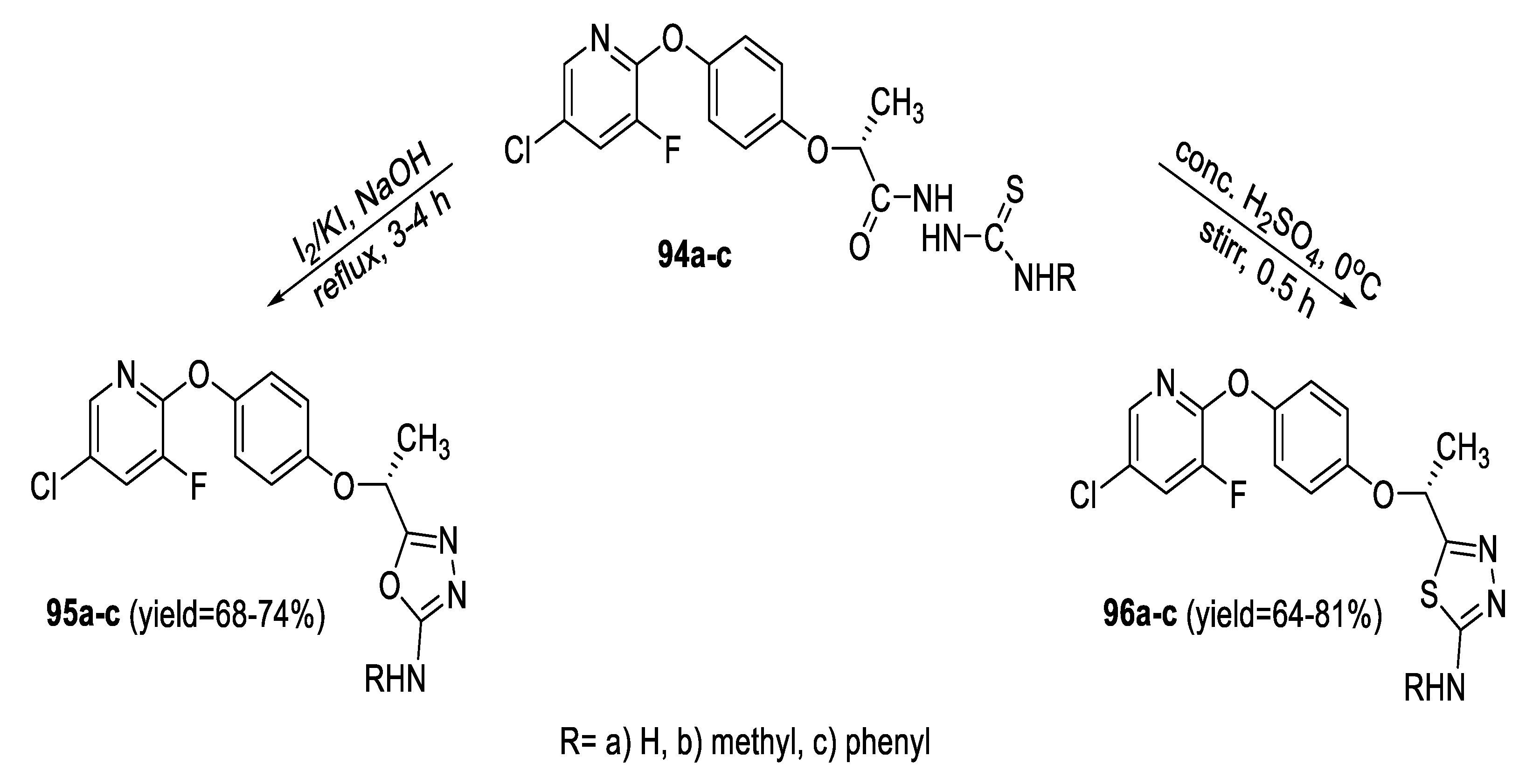
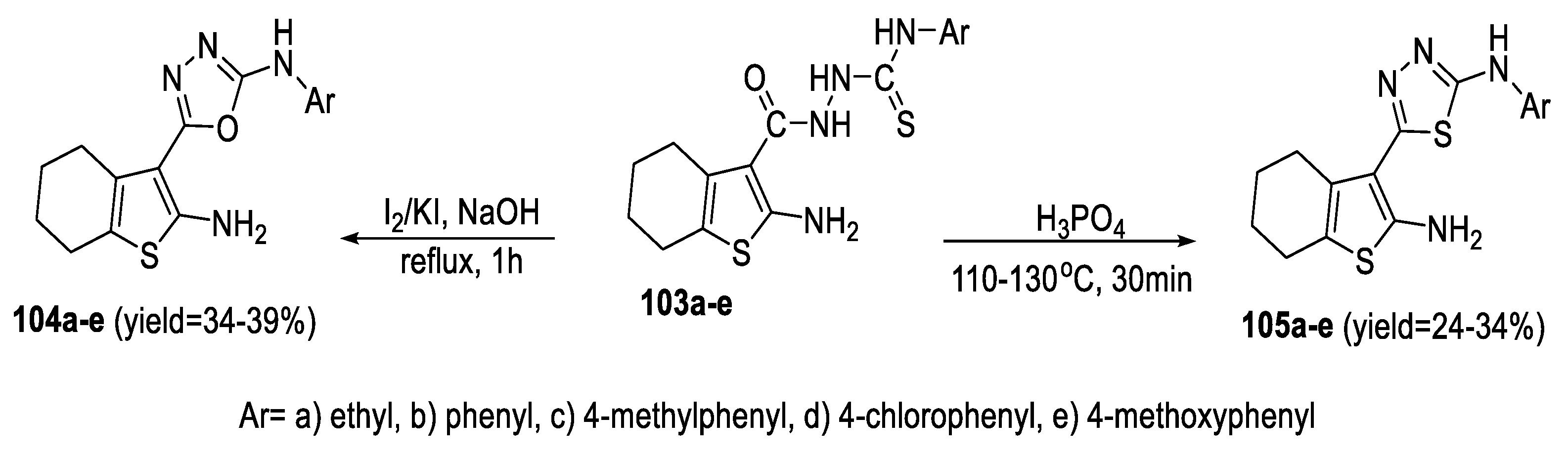
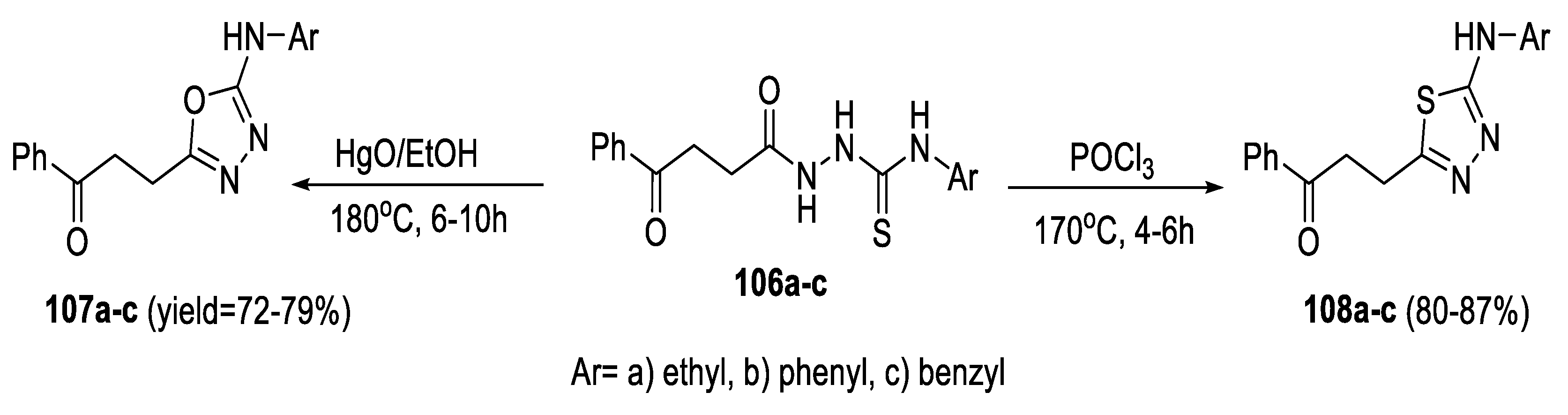
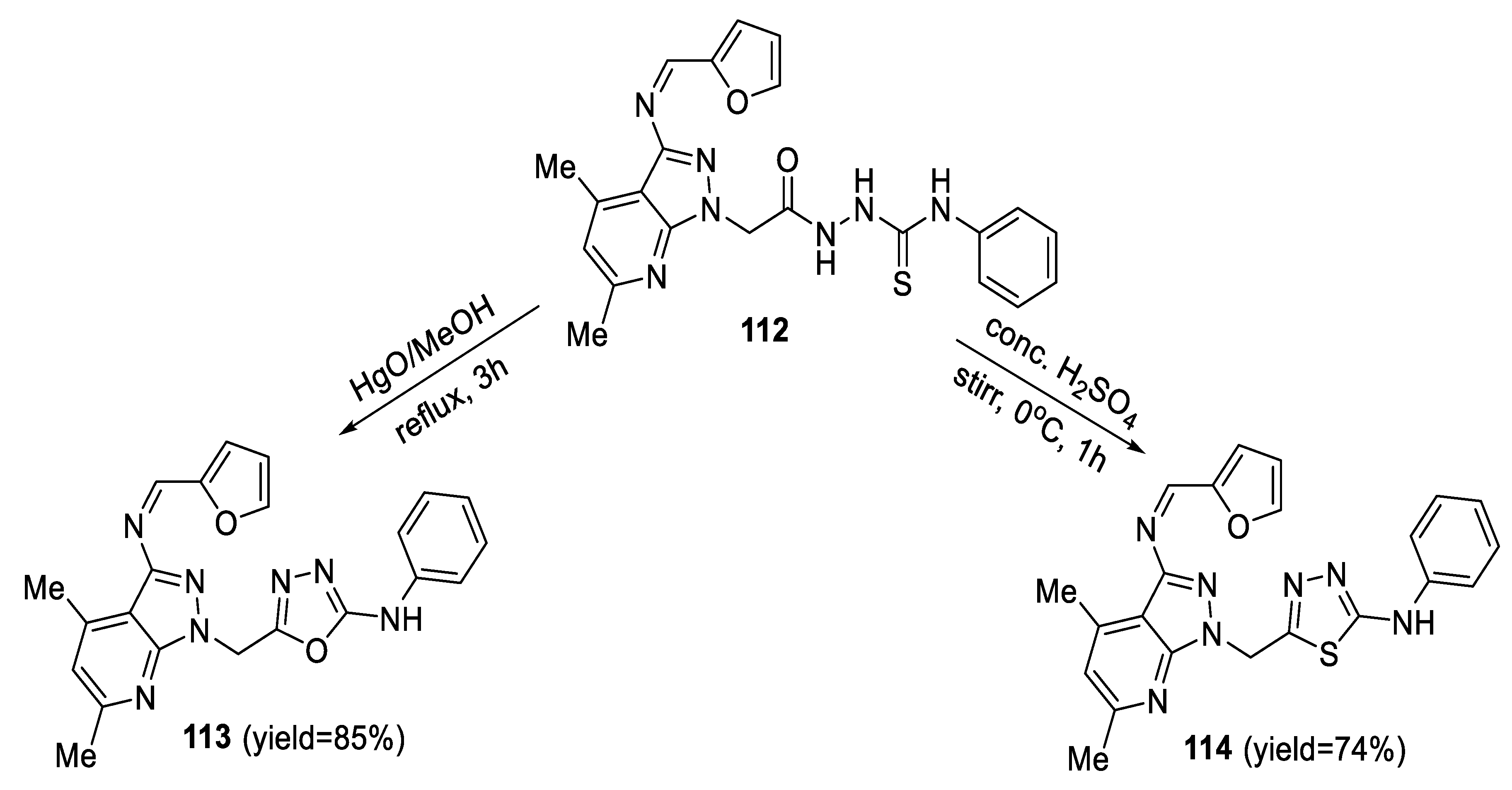
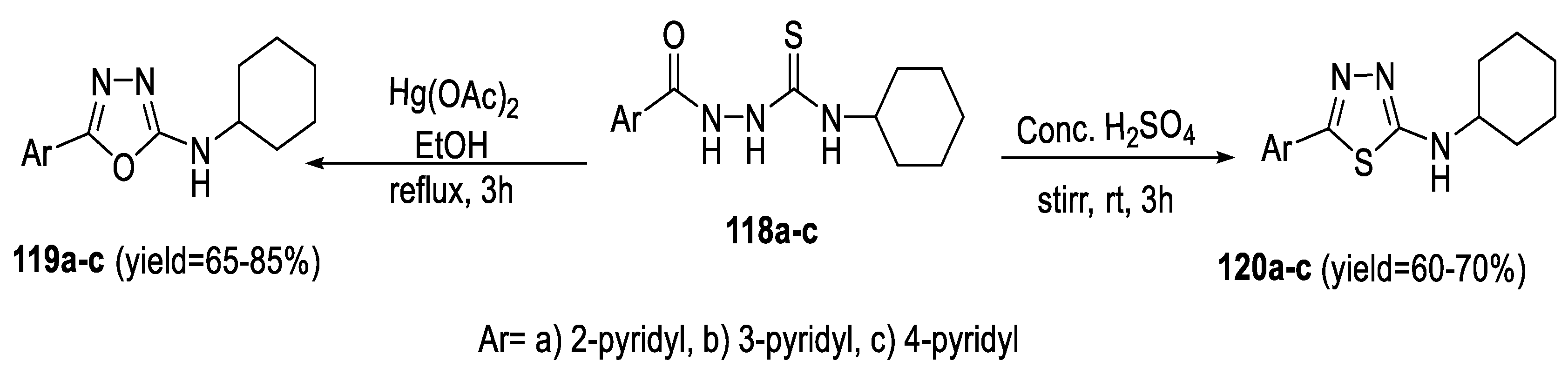
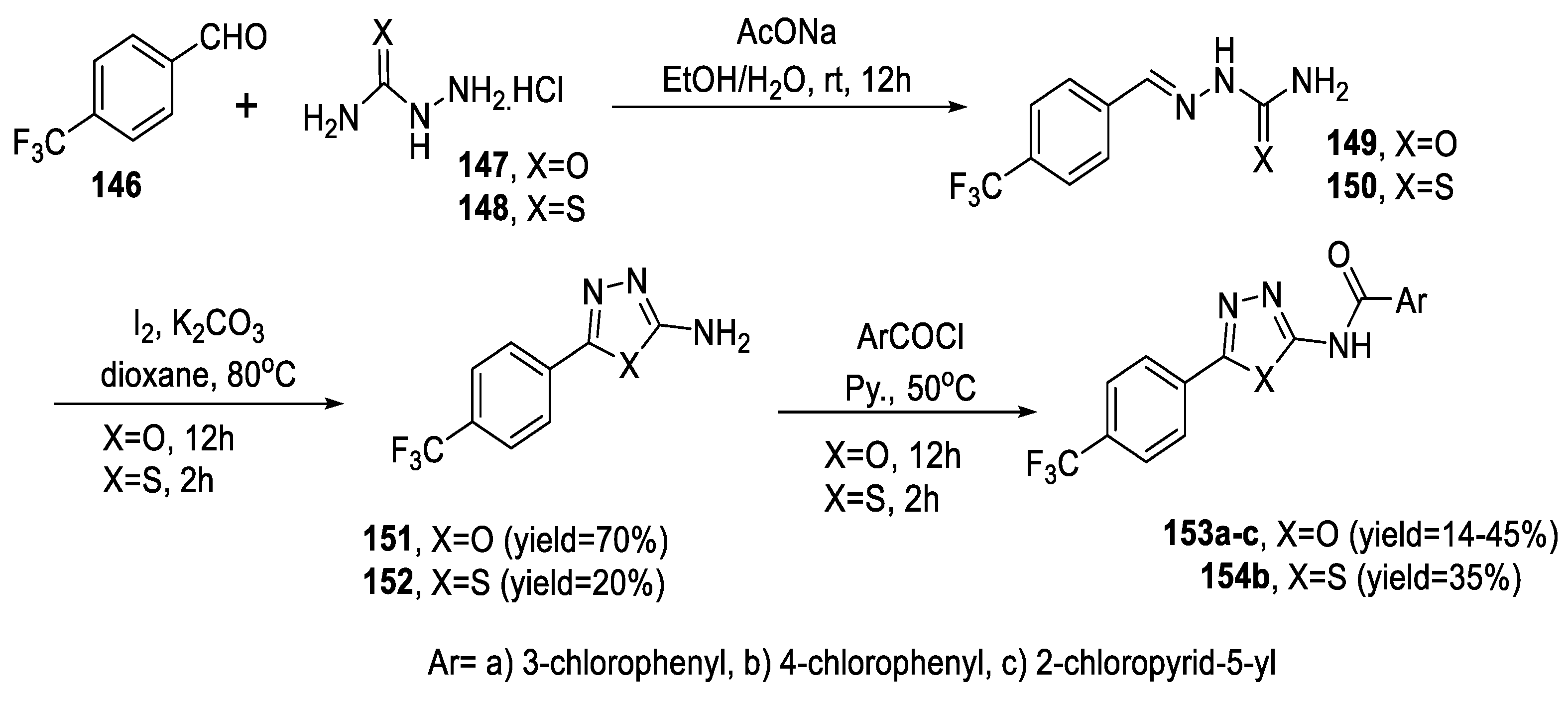
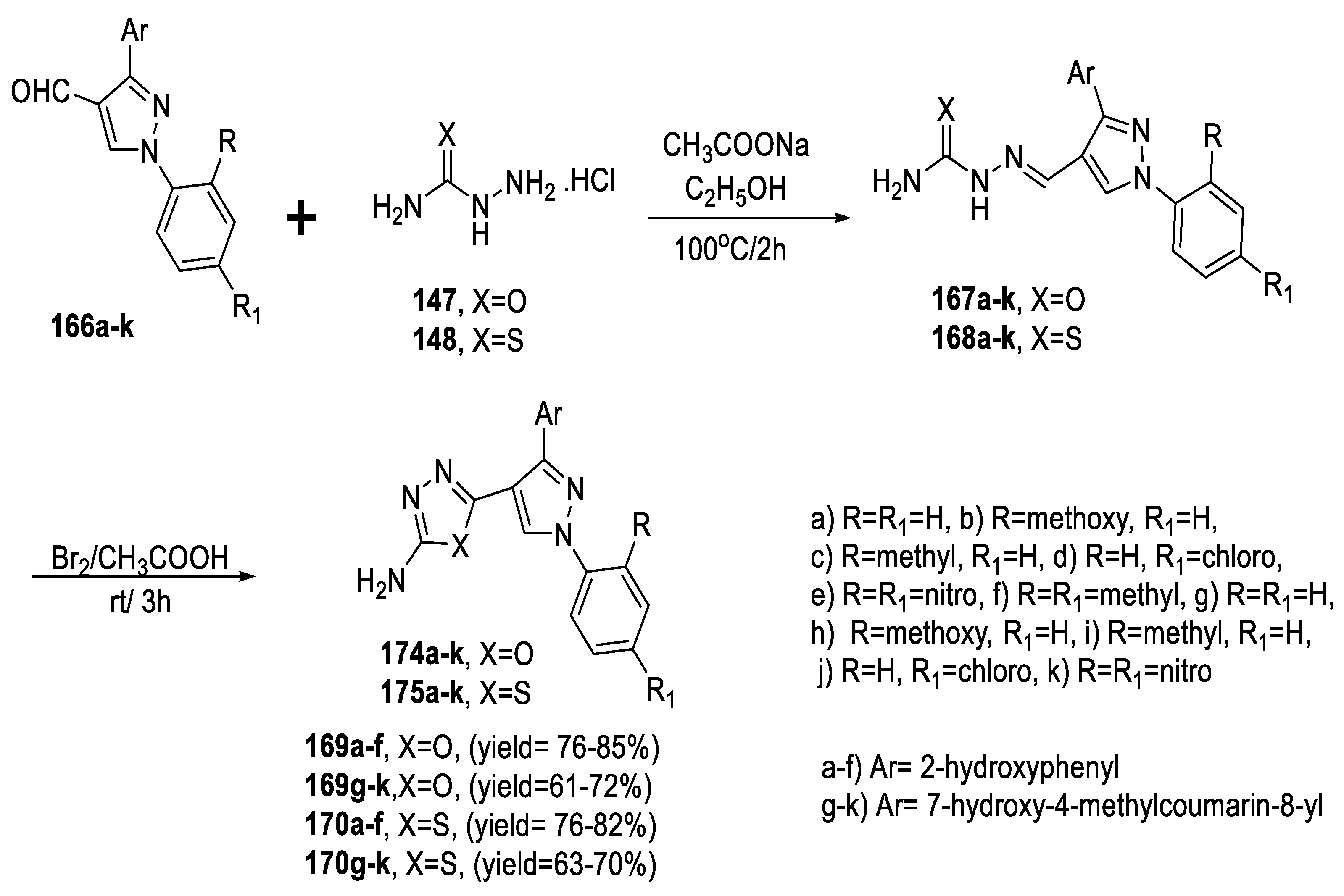
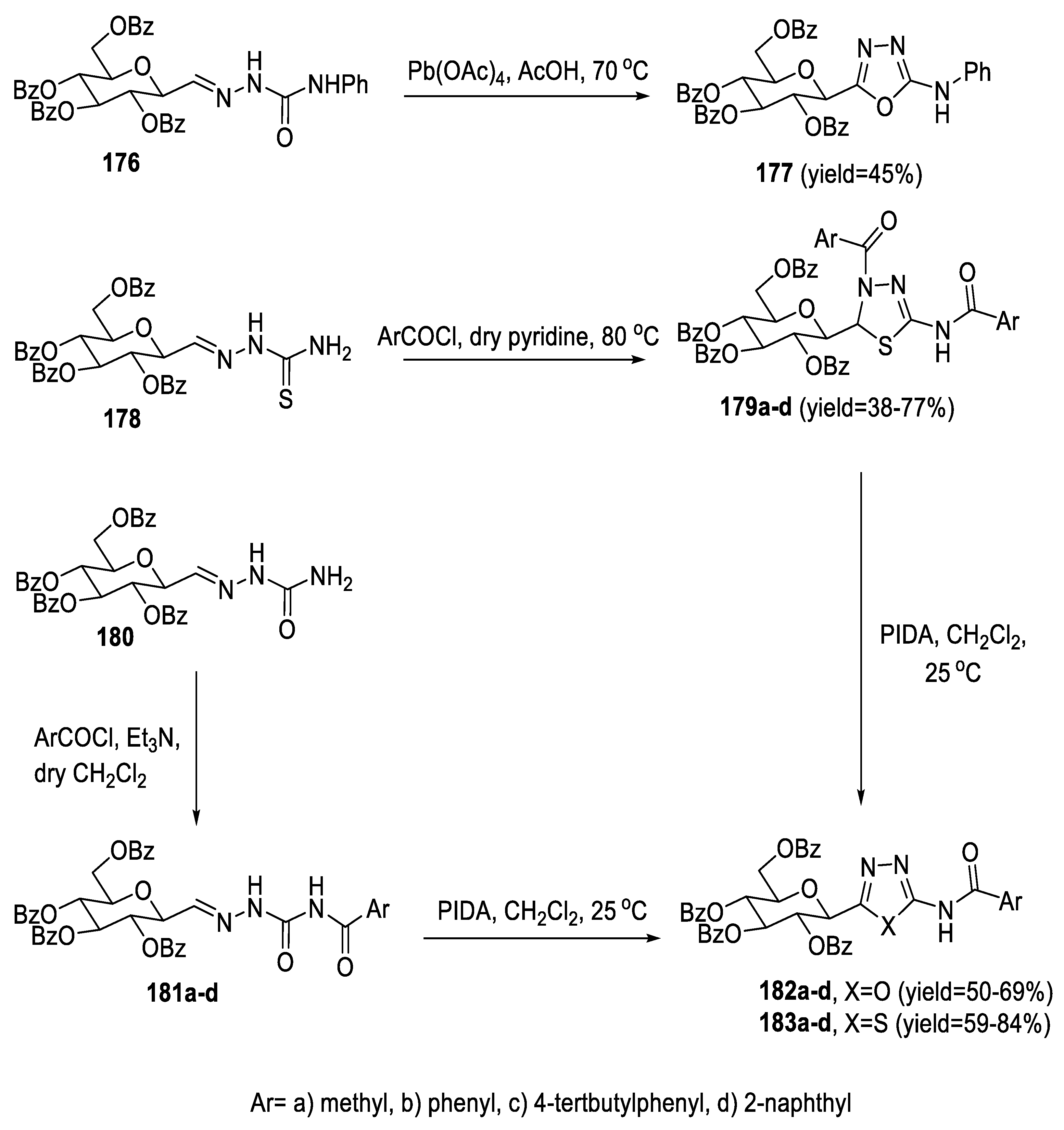
2.3. Synthesis from Acyl Semi/Thiosemicarbazides
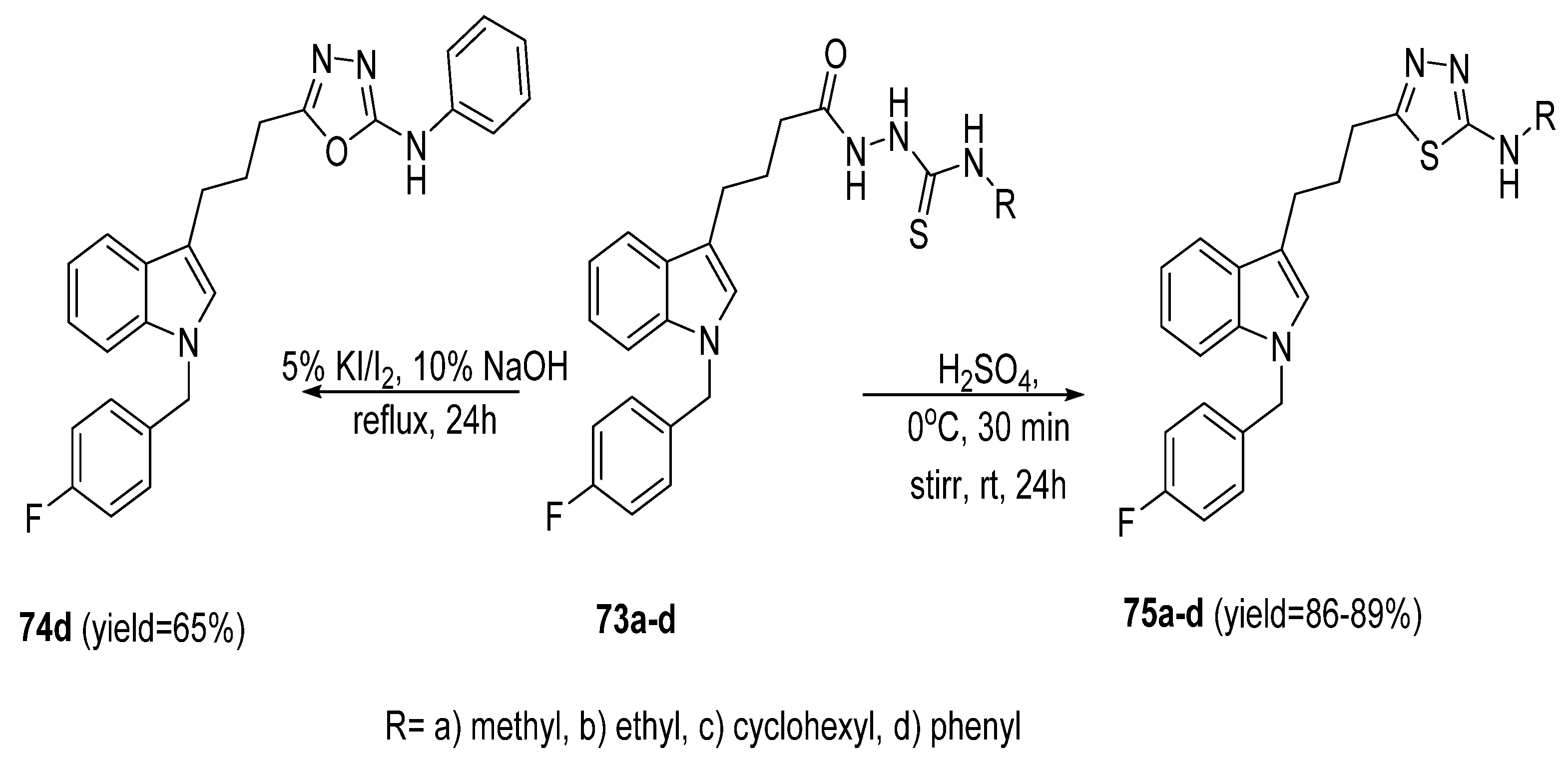
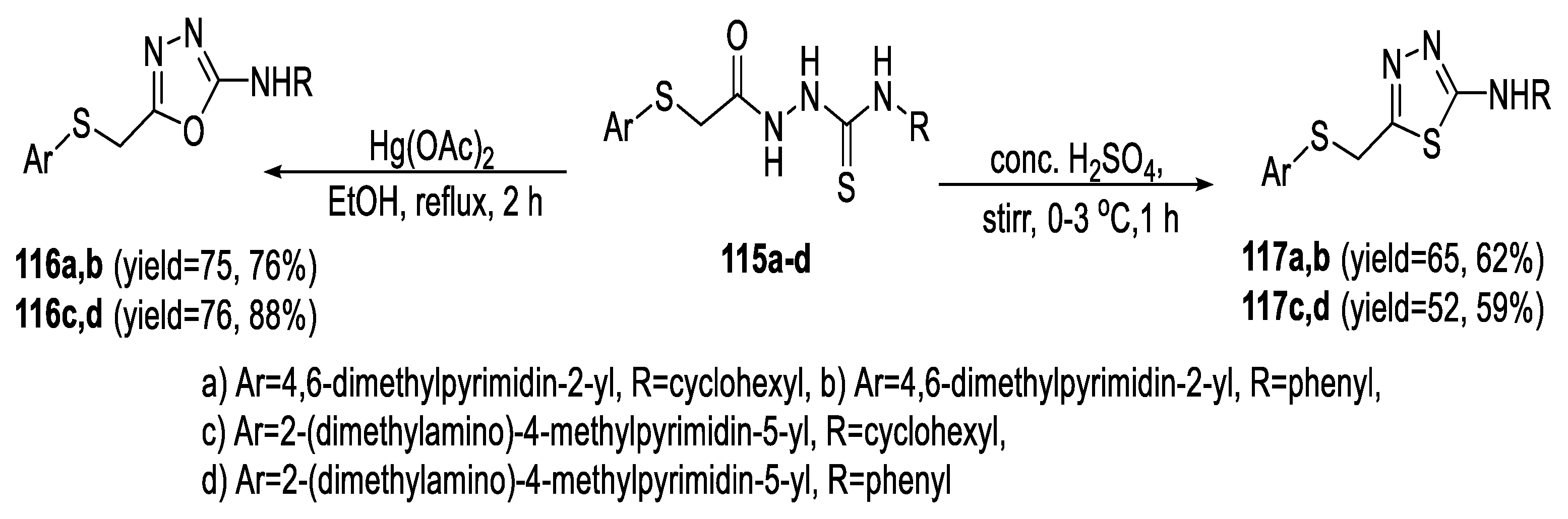
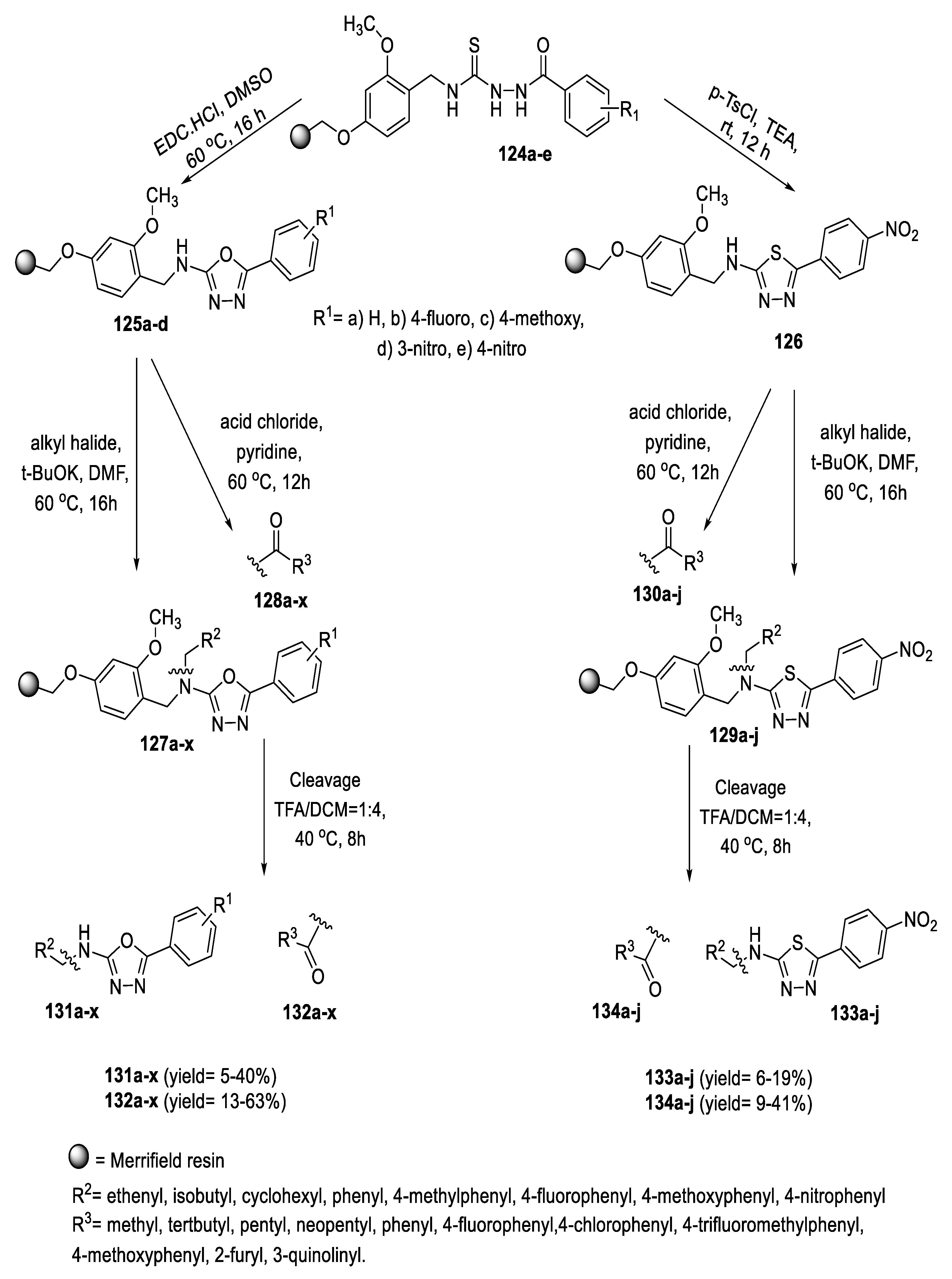
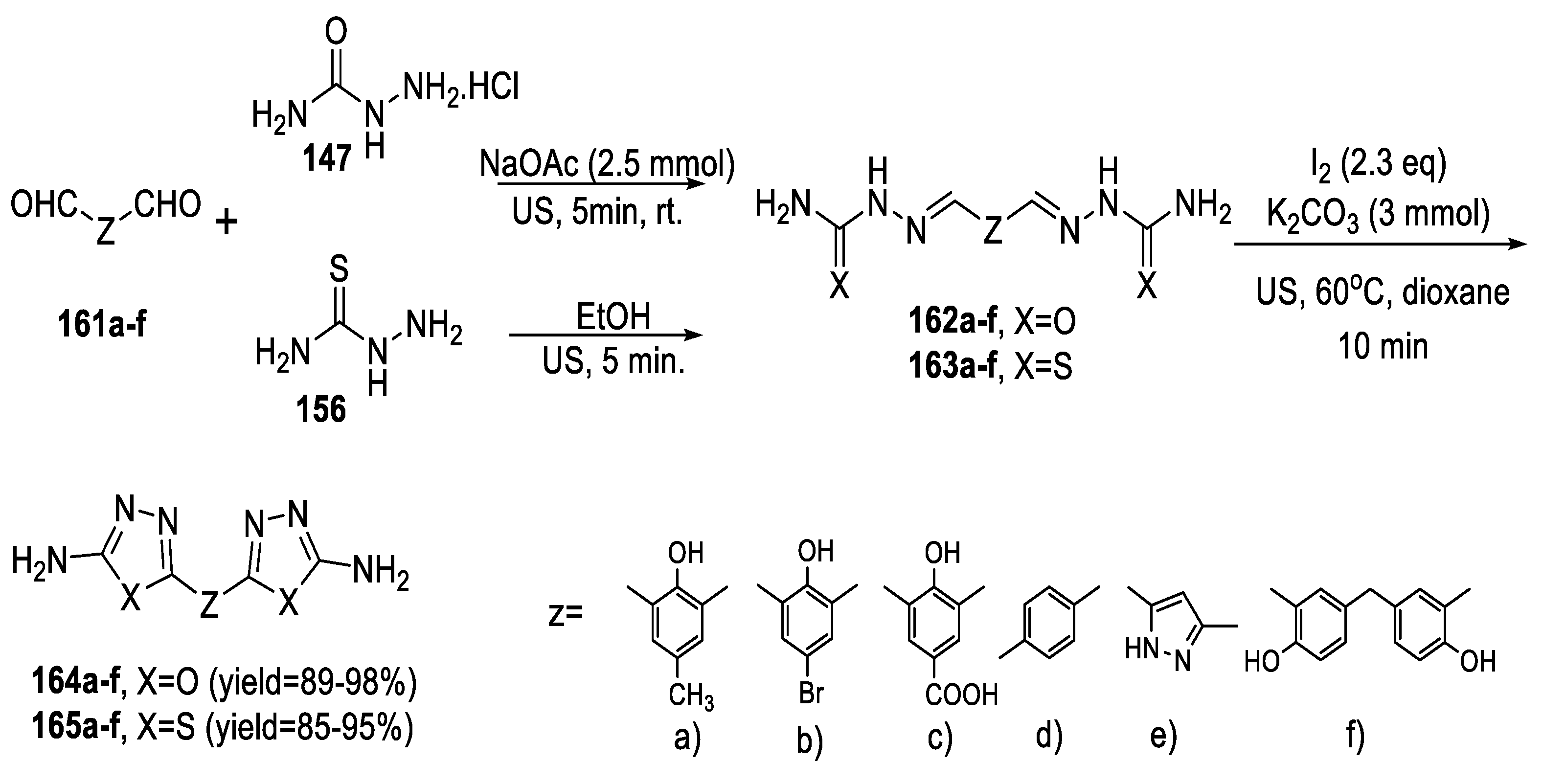
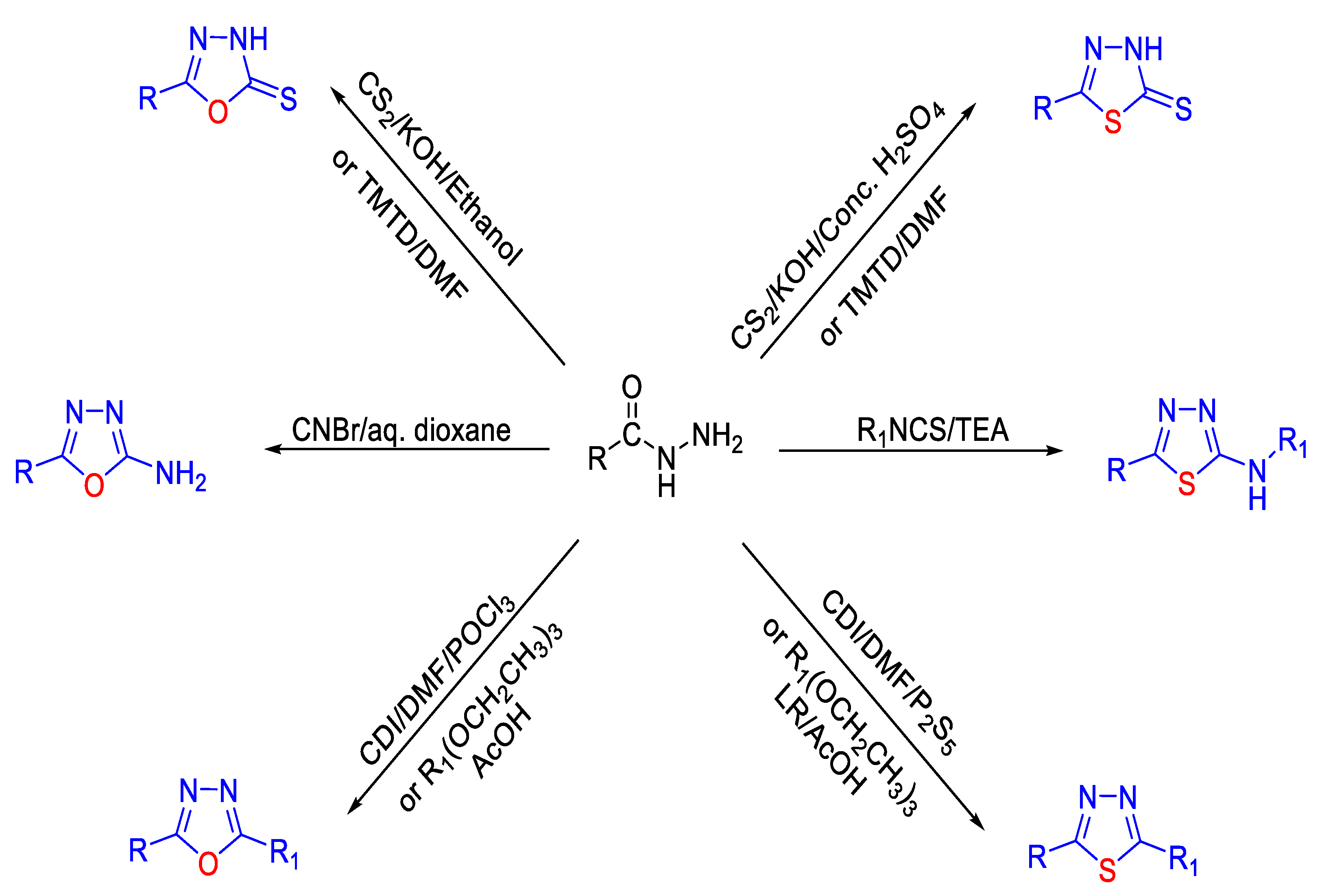
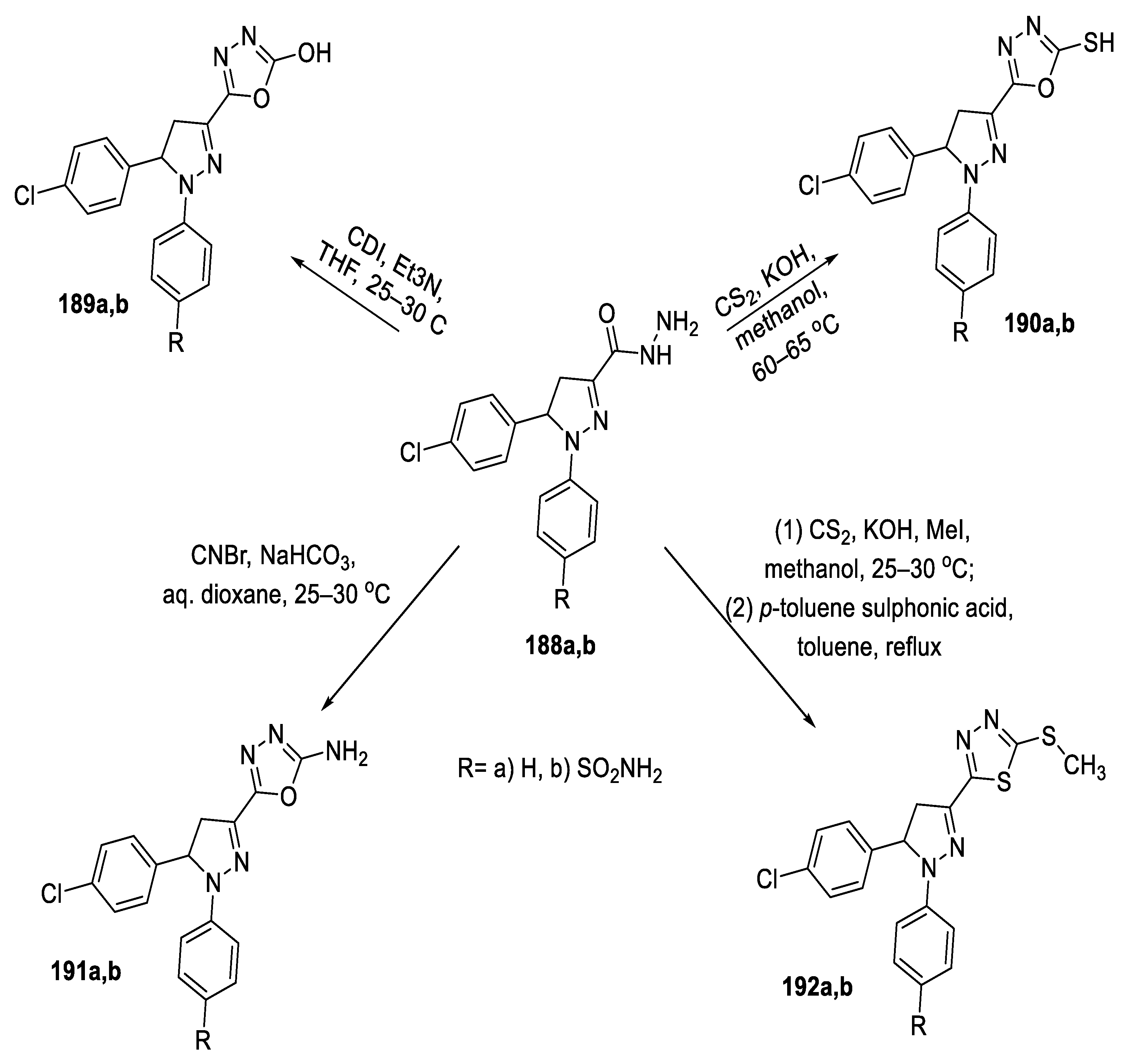
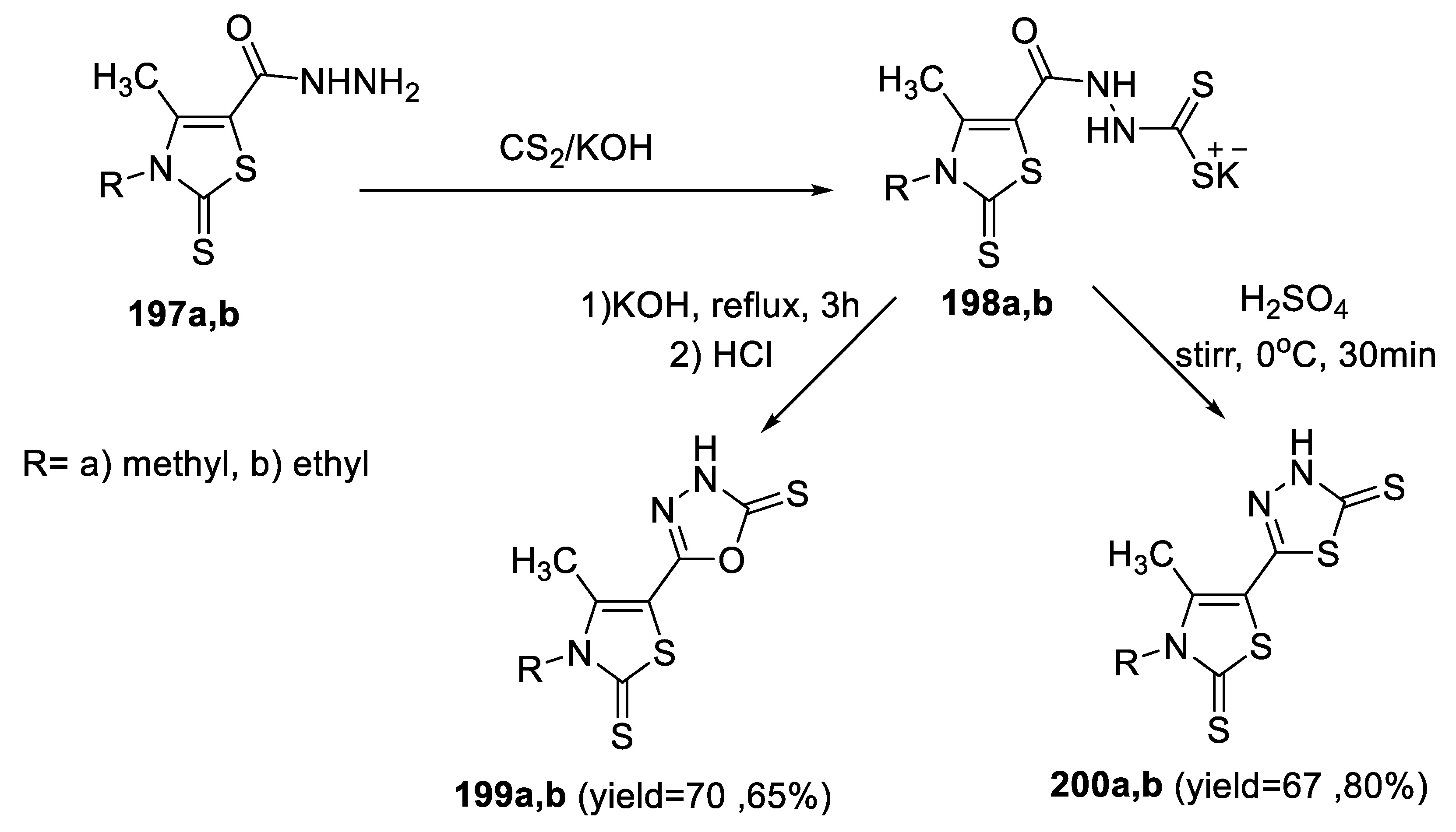
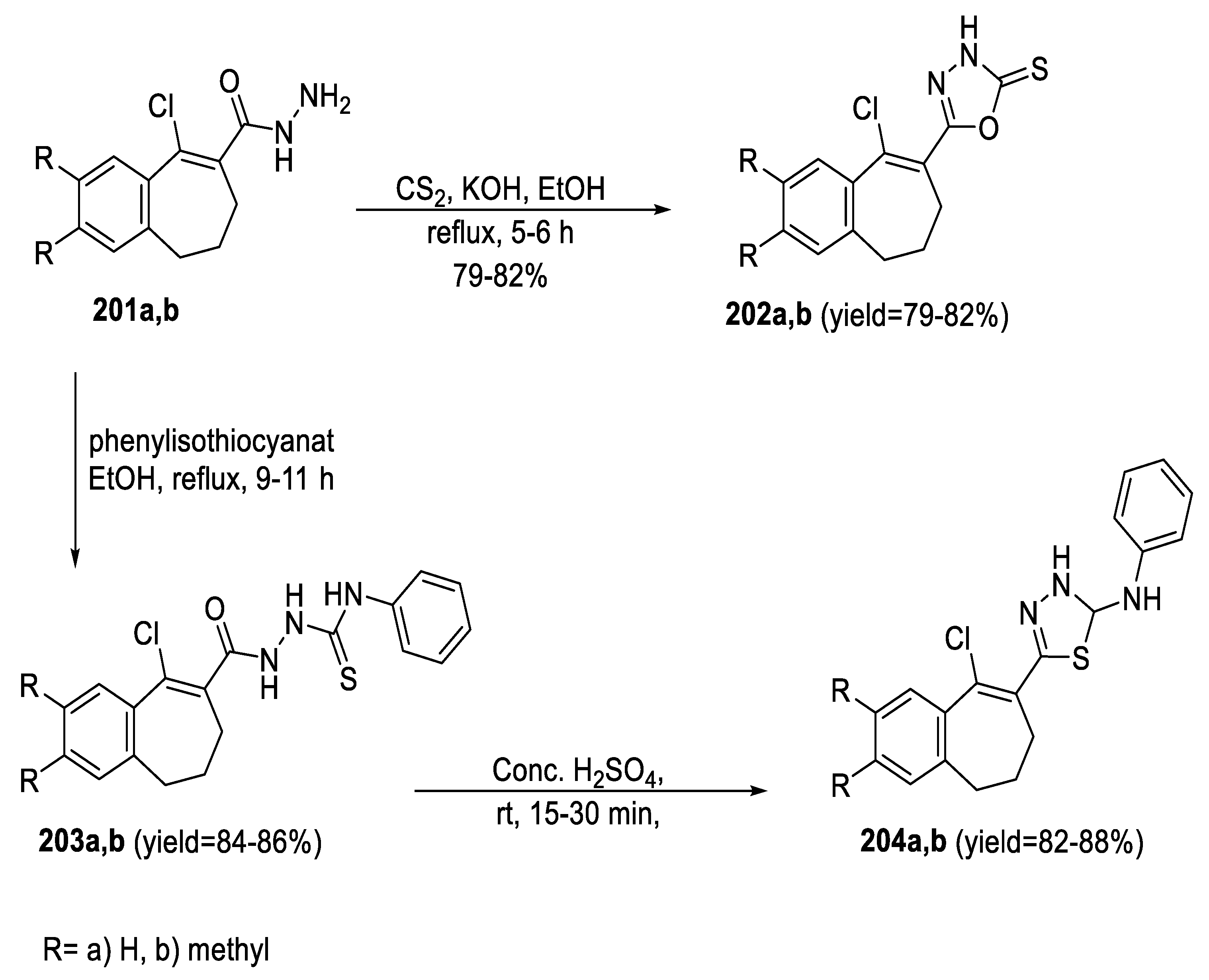
2.4. Synthesis from Semi-/Thiosemicarbazides
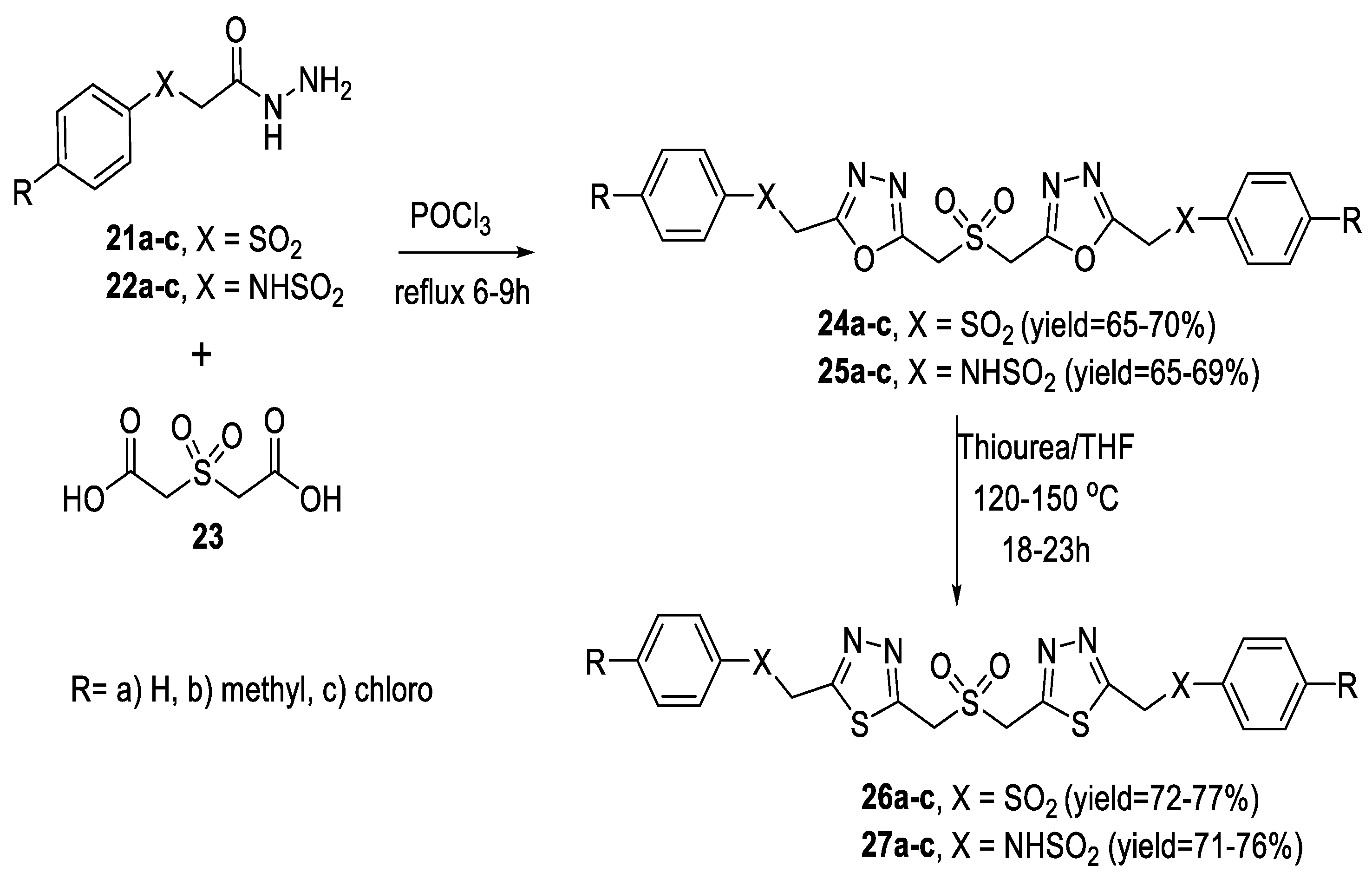
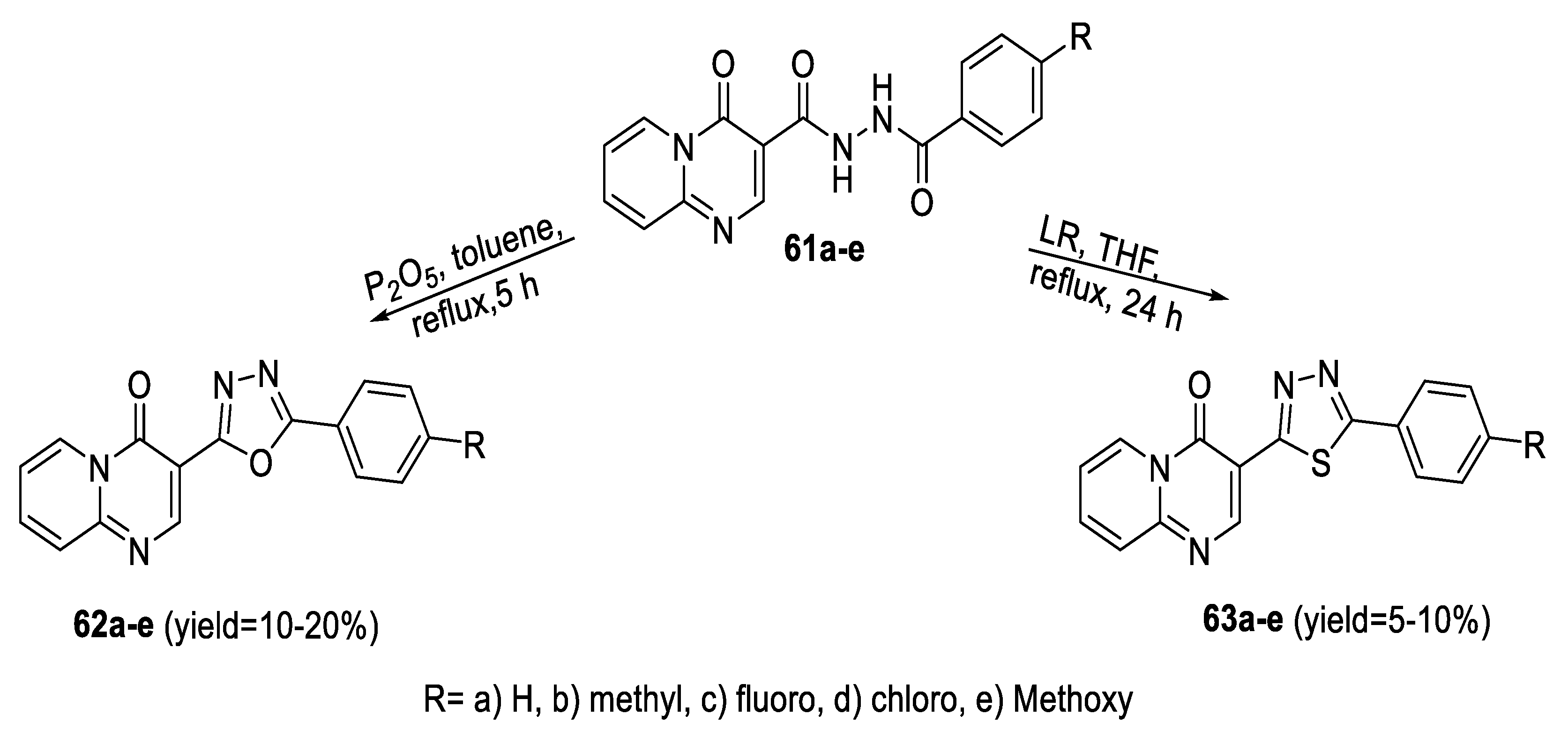
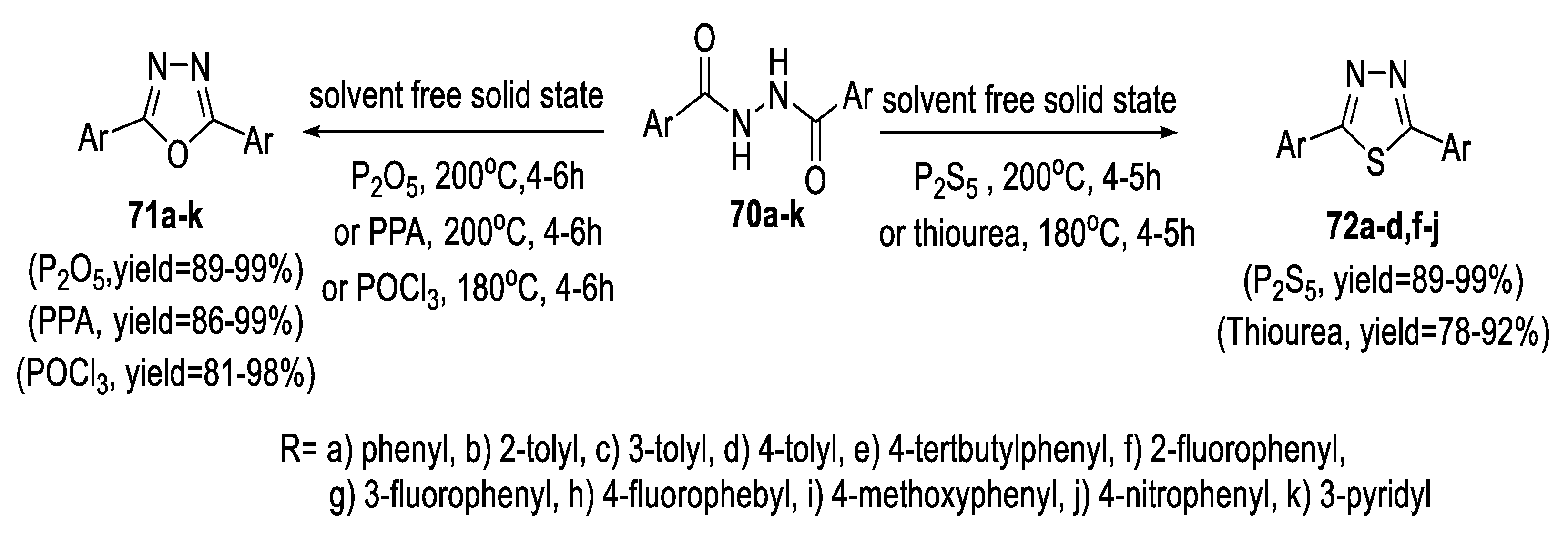
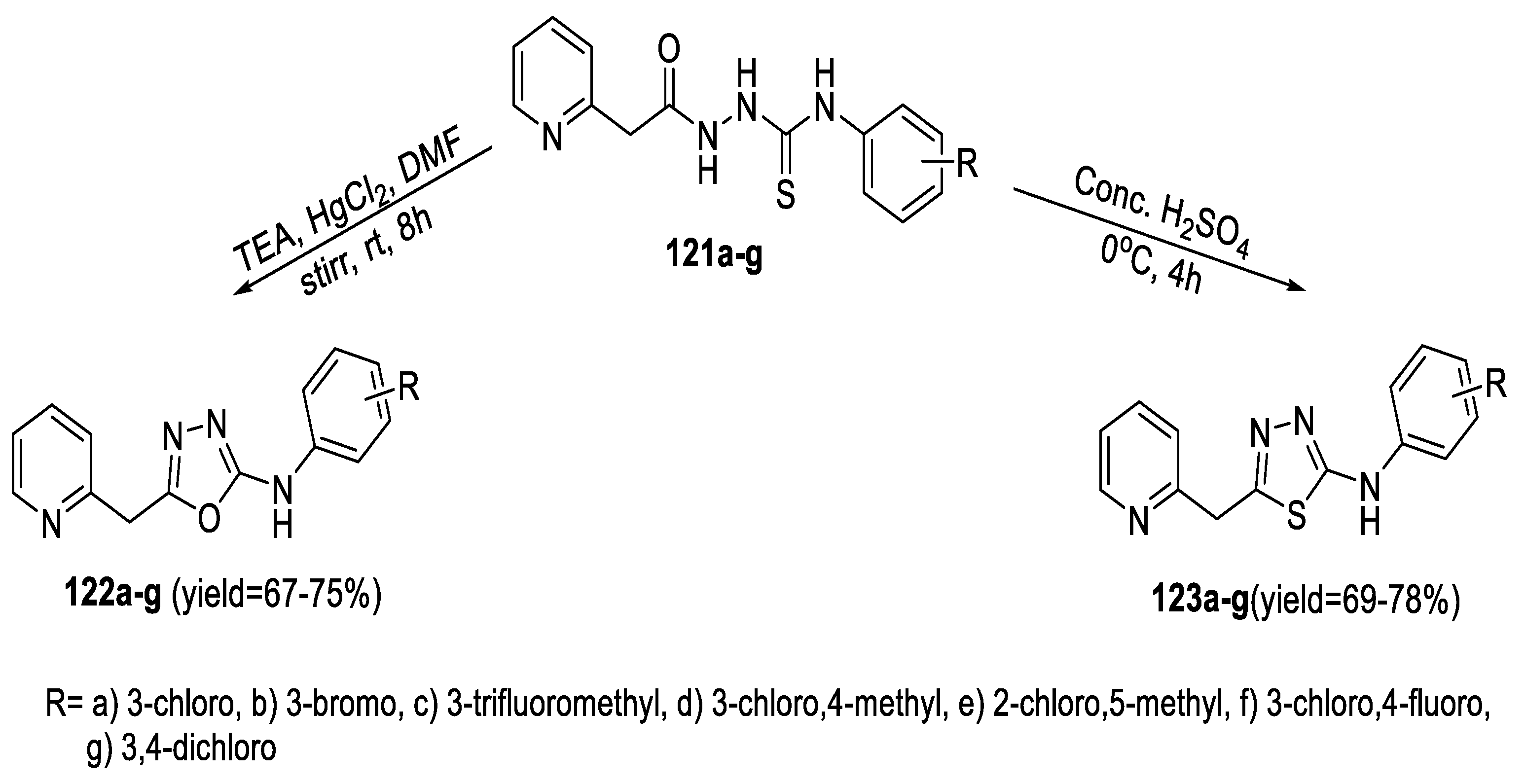
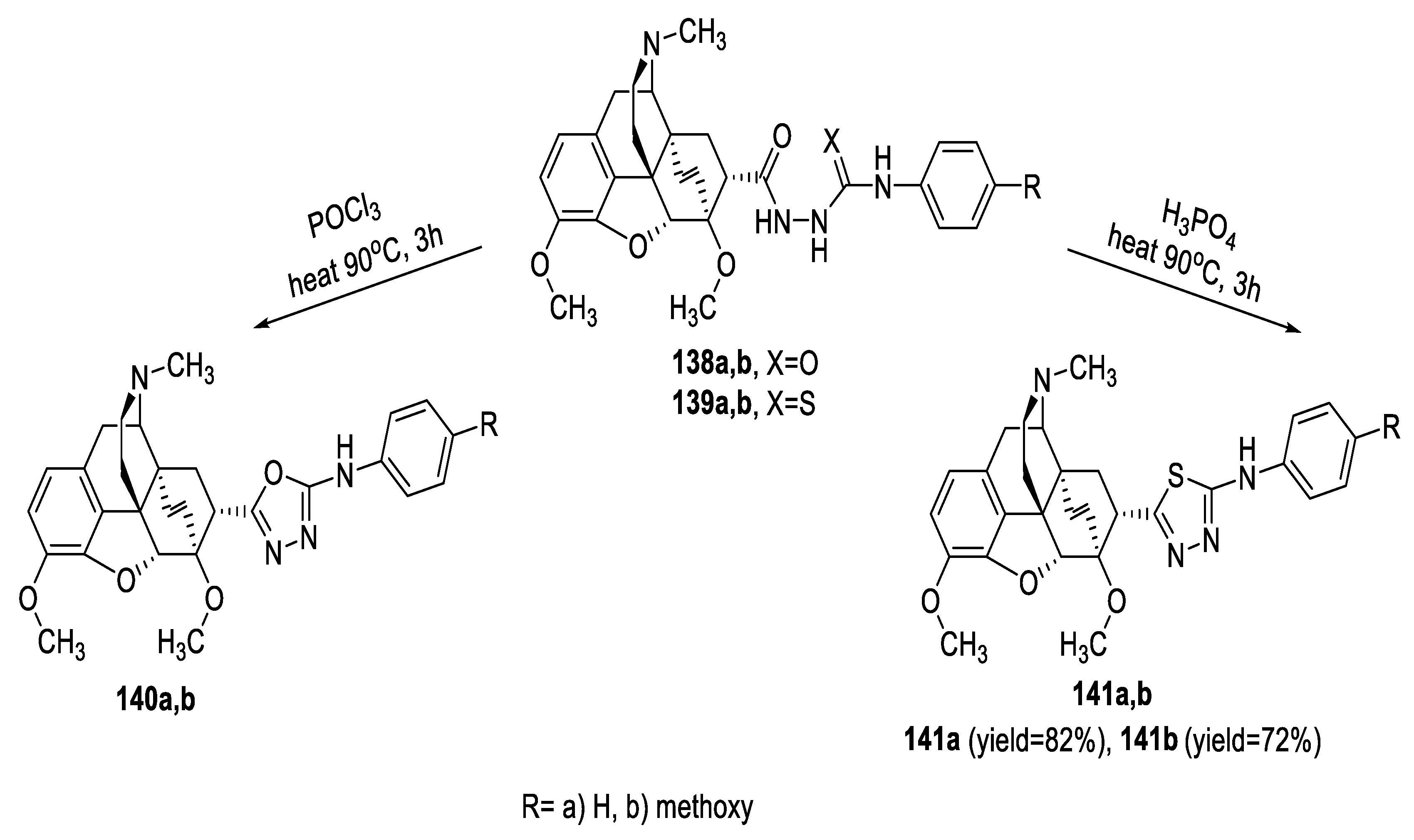
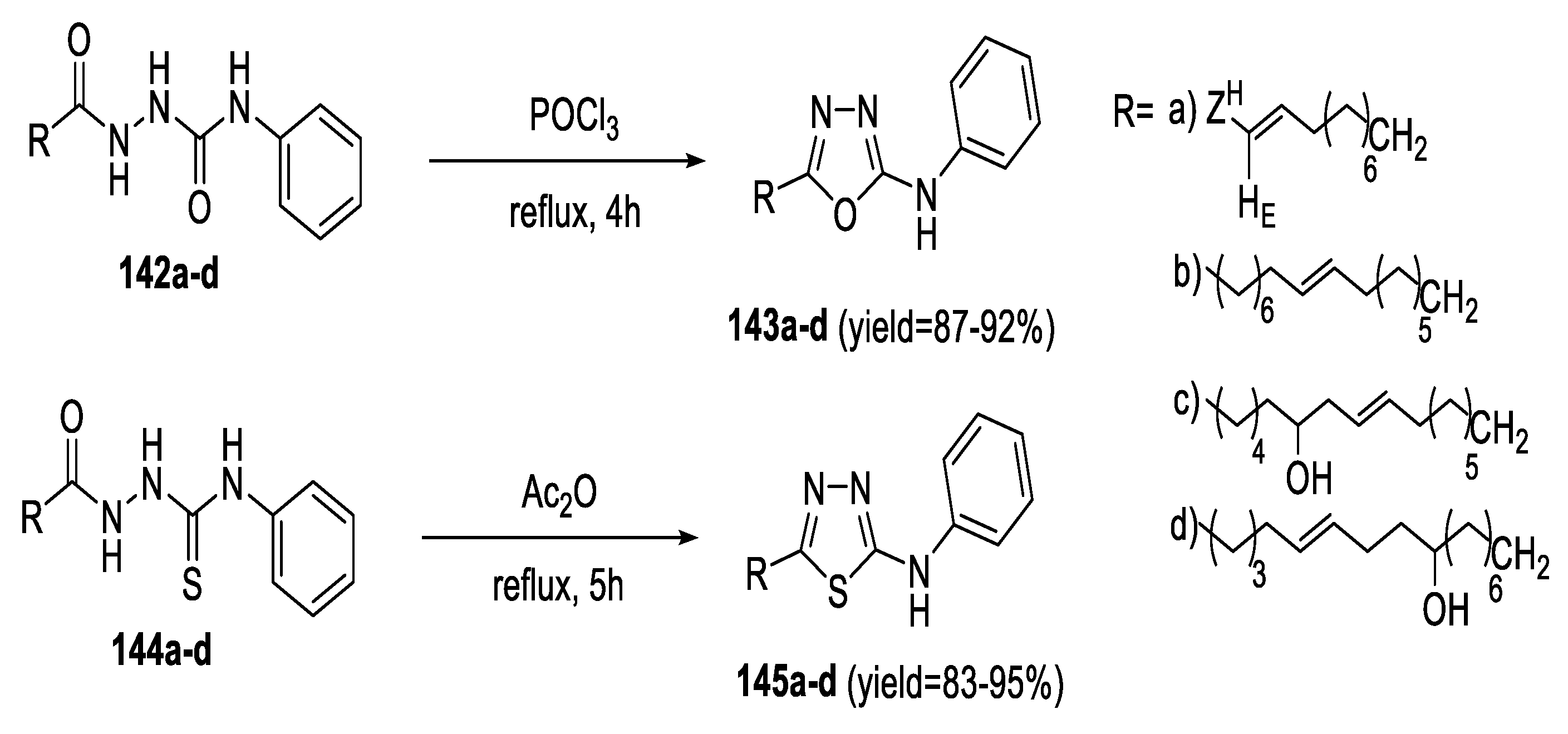
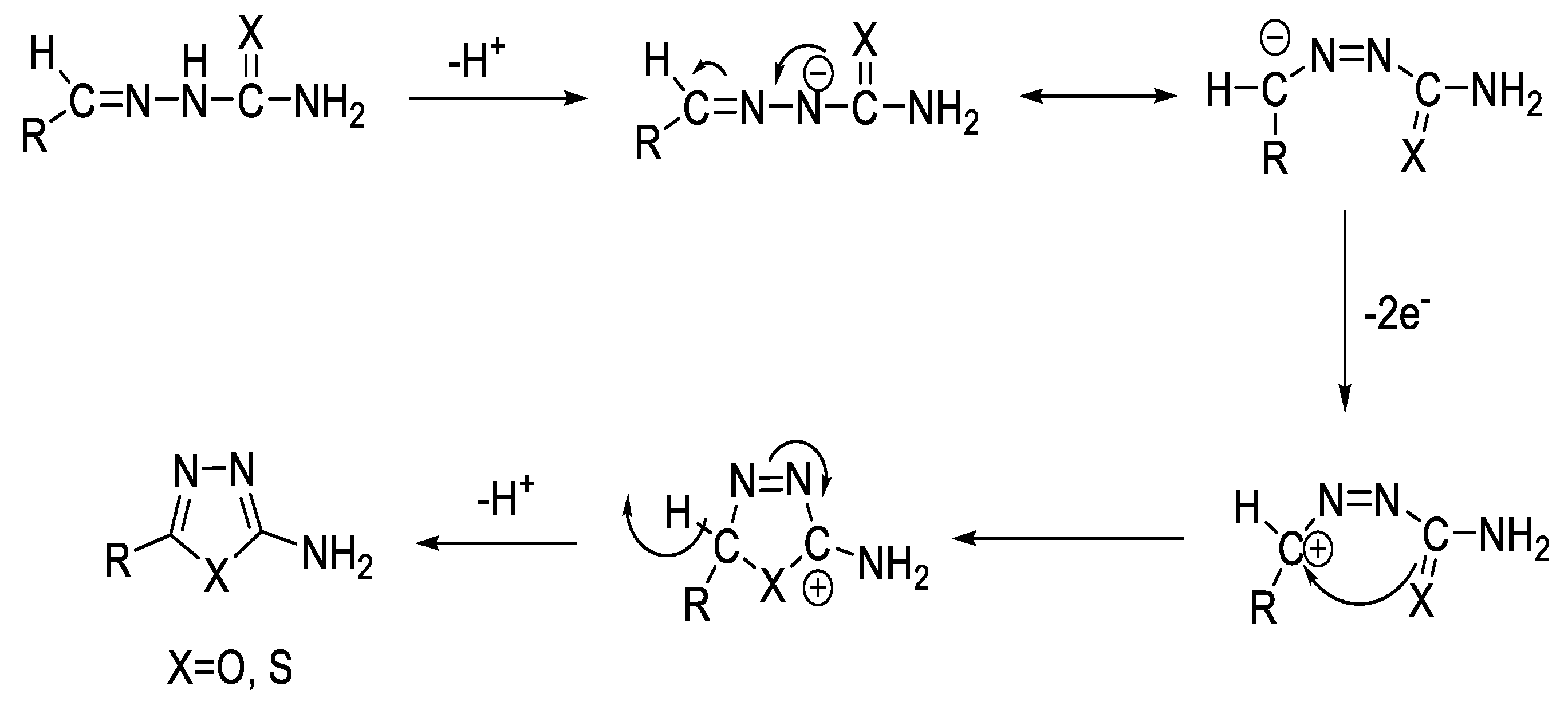
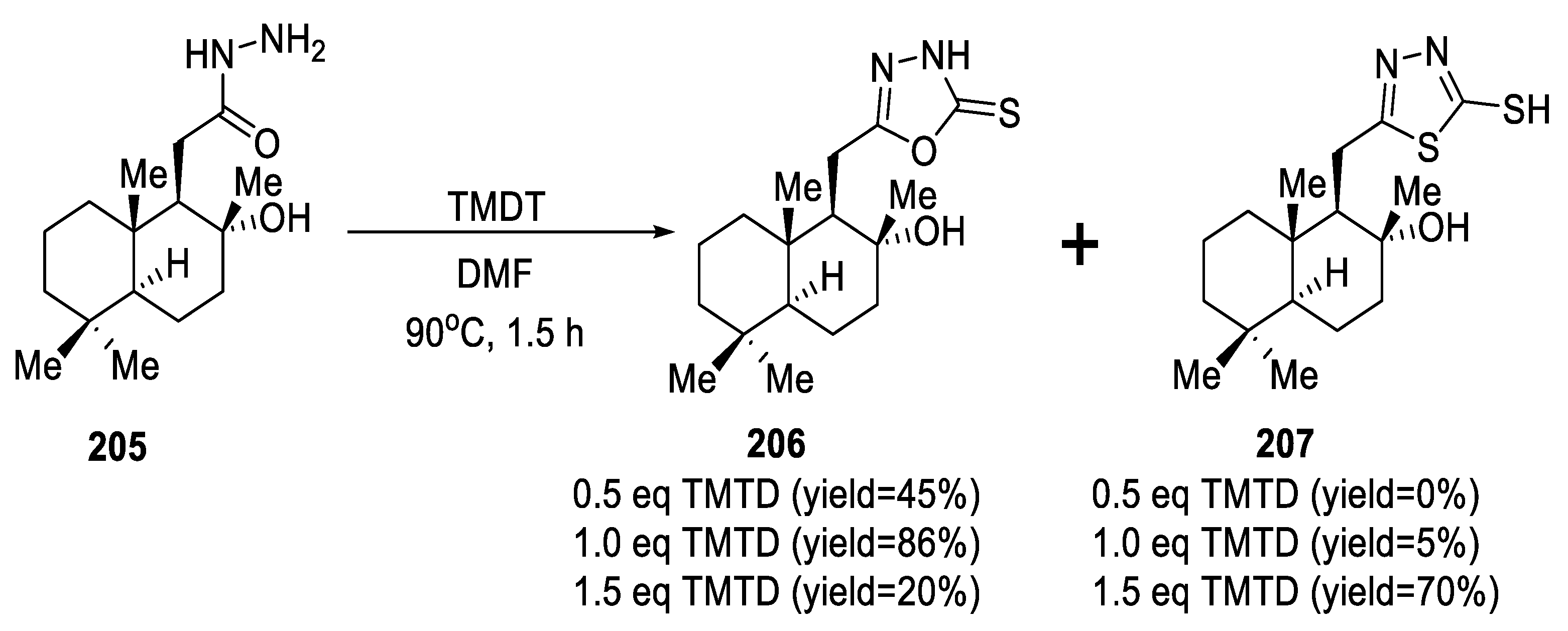
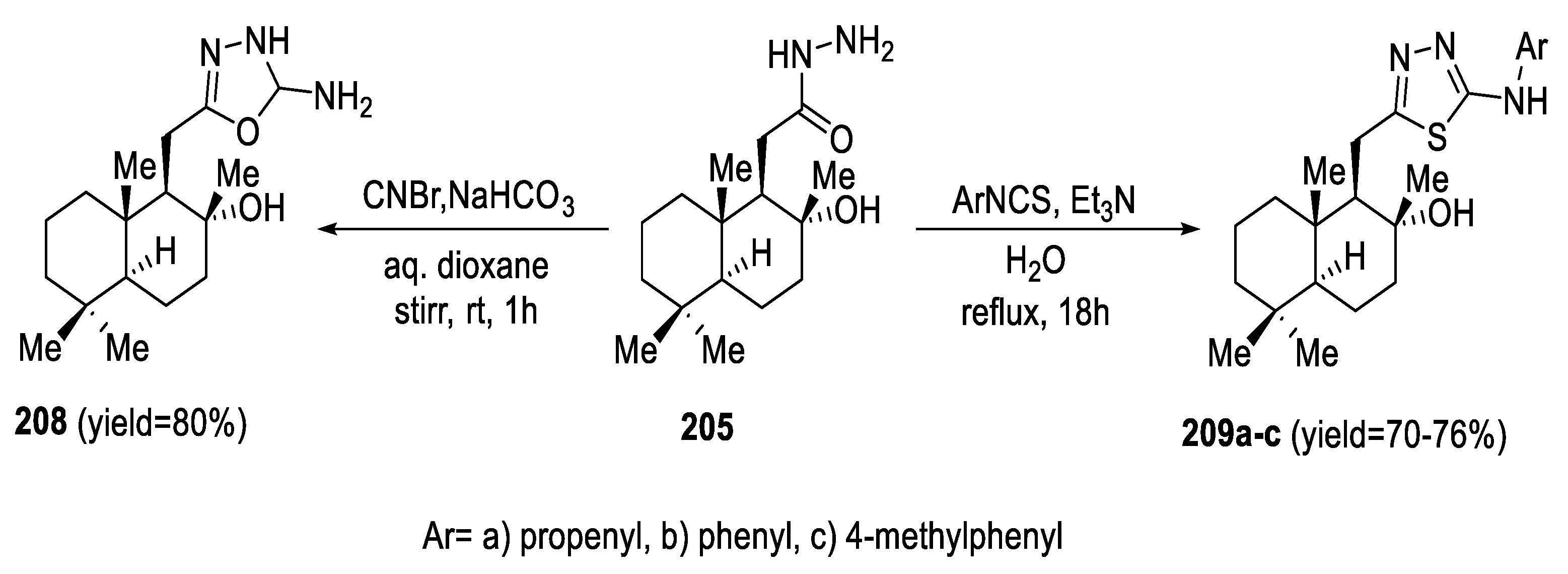
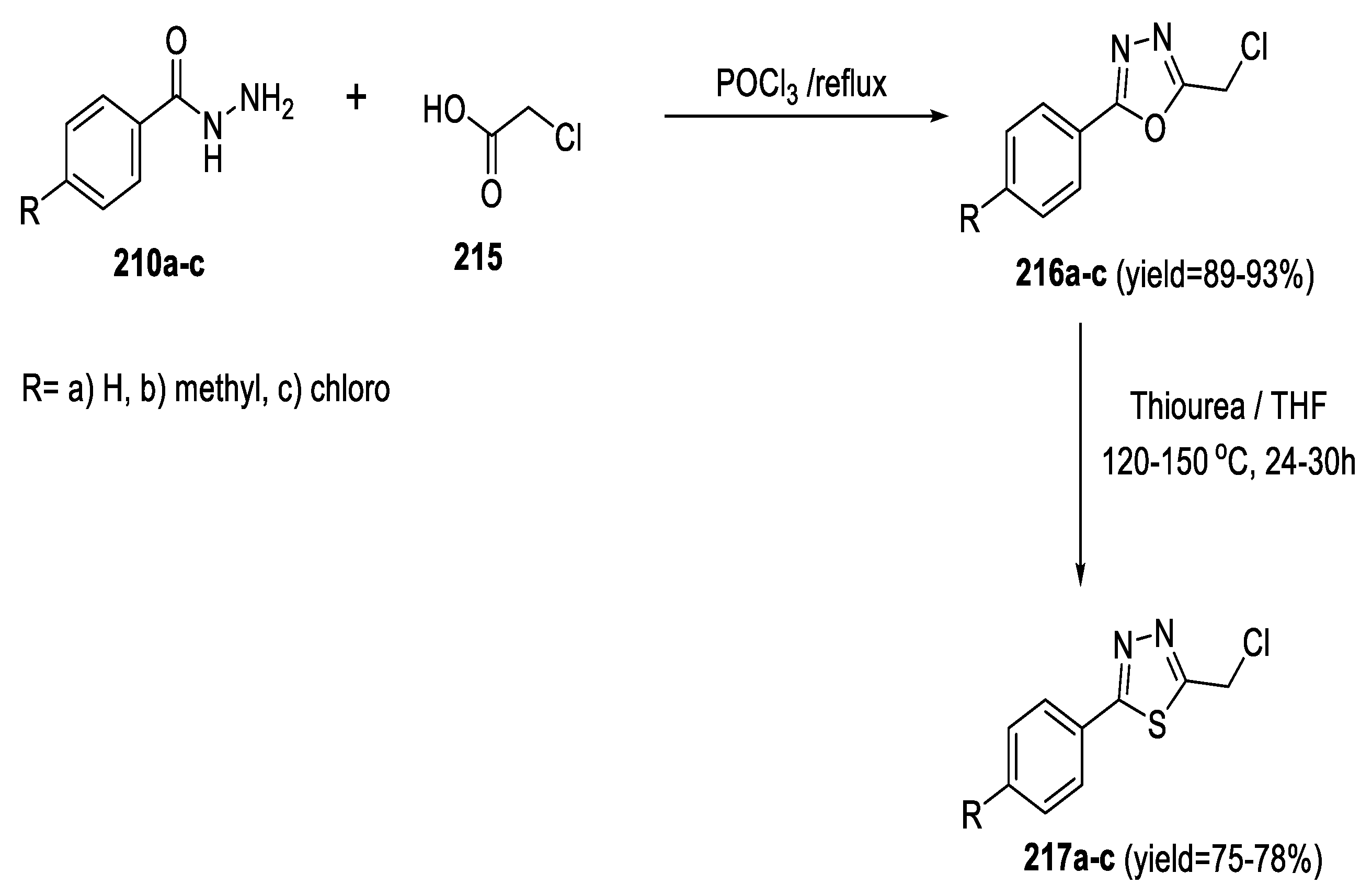
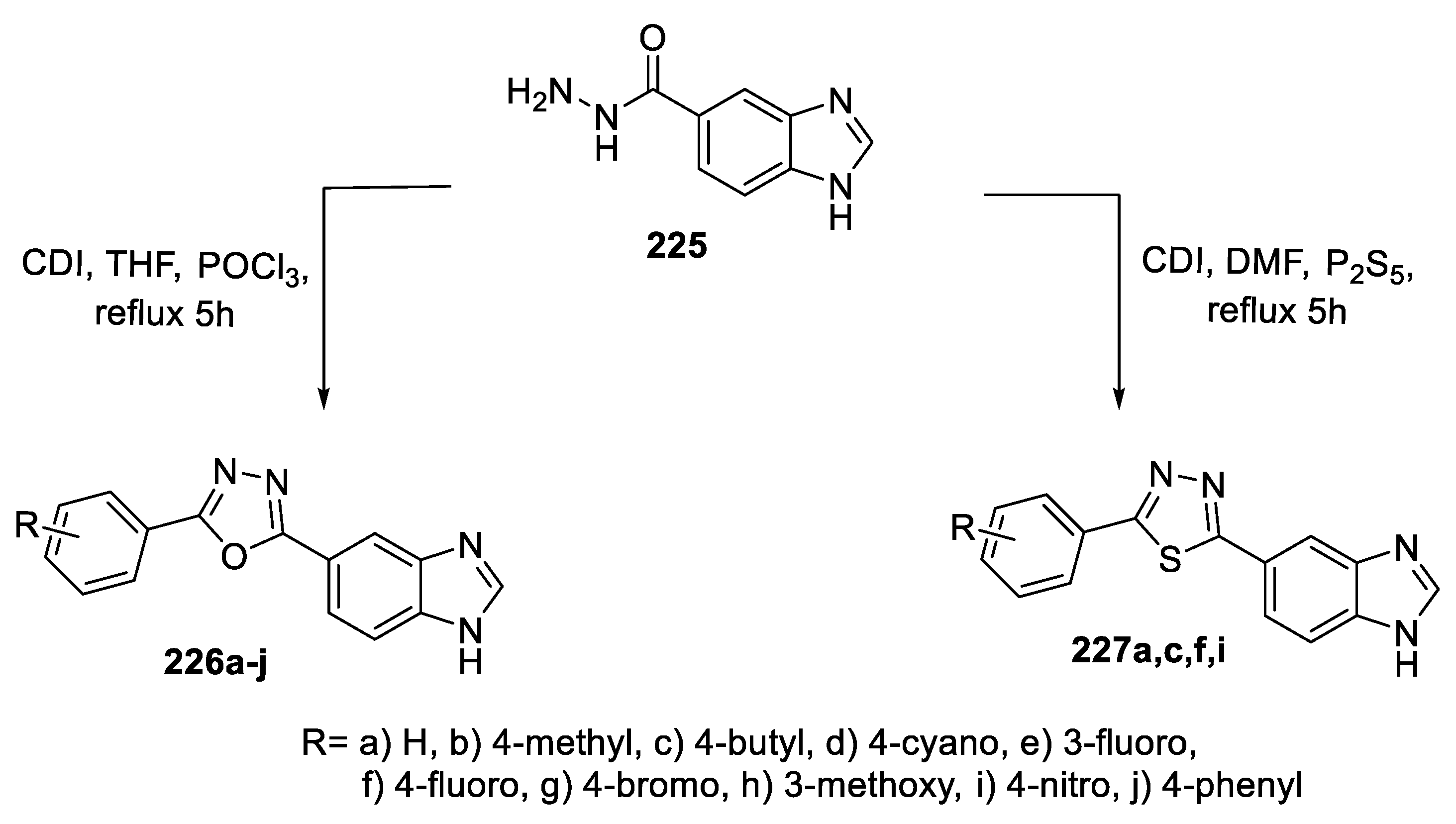
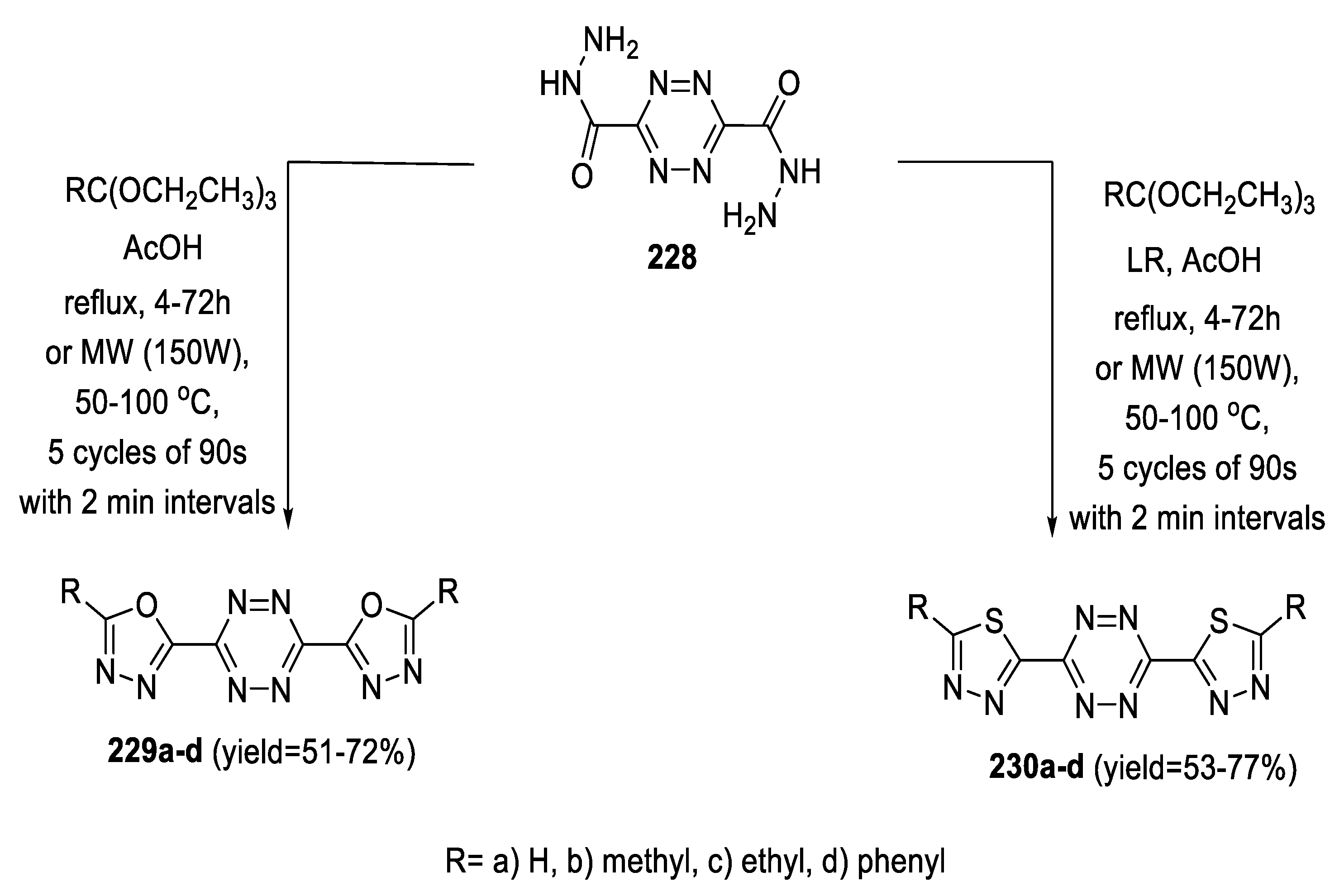
2.5. Synthesis from Acid/Thionyl Hydrazide
3. Biological Activities
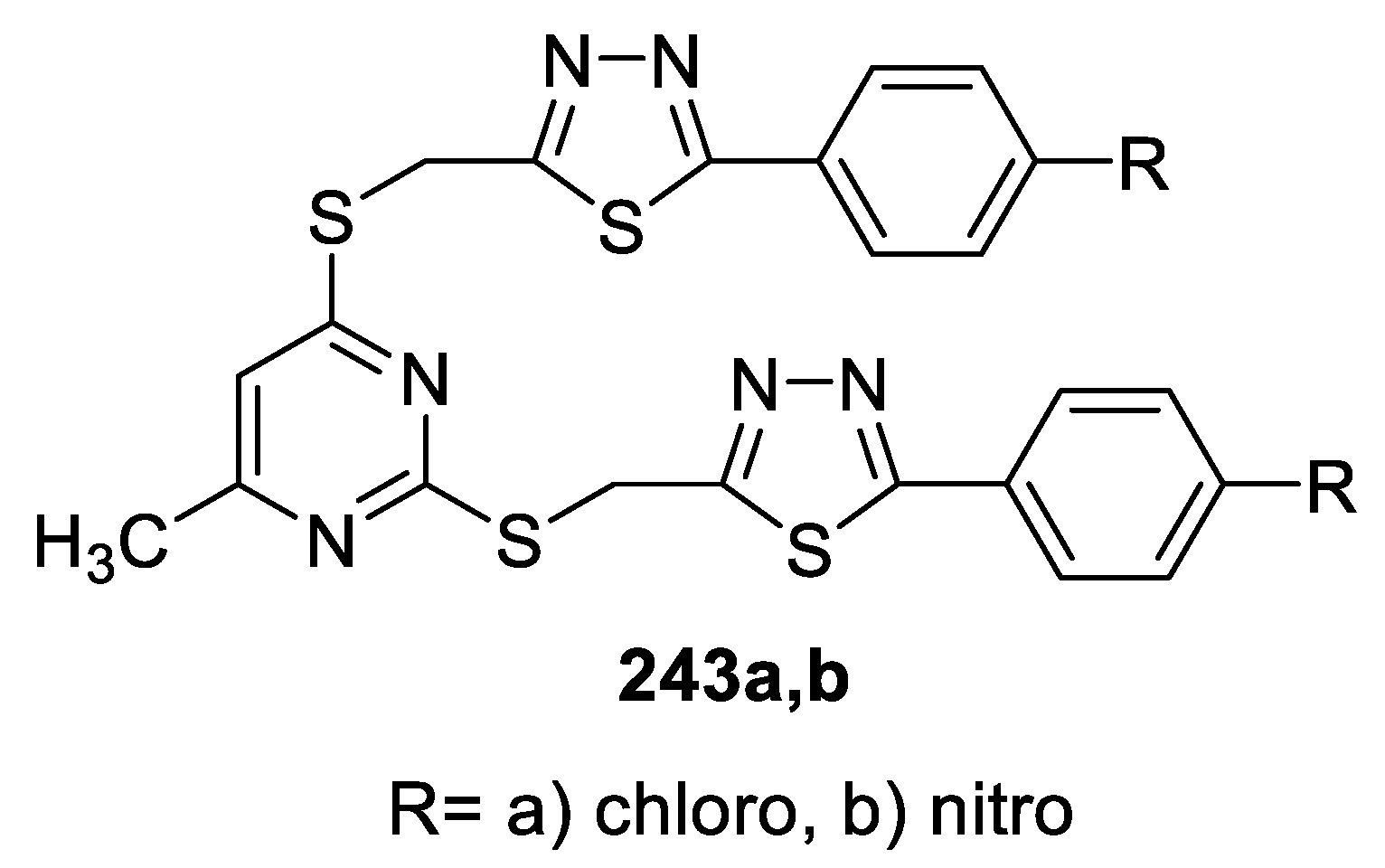
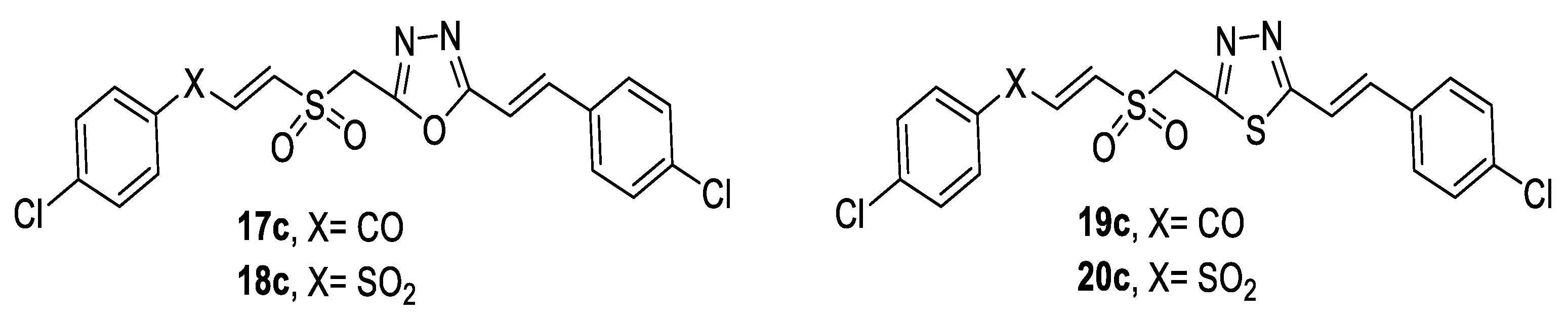
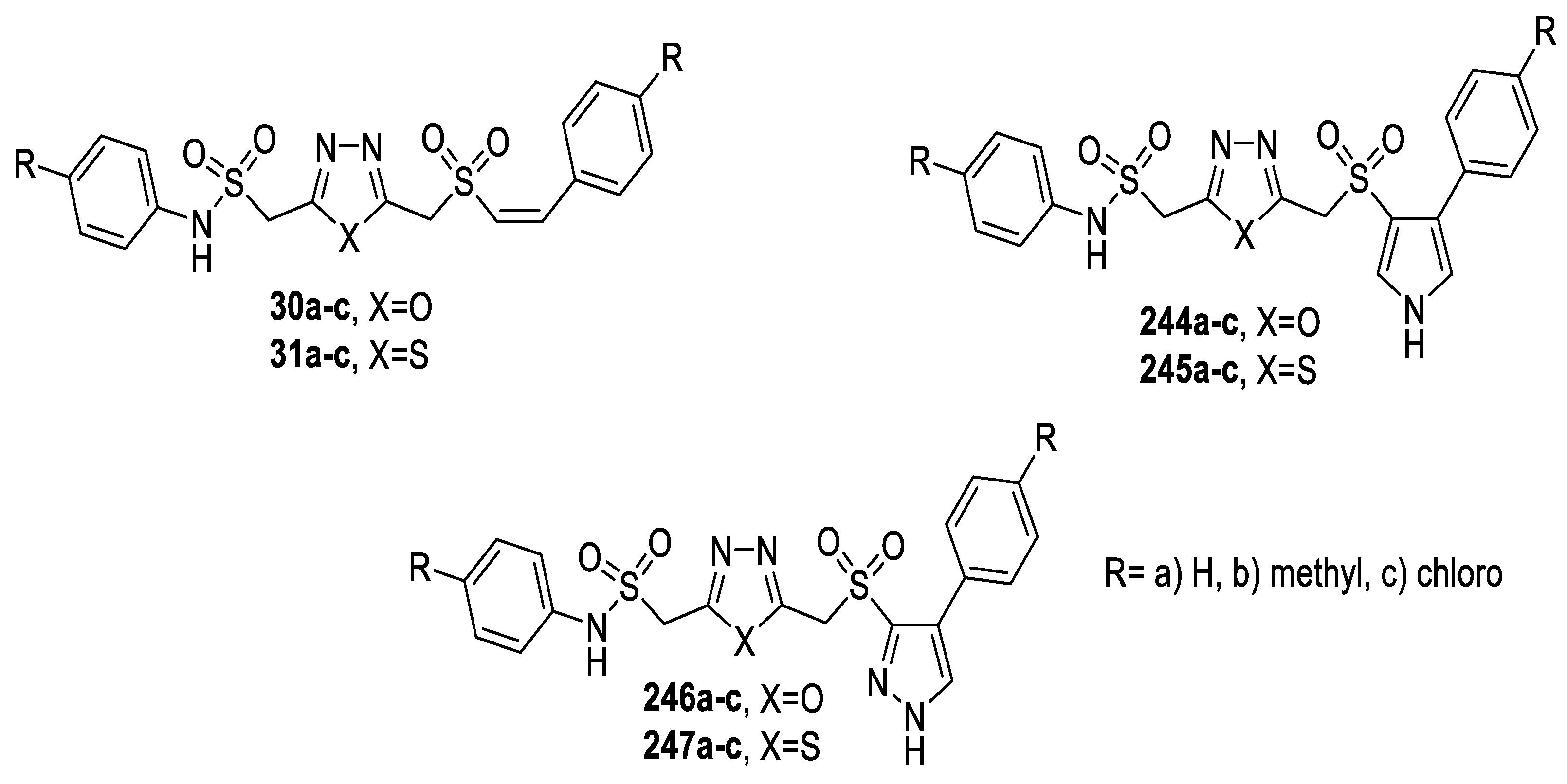
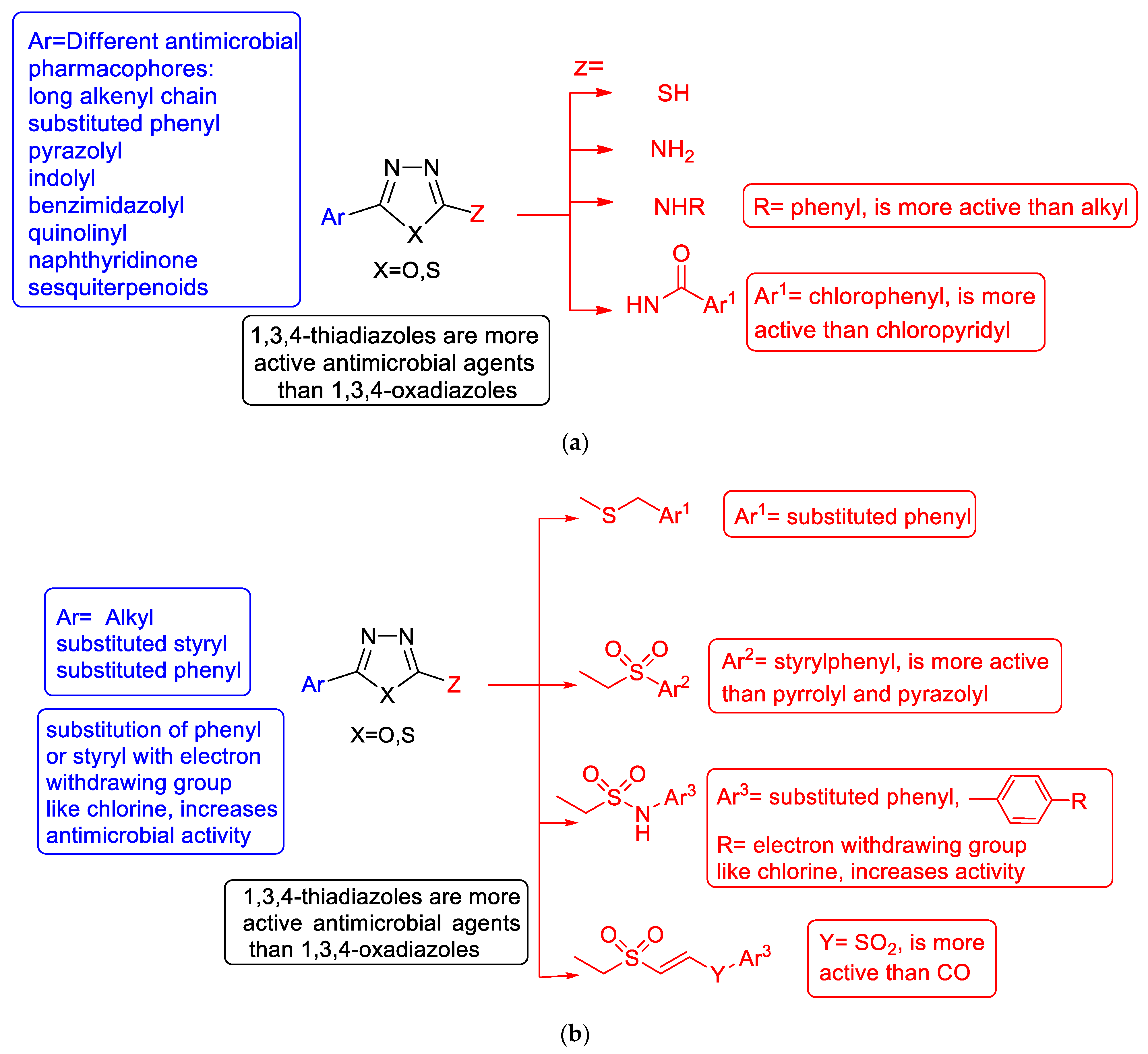
3.1. Antimicrobial Activity
3.2. Antiviral Activity
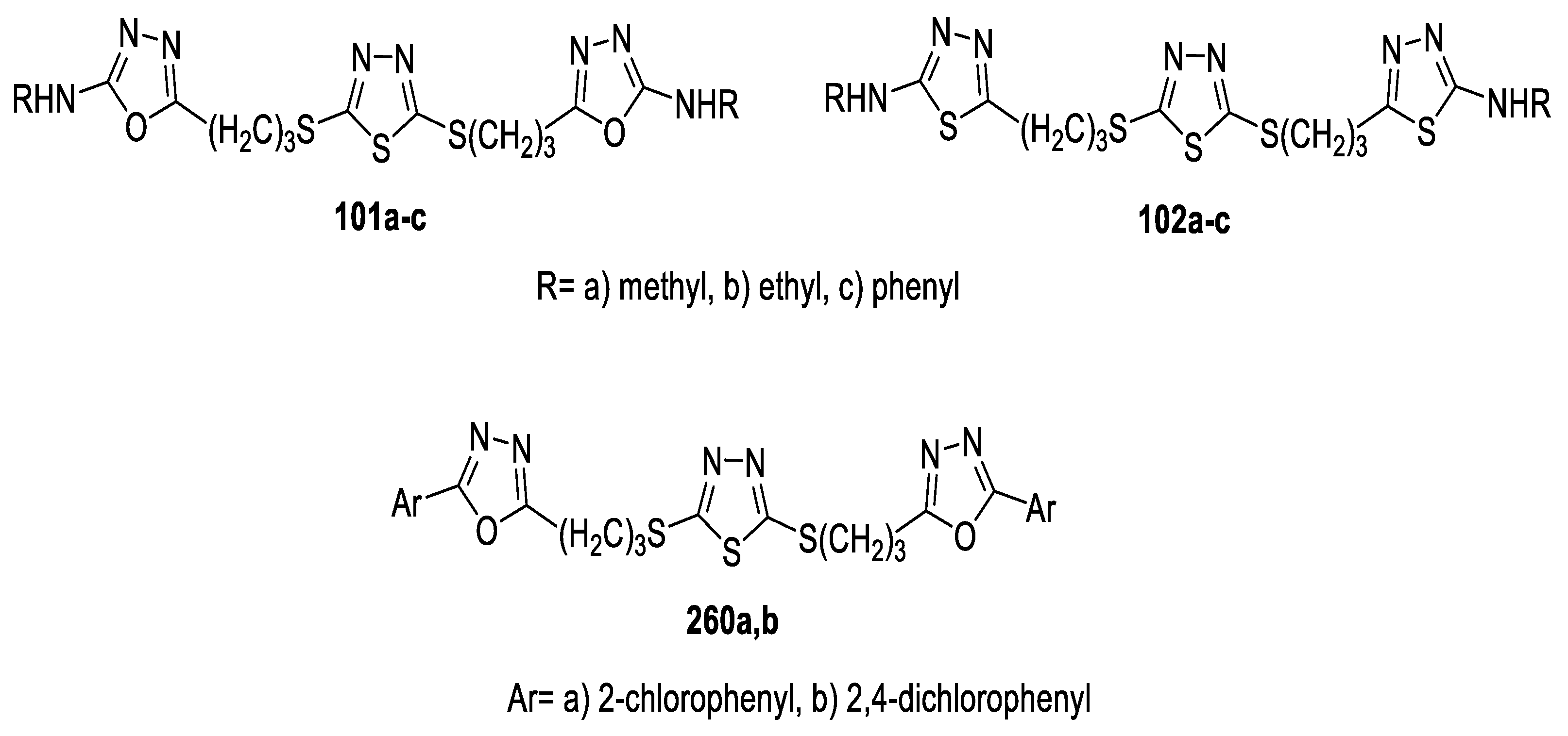
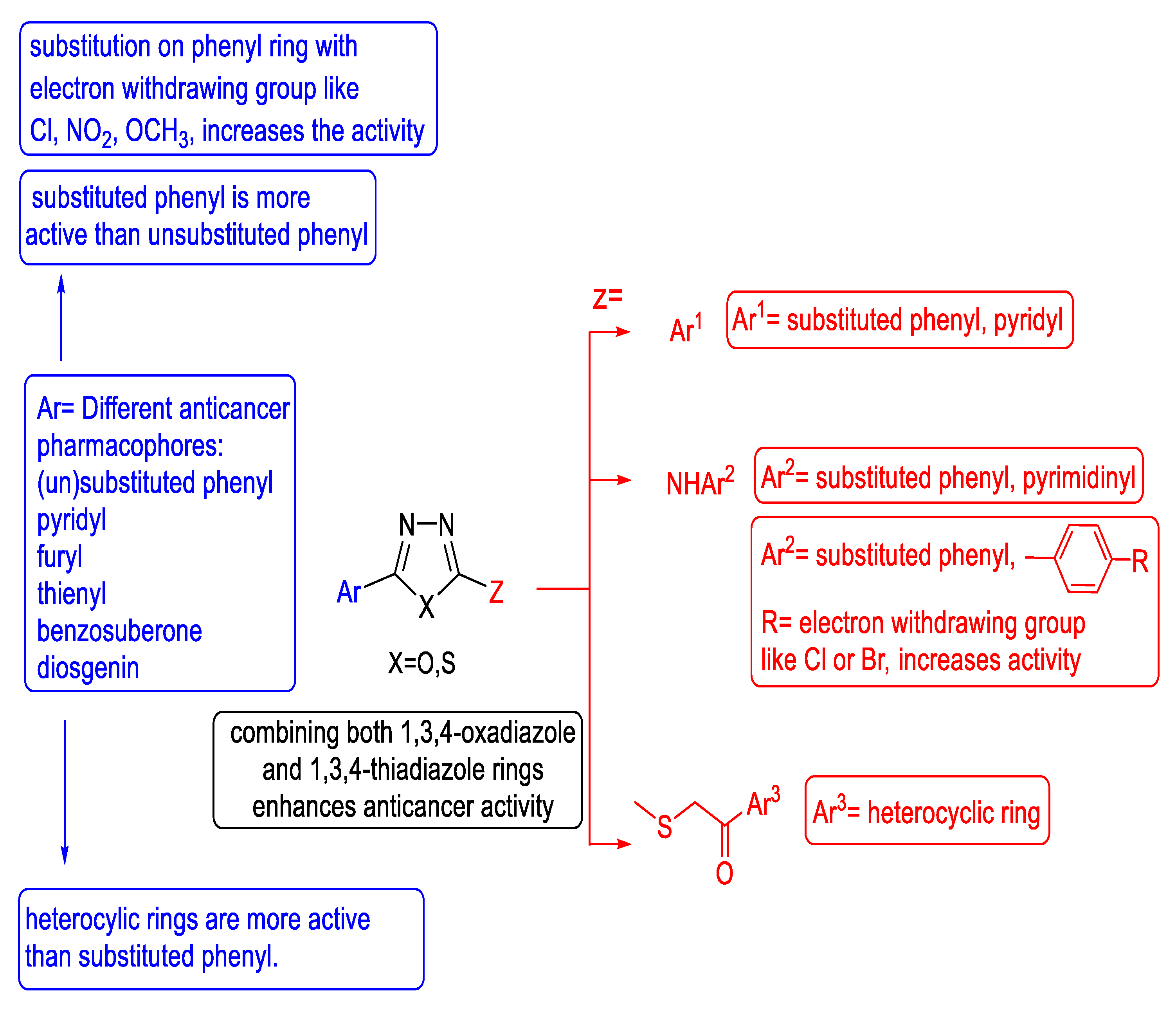
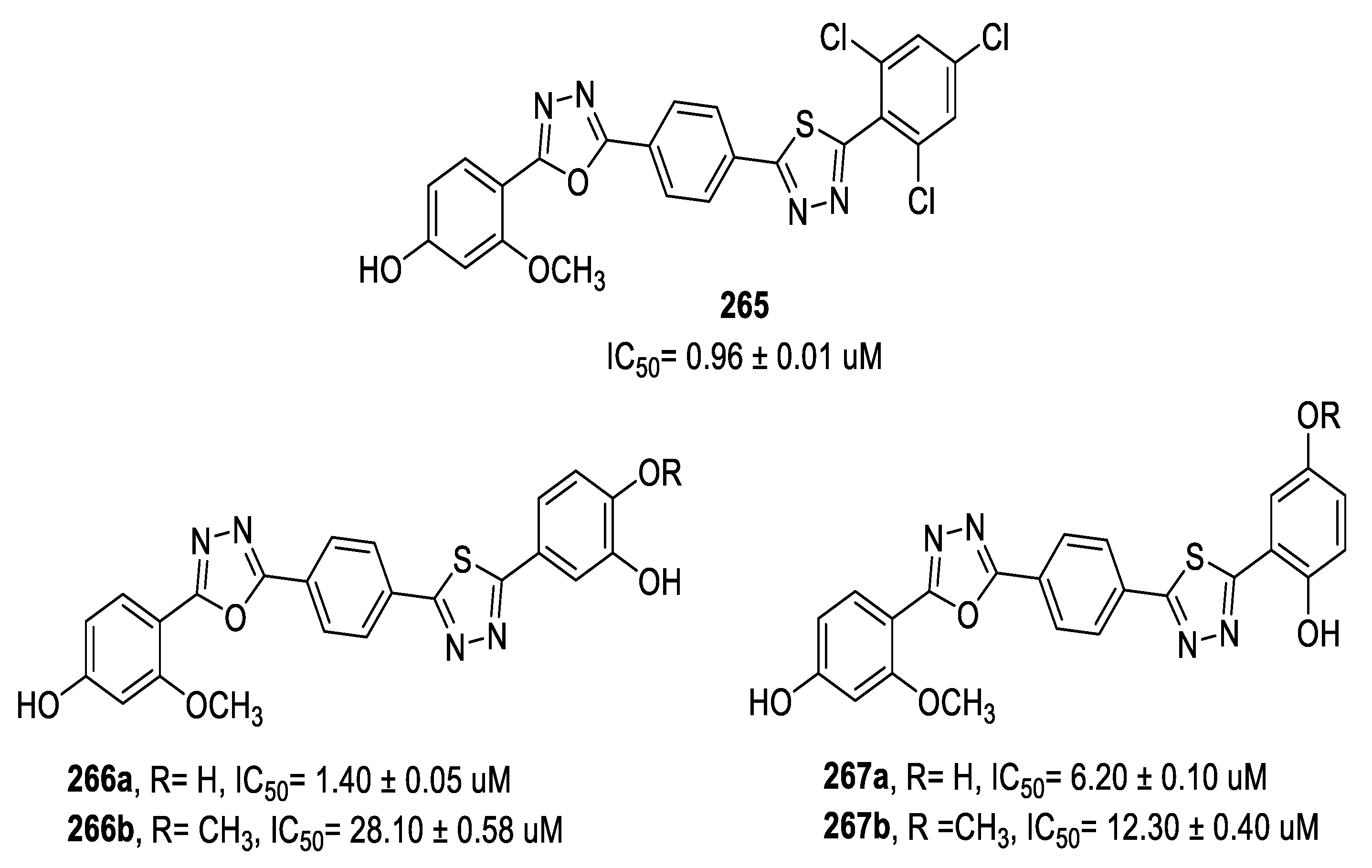
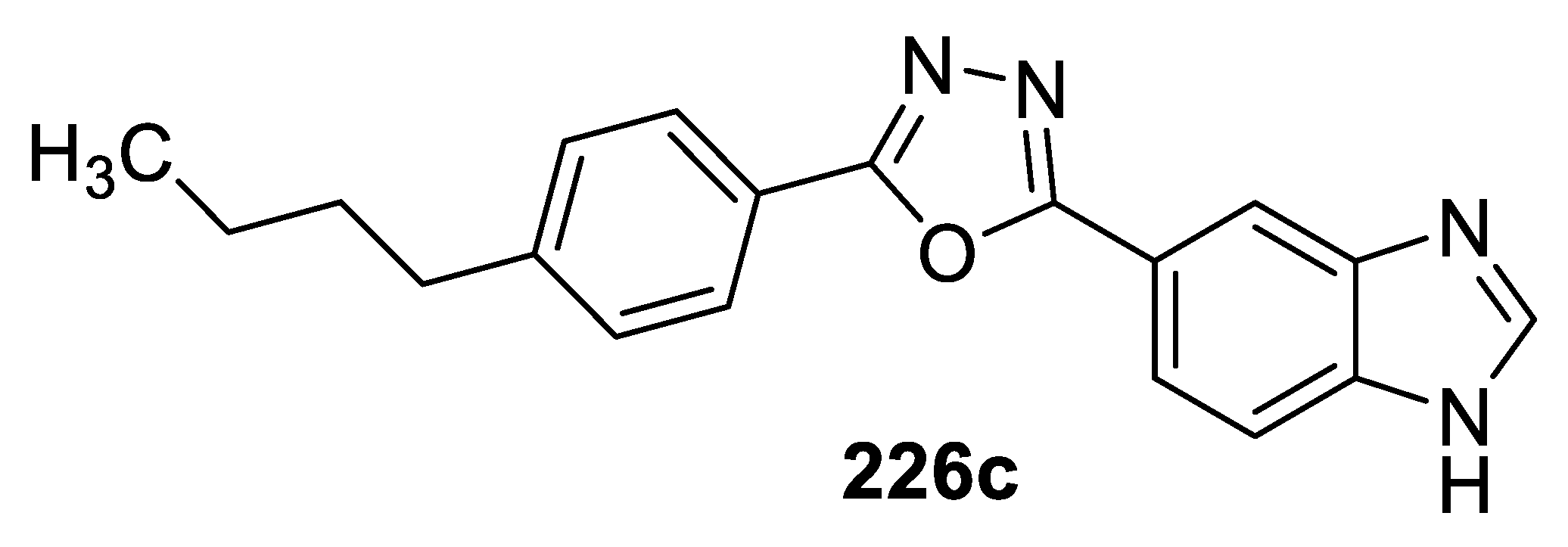
3.3. Anticancer Activity
3.4. Antidiabetic Activity
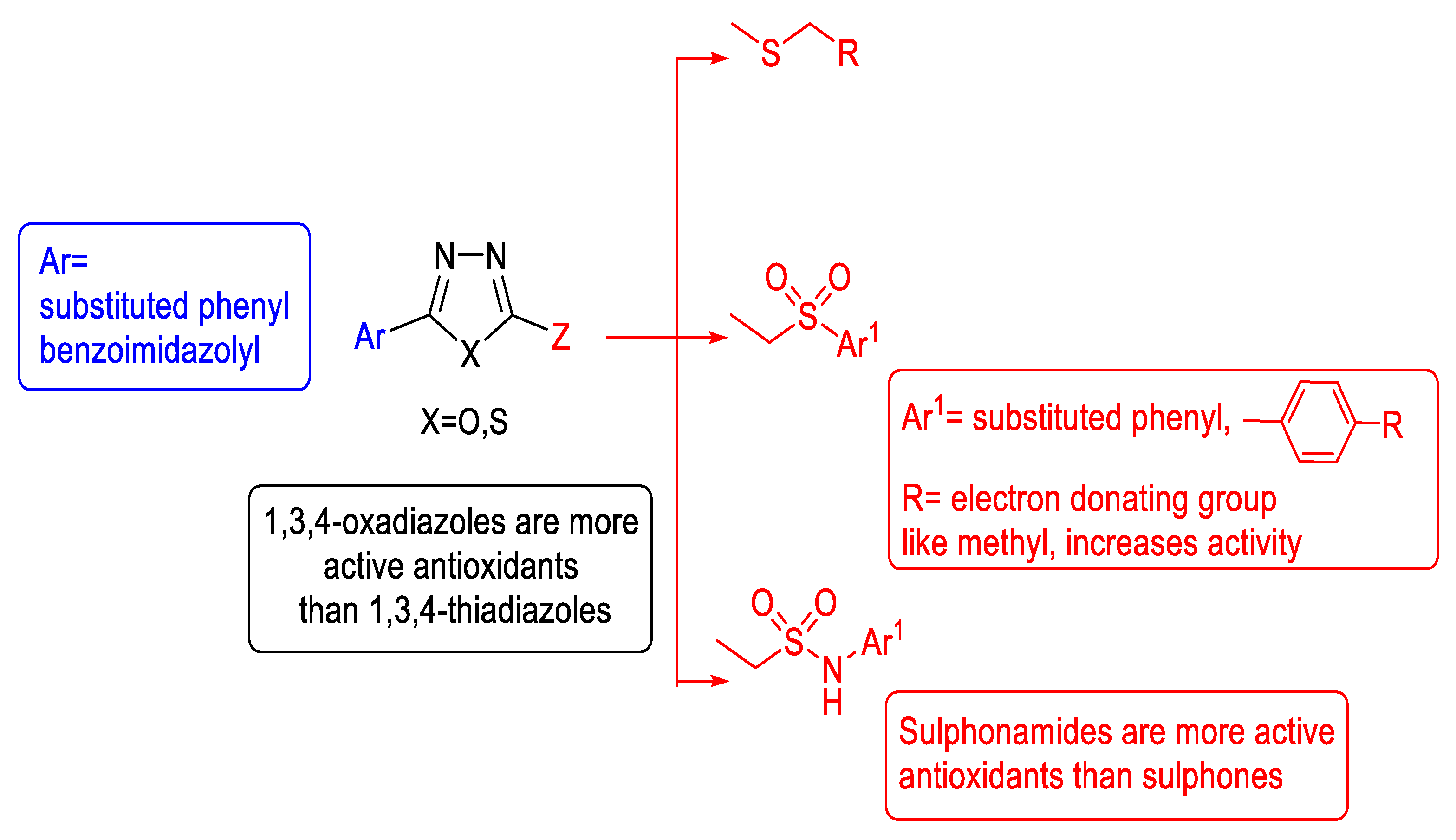
3.5. Antioxidant Activity
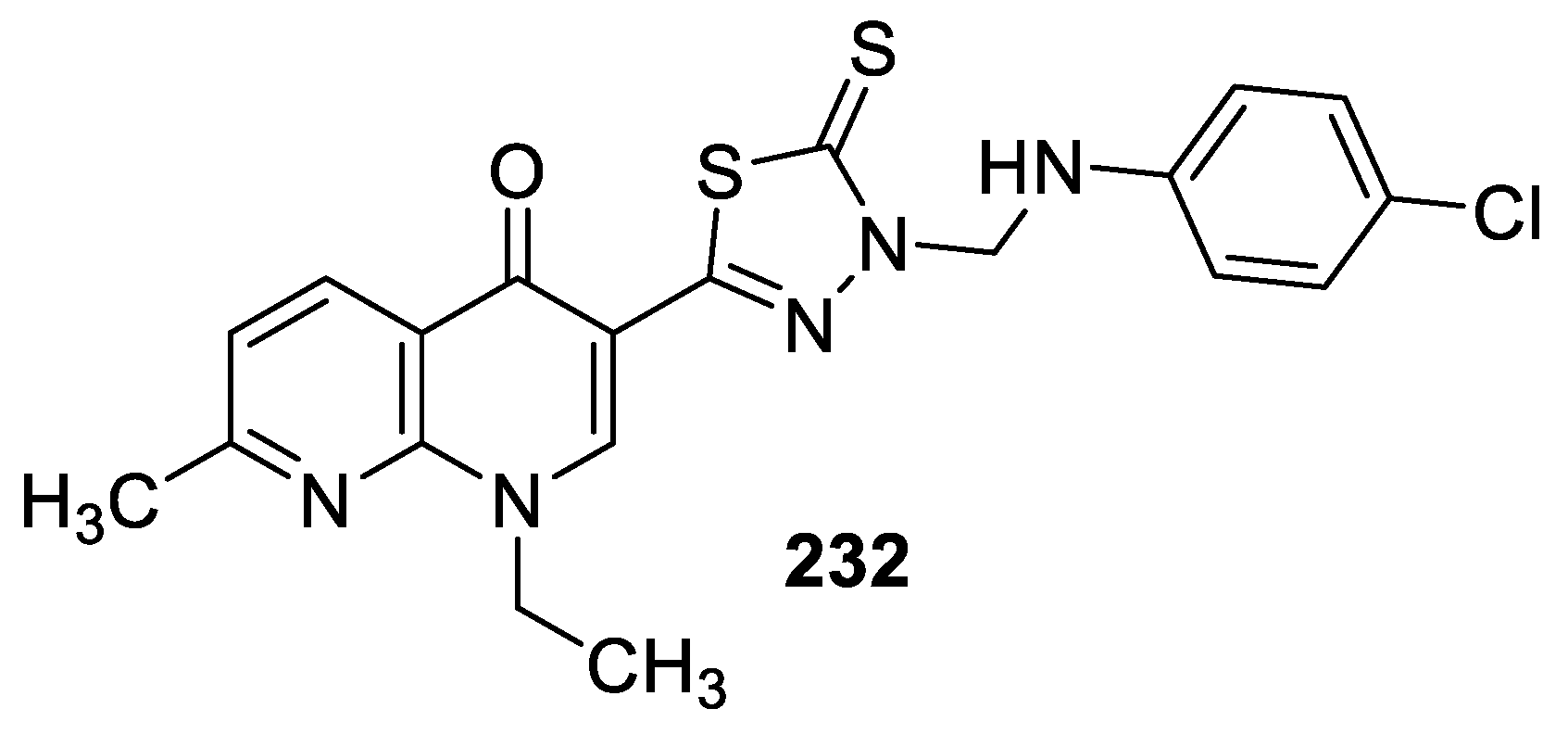
3.6. Analgesic and Anti-inflammatory Activities
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
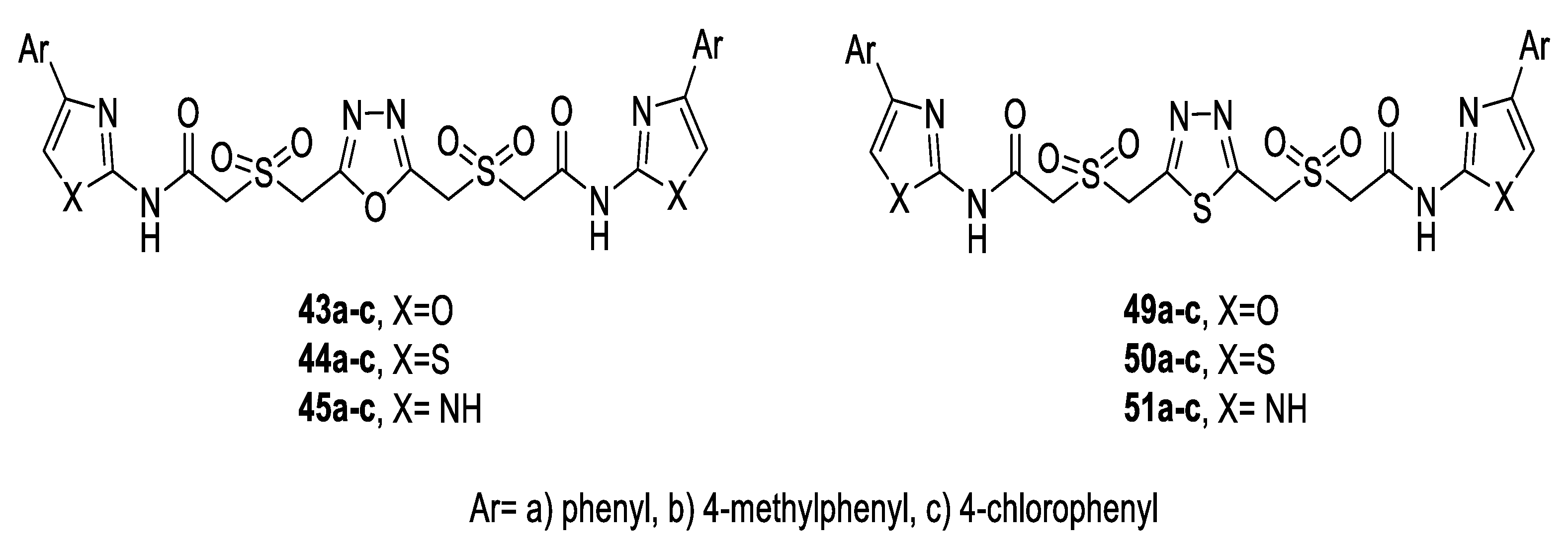
- Swapna, M.; Premakumari, C.; Reddy, S.N.; Padmaja, A.; Padmavathi, V. Synthesis and antioxidant activity of a variety of sulfonamidomethane linked 1,3,4-oxadiazoles and thiadiazoles. Chem. Pharm. Bull. 2013, 61, 611–617. [Google Scholar] [CrossRef][Green Version]
- Padmaja, A.; Pedamalakondaiah, D.; Sravya, G.; Reddy, G.M.; Kumar, M.V.J. Synthesis and antioxidant activity of a new class of sulfone/sulfonamide-linked Bis(Oxadiazoles), Bis(Thiadiazoles), and Bis(Triazoles). Med. Chem. Res. 2015, 24, 2011–2020. [Google Scholar] [CrossRef]
- Meanwell, N.A. Synopsis of some recent tactical application of bioisosteres in drug design. J. Med. Chem. 2011, 54, 2529–2591. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.M.; Salahuddin, A.; Azam, A. Synthesis of some 1,3,4-thiadiazole derivatives as inhibitors of entamoeba histolytica. Med. Chem. Res. 2013, 22, 1305–1312. [Google Scholar] [CrossRef]
- Boström, J.; Hogner, A.; Llinàs, A.; Wellner, E.; Plowright, A.T. Oxadiazoles in medicinal chemistry. J. Med. Chem. 2012, 55, 1817–1830. [Google Scholar] [CrossRef] [PubMed]
- Madhu Sekhar, M.; Yamini, G.; Divya, K.R.G.; Padmavathi, V.; Padmaja, A. Synthesis and bioassay of a new class of disubstituted 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazoles. Med. Chem. Res. 2019, 28, 1049–1062. [Google Scholar] [CrossRef]
- Wang, P.Y.; Zhou, L.; Zhou, J.; Wu, Z.B.; Xue, W.; Song, B.A.; Yang, S. Synthesis and antibacterial activity of pyridinium-tailored 2,5-substituted-1,3,4-oxadiazole thioether/sulfoxide/sulfone derivatives. Bioorg. Med. Chem. Lett. 2016, 26, 1214–1217. [Google Scholar] [CrossRef]
- Hannoun, M.H.; Hagras, M.; Kotb, A.; El-Attar, A.A.M.M.; Abulkhair, H.S. Synthesis and antibacterial evaluation of a novel library of 2-(Thiazol-5-Yl)-1,3,4-oxadiazole derivatives against Methicillin-Resistant Staphylococcus Aureus (MRSA). Bioorg. Chem. 2020, 94, 103364. [Google Scholar] [CrossRef]
- Karabanovich, G.; Němeček, J.; Valášková, L.; Carazo, A.; Konečná, K.; Stolaříková, J.; Hrabálek, A.; Pavliš, O.; Pávek, P.; Vávrová, K.; et al. S-Substituted 3,5-dinitrophenyl 1,3,4-oxadiazole-2-thiols and tetrazole-5-thiols as highly efficient antitubercular agents. Eur. J. Med. Chem. 2017, 126, 369–383. [Google Scholar] [CrossRef]
- Verma, G.; Chashoo, G.; Ali, A.; Khan, M.F.; Akhtar, W.; Ali, I.; Akhtar, M.; Alam, M.M.; Shaquiquzzaman, M. Synthesis of pyrazole acrylic acid based oxadiazole and amide derivatives as antimalarial and anticancer agents. Bioorg. Chem. 2018, 77, 106–124. [Google Scholar] [CrossRef] [PubMed]
- Almasirad, A.; Shafiee, A.; Abdollahi, M.; Noeparast, A.; Shahrokhinejad, N.; Vousooghi, N.; Tabatabai, S.A.; Khorasani, R. Synthesis and analgesic activity of new 1,3,4-oxadiazoles and 1,2,4-triazoles. Med. Chem. Res. 2011, 20, 435–442. [Google Scholar] [CrossRef]
- Nazar, S.; Siddiqui, N.; Alam, O. Recent progress of 1,3,4-oxadiazoles as anticonvulsants: Future horizons. Arch. Pharm. 2020, 353, 1900342. [Google Scholar] [CrossRef] [PubMed]
- Kumar Singh, A.; Lohani, M.; Parthsarthy, R. Synthesis, characterization and anti-inflammatory activity of various isatin derivatives. Iran. J. Pharm. Res. 2013, 12, 319–323. [Google Scholar]
- El-Essawy, F.A.; El-Sayed, W.A.; El-Kafrawy, S.A.; Morshedy, A.S.; Abdel-Rahman, A.H. Anti-hepatitis B virus activity of new 1,2,4-triazol-2-Yl- and 1,3,4-oxadiazol-2-Yl-2-pyridinone derivatives. Z. Naturforsch. Sect. C J. Biosci. 2008, 63, 667–674. [Google Scholar] [CrossRef]
- El-Masry, R.M.; Al-Karmalawy, A.A.; Alnajjar, R.; Mahmoud, S.H.; Mostafa, A.; Kadry, H.H.; Abou-Seri, S.M.; Taher, A.T. Newly synthesized series of oxoindole–oxadiazole conjugates as potential anti-SARS-CoV-2 agents: In silico and in vitro studies. New J. Chem. 2022, 46, 5078–5090. [Google Scholar] [CrossRef]
- Ahsan, M.J.; Choupra, A.; Sharma, R.K.; Jadav, S.S.; Padmaja, P.; Hassan, M.Z.; Al-Tamimi, A.B.S.; Geesi, M.H.; Bakht, M.A. Rationale design, synthesis, cytotoxicity evaluation, and molecular docking studies of 1,3,4-oxadiazole analogues. Anticancer Agents Med. Chem. 2017, 18, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Hamdi, N.; Passarelli, V.; Romerosa, A. Synthesis, spectroscopy and electrochemistry of new 4-(4-acetyl-5-substituted-4,5-dihydro-1,3,4-oxodiazol-2-yl)methoxy)-2H-chromen-2-ones as a novel class of potential antibacterial and antioxidant derivatives. Comptes Rendus Chim. 2011, 14, 548–555. [Google Scholar] [CrossRef]
- Siwach, A.; Verma, P.K. Therapeutic potential of oxadiazole or furadiazole containing compounds. BMC Chem. 2020, 14, 1–40. [Google Scholar] [CrossRef]
- Vaidya, A.; Pathak, D.; Shah, K. 1,3,4-Oxadiazole and its derivatives: A review on recent progress in anticancer activities. Chem. Biol. Drug Des. 2021, 97, 572–591. [Google Scholar] [CrossRef]
- Pouliot, M.; Angers, L.; Hamel, J. Synthesis of 1,3,4-oxadiazoles from 1,2-diacylhydrazines using [Et2NSF2]BF4 as a practical cyclodehydration agent. Org. Biomol. Chem. 2012, 10, 988–993. [Google Scholar] [CrossRef]
- Ogata, M.; Atobe, H.; Kushida, H. In vitro sensitivity of mycoplasmas isolated from various animals and sewage to antibiotics and nitrofurans. J. Antibiot. 1971, 24, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Schlecker, R.; Thieme, P. The synthesis of antihypertensive 3-(1,3,4-oxadiazol-2-Yl)phenoxypropanolahines. Tetrahedron 1988, 44, 3289–3294. [Google Scholar] [CrossRef]
- Serrao, E.; Odde, S.; Ramkumar, K.; Neamati, N. Raltegravir, elvitegravir, and metoogravir: The birth of “Me-Too” HIV-1 integrase inhibitors. Retrovirology 2009, 6, 1–14. [Google Scholar] [CrossRef]
- Partyka, R.A.; Crenshaw, R.R. 1,3,4-Oxadiazole Amide 1977. U.S. Patent 4001238, 4 January 1977. [Google Scholar]
- Vardan, S.; Smulyan, H.; Mookherjee, S.; Eich, R. Effects of tiodazosin, a new antihypertensive, hemodynamics and clinical variables. Clin. Pharmacol. Ther. 1983, 34, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Brandenberger, H.; Maes, R. Analytical toxicity for clinical, forensic and pharmaceutical chemists. In Clinical Biochemistry; Curtius, H.C., Roth, M., Eds.; De Gruyter: Berlin, Germany, 1997; Volume 95, pp. 419–421. ISBN 3110107317. [Google Scholar]
- Yarmohammadi, E.; Beyzaei, H.; Aryan, R.; Moradi, A. Ultrasound-assisted, low-solvent and acid/base-free synthesis of 5-substituted 1,3,4-oxadiazole-2-thiols as potent antimicrobial and antioxidant agents. Mol. Divers. 2021, 25, 2367–2378. [Google Scholar] [CrossRef] [PubMed]
- Haque, S.U.; Dashwood, M.R.; Heetun, M.; Shiwen, X.; Farooqui, N.; Ramesh, B.; Welch, H.; Savage, F.J.; Ogunbiyi, O.; Abraham, D.J.; et al. Efficacy of the specific endothelin a receptor antagonist Zibotentan (ZD4054) in colorectal cancer: A preclinical study. Mol. Cancer Ther. 2013, 12, 1556–1567. [Google Scholar] [CrossRef]
- Glomb, T.; Szymankiewicz, K.; Świątek, P. Anti-cancer activity of derivatives of 1,3,4-oxadiazole. Molecules 2018, 23, 3361. [Google Scholar] [CrossRef]
- Johansson, A.; Löfberg, C.; Antonsson, M.; Von Unge, S.; Hayes, M.A.; Judkins, R.; Ploj, K.; Benthem, L.; Lindén, D.; Brodin, P.; et al. Discovery of (3-(4-(2-Oxa-6-Azaspiro[3.3]Heptan-6- Ylmethyl)Phenoxy)Azetidin-1-Yl)(5-(4-Methoxyphenyl)-1,3,4-Oxadiazol-2- Yl)Methanone (AZD1979), a melanin concentrating hormone receptor 1 (MCHr1) antagonist with favourable physicochemical properties. J. Med. Chem. 2016, 59, 2497–2511. [Google Scholar] [CrossRef]
- Li, Y.; Geng, J.; Liu, Y.; Yu, S.; Zhao, G. Thiadiazole-a promising structure in medicinal chemistry. ChemMedChem 2013, 8, 27–41. [Google Scholar] [CrossRef]
- Yan, L.; Deng, M.; Chen, A.; Li, Y.; Zhang, W.; Du, Z.Y.; Dong, C.Z.; Meunier, B.; Chen, H. Synthesis of N-pyrimidin[1,3,4]oxadiazoles and N-Pyrimidin[1,3,4]-thiadiazoles from 1,3,4-oxadiazol-2-amines and 1,3,4-thiadiazol-2-amines via Pd-catalyzed heteroarylamination. Tetrahedron Lett. 2019, 60, 1359–1362. [Google Scholar] [CrossRef]
- Gomha, S.M.; Kheder, N.A.; Abdelhamid, A.O.; Mabkhot, Y.N. One pot single step synthesis and biological evaluation of some novel Bis(1,3,4-Thiadiazole) derivatives as potential cytotoxic agents. Molecules 2016, 21, 1532. [Google Scholar] [CrossRef]
- Hamama, W.S.; Gouda, M.A.; Badr, M.H.; Zoorob, H.H. Synthesis, antioxidant, and antitumor evaluation of certain new n-substituted-2-amino-1,3,4-thiadiazoles. Med. Chem. Res. 2013, 22, 3556–3565. [Google Scholar] [CrossRef]
- Chandrakantha, B.; Isloor, A.M.; Shetty, P.; Fun, H.K.; Hegde, G. Synthesis and biological evaluation of novel substituted 1,3,4-thiadiazole and 2,6-di aryl substituted imidazo [2,1-b] [1,3,4] thiadiazole derivatives. Eur. J. Med. Chem. 2014, 71, 316–323. [Google Scholar] [CrossRef]
- Khan, I.; Tantray, M.A.; Hamid, H.; Alam, M.S.; Kalam, A.; Dhulap, A. Synthesis of benzimidazole based thiadiazole and carbohydrazide conjugates as glycogen synthase kinase-3β inhibitors with anti-depressant activity. Bioorg. Med. Chem. Lett. 2016, 26, 4020–4024. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, N.; Ahuja, P.; Malik, S.; Arya, S.K. Design of benzothiazole-1,3,4-thiadiazole conjugates: Synthesis and anticonvulsant evaluation. Arch. Pharm. 2013, 346, 819–831. [Google Scholar] [CrossRef]
- Shkair, M.H.A.; Shakya, A.K.; Raghavendra, N.M.; Naik, R.R. Molecular modeling, synthesis and pharmacological evaluation of 1,3,4- thiadiazoles as anti-inflammatory and analgesic agents. Med. Chem. 2016, 12, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Hill, J. 4.23—1,3,4-oxadiazoles. In Comprehensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Eds.; Pergamon Press, Ltd.: Oxford, UK, 1984; pp. 427–446. [Google Scholar]
- Kakitani, T.; Kakitani, H. Application of self-consistent HMO theory to heteroconjugated molecules. Theor. Chim. Acta 1977, 46, 259–275. [Google Scholar] [CrossRef]
- Ha, T.-K. A Theoretical study of the electronic structure and properties of some five-membered heterocyclic compounds: Pyrazole, imidazole, furan, isoxazole, 1,2,5-oxadiazole and 1,3,4-oxadiazole. J. Mol. Struct. 1979, 51, 87–98. [Google Scholar] [CrossRef]
- Kosobutskii, V.A.; Kagan, G.I.; Betyakov, V.K. Amide-imidol tautomerism in aromatic polyamides. J. Struct. Chem. 1972, 12, 753–760. [Google Scholar] [CrossRef]
- Aydogan, F.; Turgut, Z.; Ocal, N. Synthesis and electronic structure of new aryl- and alkyl-substituted 1,3,4-oxadiazole-2-thione derivatives. Turk. J. Chem. 2002, 26, 159–169. [Google Scholar]
- Khiati, Z.; Othman, A.A.; Guessas, B. Synthesis and antibacterial activity of 1,3,4-oxadiazole and 1,2,4-triazole derivatives of salicylic acid and its synthetic intermediates. S. Afr. J. Chem. 2007, 60, 20–24. [Google Scholar]
- Sharma, B.; Verma, A.; Prajapati, S.; Sharma, U.K. Synthetic methods, chemistry, and the anticonvulsant activity of thiadiazoles. Int. J. Med. Chem. 2013, 2013, 1–16. [Google Scholar] [CrossRef]
- Goerdler, J.; Ohm, J.; Tegmeyer, O. Darstellung und eigenschaften des 1.2.4- und des 1.3.4-thiodiazols. Chem. Ber. 1956, 89, 1534–1543. [Google Scholar] [CrossRef]
- Othman, A.A.; Kihel, M.; Amara, S. 1,3,4-oxadiazole, 1,3,4-thiadiazole and 1,2,4-triazole derivatives as potential antibacterial agents. Arab. J. Chem. 2019, 12, 1660–1675. [Google Scholar] [CrossRef]
- Strzemecka, L.; Maciejewska, D.; Urbanczyk, Z. The structure of N-allyl derivatives of (5-(2′-pyridyl)- [1,3,4]thiadiazol-2-yl) amine in solution and the solid state studied by the 1H, 13C, 15N NMR spectroscopy, X-ray crystallography and DFT computations. J. Mol. Struct. 2003, 648, 107–113. [Google Scholar] [CrossRef]
- Padmaja, A.; Muralikrishna, A.; Rajasekhar, C.; Padmavathi, V. Synthesis and antimicrobial activity of pyrrolyl/pyrazolyl arylaminosulfonylmethyl 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazoles. Chem. Pharm. Bull. 2011, 59, 1509–1517. [Google Scholar] [CrossRef][Green Version]
- Padmavathi, V.; Reddy, S.N.; Reddy, G.D.; Padmaja, A. Synthesis and bioassay of aminosulfonyl-1,3,4-oxadiazoles and their interconversion to 1,3,4-thiadiazoles. Eur. J. Med. Chem. 2010, 45, 4246–4251. [Google Scholar] [CrossRef] [PubMed]
- Basha, N.M.; Reddy, P.R.; Padmaja, A.; Padmavathi, V.; Duszynska, B.; Bojarski, A.J.; Strekowski, L. Synthesis and antioxidant activity of bis-oxazolyl/thiazolyl/imidazolyl 1,3,4-oxadiazoles and 1,3,4-thiadiazoles. J. Heterocycl. Chem. 2016, 53, 1276–1283. [Google Scholar] [CrossRef]
- Du, Y.; Wan, Z.; Chen, L.; Wu, L. One pot solvent-free solid state synthesis, photophysical properties and crystal structure of substituted azole derivatives. J. Mol. Struct. 2019, 1193, 315–325. [Google Scholar] [CrossRef]
- Anna, K.; Kudelko, A.; Marcin, Ś.; Kruszynski, R. Microwave-promoted synthesis of highly luminescent s-tetrazine-1,3,4- oxadiazole and s-tetrazine-1,3,4-thiadiazole hybrids. Dye. Pigment. 2020, 172, 107865. [Google Scholar] [CrossRef]
- Nagendra, G.; Lamani, R.S.; Narendra, N.; Sureshbabu, V.V. A Convenient synthesis of 1,3,4-thiadiazole and 1,3,4-oxadiazole based peptidomimetics employing diacylhydrazines derived from amino acids. Tetrahedron Lett. 2010, 51, 6338–6341. [Google Scholar] [CrossRef]
- Mutchu, B.R.; Kotra, V.; Onteddu, S.R.; Boddapati, S.N.M.; Bollikolla, H.B. Synthesis, cytotoxicity and antimicrobial evaluation of some new 2-aryl,5-substituted 1,3,4-oxadiazoles and 1,3,4-thiadiazoles. Chem. Africa 2019, 2, 15–20. [Google Scholar] [CrossRef]
- Taylor, P.; Prajapati, A.K.; Modi, V. Mesogenic bent-shaped nitrooxadiazoles and thiadiazoles. Liq. Cryst. 2011, 38, 191–199. [Google Scholar] [CrossRef]
- Schmidt, M. A New Late-Stage Lawesson’s Cyclization Strategy Towards the Synthesis of Aryl 1,3,4-Thiadiazole-2-Carboxylate Esters; Kent State University: Kent, OH, USA, 2013. [Google Scholar]
- Zhang, J.; Wang, X.; Yang, J.; Guo, L.; Wang, X.; Song, B.; Wang, W. Novel diosgenin derivatives containing 1,3,4-oxadiazole/thiadiazole moieties as potential antitumor agents: Design, synthesis and cytotoxic evaluation. Eur. J. Med. Chem. 2019, 111897. [Google Scholar] [CrossRef] [PubMed]
- Hajimahdi, Z.; Zarghi, A.; Zabihollahi, R.; Aghasadeghi, M.R. Synthesis, biological evaluation, and molecular modeling studies of new 1,3,4-oxadiazole- and 1,3,4-thiadiazole-substituted 4-oxo-4h-pyrido[1,2-a]pyrimidines as anti-HIV-1 agents. Med. Chem. Res. 2012, 22, 2467–2475. [Google Scholar] [CrossRef]
- Yavuz, S.; Unal, Y.; Pamir, O.; Yılmazer, D.; Kurtipek, O.; Kavutcu, M.; Arslan, M.; Ark, M.; Yıldırır, Y. Synthesis and pharmacological evaluation of some novel thebaine derivatives: N-(tetrazol-1H-5-yl)-6,14- endoethenotetrahydrothebaine incorporating the 1,3,4-oxadiazole or the 1,3,4-thiadiazole moiety. Arch. Pharm. Chem. Life Sci. 2013, 346, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A.; Soliman, M.A.; Aouad, M.R.; Messali, M.; Ali, A.A.; Soliman, M.A.; Aouad, M.R. Synthesis, characterization, and antimicrobial screening of novel 1,2,4-triazoles, 1,3,4- thiadiazoles, and 1,3,4-oxadiazoles bearing the indole moiety. Org. Prep. Proced. Int. 2019, 51, 270–286. [Google Scholar] [CrossRef]
- Abu-Hashem, A.A. Synthesis and antimicrobial activity of new 1,2,4-triazole, 1,3,4-oxadiazole, 1,3,4-thiadiazole, thiopyrane, thiazolidinone, and azepine derivatives. J. Heterocycl. Chem. 2021, 58, 74–92. [Google Scholar] [CrossRef]
- Yang, S.; Choe, J.; Abdildinova, A.; Gong, Y. A Highly efficient diversification of 2-amino/amido-1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives via reagent-based cyclization of thiosemicarbazide intermediate on solid-phase. ACS Comb. Sci. 2015, 17, 732–741. [Google Scholar] [CrossRef]
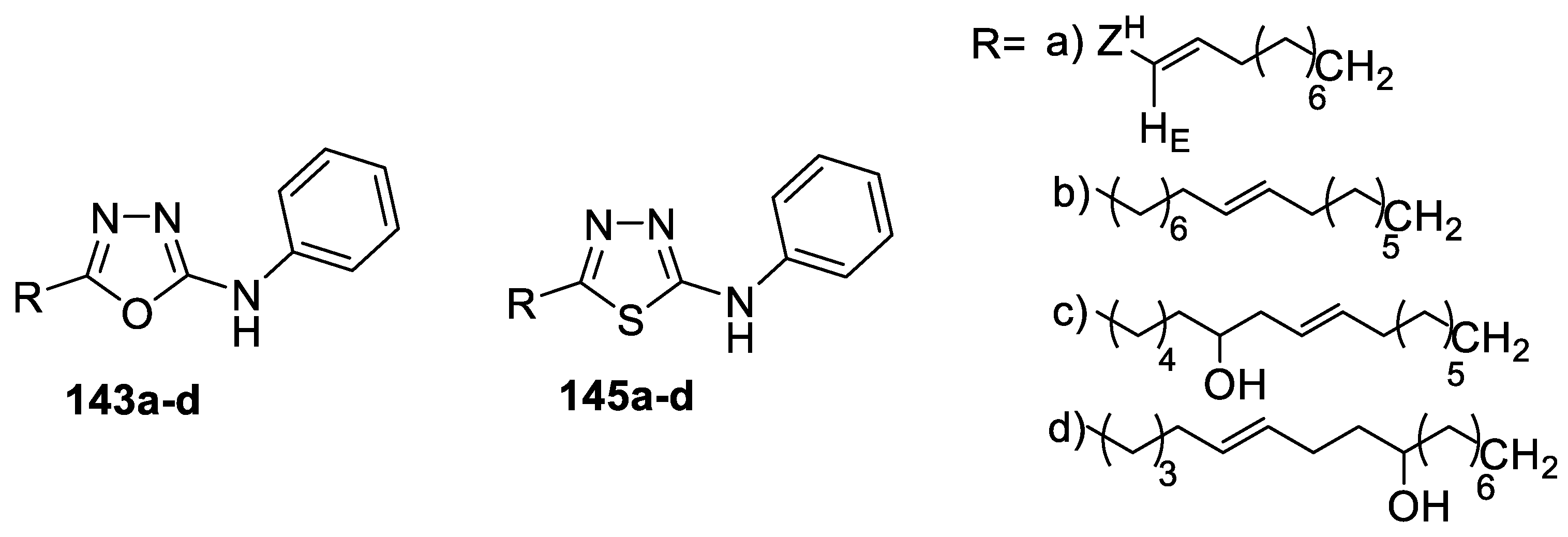
- Farshori, N.N.; Banday, M.R.; Ahmad, A.; Khan, A.U.; Rauf, A. Synthesis, characterization, and in vitro antimicrobial activities of 5-alkenyl/hydroxyalkenyl-2-phenylamine-1,3,4-oxadiazoles and thiadiazoles. Bioorg. Med. Chem. Lett. 2010, 20, 1933–1938. [Google Scholar] [CrossRef]
- Coruh, I.; Rollas, S.; Turan, S. Synthesis and evaluation of cytotoxic activities of some 1,4-disubstituted thiosemicarbazides, 2,5-disubstituted-1,3,4- thiadiazoles and 1,2,4-triazole-5-thiones derived from benzilic acid hydrazide. Marmara Pharm. J. 2012, 16, 56–63. [Google Scholar] [CrossRef]
- Burbuliene, M.; Simkus, A.; Vainilavicius, P. Synthesis of new pyrimidinylthio-substituted 1,3,4-oxa(thia)diazoles and 1,2,4-triazoles. J. Sulfur Chem. 2012, 33, 403–411. [Google Scholar] [CrossRef]
- Szulczyk, D.; Tomaszewski, P.; Michał, J.; Kozioł, A.E.; Lis, T.; Collu, D.; Iuliano, F.; Struga, M. Synthesis and biological activities of ethyl 2-(2-pyridylacetate) derivatives containing thiourea, 1,2,4-triazole, thiadiazole and oxadiazole moieties. Molecules 2017, 22, 409. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, R.; Karale, B.; Akolkar, H.; Randhavane, P. Synthesis and antibacterial screening of 1,3,4-thiadiazoles, 1,2,4-triazoles, and 1,3,4-oxadiazoles containing piperazine nucleus. J. Heterocycl. Chem. 2017, 54, 1355–1360. [Google Scholar] [CrossRef]
- Abdo, N.Y.M.; Kamel, M.M. Synthesis and anticancer evaluation of 1,3,4-oxadiazoles, 1,3,4-thiadiazoles, 1,2,4-triazoles and mannich bases. Chem. Pharm. Bull. 2015, 63, 369–376. [Google Scholar] [CrossRef]
- Reddy, K.R.; Mamatha, R.; Babu, M.S.S.; Kumar, K.S.; Jayaveera, K.N.; Narayanaswamy, G. Synthesis and antimicrobial activities of some triazole, thiadiazole, and oxadiazole substituted coumarins. J. Hetercyclic Chem. 2013, 51, 132–137. [Google Scholar] [CrossRef]
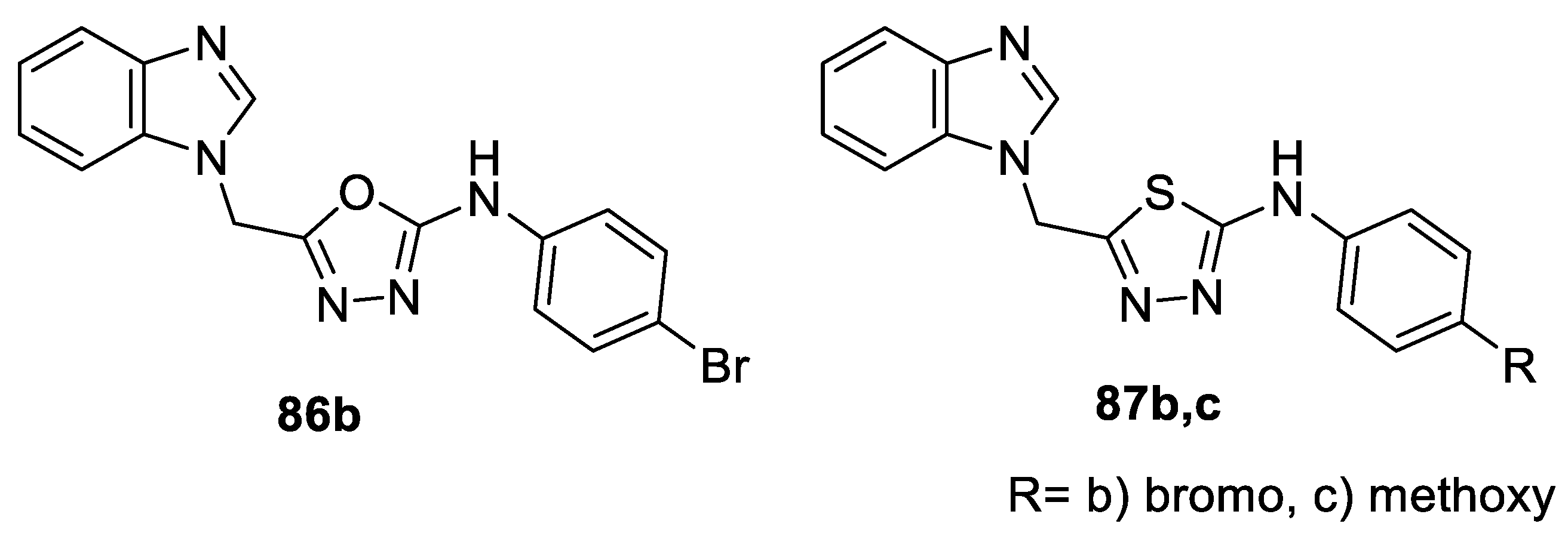
- Abdel-fattah, H.A.; El-Etrawy, A.S.; Gabr, N.M.R. Synthesis and biological evaluation of some new 1,3,4-oxa, thiadiazole and 1,2,4-triazole derivatives attached to benzimidazole. Int. J. Pharm. Chem. 2014, 4, 112–118. [Google Scholar] [CrossRef]
- El-essawy, F.A.; El-sayed, W.A. Synthesis of new 1,3,4-oxadiazol, thiadiazole, 1,2,4-triazole, and arylidene hydrazide derivatives of 4-oxo-1,4-dihydroquinoline with antimicrobial evaluation. J. Heterocycl. Chem. 2013, 50, 1–8. [Google Scholar] [CrossRef]
- Andrews, B.; Ahmed, M. Novel synthesis and characterization of some pyrimidine derivatives of oxadiazoles, triazole and 1,3,4-thiadiazoles. Asian J. Chem. 2013, 25, 2070–2072. [Google Scholar] [CrossRef]
- Kalhor, M.; Dadras, A. Synthesis, characterization, and herbicidal activities of new 1,3,4-oxadiazoles, 1,3,4-thiadiazoles, and 1,2,4-triazoles derivatives bearing (r)-5-chloro-3-fluoro-2-phenoxypyridin. J. Heterocycl. Chem. 2013, 50, 220–224. [Google Scholar] [CrossRef]
- Zoumpoulakis, P.; Camoutsis, C.; Pairas, G.; Sokovic, M.; Glamoclija, J.; Potamitis, C.; Pitsas, A. Synthesis of novel sulfonamide-1,2,4-triazoles, 1,3,4-thiadiazoles and 1,3,4-oxadiazoles, as potential antibacterial and antifungal agents. Biological evaluation and conformational analysis studies. Bioorg. Med. Chem. 2012, 20, 1569–1583. [Google Scholar] [CrossRef] [PubMed]
- Rezki, N.; Al-Yahyawi, A.M.; Bardaweel, S.K.; Al-Blewi, F.F.; Aouad, M.R. Synthesis of novel 2,5-disubstituted-1,3,4-thiadiazoles clubbed 1,2,4-triazole, 1,3,4-thiadiazole, 1,3,4-oxadiazole and/or schiff base as potential antimicrobial and antiproliferative agents. Molecules 2015, 20, 16048–16067. [Google Scholar] [CrossRef]
- Upadhyay, P.K.; Mirsha, P. Design, synthesis and antifungal evaluation of novel substituted 1,3,4-oxadiazoles, and 1,3,4-thiadiazoles. Int. J. Pharm. Pharm. Sci. 2015, 7, 466–470. [Google Scholar]
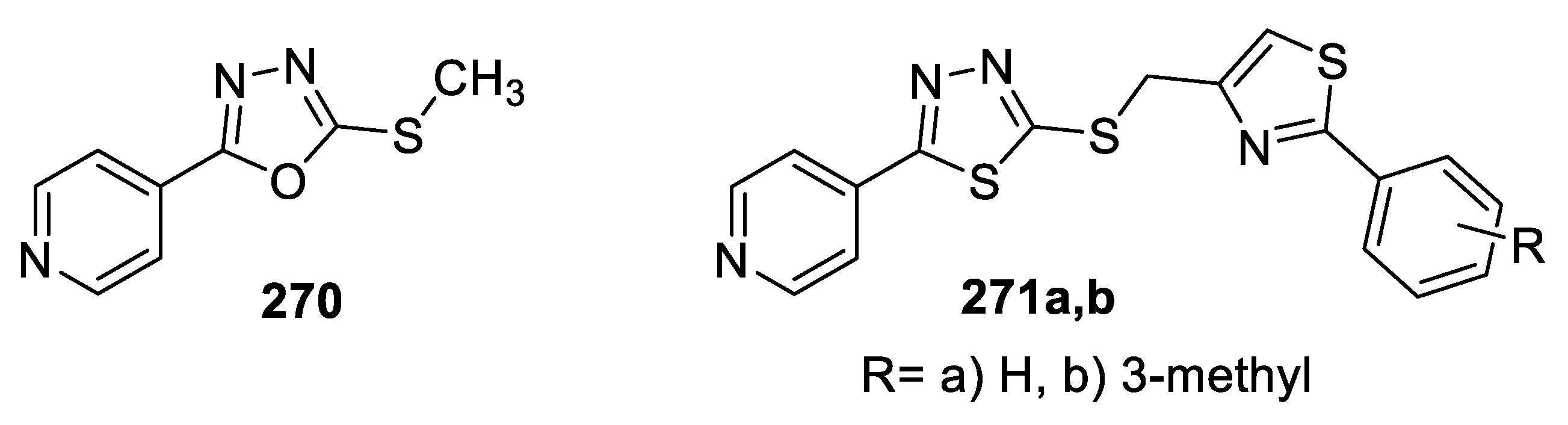
- Barbuceanu, S.; Ilies, D.C.; Radulescu, V.; Socea, L.; Draghici, C.; Saramet, G. Synthesis, Characterization and antioxidant activity evaluation of some 1,3,4-thiadiazole and 1,3,4-oxadiazole compounds. Rev. Chim. 2014, 65, 1172–1175. [Google Scholar]
- Barbuceanu, S.; Draghici, C.; Socea, L.; Enache, C.; Andreescu, C.; Saramet, G. Hydrazinecarbothioamides and 1,3,4-thia/oxadiazoles derivatives with potential biological activity synthesis and spectral characterization. Rev. Chim. 2015, 66, 1558–1562. [Google Scholar]
- El-Essawy, F.A.; Rady, S.I.M. Synthesis of some N-alkylated 1,2,4-triazoles, 1,3,4-oxadiazoles, and 1,3,4-thiadiazoles based on n-(furann-2-yl- methylidene)-4,6-dimethyl-1h-pyrazolo- [3,4-b]pyridine-3-amine. Chem. Heterocycl. Compd. 2011, 47, 497–501. [Google Scholar] [CrossRef]
- Ebrahimi, S. Synthesis of some pyridyl and cyclohexyl substituted 1,2,4 triazole, 1,3,4-thiadiazole and 1,3,4-oxadiazole derivatives. Eur. J. Chem. 2010, 4, 322–324. [Google Scholar] [CrossRef]
- Yang, S.; Lee, S.; Kwak, H.; Gong, Y. Regioselective synthesis of 2-amino-substituted 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives via reagent-based cyclization of thiosemicarbazide intermediate. J. Org. Chem. 2013, 78, 438–444. [Google Scholar] [CrossRef]
- Niu, P.; Kang, J.; Tian, X.; Song, L.; Liu, H.; Wu, J.; Yu, W.; Chang, J. Synthesis of 2-amino-1,3,4-oxadiazoles and 2-amino-1,3,4-thiadiazoles via sequential condensation and i2-mediated oxidative c-o/c-s bond formation. J. Org. Chem. 2015, 80, 1018–1024. [Google Scholar] [CrossRef]
- Renuka, N.; Vivek, H.K.; Pavithra, G.; Ajay Kumar, K. Synthesis of coumarin appended pyrazolyl-1,3,4-oxadiazoles and pyrazolyl-1,3,4-thiadiazoles: Evaluation of their in vitro antimicrobial and antioxidant activities and molecular docking studies. Russ. J. Bioorganic Chem. 2017, 43, 197–210. [Google Scholar] [CrossRef]
- Aksenov, N.A.; Arutiunov, N.A.; Kirillov, N.K.; Aksenov, D.A.; Aksenov, A.V.; Rubin, M. Preparation of 1,3,4-oxadiazoles and 1,3,4-thiadiazoles via chemoselective cyclocondensation of electrophilically activated nitroalkanes to (thio)semicarbazides or thiohydrazides. Chem. Heterocycl. Compd. 2020, 56, 1067–1072. [Google Scholar] [CrossRef]
- Kumar, S.; Srivastava, D.P. Efficient electrochemical synthesis, antimicrobial and antiinflammatory activity of 2–amino-5-substituted- 1,3,4-oxadiazole derivatives. Indian J. Pharm. Sci. 2010, 72, 458–464. [Google Scholar] [CrossRef]
- Tresse, C.; Radigue, R.; Gomes Von Borowski, R.; Thepaut, M.; Hanh Le, H.; Demay, F.; Georgeault, S.; Dhalluin, A.; Trautwetter, A.; Ermel, G.; et al. Synthesis and evaluation of 1,3,4-oxadiazole derivatives for development as broad-spectrum antibiotics. Bioorg. Med. Chem. 2019, 27, 115097. [Google Scholar] [CrossRef]
- Arafa, W.A.A.; Abdel-Magied, A.F. Utilization of ultrasonic irradiation as green and effective one-pot protocol to prepare a novel series of bis-2-amino-1,3,4-oxa(thia)diazoles and bis-tetrazoles. Arkivoc 2017, 2017, 327–340. [Google Scholar] [CrossRef]
- Gurunanjappa, P.; Kariyappa, A.K. Design, synthesis and biological evaluation of 1,3,4-oxadiazoles/thiadiazoles bearing pyrazole scaffold as antimicrobial and antioxidant candidates. Curr. Chem. Lett. 2016, 5, 109–122. [Google Scholar] [CrossRef]
- Szocs, B.; Tóth, M.; Docsa, T.; Gergely, P.; Somsák, L. Synthesis of 2-(β-d-glucopyranosyl)-5-(substituted-amino)-1,3,4-oxa- and -thiadiazoles for the inhibition of glycogen phosphorylase. Carbohydr. Res. 2013, 381, 187–195. [Google Scholar] [CrossRef]
- Lungu, L.; Ciocarlan, A.; Smigon, C.; Ozer, I.; Shova, S.; Gutu, I.; Vornicu, N.; Mangalagiu, I.; D’Ambrosio, M.; Aricu, A. Synthesis and evaluation of biological activity of homodrimane sesquiterpenoids bearing 1,3,4-oxadiazole or 1,3,4-thiadiazole units. Chem. Heterocycl. Compd. 2020, 56, 578–585. [Google Scholar] [CrossRef]
- Lin, H.; Qiao, Y.; Yang, H.; Li, Q.; Chen, Y.; Qu, W.; Liu, W.; Feng, F.; Sun, H. Design and evaluation of Nrf2 activators with 1,3,4-oxa/thiadiazole core as neuro-protective agents against oxidative stress in PC-12 cells. Bioorg. Med. Chem. Lett. 2020, 30, 126853. [Google Scholar] [CrossRef]
- Aggarwal, N.; Kumar, R.; Dureja, P.; Khurana, J.M. Synthesis of novel nalidixic acid-based 1,3,4-thiadiazole and 1,3,4-oxadiazole derivatives as potent antibacterial agents. Chem. Biol. Drug Des. 2012, 79, 384–397. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, Y.; Tang, Q.; Yuan, J.; Ran, D.; Tian, B.; Pan, T.; Gan, Z. TiCl4 mediated facile synthesis of 1,3,4-oxadiazoles and 1,3,4-thiadiazoles. Synth. Commun. 2020, 50, 423–431. [Google Scholar] [CrossRef]
- Fan, X.; Jiang, X.; Zhang, Y.; Chen, Z.; Zhu, Y. Palladium-catalyzed one-pot synthesis of diazoles via tert-butyl isocyanide insertion. Org. Biomol. Chem. 2015, 13, 10402–10408. [Google Scholar] [CrossRef] [PubMed]
- Dekhane, D.V.; Pawar, S.S.; Gupta, S.; Shingare, M.S.; Patil, C.R.; Thore, S.N. Synthesis and anti-inflammatory activity of some new 4,5-dihydro-1,5-diaryl-1h-pyrazole-3-substituted-heteroazole derivatives. Bioorg. Med. Chem. Lett. 2011, 21, 6527–6532. [Google Scholar] [CrossRef] [PubMed]
- Knyazyan, A.; Eliazyan, K.; Pivazyan, V.; Ghazaryan, E.; Harutyunyan, S.; Yengoyan, A. Synthesis and growth regulatory activity of novel 5-(3-alkyl-4-methyl-2-thioxo-2,3-dihydro-thiazol-5-Yl)-3H-[1,3,4] thiadiazole(oxadiazole)-2-thiones and their derivatives. Heterocycl. Commun. 2012, 18, 103–108. [Google Scholar] [CrossRef]
- Yadagiri, B.; Gurrala, S.; Bantu, R.; Nagarapu, L.; Polepalli, S.; Srujana, G.; Jain, N. Synthesis and evaluation of benzosuberone embedded with 1,3,4-oxadiazole, 1,3,4-thiadiazole and 1,2,4-triazole moieties as new potential anti proliferative agents. Bioorg. Med. Chem. Lett. 2015, 25, 2220–2224. [Google Scholar] [CrossRef]
- Padmavathi, V.; Reddy, G.D.; Reddy, S.N.; Mahesh, K. Synthesis and biological activity of 2-(bis((1,3,4-oxadiazolyl/1,3,4-thiadiazolyl) methylthio)methylene)malononitriles. Eur. J. Med. Chem. 2011, 46, 1367–1373. [Google Scholar] [CrossRef]
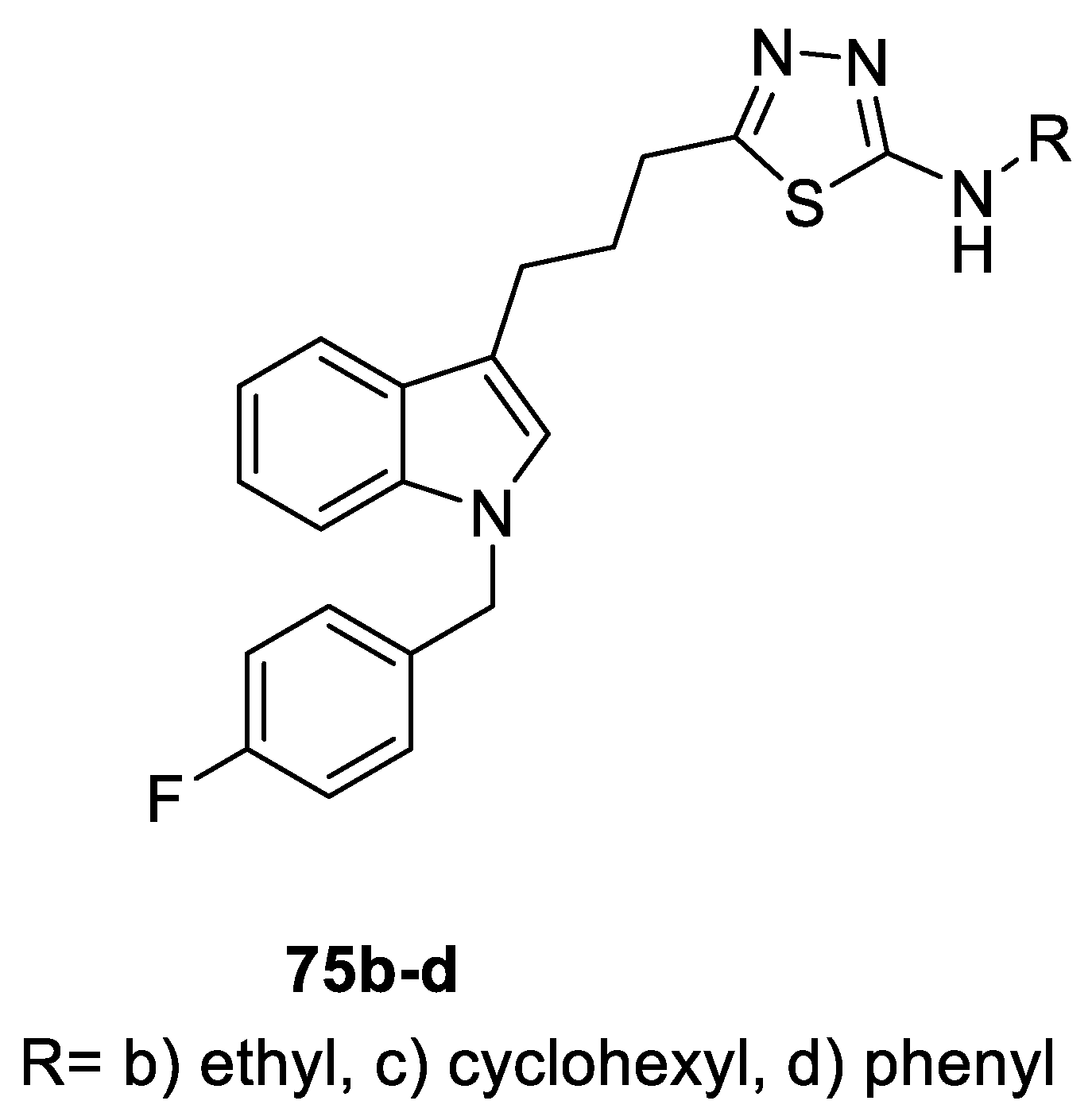
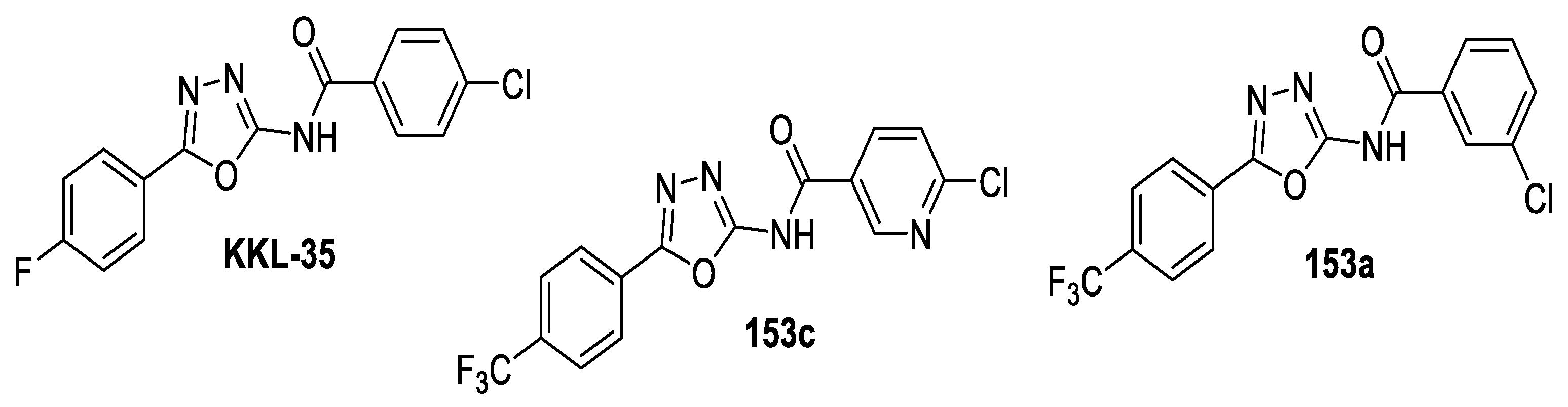
- Ramadoss, N.S.; Alumasa, J.N.; Cheng, L.; Wang, Y.; Li, S.; Chambers, B.S.; Chang, H.; Chatterjee, A.K.; Brinker, A.; Engels, I.H.; et al. Small molecule inhibitors of trans-translation have broad-spectrum antibiotic activity. Proc. Natl. Acad. Sci. USA 2013, 110, 10282–10287. [Google Scholar] [CrossRef] [PubMed]
- Karabanovich, G.; Zemanová, J.; Smutný, T.; Székely, R.; Šarkan, M.; Centárová, I.; Vocat, A.; Pávková, I.; Čonka, P.; Němeček, J.; et al. Development of 3,5-dinitrobenzylsulfanyl-1,3,4-oxadiazoles and thiadiazoles as selective antitubercular agents active against replicating and nonreplicating mycobacterium tuberculosis. J. Med. Chem. 2016, 59, 2362–2380. [Google Scholar] [CrossRef]
- Al-Azzawi, A.M.; Hamd, A.S. Synthesis, characterization and antimicrobial activity evaluation of new cyclic iimides containing 1,3,4-thiadiazole and 1,3,4- oxadiazole moieties. Int. J. Res. Pharm. Chem. 2013, 3, 2231–2781. [Google Scholar]
- Maddila, S.; Jonnalagadda, S.B. Synthesis and antimicrobial activity of new 1,3,4-thiadiazoles containing oxadiazole, thiadiazole and triazole nuclei. Pharm. Chem. J. 2013, 46, 661–666. [Google Scholar] [CrossRef]
- Madhu Sekhar, M.; Nagarjuna, U.; Padmavathi, V.; Padmaja, A.; Reddy, N.V.; Vijaya, T. Synthesis and antimicrobial activity of pyrimidinyl 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazoles. Eur. J. Med. Chem. 2018, 145, 1–10. [Google Scholar] [CrossRef]
- Xu, W.M.; Li, S.Z.; He, M.; Yang, S.; Li, X.Y.; Li, P. Synthesis and bioactivities of novel thioether/sulfone derivatives containing 1,2,3-thiadiazole and 1,3,4-oxadiazole/thiadiazole moiety. Bioorg. Med. Chem. Lett. 2013, 23, 5821–5824. [Google Scholar] [CrossRef]
- Rajak, H.; Agarawal, A.; Parmar, P.; Thakur, B.S.; Veerasamy, R.; Sharma, P.C.; Kharya, M.D. 2,5-disubstituted-1,3,4-oxadiazoles/thiadiazole as surface recognition moiety: Design and synthesis of novel hydroxamic acid based histone deacetylase inhibitors. Bioorg. Med. Chem. Lett. 2011, 21, 5735–5738. [Google Scholar] [CrossRef]
- Dawood, K.M.; Gomha, S.M. Synthesis and anti-cancer activity of 1,3,4-thiadiazole and 1,3-thiazole derivatives having 1,3,4-oxadiazole moiety. J. Heterocycl. Chem. 2015, 52, 1400–1405. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, P.; Xuan, L.N.; Fu, X.Y.; Jing, F.; Li, S.; Liu, Y.M.; Chen, B.Q. Synthesis and antitumor activities of novel hybrid molecules containing 1,3,4-oxadiazole and 1,3,4-thiadiazole bearing schiff base moiety. Bioorg. Med. Chem. Lett. 2014, 24, 5154–5156. [Google Scholar] [CrossRef]
- Özdemir, A.; Sever, B.; Altıntop, M.D.; Temel, H.E.; Atlı, Ö.; Baysal, M.; Demirci, F. Synthesis and evaluation of new oxadiazole, thiadiazole, and triazole derivatives as potential anticancer agents targeting MMP-9. Molecules 2017, 22, 1109. [Google Scholar] [CrossRef] [PubMed]
- Toma, A.; Hapău, D.; Vlase, L.; Mogoşan, C.; Zaharia, V. Synthesis and anti-inflammatory activity of 5-(pyridin-4-yl)-1,3,4-oxadiazole-2-thiol, 5-(pyridin-4-yl)-1,3,4-thiadiazole-2-thiol and 5-(pyridin-4-yl)-1,2,4-triazole-3-thiol derivatives. Clujul Med. 2013, 86, 34–39. [Google Scholar]







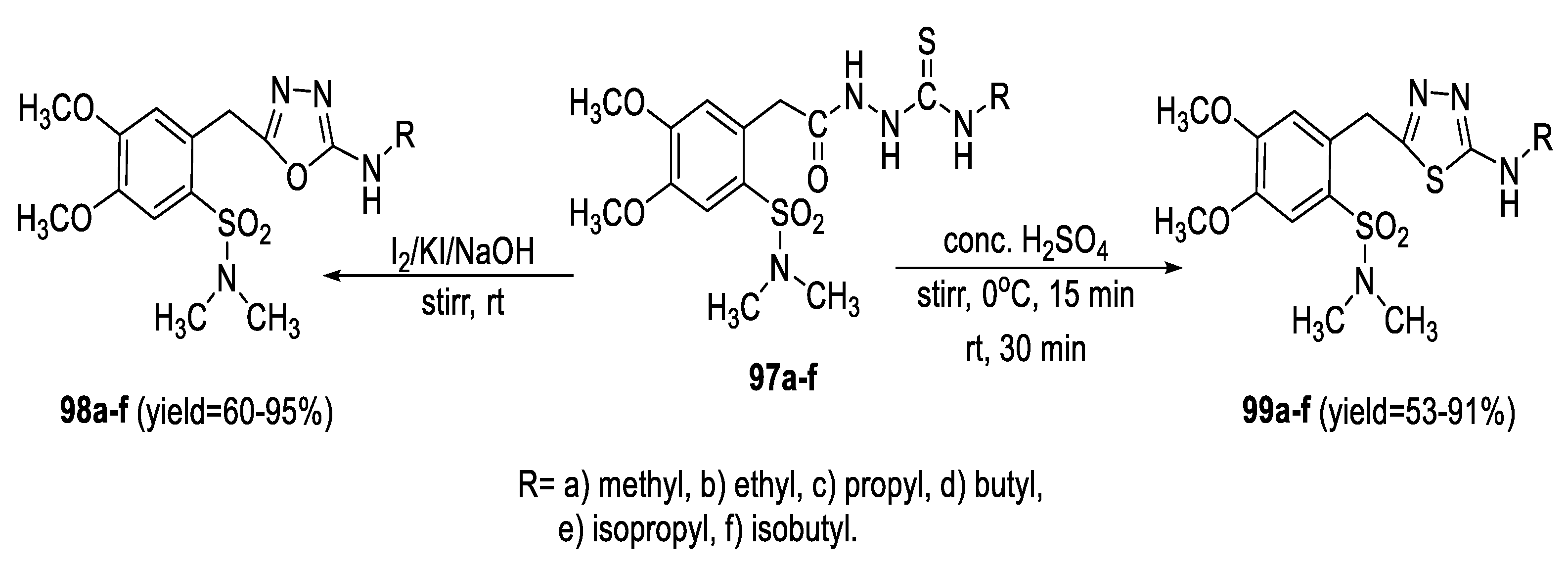
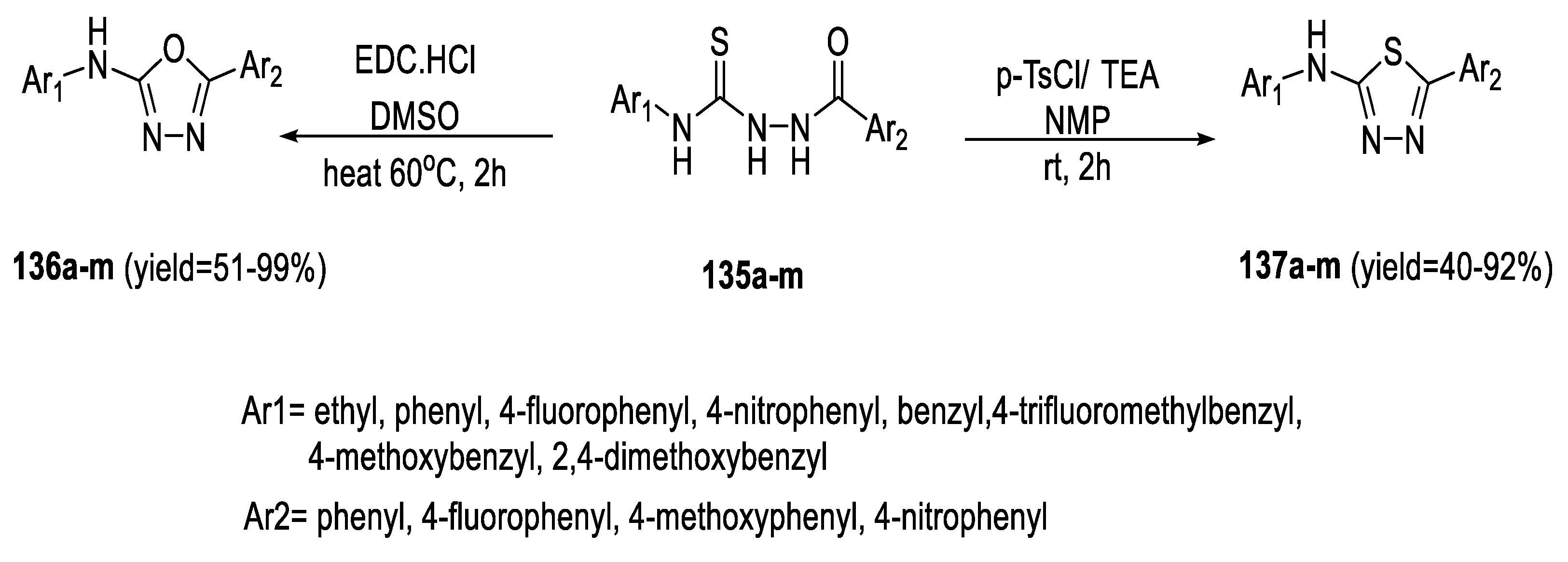





















Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Masry, R.M.; Kadry, H.H.; Taher, A.T.; Abou-Seri, S.M. Comparative Study of the Synthetic Approaches and Biological Activities of the Bioisosteres of 1,3,4-Oxadiazoles and 1,3,4-Thiadiazoles over the Past Decade. Molecules 2022, 27, 2709. https://doi.org/10.3390/molecules27092709
El-Masry RM, Kadry HH, Taher AT, Abou-Seri SM. Comparative Study of the Synthetic Approaches and Biological Activities of the Bioisosteres of 1,3,4-Oxadiazoles and 1,3,4-Thiadiazoles over the Past Decade. Molecules. 2022; 27(9):2709. https://doi.org/10.3390/molecules27092709
Chicago/Turabian StyleEl-Masry, Rana M., Hanan H. Kadry, Azza T. Taher, and Sahar M. Abou-Seri. 2022. "Comparative Study of the Synthetic Approaches and Biological Activities of the Bioisosteres of 1,3,4-Oxadiazoles and 1,3,4-Thiadiazoles over the Past Decade" Molecules 27, no. 9: 2709. https://doi.org/10.3390/molecules27092709
APA StyleEl-Masry, R. M., Kadry, H. H., Taher, A. T., & Abou-Seri, S. M. (2022). Comparative Study of the Synthetic Approaches and Biological Activities of the Bioisosteres of 1,3,4-Oxadiazoles and 1,3,4-Thiadiazoles over the Past Decade. Molecules, 27(9), 2709. https://doi.org/10.3390/molecules27092709





