Psychedelics: Alternative and Potential Therapeutic Options for Treating Mood and Anxiety Disorders
Abstract
1. Introduction
1.1. Change in Status Quo
1.2. Classification of Psychedelic Drugs
1.3. Treating Mood and Anxiety Disorders with Psychedelic Drugs
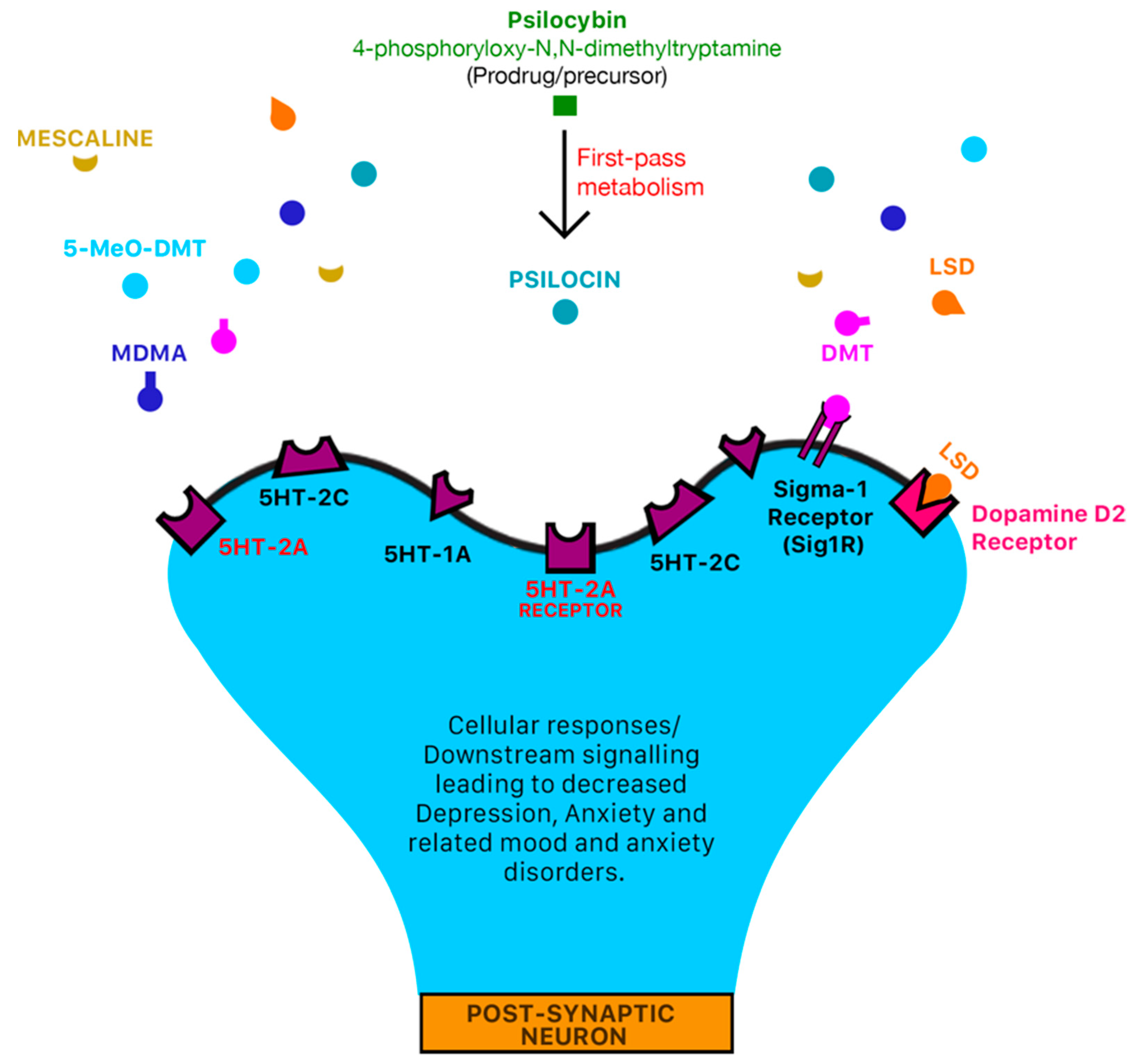
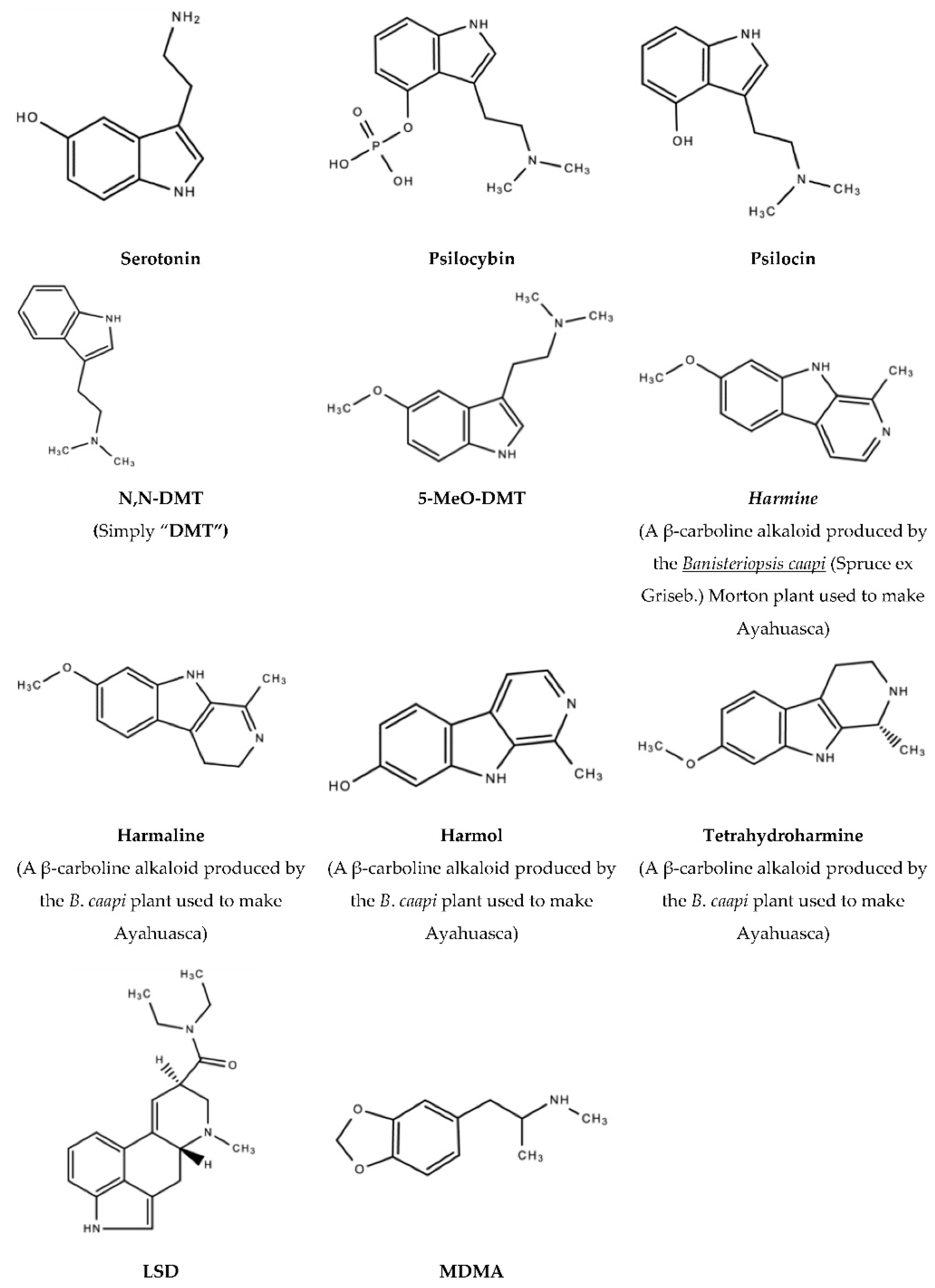
1.4. Psilocybin
1.5. N,N-DMT/DMT (The “God/Spirit Molecule”)
1.6. Ayahuasca
1.7. Ayahuasca Tourism
1.8. 5-MeO-DMT (Popularly Referred to as “Toad Venom”)
1.9. Lysergic Acid Diethylamide (LAD/LSD)
1.10. 3,4-Methylenedioxymethamphetamine (MDMA/“Ecstasy”)
2. The Economic Value of Psychedelics
3. Conclusions and Future Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Rucker, J.J.; Iliff, J.; Nutt, D.J. Psychiatry & the psychedelic drugs. past, present & future. Neuropharmacology 2018, 142, 200–218. [Google Scholar] [CrossRef] [PubMed]
- Kelley, A. Americans are Taking More Anti-Anxiety Medication and Antidepressants during CORONAVIRUS Pandemic: Report. 16 April 2020. Available online: https://thehill.com/changing-america/well-being/mental-health/493125-increase-in-anti-anxiety-medication-antidepressants (accessed on 18 February 2021).
- Harvard Health Publishing. What Are the Real Risks of Antidepressants? March 2014. Available online: https://www.health.harvard.edu/mind-and-mood/what-are-the-real-risks-of-antidepressants (accessed on 18 February 2021).
- Mayo Clinic Staff. The Most Commonly Prescribed Type of Antidepressant. 17 September 2019. Available online: https://www.mayoclinic.org/diseases-conditions/depression/in-depth/ssris/art-20044825 (accessed on 18 February 2021).
- Gable, R.S. Risk assessment of ritual use of oral dimethyltryptamine (DMT) and harmala alkaloids. Addiction 2007, 102, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.W.; Griffiths, R.R. Potential Therapeutic Effects of Psilocybin. Neurotherapeutics 2017, 14, 734–740. [Google Scholar] [CrossRef]
- Passie, T.; Seifert, J.; Schneider, U.; Emrich, H.M. The pharmacology of Psilocybin. Addict. Biol. 2002, 7, 357–364. [Google Scholar] [CrossRef]
- Oliveira e Souza, R. COVID-19 Pandemic Exacerbates Suicide Risk Factors. 10 September 2020. Available online: https://www.paho.org/en/news/10-9-2020-covid-19-pandemic-exacerbates-suicide-risk-factors (accessed on 16 January 2021).
- Griffiths, R.R.; Richards, W.A.; McCann, U.; Jesse, R. Psilocybin can occasion mystical-type experiences having substantial and sustained personal meaning and spiritual significance. Psychopharmacology 2006, 187, 268–292. [Google Scholar] [CrossRef]
- Mahapatra, A.; Gupta, R. Role of Psilocybin in the treatment of depression. Ther. Adv. Psychopharmacol. 2016, 7, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Halpern, J.H.; Pope, H.G., Jr. Do hallucinogens cause residual neuropsychological toxicity? Drug Alcohol Depend. 1999, 53, 247–256. [Google Scholar] [CrossRef]
- Lim, A. Psychedelics as Antidepressants. 30 January 2021. Available online: https://www.scientificamerican.com/article/psychedelics-as-antidepressants/ (accessed on 18 February 2021).
- Marona-Lewicka, D.; Thisted, R.A.; Nichols, D.E. Distinct temporal phases in the behavioral pharmacology of LSD: Dopamine D2 receptor-mediated effects in the rat and implications for psychosis. Psychopharmacology 2005, 180, 427–435. [Google Scholar] [CrossRef]
- Geyer, M.; Vollenweider, F. Serotonin research: Contributions to understanding psychoses. Trends Pharmacol. Sci. 2008, 29, 445–453. [Google Scholar] [CrossRef]
- Aghajanian, G.K.; Haigler, H.J.; Bloom, F.E. Lysergic acid diethylamide and serotonin: Direct actions on serotonin-containing neurons in rat brain. Life Sci. 1972, 11, 615–622. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information PubChem Compound Summary for CID 1150, Tryptamine. 2021. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Tryptamine (accessed on 16 February 2021).
- Halberstadt, A.L.; Geyer, M.A. Multiple receptors contribute to the behavioral effects of indoleamine hallucinogens. Neuropharmacology 2011, 61, 364–381. [Google Scholar] [CrossRef]
- Beliveau, V.; Ganz, M.; Feng, L.; Ozenne, B.; Højgaard, L.; Fisher, P.M.; Svarer, C.; Greve, D.N.; Knudsen, G.M. A High-Resolution In Vivo Atlas of the Human Brain’s Serotonin System. J. Neurosci. Off. J. Soc. Neurosci. 2017, 37, 120–128. [Google Scholar] [CrossRef]
- Nichols, D.E.; Nichols, C.D. Serotonin receptors. Chem. Rev. 2008, 108, 1614–1641. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, D.; Clarke, D.E.; Fozard, J.R.; Hartig, P.R.; Martin, G.R.; Mylecharane, E.J.; Saxena, P.R.; Humphrey, P.P. International Union of Pharmacology classification of receptors for 5-hydroxytryptamine (Serotonin). Pharmacol. Rev. 1994, 46, 157–203. [Google Scholar]
- Frazer, A.; Hensler, J.G. Serotonin Involvement in Physiological Function and Behavior. Basic Neurochemistry: Molecular, Cellular and Medical Aspects, 6th ed.; Lippincott-Raven: Philadelphia, PA, USA, 1999. Available online: https://www.ncbi.nlm.nih.gov/books/NBK27940/ (accessed on 9 March 2021).
- Su, T.P.; Hayashi, T.; Vaupel, D.B. When the endogenous hallucinogenic trace amine N,N-dimethyltryptamine meets the sigma-1 receptor. Sci. Signal. 2009, 2, pe12. [Google Scholar] [CrossRef] [PubMed]
- Fontanilla, D.; Johannessen, M.; Hajipour, A.R.; Cozzi, N.V.; Jackson, M.B.; Ruoho, A.E. The hallucinogen N,N-dimethyltryptamine (DMT) is an endogenous sigma-1 receptor regulator. Science 2009, 323, 934–937. [Google Scholar] [CrossRef] [PubMed]
- Canal, C.E. Serotonergic Psychedelics: Experimental Approaches for Assessing Mechanisms of Action. Handb. Exp. Pharmacol. 2018, 252, 227–260. [Google Scholar] [CrossRef]
- Berry, M.D. The potential of trace amines and their receptors for treating neurological and psychiatric diseases. Rev. Recent Clin. Trials 2007, 2, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.D.; Gainetdinov, R.R.; Hoener, M.C.; Shahid, M. Pharmacology of human trace amine-associated receptors: Therapeutic opportunities and challenges. Pharmacol. Ther. 2017, 180, 161–180. [Google Scholar] [CrossRef]
- Gainetdinov, R.R.; Hoener, M.C.; Berry, M.D. Trace Amines and Their Receptors. Pharmacol. Rev. 2018, 70, 549–620. [Google Scholar] [CrossRef]
- Simmler, L.D.; Buchy, D.; Chaboz, S.; Hoener, M.C.; Liechti, M.E. In Vitro Characterization of Psychoactive Substances at Rat, Mouse, and Human Trace Amine-Associated Receptor 1. J. Pharmacol. Exp. Ther. 2016, 357, 134–144. [Google Scholar] [CrossRef]
- Nichols, D.E. Psychedelics. Pharmacol. Rev. 2016, 68, 264–355. [Google Scholar] [CrossRef] [PubMed]
- Albert, P.R.; Benkelfat, C.; Descarries, L. The neurobiology of depression--revisiting the serotonin hypothesis. I. Cellular and molecular mechanisms. Philos. Trans. R. Soc. London Ser. B Biol. Sci. 2012, 367, 2378–2381. [Google Scholar] [CrossRef]
- Lowe, H.; Toyang, N.; Steele, B.; Valentine, H.; Grant, J.; Ali, A.; Ngwa, W.; Gordon, L. The Therapeutic Potential of Psilocybin. Molecules 2021, 26, 2948. [Google Scholar] [CrossRef] [PubMed]
- Palhano-Fontes, F.; Barreto, D.; Onias, H.; Andrade, K.C.; Novaes, M.M.; Pessoa, J.A.; Mota-Rolim, S.A.; Osório, F.L.; Sanches, R.; dos Santos, R.G. Rapid antidepressant effects of the psychedelic Ayahuasca in treatment-resistant depression: A randomized placebo-controlled trial. Psychol. Med. 2019, 49, 655–663. [Google Scholar] [CrossRef]
- Palhano-Fontes, F.; Alchieri, J.C.; Oliveira, J.P.M.; Soares, B.L.; Hallak, J.E.C.; Galvao-Coelho, N.; de Araujo, D.B. The Therapeutic Potentials of Ayahuasca in the Treatment of Depression. In The Therapeutic Use of Ayahuasca; Labate, B., Cavnar, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar] [CrossRef]
- Dos Santos, R.G.; Sanches, R.F.; Osório, F.D.; Hallak, C.J.E. Long-term effects of Ayahuasca in patients with recurrent depression: A 5-year qualitative follow-up. Arch. Clin. Psychiatry (São Paulo) 2018, 45, 22–24. [Google Scholar] [CrossRef]
- Dos Santos, R.G.; Osório, F.L.; Crippa, J.A.; Riba, J.; Zuardi, A.W.; Hallak, J.E. Antidepressive, anxiolytic, and antiaddictive effects of Ayahuasca, Psilocybin and lysergic acid diethylamide (LSD): A systematic review of clinical trials published in the last 25 years. Ther. Adv. Psychopharmacol. 2016, 6, 193–213. [Google Scholar] [CrossRef] [PubMed]
- Osório, F.; Sanches, R.F.; Macedo, L.R.; Santos, R.G.; Maia-de-Oliveira, J.P.; Wichert-Ana, L.; Araujo, D.B.; Riba, J.; Crippa, J.A.; Hallak, J.E. Antidepressant effects of a single dose of Ayahuasca in patients with recurrent depression: A preliminary report. Rev. Bras. Psiquiatr. 2015, 37, 13–20. [Google Scholar] [CrossRef]
- Johnson, F.G. LSD in the treatment of alcoholism. Am. J. Psychiatry 1969, 126, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.; Levine, J.; Stark, L.; Lazar, R. A clinical study of LSD treatment in alcoholism. Am. J. Psychiatry 1969, 126, 59–69. [Google Scholar] [CrossRef]
- Smart, R.G.; Storm, T. The Efficacy of LSD in the Treatment of Alcoholism. Q. J. Stud. Alcohol 1964, 25, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Visotsky, H.M. LSD and alcoholism: A clinical study of treatment efficacy. J. Chronic Dis. 1971, 24, 597. [Google Scholar] [CrossRef]
- Dahlberg, C.C. Lysergic Acid Diethylamide (LSD) in the Treatment of Alcoholism. Arch. Gen. Psychiatry 1968, 19, 508. [Google Scholar] [CrossRef]
- Simmons, J.Q.; Benor, D.; Daniel, D. The variable effects of LSD-25 on the behavior of a heterogeneous group of childhood schizophrenics. Behav. Neuropsychiatry 1972, 4, 10–24. [Google Scholar] [PubMed]
- Ball, J.R.; Armstrong, J.J. The use of L.S.D. 25 (D-lysergic acid diethylamide) in the treatment of the sexual perversions. Can. Psychiatr. Assoc. J. 1961, 6, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Sigafoos, J.; Green, V.A.; Edrisinha, C.; Lancioni, G.E. Flashback to the 1960s: LSD in the treatment of autism. Dev. Neurorehabilit. 2007, 10, 75–81. [Google Scholar] [CrossRef]
- Zamaria, J.A. A Phenomenological Examination of Psilocybin and its Positive and Persisting Aftereffects. NeuroQuantology 2016, 14. [Google Scholar] [CrossRef]
- Mithoefer, M.C.; Grob, C.S.; Brewerton, T.D. Novel psychopharmacological therapies for psychiatric disorders: Psilocybin and MDMA. Lancet Psychiatry 2016, 3, 481–488. [Google Scholar] [CrossRef]
- Sessa, B.; Sakal, C.; O’Brien, S.; Nutt, D. First study of safety and tolerability of 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy in patients with alcohol use disorder: Preliminary data on the first four participants. BMJ Case Rep. 2019, 12, e230109. [Google Scholar] [CrossRef]
- Zarley, B.D. First-of-Its-Kind Pilot Study Uses MDMA for Alcohol Addiction. 28 February 2021. Available online: https://www.freethink.com/articles/mdma-for-alcohol-addiction (accessed on 10 March 2021).
- Ot’alora, G.M.; Grigsby, J.; Poulter, B.; Van Derveer, J.W., 3rd; Giron, S.G.; Jerome, L.; Feduccia, A.A.; Hamilton, S.; Yazar-Klosinski, B.; Emerson, A.; et al. 3,4-Methylenedioxymethamphetamine-assisted psychotherapy for treatment of chronic posttraumatic stress disorder: A randomized phase 2 controlled trial. J. Psychopharmacol. 2018, 32, 1295–1307. [Google Scholar] [CrossRef]
- Davis, A.K.; Barsuglia, J.P.; Lancelotta, R.; Grant, R.M.; Renn, E. The epidemiology of 5-methoxy-N, N-dimethyltryptamine (5-MeO-DMT) use: Benefits, consequences, patterns of use, subjective effects, and reasons for consumption. J. Psychopharmacol. 2018, 32, 779–792. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.K.; So, S.; Lancelotta, R.; Barsuglia, J.P.; Griffiths, R.R. 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT) used in a naturalistic group setting is associated with unintended improvements in depression and anxiety. Am. J. Drug Alcohol Abus. 2019, 45, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Lima da Cruz, R.V.; Moulin, T.C.; Petiz, L.L.; Leão, R.N. A Single Dose of 5-MeO-DMT Stimulates Cell Proliferation, Neuronal Survivability, Morphological and Functional Changes in Adult Mice Ventral Dentate Gyrus. Front. Mol. Neurosci. 2018, 11, 312. [Google Scholar] [CrossRef] [PubMed]
- Lancelotta, R.L.; Davis, A.K. Use of Benefit Enhancement Strategies among 5-Methoxy-N,N-Dimethyltryptamine (5-MeO-DMT) Users: Associations with Mystical, Challenging, and Enduring Effects. J. Psychoact. Drugs 2020, 52, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Sherwood, A.M.; Claveau, R.; Lancelotta, R.; Kaylo, K.W.; Lenoch, K. Synthesis and Characterization of 5-MeO-DMT Succinate for Clinical Use. ACS Omega 2020, 5, 32067–32075. [Google Scholar] [CrossRef] [PubMed]
- Bahji, A.; Forsyth, A.; Groll, D.; Hawken, E.R. Efficacy of 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy for posttraumatic stress disorder: A systematic review and meta-analysis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2020, 96, 109735. [Google Scholar] [CrossRef]
- Dos Santos, R.G.; Osório, F.L.; Rocha, J.M.; Rossi, G.N.; Bouso, J.C.; Rodrigues, L.S.; de Oliveira Silveira, G.; Yonamine, M.; Hallak, J. Ayahuasca Improves Self-perception of Speech Performance in Subjects with Social Anxiety Disorder: A Pilot, Proof-of-Concept, Randomized, Placebo-Controlled Trial. J. Clin. Psychopharmacol. 2021. [Google Scholar] [CrossRef]
- Leonard, J. Ayahuasca: What It Is, Effects, and Usage (1182927539 885508185 Warwick, K.W., Ed.). 31 January 2021. Available online: https://www.medicalnewstoday.com/articles/Ayahuasca (accessed on 7 February 2021).
- Eisner, B. Set, setting, and matrix. J. Psychoact. Drugs 1997, 29, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Tupper, K.W. The globalization of Ayahuasca: Harm reduction or benefit maximization? Int. J. Drug Policy 2008, 19, 297–303. [Google Scholar] [CrossRef]
- Scaccia, A. What Psychedelics Really Do to Your Brain. Rolling Stone. 25 June 2018. Available online: https://www.rollingstone.com/culture/culture-news/what-psychedelics-really-do-to-your-brain-112948/ (accessed on 10 March 2021).
- Stafford, P.G.; Bigwood, J. Psychedelics Encyclopedia; Ronin Publishing: Berkeley, CA, USA, 2013. [Google Scholar]
- Inaba, D.; Cohen, W.E. Uppers, Downers, All Arounders: Physical and Mental Effects of Psychoactive Drugs, 7th ed.; CNS Productions: Medford, OR, USA, 2014. [Google Scholar]
- Nichols, D.E. Psilocybin: From ancient magic to modern medicine. J. Antibiot. 2020, 73, 679–686. [Google Scholar] [CrossRef]
- Hollister, L.E. Some general thoughts about endogenous psychotogens. In Neuroregulators and Psychiatric Disorders; Usdin, E., Hamburg, D.A., Barchassca, J.D., Eds.; Oxford University Press: New York, NY, USA, 1977; pp. 550–556. [Google Scholar]
- Saavedra, J.M.; Axelrod, J. Psychotomimetic N-methylated tryptamines: Formation in brain in vivo and in vitro. Science 1972, 175, 1365–1366. [Google Scholar] [CrossRef]
- Checkley, S.A.; Murray, R.M.; Oon, M.C.; Rodnight, R.; Birley, J.L. A longitudinal study of urinary excretion of N,N,-dimethyltryptamine in psychotic patients. Br. J. Psychiatry J. Ment. Sci. 1980, 137, 236–239. [Google Scholar] [CrossRef]
- Dean, J.; Liu, T.; Huff, S.; Sheler, B.; Barker, S.A.; Strassman, R.J.; Wang, M.M.; Borjigin, J. Biosynthesis and Extracellular Concentrations of N,N-dimethyltryptamine (DMT) in Mammalian Brain. Sci. Rep. 2019, 9, 9333. [Google Scholar] [CrossRef] [PubMed]
- Gillin, J.C.; Kaplan, J.; Stillman, R.; Wyatt, R.J. The psychedelic model of schizophrenia: The case of N,N-dimethyltryptamine. Am. J. Psychiatry 1976, 133, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.S.; Presti, D.E. Endogenous psychoactive tryptamines reconsidered: An anxiolytic role for dimethyltryptamine. Med. Hypotheses 2005, 64, 930–937. [Google Scholar] [CrossRef]
- Murray, R.M.; Oon, M.C.; Rodnight, R.; Birley, J.L.; Smith, A. Increased excretion of dimethyltryptamine and certain features of psychosis: A possible association. Arch. Gen. Psychiatry 1979, 36, 644–649. [Google Scholar] [CrossRef]
- Pomilio, A.B.; Vitale, A.A.; Ciprian-Ollivier, J.; Cetkovich-Bakmas, M.; Gómez, R.; Vázquez, G. Ayahoasca: An experimental psychosis that mirrors the transmethylation hypothesis of schizophrenia. J. Ethnopharmacol. 1999, 65, 29–51. [Google Scholar] [CrossRef]
- Grammenos, D.; Barker, S.A. On the transmethylation hypothesis: Stress, N,N-dimethyltryptamine, and positive symptoms of psychosis. J. Neural Transm. 2015, 122, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Smythies, J.R. The transmethylation hypotheses of schizophrenia re-evaluated. Trends Neurosci. 1984, 7, 45–47. [Google Scholar] [CrossRef]
- Marazziti, D. Understanding the role of serotonin in psychiatric diseases. F1000Research 2017, 6, 180. [Google Scholar] [CrossRef]
- Strassman, R.J. Human psychopharmacology of N,N-dimethyltryptamine. Behav. Brain Res. 1996, 73, 121–124. [Google Scholar] [CrossRef]
- Gouzoulis-Mayfrank, E.; Heekeren, K.; Neukirch, A.; Stoll, M.; Stock, C.; Obradovic, M.; Kovar, K.A. Psychological effects of (S)-ketamine and N,N-dimethyltryptamine (DMT): A double-blind, cross-over study in healthy volunteers. Pharmacopsychiatry 2005, 38, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Paterson, N.E.; Darby, W.C.; Sandhu, P.S. N,N-Dimethyltryptamine-Induced Psychosis. Clin. Neuropharmacol. 2015, 38, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Strassman, R.J.; Qualls, C.R. Dose-response study of N,N-dimethyltryptamine in humans. I. Neuroendocrine, autonomic, and cardiovascular effects. Arch. Gen. Psychiatry 1994, 51, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Dmt. 26 June 2015. Available online: https://www.release.org.uk/drugs/dmt/pharmacology (accessed on 7 February 2021).
- McKenna, D.J.; Towers, G.H.; Abbott, F. Monoamine oxidase inhibitors in South American hallucinogenic plants: Tryptamine and beta-carboline constituents of Ayahuasca. J. Ethnopharmacol. 1984, 10, 195–223. [Google Scholar] [CrossRef]
- Halberstadt, A.L. Behavioral and pharmacokinetic interactions between monoamine oxidase inhibitors and the hallucinogen 5-methoxy-N,N-dimethyltryptamine. Pharmacol. Biochem. Behav. 2016, 143, 1–10. [Google Scholar] [CrossRef]
- An Option if Other Antidepressants Haven’t Helped. 12 September 2019. Available online: https://www.mayoclinic.org/diseases-conditions/depression/in-depth/maois/art-20043992 (accessed on 7 February 2021).
- Szabo, A.; Kovacs, A.; Riba, J.; Djurovic, S.; Rajnavolgyi, E.; Frecska, E. The Endogenous Hallucinogen and Trace Amine N,N-Dimethyltryptamine (DMT) Displays Potent Protective Effects against Hypoxia via Sigma-1 Receptor Activation in Human Primary iPSC-Derived Cortical Neurons and Microglia-Like Immune Cells. Front. Neurosci. 2016, 10, 423. [Google Scholar] [CrossRef]
- Frecska, E.; Bokor, P.; Winkelman, M. The Therapeutic Potentials of Ayahuasca: Possible Effects against Various Diseases of Civilization. Front. Pharmacol. 2016, 7, 35. [Google Scholar] [CrossRef]
- Inserra, A. Hypothesis: The Psychedelic Ayahuasca Heals Traumatic Memories via a Sigma 1 Receptor-Mediated Epigenetic-Mnemonic Process. Front. Pharmacol. 2018, 9, 330. [Google Scholar] [CrossRef]
- Pal, A.; Fontanilla, D.; Gopalakrishnan, A.; Chae, Y.K.; Markley, J.L.; Ruoho, A.E. The sigma-1 receptor protects against cellular oxidative stress and activates antioxidant response elements. Eur. J. Pharmacol. 2012, 682, 12–20. [Google Scholar] [CrossRef]
- Carbonaro, T.M.; Gatch, M.B. Neuropharmacology of N,N-Dimethyltryptamine. Brain Res. Bull. 2016, 126, 1–61. [Google Scholar] [CrossRef]
- Malcolm, B.J.; Lee, K.C. Ayahuasca: An ancient sacrament for treatment of contemporary psychiatric illness? Ment. Health Clin. 2018, 7, 39–45. [Google Scholar] [CrossRef]
- Ruffell, S.; Netzband, N.; Bird, C.; Young, A.H.; Juruena, M.F. The pharmacological interaction of compounds in Ayahuasca: A systematic review. Rev. Bras. Psiquiatr. 2020, 42, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.J.; Albarracin-Jordan, J.; Moore, C.; Capriles, J.M. Chemical evidence for the use of multiple psychotropic plants in a 1,000-year-old ritual bundle from South America. Proc. Natl. Acad. Sci. USA 2019, 116, 11207–11212. [Google Scholar] [CrossRef] [PubMed]
- Savoldi, R.; Polari, D.; Pinheiro-da-Silva, J.; Silva, P.F.; Lobao-Soares, B.; Yonamine, M.; Freire, F.; Luchiari, A.C. Behavioral Changes Over Time Following Ayahuasca Exposure in Zebrafish. Front. Behav. Neurosci. 2017, 11, 139. [Google Scholar] [CrossRef] [PubMed]
- Labate, B.C.; Macrae, E. Ayahuasca, Ritual and Religion in Brazil; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Herraiz, T.; González, D.; Ancín-Azpilicueta, C.; Arán, V.J.; Guillén, H. β-Carboline alkaloids in Peganum harmala and inhibition of human monoamine oxidase (MAO). Food Chem. Toxicol. 2010, 48, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Morales-García, J.A.; de la Fuente Revenga, M.; Alonso-Gil, S.; Rodríguez-Franco, M.I.; Feilding, A.; Perez-Castillo, A.; Riba, J. The alkaloids of Banisteriopsis caapi, the plant source of the Amazonian hallucinogen Ayahuasca, stimulate adult neurogenesis in vitro. Sci. Rep. 2017, 7, 5309. [Google Scholar] [CrossRef]
- Jiménez-Garrido, D.F.; Gómez-Sousa, M.; Ona, G.; Dos Santos, R.G.; Hallak, J.; Alcázar-Córcoles, M.Á.; Bouso, J.C. Effects of Ayahuasca on mental health and quality of life in naïve users: A longitudinal and cross-sectional study combination. Sci. Rep. 2020, 10, 4075. [Google Scholar] [CrossRef]
- McKenna, D.J. Clinical investigations of the therapeutic potential of Ayahuasca: Rationale and regulatory challenges. Pharmacol. Ther. 2004, 102, 111–129. [Google Scholar] [CrossRef]
- Manske, R.H. A synthesis of the methyltryptamines and some derivatives. Can. J. Res. 1931, 5, 592–600. [Google Scholar] [CrossRef]
- Bigwood, J.; Ott, J. GWU Event on Colombia. for Wola if You Need Higher Resolution, Please e-mail Me, and I Will Send Them in Hi-res. November 1977. Available online: https://web.archive.org/web/20060127003553/ (accessed on 4 March 2021).
- Sanches, R.F.; de Lima Osório, F.; Dos Santos, R.G.; Macedo, L.R.; Maia-de-Oliveira, J.P.; Wichert-Ana, L.; de Araujo, D.B.; Riba, J.; Crippa, J.A.; Hallak, J.E. Antidepressant Effects of a Single Dose of Ayahuasca in Patients with Recurrent Depression: A SPECT Study. J. Clin. Psychopharmacol. 2016, 36, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.G.; Landeira-Fernandez, J.; Strassman, R.J.; Motta, V.; Cruz, A.P. Effects of Ayahuasca on psychometric measures of anxiety, panic-like and hopelessness in Santo Daime members. J. Ethnopharmacol. 2007, 112, 507–513. [Google Scholar] [CrossRef]
- Zeifman, R.J.; Palhano-Fontes, F.; Hallak, J.; Arcoverde, E.; Maia-Oliveira, J.P.; Araujo, D.B. The Impact of Ayahuasca on Suicidality: Results From a Randomized Controlled Trial. Front. Pharmacol. 2019, 10, 1325. [Google Scholar] [CrossRef]
- Uthaug, M.V.; van Oorsouw, K.; Kuypers, K.; van Boxtel, M.; Broers, N.J.; Mason, N.L.; Toennes, S.W.; Riba, J.; Ramaekers, J.G. Sub-acute and long-term effects of Ayahuasca on affect and cognitive thinking style and their association with ego dissolution. Psychopharmacology 2018, 235, 2979–2989. [Google Scholar] [CrossRef] [PubMed]
- Soler, J.; Elices, M.; Dominguez-Clavé, E.; Pascual, J.C.; Feilding, A.; Navarro-Gil, M.; García-Campayo, J.; Riba, J. Four Weekly Ayahuasca Sessions Lead to Increases in “Acceptance” Capacities: A Comparison Study with a Standard 8-Week Mindfulness Training Program. Front. Pharmacol. 2018, 9, 224. [Google Scholar] [CrossRef]
- Galvão, A.; de Almeida, R.N.; Silva, E.; Freire, F.; Palhano-Fontes, F.; Onias, H.; Arcoverde, E.; Maia-de-Oliveira, J.P.; de Araújo, D.B.; Lobão-Soares, B.; et al. Cortisol Modulation by Ayahuasca in Patients with Treatment Resistant Depression and Healthy Controls. Front. Psychiatry 2018, 9, 185. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, R.G.; Hallak, J.E. Effects of the Natural β-Carboline Alkaloid Harmine, a Main Constituent of Ayahuasca, in Memory and in the Hippocampus: A Systematic Literature Review of Preclinical Studies. J. Psychoact. Drugs 2017, 49, 1–10. [Google Scholar] [CrossRef]
- Loizaga-Velder, A.; Verres, R. Therapeutic effects of ritual Ayahuasca use in the treatment of substance dependence—qualitative results. J. Psychoact. Drugs 2014, 46, 63–72. [Google Scholar] [CrossRef]
- Thomas, G.; Lucas, P.; Capler, N.R.; Tupper, K.W.; Martin, G. Ayahuasca-assisted therapy for addiction: Results from a preliminary observational study in Canada. Curr. Drug Abus. Rev. 2013, 6, 30–42. [Google Scholar] [CrossRef]
- Palhano-Fontes, F.; Andrade, K.C.; Tofoli, L.F.; Santos, A.C.; Crippa, J.A.S.; Hallak, J.E.C.; Ribeiro, S.; de Araujo, D.B. The psychedelic state induced by Ayahuasca modulates the activity and connectivity of the default mode network. PLoS ONE 2015, 10, e0118143. [Google Scholar] [CrossRef] [PubMed]
- Uddin, L.Q.; Kelly, A.M.; Biswal, B.B.; Castellanos, F.X.; Milham, M.P. Functional connectivity of default mode network components: Correlation, anticorrelation, and causality. Hum. Brain Mapp. 2009, 30, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Andrews-Hanna, J.R.; Schacter, D.L. The brain’s default network: Anatomy, function, and relevance to disease. Ann. N. Y. Acad. Sci. 2008, 1124, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Jung, W.H.; Kang, D.H.; Byun, M.S.; Kwon, S.J.; Choi, C.H.; Kwon, J.S. Increased default mode network connectivity associated with meditation. Neurosci. Lett. 2011, 487, 358–362. [Google Scholar] [CrossRef] [PubMed]
- Greicius, M.D.; Kiviniemi, V.; Tervonen, O.; Vainionpää, V.; Alahuhta, S.; Reiss, A.L.; Menon, V. Persistent default-mode network connectivity during light sedation. Hum. Brain Mapp. 2008, 29, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Horovitz, S.G.; Braun, A.R.; Carr, W.S.; Picchioni, D.; Balkin, T.J.; Fukunaga, M.; Duyn, J.H. Decoupling of the brain’s default mode network during deep sleep. Proc. Natl. Acad. Sci. USA 2009, 106, 11376–11381. [Google Scholar] [CrossRef]
- Schrouff, J.; Perlbarg, V.; Boly, M.; Marrelec, G.; Boveroux, P.; Vanhaudenhuyse, A.; Bruno, M.A.; Laureys, S.; Phillips, C.; Pélégrini-Issac, M.; et al. Brain functional integration decreases during propofol-induced loss of consciousness. NeuroImage 2011, 57, 198–205. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Erritzoe, D.; Williams, T.; Stone, J.M.; Reed, L.J.; Colasanti, A.; Tyacke, R.J.; Leech, R.; Malizia, A.L.; Murphy, K.; et al. Neural correlates of the psychedelic state as determined by fMRI studies with Psilocybin. Proc. Natl. Acad. Sci. USA 2012, 109, 2138–2143. [Google Scholar] [CrossRef] [PubMed]
- Carhart-Harris, R.L.; Leech, R.; Erritzoe, D.; Williams, T.M.; Stone, J.M.; Evans, J.; Sharp, D.J.; Feilding, A.; Wise, R.G.; Nutt, D.J. Functional connectivity measures after Psilocybin inform a novel hypothesis of early psychosis. Schizophr. Bull. 2013, 39, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Mason, M.F.; Norton, M.I.; Van Horn, J.D.; Wegner, D.M.; Grafton, S.T.; Macrae, C.N. Wandering minds: The default network and stimulus-independent thought. Science 2007, 315, 393–395. [Google Scholar] [CrossRef]
- Brewer, J.A.; Worhunsky, P.D.; Gray, J.R.; Tang, Y.Y.; Weber, J.; Kober, H. Meditation experience is associated with differences in default mode network activity and connectivity. Proc. Natl. Acad. Sci. USA 2011, 108, 20254–20259. [Google Scholar] [CrossRef]
- Sämann, P.G.; Wehrle, R.; Hoehn, D.; Spoormaker, V.I.; Peters, H.; Tully, C.; Holsboer, F.; Czisch, M. Development of the brain’s default mode network from wakefulness to slow wave sleep. Cereb. Cortex 2011, 21, 2082–2093. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J. Ayahuasca Tourism: The Commercialization of Culture. 2 August 2019. Available online: https://worldfootprints.com/Ayahuasca-tourism-the-commercialization-of-culture/ (accessed on 8 February 2021).
- Draven, J. Hot Topic: Is Ayahuasca Tourism a Bad Trip? 8 April 2019. Available online: https://www.nationalgeographic.co.uk/travel/2017/03/hot-topic-Ayahuasca-tourism-bad-trip (accessed on 8 February 2021).
- Kavenská, V.; Simonová, H. Ayahuasca Tourism: Participants in Shamanic Rituals and their Personality Styles, Motivation, Benefits and Risks. J. Psychoact. Drugs 2015, 47, 351–359. [Google Scholar] [CrossRef]
- Hill, D. Peru’s Ayahuasca Industry Booms as Westerners Search for Alternative Healing. 7 June 2016. Available online: https://www.theguardian.com/travel/2016/jun/07/peru-Ayahuasca-drink-boom-amazon-spirituality-healing (accessed on 9 February 2021).
- Braczkowski, A.; Ruzo, A.; Sanchez, F.; Castagnino, R.; Brown, C.; Guynup, S.; Winter, S.; Gandy, D.; O’Bryan, C.J. The Ayahuasca tourism boom: An undervalued demand driver for jaguar body parts? Conserv. Sci. Pract. 2019, 1, Ee126. [Google Scholar] [CrossRef]
- Crisafulli, A. Ayahuasca Tourism: Shamans, Charlatans AND Thousand-Dollar Retreats. 2019. Available online: https://www.vergemagazine.com/travel-intelligence/beyond-the-guidebook/2495-is-Ayahuasca-tourism-safe-and-ethical.html (accessed on 9 February 2021).
- Babe, A. Ayahuasca Tourism is Ripping off Indigenous Amazonians. May 2016. Available online: https://www.vice.com/en/article/qbn8vq/Ayahuasca-tourism-is-ripping-off-indigenous-amazonians (accessed on 8 February 2021).
- Vaughn, C. Ayahuasca Ceremonies and Tourism Return to Costa Rica. 25 October 2020. Available online: https://news.co.cr/Ayahuasca-ceremonies-and-tourism-return-to-costa-rica/82489/ (accessed on 9 February 2021).
- Matthews, K. Visiting Brazil for an Ayahuasca Ceremony. 20 December 2019. Available online: https://thetravelmanuel.com/visiting-brazil-for-an-Ayahuasca-ceremony/ (accessed on 9 February 2021).
- Fraser, B. The Perils and Privileges of an Amazonian Hallucinogen. 8 August 2017. Available online: https://www.sapiens.org/culture/Ayahuasca-tourism-amazon/ (accessed on 9 February 2021).
- Om Spirit. Ayahuasca Retreat Sacred Valley Tribe. 29 December 2018. Available online: https://omspirit.net/retrets-centers/ayahuasca-retreat-sacred-valley-tribe/ (accessed on 9 February 2021).
- Amor, B. Ayahuasca is the Latest TRENDY Tonic for White People Problems. 21 June 2019. Available online: https://www.bitchmedia.org/article/heart-of-whiteness-spiritual-tourism-colonization-Ayahuasca (accessed on 9 February 2021).
- Hay, M. The Colonization of the Ayahuasca Experience. JSTOR Daily. 20 November 2020. Available online: https://daily.jstor.org/the-colonization-of-the-ayahuasca-experience/ (accessed on 9 February 2021).
- Krebs-Thomson, K.; Ruiz, E.M.; Masten, V.; Buell, M.; Geyer, M.A. The roles of 5-HT1A and 5-HT2 receptors in the effects of 5-MeO-DMT on locomotor activity and prepulse inhibition in rats. Psychopharmacology 2006, 189, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Ray, T.S. Psychedelics and the human receptorome. PLoS ONE 2010, 5, e9019. [Google Scholar] [CrossRef]
- Shen, H.W.; Jiang, X.L.; Winter, J.C.; Yu, A.M. Psychedelic 5-methoxy-N,N-dimethyltryptamine: Metabolism, pharmacokinetics, drug interactions, and pharmacological actions. Curr. Drug Metab. 2010, 11, 659–666. [Google Scholar] [CrossRef]
- Malaca, S.; Lo Faro, A.F.; Tamborra, A.; Pichini, S.; Busardò, F.P.; Huestis, M.A. Toxicology and Analysis of Psychoactive Tryptamines. Int. J. Mol. Sci. 2020, 21, 9279. [Google Scholar] [CrossRef]
- Hoshino, T.; Shimodaira, K. Über die synthese des bufotenin-methyl-äthers (5-methoxy-n-dimethyl-tryptamin) und bufotenins (synthesen in der indol-gruppe. xv). Bull. Chem. Soc. Jpn. 1936, 11, 221–224. [Google Scholar] [CrossRef]
- Pachter, I.J.Z.; Ribeiro, D.E.; Ribeiro, O. Indole alkaloids of acer saccharinum (the silver maple), Dictyoloma incanescens, Piptadenia colubrina, and Mimosa hostilis. J. Org. Chem. 1959, 24, 1285–1287. [Google Scholar] [CrossRef]
- Weil, A.T.; Davis, W. Bufo alvarius: A potent hallucinogen of animal origin. J. Ethnopharmacol. 1994, 41, 1–8. [Google Scholar] [CrossRef]
- Guide to 5-MeO-DMT-EXPERIENCE, BENEFITS, & Side Effects. 5 January 2021. Available online: https://thethirdwave.co/psychedelics/5-meo-dmt/ (accessed on 5 February 2021).
- Uthaug, M.V.; Lancelotta, R.; van Oorsouw, K.; Kuypers, K.; Mason, N.; Rak, J.; Šuláková, A.; Jurok, R.; Maryška, M.; Kuchař, M.; et al. A single inhalation of vapor from dried toad secretion containing 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT) in a naturalistic setting is related to sustained enhancement of satisfaction with life, mindfulness-related capacities, and a decrement of psychopathological symptoms. Psychopharmacology 2019, 236, 2653–2666. [Google Scholar] [CrossRef]
- Araújo, A.M.; Carvalho, F.; Bastos, M.; Guedes de Pinho, P.; Carvalho, M. The hallucinogenic world of tryptamines: An updated review. Arch. Toxicol. 2015, 89, 1151–1173. [Google Scholar] [CrossRef] [PubMed]
- Halberstadt, A.L.; Buell, M.R.; Masten, V.L.; Risbrough, V.B.; Geyer, M.A. Modification of the effects of 5-methoxy-N,N-dimethyltryptamine on exploratory behavior in rats by monoamine oxidase inhibitors. Psychopharmacology 2008, 201, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.L.; Shen, H.W.; Mager, D.E.; Yu, A.M. Pharmacokinetic interactions between monoamine oxidase A inhibitor harmaline and 5-methoxy-N,N-dimethyltryptamine, and the impact of CYP2D6 status. Drug Metab. Dispos. Biol. Fate Chem. 2013, 41, 975–986. [Google Scholar] [CrossRef]
- Gale, E. Bufo Alvarius-Colorado River Toad Sonoran Desert Toad. 11 April 2003. Available online: https://www.erowid.org/archive/sonoran_desert_toad/deeptoad.htm (accessed on 10 March 2021).
- Pochettino, M.L.; Cortella, A.R.; Ruiz, M. Hallucinogenic snuff from Northwestern Argentina: Microscopical identification of anadenanthera colubrina var. cebil (fabaceae) in powdered archaeological material. Econ. Bot. 1999, 53, 127–132. [Google Scholar] [CrossRef]
- Carod-Artal, F.J.; Vázquez Cabrera, C.B. Usos rituales de la semilla de Anadenanthera sp entre los indígenas sudamericanos [Ritual use of Anadenanthera seeds among South America natives]. Neurologia 2007, 22, 410–415. [Google Scholar]
- Schultes, R.E. The Plant Kingdom and Hallucinogens (Part II). 1 January 1969. Available online: https://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1969-01-01_4_page004.html (accessed on 5 February 2021).
- Pipeline: GH Research. GH Research Limited (n.d.). Available online: https://www.ghres.com/pipeline (accessed on 10 March 2021).
- Lysergide (LSD) drug profile. European Monitoring Centre for Drugs and Drug Addiction. 1 January 2021. Available online: https://www.emcdda.europa.eu/publications/drug-profiles/lsd_en (accessed on 10 March 2021).
- Whelan, A.; Johnson, M.I. Lysergic acid diethylamide and Psilocybin for the management of patients with persistent pain: A potential role? Pain Manag. 2018, 8, 217–229. [Google Scholar] [CrossRef]
- Brandt, S.D.; Kavanagh, P.V.; Twamley, B.; Westphal, F.; Elliott, S.P.; Wallach, J.; Stratford, A.; Klein, L.M.; McCorvy, J.D.; Nichols, D.E.; et al. Return of the lysergamides. Part IV: Analytical and pharmacological characterization of lysergic acid morpholide (LSM-775). Drug Test. Anal. 2018, 10, 310–322. [Google Scholar] [CrossRef]
- Halberstadt, A.L.; Klein, L.M.; Chatha, M.; Valenzuela, L.B.; Stratford, A.; Wallach, J.; Nichols, D.E.; Brandt, S.D. Pharmacological characterization of the LSD analog N-ethyl-N-cyclopropyl lysergamide (ECPLA). Psychopharmacology 2019, 236, 799–808. [Google Scholar] [CrossRef]
- Watts, V.J.; Lawler, C.P.; Fox, D.R.; Neve, K.A.; Nichols, D.E.; Mailman, R.B. LSD and structural analogs: Pharmacological evaluation at D1 dopamine receptors. Psychopharmacology 1995, 118, 401–409. [Google Scholar] [CrossRef]
- Hoffman, A.J.; Nichols, D.E. Synthesis and LSD-like discriminative stimulus properties in a series of N(6)-alkyl norlysergic acid N,N-diethylamide derivatives. J. Med. Chem. 1985, 28, 1252–1255. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Marona-Lewicka, D.; Pfaff, R.C.; Nichols, D.E. Drug discrimination and receptor binding studies of N-isopropyl lysergamide derivatives. Pharmacol. Biochem. Behav. 1994, 47, 667–673. [Google Scholar] [CrossRef]
- Fuentes, J.J.; Fonseca, F.; Elices, M.; Farré, M.; Torrens, M. Therapeutic Use of LSD in Psychiatry: A Systematic Review of Randomized-Controlled Clinical Trials. Front. Psychiatry 2020, 10, 943. [Google Scholar] [CrossRef]
- Davies, B. Lysergic acid (LSD 25) and ritalin in the treatment of neurosis. J. Psychosom. Res. 1964, 8, 164–165. [Google Scholar] [CrossRef]
- Das, S.; Barnwal, P.; Ramasamy, A.; Sen, S.; Mondal, S. Lysergic acid diethylamide: A drug of ‘use’? Ther. Adv. Psychopharmacol. 2016, 6, 214–228. [Google Scholar] [CrossRef]
- Jordy, S.S. Book Reviews-LSD and alcoholism; a clinical study of treatment efficacy. Q. J. Stud. Alcohol 1971, 32, 589–592. [Google Scholar] [CrossRef]
- McBroom, P. LSD vs. Alcoholism. Sci. News 1968, 93, 578. [Google Scholar] [CrossRef]
- Szabo, A. Psychedelics and Immunomodulation: Novel Approaches and Therapeutic Opportunities. Front. Immunol. 2015, 6, 358. [Google Scholar] [CrossRef] [PubMed]
- Family, N.; Maillet, E.L.; Williams, L.T.J.; Krediet, E.; Carhart-Harris, R.L.; Williams, T.M.; Nichols, C.D.; Goble, D.J.; Raz, S. Safety, tolerability, pharmacokinetics, and pharmacodynamics of low dose lysergic acid diethylamide (LSD) in healthy older volunteers. Psychopharmacology 2020, 237, 841–853. [Google Scholar] [CrossRef]
- Bender, L.; Goldschmidt, L.; Sankar, D.V.S.; Freedman, A.M. Treatment of Autistic Schizophrenic Children with LSD-25 and UML-491. In Recent Advances in Biological Psychiatry; Wortis, J., Ed.; Springer: Boston, MA, USA, 1962. [Google Scholar] [CrossRef]
- Bender, L. D-lysergic acid in the treatment of the biological features of childhood schizophrenia. Dis. Nerv. Syst. 1966, 7 (Suppl. 7), 43–46. [Google Scholar]
- Freedman, A.M.; Ebin, E.V.; Wilson, E.A. Autistic Schizophrenic Children; An Experiment in the Use of D-Lysergic Acid Diethylamide (LSD-25). Arch. Gen. Psychiatry 1962, 6, 203–213. [Google Scholar] [CrossRef]
- Moller, H. The treatment of childhood schizophrenia in a public school system. Psychol. Sch. 1964, 1, 297–304. [Google Scholar] [CrossRef]
- Rice, M.E.; Harris, G.T.; Cormier, C.A. An evaluation of a maximum security therapeutic community for psychopaths and other mentally disordered offenders. Law Hum. Behav. 1992, 16, 399–412. [Google Scholar] [CrossRef]
- Harris, G.T.; Rice, M.E.; Cormier, C.A. Psychopaths: Is a therapeutic community therapeutic? Ther. Communities 1994, 15, 283–299. [Google Scholar]
- Reidy, D.E.; Kearns, M.C.; DeGue, S. Reducing psychopathic violence: A review of the treatment literature. Aggress. Violent Behav. 2013, 18, 527–538. [Google Scholar] [CrossRef]
- Honigfeld, G. Temporal effects of LSD-25 and epinephrine on verbal behavior. PsycEXTRA Dataset. 1965, 70, 303. [Google Scholar] [CrossRef] [PubMed]
- Ling, T.M.; Buckman, J. The Treatment of Frigidity with LSD and Ritalin. In The Psychedelic Reader: Selected from the Psychedelic Review; Weil, G.M., Metzner, R., Leary, T., Eds.; Place of publication not identified; University Books: New York, NY, USA, 1965; pp. 231–239. [Google Scholar]
- Davenport, W.J. Psychedelic and nonpsychedelic LSD and Psilocybin for cluster headache. Can. Med. Assoc. J. 2016, 188, 217. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sewell, R.A.; Halpern, J.H.; Pope, H.G. Response of cluster headache to Psilocybin and lsd. Neurology 2006, 66, 1920–1922. [Google Scholar] [CrossRef] [PubMed]
- Liechti, M.; Schmid, Y. Lysergic Acid Diethylamide (LSD) as Treatment for Cluster Headache (LCH). Identifier NCT03781128. (2 January 2019–December 2023). Available online: https://clinicaltrials.gov/ct2/show/NCT03781128 (accessed on 11 March 2021).
- Karst, M.; Halpern, J.H.; Bernateck, M.; Passie, T. The non-hallucinogen 2-bromo-lysergic acid diethylamide as preventative treatment for cluster headache: An open, non-randomized case series. Cephalalgia 2010, 30, 1140–1144. [Google Scholar] [CrossRef] [PubMed]
- Schindler, E.A.; Gottschalk, C.H.; Weil, M.J.; Shapiro, R.E.; Wright, D.A.; Sewell, R.A. Indoleamine hallucinogens in cluster headache: Results of the clusterbusters medication use survey. J. Psychoact. Drugs 2015, 47, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Sicuteri, F. Prophylactic treatment of migraine by means of lysergic acid derivatives. Triangle 1963, 6, 116–125. [Google Scholar]
- Crowther, D.L. The prophylactic effect of 1-methyl-D-lysergic acid butanolamide (methysergide) in the treatment of vascular headache. A clinical study. Med. Exp. Int. J. Exp. Med. 1964, 10, 137–143. [Google Scholar]
- Fanciullacci, M.; Bene, E.D.; Franchi, G.; Sicuteri, F. Brief report: Phantom limp pain: Sub-hallucinogenic treatment with lysergic acid diethylamide (LSD-25). Headache 1977, 17, 118–119. [Google Scholar] [CrossRef]
- Liester, M.B. A review of lysergic acid diethylamide (LSD) in the treatment of addictions: Historical perspectives and future prospects. Curr. Drug Abus. Rev. 2014, 7, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Winkelman, M. Psychedelics as medicines for substance Abuse Rehabilitation: Evaluating treatments with lsd, Peyote, ibogaine and Ayahuasca. Curr. Drug Abus. Rev. 2015, 7, 101–116. [Google Scholar] [CrossRef]
- Oehen, P.; Traber, R.; Widmer, V.; Schnyder, U. A randomized, controlled pilot study of MDMA (± 3,4-Methylenedioxymethamphetamine)-assisted psychotherapy for treatment of resistant, chronic Post-Traumatic Stress Disorder (PTSD). J. Psychopharmacol. 2013, 27, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Mithoefer, M.C.; Wagner, M.T.; Mithoefer, A.T.; Jerome, L.; Doblin, R. The safety and efficacy of {+/-}3,4-methylenedioxymethamphetamine-assisted psychotherapy in subjects with chronic, treatment-resistant posttraumatic stress disorder: The first randomized controlled pilot study. J. Psychopharmacol. 2011, 25, 439–452. [Google Scholar] [CrossRef] [PubMed]
- Amoroso, T.; Workman, M. Treating posttraumatic stress disorder with MDMA-assisted psychotherapy: A preliminary meta-analysis and comparison to prolonged exposure therapy. J. Psychopharmacol. 2016, 30, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Psychedelic Drugs Market Projected to Reach $6859.95 Million by 2027. 3 June 2020. Available online: https://www.prnewswire.com/news-releases/psychedelic-drugs-market-projected-to-reach-6-859-95-million-by-2027--301069861.html (accessed on 16 February 2021).
- Europe Psychedelic Drugs Market Global Survey Report with COVID-19 Impact and Overwhelming Hike of BILLION-DOLLAR INDUSTRY: COMPASS, Johnson & JOHNSON, NeuroRX, Hikma Pharmaceuticals, Jazz Pharmaceuticals. 17 November 2020. Available online: https://apnews.com/press-release/wired-release/business-technology-lifestyle-products-and-services-mental-health-04591bf30586e804c554fe61fafddaf7 (accessed on 16 February 2021).
- Winkelman, M. Drug tourism or spiritual healing? Ayahuasca seekers in Amazonia. J. Psychoact. Drugs 2005, 37, 209–218. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lowe, H.; Toyang, N.; Steele, B.; Grant, J.; Ali, A.; Gordon, L.; Ngwa, W. Psychedelics: Alternative and Potential Therapeutic Options for Treating Mood and Anxiety Disorders. Molecules 2022, 27, 2520. https://doi.org/10.3390/molecules27082520
Lowe H, Toyang N, Steele B, Grant J, Ali A, Gordon L, Ngwa W. Psychedelics: Alternative and Potential Therapeutic Options for Treating Mood and Anxiety Disorders. Molecules. 2022; 27(8):2520. https://doi.org/10.3390/molecules27082520
Chicago/Turabian StyleLowe, Henry, Ngeh Toyang, Blair Steele, Justin Grant, Amza Ali, Lorenzo Gordon, and Wilfred Ngwa. 2022. "Psychedelics: Alternative and Potential Therapeutic Options for Treating Mood and Anxiety Disorders" Molecules 27, no. 8: 2520. https://doi.org/10.3390/molecules27082520
APA StyleLowe, H., Toyang, N., Steele, B., Grant, J., Ali, A., Gordon, L., & Ngwa, W. (2022). Psychedelics: Alternative and Potential Therapeutic Options for Treating Mood and Anxiety Disorders. Molecules, 27(8), 2520. https://doi.org/10.3390/molecules27082520





