2.1. Propolis Characterization
Previous studies have shown that propolis extract in water/ethanol mixtures does not dissolve but could be dissolved in 10–15% PVA water/ethanol solution and nanofibers could be obtained from this solution [
7]. Some other investigations show ethanol solubilized propolis in a range of concentrations from 3 to 20% could be mixed with the 10 to 15% PVA water/ethanol solution and obtain fibers from solutions with various morphology [
11,
12,
13,
14]. In this study laboratory made propolis water (PW/BRA Lab) and ethyl alcohol propolis extract (PEx/BRA Lab) were prepared (
Section 3.3) and used in view of the fact that the largest amount of spinning solution is water (90–94%), so propolis water is one of the most suitable for integration into the solution. Propolis is only partially soluble in water, and there are limited possibilities to obtain propolis water with the highest propolis content. This study analyzed two propolis water types obtained in a laboratory and two industrially produced with different propolis contents to evaluate their suitability for integration in a spinning solution (
Table 1). The combination of different propolis waters makes it possible to obtain fibers with customized properties depending on the planned use.
The pH of propolis water affects the total pH of the solution and may result from the effect of fiber diameters, a slight decrease at a pH of 2 due to the decrease in viscosity in an acidic environment [
9]. As can be seen from
Table 1, the pH of all four propolis water samples are in range from 3.4 to 4.6.
As part of this study, it has been found that the density of propolis water affects the total viscosity of the solution. The obtained data indicate that the highest density is for propolis water PW/LT (1.01 g/mL), which also contains the highest amount of propolis, and for laboratory-made propolis water obtained from propolis in Brazil and containing Arabic Gum (1.032 g/mL). The increase in density affects not only the total viscosity of the solution but also the mixing time required to dissolve the PVA. Arabic Gum (Gum Acacia) is used as a stabilizer and a thickener in the food and pharmaceutic industries; therefore, an effect on density was expected. Arabic Gum is also useful for PVA film blends tensile properties improvement [
20].
The propolis water added to the electrospinning solution effect on electrical conductivity is significant. The conductivity of propolis water samples was in a range from 352 to 1526 µS/cm. A too-high electrical conductivity (up to 1000 µS/cm) can result in an orientation of fibers perpendicular to the support material. As shown in
Table 1, PW/LT and PW/BRA Lab samples have high conductivity, but it can be balanced by the addition of low-conductivity propolis extracts in the spinning solution.
High propolis content does not ensure high flavonoid content in propolis water. As shown in
Table 1, PW/BRA Lab propolis contains the highest concentration of flavonoids (1.25 mg/g), but its propolis content is only 10%, in comparison with the PW/LT sample, which contains 30.8% propolis but only 0.087 mg/g of flavonoids. This can be affected by both the method of propolis water preparation and the location and time of propolis collection [
1]. Comparing the proportions of propolis and flavonoids in aqueous solution and powder, it can be seen that propolis powder (PPa/BRA) has a high solubility in water by boiling, according to the mixing scheme shown in
Section 3.3, because propolis powder (~100%) contains 5.43 mg/g of flavonoid, but 10% propolis water (PW/BRA Lab) 1.25 mg/g of flavonoids (
Table 1 and
Table 2). The sample PW/BRA Lab has the highest concentration of flavonoids in proportion to the propolis content compared to other water samples of propolis–0.0139 mg/g.
Compared with propolis waters, hydroglyceric propolis extract (20–25%) has a higher density (1.220 g/mL) and low electrical conductivity (3 µS/cm), and flavonoid content (1.15 mg/g) is similar to PW/BRA Lab (1.25 mg/g),
Table 2. Due to the low electrical conductivity, hydroglyceric propolis extract is suitable for combination with propolis water, which has high electrical conductivity, but care should be taken not to increase the viscosity too much—it is better to use lower PVA concentrations. Glycerol acts as a plasticizer for PVA [
21]; it can promote a more even mixing between the water-insoluble part of propolis and affect the mechanical properties of the fiber web.
Due to the very high molecular complex composition of propolis, different extractive processes are needed to remove the inert material and preserve the polyphenolic fraction-flavonoids and phenolic acids. Ethanolic extraction is commonly used for this purpose [
6]. The pH of ethyl alcohol propolis extracts is more alkaline than that of propolis water, and no significant effect on fiber diameters is expected due to the excessively acidic environment in electrospinning solutions. Rapid reductions in viscosity can only be observed by adding high levels of ethyl alcohol extracts in low-concentration PVA spinning solutions due to the density rising from 0.885 to 0.905 g/mL (
Table 3). Depending on the concentration of PVA, the conductivity of the electrospinning solution may decrease slightly. The main problem with the addition of propolis ethyl alcohol to PVA solutions is the formation of propolis agglomerates due to the reduction of alcohol concentration. It can be seen that the use of a higher alcohol concentration improves the content of flavonoids: 98% ethyl alcohol extract contains 8.87 and 7.53 mg/g, respectively, and 70% extract contains 5.75 mg/g (
Table 3). The propolis harvested in Latvia (LV) has a higher flavonoids content in its alcohol extract (PEx/LV Lab) than that harvested in Brazil (BRA) when the alcohol extract (PEx/LV Lab) is obtained, but it is not fully comparable to the PEx/BRA Lab extract due to the small amount of Arabic Gum content in the propolis powder. The wax component of LV propolis has been previously removed. The Arabic Gum swells and dissolves in water, and it has the potential to be used as a wound dressing due to its antibacterial and hemostatic activity [
22].
2.2. PVA/Propolis Extract Solution Characteristic
The 125 kDa and 130 kDa PVA polymers have short mixing time and dissolution temperature (not exceeding 100 °C). Their viscosity and electrical conductivity are also suitable for all tested concentrations (6, 8, and 10 wt%). After the range of properties of PVA colloidal solutions were determined, possible strategies for the integration of propolis were developed and nanowebs for morphology analysis were made.
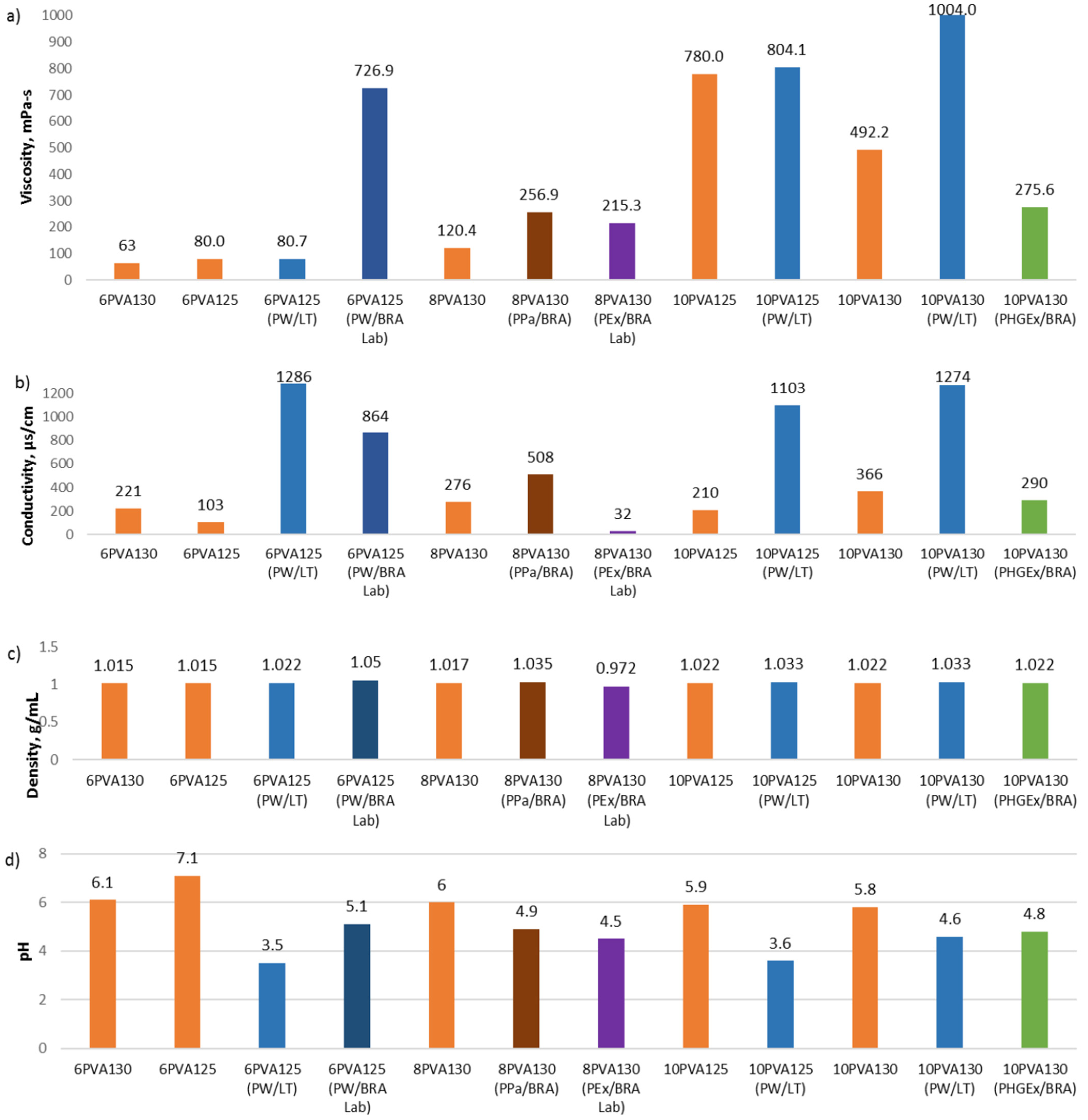
We compared 6 wt% and 10 wt% PVA solutions to solutions to which propolis water has been added. As could be expected from the analysis of the properties, the addition of propolis water increases the viscosity, conductivity, and density for all samples (
Figure 1). In particular, the viscosity of the 6PVA130 is close to a value of 60 mPa s, which is practically the lower limit of viscosity for spinning fibers with a cylindrical electrode. However, 6PVA125 viscosity is 80 mPa s, and PW/LT additive does not increase it substantially, but PW/BRA Lab increases viscosity dramatically up to 727 mPa s. The additive of PW/LT increases viscosity of 10PVA125 from 780 to 804 mPa s and 10PVA130 from 492 to 1004 mPa s. All solutions with the addition of propolis water have the appropriate viscosity for electrospinning in the range from 81 to 1004 mPa s (
Figure 1a).
The PVA solutions 125 KDa and 130 kDa have the same density, i.e., 1.015 (6 w%) and 1.022 g/mL (10 wt%), respectively, and replacing distilled water with propolis water PW/LT increases the density to 1.022 g/mL (6PVA125 and 6PVA130) and 1.033 g/mL (10PVA130 and 10PVA125), and replacing distilled water with PW/BRA Lab increases the density to 1.05 g/mL (6PVA125). (
Figure 1c).
Propolis water significantly increases the electrical conductivity of the spinning solutions (
Figure 1b). The 6PVA126 conductivity increases from 103 to 1286 µS/cm when PW/LT additive is used and to 864 µS/cm when PW/BRA Lab is used. The conductivity of 10PVA125 increases from 210 to 1103 µS/cm and 10PVA130 from 366 to 1274 µS/cm when distilled water is replaced with PW/LT. All prepared solutions, with and without propolis water, have sufficient electrical conductivity to form fibers.
The acidic environment of the propolis additives also significantly reduces the total pH of the spinning solutions but does not reach pH 2, which could significantly affect the fiber size (
Figure 1d).
Hydroglyceric propolis additive (PHGEx/BRA) decreases the viscosity of the 10PVA130 solution by 44% (
Figure 1a) and the pH from 5.9 to 4.8 (
Figure 1d), but the density does not change (
Figure 1c) because the densities of the additive and the PVA solution are the same. The electrical conductivity also decreases from 366 to 290 μs/cm, but this still is sufficient for electrospinning (
Figure 1b).
The addition of 3.85 wt% propolis powder (PPa/BRA) increases the viscosity, electrical conductivity, and density of 8PVA130 solution but decreases the pH from 6 to 4.9 (
Figure 1).
Ethyl alcohol propolis extract (PEx/BRA Lab) additive increases the viscosity of the 8PVA130 solution and significantly reduces conductivity, but the density decreases under the influence of alcohol (
Figure 1a–c). The propolis powder and alcohol extract decrease the pH correspondingly to 4.9 and 4.5 (
Figure 1d).
2.3. Nanoweb Morphology
All propolis-containing solutions were spun stably, and no web defects such as film areas were visible. As mentioned in
Section 2.2, the addition of propolis water increased the viscosity for all samples and the resulting fiber diameters. The only exception is hydroglyceric propolis extract/PVA solution, which turned into flat fibers instead of cylindrical ones, and the fiber width was not as homogeneous as that of the other samples due to the high glycerol content. The web of 10PVA130 (
Figure 2a) shows that the fibers’ average diameter was of 286 ± 10 nm, the addition of propolis water (
Figure 2b) increased the diameters to 664 ± 11 nm, hydroglyceric propolis extracts (
Figure 2c) increased them to 606 ± 11 nm, and combination of hydroglyceric propolis extracts and propolis alcohol extract increased them to 491 ± 8 nm. The sample 10PVA130(PHGEx/BRA+ PEx/LV) fibers tended to form bundles; 2–6 fibers were stuck together, glued in the parallel direction (
Figure 2e). The web of 8PVA130 (
Figure 2d) showed fibers with an average diameter of 255 ± 4 nm, and the micrograph of sample 8PVA130 (PPa/BRA) shows inclusions of spherical propolis powder particles not exceeding 7000 nm in size (
Figure 2f) and fibers with an average diameter of 218 ± 4 nm (
Table 4).
2.4. Total Flavonoids and Propolis Content of Nanowebs
The flavonoids content of fibers containing propolis water PW/LT (4.74–4.96 mg/g) is close to the calculated flavonoid content (4.42–4.96 mg/g). It allows a relatively accurate prediction of the flavonoids content in the fibers and shows that propolis is present not only in the spinning solution but also in the same amount in the fibers. The content of propolis in the fibers is 73.5 % (
Table 5) and may be higher if a 6 wt% PVA concentration solution is used.
With the addition of propolis powder, it is possible to obtain fibers with a propolis content of 33%. In future studies, combining propolis water and propolis powder has the potential to achieve even higher propolis content in the fibers. The flavonoids content of fibers containing propolis particles PPa/BRA (8.3–8.88 mg/g) is lower than calculated (12.86–13.2 mg/g), which means that fewer propolis particles from the solution enter the web, such that it is possible that the solution is too saturated with propolis particles (
Table 5).
The flavonoids content of fibers containing propolis glycerol extract PHGEx/BRA (4.91–5.34 mg/g) is slightly higher than calculated (4.49–5.17 mg/g), but it still allows a relatively accurate prediction of total flavonoid content in the fiber web. As glycerol acts as a plasticizer, and in view of SEM micrographs showing the formation of flat fibers (
Figure 2c), the 7 wt% amount of hydroglyceric propolis extract in the 10PVA130 spinning solution results in only 12% propolis content in fibers (
Table 5) and should not be exceeded. The hydroglyceric propolis extract datasheet indicates that it contains 20–25% propolis, but a concentration of 20% was used for the calculation, which may explain the slightly higher content of flavonoids, and the content of propolis in the fibers may reach to 15% if the 25% extract concentration is used.
The addition of propolis hydroglycerol extract and alcoholic extract in a 10 wt% PVA spinning solution allows to incorporate 26% of propolis in the electrospun fibers (
Table 5), there are potential to add a higher amount of alcohol extract and reduce the PVA concentration to 8 wt% to achieve a higher propolis concentration in the electrospun fibers (depending on solution viscosity). The flavonoids content of fibers containing propolis extracts PHGEx/BRA and PEx/LV (32.72–33.54 mg/g) is a bit higher than calculated (30.33–31.01 mg/g), and the fiber web containing glycerol extract also had a slightly higher flavonoid content than expected, so the presence of glycerol in this sample is also likely to be due to a slightly higher actual flavonoid content than calculated.
2.5. Solution Stability and Shelf Life
The sample 6PVA125(PW/LT) is homogeneous and can be spun even after 6 months (
Figure S1a). The solution 6PVA125(PW/BRA Lab) precipitated 5 min after mixing with a magnetic stirrer (
Figure S1c), and 3 h ultrasound mixing temporarily improved uniformity and particle size reduction, but the solution was also stratified after 5 min (
Figure S1d–f). Therefore, premixing is required. The samples 10PVA125(PW/LT) and 10PVA130(PW/LT) have excellent stability of solutions: their spinnability time is 6 months and premixing is not required, nor was precipitate or mold observed.
After the solution 8PVA130(PPa/BRA) had settled for a week, a small precipitate was visible (
Figure S2b). Additional ultrasonic mixing was also used to both reduce the particle size and improve the dispersion (
Figure S2c–f). After the solution was mixed with a magnetic stirrer, large agglomerates (6–59 µm) were visible in the micrographs (
Figure S2a), but after ultrasonic treatment for 3 h, their size decreased, and the largest particles were 2–12 µm (
Figure S2d–f). No improvement was seen with processing longer than 3 h. After two weeks of storage at room temperature, mold appeared on the surface of the solution 8PVA130(PPa/BRA).
The shelf life of the solution 10PVA130(PHGEx/BRA) (yellow, transparent, clear liquid) is approximately 6 months at room temperature (
Figure S3), after which visual changes are visible. The solution was still spinnable but premixing was needed.
In the solution 8PVA130(PEx/BRA Lab), the propolis particles were of different sizes and required sonication to decompose the propolis agglomerates. A small precipitate of propolis was visible after 1 h, and a clear precipitate of propolis was visible after 60 days (
Figure S4), but spinnability time was only 1 week.
The 10PVA130(PHGEx/BRA+PEx/LV) sample with propolis alcohol extract and hydroglyceric propolis extract spun more stable than without glycerol, and its shelf life was significantly increased (1 week to 3 months (
Figure S3)).
The stability of the solutions and shelf life are summarized in
Table 6. Photographs and optical microscope images of the obtained solutions are shown in
Figures S1–S4.