Natural Small Molecules in Breast Cancer Treatment: Understandings from a Therapeutic Viewpoint
Abstract
:1. Introduction
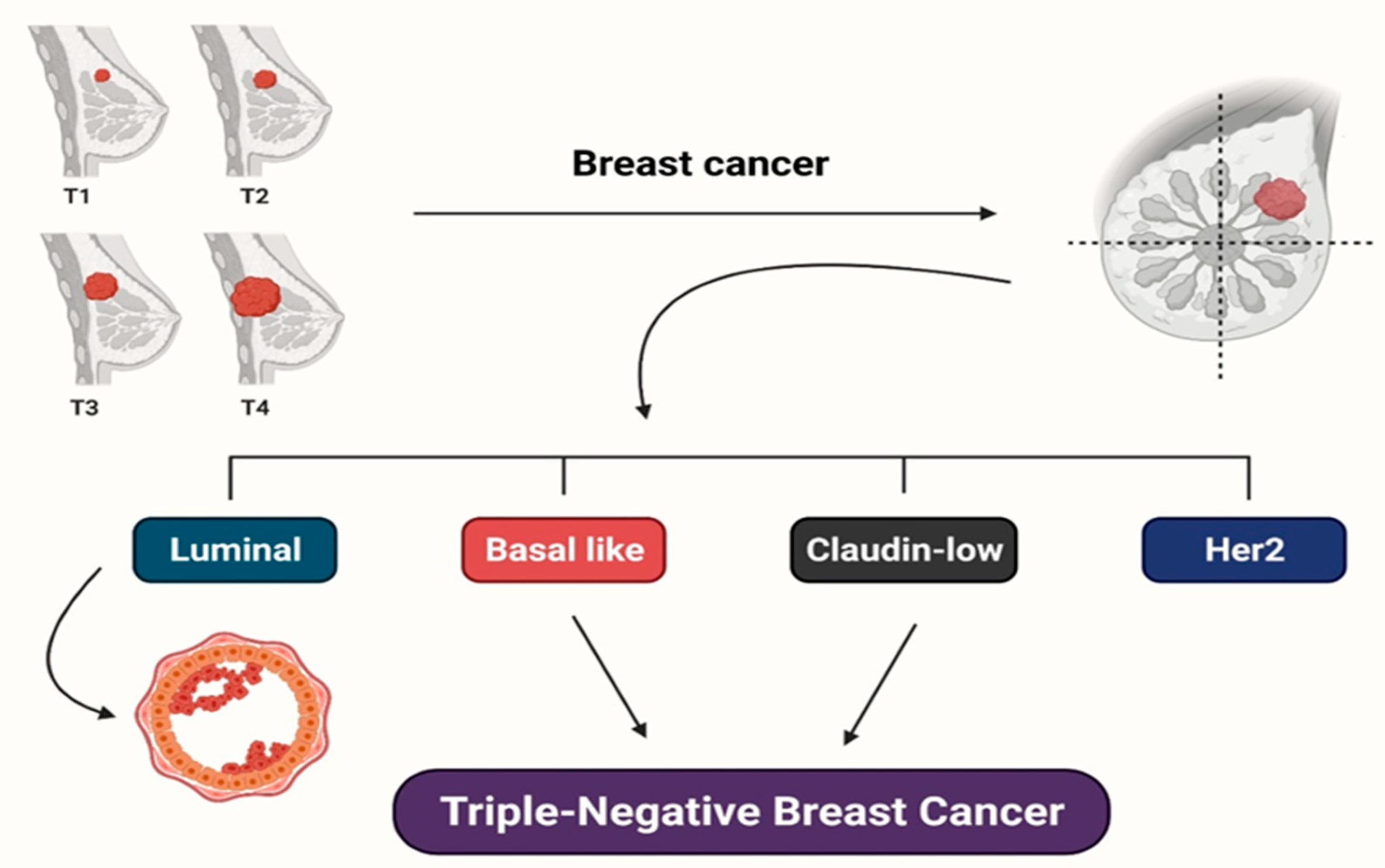
2. Various Subtypes of Breast Cancer
3. Risk Factors for Breast Cancer
4. Mechanisms Inherent in the Clinical Actions of Cholesterol in BrCa
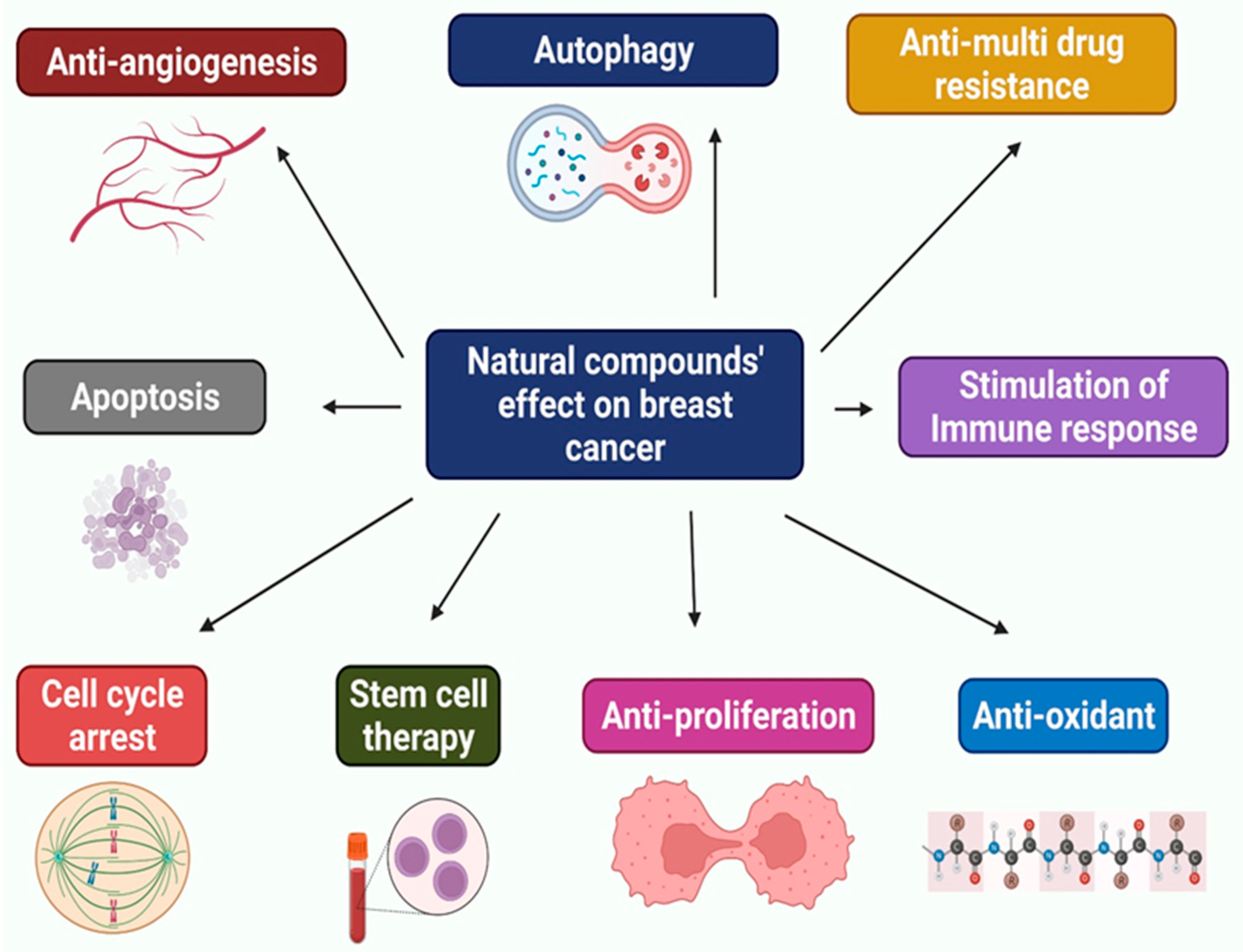
5. Natural Compounds against Breast Cancer
5.1. Quercetin
5.2. Tetrandrine
5.3. Thymoquinone
| Natural Compound and Class | Chemical Formulas | Source | Mechanism of Action | References |
|---|---|---|---|---|
| Quercetin (flavonoid) | C15H10O7 | Allagopappus viscosissimus, Opuntia ficus-indica var. saboten, Lychnophora staavioides, and Rhamnus species | Programmed cell death and the cell cycle are promoted, and breast cancer stem cells (BCSCs) are kept from invasion | [45,53,55,81,82,83,84] |
| Tetrandrine (alkaloid) | C38H42N2O6 | Stephania tetrandra | Blocks positive ion channels, overcomes drug resistance, boosts autophagy, and triggers cell death | [61,62,65] |
| Thymoquinone (4-benzoquinone) | C10H12O2 | Nigella Sativa | Both p53-dependent and p53-independent mechanisms increase apoptosis; cell cycle arrest triggers p38 and ROS signaling; NF-κB is a tumor-suppressing protein. The peroxisome proliferator-activated receptor (PPAR) activation pathway has improved, as has PPAR activity; phosphorylation of Akt, 4E-BP1, eIF4E, S6R, and p70S6K has decreased | [70,72,74,76,78,80] |
| Resveratrol (phytoalexin) | C14H12O3 | Polygonum cuspidatum | Encourages cell cycle arrest and death; prevents carcinogenesis, DNA damage, and cancer spread; Cells genetic and epigenetic profiles are altered, and COX activity is inhibited; NF-κB DNA’s binding activity is reduced, and cell viability, glucose ingesting, and ATP content are all reduced; TGFβ1 expression is suppressed; BCSC survival is reduced; Wnt/β-catenin signaling pathway is inhibited, resulting in autophagy; signaling between PI3K, Akt, and mTOR is suppressed | [85,86,87,88,89,90,91,92] |
| Honokiol (neolignan biphenols) | C18H18O2 | Magnolia grandiflora | Autophosphorylation inhibits angiogenesis, tumor cell proliferation, and programmed cell death; the PI3K/mTOR pathway governs immune resistance; inhibits angiogenesis, tumor cell proliferation, and death; suppresses Wnt1-MTA1-β-catenin signaling induced by leptin; STAT3 phosphorylation is reduced, and phospholipase D (PLD) activity is inhibited; induces cell cycle arrest and decreases mammosphere development, aldehyde dehydrogenases (ALDH) activity, and expression of iPSC inducers; EGFR is inhibited, and c-Src phosphorylation is suppressed | [93,94,95,96,97,98] |
| Garcinol (polyisoprenylated benzophenone) | C38H50O6, | Garcinia indica | Regulates the NF-κB signaling pathway; reduces histone acetyltransferases and ROS; induces cell cycle arrest; reverses EMT markers, and governs the β-catenin and Wnt signaling pathways | [99,100,101,102] |
| Biochanin A (flavonoid) | C16H12O5 | Trifolium pratense | Biochanin A inhibited the aromatase enzyme activity and prevented cell proliferation in MCF-7 cells that had been stably transfected with the CYP19 gene. Biochanin A was reported to reduce aromatase enzyme activity and mRNA expression in SK-BR3 cells (ER-negative BrCa cells) | [103,104,105] |
| Lycopene (tetraterpenoids) | C40H56 | Tomatoes, carrots, watermelon, papaya, and cherries all contain lycopene, a vivid red carotene pigment that belongs to the tetra terpenoids family | BrCa cells regulate several genes involved in DNA repair, cell cycle control, and apoptosis, making them potent antioxidants | [106,107,108] |
| Shikonin (hydroxy-1,4-naphthoquinone) | C16H16O5 | Lithospermum erythrorhizon’s root extract | Shikonin inhibits estrogen-encouraged cell production and initiates ER ubiquitination, promoting ER breakdown in ER-positive breast cells. It induces necroptosis-like death in ER-positive BrCa cells | [109,110] |
| Sulforaphane (isothiocyanate) | C6H11NOS2 | Broccoli, water lily, broccoli sprouts, cabbage, and kale | In BrCa cells, sulforaphane has been shown to prevent tubulin polymerization. It can cause both cell cycle detention and apoptosis in BrCa cells | [111,112] |
| Caffeic acid (phenolic compound) | C9H8O4 | Echinacea purpurea | Echinacea includes flavonoids, which stimulate the immune system. It boosts lymphocyte activity, which encourages macrophage phagocytosis and natural killer cell activity, triggering interferon assembly and minimizing the adverse effects of chemotherapy and radiation therapy. It also helps people extend their life expectancy as their cancer advances. Echinacea juice in commercial formulations has been demonstrated to increase macrophage cytokine production. The activation and proliferation of T-cells and B-cells has fewer apparent implications. Several components of echinacea have been shown to contribute to the immune system’s unique effects | [82,113] |
| Alliin, and Allicin (sulfoxide) | C6H11NO3S, C6H10OS2 | Allium sativum | Garlic’s anticancer benefits come from its high organic sulfides and polysulfides composition. The mechanisms of antitumor activity activating lymphocytes and macrophages are the destruction of malignant cells and interfering with tumor cell metabolism | [114] |
| Curcumin (flavonoid) | C21H20O6 | Curcuma longa | Curcuma longa (turmeric) gives food a dark yellow color. The active element in turmeric, curcumin, can be found in the rhizome and rootstock. Curcumin’s phenolic compounds have been demonstrated to have anticancer properties. Turmeric protects against lung, breast, skin, and stomach cancers | [114,115,116,117] |
| Luteolin (flavonoid) | C15H10O6 | Arctium lappa | Antioxidants of the flavonoid and polyphenol are found in burdock root, suppressing tumor growth. Root extract protects normal physiological cells from toxic substances and helps to prevent cell mutations. The most crucial active element in burdock is tannin, a phenolic substance. It activates macrophages, inhibits cancer spread, and maintains immune-modulatory capacities | [118,119] |
| Carotenoids (Tetraterpenoids) | C40H64 | Rosehips | Carotenoids are potent antioxidants with therapeutic properties, such as scavenging free radicals, protecting cells from oxidative stress, illuminating gap intersections, stimulating the immune system, and regulating enzyme activity, all of which contribute to cancer production and boost the body’s immune system activity | [120] |
| Epigallocatechin gallate (catechin) | C22H18O11 | Camellia sinensis | Green tea possesses cancer-fighting and antimutagenic properties. EGCG protects cells against DNA damage caused by reactive oxygen species. Green tea polyphenols, according to animal studies, inhibit cancer cell division and cause tumor cell necrosis and death | [121,122,123,124] |
5.4. Resveratrol
5.5. Honokiol
5.6. Garcinol
5.7. Biochanin A
5.8. Lycopene
5.9. Shikonin
5.10. Sulforaphane
5.11. Echinacea
5.12. Garlic
5.13. Turmeric
5.14. Burdock
5.15. Carotenoids
5.16. Green Tea
6. Combination Effects
7. Future Prospect of Herbal Management
8. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, Y.; Li, S.; Meng, X.; Gan, R.Y.; Zhang, J.J.; Bin, H.L. Dietary natural products for prevention and treatment of breast cancer. Nutrients 2017, 9, 728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strunk, M.A.; Zopf, E.M.; Steck, J.; Hamacher, S.; Hallek, M.; Baumann, F.T. Effects of kyusho jitsu on physical activity-levels and quality of life in breast cancer patients. In Vivo 2018, 32, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA. Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamaruzman, N.I.; Tiash, S.; Ashaie, M.; Chowdhury, E.H. siRNAs targeting growth factor receptor and anti-apoptotic genes synergistically kill breast cancer cells through inhibition of MAPK and PI-3 kinase pathways. Biomedicines 2018, 6, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, E.-Y.; Moon, A. Natural Products for Chemoprevention of Breast Cancer. J. Cancer Prev. 2015, 20, 223–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitra, S.; Lami, M.S.; Ghosh, A.; Das, R.; Tallei, T.E.; Islam, F.; Dhama, K.; Begum, M.Y.; Aldahish, A.; Chidambaram, K. Hormonal Therapy for Gynecological Cancers: How Far Has Science Progressed toward Clinical Applications? Cancers 2022, 14, 759. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Singh, S.; Wlillard, J.; Singh, R. Drug delivery approaches for breast cancer. Int. J. Nanomed. 2017, 12, 6205–6218. [Google Scholar] [CrossRef] [Green Version]
- Ouyang, L.; Luo, Y.; Tian, M.; Zhang, S.Y.; Lu, R.; Wang, J.H.; Kasimu, R.; Li, X. Plant natural products: From traditional compounds to new emerging drugs in cancer therapy. Cell Prolif. 2014, 47, 506–515. [Google Scholar] [CrossRef]
- Rahman, M.; Islam, F.; Afsana Mim, S.; Khan, M.; Islam, M.; Haque, M.; Mitra, S.; Emran, T.B.; Rauf, A. Multifunctional Therapeutic Approach of Nanomedicines against Inflammation in Cancer and Aging. J. Nanomater. 2022, 2022, 4217529. [Google Scholar] [CrossRef]
- Ijaz, S.; Akhtar, N.; Khan, M.S.; Hameed, A.; Irfan, M.; Arshad, M.A.; Ali, S.; Asrar, M. Plant derived anticancer agents: A green approach towards skin cancers. Biomed. Pharmacother. 2018, 103, 1643–1651. [Google Scholar] [CrossRef]
- Aung, T.N.; Qu, Z.; Kortschak, R.D.; Adelson, D.L. Understanding the effectiveness of natural compound mixtures in cancer through their molecular mode of action. Int. J. Mol. Sci. 2017, 18, 656. [Google Scholar] [CrossRef] [PubMed]
- Noel, B.; Singh, S.K.; Lillard, J.W.; Singh, R. Role of natural compounds in preventing and treating breast cancer. Front. Biosci. 2020, 12, 137–160. [Google Scholar] [CrossRef]
- Rauf, A.; Abu-Izneid, T.; Khalil, A.A.; Imran, M.; Shah, Z.A.; Emran, T.B.; Mitra, S.; Khan, Z.; Alhumaydhi, F.A.; Aljohani, A.S.; et al. Berberine as a potential anticancer agent: A comprehensive review. Molecules 2021, 26, 7368. [Google Scholar] [CrossRef] [PubMed]
- Nagini, S. Breast cancer: Current molecular therapeutic targets and new players. Anti-Cancer Agents Med. Chem. 2017, 17, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; Van De Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Sørlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; Van De Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 11, 19. [Google Scholar] [CrossRef] [Green Version]
- Prat, A.; Parker, J.S.; Karginova, O.; Fan, C.; Livasy, C.; Herschkowitz, J.I.; He, X.; Perou, C.M. Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res. 2010, 12, R68. [Google Scholar] [CrossRef] [Green Version]
- Herschkowitz, J.I.; Simin, K.; Weigman, V.J.; Mikaelian, I.; Usary, J.; Hu, Z.; Rasmussen, K.E.; Jones, L.P.; Assefnia, S.; Chandrasekharan, S.; et al. Identification of conserved gene expression features between murine mammary carcinoma models and human breast tumors. Genome Biol. 2007, 8, R76. [Google Scholar] [CrossRef] [Green Version]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef] [Green Version]
- Mitra, S. MicroRNA Therapeutics in Triple Negative Breast Cancer. Arch. Pathol. Clin. Res. 2017, 1, 009–017. [Google Scholar] [CrossRef]
- Shah, R.; Rosso, K.; Pathogenesis, N.S.D. Prevention, diagnosis and treatment of breast cancer. World J. Clin. Oncol. 2014, 10, 5. [Google Scholar]
- Petracci, E.; Decarli, A.; Schairer, C.; Pfeiffer, R.M.; Pee, D.; Masala, G.; Palli, D.; Gail, M.H. Risk factor modification and projections of absolute breast cancer risk. J. Natl. Cancer Inst. 2011, 103, 1037–1048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bianchini, F.; Kaaks, R.; Overweight, V.H. Obesity, and cancer risk. Lancet Oncol. 2002, 1, 9. [Google Scholar]
- Capasso, I.; Esposito, E.; Pentimalli, F.; Crispo, A.; Montella, M.; Grimaldi, M.; De Marco, M.; Cavalcanti, E.; D’Aiuto, M.; Fucito, A.; et al. Metabolic syndrome affects breast-cancer risk in postmenopausal women: National Cancer Institute of Naples experience. Cancer Biol. Ther. 2010, 10, 1240–1243. [Google Scholar] [CrossRef] [Green Version]
- Renehan, A.G.; Roberts, D.L.; Dive, C. Obesity and cancer: Pathophysiological and biological mechanisms. Arch. Physiol. Biochem. 2008, 114, 71–83. [Google Scholar] [CrossRef]
- Mitra, S.; Rauf, A.; Tareq, A.M.; Jahan, S.; Emran, T.B.; Shahriar, T.G.; Dhama, K.; Alhumaydhi, F.A.; Aljohani, A.S.; Rebezov, M.; et al. Potential health benefits of carotenoid lutein: An updated review. Food Chem. Toxicol. 2021, 154, 112328. [Google Scholar] [CrossRef]
- Must, A.; Spadano, J.; Coakley, E.H.; Field, A.E.; Colditz, G.; Dietz, W.H. The disease burden associated with overweight and obesity. J. Am. Med. Assoc. 1999, 282, 1523–1529. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Prevalence of abnormal lipid levels among youths—United States, 1999–2006. MMWR. Morb. Mortal. Wkly. Rep. 2010, 59, 29–33. [Google Scholar]
- Gostynski, M.; Gutzwiller, F.; Kuulasmaa, K.; Döring, A.; Ferrario, M.; Grafnetter, D.; Pajak, A. Analysis of the relationship between total cholesterol, age, body mass index among males and females in the WHO MONICA Project. Int. J. Obes. 2004, 28, 1082–1090. [Google Scholar] [CrossRef] [Green Version]
- Nelson, E.R.; Chang, C.Y.; Cholesterol, M.D.P. Cholesterol and breast cancer pathophysiology. Trends Endocrinol. Metab. 2014, 25, 649–655. [Google Scholar] [CrossRef] [Green Version]
- Das, A.; Brown, M.S.; Anderson, D.D.; Goldstein, J.L.; Radhakrishnan, A. Three pools of plasma membrane cholesterol and their relation to cholesterol homeostasis. eLife 2014, 3, e02882. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Wu, J.; Goldstein, J.L.; Brown, M.S.; Hobbs, H.H. Structure of the human gene encoding sterol regulatory element binding protein-1 (SREBF1) and localization of SREBF1 and SREBF2 to chromosomes 17p11.2 and 22q13. Genomics 1995, 25, 667–673. [Google Scholar] [CrossRef]
- Cheskis, B.J.; Greger, J.G.; Nagpal, S.; Freedman, L.P. Signaling by estrogens. J. Cell. Physiol. 2007, 213, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, A.; Ikeda, Y.; Hyock, J.K.; Brown, M.S.; Goldstein, J.L. Sterol-regulated transport of SREBPs from endoplasmic reticulum to Golgi: Oxysterols block transport by binding to Insig. Proc. Natl. Acad. Sci. USA 2007, 104, 6511–6518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szanto, A.; Benko, S.; Szatmari, I.; Balint, B.L.; Furtos, I.; Rühl, R.; Molnar, S.; Csiba, L.; Garuti, R.; Calandra, S.; et al. Transcriptional Regulation of Human CYP27 Integrates Retinoid, Peroxisome Proliferator-Activated Receptor, and Liver X Receptor Signaling in Macrophages. Mol. Cell. Biol. 2004, 24, 8154–8166. [Google Scholar] [CrossRef] [Green Version]
- Zelcer, N.; Hong, C.; Boyadjian, R.; Tontonoz, P. LXR regulates cholesterol uptake through idol-dependent ubiquitination of the LDL receptor. Science 2009, 325, 100–104. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Reue, K.; Fong, L.G.; Young, S.G.; Tontonoz, P. Feedback regulation of cholesterol uptake by the LXR-IDOL-LDLR axis. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2541–2546. [Google Scholar] [CrossRef] [Green Version]
- Bensinger, S.J.; Bradley, M.N.; Joseph, S.B.; Zelcer, N.; Janssen, E.M.; Hausner, M.A.; Shih, R.; Parks, J.S.; Edwards, P.A.; Jamieson, B.D.; et al. LXR Signaling Couples Sterol Metabolism to Proliferation in the Acquired Immune Response. Cell 2008, 134, 97–111. [Google Scholar] [CrossRef] [Green Version]
- Nelson, E.R.; Wardell, S.E.; Jasper, J.S.; Park, S.; Suchindran, S.; Howe, M.K.; Carver, N.J.; Pillai, R.V.; Sullivan, P.M.; Sondhi, V.; et al. 27-Hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science 2013, 342, 1094–1098. [Google Scholar] [CrossRef] [Green Version]
- Pommier, A.J.C.; Dufour, J.; Alves, G.; Viennois, E.; De Boussac, H.; Trousson, A.; Volle, D.H.; Caira, F.; Val, P.; Arnaud, P.; et al. Liver X Receptors Protect from Development of Prostatic Intra-Epithelial Neoplasia in Mice. PLoS Genet. 2013, 9, e1003483. [Google Scholar] [CrossRef] [Green Version]
- Alikhani, N.; Ferguson, R.D.; Novosyadlyy, R.; Gallagher, E.J.; Scheinman, E.J.; Yakar, S.; Leroith, D. Mammary tumor growth and pulmonary metastasis are enhanced in a hyperlipidemic mouse model. Oncogene 2013, 32, 961–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, S.; Nelson, E.R. 27-Hydroxycholesterol, an endogenous selective estrogen receptor modulator. Maturitas 2017, 104, 29–35. [Google Scholar] [CrossRef] [PubMed]
- DuSell, C.D.; Umetani, M.; Shaul, P.W.; Mangelsdorf, D.J.; McDonnell, D.P. 27-hydroxycholesterol is an endogenous selective estrogen receptor modulator. Mol. Endocrinol. 2008, 22, 65–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaffe, R.B. 27-Hydroxycholesterol is an endogenous SERM that inhibits the cardiovascular effects of estrogen. Obstet. Gynecol. Surv. 2008, 63, 160–161. [Google Scholar] [CrossRef] [Green Version]
- Umetani, M.; Domoto, H.; Gormley, A.K.; Yuhanna, I.S.; Cummins, C.L.; Javitt, N.B.; Korach, K.S.; Shaul, P.W.; Mangelsdorf, D.J. 27-Hydroxycholesterol is an endogenous SERM that inhibits the cardiovascular effects of estrogen. Nat. Med. 2007, 13, 1185–1192. [Google Scholar] [CrossRef]
- Nelson, E.R.; DuSell, C.D.; Wang, X.; Howe, M.K.; Evans, G.; Michalek, R.D.; Umetani, M.; Rathmell, J.C.; Khosla, S.; Gesty-Palmer, D.; et al. The oxysterol, 27-hydroxycholesterol, links cholesterol metabolism to bone homeostasis through its actions on the estrogen and liver X receptors. Endocrinology 2011, 152, 4691–4705. [Google Scholar] [CrossRef]
- DuSell, C.D.; Nelson, E.R.; Wang, X.; Abdo, J.; Mödder, U.I.; Umetani, M.; Gesty-Palmer, D.; Javitt, N.B.; Khosla, S.; McDonnell, D.P. The endogenous selective estrogen receptor modulator 27-hydroxycholesterol is a negative regulator of bone homeostasis. Endocrinology 2010, 151, 3675–3685. [Google Scholar] [CrossRef] [Green Version]
- Umetani, M.; Ghosh, P.; Ishikawa, T.; Umetani, J.; Ahmed, M.; Mineo, C.; Shaul, P.W. The cholesterol metabolite 27-hydroxycholesterol promotes atherosclerosis via proinflammatory processes mediated by estrogen receptor alpha. Cell Metab. 2014, 20, 172–182. [Google Scholar] [CrossRef] [Green Version]
- Nachtergaele, S.; Mydock, L.K.; Krishnan, K.; Rammohan, J.; Schlesinger, P.H.; Covey, D.F.; Rohatgi, R. Oxysterols are allosteric activators of the oncoprotein Smoothened. Nat. Chem. Biol. 2012, 8, 211–220. [Google Scholar] [CrossRef] [Green Version]
- Villablanca, E.J.; Raccosta, L.; Zhou, D.; Fontana, R.; Maggioni, D.; Negro, A.; Sanvito, F.; Ponzoni, M.; Valentinis, B.; Bregni, M.; et al. Tumor-mediated liver X receptor-α activation inhibits CC chemokine receptor-7 expression on dendritic cells and dampens antitumor responses. Nat. Med. 2010, 16, 98–105. [Google Scholar] [CrossRef]
- Rauf, A.; Shariati, M.A.; Imran, M.; Bashir, K.; Khan, S.A.; Mitra, S.; Emran, T.B.; Badalova, K.; Uddin, M.; Mubarak, M.S.; et al. Comprehensive review on naringenin and naringin polyphenols as a potent anticancer agent. Environ. Sci. Pollut. Res. 2022, 2022, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Aghapour, F.; Moghadamnia, A.A.; Nicolini, A.; Kani, S.N.M.; Barari, L.; Morakabati, P.; Rezazadeh, L.; Kazemi, S. Quercetin conjugated with silica nanoparticles inhibits tumor growth in MCF-7 breast cancer cell lines. Biochem. Biophys. Res. Commun. 2018, 500, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhu, F.; Lubet, R.A.; De Luca, A.; Grubbs, C.; Ericson, M.E.; D’Alessio, A.; Normanno, N.; Dong, Z.; Bode, A.M. Quercetin-3-methyl ether inhibits lapatinib-sensitive and -resistant breast cancer cell growth by inducing G2/M arrest and apoptosis. Mol. Carcinog. 2013, 52, 134–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef] [Green Version]
- Manouchehri, J.M.; Turner, K.A.; Kalafatis, M. TRAIL-induced apoptosis in TRAIL-resistant breast carcinoma through quercetin cotreatment. Breast Cancer Basic Clin. Res. 2018, 12, 1178223417749855. [Google Scholar] [CrossRef]
- Patra, A.; Satpathy, S.; Shenoy, A.K.; Bush, J.A.; Kazi, M.; Hussain, M.D. Formulation and evaluation of mixed polymeric micelles of quercetin for treatment of breast, ovarian, and multidrug resistant cancers. Int. J. Nanomed. 2018, 13, 2869–2881. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.; Yang, L.; Li, S.; Ye, D.; Yang, L.; Liu, Q.; Zhao, Z.; Cai, Q.; Tan, J.; Li, X. Quercetin inhibits breast cancer stem cells via downregulation of aldehyde dehydrogenase 1A1 (ALDH1A1), chemokine receptor type 4 (CXCR4), mucin 1 (MUC1), and epithelial cell adhesion molecule (EpCAM). Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 412–420. [Google Scholar] [CrossRef] [Green Version]
- Manouchehri, J.M.; Kalafatis, M.; Lindner, D. Abstract 1295: Evaluation of the efficacy of TRAIL plus quercetin as a potential breast carcinoma therapeutic. Cancer Res. 2016, 76 (Suppl. S14). [Google Scholar] [CrossRef]
- Srinivasan, A.; Thangavel, C.; Liu, Y.; Shoyele, S.; Den, R.B.; Selvakumar, P.; Lakshmikuttyamma, A. Quercetin regulates β-catenin signaling and reduces the migration of triple negative breast cancer. Mol. Carcinog. 2016, 55, 743–756. [Google Scholar] [CrossRef]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef]
- Lan, J.; Huang, L.; Lou, H.; Chen, C.; Liu, T.; Hu, S.; Yao, Y.; Song, J.; Luo, J.; Liu, Y.; et al. Design and synthesis of novel C14-urea-tetrandrine derivatives with potent anti-cancer activity. Eur. J. Med. Chem. 2018, 143, 1968–1980. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Debeb, B.G.; Lacerda, L.; Li, J.; Woodward, W.A. Tetrandrine, a compound common in Chinese traditional medicine, preferentially kills breast cancer tumor initiating cells (TICs) in vitro. Cancers 2011, 3, 2274–2285. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Zhang, R.; Wang, Y.; Jing, W.; Liu, Y.; Ma, Y.; Sun, B.; Wang, M.; Chen, P.; Liu, H.; et al. Reduction-sensitive Paclitaxel Prodrug Self-assembled Nanoparticles with Tetrandrine Effectively Promote Synergistic Therapy Against Drug-sensitive and Multidrug-resistant Breast Cancer. Mol. Pharm. 2017, 14, 3628–3635. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Chen, X.Y. Tetrandrine reversed the resistance of tamoxifen in human breast cancer MCF-7/TAM cells: An experimental research. Zhongguo Zhong Xi Yi Jie He Za Zhi Zhongguo Zhongxiyi Jiehe Zazhi = Chin. J. Integr. Tradit. West. Med. 2013, 33, 488–491. [Google Scholar]
- Wong, V.K.W.; Zeng, W.; Chen, J.; Yao, X.J.; Leung, E.L.H.; Wang, Q.Q.; Chiu, P.; Ko, B.C.B.; Law, B.Y.K. Tetrandrine, an activator of autophagy, induces autophagic cell death via PKC-α inhibition and mTOR-dependent mechanisms. Front. Pharmacol. 2017, 8, 351. [Google Scholar] [CrossRef] [Green Version]
- Antonescu, A.-I.; Miere, F.; Fritea, L.; Ganea, M.; Zdrinca, M.; Dobjanschi, L.; Antonescu, A.; Vicas, S.I.; Bodog, F.; Sindhu, R.K. Perspectives on the combined effects of Ocimum basilicum and Trifolium pratense extracts in terms of phytochemical profile and pharmacological effects. Plants 2021, 10, 1390. [Google Scholar] [CrossRef]
- Kuo, P.L.; Lin, C.C. Tetrandrine-induced cell cycle arrest and apoptosis in Hep G2 cells. Life Sci. 2003, 73, 243–252. [Google Scholar] [CrossRef]
- Chen, T.; Ji, B.; Chen, Y. Tetrandrine triggers apoptosis and cell cycle arrest in human renal cell carcinoma cells. J. Nat. Med. 2014, 68, 46–52. [Google Scholar] [CrossRef]
- Lee, D.E.; Lee, K.W.; Song, N.R.; Seo, S.K.; Heo, Y.S.; Kang, N.J.; Bode, A.M.; Lee, H.J.; Dong, Z. 7,3′,4′-trihydroxyisoflavone inhibits epidermal growth factor-induced proliferation and transformation of JB6 P+ mouse epidermal cells by suppressing cyclin-dependent kinases and phosphatidylinositol 3-kinase. J. Biol. Chem. 2010, 285, 21458–21466. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, S.; Ahir, M.; Patra, P.; Mukherjee, S.; Ghosh, S.; Mazumdar, M.; Chattopadhyay, S.; Das, T.; Chattopadhyay, D.; Adhikary, A. PEGylated-thymoquinone-nanoparticle mediated retardation of breast cancer cell migration by deregulation of cytoskeletal actin polymerization through miR-34a. Biomaterials 2015, 51, 91–107. [Google Scholar] [CrossRef]
- Dehghani, H.; Hashemi, M.; Entezari, M.; Mohsenifar, A. The comparison of anticancer activity of thymoquinone and nanothymoquinone on human breast adenocarcinoma. Iran. J. Pharm. Res. 2015, 14, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Motaghed, M.; Muti Al-Hassan, F.; Hamid, S.S. Thymoquinone regulates gene expression levels in the estrogen metabolic and interferon pathways in MCF7 breast cancer cells. Int. J. Mol. Med. 2014, 33, 8–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Odeh, F.; Ismail, S.I.; Abu-Dahab, R.; Mahmoud, I.S.; Al Bawab, A. Thymoquinone in liposomes: A study of loading efficiency and biological activity towards breast cancer. Drug Deliv. 2012, 19, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Woo, C.C.; Hsu, A.; Kumar, A.P.; Sethi, G.; Tan, K.H.B. Thymoquinone Inhibits Tumor Growth and Induces Apoptosis in a Breast Cancer Xenograft Mouse Model: The Role of p38 MAPK and ROS. PLoS ONE 2013, 8, e75356. [Google Scholar] [CrossRef] [Green Version]
- Vicas, S.I.; Cavalu, S.; Laslo, V.; Tocai, M.; Costea, T.O.; Moldovan, L. Growth, photosynthetic pigments, phenolic, glucosinolates content and antioxidant capacity of broccoli sprouts in response to nanoselenium particles supply. Not. Bot. Horti Agrobot. Cluj-Napoca 2019, 47, 821–828. [Google Scholar] [CrossRef]
- Motaghed, M.; Al-Hassan, F.M.; Hamid, S.S. Cellular responses with thymoquinone treatment in human breast cancer cell line MCF-7. Pharmacogn. Res. 2013, 5, 200–206. [Google Scholar] [CrossRef] [Green Version]
- Sutton, K.M.; Greenshields, A.L.; Hoskin, D.W. Thymoquinone, a bioactive component of black caraway seeds, causes G1 phase cell cycle arrest and apoptosis in triple-negative breast cancer cells with mutant p53. Nutr. Cancer 2014, 66, 408–418. [Google Scholar] [CrossRef]
- Woo, C.C.; Loo, S.Y.; Gee, V.; Yap, C.W.; Sethi, G.; Kumar, A.P.; Benny Tan, K.H. Anticancer activity of thymoquinone in breast cancer cells: Possible involvement of PPAR-γ pathway. Biochem. Pharmacol. 2011, 82, 464–475. [Google Scholar] [CrossRef]
- Arafa, E.S.A.; Zhu, Q.; Shah, Z.I.; Wani, G.; Barakat, B.M.; Racoma, I.; El-Mahdy, M.A.; Wani, A.A. Thymoquinone up-regulates PTEN expression and induces apoptosis in doxorubicin-resistant human breast cancer cells. Mutat. Res./Fundam. Mol. Mech. Mutagenes. 2011, 706, 28–35. [Google Scholar] [CrossRef] [Green Version]
- Rajput, S.; Kumar, B.N.P.; Dey, K.K.; Pal, I.; Parekh, A.; Mandal, M. Molecular targeting of Akt by thymoquinone promotes G1 arrest through translation inhibition of cyclin D1 and induces apoptosis in breast cancer cells. Life Sci. 2013, 93, 783–790. [Google Scholar] [CrossRef]
- Rahman, M.; Islam, F.; Saidur Rahaman, M.; Sultana, N.A.; Fahim, N.F.; Ahmed, M. Studies on the prevalence of HIV/AIDS in Bangladesh including other developing countries. Adv. Tradit. Med. 2021, 2021, 1–12. [Google Scholar] [CrossRef]
- Rahman, M.; Majumder, S.; Akter, F.; Islam, F.; Shahriar, M.; Alam, J. Pre-clinical investigation of analgesic, anti-diarrheal and CNS depressant effect of Pterocarpus indicus in Swiss albino mice. Jordan J. Pharm. Sci. 2021, 14, 85–94. [Google Scholar]
- Deng, X.H.; Song, H.Y.; Zhou, Y.F.; Yuan, G.Y.; Zheng, F.J. Effects of quercetin on the proliferation of breast cancer cells and expression of survivin in vitro. Exp. Ther. Med. 2013, 6, 1155–1158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahaman, M.M.; Rakib, A.; Mitra, S.; Tareq, A.M.; Emran, T.B.; Shahid-Ud-daula, A.F.M.; Amin, M.N.; Simal-Gandara, J. The genus curcuma and inflammation: Overview of the pharmacological perspectives. Plants 2020, 10, 63. [Google Scholar] [CrossRef]
- Carter, L.G.; D’Orazio, J.A.; Pearson, K.J. Resveratrol and cancer: Focus on in vivo evidence. Endocr. Relat. Cancer 2014, 21, R209–R225. [Google Scholar] [CrossRef] [Green Version]
- Kala, R.; Shah, H.N.; Martin, S.L.; Epigenetic-based, T.T. Combinatorial resveratrol and ptero-stilbene alters DNA damage response by affecting SIRT1 and DNMT enzyme expression. Incl. SIRT 2015, 15, 672. [Google Scholar] [CrossRef] [Green Version]
- Chin, Y.T.; Hsieh, M.T.; Yang, S.H.; Tsai, P.W.; Wang, S.H.; Wang, C.C.; Lee, Y.S.; Cheng, G.Y.; HuangFu, W.C.; London, D.; et al. Anti-proliferative and gene expression actions of resveratrol in breast cancer cells in vitro. Oncotarget 2014, 5, 12891–12907. [Google Scholar] [CrossRef] [Green Version]
- Lee-Chang, C.; Bodogai, M.; Martin-Montalvo, A.; Wejksza, K.; Sanghvi, M.; Moaddel, R.; de Cabo, R.; Biragyn, A. Inhibition of Breast Cancer Metastasis by Resveratrol-Mediated Inactivation of Tumor-Evoked Regulatory B Cells. J. Immunol. 2013, 191, 4141–4151. [Google Scholar] [CrossRef]
- Gomez, L.S.; Zancan, P.; Marcondes, M.C.; Ramos-Santos, L.; Meyer-Fernandes, J.R.; Sola-Penna, M.; Da Silva, D. Resveratrol decreases breast cancer cell viability and glucose metabolism by inhibiting 6-phosphofructo-1-kinase. Biochimie 2013, 95, 1336–1343. [Google Scholar] [CrossRef] [Green Version]
- Venkatadri, R.; Muni, T.; Iyer, A.K.V.; Yakisich, J.S.; Azad, N. Role of apoptosis-related miRNAs in resveratrol-induced breast cancer cell death. Cell Death Dis. 2016, 7, e2104. [Google Scholar] [CrossRef] [Green Version]
- Casanova, F.; Quarti, J.; Ferraz Da Costa, D.C.; Ramos, C.A.; Da Silva, J.L.; Fialho, E. Resveratrol chemosensitizes breast cancer cells to melphalan by cell cycle arrest. J. Cell. Biochem. 2012, 113, 2586–2596. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Aljarbou, A.N.; Aldebasi, Y.H.; Faisal, S.M.; Khan, M.A. Resveratrol suppresses the proliferation of breast cancer cells by inhibiting fatty acid synthase signaling pathway. Cancer Epidemiol. 2014, 38, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Zheng, Y.; Zhao, C.; Toschi, A.; Fan, J.; Shraibman, N.; Brown, H.A.; Bar-Sagi, D.; Foster, D.A.; Arbiser, J.L. Honokiol suppresses survival signals mediated by ras-dependent phospholipase D activity in human cancer cells. Clin. Cancer Res. 2008, 14, 4267–4274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, D.; Lu, Q.; Hu, X. Down-regulation of P-glycoprotein expression in MDR breast cancer cell MCF-7/ADR by honokiol. Cancer Lett. 2006, 243, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Crane, C.; Panner, A.; Pieper, R.O.; Arbiser, J.; Parsa, A.T. Honokiol-mediated inhibition of PI3K/mTOR pathway: A potential strategy to overcome immunoresistance in glioma, breast, and prostate carcinoma without impacting T cell function. J. Immunother. 2009, 32, 585–592. [Google Scholar] [CrossRef] [Green Version]
- Avtanski, D.B.; Nagalingam, A.; Kuppusamy, P.; Bonner, M.Y.; Arbiser, J.L.; Saxena, N.K.; Sharma, D. Honokiol abrogates leptin-induced tumor progression by inhibiting Wnt1-MTA1-β-catenin signaling axis in a microRNA-34a dependent manner. Oncotarget 2015, 6, 16396–16410. [Google Scholar] [CrossRef] [Green Version]
- Sengupta, S.; Nagalingam, A.; Muniraj, N.; Bonner, M.Y.; Mistriotis, P.; Afthinos, A.; Kuppusamy, P.; Lanoue, D.; Cho, S.; Korangath, P.; et al. Activation of tumor suppressor LKB1 by honokiol abrogates cancer stem-like phenotype in breast cancer via inhibition of oncogenic Stat3. Oncogene 2017, 36, 5709–5721. [Google Scholar] [CrossRef] [Green Version]
- Park, E.J.; Min, H.Y.; Chung, H.J.; Hong, J.Y.; Kang, Y.J.; Hung, T.M.; Youn, U.J.; Kim, Y.S.; Bae, K.H.; Kang, S.S.; et al. Down-regulation of c-Src/EGFR-mediated signaling activation is involved in the honokiol-induced cell cycle arrest and apoptosis in MDA-MB-231 human breast cancer cells. Cancer Lett. 2009, 277, 133–140. [Google Scholar] [CrossRef]
- Munna, M.M.R.; Azad, A.K.; Islam, F.; Ahmed, M.F.; Parvez, A.; Rahman, M.M.; Hossain, M.S. Phytochemical Investigation, Evaluation of Antioxidant and Cytotoxic Activity of the Methanol Extract of Cuscuta reflexa (Stem). Pharmacol. Online 2019, 3, 135–143. [Google Scholar]
- Islam, F.; Azad, A.K.; Faysal, M.; Islam, S.; Sugandha, N.J.; Saha, S.; Isah, S.I. Phytochemical investigation and comparative anthelmintic activity of between methanol and acetone extract of Limonia acidissima L. (fruit PEEL). Pharmacologyonline 2019, 2, 241–246. [Google Scholar]
- Ahmad, A.; Sarkar, S.H.; Bitar, B.; Ali, S.; Aboukameel, A.; Sethi, S.; Li, Y.; Bao, B.; Kong, D.; Banerjee, S.; et al. Garcinol regulates EMT and Wnt signaling pathways in vitro and in vivo, leading to anticancer activity against breast cancer cells. Mol. Cancer Ther. 2012, 11, 2193–2201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, A.; Wang, Z.; Ali, R.; Maitah, M.Y.; Kong, D.; Banerjee, S.; Padhye, S.; Sarkar, F.H. Apoptosis-inducing effect of garcinol is mediated by NF-κB signaling in breast cancer cells. J. Cell. Biochem. 2010, 109, 1134–1141. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Man Gho, W.; Chan, F.I.; Chen, S.; Leung, L.K. The red clover (Trifolium pratense) isoflavone biochanin A inhibits aromatase activity and expression. Br. J. Nutr. 2008, 99, 303–310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhushan, A.; Sehdev, V.; Lai, J.C.K. Biochanin A modulates cell viability, invasion, and growth promoting signaling pathways in HER-2-positive breast cancer cells. J. Oncol. 2009, 2009, 121458. [Google Scholar] [CrossRef] [Green Version]
- Atwell, L.L.; Zhang, Z.; Mori, M.; Farris, P.E.; Vetto, J.T.; Naik, A.M.; Oh, K.Y.; Thuillier, P.; Ho, E.; Shannon, J. Sulforaphane bioavailability and chemopreventive activity in women scheduled for breast biopsy. Cancer Prev. Res. 2015, 8, 1184–1191. [Google Scholar] [CrossRef] [Green Version]
- Miere, F.; Vicas, S.I.; Timar, A.V.; Ganea, M.; Zdrinca, M.; Cavalu, S.; Fritea, L.; Vicas, L.; Muresan, M.; Pallag, A. Preparation and characterization of two different liposomal formulations with bioactive natural extract for multiple applications. Processes 2021, 9, 432. [Google Scholar] [CrossRef]
- Holzer, T.R.; McMaster, W.R.; Forney, J.D. Expression profiling by whole-genome interspecies microarray hybridization reveals differential gene expression in procyclic promastigotes, lesion-derived amastigotes, and axenic amastigotes in Leishmania mexicana. Mol. Biochem. Parasitol. 2006, 146, 198–218. [Google Scholar] [CrossRef]
- Chalabi, N.; Delort, L.; Le Corre, L.; Satih, S.; Bignon, Y.J.; Bernard-Gallon, D. Gene signature of breast cancer cell lines treated with lycopene. Pharmacogenomics 2006, 7, 663–672. [Google Scholar] [CrossRef]
- Yao, Y.; Zhou, Q. A novel antiestrogen agent Shikonin inhibits estrogen-dependent gene transcription in human breast cancer cells. Breast Cancer Res. Treat. 2010, 121, 233–240. [Google Scholar] [CrossRef]
- Han, W.; Li, L.; Qiu, S.; Lu, Q.; Pan, Q.; Gu, Y.; Luo, J.; Hu, X. Shikonin circumvents cancer drug resistance by induction of a necroptotic death. Mol. Cancer Ther. 2007, 6, 1641–1649. [Google Scholar] [CrossRef] [Green Version]
- Bishayee, A. Cancer prevention and treatment with resveratrol: From rodent studies to clinical trials. Cancer Prev. Res. 2009, 2, 409–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jackson, S.J.T.; Singletary, K.W. Sulforaphane: A naturally occurring mammary carcinoma mitotic inhibitor, which disrupts tubulin polymerization. Carcinogenesis 2004, 25, 219–227. [Google Scholar] [CrossRef] [Green Version]
- Luettig, B.; Steinmüller, C.; Gifford, G.E.; Wagner, H.; Lohmann-matthes, M.L. Macrophage activation by the polysaccharide arabinogalactan isolated from plant cell cultures of Echinacea purpurea. J. Natl. Cancer Inst. 1989, 81, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.J. Health-promoting properties of common herbs. Am. J. Clin. Nutr. 1999, 70, 491s–499s. [Google Scholar] [CrossRef] [PubMed]
- Islam, F.; Faysal, M.; Trina, T.A.; Begh, M.Z.A.; Al Amin, M.; Rashid, M.M.; Rahman, M.M. Phytochemical screening and anthelmintic activity of alcoholic extract of fruits of Eleocharis dulcis. Pharmacol. Online 2019, 3, 94–99. [Google Scholar]
- Tagde, P.; Tagde, P.; Islam, F.; Tagde, S.; Shah, M.; Hussain, Z.D.; Rahman, M.; Najda, A.; Alanazi, I.S.; Germoush, M.O. The Multifaceted Role of Curcumin in Advanced Nanocurcumin Form in the Treatment and Management of Chronic Disorders. Molecules 2021, 26, 7109. [Google Scholar] [CrossRef]
- Islam, F.; Bibi, S.; Meem, A.F.K.; Islam, M.; Rahaman, M.; Bepary, S.; Rahman, M.; Elzaki, A.; Kajoak, S.; Osman, H. Natural Bioactive Molecules: An Alternative Approach to the Treatment and Control of COVID-19. Int. J. Mol. Sci. 2021, 22, 12638. [Google Scholar] [CrossRef]
- Islam, F.; Fahim, N.F.; Trina, T.A.; Mishu, I.J. Evaluation of Antioxidant, Antimicrobial and Thrombolytic Activity of Eleocharis Dulcis (Cyperaceae) Fruits of Methanol Extract. Evaluation 2019, 2, 39–49. [Google Scholar]
- Potter, J.D. Cancer prevention: Epidemiology and experiment. Cancer Lett. 1997, 114, 7–9. [Google Scholar] [CrossRef]
- Freudenheim, J.L.; Marshall, J.R.; Vena, J.E.; Laughlin, R.; Brasure, J.R.; Swanson, M.K.; Nemoto, T.; Graham, S. Premenopausal breast cancer risk and intake of vegetables, fruits, and related nutrients. J. Natl. Cancer Inst. 1996, 88, 340–348. [Google Scholar] [CrossRef]
- Rahman, M.M.; Islam, M.R.; Shohag, S.; Hossain, M.E.; Rahaman, M.S.; Islam, F.; Ahmed, M.; Mitra, S.; Khandaker, M.U.; Idris, A.M. The Multifunctional Role of Herbal Products in the Management of Diabetes and Obesity: A Comprehensive Review. Molecules 2022, 27, 1713. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Islam, F.; Anwar Parvez, M.A.K.; Ashraf, G.M.; Ullah, M.F.; Ahmed, M. Citrus limon L.(lemon) seed extract shows neuro-modulatory activity in an in vivo thiopental-sodium sleep model by reducing the sleep onset and enhancing the sleep duration. J. Integr. Neurosci. 2022, 21, 042. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Yang, C.S. Mechanisms of cancer prevention by tea constituents. J. Nutr. 2003, 133, 3262S–3267S. [Google Scholar] [CrossRef] [PubMed]
- Zaveri, N.T. Green tea and its polyphenolic catechins: Medicinal uses in cancer and noncancer applications. Life Sci. 2006, 78, 2073–2080. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Chang, H.; Peng, X.; Bai, Q.; Yi, L.; Zhou, Y.; Zhu, J.; Mi, M. Resveratrol inhibits breast cancer stem-like cells and induces autophagy via suppressing Wnt/β-catenin signaling pathway. PLoS ONE 2014, 9, e102535. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.K.; Banerjee, S.; Acosta, E.P.; Lillard, J.W.; Singh, R. Resveratrol induces cell cycle arrest and apoptosis with docetaxel in prostate cancer cells via a p53/p21WAF1/CIP1 and p27KIP1 pathway. Oncotarget 2017, 8, 17216–17228. [Google Scholar] [CrossRef] [Green Version]
- Chopra, H.; Bibi, S.; Mishra, A.K.; Tirth, V.; Yerramsetty, S.V.; Murali, S.V.; Ahmad, S.U.; Mohanta, Y.K.; Attia, M.S.; Algahtani, A. Nanomaterials: A Promising Therapeutic Approach for Cardiovascular Diseases. J. Nanomater. 2022, 2022, 4155729. [Google Scholar] [CrossRef]
- Chopra, H.; Bibi, S.; Islam, F.; Ahmad, S.U.; Olawale, O.A.; Alhumaydhi, F.A.; Marzouki, R.; Baig, A.A.; Emran, T.B. Emerging Trends in the Delivery of Resveratrol by Nanostructures: Applications of Nanotechnology in Life Sciences. J. Nanomater. 2022, 2022, 3083728. [Google Scholar] [CrossRef]
- Sprouse, A.A.; Herbert, B.S. Resveratrol augments paclitaxel treatment in MDA-MB-231 and paclitaxel-resistant MDA-MB-231 breast cancer cells. Anticancer Res. 2014, 34, 5363–5374. [Google Scholar]
- Singh, S.K.; Lillard, J.W.; Singh, R. Reversal of drug resistance by planetary ball milled (PBM) nanoparticle loaded with resveratrol and docetaxel in prostate cancer. Cancer Lett. 2018, 427, 49–62. [Google Scholar] [CrossRef]
- Vinod, B.S.; Nair, H.H.; Vijayakurup, V.; Shabna, A.; Shah, S.; Krishna, A.; Pillai, K.S.; Thankachan, S.; Anto, R.J. Resveratrol chemosensitizes HER-2-overexpressing breast cancer cells to docetaxel chemoresistance by inhibiting docetaxel-mediated activation of HER-2–Akt axis. Cell Death Discov. 2015, 1, 15061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, Y.; Yang, S.; Troup, S.; Lu, X.; Callaghan, S.; Park, D.S.; Xing, Y.; Yang, X. Resveratrol induces apoptosis in breast cancer cells by E2F1-mediated up-regulation of ASPP1. Oncol. Rep. 2011, 25, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Woodbury, A.; Yu, S.P.; Wei, L.; García, P. Neuro-modulating effects of honokiol: A review. Front. Neurol. 2013, 4, 130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, W.; Chen, L.; Yang, G.; Zhou, H.; Jiang, Q.; Zhong, Z.; Hu, J.; Chen, X.; Wang, X.; Yuan, Y.; et al. Synergistic antitumor effects of liposomal honokiol combined with adriamycin in breast cancer models. Phyther. Res. 2008, 22, 1125–1132. [Google Scholar] [CrossRef]
- Akter, A.; Islam, F.; Bepary, S.; Al-Amin, M.; Begh, M.; Alam, Z.; Islam, M.; Ashraf, G.M.; Baeesa, S.S.; Ullah, M.F. CNS depressant activities of Averrhoa carambola leaves extract in thiopental-sodium model of Swiss albino mice: Implication for neuro-modulatory properties. Biologia 2022, 77, 1–10. [Google Scholar] [CrossRef]
- Wolf, I.; O’Kelly, J.; Wakimoto, N.; Nguyen, A.; Amblard, F.; Karlan, B.Y.; Arbiser, J.L.; Koeffler, H.P. Honokiol, a natural biphenyl, inhibits in vitro and in vivo growth of breast cancer through induction of apoptosis and cell cycle arrest. Int. J. Oncol. 2007, 30, 1529–1537. [Google Scholar] [CrossRef]
- Alonso-Castro, A.J.; Domínguez, F.; García-Regalado, A.; González-Sánchez, I.; Cerbón, M.A.; García-Carrancá, A. Magnolia dealbata seeds extract exert cytotoxic and chemopreventive effects on MDA-MB231 breast cancer cells. Pharm. Biol. 2014, 52, 621–627. [Google Scholar] [CrossRef]
- Saadat, N.; Gupta, S.V. Potential role of garcinol as an anticancer agent. J. Oncol. 2012, 2012, 647206. [Google Scholar] [CrossRef] [Green Version]
- Ye, X.; Yuan, L.; Zhang, L.; Zhao, J.; Zhang, C.M.; Deng, H.Y. Garcinol, an acetyltransferase inhibitor, suppresses proliferation of breast cancer cell line MCF-7 promoted by 17β-estradiol. Asian Pac. J. Cancer Prev. 2014, 15, 5001–5007. [Google Scholar] [CrossRef] [Green Version]
- Heinonen, S.M.; Wähälä, K.; Adlercreutz, H. Identification of urinary metabolites of the red clover isoflavones formononetin and biochanin A in human subjects. J. Agric. Food Chem. 2004, 52, 6802–6809. [Google Scholar] [CrossRef]
- Young, J.M.; Brazeau, D.A.; Morris, M.E. Effects of flavonoids genistein and biochanin A on gene expression and their metabolism in human mammary cells. Nutr. Cancer 2007, 57, 48–58. [Google Scholar] [CrossRef]
- Moon, Y.J.; Shin, B.S.; An, G.; Morris, M.E. Biochanin A inhibits breast cancer tumor growth in a murine xenograft model. Pharm. Res. 2008, 25, 2158–2163. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Zheng, B.; Sun, J.; Yang, J. Biochanin a induces anticancer effects in SK-Mel-28 human malignant melanoma cells via induction of apoptosis, inhibition of cell invasion and modulation of Nf-κB and MAPK signaling pathways. Oncol. Lett. 2017, 14, 5989–5993. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Lai, J.C.K.; Bhushan, A. Abstract 3523: Biochanin A enhances anticancer activity of temozolomide in glioblastoma multiforme. Cancer Res. 2011, 71 (Suppl. S8), 3523. [Google Scholar] [CrossRef]
- King-Batoon, A.; Leszczynska, J.M.; Klein, C.B. Modulation of gene methylation by genistein or lycopene in breast cancer cells. Environ. Mol. Mutagen. 2008, 49, 36–45. [Google Scholar] [CrossRef]
- Bishop, K.S.; Ferguson, L.R. The interaction between epigenetics, nutrition and the development of cancer. Nutrients 2015, 7, 922–947. [Google Scholar] [CrossRef] [Green Version]
- Takeshima, M.; Ono, M.; Higuchi, T.; Chen, C.; Hara, T.; Nakano, S. Anti-proliferative and apoptosis-inducing activity of lycopene against three subtypes of human breast cancer cell lines. Cancer Sci. 2014, 105, 252–257. [Google Scholar] [CrossRef] [Green Version]
- Peng, S.J.; Li, J.; Zhou, Y.; Tuo, M.; Qin, X.X.; Yu, Q.; Cheng, H.; Li, Y.M. In vitro effects and mechanisms of lycopene in MCF-7 human breast cancer cells. Genet. Mol. Res. 2017, 16, 13. [Google Scholar] [CrossRef]
- Zhang, X.; Spiegelman, D.; Baglietto, L.; Bernstein, L.; Boggs, D.A.; Van Den Brandt, P.A.; Buring, J.E.; Gapstur, S.M.; Giles, G.G.; Giovannucci, E.; et al. Carotenoid intakes and risk of breast cancer defined by estrogen receptor and progesterone receptor status: A pooled analysis of 18 prospective cohort studies. Am. J. Clin. Nutr. 2012, 95, 713–725. [Google Scholar] [CrossRef]
- Eliassen, A.H.; Hendrickson, S.J.; Brinton, L.A.; Buring, J.E.; Campos, H.; Dai, Q.; Dorgan, J.F.; Franke, A.A.; Gao, Y.T.; Goodman, M.T.; et al. Circulating carotenoids and risk of breast cancer: Pooled analysis of eight prospective studies. J. Natl. Cancer Inst. 2012, 104, 1905–1916. [Google Scholar] [CrossRef] [Green Version]
- Rao, A.V.; Shen, H. Effect of low dose lycopene intake on lycopene bioavailability and oxidative stress. Nutr. Res. 2002, 22, 1125–1131. [Google Scholar] [CrossRef]
- Basu, A.; Imrhan, V. Tomatoes versus lycopene in oxidative stress and carcinogenesis: Conclusions from clinical trials. Eur. J. Clin. Nutr. 2007, 61, 295–303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dash, R.; Junaid, M.; Islam, N.; Akash, C.; Forhad, M.; Khan, M.; Arifuzzaman, M.; Khatun, M.; Zahid Hosen, S.M. Molecular insight and binding pattern analysis of Shikonin as a potential VEGFR-2 inhibitor. Curr. Enzym. Inhib. 2017, 13, 235–244. [Google Scholar] [CrossRef]
- Yao, Y.; Brodie, A.M.H.; Davidson, N.E.; Kensler, T.W.; Zhou, Q. Inhibition of estrogen signaling activates the NRF2 pathway in breast cancer. Breast Cancer Res. Treat. 2010, 124, 585–591. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Qian, R.Q.; Li, P.P. Shikonin, an ingredient of Lithospermum erythrorhizon, down-regulates the expression of steroid sulfatase genes in breast cancer cells. Cancer Lett. 2009, 284, 47–54. [Google Scholar] [CrossRef]
- Gernapudi, R. Chemopreventive Activities of Shikonin in Breast Cancer. Biochem. Pharmacol. Open Access 2014, 03. [Google Scholar] [CrossRef] [Green Version]
- Jang, S.Y.; Lee, J.K.; Jang, E.H.; Jeong, S.Y.; Kim, J.H. Shikonin blocks migration and invasion of human breast cancer cells through inhibition of matrix metalloproteinase-9 activation. Oncol. Rep. 2014, 31, 2827–2833. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Dai, M.; Zhu, C.; Zhang, J.; Lin, L.; Ding, J.; Duan, W. Synthesis and biological activity of novel shikonin analogues. Bioorg. Med. Chem. Lett. 2009, 19, 735–737. [Google Scholar] [CrossRef]
- Li, W.; Liu, J.; Jackson, K.; Shi, R.; Zhao, Y. Sensitizing the therapeutic efficacy of taxol with shikonin in human breast cancer cells. PLoS ONE 2014, 9, e94079. [Google Scholar] [CrossRef]
- Zhang, C.H.; Wang, J.; Zhang, L.X.; Lu, Y.H.; Ji, T.H.; Xu, L.; Ling, L.J. Shikonin reduces tamoxifen resistance through long non-coding RNA uc.57. Oncotarget 2017, 8, 88658–88669. [Google Scholar] [CrossRef] [Green Version]
- Su, L.; Liu, L.H.; Wang, Y.L.; Yan, G.Z.; Zhang, Y. Long-term systemic toxicity of shikonin derivatives in Wistar rats. Pharm. Biol. 2014, 52, 486–490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Assimopoulou, A.N.; Papageorgiou, V.P. Encapsulation of isohexenylnaphthazarins in cyclodextrins. Biomed. Chromatogr. 2004, 18, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.L.; Kong, A.N. Molecular targets of dietary phenethyl isothiocyanate and sulforaphane for cancer chemoprevention. AAPS J. 2010, 12, 87–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pledgie-Tracy, A.; Sobolewski, M.D.; Davidson, N.E. Sulforaphane induces cell type-specific apoptosis in human breast cancer cell lines. Mol. Cancer Ther. 2007, 6, 1013–1021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jackson, S.J.T.; Singletary, K.W. Sulforaphane inhibits human MCF-7 mammary cancer cell mitotic progression and tubulin polymerization. J. Nutr. 2004, 134, 2229–2236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.H.; Park, H.J.; Moon, D.O. Sulforaphane sensitizes human breast cancer cells to paclitaxel-induced apoptosis by downregulating the NF-κB signaling pathway. Oncol. Lett. 2017, 13, 4427–4432. [Google Scholar] [CrossRef] [Green Version]
- Lewinska, A.; Bednarz, D.; Adamczyk-Grochala, J.; Wnuk, M. Phytochemical-induced nucleolar stress results in the inhibition of breast cancer cell proliferation. Redox Biol. 2017, 12, 469–482. [Google Scholar] [CrossRef]
- Li, Y.; Meeran, S.M.; Tollefsbol, T.O. Combinatorial bioactive botanicals re-sensitize tamoxifen treatment in ER-negative breast cancer via epigenetic reactivation of ERα expression. Sci. Rep. 2017, 7, 9345. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Yuan, Y.Y.; Meeran, S.M.; Tollefsbol, T.O. Synergistic epigenetic reactivation of estrogen receptor-α (ERα) by combined green tea polyphenol and histone deacetylase inhibitor in ERα-negative breast cancer cells. Mol. Cancer 2010, 9, 274. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.N.; Kim, D.H.; Kim, E.H.; Lee, M.H.; Kundu, J.K.; Na, H.K.; Cha, Y.N.; Surh, Y.J. Sulforaphane inhibits phorbol ester-stimulated IKK-NF-κB signaling and COX-2 expression in human mammary epithelial cells by targeting NF-κB activating kinase and ERK. Cancer Lett. 2014, 351, 41–49. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, T.; Korkaya, H.; Liu, S.; Lee, H.F.; Newman, B.; Yu, Y.; Clouthier, S.G.; Schwartz, S.J.; Wicha, M.S.; et al. Sulforaphane, a dietary component of broccoli/broccoli sprouts, inhibits breast cancer stem cells. Clin. Cancer Res. 2010, 16, 2580–2590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Yao, Y.; Eades, G.; Liu, Z.; Zhang, Y.; Zhou, Q. Downregulation of miR-140 promotes cancer stem cell formation in basal-like early stage breast cancer. Oncogene 2014, 33, 2589–2600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Eades, G.; Yao, Y.; Zhang, Y.; Zhou, Q. Characterization of a stem-like subpopulation in basal-like ductal carcinoma in situ (DCIS) lesions. J. Biol. Chem. 2014, 289, 1303–1312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dandawate, P.R.; Subramaniam, D.; Jensen, R.A.; Anant, S. Targeting cancer stem cells and signaling pathways by phytochemicals: Novel approach for breast cancer therapy. Semin. Cancer Biol. 2016, 40, 192–208. [Google Scholar] [CrossRef] [Green Version]
- Mitra, S.; Tareq, A.M.; Das, R.; Emran, T.B.; Nainu, F.; Chakraborty, A.J.; Ahmad, I.; Tallei, T.E.; Idris, A.M.; Simal-Gandara, J. Polyphenols: A first evidence in the synergism and bioactivities. Food Rev. Int. 2022, 2022, 1–23. [Google Scholar] [CrossRef]
- Galeone, C.; Pelucchi, C.; Levi, F.; Negri, E.; Franceschi, S.; Talamini, R.; Giacosa, A.; La Vecchia, C. Onion and garlic use and human cancer. Am. J. Clin. Nutr. 2006, 84, 1027–1032. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.S.; Chhabra, S.K.; Hong, J.Y.; Smith, T.J. Mechanisms of inhibition of chemical toxicity and carcinogenesis by diallyl sulfide (DAS) and related compounds from garlic. J. Nutr. 2001, 131, 1041S–1045S. [Google Scholar] [CrossRef] [Green Version]
- Shareef, M.; Ashraf, M.A.; Sarfraz, M. Natural cures for breast cancer treatment. Saudi Pharm. J. 2016, 24, 233–240. [Google Scholar] [CrossRef]
- Donaldson, M.S. Nutrition and cancer: A review of the evidence for an anti-cancer diet. Nutr. J. 2004, 3, 19. [Google Scholar] [CrossRef] [Green Version]
- Keum, Y.S.; Park, K.K.; Lee, J.M.; Chun, K.S.; Park, J.H.; Lee, S.K.; Kwon, H.; Surh, Y.J. Antioxidant and anti-tumor promoting activities of the methanol extract of heat-processed ginseng. Cancer Lett. 2000, 150, 41–48. [Google Scholar] [CrossRef]
- Rauf, A.; Khan, S.A.; Olatunde, A.; Imran, M.; Alhumaydhi, F.A.; Aljohani, A.S.M.; Uddin, M.S.; Mitra, S.; Emran, T.B.; Khayrullin, M.; et al. Honokiol: A review of its pharmacological potential and therapeutic insights. Phytomedicine 2021, 90, 153647. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Anand, A.; Garg, S.; Khan, M.S.; Bhasin, S.; Asghar, M.N.; Emran, T.B. Structure-Based In Silico Investigation of Agonists for Proteins Involved in Breast Cancer. Evid.-Based Complement. Altern. Med. 2022, 2022, 7278731. [Google Scholar] [CrossRef]
- Yuan, B.; Yao, M.; Wang, X.; Sato, A.; Okazaki, A.; Komuro, H.; Hayashi, H.; Toyoda, H.; Pei, X.; Hu, X.; et al. Antitumor activity of arsenite in combination with tetrandrine against human breast cancer cell line MDA-MB-231 in vitro and in vivo. Cancer Cell Int. 2018, 18, 113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, M.; Yuan, B.; Wang, X.; Sato, A.; Sakuma, K.; Kaneko, K.; Komuro, H.; Okazaki, A.; Hayashi, H.; Toyoda, H.; et al. Synergistic cytotoxic effects of arsenite and tetrandrine in human breast cancer cell line MCF-7. Int. J. Oncol. 2017, 51, 587–598. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.; Zhang, C.; Bao, J.; Jia, X.; Liang, Y.; Wang, X.; Chen, M.; Su, H.; Li, P.; Wan, J.B.; et al. Synergistic chemopreventive effects of curcumin and berberine on human breast cancer cells through induction of apoptosis and autophagic cell death. Sci. Rep. 2016, 6, 26064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, S.; Rakib, A.; Islam, M.; Khanam, B.H.; Faiz, F.B.; Paul, A.; Chy, M.; Uddin, N.; Bhuiya, N.M.; Uddin, M.M.N.; et al. In vivo and in vitro pharmacological activities of Tacca integrifolia rhizome and investigation of possible lead compounds against breast cancer through in silico approaches. Clin. Phytosci. 2019, 5, 1–13. [Google Scholar] [CrossRef]
- Mohamed, A.A.R.; Khater, S.I.; Metwally, M.M.; Emran, T.B.; Nassan, M.A.; Abd El-Emam, M.M.; Mostafa-Hedeab, G.; El-Shetry, E.S. TGF-β1, NAG-1, and antioxidant enzymes expression alterations in Cisplatin-induced nephrotoxicity in a rat model: Comparative modulating role of Melatonin, Vit. E and Ozone. Gene 2022, 820, 146293. [Google Scholar] [CrossRef]
- Uddin, M.; Kabir, M.; Mamun, A.A.; Sarwar, M.; Nasrin, F.; Emran, T.B.; Alanazi, I.S.; Rauf, A.; Albadrani, G.M.; Sayed, A.A.; et al. Natural small molecules targeting NF-κB signaling in glioblastoma. Front. Pharmacol. 2021, 12, 2174. [Google Scholar] [CrossRef]
- Tu, S.H.; Chiou, Y.S.; Kalyanam, N.; Ho, C.T.; Chen, L.C.; Pan, M.H. Garcinol sensitizes breast cancer cells to Taxol through the suppression of caspase-3/iPLA2 and NF-κB/Twist1 signaling pathways in a mouse 4T1 breast tumor model. Food Funct. 2017, 8, 1067–1079. [Google Scholar] [CrossRef]
- Liu, H.; Zang, C.; Emde, A.; Planas-Silva, M.D.; Rosche, M.; Kühnl, A.; Schulz, C.O.; Elstner, E.; Possinger, K.; Eucker, J. Anti-tumor effect of honokiol alone and in combination with other anti-cancer agents in breast cancer. Eur. J. Pharmacol. 2008, 591, 43–51. [Google Scholar] [CrossRef]
- Xue, J.P.; Wang, G.; Zhao, Z.-B.; Wang, Q.; Shi, Y. Synergistic cytotoxic effect of genistein and doxorubicin on drug-resistant human breast cancer MCF-7/Adr cells. Oncol. Rep. 2014, 32, 1647–1653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charalambous, C.; Pitta, C.A.; Constantinou, A.I. Equol enhances tamoxifen’s anti-tumor activity by induction of caspase-mediated apoptosis in MCF-7 breast cancer cells. BMC Cancer 2013, 13, 238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nainu, F.; Masyita, A.; Bahar, M.; Raihan, M.; Prova, S.R.; Mitra, S.; Emran, T.B.; Simal-Gandara, J. Pharmaceutical prospects of bee products: Special focus on anticancer, antibacterial, antiviral, and antiparasitic properties. Antibiotics 2021, 10, 822. [Google Scholar] [CrossRef] [PubMed]
- González-Vallinas, M.; Molina, S.; Vicente, G.; Sánchez-Martínez, R.; Vargas, T.; García-Risco, M.R.; Fornari, T.; Reglero, G.; Ramírez de Molina, A. Modulation of estrogen and epidermal growth factor receptors by rosemary extract in breast cancer cells. Electrophoresis 2014, 35, 1719–1727. [Google Scholar] [CrossRef]
- Holohan, C.; Van Schaeybroeck, S.; Longley, D.B.; Johnston, P.G. Cancer drug resistance: An evolving paradigm. Nat. Rev. Cancer 2013, 13, 714–726. [Google Scholar] [CrossRef]
- Mitra, S.; Prova, S.R.; Sultana, S.A.; Das, R.; Nainu, F.; Emran, T.B.; Tareq, A.M.; Uddin, M.S.; Alqahtani, A.M.; Dhama, K.; et al. Therapeutic potential of indole alkaloids in respiratory diseases: A comprehensive review. Phytomedicine 2021, 90, 153649. [Google Scholar] [CrossRef]
- Islam, M.N.; Rauf, A.; Fahad, F.I.; Emran, T.B.; Mitra, S.; Olatunde, A.; Shariati, M.A.; Rebezov, M.; Rengasamy, K.R.; Mubarak, M.S. Superoxide dismutase: An updated review on its health benefits and industrial applications. Crit. Rev. Food Sci. Nutr. 2021, 2021, 1–19. [Google Scholar] [CrossRef]
- Eva, T.A.; Barua, N.; Chowdhury, M.M.; Yeasmin, S.; Rakib, A.; Islam, M.R.; Emran, T.B.; Simal-Gandara, J. Perspectives on signaling for biological-and processed food-related advanced glycation end-products and its role in cancer progression. Crit. Rev. Food Sci. Nutr. 2020, 2020, 1–18. [Google Scholar] [CrossRef]
- Millimouno, F.M.; Dong, J.; Yang, L.; Li, J.; Li, X. Targeting apoptosis pathways in cancer and perspectives with natural compounds from mother nature. Cancer Prev. Res. 2014, 7, 1081–1107. [Google Scholar] [CrossRef] [Green Version]
| Combinations and Their Classes | Chemical Formulas | Mode of Action | References |
|---|---|---|---|
| Tetrandrine (alkaloid) and Arsenic (metalloid) | C38H42N2O6, and H3AsO4 | Increases FOXO3a, p21, and p27 expression; suppresses cyclin D1 expression; induces G0/G1 phase arrest; promotes autophagy. Survivin expression is also reduced | [183,184] |
| Curcumin (flavonoid) and Berberine (alkaloid) | C21H20O6, and C20H18NO4+ | Activates ERK pathways, promoting caspase-dependent apoptosis; induces autophagy; increases JNK and beclin1 phosphorylation; decreases Bcl-2 phosphorylation | [185] |
| Thymoquinone (1,4-benzoquinone) and Tamoxifen (triphenylethylene) | C10H12O2, and C26H29NO | Reduces relapse rates, TNF-α, IL-6, and TGF-1β levels; upregulates caspase-3 expression; downregulates Bcl-2 expression; inhibits cell survival via the PI3-K/Akt pathway by suppressing Akt phosphorylation; stimulates XIAP degradation; activates caspase-9, and promotes apoptosis via the PI3-K/Akt pathway; inhibits cell survival by suppressing Akt phosphorylation | [186] |
| Silibinin (flavonoid)and Chrysin (flavonoid) | C25H22O10, and C15H10O4 | Stops proliferation of BrCa cells, and reduces the expression of hTERT and cyclin D1 mRNA | [187] |
| Resveratrol (phytoalexin)and Salinomycin (polyketide and a spiroketal) | C14H12O3, and C42H70O11 | Reduces Wnt signaling protein synthesis, increases E-cadherin and lowers vimentin, slows cell migration and invasion, activates caspase-8 and 9, and downregulates Wnt/EMT signaling | [188] |
| Garcinol (polyisoprenylated benzophenone) and Paclitaxel (alkaloid) | C38H50O6, and C47H51NO14 | Promotes cell cycle arrest, inhibits the (NF-κB)/Twist-related protein 1 (Twist1) signaling system, and suppresses the caspase-3/cytosolic Ca2+-independent phospholipase A2 (iPLA2) signaling pathway | [189] |
| Honokiol (neolignan biphenols) and Lapatinib (4-anilinoquinazoline) | C18H18O2, and C29H26ClFN4O4S | Inhibits tumor cell proliferation by suppressing HER-2 expression | [190] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Islam, M.R.; Islam, F.; Nafady, M.H.; Akter, M.; Mitra, S.; Das, R.; Urmee, H.; Shohag, S.; Akter, A.; Chidambaram, K.; et al. Natural Small Molecules in Breast Cancer Treatment: Understandings from a Therapeutic Viewpoint. Molecules 2022, 27, 2165. https://doi.org/10.3390/molecules27072165
Islam MR, Islam F, Nafady MH, Akter M, Mitra S, Das R, Urmee H, Shohag S, Akter A, Chidambaram K, et al. Natural Small Molecules in Breast Cancer Treatment: Understandings from a Therapeutic Viewpoint. Molecules. 2022; 27(7):2165. https://doi.org/10.3390/molecules27072165
Chicago/Turabian StyleIslam, Md. Rezaul, Fahadul Islam, Mohamed H. Nafady, Muniya Akter, Saikat Mitra, Rajib Das, Humaira Urmee, Sheikh Shohag, Aklima Akter, Kumarappan Chidambaram, and et al. 2022. "Natural Small Molecules in Breast Cancer Treatment: Understandings from a Therapeutic Viewpoint" Molecules 27, no. 7: 2165. https://doi.org/10.3390/molecules27072165
APA StyleIslam, M. R., Islam, F., Nafady, M. H., Akter, M., Mitra, S., Das, R., Urmee, H., Shohag, S., Akter, A., Chidambaram, K., Alhumaydhi, F. A., Emran, T. B., & Cavalu, S. (2022). Natural Small Molecules in Breast Cancer Treatment: Understandings from a Therapeutic Viewpoint. Molecules, 27(7), 2165. https://doi.org/10.3390/molecules27072165










