Griseofulvin: An Updated Overview of Old and Current Knowledge
Abstract
1. Introduction
2. Discovery of Griseofulvin and Its Biosynthetic Gene Cluster in Fungi
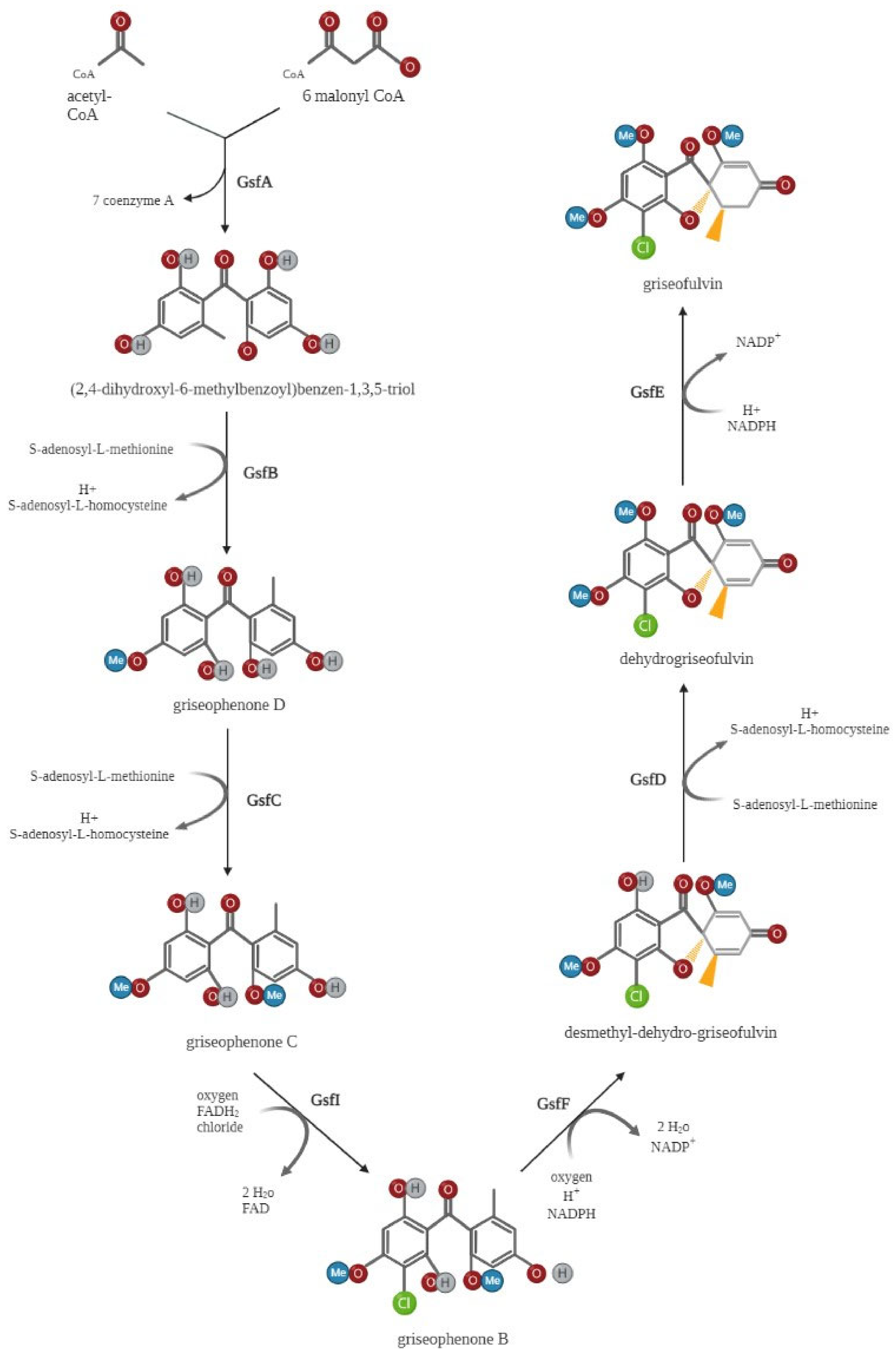
3. Biosynthesis of Griseofulvin
3.1. Gene Cluster Evolution Hypothesis
3.2. Griseofulvin Biosynthetic Gene Cluster
3.3. Genetic Regulation of Griseofulvin Biosynthesis
3.4. Environmental Factors in Producing Griseofulvin
4. Physiochemical Properties of Griseofulvin
5. Medical Applications
5.1. Antifungal Application
5.2. Non-fungal Inflammatory Diseases
5.3. Cardiovascular Applications
5.4. Antitumor Applications
5.5. Antiviral Applications
6. Pharmacokinetics of Griseofulvin
7. Characterized Interactions between Griseofulvin and Cellular Components
8. Changes in Keratins 8 and 18 in Griseofulvin-Induced Liver Injury
9. Repurposing of Griseofulvin
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Whalley, A. The xylariaceous way of life. Mycol. Res. 1996, 100, 897–922. [Google Scholar] [CrossRef]
- Mead, M.E.; Raja, H.A.; Steenwyk, J.L.; Knowles, S.L.; Oberlies, N.H.; Rokas, A. Draft genome sequence of the griseofulvin-producing fungus xylaria flabelliformis strain G536. Microbiol. Resour. Announc. 2019, 8, e00890-19. [Google Scholar] [CrossRef]
- Lee, H.B.; Mun, H.Y.; Nguyen, T.T.T.; Kim, J.-C.; Stone, J.K. Abieticola koreana Gen. Et Sp. Nov.; A griseofulvin-producing endophytic xylariaceous ascomycete from korea. Mycotaxon 2016, 131, 749–764. [Google Scholar] [CrossRef]
- Ribeiro, A.I.; Costa, E.S.; Thomasi, S.S.; Brandão, D.F.R.; Vieira, P.C.; Fernandes, J.O.B.; Forim, M.R.; Ferreira, A.G.; Pascholati, S.R.F.; Gusmão, L.F.P. Biological and chemical control of sclerotinia sclerotiorum using stachybotrys levispora and its secondary metabolite griseofulvin. J. Agric. Food Chem. 2018, 66, 7627–7632. [Google Scholar] [CrossRef]
- Dayan, F.E.; Cantrell, C.L.; Duke, S.O. Natural products in crop protection. Bioorganic Med. Chem. 2009, 17, 4022–4034. [Google Scholar] [CrossRef]
- Oxford, A.E.; Raistrick, H.; Simonart, P. Studies in the biochemistry of micro-organisms: Griseofulvin, c17h17o6cl, a metabolic product of penicillium griseo-fulvum dierckx. Biochem. J. 1939, 33, 240. [Google Scholar] [CrossRef]
- Lambert, D.R.; Siegle, R.J.; Camisa, C. Griseofulvin and ketoconazole in the treatment of dermatophyte infections. Int. J. Dermatol. 1989, 28, 300–304. [Google Scholar] [CrossRef]
- Rebacz, B.; Larsen, T.O.; Clausen, M.H.; Rønnest, M.H.; Löffler, H.; Ho, A.D.; Krämer, A. Identification of griseofulvin as an inhibitor of centrosomal clustering in a phenotype-based screen. Cancer Res. 2007, 67, 6342–6350. [Google Scholar] [CrossRef]
- Jin, H.; Yamashita, A.; Maekawa, S.; Yang, P.; He, L.; Takayanagi, S.; Wakita, T.; Sakamoto, N.; Enomoto, N.; Ito, M. Griseofulvin, an oral antifungal agent, suppresses hepatitis c virus replication In Vitro. Hepatol. Res. 2008, 38, 909–918. [Google Scholar]
- Rubin, A.A. Coronary vascular effects of griseofulvin. JAMA 1963, 185, 971–972. [Google Scholar] [CrossRef]
- Aldinger, E.E. Cardiovascular effects of griseofulvin. Circ. Res. 1968, 22, 589–593. [Google Scholar] [CrossRef]
- Aris, P.; Mohamadzadeh, M.; Wei, Y.; Xia, X. In Silico molecular dynamics of griseofulvin and its derivatives revealed potential therapeutic applications for COVID-19. Int. J. Mol. Sci. 2022, 23, 6889. [Google Scholar] [CrossRef]
- Atlas, S.A. The Renin-angiotensin aldosterone system: Pathophysiological role and pharmacologic inhibition. J. Manag. Care Pharm. 2007, 13, 9–20. [Google Scholar] [CrossRef]
- Brian, P.; Hemming, H. Production of antifungal and antibacterial substances by fungi; preliminary examination of 166 strains of fungi imperfecti. Microbiology 1947, 1, 158–167. [Google Scholar] [CrossRef]
- Grove, J.F.; Macmillan, J.; Mulholland, T.; Rogers, M.T. Griseofulvin. Part I. J. Chem. Soc. 1952, 759, 3949–3958. [Google Scholar] [CrossRef]
- Chooi, Y.-H.; Cacho, R.; Tang, Y. Identification of the viridicatumtoxin and griseofulvin gene clusters from Penicillium aethiopicum. Chem. Biol. 2010, 17, 483–494. [Google Scholar] [CrossRef]
- Tang, H.-Y.; Zhang, Q.; Li, H.; Gao, J.-M. Antimicrobial and allelopathic metabolites produced by Penicillium brasilianum. Nat. Prod. Res. 2015, 29, 345–348. [Google Scholar] [CrossRef]
- Roullier, C.; Bertrand, S.; Blanchet, E.; Peigné, M.; Robiou Du Pont, T.; Guitton, Y.; Pouchus, Y.F.; Grovel, O. Time dependency of chemodiversity and biosynthetic pathways: An lc-ms metabolomic study of marine-sourced Penicillium. Mar. Drugs 2016, 14, 103. [Google Scholar] [CrossRef]
- Jadulco, R.; Edrada, R.A.; Ebel, R.; Berg, A.; Schaumann, K.; Wray, V.; Steube, K.; Proksch, P. New communesin derivatives from the fungus Penicillium sp. derived from the mediterranean sponge Axinella verrucosa. J. Nat. Prod. 2004, 67, 78–81. [Google Scholar] [CrossRef]
- Petit, K.; Mondeguer, F.; Roquebert, M.; Biard, J.; Pouchus, Y. Detection of griseofulvin in a marine strain of Penicillium waksmanii by ion trap mass spectrometry. J. Microbiol. Methods 2004, 58, 59–65. [Google Scholar] [CrossRef]
- Xue, C.; Li, T.; Deng, Z.; Fu, H.; Lin, W. Janthinolide Ab, Two New 2, 5-piperazinedione derivatives from the endophytic Penicillium janthinellum isolated from the soft coral dendronephthya sp. Die Pharm. 2006, 61, 1041–1044. [Google Scholar]
- Clarke, S.; Mckenzie, M. Penicillium sclerotigenum: A new source of griseofulvin. Nature 1967, 213, 504–505. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, H.-B.; Frisvad, J.C.; Samson, R.A. Penicillium persicinum, a new griseofulvin, chrysogine and roquefortine c producing species from qinghai province, china. Antonie Van Leeuwenhoek 2004, 86, 173–179. [Google Scholar] [CrossRef]
- Nielsen, J.C.; Grijseels, S.; Prigent, S.; Ji, B.; Dainat, J.; Nielsen, K.F.; Frisvad, J.C.; Workman, M.; Nielsen, J. Global analysis of biosynthetic gene clusters reveals vast potential of secondary metabolite production in Penicillium species. Nat. Microbiol. 2017, 2, 17044. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Samson, R.A. Polyphasic taxonomy of Penicillium subgenus Penicillium. a guide to identification of food and air-borne terverticillate penicillia and their mycotoxins. Stud. Mycol. 2004, 49, 1–174. [Google Scholar]
- Larsen, T.O.; Smedsgaard, J.; Nielsen, K.F.; Hansen, M.E.; Frisvad, J.C. Phenotypic taxonomy and metabolite profiling in microbial drug discovery. Nat. Prod. Rep. 2005, 22, 672–695. [Google Scholar] [CrossRef]
- Samson, R.A.; Pitt, J.I. Modern Concepts in Penicillium and Aspergillus Classification; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Allawi, M.Y.A.; Al-Taee, W.S.Q. Investigation of a new local isolate of Penicillium lanosocoeruleum that produces the antifungal griseofulvin. Pak. J. Med. Health Sci. 2022, 16, 380. [Google Scholar]
- Park, J.-H.; Choi, G.-J.; Lee, S.-W.; Lee, H.-B.; Kim, K.-M.; Jung, H.-S.; Jang, K.-S.; Cho, K.-Y.; Kim, J.-C. Griseofulvin from xylaria sp. strain f0010, an endophytic fungus of abies holophylla and its antifungal activity against plant pathogenic fungi. J. Microbiol. Biotechnol. 2005, 15, 112–117. [Google Scholar]
- Petrini, O. Fungal endophytes of tree leaves. In Microbial Ecology of Leaves; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Grove, J.F.; Macmillan, J.; Mulholland, T.; Rogers, M.T. Griseofulvin. Part IV. Structure. J. Chem. Soc. 1952, 762, 3977–3987. [Google Scholar] [CrossRef]
- Birch, A.; Massy-Westropp, R.; Rickards, R.; Smith, H. Studies in relation to biosynthesis. Part XIII. Griseofulvin. J. Chem. Soc. 1958, 66, 360–365. [Google Scholar] [CrossRef]
- Birch, A.; Donovan, F. Studies In relation to biosynthesis. I. Some possible routes to derivatives of orcinol and phloroglucinol. Aust. J. Chem. 1953, 6, 360–368. [Google Scholar] [CrossRef]
- Simpson, T.J.; Holker, J.S. 13c-Nmr studies on griseofulvin biosynthesis and acetate metabolism in penicillium patulum. Phytochemistry 1977, 16, 229–233. [Google Scholar] [CrossRef]
- Nirmala, K.; Sakegowda, D.; Duax, W.L. Crystal structure of griseofulvin. J. Crystallogr. Spectrosc. Res. 1982, 12, 415–423. [Google Scholar]
- Brown, W.; Sim, G. Fungal metabolites. Part I. The stereochemistry of griseofulvin: X-ray analysis of 5-bromogriseofulvin. J. Chem. Soc. 1963, 193, 1050–1059. [Google Scholar] [CrossRef]
- Levine, S.G.; Hicks, R.E. The conformation of griseofulvin. Application of an nmr shift reagent. Tetrahedron Lett. 1971, 12, 311–314. [Google Scholar] [CrossRef]
- Aris, P.; Yan, L.; Wei, Y.; Chang, Y.; Shi, B.; Xia, X. Conservation of griseofulvin genes in the gsf gene cluster among fungal genomes. G3 2021, 2, jkab399. [Google Scholar] [CrossRef]
- Cimermancic, P.; Medema, M.H.; Claesen, J.; Kurita, K.; Brown, L.C.W.; Mavrommatis, K.; Pati, A.; Godfrey, P.A.; Koehrsen, M.; Clardy, J.; et al. Insights into Secondary Metabolism from a Global Analysis of Prokaryotic Biosynthetic Gene Clusters. Cell 2014, 158, 412–421. [Google Scholar] [CrossRef]
- Slot, J.C.; Rokas, A. Horizontal transfer of a large and highly toxic secondary metabolic gene cluster between fungi. Curr. Biol. 2011, 21, 134–139. [Google Scholar] [CrossRef]
- Dobrindt, U.; Hochhut, B.; Hentschel, U.; Hacker, J. Genomic Islands in Pathogenic and Environmental Microorganisms. Nat. Rev. Microbiol. 2004, 2, 414–424. [Google Scholar] [CrossRef]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal Secondary Metabolism—From Biochemistry to Genomics. Nat. Rev. Microbiol. 2005, 3, 937–947. [Google Scholar] [CrossRef]
- Wisecaver, J.H.; Slot, J.C.; Rokas, A. The Evolution of Fungal Metabolic Pathways. PLoS Genet. 2014, 10, e1004816. [Google Scholar] [CrossRef]
- Cacho, R.A.; Chooi, Y.-H.; Zhou, H.; Tang, Y. Complexity generation in fungal polyketide biosynthesis: A spirocycle-forming p450 in the concise pathway to the antifungal drug griseofulvin. Acs Chem. Biol. 2013, 8, 2322–2330. [Google Scholar] [CrossRef]
- Banani, H.; Marcet-Houben, M.; Ballester, A.-R.; Abbruscato, P.; González-Candelas, L.; Gabaldón, T.; Spadaro, D. Genome sequencing and secondary metabolism of the postharvest pathogen penicillium griseofulvum. BMC Genom. 2016, 17, 19. [Google Scholar] [CrossRef]
- Valente, S.; Cometto, A.; Piombo, E.; Meloni, G.R.; Ballester, A.-R.; González-Candelas, L.; Spadaro, D. Elaborated regulation of griseofulvin biosynthesis in penicillium griseofulvum and its role on conidiation and virulence. Int. J. Food Microbiol. 2020, 328, 108687. [Google Scholar] [CrossRef]
- Wright, J.M. The production of antibiotics in soil: II. Production of griseofulvin by penicillium nigricans. Ann. Appl. Biol. 1955, 43, 288–296. [Google Scholar] [CrossRef]
- Soloveva, N.; Malkov, M. Kil® N, Gi; Golubeva, La: Role of nitrogen nutrition in the cultivation of penicillium nigricans producing griseofulvin on a synthetic medium. Antibiot 1972, 9, 104–106. [Google Scholar]
- Rogal, I.; Malkov, M. Effect Of The cultivation temperature on the dynamics of the atp and adp levels and on the growth of p. nigricans thom. strains in the presence of various carbon sources. Antibiotiki 1978, 23, 971. [Google Scholar]
- Dasu, V.V.; Panda, T. Studies on production of griseofulvin. Bioprocess Eng. 1999, 21, 489–495. [Google Scholar] [CrossRef]
- Alan, R.; Rodger, C.; Primrose, F.T. Production of Griseofulvin in Low Nitrogen Level Medium. U.S. Patent 2843527A, 15 July 1958. [Google Scholar]
- Niu, G.; Tan, H. Biosynthesis and Regulation of Secondary Metabolites in Microorganisms. Sci China Life Sci. 2013, 56, 581–583. [Google Scholar] [CrossRef][Green Version]
- Vining, L. Functions of Secondary Metabolites. Annu. Rev. Microbiol. 1990, 44, 395–427. [Google Scholar] [CrossRef]
- Hurst, L. Genetics and the understanding of selection. Nat. Rev. Genet. 2009, 10, 83–93. [Google Scholar] [CrossRef]
- Kimura, M. The Neutral Theory of Molecular Evolution and the World View of the Neutralists. Genome 1989, 31, 24–31. [Google Scholar] [CrossRef]
- Gentles, J. Experimental ringworm in guinea pigs: Oral treatment with griseofulvin. Nature 1958, 182, 476–477. [Google Scholar] [CrossRef]
- Gupta, A.K.; Adam, P.; Dlova, N.; Lynde, C.W.; Hofstader, S.; Morar, N.; Aboobaker, J.; Summerbell, R.C. Therapeutic options for the treatment of tinea capitis caused by trichophyton species: Griseofulvin versus the new oral antifungal agents, terbinafine, itraconazole, and fluconazole. Pediatr. Dermatol. 2001, 18, 433–438. [Google Scholar] [CrossRef]
- Sehgal, V.; Bikhchandani, R.; Koranne, R.; Nayar, M.; Saxena, H. Histopathological Evaluation of griseofulvin therapy in lichen planus. Dermatology 1980, 161, 22–27. [Google Scholar] [CrossRef]
- Tamaki, K.; Yasaka, N.; Osada, A.; Shibagaki, N.; Furue, M. Successful Treatment of pigmented purpuric dermatosis with griseoiulvin. Br. J. Dermatol. 1995, 132, 159–160. [Google Scholar] [CrossRef]
- Cohen, A.; Goldman, J.; Daniels, R.; Kanenson, W. Treatment of shoulder-hand syndrome with griseofulvin. J. Am. Med. Assoc. 1960, 173, 542–543. [Google Scholar] [CrossRef]
- Serre, H.; Simon, L. Action Therapeutique inattendue en rhumatologie dun antibiotique antifongique-la griseofulvine. Presse Med. 1962, 70, 2263. [Google Scholar]
- Rathinasamy, K.; Jindal, B.; Asthana, J.; Singh, P.; Balaji, P.V.; Panda, D. Griseofulvin stabilizes microtubule dynamics, activates p53 and inhibits the proliferation of mcf-7 cells synergistically with vinblastine. BMC Cancer 2010, 10, 213. [Google Scholar] [CrossRef]
- Livingston, W.A. Griseofulvin in treatment of zoster. Arch. Dermatol. 1965, 92, 761. [Google Scholar] [CrossRef]
- Crounse, R. Effective Use of Griseofulvin. Arch. Dermatol. Res. 1963, 87, 176–178. [Google Scholar] [CrossRef]
- Ong, S.G.M.; Ming, L.C.; Lee, K.S.; Yuen, K.H. Influence of the encapsulation efficiency and size of liposome on the oral bioavailability of griseofulvin-loaded liposomes. Pharmaceutics 2016, 8, 25. [Google Scholar] [CrossRef]
- Lin, C.; Symchowicz, S. Absorption, Distribution, Metabolism, and Excretion of Griseofulvin in Man and Animals. Drug Metab. Rev. 1975, 4, 75–95. [Google Scholar] [CrossRef] [PubMed]
- Bedford, C.; Busfield, D.; Child, K.J.; Mac, G.; Sutherland, P.; Tomich, E.G. Studies on the biological disposition of griseofulvin, an oral antifungal agent. AMA Arch. Dermatol. 1960, 81, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Araujo, O.E.; Flowers, F.P.; King, M.M. Griseofulvin: A new look at an old drug. DICP 1990, 24, 851–854. [Google Scholar] [CrossRef]
- Roth, J.; Frank, J. Griseofulvin. Ann. N. Y. Acad Sci. 1960, 89, 247–253. [Google Scholar] [CrossRef]
- Develoux, M.G. Paper presented at the Annales de dermatologie et de venereologie. Ann. Dermatol. Venereol. 2001, 128, 910–989. [Google Scholar]
- Gull, K.; Trinci, A.P.J. Griseofulvin Inhibits Fungal Mitosis. Nature 1973, 244, 292–294. [Google Scholar] [CrossRef]
- Panda, D.; Rathinasamy, K.; Santra, M.K.; Wilson, L. Kinetic suppression of microtubule dynamic instability by griseofulvin: Implications for its possible use in the treatment of cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 9878–9883. [Google Scholar] [CrossRef]
- Uen, Y.H.; Liu, D.Z.; Weng, M.S.; Ho, Y.S.; Lin, S.Y. Nf-Κb pathway is involved in griseofulvin-induced g2/m arrest and apoptosis in hl-60 cells. J. Cell. Biochem. 2007, 101, 1165–1175. [Google Scholar] [CrossRef]
- Zomorodian, K.; Uthman, U.; Tarazooie, B.; Rezaie, S. The effect of griseofulvin on the gene regulation of β-tubulin in the dermatophyte pathogen trichophyton rubrum. J. Infect. Chemother. 2007, 13, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Asahina, A.; Tada, Y.; Nakamura, K.; Tamaki, K. Colchicine and griseofulvin inhibit VCAM-1 expression on human vascular endothelial cells—Evidence for the association of VCAM-1 expression with microtubules. J. Dermatol. Sci. 2001, 25, 1–9. [Google Scholar] [CrossRef]
- Chiprut, R.O.; Viteri, A.; Jamroz, C.; Dyck, W.P. Intrahepatic cholestasis after griseofulvin administration. Gastroenterology 1976, 70, 1141–1143. [Google Scholar] [CrossRef]
- Watanabe, M.; Akagi, S.; Kohge, N.; Uchida, Y.; Nguyen, T.; Hirakawa, K.; Fukumoto, S. Laparoscopy of griseofulvin-induced liver injury presenting a wide depression. Endoscopy 1994, 26, 514–515. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, H.J. Hepatotoxicity: The Adverse Effects of Drugs and Other Chemicals on the Liver; Lippincott: Philadelphia, PA, USA, 1999. [Google Scholar]
- Moseley, R.H. Hepatotoxicity of antimicrobials and antifungal agents. In Drug-Induced Liver Disease, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Zatloukal, K.; French, S.W.; Stumptner, C.; Strnad, P.; Harada, M.; Toivola, D.M.; Cadrin, M.; Omary, M.B. From mallory to mallory–denk bodies: What, how and why? Exp. Cell Res. 2007, 313, 2033–2049. [Google Scholar] [CrossRef]
- Fortier, A.-M.; Riopel, K.; Désaulniers, M.; Cadrin, M. Novel insights into changes in biochemical properties of keratins 8 and 18 in griseofulvin-induced toxic liver injury. Exp. Mol. Pathol. 2010, 89, 117–125. [Google Scholar] [CrossRef]
- Fortier, A.-M.; Asselin, E.; Cadrin, M. Keratin 8 and 18 loss in epithelial cancer cells increases collective cell migration and cisplatin sensitivity through claudin1 up-regulation. J. Biol. Chem. 2013, 288, 11555–11571. [Google Scholar] [CrossRef]
- Golob-Schwarzl, N.; Bettermann, K.; Mehta, A.K.; Kessler, S.M.; Unterluggauer, J.; Krassnig, S.; Kojima, K.; Chen, X.; Hoshida, Y.; Bardeesy, N.M. High keratin 8/18 ratio predicts aggressive hepatocellular cancer phenotype. Transl. Oncol. 2019, 12, 256–268. [Google Scholar] [CrossRef]
- French, S.W.; Bardag-Gorce, F.; Li, J.; French, B.A.; Oliva, J. Mallory-denk body pathogenesis revisited. World J. Hepatol. 2010, 2, 295. [Google Scholar] [CrossRef]
- Redeker, A.G.; Sterling, R.E.; Bronow, R.S. Effect of griseofulvin in acute intermittent porphyria. JAMA 1964, 188, 466–468. [Google Scholar] [CrossRef]
- Berman, A.; Franklin, R.L. Precipitation of acute intermittent porphyria by griseofulvin therapy. JAMA 1965, 192, 1005–1007. [Google Scholar] [CrossRef] [PubMed]
- Maitra, D.; Cunha, J.B.; Elenbaas, J.S.; Bonkovsky, H.L.; Shavit, J.A.; Omary, M.B. Porphyrin-induced protein oxidation and aggregation as a mechanism of porphyria-associated cell injury. Cell. Mol. Gastroenterol. Hepatol. 2019, 8, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Knasmüller, S.; Parzefall, W.; Helma, C.; Kassie, F.; Ecker, S.; Schulte-Hermann, R. Toxic Effects of griseofulvin: Disease models, mechanisms, and risk assessment. Crit. Rev. Toxicol. 1997, 27, 495–537. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Qian, S.-B. Chaperone-mediated hierarchical control in targeting misfolded proteins to aggresomes. Mol. Biol. Cell 2011, 22, 3277–3288. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. Autodock4 and autodocktools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Rosenbaum, M.B.; Chiale, P.A.; Halpern, M.S.; Nau, G.J.; Przybylski, J.; Levi, R.J.; Lázzari, J.O.; Elizari, M.V. Clinical efficacy of amiodarone as an antiarrhythmic agent. Am. J. Cardiol. 1976, 38, 934–944. [Google Scholar] [CrossRef]
- Poucell, S.; Ireton, J.; Valencia-Mayoral, P.; Downar, E.; Larratt, L.; Patterson, J.; Blendis, L.; Phillips, M.J. Amiodarone-associated phospholipidosis and fibrosis of the liver. Light, immunohistochemical, and electron microscopic studies. Gastroenterology 1984, 86, 926–936. [Google Scholar]
- Mason, J.W. Amiodarone. N. Engl. J. Med. 1987, 316, 455–466. [Google Scholar]
- March-Vila, E.; Pinzi, L.; Sturm, N.; Tinivella, A.; Engkvist, O.; Chen, H.; Rastelli, G. On the integration of in silico drug design methods for drug repurposing. Front. Pharmacol. 2017, 8, 298. [Google Scholar] [CrossRef]
- Ashburn, T.T.; Thor, K.B. Drug repositioning: Identifying and developing new uses for existing drugs. Nat. Rev. Drug Discov. 2004, 3, 673–683. [Google Scholar] [CrossRef]

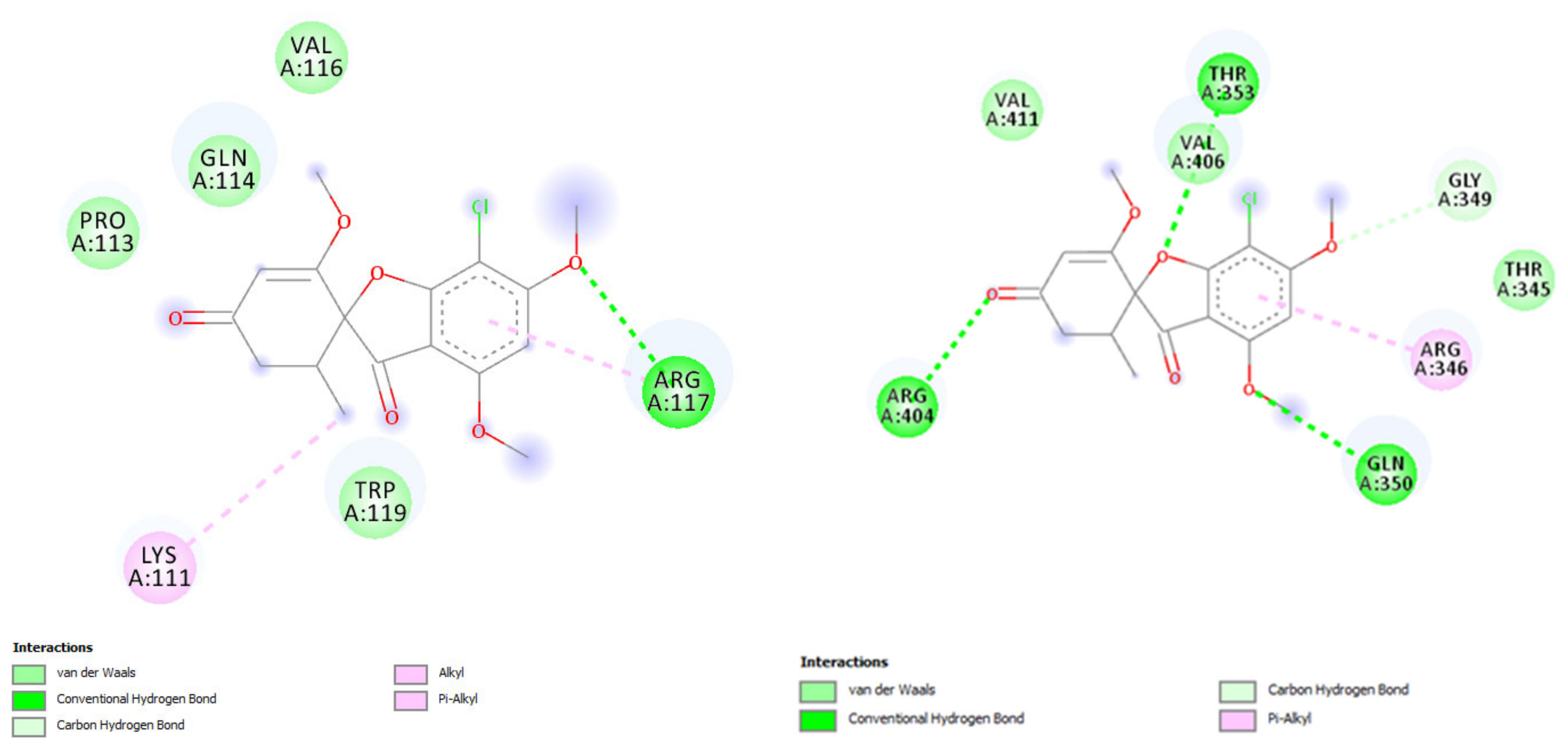
| Keratin | Species | PDB or Alpha Fold IDs | ΔGbinding (Kcal/mol) | Hydrogen Bond | Hydrophobic Interaction |
|---|---|---|---|---|---|
| K8 | Human | 3K3C | −3.17 | ILE 63 | GLY 61, GLY 62, THR 64 |
| 7K3X | −3.2 | ILE 63 | GLY 61 | ||
| 7K3Y | −3.61 | - | SER 58 | ||
| P05787F1 | −4.6 | GLN 85, LYS 92 | ARG 88, LYS 92 | ||
| Mouse | P11679F1 | −4.1 | - | LYS 420, THR 421 | |
| Rat | Q10758F1 | −4.55 | - | SER 15, GLY 16, PRO 17 | |
| K18 | Human | A0A024RAY2F1 | −4.72 | - | LEU 87, ARG 90, SER 93, TYR94 |
| P05783F1 | −4.99 | GLN 243 | SER 242, GLN 243, ASP 244, LYS 247, ILE 248, ASP 251 | ||
| Mouse | P05784F1 | −5.54 | ARG 117 | LYS 111, ARG 117 | |
| Rat | Q5BJY9F1 | −5.23 | GLN 350, THR 353, ARG 404 | ARG 346, GLY 349 |
| Keratin | Species | PDB or Alpha Fold IDs | ΔGbinding (Kcal/mol) | Hydrogen Bond | Hydrophobic Interaction |
|---|---|---|---|---|---|
| K8 | Human | 3K3C | −3.38 | GLY 61, ILE 63 | MET 60, GLY 61, ILE 63 |
| 7K3X | >0 | - | - | ||
| 7K3Y | >0 | - | - | ||
| P05787F1 | −4.1 | - | MET 406, SER 410, HIS 412 | ||
| Mouse | P11679F1 | -4.14 | - | MET 412, MET 415, ILE 417 | |
| Rat | Q10758F1 | −3.64 | ARG 49 | SER 51, LEU 52, PHE 55 | |
| K18 | Human | A0A024RAY2F1 | −4.69 | ARG 90 | LEU 87, ARG 90, LEU 91, TYR 94 |
| P05783F1 | −4.85 | - | LEU 87, ARG 90, LEU 91, TYR 94 | ||
| Mouse | P05784F1 | −2.51 | - | GLU 414 | |
| Rat | Q5BJY9F1 | −0.21 | - | ARG 374, GLU 378 |
| Keratin | Species | PDB or Alpha Fold IDs | ΔGbinding (Kcal/mol) | Hydrogen Bond | Hydrophobic Interaction |
|---|---|---|---|---|---|
| K8 | Human | 3K3C | −3.34 | GLY 62, THR 64 | GLY 61, GLY 62 |
| 7K3X | −3.34 | GLY 61, ILE 63 | GLY 61, ILE 63 | ||
| 7K3Y | −3.8 | SER 58 | SER 58, ALA 57 | ||
| P05787F1 | −5.43 | THR 64, ALA 65 | THR 64, ALA 65,VAL 66, THR 67 | ||
| Mouse | P11679F1 | −4.14 | THR 419, LYS 420, THR 421 | THR 421, THR 422 | |
| Rat | Q10758F1 | −3.93 | SER 21, ARG 23 | ALA 19, PHE 20, SER 21 | |
| K18 | Human | A0A024RAY2F1 | −4.48 | ARG 90 | ARG 90 |
| P05783F1 | −4.96 | SER 242, ASP 244 | GLN 243, LYS 247, ASP 251 | ||
| Mouse | P05784F1 | −5.28 | GLN 114, ARG 117 | LYS 111, PRO 113, GLN 114, ARG 117 | |
| Rat | Q5BJY9F1 | −5.22 | THR 353, ARG 404 | ARG 346, GLN 350, VAL 411 |
| Keratin | Species | PDB or Alpha Fold IDs | ΔGbinding (Kcal/mol) | Hydrogen Bond | Hydrophobic Interaction |
|---|---|---|---|---|---|
| K8 | Human | 3K3C | −4.04 | GLY 62, THR 64 | GLY 61, GLY 62 |
| 7K3X | −3.71 | ILE 63 | ILE 63 | ||
| 7K3Y | −3.95 | GLY 56, SER 58 | GLY 56, SER 58 | ||
| P05787F1 | −4.85 | LEU 72 | LEU 72, PRO 75, VAL 77 | ||
| Mouse | P11679F1 | −4.29 | GLN 134, SER 138 | LEU 131, GLN 134, GLN 135, SER 138 | |
| Rat | Q10758F1 | −4.50 | SER 21 | ARG 18, ALA 19 | |
| K18 | Human | A0A024RAY2F1 | −4.88 | TYR94 | LEU 91, TYR 94 |
| P05783F1 | −4.81 | GLU 236 | SER230, SER 231, LEU 233, VAL 235, GLU 236 | ||
| Mouse | P05784F1 | −5.61 | LEU 108, TPR 119 | LYS 111, PRO 113, VAL 116 | |
| Rat | Q5BJY9F1 | −5.33 | ARG 346, GLN 350, ARG 404 | ARG 346, GLN 350, VAL 411 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aris, P.; Wei, Y.; Mohamadzadeh, M.; Xia, X. Griseofulvin: An Updated Overview of Old and Current Knowledge. Molecules 2022, 27, 7034. https://doi.org/10.3390/molecules27207034
Aris P, Wei Y, Mohamadzadeh M, Xia X. Griseofulvin: An Updated Overview of Old and Current Knowledge. Molecules. 2022; 27(20):7034. https://doi.org/10.3390/molecules27207034
Chicago/Turabian StyleAris, Parisa, Yulong Wei, Masoud Mohamadzadeh, and Xuhua Xia. 2022. "Griseofulvin: An Updated Overview of Old and Current Knowledge" Molecules 27, no. 20: 7034. https://doi.org/10.3390/molecules27207034
APA StyleAris, P., Wei, Y., Mohamadzadeh, M., & Xia, X. (2022). Griseofulvin: An Updated Overview of Old and Current Knowledge. Molecules, 27(20), 7034. https://doi.org/10.3390/molecules27207034









