CO2 to CO Electroreduction, Electrocatalytic H2 Evolution, and Catalytic Degradation of Organic Dyes Using a Co(II) meso-Tetraarylporphyrin †
Abstract
:1. Introduction
2. Results and Discussion
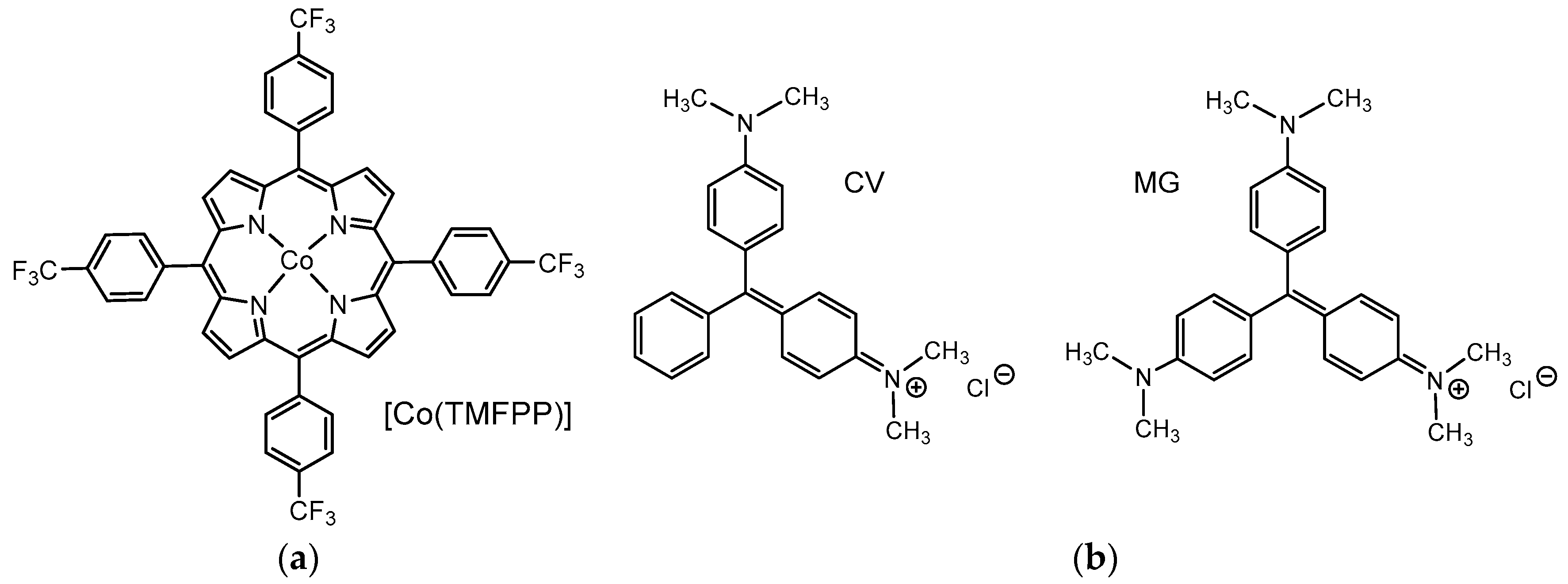
2.1. Synthesis and 1H NMR Spectroscopy
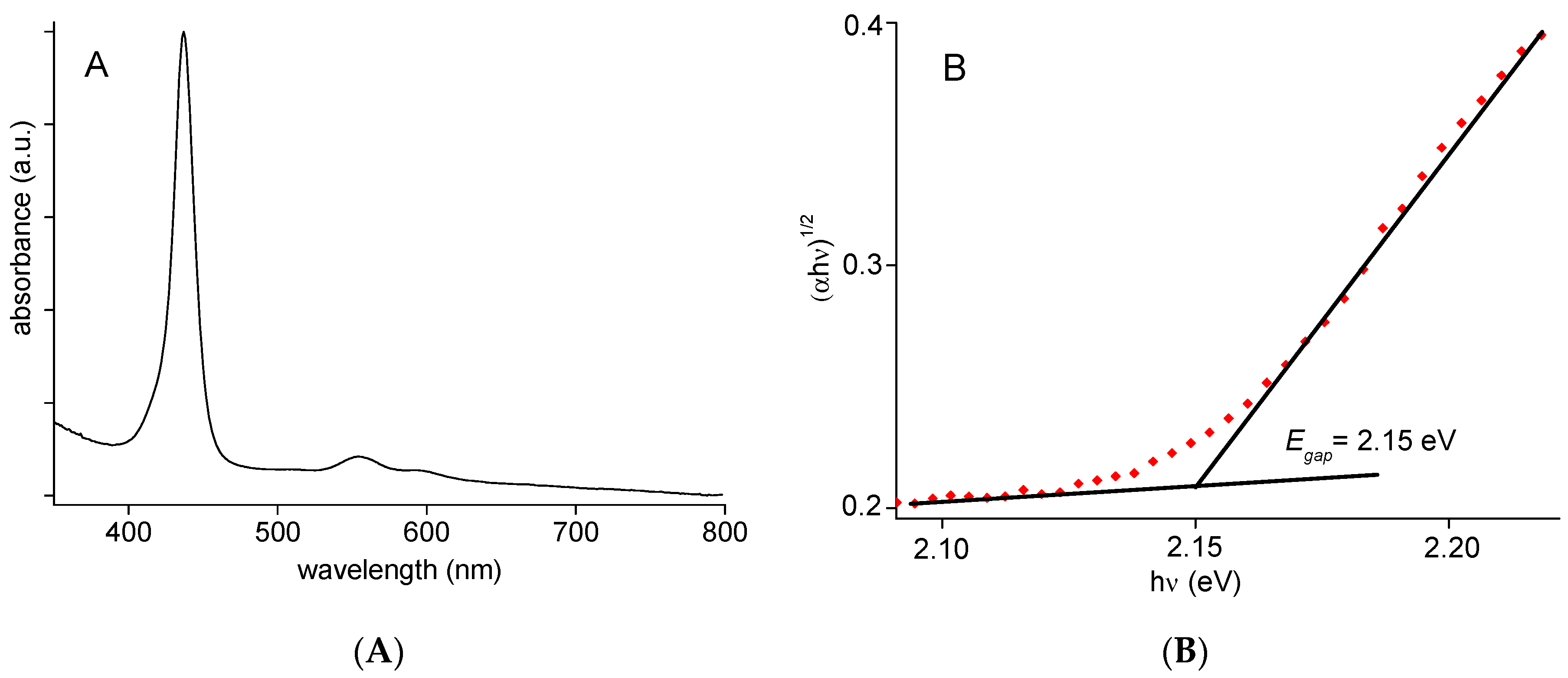
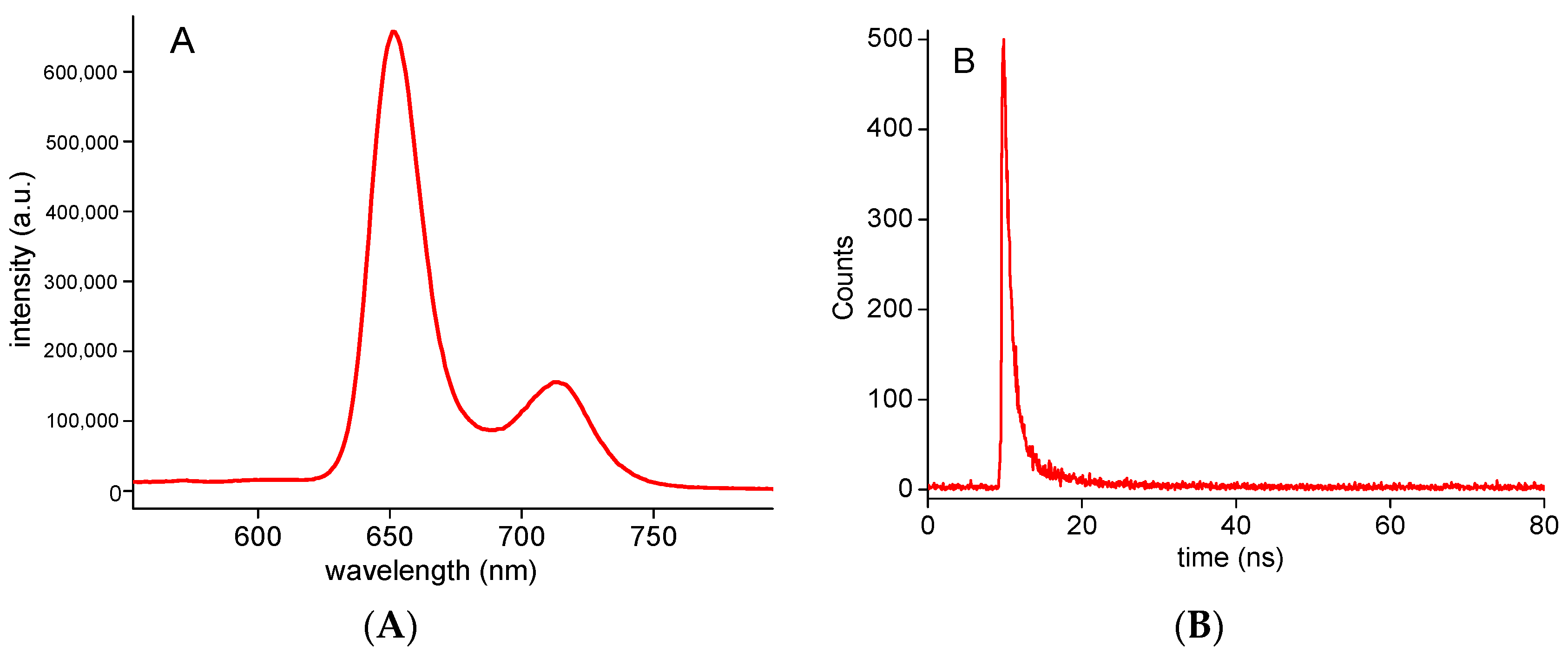
2.2. Photophysical Properties
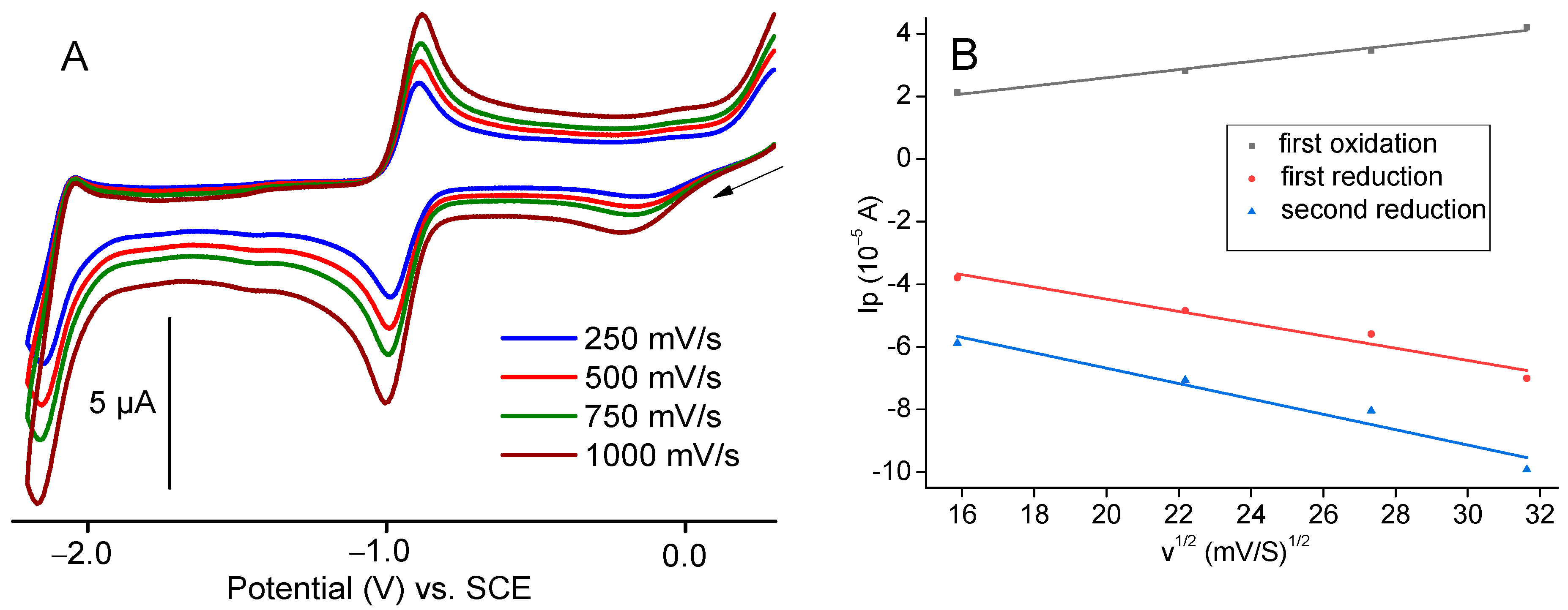
2.3. Electrochemical Characterisation
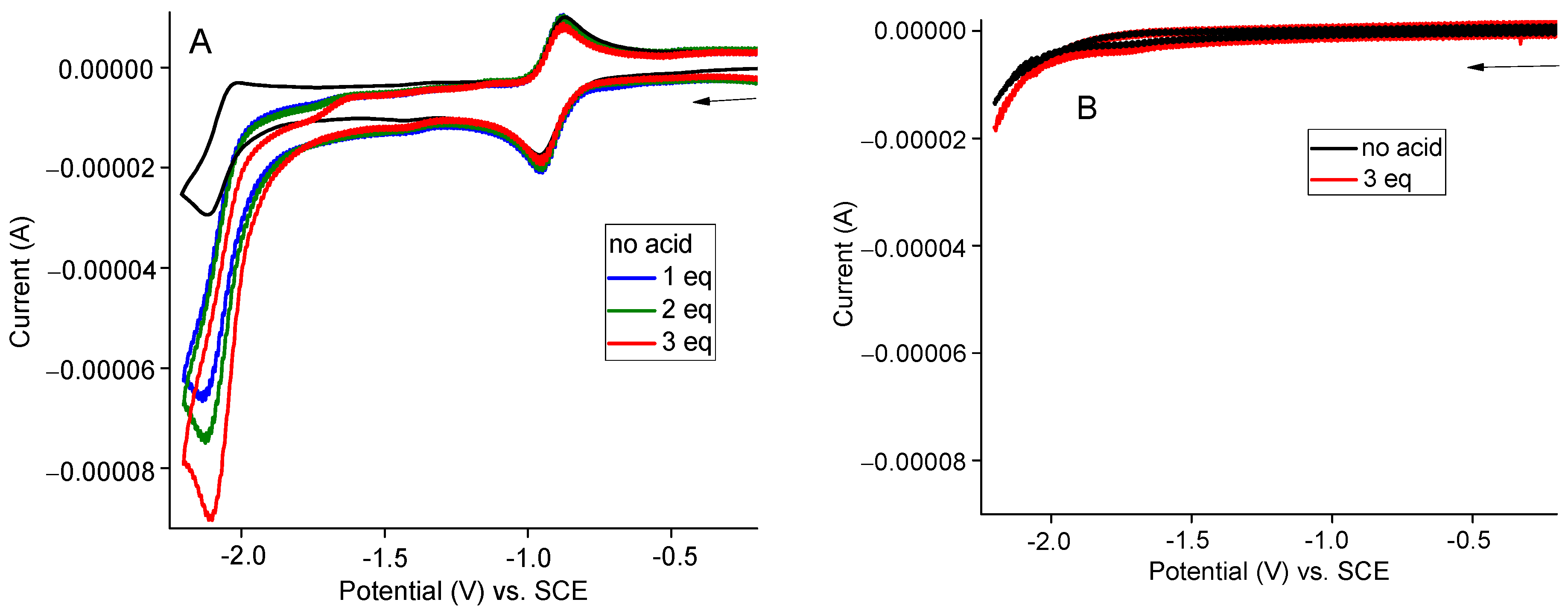
2.4. Electrocatalytic H2 Production in the Presence of Acetic Acid (AcOH)
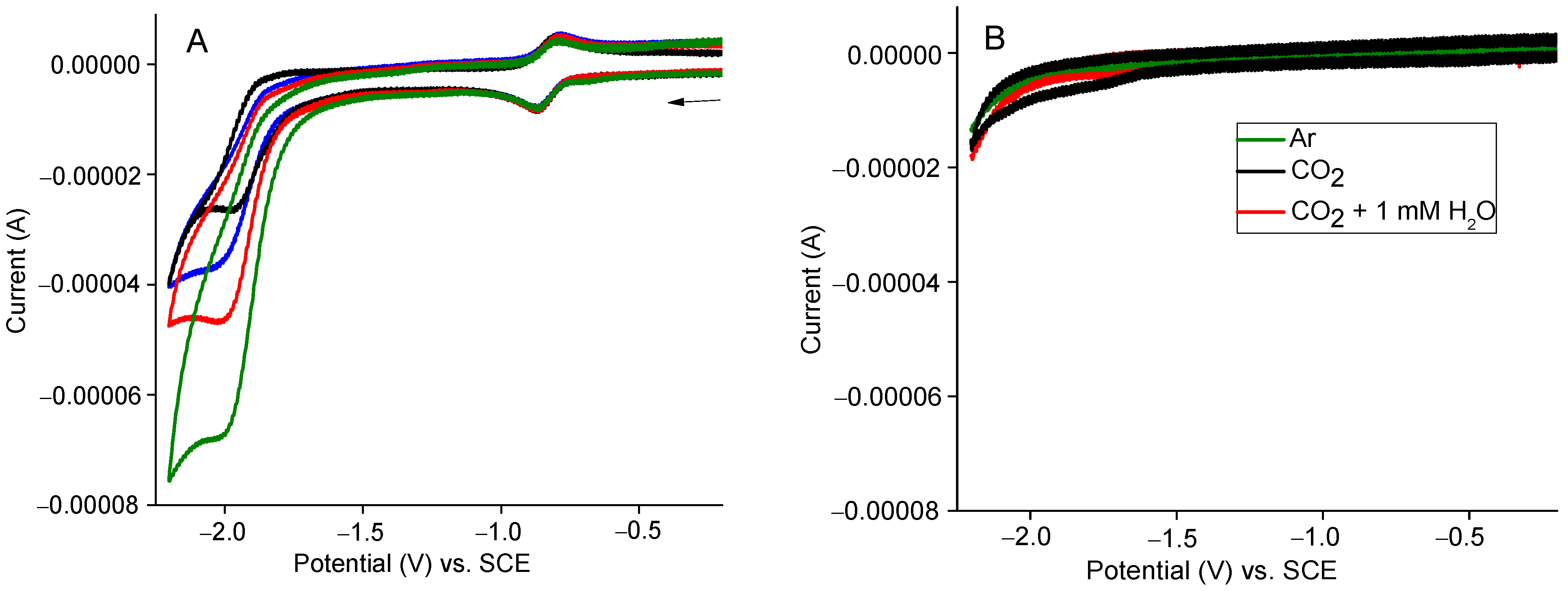
2.5. Electroreduction CO2 to CO
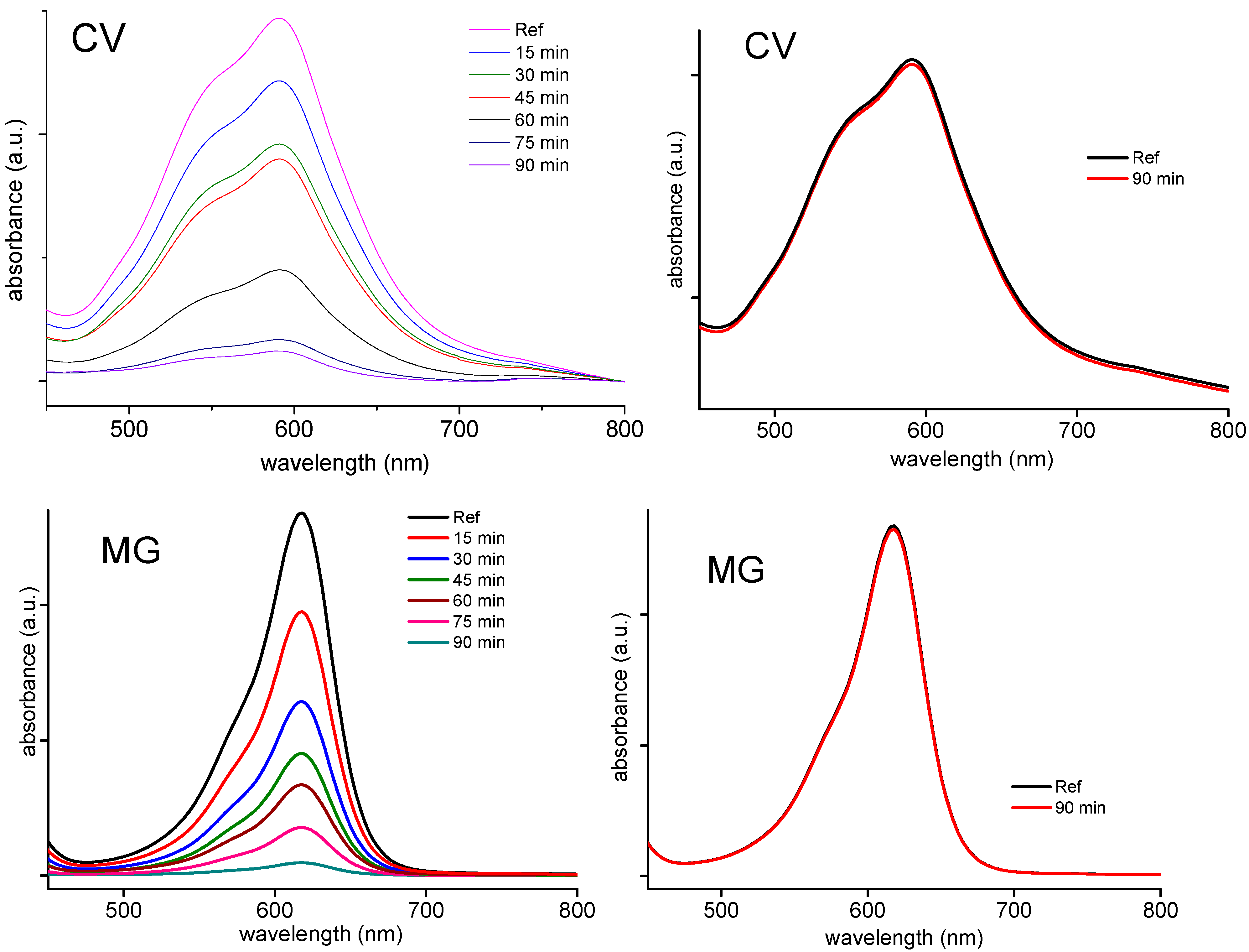
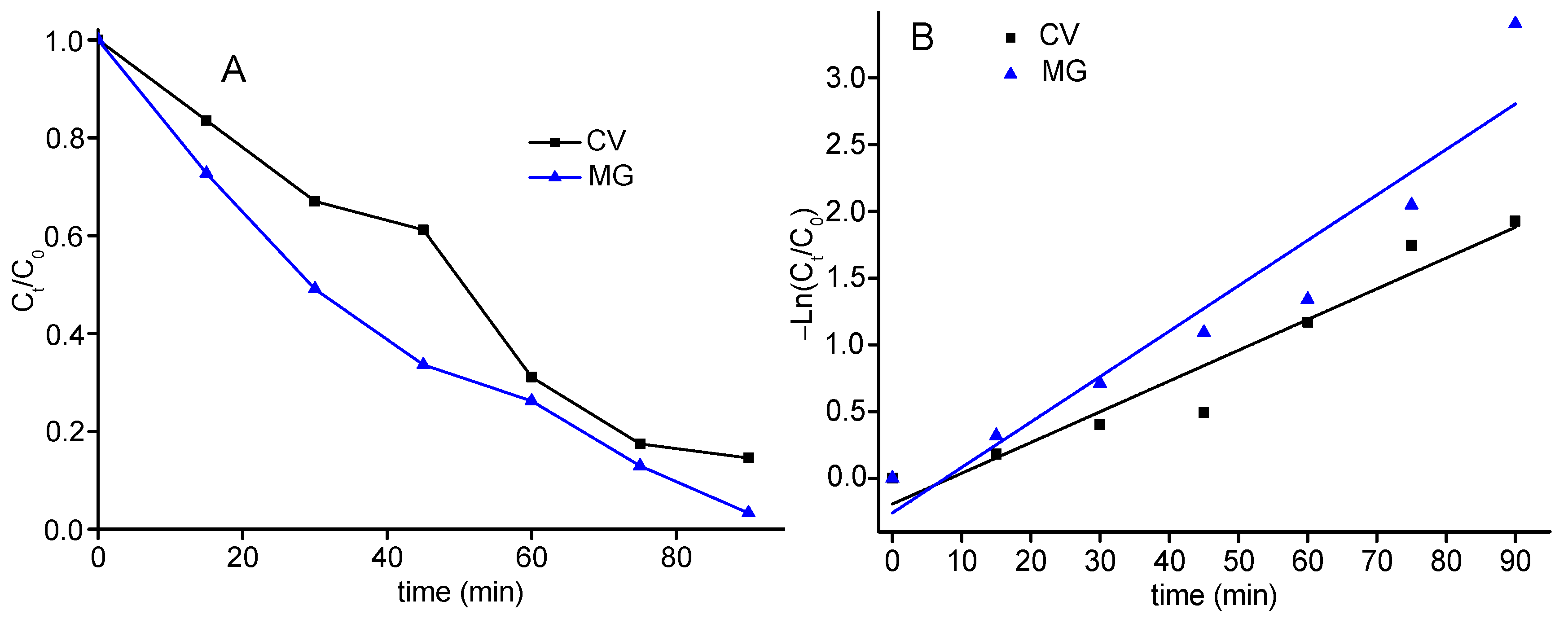
2.6. Catalytic Oxidative Degradation of Dyes
3. Experimental Section
3.1. Materials
3.2. Synthetic Procedures
3.2.1. Synthesis of the meso-Tetrakis(4-(trifluoromethyl)phenyl)porphyrin (H2TMFPP)
3.2.2. Synthesis of the meso-Tetrakis(4-(trifluoromethyl)phenyl)porphyrinato Co(II) [Co(TMFPP)]
3.3. Instrumentation
3.4. Electrochemistry
3.5. Electrocatalytic CO2 Reduction
3.6. Gas Detection
3.7. Faradaic Efficiency Calculation
3.8. Catalytic Dye Degradation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaim, W.; Schwederski, B.; Klein, A. Bioinorganic Chemistry: Inorganic Elements in the Chemistry of Life—An. Introduction and Guide, 2nd ed.; John Wiley & Sons: Chichester, UK, 2013; ISBN 978-0-470-97523-7. [Google Scholar]
- Verma, P.K.; Sawant, S.D. Unravelling reaction selectivities via bio-inspired porphyrinoid tetradentate frameworks. Coord. Chem. Rev. 2022, 450, 214239. [Google Scholar] [CrossRef]
- Nishiori, D.; Wadsworth, B.L.; Reyes Cruz, E.A.; Nguyen, N.P.; Hensleigh, L.K.; Karcher, T.; Moore, G.F. Photoelectrochemistry of metalloporphyrin--modified GaP semiconductors. Photosynth. Res. 2021. online. [Google Scholar] [CrossRef] [PubMed]
- Lomova, T. Recent progress in organometallic porphyrin-based molecular materials for optical sensing, light conversion, and magnetic cooling. Appl. Organomet. Chem. 2021, 35, e6254. [Google Scholar] [CrossRef]
- Harvey, P.D. Porphyrin-based MOFs as heterogeneous photocatalysts for the eradication of organic pollutants and toxins. J. Porphyr. Phthalocyanines 2021, 25, 583–604. [Google Scholar] [CrossRef]
- Mathew, D.; Sujatha, S. Interactions of porphyrins with DNA: A review focusing recent advances inchemical modifications on porphyrins as artificial nucleases. J. Inorg. Biochem. 2021, 219, 111434. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Warren, J.J. Recent Developments in Metalloporphyrin Electrocatalysts for Reduction of Small Molecules: Strategies for Managing Electron and Proton Transfer Reactions. ChemSusChem 2021, 14, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Faustova, M.; Nikolskaya, E.; Sokol, M.; Fomicheva, M.; Petrov, R.; Yabbarov, N. Metalloporphyrins in Medicine: From History to Recent Trends. ACS Appl. Bio Mater. 2020, 3, 8146–8171. [Google Scholar] [CrossRef]
- Sun, T.; Zhang, Z.; Xu, J.; Liang, L.; Mai, C.-L.; Ren, L.; Zhou, Q.; Yu, Y.; Zhang, B.; Gao, P. Structural, photophysical, electrochemical and spintronic study of first-row metal Tetrakis(meso-triphenylamine)-porphyrin complexes: A combined experimental and theoretical study. Dyes Pigm. 2021, 193, 109469. [Google Scholar] [CrossRef]
- Guergueb, M.; Nasri, S.; Brahmi, J.; Al-Ghamdi, Y.O.; Loiseau, F.; Molton, F.; Roisnel, T.; Guerineau, V.; Nasri, H. Spectroscopic characterization, X-ray molecular structures and cyclic voltammetry study of two (piperazine) cobalt(II) meso-arylporphyin complexes. Application as a catalyst for the degradation of 4-nitrophenol. Polyhedron 2021, 209, 115468. [Google Scholar] [CrossRef]
- Guergueb, M.; Nasri, S.; Brahmi, J.; Loiseau, F.; Molton, F.; Roisnel, T.; Guerineau, V.; Turowska-Tyrk, I.; Aouadi, K.; Nasri, H. Effect of the coordination of π-acceptor 4-cyanopyridine ligand on the structural and electronic properties of meso-tetra(para-methoxy) and meso-tetra(para-chlorophenyl) porphyrin cobalt(II) coordination compounds. Application in the catalytic degradation of methylene blue dye. RSC Adv. 2020, 10, 6900–6918. [Google Scholar] [CrossRef] [Green Version]
- Guergueb, M.; Brahmi, J.; Nasri, S.; Loiseau, F.; Aouadi, K.; Guerineau, V.; Najmudin, S.; Nasri, H. Zinc(II) triazole meso-arylsubstituted porphyrins for UV-visible chloride and bromide detection. Adsorption and catalytic degradation of malachite green dye. RSC Adv. 2020, 10, 22712–22725. [Google Scholar] [CrossRef]
- Chaudhri, N.; Cong, L.; Bulbul, A.S.; Grover, N.; Osterloh, W.R.; Fang, Y.; Sankar, M.; Kadish, K.M. Structural, Photophysical, and Electrochemical Properties of Doubly Fused Porphyrins and Related Fused Chlorins. Inorg. Chem. 2020, 59, 1481–1495. [Google Scholar] [CrossRef] [PubMed]
- Pamin, K.; Tabor, E.; Gjrecka, S.; Kubiak, W.W.; Rutkowska-Zbik, D.; Połtowicz, J. Three Generations of Cobalt Porphyrins as Catalysts in the Oxidation of Cycloalkanes. ChemSusChem 2019, 12, 684–691. [Google Scholar] [CrossRef]
- Soury, R.; Jabli, M.; Saleh, T.A.; Abdul-Hassan, W.S.; Saint-Aman, E.; Loiseau, F.; Philouze, C.; Nasri, H. Tetrakis(ethyl-4(4-butyryl)oxyphenyl)porphyrinato zinc complexes with 4,4′-bpyridin: Synthesis, characterization, and its catalytic degradation of Calmagite. RSC Adv. 2018, 8, 20143–20156. [Google Scholar] [CrossRef] [Green Version]
- Ke, X.; Kumar, R.; Sankar, M.; Kadish, K.M. Electrochemistry and Spectroelectrochemistry of Cobalt Porphyrins with π--Extending and/or Highly Electron-Withdrawing Pyrrole Substituents, In Situ Electrogeneration of σ--Bonded Complexes. Inorg. Chem. 2018, 57, 1490–1503. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Fang, Y.; Ou, Z.; Xue, S.; Kadish, K.M. Cobalt Tetrabutano- and Tetrabenzotetraarylporphyrin Complexes: Effect of Substituents on the Electrochemical Properties and Catalytic Activity of Oxygen Reduction Reactions. Inorg. Chem. 2017, 56, 13613–13626. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Fu, L.-Z.; Yang, L.-M.; Liu, X.-P.; Zhan, S.-Z.; Ni, C.-L. The impact of modifying the ligands on hydrogen production electro-catalyzed by meso-tetra-p-X-phenylporphin cobalt complexes, CoT(X)PP. J. Mol. Catal. A Chem. 2016, 417, 101–106. [Google Scholar] [CrossRef] [Green Version]
- Zhutaeva, G.V.; Tarasevich, M.R.; Radina, M.V.; Chernyshova, I.S. Composites Based on Phenyl Substituted Cobalt Porphyrins with Nafion as Catalysts for Oxygen Electroreduction. Russ. J. Electrochem. 2009, 45, 1080–1088. [Google Scholar] [CrossRef]
- Klein, A. Spectroelectrochemistry of Metalloporphyrins. In Spectroelectrochemistry; Kaim, W., Klein, A., Eds.; RSC Publishing: Cambridge UK, 2008; pp. 91–122. [Google Scholar]
- Ryabova, V.; Schulte, A.; Erichsen, T.; Schuhmann, W. Robotic sequential analysis of a library of metalloporphyrins as electrocatalysts for voltammetric nitric oxide sensors. Analyst 2005, 130, 1245–1252. [Google Scholar] [CrossRef]
- Sun, H.; Smirnov, V.V.; DiMagno, S.G. Slow Electron Transfer Rates for Fluorinated Cobalt Porphyrins: Electronic and Conformational Factors Modulating Metalloporphyrin ET. Inorg. Chem. 2003, 42, 6032–6040. [Google Scholar] [CrossRef]
- Kobayashi, N.; Nevin, W.A. Electrocatalytic Reduction of Oxygen Using Water-Soluble Iron and Cobalt Phthalocyanines and Porphyrins. Appl. Organomet. Chem. 1996, 10, 579–590. [Google Scholar] [CrossRef]
- Walker, F.A.; Beroiz, D.; Kadish, K.M. Electronic Effects in Transition Metal Porphyrins. 2. The Sensitivity of Redox and Ligand Addition Reactions in Para-Substituted Tetraphenylporphyrin Complexes of Cobalt(II). J. Am. Chem. Soc. 1976, 98, 3484–3489. [Google Scholar] [CrossRef] [PubMed]
- Tezuka, M.; Ohkatsu, Y.; Osa, T. Reduction and Oxidation Potentials of Metal-free and Cobalt Tetra(p-substituted phenyl)porphyrins. Bull. Chem. Soc. Jpn. 1976, 49, 1435–1436. [Google Scholar] [CrossRef] [Green Version]
- Nasri, S.; Hajji, M.; Guergueb, M.; Dhifaoui, S.; Marvaud, V.; Loiseau, F.; Molton, F.; Roisnel, T.; Guerfel, T.; Nasri, H. Spectroscopic, Electrochemical, Magnetic and Structural Characterization of an hexamethylenetetramine Co(II) Porphyrin Complex – Application in the Catalytic Degradation of Vat Yellow 1 dye. J. Mol. Struct. 2021, 1231, 129676. [Google Scholar] [CrossRef]
- Zhou, Y.; Xing, Y.-F.; Wen, J.; Ma, H.B.; Wang, F.-B.; Xia, X.-H. Axial ligands tailoring the ORR activity of cobalt porphyrin. Sci. Bull. 2019, 64, 1158–1166. [Google Scholar] [CrossRef] [Green Version]
- Doctorovich, F.; Bikiel, D.; Pellegrino, J.; Suárez, S.A.; Martí, M.A. Stabilization and detection of nitroxyl by iron and cobalt porphyrins in solution and on surfaces. J. Porphyr. Phthalocyanines 2010, 14, 1012–1018. [Google Scholar] [CrossRef]
- Richter-Addo, G.B.; Hodge, S.J.; Yi, G.-B.; Khan, M.A.; Ma, T.; Van Caemelbecke, E.; Guo, N.; Kadish, K.M. Synthesis, Characterization, and Spectroelectrochemistry of Cobalt Porphyrins Containing Axially Bound Nitric Oxide. Inorg. Chem. 1996, 35, 6530–6538. [Google Scholar] [CrossRef]
- Usman, M.; Humayun, M.; Garba, M.D.; Ullah, L.; Zeb, Z.; Helal, A.; Suliman, M.H.; Alfaifi, B.Y.; Iqbal, N.; Abdinejad, M.; et al. Electrochemical Reduction of CO2: A Review of Cobalt Based Catalysts for Carbon Dioxide Conversion to Fuels. Nanomaterials 2021, 11, 2029. [Google Scholar] [CrossRef]
- Marianov, A.N.; Kochubei, A.S.; Roman, T.; Conquest, O.J.; Stampfl, C.; Jiang, Y. Modeling and Experimental Study of the Electron Transfer Kinetics for Non-ideal Electrodes Using Variable-Frequency Square Wave Voltammetry. Anal. Chem. 2021, 93, 10175–10186. [Google Scholar] [CrossRef]
- Dou, S.; Sun, L.; Xi, S.; Li, X.; Su, T.; Fan, H.J.; Wang, X. Enlarging the π-Conjugation of Cobalt Porphyrin for Highly Active and Selective CO2 Electroreduction. ChemSusChem 2021, 14, 2126–2132. [Google Scholar] [CrossRef]
- Marianov, A.N.; Kochubei, A.S.; Roman, T.; Conquest, O.J.; Stampfl, C.; Jiang, Y. Resolving Deactivation Pathways of Co Porphyrin-Based Electrocatalysts for CO2 Reduction in Aqueous Medium. ACS Catal. 2021, 11, 3715–3729. [Google Scholar] [CrossRef]
- Chen, X.; Hu, X.-M.; Daasbjerg, K.; Ahlquist, M.S.G. Understanding the Enhanced Catalytic CO2 Reduction upon Adhering Cobalt Porphyrin to Carbon Nanotubes and the Inverse Loading Effect. Organometallics 2020, 39, 1634–1641. [Google Scholar] [CrossRef]
- Jack, J.; Park, E.; Maness, P.-C.; Huang, S.; Zhang, W.; Ren, Z.J. Selective ligand modification of cobalt porphyrins for carbon dioxide electrolysis: Generation of a renewable H2/CO feedstock for downstream catalytic hydrogenation. Inorg. Chim. Acta 2020, 507, 119594. [Google Scholar] [CrossRef]
- Wang, Z.-j.; Song, H.; Liu, H.; Ye, J. Coupling of Solar Energy and Thermal Energy for Carbon Dioxide Reduction: Status and Prospects. Angew. Chem. Int. Ed. 2020, 59, 8016–8035. [Google Scholar] [CrossRef]
- Sinha, S.; Zhang, R.; Warren, J.J. Low Overpotential CO2 Activation by a Graphite-Adsorbed Cobalt Porphyrin. ACS Catal. 2020, 10, 12284–12291. [Google Scholar] [CrossRef]
- Abdinejad, M.; Seifitokaldani, A.; Dao, C.; Sargent, E.H.; Zhang, X.-a.; Kraatz, H.B. Enhanced Electrochemical Reduction of CO2 Catalyzed by Cobalt and Iron Amino Porphyrin Complexes. ACS Appl. Energy Mater. 2019, 2, 1330–1335. [Google Scholar] [CrossRef]
- Hu, B.; Xie, W.; Li, R.; Pan, Z.; Song, S.; Wang, Y. How does the ligands structure surrounding metal-N4 of Co-based macrocyclic compounds affect electrochemical reduction of CO2 performance? Electrochim. Acta 2019, 331, 135283. [Google Scholar] [CrossRef]
- Miyamoto, K.; Asahi, R. Water Facilitated Electrochemical Reduction of CO2 on Cobalt-Porphyrin Catalysts. J. Phys. Chem. C 2019, 123, 9944–9948. [Google Scholar] [CrossRef] [Green Version]
- Smith, P.T.; Benke, B.P.; An, L.; Kim, Y.; Kim, K.; Chang, C.J. A Supramolecular Porous Organic Cage Platform Promotes Electrochemical Hydrogen Evolution from Water Catalyzed by Cobalt Porphyrins. ChemElectroChem 2021, 8, 1653–1657. [Google Scholar] [CrossRef]
- Lv, X.; Chen, Y.; Wu, Y.; Wang, H.; Wang, X.; Wei, C.; Xiao, Z.; Yang, G.; Jiang, J. A Br-regulated transition metal active-site anchoring and exposure strategy in biomass derived carbon nanosheets for obtaining robust ORR/HER electrocatalysts at all pH values. J. Mater. Chem. A 2019, 7, 27089–27098. [Google Scholar] [CrossRef]
- Zhao, Z.; Ozoemena, K.I.; Maree, D.M.; Nyokong, T. Synthesis and electrochemical studies of a covalently linked cobalt(II) phthalocyanine–cobalt(II) porphyrin conjugate. Dalton Trans. 2005, 1241–1248. [Google Scholar] [CrossRef] [PubMed]
- Atoguchi, T.; Aramata, A.; Kazusaka, A.; Enyo, M. Electrocatalytic activity of CoII TPP-pyridine complex modified carbon electrode for CO2 reduction. J. Electroanal. Chem. 1991, 318, 309–320. [Google Scholar] [CrossRef]
- Yuan, Y.-J.; Yu, Z.-T.; Chen, D.-Q.; Zou, Z.-G. Metal-complex chromophores for solar hydrogen generation. Chem. Soc. Rev. 2017, 46, 603–631. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.A.W.; Celestine, M.J.; Artis, E.T.; Joseph, L.S.; Esquivel, D.L.; Ledbetter, A.J.; Cropek, D.M.; Jarrett, W.L.; Bayse, C.A.; Brewer, M.I.; et al. Computational, electrochemical, and spectroscopic studies of two mononuclear cobaloximes: The influence of an axial pyridine and solvent on the redox behaviour and evidence for pyridine coordination to cobalt(I) and cobalt(II) metal centres. Dalton Trans. 2016, 45, 10326–10342. [Google Scholar] [CrossRef]
- McKone, J.R.; Marinescu, S.C.; Brunschwig, B.S.; Winkler, J.J.; Gray, H.B. Earth-abundant hydrogen evolution electrocatalysts. Chem. Sci. 2014, 5, 865–878. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Veleta, J.M.; Tang, D.; Price, A.D.; Botez, C.E.; Villagrán, D. Efficient electrocatalytic hydrogen gas evolution by a cobalt–porphyrin-based crystalline polymer. Dalton Trans. 2018, 47, 8801–8806. [Google Scholar] [CrossRef]
- Attatsi, I.K.; Weihua Zhu, W.; Liang, X. Noncovalent immobilization of Co(II)porphyrin through axial coordination as an enhanced electrocatalyst on carbon electrodes for oxygen reduction and evolution. New J. Chem. 2020, 44, 4340–4345. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Schneider, P.E.; Goldsmith, Z.K.; Mondal, B.; Hammes-Schiffer, S.; Stahl, S.S. Brønsted Acid Scaling Relationships Enable Control Over Product Selectivity from O2 Reduction with a Mononuclear Cobalt Porphyrin Catalyst. ACS Cent. Sci. 2019, 5, 1024–1034. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.-S.; Chen, C.; Liu, J.; Parvez, K.; Liang, H.; Shu, J.; Sachdev, H.; Graf, R.; Feng, X.; Müllen, K. High-Performance Electrocatalysts for Oxygen Reduction Derived from Cobalt Porphyrin-Based Conjugated Mesoporous Polymers. Adv. Mater. 2014, 26, 1450–1455. [Google Scholar] [CrossRef]
- Pessoa, C.A.; Gushikem, Y. Cobalt porphyrins immobilized on niobium(V) oxide grafted on a silica gel surface: Study of the catalytic reduction of dissolved dioxygen. J. Porphyr. Phthalocyanines 2001, 5, 537–544. [Google Scholar] [CrossRef]
- Biloul, A.; Gouerec, P.; Savy, M.; Scarbeck, G.; Besse, S.; Riga, J. Oxygen electrocatalysis under fuel cell conditions: Behaviour of cobalt porphyrins and tetraazaannulene analogues. J. Appl. Electrochem. 1996, 26, 1139–1146. [Google Scholar] [CrossRef]
- Gouerec, P.; Bilou, A.; Contamin, O.; Scarbeck, G.; Savy, M.; Barbe, J.M.; Guilard, R. Dioxygen reduction electrocatalysis in acidic media: Effect of peripheral ligand substitution on cobalt tetraphenylporphyrin. J. Electroanal. Chem. 1995, 398, 67–75. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Basu, O.; Das, S.K. ZIF-8 MOF Encapsulated Co-porphyrin, an Efficient Electrocatalyst for Water Oxidation in a Wide pH Range: Works Better at Neutral pH. ChemCatChem 2020, 12, 5430–5438. [Google Scholar] [CrossRef]
- Xie, J.; Xu, P.; Zhu, Y.; Wang, J.; Lee, W.-C.C.; Zhang, X.P. New Catalytic Radical Process Involving 1,4-Hydrogen Atom Abstraction: Asymmetric Construction of Cyclobutanones. J. Am. Chem. Soc. 2021, 143, 11670–11678. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.-J.; Mao, M.-J.; Chen, J.-X.; Huang, Y.-B.; Cao, R. Integration of metalloporphyrin into cationic covalent triazine frameworks for the synergistically enhanced chemical fixation of CO2. Catal. Sci. Technol. 2020, 10, 8026–8033. [Google Scholar] [CrossRef]
- Li, C.; Lang, K.; Lu, H.; Hu, Y.; Cui, X.; Wojtas, L.; Zhang, X.P. Catalytic Radical Process for Enantioselective Amination of C(sp3)–H Bonds. Angew. Chem. Int. Ed. 2018, 57, 16837–16841. [Google Scholar] [CrossRef] [PubMed]
- Zardi, P.; Intrieri, D.; Caselli, A.; Gallo, E. Co(porphyrin)-catalysed amination of 1,2-dihydronaphthalene derivatives by aryl azides. J. Organomet. Chem 2012, 716, 269–274. [Google Scholar] [CrossRef]
- Chan, T.L.; To, C.T.; Liao, B.-S.; Liu, S.-T.; Chan, K.S. Electronic Effects of Ligands on the Cobalt(II)–Porphyrin-Catalyzed Direct C–H Arylation of Benzene. Eur. J. Inorg. Chem. 2012, 2012, 485–489. [Google Scholar] [CrossRef]
- Lin, X.Q.; Boisselier-Cocolios, B.; Kadish, K.M. Electrochemistry, Spectroelectrochemistry, and Ligand Addition Reactions of an Easily Reducible Cobalt Porphyrin. Reactions of Tetracyanotetraphenylporphinato)cobalt(II) ((CN)4TPP)CoII) in Pyridine and in Pyridine/Methylene Chloride Mixtures. Inorg. Chem. 1986, 25, 3242–3248. [Google Scholar] [CrossRef]
- Kadish, K.M. The Electrochemistry of Metalloporphyrins in Nonaqueous Media. In Progress in Inorganic Chemistry; Lippard, S.J., Ed.; John Wiley & Sons: Chichester, UK, 1986; Volume 34, pp. 345–605. ISBN 978-0-470-16691-8. [Google Scholar]
- Connelly, N.G.; Geiger, W.E. Chemical Redox Agents for Organometallic Chemistry. Chem. Rev. 1996, 96, 877–910. [Google Scholar] [CrossRef]
- Long, C.; Wan, K.; Qiu, X.; Zhang, X.; Han, J.; An, P.; Yang, Z.; Li, X.; Guo, J.; Shi, X.; et al. Single site catalyst with enzyme-mimic micro-environment for electroreduction of CO2. Nano Res. 2021. [Google Scholar] [CrossRef]
- Du, P.; Eisenberg, R. Catalysts made of earth-abundant elements (Co, Ni, Fe) for water splitting: Recent progress and future challenges. Energy Environ. Sci. 2012, 5, 6012–6021. [Google Scholar] [CrossRef]
- Artero, V.; Chavarot-Kerlidou, M.; Fontecave, M. Splitting water with cobalt. Angew. Chem. Int. Ed. 2011, 50, 7238–7266. [Google Scholar] [CrossRef] [PubMed]
- Zee, D.Z.; Chantarojsiri, T.; Long, J.R.; Chang, C.J. Metal polypyridyl catalysts for electro- and photochemical reduction of water to hydrogen. Acc. Chem. Res. 2015, 48, 2027–2036. [Google Scholar] [CrossRef] [Green Version]
- Pal, R.; Laureanti, J.A.; Groy, T.L.; Jones, A.K.; Trovitch, R.J. Hydrogen production from water using a bis(imino)pyridine molybdenum electrocatalyst. Chem. Commun. 2016, 52, 11555–11558. [Google Scholar] [CrossRef]
- Roubelakis, M.M.; Bediako, D.K.; Dogutan, D.K.; Nocera, D.G. Proton-coupled electron transfer kinetics for the hydrogen evolution reaction of hangman porphyrins. Energy Environ. Sci. 2012, 5, 7737–7740. [Google Scholar] [CrossRef]
- Kellett, R.M.; Spiro, T.G. Cobalt(I) porphyrin catalysts of hydrogen production from water. Inorg. Chem. 1985, 24, 2373–2377. [Google Scholar] [CrossRef]
- Brahmi, J.; Nasri, S.; Briki, C.; Guergueb, M.; Najmudin, S.; Aouadi, K.; Sanderson, M.R.; Winter, M.; Cruickshank, D.; Nasri, H. X-ray molecular structure characterization of a hexamethylenetetramine zinc(II) porphyrin complex, catalytic degradation of toluidine blue dye, experimental and statistical studies of adsorption isotherms. J. Mol. Liq. 2021, 341, 117394. [Google Scholar] [CrossRef]
- Soury, R.; Jabli, M.; Saleh, T.A.; Abdul-Hassan, W.S.; Saint-Aman, E.; Loiseau, F.; Philouze, F.; Bujacz, A.; Nasri, H. Synthesis of the (4,4′-bipyridine)(5,10,15,20-tetratolylphenylporphyrinato)zinc(II) bis(4,4-bipyridine) disolvate dehydrate and evaluation of its interaction with organic dyes. J. Mol. Liq. 2018, 264, 134–142. [Google Scholar] [CrossRef]
- Simonova, O.R.; Zdanovich, S.A.; Zaitseva, S.V.; Koifman, O.I. Kinetic Study of the Redox Properties of [5,10,15,20-Tetrakis(2,5-dimethoxyphenyl)porphyrinato]cobalt(II) in the Reaction with Hydrogen Peroxide. Russ. J. Gen. Chem. 2020, 90, 863–869. [Google Scholar] [CrossRef]
- Eaton, S.S.; Boymel, P.M.; Sawant, B.M.; More, J.K.; Eaton, G.R. Metal-Nitroxyl Interactions. 32. Spin-Spin Splitting in EPR Spectra of Spin-Labeled Pyridine Adducts of a Cobalt(II) Porphyrin in Frozen Solution. J. Magnet. Res. 1984, 56, 183–199. [Google Scholar] [CrossRef]
- More, K.M.; Eaton, G.R.; Eaton, S.S.; Hankovszky, O.H.; Hideg, K. Metal-Nitroxyl Interactions. 53. Effect of the Metal-Nitroxyl Linkage on the Electron-Electron Exchange Interaction in Spin-Labeled Complexes of Copper(II), Low-Spin Cobalt(II), Vanadyl, and Chromium(III). Inorg. Chem. 1989, 28, 1734–1743. [Google Scholar] [CrossRef]
- Adler, A.D.; Longo, F.R.; Finarelli, J.D.; Goldmacher, J.; Assour, J.; Korsakoff, L. A Simplified Synthesis for meso-Tetraphenylporphin. J. Org. Chem. 1967, 32, 476. [Google Scholar] [CrossRef]
- Adler, A.D.; Longo, F.R.; Kampas, F.; Kim, J. On the preparation of metalloporphyrins. J. Inorg. Nucl. Chem. 1970, 32, 2445–2448. [Google Scholar] [CrossRef]
- Mansour, A.; Belghith, Y.; Belkhiria, M.S.; Bujacz, A.; Guérineau, V.; Nasri, H. Synthesis, crystal structures and spectroscopic characterization of Co(II) bis(4,4′-bipyridine) with mesoporphyrins α,β,α,β-tetrakis(o-pivalamidophenyl) porphyrin (α,β,α,β-TpivPP) and tetraphenylporphyrin (TPP). J. Porphyr. Phthalocyanines 2013, 17, 1094–1103. [Google Scholar] [CrossRef]
- Francis, S.; Rajith, L. Selective Fluorescent Sensing of Adenine Via the Emissive Enhancement of a Simple Cobalt Porphyrin. J. Fluoresc. 2021, 31, 577–586. [Google Scholar] [CrossRef]
- Rathi, P.; Butcher, R.; Sankar, M. Unsymmetrical nonplanar ‘push–pull’ β-octasubstituted porphyrins: Facile synthesis, structural, photophysical and electrochemical redox properties. Dalton Trans. 2019, 48, 15002–15011. [Google Scholar] [CrossRef]
- Kobayashi, H.; Kaizu, Y. Photodynamics and Electronic Structures of Metal Complexes. Coord. Chem. Rev. 1985, 64, 53–64. [Google Scholar] [CrossRef]
- Antipas, A.; Gouterman, M. Porphyrins. 44. Electronic States of Co, Ni, Rh, and Pd Complexes. J. Am. Chem. Soc. 1983, 105, 4896–4901. [Google Scholar] [CrossRef]
- Guo, X.; Guo, B.; Shi, T. The photochemical and electrochemical properties of chiral porphyrin dimer and self-aggregate nanorods of cobalt(II) porphyrin dimer. Inorg. Chim. Acta 2010, 363, 317–323. [Google Scholar] [CrossRef]
- Pu, G.; Yang, Z.; Wu, Y.; Wang, Z.; Deng, Y.; Gao, Y.J.; Zhang, Z.; Lu, X. Investigation into the Oxygen-Involved Electrochemiluminescence of Porphyrins and Its Regulation by Peripheral Substituents/Central Metals. Anal. Chem. 2019, 91, 2319–2328. [Google Scholar] [CrossRef] [PubMed]
- Kadish, K.M.; Li, J.; Van Caemelbecke, E.; Ou, Z.; Guo, N.; Autret, M.; D’Souza, F.; Tagliatesta, P. Electrooxidation of Cobalt(II) α-Brominated-Pyrrole Tetraphenylporphyrins in CH2Cl2 under an N2 or a CO Atmosphere. Inorg. Chem. 1997, 36, 6292–6298. [Google Scholar] [CrossRef]
- D’Souza, F.; Villard, A.; Caemelbecke, E.; Franzen, M.; Boschi, T.; Tagliatesta, P.; Kadish, K.M. Electrochemical and Spectroelectrochemical Behavior of Cobalt(III), Cobalt(II), and Cobalt(I) Complexes of meso-tetraphenylporphyrinate Bearing Bromides on the β-Pyrrole Positions. Inorg. Chem. 1993, 32, 4042–4048. [Google Scholar] [CrossRef]
- Ke, X.; Yadav, P.; Cong, L.; Kumar, R.; Sankar, M.; Kadish, K.M. Facile and Reversible Electrogeneration of Porphyrin Trianions and Tetraanions in Nonaqueous Media. Inorg. Chem. 2017, 56, 8527–8537. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.M.; Rønne, M.H.; Pedersen, S.U.; Skrydstrup, T.; Daasbjerg, K. Enhanced Catalytic Activity of Cobalt Porphyrin in CO2 Electroreduction Upon Immobilization on Carbon Materials. Angew. Chem. Int. Ed. 2017, 56, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Behar, D.; Dhanasekaran, T.; Neta, P.; Hosten, C.M.; Ejeh, D.; Hambright, P.; Fujita, E. Cobalt Porphyrin Catalyzed Reduction of CO2, Radiation Chemical, Photochemical, and Electrochemical Studies. J. Phys. Chem. A 1998, 102, 2870–2877. [Google Scholar] [CrossRef]
- Perrin, D.D.; Armarego, W.L.F. Purification of Organic Solvents; Pergamon Press: Oxford, UK, 1988; ISBN 0-7506-3761-7. [Google Scholar]
- Denden, Z.; Ezzayani, K.; Saint-Aman, E.; Loiseau, F.; Najmudin, S.; Bonifácio, C.; Daran, J.C.; Nasri, H. Insights on the UV/Vis, Fluorescence, and Cyclic Voltammetry Properties and the Molecular Structures of ZnII Tetraphenylporphyrin Complexes with Pseudohalide Axial Azido, Cyanato-N, Thiocyanato-N, and Cyanido Ligands. Eur. J. Inorg. Chem. 2015, 2015, 2596–2610. [Google Scholar] [CrossRef]
- Ezzayani, K.; Denden, Z.; Najmudin, S.; Bonifácio, C.; Saint-Aman, E.; Loiseau, F.; Nasri, H. Exploring the Effects of Axial Pseudohalide Ligands on the Photophysical and Cyclic Voltammetry Properties and Molecular Structures of MgII Tetraphenyl/porphyrin Complexes. Eur. J. Inorg. Chem. 2014, 2014, 5348–5361. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guergueb, M.; Loiseau, F.; Molton, F.; Nasri, H.; Klein, A. CO2 to CO Electroreduction, Electrocatalytic H2 Evolution, and Catalytic Degradation of Organic Dyes Using a Co(II) meso-Tetraarylporphyrin. Molecules 2022, 27, 1705. https://doi.org/10.3390/molecules27051705
Guergueb M, Loiseau F, Molton F, Nasri H, Klein A. CO2 to CO Electroreduction, Electrocatalytic H2 Evolution, and Catalytic Degradation of Organic Dyes Using a Co(II) meso-Tetraarylporphyrin. Molecules. 2022; 27(5):1705. https://doi.org/10.3390/molecules27051705
Chicago/Turabian StyleGuergueb, Mouhieddinne, Frédérique Loiseau, Florian Molton, Habib Nasri, and Axel Klein. 2022. "CO2 to CO Electroreduction, Electrocatalytic H2 Evolution, and Catalytic Degradation of Organic Dyes Using a Co(II) meso-Tetraarylporphyrin" Molecules 27, no. 5: 1705. https://doi.org/10.3390/molecules27051705
APA StyleGuergueb, M., Loiseau, F., Molton, F., Nasri, H., & Klein, A. (2022). CO2 to CO Electroreduction, Electrocatalytic H2 Evolution, and Catalytic Degradation of Organic Dyes Using a Co(II) meso-Tetraarylporphyrin. Molecules, 27(5), 1705. https://doi.org/10.3390/molecules27051705







