Anti-Cancer Activity of Buthus occitanus Venom on Hepatocellular Carcinoma in 3D Cell Culture
Abstract
:1. Introduction
2. Results
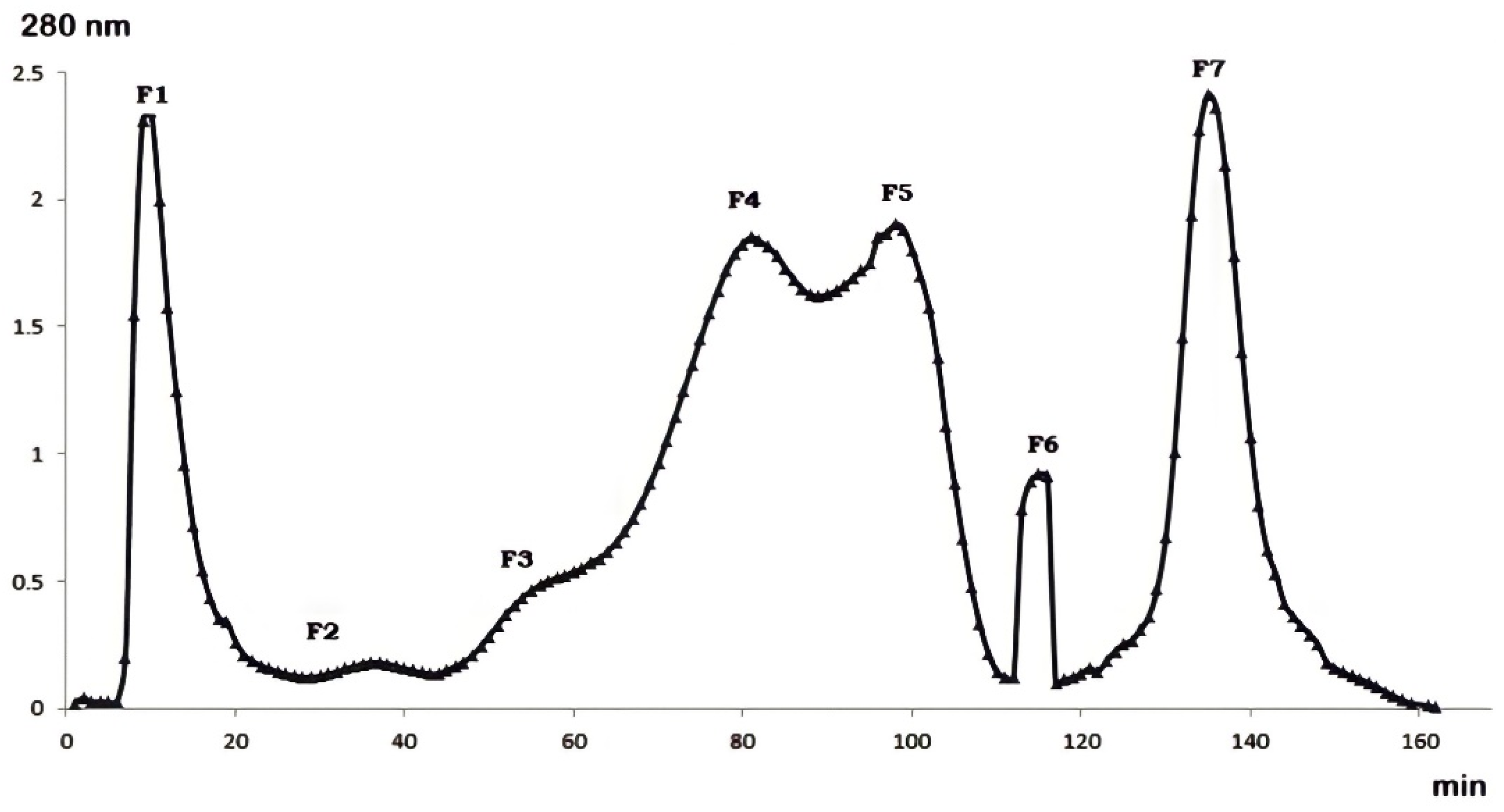
2.1. Gel Filtration Purification of Buthus occitanus Venom
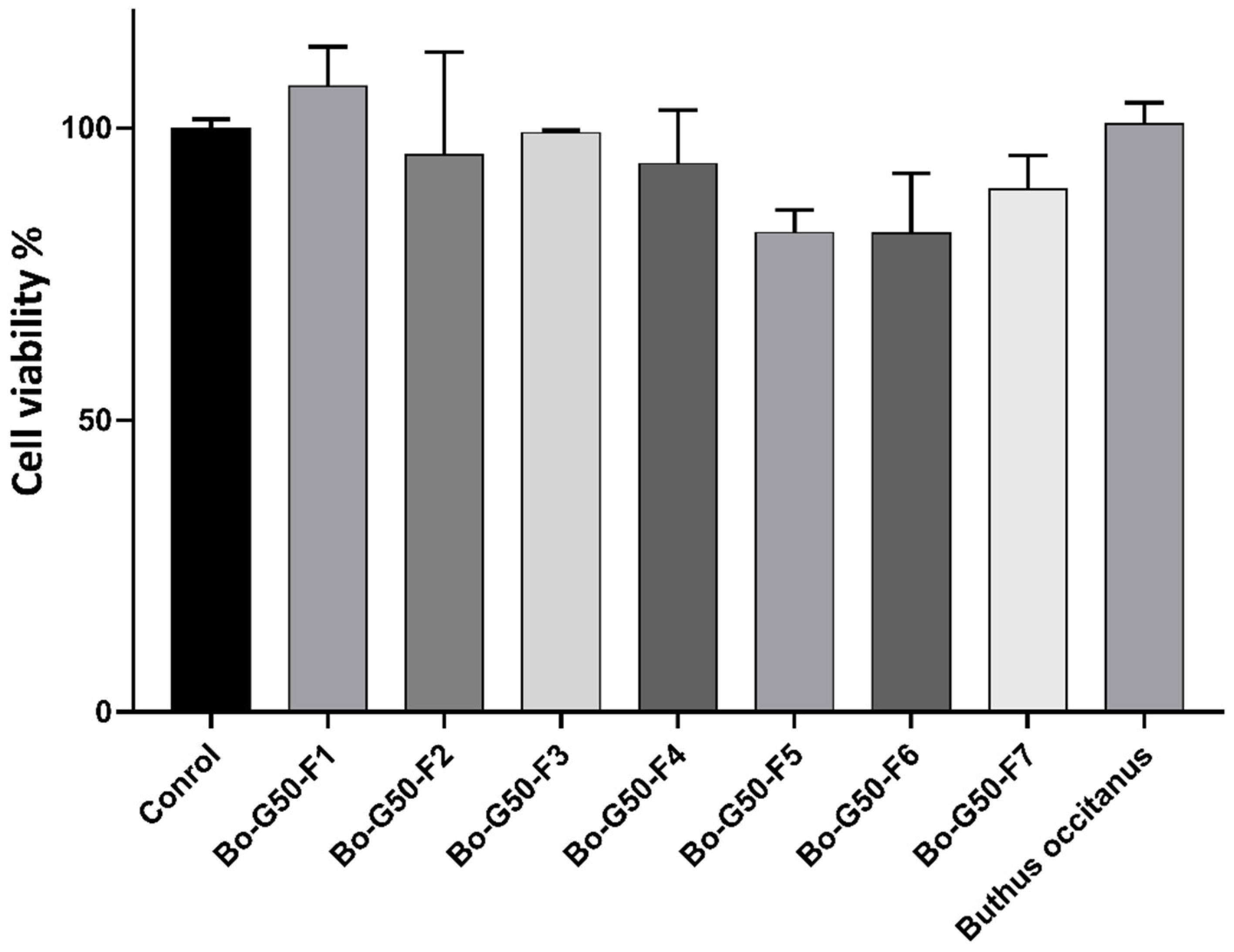
2.2. Cellular Toxicity Test against Fa2N-4 Cells (Normal Hepatocytes)
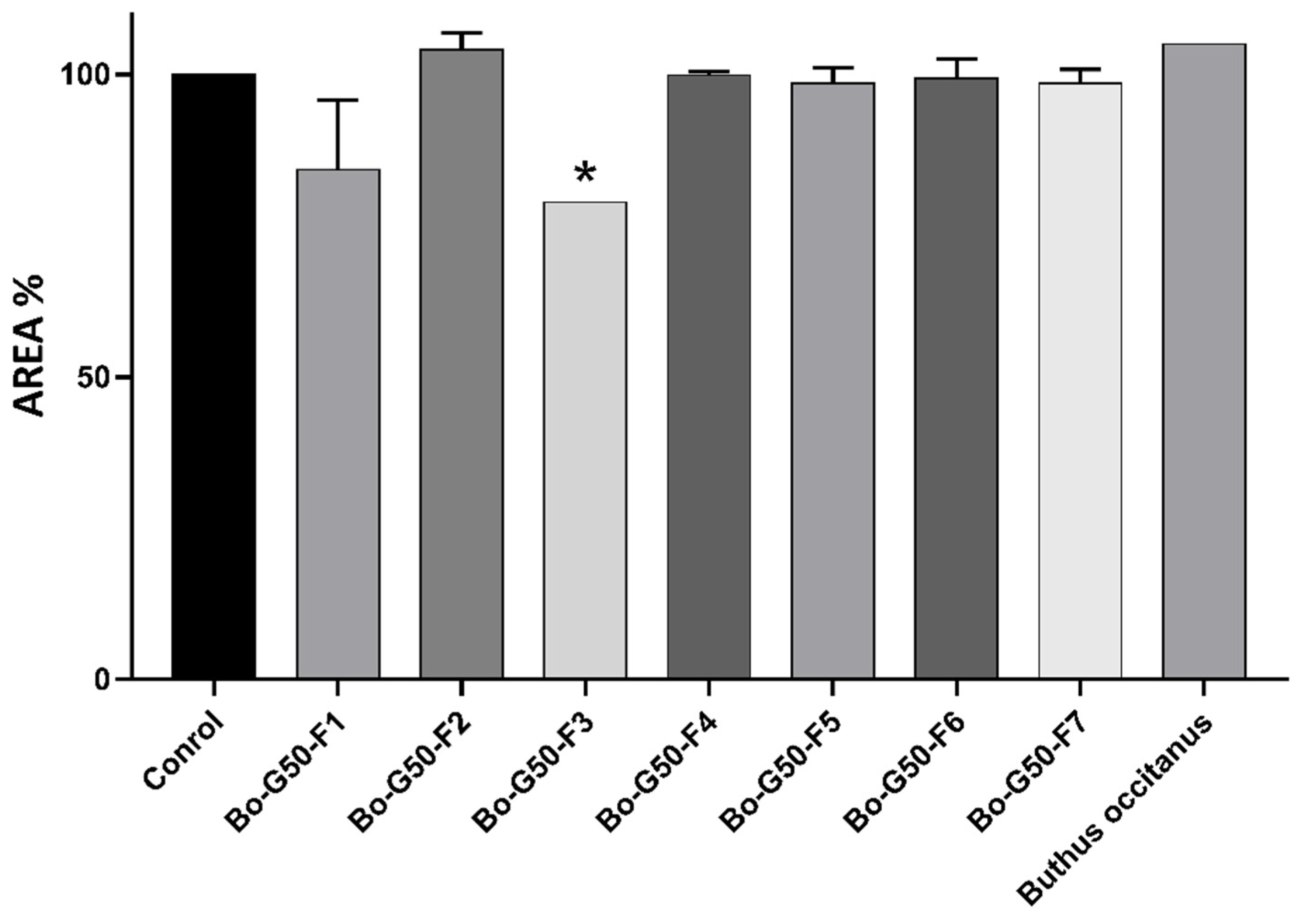
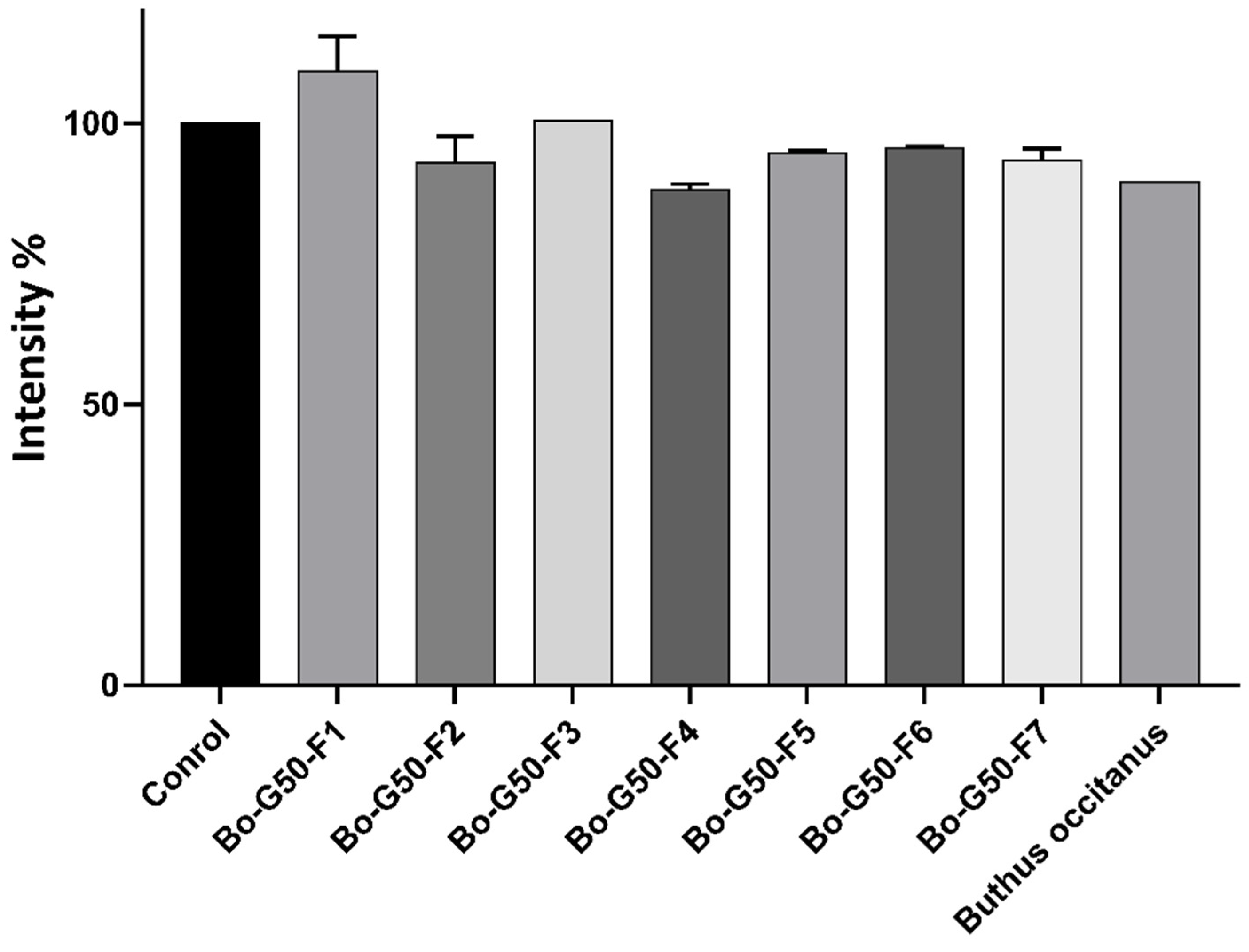
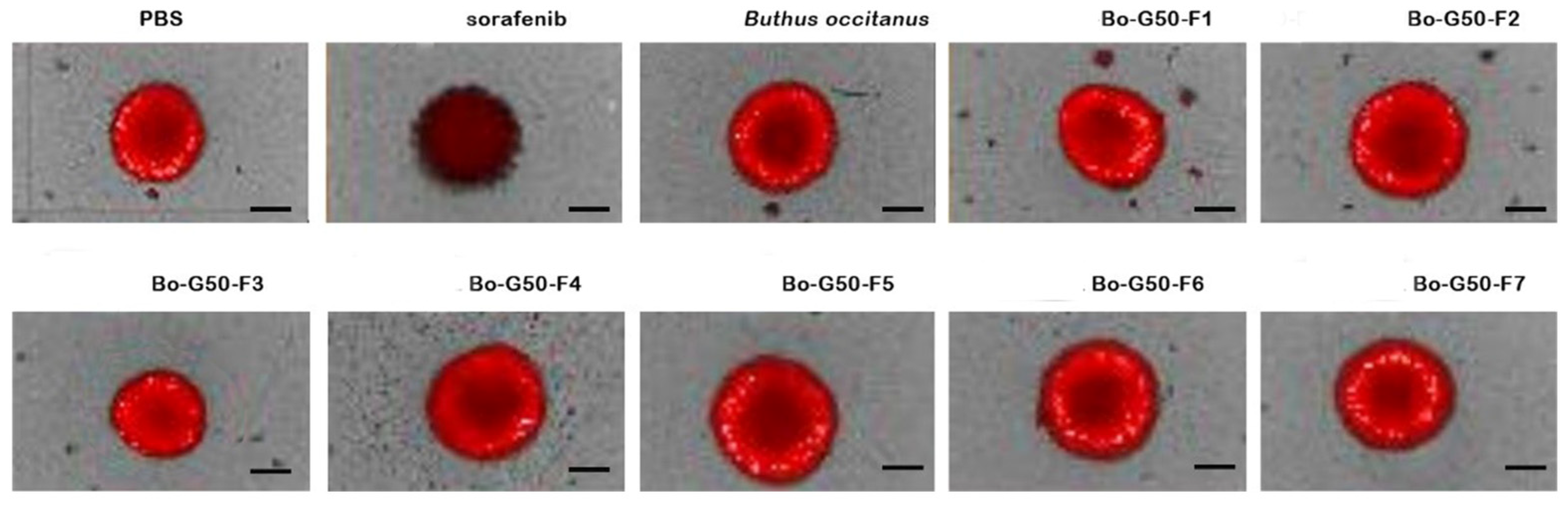
2.3. Cytotoxicity Test of Venoms and Its Fractions against MCTS (Multicellular Tumor Spheroid)
2.4. Characterization of F3 Fraction Using Liquid Chromatography Coupled with Tandem Mass Spectrometry (LC-MS/MS)
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Venom Milking
4.3. Fractionation of Buthus occitanus Venom by Gel Filtration
4.4. Cytotoxicity Assay of Buthus occitanus Venom and Its Fractions against Fa2N-4
4.5. Anticancer Activity of Buthus occitanus Venom and Its Fractions against MCTS
4.6. Characterization of F3 Fraction Using Liquid Chromatography Coupled with Tandem Mass Spectrometry (LC-MS/MS)
4.7. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Global Burden of Disease Cancer Collaboration; Fitzmaurice, C.; Allen, C.; Barber, R.M.; Barregard, L.; Bhutta, Z.A.; Brenner, H.; Dicker, D.J.; Chimed-Orchir, O.; Dandona, R.; et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: A systematic analysis for the global burden of disease study. JAMA Oncol. 2017, 3, 524–548. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, S.; Kudo, M.; Nagai, T.; Inoue, T.; Ueshima, K.; Nishida, N.; Watanabe, T.; Sakurai, T. Activation of JNK and High Expression Level of CD133 Predict a Poor Response to Sorafenib in Hepatocellular Carcinoma. Br. J. Cancer 2012, 106, 1997–2003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Serag, H.B. Epidemiology of Viral Hepatitis and Hepatocellular Carcinoma. Gastroenterology 2012, 142, 1264–1273.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.-W.; Chen, M.; Colombo, M.; Roberts, L.R.; Schwartz, M.; Chen, P.-J.; Kudo, M.; Johnson, P.; Wagner, S.; Orsini, L.S.; et al. Global patterns of hepatocellular carcinoma management from diagnosis to death: The BRIDGE study. Liver Int. 2015, 35, 2155–2166. [Google Scholar] [CrossRef] [Green Version]
- Younossi, Z.M.; Blissett, D.; Blissett, R.; Henry, L.; Stepanova, M.; Younossi, Y.; Racila, A.; Hunt, S.; Beckerman, R. The economic and clinical burden of nonalcoholic fatty liver disease in the united states and europe. Hepatology 2016, 64, 1577–1586. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Otgonsuren, M.; Henry, L.; Venkatesan, C.; Mishra, A.; Erario, M.; Hunt, S. Association of nonalcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009. Hepatology 2015, 62, 1723–1730. [Google Scholar] [CrossRef]
- Welzel, T.M.; Graubard, B.I.; Quraishi, S.; Zeuzem, S.; Davila, J.A.; El-Serag, H.B.; McGlynn, K.A. Population-attributable fractions of risk factors for hepatocellular carcinoma in the United States. Am. J. Gastroenterol. 2013, 108, 1314–1321. [Google Scholar] [CrossRef] [Green Version]
- Feo, F.; De Miglio, M.R.; Simile, M.M.; Muroni, M.R.; Calvisi, D.F.; Frau, M.; Pascale, R.M. hepatocellular carcinoma as a complex polygenic disease. Interpretive analysis of recent developments on genetic predisposition. Biochim. Biophys. Acta 2006, 1765, 126–147. [Google Scholar] [CrossRef]
- Pinyol, R.; Montal, R.; Bassaganyas, L.; Sia, D.; Takayama, T.; Chau, G.-Y.; Mazzaferro, V.; Roayaie, S.; Lee, H.C.; Kokudo, N.; et al. Molecular predictors of prevention of recurrence in HCC with sorafenib as adjuvant treatment and prognostic factors in the phase 3 STORM Trial. Gut 2019, 68, 1065–1075. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.D.; Hainaut, P.; Gores, G.J.; Amadou, A.; Plymoth, A.; Roberts, L.R. A global view of hepatocellular carcinoma: Trends, risk, prevention and management. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 589–604. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmadi, S.; Knerr, J.M.; Argemi, L.; Bordon, K.C.F.; Pucca, M.B.; Cerni, F.A.; Arantes, E.C.; Çalışkan, F.; Laustsen, A.H. Scorpion venom: Detriments and benefits. Biomedicines 2020, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, I.S.; Ferreira, I.G.; Alexandre-Silva, G.M.; Cerni, F.A.; Cremonez, C.M.; Arantes, E.C.; Zottich, U.; Pucca, M.B. Scorpion Toxins Targeting Kv1.3 Channels: Insights into immunosuppression. J. Venom. Anim. Toxins Incl. Trop. Dis. 2019, 25, e148118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ward, M.J.; Ellsworth, S.A.; Nystrom, G.S. A global accounting of medically significant scorpions: Epidemiology, major toxins, and comparative resources in harmless counterparts. Toxicon 2018, 151, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez de la Vega, R.C.; Possani, L.D. Overview of scorpion toxins specific for na+ channels and related peptides: Biodiversity, structure-function relationships and evolution. Toxicon 2005, 46, 831–844. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Harrison, P.L.; Strong, P.N. Snapshots of scorpion venomics. J. Arid Environ. 2015, 112, 170–176. [Google Scholar] [CrossRef]
- Pennington, M.W.; Czerwinski, A.; Norton, R.S. Peptide therapeutics from venom: Current status and potential. Bioorg. Med. Chem. 2018, 26, 2738–2758. [Google Scholar] [CrossRef]
- González, J.A.; Vallejo, J.R. The scorpion in Spanish folk medicine: A review of traditional remedies for stings and its use as a therapeutic resource. J. Ethnopharmacol. 2013, 146, 62–74. [Google Scholar] [CrossRef]
- Santibáñez-López, C.E.; Cid-Uribe, J.I.; Batista, C.V.F.; Ortiz, E.; Possani, L.D. Venom gland transcriptomic and proteomic analyses of the enigmatic scorpion superstitionia donensis (scorpiones: Superstitioniidae), with insights on the evolution of its venom components. Toxins 2016, 8, 367. [Google Scholar] [CrossRef] [Green Version]
- Gomes, A.; Bhattacharjee, P.; Mishra, R.; Biswas, A.K.; Dasgupta, S.C.; Giri, B. Anticancer potential of animal venoms and toxins. Indian J. Exp. Biol. 2010, 48, 93–103. [Google Scholar]
- D’Suze, G.; Rosales, A.; Salazar, V.; Sevcik, C. Apoptogenic Peptides from tityus discrepans scorpion venom acting against the SKBR3 breast cancer cell line. Toxicon 2010, 56, 1497–1505. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Chua, P.-J.; Bay, B.-H.; Gopalakrishnakone, P. Scorpion venoms as a potential source of novel cancer therapeutic compounds. Exp. Biol. Med. 2014, 239, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Srairi-Abid, N.; Othman, H.; Aissaoui, D.; BenAissa, R. Anti-tumoral effect of scorpion peptides: Emerging new cellular targets and signaling pathways. Cell Calcium 2019, 80, 160–174. [Google Scholar] [CrossRef]
- Salem, M.L.; Shoukry, N.M.; Teleb, W.K.; Abdel-Daim, M.M.; Abdel-Rahman, M.A. In vitro and in vivo antitumor effects of the egyptian scorpion androctonus amoreuxi venom in an ehrlich ascites tumor model. Springerplus 2016, 5, 570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Asmari, A.K.; Islam, M.; Al-Zahrani, A.M. In Vitro analysis of the anticancer properties of scorpion venom in colorectal and breast cancer cell lines. Oncol. Lett. 2016, 11, 1256–1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinen, T.E.; Gorini da Veiga, A.B. Arthropod venoms and cancer. Toxicon 2011, 57, 497–511. [Google Scholar] [CrossRef]
- Rapôso, C. Scorpion and spider venoms in cancer treatment: State of the art, challenges, and perspectives. J. Clin. Transl. Res. 2017, 3, 233–249. [Google Scholar] [CrossRef]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2021, 7, 6. [Google Scholar] [CrossRef]
- Llovet, J.M.; De Baere, T.; Kulik, L.; Haber, P.K.; Greten, T.F.; Meyer, T.; Lencioni, R. Locoregional therapies in the era of molecular and immune treatments for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 293–313. [Google Scholar] [CrossRef]
- Daher, S.; Massarwa, M.; Benson, A.A.; Khoury, T. Current and future treatment of hepatocellular carcinoma: An updated comprehensive review. J. Clin. Transl. Hepatol. 2018, 6, 69–78. [Google Scholar] [CrossRef] [Green Version]
- Lewis, R.J.; Garcia, M.L. Therapeutic potential of venom peptides. Nat. Rev. Drug Discov. 2003, 2, 790–802. [Google Scholar] [CrossRef] [PubMed]
- Lv, D.; Hu, Z.; Lu, L.; Lu, H.; Xu, X. Three-dimensional cell culture: A powerful tool in tumor research and drug discovery (review). Oncol. Lett. 2017, 14, 6999–7010. [Google Scholar] [CrossRef] [Green Version]
- Lu, H.; Stenzel, M.H. Multicellular tumor spheroids (MCTS) as a 3D in vitro evaluation tool of nanoparticles. Small 2018, 14, 1702858. [Google Scholar] [CrossRef] [PubMed]
- Lafnoune, A.; Lee, S.-Y.; Heo, J.-Y.; Gourja, I.; Darkaoui, B.; Abdelkafi-Koubaa, Z.; Chgoury, F.; Daoudi, K.; Chakir, S.; Cadi, R.; et al. Anti-cancer effect of Moroccan Cobra Naja Haje venom and its fractions against hepatocellular carcinoma in 3D cell culture. Toxins 2021, 13, 402. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Zhang, G.; Sun, A.; Guo, J.; Tian, Z.; Wang, H.; Liu, Y. Scorpion venom component iii inhibits cell proliferation by modulating NF-ΚB activation in human leukemia cells. Exp. Ther. Med. 2012, 4, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz Alikhani, H.; Bidmeshkipour, A.; Zargan, J. Cytotoxic and apoptotic induction effects of the venom of iranian scorpion (odontobuthus bidentatus) in the hepatocellular carcinoma cell line (HepG2). Int. J. Pept. Res. Ther. 2020, 26, 2475–2484. [Google Scholar] [CrossRef]
- Chen, H.; Zhidan, W.; Xia, R.; Zhaoxia, W.; Qing, J.; Qiang, G.; Haipeng, Y.; Hengxiao, W. Scorpion venom activates natural killer cells in hepatocellular carcinoma via the NKG2D-MICA pathway. Int. Immunopharmacol. 2016, 35, 307–314. [Google Scholar] [CrossRef]
- Lian, W.; Lian, H.; Li, Q.; Hu, A.; Liu, S. The venom of spider haplopelma hainanum suppresses proliferation and induces apoptosis in hepatic cancer cells by caspase activation in vitro. J. Ethnopharmacol. 2018, 225, 169–177. [Google Scholar] [CrossRef]
- Hou, Y.; Zhao, X.; Chen, J.; Zhou, J.; Chen, W.; Mao, H.; Chen, R. Effects of macrothele raven venom on intrarenal invasion and metastasis of H22 liver cancer cells in mice. J. Can. Res. Ther. 2017, 13, 725–729. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Bae, S.K.; Kim, M.; Pyo, M.J.; Kim, M.; Yang, S.; Won, C.; Yoon, W.D.; Han, C.H.; Kang, C.; et al. Anticancer effect of nemopilema nomurai jellyfish venom on HepG2 cells and a tumor xenograft animal model. Evid. Based Complement. Altern. Med. 2017, 2017, e2752716. [Google Scholar] [CrossRef] [Green Version]
- Seydi, E.; Babaei, S.; Fakhri, A.; Pourahmad, J. Selective toxicity of caspian cobra (Naja Oxiana) venom on liver cancer cell mitochondria. Asian Pac. J. Trop. Biomed. 2017, 7, 460–465. [Google Scholar] [CrossRef]
- Mahmoud, G.H.; Saber, S.A.; El-Fiky, A.A.E.-F.; Mohamed, A.F. In vitro evaluation of anticancer potential of echispyramidum venom (viperidae) and related genetic and apoptotic profile alterations. Egypt. J. Hosp. Med. 2019, 76, 3891–3900. [Google Scholar] [CrossRef]
- Prinholato da Silva, C.; Costa, T.R.; Paiva, R.M.A.; Cintra, A.C.O.; Menaldo, D.L.; Antunes, L.M.G.; Sampaio, S.V. Antitumor potential of the myotoxin BthTX-I from bothrops jararacussu snake venom: Evaluation of cell cycle alterations and death mechanisms induced in tumor cell lines. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 44. [Google Scholar] [CrossRef] [PubMed]
- Oukkache, N.; Chgoury, F.; Lalaoui, M.; Cano, A.A.; Ghalim, N. Comparison between two methods of scorpion venom milking in Morocco. J. Venom. Anim. Toxins Incl. Trop. Dis. 2013, 19, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malih, I.; Ahmad Rusmili, M.R.; Tee, T.Y.; Saile, R.; Ghalim, N.; Othman, I. Proteomic analysis of moroccan cobra naja haje legionis venom using tandem mass spectrometry. J. Proteom. 2014, 96, 240–252. [Google Scholar] [CrossRef] [PubMed]
| Protein Name | Molecular Weight (Da) | UniProt ID | Molecular Function | Homology Degree % | Sequence |
|---|---|---|---|---|---|
| Alpha-insect toxin Lqq3 | 7334.93 ± 0.23 | P01487 | Sodium channel inhibitor activity | 91.3 | VRDAYIAKNY NCVYECFRDS YCNDLCTKNG ASSGYCQWAG KYGNACWCYA LPDNVPIRVP GKCH |
| Alpha-like toxin Bom4 | 7287.96 ± 0.37 | P59354 | Sodium channel inhibitor activity | 97.7 | GRDAYIAQPE NCVYECAKNS YCNDLCTKNG AKSGYCQWLG KYGNACWCED LPDNVPIRIP GKCHF |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lafnoune, A.; Lee, S.-Y.; Heo, J.-Y.; Daoudi, K.; Darkaoui, B.; Chakir, S.; Cadi, R.; Mounaji, K.; Shum, D.; Seo, H.-R.; et al. Anti-Cancer Activity of Buthus occitanus Venom on Hepatocellular Carcinoma in 3D Cell Culture. Molecules 2022, 27, 2219. https://doi.org/10.3390/molecules27072219
Lafnoune A, Lee S-Y, Heo J-Y, Daoudi K, Darkaoui B, Chakir S, Cadi R, Mounaji K, Shum D, Seo H-R, et al. Anti-Cancer Activity of Buthus occitanus Venom on Hepatocellular Carcinoma in 3D Cell Culture. Molecules. 2022; 27(7):2219. https://doi.org/10.3390/molecules27072219
Chicago/Turabian StyleLafnoune, Ayoub, Su-Yeon Lee, Jin-Yeong Heo, Khadija Daoudi, Bouchra Darkaoui, Salma Chakir, Rachida Cadi, Khadija Mounaji, David Shum, Haeng-Ran Seo, and et al. 2022. "Anti-Cancer Activity of Buthus occitanus Venom on Hepatocellular Carcinoma in 3D Cell Culture" Molecules 27, no. 7: 2219. https://doi.org/10.3390/molecules27072219
APA StyleLafnoune, A., Lee, S.-Y., Heo, J.-Y., Daoudi, K., Darkaoui, B., Chakir, S., Cadi, R., Mounaji, K., Shum, D., Seo, H.-R., & Oukkache, N. (2022). Anti-Cancer Activity of Buthus occitanus Venom on Hepatocellular Carcinoma in 3D Cell Culture. Molecules, 27(7), 2219. https://doi.org/10.3390/molecules27072219









