Assessment of Performance of Photocatalytic Nanostructured Materials with Varied Morphology Based on Reaction Conditions
Abstract
1. Introduction
2. Discussion on Various Nanomaterials
2.1. Titanium Dioxide (TiO2) Nanostructures

| S. NO. | Material & Morphology | Method | Application | Photocatalytic Activity | Reference |
|---|---|---|---|---|---|
| Nanorod | |||||
| 1 | TiO2/Nanorod | Sol-gel | P-nitrophenol, under 15 W UV Philips lamp | 69% in 20 min | [30] |
| 2 | TiO2/Nanorod | Chemical vapour deposition | Methyl Orange (MO) & Methylene Blue (MB) under 100 W UV mercury lamp | 97% of MO in 100 min 99% of MB in 50 min | [18] |
| 3 | TiO2/Nanorod | Sol-gel | Phenol under18 W UV lamp, | 48% in 360 min | [23] |
| 4 | TiO2/Nanorod | Hydrothermal Method at 120 °C for 15 h | Phenol under UV light | 87% in 360 min | [31] |
| 5 | TiO2/Nanorod | Hydrothermal Method at 200 °C for 12 h | MO under 300 W UV Xenon lamp | 100% in 95 min | [32] |
| 6 | TiO2/Nanorod | Hydrothermal Method at 225 °C for 24 h | MO & MB under 6 W UV lamp | 100% of MO & 88% of MB in 120 min | [33] |
| 7 | TiO2/Nanorod | Hydrothermal Method at 180 °C for 12 h | Phenol, 20 W UV lamp, 365 | 55% in 360 min | [21] |
| 8 | TiO2/rod | Hydrothermal, 200 °C for 18 h | MB, under 6 W UV lamp | 80% in 100 min | [34] |
| 9 | TiO2/Nanorod | Hydrothermal Method at 180 °C for 24 h | MO, under UV mercury lamp | 51% in 150 min | [35] |
| Flower | |||||
| 10 | TiO2/Flower | Sol-gel | Phenol, under 18 W UV lamp | 70% within 360 min | [23] |
| 11 | TiO2/Flower | Hydrothermal, 180 °C for 12 h | Phenol, 20 W UV lamp | 97% in120 min | [21] |
| 12 | TiO2/Flower | Hydrothermal, 180 °C for 6 h | Rhodamine B (RhB), under 450 W UV Xenon lamp | 69% in 160 min | [36] |
| 13 | TiO2/Flower | Sol-gel | RhB under 300 W UV lamp | 91.4% in 50 min | [37] |
| 14 | TiO2/Flower | Hydrothermal, 150 °C for 24 h | RhB, under 350 W Xenon Visible lamp | 63% in 60 min | [38] |
| 15 | TiO2/Flower | Hydrothermal, 150 °C for 3 h | MB under UV lamp | 78% in 60 min | [39] |
| 16 | TiO2/Flower | Hydrothermal, 120 °C for 48 h | MO under sunlight | 60% in 60 min | [19] |
| 17 | TiO2/Flower | Hydrothermal, 150 °C for 24 h | MB under UV 300 W high-pressure mercury (Hg) lamp | 75% in 60 min | [40] |
| Tube | |||||
| 18 | TiO2/Tube | Furnace 500 °C for 4 h | Papermaking wastewater, under 375 W high-pressure Hg lamp | 99.5% in 720 min | [41] |
| 19 | TiO2/Tube | Electrochemical Method | MB, under UV lamp | 98% in 60 min | [20] |
| 20 | TiO2/Tube | Hydrothermal, 160 °C for 24 h | MO, under 300 W UV lamp | 50.2% in 60 min | [42] |
| 21 | TiO2/Tube | Sol-gel stirring at 40 °C for 24 h | RhB & Dibutyl phthalate (DBP) 125 W high-pressure Hg UV lamp | 20% of RhB in 60 min & 15% of DBP in 60 min | [43] |
| 22 | TiO2/Tube | Solvothermal, 180 °C for 24 h | Orange II, under 18 high-pressure Hg lamps | 97.98% in 3000 min | [44] |
| 23 | TiO2/Tube | Electrochemical method | MB, under UV light lamp | 72% in 200 min | [45] |
| 24 | TiO2/Tube | Electrochemical Method | Phenol, under 1000 W Xenon lamp visible light lamp | 99.5% in 40 min | [46] |
| Sphere | |||||
| 25 | TiO2/Sphere | Hydrothermal, 180 °C for 24 h | Phenol, under 20 W UV lamp, | 60% in 120 min | [21] |
| 26 | TiO2/Sphere | Hydrothermal, 130 °C for 48 h | MB, under UV lamp | 96% in 80 min | [47] |
| 27 | TiO2/Sphere | Hydrothermal, 200 °C for 18 h | MB, under 6 W UV lamp | 90% in 100 min | [34] |
| 28 | TiO2/Sphere | Hydrothermal, 80 °C for 24 h | MB, under UV light | 96% in 100 min | [48] |
| 29 | TiO2/Sphere | Hydrothermal, 160 °C for 24 h | MO, under 4 W UV lamp | 50% in 60 min | [49] |
| 30 | TiO2/Sphere | Hydrothermal, 150 °C for 72 h | MO, under 8 W UV lamp | 91.6% in 60 min | [50] |
| Spindle | |||||
| 31 | TiO2/spindle | Hydrothermal, 180 °C for 12 h | MO, under 300 W visible light | 38% in 120 min, | [51] |
| 32 | TiO2/spindle | Hydrothermal, 200 °C for 24 h | RhB, under 350 W Xenon visible lamp | 23% in 60 min | [52] |
| 33 | TiO2/spindle | Reverse micellar method | RhB, under UV lamp | 90% in 130 min | [53] |
| 34 | TiO2/spindle | Hydrothermal, 180 °C for 12 h | MO, under 250 W UV high-voltage Hg lamp | 91% in 300 min | [54] |
| 35 | TiO2/spindle | Hydrothermal, 200 °C for 24 h | RhB, visible light | 25% in 60 min | [55] |
2.2. Zinc Oxide (ZnO) Nanostructures

| S. NO. | Material & Morphology | Method | Application | Performance | Reference |
|---|---|---|---|---|---|
| Rod | |||||
| 1 | ZnO/Rod | Microwave reactor (heated to 80 °C for 10 min) | MO, under 300 W Hg lamp | 86.3% in 180 min | [69] |
| 2 | ZnO/Rod | Hydrothermal, 180 °C for 24 h | Resorcinol, under 15 W UV lamp | 100% in 120 min | [70] |
| 3 | ZnO/Rod | Atmospheric self-induction method | RhB, under 400 W Xenon visible lamp | 36.8% in 300 min | [71] |
| 4 | ZnO/Rod | Solvothermal, 80 °C for 5 h | MB under 300 W UV lamp | 100% in 20 min | [72] |
| 5 | ZnO/Rod | Hydrothermal, 140 °C for 12 h | MB under 6 W UV lamp | 98.5% in 100 min | [73] |
| 6 | ZnO/Rod | Hydrothermal, 120 °C for 20 h | Phenol under l 15 W UV lamp | 100% in 40 min | [74] |
| 7 | ZnO/Rod | Hydrothermal, 95 °C for 30 h | RhB under 500 W visible Xenon lamp | 50% in 300 min | [75] |
| Flower | |||||
| 8 | ZnO/Flower | Hydrothermal, 100 °C for 12 h | RhB, under 300 W Hg lamp | 99.84% in 25 min | [64] |
| 9 | ZnO/Flower | Thermal decomposition at 300 °C for 20 min | RhB, under 36 W UV lamp | 100% in 90 min | [76] |
| 10 | ZnO/Flower | Sol-gel at 80 °C | RhB, under 200 W high-pressure Hg UV lamp | 99.8% in 100 min | [77] |
| 11 | ZnO/Flower | Hydrothermal, 140 °C for 12 h | MB, under 6 W UV lamp | 94% in 100 min | [73] |
| 12 | ZnO/Flower | Precipitation method | RhB, under Hg UV lamp | 30% in 180 min | [78] |
| 13 | ZnO/Flower | Hydrothermal, at 190 °C for 1 h | MB, 125 W Hg UV lamp | 98% in 60 min | [77] |
| 14 | ZnO/Flower | Solution based at 97 °C for 4 h. | MB, under 30 W Hg UV lamp | 99.9% in 180 min | [79] |
| 15 | ZnO/Flower | Microwave, at 300 W for 12 s | MB, under high-pressure Hg UV lamp | 80% in 60 min | [63] |
| 16 | ZnO/Flower | Hydrothermal, at 90 ° C for 24 h | MB, under BLB UV lamp | 100% in 105 min | [80] |
| 17 | ZnO/Flower | Sol-gel at room temperature for 16 h. | RhB, under 300 W Xenon UV lamp | 100% in 100 min | [81] |
| Sphere | |||||
| 18 | ZnO/Sphere | Hydrothermal, at 140 °C for 12 h | MB, under 6 W UV lamp | 74% in 100 min | [73] |
| 19 | ZnO/Sphere | Hydrothermal, at 180 °C for 24 h | RhB, under 15 W UV lamp | 100% in 240 min | [82] |
| 20 | ZnO/Sphere | Heated in a silicone bath at 120−140 °C for 4 h | MO, under 24 W UV lamp | 90% in 300 min | [83] |
| 21 | ZnO/Sphere | Hydrothermal, at 180 °C for 4 h | Congo Red, under 30 W UV lamp | 99.2%, in 90 min | [84] |
| 22 | ZnO/Sphere | Hydrothermal, at 120 °C for 6 h | MB, under 80 W UV lamp | 95% in 60 min | [85] |
| Hexagonal | |||||
| 23 | ZnO/Hexagonal | Heated at 150 °C on a hotplate | MB, under 450 W medium pressure Hg UV lamp | 100% in 16 min | [68] |
| 24 | ZnO/Hexagonal | Calcined at 400 °C | MB, under 16 W UV lamp | 100% in 75 min | [86] |
| 25 | ZnO/Hexagonal | Solvothermal, at 110 °C for 10 h | RhB, under Hg UV lamp | 80% in 60 min | [87] |
| 26 | ZnO/Hexagonal | Sol-gel at 80 °C for 3 h | MB, under 100 W UV lamp | 100% in 20 min | [88] |
| 27 | ZnO/Hexagonal | Hydrothermal, 120 °C for 20 h | MB, under UV lamp | 100% in 60 min | [74] |
| 28 | ZnO/Hexagonal | solid-phase method | MO, under 300 W UV lamp | 96.4% in 60 min | [89] |
| 29 | ZnO/Hexagonal | Hydrothermal, at 200 °C for 24 h | MB, under 300 W Hg UV lamp | 60% in 180 min | [90] |
| 30 | ZnO/Hexagonal | Sol-gel at 80 °C for 12 h | MB, under UV lamp | 95% in 60 min | [65] |
| 31 | ZnO/Hexagonal | Sonochemical method | MB, under 400 W Xenon UV lamp | 97% in 30 min | [91] |
| Spindle | |||||
| 32 | ZnO/Spindle | Hydrothermal, 140 °C for 12 h | MB, under 365 UV lamp | 62% in 100 min | [73] |
| 33 | ZnO/Spindle | Hydrothermal, 120 °C for 8 h | MO, under UV lamp | 55% in 180 min | [67] |
| 34 | ZnO/Spindle | Hydrothermal, 150 °C for 3 h | RhB, under 8W HG UV lamp | 73% in 120 min | [92] |
| 35 | ZnO/Spindle | Microwave, at 110 °C for 17 min | MB, under 300 W high-pressure Hg UV lamp | 98% in 120 min | [93] |
| 36 | ZnO/Spindle | Hydrothermal, at 140 °C for 12 h | MB, under 6 W high-pressure Hg UV lamp | 72% in 100 min | [73] |
| 37 | ZnO/Spindle | Hydrothermal, 95 °C for 24 h | MB, under 60 W Hg UV lamp | 83% in 55 min | [94] |
2.3. Cadmium Sulphide (CdS) Nanostructures

| S. NO. | Material & Morphology | Method | Application | Performance | Reference |
|---|---|---|---|---|---|
| Rod | |||||
| 1 | CdS/Rod | Hydrothermal, 180 °C for 6 h | MB, under 300 W Xenon visible lamp | 70% in 80 min | [115] |
| 2 | CdS/Rod | Reflux method for 13 h | MB, under 300 W Xenon visible lamp | 95% in 50 min | [106] |
| 3 | CdS/Rod | Hydrothermal, 160 °C for 48 h | Malachite green (MG) & MO, under 300 W Xenon visible lamp | 67% of MG in 30 min & 58% of MO in 45 min | [116] |
| 4 | CdS/Rod | Hydrothermal, 120 °C for 10 h | Salicylic acid and p-nitrophenol under 125 W Hg UV lamp | 70% Salicylic acid & 43.7% p-nitrophenol in 240 min | [117] |
| 5 | CdS/Rod | Hydrothermal, 180 °C for 12 h | Congo red (CR), under a visible tungsten lamp | 40% in 25 min | [118] |
| 6 | CdS/Rod | Hydrothermal, 180 °C for 24 h | MB, under a 100 W visible lamp | 62% in 180 min | [119] |
| 7 | CdS/Rod | Hydrothermal, 160 °C for 12 h | MB, under Hg UV lamp | 35% in 120 min | [114] |
| 8 | CdS/Rod | Hydrothermal, 160 °C for 24 h | Cr (VI), under a 1 kW Xenon visible light lamp | 19% in 120 min | [120] |
| 9 | CdS/Rod | 400 °C for 1 h in an N2 atmosphere | RhB, under a 200 W tungsten halogen visible lamp | 100% in 55 min | [121] |
| 10 | CdS/Rod | Hydrothermal, 200 °C for 10 h | MB, under a Xenon visible lamp | 50% in 120 min | [122] |
| 11 | CdS/Rod | Hydrothermal, 180 °C for 1 h | Ciprofloxacin (CIP), under a 300 W Xenon visible lamp | 57% in 60 min | [123] |
| 12 | CdS/Rod | Wet chemical method under reflux condition (100 °C for 7 h) | MO under a 300 W UV mercury lamp | 93% within 40 min | [124] |
| Flower | |||||
| 13 | CdS/Flower | Hydrothermal 200 °C for 12 h | MB, under a 125 W Hg visible lamp | 100% in 220 min | [125] |
| 14 | CdS/Flower | Sol-gel method | MB, under a 300 W Xenon visible lamp | 80% in 60 min | [126] |
| 15 | CdS/Flower | Hydrothermal 260 °C for 12 h | RhB, under a 300 W Xenon visible lamp | 70%, in 180 min | [127] |
| 16 | CdS/Flower | Hydrothermal, 180 °C for 12 h, | Acid fuchsine, under a 125 W Hg UV lamp | 100% in 40 min | [109] |
| 17 | CdS/Flower | Hydrothermal, 160 °C for 12 h | MB, under a 300 W Xenon visible lamp | 100% in 180 min | [128] |
| 18 | CdS/Flower | Hydrothermal, 160 °C for 4 h | MB, MO & RhB, under a 500 W Xenon visible lamp | 100% of MB, 91% of MO, and 85% of RhB in 150 min | [113] |
| 19 | CdS/Flower | Hydrothermal, 200 °C for 5 h | RhB, under visible light irradiation | 93% in 120 min | [100] |
| Sheet | |||||
| 20 | CdS/Sheet | Hydrothermal, 80 °C for 72 h | H2 production under a visible-light, AM 1.5 G solar simulator | 20 μmol within 480 min | [129] |
| 21 | CdS/Sheet | Electrochemical deposition for 15 min | CO2 reduction under sunlight | 2.1 μmol/g of C2H5OH and 62.8 μmol/g of HCOOH, 0.25% in 300 min | [130] |
| 22 | CdS/Sheet | Microwave method, at 80 °C for 30 min | H2 production, under visible light irradiation | 27.4 μmol/g in 240 min | [131] |
| 23 | CdS/Sheet | Heated in an oil bath at 60 °C for 3 h | H2 production under a 350 W Xenon visible lamp | 582 μmol/g in 240 min | [103] |
| 24 | CdS/Sheet | Ultrasonication at 90 °C for 2.5 h | RhB, under a 500 W Xenon visible lamp | 50% in 180 min | [112] |
| Sphere | |||||
| 25 | CdS/Sphere | Hydrothermal, at 120 °C for 10 h | Salicylic acid & p-nitrophenol, under a 125 W Hg UV lamp | 20% Salicylic acid & 6.25% p-nitrophenol in 240 min | [117] |
| 26 | CdS/Sphere | Hydrothermal, at 160 °C for 12 h | MB, under a Hg UV lamp | 38% in 120 min | [114] |
| 27 | CdS/Sphere | Hydrothermal, 180 °C for 4 h | Eosin Y, under a 500 W iodine tungsten lamp | 100% in 120 min | [107] |
| 28 | CdS/Sphere | Ultrasonic method | MB, under a 125 W UV lamp | 87% in 90 min | [132] |
| 29 | CdS/Sphere | Hydrothermal, at 100 °C for 2 h | 4-Chlorophenol, under 65 W fluorescent visible lamps | 52% in 150 min | [133] |
| 30 | CdS/Sphere | Microwave for 30 min | MB & RhB, under a 300 W Xenon visible lamp | 95% of MB in 150 min, 90% of RhB in 180 min | [102] |
| 31 | CdS/Sphere | Hydrothermal, at 200 °C for 3.5 h | RhB, under a 300 W tungsten halide visible lamp | 90% in 180 min | [134] |
| Particles | |||||
| 32 | CdS/Particle | Hydrothermal, at 160 °C for 12 h | MO, under a 350 W Xenon visible lamp | 12% in 60 min | [101] |
| 33 | CdS/Particle | Microwave for 20 s | Selective oxidation of alcohols to corresponding aldehydes under a 300 W Xenon visible lamp | 94% in 60 min | [108] |
| 34 | CdS/Particle | Hydrothermal, at 160 °C for 12 h | MB, under a Hg UV lamp | 29% in 120 min | [114] |
| 35 | CdS/Particle | Heating at 120 °C in an N2 environment | RhB, MB, & Cr (VI), under a 300 W Xenon visible lamp | 21% of RhB, 16% of Cr (VI) in 20 min & 24% of MB in 40 min | [135] |
| 36 | CdS/Particle | Hydrothermal, at 160 °C for 24 h | RhB, under a 250 W visible lamp | 72% in 240 min | [136] |
| 37 | CdS/Particle | Sol-gel method | MB, under a 300 W Xenon visible lamp | 48% in 60 min | [126] |
2.4. Silver Phosphate (Ag3PO4) Nanostructures

| S. NO. | Material & Morphology | Method | Application | Performance | Reference |
|---|---|---|---|---|---|
| Spherical | |||||
| 1 | Ag3PO4/Spherical | Continuous flow synthesis | Microfluidic photocatalytic dye-degradation, microreactor under visible light illumination | 97% within 15 min | [145] |
| 2 | Ag3PO4/Spherical | Precipitation method | Phenol, BSP, visible light, 400-W metal halide lamp | 82% phenol within 12 min, 81% Bisphenol within 10 min | [146] |
| 3 | Ag3PO4/Spherical | Precipitation | Rh.B. Xenon lamp (15 W), visible light | 88% within 35 min, | [147] |
| 4 | Ag3PO4/Spherical | Sol-gel | phenol under visible light irradiation with a 1000 W Xenon lamp | 42% within 60 min, | [142] |
| 5 | Ag3PO4/Spherical | Precipitation method at room temperature, 500 W Xenon lamp | MO under visible light irradiation | 35%, within 15 min | [141] |
| 6 | Ag3PO4/Spherical | Precipitation method | MB, 5 W compact fluorescent lamp, visible light | 78%, within 70 min | [13] |
| 7 | Ag3PO4/Spherical | Sol-gel | 6 W/649 fluorescent lamp | Sulfamethoxazole, 100% after 15 min | [148] |
| 8 | Ag3PO4/Spherical | Coprecipitation method at 20 °C | RhB, visible light, 300 W Xenon lamp | 66% within 6 min | [149] |
| 9 | Ag3PO4/Spherical | Precipitation method | Congo red (CR) under visible light irradiation, 400 W metal halogen lamp | 96%, within 210 min | [150] |
| 10 | Ag3PO4/Spherical | Precipitation method | CR, visible light irradiation with a 350 W Xenon lamp | 91%, within 14 min | [151] |
| 11 | Ag3PO4/Spherical | Precipitation method | RhB and MO dyes in 10 mgL−1, WLED with a luminous flux (Φv) of 85 l m | 100% within 30 min | [152] |
| Tetrahedral | |||||
| 12 | Ag3PO4/Tetrahedral | Sol-gel | phenol under visible light irradiation with a 1000 W Xenon lamp | 70% within 60 min, | [142] |
| 13 | Ag3PO4/Tetrahedral | Sol-gel method | MO, visible light, 500 W Xenon lamp | 100% within 90 min | [143] |
| 14 | Ag3PO4/Tetrahedral | Precipitation method | MB, MO, RhB Visible light, 500 W Xenon lamp | 100% MB, 93% MO, 100% RhB within 6 min | [153] |
| 15 | Ag3PO4/Tetrahedral | Precipitation method | MB, under visible light irradiation, 300 W Xenon lamp | 88% within 12 min | [154] |
| 16 | Ag3PO4/Tetrahedral | Ion exchange in the ethanol-water solvent at room temperature | RhB, visible-light provided by a 250 W Xenon lamp | 100% within 24 min | [155] |
| Dodecahedral | |||||
| 17 | Ag3PO4/dodecahedral | Sol-gel | phenol under visible light irradiation with a 1000 W Xenon lamp | 100% within 60 min, | [142] |
| 18 | Ag3PO4/dodecahedral | Precipitation method | CR under visible light irradiation, 400 W metal halogen lamp | 85%, within 210 min | [150] |
| 19 | Ag3PO4/dodecahedral | Precipitation method | MB), RhB, and reactive orange (RO), visible light, TL-D/35 fluorescent tube (18 W, Philips) | 90% MB, 82% RhB, 22% RO within 60 min | [156] |
| 20 | Ag3PO4/dodecahedral | Sol-gel method | MO, visible light, 500 W Xenon lamp | 78% within 90 min | [143] |
| 21 | Ag3PO4/dodecahedral | Hydrothermally processed at 150 °C for 24 h | RhB, UV illumination, 15 W UV germicidal irradiation lamps | 99.55% within 120 min | [157] |
| 22 | Ag3PO4/dodecahedral | Precipitation method | MB, MO, RhB Visible light, 500 W Xenon lamp | 93% MB, 62% MO, 100% RhB within 18 min | [153] |
| Polyhedral | |||||
| 23 | Ag3PO4/Polyhedral | Hydrothermal at 120 °C for 12 h | RhB, visible light, 300 W Xenon lamp | 97.83% in 6 min | [149] |
| 24 | Ag3PO4/Polyhedral | Sol-gel | RhB, visible light, 350 W Xenon lamp | 100% in 4 min | [137] |
| 25 | Ag3PO4/Polyhedral | Precipitation method | MO, 300 W halogen lamp | 85% of MO within 15 min | [158] |
| 26 | Ag3PO4/Polyhedral | Sol-gel | phenol under visible light irradiation with a 1000 W Xenon lamp | 100% within 60 min | [142] |
| 27 | Ag3PO4/Polyhedral | Conventional ion exchange/precipitation method | phenol under visible light irradiation with a 35 W Xenon lamp | 100% within 120 min | [159] |
| Cubic | |||||
| 28 | Ag3PO4/Cubic | Ion exchange method | RhB, Sunlight | 100% within 10 min | [144] |
| 29 | Ag3PO4/Cubic | Sol-gel method | MO, visible light, 500 W Xenon lamp | 65% within 90 min | [143] |
| 30 | Ag3PO4/Cubic | Precipitation method | crystal violet (CV) and MB, MO and orange G (OG) with visible irradiation, 125 W high-pressure HG lamp | 93.0% of CV, within 30 min, 98% MB, MO 79.4%, OG 57.3% within 50 min | [160] |
| 31 | Ag3PO4/Cubic | Precipitation method | MB, MO, RhB Visible light, 500 W Xenon lamp | 91% MB, 32% MO, 78% RhB within 18 min | [153] |
| 32 | Ag3PO4/Cubic | Simple ion-exchange deposition method | RhB, Visible light, 300 W Xenon | 92% within 30 min | [161] |
| 33 | Ag3PO4/Cubic | Hydrothermal treatment at 160 °C for 3 h | MB and RhB, sunny light between 10 am to 2 pm in the summer | 81% within 90 min | [162] |
2.5. Zinc Titanate (ZnTiO3) Nanostructures

| S. NO. | Material & Morphology | Method | Application | Performance | Reference |
|---|---|---|---|---|---|
| Particles | |||||
| 1 | ZnTiO3/Particle | Sol-gel | Phenol, under a 100 W incandescent visible lamp | 100% in 300 min | [170] |
| 2 | ZnTiO3/Particle | Hydrothermal, at 180 °C for 8 h | MB, under a 350 W Xenon visible lamp | 29.7% in 120 min | [168] |
| 3 | ZnTiO3/Particle | Sol-gel | 4-chlorophenol, under natural sunlight | 67% in 45 min | [171] |
| 4 | ZnTiO3/Particle | Sonochemical method | Rh), under a 70 W LED visible lamp | 36% in 150 min | [172] |
| 5 | ZnTiO3/Particle | Sol-gel | MO, under a 400 W UV lamp | 70% in 60 min | [167] |
| 6 | ZnTiO3/Particle | Solvothermal, at 180 °C 24 h | RhB & MO, under a 400 W halide visible lamp | 17% of Rh B, 3% of MO in 90 min | [157] |
| Rod | |||||
| 7 | ZnTiO3/Rod | Microwave | RhB, under a 150 W Xenon visible lamp | 93% in 180 min | [173] |
| 8 | ZnTiO3/Rod | Sol-gel | RhB, under a 50 W high-pressure Hg lamp | 97% in 70 min | [174] |
| 9 | ZnTiO3/Rod | Precipitation method | RhB, under sunlight | 71% in 60 min | [164] |
| 10 | ZnTiO3/Rod | Hydrothermal, 120 °C for 24 h | MO, under a 500 W Xenon lamp | 99.3% in 20 min | [175] |
| 11 | ZnTiO3/Rod | Sol-gel | RhB & crystal violet, under sunlight | 98% of CV in 60 min & 77% of RhB in 90 min | [166] |
| Spherical | |||||
| 12 | ZnTiO3/Spherical | Sol-gel | Methyl violet, under sunlight | 97% in 120 min | [169] |
| 13 | ZnTiO3/Spherical | Sol-gel | H2 production, under a 125 W high-pressure Hg UV lamp | 110 µmol/h, 60% in 3600 min | [176] |
| 14 | ZnTiO3/Spherical | Sol-gel | MB, under a 150 W UV lamp | 33% in 3600 min | [177] |
| 15 | ZnTiO3/Spherical | Sol-gel | Norfloxacin (NOR) and MO, under a 300 W Xenon visible lamp | 95% of NOR & 46% of MB in 60 min | [178] |
| 16 | ZnTiO3/Spherical | Sol-gel | MB, under sunlight | 76% in 60 min | [179] |
| Hexagonal | |||||
| 17 | ZnTiO3/Hexagonal | Sol-gel | p-nitrophenol, under sonocatalytic activity | 74.8% in 180 min | [180] |
| 18 | ZnTiO3/Hexagonal | Sol-gel | RhB, under sunlight | 35% in 180 min | [165] |
| S. No. | Materials | Photocatalysis Mechanism | Ref. |
|---|---|---|---|
| 1. | TiO2 |  | [183] |
| 2. | ZnO | 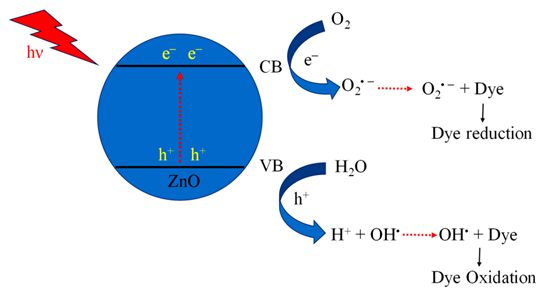 | [184] |
| 3. | CdS | 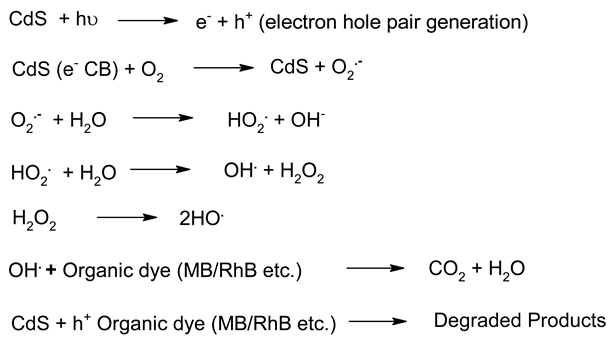 | [185] |
| 4. | Ag3PO4 |  | [186] |
| 5. | ZnTiO3 | 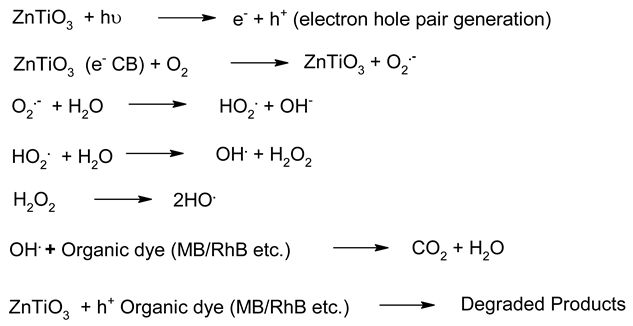 | [187] |
| S. No. | Photocatalytic Materials | Band Gap Energy (Eg) eV | Photocatalytic Performances | Ref. |
|---|---|---|---|---|
| 1. | TiO2 | Eg ~ 3.1 | The synthesised TiO2 nanotubes possess better photocatalytic activity than the as-prepared counterparts because of the larger surface area and good crystallinity. The emission stability of the catalyst also validates that TiO2 nanotubes could find potential applications in cold-cathode-based electronics. Literature shows that the decoration of TiO2 nanotubes by noble metal nanoparticles (such as Au, Ag, and Pt) also enhances its photocatalytic activity. | [45,188,189] |
| 2. | ZnO | Eg ~ 3.35 | Qu et al. Synthesised various shapes and morphology of ZnO nanomaterials using different ultrasonic processes. The study revealed that ZnO nanoflower morphology shows excellent photocatalytic activity. | [97,190] |
| 3. | Ag3PO4 | Eg ~ 2.45 | Morales et al. synthesised silver phosphate microcrystals with polyhedral morphologies and its higher surface area played an important role in higher photocatalytic activity.Geng et al. successfully prepared the polyhedral morphology of the Ag3PO4 microcrystal structure. The photocatalytic activity study confirmed its excellent photocatalytic ability. | [191,192] |
| 4. | CdS | Eg ~ 2.4 | Ganesh et al. synthesised CdS nanoflowers. The nano flowers of the CdS materials showed better photocatalytic activity in visible light. Wang et al. recently studied Ti3C2 MXene@CdS based nanoflowers composites heterostructures. It shows steady photoluminescence intensity and a longer fluorescence lifetime. | [100,193] |
| 5. | ZnTiO3 | Eg ~ 2.9 (indirect) 3.59 (direct) | Dutta et al. reported the Ag-doped ZnTiO3 nanorods. The photocatalytic results show improved photocatalytic activity. Chuaicham et al. recently studied ZnTiO3 mixed metal oxide. The higher photocatalytic activity was observed for phenol photodegradation. | [173,194] |
| Type of NPs | Stability and Recyclability | Biocompatibility | Ref. |
|---|---|---|---|
| TiO2 | The photostability of TiO2 for phenol degradation in four cycles remained constant. | Biocompatible, supports osteoblast-like cell formation, and can be used in biomedical applications. | [195,196,197] |
| ZnO | RhB aqueous solution for five cycles, good recyclability. | high bactericidal efficacy along with good cytocompatibility | [64,76,98,198] |
| Ag3PO4 | Photocatalytic efficiency remained consistently high after four cycles [11]. | Spectacular biocompatibility and good immunosensor sensitivity, low toxicity. | [137,199] |
| CdS | The catalytic activity of the photocatalyst remained constant. | Environment-friendly and economical photocatalyst | [200,201] |
| ZnTiO3 | The efficacy of the photocatalyst remained constant. | Excellent antitumor ability and good biocompatibility | [202,203,204] |
2.6. Challenges during the Application of NPs for Photochemical Reactions
2.7. Future Direction for the Application of Nanomaterial Photocatalyst
3. Conclusions
- (i)
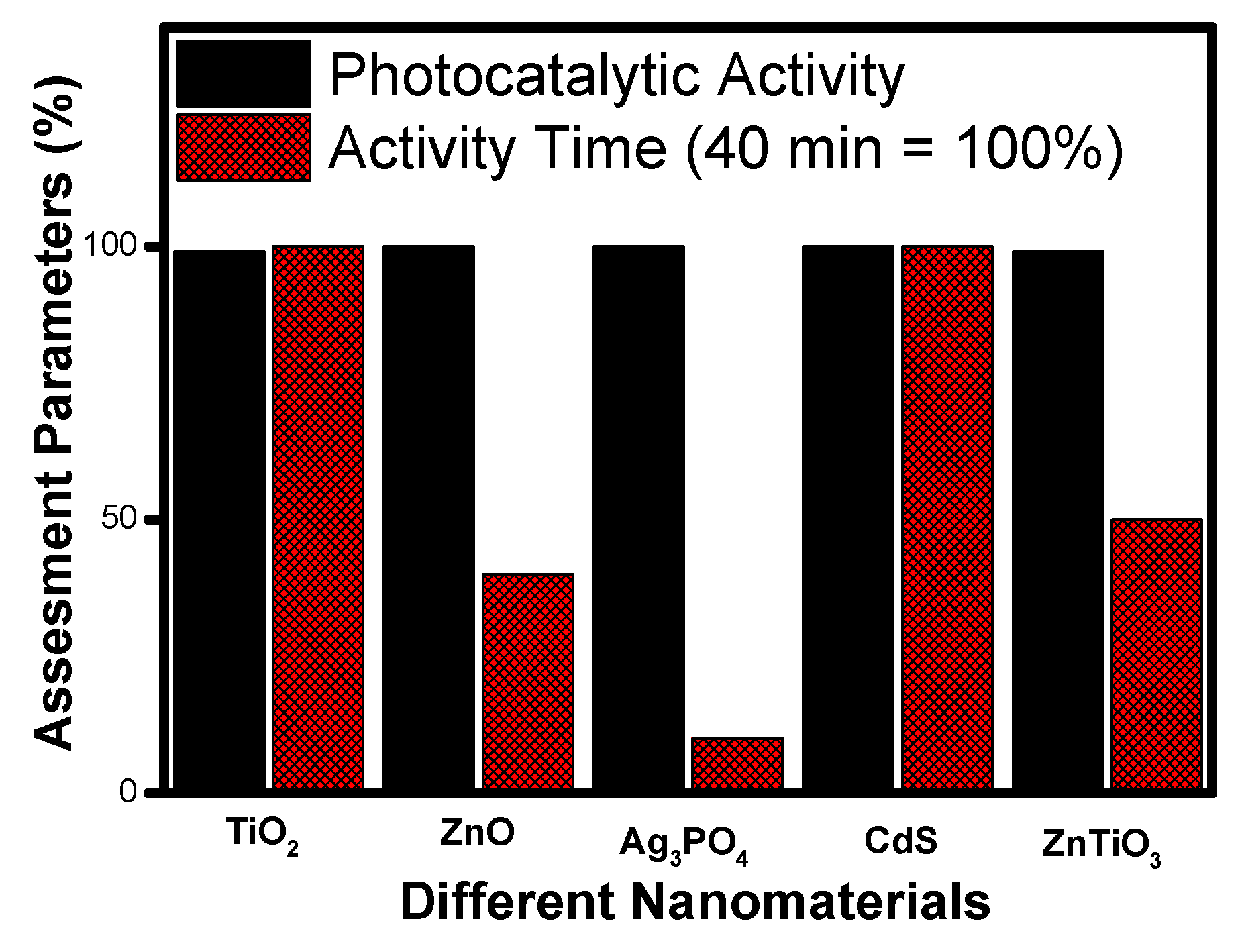
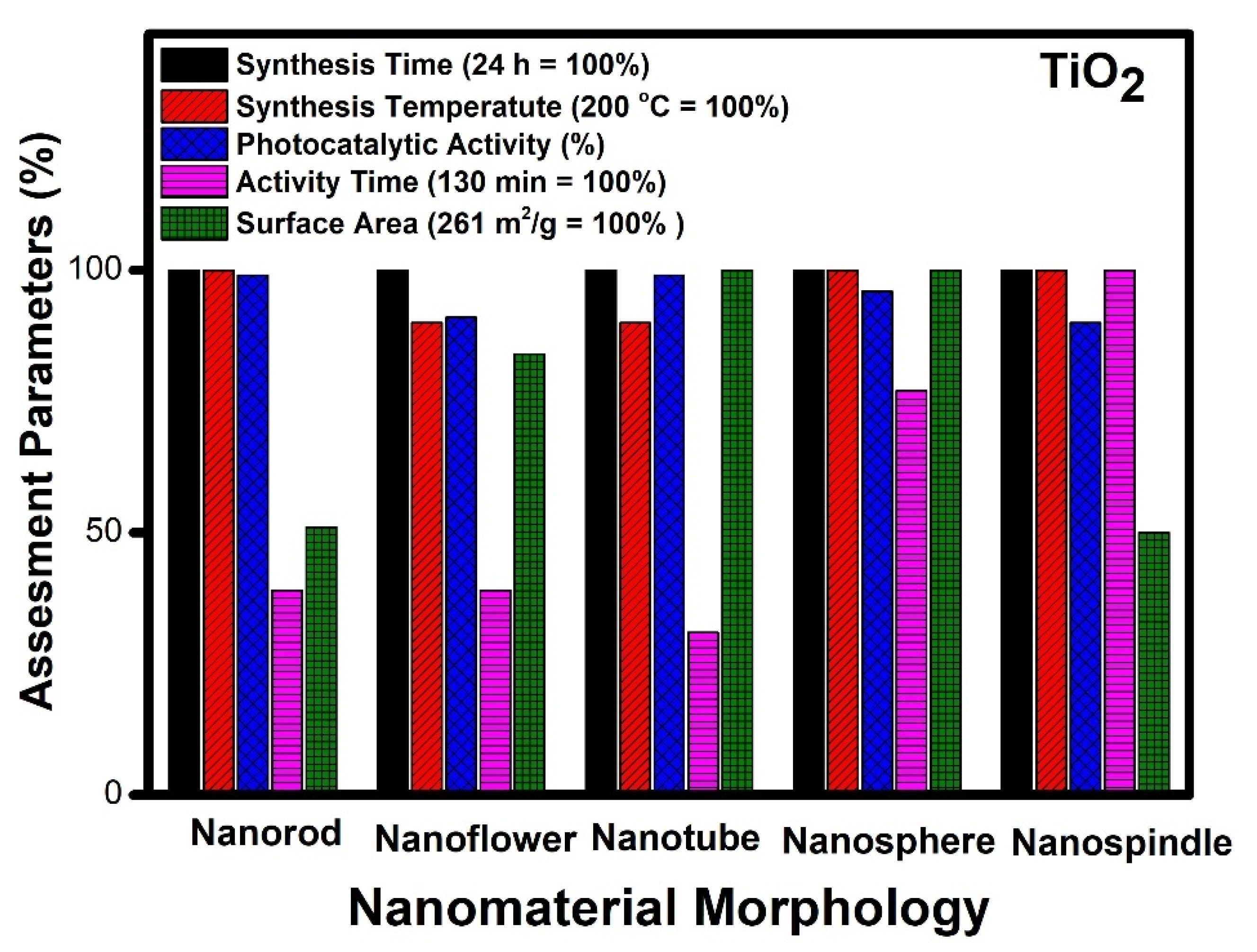
- The analysis of literature data indicates that titanium oxide (TiO2) nanotube morphology emerged as the best material. It has excellent photocatalytic performance and takes minimum time for the degradation of contaminants. Due to its high surface area, the active centres are easily accessible during photocatalysis;
- (ii)
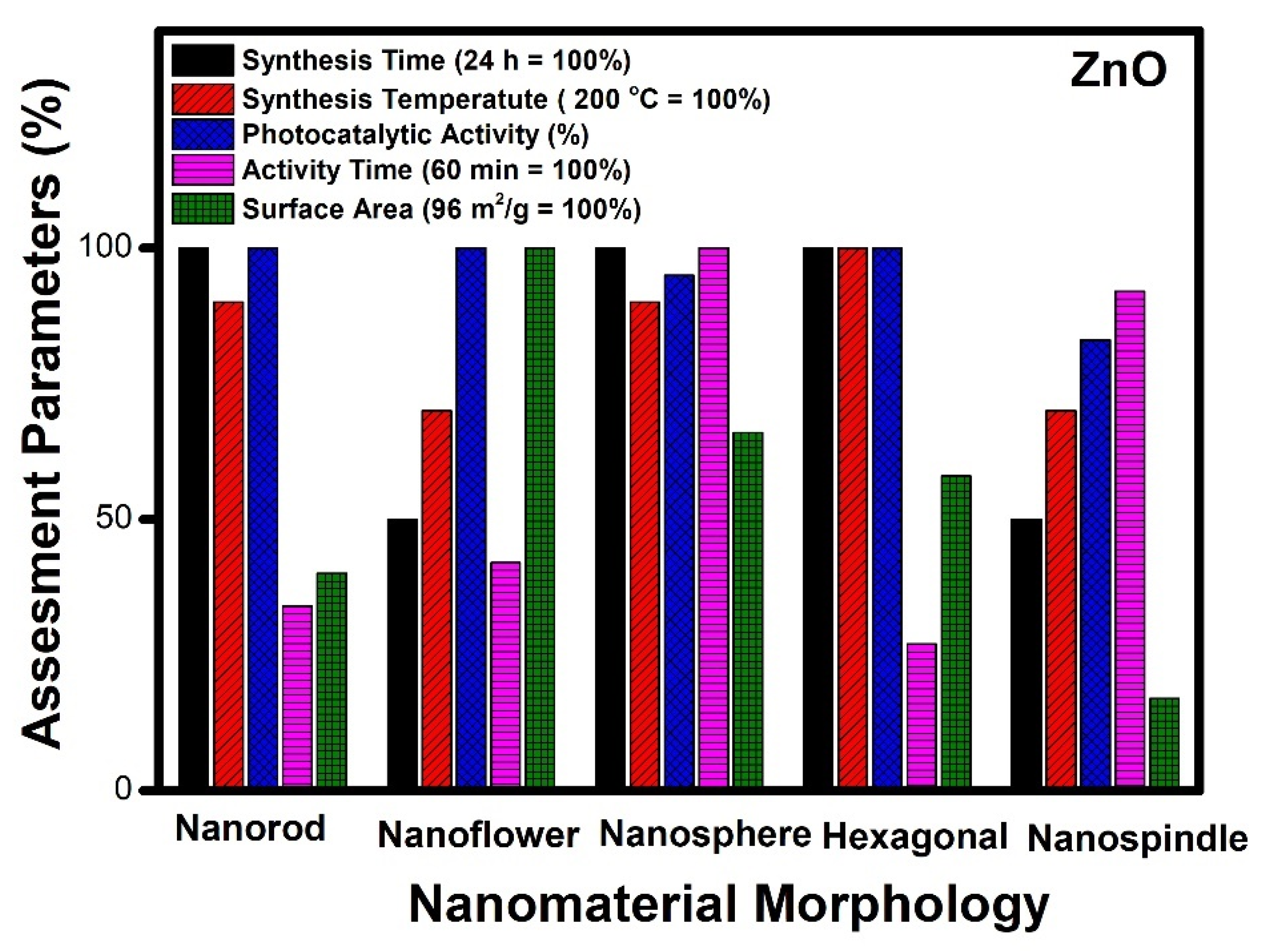
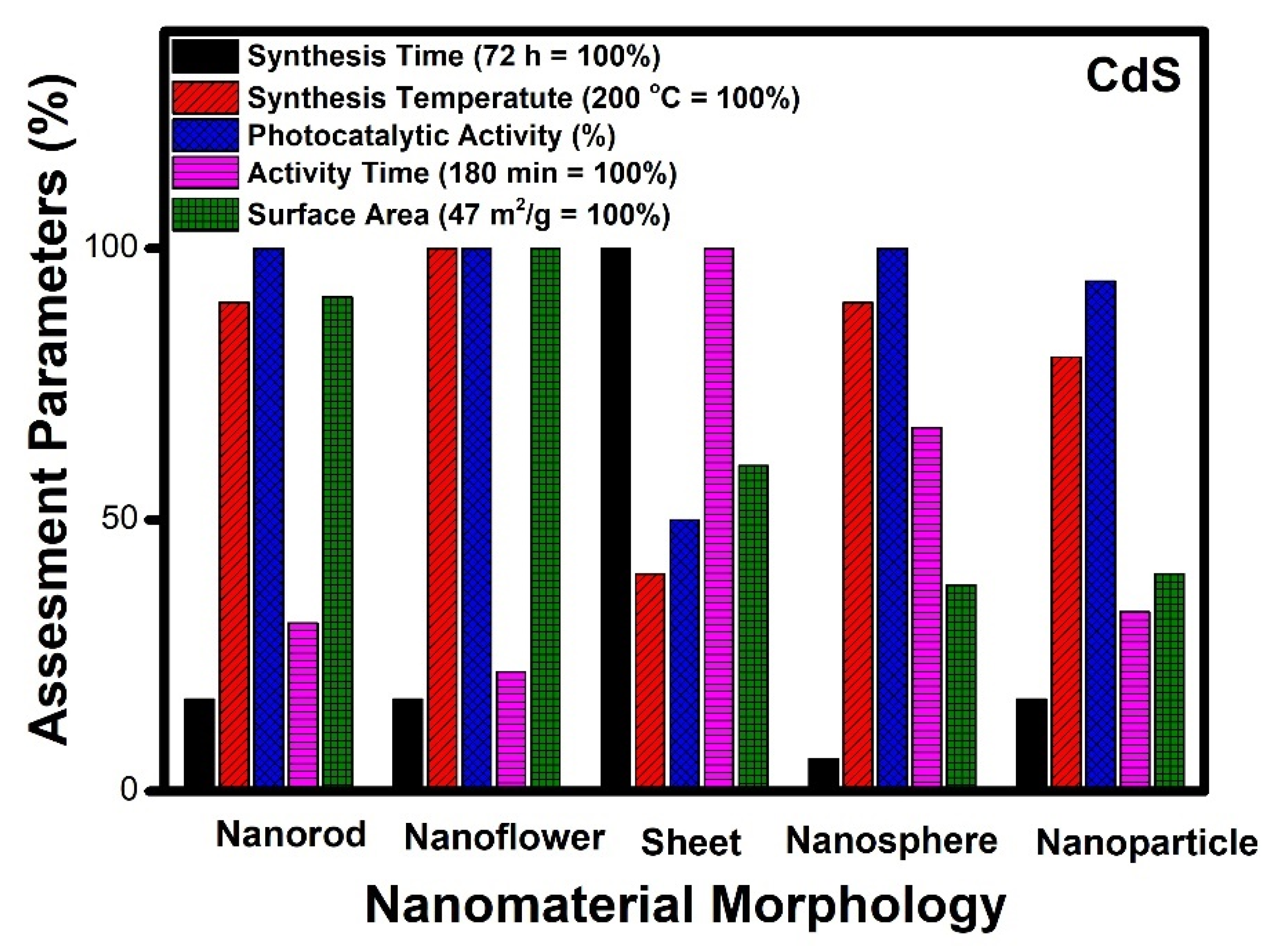
- Similar criteria, when applied to other nanomaterials, show zinc oxide (ZnO) and cadmium sulphide (CdS) nanomaterial having nanoflower morphology as the best among other reported;
- (iii)
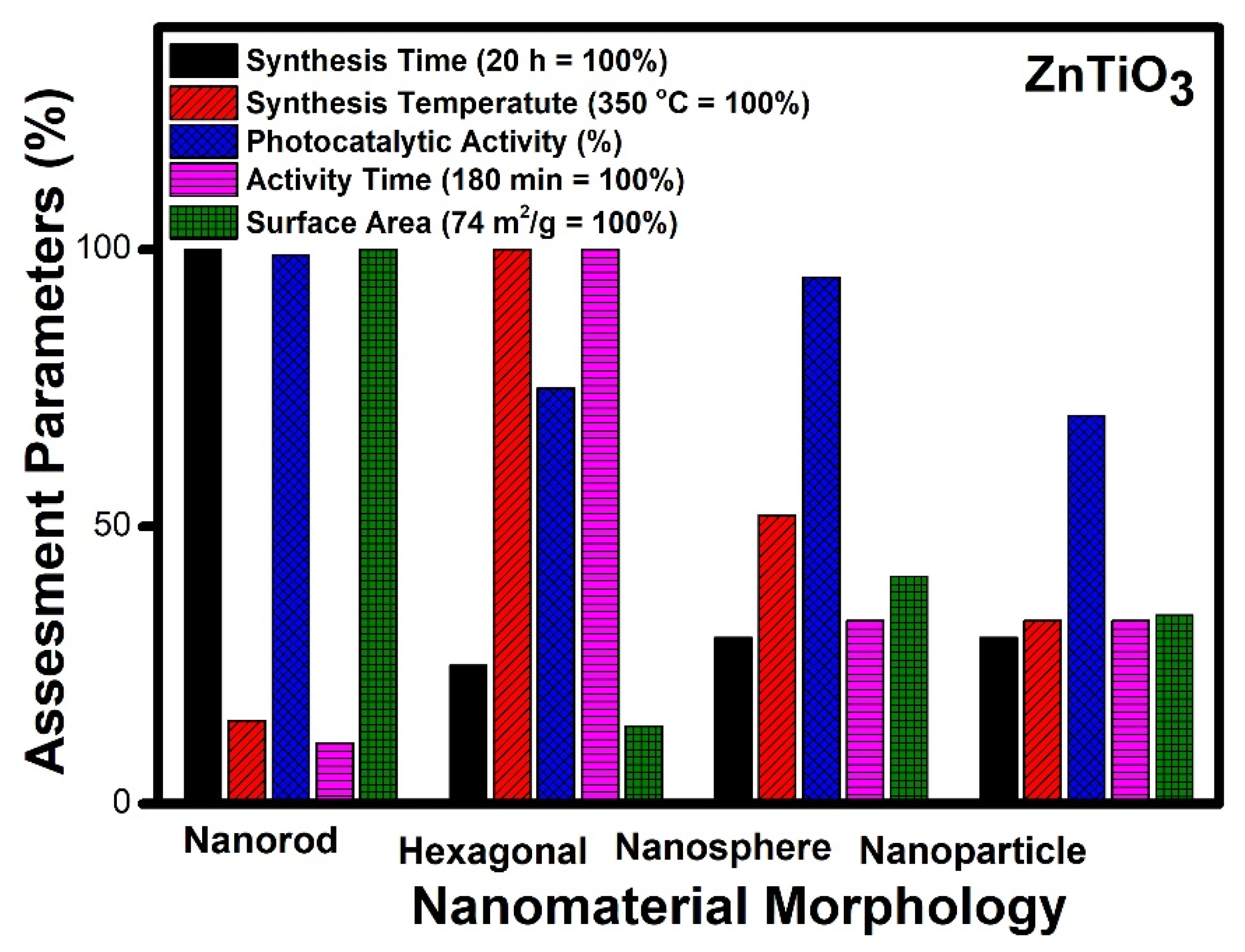
- Nanorod morphology appeared as the best morphology for zinc titanate (ZnTiO3) for photocatalytic applications;
- (iv)
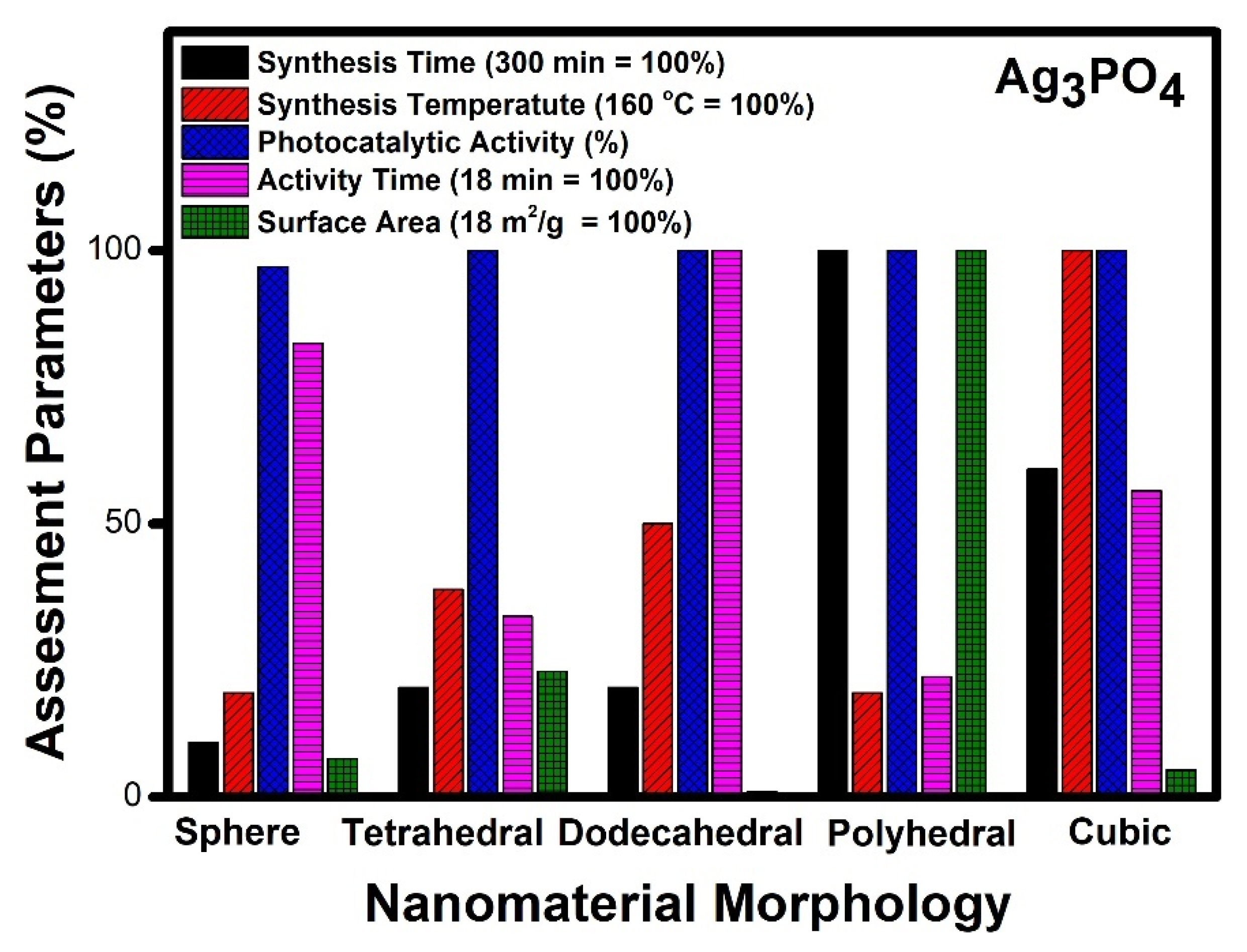
- Silver phosphate (Ag3PO4) shows polyhedral morphology as the best performer on all given counts and appears to be the best morphology for a photocatalyst.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inshakova, E.; Inshakov, O. World Market for Nanomaterials: Structure and Trends. MATEC Web Conf. 2017, 129, 02013. [Google Scholar] [CrossRef]
- Yashwant, S. Nanomaterials Market-Global Opportunity Analysis and Industry Forecast 2014–2022. Allied Market Research report No. A01419. 2016. Available online: https://www.alliedmarketresearch.com/europe-nanomaterials-market (accessed on 1 October 2022).
- Zhao, C.; Xi, M.; Huo, J.; He, C. B-Doped 2D-InSe as a Bifunctional Catalyst for CO2/CH4separation under the Regulation of an External Electric Field. Phys. Chem. Chem. Phys. 2021, 23, 23219–23224. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.R.; Deng, L.; Liu, G.C.; Wen, L.; Wang, J.G.; Huang, K.B.; Tang, H.T.; Pan, Y.M. Porous Organic Polymer-Derived Nanopalladium Catalysts for Chemoselective Synthesis of Antitumor Benzofuro[2,3-b]Pyrazine from 2-Bromophenol and Isonitriles. Org. Lett. 2019, 21, 4929–4932. [Google Scholar] [CrossRef] [PubMed]
- Nikazar, S.; Barani, M.; Rahdar, A.; Zoghi, M.; Kyzas, G. Photo-and Magnetothermally Responsive Nanomaterials for Therapy, Controlled Drug Delivery and Imaging Applications. ChemistrySelect 2020, 5, 12590–12609. [Google Scholar] [CrossRef]
- Pokropivny, V.V.; Skorokhod, V.V. Classification of Nanostructures by Dimensionality and Concept of Surface Forms Engineering in Nanomaterial Science. Mater. Sci. Eng. C 2007, 27, 990–993. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, D.; Ji, Y.; Ma, X.; Xu, J.; Que, D. Selenium Nanotubes Synthesized by a Novel Solution Phase Approach. J. Phys. Chem. B 2004, 108, 1179–1182. [Google Scholar] [CrossRef]
- Lao, J.Y.; Wen, J.G.; Ren, Z.F. Hierarchical ZnO Nanostructures. Nano Lett. 2002, 2, 1287–1291. [Google Scholar] [CrossRef]
- Ye, C.; Meng, G.; Wang, Y.; Jiang, Z.; Zhang, L. On the Growth of CdS Nanowires by the Evaporation of CdS Nanopowders. J. Phys. Chem. B 2002, 106, 10338–10341. [Google Scholar] [CrossRef]
- Ghoderao, K.P.; Jamble, S.N.; Kale, R.B. PEG—Assisted Morphological Transformation of 3D Flower-like ZnO to 1D Micro-/Nanorods and Nanoparticles for Enhanced Photocatalytic Activity. Mater. Res. Express 2017, 4, 105009. [Google Scholar] [CrossRef][Green Version]
- Nandiyanto, A.B.D.; Iskandar, F.; Okuyama, K. Macroporous Anatase Titania Particle: Aerosol Self-Assembly Fabrication with Photocatalytic Performance. Chem. Eng. J. 2009, 152, 293–296. [Google Scholar] [CrossRef]
- Vehring, R. Pharmaceutical Particle Engineering via Spray Drying. Pharm. Res. 2008, 25, 999–1022. [Google Scholar] [CrossRef]
- Yang, Z.M.; Liu, Y.Y.; Xu, L.; Huang, G.F.; Huang, W.Q. Facile Shape-Controllable Synthesis of Ag3PO4 Photocatalysts. Mater. Lett. 2014, 133, 139–142. [Google Scholar] [CrossRef]
- Tian, J.; Sang, Y.; Yu, G.; Jiang, H.; Mu, X.; Liu, H. A Bi2WO6-Based Hybrid Photocatalyst with Broad Spectrum Photocatalytic Properties under UV, Visible, and near-Infrared Irradiation. Adv. Mater. 2013, 25, 5075–5080. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Hu, H.; Jiao, Z.; Yu, H.; Lu, G.; Ye, J. Two-Dimensional Dendritic Ag3PO4 Nanostructures and Their Photocatalytic Properties. Phys. Chem. Chem. Phys. 2012, 14, 14486–14488. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Liu, H.; Wang, J.; Liu, D.; Du, G.; Cui, J. Ag2O/TiO2 Nanobelts Heterostructure with Enhanced Ultraviolet and Visible Photocatalytic Activity. ACS Appl. Mater. Interfaces 2010, 2, 2385–2392. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.X.; Miao, J.; Tao, H.B.; Hung, S.F.; Wang, H.Y.; Yang, H.B.; Chen, J.; Chen, R.; Liu, B. One-Dimensional Hybrid Nanostructures for Heterogeneous Photocatalysis and Photoelectrocatalysis. Small 2015, 11, 2115–2131. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, Y.; Li, Y.; Jia, D.; Xie, J. Directed Synthesis of TiO2 Nanorods and Their Photocatalytic Activity. Ceram. Int. 2014, 40, 11735–11742. [Google Scholar] [CrossRef]
- Tao, Y.G.; Xu, Y.Q.; Pan, J.; Gu, H.; Qin, C.Y.; Zhou, P. Glycine Assisted Synthesis of Flower-like TiO2 Hierarchical Spheres and Its Application in Photocatalysis. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2012, 177, 1664–1671. [Google Scholar] [CrossRef]
- Lamberti, A.; Chiodoni, A.; Shahzad, N.; Bianco, S.; Quaglio, M.; Pirri, C.F. Ultrafast Room-Temperature Crystallization of TiO2 Nanotubes Exploiting Water-Vapor Treatment. Sci. Rep. 2015, 5, 7808. [Google Scholar] [CrossRef]
- Tian, G.; Chen, Y.; Zhou, W.; Pan, K.; Tian, C.; Huang, X.R.; Fu, H. 3D Hierarchical Flower-like TiO2 Nanostructure: Morphology Control and Its Photocatalytic Property. CrystEngComm 2011, 13, 2994–3000. [Google Scholar] [CrossRef]
- Zhao, Z.; Kou, T.; Zhang, L.; Zhai, S.; Wang, W.; Wang, Y. Dealloying Induced N-Doping in Spindle-like Porous Rutile TiO2 for Enhanced Visible Light Photocatalytic Activity. Corros. Sci. 2018, 137, 204–211. [Google Scholar] [CrossRef]
- Zhao, Q.E.; Wen, W.; Xia, Y.; Wu, J.M. Photocatalytic Activity of TiO2 Nanorods, Nanowires and Nanoflowers Filled with TiO2 Nanoparticles. Thin Solid Films 2018, 648, 103–107. [Google Scholar] [CrossRef]
- Hagfeldt, A.; Grätzel, M. Molecular Photovoltaics. Acc. Chem. Res. 2000, 33, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Qi, L.; Jaroniec, M. Hydrogen Production by Photocatalytic Water Splitting over Pt/TiO2 Nanosheets with Exposed (001) Facets. J. Phys. Chem. C 2010, 114, 13118–13125. [Google Scholar] [CrossRef]
- Ganesh, V.A.; Raut, H.K.; Nair, A.S.; Ramakrishna, S. A Review on Self-Cleaning Coatings. J. Mater. Chem. 2011, 21, 16304–16322. [Google Scholar] [CrossRef]
- Orilall, M.C.; Wiesner, U. Block Copolymer Based Composition and Morphology Control in Nanostructured Hybrid Materials for Energy Conversion and Storage: Solar Cells, Batteries, and Fuel Cells. Chem. Soc. Rev. 2011, 40, 520–535. [Google Scholar] [CrossRef]
- Ghezelbash, A.; Koo, B.; Korgel, B.A. Self-Assembled Stripe Patterns of CdS Nanorods. Nano Lett. 2006, 6, 1832–1836. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, X.; Liu, J.; Zhai, Z.; Yang, Z.; Xia, J.; Deng, S.; Qu, X.; Zhang, H.; Wu, D.; et al. Mo-Modified Band Structure and Enhanced Photocatalytic Properties of Tin Oxide Quantum Dots for Visible-Light Driven Degradation of Antibiotic Contaminants. J. Environ. Chem. Eng. 2021, 10, 107091. [Google Scholar] [CrossRef]
- Sarkar, S.; Biswas, S.; Sarkar, M.; Ray, T.; Sharma, A.; Won, S.O.; Saha, A.; De, S. Ag Deposition Effects on the Photocatalytic Activity of Nanoparticulate TiO2—Comparison of Gamma Irradiation and UV Irradiation Methods. Nano-Struct. Nano-Objects 2018, 16, 134–143. [Google Scholar] [CrossRef]
- Liao, J.; Shi, L.; Yuan, S.; Zhao, Y.; Fang, J. Solvothermal Synthesis of TiO2 Nanocrystal Colloids from Peroxotitanate Complex Solution and Their Photocatalytic Activities. J. Phys. Chem. C 2009, 113, 18778–18783. [Google Scholar] [CrossRef]
- Zheng, X.; Kuang, Q.; Yan, K.; Qiu, Y.; Qiu, J.; Yang, S. Mesoporous TiO2 Single Crystals: Facile Shape-, Size-, and Phase-Controlled Growth and Efficient Photocatalytic Performance. ACS Appl. Mater. Interfaces 2013, 5, 11249–11257. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.; Park, Y.; Sohn, Y.; Leung, K.T.; Pradhan, D. Green Synthesis of Anatase TiO2 Nanocrystals with Diverse Shapes and Their Exposed Facets-Dependent Photoredox Activity. ACS Appl. Mater. Interfaces 2014, 6, 16498–16507. [Google Scholar] [CrossRef] [PubMed]
- Degabriel, T.; Colaço, E.; Domingos, R.F.; El Kirat, K.; Brouri, D.; Casale, S.; Landoulsi, J.; Spadavecchia, J. Factors impacting the aggregation/agglomeration and photocatalytic activity of highly crystalline spheroid- and rod-shaped TiO2 nanoparticles in aqueous solutions. Phys. Chem. Chem. Phys. 2018, 20, 12898–12907. [Google Scholar] [CrossRef] [PubMed]
- Santhi, K.; Navaneethan, M.; Harish, S.; Ponnusamy, S.; Muthamizhchelvan, C. Synthesis and characterization of TiO2 nanorods by hydrothermal method with different pH conditions and their photocatalytic activity. Appl. Surf. Sci. 2019, 500, 144058. [Google Scholar] [CrossRef]
- Wang, W.; Lu, C.; Ni, Y.; Su, M.; Xu, Z. Hydrothermal synthesis and enhanced photocatalytic activity of flower-like TiO2 on carbon nanotubes. Mater. Lett. 2012, 79, 11–13. [Google Scholar] [CrossRef]
- Song, H.; Chen, T.; Sun, Y.-L.; Zhang, X.-Q.; Jia, X.-H. Controlled synthesis of porous flower-like TiO2 nanostructure with enhanced photocatalytic activity. Ceram. Int. 2014, 40, 11015–11022. [Google Scholar] [CrossRef]
- Ge, M.; Li, J.W.; Liu, L.; Zhou, Z. Template-Free Synthesis and Photocatalytic Application of Rutile TiO2 Hierarchical Nanostructures. Ind. Eng. Chem. Res. 2011, 50, 6681–6687. [Google Scholar] [CrossRef]
- Liu, M.; Lu, W.-M.; Zhao, L.; Zhou, C.-L.; Li, H.-L.; Wang, W.-J. Fabrication and photocatalytical properties of flower-like TiO2 nanostructures. Trans. Nonferrous Met. Soc. China 2010, 20, 2299–2302. [Google Scholar] [CrossRef]
- Wang, Y.; Mo, Z.; Zhang, C.; Zhang, P.; Guo, R.; Gou, H.; Hu, R.; Wei, X. Morphology-controllable 3D flower-like TiO2 for UV shielding application. J. Ind. Eng. Chem. 2015, 32, 172–177. [Google Scholar] [CrossRef]
- Shao, C.; Zhou, G.; Li, Z.; Wu, Y.; Xu, D.; Sun, B. Fabrication of large-diameter tube-like mesoporous TiO2 via homogeneous precipitation and photocatalytic decomposition of papermaking wastewater. Chem. Eng. J. 2013, 230, 227–235. [Google Scholar] [CrossRef]
- Huang, F.; Guo, Y.; Wang, S.; Zhang, S.; Cui, M. Solgel-hydrothermal synthesis of Tb/Tourmaline/TiO2 nano tubes and enhanced photocatalytic activity. Solid State Sci. 2017, 64, 62–68. [Google Scholar] [CrossRef]
- He, G.; Zhang, J.; Hu, Y.; Bai, Z.; Wei, C. Dual-template synthesis of mesoporous TiO2 nanotubes with structure-enhanced functional photocatalytic performance. Appl. Catal. B Environ. 2019, 250, 301–312. [Google Scholar] [CrossRef]
- Zulfiqar, M.; Chowdhury, S.; Sufian, S.; Omar, A.A. Enhanced photocatalytic activity of Orange II in aqueous solution using solvent-based TiO2 nanotubes: Kinetic, equilibrium and thermodynamic studies. J. Clean. Prod. 2018, 203, 848–859. [Google Scholar] [CrossRef]
- Xu, X.; Tang, C.; Zeng, H.; Zhai, T.; Zhang, S.; Zhao, H.; Bando, Y.; Golberg, D. Structural Transformation, Photocatalytic, and Field-Emission Properties of Ridged TiO2 Nanotubes. ACS Appl. Mater. Interfaces 2011, 3, 1352–1358. [Google Scholar] [CrossRef] [PubMed]
- Pancielejko, A.; Mazierski, P.; Lisowski, W.; Zaleska-Medynska, A.; Kosek, K.; Łuczak, J. Facile Formation of Self-Organized TiO2 Nanotubes in Electrolyte Containing Ionic Liquid-Ethylammonium Nitrate and Their Remarkable Photocatalytic Properties. ACS Sustain. Chem. Eng. 2018, 6, 14510–14522. [Google Scholar] [CrossRef]
- Manjunath, K.; Yadav, L.S.R.; Jayalakshmi, T.; Reddy, V.; Rajanaika, H.; Nagaraju, G. Ionic liquid assisted hydrothermal synthesis of TiO2 nanoparticles: Photocatalytic and antibacterial activity. J. Mater. Res. Technol. 2018, 7, 7–13. [Google Scholar] [CrossRef]
- Mohammadi, N.N.; Pajootan, E.; Bahrami, H.; Arami, M. Magnetization of TiO2 nanofibrous spheres by one-step ultrasonic-assisted electrochemical technique. J. Mol. Liq. 2018, 265, 251–259. [Google Scholar] [CrossRef]
- Zheng, Z.; Wang, Z.; Xie, L.; Fang, Z.; Feng, W.; Huang, M.; Liu, P. Synthesis of single-crystal-like TiO2 hierarchical spheres with exposed {101} and {111} facets via lysine-inspired method. Appl. Surf. Sci. 2015, 353, 714–722. [Google Scholar] [CrossRef]
- An, T.; Chen, J.; Nie, X.; Li, G.; Zhang, H.; Liu, X.; Zhao, H. Synthesis of Carbon Nanotube-Anatase TiO2 Sub-Micrometer-Sized Sphere Composite Photocatalyst for Synergistic Degradation of Gaseous Styrene. ACS Appl. Mater. Interfaces 2012, 4, 5988–5996. [Google Scholar] [CrossRef]
- Sun, D.; Liu, J.; Li, J.; Feng, Z.; He, L.; Zhao, B.; Wang, T.; Li, R.; Yin, S.; Sato, T. Solvothermal synthesis of spindle-like WO3–TiO2 particles with enhanced photocatalytic activity. Mater. Res. Bull. 2014, 53, 163–168. [Google Scholar] [CrossRef]
- Tang, H.; Chang, S.; Jiang, L.; Tang, G.; Liang, W. Novel spindle-shaped nanoporous TiO2 coupled graphitic g-C3N4 nanosheets with enhanced visible-light photocatalytic activity. Ceram. Int. 2016, 42, 18443–18452. [Google Scholar] [CrossRef]
- Das, D.; Shivhare, A.; Saha, S.; Ganguli, A.K. Room temperature synthesis of mesoporous TiO2 nanostructures with high photocatalytic efficiency. Mater. Res. Bull. 2012, 47, 3780–3785. [Google Scholar] [CrossRef]
- Zhao, Z.; Xu, J.; Shang, C.; Ye, R.; Wang, Y. Dealloying-driven synthesis of sea-urchin like titanate nanowires and hierarchically porous anatase TiO2 nanospindles with enhanced photocatalytic performance. Corros. Sci. 2015, 98, 651–660. [Google Scholar] [CrossRef]
- Gao, P.; Liu, J.; Zhang, T.; Sun, D.D.; Ng, W. Hierarchical TiO2/CdS “spindle-like” composite with high photodegradation and antibacterial capability under visible light irradiation. J. Hazard. Mater. 2012, 229, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Raza, W.; Haque, M.; Muneer, M.; Fleisch, M.; Hakki, A.; Bahnemann, D.B.D. Photocatalytic degradation of different chromophoric dyes in aqueous phase using La and Mo doped TiO2 hybrid carbon spheres. J. Alloys Compd. 2015, 632, 837–844. [Google Scholar] [CrossRef]
- Ali, T.; Tripathi, P.; Azam, A.; Raza, W.; Ahmed, A.S.; Ahmed, A.; Muneer, M. Photocatalytic performance of Fe-doped TiO2nanoparticles under visible-light irradiation. Mater. Res. Express 2017, 4, 015022. [Google Scholar] [CrossRef]
- Allende-González, P.; Laguna-Bercero, M.Á.; Barrientos, L.; Valenzuela, M.L.; Díaz, C. Solid State Tuning of TiO2 Morphology, Crystal Phase, and Size through Metal Macromolecular Complexes and Its Significance in the Photocatalytic Response. ACS Appl. Energy Mater. 2018, 1, 3159–3170. [Google Scholar] [CrossRef]
- Perera, S.D.; Mariano, R.G.; Vu, K.; Nour, N.; Seitz, O.; Chabal, Y.; Balkus, K.J. Hydrothermal Synthesis of Graphene-TiO2 Nanotube Composites with Enhanced Photocatalytic Activity. ACS Catal. 2012, 2, 949–956. [Google Scholar] [CrossRef]
- Abbas, N.; Shao, G.N.; Haider, M.S.; Imran, S.M.; Park, S.S.; Jeon, S.J.; Kim, H.T. Inexpensive Sol-Gel Synthesis of Multiwalled Carbon Nanotube-TiO2 Hybrids for High Performance Antibacterial Materials. Mater. Sci. Eng. C 2016, 68, 780–788. [Google Scholar] [CrossRef]
- Ramakrishnan, V.M.; Muthukumarasamy, N.; Balraju, P.; Pitchaiya, S.; Velauthapillai, D.; Pugazhendhi, A. Transformation of TiO2 Nanoparticles to Nanotubes by Simple Solvothermal Route and Its Performance as Dye-Sensitized Solar Cell (DSSC) Photoanode. Int. J. Hydrog. Energy 2020, 45, 15441–15452. [Google Scholar] [CrossRef]
- Liu, N.; Chen, X.; Zhang, J.; Schwank, J.W. A Review on TiO2-Based Nanotubes Synthesized via Hydrothermal Method: Formation Mechanism, Structure Modification, and Photocatalytic Applications. Catal. Today 2014, 225, 34–51. [Google Scholar] [CrossRef]
- Ahmed, F.; Arshi, N.; Anwar, M.S.; Danish, R.; Koo, B.H. Morphological Evolution of ZnO Nanostructures and Their Aspect Ratio-Induced Enhancement in Photocatalytic Properties. RSC Adv. 2014, 4, 29249–29263. [Google Scholar] [CrossRef]
- Miao, Y.; Zhang, H.; Yuan, S.; Jiao, Z.; Zhu, X. Preparation of Flower-like ZnO Architectures Assembled with Nanosheets for Enhanced Photocatalytic Activity. J. Colloid Interface Sci. 2016, 462, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, H.; Zhang, Q.; Li, T. Adjusting the Proportions of {0001} Facets and High-Index Facets of ZnO Hexagonal Prisms and Their Photocatalytic Activity. RSC Adv. 2017, 7, 3515–3520. [Google Scholar] [CrossRef]
- Patrinoiu, G.; Hussien, M.D.; Calderón-Moreno, J.M.; Atkinson, I.; Musuc, A.M.; Ion, R.N.; Cimpean, A.; Chifiriuc, M.C.; Carp, O. Eco-Friendly Synthesized Spherical ZnO Materials: Effect of the Core-Shell to Solid Morphology Transition on Antimicrobial Activity. Mater. Sci. Eng. C 2019, 97, 438–450. [Google Scholar] [CrossRef]
- Lv, X.; Du, Y.; Li, Z.; Chen, Z.; Yang, K.; Liu, T.; Zhu, C.; Du, M.; Feng, Y. High Photocatalytic Property and Crystal Growth of Spindle-like ZnO Microparticles Synthesized by One-Step Hydrothermal Method. Vacuum 2017, 144, 229–236. [Google Scholar] [CrossRef]
- Messali, M.; Al Wadaani, F.; Oudghiri-Hassani, H.; Rakass, S.; Al Amri, S.; Benaissa, M.; Abboudi, M. Preparation, Characterization and Photocatalytic Activity of Hexagonal ZnO Nanoparticles. Mater. Lett. 2014, 128, 187–190. [Google Scholar] [CrossRef]
- Tong, L.; Li, W.; Shen, O.; Rong, H.; Gong, L. Microwave-Assisted the Facile Synthesis and Photocatalytic Properties of Rhombic ZnO Microstructures. Mater. Lett. 2018, 218, 1–4. [Google Scholar] [CrossRef]
- Lam, S.-M.; Sin, J.-C. A Green and Facile Hydrothermal Synthesis of ZnO Nanorods for Photocatalytic Application. JOJ Mater. Sci. 2018, 4, 8–10. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, J.K.; Wang, J.D.; Luo, H.X.; Lu, Y.; Yang, X.H. Atmospheric Self-Induction Synthesis and Enhanced Visible Light Photocatalytic Performance of Fe3+ Doped Ag-ZnO Mesocrystals. Ind. Eng. Chem. Res. 2014, 53, 13236–13246. [Google Scholar] [CrossRef]
- Deng, Q.; Duan, X.; Ng, D.H.L.; Tang, H.; Yang, Y.; Kong, M.; Wu, Z.; Cai, W.; Wang, G. Ag Nanoparticle Decorated Nanoporous ZnO Microrods and Their Enhanced Photocatalytic Activities. ACS Appl. Mater. Interfaces 2012, 4, 6030–6037. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, Q.; Wan, Q.; Dai, G.; Zhou, C.; Zou, B. Controllable ZnO Architectures by Ethanolamine-Assisted Hydrothermal Reaction for Enhanced Photocatalytic Activity. J. Phys. Chem. C 2011, 115, 2769–2775. [Google Scholar] [CrossRef]
- Xu, L.; Hu, Y.-L.; Pelligra, C.; Chen, C.-H.; Jin, L.; Huang, H.; Sithambaram, S.; Aindow, M.; Joesten, R.; Suib, S.L. ZnO with Different Morphologies Synthesized by Solvothermal Methods for Enhanced Photocatalytic Activity. Chem. Mater. 2009, 21, 2875–2885. [Google Scholar] [CrossRef]
- Wang, G.; Chen, D.; Zhang, H.; Zhang, J.Z.; Li, J. Tunable Photocurrent Spectrum in Well-Oriented Zinc Oxide Nanorod Arrays with Enhanced Photocatalytic Activity. J. Phys. Chem. C 2008, 112, 8850–8855. [Google Scholar] [CrossRef]
- Han, Z.; Liao, L.; Wu, Y.; Pan, H.; Shen, S.; Chen, J. Synthesis and photocatalytic application of oriented hierarchical ZnO flower-rod architectures. J. Hazard. Mater. 2012, 217, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Hezam, A.; Namratha, K.; Drmosh, Q.A.; Chandrashekar, B.N.; Sadasivuni, K.K.; Yamani, Z.H.; Cheng, C.; Byrappa, K. Heterogeneous Growth Mechanism of ZnO Nanostructures and the Effects of Their Morphology on Optical and Photocatalytic Properties. CrystEngComm 2017, 19, 3299–3312. [Google Scholar] [CrossRef]
- Liang, Y.; Guo, N.; Li, L.; Li, R.; Ji, G.; Gan, S. Facile Synthesis of Ag/ZnO Micro-Flowers and Their Improved Ultraviolet and Visible Light Photocatalytic Activity. New J. Chem. 2015, 40, 1587–1594. [Google Scholar] [CrossRef]
- Ranjith, K.S.; Kumar, R.T.R. Regeneration of an efficient, solar active hierarchical ZnO flower photocatalyst for repeatable usage: Controlled desorption of poisoned species from active catalytic sites. RSC Adv. 2017, 7, 4983–4992. [Google Scholar] [CrossRef]
- Silva, H.; Mateos-Pedrero, C.; Magén, C.; Tanaka, D.A.P.; Mendes, A. Simple hydrothermal synthesis method for tailoring the physicochemical properties of ZnO: Morphology, surface area and polarity. RSC Adv. 2014, 4, 31166–31176. [Google Scholar] [CrossRef]
- Sun, H.; Yu, Y.; Luo, J.; Ahmad, M.; Zhu, J. Morphology-controlled synthesis of ZnO 3D hierarchical structures and their photocatalytic performance. CrystEngComm 2012, 14, 8626–8632. [Google Scholar] [CrossRef]
- Yu, J.; Yu, X. Hydrothermal Synthesis and Photocatalytic Activity of Zinc Oxide Hollow Spheres. Environ. Sci. Technol. 2008, 42, 4902–4907. [Google Scholar] [CrossRef] [PubMed]
- de Lucas-Gil, E.; Leret, P.; Monte-Serrano, M.; Reinosa, J.J.; Enríquez, E.; Del Campo, A.; Cañete, M.; Menéndez, J.; Fernández, J.F.; Rubio-Marcos, F. ZnO Nanoporous Spheres with Broad-Spectrum Antimicrobial Activity by Physicochemical Interactions. ACS Appl. Nano Mater. 2018, 1, 3214–3225. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Mahjoub, A.R.; Abazari, R. Green synthesis of ZnO hollow sphere nanostructures by a facile route at room temperature with efficient photocatalytic dye degradation properties. RSC Adv. 2015, 5, 107378–107388. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Zhou, G.-Q.; Guo, J.; Liu, T.-Q. Controllable preparation of porous ZnO microspheres with a niosome soft template and their photocatalytic properties. Ceram. Int. 2016, 42, 12467–12474. [Google Scholar] [CrossRef]
- Pudukudy, M.; Yaakob, Z. Facile solid state synthesis of ZnO hexagonal nanogranules with excellent photocatalytic activity. Appl. Surf. Sci. 2014, 292, 520–530. [Google Scholar] [CrossRef]
- Mao, Y.; Li, Y.; Zou, Y.; Shen, X.; Zhu, L.; Liao, G. Solvothermal synthesis and photocatalytic properties of ZnO micro/nanostructures. Ceram. Int. 2018, 45, 1724–1729. [Google Scholar] [CrossRef]
- Zhang, L.; Yin, L.; Wang, C.; Lun, N.; Qi, Y. Sol−Gel Growth of Hexagonal Faceted ZnO Prism Quantum Dots with Polar Surfaces for Enhanced Photocatalytic Activity. ACS Appl. Mater. Interfaces 2010, 2, 1769–1773. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Y.; Hao, C.; Feng, F.; Yin, H.; Si, N. Solid-Phase Synthesis of Mesoporous ZnO Using Lignin-Amine Template and Its Photocatalytic Properties. Ind. Eng. Chem. Res. 2014, 53, 6585–6592. [Google Scholar] [CrossRef]
- Song, L.; Zhang, S.; Wu, X.; Wei, Q. Controllable Synthesis of Hexagonal, Bullet-Like ZnO Microstructures and Nanorod Arrays and Their Photocatalytic Property. Ind. Eng. Chem. Res. 2012, 51, 4922–4926. [Google Scholar] [CrossRef]
- Warule, S.S.; Chaudhari, N.S.; Kale, B.B.; More, M.A. Novel sonochemical assisted hydrothermal approach towards the controllable synthesis of ZnO nanorods, nanocups and nanoneedles and their photocatalytic study. CrystEngComm 2009, 11, 2776–2783. [Google Scholar] [CrossRef]
- Kumaresan, N.; Ramamurthi, K.; Babu, R.R.; Sethuraman, K.; Babu, S.M. Hydrothermally grown ZnO nanoparticles for effective photocatalytic activity. Appl. Surf. Sci. 2017, 418, 138–146. [Google Scholar] [CrossRef]
- Li, Q.; Li, H.; Wang, R.; Li, G.; Yang, H.; Chen, R. Controllable microwave and ultrasonic wave combined synthesis of ZnO micro-/nanostructures in HEPES solution and their shape-dependent photocatalytic activities. J. Alloys Compd. 2013, 567, 1–9. [Google Scholar] [CrossRef]
- Das, S.; Ghosh, S. Fabrication of different morphologies of ZnO superstructures in presence of synthesized ethylammonium nitrate (EAN) ionic liquid: Synthesis, characterization and analysis. Dalton Trans. 2012, 42, 1645–1656. [Google Scholar] [CrossRef]
- Arzac, G.M.; Fernández, A. Hydrogen Production through Sodium Borohydride Ethanolysis. Int. J. Hydrogen Energy 2015, 40, 5326–5332. [Google Scholar] [CrossRef]
- Raza, W.; Faisal, S.M.; Owais, M.; Bahnemann, D.; Muneer, M. Facile Fabrication of Highly Efficient Modified ZnO Photocatalyst with Enhanced Photocatalytic, Antibacterial and Anticancer Activity. RSC Adv. 2016, 6, 78335–78350. [Google Scholar] [CrossRef]
- Qu, Y.; Huang, R.; Qi, W.; Shi, M.; Su, R.; He, Z. Controllable Synthesis of ZnO Nanoflowers with Structure-Dependent Photocatalytic Activity. Catal. Today 2020, 355, 397–407. [Google Scholar] [CrossRef]
- Temel, S.; Gökmen, F.Ö.; Yaman, E. Investigation of Structural and Morphological Properties of ZnO Nanoflowers on Biocompatible Polymeric Substrate. Acad. Platf. J. Eng. Sci. 2020, 8, 36–40. [Google Scholar] [CrossRef]
- Li, Q.; Shi, T.; Li, X.; Lv, K.; Li, M.; Liu, F.; Li, H.; Lei, M. Remarkable Positive Effect of Cd(OH)2 on CdS Semiconductor for Visible-Light Photocatalytic H2 Production. Appl. Catal. B Environ. 2018, 229, 8–14. [Google Scholar] [CrossRef]
- Ganesh, R.S.; Sharma, S.K.; Durgadevi, E.; Navaneethan, M.; Ponnusamy, S.; Muthamizhchelvan, C.; Hayakawa, Y.; Kim, D.Y. Growth, Microstructure, Structural and Optical Properties of PVP-Capped CdS Nanoflowers for Efficient Photocatalytic Activity of Rhodamine, B. Mater. Res. Bull. 2017, 94, 190–198. [Google Scholar] [CrossRef]
- Alomar, M.; Liu, Y.; Chen, W.; Fida, H. Controlling the Growth of Ultrathin MoS2 Nanosheets/CdS Nanoparticles by Two-Step Solvothermal Synthesis for Enhancing Photocatalytic Activities under Visible Light. Appl. Surf. Sci. 2019, 480, 1078–1088. [Google Scholar] [CrossRef]
- Deng, C.; Tian, X. Facile Microwave-Assisted Aqueous Synthesis of CdS Nanocrystals with Their Photocatalytic Activities under Visible Lighting. Mater. Res. Bull. 2013, 48, 4344–4350. [Google Scholar] [CrossRef]
- Bie, C.; Fu, J.; Cheng, B.; Zhang, L. Ultrathin CdS Nanosheets with Tunable Thickness and Efficient Photocatalytic Hydrogen Generation. Appl. Surf. Sci. 2018, 462, 606–614. [Google Scholar] [CrossRef]
- Ouyang, L.; Maher, K.N.; Yu, C.L.; McCarty, J.; Park, H. Catalyst-Assisted Solution-Liquid-Solid Synthesis of CdS/CdSe Nanorod Heterostructures. J. Am. Chem. Soc. 2007, 129, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Onsager, L. The Effects Of Shape On The Interaction Of Colloidal Particles. Ann. N. Y. Acad. Sci. 1949, 51, 627–659. [Google Scholar] [CrossRef]
- Wang, J.; Li, B.; Chen, J.; Li, N.; Zheng, J.; Zhao, J.; Zhu, Z. Diethylenetriamine-Assisted Synthesis of CdS Nanorods under Reflux Condition and Their Photocatalytic Performance. J. Alloys Compd. 2012, 535, 15–20. [Google Scholar] [CrossRef]
- Wang, Z.; Pan, L.; Wang, L.; Wang, H. Urchin-like CdS microspheres self-assembled from CdS nanorods and their photocatalytic properties. Solid State Sci. 2011, 13, 970–975. [Google Scholar] [CrossRef]
- She, H.; Li, L.; Sun, Y.; Wang, L.; Huang, J.; Zhu, G.; Wang, Q. Facile preparation of mixed-phase CdS and its enhanced photocatalytic selective oxidation of benzyl alcohol under visible light irradiation. Appl. Surf. Sci. 2018, 457, 1167–1173. [Google Scholar] [CrossRef]
- Yao, W.-T.; Yu, S.-H.; Liu, S.-J.; Chen, J.-P.; Liu, X.-M.; Li, F.-Q. Architectural Control Syntheses of CdS and CdSe Nanoflowers, Branched Nanowires, and Nanotrees via a Solvothermal Approach in a Mixed Solution and Their Photocatalytic Property. J. Phys. Chem. B 2006, 110, 11704–11710. [Google Scholar] [CrossRef]
- Alivisatos, A.P.; Kadavanich, A.V.; Schlamp, M.C.; Peng, X. Epitaxial Growth of Highly Luminescent CdSe/CdS Core/Shell Nanocrystals with Photostability and Electronic Accessibility. J. Am. Chem. Soc. 1997, 119, 7019–7029. [Google Scholar] [CrossRef]
- Duan, X.; Lieber, C.M. General synthesis of compound semiconductor nanowires. Adv. Mater. 2000, 12, 298–302. [Google Scholar] [CrossRef]
- Zou, L.; Wang, H.; Wang, X. High Efficient Photodegradation and Photocatalytic Hydrogen Production of CdS/BiVO4 Heterostructure through Z-Scheme Process. ACS Sustain. Chem. Eng. 2016, 5, 303–309. [Google Scholar] [CrossRef]
- Wang, W.; Lu, Y.; Xu, Y.; Wu, K.; Huang, J.; Ji, C.; Ryu, S.O. A facile template-free approach for fabrication of flower-like CdS: The evolutionary process of the structure and the performance of photocatalytic activity. CrystEngComm 2016, 18, 4681–4687. [Google Scholar] [CrossRef]
- Ahmed, B.; Kumar, S.; Kumar, S.; Ojha, A.K. Shape induced (spherical, sheets and rods) optical and magnetic properties of CdS nanostructures with enhanced photocatalytic activity for photodegradation of methylene blue dye under ultra-violet irradiation. J. Alloys Compd. 2016, 679, 324–334. [Google Scholar] [CrossRef]
- Wang, X.; Mu, B.; An, X.; Wang, A. Insights into the Relationship between the Color and Photocatalytic Property of Attapulgite/CdS Nanocomposites. Appl. Surf. Sci. 2018, 439, 202–212. [Google Scholar] [CrossRef]
- Liang, Q.; Cui, S.; Liu, C.; Xu, S.; Yao, C.; Li, Z. Construction of CdS@UIO-66-NH2 core-shell nanorods for enhanced photocatalytic activity with excellent photostability. J. Colloid Interface Sci. 2018, 524, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Pal, B. Study of excited charge carrier’s lifetime for the observed photoluminescence and photocatalytic activity of CdS nanostructures of different shapes. J. Mol. Catal. A Chem. 2013, 371, 77–85. [Google Scholar] [CrossRef]
- Kumar, P.S.; Prabavathi, S.L.; Indurani, P.; Karuthapandian, S.; Muthuraj, V. Light assisted synthesis of hierarchically structured Cu/CdS nanorods with superior photocatalytic activity, stability and photocatalytic mechanism. Sep. Purif. Technol. 2017, 172, 192–201. [Google Scholar] [CrossRef]
- Shen, Q.; Xue, J.; Zhao, H.; Shao, M.; Liu, X.; Jia, H. The role of crystalline TiO2 nanoparticle in enhancing the photocatalytic and photoelectrocatalytic properties of CdS nanorods. J. Alloys Compd. 2017, 695, 1080–1087. [Google Scholar] [CrossRef]
- Fang, S.; Zhou, Y.; Zhou, M.; Li, Z.; Xu, S.; Yao, C. Facile synthesis of novel ZnFe2O4/CdS nanorods composites and its efficient photocatalytic reduction of Cr(VI) under visible-light irradiation. J. Ind. Eng. Chem. 2018, 58, 64–73. [Google Scholar] [CrossRef]
- Maji, S.K.; Dutta, A.K.; Dutta, S.; Srivastava, D.N.; Paul, P.; Mondal, A.; Adhikary, B. Single-Source Precursor Approach for the Preparation of CdS Nanoparticles and Their Photocatalytic and Intrinsic Peroxidase like Activity. Appl. Catal. B Environ. 2012, 126, 265–274. [Google Scholar] [CrossRef]
- Singh, S.; Khare, N. Magnetically Separable, CoFe2O4 Decorated CdS Nanorods for Enhanced Visible Light Driven Photocatalytic Activity. Mater. Lett. 2015, 161, 64–67. [Google Scholar] [CrossRef]
- Huo, P.; Zhou, M.; Tang, Y.; Liu, X.; Ma, C.; Yu, L.; Yan, Y. Incorporation of N–ZnO/CdS/Graphene oxide composite photocatalyst for enhanced photocatalytic activity under visible light. J. Alloys Compd. 2016, 670, 198–209. [Google Scholar] [CrossRef]
- Chen, F.; Jia, D.; Cao, Y.; Jin, X.; Liu, A. Facile synthesis of CdS nanorods with enhanced photocatalytic activity. Ceram. Int. 2015, 41, 14604–14609. [Google Scholar] [CrossRef]
- Jamble, S.N.; Ghoderao, K.P.; Kale, R.B. Studies on growth mechanism and physical properties of hydrothermally synthesized CdS with novel hierarchical superstructures and their photocatalytic activity. J. Phys. Chem. Solids 2018, 114, 109–120. [Google Scholar] [CrossRef]
- Bera, R.; Kundu, S.; Patra, A. 2D Hybrid Nanostructure of Reduced Graphene Oxide–CdS Nanosheet for Enhanced Photocatalysis. ACS Appl. Mater. Interfaces 2015, 7, 13251–13259. [Google Scholar] [CrossRef]
- Chen, Y.; Zhai, B.; Liang, Y.; Li, Y.; Li, J. Preparation of CdS/g-C3N4/MOF composite with enhanced visible-light photocatalytic activity for dye degradation. J. Solid State Chem. 2019, 274, 32–39. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Qin, Y.; Rao, J.; Chen, G.; Lv, C.; Liu, B. The synthesis of elegant hierarchical CdS via a facile hydrothermal method assisted by inorganic salt, with photocorrosion inhibition. CrystEngComm 2016, 18, 7523–7529. [Google Scholar] [CrossRef]
- Zhu, C.; Liu, C.; Fu, Y.; Gao, J.; Huang, H.; Liu, Y.; Kang, Z. Construction of CDs/CdS photocatalysts for stable and efficient hydrogen production in water and seawater. Appl. Catal. B Environ. 2019, 242, 178–185. [Google Scholar] [CrossRef]
- Kandy, M.M.; Gaikar, V.G. Enhanced photocatalytic reduction of CO2 using CdS/Mn2O3 nanocomposite photocatalysts on porous anodic alumina support with solar concentrators. Renew. Energy 2019, 139, 915–923. [Google Scholar] [CrossRef]
- Ke, X.; Dai, K.; Zhu, G.; Zhang, J.; Liang, C. In situ photochemical synthesis noble-metal-free NiS on CdS-diethylenetriamine nanosheets for boosting photocatalytic H2 production activity. Appl. Surf. Sci. 2019, 481, 669–677. [Google Scholar] [CrossRef]
- Mohanraj, V.; Jayaprakash, R.; Sangaiya, P.; Gopi, S.; Dilip, R.; Suresh, R. Study of photocatalytic activity and inherent property of surfactant (PVA) on CdS nanoparticles by ultrasonic wave irradiation method. Int. J. Hydrogen Energy 2017, 42, 28266–28277. [Google Scholar] [CrossRef]
- Lavand, A.B.; Malghe, Y.S. Visible light photocatalytic degradation of 4-chlorophenol using C/ZnO/CdS nanocomposite. J. Saudi Chem. Soc. 2015, 19, 471–478. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, Y.-X.; Zhang, W.-D. Carbon quantum dots-doped CdS microspheres with enhanced photocatalytic performance. J. Alloys Compd. 2013, 569, 102–110. [Google Scholar] [CrossRef]
- Saha, M.; Ghosh, S.; De, S.K. Nanoscale Kirkendall Effect Driven Au Decorated CdS/CdO Colloidal Nanocomposites for Efficient Hydrogen Evolution, Photocatalytic Dye Degradation and Cr (VI) Reduction. Catal. Today 2018, 340, 253–267. [Google Scholar] [CrossRef]
- Xiong, S.; Xi, B.; Qian, Y. CdS Hierarchical Nanostructures with Tunable Morphologies: Preparation and Photocatalytic Properties. J. Phys. Chem. C 2010, 114, 14029–14035. [Google Scholar] [CrossRef]
- Yang, X.; Cui, H.; Li, Y.; Qin, J.; Zhang, R.; Tang, H. Fabrication of Ag3PO4-Graphene Composites with Highly Efficient and Stable Visible Light Photocatalytic Performance. ACS Catal. 2013, 3, 363–369. [Google Scholar] [CrossRef]
- Liu, Y.; Fang, L.; Lu, H.; Li, Y.; Hu, C.; Yu, H. One-Pot Pyridine-Assisted Synthesis of Visible-Light-Driven Photocatalyst Ag/Ag3PO4. Appl. Catal. B Environ. 2012, 115, 245–252. [Google Scholar] [CrossRef]
- Yi, Z.; Ye, J.; Kikugawa, N.; Kako, T.; Ouyang, S.; Stuart-Williams, H.; Yang, H.; Cao, J.; Luo, W.; Li, Z.; et al. An Orthophosphate Semiconductor with Photooxidation Properties under Visible-Light Irradiation. Nat. Mater. 2010, 9, 559–564. [Google Scholar] [CrossRef]
- Chen, X.; Dai, Y.; Wang, X. Methods and Mechanism for Improvement of Photocatalytic Activity and Stability of Ag3PO4: A Review. J. Alloys Compd. 2015, 649, 910–932. [Google Scholar] [CrossRef]
- Wang, L.; Liu, J.; Wang, Y.; Zhang, X.; Duan, D.; Fan, C.; Wang, Y. Insight into the Enhanced Photocatalytic Performance of Ag3PO4 Modified Metastable Hexagonal WO3. Colloids Surfaces A Physicochem. Eng. Asp. 2018, 541, 145–153. [Google Scholar] [CrossRef]
- Zwara, J.; Grabowska, E.; Klimczuk, T.; Lisowski, W.; Zaleska-Medynska, A. Shape-Dependent Enhanced Photocatalytic Effect under Visible Light of Ag3PO4 Particles. J. Photochem. Photobiol. A Chem. 2018, 367, 240–252. [Google Scholar] [CrossRef]
- Hsieh, M.S.; Su, H.J.; Hsieh, P.L.; Chiang, Y.W.; Huang, M.H. Synthesis of Ag3PO4 Crystals with Tunable Shapes for Facet-Dependent Optical Property, Photocatalytic Activity, and Electrical Conductivity Examinations. ACS Appl. Mater. Interfaces 2017, 9, 39086–39093. [Google Scholar] [CrossRef]
- Guo, X.; Chen, C.; Yin, S.; Huang, L.; Qin, W. Controlled synthesis and photocatalytic properties of Ag3PO4 microcrystals. J. Alloys Compd. 2015, 619, 293–297. [Google Scholar] [CrossRef]
- Singh, A.; Baruah, A.; Katoch, V.; Vaghasiya, K.; Prakash, B.; Ganguli, A.K. Continuous Flow Synthesis of Ag3PO4 Nanoparticles with Greater Photostability and Photocatalytic Dye Degradation Efficiency. J. Photochem. Photobiol. A Chem. 2018, 364, 382–389. [Google Scholar] [CrossRef]
- Liu, L.; Hu, P.; Li, Y.; An, W.; Lu, J.; Cui, W. P3HT-Coated Ag3PO4 Core-Shell Structure for Enhanced Photocatalysis under Visible Light Irradiation. Appl. Surf. Sci. 2018, 466, 928–936. [Google Scholar] [CrossRef]
- Xie, Y.; Luo, S.; Huang, H.; Huang, Z.; Liu, Y.; Fang, M.; Wu, X.; Min, X. Construction of an Ag3PO4 Morphological Homojunction for Enhanced Photocatalytic Performance and Mechanism Investigation. Colloids Surf. A Physicochem. Eng. Asp. 2018, 546, 99–106. [Google Scholar] [CrossRef]
- Petala, A.; Spyrou, D.; Frontistis, Z.; Mantzavinos, D.; Kondarides, D.I. Immobilized Ag3PO4 photocatalyst for micro-pollutants removal in a continuous flow annular photoreactor. Catal. Today 2018, 328, 223–229. [Google Scholar] [CrossRef]
- Cui, X.; Zheng, Y.F.; Zhou, H.; Yin, H.Y.; Song, X.C. The effect of synthesis temperature on the morphologies and visible light photocatalytic performance of Ag3POJ. Taiwan Inst. Chem. Eng. 2016, 60, 328–334. [Google Scholar] [CrossRef]
- Song, X.; Li, R.; Xiang, M.; Hong, S.; Yao, K.; Huang, Y. Morphology and photodegradation performance of Ag3PO4 prepared by (NH4)3PO4, (NH4)2HPO4 and NH4H2PO. Ceram. Int. 2017, 43, 4692–4701. [Google Scholar] [CrossRef]
- Li, R.; Song, X.; Huang, Y.; Fang, Y.; Jia, M.; Ma, W. Visible-light photocatalytic degradation of azo dyes in water by Ag3PO4: An unusual dependency between adsorption and the degradation rate on pH value. J. Mol. Catal. A Chem. 2016, 421, 57–65. [Google Scholar] [CrossRef]
- Cruz-Filho, J.; Costa, T.; Lima, M.; Silva, L.; Santos, R.; Cavalcante, L.; Longo, E.; Luz, G. Effect of different synthesis methods on the morphology, optical behavior, and superior photocatalytic performances of Ag3PO4 sub-microcrystals using white-light-emitting diodes. J. Photochem. Photobiol. A Chem. 2019, 377, 14–25. [Google Scholar] [CrossRef]
- Zheng, B.; Wang, X.; Liu, C.; Tan, K.; Xie, Z.; Zheng, L. High-efficiently visible light-responsive photocatalysts: Ag3PO4 tetrahedral microcrystals with exposed {111} facets of high surface energy. J. Mater. Chem. A 2013, 1, 12635–12640. [Google Scholar] [CrossRef]
- Tang, C.; Liu, E.; Fan, J.; Hu, X.; Ma, Y.; Wan, J. A graphitic-C3N4-hybridized Ag3PO4 tetrahedron with reactive {111} facets to enhance the visible-light photocatalytic activity. RSC Adv. 2015, 5, 91979–91987. [Google Scholar] [CrossRef]
- Zhang, G.-Y.; Wei, X.-M.; Bai, X.; Liu, C.-M.; Wang, B.-Y.; Liu, J.-W. Ethanol–water ambient precipitation of {111} facets exposed Ag3PO4 tetrahedra and its hybrid with graphene oxide for outstanding photoactivity and stability. Inorg. Chem. Front. 2018, 5, 951–961. [Google Scholar] [CrossRef]
- Amornpitoksuk, P.; Intarasuwan, K.; Suwanboon, S.; Baltrusaitis, J. Effect of Phosphate Salts (Na3PO4, Na2HPO4, and NaH2PO4) on Ag3PO4 Morphology for Photocatalytic Dye Degradation under Visible Light and Toxicity of the Degraded Dye Products. Ind. Eng. Chem. Res. 2013, 52, 17369–17375. [Google Scholar] [CrossRef]
- Krungchanuchat, S.; Ekthammathat, N.; Phuruangrat, A.; Thongtem, S.; Thongtem, T. High UV-Visible Photocatalytic Activity of Ag3PO4 Dodecahedral Particles Synthesized by a Simple Hydrothermal Method. Mater. Lett. 2017, 201, 58–61. [Google Scholar] [CrossRef]
- Yan, T.; Zhang, H.; Liu, Y.; Guan, W.; Long, J.; Li, W.; You, J. Fabrication of Robust M/Ag3PO4 (M = Pt, Pd, Au) Schottky-Type Heterostructures for Improved Visible-Light Photocatalysis. RSC Adv. 2014, 4, 37220–37230. [Google Scholar] [CrossRef]
- Hewer, T.L.R.; Machado, B.C.; Freire, R.S.; Guardani, R. Ag3PO4 Sunlight-Induced Photocatalyst for Degradation of Phenol. RSC Adv. 2014, 4, 34674–34680. [Google Scholar] [CrossRef]
- Luo, L.; Li, Y.; Hou, J.; Yang, Y. Visible Photocatalysis and Photostability of Ag3PO4 Photocatalyst. Appl. Surf. Sci. 2014, 319, 332–338. [Google Scholar] [CrossRef]
- Zhou, T.; Zhang, G.; Yang, H.; Zhang, H.; Suo, R.; Xie, Y.; Liu, G. Fabrication of Ag3PO4/GO/NiFe2O4 Composites with Highly Efficient and Stable Visible-Light-Driven Photocatalytic Degradation of Rhodamine, B. RSC Adv. 2018, 8, 28179–28188. [Google Scholar] [CrossRef]
- Abroushan, E.; Farhadi, S.; Zabardasti, A. Ag3PO4/CoFe2O4 Magnetic Nanocomposite: Synthesis, Characterization and Applications in Catalytic Reduction of Nitrophenols and Sunlight-Assisted Photocatalytic Degradation of Organic Dye Pollutants. RSC Adv. 2017, 7, 18293–18304. [Google Scholar] [CrossRef]
- Ren, Y.; Li, H.; Yang, W.; Shi, D.; Wu, Q.; Zhao, Y.; Feng, C.; Liu, H.; Jiao, Q. Alkaline Ionic Liquids Immobilized on Protective Copolymers Coated Magnetic Nanoparticles: An Efficient and Magnetically Recyclable Catalyst for Knoevenagel Condensation. Ind. Eng. Chem. Res. 2019, 58, 2824–2834. [Google Scholar] [CrossRef]
- Li, J.; Cui, H.; Mu, D.; Liu, Y.; Guan, T.; Xia, Z.; Jiang, L.; Zuo, J.; Tan, C.; You, H. Synthesis and Characterization of RGO Decorated Cubic ZnTiO3 Rods for Solar Light-Induced Photodegradation of Rhodamine B. New J. Chem. 2019, 43, 3374–3382. [Google Scholar] [CrossRef]
- Surendar, T.; Kumar, S.; Shanker, V. Influence of La-Doping on Phase Transformation and Photocatalytic Properties of ZnTiO3 nanoparticles Synthesized via Modified Sol-Gel Method. Phys. Chem. Chem. Phys. 2013, 16, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Sharma, U.; Jeevanandam, P. Synthesis Temperature Dependent Morphological Evolution in Zinc Titanate Heteronanostructures and Their Application in Environmental Remediation. ChemistrySelect 2016, 1, 6382–6395. [Google Scholar] [CrossRef]
- Salavati-Niasari, M.; Soofivand, F.; Sobhani-Nasab, A.; Shakouri-Arani, M.; Yeganeh Faal, A.; Bagheri, S. Synthesis, Characterization, and Morphological Control of ZnTiO3 Nanoparticles through Sol-Gel Processes and Its Photocatalyst Application. Adv. Powder Technol. 2016, 27, 2066–2075. [Google Scholar] [CrossRef]
- Li, X.; Xiong, J.; Huang, J.; Feng, Z.; Luo, J. Novel g-C3N4/h′ZnTiO3-a′TiO2 direct Z-scheme heterojunction with significantly enhanced visible-light photocatalytic activity. J. Alloys Compd. 2018, 774, 768–778. [Google Scholar] [CrossRef]
- Kong, J.-Z.; Li, A.-D.; Zhai, H.; Li, H.; Yan, Q.-Y.; Ma, J.; Wu, D. Preparation, characterization and photocatalytic properties of ZnTiO3 powders. J. Hazard. Mater. 2009, 171, 918–923. [Google Scholar] [CrossRef]
- Perween, S.; Ranjan, A. Improved visible-light photocatalytic activity in ZnTiO3 nanopowder prepared by sol-electrospinning. Sol. Energy Mater. Sol. Cells 2017, 163, 148–156. [Google Scholar] [CrossRef]
- Ozturk, B.; Soylu, G.S.P. Promoting role of transition metal oxide on ZnTiO3–TiO2 nanocomposites for the photocatalytic activity under solar light irradiation. Ceram. Int. 2016, 42, 11184–11192. [Google Scholar] [CrossRef]
- Wattanawikkam, C.; Kansa-Ard, T.; Pecharapa, W. X-ray absorption spectroscopy analysis and photocatalytic behavior of ZnTiO3 nanoparticles doped with Co and Mn synthesized by sonochemical method. Appl. Surf. Sci. 2019, 474, 169–176. [Google Scholar] [CrossRef]
- Dutta, D.P.; Singh, A.; Tyagi, A.K. Ag Doped and Ag Dispersed Nano ZnTiO3: Improved Photocatalytic Organic Pollutant Degradation under Solar Irradiation and Antibacterial Activity. J. Environ. Chem. Eng. 2014, 2, 2177–2187. [Google Scholar] [CrossRef]
- Chi, Y.; Yuan, Q.; Hou, S.; Zhao, Z. Synthesis and Characterization of Mesoporous ZnTiO3 Rods via a Polyvinylpyrrolidone Assisted Sol-Gel Method. Ceram. Int. 2016, 42, 5094–5099. [Google Scholar] [CrossRef]
- Xin, Y.A.N.; Zhao, C.L.; Zhou, Y.L.; Wu, Z.J.; Yuan, J.M.; Li, W.S. Synthesis and characterization of ZnTiO3 with high photocatalytic activity. Trans. Nonferrous Met. Soc. China 2015, 25, 2272–2278. [Google Scholar] [CrossRef]
- Reddy, K.H.; Martha, S.; Parida, K.M. Erratic charge transfer dynamics of Au/ZnTiO3 nanocomposites under UV and visible light irradiation and their related photocatalytic activities. Nanoscale 2018, 10, 18540–18554. [Google Scholar] [CrossRef]
- Yang, J.; Akbarzadeh, J.; Maurer, C.; Peterlik, H.; Schubert, U. Sol–gel synthesis of ZnTiO3 using a single-source precursor based on p-carboxybenzaldehyde oxime as a linker. J. Mater. Chem. 2012, 22, 24034–24041. [Google Scholar] [CrossRef]
- Wu, H.; Min, Y.; Zhang, Q.; Li, W.; Yuan, J.; Wu, Z.; Wang, S. Low-temperature synthesis of mesoporous ZnTiO3–graphene composite for the removal of norfloxacin in aqueous solution. RSC Adv. 2016, 6, 103822–103829. [Google Scholar] [CrossRef]
- Gayathri, S.; Jayabal, P.; Kottaisamy, M.; Ramakrishnan, V. Synthesis of the graphene-ZnTiO3 nanocomposite for solar light assisted photodegradation of methylene blue. J. Phys. D Appl. Phys. 2015, 48, 415305. [Google Scholar] [CrossRef]
- Eskandarloo, H.; Badiei, A.; Behnajady, M.A.; Tavakoli, A.; Ziarani, G.M. Ultrasonic-assisted synthesis of Ce doped cubic–hexagonal ZnTiO3 with highly efficient sonocatalytic activity. Ultrason. Sonochem. 2016, 29, 258–269. [Google Scholar] [CrossRef]
- Yu, C.; Chen, F.; Zeng, D.; Xie, Y.; Zhou, W.; Liu, Z.; Wei, L.; Yang, K.; Li, D. A facile phase transformation strategy for fabrication of novel Z-scheme ternary heterojunctions with efficient photocatalytic properties. Nanoscale 2019, 11, 7720–7733. [Google Scholar] [CrossRef]
- Raza, W.; Haque, M.; Muneer, M. Synthesis of visible light driven ZnO: Characterization and photocatalytic performance. Appl. Surf. Sci. 2014, 322, 215–224. [Google Scholar] [CrossRef]
- Guo, Q.; Zhou, C.; Ma, Z.; Yang, X. Fundamentals of TiO2 Photocatalysis: Concepts, Mechanisms, and Challenges. Adv. Mater. 2019, 31, 1901997. [Google Scholar] [CrossRef] [PubMed]
- Mirzaeifard, Z.; Shariatinia, Z.; Jourshabani, M.; Rezaei Darvishi, S.M. ZnO Photocatalyst Revisited: Effective Photocatalytic Degradation of Emerging Contaminants Using S-Doped ZnO Nanoparticles under Visible Light Radiation. Ind. Eng. Chem. Res. 2020, 59, 15894–15911. [Google Scholar] [CrossRef]
- Yang, X.; Ma, J.; Wang, T.; Wang, B.; Meng, D.; Wang, Y. Synthesis, growth mechanism and photocatalytic property of CdS with different kinds of surfactants. New J. Chem. 2019, 43, 10126–10133. [Google Scholar] [CrossRef]
- Santos, R.K.; Martins, T.A.; Silva, G.N.; Conceição, M.V.S.; Nogueira, I.C.; Longo, E.; Botelho, G. Ag3PO4/NiO Composites with Enhanced Photocatalytic Activity under Visible Light. ACS Omega 2020, 5, 21651–21661. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Lu, S.; He, L.; Zhu, X.; Feng, W.; Zhang, W. Preparation, Characterization of ZnTiO3/ZnO Composite Materials and Their Photocatalytic Performance. Nanomaterials 2022, 12, 1345. [Google Scholar] [CrossRef]
- Esrafili, A.; Salimi, M.; Jafari, A.J.; Sobhi, H.R.; Gholami, M.; Kalantary, R.R. Pt-based TiO2 photocatalytic systems: A systematic review. J. Mol. Liq. 2022, 352, 118685. [Google Scholar] [CrossRef]
- Nah, Y.-C.; Paramasivam, I.; Schmuki, P. Doped TiO2 and TiO2 Nanotubes: Synthesis and Applications. ChemPhysChem 2010, 11, 2698–2713. [Google Scholar] [CrossRef]
- Vinayagam, R.; Pai, S.; Varadavenkatesan, T.; Pugazhendhi, A.; Selvaraj, R. Characterization and photocatalytic activity of ZnO nanoflowers synthesized using Bridelia retusa leaf extract. Appl. Nanosci. 2021, 1–10. [Google Scholar] [CrossRef]
- Morales, M.; Fernández-Cervantes, I.; Agustín-Serrano, R.; Ruíz-Salgado, S.; Sampedro, M.; Varela-Caselis, J.; Portillo, R.; Rubio, E. AgPO microcrystals with complex polyhedral morphologies diversity obtained by microwave-hydrothermal synthesis for MB degradation under sunlight. Results Phys. 2018, 12, 1344–1356. [Google Scholar] [CrossRef]
- Geng, T.; Zhang, S.; Chen, Y. Study on the synthesis and photocatalysis of Ag3PO4 polyhedral microcrystals. Bull. Mater. Sci. 2020, 43, 223. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Ji, Y.; Bian, R.; Li, J.; Zhang, X.; Tian, J.; Yang, Q.; Shi, F. Ti3C2 MXene coupled with CdS nanoflowers as 2D/3D heterostructures for enhanced photocatalytic hydrogen production activity. Int. J. Hydrogen Energy 2022, 47, 22045–22053. [Google Scholar] [CrossRef]
- Chuaicham, C.; Karthikeyan, S.; Song, J.T.; Ishihara, T.; Ohtani, B.; Sasaki, K. Importance of ZnTiO3 Phase in ZnTi-Mixed Metal Oxide Photocatalysts Derived from Layered Double Hydroxide. ACS Appl. Mater. Interfaces 2020, 12, 9169–9180. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wen, C.; Hodgson, P.; Li, Y. Biocompatibility of TiO2 nanotubes with different topographies. J. Biomed. Mater. Res. Part A 2013, 102, 743–751. [Google Scholar] [CrossRef]
- Fontes, A.C.C.A.; Sopchenski, L.; Laurindo, C.A.H.; Torres, R.D.; Popat, K.C.; Soares, P. Annealing Temperature Effect on Tribocorrosion and Biocompatibility Properties of TiO2 Nanotubes. J. Bio- Tribo-Corros. 2020, 6, 64. [Google Scholar] [CrossRef]
- Kunrath, M.F.; Hubler, R.; Shinkai, R.S.A.; Teixeira, E.R. Application of TiO2 Nanotubes as a Drug Delivery System for Biomedical Implants: A Critical Overview. ChemistrySelect 2018, 3, 11180–11189. [Google Scholar] [CrossRef]
- Ray, P.G.; Biswas, S.; Roy, T.; Ghosh, S.; Majumder, D.; Basak, P.; Roy, S.; Dhara, S. Sonication Assisted Hierarchical Decoration of Ag-NP on Zinc Oxide Nanoflower Impregnated Eggshell Membrane: Evaluation of Antibacterial Activity and in Vitro Cytocompatibility. ACS Sustain. Chem. Eng. 2019, 7, 13717–13733. [Google Scholar] [CrossRef]
- Khan, M.S.; Ameer, H.; Ali, A.; Zhu, W.; Li, X.; Yang, L.; Wang, H.; Feng, R.; Wei, Q. Label-free electrochemiluminescent immunosensor for prostate specific antigen ultrasensitive detection based on novel luminophore Ag3PO4 decorated GO. J. Electroanal. Chem. 2019, 847, 113266. [Google Scholar] [CrossRef]
- Bhavsar, K.; Labhane, P.; Dhake, R.; Sonawane, G. Solvothermal synthesis of activated carbon loaded CdS nanoflowers: Boosted photodegradation of dye by adsorption and photocatalysis synergy. Chem. Phys. Lett. 2020, 744, 137202. [Google Scholar] [CrossRef]
- Al-Jawad, S.M.H.; Imran, N.J.; Aboud, K.H. Synthesis and characterization of Mn:CdS nanoflower thin films prepared by hydrothermal method for photocatalytic activity. J. Sol-Gel Sci. Technol. 2021, 100, 423–439. [Google Scholar] [CrossRef]
- Mathew, A.M.; Chukwuike, V.; Venkatesan, K.; Raveendran, S.; Barik, R.C.; Pattanayak, D.K. Zinc oxide decorated titania nanostructured layer over Ti metal as a biocompatible and antimicrobial surface for biomedical application. Surf. Interfaces 2022, 33, 102275. [Google Scholar] [CrossRef]
- Zhang, M.; Gong, Z.; Zhang, J.; Cheng, H.; Chen, J.; Zeng, Y.; Zhu, Z.; Wan, Y. Engineered Zinc Titanate Coatings on the Titanium Surface with Enhanced Antitumor Properties and Biocompatibility. ACS Biomater. Sci. Eng. 2019, 5, 5935–5946. [Google Scholar] [CrossRef] [PubMed]
- Abirami, R.; Senthil, T.; Keerthana, S.; Yuvakkumar, R.; Ravi, G.; Pannipara, M.; Al-Sehemi, A.G. An approach to enhance the photocatalytic activity of ZnTiO3. Ceram. Int. 2021, 47, 18122–18131. [Google Scholar] [CrossRef]
- Grilli, M.L. Metal Oxides. Metals 2020, 10, 820. [Google Scholar] [CrossRef]
- Gautam, S.; Agrawal, H.; Thakur, M.; Akbari, A.; Sharda, H.; Kaur, R.; Amini, M. Metal oxides and metal organic frameworks for the photocatalytic degradation: A review. J. Environ. Chem. Eng. 2020, 8, 103726. [Google Scholar] [CrossRef]
- Bel Hadjltaief, H.; Ben Zina, M.; Galvez, M.E.; Da Costa, P. Photocatalytic Degradation of Methyl Green Dye in Aqueous Solution over Natural Clay-Supported ZnO-TiO2 Catalysts. J. Photochem. Photobiol. A Chem. 2016, 315, 25–33. [Google Scholar] [CrossRef]
- Theerthagiri, J.; Chandrasekaran, S.; Salla, S.; Elakkiya, V.; Senthil, R.A.; Nithyadharseni, P.; Maiyalagan, T.; Micheal, K.; Ayeshamariam, A.; Arasu, M.V.; et al. Recent Developments of Metal Oxide Based Heterostructures for Photocatalytic Applications towards Environmental Remediation. J. Solid State Chem. 2018, 267, 35–52. [Google Scholar] [CrossRef]
- Yumashev, A.; Ślusarczyk, B.; Kondrashev, S.; Mikhaylov, A. Global Indicators of Sustainable Development: Evaluation of the Influence of the Human Development Index on Consumption and Quality of Energy. Energies 2020, 13, 2768. [Google Scholar] [CrossRef]
- Huang, T.; Crews, J.B. Wastewater Purification with Nanoparticle Treated Bed. PCT/US2009/040597, 15 April 2009. [Google Scholar]
- Shalabayev, Z.; Baláž, M.; Khan, N.; Nurlan, Y.; Augustyniak, A.; Daneu, N.; Tatykayev, B.; Dutková, E.; Burashev, G.; Casas-Luna, M.; et al. Sustainable Synthesis of Cadmium Sulfide, with Applicability in Photocatalysis, Hydrogen Production, and as an Antibacterial Agent, Using Two Mechanochemical Protocols. Nanomaterials 2022, 12, 1250. [Google Scholar] [CrossRef]
- Li, X.; Xu, P.; Chen, M.; Zeng, G.; Wang, D.; Chen, F.; Tang, W.; Chen, C.; Zhang, C.; Tan, X. Application of Silver Phosphate-Based Photocatalysts: Barriers and Solutions. Chem. Eng. J. 2019, 366, 339–357. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganguli, A.K.; Kunde, G.B.; Raza, W.; Kumar, S.; Yadav, P. Assessment of Performance of Photocatalytic Nanostructured Materials with Varied Morphology Based on Reaction Conditions. Molecules 2022, 27, 7778. https://doi.org/10.3390/molecules27227778
Ganguli AK, Kunde GB, Raza W, Kumar S, Yadav P. Assessment of Performance of Photocatalytic Nanostructured Materials with Varied Morphology Based on Reaction Conditions. Molecules. 2022; 27(22):7778. https://doi.org/10.3390/molecules27227778
Chicago/Turabian StyleGanguli, Ashok Kumar, Gajanan B. Kunde, Waseem Raza, Sandeep Kumar, and Priyanka Yadav. 2022. "Assessment of Performance of Photocatalytic Nanostructured Materials with Varied Morphology Based on Reaction Conditions" Molecules 27, no. 22: 7778. https://doi.org/10.3390/molecules27227778
APA StyleGanguli, A. K., Kunde, G. B., Raza, W., Kumar, S., & Yadav, P. (2022). Assessment of Performance of Photocatalytic Nanostructured Materials with Varied Morphology Based on Reaction Conditions. Molecules, 27(22), 7778. https://doi.org/10.3390/molecules27227778







