An Improved Approach for Practical Synthesis of 5-Hydroxymethyl-2′-deoxycytidine (5hmdC) Phosphoramidite and Triphosphate
Abstract
1. Introduction
2. Results and Discussion
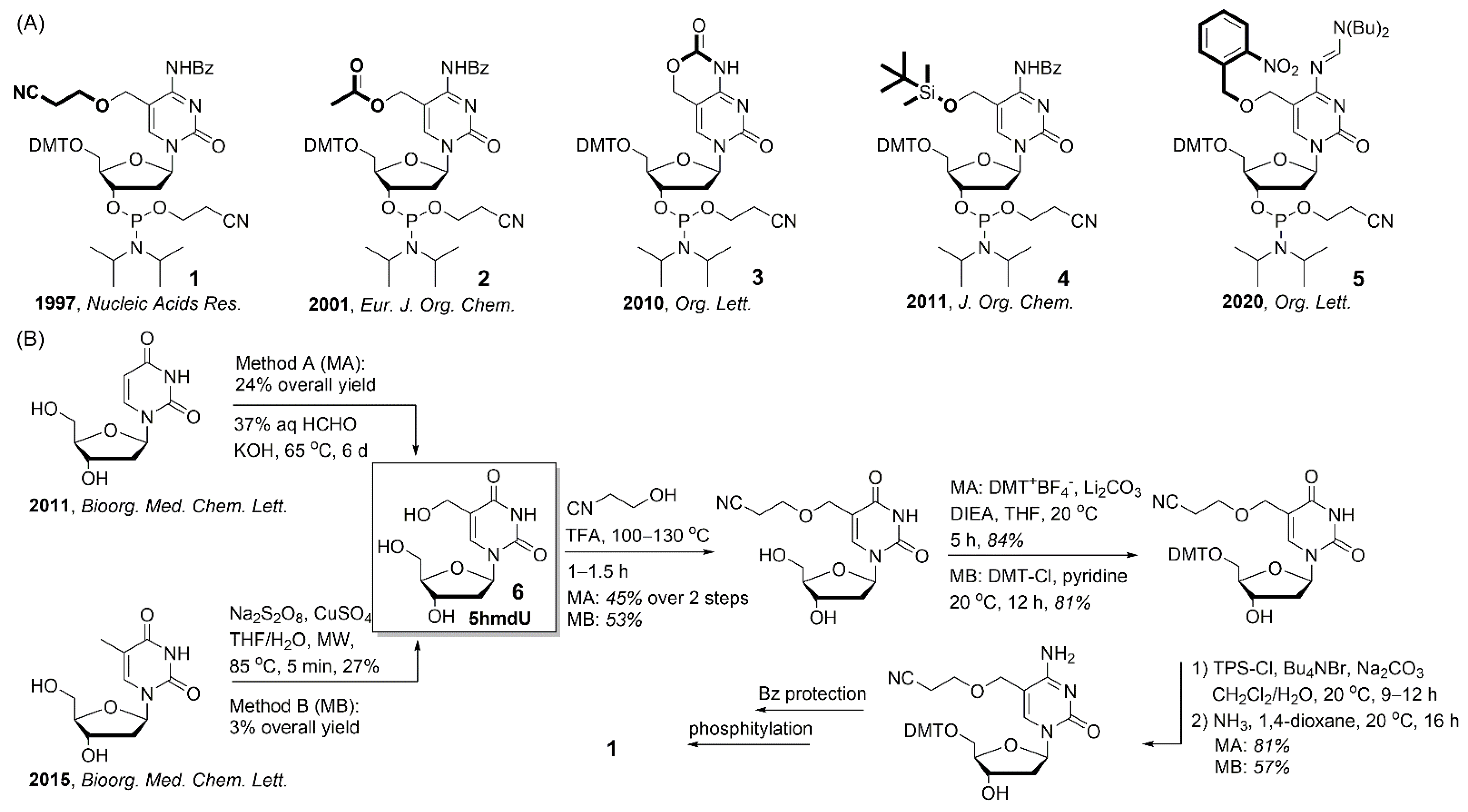
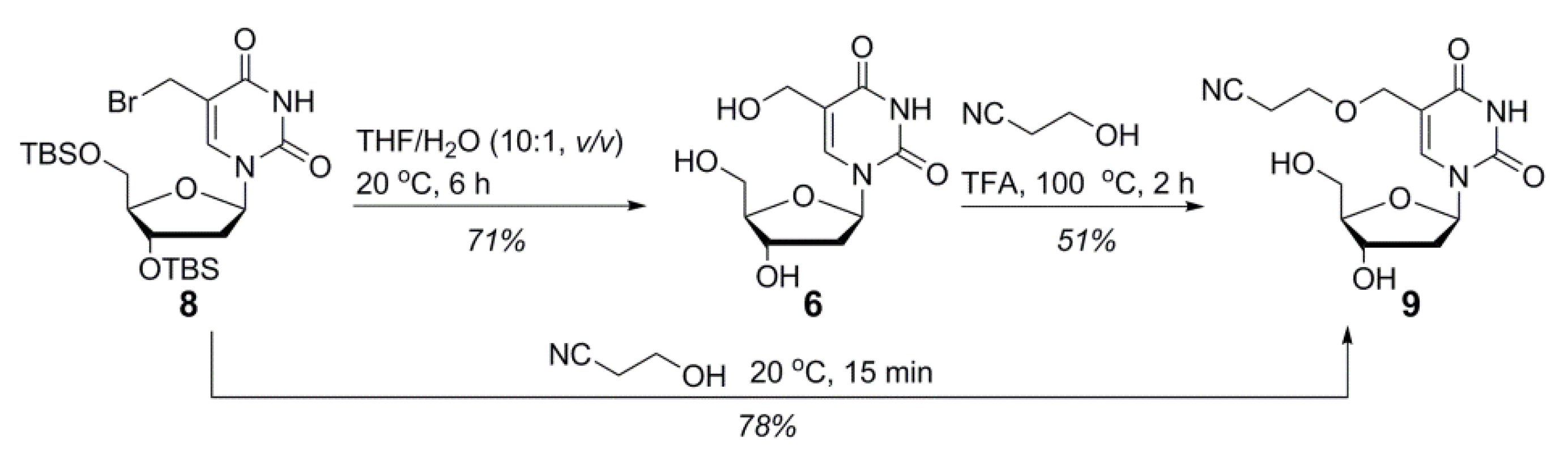
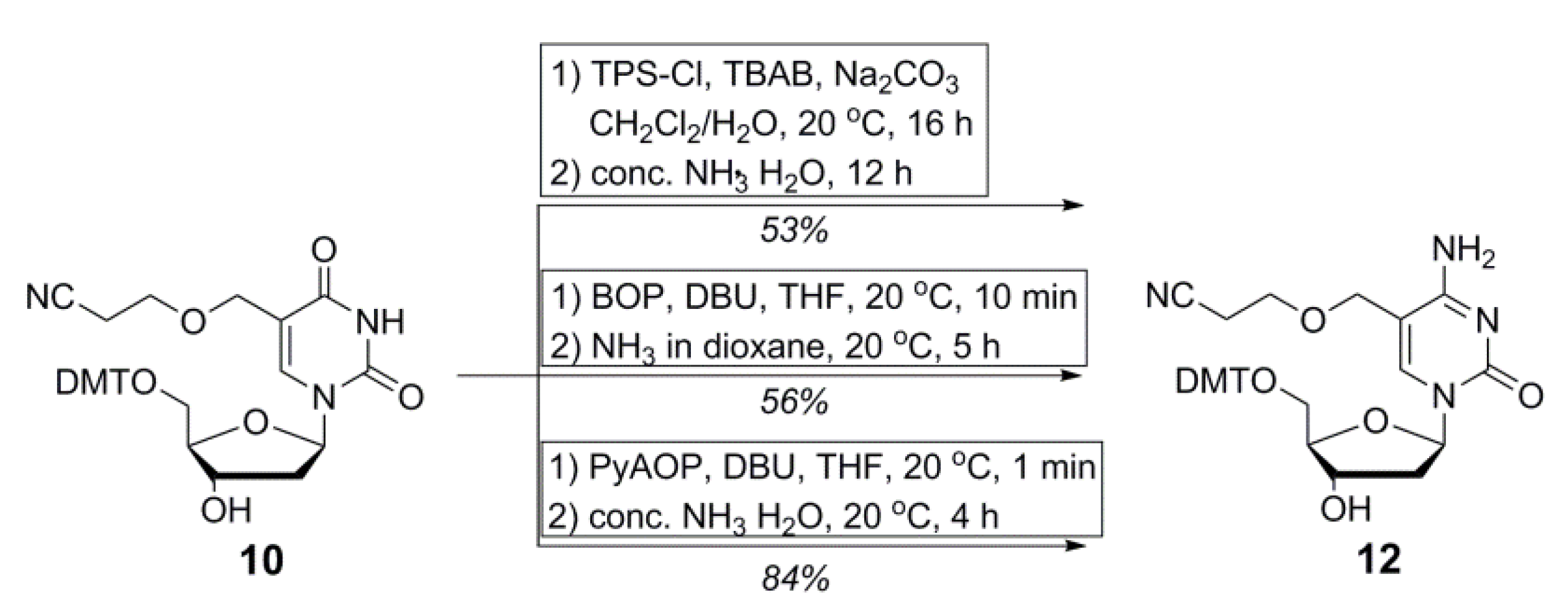
2.1. Synthesis of Cyanoethyl-Protected 5hmdC Phosphoramidite (1)
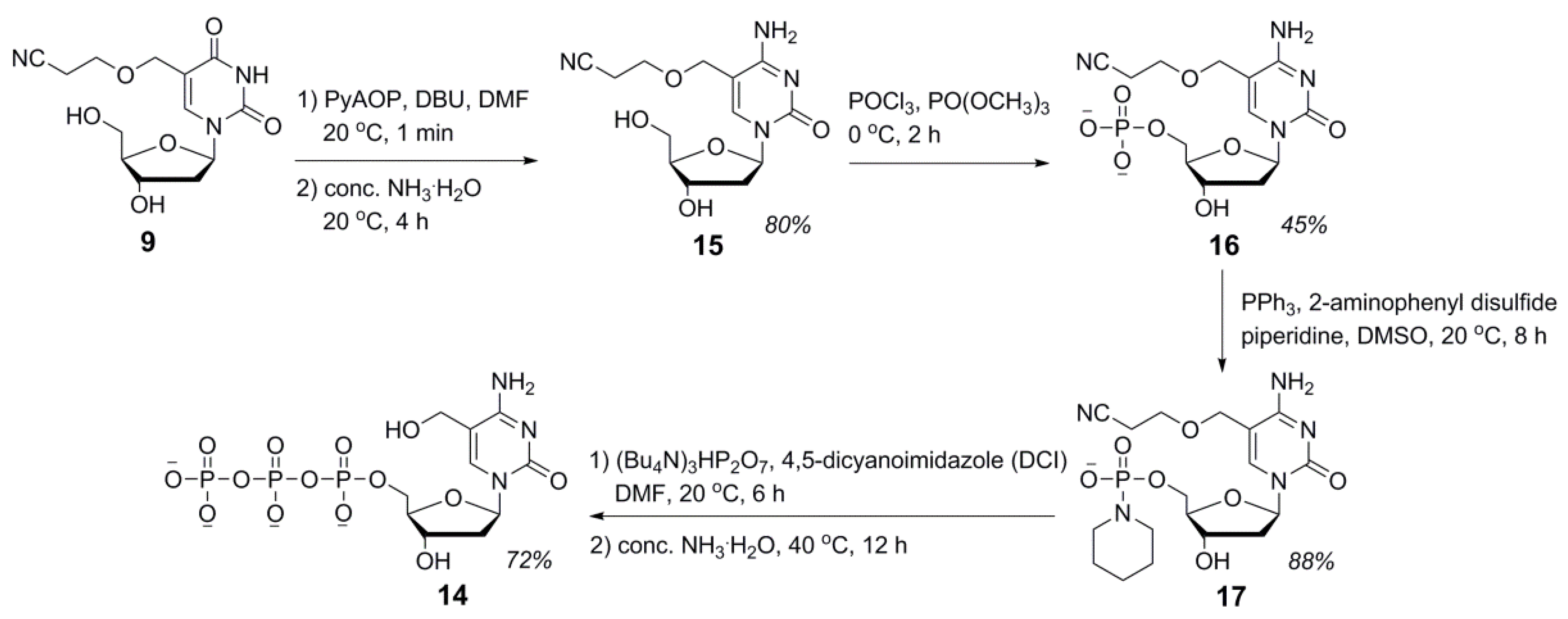
2.2. Synthesis of 5hmdC Triphosphate (14)
3. Materials and Methods
3.1. General Methods
3.2. Synthetic Procedures of 5hmdC Phosphoramidite (1) and Intermediates (9–13)
3.3. Synthetic Procedures and Characterization Data of 5hmdC Triphosphate (14) and Intermediates (15–20)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kriaucionis, S.; Heintz, N. The nuclear DNA base 5-hydroxymethylcytosine is present in purkinje neurons and the brain. Science 2009, 324, 929–930. [Google Scholar] [CrossRef] [PubMed]
- Tahiliani, M.; Koh, K.P.; Shen, Y.-H.; Pastor, W.A.; Bandukwala, H.; Brudno, Y.; Agarwal, S.; Iyer, L.M.; Liu, D.R.; Aravind, L.; et al. Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 2009, 324, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.J.; Zhang, Y. TET-mediated active DNA demethylation: Mechanism, function and beyond. Nat. Rev. Genet. 2017, 18, 517–534. [Google Scholar] [CrossRef]
- Munzel, M.; Globisch, D.; Carell, T. 5-Hydroxymethylcytosine, the sixth base of the genome. Angew. Chem. Int. Ed. 2011, 50, 6460–6468. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhang, Y. Reversing DNA methylation: Mechanisms, genomics, and biological functions. Cell 2014, 156, 45–68. [Google Scholar] [CrossRef] [PubMed]
- He, Y.-F.; Li, B.-Z.; Li, Z.; Liu, P.; Wang, T.; Tang, Q.-Y.; Ding, J.; Jia, Y.; Chen, Z.; Li, L.; et al. Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Science 2011, 333, 1303–1307. [Google Scholar] [CrossRef] [PubMed]
- Spada, F.; Schiffers, S.; Kirchner, A.; Zhang, Y.; Arista, G.; Kosmatchev, O.; Korytiakova, E.; Rahimoff, R.; Ebert, C.; Carell, T. Active turnover of genomic methylcytosine in pluripotent cells. Nat. Chem. Biol. 2020, 16, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Stroud, H.; Feng, S.; Kinney, S.M.; Pradhan, S.; Jacobsen, S.E. 5-Hydroxymethylcytosine is associated with enhancers and gene bodies in human embryonic stem cells. Genome Biol. 2011, 12, R54. [Google Scholar] [CrossRef] [PubMed]
- Munzel, M.; Globisch, D.; Bruckl, T.; Wagner, M.; Welzmiller, V.; Michalakis, S.; Muller, M.; Biel, M.; Carell, T. Quantification of the sixth DNA base hydroxymethylcytosine in the brain. Angew. Chem. Int. Ed. 2010, 49, 5375–5377. [Google Scholar] [CrossRef] [PubMed]
- Stricker, S.H.; Götz, M. DNA-methylation: Master or slave of neural fate decisions? Front. Neurosci. 2018, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Jiang, H.-P.; Peng, C.-Y.; Deng, Q.-Y.; Lan, M.-D.; Zeng, H.; Zheng, F.; Feng, Y.-Q.; Yuan, B.-F. DNA hydroxymethylation age of human blood determined by capillary hydrophilic-interaction liquid chromatography/mass spectrometry. Clin. Epigenet. 2015, 7, 72. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ito, S.; D’Alessio, A.C.; Taranova, O.V.; Hong, K.; Sowers, L.C.; Zhang, Y. Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nature 2010, 466, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Scourzic, L.; Mouly, E.; Bernard, O.A. TET proteins and the control of cytosine demethylation in cancer. Genome Med. 2015, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-L.; Shen, F.; Huang, W.; Qi, J.-H.; Wang, Y.; Feng, Y.-Q.; Liu, S.-M.; Yuan, B.-F. Quantification of 5-methylcytosine and 5-hydroxymethyl cytosine in genomic DNA from hepatocellular carcinoma tissues by capillary hydrophilic-interaction liquid chromatography/quadrupole TOF mass spectrometry. Clin. Chem. 2013, 59, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Olinski, R.; Starczak, M.; Gackowski, D. Enigmatic 5-hydroxymethyluracil: Oxidatively modified base, epigenetic mark or both? Mutat. Res. Rev. Mutat. Res. 2016, 767, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.-Q.; Ali, I.; Tang, J.; Yang, W.-C. New insights into 5hmC DNA modification: Generation, distribution and function. Front. Genet. 2017, 8, 100. [Google Scholar] [CrossRef]
- Pfaffeneder, T.; Sadeghian, K.; Hackner, B.; Steigenberger, B.; Schröder, A.S.; Steinbacher, J.; Kashiwazaki, G.; Höfner, G.; Wanner, K.T.; Ochsenfeld, C.; et al. Deamination, oxidation, and C–C bond cleavage reactivity of 5-hydroxymethylcytosine, 5-formylcytosine, and 5-carboxycytosine. J. Am. Chem. Soc. 2013, 135, 14593–14599. [Google Scholar]
- Schröder, A.S.; Raddaoui, N.; Carell, T. Synthesis of (R)-configured 2′-fluorinated mC, hmC, fC, and caC phosphoramidites and oligonucleotides. Org. Lett. 2016, 18, 4368–4371. [Google Scholar] [CrossRef]
- Steigenberger, B.; Schiesser, S.; Hackner, H.; Brandmayr, C.; Laube, S.K.; Steinbacher, J.; Pfaffeneder, T.; Carell, T. Synthesis of 5-hydroxymethyl-, 5-formyl-, and 5-carboxycytidine triphosphates and their incorporation into oligonucleotides by polymerase chain reaction. Org. Lett. 2013, 15, 366–369. [Google Scholar] [CrossRef]
- Vaníková, Z.; Janoušková, M.; Kambová, M.; Krásný, L.; Hocek, M. Switching transcription with bacterial RNA polymerase through photocaging, photorelease and phosphorylation reactions in the major groove of DNA. Chem. Sci. 2019, 10, 3937–3942. [Google Scholar] [CrossRef]
- Tardy-Planechaud, S.; Fujimoto, J.; Lin, S.S.; Sowers, L.C. Solid phase synthesis and restriction endonuclease cleavage of oligodeoxynucleotides containing 5-(hydroxymethyl)-cytosine. Nucleic Acids Res. 1997, 25, 553–558. [Google Scholar] [CrossRef] [PubMed]
- de Kort, M.; de Visser, P.C.; Kurzeck, J.; Meeuwenoord, N.J.; van der Marel, G.A.; Rüger, W.; van Boom, J.H. Chemical and enzymatic synthesis of DNA fragments containing 5-(β-D-glucopyranosyloxymethyl)-2′-deoxycytidine-a modified nucleoside in T4 phage DNA. Eur. J. Org. Chem. 2001, 11, 2075–2082. [Google Scholar] [CrossRef]
- Münzel, M.; Globisch, D.; Trindler, C.; Carell, T. Efficient synthesis of 5-hydroxymethylcytosine containing DNA. Org. Lett. 2010, 12, 5671–5673. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Song, C.-X.; Pan, T.; He, C. Syntheses of two 5-hydroxymethyl-2’-deoxycytidine phosphoramidites with TBDMS as the 5-hydroxymethyl protecting group and their incorporation into DNA. J. Org. Chem. 2011, 76, 4182–4188. [Google Scholar] [CrossRef] [PubMed]
- Chakrapani, A.; Hausnerová, V.V.; Ruiz-Larrabeiti, O.; Pohl, R.; Krásný, L.; Hocek, M. Photocaged 5-(hydroxymethyl)pyrimidine nucleoside phosphoramidites for specific photoactivatable epigenetic labeling of DNA. Org. Lett. 2020, 22, 9081–9085. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.S.; Thalhammer, A.; El-Sagheer, A.H.; Brown, T.; Schofield, C.J. Improved synthesis of 5-hydroxymethyl-2’-deoxy-cytidine phosphoramidite using a 2’-deoxyuridine to 2’-deoxycytidine conversion without temporary protecting groups. Bioorg. Med. Chem. Lett. 2011, 21, 1181–1184. [Google Scholar] [CrossRef]
- Xuan, S.; Wu, Q.; Cui, L.; Zhang, D.; Shao, F. 5-Hydroxymethylcytosine and 5-formylcytosine containing deoxyoligonucleotides: Facile syntheses and melting temperature studies. Bioorg. Med. Chem. Lett. 2015, 25, 1186–1191. [Google Scholar] [CrossRef]
- Shahsavari, S.; Chen, J.; Wigstrom, T.; Gooding, J.; Gauronskas, A.; Fang, S. Tritylation of alcohols under mild conditions without using silver salts. Tetrahedron Lett. 2016, 57, 3877–3880. [Google Scholar] [CrossRef]
- Akula, H.K.; Kokatla, H.; Andrei, G.; Snoeck, R.; Schols, D.; Balzarini, J.; Yang, L.-J.; Lakshman, M.K. Facile functionalization at the C4 position of pyrimidine nucleosides via amide group activation with (benzotriazol-1-yloxy)tris(dimethylamino)phos- phonium hexafluorophosphate (BOP) and biological evaluations of the products. Org. Biomol. Chem. 2017, 15, 1130–1139. [Google Scholar] [CrossRef]
- Zheng, X.-A.; Huang, H.-S.; Kong, R.; Chen, W.-J.; Gong, S.-S.; Sun, Q. An efficient PyAOP-based C4-amination method for direct access of oxidized 5MedC derivatives. Tetrahedron 2018, 74, 7095–7101. [Google Scholar] [CrossRef]
- Sun, Q.; Sun, J.; Gong, S.-S.; Wang, C.-J.; Pu, S.-Z.; Feng, F.-D. Efficient synthesis of 5-hydroxymethyl-, 5-formyl-, and 5-carboxyl-2’-deoxycytidine and their triphosphates. RSC Adv. 2014, 4, 36036–36039. [Google Scholar] [CrossRef]
- Sun, Q.; Gong, S.; Sun, J.; Wang, C.; Liu, S.; Liu, G.; Ma, C. Efficient synthesis of nucleoside 5’-triphosphates and their β,γ-bridging oxygen-modified analogs from nucleoside 5’-phosphates. Tetrahedron Lett. 2014, 55, 2114–2118. [Google Scholar] [CrossRef]
- Sun, Q.; Gong, S.; Sun, J.; Liu, S.; Xiao, Q.; Pu, S. A P(V)–N activation strategy for the synthesis of nucleoside polyphophates. J. Org. Chem. 2013, 78, 8417–8426. [Google Scholar] [CrossRef] [PubMed]


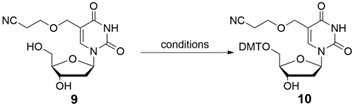 | ||
|---|---|---|
| Entry | Conditions | Yield (%) |
| 1 | DMT-Cl (1.5 eq), 20 °C, Pyr, 12 h | 51 a |
| 2 | DMT-Cl (1.5 eq), 40 °C, Pyr, 12 h | 46 a |
| 3 | DMT-Cl (1.5 eq), 60 °C, Pyr, 12 h | 34 a,b |
| 4 | DMT-Cl (1.5 eq), DMAP (0.15 eq), 20 °C, Pyr, 6 h | 48 a |
| 5 | DMT-Cl (1.5 eq), DMAP (0.15 eq), DIPEA (2 eq), 20 °C, Pyr, 6 h | 54 a |
| 6 | DMT-trifluoroacetate (1.5 eq), 20 °C, THF, 12 h | No reaction |
| 7 | DMT-trifluoroacetate (1.5 eq), 20 °C, Pyr, 12 h | No reaction |
| 8 | DMT+BF4−(1.5 eq), Li2CO3, (5 eq), DIPEA (3 eq), 20 °C, THF, 5 h | 22 a |
| 9 | DMT+BF4− (1.5 eq), 20 °C, Pyr, 1 h | 85 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, D.-Z.; Chen, Z.-Z.; Chi, M.; Dong, Y.-Y.; Pu, S.-Z.; Sun, Q. An Improved Approach for Practical Synthesis of 5-Hydroxymethyl-2′-deoxycytidine (5hmdC) Phosphoramidite and Triphosphate. Molecules 2022, 27, 749. https://doi.org/10.3390/molecules27030749
Yang D-Z, Chen Z-Z, Chi M, Dong Y-Y, Pu S-Z, Sun Q. An Improved Approach for Practical Synthesis of 5-Hydroxymethyl-2′-deoxycytidine (5hmdC) Phosphoramidite and Triphosphate. Molecules. 2022; 27(3):749. https://doi.org/10.3390/molecules27030749
Chicago/Turabian StyleYang, Dong-Zhao, Zhen-Zhen Chen, Mei Chi, Ying-Ying Dong, Shou-Zhi Pu, and Qi Sun. 2022. "An Improved Approach for Practical Synthesis of 5-Hydroxymethyl-2′-deoxycytidine (5hmdC) Phosphoramidite and Triphosphate" Molecules 27, no. 3: 749. https://doi.org/10.3390/molecules27030749
APA StyleYang, D.-Z., Chen, Z.-Z., Chi, M., Dong, Y.-Y., Pu, S.-Z., & Sun, Q. (2022). An Improved Approach for Practical Synthesis of 5-Hydroxymethyl-2′-deoxycytidine (5hmdC) Phosphoramidite and Triphosphate. Molecules, 27(3), 749. https://doi.org/10.3390/molecules27030749






