Abstract
β-cyclocitral (βCC), a main apocarotenoid of β-carotene, increases plants’ resistance against stresses. It has recently appeared as a novel bioactive composite in a variety of organisms from plants to animals. In plants, βCC marked as stress signals that accrue under adverse ecological conditions. βCC regulates nuclear gene expression through several signaling pathways, leading to stress tolerance. In this review, an attempt has been made to summarize the recent findings of the potential role of βCC. We emphasize the βCC biosynthesis, signaling, and involvement in the regulation of abiotic stresses. From this review, it is clear that discussing compound has great potential against abiotic stress tolerance and be used as photosynthetic rate enhancer. In conclusion, this review establishes a significant reference base for future research.
1. Introduction
Plants have evolved several types of secondary metabolites as a defensive shield to protect themselves from phytophagous herbivores [1]. Miscellaneous bioactive compounds induce fuel in various beneficial activities such as wound healing and antifungal, anti-inflammatory, and antimicrobial effects. For eco-friendly and substantial environments, the use of biomolecules is incrementally increasing [2]. β-cyclocitral (βCC) 2,6,6-trimethyl-1-cyclohexene-1-carboxaldehyde is an endogenous volatile compound which is derived from the carotenoid β-carotene [3]. Plant protection proficiency from injury of plants due to free radicals, stimulation of enzymes, and extinguishing singlet oxygen function is controlled by bioactive compounds [4]. The functional characterization of carotenoid compounds like hormones, signals, and biosynthesis through the non-enzymatic method is observed in apocarotenoid [5].
Carotenoids are tetraterpene pigments, which extensively distributed in foodstuffs that have always been part of the human diet. Few carotenoids can be converted into retinoids exhibiting vitamin A properties, which is important for humans. Moreover, they are very versatile as they are present in food not only as a vitamin A component, but also as natural pigment, antioxidant, and health curing compound [6]. They play an important role in lethal reactive oxygen species (ROS) scavenging and also actively participate in light absorption and the protection of photosynthetic supplies and functions [7]. The β-ring holding carotenoids like apocarotenoid obtained from carotenoid cleavage dioxygenases-4 (CCD-4 clade), which generate volatile apocarotenoids are known as βCC. It is derived from singlet oxygen 1O2 invasion, and is present in higher amounts during the chemical reactions in photosynthesis performing cells. Poly-unsaturated fatty acid oxidation is found to function as a co-substrate for performing enzymatic activities [8,9]. Interestingly, βCC contain vital apocarotenoids of β-carotene [10] that play important roles under abiotic stress conditions [11,12]. Plant genomic effect, apoptosis, gene activation processes, and transcription activation are encountered due to low RES and excess ROS oxidation [13,14,15]. Carotenoid degradation involves βCC, β-ionone, β-ionone rendered in water odor. Organoleptic properties (flowery flavor or fragrance) of βCC in beverages, pharmacy and industries are also economically beneficial [16,17]. In many plant species, βCC has been found in leaves, flowers, fruits and roots [18,19]. Moreover, a substantial source of βCC is found in lichens and mosses [20,21].
Although it acts as a powerful repellent and a signal of poor quality food to grazers, such as Daphnia [22] and the cell dissolution of Microcystis [23], βCC is beneficial to vascular plant growth. One of the major benefits of βCC is that it plays an important role in growth regulation, enhancing branching, the emergence of lateral roots, and cell division [24]. The production of provitamin carotenoid is highly dependent on βCC, which helps to improve the yield of the bioreactor and growth index [25]. The quantity of βCC is proportional to the amount of LOX 13S- lipoxygenase process which influences the decrease of apocarotenoid in Solanum lycopersicum and A. thaliana mutants in comparison with the wild type [26]. The deterioration of aquatic and marine organisms in freshwater due to carotenoid compound degeneration lead to the formation of malodor. One of the major pretenses of this odor and taste compound is βCC along with dimethyl sulfide, β-ionone, etc [27]. A number of vegetables and fruits are the sources of violet and raspberry type fragrance in carotenoid compounds.
βCC is capable of eliciting multiple stress signals and gives strength to survive under unfavorable conditions. Stress signaling of βCC is induced by oxidation and degradation of chloroplasts which enhances the production of ROS and the activity of auxin and brassinosteroids. βCC is able to promote root growth in rice, reducing the toxicity of salinity to rice seedlings [24]. Recent studies have found that βCC can enhance the tolerance of Arabidopsis thaliana to high light stress [10]. The acclimatization of photo-oxidative stress causes the inhibition of signal modulation of 1O2 through methylene blue sensitivity, which acts as a zinc finger protein. This function was directed by transcriptome reprogramming [28]. Detoxification activities for resisting oxidative cell damage are increased by βCC [10]. The βCC exhaustion is influenced by foliose lichen during heat and wounding stress [29]. It acts as a secondary precursor for stress signal relocation in A. thaliana. Analogous activities of βCC were also found to occur in dihydroactinidiolide during 1O2 oxidation [10]. The excess amount of light interference persuades the increasing amount of glycosylated βCC, which immobilizes the signaling molecule [30,31]. As a consequence, drought tolerance is achieved by signaling and pathway activation by using βCC exploitation. The exogenous application of βCC also ameliorates the drought and light stress in A. thaliana, Viola tricolor and Capsicum sp. [31]. Antioxidant signaling and crosstalk are imitated during stress, which causes the upregulation of various enzymes like superoxide dismutases (SOD), catalase (CAT) and peroxidase (POD) [32]. Inhibiting traits of βCC on hydroxylase enzymes causes multiple loop-hole creations for establishing a homeostasis pathway. The inter-connectedness between metabolic activities in primary and secondary metabolism is observed which agitate the consistent state of miscellaneous metabolites [33]. Previous studies revealed that βCC is a volatile compound derived from β-carotene oxidation, which mediates the response of cells to singlet oxygen stress. Beside these well-known examples, the latest research unraveled novel apocarotenoid growth regulators and suggested the presence of yet unidentified ones. However, knowledge of βCC involvement in the complex stress signaling network is very limited. This review highlights the structure and functions of βCC in plants. It presents the βCC mediated stress mitigation, as well as signaling cascades in plants. The authors also demonstrated the βCC-mediated involvement in the regulation of the stress response.
2. Biosynthesis of βCC
The formation of βCC occurs either by direct oxidation of β-carotene through ROS (1O2) or by an enzymatic pathway. A family of non-heme iron-dependent enzymes in plants catalyzes the carotenoids by an enzymatic cleavage via 9-cis-epoxycarotenoid cleavage dioxygenases (NCEDs) and carotenoid cleavage dioxygenases (CCDs), resulting in apocarotenoids, an oxidation product [34,35]. The first step in abscisic acid (ABA) production is catalyzed by NCED enzymes cleaving the 11, 12 (11′, 12′) double bond of 9-cis-violaxanthin or 9-cisneoxanthin [36]. Furthermore, CCD enzymes and NCED enzymes do not share cleavage specificities. In Arabidopsis, there are four CCDs (CCD1, CCD4, CCD7, and CCD8). It is unknown whether one of these CCDs creates βCC from carotene in plant leaves. In each of the four CCDs in Arabidopsis deficient mutants, the accumulation of βCC was not affected, which suggests that β-carotene oxidation mediated by CCD in this species is not a major source of this apocarotenoid [37], despite the fact that between 4 CCDs functional redundancy cannot be ruled out. This is similar in cyanobacteria, where βCC formation aided by CCD was not found [38]. Unlike CCDs that are plastidial, cytosolic CCD1 cleaves the double bonds of 9, 10 (9′, 10′) to produce varying volatiles and apocarotenoids of extensive acyclic or monocyclic apocarotenoids and carotenoids.
The strigolactones biosynthesis is dependent on CCD8 and CCD7 [39]. Since CCD4 has a specific cleavage activity at 9, 10 (9′, 10′) and 5, 6 (5′, 6′) double bond, it does not generate βCC [34,35]. Furthermore, in high light conditions, CCD4 is highly downregulated, which activates the accumulation of βCC [37]. However, the cleavage of β-carotene in citrus from the location 7, 8 (7′, 8′), CCD4b is reported under CCD4 enzyme, which results in the production of βCC [8]. Similarly, another CCD4c in the Crocus stigma from CCD4 can cleave carotenoids at 9-10 (9′, 10′), resulting into β-ionone and produces βCC with low efficiency at 7 and 8 (7′, 8′) [40]. For the production of βCC, CCD4b gene in Vitis vinifera in the carotenoid-accumulating yeast strain is also reported [41]. Another way for the oxidation of carotenoids can be provided by lipoxygenase [9]. Similarly, in leaves of Solanum lycopersicum and Arabidopsis, knockout mutants for 13-lipoxygenase LOX2 were reported to have low levels of βCC [26]. On the other hand, in the βCC accumulation under high light and 1O2 stresses, it is unknown if this enzyme is involved despite the fact that LOX2 is induced under these circumstances [37]. Eventually, from the fungus Lepista irina, extracellular fluid purified a peroxidase which produces βCC and other unstable apocarotenoids from the cleavage of β-carotene [42].
When compared to photosystem II, it is thought that photosystem I does not produce considerable amounts of 1O2. Auto-oxidation of β-carotene can also produce βCC, especially when attacked by the reactive specie 1O2 [31]. Carotenoids quench 1O2 through a physical mechanism that involves energy transfer from 1O2 to the carotenoid, followed by the excited quencher’s thermal decay [43]. However, carotene can be oxidised by 1O2, allowing 1O2 to be chemically quenched. 1O2 is an electrophilic molecule that has a strong affinity for double bonds in carotenoid molecules, oxidizing them and creating a range of apocarotenoids, including βCC [10]. In microalgae, the principal oxidation products of β-carotene are β-ionone and βCC, which release large amounts of these chemicals during summer blooms [44].
3. Derivatives of βCC
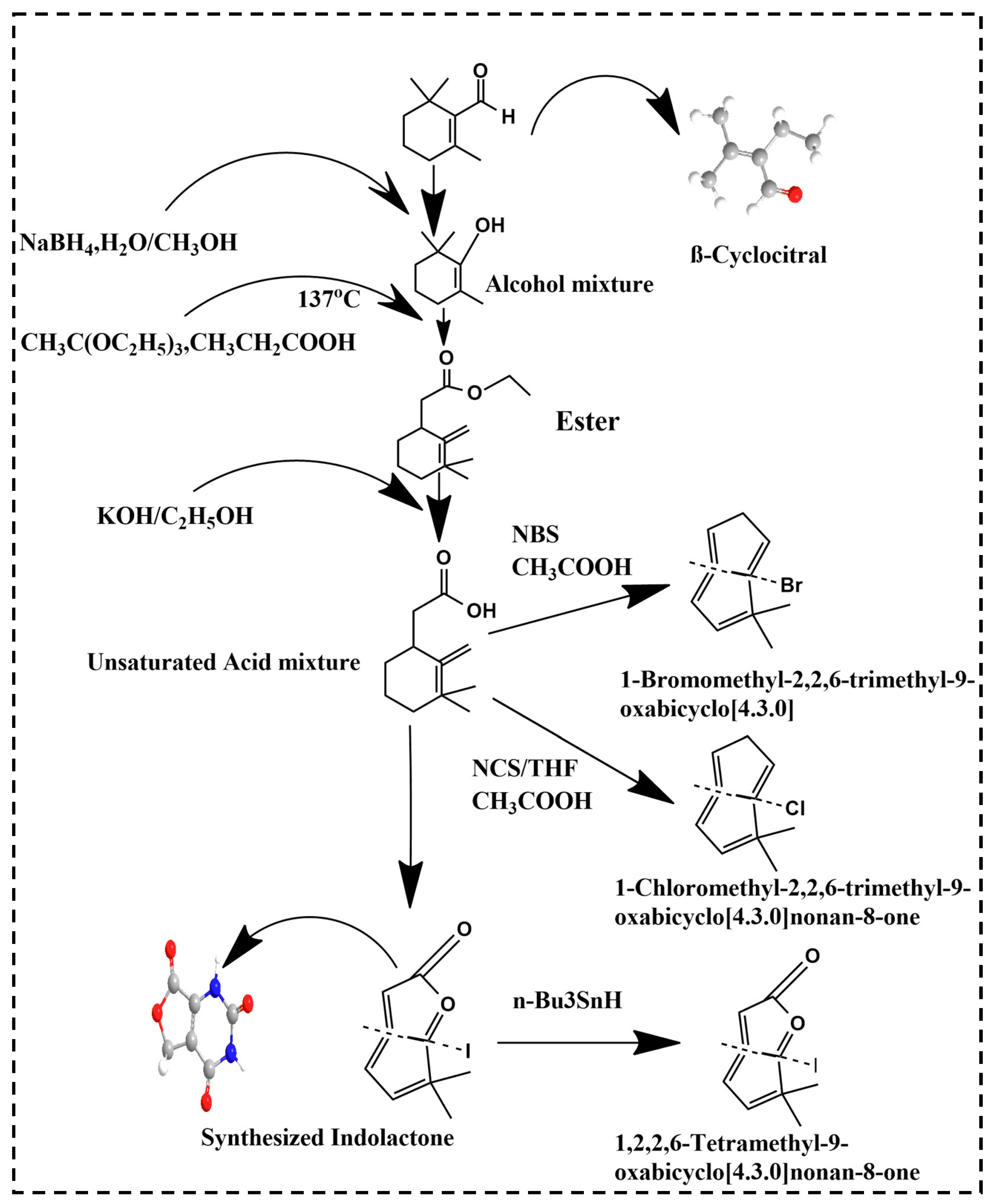
Recently, a new bioactive compound, βCC, has emerged in a variety of living organisms varying from plants and cyanobacteria to animals and fungi. It is a volatile compound consisting of a short chain of apocarotenoids produced by the non-enzymatic and enzymatic oxidation of the β-carotene. Derivatives of βCC such as lactones, β-cyclocitric acid, glycosylated βCC, 2,2,6-trimethyl-cyclohexanone and 2,6-dimethyl-cyclohexanol are briefly discussed. Lactone, being a potent biologically active compound, occurs naturally and possesses anticancer, antiplasmodial, antifungal and antimicrobial effects. Figure 1 showed the schematic synthesis of lactone. In a previous study, lactone synthesis stated by the addition of NaBH4 in the presence of H2O/CH3OH in βCC (10 g, 0.06 mol) which yielded an alcoholic compound βCC (9.65 g, yield 97%) [45]. This alcohol was treated with CH3C(OC2H5)3, CH3CH2COOH at 137 °C and resulted in (1,3,3-Trimethyl-2-methylene-cyclohex-1-yl) acetic acid ethyl ester. In the presence of KOH/C2H5OH, this acetic acid ethyl ester was transformed into (1,3,3-Trimethyl-2-methylene-cyclohexyl) acetic acid [46]. Furthermore, this acetic acid was converted to either 1-Bromomethyl-2,2,6-trimethyl-9-oxabicyclo [4.3.0] nonan-8-one with the reagent NBS/THF, CH3COOH or to 1-Chloromethyl-2,2,6-trimethyl-9-oxabicyclo [4.3.0] nonan-8-one with the help of NCS/THF, CH3COOH. It can also be converted to 1-Iodomethyl-2,2,6-trimethyl-9-oxabicyclo [4.3.0] nonan-8-one with the help of I2/KI, NaHCO3 which can be further converted to 1,2,2,6-Tetramethyl-9-oxabicyclo [4.3.0] nonan-8-one in the presence of n-Bu3SnH. The product 1,2,2,6-Tetramethyl-9-oxabicyclo [4.3.0] nonan-8-one, 1-Iodomethyl-2,2,6-trimethyl-9-oxabicyclo [4.3.0] nonan-8-one, 1-Bromomethyl-2,2,6-trimethyl-9-oxabicyclo [4.3.0] nonan-8-one and 1-Chloromethyl-2,2,6-trimethyl-9-oxabicyclo [4.3.0] nonan-8-one are all lactones derived from βCC [46].
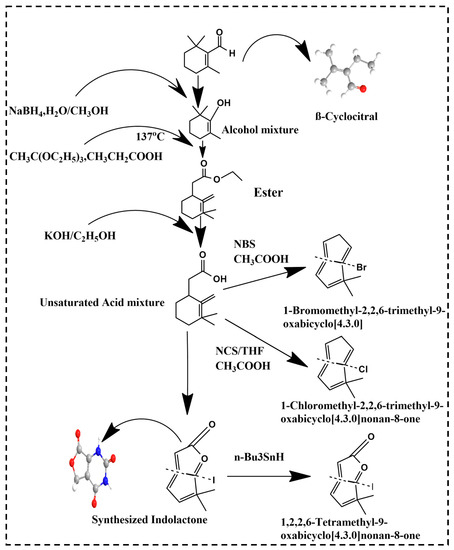
Figure 1.
Lactone synthesis from βCC.
In the leaves of Arabidopsis, the oxidation of βCC results in β-cyclocitric acid (2,2,6-trimethyl cyclohexene-1-carboxylic acid) which is responsible for the accumulation under strain conditions [31]. The level of β-cyclocitric acid in plants under drought stress increased by an aspect of 15 and doubled the content of βCC only. An accumulation of β-cyclocitric acid in plants exposed to volatile βCC was observed [31]. In the leaves and fruits of tomato, the levels of β-cyclocitric acid are remarkably more than that of βCC. Unlike in water, the conversion of βCC to β-cyclocitric acid was much faster in plants, suggesting the role of enzymatic catalysis [47]. As it remains undiscovered, there might be a connection of a Baeyer-Villiger monooxygenases that yields esters from carboxylic acids and ketones from aldehydes [48]. In brassinosteroid biosynthesis, this type of enzyme responsible for the oxidation of castasterone to brassinolide has been reported [49]. A regulatory mechanism that modulates the βCC-mediated signaling could be represented by the process of glycosylation of βCC. However, from the total βCC pool, glycosylated βCC was found to represent only a small portion (<2%) [30]. The βCC converts to various compounds like 2,2,6-trimethyl-cyclohexanone and 2,6-dimethyl-cyclohexanol after UV-light exposure [50]. In glycosylated form, βCC can occur in plants, and several glycosyl transferases are induced by the βCC treatment [10].
4. Signalling of βCC in Plants
The βCC has emerged as a new dimension for acting as a stress tolerant molecule in adverse conditions. The signaling pathway has been disclosed along with the transportation function within plants. The βCC performs in a hormone-induced marker line and the corresponding mutant responds to phytohormone pathway signals like auxin and brassinosteroids (BRs) and eventually results in cell enlargement [51]. Enzymatic action produces CCD4b from the genetic variance of Crocus sativus through β-carotene cleavage in the model plant Arabidopsis, which hastens to reduce the dehydration, salinity and oxidation rate [40,52]. The endo-metabolic substances in vascular plants took part in the xenobiotic response with diverse detoxifying agents, such as SCL14, ANAC102, ANAC001 and ANAC031 for oxidation resistance [11]. The βCC induced plants build interdependence with PAP signaling and down-regulate carotenoid substances while ST2A acts as a sulfate donor, and SAL1 has a deleterious effect during the plethora of light and drought stress [53,54]. Along with PAP, Methylerythritol cyclodiphosphate (MEcPP) substrates are also redox regularized and trigger the augmentation rate of the ROS level [55,56,57]. Sustaining photosensitivity during oxidative stress environment in mbs1 mutant crops for signaling pathway, procurement of protein and partial replacement in the nuclei occurs [28].
5. Functions of βCC in Plants
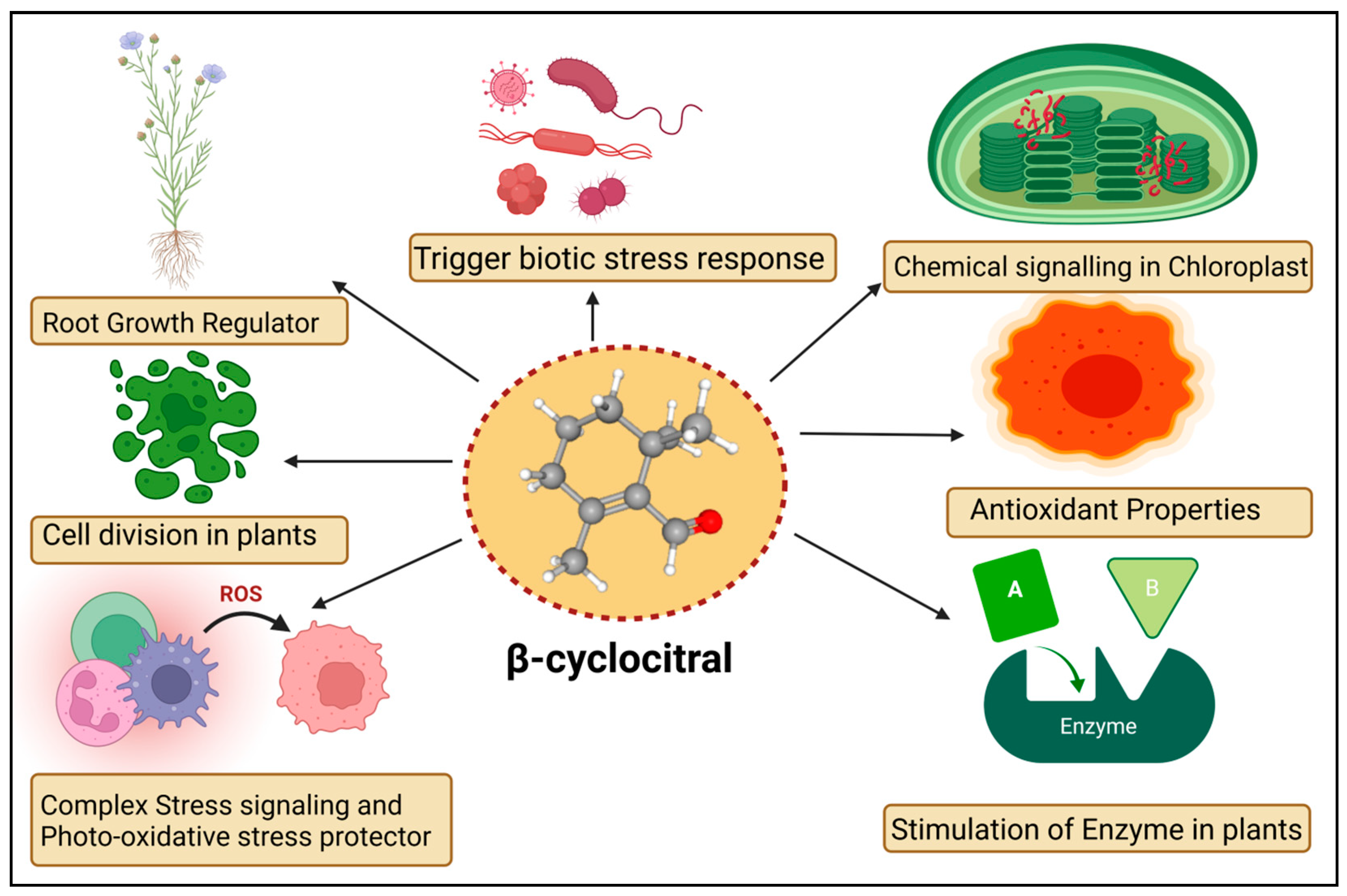
The βCC is a volatile organic compound that has been reported to have multiple functions in non-vascular plants (Figure 2). Microalgae discharge βCC, which is responsible for transferring stress signals to homogeneous algae and inducing defence. The former compound plays an allelopathic function on heterogeneous algae and aquatic macrophyte for opposite nutrients, as well as providing protection in opposition to predators [58]. The βCC has been reported to make cell rupture in Nitzschia palea, a diatom [59]. Ikawa et al. (2001) [60] reported that in cyanobacterium Microcystis, βCC is one of the main emitted volatile organic compounds. Sun et al. (2020) [61] suggested that the toxicity of βCC to cells might be associated with nuclear variation, DNA laddering, caspase 9- and caspase 3-like performance, signifying the initiation of a programmed cell death mechanism. In the case of cyanobacterial bloom, β-cyclocitric acid is produced by the oxidation of the βCC compound by Microcystis, indirectly causing toxicity. The production of this acidic compound leads to water acidification, causing chlorophyll loss, cell lysis, and phycocyanin pigment release, resulting in a characteristic blue colour [21]. These studies suggest that βCC and other volatile apocarotenoids are the principal allelopathic agents in cyanobacterial volatile organic compounds, but that at high concentrations, these compounds may be harmful to the emitters. However, no evidence has been found that low levels of βCC can elicit defence mechanisms in photosynthetic bacteria, such as those found in vascular plants. Mosses have also been observed to release volatile chemical compounds that could be used in interspecies communication [62]. Experimental evidence exhibited that photosynthetic activators and enzymatic variance treated with βCC in plants increased the photosynthetic rate, root-shoot expansion and carbon assimilation [63].
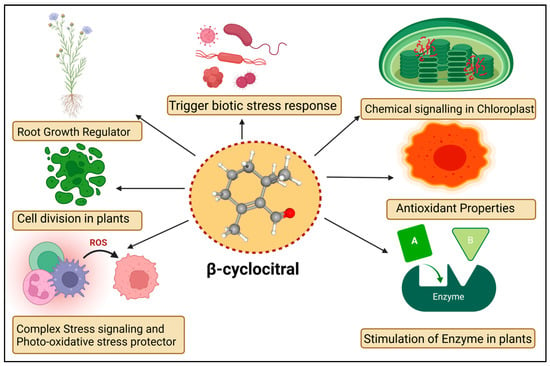
Figure 2.
Diagrammatic sketch describing different functions of β-cyclocitral in plants.
The mosses Hamatocaulis vernicosus and Sphagnum fexuosum compete for the release of volatile organic compounds via increasing emission of a molecule called methyl 2,6, 6-trimethyl-1-cyclohexene-1-carboxylate, which is chemically linked to βCC. As a result, an alarmed mechanism could be set off, signalling the competitive strength of their neighboring moss species. The former is an enzyme that can convert carotene to βCC. The βCC is an intermediary in the 1O2 signalling pathway, which controls gene expression reprogramming. It eventually causes plant cells to shift from active growth to cellular defence, resulting in stress and adaptation. The bulk of the downregulated gene encoded proteins involved in the development, growth, and biogenesis of cellular components [10]. Upregulated genes, on the other hand, were linked to environmental interactions, stress responses, and cellular mobility. Under normal or light stress conditions, βCC produces a tiny zinc finger protein (MBS1; methylene blue sensitivity 1) that is needed for the proper expression of 1O2-responsive genes [64]. The βCC is said to have increased the former protein levels while also causing the protein to relocalize to the nucleus [28]. Further research revealed that the Arabidopsis mbs1 mutant (deficient in MBS1) was insensitive to βCC and therefore lacked an increase in photo-tolerance after treatment with βCC [28]. MBS1 is thought to be downstream of βCC in the 1O2 signalling pathway, although it’s precise function remains uncertain. Exogenous reactive substances are inactivated by typical detoxifying enzymes in vascular plants, which remove these molecules in three phases. In the first stage, side groups are introduced or modified in harmful substances such as herbicides, pollutants, and so on. The modified molecule is conjugated to sugar moieties or glutathione in the second stage. Finally, inactivated chemical compounds are compartmentalized [65]. The transcriptome of βCC-treated Arabidopsis plants showed activation of detoxification pathways [66]. Several glutathione-S-transferases (GST) and UDP-glycosyltransferases (UDP-glycosyltransferases) were involved in the xenobiotic detoxification process. The GRAS protein (SCL14; SCARECROW LIKE 14) and the glutaredoxin (GRX480/ROXY19) fight for interaction with TGAII transcription factors and mediate the activation/inhibition of a large number of detoxifying enzymes during the modification phase [67]. SCL14-controlled xenobiotic detoxification was induced by βCC and photooxidative stress conditions. Similarly, the scl14 knockout mutant did not respond to βCC and remained susceptible to high light stress even after treatment with βCC [68]. A few signalling cascade components downstream of βCC have been identified. The SCL14/TGA II complex, in particular, modulates the transcriptional levels of a transcription factor that regulates other downstream transcription regulators and, eventually, the redox enzymes of the first phase of the detoxification response [68], thus improving cellular detoxification capabilities. Surprisingly, the loss of MBS1 had no effect on βCC-induced cellular detoxification, indicating that there are two pathways in βCC signalling, one regulated by SCL14 and the other dependent on MBS1 [68]. Lipid peroxidation produces the chemicals that are characteristic of photooxidation and contributes unwaveringly to its toxicity. They decompose into reactive aldehydes (acrolein or 4-hydroxynonenal), which obstruct macromolecule function and cause cell death [69]. Diversified functions of βCC in plants are given in Table 1.
In plants including Solanum lycopersicum, Piper nigrum, and Arabidopsis thaliana, chemical βCC can cause changes in gene expression and promote drought tolerance [70]. The available literature on βCC shows that it may activate a signalling cascade that has yet to be fully described. Dickinson et al. (2019) [24] found that Arabidopsis seedlings grown in Petri plates treated with βCC stimulated the growth of primary roots. Increased root lengths may be beneficial under salt and water stress conditions, allowing for better soil exploration and water uptake. βCC has the ability to influence root development in Solanum lycopersicum and Oryza sativa without relying on auxin or brassinosteroid signalling. However, it is unclear whether βCC causes root growth directly through cell division and elongation or indirectly through the activation of cellular detoxification and resistance to oxidative stress. The molecular processes underlying βCC control of root development will need to be clarified in future research [31].

Table 1.
Various functions of βCC in Plants.
6. βCC Involved in the Regulation of Stress Response
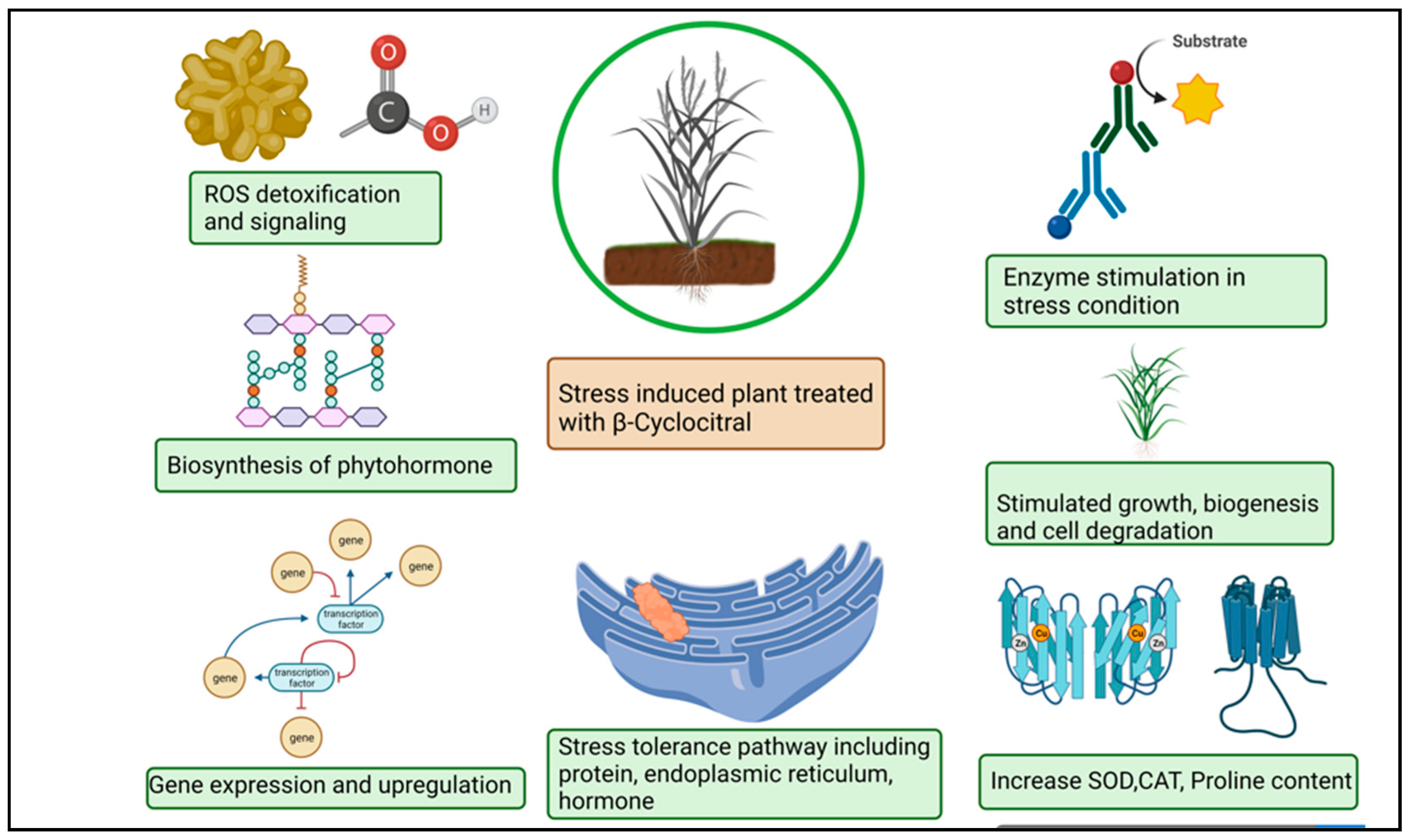
The βCC has recently emerged as a unique plant signal in vascular and non-vascular plants that triggers stress tolerance (Figure 3). Ramel et al. (2012) [10] found that βCC increased tolerance to drought stress and high light-induced oxidative stress in Arabidopsis thaliana by altering the expression of several nuclear-encoded genes. Overexpression of the Crocus CCD4b gene in Arabidopsis confers tolerance to environmental and oxidative stressors, according to Baba et al. (2015) [52]. βCC plays important role in drought stress tolerance. Drought stress hampers photosynthesis, stomatal conductance, and the respiration and transduction system. In plants, leaf senescence, cell expansion, and yield significantly decreased under drought stress [78,79]. ROS provokes drought stress which puts the oxidation process, assimilation and antioxidant systems at risk [80]. The correlation between βCC and ROS during drought merely demonstrated that increase the rate of βCC aid the reduction of β-carotene in Solanum lycopersicum. Simulating stress tolerance, SOD and CAT upregulation altered transcription levels in plants during drought stress (Figure 3). The 1O2 simulate as a signaling particle, attacking lipid peroxidation within stressed plants [81,82]. In Arabidopsis thaliana, elevated levels of singlet oxygen-regulated genes (SORGs) are a crucial indicator in the early stages of stress. Dispensation of βCC or dhA protein aid in lowering photoinhibition in comparison with plants grown in a control or natural stress environment βCC acts as a secondary messenger imparting towards the nucleus through methylene blue sensitive [28]. The uncoupling of chlorophyll compound produces ROS accordingly. The plethora of light stress in the environment decrement the pace of the photosynthetic electron transport chain (PETC) immobilizes the photosystem and inhibits photosynthesis [83,84]. Photoinhibition, incompatibility of photochemical reaction and the D1 protein in photodamage system degrade [85,86]. A copious amount of light energy is mechanized with a non-photochemical extinguishing method that stimulates 1O2 production in stress conditions and 1O2 mitigation in the dark light [87]. The excessive re-occurrence of mutagenesis took place in the chlorophyll content [88]. Signaling molecules helps to overcome photodegradation in the light stress condition using a wide range of carotenoid content. In high light stress, carotenoid oxidation took place in PS–II which alters the nuclear gene expression [89]. Moreover, low temperature caused the progressive mitigation of βCC content and the substantial increase in H2O2 in Solanum lycopersicum [90,91]. Plant reaction after treatment with βCC under stress condition is described in Table 2.
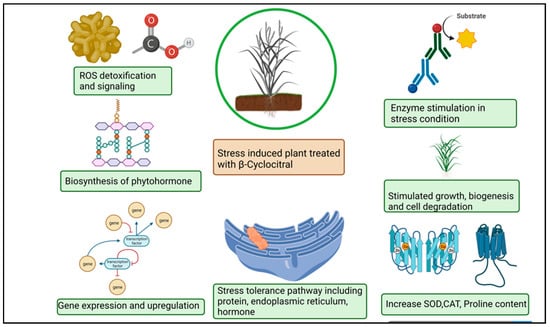
Figure 3.
Schematic representation of β-cyclocitral effects on plants under abiotic stress.
Salt stress hampers plant growth amplification, ionic balance, nutritional symmetry and stomatal function in shoots [92,93,94]. Water absorption blockage, water potential alleviation, oxidative stress occurrence and programmed cell death (PCD) strike the plants functioning during salinity stress [95]. Executer 1 and 2 proteins are accountable for cell death in each route [82]. Enhanced root ontogenesis illustrates a favorable impact on salinity stress via βCC application. Rice and Arabidopsis roots in considerable depth stimulate plant vigor and genomic functions which help to defeat stress through nuclear transportation and ion activation [24]. In the presence of carotenoid compounds, the S. lycopersicum crop demonstrated enhanced oxidation and biochemical synthesis [31]. The inhibition of curly leaves, prevention of leaves wilting, ameliorating relative water content (RWC) and the stomatal opening is accelerated in βCC treated plants [7]. However, the functioning mechanism of βCC in plants has yet to be extensively studied in both long and short-duration experiments. Photo-oxidative stress is a consecutive production of ROS in a pattern which is harmful to the antioxidant defense system. As a result, the photo-oxidation process continues to develop toward a pessimistic function, hazardous chemicals get acclimated, and chloroplast damage happens and eventually yields losses [96]. Photo-oxidation took place in leaves, ontogeny led to senescence and oxidative cascade occurred under stressed conditions [97,98]. The abundance of stress-tolerant genes becomes active in the raised ROS [99,100,101]. In the presence of elevated ROS, singlet oxygen oxidizes promptly in the environment and occurs in PS-II complexes, enabling photochemical quenching of 3Chl* transfer [102]. ROS is responsible for leaf damage, mitigating photosynthesis competence, photoproduction and photo-oxidative injury [6,70,103,104,105]. Lichens and cyanobacterial compounds contain reactive radicals in the physiologically active stress condition [29].

Table 2.
βCC mediated abiotic alteration in plants.
7. Conclusions and Future Perspective
As an emerging molecule, βCC has been gaining increasing interest to provide stress tolerance due to their excellent features resulting from the altering of the expression of several nuclear-encoded genes. In this work, novel apocarotenoids of β-carotene and βCC applications for stress tolerance were systematically reviewed. Their biosynthesis, derivatives and signalling were discussed. Furthermore, the promising functions of βCC were also discussed and potential directions for future work were suggested.
Author Contributions
Conceptualization, M.F. and P.A.; resources, M.F., S.H.T. and S.A.; writing-original draft preparation, M.F., S.H.T. and S.A.; writing-review and editing, Z.F., S.M.A., F.Y. and S.H.; visualization, P.A. and S.H., supervision, S.H. and S.M.A.; funding acquisition, P.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Bernhoft, A. A brief review on bioactive compounds in plants. In Bioactive Compounds in Plants—Benefits and Risks for Man and Animals; The Norwegian Academy of Science and Letters: Oslo, Norway, 2010; Volume 50, pp. 11–17. [Google Scholar]
- Sasidharan, S.; Chen, Y.; Saravanan, D.; Sundram, K.M.; Latha, L.Y. Extraction, isolation and characterization of bioactive compounds from plants’ extracts. Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S. Plants: A rich source of herbal medicine. J. Nat. Prod. 2008, 1, 27–35. [Google Scholar]
- Couladis, M.; Tzakou, O.; Verykokidou, E.; Harvala, C. Screening of some Greek aromatic plants for antioxidant activity. Phyther. Res. 2003, 17, 194–195. [Google Scholar] [CrossRef] [PubMed]
- Havaux, M. Carotenoid oxidation products as stress signals in plants. Plant J. 2014, 79, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Galasso, C.; Corinaldesi, C.; Sansone, C. Carotenoids from marine organisms: Biological functions and inductrial applications. Antioxidants 2017, 6, 96. [Google Scholar] [CrossRef]
- Deshpande, S.; Manoharan, R.; Mitra, S. Exogenous β-cyclocitral treatment primes tomato plants against drought by inducing tolerance traits, independent of abscisic acid. Plant Biol. 2021, 23, 170–180. [Google Scholar] [CrossRef]
- Rodrigo, M.J.; Alquézar, B.; Alós, E.; Medina, V.; Carmona, L.; Bruno, M.; Al-Babili, S.; Zacarías, L. A novel carotenoid cleavage activity involved in the biosynthesis of Citrus fruit-specific apocarotenoid pigments. J. Exp. Bot. 2013, 64, 4461–4478. [Google Scholar] [CrossRef]
- Hayward, S.; Cilliers, T.; Swart, P. Lipoxygenases: From isolation to application. Compr. Rev. Food Sci. Food Saf. 2017, 16, 199–211. [Google Scholar] [CrossRef]
- Ramel, F.; Birtic, S.; Ginies, C.; Soubigou-Taconnat, L.; Triantaphylidès, C.; Havaux, M. Carotenoid oxidation products are stress signals that mediate gene responses to singlet oxygen in plants. Proc. Natl. Acad. Sci. USA 2012, 109, 5535–5540. [Google Scholar] [CrossRef]
- Moretto, J.A.S.; de Freitas, P.N.N.; de Almeida, E.C.; Altarugio, L.M.; da Silva, S.V.; Fiore, M.F.; Pinto, E. Effects of different cultivation conditions on the production of β-cyclocitral and β-ionone in Microcystis aeruginosa. BMC Microbiol. 2022, 22, 78. [Google Scholar] [CrossRef]
- Mitra, S.; Estrada-Tejedor, R.; Volke, D.C.; Phillips, M.A.; Gershenzon, J.; Wright, L.P. Negative regulation of plastidial isoprenoid pathway by herbivore-induced β-cyclocitral in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2021, 118, e2008747118. [Google Scholar] [CrossRef] [PubMed]
- Sharoni, Y.; Danilenko, M.; Dubi, N.; Ben-Dor, A.; Levy, J. Carotenoids and transcription. Arch. Biochem. Biophys. 2004, 430, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, E.; Hoeller, U.; Greatrix, B.; Lankin, C.; Seifert, N.; Acharya, S.; Riss, G.; Buchwald-Hunziker, P.; Hunziker, W.; Goralczyk, R. β-Carotene and apocarotenals promote retinoid signaling in BEAS-2B human bronchioepithelial cells. Arch. Biochem. Biophys. 2006, 455, 48–60. [Google Scholar] [CrossRef]
- Liu, J.; Sun, X.; Dong, H.; Sun, C.; Sun, W.; Chen, B.; Song, Y.; Yang, B. β-Ionone suppresses mammary carcinogenesis, proliferative activity and induces apoptosis in the mammary gland of the Sprague-Dawley rat. Int. J. Cancer 2008, 122, 2689–2698. [Google Scholar] [CrossRef] [PubMed]
- Mahattanatawee, K.; Rouseff, R.; Valim, M.F.; Naim, M. Identification and aroma impact of norisoprenoids in orange juice. J. Agric. Food Chem. 2005, 53, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Lalko, J.; Lapczynski, A.; McGinty, D.; Bhatia, S.; Letizia, C.S.; Api, A.M. Fragrance material review on ionone. Food Chem. Toxicol. 2007, 45, S251–S257. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Shi, C.; Ji, M.; Xu, X.; Zhang, Z.; Ma, J.; Wang, G. Taste and odor compounds associated with aquatic plants in Taihu Lake: Distribution and producing potential. Environ. Sci. Pollut. Res. 2019, 26, 34510–34520. [Google Scholar] [CrossRef]
- Moraga, Á.R.; Rambla, J.L.; Ahrazem, O.; Granell, A.; Gómez-Gómez, L. Metabolite and target transcript analyses during Crocus sativus stigma development. Phytochemistry 2009, 70, 1009–1016. [Google Scholar] [CrossRef]
- Lewinsohn, E.; Sitrit, Y.; Bar, E.; Azulay, Y.; Meir, A.; Zamir, D.; Tadmor, Y. Carotenoid pigmentation affects the volatile composition of tomato and watermelon fruits, as revealed by comparative genetic analyses. J. Agric. Food Chem. 2005, 53, 3142–3148. [Google Scholar] [CrossRef]
- Arii, S.; Tsuji, K.; Tomita, K.; Hasegawa, M.; Bober, B.; Harada, K.-I. Cyanobacterial blue color formation during lysis under natural conditions. Appl. Environ. Microbiol. 2015, 81, 2667–2675. [Google Scholar] [CrossRef]
- Juttner, F.; Watson, S.B.; von Elert, E.; Koster, O.J. β-cyclocitral, a grazer defense signal unique to the cyanobacterium Microcystis. Chem. Ecol. 2010, 36, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, K.; Ohta, A.; Iwata, C.; Horikawa, A.; Tsuji, K.; Ito, E.; Ikai, Y.; Harada, K.I. Lysis of cyanobacteria with volatile organic compounds. Chemosphere 2008, 71, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, A.J.; Lehner, K.; Mi, J.; Jia, K.-P.; Mijar, M.; Dinneny, J.; Al-Babili, S.; Benfey, P.N. β-Cyclocitral is a conserved root growth regulator. Proc. Natl. Acad. Sci. USA 2019, 116, 10563–10567. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Zhu, K.; Ye, J.; Price, E.J.; Deng, X.; Fraser, P.D. The effect of β-cyclocitral treatment on the carotenoid content of transgenic Marsh grapefruit (Citrus paradisi Macf.) suspension-cultured cells. Phytochemistry 2020, 180, 112509. [Google Scholar] [CrossRef] [PubMed]
- Miras-Moreno, B.; Pedreño, M.A.; Fraser, P.D.; Sabater-Jara, A.B.; Almagro, L. Effect of diflufenican on total carotenoid and phytoene production in carrot suspension-cultured cells. Planta 2019, 249, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Havaux, M. β-Cyclocitral and Derivatives: Emerging Molecular Signals Serving Multiple Biological Functions. Plant Physiol. Biochem. 2020, 155, 35–41. [Google Scholar] [CrossRef]
- Shumbe, L.; d’Alessandro, S.; Shao, N.; Chevalier, A.; Ksas, B.; Bock, R.; Havaux, M. Methylene Blue Sensitivity 1 (MBS1) is required for acclimation of Arabidopsis to singlet oxygen and acts downstream of β-cyclocitral. Plant Cell Environ. 2017, 40, 216–226. [Google Scholar] [CrossRef]
- García-Plazaola, J.I.; Portillo-Estrada, M.; Fernández-Marín, B.; Kännaste, A.; Niinemets, Ü. Emissions of carotenoid cleavage products upon heat shock and mechanical wounding from a foliose lichen. Environ. Exp. Bot. 2017, 133, 87–97. [Google Scholar] [CrossRef]
- Swapnil, P.; Meena, M.; Singh, S.K.; Dhuldhaj, U.P.; Harish Marwal, A. Vital roles of carotenoids in plants and humans to deteriorate stress with its structure, biosynthesis, metabolic engineering and functional aspects. Curr. Plant Biol. 2021, 26, 100203. [Google Scholar] [CrossRef]
- D’Alessandro, S.; Havaux, M. Sensing β-carotene oxidation in photosystem II to master plant stress tolerance. New Phytol. 2019, 223, 1776–1783. [Google Scholar] [CrossRef]
- Noctor, G.; Arisi, A.-C.M.; Jouanin, L.; Kunert, K.J.; Rennenberg, H.; Foyer, C.H. Glutathione: Biosynthesis, metabolism and relationship to stress tolerance explored in transformed plants. J. Exp. Bot. 1998, 49, 623–647. [Google Scholar] [CrossRef]
- Jamaluddin, N.D.; Rohani, E.R.; Mohd Noor, N.; Goh, H.-H. Transcriptome-wide effect of DE-ETIOLATED1 (DET1) suppression in embryogenic callus of Carica papaya. J. Plant Res. 2019, 132, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, G.; Al-Babili, S.; Von Lintig, J. Carotenoid oxygenases: Cleave it or leave it. Trends Plant Sci. 2003, 8, 145–149. [Google Scholar] [CrossRef]
- Hou, X.; Rivers, J.; León, P.; McQuinn, R.P.; Pogson, B.J. Synthesis and function of apocarotenoid signals in plants. Trends Plant Sci. 2016, 21, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Nambara, E.; Marion-Poll, A. Abscisic acid biosynthesis and catabolism. Annu. Rev. Plant Biol. 2005, 56, 165. [Google Scholar] [CrossRef]
- Ramel, F.; Mialoundama, A.S.; Havaux, M. Nonenzymic carotenoid oxidation and photooxidative stress signalling in plants. J. Exp. Bot. 2013, 64, 799–805. [Google Scholar] [CrossRef]
- Cui, H.; Wang, Y.; Qin, S. Genomewide analysis of carotenoid cleavage dioxygenases in unicellular and filamentous cyanobacteria. Comp. Funct. Genom. 2012, 2012, 164690. [Google Scholar] [CrossRef]
- Al-Babili, S.; Bouwmeester, H.J. Strigolactones, a novel carotenoid-derived plant hormone. Annu. Rev. Plant Biol. 2015, 66, 161–186. [Google Scholar] [CrossRef]
- Rubio-Moraga, A.; Rambla, J.L.; Fernández-de-Carmen, A.; Trapero-Mozos, A.; Ahrazem, O.; Orzáez, D.; Granell, A.; Gómez-Gómez, L. New target carotenoids for CCD4 enzymes are revealed with the characterization of a novel stress-induced carotenoid cleavage dioxygenase gene from Crocus sativus. Plant Mol. Biol. 2014, 86, 555–569. [Google Scholar] [CrossRef]
- Meng, N.; Yan, G.-L.; Zhang, D.; Li, X.-Y.; Duan, C.-Q.; Pan, Q.-H. Characterization of two Vitis vinifera carotenoid cleavage dioxygenases by heterologous expression in Saccharomyces cerevisiae. Mol. Biol. Rep. 2019, 46, 6311–6323. [Google Scholar] [CrossRef]
- Zorn, H.; Langhoff, S.; Scheibner, M.; Nimtz, M.; Berger, R.G. A peroxidase from Lepista irina cleaves β, β-carotene to flavor compounds. Biol. Chem. 2003, 384, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Edge, R.; McGarvey, D.J.; Truscott, T.G. The carotenoids as anti-oxidants—A review. J. Photochem. Photobiol. B Biol. 1997, 41, 189–200. [Google Scholar] [CrossRef]
- Ma, Z.; Niu, Y.; Xie, P.; Chen, J.; Tao, M.; Deng, X. Off-flavor compounds from decaying cyanobacterial blooms of Lake Taihu. J. Environ. Sci. 2013, 25, 495–501. [Google Scholar] [CrossRef]
- Crombie, B.S.; Smith, C.; Varnavas, C.Z.; Wallace, T.W. A conjugate addition–radical cyclisation approach to sesquiterpene-phenol natural products. J. Chem. Soc. Perkin Trans. 2001, 1, 206–215. [Google Scholar] [CrossRef]
- Mazur, M.; Gładkowski, W.; Podkowik, M.; Bania, J.; Nawrot, J.; Białońska, A.; Wawrzeńczyk, C. Lactones 43. New biologically active lactones: β-cyclocitral derivatives. Pest Manag. Sci. 2014, 70, 286–294. [Google Scholar] [CrossRef]
- Tomita, K.; Hasegawa, M.; Arii, S.; Tsuji, K.; Bober, B.; Harada, K. Characteristic oxidation behavior of β-cyclocitral from the cyanobacterium Microcystis. Environ. Sci. Pollut. Res. 2016, 23, 11998–12006. [Google Scholar] [CrossRef]
- Van Berkel, W.J.H.; Kamerbeek, N.M.; Fraaije, M. Flavoprotein monooxygenases, a diverse class of oxidative biocatalysts. J. Biotechnol. 2006, 124, 670–689. [Google Scholar] [CrossRef]
- Kim, T.-W.; Hwang, J.-Y.; Kim, Y.-S.; Joo, S.-H.; Chang, S.C.; Lee, J.S.; Takatsuto, S.; Kim, S.-K. Arabidopsis CYP85A2, a cytochrome P450, mediates the Baeyer-Villiger oxidation of castasterone to brassinolide in brassinosteroid biosynthesis. Plant Cell 2005, 17, 2397–2412. [Google Scholar] [CrossRef]
- Kim, T.; Kim, T.-K.; Zoh, K.-D. Degradation kinetics and pathways of β-cyclocitral and β-ionone during UV photolysis and UV/chlorination reactions. J. Environ. Manag. 2019, 239, 8–16. [Google Scholar] [CrossRef]
- Felemban, A.; Braguy, J.; Zurbriggen, M.D.; Al-Babili, S. Apocarotenoids involved in plant development and stress response. Front. Plant Sci. 2019, 10, 1168. [Google Scholar] [CrossRef]
- Baba, S.A.; Jain, D.; Abbas, N.; Ashraf, N. Overexpression of Crocus carotenoid cleavage dioxygenase, CsCCD4b, in Arabidopsis imparts tolerance to dehydration, salt and oxidative stresses by modulating ROS machinery. J. Plant Physiol. 2015, 189, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Estavillo, G.M.; Crisp, P.A.; Pornsiriwong, W.; Wirtz, M.; Collinge, D.; Carrie, C.; Giraud, E.; Whelan, J.; David, P.; Javot, H. Evidence for a SAL1-PAP chloroplast retrograde pathway that functions in drought and high light signaling in Arabidopsis. Plant Cell 2011, 23, 3992–4012. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.B.; Estavillo, G.M.; Field, K.J.; Pornsiriwong, W.; Carroll, A.J.; Howell, K.A.; Woo, N.S.; Lake, J.A.; Smith, S.M.; Harvey Millar, A. The nucleotidase/phosphatase SAL1 is a negative regulator of drought tolerance in Arabidopsis. Plant J. 2009, 58, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Rossel, J.B.; Walter, P.B.; Hendrickson, L.; Chow, W.S.; Poole, A.; Mullineaux, P.M.; Pogson, B.J. A mutation affecting ASCORBATE PEROXIDASE 2 gene expression reveals a link between responses to high light and drought tolerance. Plant Cell Environ. 2006, 29, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Von Arnim, A.G. FIERY1 regulates light-mediated repression of cell elongation and flowering time via its 3′(2′), 5′-bisphosphate nucleotidase activity. Plant J. 2009, 58, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Sharkey, T.D. Metabolic profiling of the methylerythritol phosphate pathway reveals the source of post-illumination isoprene burst from leaves. Plant Cell Environ. 2013, 36, 429–437. [Google Scholar] [CrossRef]
- Zuo, Z. Why algae release volatile organic compounds—The emission and roles. Front. Microbiol. 2019, 10, 491. [Google Scholar] [CrossRef]
- Chang, D.W.; Hsieh, M.-L.; Chen, Y.-M.; Lin, T.-F.; Chang, J.-S. Kinetics of cell lysis for Microcystis aeruginosa and Nitzschia palea in the exposure to β-cyclocitral. J. Hazard. Mater. 2011, 185, 1214–1220. [Google Scholar] [CrossRef]
- Ikawa, M.; Sasner, J.J.; Haney, J.F. Activity of cyanobacterial and algal odor compounds found in lake waters on green alga Chlorella pyrenoidosa growth. Hydrobiologia 2001, 443, 19–22. [Google Scholar] [CrossRef]
- Sun, Q.; Zhou, M.; Zuo, Z. Toxic mechanism of eucalyptol and β-cyclocitral on Chlamydomonas reinhardtii by inducing programmed cell death. J. Hazard. Mater. 2020, 389, 121910. [Google Scholar] [CrossRef]
- Saritas, Y.; Sonwa, M.M.; Iznaguen, H.; König, W.A.; Muhle, H.; Mues, R. Volatile constituents in mosses (Musci). Phytochemistry 2001, 57, 443–457. [Google Scholar] [CrossRef]
- Schwachtje, J.; Minchin, P.E.H.; Jahnke, S.; van Dongen, J.T.; Schittko, U.; Baldwin, I.T. SNF1-related kinases allow plants to tolerate herbivory by allocating carbon to roots. Proc. Natl. Acad. Sci. USA 2006, 103, 12935–12940. [Google Scholar] [CrossRef] [PubMed]
- Shao, N.; Duan, G.Y.; Bock, R. A mediator of singlet oxygen responses in Chlamydomonas reinhardtii and Arabidopsis identified by a luciferase-based genetic screen in algal cells. Plant Cell 2013, 25, 4209–4226. [Google Scholar] [CrossRef] [PubMed]
- Riechers, D.E.; Kreuz, K.; Zhang, Q. Detoxification without intoxication: Herbicide safeners activate plant defense gene expression. Plant Physiol. 2010, 153, 3–13. [Google Scholar] [CrossRef]
- Sandermann, H., Jr. Plant metabolism of xenobiotics. Trends Biochem. Sci. 1992, 17, 82–84. [Google Scholar] [CrossRef]
- Huang, L.J.; Li, N.; Thurow, C.; Wirtz, M.; Hell, R.; Gatz, C. Ectopically expressed glutaredoxin ROXY19 negatively regulates the detoxification pathway in Arabidopsis thaliana. BMC Plant Biol. 2016, 16, 200. [Google Scholar] [CrossRef] [PubMed]
- D’alessandro, S.; Ksas, B.; Havaux, M. Decoding β-cyclocitral-mediated retrograde signaling reveals the role of a detoxification response in plant tolerance to photooxidative stress. Plant Cell 2018, 30, 2495–2511. [Google Scholar] [CrossRef]
- Mano, J.; Biswas, M.S.; Sugimoto, K. Reactive carbonyl species: A missing link in ROS signaling. Plants 2019, 8, 391. [Google Scholar] [CrossRef]
- D’Alessandro, S.; Mizokami, Y.; Légeret, B.; Havaux, M. The apocarotenoid β-cyclocitric acid elicits drought tolerance in plants. iScience 2019, 19, 461–473. [Google Scholar] [CrossRef]
- Xu, Q.; Yang, L.; Yang, W.; Bai, Y.; Hou, P.; Zhao, J.; Zhou, L.; Zuo, Z. Volatile organic compounds released from Microcystis flos-aquae under nitrogen sources and their toxic effects on Chlorella vulgaris. Ecotoxicol. Environ. Saf. 2017, 135, 191–200. [Google Scholar] [CrossRef]
- Hasegawa, M.; Nishizawa, A.; Tsuji, K.; Kimura, S.; Harada, K. Volatile organic compounds derived from 2-keto-acid decarboxylase in Microcystis aeruginosa. Microbes Environ. 2012, 27, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Tieman, D.; Bliss, P.; McIntyre, L.M.; Blandon-Ubeda, A.; Bies, D.; Odabasi, A.Z.; Rodríguez, G.R.; van der Knaap, E.; Taylor, M.G.; Goulet, C. The chemical interactions underlying tomato flavor preferences. Curr. Biol. 2012, 22, 1035–1039. [Google Scholar] [CrossRef] [PubMed]
- Hinge, V.; Patil, H.; Nadaf, A. Comparative characterization of aroma volatiles and related gene expression analysis at vegetative and mature stages in basmati and non-basmati rice (Oryza sativa L.) cultivars. Appl. Biochem. Biotechnol. 2016, 178, 619–639. [Google Scholar] [CrossRef] [PubMed]
- Linde, G.A.; Gazim, Z.C.; Cardoso, B.K.; Jorge, L.F.; Tešević, V.; Glamočlija, J.; Soković, M.; Colauto, N.B. Antifungal and antibacterial activities of Petroselinum crispum essential oil. Genet. Mol. Res. 2016, 15, gmr.15038538. [Google Scholar] [CrossRef] [PubMed]
- Ojha, P.K.; Roy, K. PLS regression-based chemometric modeling of odorant properties of diverse chemical constituents of black tea and coffee. RSC Adv. 2018, 8, 2293–2304. [Google Scholar] [CrossRef] [PubMed]
- Lazazzara, V.; Bueschl, C.; Parich, A.; Pertot, I.; Schuhmacher, R.; Perazzolli, M. Downy mildew symptoms on grapevines can be reduced by volatile organic compounds of resistant genotypes. Sci. Rep. 2018, 8, 1618. [Google Scholar] [CrossRef] [PubMed]
- Misra, V.; Solomon, S.; Mall, A.K.; Prajapati, C.P.; Hashem, A.; Abd_Allah, E.F.; Ansari, M.I. Morphological assessment of water stressed sugarcane: A comparison of waterlogged and drought affected crop. Saudi J. Biol. Sci. 2020, 27, 1228–1236. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B.; Zohaib, A.; Abbas, F.; Saleem, M.F.; Ali, I. Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front. Plant Sci. 2017, 8, 69. [Google Scholar] [CrossRef]
- Tanveer, M.; Shahzad, B.; Sharma, A.; Khan, E.A. 24-Epibrassinolide application in plants: An implication for improving drought stress tolerance in plants. Plant Physiol. Biochem. 2019, 135, 295–303. [Google Scholar] [CrossRef]
- Laloi, C.; Havaux, M. Key players of singlet oxygen-induced cell death in plants. Front. Plant Sci. 2015, 6, 39. [Google Scholar] [CrossRef]
- Dogra, V.; Rochaix, J.; Kim, C. Singlet oxygen-triggered chloroplast-to-nucleus retrograde signalling pathways: An emerging perspective. Plant. Cell Environ. 2018, 41, 1727–1738. [Google Scholar] [CrossRef] [PubMed]
- Murata, N.; Takahashi, S.; Nishiyama, Y.; Allakhverdiev, S.I. Photoinhibition of photosystem II under environmental stress. Biochim. Biophys. Acta (BBA)-Bioenerg. 2007, 1767, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Roach, T.; Krieger-Liszkay, A. Regulation of photosynthetic electron transport and photoinhibition. Curr. Protein Pept. Sci. 2014, 15, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Aro, E.M.; Suorsa, M.; Rokka, A.; Allahverdiyeva, Y.; Paakkarinen, V.; Saleem, A.; Battchikova, N.; Rintamäki, E. Dynamics of photosystem II: A proteomic approach to thylakoid protein complexes. J. Exp. Bot. 2005, 56, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Murata, N. How do environmental stresses accelerate photoinhibition? Trends Plant Sci. 2008, 13, 178–182. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Garab, G.; Adams III, W.; Govindjee, U. Non-Photochemical Quenching and Energy Dissipation in Plants, Algae and Cyanobacteria; Springer: Dordrecht, The Netherlands, 2014. [Google Scholar]
- Ouchane, S.; Picaud, M.; Vernotte, C.; Astier, C. Photooxidative stress stimulates illegitimate recombination and mutability in carotenoid-less mutants of Rubrivivax gelatinosus. EMBO J. 1997, 16, 4777–4787. [Google Scholar] [CrossRef]
- Shumbe, L.; Bott, R.; Havaux, M. Dihydroactinidiolide, a high light-induced β-carotene derivative that can regulate gene expression and photoacclimation in Arabidopsis. Mol. Plant 2014, 7, 1248–1251. [Google Scholar] [CrossRef]
- Liu, T.; Ye, X.; Li, M.; Li, J.; Qi, H.; Hu, X. H2O2 and NO are involved in trehalose-regulated oxidative stress tolerance in cold-stressed tomato plants. Environ. Exp. Bot. 2020, 171, 103961. [Google Scholar] [CrossRef]
- Khan, T.A.; Fariduddin, Q.; Yusuf, M. Lycopersicon esculentum under low temperature stress: An approach toward enhanced antioxidants and yield. Environ. Sci. Pollut. Res. 2015, 22, 14178–14188. [Google Scholar] [CrossRef]
- Hernández, J.A.; Ferrer, M.A.; Jiménez, A.; Barceló, A.R.; Sevilla, F. Antioxidant systems and O2−/H2O2 production in the apoplast of pea leaves. Its relation with salt-induced necrotic lesions in minor veins. Plant Physiol. 2001, 127, 817–831. [Google Scholar] [CrossRef]
- Isayenkov, S.V. Physiological and molecular aspects of salt stress in plants. Cytol. Genet. 2012, 46, 302–318. [Google Scholar] [CrossRef]
- Rajendran, K.; Tester, M.; Roy, S.J. Quantifying the three main components of salinity tolerance in cereals. Plant. Cell Environ. 2009, 32, 237–249. [Google Scholar] [CrossRef]
- Demidchik, V.; Cuin, T.A.; Svistunenko, D.; Smith, S.J.; Miller, A.J.; Shabala, S.; Sokolik, A.; Yurin, V. Arabidopsis root K+-efflux conductance activated by hydroxyl radicals: Single-channel properties, genetic basis and involvement in stress-induced cell death. J. Cell Sci. 2010, 123, 1468–1479. [Google Scholar] [CrossRef] [PubMed]
- Borisova, M.M.M.; Kozuleva, M.A.; Rudenko, N.N.; Naydov, I.A.; Klenina, I.B.; Ivanov, B.N. Photosynthetic electron flow to oxygen and diffusion of hydrogen peroxide through the chloroplast envelope via aquaporins. Biochim. Biophys. Acta (BBA)-Bioenerg. 2012, 1817, 1314–1321. [Google Scholar] [CrossRef] [PubMed]
- Juvany, M.; Müller, M.; Munné-Bosch, S. Photo-oxidative stress in emerging and senescing leaves: A mirror image? J. Exp. Bot. 2013, 64, 3087–3098. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, P.; Jaleel, C.A.; Salem, M.A.; Nabi, G.; Sharma, S. Roles of enzymatic and nonenzymatic antioxidants in plants during abiotic stress. Crit. Rev. Biotechnol. 2010, 30, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Queval, G.; Foyer, C.H. Redox regulation of photosynthetic gene expression. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 3475–3485. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Queval, G.; Foyer, C.H. The impact of global change factors on redox signaling underpinning stress tolerance. Plant Physiol. 2013, 161, 5–19. [Google Scholar] [CrossRef]
- Mozzo, M.; Dall’Osto, L.; Hienerwadel, R.; Bassi, R.; Croce, R. Photoprotection in the antenna complexes of photosystem II: Role of individual xanthophylls in chlorophyll triplet quenching. J. Biol. Chem. 2008, 283, 6184–6192. [Google Scholar] [CrossRef]
- Fischer, B.B.; Hideg, E.; Krieger-Liszkay, A. Production, detection, and signaling of singlet oxygen in photosynthetic organisms. Antioxid. Redox Signal. 2013, 18, 2145–2162. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, S.; Purkar, V.; Mitra, S. β-Cyclocitral, a Master Regulator of Multiple Stress-Responsive Genes in Solanum lycopersicum L. Plants. Plants 2021, 10, 2465. [Google Scholar] [CrossRef] [PubMed]
- Roach, T.; Baur, T.; Kranner, I. β-Cyclocitral Does Not Contribute to Singlet Oxygen-Signalling in Algae, but May Down-Regulate Chlorophyll Synthesis. Plants 2022, 11, 2155. [Google Scholar] [CrossRef] [PubMed]
- Sunkar, R.; Kapoor, A.; Zhu, J.-K. Posttranscriptional induction of two Cu/Zn superoxide dismutase genes in Arabidopsis is mediated by downregulation of miR398 and important for oxidative stress tolerance. Plant Cell 2006, 18, 2051–2065. [Google Scholar] [CrossRef]
- Koh, E.; Carmieli, R.; Mor, A.; Fluhr, R. Singlet oxygen-induced membrane disruption and serpin-protease balance in vacuolar-driven cell death. Plant Physiol. 2016, 171, 1616–1625. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).