Microalgae Biomass as a New Potential Source of Sustainable Green Lubricants
Abstract
1. Introduction
2. State-of-the-Art of Microalgae-Based Biolubricants
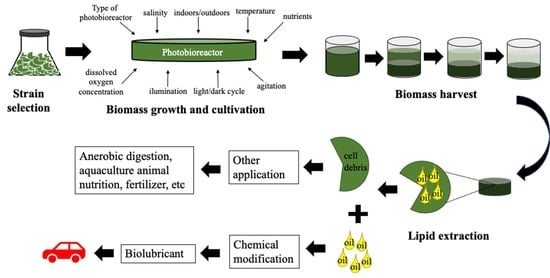
3. Microalgae Oil Production
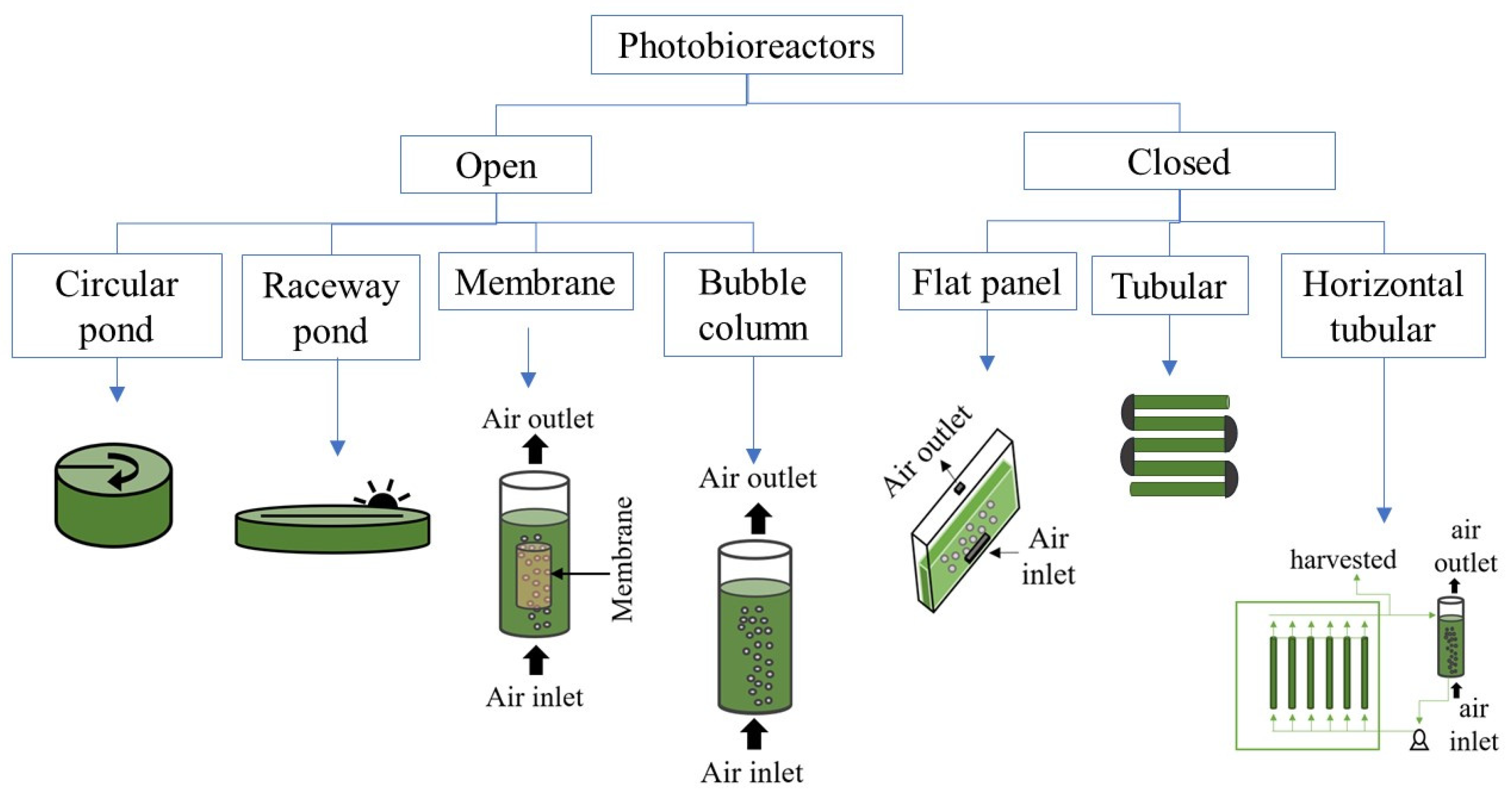
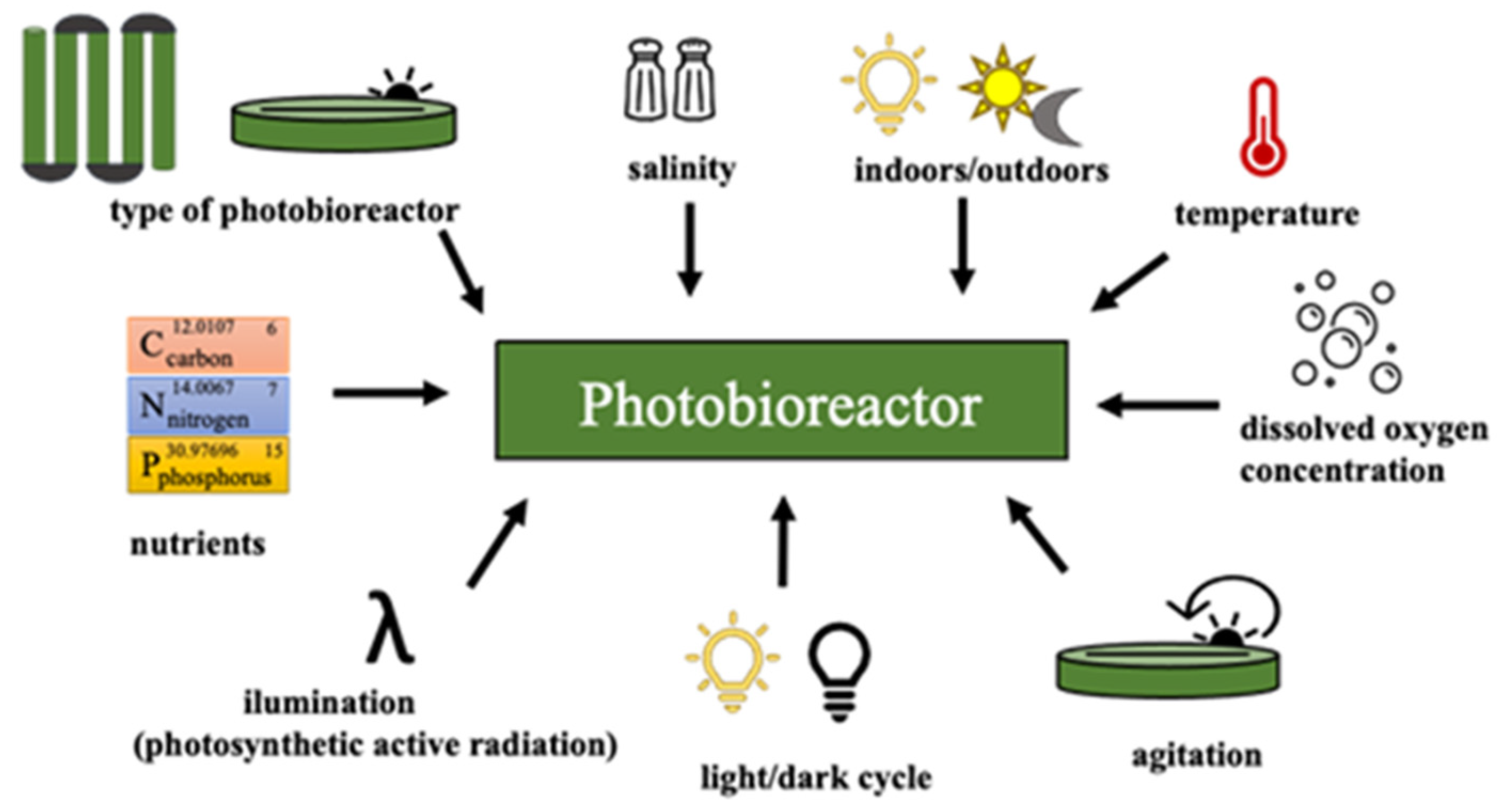
3.1. Cultivation (Photobioreactors and Growth Conditions)
3.2. Lipids (Oil) Accumulation Operational Strategies
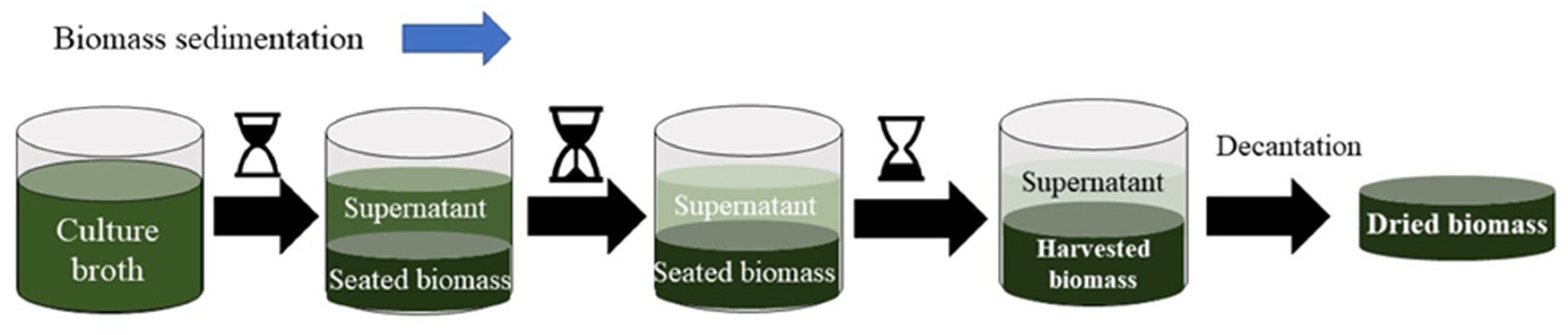
3.3. Microalgae Biomass Harvesting
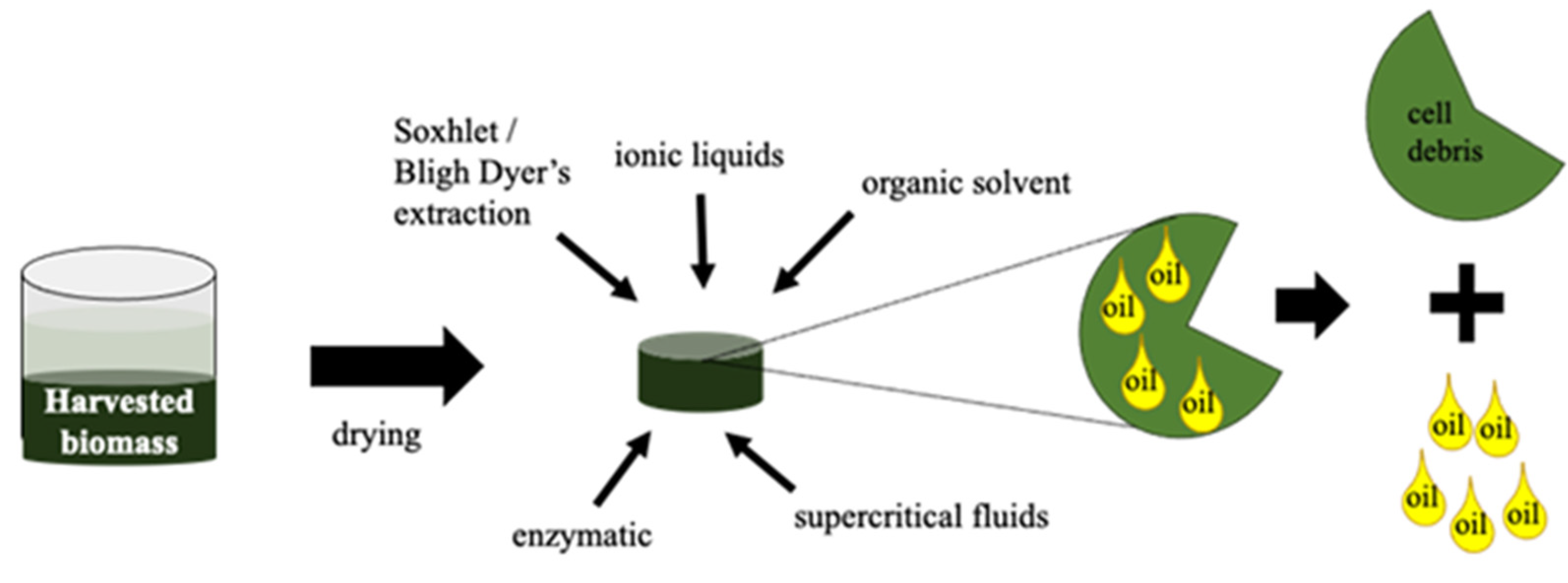
3.4. Microalgae Oil Extraction
4. Considerations of Microalgae Oils as Lubricants
4.1. Relation of Oil Chemical Structure with Its Lubricating Properties
4.1.1. Lubricity
4.1.2. Viscosity
4.1.3. Pour Point
4.1.4. Oxidation Stability
4.2. Chemical Processes for Improving Lubricant Properties of Microalgae Oil
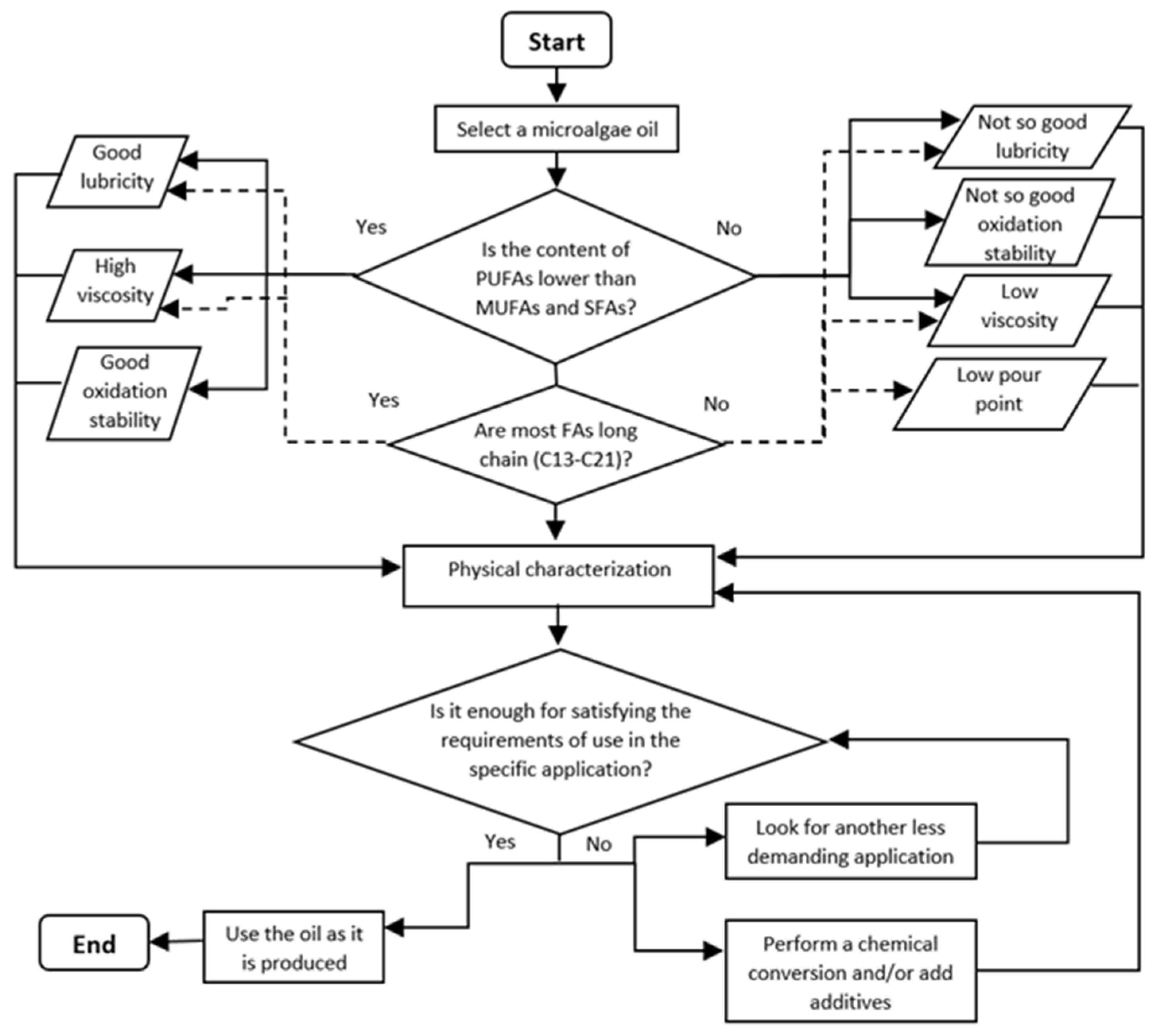
5. A Guide for Selection of Microalgae Oils for Producing Biolubricants for Different Applications
6. Challenges in the Microalgae-to-Biolubricant Production and Use
- -
- Design and construction of photobioreactors (open and close). Deviations from design specifications of photobioreactors due to little experience of construction companies and biologists on these new facilities cause substantial delays to the inception of microalgae biomass production projects.
- -
- Harvesting, seed culture preparation and inoculation for scales up to 300,000 L of microalgae. Lack of skills in handling microalgal cultivation, harvesting and oil extraction operations in large scales is a considerable obstacle for efficient operations.
- -
- Climate changes and environmental and land use regulations. Site specific problems such as unexpected power and water outages, seepages, contamination, water evaporation, drastic weather variations and requirement of special land use permissions can cause production stops, or even, death of the culture broth.
- -
- Assuring product quality and consistency by using green processes. Daily culture sampling and analyses is required to assure consistency and quality of the product. Reaching an acceptable quality and consistency of the product by incorporating green processes for harvesting and oil extraction is currently a research challenge.
- -
- On the other hand, akin to other VOs considered as potential candidates for biolubricants production [24], some challenges must be overcome to use confidently and extensively microalgae biolubricants for industry and vehicle applications. The main challenges are:
- -
- Guaranty homogeneity and continuous availability of the product. It depends on the supplier, feed stocks, and production methods, which is line with the microalgae-based oil production challenges.
- -
- -
- Guaranty of acceptable biodegradability and low toxicity of microalgae biolubricants in case of chemical modification of the base stock oils. Biodegradability of the biolubricants can be worsened by chemical modification and additivation. It must be further evaluated and treated to guaranty the eco-friendly characteristics.
- -
- Acceptance by machine manufacturers. Machine performance, emissions, durability, and biolubricant oxidation in a wide range of machines and sizes need to be demonstrated feasibly and widespread to increase consumer and manufacturer confidence. In addition, the environmental benefits offered by microalgae oil over petroleum lubricating oil, or even VOs, need to be popularized.
7. Conclusions
Authors contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| VO | Vegetable oil |
| MO | Mineral oil |
| SO | Synthetic oil |
| CoF | Coefficient of friction |
| PAO | Poly-alpha-olefin |
| HRAP | High-rate algal pond |
| ATP | Adenosine triphosphate |
| TAG | Triacyclglycerol |
| FA | Fatty acid |
| PUFA | Polyunsaturated fatty acid |
| MUFA | Monounsaturated fatty acid |
| SFA | Saturated fatty acid |
| BL | Boundary lubrication |
References
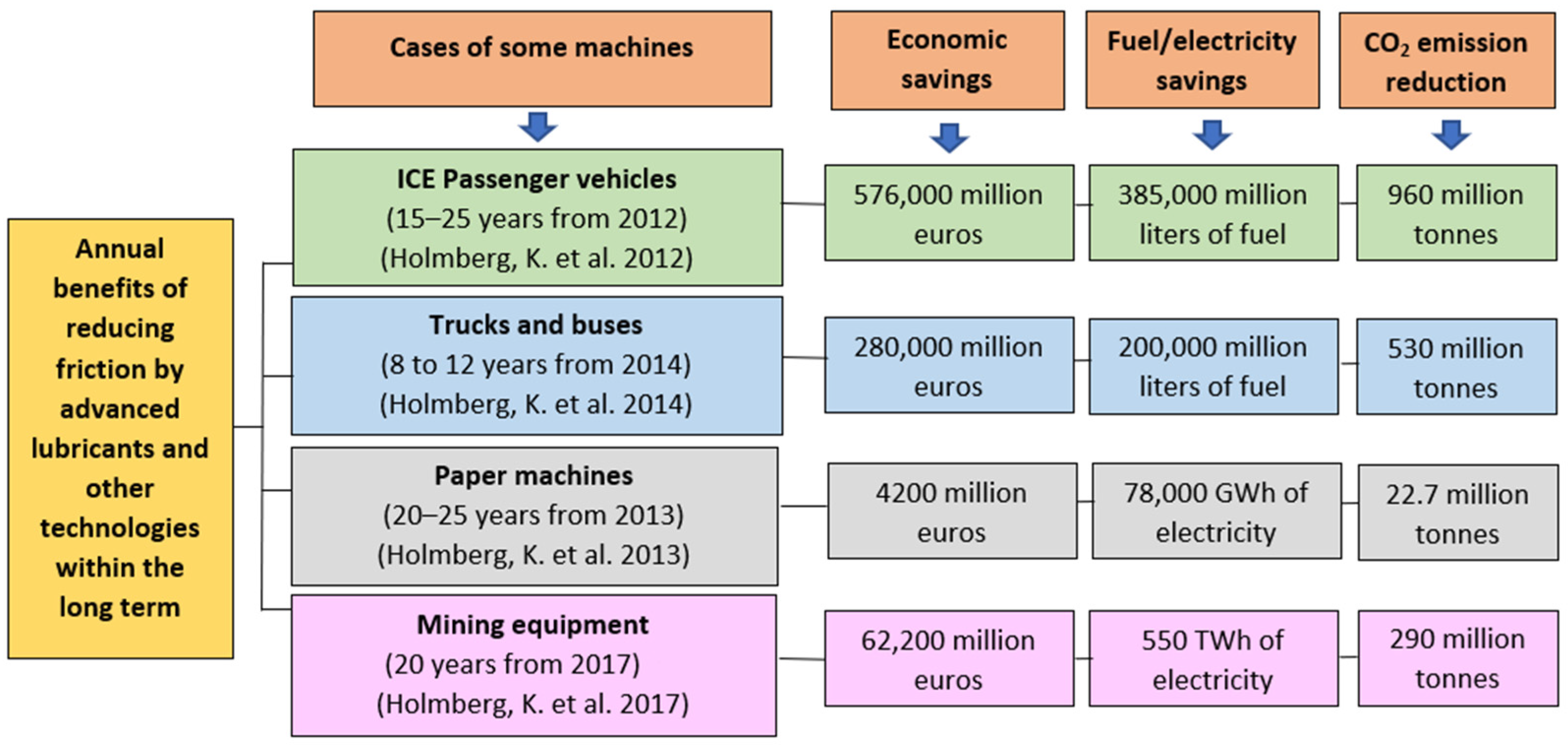
- Jost, H.P. James Clayton Lecture Energy Saving through Tribology: A Techno-Economic Study. Proc. Inst. Mech. Eng. 1981, 195, 151–173. [Google Scholar] [CrossRef]
- Holmberg, K.; Andersson, P.; Erdemir, A. Global energy consumption due to friction in passenger cars. Tribol. Int. 2012, 47, 221–234. [Google Scholar] [CrossRef]
- Holmberg, K.; Siilasto, R.; Laitinen, T.; Andersson, P.; Jäsberg, A. Global energy consumption due to friction in paper machines. Tribol. Int. 2013, 62, 58–77. [Google Scholar] [CrossRef]
- Holmberg, K.; Andersson, P.; Nylund, N.O.; Mäkelä, K.; Erdemir, A. Global energy consumption due to friction in trucks and buses. Tribol. Int. 2014, 78, 94–114. [Google Scholar] [CrossRef]
- Holmberg, K.; Kivikytö-Reponen, P.; Härkisaari, P.; Valtonen, K.; Erdemir, A. Global energy consumption due to friction and wear in the mining industry. Tribol. Int. 2017, 115, 116–139. [Google Scholar] [CrossRef]
- Cunningham, N.; Kool, T.; Owens, J. Oil Price. Available online: https://oilprice.com/oil-price-charts/ (accessed on 21 January 2022).
- Marinescu, I.D.; Rowe, W.B.; Dimitrov, B.; Hitoshi, O. Tribology of Abrasive Machining Processes, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; ISBN 978-1-4377-3467-6. [Google Scholar]
- Owuna, F.J.; Dabai, M.U.; Sokoto, M.A.; Dangoggo, S.M.; Bagudo, B.U.; Birnin-Yauri, U.A.; Hassan, L.G.; Sada, I.; Abubakar, A.L.; Jibrin, M.S. Chemical modification of vegetable oils for the production of biolubricants using trimethylolpropane: A review. Egypt. J. Pet. 2020, 29, 75–82. [Google Scholar] [CrossRef]
- Bloch, P.H.; Geinter, K.F. Practical Machinery Management for Process Plants, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 1997; Volume 2, ISBN 9780080510583. [Google Scholar]
- Luna, F.M.T.; Rocha, B.S.; Rola, E.M.; Albuquerque, M.C.G.; Azevedo, D.C.S.; Cavalcante, C.L. Assessment of biodegradability and oxidation stability of mineral, vegetable and synthetic oil samples. Ind. Crops Prod. 2011, 33, 579–583. [Google Scholar] [CrossRef]
- Willing, A. Lubricants based on renewable resources—An environmentally compatible alternative to mineral oil products. Chemosphere 2001, 43, 89–98. [Google Scholar] [CrossRef]
- Rudnick, L.R. Synthetics, Mineral Oils, and Bio-Based Lubricants Chemistry and Technology, 2nd ed.; Rudnick, L.R., Ed.; CRC Press: Boca Raton, FL, USA, 2013; ISBN 9781138068216. [Google Scholar]
- Zainal, N.A.; Zulkifli, N.W.M.; Gulzar, M.; Masjuki, H.H. A review on the chemistry, production, and technological potential of bio-based lubricants. Renew. Sustain. Energy Rev. 2018, 82, 80–102. [Google Scholar] [CrossRef]
- Global Bio-Lubricants Market to 2024: Regulatory Norms Driving the Demand for Bio-Based Lubricants & Increasing Adoption in the Automotive Industry. Available online: https://www.globenewswire.com/news-release/2019/12/23/1963974/0/en/Global-Bio-lubricants-Market-to-2024-Regulatory-Norms-Driving-the-Demand-for-Bio-based-Lubricants-Increasing-Adoption-in-the-Automotive-Industry.html (accessed on 9 October 2021).
- Mobarak, H.M.; Niza Mohamad, E.; Masjuki, H.H.; Kalam, M.A.; Al Mahmud, K.A.H.; Habibullah, M.; Ashraful, A.M. The prospects of biolubricants as alternatives in automotive applications. Renew. Sustain. Energy Rev. 2014, 33, 34–43. [Google Scholar] [CrossRef]
- Silva, J. Biolubricant Production Catalyzed by Enzymes. In Environmentally Friendly and Biobased Lubricants; Sharma, B., Biresaw, G., Eds.; CRC Press: Boca Raton, FL, USA, 2016; pp. 169–185. [Google Scholar]
- Igartua, A.; Fernández, X.; Areitioaurtena, O.; Luther, R.; Seyfert, C.; Rausch, J.; Illarramendi, I.; Berg, M.; Schultheiß, H.; Duffau, B.; et al. Biolubricants and triboreactive materials for automotive applications. Tribol. Int. 2009, 42, 561–568. [Google Scholar] [CrossRef]
- Heikal, E.K.; Elmelawy, M.S.; Khalil, S.A.; Elbasuny, N.M. Manufacturing of environment friendly biolubricants from vegetable oils. Egypt. J. Pet. 2017, 26, 53–59. [Google Scholar] [CrossRef]
- Ho, C.K.; McAuley, K.B.; Peppley, B.A. Biolubricants through renewable hydrocarbons: A perspective for new opportunities. Renew. Sustain. Energy Rev. 2019, 113, 109261. [Google Scholar] [CrossRef]
- Chan, C.H.; Tang, S.W.; Mohd, N.K.; Lim, W.H.; Yeong, S.K.; Idris, Z. Tribological behavior of biolubricant base stocks and additives. Renew. Sustain. Energy Rev. 2018, 93, 145–157. [Google Scholar] [CrossRef]
- Masudi, A.; Muraza, O. Vegetable Oil to Biolubricants: Review on Advanced Porous Catalysts. Energy Fuels 2018, 32, 10295–10310. [Google Scholar] [CrossRef]
- Singh, Y.; Sharma, A.; Singla, A. Non-edible vegetable oil–based feedstocks capable of bio-lubricant production for automotive sector applications—A review. Environ. Sci. Pollut. Res. 2019, 26, 14867–14882. [Google Scholar] [CrossRef] [PubMed]
- Salimon, J.; Salih, N.; Yousif, E. Biolubricants: Raw materials, chemical modifications and environmental benefits. Eur. J. Lipid Sci. Technol. 2010, 112, 519–530. [Google Scholar] [CrossRef]
- Soni, S.; Agarwal, M. Lubricants from renewable energy sources—A review. Green Chem. Lett. Rev. 2014, 7, 359–382. [Google Scholar] [CrossRef]
- Syahir, A.Z.; Zulkifli, N.W.M.; Masjuki, H.H.; Kalam, M.A.; Alabdulkarem, A.; Gulzar, M.; Khuong, L.S.; Harith, M.H. A review on bio-based lubricants and their applications. J. Clean. Prod. 2017, 168, 997–1016. [Google Scholar] [CrossRef]
- Atabani, A.E.; Silitonga, A.S.; Ong, H.C.; Mahlia, T.M.I.; Masjuki, H.H.; Badruddin, I.A.; Fayaz, H. Non-edible vegetable oils: A critical evaluation of oil extraction, fatty acid compositions, biodiesel production, characteristics, engine performance and emissions production. Renew. Sustain. Energy Rev. 2013, 18, 211–245. [Google Scholar] [CrossRef]
- Issariyakul, T.; Dalai, A.K. Biodiesel from vegetable oils. Renew. Sustain. Energy Rev. 2014, 31, 446–471. [Google Scholar] [CrossRef]
- Paul Kenney, V.; Erichsen, R.L. Conflict between fuel and food: The ethical dimension. Energy Agric. 1983, 2, 285–306. [Google Scholar] [CrossRef]
- Farfan-Cabrera, L.I.; Gallardo-Hernández, E.A.; Gómez-Guarneros, M.; Pérez-González, J.; Godínez-Salcedo, J.G. Alteration of lubricity of Jatropha oil used as bio-lubricant for engines due to thermal ageing. Renew. Energy 2020, 149, 1197–1204. [Google Scholar] [CrossRef]
- Sajeeb, A.; Rajendrakumar, P.K. Comparative evaluation of lubricant properties of biodegradable blend of coconut and mustard oil. J. Clean. Prod. 2019, 240, 118255. [Google Scholar] [CrossRef]
- Singh, Y. Tribological behavior as lubricant additive and physiochemical characterization of Jatropha oil blends. Friction 2015, 3, 320–332. [Google Scholar] [CrossRef]
- Bahari, A.; Lewis, R.; Slatter, T. Friction and wear response of vegetable oils and their blends with mineral engine oil in a reciprocating sliding contact at severe contact conditions. Proc. Inst. Mech. Eng. Part J J. Eng. Tribol. 2018, 232, 244–258. [Google Scholar] [CrossRef]
- Hisham, S.; Kadirgama, K.; Ramasamy, D.; Noor, M.M.; Amirruddin, A.K.; Najafi, G.; Rahman, M.M. Waste cooking oil blended with the engine oil for reduction of friction and wear on piston skirt. Fuel 2017, 205, 247–261. [Google Scholar] [CrossRef]
- Global Industry Analysts Inc. Biolubricants—A Global Market Overview. Available online: https://www.researchandmarkets.com/reports/2819567/biolubricants-a-global-market-overview (accessed on 25 January 2022).
- Farfan-Cabrera, L.I.; Gallardo-Hernández, E.A.; Pérez-González, J.; Marín-Santibáñez, B.M.; Lewis, R. Effects of Jatropha lubricant thermo-oxidation on the tribological behaviour of engine cylinder liners as measured by a reciprocating friction test. Wear 2019, 426–427, 910–918. [Google Scholar] [CrossRef]
- Farfan-Cabrera, L.I.; Gallardo-Hernández, E.A.; Pérez-González, J.; Marín-Santibáñez, B.M.; Lewis, R.; González-Lamas, A.K. Evaluation of thermo-oxidized Jatropha bio-oil in lubrication of actual wet clutch materials. Proc. Inst. Mech. Eng. Part J J. Eng. Tribol. 2021 235, 2021–2033. [CrossRef]
- Bellini, M.; Bianchi, S.; Zaccheria, F.; Ravasio, N. Vegetable oils as Triple Bottom Line compliant lubricants. Tribol. Int. 2021, 161, 107103. [Google Scholar] [CrossRef]
- Zulhanafi, P.; Syahrullail, S. The tribological performances of Super Olein as fluid lubricant using four-ball tribotester. Tribol. Int. 2019, 130, 85–93. [Google Scholar] [CrossRef]
- Zulkifli, N.W.M.; Azman, S.S.N.; Kalam, M.A.; Masjuki, H.H.; Yunus, R.; Gulzar, M. Lubricity of bio-based lubricant derived from different chemically modified fatty acid methyl ester. Tribol. Int. 2016, 93, 555–562. [Google Scholar] [CrossRef]
- Ruggiero, A.; D’Amato, R.; Merola, M.; Valašek, P.; Müller, M. Tribological characterization of vegetal lubricants: Comparative experimental investigation on Jatropha curcas L. oil, Rapeseed Methyl Ester oil, Hydrotreated Rapeseed oil. Tribol. Int. 2017, 109, 529–540. [Google Scholar] [CrossRef]
- Bouabidi, Z.B.; El-Naas, M.H.; Zhang, Z. Immobilization of microbial cells for the biotreatment of wastewater: A review. Environ. Chem. Lett. 2019, 17, 241–257. [Google Scholar] [CrossRef]
- Farrelly, D.J.; Everard, C.D.; Fagan, C.C.; McDonnell, K.P. Carbon sequestration and the role of biological carbon mitigation: A review. Renew. Sustain. Energy Rev. 2013, 21, 712–727. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, J.; Chen, P.; Ji, C.; Kang, Q.; Lu, B.; Li, K.; Liu, J.; Ruan, R. Bio-mitigation of carbon dioxide using microalgal systems: Advances and perspectives. Renew. Sustain. Energy Rev. 2017, 76, 1163–1175. [Google Scholar] [CrossRef]
- Li, K.; Liu, Q.; Fang, F.; Luo, R.; Lu, Q.; Zhou, W.; Huo, S.; Cheng, P.; Liu, J.; Addy, M.; et al. Microalgae-based wastewater treatment for nutrients recovery: A review. Bioresour. Technol. 2019, 291, 121934. [Google Scholar] [CrossRef]
- Serejo, M.L.; Franco Morgado, M.; García, D.; González-Sánchez, A.; Méndez-Acosta, H.O.; Toledo-Cervantes, A. Environmental Resilience by Microalgae. In Microalgae Cultivation for Biofuels Production; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 293–315. ISBN 9780128175361. [Google Scholar]
- Posadas, E.; Marín, D.; Blanco, S.; Lebrero, R.; Muñoz, R. Simultaneous biogas upgrading and centrate treatment in an outdoors pilot scale high rate algal pond. Bioresour. Technol. 2017, 232, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Franco-Morgado, M.; Toledo-Cervantes, A.; González-Sánchez, A.; Lebrero, R.; Muñoz, R. Integral (VOCs, CO2, mercaptans and H2S) photosynthetic biogas upgrading using innovative biogas and digestate supply strategies. Chem. Eng. J. 2018, 354, 363–369. [Google Scholar] [CrossRef]
- Bahr, M.; Díaz, I.; Dominguez, A.; González Sánchez, A.; Muñoz, R. Microalgal-biotechnology as a platform for an integral biogas upgrading and nutrient removal from anaerobic effluents. Environ. Sci. Technol. 2014, 48, 573–581. [Google Scholar] [CrossRef]
- Meier, L.; Pérez, R.; Azócar, L.; Rivas, M.; Jeison, D. Photosynthetic CO2 uptake by microalgae: An attractive tool for biogas upgrading. Biomass Bioenergy 2015, 73, 102–109. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.T.; Show, P.L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Yaakob, Z.; Ali, E.; Zainal, A.; Mohamad, M.; Takriff, M.S. An overview: Biomolecules from microalgae foranimal feed and aquaculture. J. Biol. Res.-Thessalon. 2014, 21, 6. [Google Scholar] [CrossRef] [PubMed]
- Matos, J.; Cardoso, C.; Bandarra, N.M.; Afonso, C. Microalgae as healthy ingredients for functional food: A review. Food Funct. 2017, 8, 2672–2685. [Google Scholar] [CrossRef]
- Deniz, I.; García-Vaquero, M.; Imamoglu, E. Trends in Red Biotechnology: Microalgae for Pharmaceutical Applications. In Microalgae-Based Biofuels and Bioproducts; Muñoz, R., Gonzalez-Fernandez, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 9780081010273. [Google Scholar]
- Ariede, M.B.; Candido, T.M.; Jacome, A.L.M.; Velasco, M.V.R.; de Carvalho, J.C.M.; Baby, A.R. Cosmetic attributes of algae—A review. Algal Res. 2017, 25, 483–487. [Google Scholar] [CrossRef]
- Arad, S.M.; Yaron, A. Natural pigments from red microalgae for use in foods and cosmetics. Trends Food Sci. Technol. 1992, 3, 92–97. [Google Scholar] [CrossRef]
- Khanra, S.; Mondal, M.; Halder, G.; Tiwari, O.N.; Gayen, K.; Bhowmick, T.K. Downstream processing of microalgae for pigments, protein and carbohydrate in industrial application: A review. Food Bioprod. Process. 2018, 110, 60–84. [Google Scholar] [CrossRef]
- Soto-Sierra, L.; Stoykova, P.; Nikolov, Z.L. Extraction and fractionation of microalgae-based protein products. Algal Res. 2018, 36, 175–192. [Google Scholar] [CrossRef]
- Uysal, O.; Uysal, F.O.; Ekinci, K. Evaluation of Microalgae as Microbial Fertilizer. Eur. J. Sustain. Dev. 2015, 4, 77–82. [Google Scholar] [CrossRef]
- Peng, L.; Fu, D.; Chu, H.; Wang, Z.; Qi, H. Biofuel production from microalgae: A review. Environ. Chem. Lett. 2020, 18, 285–297. [Google Scholar] [CrossRef]
- Shuba, E.S.; Kifle, D. Microalgae to biofuels: ‘Promising’ alternative and renewable energy, review. Renew. Sustain. Energy Rev. 2018, 81, 743–755. [Google Scholar] [CrossRef]
- Yousuf, A. (Ed.) Microalgae Cultivation for Biofuels Production; Academic Press: Cambridge, MA, USA, 2020; ISBN 978-0-12-817536-1. [Google Scholar]
- Valdovinos-García, E.M.; Barajas-Fernández, J.; Olán-Acosta, M.d.l.Á.; Petriz-Prieto, M.A.; Guzmán-López, A.; Bravo-Sánchez, M.G. Techno-Economic Study of CO2 Capture of a Thermoelectric Plant Using Microalgae (Chlorella vulgaris) for Production of Feedstock for Bioenergy. Energies 2020, 13, 413. [Google Scholar] [CrossRef]
- Encinar, J.M.; Nogales, S.; González, J.F. Biodiesel and biolubricant production from different vegetable oils through transesterification. Eng. Rep. 2020, 2, e12190. [Google Scholar] [CrossRef]
- Chandra, R.; Iqbal, H.M.N.; Vishal, G.; Lee, H.-S.; Nagra, S. Algal biorefinery: A sustainable approach to valorize algal-based biomass towards multiple product recovery. Bioresour. Technol. 2019, 278, 346–359. [Google Scholar] [CrossRef] [PubMed]
- Chew, K.W.; Yap, J.Y.; Show, P.L.; Suan, N.H.; Juan, J.C.; Ling, T.C.; Lee, D.-J.; Chang, J.-S. Microalgae biorefinery: High value products perspectives. Bioresour. Technol. 2017, 229, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Hu, X.; Yuan, K.; Zhu, G.; Wang, W. Friction and wear behaviors of catalytic methylesterified bio-oil. Tribol. Int. 2014, 71, 168–174. [Google Scholar] [CrossRef]
- Xu, Y.; Zheng, X.; Hu, X.; Dearn, K.D.; Xu, H. Effect of catalytic esterification on the friction and wear performance of bio-oil. Wear 2014, 311, 93–100. [Google Scholar] [CrossRef]
- Dziosa, K.; Makowska, M. A method for the preparation of lubricating oil from microalgae biomass. Tribologia 2016, 270, 33–44. [Google Scholar] [CrossRef]
- Xu, Y.; Peng, Y.; Dearn, K.D.; Zheng, X.; Yao, L.; Hu, X. Synergistic lubricating behaviors of graphene and MoS2 dispersed in esterified bio-oil for steel/steel contact. Wear 2015, 342–343, 297–309. [Google Scholar] [CrossRef]
- Xu, Y.; Geng, J.; Zheng, X.; Dearn, K.D.; Hu, X. Friction-Induced Transformation from Graphite Dispersed in Esterified Bio-Oil to Graphene. Tribol. Lett. 2016, 63, 18. [Google Scholar] [CrossRef]
- Xu, Y.; Peng, Y.; Zheng, X.; Wang, H.; Hu, X. Influence of Microalgal Bio-Oil on the Lubrication Properties of Engine Oil. Oil Gas Sci. Technol. 2016, 71, 29. [Google Scholar] [CrossRef]
- Cheah, M.Y.; Ong, H.C.; Zulkifli, N.W.M.; Masjuki, H.H.; Salleh, A. Physicochemical and tribological properties of microalgae oil as biolubricant for hydrogen-powered engine. Int. J. Hydrogen Energy 2020, 45, 22364–22381. [Google Scholar] [CrossRef]
- Vijayendran, B.; Randall, M.; Schmid, E. Algal Oil Based Bio-Lubricants. Patent WO2016130863A3, 18 August 2016. [Google Scholar]
- Medipally, S.R.; Yusoff, F.; Banerjee, S.; Shariff, M. Microalgae as Sustainable Renewable Energy Feedstock. BioMed Res. Int. 2015, 2015, 519513. [Google Scholar] [CrossRef] [PubMed]
- Ras, M.; Steyer, J.P.; Bernard, O. Temperature effect on microalgae: A crucial factor for outdoor production. Rev. Environ. Sci. Biotechnol. 2013, 12, 153–164. [Google Scholar] [CrossRef]
- Lee, K.K.; Lim, P.E.; Poong, S.W.; Wong, C.Y.; Phang, S.M.; Beardall, J. Growth and photosynthesis of Chlorella strains from polar, temperate and tropical freshwater environments under temperature stress. J. Oceanol. Limnol. 2018, 36, 1266–1279. [Google Scholar] [CrossRef]
- Franco-Morgado, M.; Alcántara, C.; Noyola, A.; Muñoz, R.; González-Sánchez, A. A study of photosynthetic biogas upgrading based on a high rate algal pond under alkaline conditions: Influence of the illumination regime. Sci. Total Environ. 2017, 592, 419–425. [Google Scholar] [CrossRef]
- Ishika, T.; Moheimani, N.R.; Laird, D.W.; Bahri, P.A. Stepwise culture approach optimizes the biomass productivity of microalgae cultivated using an incremental salinity increase strategy. Biomass Bioenergy 2019, 127, 105274. [Google Scholar] [CrossRef]
- Acién, G.; Molina-Grima, E.; Fernandez-Sevilla, J.M. Photobioreactors for the production of microalgae. Rev. Environ. Sci. Bio-Technol. 2013, 12, 131–151. [Google Scholar] [CrossRef]
- Torzillo, G.; Zittelli, G.C. Tubular Photobioreactors. In Algal Biorefineries: Volume 2: Products and Refinery Design; Prokop, A., Bajpai, R.K., Zappi, M.E., Eds.; Springer: Cham, Switzerland, 2015; pp. 187–212. ISBN 9783319202006. [Google Scholar]
- Camacho Rubio, F.; Acién Fernández, F.G.; Sánchez Pérez, J.A.; García Camacho, F.; Molina Grima, E. Prediction of dissolved oxygen and carbon dioxide concentration profiles in tubular photobioreactors for microalgal culture. Biotechnol. Bioeng. 1999, 62, 71–86. [Google Scholar] [CrossRef]
- Yen, H.-W.; Hu, I.-C.; Chen, C.-Y.; Nagarajan, D.; Chang, J.-S. Design of photobioreactors for algal cultivation. In Biofuels from Algae; Pandey, A., Chisti, Y., Lee, D.-J., Soccol, C.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 225–256. ISBN 9780444641922. [Google Scholar]
- Borowitzka, M.A. Commercial production of microalgae: Ponds, tanks, and fermenters tubes Michael. J. Biotechnol. 1999, 70, 313–321. [Google Scholar] [CrossRef]
- Oswald, W.J. Large Scale Algal Culture Systems. In Micro-Algal Biotechnology; Borowitzka, M.A., Borowitzka, L.J., Eds.; Cambridge University Press: Cambridge, UK, 1988; pp. 357–394. [Google Scholar]
- Acién, F.G.; Molina, E.; Reis, A.; Torzillo, G.; Zittelli, G.C.; Sepúlveda, C.; Masojídek, J. Photobioreactors for the production of microalgae. In Microalgae-Based Biofuels and Bioproducts: From Feedstock Cultivation to End-Products; Gonzalez-Fernandez, C., Muñoz, R., Eds.; Woodhead Publishing: Cambridge, UK, 2017; pp. 1–44. ISBN 9780081010273. [Google Scholar]
- Franco-Morgado, M.; Tabaco-Angoa, T.; Ramírez-García, M.A.; González-Sánchez, A. Strategies for decreasing the O2 content in the upgraded biogas purified via microalgae-based technology. J. Environ. Manag. 2021, 279, 111813. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.M.; Geng, L.J.; Ren, L.J.; Ji, X.J.; Hao, N.; Chen, K.Q.; Huang, H. Influence of oxygen on the biosynthesis of polyunsaturated fatty acids in microalgae. Bioresour. Technol. 2018, 250, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Grobbelaar, J.U. Algal Nutrition. In Microalgae Culture: Biotechnology and Applied Phycology; Richmond, A., Ed.; Blackwell Publishing: Oxford, UK, 2004; pp. 97–115. [Google Scholar]
- Ogbonna, J.C.; Tanaka, H. Light requirement and photosynthetic cell cultivation—Development of processes for efficient light utilization in photobioreactors. J. Appl. Phycol. 2000, 12, 207–218. [Google Scholar] [CrossRef]
- Grima, E.M.; Acie, F.G.; Chisti, Y. Photobioreactors: Light regime, mass transfer, and scaleup. J. Biotechnol. 1999, 70, 231–247. [Google Scholar] [CrossRef]
- Benavente-Valdés, J.R.; Aguilar, C.; Contreras-Esquivel, J.C.; Méndez-Zavala, A.; Montañez, J. Strategies to enhance the production of photosynthetic pigments and lipids in chlorophycae species. Biotechnol. Rep. 2016, 10, 117–125. [Google Scholar] [CrossRef]
- Geremia, E.; Ripa, M.; Catone, C.M.; Ulgiati, S. A Review about Microalgae Wastewater Treatment for Bioremediation and Biomass Production. A New Challenge for Europe. Environments 2021, 8, 136. [Google Scholar] [CrossRef]
- Debowski, M.; Zielinski, M.; Kisielewska, M.; Kazimierowicz, J.; Dudek, M.; Swica, I.; Rudnicka, A. The Cultivation of Lipid-Rich Microalgae Biomass as Anaerobic Digestate Valorization Technology—A Pilot-Scale Study. Processes 2020, 8, 517. [Google Scholar] [CrossRef]
- Amenorfenyo, D.K.; Huang, X.; Zhang, Y.; Zeng, Q.; Zhang, N.; Ren, J.; Huang, Q. Microalgae brewery wastewater treatment: Potentials, benefits and the challenges. Int. J. Environ. Res. Public Health 2019, 16, 1910. [Google Scholar] [CrossRef]
- Tsolcha, O.N.; Tekerlekopoulou, A.G.; Akratos, C.S.; Aggelis, G.; Genitsaris, S.; Moustaka-Gouni, M.; Vayenas, D.V. Agroindustrial wastewater treatment with simultaneous biodiesel production in attached growth systems using a mixed microbial culture. Water 2018, 10, 1693. [Google Scholar] [CrossRef]
- Patel, A.K.; Joun, J.M.; Hong, M.E.; Sim, S.J. Effect of light conditions on mixotrophic cultivation of green microalgae. Bioresour. Technol. 2019, 282, 245–253. [Google Scholar] [CrossRef]
- Da Silva, T.L.; Moniz, P.; Silva, C.; Reis, A. The role of heterotrophic microalgae in waste conversion to biofuels and bioproducts. Processes 2021, 9, 1090. [Google Scholar] [CrossRef]
- Park, J.E.; Zhang, S.; Han, T.H.; Hwang, S.J. The contribution ratio of autotrophic and heterotrophic metabolism during a mixotrophic culture of chlorella sorokiniana. Int. J. Environ. Res. Public Health 2021, 18, 1353. [Google Scholar] [CrossRef] [PubMed]
- Kujawska, N.; Talbierz, S.; Dębowski, M.; Kazimierowicz, J.; Zieliński, M. Cultivation method effect on schizochytrium sp. Biomass growth and docosahexaenoic acid (dha) production with the use of waste glycerol as a source of organic carbon. Energies 2021, 14, 2952. [Google Scholar] [CrossRef]
- Benasla, A.; Hausler, R. A Two-Step Cultivation Strategy for High Biomass Production and Lipid Accumulation of Raphidocelis subcapitata Immobilized in Alginate Gel. Biomass 2021, 1, 7. [Google Scholar] [CrossRef]
- Savvidou, M.G.; Ferraro, A.; Schinas, P.; Mamma, D.; Kekos, D.; Hristoforou, E.; Kolisis, F.N. Magnetic Immobilization and Growth of Nannochloropsis oceanica and Scenedasmus almeriensis. Plants 2021, 11, 72. [Google Scholar] [CrossRef]
- Li-Beisson, Y.; Nakamura, Y.; Harwood, J. Lipids: From Chemical Structures, Biosynthesis, and Analyses to Industrial Applications. In Lipids in Plant and Algae Development; Nakamura, Y., Li-Beisson, Y., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 86, ISBN 978-3-319-25977-2. [Google Scholar]
- Durrett, T.P.; Benning, C.; Ohlrogge, J. Plant triacylglycerols as feedstocks for the production of biofuels. Plant J. 2008, 54, 593–607. [Google Scholar] [CrossRef]
- Liu, B.; Benning, C. Lipid metabolism in microalgae distinguishes itself. Curr. Opin. Biotechnol. 2013, 24, 300–309. [Google Scholar] [CrossRef]
- Du, Z.-Y.; Benning, C. Triacylglycerol Accumulation in Photosynthetic Cells in Plants and Algae. In Lipids in Plant and Algae Development; Nakamura, Y., Li-Beisson, Y., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 86, pp. 179–205. ISBN 978-3-319-25977-2. [Google Scholar]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef]
- Abida, H.; Dolch, L.J.; Meï, C.; Villanova, V.; Conte, M.; Block, M.A.; Finazzi, G.; Bastien, O.; Tirichine, L.; Bowler, C.; et al. Membrane glycerolipid remodeling triggered by nitrogen and phosphorus starvation in Phaeodactylum tricornutum. Plant Physiol. 2015, 167, 118–136. [Google Scholar] [CrossRef]
- Khozin-Goldberg, I.; Cohen, Z. Unraveling algal lipid metabolism: Recent advances in gene identification. Biochimie 2011, 93, 91–100. [Google Scholar] [CrossRef]
- Khozin-Goldberg, I.; Cohen, Z. The effect of phosphate starvation on the lipid and fatty acid composition of the fresh water eustigmatophyte Monodus subterraneus. Phytochemistry 2006, 67, 696–701. [Google Scholar] [CrossRef]
- Daliry, S.; Hallajisani, A.; Mohammadi Roshandeh, J.; Nouri, H.; Golzary, A. Investigation of optimal condition for Chlorella vulgaris microalgae growth. Glob. J. Environ. Sci. Manag. 2017, 3, 217–230. [Google Scholar] [CrossRef]
- Niki, E. Lipid peroxidation: Physiological levels and dual biological effects. Free Radic. Biol. Med. 2009, 47, 469–484. [Google Scholar] [CrossRef]
- Kitaya, Y.; Azuma, H.; Kiyota, M. Effects of temperature, CO2/O2 concentrations and light intensity on cellular multiplication of microalgae, Euglena gracilis. Adv. Space Res. 2005, 35, 1584–1588. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yuan, C.; Hu, G.; Li, F. Effects of light intensity on the growth and lipid accumulation of microalga Scenedesmus sp. 11-1 under nitrogen limitation. Appl. Biochem. Biotechnol. 2012, 166, 2127–2137. [Google Scholar] [CrossRef] [PubMed]
- Che, C.A.; Kim, S.H.; Hong, H.J.; Kityo, M.K.; Sunwoo, I.Y.; Jeong, G.T.; Kim, S.K. Optimization of Light Intensity and Photoperiod for Isochrysis Galbana Culture to Improve the Biomass and Lipid Production using 14-L Photobioreactors with Mixed Light Emitting Diodes (LEDs) Wavelength under Two-Phase Culture System. Bioresour. Technol. 2019, 285, 121323. [Google Scholar] [CrossRef]
- Chen, B.; Wan, C.; Mehmood, M.A.; Chang, J.S.; Bai, F.; Zhao, X. Manipulating environmental stresses and stress tolerance of microalgae for enhanced production of lipids and value-added products—A review. Bioresour. Technol. 2017, 244, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Rong, J.; Yang, H.; He, Q.; Zhang, D.; Hu, C. NaCl as an effective inducer for lipid accumulation in freshwater microalgae Desmodesmus abundans. Bioresour. Technol. 2014, 161, 402–409. [Google Scholar] [CrossRef]
- Wang, T.; Ge, H.; Liu, T.; Tian, X.; Wang, Z.; Guo, M.; Chu, J.; Zhuang, Y. Salt stress induced lipid accumulation in heterotrophic culture cells of Chlorella protothecoides: Mechanisms based on the multi-level analysis of oxidative response, key enzyme activity and biochemical alteration. J. Biotechnol. 2016, 228, 18–27. [Google Scholar] [CrossRef]
- Ho, S.H.; Nakanishi, A.; Kato, Y.; Yamasaki, H.; Chang, J.S.; Misawa, N.; Hirose, Y.; Minagawa, J.; Hasunuma, T.; Kondo, A. Dynamic metabolic profiling together with transcription analysis reveals salinity-induced starch-To-lipid biosynthesis in alga Chlamydomonas sp. JSC4. Sci. Rep. 2017, 7, 45471. [Google Scholar] [CrossRef]
- Yuan, W.; Ma, Y.; Wei, W.; Liu, W.; Ding, Y.; Balamurugan, S. Sequential treatment with bicarbonate and low-temperature to potentiate both biomass and lipid productivity in Nannochloropsis oceanica. J. Chem. Technol. Biotechnol. 2019, 94, 3413–3419. [Google Scholar] [CrossRef]
- Iwai, M.; Hori, K.; Sasaki-Sekimoto, Y.; Shimojima, M.; Ohta, H. Manipulation of oil synthesis in Nannochloropsis strain NIES-2145 with a phosphorus starvation-inducible promoter from Chlamydomonas reinhardtii. Front. Microbiol. 2015, 6, 912. [Google Scholar] [CrossRef] [PubMed]
- Pragya, N.; Pandey, K.K.; Sahoo, P.K. A review on harvesting, oil extraction and biofuels production technologies from microalgae. Renew. Sustain. Energy Rev. 2013, 24, 159–171. [Google Scholar] [CrossRef]
- Amaro, H.M.; Guedes, A.C.; Malcata, F.X. Advances and perspectives in using microalgae to produce biodiesel. Appl. Energy 2011, 88, 3402–3410. [Google Scholar] [CrossRef]
- Yin, Z.; Zhu, L.; Li, S.; Hu, T.; Chu, R.; Mo, F.; Hu, D.; Liu, C.; Li, B. A comprehensive review on cultivation and harvesting of microalgae for biodiesel production: Environmental pollution control and future directions. Bioresour. Technol. 2020, 301, 122804. [Google Scholar] [CrossRef]
- Li, S.; Hu, T.; Xu, Y.; Wang, J.; Chu, R.; Yin, Z.; Mo, F.; Zhu, L. A review on flocculation as an efficient method to harvest energy microalgae: Mechanisms, performances, influencing factors and perspectives. Renew. Sustain. Energy Rev. 2020, 131, 110005. [Google Scholar] [CrossRef]
- Letelier-Gordo, C.O.; Holdt, S.L.; De Francisci, D.; Karakashev, D.B.; Angelidaki, I. Effective harvesting of the microalgae Chlorella protothecoides via bioflocculation with cationic starch. Bioresour. Technol. 2014, 167, 214–218. [Google Scholar] [CrossRef]
- Milledge, J.J.; Heaven, S. A review of the harvesting of micro-algae for biofuel production. Rev. Environ. Sci. Biotechnol. 2013, 12, 165–178. [Google Scholar] [CrossRef]
- Enamala, M.K.; Enamala, S.; Chavali, M.; Donepudi, J.; Yadavalli, R.; Kolapalli, B.; Aradhyula, T.V.; Velpuri, J.; Kuppam, C. Production of biofuels from microalgae—A review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renew. Sustain. Energy Rev. 2018, 94, 49–68. [Google Scholar] [CrossRef]
- Show, K.Y.; Lee, D.J. Algal Biomass Harvesting. In Biofuels from Algae; Pandey, A., Lee, D.-J., Chisti, Y., Soccol, C.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 85–110. ISBN 9780444595584. [Google Scholar]
- Kromkamp, J.; Walsby, A.E. A computer model of buoyancy and vertical migration in cyanobacteria. J. Plankton Res. 1990, 12, 161–183. [Google Scholar] [CrossRef]
- Zhou, W.; Cheng, Y.; Li, Y.; Wan, Y.; Liu, Y.; Lin, X.; Ruan, R. Novel fungal pelletization-assisted technology for algae harvesting and wastewater treatment. Appl. Biochem. Biotechnol. 2012, 167, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Ummalyma, S.B.; Gnansounou, E.; Sukumaran, R.K.; Sindhu, R.; Pandey, A.; Sahoo, D. Bioflocculation: An alternative strategy for harvesting of microalgae—An overview. Bioresour. Technol. 2017, 242, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Rawat, I.; Ranjith Kumar, R.; Mutanda, T.; Bux, F. Dual role of microalgae: Phycoremediation of domestic wastewater and biomass production for sustainable biofuels production. Appl. Energy 2011, 88, 3411–3424. [Google Scholar] [CrossRef]
- Rodolfi, L.; Zittelli, G.C.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef]
- Orr, V.C.A.; Rehmann, L. Ionic liquids for the fractionation of microalgae biomass. Curr. Opin. Green Sustain. Chem. 2016, 2, 22–27. [Google Scholar] [CrossRef]
- Lee, S.Y.; Khoiroh, I.; Vo, D.V.N.; Senthil Kumar, P.; Show, P.L. Techniques of lipid extraction from microalgae for biofuel production: A review. Environ. Chem. Lett. 2020, 19, 231–251. [Google Scholar] [CrossRef]
- Kumar, R.R.; Rao, P.H.; Arumugam, M. Lipid extraction methods from microalgae: A comprehensive review. Front. Energy Res. 2015, 3, 1–9. [Google Scholar] [CrossRef]
- Reeves, C.J.; Menezes, P.L.; Jen, T.C.; Lovell, M.R. The influence of fatty acids on tribological and thermal properties of natural oils as sustainable biolubricants. Tribol. Int. 2015, 90, 123–134. [Google Scholar] [CrossRef]
- Askwith, T.C.; Cameron, A.; Crouch, R.F. Chain length of additives in relation to lubricants in thin film and boundary lubrication. Proc. R. Soc. A Math. Phys. Sci. 1966, 291, 500–519. [Google Scholar] [CrossRef]
- Konishi, T.; Perez, J.M. Properties of polyol esters—Lubrication of an aluminum silicon alloy. Tribol. Trans. 1997, 40, 500–506. [Google Scholar] [CrossRef]
- Fox, N.J.; Tyrer, B.; Stachowiak, G.W. Boundary lubrication performance of free fatty acids in sunflower oil. Tribol. Lett. 2004, 16, 275–281. [Google Scholar] [CrossRef]
- Vizintin, J.; Arnek, A.; Ploj, T. Lubricating properties of rapeseed oils compared to mineral oils under a high load oscillating movement. J. Synth. Lubr. 2000, 17, 201–217. [Google Scholar] [CrossRef]
- Adhvaryu, A.; Erhan, S.Z.; Perez, J.M. Tribological studies of thermally and chemically modified vegetable oils for use as environmentally friendly lubricants. Wear 2004, 257, 359–367. [Google Scholar] [CrossRef]
- Aziz, N.A.M.; Yunus, R.; Rashid, U.; Zulkifli, N.W.M. Temperature effect on tribological properties of polyol ester-based environmentally adapted lubricant. Tribol. Int. 2016, 93, 43–49. [Google Scholar] [CrossRef]
- Eychenne, V.; Mouloungui, Z. Relationships between structure and lubricating properties of neopentylpolyol esters. Ind. Eng. Chem. Res. 1998, 37, 4835–4843. [Google Scholar] [CrossRef]
- Knothe, G.; Steidley, K.R. Kinematic viscosity of biodiesel fuel components and related compounds. Influence of compound structure and comparison to petrodiesel fuel components. Fuel 2005, 84, 1059–1065. [Google Scholar] [CrossRef]
- Rodrigues, J.D.A.; Cardoso, F.D.P.; Lachter, E.R.; Estevão, L.R.M.; Lima, E.; Nascimento, R.S.V. Correlating chemical structure and physical properties of vegetable oil esters. J. Am. Oil Chem. Soc. 2006, 83, 353–357. [Google Scholar] [CrossRef]
- Jayadas, N.H.; Nair, K.P. Coconut oil as base oil for industrial lubricants-evaluation and modification of thermal, oxidative and low temperature properties. Tribol. Int. 2006, 39, 873–878. [Google Scholar] [CrossRef]
- Sherwin, R.E. Oxidation and antioxidants in fat and oil processing. J. Am. Oil Chem. Soc. 1978, 55, 809–814. [Google Scholar] [CrossRef]
- Mang, T. Rheology of Lubricants. In Lubricants and Lubrication, 2nd ed.; Dresel, W., Mang, T., Eds.; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2007; pp. 23–33. ISBN 9783527314973. [Google Scholar]
- Rhodes, B.N. Vegetable Oil. In Synthetics, Mineral Oils, and Bio-Based Lubricants; Rudnick, L.R., Ed.; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Torbacke, M.; Rudolphi, Å.K.; Kassfeldt, E. (Eds.) Lubricants: Introduction to Properties and Performance; Wiley: Hoboken, NJ, USA, 2014; ISBN 978-1-118-79974-1. [Google Scholar]
- Holmberg, K.; Erdemir, A. The impact of tribology on energy use and CO2 emission globally and in combustion engine and electric cars. Tribol. Int. 2019, 135, 389–396. [Google Scholar] [CrossRef]
- Farfan-Cabrera, L.I. Tribology of electric vehicles: A review of critical components, current state and future improvement trends. Tribol. Int. 2019, 138, 473–486. [Google Scholar] [CrossRef]
- Perez, J.M.; Rudnick, L.R.; Erhan, S.Z.; Sharma, B.K.; Kohli, K. Natural Oils as Lubricants. In Synthetics, Mineral Oils, and Bio-Based Lubricants Chemistry and Technology; Rudnick, L.R., Ed.; CRC Press: Boca Raton, FL, USA, 2020; ISBN 9781138068216. [Google Scholar]
- Gnanasekaran, D.; Chavidi, V.P. Vegetable Oil Based Bio-Lubricants and Transformer Fluids: Application in Power Plants, 1st ed.; Springer: Singapore, 2018; ISBN 2195-0911. [Google Scholar]
- Cao-Romero-Gallegos, J.A.; Farfan-Cabrera, L.I.; Pérez-González, J.; Marín-Santibáñez, B.M. Tribological and rheological evaluation of a graphene nanosheets-based lubricant for metal-on-metal and wet clutch interfaces. Mater. Lett. 2022, 309, 131441. [Google Scholar] [CrossRef]
- Cermak, S.C.; Isbell, T.A.; Bredsguard, J.W.; Thompson, T.D. Estolides: Synthesis and Applications. In Fatty Acids: Chemistry, Synthesis, and Application; Ahmad, M.U., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 431–475. ISBN 9780128095218. [Google Scholar]
- Ayan, E.D.; Sert, E.; Atalay, F.S. An investigation of the production and lubricity characteristics of fatty acid esters. Energy Sources Part A Recover. Util. Environ. Eff. 2014, 36, 64–72. [Google Scholar] [CrossRef]
- Romsdahl, T.; Shirani, A.; Minto, R.E.; Zhang, C.; Cahoon, E.B.; Chapman, K.D.; Berman, D. Nature-Guided Synthesis of Advanced Bio-Lubricants. Sci. Rep. 2019, 9, 11711. [Google Scholar] [CrossRef] [PubMed]
- Wadumesthrige, K.; Ara, M.; Salley, S.O.; Ng, K.Y.S. Investigation of lubricity characteristics of biodiesel in petroleum and synthetic fuel. Energy Fuels 2009, 23, 2229–2234. [Google Scholar] [CrossRef]
- Becker, R.; Knorr, A. An evaluation of antioxidants for vegetable oils at elevated temperatures. Lubr. Sci. 1996, 8, 95–117. [Google Scholar] [CrossRef]
- Asadauskas, S.; Perez, J.M.; Duda, J.L. Lubrication properties of castor oil—Potential basestock for biodegradable lubricants. Lubr. Eng. 1997, 53, 35–40. [Google Scholar]
- Karmakar, G.; Ghosh, P.; Sharma, B.K. Chemically modifying vegetable oils to prepare green lubricants. Lubricants 2017, 5, 44. [Google Scholar] [CrossRef]
- Shomchoam, B.; Yoosuk, B. Eco-friendly lubricant by partial hydrogenation of palm oil over Pd/γ-Al2O3 catalyst. Ind. Crop. Prod. 2014, 62, 395–399. [Google Scholar] [CrossRef]
- Fernández, M.B.; Sánchez, M.J.F.; Tonetto, G.M.; Damiani, D.E. Hydrogenation of sunflower oil over different palladium supported catalysts: Activity and selectivity. Chem. Eng. J. 2009, 155, 941–949. [Google Scholar] [CrossRef]
- Yunus, R.; Fakhru’l-Razi, A.; Ooi, T.L.; Omar, R.; Idris, A. Synthesis of Palm Oil Based Trimethylolpropane Esters with Improved Pour Points. Ind. Eng. Chem. Res. 2005, 44, 8178–8183. [Google Scholar] [CrossRef]
- Wagner, H.; Luther, R.; Mang, T. Lubricant base fluids based on renewable raw materials: Their catalytic manufacture and modification. Appl. Catal. A Gen. 2001, 221, 429–442. [Google Scholar] [CrossRef]
- Abdullah, B.M.; Salimon, J. Epoxidation of Vegetable Oils- Catalyst, Methods and Advantages. J. Appl. Sci. 2010, 10, 1545–1553. [Google Scholar]
- Chen, J.; De Liedekerke Beaufort, M.; Gyurik, L.; Dorresteijn, J.; Otte, M.; Klein Gebbink, R.J.M. Highly efficient epoxidation of vegetable oils catalyzed by a manganese complex with hydrogen peroxide and acetic acid. Green Chem. 2019, 21, 2436–2447. [Google Scholar] [CrossRef]
- Danov, S.M.; Kazantsev, O.A.; Esipovich, A.L.; Belousov, A.S.; Rogozhin, A.E.; Kanakov, E.A. Recent advances in the field of selective epoxidation of vegetable oils and their derivatives: A review and perspective. Catal. Sci. Technol. 2017, 7, 2–6. [Google Scholar] [CrossRef]
- Balamurugan, K.; Kanagasabapathy, N.; Mayilsamy, K. Studies on soya bean oil based lubricant for diesel engines. J. Sci. Ind. Res. 2010, 69, 794–797. [Google Scholar]
- Roegiers, M.; Zhmud, B. Tribological performance of ionised vegetable oils as lubricity and fatty oiliness additives in lubricants and fuels. Lubr. Sci. 2009, 21, 169–182. [Google Scholar] [CrossRef]
- Quinchia, L.A.; Delgado, M.A.; Valencia, C.; Franco, J.M.; Gallegos, C. Natural and synthetic antioxidant additives for improving the performance of new biolubricant formulations. J. Agric. Food Chem. 2011, 59, 12917–12924. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.K.; Perez, J.M.; Erhan, S.Z. Soybean oil-based lubricants: A search for synergistic antioxidants. Energy Fuels 2007, 21, 2408–2414. [Google Scholar] [CrossRef]
- Singh, R.K.; Kukrety, A.; Chatterjee, A.K.; Thakre, G.D.; Bahuguna, G.M.; Saran, S.; Adhikari, D.K.; Atray, N. Use of an acylated chitosan Schiff base as an ecofriendly multifunctional biolubricant additive. Ind. Eng. Chem. Res. 2014, 53, 18370–18379. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, R.K. A search for ecofriendly detergent/dispersant additives for vegetable-oil based lubricants. J. Surfactants Deterg. 2012, 15, 399–409. [Google Scholar] [CrossRef]
- Rudnick, L.R. Lubricant Additives: Chemistry and Applications; Rudnick, L.R., Ed.; CRC Press: Boca Raton, FL, USA, 2010; ISBN 9781498731720. [Google Scholar]
- Quinchia, L.A.; Delgado, M.A.; Valencia, C.; Franco, J.M.; Gallegos, C. Viscosity modification of high-oleic sunflower oil with polymeric additives for the design of new biolubricant formulations. Environ. Sci. Technol. 2009, 43, 2060–2065. [Google Scholar] [CrossRef]
- Quinchia, L.A.; Delgado, M.A.; Valencia, C.; Franco, J.M.; Gallegos, C. Viscosity modification of different vegetable oils with EVA copolymer for lubricant applications. Ind. Crops Prod. 2010, 32, 607–612. [Google Scholar] [CrossRef]
- Chiu, I.C.; Gunsel, C.; Lacey, P. Environmentally friendly lubricants. U.S. Patent 20030186824A1, 15 February 2006. [Google Scholar]
- Bart, J.C.; Cavallaro, S.; Gucciardi, E. Biolubricants: Science and Technology; Woodhead Publishing: Cambridge, UK, 2013. [Google Scholar]
- Hwang, H.S.; Erhan, S.Z. Modification of epoxidized soybean oil for lubricant formulations with improved oxidative stability and low pour point. JAOCS J. Am. Oil Chem. Soc. 2001, 78, 1179–1184. [Google Scholar] [CrossRef]
- Asadauskas, S.; Erhan, S.Z. Depression of pour points of vegetable oils by blending with diluents used for biodegradable lubricants. J. Am. Oil Chem. Soc. 1999, 76, 313–316. [Google Scholar] [CrossRef]
- Madihalli, C.; Sudhakar, H.; Doble, M. Mannosylerythritol Lipid-A as a Pour Point Depressant for Enhancing the Low-Temperature Fluidity of Biodiesel and Hydrocarbon Fuels. Energy Fuels 2016, 30, 4118–4125. [Google Scholar] [CrossRef]
- Eisentraeger, A.; Schmidt, M.; Murrenhoff, H.; Dott, W.; Hahn, S. Biodegradability testing of synthetic ester lubricants—Effects of additives and usage. Chemosphere 2002, 48, 89–96. [Google Scholar] [CrossRef]
- Hahn, S.; Dott, W.; Eisentraeger, A. The influence of application and additives on ecotoxicity and biodegradability of environmentally acceptable ester-based lubricants. J. Synth. Lubr. 2003, 20, 123–137. [Google Scholar] [CrossRef]
- Cecutti, C.; Agius, D. Ecotoxicity and biodegradability in soil and aqueous media of lubricants used in forestry applications. Bioresour. Technol. 2008, 99, 8492–8496. [Google Scholar] [CrossRef][Green Version]
- Luna, F.M.T.; Cavalcante, J.B.; Silva, F.O.N.; Cavalcante, C.L. Studies on biodegradability of bio-based lubricants. Tribol. Int. 2015, 92, 301–306. [Google Scholar] [CrossRef]
- Briassoulis, D.; Mistriotis, A.; Mortier, N.; Tosin, M. A horizontal test method for biodegradation in soil of bio-based and conventional plastics and lubricants. J. Clean. Prod. 2020, 242, 118392. [Google Scholar] [CrossRef]
- Beran, E. Experience with evaluating biodegradability of lubricating base oils. Tribol. Int. 2008, 41, 1212–1218. [Google Scholar] [CrossRef]
- Cabanelas, I.T.D.; Marques, S.S.I.; de Souza, C.O.; Druzian, J.I.; Nascimento, I.A. Botryococcus, what to do with it? Effect of nutrient concentration on biorefinery potential. Algal Res. 2015, 11, 43–49. [Google Scholar] [CrossRef]
- Hempel, N.; Petrick, I.; Behrendt, F. Biomass productivity and productivity of fatty acids and amino acids of microalgae strains as key characteristics of suitability for biodiesel production. J. Appl. Phycol. 2012, 24, 1407–1418. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.J.; Ji, X.J.; Huang, H.; Qu, L.; Feng, Y.; Tong, Q.Q.; Ouyang, P.K. Development of a stepwise aeration control strategy for efficient docosahexaenoic acid production by Schizochytrium sp. Appl. Microbiol. Biotechnol. 2010, 87, 1649–1656. [Google Scholar] [CrossRef]
- Ren, L.J.; Sun, L.N.; Zhuang, X.Y.; Qu, L.; Ji, X.J.; Huang, H. Regulation of docosahexaenoic acid production by Schizochytrium sp.: Effect of nitrogen addition. Bioprocess Biosyst. Eng. 2014, 37, 865–872. [Google Scholar] [CrossRef]
- Meiser, A.; Schmid-Staiger, U.; Trösch, W. Optimization of eicosapentaenoic acid production by Phaeodactylum tricornutum in the flat panel airlift (FPA) reactor. J. Appl. Phycol. 2004, 16, 215–225. [Google Scholar] [CrossRef]
- Guo, D.-S.; Ji, X.J.; Ren, L.J.; Li, G.L.; Huang, H. Improving Docosahexaenoic Acid Production by Schizochytrium sp. using a Newly Designed High-Oxygen-Supply Bioreactor. AIChE J. 2017, 63, 4278–4286. [Google Scholar] [CrossRef]
- Mohammad Mirzaie, M.A.; Kalbasi, M.; Ghobadian, B.; Mousavi, S.M. Kinetic modeling of mixotrophic growth of Chlorella vulgaris as a new feedstock for biolubricant. J. Appl. Phycol. 2016, 28, 2707–2717. [Google Scholar] [CrossRef]
- Da Silva, A.P.T.; Bredda, E.H.; de Castro, H.F.; Da Rós, P.C.M. Enzymatic catalysis: An environmentally friendly method to enhance the transesterification of microalgal oil with fusel oil for production of fatty acid esters with potential application as biolubricants. Fuel 2020, 273, 117786. [Google Scholar] [CrossRef]
- Bredda, E.H.; Silva, M.B.; De Castro, H.F.; Silva, A.P.T.; Da Rós, P.C.M. Microalgae as a Source of Functional PUFAs: A Green Low-cost Pathway via Enzymatic Hydrolysis. J. Adv. Biol. Biotechnol. 2019, 20, 1–13. [Google Scholar] [CrossRef]
- da Silva, T.; Reis, A. Scale-up Problems for the Large Scale Production of Algae. In Algal Biorefinery: An Integrated Approach; Das, D., Ed.; Springer International Publishing: Cham, Switzerland, 2015; ISBN 978-3-319-22813-6. [Google Scholar]
- Farfán-Cabrera, L.I.; Gallardo-Hernández, E.A.; Reséndiz-Calderón, C.D.; Sedano de la Rosa, C. Physical and Tribological Properties Degradation of Silicone Rubber Using Jatropha Biolubricant. Tribol. Trans. 2018, 61, 640–647. [Google Scholar] [CrossRef]
- Farfan-Cabrera, L.I.; Gallardo-Hernández, E.A.; Pérez-González, J. Compatibility study of common sealing elastomers with a biolubricant (Jatropha oil). Tribol. Int. 2017, 116, 1–8. [Google Scholar] [CrossRef]
- Carrell, J.; Lewis, R.; Slatter, T. Elastomer solubility and stress relaxation in bio-lubricants. Tribol. Int. 2020, 141, 105947. [Google Scholar] [CrossRef]
| Stress Condition | Effect of the Stress Condition on Growth and Lipids Accumulation | References |
|---|---|---|
| Deprivation of nutrients | Deprivation of nitrogen and phosphorus promote larger lipid accumulation in microalgae cells. | [107,108,109,110] |
| Oxygen saturation | Higher accumulation of dissolved oxygen in the culture broth promotes lipid degradation and decreases biomass productivity. So, it is important to regulate oxygen accumulation to enhance lipids increase. | [87,111] |
| Light intensity, illuminated/dark cycles and CO2 supply | Supply of appropriate light intensity and illuminated/dark cycles improves growth of different species of microalgae and lipid accumulation concomitant with CO2 supply at different concentrations. | [110,112,113,114] |
| Salinity | Salinity stimulates the accumulation of lipids in microalgae and avoids cell damage. | [115,116,117,118] |
| Temperature | Temperature changes promote enhancement in biomass productivity and carbon precursors for lipid hyper accumulation. | [119] |
| Mutant genes | Application of mutant genes increases lipid accumulation in microalgae cells due to changes in its metabolism. | [120] |
| Type | Method | Description | Examples | Advantage | Disadvantage | References |
|---|---|---|---|---|---|---|
| Chemical | Flocculation/ Coagulation | Separation by interaction between negative charged cells and flocculant ions producing scattered units that settling down. |
|
|
| [123,124,125] |
| Physical | Centrifugation | Separation by centrifugal force. |
|
|
| [123,126,127] |
| Filtration | Separation by using a filter medium through which only the fluid can pass. |
|
|
| [124,127,128] | |
| Sedimentation | Separation by the action of gravity leaving a supernatant. |
|
|
| [123,129] | |
| Flotation | Separation by using a thick foamed bed from which the solids are recovered by skimming. |
|
|
| [123,128] | |
| Electrical methods | Separation by application of an electric field to the microalgae cells with metallic electrodes. |
|
|
| [123,128] | |
| Biological | Bioflocculation Autobioflocculation | Separation by the addition of micro- or macro-organisms to induce extra cellular polymer substances that promote flocculation through changes in pH, carbon sources supply, etc. |
|
|
| [123,128,130,131] |
| Oil Extraction Technique | Description | Advantages | Disadvantage | References |
|---|---|---|---|---|
| Organic Solvents | Use of non-polar solvents to disrupt interactions between non-polar/neutral lipids. |
|
| [121,132] |
| Soxhlet extraction/Bligh and Dyer’s method | Use of hexane/mixture of chloroform and methanol as solvent to disrupt interactions between non-polar/neutral lipids. |
|
| [121] |
| Ionic liquids | Use of ionic solvents to disrupt microalgae cells and extract lipids. |
|
| [134] |
| Supercritical fluids | Use of supercritical fluids as solvents to extract lipids. |
|
| [135] |
| Mechanical process | Use of mechanical methods as bead beating, pressing, ultrasonic-assisted extraction, electroporation, etc. for cell disruption and simultaneous oil extraction |
|
| [136] |
| Property | Requirement | Chemical Factor | Characteristics | References |
|---|---|---|---|---|
| Lubricity | Low friction and wear under boundary lubrication. | Carbon chain length | Long (n ≥ 9) and linear (SFA, MUFA) | [138,139] |
| FAs | Low unsaturation degree (SFA, MUFA) | [140,141] | ||
| Polarity | High | [139,142,143] | ||
| Viscosity | Low viscosity | Carbon chain length | Short (viscosity increases with chain length) | [144,145] |
| FAs | High unsaturation degree (PUFA) | [146] | ||
| High viscosity | Carbon chain length | Long (viscosity increases with chain length) | [144,145] | |
| FAs | Low unsaturation degree (SFA, MUFA) | [146] | ||
| Pour point | Low pour point | FAs | High unsaturation degree (PUFA) | [144,147] |
| Oxidation stability | High oxidation stability | FAs | Low unsaturation degree (SFA, MUFA) | [148] |
| Additive | Function | Reported Effective Chemical Additives for Bio-Lubricants | References |
|---|---|---|---|
| Antioxidants | Interrupt or prevent the oxidation process without modifying other lubricant properties required. The process occurs in different ways depending on the structure and antioxidant mechanism. | Tocopherol, propyl gallate, l-ascorbic acid 6-palmitate, synthetic antioxidants (4,4′-methylenebis (2,6-di-tert-butylphenol), zinc diamyl dithiocarbamate, butylated hydroxy toluene, alkylated phenol/dithiophosphoric acid ester/diphenylamine and acylated chitosan schiff | [173,174,175] |
| Detergents and dispersants | Prevent the accumulation of sludge particles or other oil-insoluble substances. by dispersing and keeping them suspended in the oil. | Metal sulfonate, ash-less sulfonate, over based sulfonate, salicylates, alkyl phenolates, overbased carboxylate, polyisobu-tylene succinimides, glycidol modified succinimides, Mannich adducts, polyethylene glycol esters, polyol poly- (12-hydroxy stearic acid), piperazine derivatives, butyl citrate and ethyl oleate. | [176,177] |
| Viscosity modifiers | Provide the bio-lubricant with the viscosity magnitude required for both low and high temperatures keeping appropriate lubricity. | Olefin copolymer, ethylene-vinyl acetate (EVA) copolymer, olymethacrylates, styrene-diene copolymers and styrene-ester copolymers. | [178,179,180,181] |
| Pour point depressants | Limit the formation of large crystals during solidification process to provide oil flow at low temperatures. | Polymethacrylate backbone with a certain type of branching, 2-ethylhexyl oleate, isobutyl oleate, trimethylolpropane trioleate, pentaerythritol tetraoleate, diisodecyl adipate and Mannosylerythritol lipid. | [182,183,184] |
| Strain | Biomass Productivity (g L−1 h−1) | g Lipids/100 g DW | g Fatty Acid/100 g Lipids | Fatty Acids Profileg x-Fatty Acid/100 g Lipids | Photobioreactor/Illumination | Operational Conditions | Medium | Oil Extraction Method | References |
|---|---|---|---|---|---|---|---|---|---|
| B. braunii (UTEX LB 572) | 0.18–0.22 | 14.72–15.64 | 7.26–11.69 SFA 63.10–82.9 MUFA 9.45–25.21 PUFA | (C4:0) 0.37–0.46 (C12:0) 0.20 (C14:0) 0.20–0.68 (C16:0) 6.40–9.36 (C16:1w7) 0.17–0.32 (C17:1w5) 1.22–2.68 (C18:0) 0.56–1.27 (C18:1w9c) 59.55–81.79 (C18:2w6c) 2.26–7.00 (C18:3w3) 5.27–11.20 (C20:1w9) 0.45–0.55 (C20:3w3) 1.21 (C20:4w6) 0.71–0.92 (C20:5w3) 1.56–2.23 (C22:1w9) 0.28 (C22:6w3) 3.87–3.88 | Erlenmeyer flasks 170 μEm−2 s−1. | 12 h light:12 h dark. 25 °C | CHU-medium, (g L−1) (0.2 KNO3; 0.04 K2HPO4; 0.08 CaCl2·2H2O; 0.1 MgSO4·7H2O; 0.1 C6H8O7·H2O; 0.1 C6H5FeO7·xH2O) | Chloroform-methanol (1:2) | [191] |
| B. braunii (IBL-C117) | 0.10–0.15 | 7.037–7.822 | 17.30–20.46 SFA 21.95–65.60 MUFA 15.01–60.75 PUFA | (C4:0) 1.28–4.01 (C8:0) 0.47 (C12:0) 0.66 (C14:0) 0.61–1.26 (C16:0) 12.02–17.15 (C16:1w7) 0.52–1.13 (C17:0) 0.32 (C18:0) 0.74–1.26 (C18:1w9c) 14.78–24.60 (C18:2w6c) 4.63–9.36 (C18:3w3) 10.38–19.54 (C20:1w9) 0.90–1.30 (C20:5w3) 2.46 (C22:1w9) 23.34–49.35 (C22:6w3) 34.78 | Erlenmeyer flasks 170 μEm−2 s−1. | 12 h light:12 h dark. 25 °C | [191] | ||
| B. terribilis (IBL-C115) | 0.10–0.18 | 15.33–16.79 | 9.91–10.60 SFA 71.66–83.56 MUFA 6.53–17.85 PUFA | (C4:0) 0.49–2.98 (C14:0) 0.34–0.64 (C16:0) 5.71–8.25 (C16:1w7) 0.23 (C17:0) 0.27 (C17:1w5) 0.70–2.35 (C18:0) 0.87–1.28 (C18:1w9c) 69.31–81.77 (C18:2w6c)1.92–5.84 (C18:3w3) 3.49–9.66 (C20:1w9) 0.66–0.69 (C20:4w6) 0.28–0.59 (C20:5w3) 0.84–1.77 (C22:1w9) 0.41 (C22:6w3) 2.26 | Erlenmeyer flasks 170 μEm−2 s−1. | 12 h light:12 h dark. 25 °C | [191] | ||
| Chlorella sp. 800 | 0.020 | 25.20 | 2.46 SFA 2.64 MUFA 2.64 PUFA | (C16:0) 1.86 (C16:1) 0.055 (C18:0) 0.60 (C18:1ω9) 2.59 (C18:2ω6) 1.97 (C18:3ω3) 0.67 | 2 L glass cylinder photobioreactors 200 μmol E m−2 s−1. | 25 °C air enriched with 2% (v/v) CO2 | Modified Tamiya medium (g L−1) (KNO3, 2.5; KH2PO4, 0.625; MgSO4·7H2O, 1.25; FeSO4·7H2O, 0.0045; Na2EDTA, 0.0186; and trace elements solution, 0.5 mL L−1). The trace elements solution (g L−1): H3BO3, 2.86; MnCl2·4H2O, 1.81; ZnSO4·7H2O, 0.222; NH4VO3, 0.023; and MoO3, 0.018. | Chloroform/methanol (2:1) and ball mill | [192] |
| Chlorella saccharophila 477 | 0.008 | 27.6 | 1.91 SFA 1.905 MUFA 4.23 PUFA | (C16:0) 1.56 (C16:1) 0.045 (C18:0) 0.35 (C18:1ω9) 1.86 (C18:2ω6) 4.04 (C18:3ω3) 0.19 | 2 L glass cylinder photobioreactors containing 200 μmol E m−2 s−1. | 25 °C air enriched with 2% (v/v) CO2 | [192] | ||
| Chlorella minutissima 494 | 0.016 | NR | 1.216 SFA 0.217 MUFA 2.3 PUFA | (C16:0) 1.18 (C16:1) 0.037 (C18:0) 0.036 (C18:1ω9) 0.18 (C18:2ω6) 2.05 (C18:3ω3) 0.25 | 2 L glass cylinder photobioreactors containing 200 μmol photons m−2 s−1. | 25 °C air enriched with 2% (v/v) CO2 | [192] | ||
| Chlorella sp. 313 | 0.018 | NR | 1.12 SFA 0.192 MUFA 1.6 PUFA | (C16:0) 1.08 (C16:1) 0.032 (C18:0) 0.04 (C18:1ω9) 0.16 (C18:2ω6) 1.41 (C18:3ω3) 0.19 | 2 L glass cylinder photobioreactors containing 200 μmol photons m−2 s−1. | 25 °C air enriched with 2% (v/v) CO2 | [192] | ||
| Chlorella minutissima 444 | 0.016 | NR | 1.04 SFA 0.18 MUFA 2.18 PUFA | (C16:0) 1.00 (C16:1) 0.04 (C18:0) 0.04 (C18:1ω9) 0.14 (C18:2ω6)1.95 (C18:3ω3) 0.23 | 2 L glass cylinder photobioreactors containing 200 μmol E m−2s−1. | 25 °C air enriched with 2% (v/v) CO2 | [192] | ||
| Schizochytrium sp. | 0.537 | 49.53 | 44.81 PUFA | (C22:6) 100 | 1.500-L fermenter. | Control aeration 24 and 36 m3 h−1 | 40 g L−1 glucose and 0.4 g L-1 yeast extract dissolved in artificial sea water. | High-pressure homogenizer and n-hexane/ethanol (2:1). | [193] |
| Schizochytrium sp. HX-308 | 0.479 | 69.98 | 32.5 SFA 59.58 PUFA | (C14:0) 9.35 (C16:0) 23.15 (C22:5) 17.94 (C22:6) 41.64 | 10 L fermenter. | 25 °C fed-batch cultivation. | Artificial sea water. (g/L): Na2SO4 10; (NH4)2SO4 0.8; KH2PO4 4; KCl 0.2; MgSO4 2; Monosodium glutamate 20 (g L−1); CaCl2 0.1 (g L−1) and the trance elements (g/L): Na2EDTA 6, FeSO4 0.29, MnCl2. 4H2O 0.86, ZnSO4 0.8, CoCl2.6H2O 0.01, Na2MoO4.2H2O 0.01, NiSO4.6H2O 0.06 and CuSO4 5H2O 0.6. | [193,194] | |
| Phaeodactylum tricornutum | 0.043 | NR | 4.9 SFA 2 MUFA 5.6 PUFA | (C14:0) 0.7 (C16:0) 2.7 (C16:1) 2 (C16:3) 0.6 (C18:0) 1.5 (C20:5 n-3) 5 | Flat panel airlift 200–1000 µmol E m−2 s−1). | 1.25% (v/v) CO2 16 h light per day 20 °C during the light period and 12–14 °C during the dark period. | Modified Mann and Myers medium (g L−1) 10 NaCl, 1.8 KCl, 2.4 MgSO4·7 H2O, and different N source urea, KNO3, NH4Cl, | NR | [195] |
| Schizochytrium sp. | 1.25 | 68.6 | 20.69-34.32 SFA 0.43-1.88 MUFA 53.23-64.54 PUFA | (C14:0) 5.24–9.96 (C14:1) 0.43–1.88 (C16:0) 14.94–22.85 (C18:0) 0.51–1.51 (C22:5, n-6) 15.2–20.96 (C22:6, n-3) 38.03–43.58 | 50 L porous membrane-impeller bioreactor. | 30 °C | Glucose (70 g L−1) and yeast extract. (8 g L−1) in artificial seawater. | Petroleum ether/diethyl ether (9:1) and methanol. | [196] |
| Chlorella vulgaris | 0.005 | 30 | 38.8 SFA 45.4 MUFA 13.3 PUFA | (C16:0) 32.3 (C16:1) 17.7 (C18:0) 6.5 (C18:1) 27.7 (C18:2) 10.2 (C18:3) 3.1 | 5 L cylindrical algal photobioreactor 8-28 μmol photons m−2 s−1. | air sparger 30 °C light/dark period was 16/8 h | Waste industrial cane molasses. Gloucosan corn industry. | Bligh and Dyer method | [197] |
| Dunaliella salina | 0.068 | 19.04 | 37.8 SFA 41.2 MUFA 20.9 PUFA | (C16:0) 15.37 (C16:1) 3.80 (C18:0) 7.15 (C18:1) 37.48 (C18:2)15.40 (C18:3) 2.31 | 10 L working bubble column photobioreactors 150 klux light intensity. | 25 °C | Modified Guillard f/2 medium (without silica). Supplemented with NaNO3 (225 mg L−1) and sodium acetate (4 g L−1). | Ultrasound bath | [198] |
| Nannochloropsis gaditana | 0.0036–0.0078 | 14.72–11.66 | 31.17 SFA 30.27 MUFA 20.79 PUFA | (C18:3 ω) 3.72 (C18:2) 17.07 (C18:1) 23.24 (C18:0) 8.99 (C16:1) 7.03 (C16:0) 22.18 | 4 L tank photo bioreactors 150 klux light intensity for 7 days. | 24 °C sterile air at 1.4 L.min−1 | Modified Guillard f/2 medium (without silica). 6 g L−1 of acetate and 225 mg L−1 of nitrate. | Modified Folch method and ultrasound, with a mixture of chloroform: methanol (3:1). | [199] |
| Microalgae Strain | Main Fatty Acids | Main Lubricant Properties * | Possible Applications | |||
|---|---|---|---|---|---|---|
| Lubricity | Viscosity | Pour Point | Oxidation Stability | |||
| B. braunii (UTEX LB 572) | MUFA (C:18) | ✓✓✓ | ✓✓ | ✓ | ✓✓✓ | Engine oil, gear oil, grease, metalworking fluid, insulating oil, refrigeration compressor oil, air mist lubricant, rock drill oil, vacuum pump oil, etc. |
| B. braunii (IBL-C117) | MUFA (C:22) | ✓✓✓ | ✓✓✓ | ✓ | ✓✓✓ | Engine oil, gear oil, grease, metalworking fluid, insulating oil, refrigeration compressor oil, air mist lubricant, rock drill oil, vacuum pump oil, etc. |
| B. terribilis (IBL-C115) | MUFA (C:18) | ✓✓✓ | ✓✓ | ✓ | ✓✓✓ | Engine oil, gear oil, grease, metalworking fluid, insulating oil, refrigeration compressor oil, air mist lubricant, rock drill oil, vacuum pump oil, etc. |
| Chlorella sp. 800 | MUFA PUFA (C:18) | ✓✓ | ✓✓ | ✓✓ | ✓✓ | Concrete demolding oil, chainsaw oil, gear oil, grease, metalworking fluid, air mist lubricant, rock drill oil, etc. |
| Chlorella saccharophila 477 | PUFA (C:18) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Chlorella minutissima 494 | PUFA (C:18) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Chlorella sp. 313 | PUFA (C:18) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Chlorella minutissima 444 | PUFA (C:18) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Schizochytrium sp. | PUFA (C:22) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Schizochytrium sp. HX-308 | PUFA (C:22) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Phaeodactylum tricornutum | PUFA (C:20) | ✓ | ✓ | ✓✓✓ | ✓ | Concrete demolding oil, hydraulic fluid, chainsaw oil, air mist lubricant, etc. |
| Chlorella vulgaris | MUFA (C:16) | ✓✓✓ | ✓✓ | ✓ | ✓✓✓ | Engine oil, gear oil, grease, metalworking fluid, insulating oil, refrigeration compressor oil, air mist lubricant, rock drill oil, vacuum pump oil, etc. |
| Dunaliella salina | MUFA (C:18) | ✓✓✓ | ✓✓ | ✓ | ✓✓✓ | Engine oil, gear oil, grease, metalworking fluid, insulating oil, refrigeration compressor oil, air mist lubricant, rock drill oil, vacuum pump oil, etc. |
| Nannochloropsis gaditana | SFA (C:18) | ✓✓✓ | ✓✓ | ✓ | ✓✓✓ | Engine oil, gear oil, grease, metalworking fluid, insulating oil, refrigeration compressor oil, air mist lubricant, rock drill oil, vacuum pump oil, etc. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farfan-Cabrera, L.I.; Franco-Morgado, M.; González-Sánchez, A.; Pérez-González, J.; Marín-Santibáñez, B.M. Microalgae Biomass as a New Potential Source of Sustainable Green Lubricants. Molecules 2022, 27, 1205. https://doi.org/10.3390/molecules27041205
Farfan-Cabrera LI, Franco-Morgado M, González-Sánchez A, Pérez-González J, Marín-Santibáñez BM. Microalgae Biomass as a New Potential Source of Sustainable Green Lubricants. Molecules. 2022; 27(4):1205. https://doi.org/10.3390/molecules27041205
Chicago/Turabian StyleFarfan-Cabrera, Leonardo I., Mariana Franco-Morgado, Armando González-Sánchez, José Pérez-González, and Benjamín M. Marín-Santibáñez. 2022. "Microalgae Biomass as a New Potential Source of Sustainable Green Lubricants" Molecules 27, no. 4: 1205. https://doi.org/10.3390/molecules27041205
APA StyleFarfan-Cabrera, L. I., Franco-Morgado, M., González-Sánchez, A., Pérez-González, J., & Marín-Santibáñez, B. M. (2022). Microalgae Biomass as a New Potential Source of Sustainable Green Lubricants. Molecules, 27(4), 1205. https://doi.org/10.3390/molecules27041205