Development of a Polymeric Membrane Impregnated with Poly-Lactic Acid (PLA) Nanoparticles Loaded with Red Propolis (RP)
Abstract
1. Introduction
2. Results
2.1. The Yield of Red Propolis Extract
2.2. Nanoparticles and the Production of Membranes Loaded with Red Propolis Extract
2.3. Determination of Total Phenols and Total Flavonoids Content
2.4. Antioxidant Activity Using DPPH and FRAP
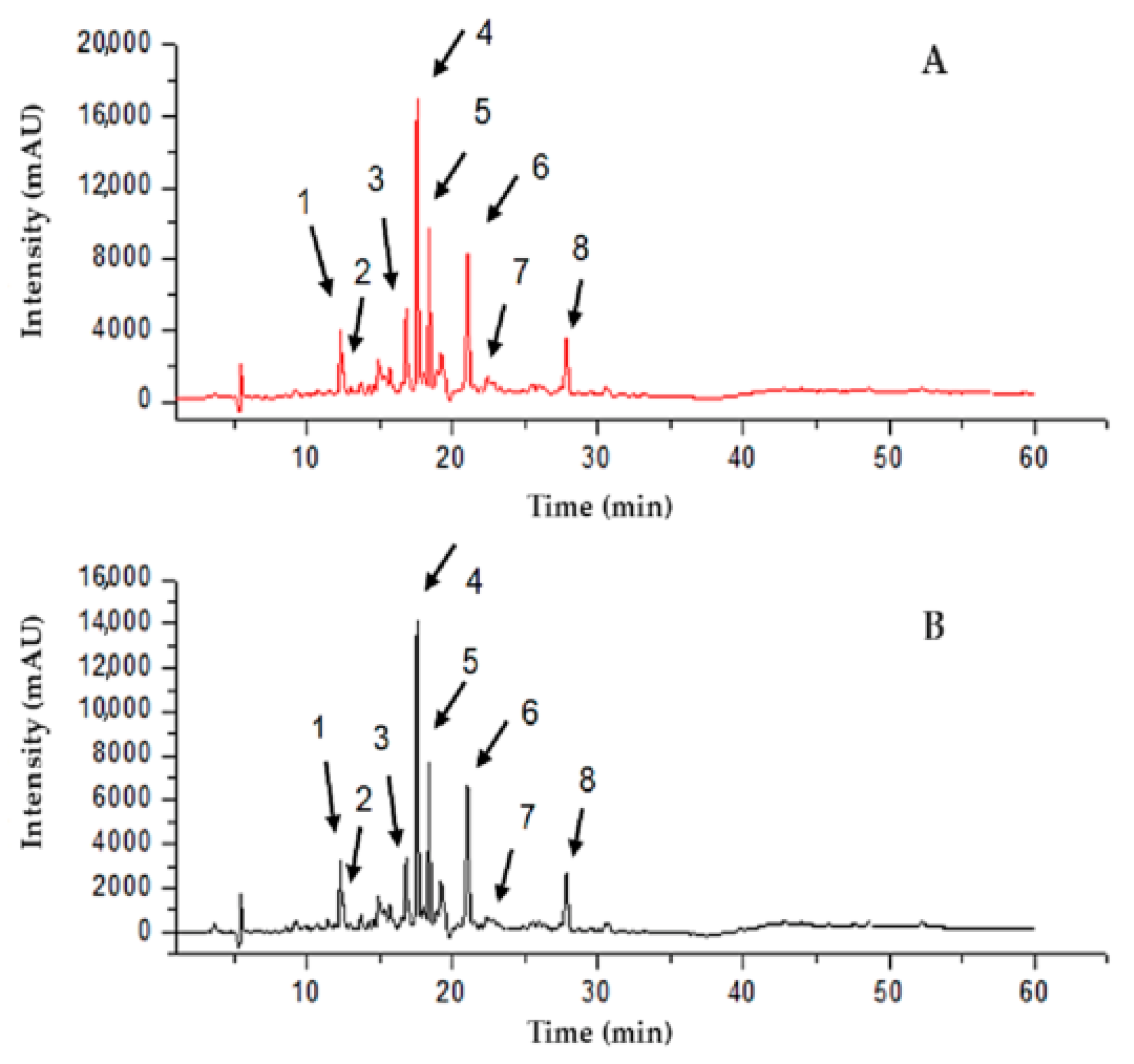
2.5. Determination of the RPE Chemical Markers
2.6. Encapsulation Efficiency (% EE)
2.7. Particle Size, Polydispersion Index, and Zeta Potential
2.8. Differential Exploratory Calorimetry
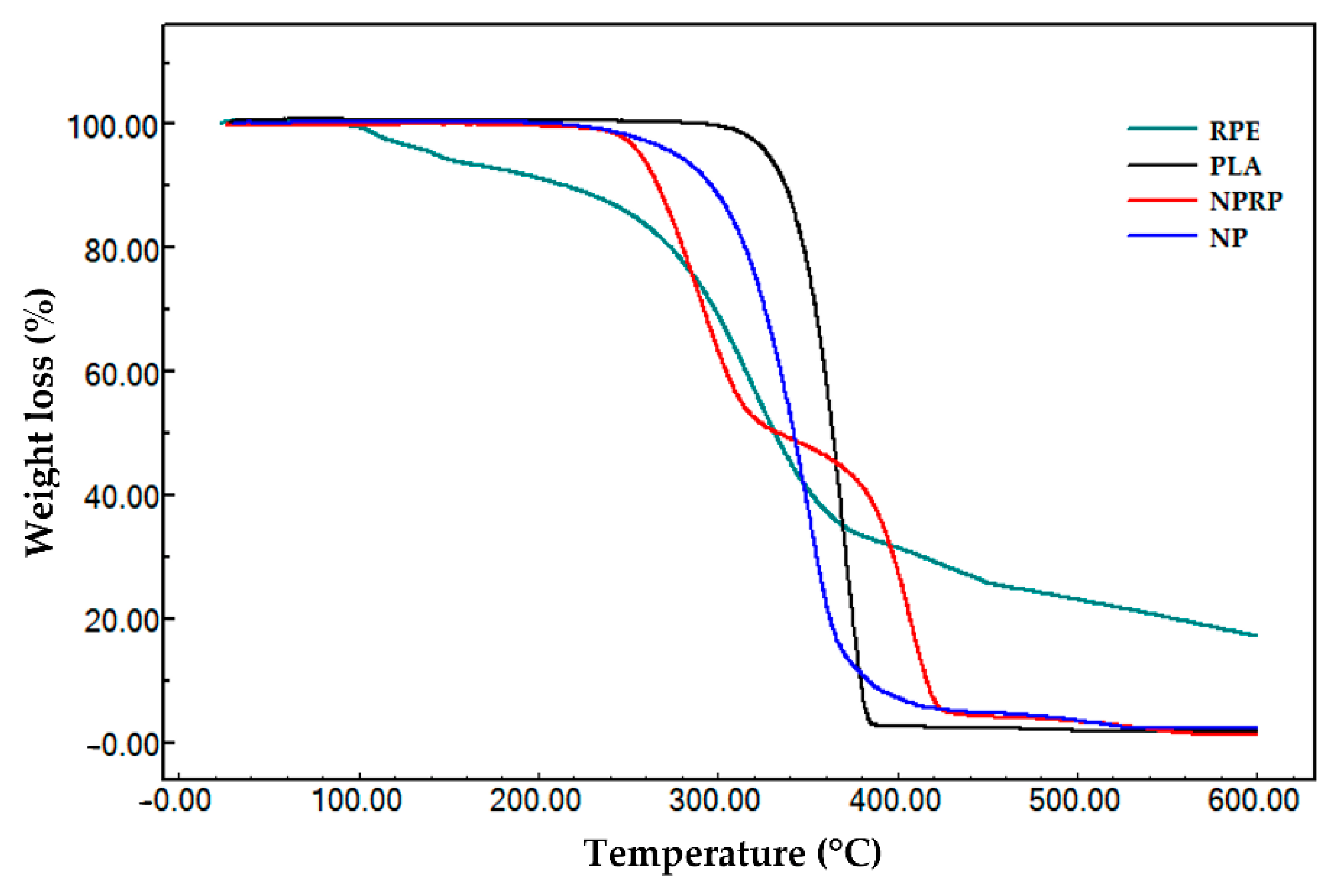
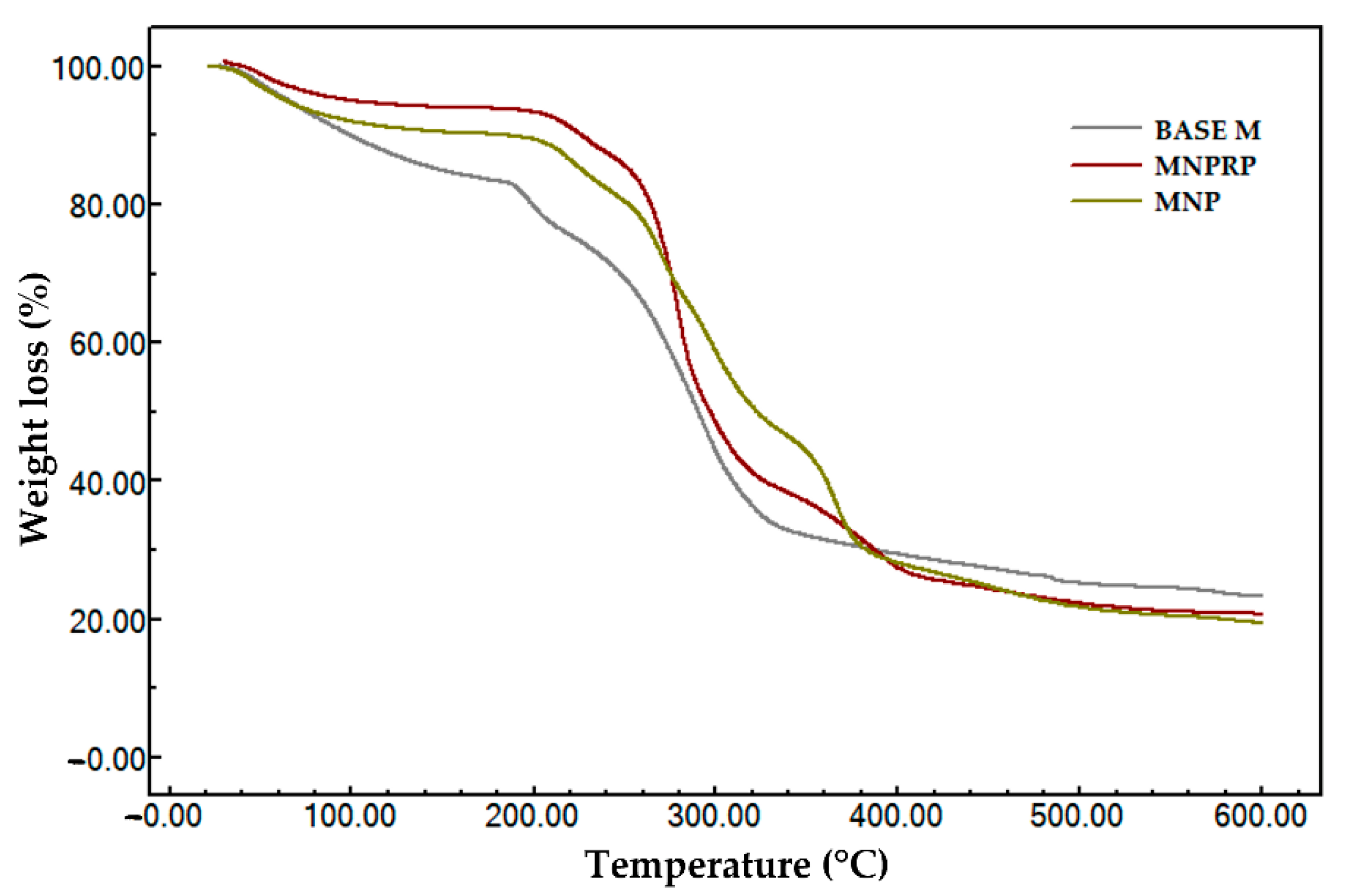
2.9. Thermogravimetric Analysis
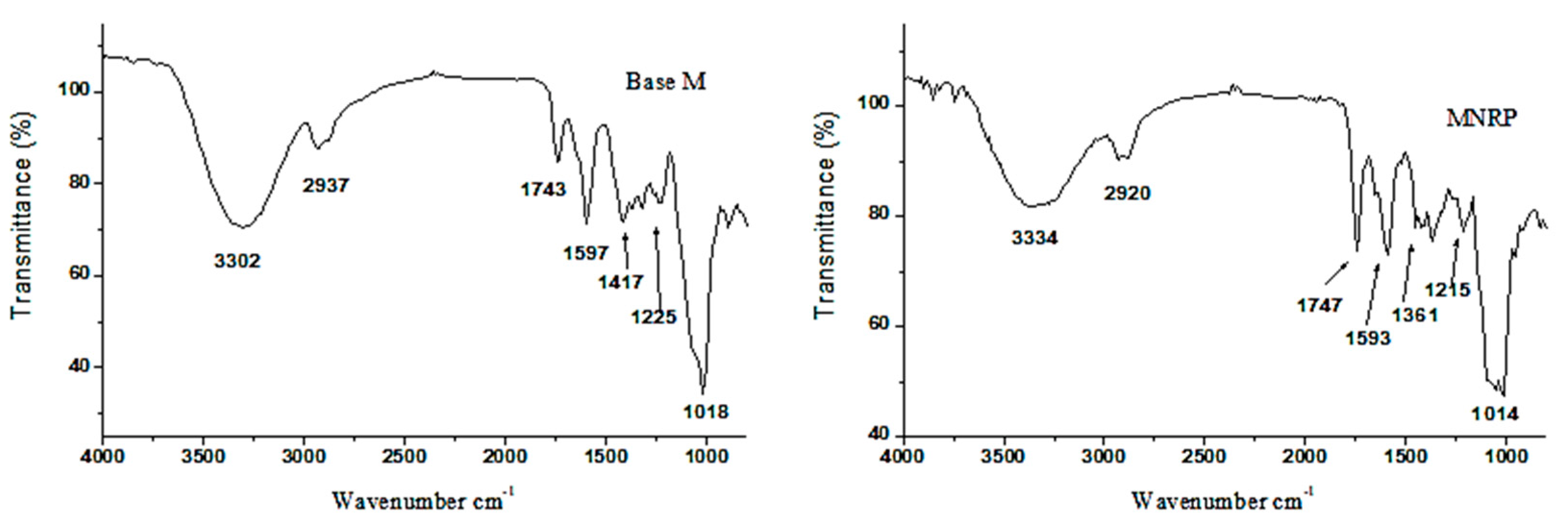
2.10. ATR Coupled to Fourier Transform Infrared (ATR-FTIR)
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Red Propolis Extract (RPE)
4.3. Preparation of the PLA Nanoparticles Loaded with Red Propolis (NPRP) and Membrane
4.3.1. Preparation of the PLA Nanoparticles Loaded with Red Propolis (NPRP)
4.3.2. Development of the Polymeric Membrane Loaded with NPRP
4.4. Size, Polydispersity Index, and Zeta Potential of the Nanoparticles
4.5. Rupture of the Nanoparticles Loaded with Red Propolis (NPRP)
4.6. Extraction of the Red Propolis Impregnated as Nanoparticles in the Polymeric Membrane
4.7. Total Phenol Content (TPC)
4.8. Determination of the Total Flavonoid Content (TFC)
4.9. Antioxidant Activity Assay
4.9.1. Evaluation of the Antioxidant Activity by the DPPH Method
4.9.2. FRAP Assay
4.10. Identification of RPE Markers Using the UPLC-DAD Method
4.11. Encapsulation Efficiency (%)
4.12. Thermal Analyses (DSC and TG)
4.13. ATR- FTIR Spectroscopy
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Pugliese, R.; Beltrami, B.; Regondi, S.; Lunetta, C. Polymeric Biomaterials for 3D Printing in Medicine: An Overview. J. 3D Print Med. 2021, 2, 3–10. [Google Scholar] [CrossRef]
- Banik, B.L.; Brown, J.L. Polymeric Biomaterials in Nanomedicine. In Natural and Synthetic Biomedical Polymers, 1st ed.; Sangamesh, G.K., Cato, T.L., Meng, D., Eds.; Elsevier Inc.: San Diego, CA, USA, 2014; Volume 1, pp. 387–395. [Google Scholar]
- Ibrahim, H.M.; Reda, M.M.; Klingner, A. Preparation and Characterization of Green Carboxymethylchitosan (CMCS)—Polyvinyl Alcohol (PVA) Electrospun Nanofibers Containing Gold Nanoparticles (AuNPs) and Its Potential Use as Biomaterials. Int. J. Biol. Macromol. 2020, 151, 821–829. [Google Scholar] [CrossRef]
- Silva, V.C.; Silva, A.M.G.S.; Basílio, J.A.D.; Xavier, J.A.; do Nascimento, T.G.; Naal, R.M.Z.G.; del Lama, M.P.; Leonelo, L.A.D.; Mergulhão, N.L.O.N.; Maranhão, F.C.A.; et al. New Insights for Red Propolis of Alagoas-Chemical Constituents, Topical Membrane Formulations and Their Physicochemical and Biological Properties. Molecules 2020, 25, 5811. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, N.K.; Kumar, S.S.D.; Houreld, N.N.; Abrahamse, H. A Review on Nanoparticle Based Treatment for Wound Healing. J. Drug Deliv. Sci. Technol. 2018, 44, 421–430. [Google Scholar] [CrossRef]
- Berthet, M.; Gauthier, Y.; Lacroix, C.; Verrier, B.; Monge, C. Nanoparticle-Based Dressing: The Future of Wound Treatment? Trends Biotechnol. 2017, 35, 770–784. [Google Scholar] [CrossRef]
- Lasprilla, A.J.R.; Martinez, G.A.R.; Lunelli, B.H.; Jardini, A.L.; Filho, R.M. Poly-Lactic Acid Synthesis for Application in Biomedical Devices—A Review. Biotechnol. Adv. 2012, 30, 321–328. [Google Scholar] [CrossRef]
- Rufatto, L.C.; dos Santos, D.A.; Marinho, F.; Henriques, J.A.P.; Ely, M.R.; Moura, S. Red Propolis: Chemical Composition and Pharmacological Activity. Asian Pac. J. Trop. Biomed. 2017, 7, 591–598. [Google Scholar] [CrossRef]
- Azevedo, L.F.; da Fonseca Silva, P.; Brandão, M.P.; da Rocha, L.G.; Aragão, C.F.S.; da Silva, S.A.S.; Porto, I.C.C.M.; Basílio-Júnior, I.D.; da Silva Fonseca, E.J.; de Moura, M.A.B.F.; et al. Sistemas de Polímeros de Nanopartículas Cargados Con Extracto de Propóleo Rojo: Un Estudio Comparativo de Sistemas de Encapsulamiento, PCL-Pluronic versus Eudragit®E100-Pluronic. J. Apic. Res. 2018, 57, 255–270. [Google Scholar] [CrossRef]
- Daugsch, A.; Moraes, C.S.; Fort, P.; Park, Y.K. Brazilian Red Propolis—Chemical Composition and Botanical Origin. Evid. Based Complement. Altern. Med. 2008, 5, 435–441. [Google Scholar] [CrossRef]
- Batista, C.M.; Alves, A.V.F.; Queiroz, L.A.; Lima, B.S.; Filho, R.N.P.; Araújo, A.A.S.; de Albuquerque Júnior, R.L.C.; Cardoso, J.C. The Photoprotective and Anti-Inflammatory Activity of Red Propolis Extract in Rats. J. Photochem. Photobiol. B 2018, 180, 198–207. [Google Scholar] [CrossRef]
- Freires, I.A.; de Alencar, S.M.; Rosalen, P.L. A Pharmacological Perspective on the Use of Brazilian Red Propolis and Its Isolated Compounds against Human Diseases. Eur. J. Med. Chem. 2016, 110, 267–279. [Google Scholar] [CrossRef]
- Corrêa, F.R.S.; Schanuel, F.S.; Moura-Nunes, N.; Monte-Alto-Costa, A.; Daleprane, J.B. Brazilian Red Propolis Improves Cutaneous Wound Healing Suppressing Inflammation-Associated Transcription Factor NFκB. Biomed. Pharmacothe. 2017, 86, 162–171. [Google Scholar] [CrossRef]
- Nascimento, T.G.; Redondo, G.D.P.; Araújo, A.C.T.; Silva, V.C.; Lira, G.M.; Meireles, G.L.A.; Conceição, M.M.; Freitas, J.D.; Souza, J.S.; Júnior, J.X.A. Modified Release Microcapsules Loaded with Red Propolis Extract Obtained by Spray-Dryer Technique: Phytochemical, Thermal and Physicochemical Characterizations. J. Therm. Anal. Calorim. 2019, 138, 3559–3569. [Google Scholar] [CrossRef]
- Righi, A.A.; Alves, T.R.; Negri, G.; Marques, L.M.; Breyer, H.; Salatino, A. Brazilian Red Propolis: Unreported Substances, Antioxidant and Antimicrobial Activities. J. Sci. Food Agric. 2011, 91, 2363–2370. [Google Scholar] [CrossRef] [PubMed]
- Devequi-Nunes, D.; Machado, B.A.S.; de Abreu Barreto, G.; Silva, J.R.; da Silva, D.F.; da Rocha, J.L.C.; Brandão, H.N.; Borges, V.M.; Umsza-Guez, M.A. Chemical Characterization and Biological Activity of Six Different Extracts of Propolis through Conventional Methods and Supercritical Extraction. PLoS ONE 2018, 13, e0207676. [Google Scholar] [CrossRef]
- Marcucci, M.C.; Ferreres, F.; García-Viguera, C.; Bankova, V.S.; de Castro, S.L.; Dantas, A.P.; Valente, P.H.M.; Paulino, N. Phenolic Compounds from Brazilian Propolis with Pharmacological Activities. J. Ethnopharmacol. 2001, 74, 105–112. [Google Scholar] [CrossRef]
- Hatano, A.; NonaKa, T.; Yoshino, M.; Ahn, M.-R.; Tazawa, S.; Araki, Y.; Kumazawa, S. Antioxidant Activity and Phenolic Constituents of Red Propolis from Shandong, China. Food Sci. Technol. Res. 2012, 18, 577–584. [Google Scholar] [CrossRef]
- Lopez, G.C. Analysis of the Composition of Samples of Red Brazilian Propolis by Mass Spectrometry with Electrospray Ionization and Ultra High Performance Liquid Chromatography (UPLC-ESI-MS) and Evaluation of the Antioxidant and Antimicrobial Activity. Master’s Thesis, Universidade Estadual de Campinas, Campinas, Brazil. Available online: https://repositorioslatinoamericanos.uchile.cl/handle/2250/1349845 (accessed on 8 September 2022).
- de Mendonça, I.C.G.; de Moraes Porto, I.C.C.; do Nascimento, T.G.; de Souza, N.S.; dos Santos Oliveira, J.M.; Arruda, R.E.d.S.; Mousinho, K.C.; dos Santos, A.F.; Basílio-Júnior, I.D.; Parolia, A.; et al. Brazilian Red Propolis: Phytochemical Screening, Antioxidant Activity and Effect against Cancer Cells. BMC Complement. Altern. Med. 2015, 15, 357. [Google Scholar] [CrossRef] [PubMed]
- Gomes, N.T.; Fonseca, S.P.; Farias, A.L.; Guerra, R.L.; Cristina, C.M.P.I.; Flávio, A.L.M.T.; Diniz, B.J.I.; Aparecido, M.G.L.; Braga, D.C.; Jorge, S.F.E. Polymeric Nanoparticles of Brazilian Red Propolis Extract: Preparation, Characterization, Antioxidant and Leishmanicidal Activity. Nanoscale Res. Lett. 2016, 11, 301. [Google Scholar] [CrossRef]
- Pandey, S.K.; Patel, D.K.; Thakur, R.; Mishra, D.P.; Maiti, P.; Haldar, C. Anti-Cancer Evaluation of Quercetin Embedded PLA Nanoparticles Synthesized by Emulsified Nanoprecipitation. Int. J. Biol. Macromol. 2015, 75, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, T.; Ventura, C.A.; Giannone, I.; Ruozi, B.; Montenegro, L.; Pignatello, R.; Puglisi, G. PLA/PLGA Nanoparticles for Sustained Release of Docetaxel. Int. J. Pharm. 2006, 325, 172–179. [Google Scholar] [CrossRef]
- Auras, R.; Harte, B.; Selke, S. An Overview of Polylactides as Packaging Materials. Macromol. Biosci. 2004, 4, 835–864. [Google Scholar] [CrossRef] [PubMed]
- Correia, C.A. Obtainment and Characterization of Nanocellulose-Reinforced PLA. Master’s Thesis, University of São Paulo, São Paulo, Brazil, 15 April 2015. [Google Scholar] [CrossRef]
- Nalbandi, A. Kinetics of Thermal Degradation of Polylactic Acid Under N2 Atmosphere. Iran. Polym. J. 2001, 10, 371–376. [Google Scholar]
- Restrepo, I.; Medina, C.; Meruane, V.; Akbari-Fakhrabadi, A.; Flores, P.; Rodríguez-Llamazares, S. The Effect of Molecular Weight and Hydrolysis Degree of Poly(Vinyl Alcohol)(PVA) on the Thermal and Mechanical Properties of Poly(Lactic Acid)/PVA Blends. Polímeros 2018, 28, 169–177. [Google Scholar] [CrossRef]
- Choksi, N.; Desai, H. Synthesis of Biodegradable Polylactic Acid Polymer By Using Lactic Acid Monomer. Int. J. Appl. Chem 2017, 13, 377–384. [Google Scholar]
- Giita, S.V.S.; Ibrahim, N.A.; Zainuddin, N.; Yunus, W.M.Z.W.; Hassan, H.A. Mechanical, Thermal and Morphological Properties of Poly(Lactic Acid)/Epoxidized Palm Olein Blend. Molecules 2012, 17, 11729–11747. [Google Scholar] [CrossRef]
- Cukrowicz, S.; Grabowska, B.; Kaczmarska, K.; Bobrowski, A.; Sitarz, M.; Tyliszczak, B. Structural Studies (FTIR, XRD) of Sodium Carboxymethyl Cellulose Modified Bentonite. Arch. Foundry Eng. 2020, 20, 119–125. [Google Scholar] [CrossRef]
- Cuadro, P.; Belt, T.; Kontturi, K.S.; Reza, M.; Kontturi, E.; Vuorinen, T.; Hughes, M. Cross-Linking of Cellulose and Poly(Ethylene Glycol) with Citric Acid. React. Funct. Polym. 2015, 90, 21–24. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Kenawy, E.R.S.; Chen, X. A Review on Polymeric Hydrogel Membranes for Wound Dressing Applications: PVA-Based Hydrogel Dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef]
- Yazdi, M.K.; Vatanpour, V.; Taghizadeh, A.; Taghizadeh, M.; Ganjali, M.R.; Munir, M.T.; Habibzadeh, S.; Saeb, M.R.; Ghaedi, M. Hydrogel Membranes: A Review. Mater. Sci. Eng. C 2020, 114, 111023. [Google Scholar] [CrossRef]
- Pahlevanneshan, Z.; Deypour, M.; Kefayat, A.; Rafienia, M.; Sajkiewicz, P.; Neisiany, R.E.; Enayati, M.S. Polyurethane-Nanolignin Composite Foam Coated with Propolis as a Platform for Wound Dressing: Synthesis and Characterization. Polymers 2021, 13, 3191. [Google Scholar] [CrossRef]
- Valle, K.Z.M.; Acuña, R.A.S.; Arana, J.V.R.; Lobo, N.; Rodriguez, C.; Cuevas-Gonzalez, J.C.; Tovar-Carrillo, K.L. Natural Film Based on Pectin and Allantoin for Wound Healing: Obtaining, Characterization, and Rat Model. Biomed. Res. Int. 2020, 2020, 6897497. [Google Scholar] [CrossRef]
- Oliveira, R.N.; McGuinness, G.B.; Rouze, R.; Quilty, B.; Cahill, P.; Soares, G.D.A.; Thiré, R.M.S.M. PVA Hydrogels Loaded with a Brazilian Propolis for Burn Wound Healing Applications. J. Appl. Polym. Sci. 2015, 132, 1–12. [Google Scholar] [CrossRef]
- Guadarrama-Acevedo, M.C.; Mendoza-Flores, R.A.; del Prado-Audelo, M.L.; Urbán-Morlán, Z.; Giraldo-Gomez, D.M.; Magaña, J.J.; González-Torres, M.; Reyes-Hernández, O.D.; Figueroa-González, G.; Caballero-Florán, I.H.; et al. Development and Evaluation of Alginate Membranes with Curcumin-Loaded Nanoparticles for Potential Wound-Healing Applications. Pharmaceutics 2019, 11, 389. [Google Scholar] [CrossRef]
- Fernandes, D.M.; Barbosa, W.S.; Rangel, W.S.P.; Valle, I.M.M.; dos S. Matos, A.P.; Melgaço, F.G.; Dias, M.L.; Júnior, E.R.; da Silva, L.C.P.; de Abreu, L.C.L.; et al. Polymeric Membrane Based on Polyactic Acid and Babassu Oil for Wound Healing. Mater. Today Commun. 2021, 26, 102173. [Google Scholar] [CrossRef]
- Bi, H.; Feng, T.; Li, B.; Han, Y. In Vitro and In Vivo Comparison Study of Electrospun PLA and PLA/PVA/SA Fiber Membranes for Wound Healing. Polymers 2020, 12, 839. [Google Scholar] [CrossRef] [PubMed]
- Ulloa, P.A.; Vidal, J.; de Dicastillo, C.L.; Rodriguez, F.; Guarda, A.; Cruz, R.M.S.; Galotto, M.J. Development of Poly(Lactic Acid) Films with Propolis as a Source of Active Compounds: Biodegradability, Physical, and Functional Properties. J. Appl. Polym. Sci. 2019, 136, 47090. [Google Scholar] [CrossRef]
- Lima, C.R.; Souza, S.J.; Belo, N.R.; Oliveira, P.A.; Valéria, O.J.; Divino, A.E.; Berretta, S.A.A.; Maria, T.S.; Cordeiro, C.J.; Zanardo, G.M. Antinociceptive and Anti-Inflammatory Effects of Brazilian Red Propolis Extract and Formononetin in Rodents. J. Ethnopharmacol. 2015, 173, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Kole, L.; Giri, B.; Manna, S.K.; Pal, B.; Ghosh, S. Biochanin-A, an Isoflavon, Showed Anti-Proliferative and Anti-Inflammatory Activities through the Inhibition of INOS Expression, P38-MAPK and ATF-2 Phosphorylation and Blocking NFκB Nuclear Translocation. Eur. J. Pharmacol. 2011, 653, 8–15. [Google Scholar] [CrossRef]
- Nascimento, T.G.; Silva, A.S.; Lessa, C.P.B.; Silva, S.A.S.; Fidelis, M.M.A.B.; Almeida, C.P.; Silva, V.C.; Wanderley, A.B.; Basílio, J.I.D.; Escodro, P.B. Examen de Las Actividades Fitoquímica, Antioxidante y Antibacteriana de Extractos Comerciales de Propóleos. J. Apic. Res. 2018, 57, 246–254. [Google Scholar] [CrossRef]
- Morais, D.V.; Rosalen, P.L.; Ikegaki, M.; de Souza Silva, A.P.; Massarioli, A.P.; de Alencar, S.M. Active Antioxidant Phenolics from Brazilian Red Propolis: An Optimization Study for Their Recovery and Identification by Lc-Esi-Qtof-Ms/Ms. Antioxidants 2021, 10, 297. [Google Scholar] [CrossRef]
- Oldoni, T.L.C.; Oliveira, S.C.; Andolfatto, S.; Karling, M.; Calegari, M.A.; Sado, R.Y.; Maia, F.M.C.; Alencar, S.M.; Lima, V.A. Chemical Characterization and Optimization of the Extraction Process of Bioactive Compounds from Propolis Produced by Selected Bees Apis Mellifera. J. Braz. Chem. Soc. 2015, 26, 2054–2062. [Google Scholar] [CrossRef]
- Andrade, J.K.S.; Denadai, M.; de Oliveira, C.S.; Nunes, M.L.; Narain, N. Evaluation of Bioactive Compounds Potential and Antioxidant Activity of Brown, Green and Red Propolis from Brazilian Northeast Region. Int. Food Res. J. 2017, 101, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Gaumet, M.; Vargas, A.; Gurny, R.; Delie, F. Nanoparticles for Drug Delivery: The Need for Precision in Reporting Particle Size Parameters. Eur. J. Pharm. Biophar. 2008, 69, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Roussaki, M.; Gaitanarou, A.; Diamanti, P.C.; Vouyiouka, S.; Papaspyrides, C.; Kefalas, P.; Detsi, A. Encapsulation of the Natural Antioxidant Aureusidin in Biodegradable PLA Nanoparticles. Polym. Degrad. Stab. 2014, 108, 182–187. [Google Scholar] [CrossRef]
- Sarasua, J.-R.; Prud’homme, R.E.; Wisniewski, M.; le Borgne, A.; Spassky, N. Crystallization and Melting Behavior of Polylactides. Macromolecules 1998, 31, 3895–3905. [Google Scholar] [CrossRef]
- Raquez, J.M.; Murena, Y.; Goffin, A.L.; Habibi, Y.; Ruelle, B.; DeBuyl, F.; Dubois, P. Surface-Modification of Cellulose Nanowhiskers and Their Use as Nanoreinforcers into Polylactide: A Sustainably-Integrated Approach. Compos. Sci. Technol. 2012, 72, 544–549. [Google Scholar] [CrossRef]
- Alam, A.K.M.M.; Beg, M.D.H.; Mina, M.F.; Mamun, A.A.; Bledzki, A.K. Degradation and Stability of Green Composites Fabricated from Oil Palm Empty Fruit Bunch Fiber and Polylactic Acid: Effect of Fiber Length. J. Compos. Mater. 2015, 49, 3103–3114. [Google Scholar] [CrossRef]
- Naga, N.; Yoshida, Y.; Noguchi, K.; Murase, S. Crystallization of Amorphous Poly(Lactic Acid) Induced by Vapor of Acetone to Form High Crystallinity and Transparency Specimen. Open J. Polym. Chem. 2013, 03, 29–33. [Google Scholar] [CrossRef]
- Ding, J.; Chen, S.C.; Wang, X.L.; Wang, Y.Z. Synthesis and Properties of Thermoplastic Poly(Vinyl Alcohol)-Graft-Lactic Acid Copolymers. Ind. Eng. Chem. Res. 2009, 48, 788–793. [Google Scholar] [CrossRef]
- Singh, S.K.; Anthony, P.; Chowdhury, A. High Molecular Weight Poly(Lactic Acid) Synthesized with Apposite Catalytic Combination and Longer Time. Orient. J. Chem. 2018, 34, 1984–1990. [Google Scholar] [CrossRef]
- Xavier, J.A.; Valentim, I.B.; Camatari, F.O.S.; de Almeida, A.M.M.; Goulart, H.F.; de Souza Ferro, J.N.; de Oliveira Barreto, E.; Cavalcanti, B.C.; Bottoli, C.B.G.; Goulart, M.O.F. Polyphenol Profile by Uhplc-Ms/Ms, Anti-Glycation, Antioxidant and Cytotoxic Activities of Several Samples of Propolis from the Northeastern Semi-Arid Region of Brazil. Pharm. Biol. 2017, 55, 1884–1893. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]





| Composition % (Proportion, w/w) | |||
|---|---|---|---|
| Component | Base M | MNP | MNPRP |
| NaCMC | 0.9 | 0.9 | 0.9 |
| Propylene glycol | 1.6 | 1.6 | 1.6 |
| Citrus pectin | 0.6 | 0.6 | 0.6 |
| Distilled water | 96.9 | 96.8 | 96.8 |
| NPs | - | 0.1 | - |
| NPRP | - | - | 0.1 |
| DPPH● | |||||
|---|---|---|---|---|---|
| Sample | TPC (mg GAE g−1) * | TFC (mg EQ g−1) ** | RSA (%) | IC50 (µg mL−1) | FRAP (μmol Fe2+ g−1) *** |
| RPE | 240.3 ± 3.4 | 37.8 ± 0.9 | 92.8 ± 0.1 | 13.1 | 1400.2 ± 2.6 |
| NPRP | 191.7 ± 0.3 | 35.8 ± 3.9 | 75.8 ± 3.9 | 16.1 | 1294.2 ± 0.5 |
| MNPRP | 183.4 ± 2.1 | 26.4 ± 1.9 | 67.8 ± 1.0 | 18.4 | 696 ± 0.8 |
| RPE (400 µg mL−1) | NPRP (400 µg mL−1) | Recovery | |||||
|---|---|---|---|---|---|---|---|
| Compounds | λ | Peak | RT (min) | Area | RT (min) | Area | (%) * |
| Liquiritigenin | 275 | 1 | 12.3 | 34,750 | 12.31 | 28,807 | 82.9 |
| Daidzein | 249 | 2 | 12.51 | 18,096 | 12.5 | 16,785 | 92.8 |
| Isoliquiritigenin | 366 | 3 | 16.82 | 71,587 | 16.81 | 58,239 | 81.4 |
| Formononetin | 249 | 4 | 17.64 | 120,059 | 17.63 | 102,498 | 85.4 |
| Unk 1 | 281 | 5 | 18.42 | 79,979 | 18.41 | 63,478 | 79.4 |
| Unk 2 | 286 | 6 | 21.07 | 109,356 | 21.06 | 92,883 | 84.9 |
| Biochanin A | 249 | 7 | 23.25 | 6540 | 23.21 | 5239 | 80.1 |
| Unk 3 | 281 | 8 | 27.83 | 44,042 | 27.86 | 36,366 | 82.6 |
| Nanoparticles | Particle Size (nm) | Polydispersion Index (PDI) | Zeta Potential (mV) | EE ** (%) |
|---|---|---|---|---|
| NPs | 194.1 ± 0.082 * | 0.289 ± 0.024 * | −11.2 | - |
| NPRP | 361.2 ± 2.71 * | 0.239 ± 0.019 * | −16.4 | 85.4 |
| Sample | Stage(s) | Temperature | Δm (%) | |
|---|---|---|---|---|
| Onset (°C) * | Endset (°C) ** | |||
| RPE | I | 88.8 | 118.8 | 5.98 |
| II | 278.0 | 353.4 | 62.79 | |
| III | 427.4 | 503.6 | 14.20 | |
| I–III | 24.0 | 600.0 | 82.96 | |
| PLA | I | 343.7 | 382.7 | 98.19 |
| NP | I | 316.3 | 374.0 | 97.48 |
| NPRP | I | 260.4 | 312.0 | 51.55 |
| II | 387.8 | 444.2 | 47.18 | |
| I–II | 24.0 | 600.0 | 98.73 | |
| Sample | Stage(s) | Temperature | Δm (%) | |
|---|---|---|---|---|
| Onset (°C) * | Endset (°C) ** | |||
| Base M | I | 37.4 | 113.7 | 15.94 |
| II | 189.6 | 206.3 | 7.85 | |
| III | 258.9 | 336.8 | 52.91 | |
| I–III | 24.0 | 600.0 | 76.60 | |
| MNP | I | 33.7 | 86.6 | 9.74 |
| II | 206.4 | 228.9 | 7.17 | |
| III | 260.9 | 271.8 | 26.38 | |
| IV | 390.0 | 414.4 | 37.18 | |
| I–IV | 24.0 | 600.0 | 80.48 | |
| MNPRP | I | 40.1 | 82.5 | 6.86 |
| II | 211.7 | 235.7 | 6.80 | |
| III | 265.9 | 289.6 | 43.76 | |
| IV | 310.1 | 386.4 | 22.69 | |
| I–IV | 24.0 | 600.0 | 80,11 | |
| Type of Polymeric Dressings | Polymer Matrix | Characteristics | Advantages | Disadvantages | Reference |
|---|---|---|---|---|---|
| Foam | Polyurethane | Flexible polyurethane foam and lignine nanoparticles enriched with green propolis | Antibacterial activity due to propolis allows for cell adhesion, is hydrophilic, and accelerates healing | Very adherent, forming an opaque layer that makes it difficult to monitor the wound, not applicable to dry wounds and with low stability | [34] |
| Film | Pectin | Pectin film enriched with allantoin, plasticized with glycerol | Absorbs exudate, flexible, proper contact angle, and accelerates healing | Opaque layer formation, intermediate swelling ability | [35] |
| Hydrogel membrane | Polyvinyl alcohol (PVA) | PVA membrane enriched with red propolis | Antibacterial activity due to the propolis, high swelling and traction capacities | Variable mechanical and swelling stability, opaque, semipermeable to gases and to water vapor | [36] |
| Sodium carboxymethylcellulose (NaCMC) | Sodium carboxymethylcellulose membrane enriched with red propolis | Antibacterial activity due to propolis, high swelling capacity, antiallergic, low cost | [4] | ||
| Alginate membrane | Alginate | Alginate and PVA membrane enriched with curcumin nanoparticles | Antibacterial activity due to the curcumin, high swelling capacity, transparent, allows for gas exchange | Swelling, stability, and variable mechanics | [37] |
| Poly(lactic acid) (PVA) membrane | Poly(lactic acid) (PLA) | Polylactic acid membrane enriched with babassu oil | Formed by pores, it maintains moisture on top of the wound, is nontoxic, promotes cell migration, and has antimicrobial activity | Low swelling capacity, formation of opaque layer | [38] |
| Poly(lactic acid) (PLA)/sodium alginate (SA)/poly(vinyl alcohol) (PVA) | PLA/PVA/SA fiber membranes | Mechanical stability, biocompatible and biodegradable, accelerates wound healing | [39] | ||
| Poly(lactic acid) | PLA membrane enriched with green propolis | Mechanical stability, biocompatible and biodegradable, with antimicrobial activity | [40] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Costa Silva, V.; do Nascimento, T.G.; Mergulhão, N.L.O.N.; Freitas, J.D.; Duarte, I.F.B.; de Bulhões, L.C.G.; Dornelas, C.B.; de Araújo, J.X., Júnior; dos Santos, J.; Silva, A.C.A.; et al. Development of a Polymeric Membrane Impregnated with Poly-Lactic Acid (PLA) Nanoparticles Loaded with Red Propolis (RP). Molecules 2022, 27, 6959. https://doi.org/10.3390/molecules27206959
da Costa Silva V, do Nascimento TG, Mergulhão NLON, Freitas JD, Duarte IFB, de Bulhões LCG, Dornelas CB, de Araújo JX Júnior, dos Santos J, Silva ACA, et al. Development of a Polymeric Membrane Impregnated with Poly-Lactic Acid (PLA) Nanoparticles Loaded with Red Propolis (RP). Molecules. 2022; 27(20):6959. https://doi.org/10.3390/molecules27206959
Chicago/Turabian Styleda Costa Silva, Valdemir, Ticiano G. do Nascimento, Naianny L. O. N. Mergulhão, Johnnatan D. Freitas, Ilza Fernanda B. Duarte, Laisa Carolina G. de Bulhões, Camila B. Dornelas, João Xavier de Araújo, Júnior, Jucenir dos Santos, Anielle C. A. Silva, and et al. 2022. "Development of a Polymeric Membrane Impregnated with Poly-Lactic Acid (PLA) Nanoparticles Loaded with Red Propolis (RP)" Molecules 27, no. 20: 6959. https://doi.org/10.3390/molecules27206959
APA Styleda Costa Silva, V., do Nascimento, T. G., Mergulhão, N. L. O. N., Freitas, J. D., Duarte, I. F. B., de Bulhões, L. C. G., Dornelas, C. B., de Araújo, J. X., Júnior, dos Santos, J., Silva, A. C. A., Basílio, I. D., Júnior, & Goulart, M. O. F. (2022). Development of a Polymeric Membrane Impregnated with Poly-Lactic Acid (PLA) Nanoparticles Loaded with Red Propolis (RP). Molecules, 27(20), 6959. https://doi.org/10.3390/molecules27206959