Cross-Correlated Motions in Azidolysozyme
Abstract
1. Introduction
2. Methods
2.1. Molecular Dynamics Simulation
2.2. Dynamical Cross Correlation Maps
2.3. Infrared Spectroscopy
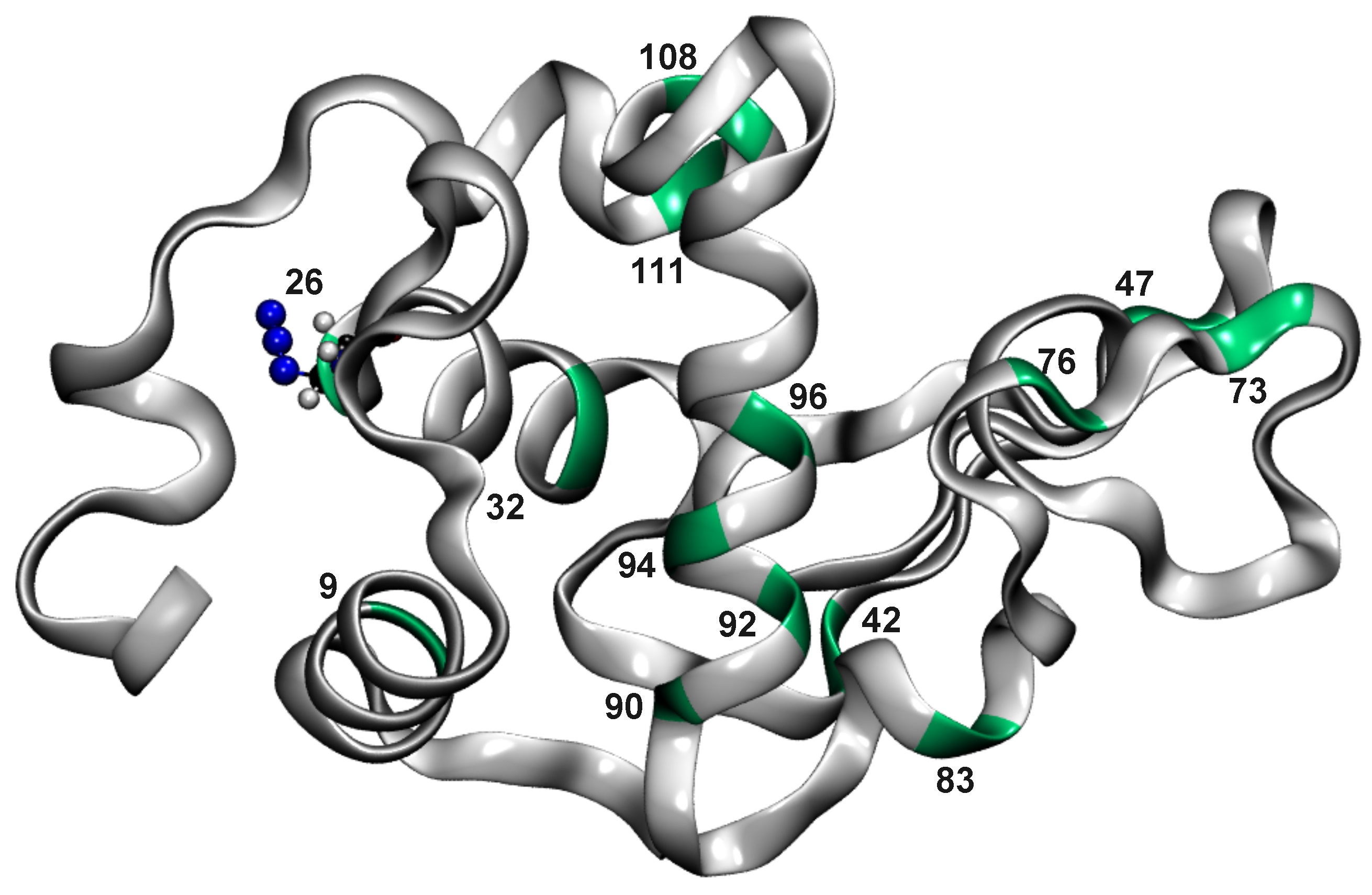
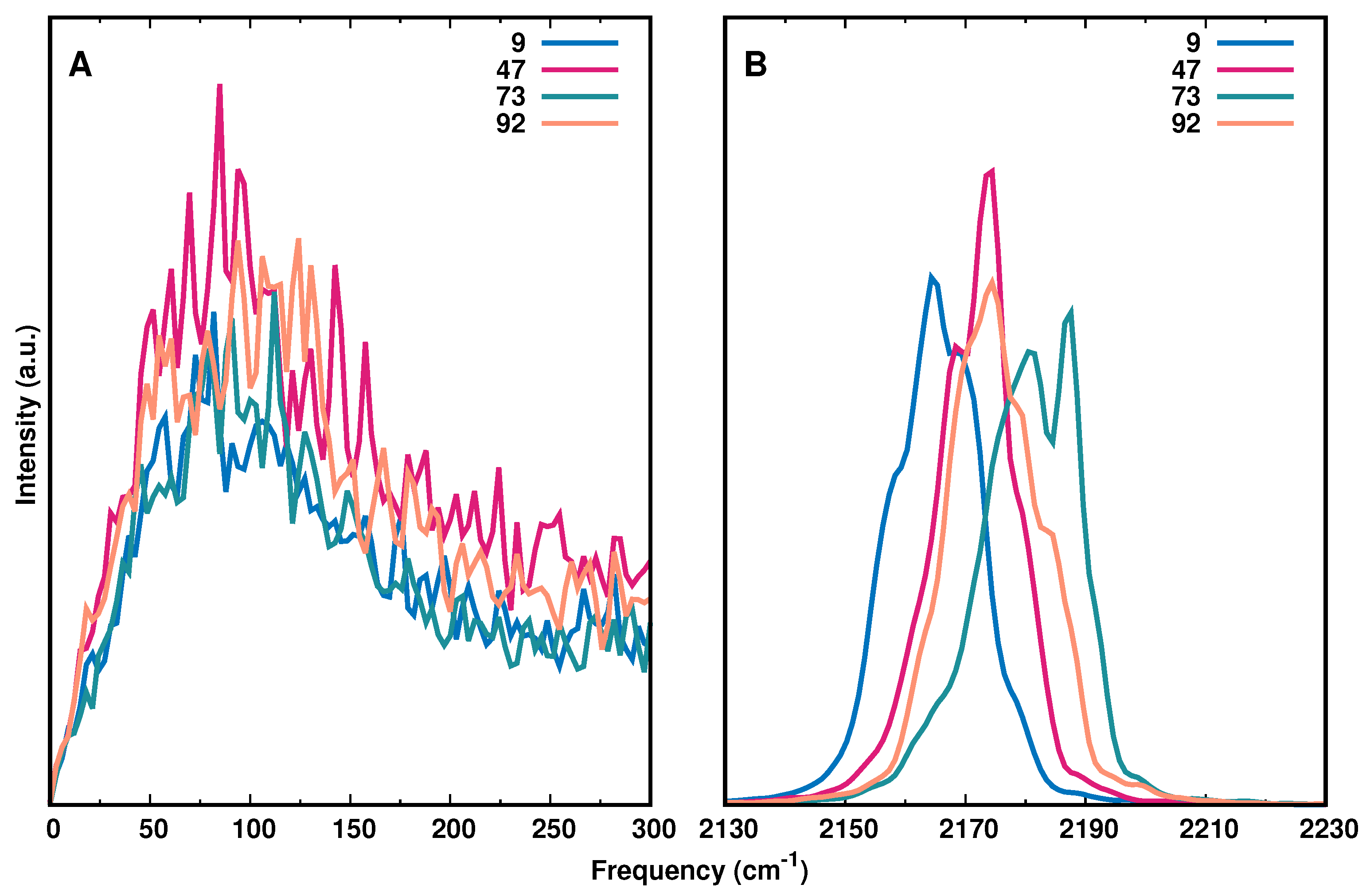
3. Results
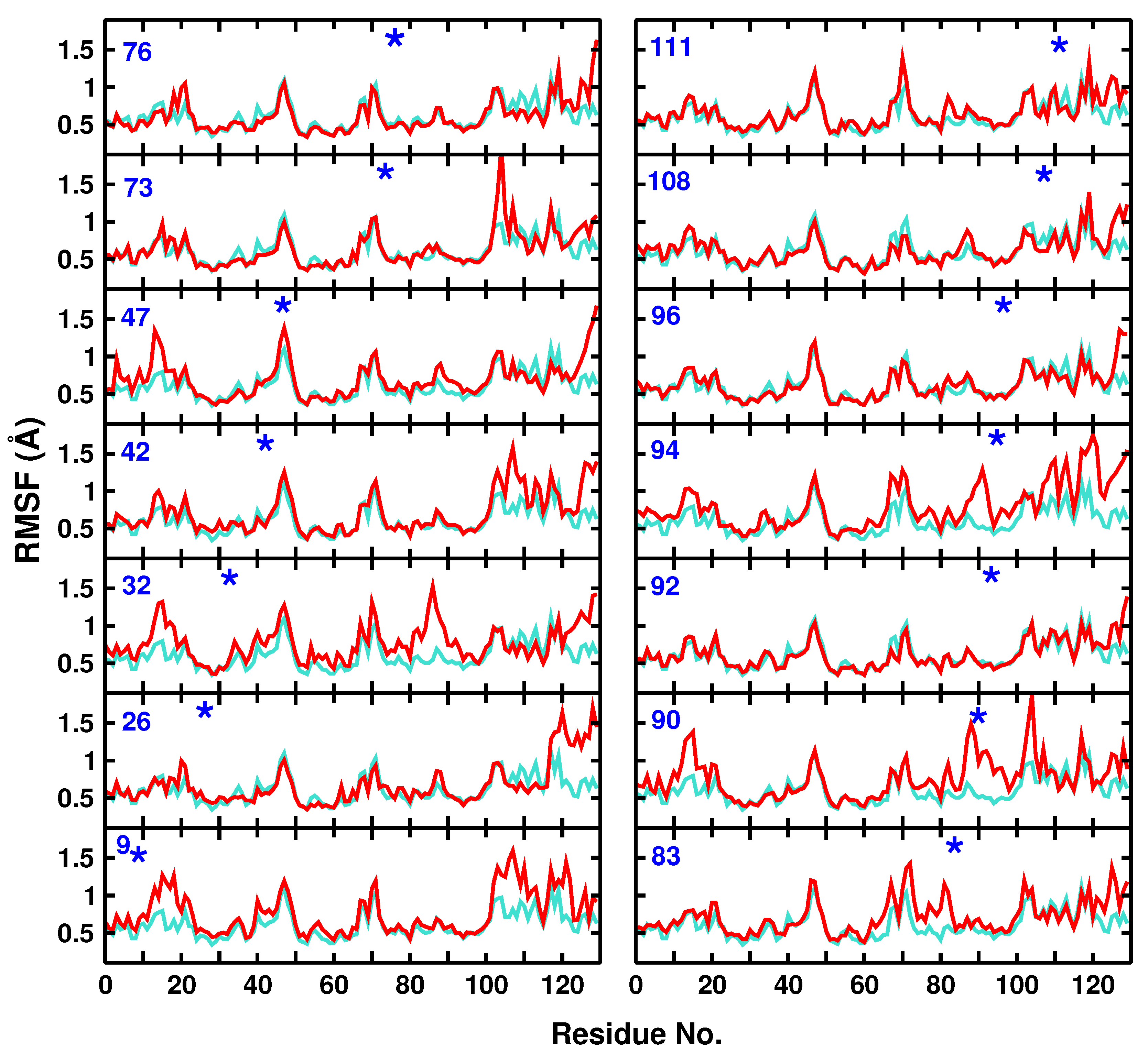
3.1. Global Dynamics (RMSF and DCCM)
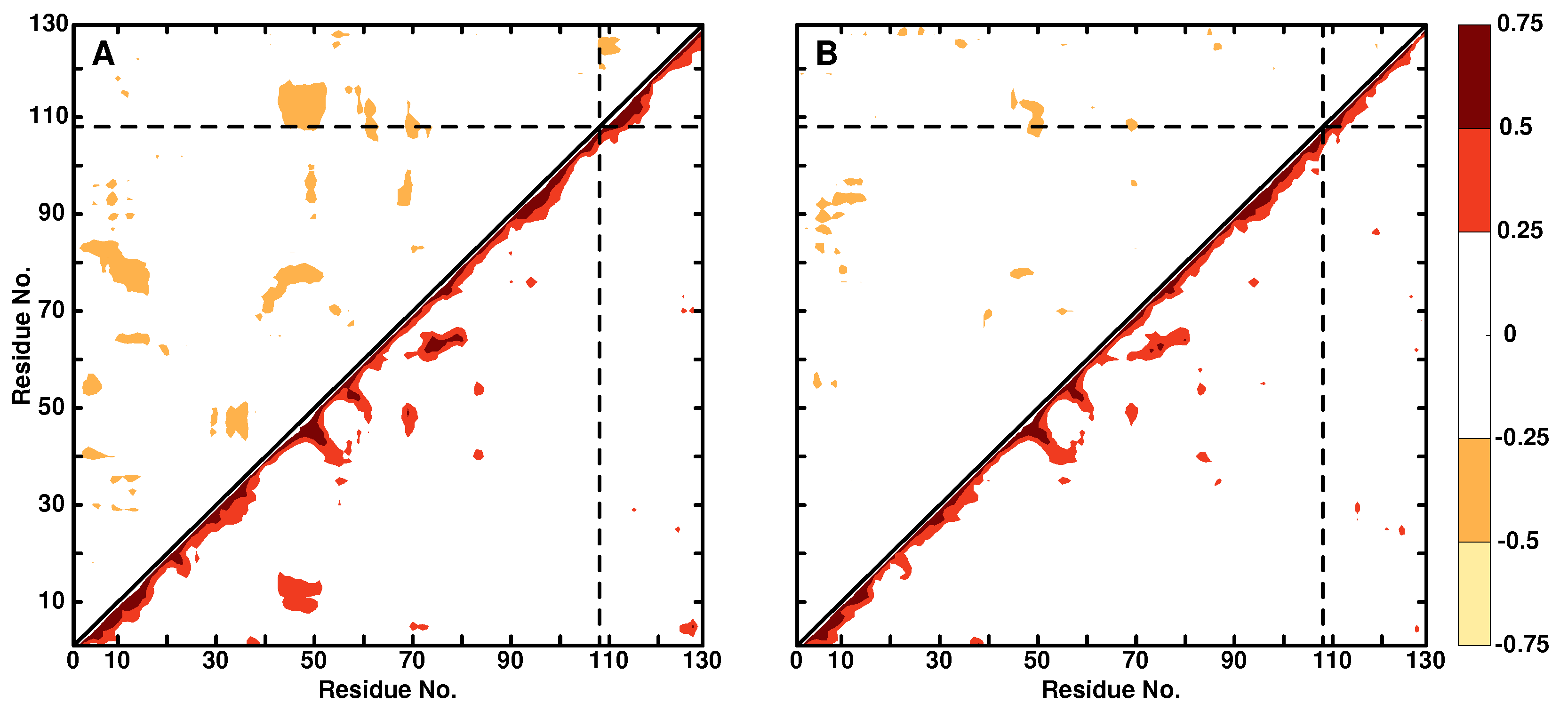
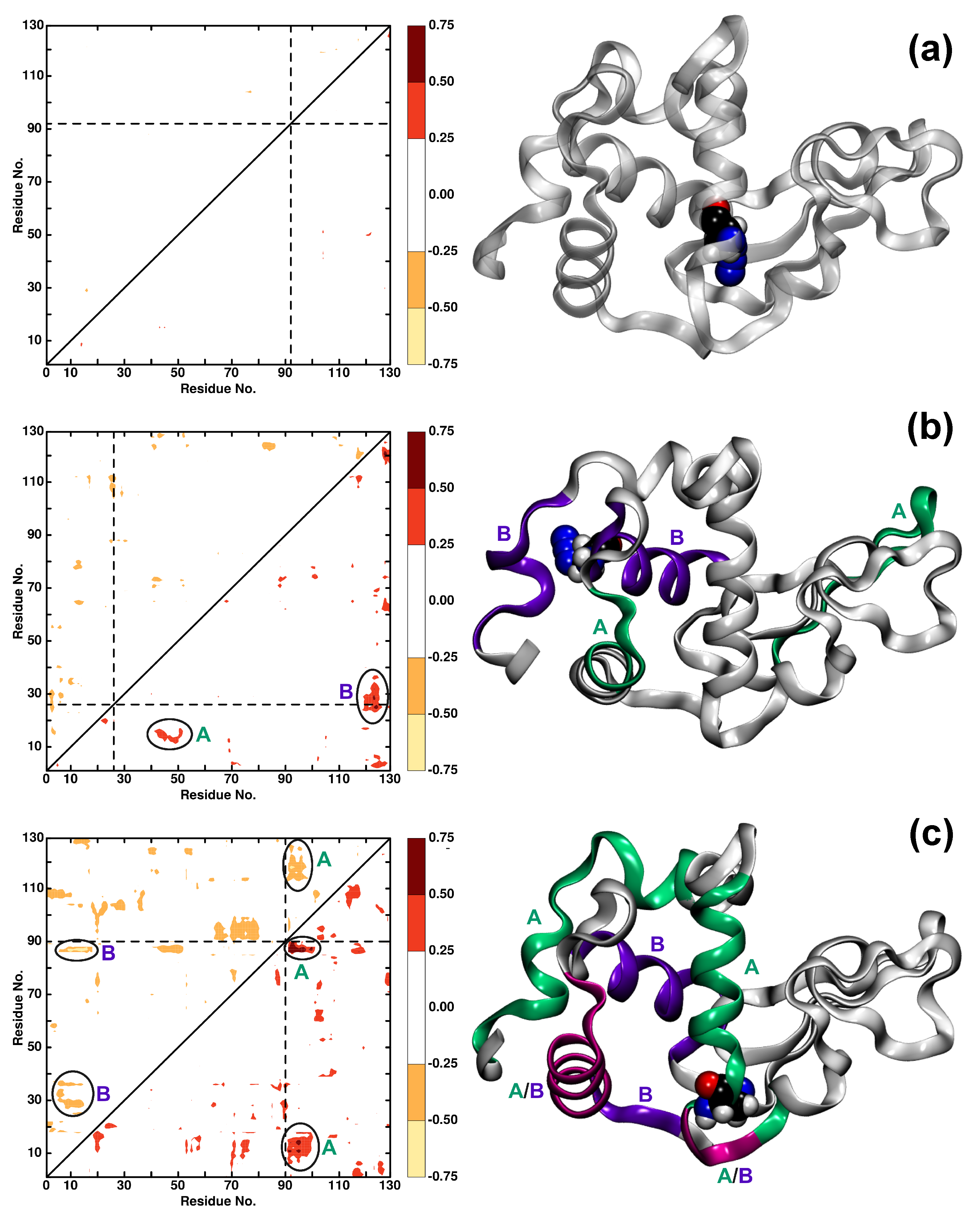
3.2. Local Dynamics (DCCM)
3.3. Spectroscopy
3.4. The Structural Dynamics of Wild Type and Azide-Labelled Lysozyme on Longer Time Scales
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Plitzko, J.M.; Schuler, B.; Selenko, P. Structural Biology outside the box—Inside the cell. Curr. Opt. Struct. Biol. 2017, 46, 110–121. [Google Scholar] [CrossRef]
- Guo, J.; Zhou, H.X. Protein Allostery and Conformational Dynamics. Chem. Rev. 2016, 116, 6503–6515. [Google Scholar] [CrossRef]
- Lu, S.; He, X.; Ni, D.; Zhang, J. Allosteric Modulator Discovery: From Serendipity to Structure-Based Design. J. Med. Chem. 2019, 62, 6405–6421. [Google Scholar] [CrossRef]
- Getahun, Z.; Huang, C.; Wang, T.; De Leon, B.; DeGrado, W.; Gai, F. Using nitrile-derivatized amino acids as infrared probes of local environment. J. Am. Chem. Soc. 2003, 125, 405–411. [Google Scholar] [CrossRef]
- Kozinski, M.; Garrett-Roe, S.; Hamm, P. 2D-IR spectroscopy of the sulfhydryl band of cysteines in the hydrophobic core of proteins. J. Phys. Chem. B 2008, 112, 7645–7650. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, J.; Thielges, M.C.; Seo, Y.J.; Dawson, P.E.; Romesberg, F.E. Cyano Groups as Probes of Protein Microenvironments and Dynamics. Angew. Chem. Int. Ed. 2011, 50, 8333–8337. [Google Scholar] [CrossRef]
- Bagchi, S.; Boxer, S.G.; Fayer, M.D. Ribonuclease S Dynamics Measured Using a Nitrile Label with 2D IR Vibrational Echo Spectroscopy. J. Phys. Chem. B 2012, 116, 4034–4042. [Google Scholar] [CrossRef] [PubMed]
- van Wilderen, L.J.G.W.; Kern-Michler, D.; Mueller-Werkmeister, H.M.; Bredenbeck, J. Vibrational dynamics and solvatochromism of the label SCN in various solvents and hemoglobin by time dependent IR and 2D-IR spectroscopy. Phys. Chem. Chem. Phys. 2014, 16, 19643–19653. [Google Scholar] [CrossRef]
- Horness, R.E.; Basom, E.J.; Thielges, M.C. Site-selective characterization of Src homology 3 domain molecular recognition with cyanophenylalanine infrared probes. Anal. Chem. 2015, 7, 7234–7241. [Google Scholar] [CrossRef]
- Lee, G.; Kossowska, D.; Lim, J.; Kim, S.; Han, H.; Kwak, K.; Cho, M. Cyanamide as an Infrared Reporter: Comparison of Vibrational Properties between Nitriles Bonded to N and C Atoms. J. Phys. Chem. B 2018, 122, 4035–4044. [Google Scholar] [CrossRef] [PubMed]
- King, J.T.; Kubarych, K.J. Site-specific coupling of hydration water and protein flexibility studied in solution with ultrafast 2D-IR spectroscopy. J. Am. Chem. Soc. 2012, 134, 18705–18712. [Google Scholar] [CrossRef] [PubMed]
- King, J.T.; Arthur, E.J.; Brooks, C.L., III; Kubarych, K.J. Site-specific hydration dynamics of globular proteins and the role of constrained water in solvent exchange with amphiphilic cosolvents. J. Phys. Chem. B 2012, 116, 5604–5611. [Google Scholar] [CrossRef]
- King, J.T.; Arthur, E.J.; Brooks, C.L., III; Kubarych, K.J. Crowding Induced Collective Hydration of Biological Macromolecules over Extended Distances. J. Am. Chem. Soc. 2014, 136, 188–194. [Google Scholar] [CrossRef] [PubMed]
- El Hage, K.; Hedin, F.; Gupta, P.K.; Meuwly, M.; Karplus, M. Valid molecular dynamics simulations of human hemoglobin require a surprisingly large box size. eLife 2018, 7, e35560. [Google Scholar] [CrossRef] [PubMed]
- Pezzella, M.; El Hage, K.; Niesen, M.J.; Shin, S.; Willard, A.P.; Meuwly, M.; Karplus, M. Water dynamics around proteins: T-and R-States of hemoglobin and melittin. J. Phys. Chem. B 2020, 124, 6540–6554. [Google Scholar] [CrossRef] [PubMed]
- Salehi, S.M.; Meuwly, M. Site-selective dynamics of azidolysozyme. J. Chem. Phys. 2021, 154, 165101. [Google Scholar] [CrossRef] [PubMed]
- Salehi, S.M.; Koner, D.; Meuwly, M. Vibrational Spectroscopy of in the Gas and Condensed Phase. J. Phys. Chem. B 2019, 123, 3282–3290. [Google Scholar] [CrossRef] [PubMed]
- Kiick, K.; Saxon, E.; Tirrell, D.; Bertozzi, C. Incorporation of azides into recombinant proteins for chemoselective modification by the Staudinger ligation. Proc. Natl. Acad. Sci. USA 2002, 99, 19–24. [Google Scholar] [CrossRef]
- MacKerell, A.D.; Bashford, D.; Bellott, M.; Dunbrack, R.L.; Evanseck, J.D.; Field, M.J.; Fischer, S.; Gao, J.; Guo, H.; Ha, S.; et al. All-atom Empirical Potential for Molecular Modeling and Dynamics Studies of Proteins. J. Phys. Chem. B 1998, 102, 3586–3616. [Google Scholar] [CrossRef]
- Brooks, B.R.; Brooks III, C.L.; Mackerell, A.D., Jr.; Nilsson, L.; Petrella, R.J.; Roux, B.; Won, Y.; Archontis, G.; Bartels, C.; Boresch, S.; et al. CHARMM: The biomolecular simulation program. J. Comp. Chem. 2009, 30, 1545–1614. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of Simple Potential Functions for Simulating Liquid Water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Chiba-Kamoshida, K.; Matsui, T.; Ostermann, A.; Chatake, T.; Ohhara, T.; Tanaka, I.; Yutani, K.; Niimura, N. X-ray Crystal Structure of Wild Type Human Lysozyme in D2O. Available online: https://www.rcsb.org/structure/3fe0 (accessed on 8 December 2009).
- Gunsteren, W.V.; Berendsen, H. Algorithms for Macromolecular Dynamics and Constraint Dynamics. Mol. Phys. 1997, 34, 1311–1327. [Google Scholar] [CrossRef]
- Steinbach, P.J.; Brooks, B.R. New Spherical-Cutoff Methods for Long-Range Forces in Macromolecular Simulation. J. Comput. Chem. 1994, 15, 667–683. [Google Scholar] [CrossRef]
- Eastman, P.; Swails, J.; Chodera, J.D.; McGibbon, R.T.; Zhao, Y.; Beauchamp, K.A.; Wang, L.P.; Simmonett, A.C.; Harrigan, M.P.; Stern, C.D.; et al. OpenMM 7: Rapid development of high performance algorithms for molecular dynamics. PLoS Comp. Biol. 2017, 13, e1005659. [Google Scholar] [CrossRef]
- Darden, T.; York, D.; Pedersen, L. Particle Mesh Ewald: An Nlog(N) Method for Ewald Sums in Large Systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef]
- Ichiye, T.; Karplus, M. Collective Motions in Proteins - A Covariance Analysis of Atomic Fluctuation in Molecular-Dynamics and Normal Mode Simulations. Protein Struct. Funct. Genet. 1991, 11, 205–217. [Google Scholar] [CrossRef]
- Arnold, G.; Ornstein, R. Molecular dynamics study of time-correlated protein domain motions and molecular flexibility: Cytochrome P450BM-3. Biophys. J. 1997, 73, 1147–1159. [Google Scholar] [CrossRef]
- Grant, B.J.; Rodrigues, A.P.C.; ElSawy, K.M.; McCammon, J.A.; Caves, L.S.D. Bio3d: An R package for the comparative analysis of protein structures. Bioinformatics 2006, 22, 2695–2696. [Google Scholar] [CrossRef]
- Schmitz, M.; Tavan, P. Vibrational spectra from atomic fluctuations in dynamics simulations. II. Solvent-induced frequency fluctuations at femtosecond time resolution. J. Chem. Phys. 2004, 121, 12247–12258. [Google Scholar] [CrossRef]
- Salehi, S.M.; Koner, D.; Meuwly, M. Dynamics and Infrared Spectrocopy of Monomeric and Dimeric Wild Type and Mutant Insulin. J. Phys. Chem. B, 2020; in print. [Google Scholar]
- Pagano, P.; Guo, Q.; Kohen, A.; Cheatum, C.M. Oscillatory enzyme dynamics revealed by two-dimensional infrared spectroscopy. J. Phys. Chem. Lett. 2016, 7, 2507–2511. [Google Scholar] [CrossRef]
- Ernst, M.; Wolf, S.; Stock, G. Identification and validation of reaction coordinates describing protein functional motion: Hierarchical dynamics of T4 lysozyme. J. Chem. Theory Comp. 2017, 13, 5076–5088. [Google Scholar] [CrossRef] [PubMed]
- Dixon, M.; Nicholson, H.; Shewchuk, L.; Baase, W.; Matthews, B. Structure of a hinge-bending bacteriophage T4 lysozyme mutant, Ile3→ Pro. J. Mol. Biol. 1992, 227, 917–933. [Google Scholar] [CrossRef]
- Zanobini, C.; Bozovic, O.; Jankovic, B.; Koziol, K.L.; Johnson, P.J.M.; Hamm, P.; Gulzar, A.; Wolf, S.; Stock, G. Azidohomoalanine: A Minimally Invasive, Versatile, and Sensitive Infrared Label in Proteins To Study Ligand Binding. J. Phys. Chem. B 2018, 122, 10118–10125. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salehi, S.M.; Meuwly, M. Cross-Correlated Motions in Azidolysozyme. Molecules 2022, 27, 839. https://doi.org/10.3390/molecules27030839
Salehi SM, Meuwly M. Cross-Correlated Motions in Azidolysozyme. Molecules. 2022; 27(3):839. https://doi.org/10.3390/molecules27030839
Chicago/Turabian StyleSalehi, Seyedeh Maryam, and Markus Meuwly. 2022. "Cross-Correlated Motions in Azidolysozyme" Molecules 27, no. 3: 839. https://doi.org/10.3390/molecules27030839
APA StyleSalehi, S. M., & Meuwly, M. (2022). Cross-Correlated Motions in Azidolysozyme. Molecules, 27(3), 839. https://doi.org/10.3390/molecules27030839





