Chemical Indices and Kinetic Evaluation of β-Sitosteryl Oleate Oxidation in a Model System of Bulk Oil
Abstract
1. Introduction
2. Results and Discussion
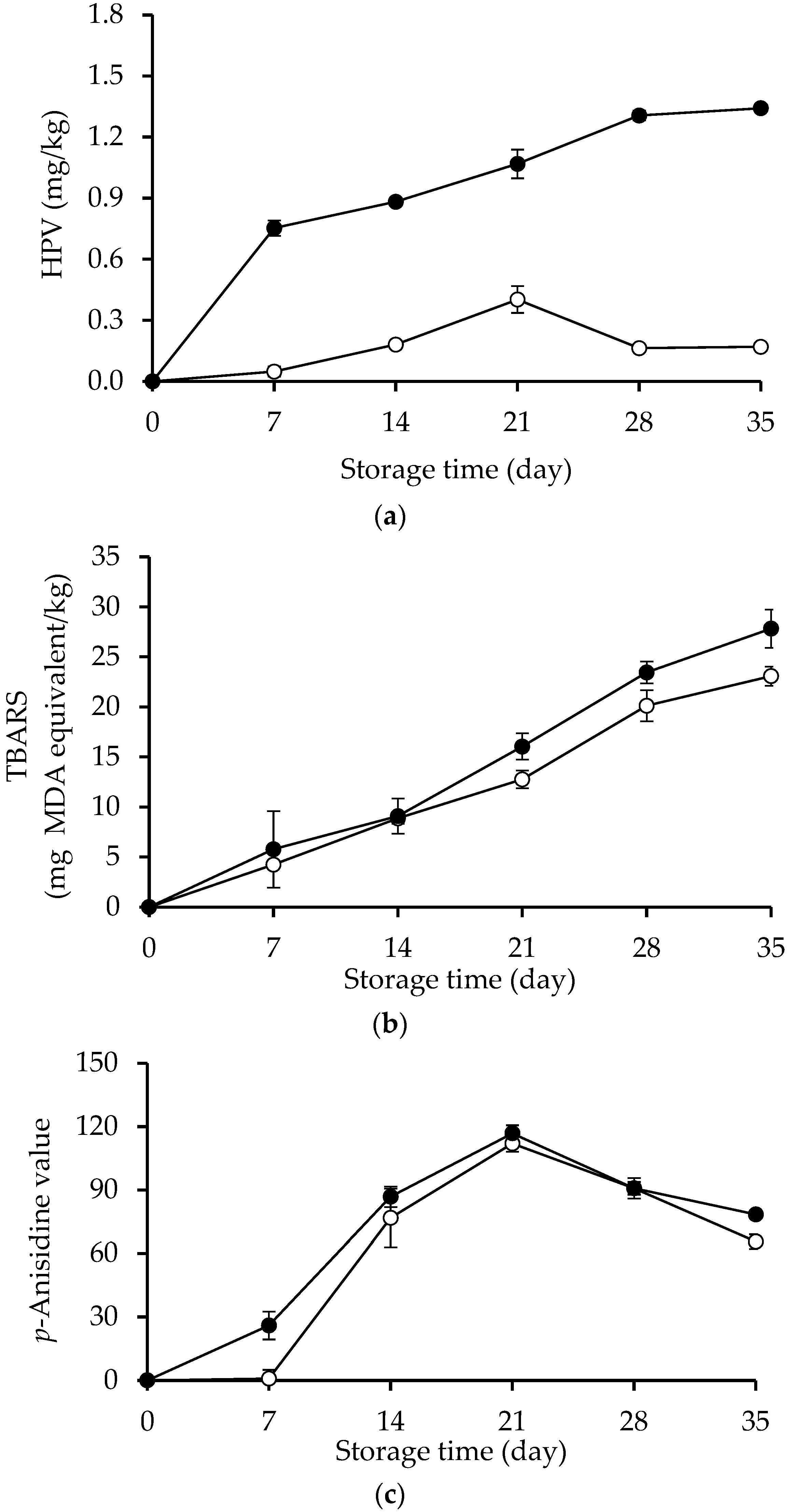
2.1. HPV, TBARS and AnV
2.2. Concentration of 7-Ketositosterol
2.3. Remaining of β-Sitosterol and β-Sitosteryl Oleate and Kinetics Evaluation
3. Materials and Methods
3.1. β-Sitosterol and β-Sitosteryl Oleate Samples
3.2. Oxidation in Bulk Oil Model System
3.3. Determination of HPV, TBARS and AnV
3.4. Determination of Phytosterol Oxide and Kinetic Evaluation for Sterol Remaining
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Matsuoka, R. Property of phytosterols and development of its containing mayonnaise-type dressing. Foods 2022, 11, 1141. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.D.S.; De Souza, A.H.; Fraga, J.L.; Villeneuve, P.; Torres, A.G.; Amaral, P.F. Lipases as effective green biocatalysts for phytosterol esters’ production: A review. Catalysts 2022, 12, 88. [Google Scholar] [CrossRef]
- Gachumi, G.; Poudel, A.; Wasan, K.M.; El-Aneed, A. Analytical strategies to analyze the oxidation products of phytosterols, and formulation-based approaches to reduce their generation. Pharmaceutics 2021, 13, 268. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Noakes, M.; Sullivan, D.; Erichsen, N.; Ross, D.; Annison, G.; Nestel, P. Cholesterol-lowering effects of plant sterol esters differ in milk, yoghurt, bread and cereal. Eur. J. Clin. Nutr. 2004, 58, 503–509. [Google Scholar] [CrossRef]
- Madsen, M.B.; Jensen, A.M.; Schmidt, E.B. The effect of a combination of plant sterol-enriched foods in mildly hypercholesterolemic subjects. Clin. Nutr. 2007, 26, 792–798. [Google Scholar] [CrossRef]
- Noakes, M.; Clifton, P.M.; Doornbos, A.M.; Trautwein, E.A. Plant sterol ester-enriched milk and yoghurt effectively reduce serum cholesterol in modestly hypercholesterolemic subjects. Eur. J. Nutr. 2005, 44, 214–222. [Google Scholar] [CrossRef]
- Abumweis, S.S.; Vanstone, C.A.; Ebine, N.; Kassis, A.; Ausman, L.M.; Jones, P.J.; Lichtenstein, A.H. Intake of a single morning dose of standard and novel plant sterol preparations for 4 weeks does not dramatically affect plasma lipid concentrations in humans. J. Nutr. 2006, 136, 1012–1016. [Google Scholar] [CrossRef]
- Miettinen, T.A.; Vanhanen, H. Dietary sitostanol related to absorption, synthesis and serum level of cholesterol in different apolipoprotein E phenotypes. Atherosclerosis 1994, 105, 217–226. [Google Scholar] [CrossRef]
- Chaijan, M.; Panpipat, W. Instability of β-sitosteryl oleate and β-sitosterol loaded in oil-in-water emulsion. NFS J. 2020, 21, 22–27. [Google Scholar] [CrossRef]
- Poudel, A.; Gachumi, G.; Purves, R.; Badea, I.; El-Aneed, A. Determination of phytosterol oxidation products in pharmaceutical liposomal formulations and plant vegetable oil extracts using novel fast liquid chromatography-Tandem mass spectrometric methods. Anal. Chim. Acta. 2022, 1194, 339404. [Google Scholar] [CrossRef]
- Lercker, G.; Rodrigues-Estrada, M.T. Cholesterol oxidation mechanism. In Cholesterol and Phytosterol Oxidation Products; Guardiola, F., Dutta, P.C., Codony, R., Savage, G.P., Eds.; AOAC Press: Champaign, IL, USA, 2002; pp. 1–25. [Google Scholar]
- Scholz, B.; Guth, S.; Engel, K.H.; Steinberg, P. Phytosterol oxidation products in enriched foods: Occurrence, exposure, and biological effects. Mol. Nutr. Food Res. 2015, 59, 1339–1352. [Google Scholar] [CrossRef] [PubMed]
- Smet, E.D.; Mensink, R.P.; Plat, J. Effects of plant sterols and stanols on intestinal cholesterol metabolism: Suggested mechanisms from past to present. Mol. Nutr. Food Res. 2012, 56, 1058–1072. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Davidson, M.H.; Umporowicz, D.M.; Chaefer, E.J.; Dicklin, M.R.; Ingram, K.A.; Perrone, G. Lipid responses to plant-sterol-enriched reduced-fat spreads incorporated into a national cholesterol education program step I diet. Am. J. Clin. Nutr. 2001, 74, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Liu, Y.; Zhao, T.; Xiao, F.; Yang, X.; Lu, B. Dietary sterols and sterol oxidation products on atherosclerosis: An insight provided by liver proteomic and lipidomic. Mol. Nutr. Food Res. 2021, 65, 2100516. [Google Scholar] [CrossRef]
- Chaiyasit, W.; Elias, R.J.; McClements, D.J.; Decker, E.A. Role of physical structures in bulk oils on lipid oxidation. Crit. Rev. Food Sci. Nutr. 2007, 47, 299–317. [Google Scholar] [CrossRef]
- Barriuso, B.; Otaegui-Arrazola, A.; Menéndez-Carreño, M.; Astiasarán, I.; Ansorena, D. Sterols heating: Degradation and formation of their ring-structure polar oxidation products. Food Chem. 2012, 135, 706–712. [Google Scholar] [CrossRef]
- Yanishlieva, N.; Marinova, E.; Schiller, H.; Seher, A. Comparison of sitosterol autoxidation in free form, as fatty acid ester and in triacylglycerol solution. Kinetics of the process and structure of the products formed. Dev. Food Sci. 1985, 11, 619–626. [Google Scholar]
- Cercaci, L.; Rodriguez-Estrada, M.T.; Lercker, G.; Decker, E.A. Phytosterol oxidation in oil-in-water emulsions and bulk oil. Food Chem. 2007, 102, 161–167. [Google Scholar] [CrossRef]
- Panpipat, W.; Chaijan, M.; Guo, Z. Oxidative stability of margarine enriched with different structures of β-sitosteryl esters during storage. Food Biosci. 2018, 22, 78–84. [Google Scholar] [CrossRef]
- Lehtonen, M.; Lampi, A.M.; Riuttamäki, M.A.; Piironen, V. Oxidation reactions of steryl esters in a saturated lipid matrix. Food Chem. 2012, 134, 2030–2039. [Google Scholar] [CrossRef]
- Panpipat, W.; Dong, M.; Xu, X.; Guo, Z. Thermal properties and nanodispersion behavior of synthesized β-sitosteryl acyl esters: A structure-activity relationship study. J. Colloid Interface Sci. 2013, 407, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Nieminen, V.; Laakso, P.; Kuusisto, P.; Niemelä, J.; Laitinen, K. Plant stanol content remains stable during storage of cholesterol-lowering functional foods. Food Chem. 2016, 196, 1325–1330. [Google Scholar] [CrossRef] [PubMed]
- Ansorena, D.; Barriuso, B.; Cardenia, V.; Astiasarán, I.; Lercker, G.; Rodriguez-Estrada, M.T. Thermo-oxidation of cholesterol: Effect of the unsaturation degree of the lipid matrix. Food Chem. 2013, 141, 2757–2764. [Google Scholar] [CrossRef] [PubMed]
- Rudzińska, M.; Przybylski, R.; Wąsowicz, E. Products formed during thermo-oxidative degradation of phytosterols. J. Am. Oil Chem. Soc. 2009, 86, 651–662. [Google Scholar] [CrossRef]
- Rudzińska, M.; Przybylski, R.; Wąsowicz, E. Degradation of phytosterols during storage of enriched margarines. Food Chem. 2014, 142, 294–298. [Google Scholar] [CrossRef]
- Shantha, N.C.; Decker, E.A. Rapid, sensitive, iron-based spectrophotometric methods for determination of peroxide values of food lipids. J. AOAC Int. 1994, 77, 421–424. [Google Scholar] [CrossRef]
- Ke, P.J.; Woyewoda, A.D. Microdetermination of thiobarbituric acid values in marine lipids by a direct spectrophotometric method with a monophasic reaction system. Anal. Chim. Acta. 1979, 106, 279–284. [Google Scholar] [CrossRef]
- IUPAC. Standard Methods for the Analysis of Oils, Fats and Derivatives., 7th ed.; Blackwell Scientific Publications: Oxford, UK, 1987. [Google Scholar]
- Soupas, L.; Juntunen, L.; Säynäjoki, S.; Lampi, A.M.; Piironen, V. GC-MS method for characterisation and quantification of sitostanol oxidation products. J. Am. Oil Chem. Soc. 2004, 81, 135–141. [Google Scholar] [CrossRef]
- Lampi, A.M.; Juntunen, L.; Toivo, J.; Piironen, V. Determination of thermo-oxidation products of plant sterols. J. Chromatogr. B. 2002, 777, 83–92. [Google Scholar] [CrossRef]

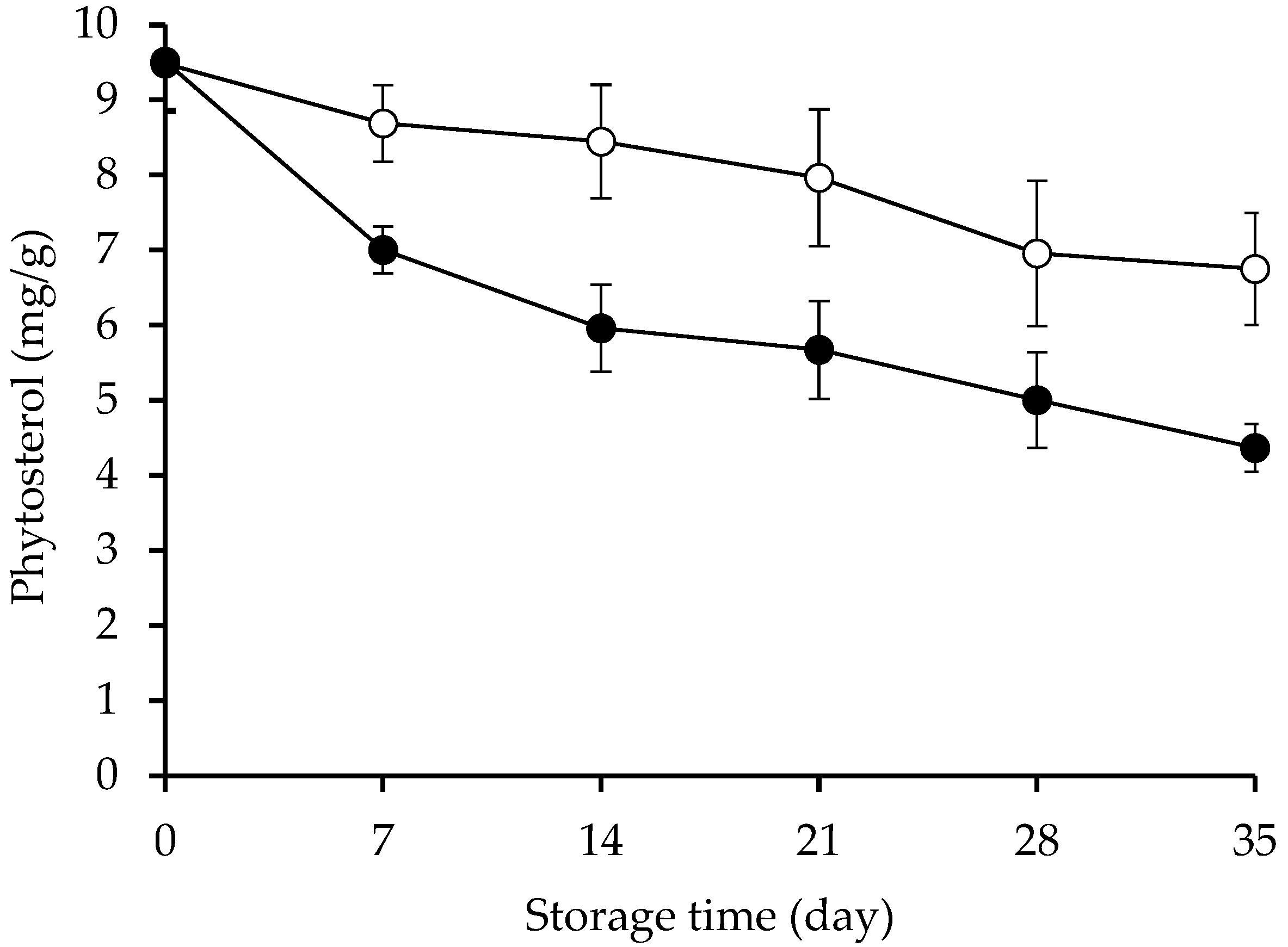

| β-Sitosterol Species | Reaction Rate Constant (k; day−1) | R2 |
|---|---|---|
| Unesterified β-sitosterol | 0.0099 | 0.9648 |
| β-sitosteryl oleate | 0.0202 | 0.9391 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaijan, M.; Panpipat, W.; Cheong, L.-Z. Chemical Indices and Kinetic Evaluation of β-Sitosteryl Oleate Oxidation in a Model System of Bulk Oil. Molecules 2022, 27, 7833. https://doi.org/10.3390/molecules27227833
Chaijan M, Panpipat W, Cheong L-Z. Chemical Indices and Kinetic Evaluation of β-Sitosteryl Oleate Oxidation in a Model System of Bulk Oil. Molecules. 2022; 27(22):7833. https://doi.org/10.3390/molecules27227833
Chicago/Turabian StyleChaijan, Manat, Worawan Panpipat, and Ling-Zhi Cheong. 2022. "Chemical Indices and Kinetic Evaluation of β-Sitosteryl Oleate Oxidation in a Model System of Bulk Oil" Molecules 27, no. 22: 7833. https://doi.org/10.3390/molecules27227833
APA StyleChaijan, M., Panpipat, W., & Cheong, L.-Z. (2022). Chemical Indices and Kinetic Evaluation of β-Sitosteryl Oleate Oxidation in a Model System of Bulk Oil. Molecules, 27(22), 7833. https://doi.org/10.3390/molecules27227833







