Synthesis of Mono-Amino Substituted γ-CD: Host–Guest Complexation and In Vitro Cytotoxicity Investigation
Abstract
:1. Introduction
2. Results and Discussion
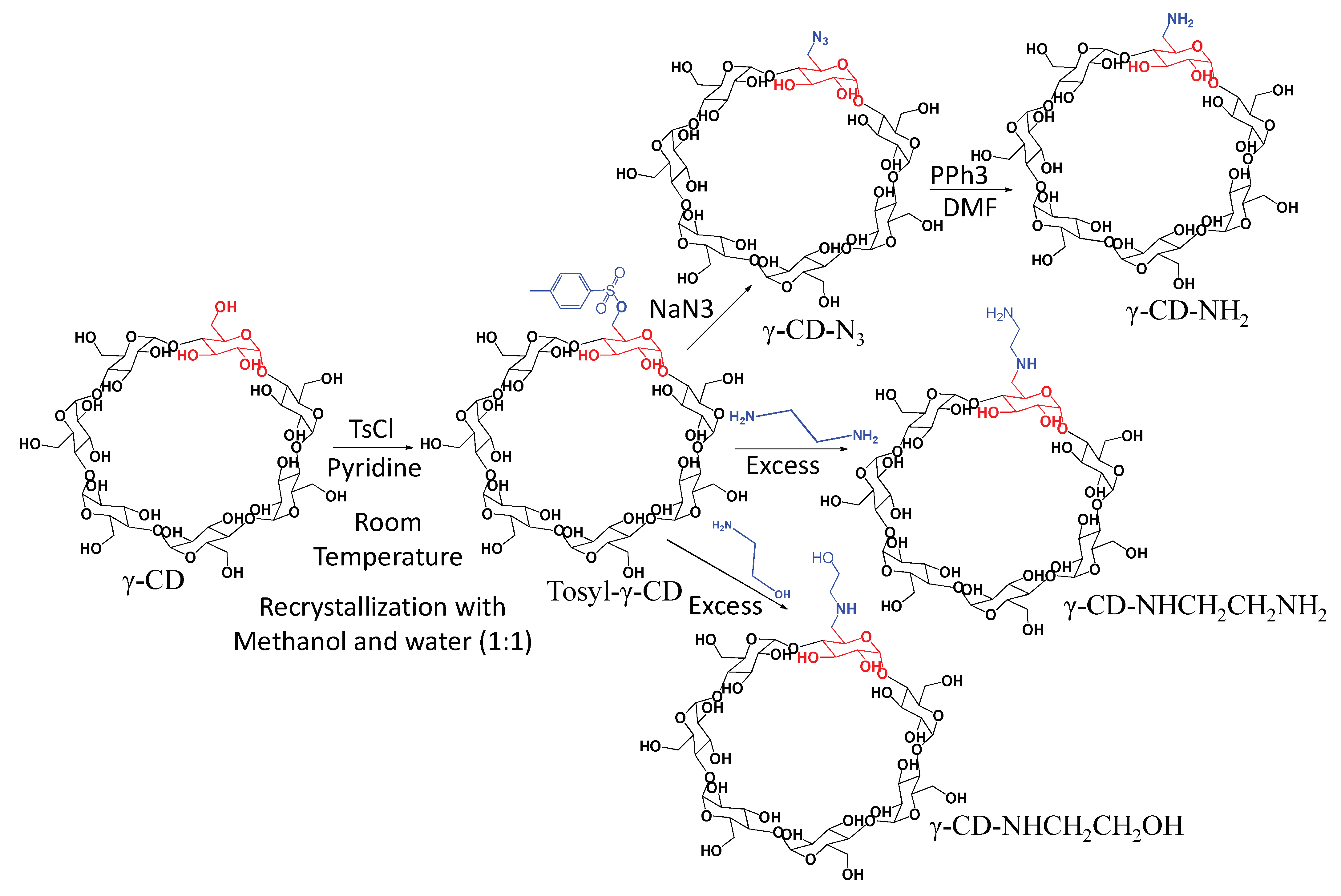
2.1. Characterization of Modified γ-CDs
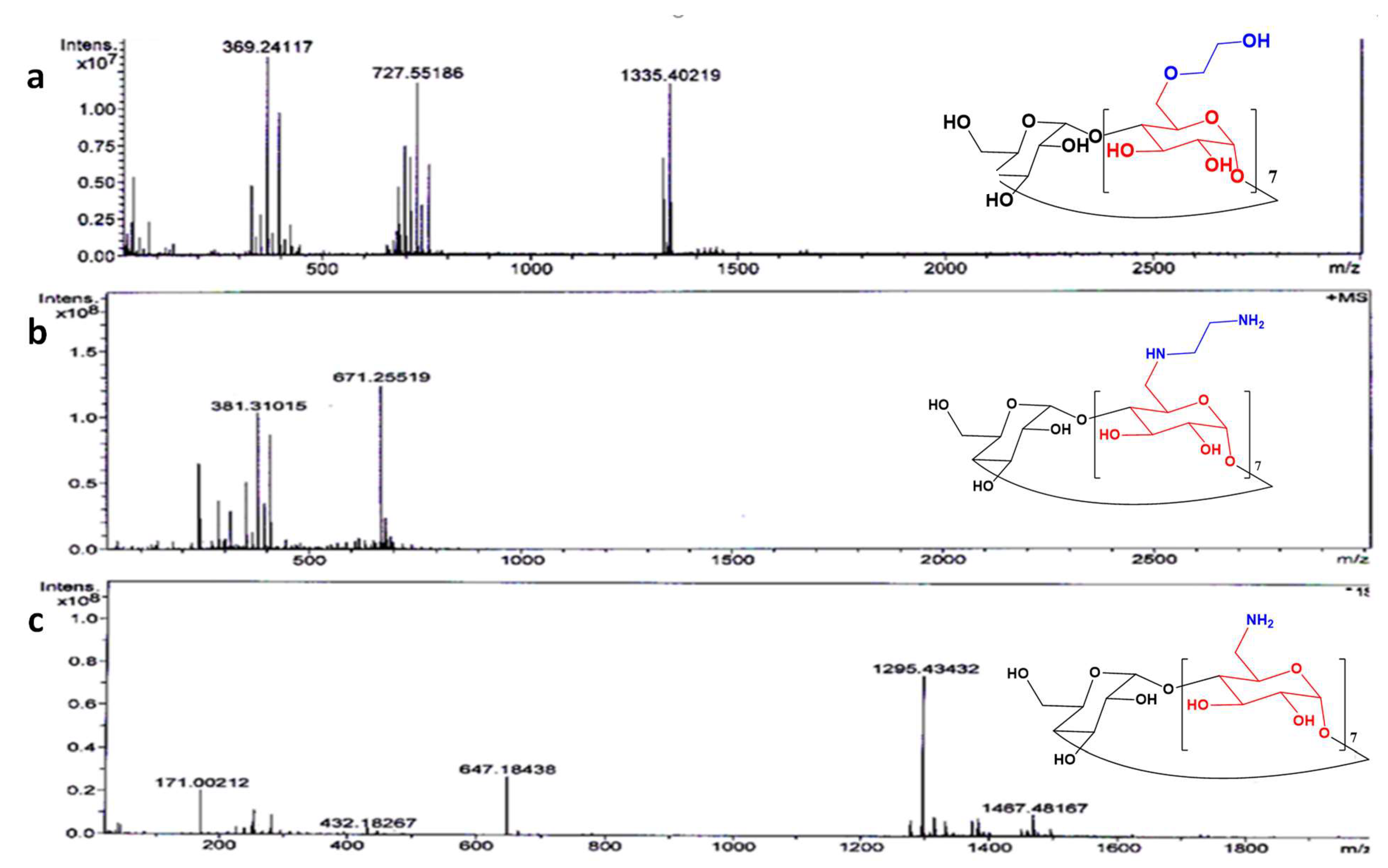
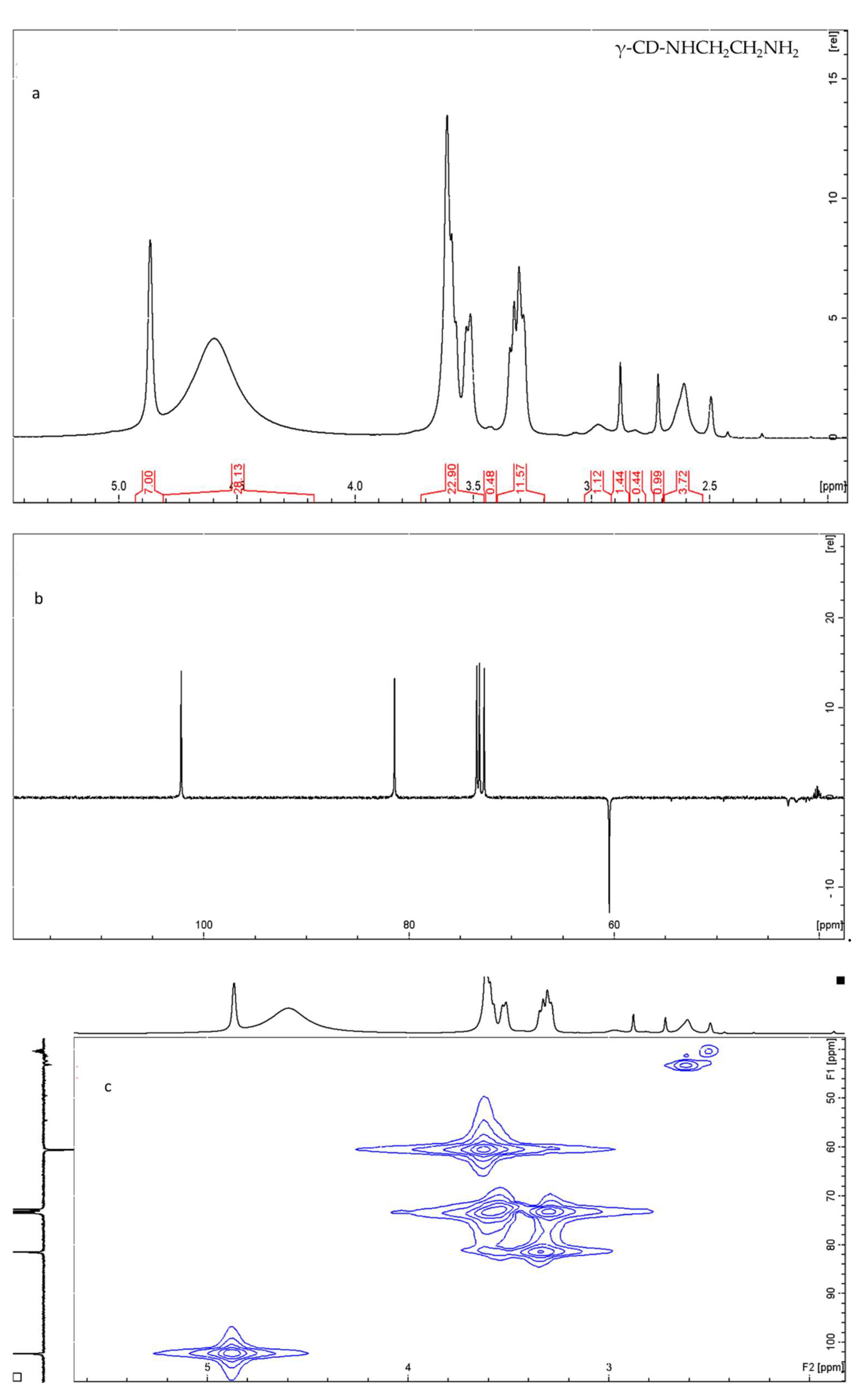
2.1.1. NMR and HR Mass Spectrometry
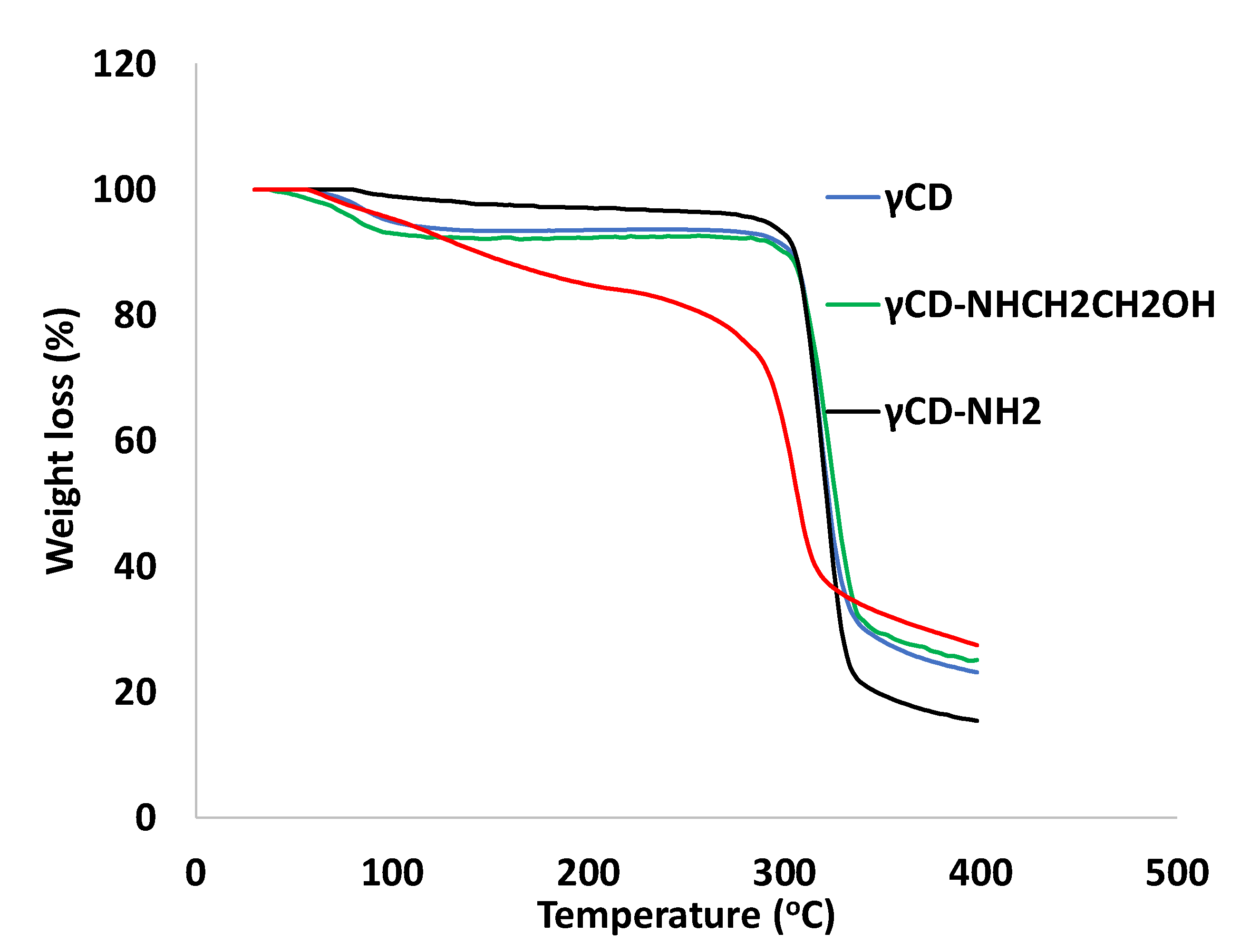
2.1.2. Thermo-Gravimetric Analysis (TGA)
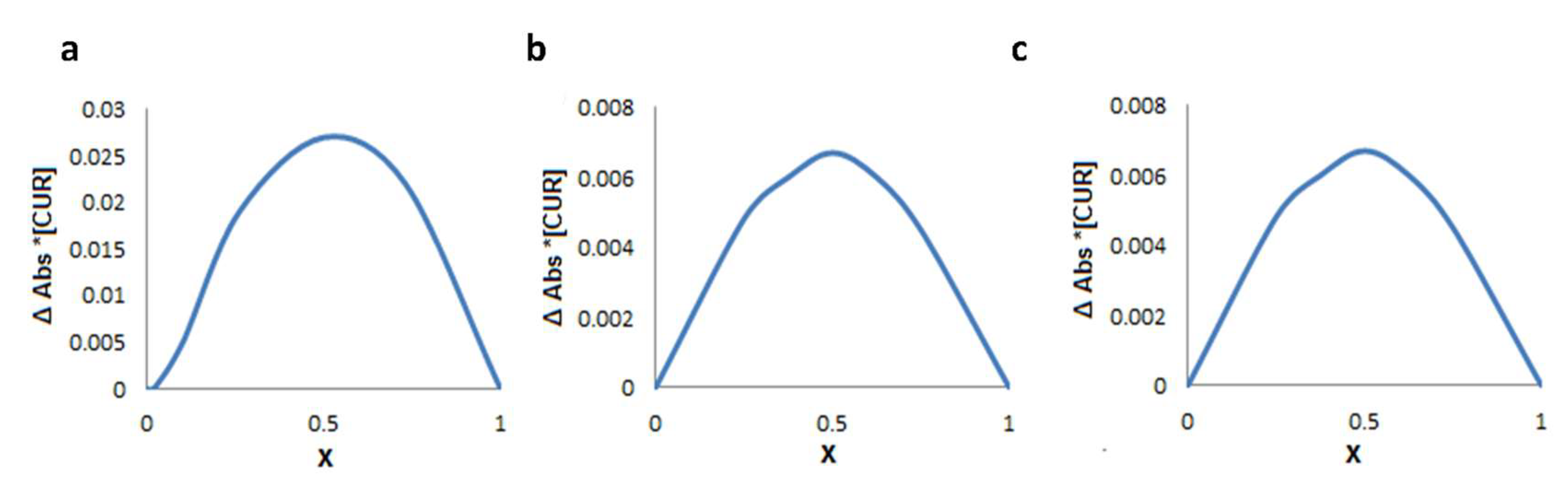
2.2. Guest–Host Interaction between CUR and Modified γ-CDs by Absorption Spectra
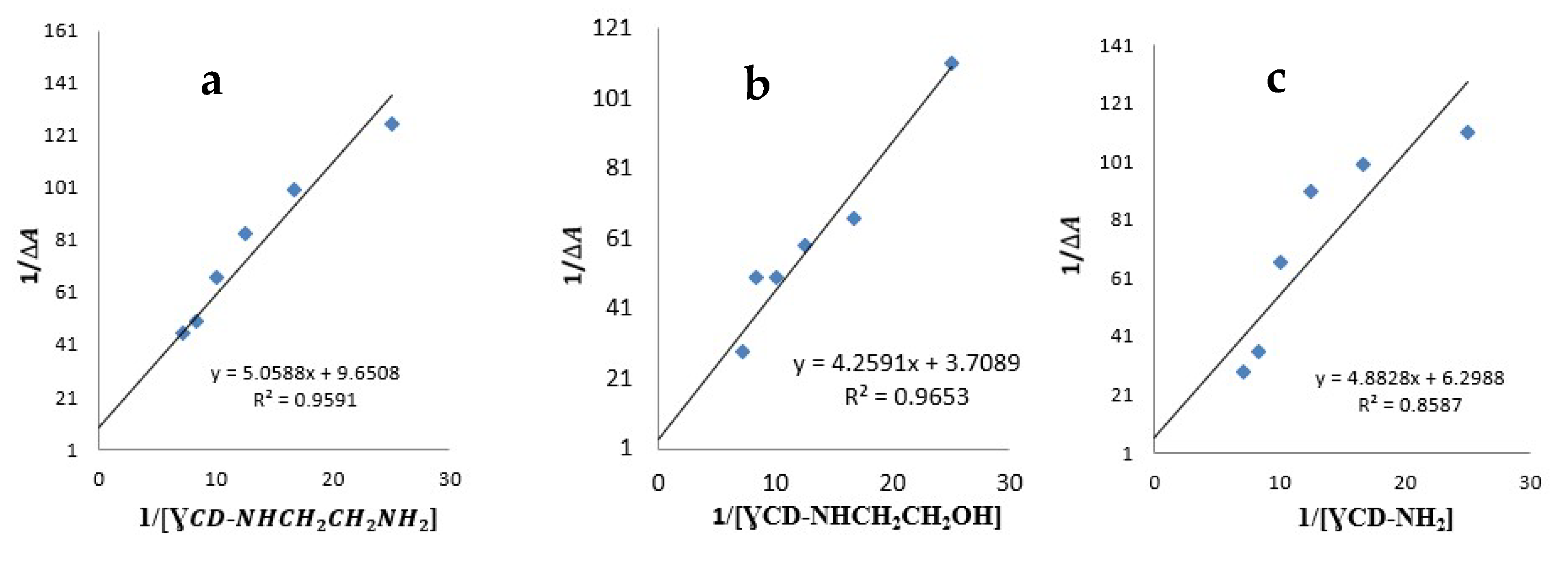
2.3. Determination of the Complexation Stoichiometry and Formation Constants
2.4. Cytotoxicity Assay
3. Materials and Methods
3.1. Chemicals
3.2. Synthesis of Mono Amino Modified γ-CDs
3.3. Nuclear Magnetic Spectroscopy (NMR)
3.4. Mass Spectrometry
3.5. Thermo-Gravimetric Analysis (TGA)
3.6. Preparation of Encapsulation Complex
3.7. Spectrophotometric Studies
3.8. Cells Culture
3.9. Cell Viability Assay
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Wupper, S.; Lüersen, K.; Rimbach, G. Cyclodextrins, Natural Compounds, and Plant Bioactives—A Nutritional Perspective. Biomolecules 2021, 11, 401. [Google Scholar] [CrossRef] [PubMed]
- Crini, G. Review: A History of Cyclodextrins. Chem. Rev. 2014, 114, 10940–10975. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.F.; Monteiro, M.; Resende, D.; Braga, S.S.; Coimbra, M.A.; Silva, A.; Cardoso, S.M. Inclusion Complex of Resveratrol with γ-Cyclodextrin as a Functional Ingredient for Lemon Juices. Foods 2020, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, B.V.K.J.; Hetzer, M.; Ritter, H.; Barner-Kowollik, C. Complex macromolecular architecture design via cyclodextrin host/guest complexes. Prog. Polym. Sci. 2014, 39, 235–249. [Google Scholar] [CrossRef]
- Kyba, E.P.; Helgeson, R.C.; Madan, K.; Gokel, G.W.; Tarnowski, T.L.; Moore, S.S.; Cram, D.J. Host-guest complexation. 1. Concept and illustration. J. Am. Chem. Soc. 1977, 99, 2564–2571. [Google Scholar] [CrossRef]
- Liu, J.Y.; Zhang, X.; Tian, B.R. Selective modifications at the different positions of cyclodextrins: A review of strategies. Turk. J. Chem. 2020, 44, 261–278. [Google Scholar] [CrossRef]
- Hsu, C.M.; Yu, S.C.; Tsai, F.J.; Tsai, Y. Characterization of in vitro and in vivo bioactivity of a ferulic acid-2-Hydroxypropyl-β-cyclodextrin inclusion complex. Colloids Surf. Biointerfaces 2019, 180, 68–74. [Google Scholar] [CrossRef]
- Mori, T.; Tsuchiya, R.; Doi, M.; Nagatani, N.; Tanaka, T. Solubilization of ultraviolet absorbers by cyclodextrin and their potential application in cosmetics. J. Incl. Phenom. Macrocycl. Chem. 2019, 93, 91–96. [Google Scholar] [CrossRef]
- Molnár, Á. Synthetic Application of Cyclodextrins in Combination with Metal Ions, Complexes, and Metal Particles. ChemCatChem 2021, 13, 1424–1474. [Google Scholar] [CrossRef]
- Saokham, P.; Loftsson, T. γ-Cyclodextrin. Int. J. Pharm. 2017, 516, 278–292. [Google Scholar] [CrossRef]
- Yadav, V.R.; Prasad, S.; Kannappan, R.; Ravindran, J.; Chaturvedi, M.M.; Vaahtera, L.; Parkkinen, J.; Aggarwal, B.B. Cyclodextrin-complexed curcumin exhibits anti-inflammatory and antiproliferative activities superior to those of curcumin through higher cellular uptake. Biochem. Pharmacol. 2010, 80, 1021–1032. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dandawate, P.R.; Vyas, A.; Ahmad, A.; Banerjee, S.; Deshpande, J.; Swamy, K.V.; Jamadar, A.; Dumhe-Klaire, A.C.; Padhye, S.; Sarkar, F.H. Inclusion complex of novel curcumin analogue CDF and β-cyclodextrin (1:2) and its enhanced in vivo anticancer activity against pancreatic cancer. Pharm. Res. 2012, 29, 1775–1786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.; Man, S.; Qiu, H.; Liu, Z.; Zhang, M.; Ma, L.; Gao, W. Curcumin-cyclodextrin complexes enhanced the anti-cancer effects of curcumin. Environ. Toxicol. Pharmacol. 2016, 48, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Liu, M.; Chen, A.; Wang, Y.; Sun, D. Solubilities of quercetin in three β-cyclodextrin derivative solutions at different temperatures. J. Mol. Liq. 2013, 177, 204–208. [Google Scholar] [CrossRef]
- Odeh, F.; Nsairat, H.; Alshaer, W.; Alsotari, S.; Buqaien, R.; Ismail, S.; Awidi, A.; Al Bawab, A. Remote loading of curcumin-in-modified β-cyclodextrins into liposomes using a transmembrane pH gradient. RSC Adv. 2019, 9, 37148–37161. [Google Scholar] [CrossRef] [Green Version]
- Albers, E.; Muller, B.W. Cyclodextrin derivatives in pharmaceutics. Crit. Rev. Ther. Drug Carr. Syst. 1995, 12, 311–337. [Google Scholar] [CrossRef]
- Kellett, K.; Kantonen, S.A.; Duggan, B.; Gilson, M.K. Toward Expanded Diversity of Host–Guest Interactions via Synthesis and Characterization of Cyclodextrin Derivatives. J. Solut. Chem. 2018, 47, 1597–1608. [Google Scholar] [CrossRef]
- Řezanka, P.; Navrátilová, K.; Řezanka, M.; Král, V.; Sýkora, D. Application of cyclodextrins in chiral capillary electrophoresis. Electrophoresis 2014, 35, 2701–2721. [Google Scholar] [CrossRef]
- Liang, W.; Huang, Y.; Lu, D.; Ma, X.; Gong, T.; Cui, X.; Yu, B.; Yang, C.; Dong, C.; Shuang, S. β-Cyclodextrin-Hyaluronic Acid Polymer Functionalized Magnetic Graphene Oxide Nanocomposites for Targeted Photo-Chemotherapy of Tumor Cells. Polymers 2019, 11, 133. [Google Scholar] [CrossRef] [Green Version]
- Nsairat, H.; Mahmoud, I.S.; Odeh, F.; Abuarqoub, D.; Al-Azzawi, H.; Zaza, R.; Qadri, M.I.; Ismail, S.; Al Bawab, A.; Awidi, A.; et al. Grafting of anti-nucleolin aptamer into preformed and remotely loaded liposomes through aptamer-cholesterol post-insertion. RSC Adv. 2020, 10, 36219–36229. [Google Scholar] [CrossRef]
- Tang, W.; Ng, S.C. Facile synthesis of mono-6-amino-6-deoxy-α-,β-, γ-cyclodextrin hydrochlorides for molecular recognition, chiral separation and drug delivery. Nat. Protoc. 2008, 3, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Kohata, S.; Jyodoi, K.; Ohyoshi, A. Thermal decomposition of cyclodextrins (α, β.and γ modified α-CYD) and of metal -(α-CYD) complexes in the solid phase. Thermochim. Acta 1993, 217, 187–198. [Google Scholar] [CrossRef]
- Rajaram, R.; Sundararajalu, K.N.; Meenakshisundaram, S. Unusual exited state characteristics of 6-aminobenzothiazole with β-cyclodextrin. J. Fluoresc. 2011, 21, 521–529. [Google Scholar] [CrossRef]
- Hashimoto, S.; Thomas, J.K. Flurescence study of pyrene and naphthalene in cyclodextrin-amphiphile complex system. J. Am. Chem. Soc. 1985, 107, 4655–4662. [Google Scholar] [CrossRef]
- Sayed, M.; Gubbala, G.K.; Pal, H. Contrasting interactions of DNA-intercalating dye acridine orange with hydroxypropyl derivatives of β-cyclodextrin and γ-cyclodextrin hosts. New J. Chem. 2019, 43, 724–736. [Google Scholar] [CrossRef]
- Saokham, P.; Muankaew, C.; Jansook, P.; Loftsson, T. solubility of cyclodextrin and drug/cyclodextrin complexes. Molecules 2018, 23, 1161. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.Y.; Han, J.; Feng, X.G. Spectroscopic of orange γ-β-cyclodextrin complex and its analytical application. Spectrochim. Acta Mol. Biomol. Spectrosc. 2007, 66, 578–785. [Google Scholar] [CrossRef]
- Srinivasan, K.; Stalin, T.; Sivakumar, K. Spectral, and electrochemical study of host-guest inclusion complex between 2,4-dinitrophenol and β-cyclodextrin. Spectrochim. Acta Mol. Biomol. Spectrosc. 2012, 94, 89–100. [Google Scholar] [CrossRef]
- De Azevedo, M.; Alderete, J.B.; Lino, A.; Loh, W.; Faljoni-Alario, A.; DurÁn, N. Violacein/β-cyclodextrin inclusion complex formation studied by measurements of diffusion coefficient and circular dichroism. J. Incl. Phenom. Macrocycl. Chem. 2000, 37, 67–74. [Google Scholar] [CrossRef]
- Lavorgna, M.; Iacovino, R.; Russo, C.; Di Donato, C.; Piscitelli, C.; Isidori, M. A new approach for improving the antibacterial and tumer cytotoxic activities of pipemidic acid by including it in trimethyl—β-cyclodextrin. Int. J. Mol. Sci. 2019, 20, 416. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odeh, F.; Adaileh, F.; Alshaer, W.; Nsairat, H.; Alqudah, D.A.; Jaber, A.M.; Al Bawab, A. Synthesis of Mono-Amino Substituted γ-CD: Host–Guest Complexation and In Vitro Cytotoxicity Investigation. Molecules 2022, 27, 1683. https://doi.org/10.3390/molecules27051683
Odeh F, Adaileh F, Alshaer W, Nsairat H, Alqudah DA, Jaber AM, Al Bawab A. Synthesis of Mono-Amino Substituted γ-CD: Host–Guest Complexation and In Vitro Cytotoxicity Investigation. Molecules. 2022; 27(5):1683. https://doi.org/10.3390/molecules27051683
Chicago/Turabian StyleOdeh, Fadwa, Fedaa Adaileh, Walhan Alshaer, Hamdi Nsairat, Dana A. Alqudah, Areej M. Jaber, and Abeer Al Bawab. 2022. "Synthesis of Mono-Amino Substituted γ-CD: Host–Guest Complexation and In Vitro Cytotoxicity Investigation" Molecules 27, no. 5: 1683. https://doi.org/10.3390/molecules27051683
APA StyleOdeh, F., Adaileh, F., Alshaer, W., Nsairat, H., Alqudah, D. A., Jaber, A. M., & Al Bawab, A. (2022). Synthesis of Mono-Amino Substituted γ-CD: Host–Guest Complexation and In Vitro Cytotoxicity Investigation. Molecules, 27(5), 1683. https://doi.org/10.3390/molecules27051683





