Exploring the Immune-Boosting Functions of Vitamins and Minerals as Nutritional Food Bioactive Compounds: A Comprehensive Review
Abstract
1. Introduction
2. Immune System and Immunomodulators
2.1. Immune System
2.2. Immune Activation in Response to Non-Self-Antigens
2.3. Regulation of Immune Responses
2.4. Immunomodulators
2.4.1. Immunostimulators
2.4.2. Immunosuppressants
2.5. The Importance of Immunomodulators in Current Clinical Practice
3. Immune-Boosting Foods
4. Insights into the Key Roles Played by Vitamins and Minerals as Immunomodulators
4.1. Vitamin A
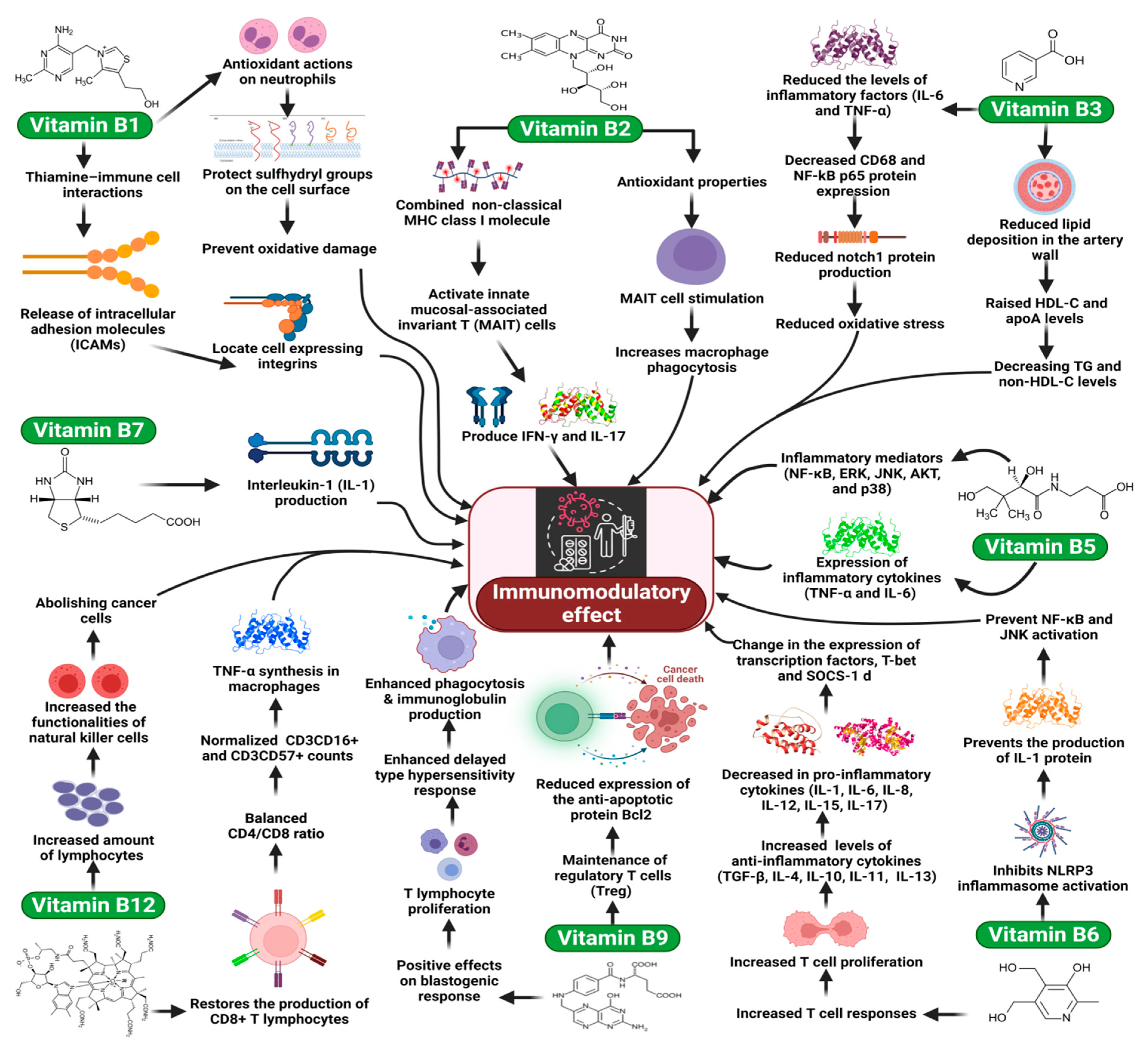
4.2. Vitamin B1
4.3. Vitamin B2
4.4. Vitamin B3 (Niacin, Nicotinic Acid, and Nicotinamide)
4.5. Vitamin B5 (Pantothenic Acid)
4.6. Vitamin B6 (Pyridoxine)
4.7. Vitamin B7 (Biotin)
4.8. Vitamin B9 (Folate)
4.9. Vitamin B12 (Cobalamin)
4.10. Vitamin C
4.11. Vitamin D
4.12. The Role of Vitamin D in Innate Immune Response
4.13. The Role of Vitamin D in Adaptive Immune Response
4.14. Vitamin E
4.15. Vitamin K
4.16. Zinc
4.17. Iron
4.18. Selenium
4.19. Iodine
4.20. Magnesium
4.21. Copper
5. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Pecora, F.; Persico, F.; Argentiero, A.; Neglia, C.; Esposito, S. The role of micronutrients in support of the immune response against viral infections. Nutrients 2020, 12, 3198. [Google Scholar] [CrossRef]
- Cai, H.; Chen, L.; Yin, C.; Liao, Y.; Meng, X.; Lu, C.; Tang, S.; Li, X.; Wang, X. The effect of micro-nutrients on malnutrition, immunity and therapeutic effect in patients with pulmonary tuberculosis: A systematic review and meta-analysis of randomised controlled trials. Tuberculosis 2020, 125, 101994. [Google Scholar] [CrossRef]
- Alpert, P.T. The Role of Vitamins and Minerals on the Immune System. Home Health Care Manag. Pract. 2017, 29, 199–202. [Google Scholar] [CrossRef]
- Tina Suksmasari, B.H. Multivitamin Supplementation Supports Immune Function and Ameliorates Conditions Triggered By Reduced Air Quality. Vitam. Miner. 2015, 4, 1–15. [Google Scholar] [CrossRef]
- Widasari, L.; Chalid, M.T.; Jafar, N.; Thaha, A.R.; Dirpan, A. The role of multimicronutrients on improving better pregnancy outcomes: A literature review. Syst. Rev. Pharm. 2020, 11, 550–553. [Google Scholar] [CrossRef]
- Gombart, A.F.; Pierre, A.; Maggini, S. A review of micronutrients and the immune system–working in harmony to reduce the risk of infection. Nutrients 2020, 12, 236. [Google Scholar] [CrossRef] [PubMed]
- Sauer, K.; Cooke, M.P. Regulation of immune cell development through soluble inositol-1,3,4,5- tetrakisphosphate. Nat. Rev. Immunol. 2010, 10, 257–271. [Google Scholar] [CrossRef] [PubMed]
- Thangasamy, T.; Subathra, M.; Sittadjody, S.; Jeyakumar, P.; Joyee, A.G.; Mendoza, E.; Chinnakkanu, P. Role of l-carnitine in the modulation of immune response in aged rats. Clin. Chim. Acta 2008, 389, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.; Mamedova, L.K.; Barton, B.; Bradford, B.J. Choline regulates the function of bovine immune cells and alters the mRNA abundance of enzymes and receptors involved in its metabolism in vitro. Front. Immunol. 2018, 9, 2448. [Google Scholar] [CrossRef]
- López-Pedrera, C.; Villalba, J.M.; Patiño-Trives, A.M.; Luque-Tévar, M.; Barbarroja, N.; Aguirre, M.Á.; Escudero-Contreras, A.; Pérez-Sánchez, C. Therapeutic potential and immunomodulatory role of coenzyme q10 and its analogues in systemic autoimmune diseases. Antioxidants 2021, 10, 600. [Google Scholar] [CrossRef] [PubMed]
- Elmadfa, I.; Meyer, A.L. The Role of the Status of Selected Micronutrients in Shaping the Immune Function. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 1100–1115. [Google Scholar] [CrossRef] [PubMed]
- Belkaid, Y.; Hand, T.W. Role of the microbiota in immunity and inflammation. Cell 2014, 157, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Luebke, R.W.; Germolec, D.R. The immune system. In Environmental Impacts on Reproductive Health and Fertility; Cambridge University Press: Cambridge, UK, 2010; pp. 188–193. [Google Scholar] [CrossRef]
- Carrillo, J.L.M.; Rodríguez, F.P.C.; Coronado, O.G.; García, M.A.M.; Cordero, J.F.C. Physiology and Pathology of Innate Immune Response Against Pathogens. In Physiology and Pathology of Immunology; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Riera Romo, M.; Pérez-Martínez, D.; Castillo Ferrer, C. Innate immunity in vertebrates: An overview. Immunology 2016, 148, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Krensky, A.M.; Bennett, W.M.; Vincenti, F. Goodman and Gilman’s the Pharmacological Basis of Therapeutics; McGraw-Hill: New York, NY, USA, 2010; Volume 7, ISBN 9780071769396. [Google Scholar]
- Marshall, J.S.; Warrington, R.; Watson, W.; Kim, H.L. An introduction to immunology and immunopathology. Allergy Asthma Clin. Immunol. 2018, 14, 1–8. [Google Scholar] [CrossRef]
- Turvey, S.E.; Broide, D.H. Innate immunity. J. Allergy Clin. Immunol. 2010, 125, S24–S32. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.; Akira, S. Innate immunity. Curr. Opin. Immunol. 2013, 25, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Negri, P.; Maggi, M.; Correa-Aragunde, N.; Brasesco, C.; Eguaras, M.; Lamattina, L. Nitric oxide participates at the first steps of Apis mellifera cellular immune activation in response to non-self recognition. Apidologie 2013, 44, 575–585. [Google Scholar] [CrossRef]
- Appay, V.; Sauce, D. Immune activation and inflammation in HIV-1 infection: Causes and consequences. J. Pathol. 2008, 214, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Bartolucci, S.; Piccirillo, C.A. Immune Regulation in Human Health and Disease. eLS 2017, 2017, 1–17. [Google Scholar] [CrossRef]
- Luckheeram, R.V.; Zhou, R.; Verma, A.D.; Xia, B. CD4 +T cells: Differentiation and functions. Clin. Dev. Immunol. 2012, 2012, 925135. [Google Scholar] [CrossRef] [PubMed]
- Robbins, P.D.; Morelli, A.E. Regulation of immune responses by extracellular vesicles. Nat. Publ. Gr. 2014, 14, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Oliver, M.A.; Bishop, T.F.A.; Marchant, B.P.; Marchant, B.P. Future prospects. Precis. Agric. Sustain. Environ. Prot. 2013, 9780203128, 265–268. [Google Scholar] [CrossRef]
- Mohan, T.; Verma, P.; Nageswara Rao, D. Novel adjuvants & delivery vehicles for vaccines development: A road ahead. Indian J. Med. Res. 2013, 138, 779–795. [Google Scholar] [PubMed]
- Heidt, S.; Roelen, D.L.; Eijsink, C.; Eikmans, M.; Van Kooten, C.; Claas, F.H.J.; Mulder, A. Calcineurin inhibitors affect B cell antibody responses indirectly by interfering with T cell help. Clin. Exp. Immunol. 2010, 159, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Mrityunjaya, M.; Pavithra, V.; Neelam, R.; Janhavi, P.; Halami, P.M.; Ravindra, P.V. Immune-Boosting, Antioxidant and Anti-inflammatory Food Supplements Targeting Pathogenesis of COVID-19. Front. Immunol. 2020, 11, 570122. [Google Scholar] [CrossRef]
- Ashaolu, T.J. Immune boosting functional foods and their mechanisms: A critical evaluation of probiotics and prebiotics. Biomed. Pharmacother. 2020, 130, 110625. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Mosaddik, A.; Alam, A.K. Traditional foods with their constituent’s antiviral and immune system modulating properties. Heliyon 2021, 7, e05957. [Google Scholar] [CrossRef]
- London, C. Functional Foods that Boost the Immune System. Funct. Food Prod. Dev. 2010, 2010, 293–321. [Google Scholar] [CrossRef]
- Sanchez-Moreno, C.; De Ancos, B.; Plaza, L.; Elez-Martinez, P.; Cano, M.P. Nutritional approaches and health-related properties of plant foods processed by high pressure and pulsed electric fields. Crit. Rev. Food Sci. Nutr. 2009, 49, 552–576. [Google Scholar] [CrossRef]
- Cilla, A.; Bosch, L.; Barberá, R.; Alegría, A. Effect of processing on the bioaccessibility of bioactive compounds—A review focusing on carotenoids, minerals, ascorbic acid, tocopherols and polyphenols. J. Food Compos. Anal. 2018, 68, 3–15. [Google Scholar] [CrossRef]
- Schoendorfer, N.; Davies, P.S.W. Micronutrient interrelationships: Synergism and antagonism. In Micronutrients, 1st ed.; Betancourt, A.I., Gaitan, H.F., Eds.; Nova Science Publishers, Inc.: New York, NY, USA, 2012; pp. 159–177. [Google Scholar]
- Borgert, C.J.; Borgert, S.A.; Findley, K.C. Synergism, antagonism, or additivity of dietary supplements: Application of theory to case studies. Thromb. Res. 2005, 117, 123–132. [Google Scholar] [CrossRef]
- Booth, S.L.; Johns, T.; Kuhnlein, H.V. Natural Food Sources of Vitamin A and Provitamin A. Food Nutr. Bull. 1992, 14, 1–15. [Google Scholar] [CrossRef]
- Grune, T.; Lietz, G.; Palou, A.; Ross, A.C.; Stahl, W.; Tang, G.; Thurnham, D.; Yin, S.A.; Biesalski, H.K. β-carotene is an important vitamin A source for humans. J. Nutr. 2010, 140, 2268S–2285S. [Google Scholar] [CrossRef]
- Saeed, A.; Anwaar, A.; Randhawa, M.A.; Sunethra, A.; Nimmathota, A.; Tariq, I.; Zulfiqar, A. Prevalence of vitamin A deficiency in South Asia: Causes, outcomes, and possible remedies. J. Health Popul. Nutr. 2013, 31, 413–423. [Google Scholar]
- WHO. WHO Vitamin A Supplementation in Infants and Children 6–59 Months of Age; WHO: Geneva, Switzerland, 2017; pp. 1–29. [Google Scholar]
- Imdad, A.; Yakoob, M.Y.; Sudfeld, C.; Haider, B.A.; Black, R.E.; Bhutta, Z.A. Impact of vitamin A supplementation on infant and childhood mortality. BMC Public Health 2011, 11, 1–15. [Google Scholar] [CrossRef]
- Wiysonge, C.; Ndze, V.; Kongnyuy, E.; Shey, M. Vitamin A supplements for reducing mother-to-child HIV transmission. Cochrane Database Syst. Rev. 2017, 2017, CD003648. [Google Scholar] [CrossRef] [PubMed]
- Acácio, S.; Nhampossa, T.; Quintó, L.; Vubil, D.; Sacoor, C.; Kotloff, K.; Farag, T.; Dilruba, N.; Macete, E.; Levine, M.M.; et al. The role of HIV infection in the etiology and epidemiology of diarrheal disease among children aged 0–59 months in Manhiça District, Rural Mozambique. Int. J. Infect. Dis. 2018, 73, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Pino-Lagos, K.; Guo, Y.; Noelle, R.J. Retinoic acid: A key player in immunity. Biofactors 2010, 36, 430–436. [Google Scholar] [CrossRef]
- Rampal, R.; Awasthi, A.; Ahuja, V. Retinoic acid-primed human dendritic cells inhibit Th9 cells and induce Th1/Th17 cell differentiation. J. Leukoc. Biol. 2016, 100, 111–120. [Google Scholar] [CrossRef]
- Lu, L.; Lan, Q.; Li, Z.; Zhou, X.; Gu, J.; Li, Q.; Wang, J.; Chen, M.; Liu, Y.; Shen, Y.; et al. Critical role of all-trans retinoic acid in stabilizing human natural regulatory T cells under inflammatory conditions. Proc. Natl. Acad. Sci. USA 2014, 111, E3432–E3440. [Google Scholar] [CrossRef]
- Roy, S.; Awasthi, A. Vitamin A and the Immune System. In Nutrition and Immunity; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar] [CrossRef]
- Zhu, B.; Buttrick, T.; Bassil, R.; Zhu, C.; Olah, M.; Wu, C. IL-4 and retinoic acid synergistically induce regulatory dendritic cells expressing Aldh1a2. J. Immunol. 2013, 191, 3139–3151. [Google Scholar] [CrossRef]
- Depaolo, R.; Abadie, V.; Tang, F.; Fehlner-Peach, H.; Hall, J.A.; Wang, W. Co-adjuvant effects of retinoic acid and IL-15 induce inflammatory immunity to dietary antigens. Nature 2011, 471, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Peterson, C.T.; Rodionov, D.A.; Peterson, S.N.; Osterman, A.L. B vitamins and their role in immune regulation and cancer. Nutrients 2020, 12, 3380. [Google Scholar] [CrossRef]
- Scalabrino, G. Vitamin-regulated cytokines and growth factors in the CNS and elsewhere. J. Neurochem. 2009, 111, 1309–1326. [Google Scholar] [CrossRef]
- Bozic, I.; Savic, D.; Laketa, D.; Bjelobaba, I.; Milenkovic, I.; Pekovic, S.; Nedeljkovic, N.; Lavrnja, I. Benfotiamine attenuates inflammatory response in LPS stimulated BV-2 microglia. PLoS ONE 2015, 10, e0118372. [Google Scholar] [CrossRef]
- Shoeb, M.; Ramana, K.V. Anti-inflammatory effects of benfotiamine are mediated through the regulation of the arachidonic acid pathway in macrophages. Free Radic. Biol. Med. 2012, 52, 182–190. [Google Scholar] [CrossRef]
- Huskisson, E.; Maggini, S.; Ruf, M. The Role of Vitamins and Minerals in Energy Metabolism and Well-Being. J. Int. Med. Res. 2007, 35, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Corbett, A.J.; Eckle, S.B.G.; Birkinshaw, R.W.; Liu, L.; Patel, O.; Mahony, J.; Chen, Z.; Reantragoon, R.; Meehan, B.; Cao, H.; et al. T-cell activation by transitory neo-antigens derived from distinct microbial pathways. Nature 2014, 509, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Cowley, S.C. MAIT cells and pathogen defense. Cell. Mol. Life Sci. 2014, 71, 4831–4840. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, K.; Apostolopoulos, V. Vitamin B1, B2, B3, B5, and B6 and the Immune System. In Nutrition and Immunity; Springer: Berlin/Heidelberg, Germany, 2019; pp. 115–125. [Google Scholar] [CrossRef]
- Mazur-Bialy, A.I.; Buchala, B.; Plytycz, B. Riboflavin deprivation inhibits macrophage viability and activity-a study on the RAW 264.7 cell line. Br. J. Nutr. 2013, 110, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Digby, J.E.; Martinez, F.; Jefferson, A.; Ruparelia, N.; Chai, J.; Wamil, M.; Greaves, D.R.; Choudhury, R.P. Anti-inflammatory effects of nicotinic acid in human monocytes are mediated by GPR109A dependent mechanisms. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.T.; Digby, J.E.; Ruparelia, N.; Jefferson, A.; Handa, A.; Choudhury, R.P. Nicotinic Acid Receptor GPR109A Is Down-Regulated in Human Macrophage-Derived Foam Cells. PLoS ONE 2013, 8, e62934. [Google Scholar] [CrossRef] [PubMed]
- Si, Y.; Zhang, Y.; Zhao, J.; Guo, S.; Zhai, L.; Yao, S.; Sang, H.; Yang, N.; Song, G.; Gu, J.; et al. Niacin inhibits vascular inflammation via downregulating nuclear transcription factor-κb signaling pathway. Mediat. Inflamm. 2014, 2014, 263786. [Google Scholar] [CrossRef]
- Zhou, E.; Li, Y.; Yao, M.; Wei, Z.; Fu, Y.; Yang, Z. Niacin attenuates the production of pro-inflammatory cytokines in LPS-induced mouse alveolar macrophages by HCA2 dependent mechanisms. Int. Immunopharmacol. 2014, 23, 121–126. [Google Scholar] [CrossRef]
- He, W.; Hu, S.; Du, X.; Wen, Q.; Zhong, X.P.; Zhou, X.; Zhou, C.; Xiong, W.; Gao, Y.; Zhang, S.; et al. Vitamin B5 reduces bacterial growth via regulating innate immunity and adaptive immunity in mice infected with Mycobacterium tuberculosis. Front. Immunol. 2018, 9, 365. [Google Scholar] [CrossRef]
- Li-Mei, W.; Jie, T.; Shan-He, W.; Dong-Mei, M.; Peng-Jiu, Y. Anti-inflammatory and Anti-oxidative Effects of Dexpanthenol on Lipopolysaccharide Induced Acute Lung Injury in Mice. Inflammation 2016, 39, 1757–1763. [Google Scholar] [CrossRef]
- Soylu Karapinar, O.; Pinar, N.; Özgür, T.; Özcan, O.; Bayraktar, H.S.; Kurt, R.K.; Nural, O. The Protective Role of Dexpanthenol on the Endometrial Implants in an Experimentally Induced Rat Endometriosis Model. Reprod. Sci. 2017, 24, 285–290. [Google Scholar] [CrossRef]
- Karadag, A.; Ozdemir, R.; Kurt, A.; Parlakpinar, H.; Polat, A.; Vardi, N.; Taslidere, E.; Karaman, A. Protective effects of dexpanthenol in an experimental model of necrotizing enterocolitis. J. Pediatr. Surg. 2015, 50, 1119–1124. [Google Scholar] [CrossRef]
- Qian, B.; Shen, S.; Zhang, J.; Jing, P. Effects of Vitamin B6 Deficiency on the Composition and Functional Potential of T Cell Populations. J. Immunol. Res. 2017, 2017, 2197975. [Google Scholar] [CrossRef]
- Zheng, X.; Feng, L.; Jiang, W.D.; Wu, P.; Liu, Y.; Jiang, J.; Kuang, S.Y.; Tang, L.; Tang, W.N.; Zhang, Y.A.; et al. Dietary pyridoxine deficiency reduced growth performance and impaired intestinal immune function associated with TOR and NF-κB signalling of young grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol. 2017, 70, 682–700. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Sun, C.; Liu, H.; Li, F.; Zhu, Y.; Li, F. Effects of dietary supplement of vitamin B6 on growth performance and non-specific immune response of weaned rex rabbits. J. Appl. Anim. Res. 2018, 46, 1370–1376. [Google Scholar] [CrossRef]
- Zhang, P.; Tsuchiya, K.; Kinoshita, T.; Kushiyama, H.; Suidasari, S.; Hatakeyama, M.; Imura, H.; Kat, N.; Suda, T. Vitamin B6 Prevents IL-1β Protein Production by Inhibiting NLRP3 Inflammasome Activation. J. Biol. Chem. 2016, 291, 24517–24527. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O. B vitamins and the brain: Mechanisms, dose and efficacy—A review. Nutrients 2016, 8, 68. [Google Scholar] [CrossRef] [PubMed]
- Kuroishi, T.; Kinbara, M.; Sato, N.; Tanaka, Y.; Nagai, Y.; Iwakura, Y.; Endo, Y.; Sugawara, S. Biotin status affects nickel allergy via regulation of interleukin-1β production in mice. J. Nutr. 2009, 139, 1031–1036. [Google Scholar] [CrossRef]
- Härtel, C.; Puzik, A.; Göpel, W.; Temming, P.; Bucsky, P.; Schultz, C. Immunomodulatory effect of vitamin C on intracytoplasmic cytokine production in neonatal cord blood cells. Neonatology 2007, 91, 54–60. [Google Scholar] [CrossRef]
- Todorova, T.T.; Ermenlieva, N.; Tsankova, G. Vitamin B12: Could It Be a Promising Immunotherapy? In Immunotherapy: Myths, Reality, Ideas, Future; Books on Demand: Norderstedt, Germany, 2017; p. 85. [Google Scholar] [CrossRef]
- Partearroyo, T.; Úbeda, N.; Montero, A.; Achón, M.; Varela-Moreiras, G. Vitamin B12 and folic acid imbalance modifies NK cytotoxicity, lymphocytes B and lymphoprolipheration in aged rats. Nutrients 2013, 5, 4836–4848. [Google Scholar] [CrossRef] [PubMed]
- Erkurt, M.A.; Aydogdu, I.; Dikilitaş, M.; Kuku, I.; Kaya, E.; Bayraktar, N.; Ozhan, O.; Ozkan, I.; Sönmez, A. Effects of cyanocobalamin on immunity in patients with pernicious anemia. Med. Princ. Pract. 2008, 17, 131–135. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Apostolopoulos, V. Vitamin B12, Folic Acid, and the Immune System. In Nutrition and Immunity; Springer: Berlin/Heidelberg, Germany, 2019; pp. 103–114. [Google Scholar] [CrossRef]
- Parahuleva, M.S.; Jung, J.; Burgazli, M.; Erdogan, A.; Parviz, B.; Hölschermann, H. Vitamin C suppresses lipopolysaccharide-induced procoagulant response of human monocyte-derived macrophages. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2174–2182. [Google Scholar]
- Woo, A.; Kim, J.-H.; Jeong, Y.-J.; Maeng, H.G.; Lee, Y.-T.; Kang, J.S.; Lee, W.J.; Hwang, Y. Vitamin C acts indirectly to modulate isotype switching in mouse B cells. Anat. Cell Biol. 2010, 43, 25. [Google Scholar] [CrossRef]
- Mousavi, S.; Bereswill, S.; Heimesaat, M.M. Immunomodulatory and antimicrobial effects of vitamin C. Eur. J. Microbiol. Immunol. 2019, 9, 73–79. [Google Scholar] [CrossRef]
- Toliopoulos, I.K.; Simos, Y.V.; Daskalou, T.A.; Verginadis, I.I.; Evangelou, A.M.; Karkabounas, S.C. Inhibition of platelet aggregation and immunomodulation of NK lymphocytes by administration of ascorbic acid. Indian J. Exp. Biol. 2011, 49, 904–908. [Google Scholar]
- Bikle, D.D. Vitamin D and immune function: Understanding common pathways. Curr. Osteoporos. Rep. 2009, 7, 58–63. [Google Scholar] [CrossRef]
- Radovic, J.; Markovic, D.; Velickov, A.; Djordjevic, B.; Stojnev, S. Vitamin D immunomodulatory effect. Acta Medica Median. 2012, 51, 58–64. [Google Scholar] [CrossRef]
- Cutolo, M. Vitamin D and autoimmune rheumatic diseases. Rheumatology 2009, 48, 210–212. [Google Scholar] [CrossRef][Green Version]
- Holick, M.F. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am. J. Clin. Nutr. 2004, 80, 1678S–1688S. [Google Scholar] [CrossRef]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; Van Etten, E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; Demay, M. Vitamin D and human health: Lessons from vitamin D receptor null mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef]
- Chang, J.H.; Cha, H.R.; Lee, D.S.; Seo, K.Y.; Kweon, M.N. 1,25-dihydroxyvitamin D3 inhibits the differentiation and migration of TH17 cells to protect against experimental autoimmune encephalomyelitis. PLoS ONE 2010, 5, e12925. [Google Scholar] [CrossRef]
- Kamen, D.L.; Tangpricha, V. Vitamin D and molecular actions on the immune system: Modulation of innate and autoimmunity. J. Mol. Med. 2010, 88, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M. Role of Toll-like receptors in Helicobacter pylori infection and immunity. World J. Gastrointest. Pathophysiol. 2014, 5, 133. [Google Scholar] [CrossRef] [PubMed]
- Yuk, J.M.; Shin, D.M.; Lee, H.M.; Yang, C.S.; Jin, H.S.; Kim, K.K.; Lee, Z.W.; Lee, S.H.; Kim, J.M.; Jo, E.K. Vitamin D3 Induces Autophagy in Human Monocytes/Macrophages via Cathelicidin. Cell Host Microbe 2009, 6, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Chun, R.F.; Liu, P.T.; Modlin, R.L.; Adams, J.S.; Hewison, M. Impact of vitamin D on immune function: Lessons learned from genome-wide analysis. Front. Physiol. 2014, 5, 151. [Google Scholar] [CrossRef]
- Carvalho, J.T.G.; Schneider, M.; Cuppari, L.; Grabulosa, C.C.; Aoike, D.T.; Redublo, B.M.Q.; Batista, M.C.; Cendoroglo, M.; Moyses, R.M.; Dalboni, M.A. Cholecalciferol decreases inflammation and improves Vitamin D regulatory enzymes in lymphocytes in the uremic environment: A randomized controlled pilot trial. PLoS ONE 2017, 12, e0179540. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Chen, J.; Zheng, C.; Wu, J.; Cheng, Y.; Zhu, S.; Lin, C.; Cao, Q.; Zhu, J.; Jin, T. 1,25-dihydroxyvitamin D3-induced dendritic cells suppress experimental autoimmune encephalomyelitis by increasing proportions of the regulatory lymphocytes and reducing T helper type 1 and type 17 cells. Immunology 2017, 152, 414–424. [Google Scholar] [CrossRef]
- Chen, S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.; Lipsky, P.E. Modulatory Effects of 1,25-Dihydroxyvitamin D 3 on Human B Cell Differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef]
- Van Etten, E.; Mathieu, C. Immunoregulation by 1,25-dihydroxyvitamin D3: Basic concepts. J. Steroid Biochem. Mol. Biol. 2005, 97, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Sassi, F.; Tamone, C.; D’amelio, P. Vitamin D: Nutrient, hormone, and immunomodulator. Nutrients 2018, 10, 1656. [Google Scholar] [CrossRef] [PubMed]
- Raederstorff, D.; Wyss, A.; Calder, P.; Weber, P.; Eggersdorfer, M. Vitamin E function and requirements in relation to PUFA. Br. J. Nutr. 2015, 114, 1113–1222. [Google Scholar] [CrossRef]
- Su, L.J.; Zhang, J.H.; Gomez, H.; Murugan, R.; Hong, X.; Xu, D.; Jiang, F.; Peng, Z.Y. Reactive Oxygen Species-Induced Lipid Peroxidation in Apoptosis, Autophagy, and Ferroptosis. Oxid. Med. Cell. Longev. 2019, 2019, 5080843. [Google Scholar] [CrossRef]
- Lee, G.Y.; Han, S.N. The role of vitamin E in immunity. Nutrients 2018, 10, 1614. [Google Scholar] [CrossRef]
- Lavrovsky, Y.; Chatterjee, B.; Clark, R.A.; Roy, A.K. Role of redox-regulated transcription factors in inflammation, aging and age-related diseases. Exp. Gerontol. 2000, 35, 521–532. [Google Scholar] [CrossRef]
- Lewis, E.D.; Meydani, S.N.; Wu, D. Regulatory role of vitamin E in the immune system and inflammation. IUBMB Life 2019, 71, 487–494. [Google Scholar] [CrossRef]
- Han, S.N.; Wu, D.; Ha, W.K.; Beharka, A.; Smith, D.E.; Bender, B.S.; Meydani, S.N. Vitamin E supplementation increases T helper 1 cytokine production in old mice infected with influenza virus. Immunology 2000, 100, 487–493. [Google Scholar] [CrossRef]
- Bou Ghanem, E.N.; Clark, S.; Du, X.; Wu, D.; Camilli, A.; Leong, J.M.; Meydani, S.N. The α-Tocopherol Form of Vitamin E Reverses Age-Associated Susceptibility to Streptococcus pneumoniae Lung Infection by Modulating Pulmonary Neutrophil Recruitment. J. Immunol. 2015, 194, 1090–1099. [Google Scholar] [CrossRef]
- Salinthone, S.; Kerns, A.R.; Tsang, V.C.D. Alpha-tocopherol (vitamin e) stimulates cyclic amp production in human peripheral mononuclear cells and alters immune function. Mol. Immunol. 2013, 53, 173–178. [Google Scholar] [CrossRef]
- Namazi, N.; Larijani, B.; Azadbakht, L. Vitamin K and the Immune System. In Nutrition and Immunity; Springer: Berlin/Heidelberg, Germany, 2019; pp. 75–79. [Google Scholar] [CrossRef]
- Hatanaka, H.; Ishizawa, H.; Nakamura, Y.; Tadokoro, H.; Tanaka, S.; Onda, K.; Sugiyama, K.; Hirano, T. Effects of vitamin K 3 and K 5 on proliferation, cytokine production, and regulatory T cell-frequency in human peripheral blood mononuclear cells. Life Sci. 2014, 99, 61–68. [Google Scholar] [CrossRef]
- Checker, R.; Sharma, D.; Sandur, S.K.; Khan, N.M.; Patwardhan, R.S.; Kohli, V.; Sainis, K.B. Vitamin K3 suppressed inflammatory and immune responses in a redox-dependent manner. Free Radic. Res. 2011, 45, 975–985. [Google Scholar] [CrossRef]
- Zhong, J.H.; Mo, X.S.; De Xiang, B.; Yuan, W.P.; Jiang, J.F.; Xie, G.S.; Li, L.Q. Postoperative Use of the Chemopreventive Vitamin K2 Analog in Patients with Hepatocellular Carcinoma. PLoS ONE 2013, 8, e58082. [Google Scholar] [CrossRef] [PubMed]
- Soye, K.J.; Trottier, C.; Di Lenardo, T.Z.; Restori, K.H.; Reichman, L.; Miller, W.H.; Ward, B.J. In vitro inhibition of mumps virus by retinoids. Virol. J. 2013, 10, 1–13. [Google Scholar] [CrossRef]
- Zeng, R.; Bscheider, M.; Lahl, K.; Lee, M.; Butcher, E.C. Generation and transcriptional programming of intestinal dendritic cells: Essential role of retinoic acid. Mucosal Immunol. 2016, 9, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Snyder, L.M.; McDaniel, K.L.; Tian, Y.; Wei, C.H.; Kennett, M.J.; Patterson, A.D.; Catharine Ross, A.; Cantorna, M.T. Retinoic acid mediated clearance of Citrobacter rodentium in vitamin A deficient mice requires CD11b+ and T cells. Front. Immunol. 2019, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Seo, G.Y.; Jang, Y.S.; Kim, J.; Choe, J.; Han, H.J.; Lee, J.M.; Kang, S.H.; Rhee, K.J.; Park, S.R.; Kim, W.S.; et al. Retinoic acid acts as a selective human IgA switch factor. Hum. Immunol. 2014, 75, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Rudraraju, R.; Jones, B.G.; Surman, S.L.; Sealy, R.E.; Thomas, P.G.; Hurwitz, J.L. Respiratory tract epithelial cells express retinaldehyde dehydrogenase ALDH1A and enhance IgA production by stimulated B cells in the presence of vitamin A. PLoS ONE 2014, 9, e86554. [Google Scholar] [CrossRef] [PubMed]
- Rudraraju, R.; Surman, S.L.; Jones, B.G.; Sealy, R.; Woodland, D.L.; Hurwitz, J.L. Reduced frequencies and heightened CD103 expression among virus-induced CD8 + T cells in the respiratory tract airways of vitamin A-deficient mice. Clin. Vacc. Immunol. 2012, 19, 757–765. [Google Scholar] [CrossRef]
- Penkert, R.R.; Jones, B.G.; Häcker, H.; Partridge, J.F.; Hurwitz, J.L. Vitamin A differentially regulates cytokine expression in respiratory epithelial and macrophage cell lines. Cytokine 2017, 91, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Penkert, R.R.; Surman, S.L.; Jones, B.G.; Sealy, R.E.; Vogel, P.; Neale, G.; Hurwitz, J.L. Vitamin A deficient mice exhibit increased viral antigens and enhanced cytokine/chemokine production in nasal tissues following respiratory virus infection despite the presence of FoxP3+ T cells. Int. Immunol. 2016, 28, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, C.A.; Spencer, S.P.; Torabi-Parizi, P.; Grainger, J.R.; Roychoudhuri, R.; Ji, Y.; Sukumar, M.; Muranski, P.; Scott, C.D.; Hall, J.A.; et al. Retinoic acid controls the homeostasis of pre-cDC-derived splenic and intestinal dendritic cells. J. Exp. Med. 2013, 210, 1961–1976. [Google Scholar] [CrossRef]
- Penkert, R.R.; Rowe, H.M.; Surman, S.L.; Sealy, R.E.; Rosch, J.; Hurwitz, J.L. Influences of Vitamin A on vaccine immunogenicity and efficacy. Front. Immunol. 2019, 10, 1–9. [Google Scholar] [CrossRef]
- Van De Pavert, S.A.; Ferreira, M.; Domingues, R.G.; Ribeiro, H.; Molenaar, R.; Moreira-Santos, L.; Almeida, F.F.; Ibiza, S.; Barbosa, I.; Goverse, G.; et al. Maternal retinoids control type 3 innate lymphoid cells and set the offspring immunity. Nature 2014, 508, 123–127. [Google Scholar] [CrossRef]
- Pantazi, E.; Marks, E.; Stolarczyk, E.; Lycke, N.; Noelle, R.J.; Elgueta, R. Cutting Edge: Retinoic Acid Signaling in B Cells Is Essential for Oral Immunization and Microflora Composition. J. Immunol. 2015, 195, 1368–1371. [Google Scholar] [CrossRef]
- Chang, H.K.; Hou, W.S. Retinoic acid modulates interferon-γ production by hepatic natural killer T cells via phosphatase 2A and the extracellular signal-regulated kinase pathway. J. Interf. Cytokine Res. 2015, 35, 200–212. [Google Scholar] [CrossRef]
- Su, Y.; Sun, Y.; Ju, D.; Chang, S.; Shi, B.; Shan, A. The detoxification effect of vitamin C on zearalenone toxicity in piglets. Ecotoxicol. Environ. Saf. 2018, 158, 284–292. [Google Scholar] [CrossRef]
- Natarajan, R. Impact of Intravenous Ascorbic Acid Infusion on Novel Biomarkers in Patients with Severe Sepsis. J. Pulm. Respir. Med. 2014, 4, 4–11. [Google Scholar] [CrossRef]
- Mohammed, B.M.; Fisher, B.J.; Kraskauskas, D.; Farkas, D.; Brophy, D.F.; Iii, A.A.F.; Natarajan, R. Vitamin C: A Novel Regulator of Neutrophil Extracellular Trap Formation. Nutrients 2013, 5, 3131–3150. [Google Scholar] [CrossRef]
- Bozonet, S.M.; Carr, A.C.; Pullar, J.M.; Vissers, M.C.M. Enhanced Human Neutrophil Vitamin C Status, Chemotaxis and Oxidant Generation Following Dietary Supplementation with Vitamin C-Rich SunGold Kiwifruit. Nutrients 2015, 7, 2574–2588. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.B.; Zhang, Y.P.; Zhang, J.; Zhang, Y.B. Evaluation of Vitamin C supplementation on kidney function and vascular reactivity following renal ischemic injury in mice. Kidney Blood Press. Res. 2016, 41, 460–470. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Luo, G.; Yuan, J.; Wang, Y.; Yang, X.; Wang, X.; Li, G.; Liu, Z.; Zhong, N. Vitamin C Mitigates Oxidative Stress and Tumor Necrosis Factor-Alpha in Severe Community-Acquired Pneumonia and LPS-Induced Macrophages. Mediat. Inflamm. 2014, 2014, 426740. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B.J.; Kraskauskas, D.; Martin, E.J.; Farkas, D.; Wegelin, J.A.; Brophy, D.; Ward, K.R.; Voelkel, N.F.; Fowler, A.A.; Natarajan, R. Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid. Am. J. Physiol. Lung Cell. Mol. Physiol. 2012, 303, 20–33. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Bae, S.; Choi, J.; Lim, S.Y.; Lee, N.; Kong, J.M.; Hwang, Y.; Kang, J.S.; Lee, W.J. Vitamin C Is an Essential Factor on the Anti-viral Immune Responses through the Production of Interferon-α/β at the Initial Stage of Influenza A Virus (H3N2) Infection. Immune Netw. 2013, 13, 70. [Google Scholar] [CrossRef]
- Hartmann, B.; Heine, G.; Babina, M.; Steinmeyer, A.; Zügel, U.; Radbruch, A.; Worm, M. Targeting the vitamin D receptor inhibits the B cell-dependent allergic immune response. Allergy Eur. J. Allergy Clin. Immunol. 2011, 66, 540–548. [Google Scholar] [CrossRef]
- Van der Eerden, B.C.J.; van der Heyden, J.C.; van Hamburg, J.P.; Schreuders-Koedam, M.; Asmawidjaja, P.S.; de Muinck Keizer-Schrama, S.M.; Boot, A.M.; Lubberts, E.; Drop, S.L.S.; van Leeuwen, J.P.T.M. A human vitamin D receptor mutation causes rickets and impaired Th1/Th17 responses. Bone 2014, 69, 6–11. [Google Scholar] [CrossRef]
- Milovanovic, M.; Heine, G.; Hallatschek, W.; Opitz, B.; Radbruch, A.; Worm, M. Vitamin D receptor binds to the ε germline gene promoter and exhibits transrepressive activity. J. Allergy Clin. Immunol. 2010, 126, 1016–1023.e4. [Google Scholar] [CrossRef]
- Heine, G.; Tabeling, C.; Hartmann, B.; González Calera, C.R.; Kühl, A.A.; Lindner, J.; Radbruch, A.; Witzenrath, M.; Worm, M. 25-Hydroxvitamin D 3 Promotes the Long-Term Effect of Specific Immunotherapy in a Murine Allergy Model. J. Immunol. 2014, 193, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; Snyder, L.; Lin, Y.D.; Yang, L. Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients 2015, 7, 3011–3021. [Google Scholar] [CrossRef] [PubMed]
- Keating, P.; Munim, A.; Hartmann, J.X. Effect of vitamin D on T-helper type 9 polarized human memory cells in chronic persistent asthma. Ann. Allergy Asthma Immunol. 2014, 112, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Schögler, A.; Muster, R.J.; Kieninger, E.; Casaulta, C.; Tapparel, C.; Jung, A.; Moeller, A.; Geiser, T.; Regamey, N.; Alves, M.P. Vitamin D represses rhinovirus replication in cystic fibrosis cells by inducing LL-37. Eur. Respir. J. 2016, 47, 520–530. [Google Scholar] [CrossRef]
- Calton, E.K.; Keane, K.N.; Raizel, R.; Rowlands, J.; Soares, M.J.; Newsholme, P. Winter to summer change in vitamin D status reduces systemic inflammation and bioenergetic activity of human peripheral blood mononuclear cells. Redox Biol. 2017, 12, 814–820. [Google Scholar] [CrossRef]
- Al-Jaderi, Z.; Maghazachi, A.A. Effects of vitamin D3, calcipotriol and FTY720 on the expression of surface molecules and cytolytic activities of human natural killer cells and dendritic cells. Toxins 2013, 5, 1932–1947. [Google Scholar] [CrossRef]
- Almerighi, C.; Sinistro, A.; Cavazza, A.; Ciaprini, C.; Rocchi, G.; Bergamini, A. 1α,25-Dihydroxyvitamin D3 inhibits CD40L-induced pro-inflammatory and immunomodulatory activity in Human Monocytes. Cytokine 2009, 45, 190–197. [Google Scholar] [CrossRef]
- Biggs, L.; Yu, C.; Fedoric, B.; Lopez, A.F.; Galli, S.J.; Grimbaldeston, M.A. Evidence that vitamin D3 promotes mast cell-dependent reduction of chronic UVB-induced skin pathology in mice. J. Exp. Med. 2010, 207, 455–463. [Google Scholar] [CrossRef]
- Coussens, A.K.; Wilkinson, R.J.; Hanifa, Y.; Nikolayevskyy, V.; Elkington, P.T.; Islam, K.; Timms, P.M.; Venton, T.R.; Bothamley, G.H.; Packe, G.E.; et al. Vitamin D accelerates resolution of inflammatory responses during tuberculosis treatment. Proc. Natl. Acad. Sci. USA 2012, 109, 15449–15454. [Google Scholar] [CrossRef]
- Ferreira, G.B.; Gysemans, C.A.; Demengeot, J.; da Cunha, J.P.M.C.M.; Vanherwegen, A.-S.; Overbergh, L.; Van Belle, T.L.; Pauwels, F.; Verstuyf, A.; Korf, H.; et al. 1,25-Dihydroxyvitamin D 3 Promotes Tolerogenic Dendritic Cells with Functional Migratory Properties in NOD Mice. J. Immunol. 2014, 192, 4210–4220. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Gu, B.; Lv, X.; Yu, Z.; Wang, R.; Zhou, X.; Qiao, W.; Mao, Z.; Zuo, G.; Li, Q.; et al. 1, 25-dihydroxy-vitamin D3 with tumor necrosis factor-alpha protects against rheumatoid arthritis by promoting p53 acetylation-mediated apoptosis via sirt1 in synoviocytes. Cell Death Dis. 2016, 7, e2423. [Google Scholar] [CrossRef]
- Khare, D.; Godbole, N.M.; Pawar, S.D.; Mohan, V.; Pandey, G.; Gupta, S.; Kumar, D.; Dhole, T.N.; Godbole, M.M. Calcitriol [1, 25[OH]2 D3] pre- and post-treatment suppresses inflammatory response to influenza A (H1N1) infection in human lung A549 epithelial cells. Eur. J. Nutr. 2013, 52, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Ota, K.; Dambaeva, S.; Kim, M.W.; Han, A.R.; Fukui, A.; Gilman-Sachs, A.; Beaman, K.; Kwak-Kim, J. 1,25-Dihydroxy-vitamin D3 regulates NK-cell cytotoxicity, cytokine secretion, and degranulation in women with recurrent pregnancy losses. Eur. J. Immunol. 2015, 45, 3188–3199. [Google Scholar] [CrossRef]
- Telcian, A.G.; Zdrenghea, M.T.; Edwards, M.R.; Laza-Stanca, V.; Mallia, P.; Johnston, S.L.; Stanciu, L.A. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antivir. Res. 2017, 137, 93–101. [Google Scholar] [CrossRef]
- Terrier, B.; Derian, N.; Schoindre, Y.; Chaara, W.; Geri, G.; Zahr, N.; Mariampillai, K.; Rosenzwajg, M.; Carpentier, W.; Musset, L.; et al. Restoration of regulatory and effector T cell balance and B cell homeostasis in systemic lupus erythematosus patients through vitamin D supplementation. Arthritis Res. Ther. 2012, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Urry, Z.; Chambers, E.S.; Xystrakis, E.; Dimeloe, S.; Richards, D.F.; Gabryšová, L.; Christensen, J.; Gupta, A.; Saglani, S.; Bush, A.; et al. The role of 1α,25-dihydroxyvitamin D3 and cytokines in the promotion of distinct Foxp3+and IL-10+ CD4+ T cells. Eur. J. Immunol. 2012, 42, 2697–2708. [Google Scholar] [CrossRef]
- Mahalingam, D.; Radhakrishnan, A.K.; Amom, Z.; Ibrahim, N.; Nesaretnam, K. Effects of supplementation with tocotrienol-rich fraction on immune response to tetanus toxoid immunization in normal healthy volunteers. Eur. J. Clin. Nutr. 2011, 65, 63–69. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin E administration may decrease the incidence of pneumonia in elderly males. Clin. Interv. Aging 2016, 11, 1379–1385. [Google Scholar] [CrossRef]
- Ren, Z.; Pae, M.; Dao, M.C.; Smith, D.; Meydani, S.N.; Wu, D. Dietary supplementation with tocotrienols enhances immune function in C57BL/6 mice. J. Nutr. 2010, 140, 1335–1341. [Google Scholar] [CrossRef]
- Uddin, M.Z.; Rana, M.S.; Hossain, S.; Ferdous, S.; Dutta, E.; Dutta, M.; Emran, T.B. In vivo neuroprotective, antinociceptive, anti-inflammatory potential in Swiss albino mice and in vitro antioxidant and clot lysis activities of fractionated Holigarna longifolia Roxb. bark extract. J. Complement. Integr. Med. 2020, 17, 20190102. [Google Scholar] [CrossRef]
- Zingg, J.M.; Han, S.N.; Pang, E.; Meydani, M.; Meydani, S.N.; Azzi, A. In vivo regulation of gene transcription by alpha- and gamma-tocopherol in murine T lymphocytes. Arch. Biochem. Biophys. 2013, 538, 111–119. [Google Scholar] [CrossRef]
- Abdala-Valencia, H.; Sergejs, B.; Soveg, F.W.; Cook-Mills, J.M. A-tocopherol supplementation of allergic female mice inhibits development of CD11c+CD11b+ dendritic cells in utero and allergic inflammation in neonates. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 307, L482–L496. [Google Scholar] [CrossRef]
- Han, S.N.; Adolfsson, O.; Lee, C.-K.; Prolla, T.A.; Ordovas, J.; Meydani, S.N. Signal transduction, transcriptional regulation, and apoptosis pathways in T cells. J. Immunol. 2006, 177, 6052–6061. [Google Scholar] [CrossRef]
- Marko, M.G.; Pang, H.J.E.; Ren, Z.; Azzi, A.; Huber, B.T.; Bunnell, S.C.; Meydani, S.N. Vitamin E reverses impaired linker for activation of T cells activation in T cells from aged C57BL/6 mice. J. Nutr. 2009, 139, 1192–1197. [Google Scholar] [CrossRef]
- Zheng, X.; Hou, Y.; He, H.; Chen, Y.; Zhou, R. Synthetic vitamin K analogs inhibit in fl ammation by targeting the NLRP3 in fl ammasome. Cell. Mol. Immunol. 2020, 18, 1–9. [Google Scholar] [CrossRef]
- Yu, Y.; Li, Y.; Gao, F.; Hu, Q.; Zhang, Y.; Chen, D.; Wang, G. Vitamin K 2 suppresses rotenone-induced microglial activation in vitro. Nat. Publ. Gr. 2016, 37, 1178–1189. [Google Scholar] [CrossRef]
- Al Mahmud, Z.; Emran, T.B.; Qais, N.; Bachar, S.C.; Sarker, M.; Uddin, M.M.N. Evaluation of analgesic, anti-inflammatory, thrombolytic and hepatoprotective activities of roots of Premna esculenta (Roxb). J. Basic Clin. Physiol. Pharmacol. 2016, 27, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Duan, F.; Yu, Y.; Guan, R.; Xu, Z.; Liang, H.; Hong, L. Vitamin K2 Induces Mitochondria-Related Apoptosis in Human Bladder Cancer Cells via ROS and JNK / p38 MAPK Signal Pathways. PLoS ONE 2016, 11, e0161886. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.S.H.; Hossain, M.M.; Kabir, M.I.; Rahman, M.M.; Hasanat, A.; Emran, T.B.; Rahman, M.A. Phytochemical screening, Antioxidant, Thrombolytic, alpha-amylase inhibition and cytotoxic activities of ethanol extract of Steudnera colocasiifolia K. Koch leaves. J. Young Pharm. 2016, 8, 391. [Google Scholar] [CrossRef]
- Liu, X.; Montissol, S.; Id, A.U.; Ganley, S.; Grossestreuer, A.V.; Berg, K.; Heydrick, S.; Donnino, M.W. The Effects of Thiamine on Breast Cancer Cells. Molecules 2018, 23, 1464. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Fan, Z.; Zhang, Y.; Yu, R.; Zhou, C.; Luo, J.; Ke, Z.; Alerts, E. Thiamine Deficiency Promotes T Cell Infiltration in Experimental Autoimmune Encephalomyelitis: The Involvement of CCL2. J. Immunol. 2014, 193, 2157–2167. [Google Scholar] [CrossRef]
- Antenor, J.; De Andrade, A.; Roberto, C.; Gayer, M.; Pereira, N.; Nogueira, D.A.; Paes, M.C.; Lúcia, V.; Cunha, F.; Bastos, C.; et al. The effect of thiamine deficiency on inflammation, oxidative stress and cellular migration in an experimental model of sepsis José. J. Inflamm. 2014, 11, 1–10. [Google Scholar]
- Rakib, A.; Nain, Z.; Sami, S.A.; Mahmud, S.; Islam, A.; Ahmed, S.; Siddiqui, A.B.F.; Babu, S.O.F.; Hossain, P.; Shahriar, A.; et al. A molecular modelling approach for identifying antiviral selenium-containing heterocyclic compounds that inhibit the main protease of SARS-CoV-2: An in silico investigation. Brief. Bioinform. 2021, 22, 1476–1498. [Google Scholar] [CrossRef] [PubMed]
- Mazur-bialy, A.I.; Pocheć, E. Vitamin B2 deficiency enhances the pro-inflammatory activity of adipocyte, consequences for insulin resistance and metabolic syndrome development. Life Sci. 2017, 178, 9–16. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Prakash, M.D.; Kuol, N.; Nurgali, K.; Stojanovska, L.; Apostolopoulos, V. Anti-Tumor E ff ects of Vitamin B2, B6 and B9 in Promonocytic Lymphoma Cells. Int. J. Mol. Sci. 2019, 20, 3763. [Google Scholar] [CrossRef]
- Tareq, A.M.; Farhad, S.; Uddin, A.N.; Hoque, M.; Nasrin, M.S.; Uddin, M.M.R.; Hasan, M.; Sultana, A.; Munira, M.S.; Lyzu, C.; et al. Chemical profiles, pharmacological properties, and in silico studies provide new insights on Cycas pectinata. Heliyon 2020, 6, e04061. [Google Scholar] [CrossRef]
- Digby, J.E.; Mcneill, E.; Dyar, O.J.; Lam, V.; Greaves, D.R.; Choudhury, R.P. Anti-inflammatory effects of nicotinic acid in adipocytes demonstrated by suppression of fractalkine, RANTES, and MCP-1 and upregulation of adiponectin. Atherosclerosis 2010, 209, 89–95. [Google Scholar] [CrossRef]
- Ganji, S.H.; Qin, S.; Zhang, L.; Kamanna, V.S.; Kashyap, M.L. Niacin inhibits vascular oxidative stress, redox-sensitive genes, and monocyte adhesion to human aortic endothelial cells. Atherosclerosis 2009, 202, 68–75. [Google Scholar] [CrossRef]
- Meyers, C.D.; Liu, P.; Kamanna, V.S.; Kashyap, M.L. Nicotinic acid induces secretion of prostaglandin D 2 in human macrophages: An in vitro model of the niacin flush. Atherosclerosis 2007, 192, 253–258. [Google Scholar] [CrossRef]
- Salem, H.A.; Wadie, W. Effect of Niacin on Inflammation and Angiogenesis in a Murine Model of Ulcerative Colitis. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Giri, B.; Malik, A.; Khodadadi, H.; Morgan, J.C.; Chong, R.K.; Baban, B. Niacin modulates macrophage polarization in Parkinson’s disease. J. Neuroimmunol. 2018, 320, 76–79. [Google Scholar] [CrossRef]
- Emran, T.B.; Nasir Uddin, M.M.; Rahman, A.; Uddin, Z.; Islam, M. Phytochemical, antimicrobial, cytotoxic, analgesic and anti-inflammatory properties of Azadirachta indica: A therapeutic study. J. Bioanal. Biomed. 2015, S12, 1–7. [Google Scholar] [CrossRef]
- Kuroishi, T.; Endo, Y.; Muramoto, K.; Sugawara, S. Biotin deficiency up-regulates TNF- production in murine macrophages. J. Leukoc. Biol. 2008, 83, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Yesmin, S.; Paul, A.; Naz, T.; Rahman, A.A.; Akhter, S.F.; Wahed, M.I.I.; Emran, T.B.; Siddiqui, S.A. Membrane stabilization as a mechanism of the anti-inflammatory activity of ethanolic root extract of Choi (Piper chaba). Clin. Phytosci. 2020, 6, 1–10. [Google Scholar] [CrossRef]
- Agrawal, S.; Agrawal, X.A.; Said, H.M. Biotin deficiency enhances the inflammatory response of human dendritic cells. Am. J. Physiol.-Cell Physiol. 2016, 311, 386–391. [Google Scholar] [CrossRef]
- Meadows, D.N.; Bahous, R.H.; Best, A.F.; Rozen, R. High dietary folate in mice alters immune response and reduces survival after malarial infection. PLoS ONE 2015, 10, e0143738. [Google Scholar] [CrossRef] [PubMed]
- Kunisawa, J.; Hashimoto, E.; Ishikawa, I.; Kiyono, H. A Pivotal Role of Vitamin B9 in the Maintenance of Regulatory T Cells In Vitro and In Vivo. PloS ONE 2012, 7, e32094. [Google Scholar] [CrossRef]
- Paniz, C.; Bertinato, J.F.; Lucena, M.R.; De Carli, E.; da Silva Amorim, P.M.; Gomes, G.W.; Palchetti, C.Z.; Figueiredo, M.S.; Pfeiffer, C.M.; Fazili, Z.; et al. A daily dose of 5 mg folic acid for 90 days is associated with increased serum unmetabolized folic acid and reduced natural killer cell cytotoxicity in healthy Brazilian adults. J. Nutr. 2017, 147, 1677–1685. [Google Scholar] [CrossRef]
- Kolb, A.F.; Petrie, L. Folate deficiency enhances the inflammatory response of macrophages. Mol. Immunol. 2013, 54, 164–172. [Google Scholar] [CrossRef]
- Li, F.; Bahnson, E.M.; Wilder, J.; Siletzky, R.; Hagaman, J.; Nickekeit, V.; Hiller, S.; Ayesha, A.; Feng, L.; Levine, J.S.; et al. Redox Biology Oral high dose vitamin B12 decreases renal superoxide and post-ischemia / reperfusion injury in mice. Redox Biol. 2020, 32, 101504. [Google Scholar] [CrossRef]
- Birch, C.S.; Brasch, N.E.; McCaddon, A.; Williams, J.H.H. A novel role for vitamin B12: Cobalamins are intracellular antioxidants in vitro. Free Radic. Biol. Med. 2009, 47, 184–188. [Google Scholar] [CrossRef]
- Politis, A.; Olgiati, P.; Malitas, P.; Albani, D.; Signorini, A.; Polito, L.; De Mauro, S.; Zisaki, A.; Piperi, C.; Stamouli, E.; et al. Vitamin B12 levels in Alzheimer’s disease: Association with clinical features and cytokine production. J. Alzheimer’s Dis. 2010, 19, 481–488. [Google Scholar] [CrossRef]
- Moreira, E.S.; Brasch, N.E.; Yun, J. Vitamin B12 protects against superoxide-induced cell injury in human aortic endothelial cells. Free Radic. Biol. Med. 2011, 51, 876–883. [Google Scholar] [CrossRef]
- Watanabe, S.; Ide, N.; Ogawara, H.; Yokohama, A.; Mitsui, T.; Handa, H.; Koiso, H.; Tsukamoto, N.; Saitoh, T.; Murakami, H. High percentage of regulatory T cells before and after vitamin B12 treatment in patients with pernicious anemia. Acta Haematol. 2015, 133, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Al-daghri, N.M.; Rahman, S.; Sabico, S.; Yakout, S.; Mcternan, P.G.; Alokail, M.S. Association of Vitamin B12 with Pro-Inflammatory Cytokines and Biochemical Markers Related to Cardiometabolic Risk in Saudi Subjects. Nutrients 2016, 8, 460. [Google Scholar] [CrossRef] [PubMed]
- Lewicki, S.; Lewicka, A.; Kalicki, B.; Kłos, A.; Bertrandt, J.; Zdanowski, R. The influence of vitamin B 12 supplementation on the level of white blood cells and lymphocytes phenotype in rats fed a low-protein diet. Cent.-Eur. J. Immunol. 2014, 39, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Kitabayashi, C.; Fukada, T.; Kanamoto, M.; Ohashi, W.; Hojyo, S.; Atsumi, T.; Ueda, N.; Azuma, I.; Hirota, H.; Murakami, M.; et al. Zinc suppresses Th17 development via inhibition of STAT3 activation. Int. Immunol. 2010, 22, 375–386. [Google Scholar] [CrossRef]
- Maywald, M.; Wang, F.; Rink, L. Zinc supplementation plays a crucial role in T helper 9 differentiation in allogeneic immune reactions and non-activated T cells. J. Trace Elem. Med. Biol. 2018, 50, 482–488. [Google Scholar] [CrossRef]
- Shankar, A.H.; Prasad, A.S. Zinc and immune function: The biological basis of altered resistance to infection. Am. J. Clin. Nutr. 1998, 68, 447S–463S. [Google Scholar] [CrossRef]
- George, M.M.; Vignesh, K.S.; Figueroa JA, L.; Caruso, J.A.; Deepe, G.S. Zinc induces dendritic cell tolerogenic phenotype and skews regulatory T Cell-Th17 balance. J. Immunol. 2016, 197, 1864–1876. [Google Scholar] [CrossRef] [PubMed]
- Gammoh, N.Z.; Rink, L. Zinc and the Immune System. In Nutrition and Immunity; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Wu, D.; Lewis, E.; Pae, M.; Meydani, S. Nutritional Modulation of Immune Function: Analysis of Evidence, Mechanisms, and Clinical Relevance. Front. Immunol. 2019, 9, 3160. [Google Scholar] [CrossRef] [PubMed]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014, 19, 164–174. [Google Scholar] [PubMed]
- Moraes, J.A.; Barcellos-de-Souza, P.; Rodrigues, G.; Nascimento-Silva, V.; Silva, S.V.; Assreuy, J.; Arruda, M.A.; Barja-Fidalgo, C. Heme modulates smooth muscle cell proliferation and migration via NADPH oxidase: A counter-regulatory role for heme oxygenase system. Atherosclerosis 2012, 224, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Chiabrando, D.; Vinchi, F.; Fiorito, V.; Tolosano, E. Haptoglobin and Hemopexin in Heme Detoxification and Iron Recycling. In Acute Phase Proteins—Regulation and Functions of Acute Phase Proteins; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Quintela-Carvalho, G.; Luz, N.F.; Celes, F.S.; Zanette, D.L.; Andrade, D.; Menezes, D.; Tavares, N.M.; Brodskyn, C.I.; Prates, D.B.; Gonçalves, M.S.; et al. Heme drives oxidative stress-associated cell death in human neutrophils infected with Leishmania infantum. Front. Immunol. 2017, 8, 1620. [Google Scholar] [CrossRef]
- Dutra, F.F.; Bozza, M.T. Heme on innate immunity and inflammation. Front. Pharmacol. 2014, 5, 115. [Google Scholar] [CrossRef]
- Ward, R.J.; Crichton, R.R.; Taylor, D.L.; Della Corte, L.; Srai, S.K.; Dexter, D.T. Iron and the immune system. J. Neural Transm. 2011, 118, 315–328. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H. Design and lab-test result of APol, a polarimeter for the Atacama sub-millimeter telescope experiment. In Ground-Based and Airborne Instrumentation for Astronomy VII. International Society for Optics and Photonics; SPIE: Bellingham, DC, USA, 2018; Volume 10702. [Google Scholar] [CrossRef]
- Zhang, Y.; Jung, Y.-G.; Zhang, J. Multiscale and multiphysics modeling of metal AM. In Multiscale Modeling of Additively Manufactured Metals; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–10. [Google Scholar] [CrossRef]
- Zhang, Q.; Cui, T.; Chang, Y.; Zhang, W.; Li, S.; He, Y.; Li, B.; Liu, L.; Wang, G.; Gao, T.; et al. HO-1 regulates the function of Treg: Association with the immune intolerance in vitiligo. J. Cell. Mol. Med. 2018, 22, 4335–4343. [Google Scholar] [CrossRef]
- Zhang, D.L.; Wu, J.; Shah, B.N.; Greutélaers, K.C.; Ghosh, M.C.; Ollivierre, H.; Su, X.Z.; Thuma, P.E.; Bedu-Addo, G.; Mockenhaupt, F.P.; et al. Erythrocytic ferroportin reduces intracellular iron accumulation, hemolysis, and malaria risk. Science 2018, 359, 1520–1523. [Google Scholar] [CrossRef]
- Kuvibidila, S.R.; Baliga, S.B.; Chandra, L.C.; French, C.L. The role of iron in immunity and inflammation: Implications for the response to infection. Diet Immun. Inflamm. 2013, 2013, 193–220. [Google Scholar] [CrossRef]
- Nkengfack, G.; Englert, H.; Haddadi, M. Selenium and Immunity. In Nutrition and Immunity; Springer: Berlin/Heidelberg, Germany, 2019; pp. 159–165. [Google Scholar] [CrossRef]
- Avery, J.; Hoffmann, P. Selenium, Selenoproteins, and Immunity. Nutrients 2018, 10, 1203. [Google Scholar] [CrossRef]
- Huang, Z.; Rose, A.H.; Hoffmann, P. The role of selenium in inflammation and immunity: From molecular mechanisms to therapeutic opportunities. Antioxid. Redox Signal. 2012, 16, 705–743. [Google Scholar] [CrossRef] [PubMed]
- Chisenga, C.C.; Kelly, P. The Role of Selenium in Human Immunity. Med. J. Zambia 2014, 41, 181–185. [Google Scholar]
- Mahmoodpoor, A.; Hamishehkar, H.; Shadvar, K.; Ostadi, Z.; Sanaie, S.; Saghaleini, S.H.; Nader, N.D. The Effect of Intravenous Selenium on Oxidative Stress in Critically Ill Patients with Acute Respiratory Distress Syndrome. Immunol. Investig. 2019, 48, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Hachimura, S.; Totsuka, M.; Hosono, A. Immunomodulation by food: Impact on gut immunity and immune cell function. Biosci. Biotechnol. Biochem. 2018, 82, 584–599. [Google Scholar] [CrossRef]
- Bilal, M.Y.; Dambaeva, S.; Kwak-Kim, J.; Gilman-Sachs, A.; Beaman, K.D. A role for iodide and thyroglobulin in modulating the function of human immune cells. Front. Immunol. 2017, 8, 1573. [Google Scholar] [CrossRef]
- Mahima; Ingle, A.M.; Verma, A.K.; Tiwari, R.; Karthik, K.; Chakraborty, S.; Deb, Q.; Rajagunalan, S.; Rathore, R.; Dhama, K. Immunomodulators in day to day life: A review. Pak. J. Biol. Sci. 2013, 16, 826–843. [Google Scholar] [CrossRef] [PubMed]
- Moshtaghi Kasaniam, G.H.; Hasennajad, M. The effects of excess iodine on immune system; an in-vitro study. J. Kerman Univ. Med. Sci. 2011, 18, 235–245. [Google Scholar] [CrossRef]
- Pickering, G.; Mazur, A.; Trousselard, M.; Bienkowski, P.; Yaltsewa, N.; Amessou, M.; Noah, L.; Pouteau, E. Magnesium status and stress: The vicious circle concept revisited. Nutrients 2020, 12, 3672. [Google Scholar] [CrossRef]
- Nielsen, F.H. Magnesium deficiency and increased inflammation: Current perspectives. J. Inflamm. Res. 2018, 11, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Almousa, L.A.; Salter, A.M.; Langley-Evans, S.C. Magnesium deficiency heightens lipopolysaccharide-induced inflammation and enhances monocyte adhesion in human umbilical vein endothelial cells. Magnes. Res. 2018, 31, 39–48. [Google Scholar] [CrossRef]
- Suzuki-Kakisaka, H.; Sugimoto, J.; Tetarbe, M.; Romani, A.M.; Ramirez Kitchen, C.M.; Bernstein, H.B. Magnesium Sulfate Increases Intracellular Magnesium Reducing Inflammatory Cytokine Release in Neonates. Am. J. Reprod. Immunol. 2013, 70, 213–220. [Google Scholar] [CrossRef]
- Da Silva Lima, F.; da Rocha Romero, A.B.; Hastreiter, A.; Nogueira-Pedro, A.; Makiyama, E.; Colli, C.; Fock, R.A. An insight into the role of magnesium in the immunomodulatory properties of mesenchymal stem cells. J. Nutr. Biochem. 2018, 55, 200–208. [Google Scholar] [CrossRef]
- Sugimoto, J.; Romani, A.M.; Valentin-Torres, A.M.; Luciano, A.A.; Ramirez Kitchen, C.M.; Funderburg, N.; Mesiano, S.; Bernstein, H.B. Magnesium Decreases Inflammatory Cytokine Production: A Novel Innate Immunomodulatory Mechanism. J. Immunol. 2012, 188, 6338–6346. [Google Scholar] [CrossRef]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary reference intakes: Vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Am. Diet. Assoc. 2001, 101, 294–301. [Google Scholar] [CrossRef]
- Higdon, J.; Drake, V.; Aschner, M. Micronutrient Information Center. Miner. Copp. 2010, 18, 80–95. [Google Scholar]
- Yin, J.J.; Fu, P.P.; Lutterodt, H.; Zhou, Y.T.; Antholine, W.E.; Wamer, W. Dual role of selected antioxidants found in dietary supplements: Crossover between anti- and pro-oxidant activities in the presence of copper. J. Agric. Food Chem. 2012, 60, 2554–2561. [Google Scholar] [CrossRef]
- Urbański, N.K.; Berȩsewicz, A. Generation of ·OH initiated by interaction of Fe2+ and Cu+ with dioxygen; comparison with the Fenton chemistry. Acta Biochim. Pol. 2000, 47, 951–962. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, E.; Maywald, M.; Hilgers, R.D.; Brieger, A.; Clarner, T.; Kipp, M.; Plümäkers, B.; Meyer, S.; Schwerdtle, T.; Rink, L. Induction of regulatory T cells in Th1-/Th17-driven experimental autoimmune encephalomyelitis by zinc administration. J. Nutr. Biochem. 2016, 29, 116–123. [Google Scholar] [CrossRef]
- Mayer, L.S.; Uciechowski, P.; Meyer, S.; Schwerdtle, T.; Rink, L.; Haase, H. Differential impact of zinc deficiency on phagocytosis, oxidative burst, and production of pro-inflammatory cytokines by human monocytes. Metallomics 2014, 6, 1288–1295. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, E.; Metz, C.H.D.; Maywald, M.; Hilgers, R.D.; Weßels, I.; Senff, T.; Haase, H.; Jager, M.; Ott, M.; Aspinall, R.; et al. Zinc supplementation induces regulatory T cells by inhibition of Sirt-1 deacetylase in mixed lymphocyte cultures. Mol. Nutr. Food Res. 2016, 60, 661–671. [Google Scholar] [CrossRef]
- Von Bülow, V.; Dubben, S.; Engelhardt, G.; Hebel, S.; Plümäkers, B.; Heine, H.; Rink, L.; Haase, H. Zinc-Dependent Suppression of TNF-α Production Is Mediated by Protein Kinase A-Induced Inhibition of Raf-1, IκB Kinase β, and NF-κB. J. Immunol. 2007, 179, 4180–4186. [Google Scholar] [CrossRef]
- Te Velthuis, A.J.W.; van den Worml, S.H.E.; Sims, A.C.; Baric, R.S.; Snijder, E.J.; van Hemert, M.J. Zn2+ inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010, 6, e1001176. [Google Scholar] [CrossRef]
- Haase, H.; Ober-Blöbaum, J.L.; Engelhardt, G.; Hebel, S.; Heit, A.; Heine, H.; Rink, L. Zinc Signals Are Essential for Lipopolysaccharide-Induced Signal Transduction in Monocytes. J. Immunol. 2008, 181, 6491–6502. [Google Scholar] [CrossRef]
- Liu, M.J.; Bao, S.; Gálvez-Peralta, M.; Pyle, C.J.; Rudawsky, A.C.; Pavlovicz, R.E.; Killilea, D.W.; Li, C.; Nebert, D.W.; Wewers, M.D.; et al. ZIP8 Regulates Host Defense through Zinc-Mediated Inhibition of NF-κB. Cell Rep. 2013, 3, 386–400. [Google Scholar] [CrossRef]
- Agoro, R.; Taleb, M.; Quesniaux, V.F.J.; Mura, C. Cell iron status influences macrophage polarization. PLoS ONE 2018, 13, e0196921. [Google Scholar] [CrossRef] [PubMed]
- Handa, P.; Morgan-Stevenson, V.; Maliken, B.D.; Nelson, J.E.; Washington, S.; Westerman, M.; Yeh, M.M.; Kowdley, K.V. Iron overload results in hepatic oxidative stress, immune cell activation, and hepatocellular ballooning injury, leading to nonalcoholic steatohepatitis in genetically obese mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 310, G117–G127. [Google Scholar] [CrossRef]
- Shen, C.C.; Liang, H.J.; Wang, C.C.; Liao, M.H.; Jan, T.R. Iron oxide nanoparticles suppressed T helper 1 cell-mediated immunity in a murine model of delayed-type hypersensitivity. Int. J. Nanomed. 2012, 7, 2729–2737. [Google Scholar] [CrossRef]
- Zanganeh, S.; Hutter, G.; Spitler, R.; Lenkov, O.; Mahmoudi, M.; Shaw, A.; Pajarinen, J.S.; Nejadnik, H.; Goodman, S.; Moseley, M.; et al. Polarization in Tumour Tissues. Nat. Nanotechnol. 2016, 11, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Gan, Z.S.; Wang, Q.Q.; Li, J.H.; Wang, X.L.; Wang, Y.Z.; Du, H.H. Iron reduces M1 macrophage polarization in RAW264.7 macrophages associated with inhibition of STAT1. Mediat. Inflamm. 2017, 2017, 8570818. [Google Scholar] [CrossRef]
- Hoffmann, F.K.W.; Hashimoto, A.C.; Shafer, L.A.; Dow, S.; Berry, M.J.; Hoffmann, P.R. Dietary selenium modulates activation and differentiation of CD4 + T cells in mice through a mechanism involving cellular free thiols. J. Nutr. 2010, 140, 1155–1161. [Google Scholar] [CrossRef]
- Smith, A.D.; Cheung, L.; Beshah, E.; Shea-Donohue, T.; Urban, J.F. Selenium status alters the immune response and expulsion of adult heligmosomoides bakeri worms in mice. Infect. Immun. 2013, 81, 2546–2553. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.M.; Lei, X.; Prabhu, K.S. Selenium levels affect the IL-4-induced expression of alternative activation markers in murine macrophages. J. Nutr. 2011, 141, 1754–1761. [Google Scholar] [CrossRef]
- Liu, Y.; Qiu, C.; Li, W.; Mu, W.; Li, C.; Guo, M. Selenium Plays a Protective Role in Staphylococcus aureus-Induced Endometritis in the Uterine Tissue of Rats. Biol. Trace Elem. Res. 2016, 173, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Kassam, S.; Goenaga-Infante, H.; Maharaj, L.; Hiley, C.T.; Juliger, S.; Joel, S.P. Methylseleninic acid inhibits HDAC activity in diffuse large B-cell lymphoma cell lines. Cancer Chemother. Pharmacol. 2011, 68, 815–821. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, Z.; Li, Y.; Shen, P.; Hu, X.; Cao, Y.; Zhang, N. Selenium Deficiency Facilitates Inflammation Following S. aureus Infection by Regulating TLR2-Related Pathways in the Mouse Mammary Gland. Biol. Trace Elem. Res. 2016, 172, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Yazdi, M.H.; Mahdavi, M.; Varastehmoradi, B.; Faramarzi, M.A.; Shahverdi, A.R. The immunostimulatory effect of biogenic selenium nanoparticles on the 4T1 breast cancer model: An in vivo study. Biol. Trace Elem. Res. 2012, 149, 22–28. [Google Scholar] [CrossRef]
- Tsuji, P.A.; Carlson, B.A.; Anderson, C.B.; Seifried, H.E.; Hatfield, D.L.; Howard, M.T. Dietary selenium levels affect selenoprotein expression and support the interferon-γ and IL-6 immune response pathways in mice. Nutrients 2015, 7, 6529–6549. [Google Scholar] [CrossRef]
- Xue, H.; Wang, W.; Li, Y.; Shan, Z.; Li, Y.; Teng, X.; Gao, Y.; Fan, C.; Teng, W. Selenium upregulates CD4(+)CD25(+) regulatory T cells in iodine-induced autoimmune thyroiditis model of NOD.H-2(h4) mice. Endocr. J. 2010, 57, 595–601. [Google Scholar] [CrossRef]
- Bentley-Hewitt, K.L.; Chen, R.K.Y.; Lill, R.E.; Hedderley, D.I.; Herath, T.D.; Matich, A.J.; Mckenzie, M.J. Consumption of selenium-enriched broccoli increases cytokine production in human peripheral blood mononuclear cells stimulated ex vivo, a preliminary human intervention study. Mol. Nutr. Food Res. 2014, 58, 2350–2357. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Xu, H.; Wang, C.; Qin, H.; An, Z. Magnesium enhances the chondrogenic differentiation of mesenchymal stem cells by inhibiting activated macrophage-induced inflammation. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Liang, R.Y.; Wu, W.; Huang, J.; Jiang, S.P.; Lin, Y. Magnesium affects the cytokine secretion of CD4+ T lymphocytes in acute asthma. J. Asthma 2012, 49, 1012–1015. [Google Scholar] [CrossRef]
- Takemoto, S.; Yamamoto, A.; Tomonaga, S.; Funaba, M.; Matsui, T. Magnesium deficiency induces the emergence of mast cells in the liver of rats. J. Nutr. Sci. Vitaminol. 2013, 59, 560–563. [Google Scholar] [CrossRef]
- Malpuech-Brugère, C.; Nowacki, W.; Daveau, M.; Gueux, E.; Linard, C.; Rock, E.; Lebreton, J.P.; Mazur, A.; Rayssiguier, Y. Inflammatory response following acute magnesium deficiency in the rat. Biochim. Biophys. Acta Mol. Basis Dis. 2000, 1501, 91–98. [Google Scholar] [CrossRef]
- Zhou, X.; Zhao, L.; Luo, J.; Tang, H.; Xu, M.; Wang, Y.; Yang, X.; Chen, H.; Li, Y.; Ye, G.; et al. The toxic effects and mechanisms of Nano-Cu on the spleen of rats. Int. J. Mol. Sci. 2019, 20, 1469. [Google Scholar] [CrossRef]
- Hien, L.T.T.; Trang, P.T.T.; Phuong, P.C.; Tam, P.T.; Xuan, N.T. Effects of nano-copper on maize yield and inflammatory response in mice. Iran. J. Basic Med. Sci. 2019, 22, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Carrascosa, J.P.; Cotán, D.; Jurado, I.; Oropesa-Ávila, M.; Sánchez-Martín, P.; Savaris, R.F.; Tan, J.; Sánchez-Alcázar, J.A.; Tan, S.L.; Horcajadas, J.A. The effect of copper on endometrial receptivity and induction of apoptosis on decidualized human endometrial stromal cells. Reprod. Sci. 2018, 25, 985–999. [Google Scholar] [CrossRef] [PubMed]

| Vitamins | Dose/Concentration | Study Model | Findings | References |
|---|---|---|---|---|
| Vitamin A | 500 nM | In vitro (peripheral blood mononuclear cells) | RA inhibits IFN-γ, IL-17, and IL-9 production in CD4+ T cells, upregulates CD103 expression and gut-homing receptors | [44] |
| 0.1 μM | In vitro (peripheral blood mononuclear cells) | RA prevents human nTregs from converting to Th1 and/or Th17 cells and sustains their Foxp3 expression and suppressive function, suppresses IL-1 receptor (IL-1R) upregulation, accelerates IL-6R downregulation | [45] | |
| 0.01 M | In vitro (myelomonocytic cells) | Retinoids inhibit mumps virus in vitro due to upregulation of type I interferon (IFN) and IFN stimulated genes | [108] | |
| Concentration in olive oil suspension 25 mg/mL and inject intraperitoneally 125 μg/gm weight | In vivo (Mice) | Retinoic acid acts intrinsically in developing gut- tropic premucosal dendritic cell, directs generation of intestine-like cDC1 and cDC2 subsets | [109] | |
| retinyl acetate (25 µg/d) | In vivo (Mice) | Effective for clearance of Citrobacter rodentium, fewer CD8αβ+ cells and more CD8αα+ and T cell receptor (TCR)γδ+ T cells, enhanced IL17 | [110] | |
| 5 or 25 nM | In vitro (Human tonsillar B cells) | Increases IgA production, the expression of germ-line IgA1, IgA2 transcripts (GLTa1 and GLTa2), and the frequency of IgA1-secreting B cell clones, and induces IgA isotype switching | [111] | |
| 1 µM retinol | In vitro (Murine respiratory tract lung epithelial cell) | Increases IgA production by lipopolysaccharide (LPS)-stimulated splenocytes cultures, and increases the expression of MCP-1, IL-6, and GM-CSF | [112] | |
| Control diet of retinol palmitate 15 IU/g | In vivo (Mice) | Causes reduction of immunodominant CD8+ T cell frequencies in the lower respiratory tract (LRT) airways of VAD animals, T cells, and shows unusually high CD10 | [113] | |
| 1 μM | In vivo (mouse splenocytes) | Lowers Th17 T-cell activity, downregulating IL-6, promoting B cell production of IgA, upregulating IL-6, and increasing transcript levels of MCP-1, GMCSF, and IL-10 in MACs | [114] | |
| 0 or 15 IU g−1 vitamin A | In vivo (Mice) | Increases cytokine/chemokine gene expression and cytokine protein production in VAD animals. Viral infection persists longer in the upper and lower respiratory tract of VAD mice | [115] | |
| 250 µg per day | In vivo (transgenic mice) | Regulates the homeostasis of pre-Dendritic cell (DC)–derived DC subsets and have implications for the management of immune deficiencies | [116] | |
| 0 or 15 IU/g vitamin A palmitate | In vivo (Mice) | VAS decreases death and diarrhea-related mortalities in children, and increases immune responses toward pediatric vaccines | [117] | |
| 0 to 25,000 IU kg−1 | In vivo (Mice) | Controls local lymphoid tissue inducer cell differentiation and maturation upstream of the transcription factor RORct | [118] | |
| 10 nM RA | In vivo (Mice) | Abrogates RA signaling in B cells; these cells were not able to induce a4b7 expression. RA signaling in B cells is important for the induction of IgA+ GC, effective gut humoral response, and to maintain a normal microbiota composition. It has a direct effect on IgA plasma cell differentiation | [119] | |
| 60 mg/kg | In vivo (Mice) | Lowers interferon (IFN)-g production, activates NKT cells, decreases extracellular signal-regulated kinase (ERK) phosphorylation, and enhances phosphatase 2A (PP2A) activity | [120] | |
| Vitamin C | 2 mg/kg, 150 mg/kg | In vivo (piglets) | Decreases vulva length, width, height, and area,. Decreses the concentrations of BUN, CRE, AST and TBIL in serum, and reduces IgA, IgG and IgM levels. Restoring serum estradiol (E2), progesterone (PROG), luteinizing hormone (LH) and follicle stimulating hormone (FSH) levels in weaning piglets | [121] |
| 50 to 200 mg/kg/24 h. | In vitro (venous blood sample) | Increases the expression of the antimicrobial proteins, ↓MtDNA levels | [122] | |
| 200 mg/kg | In vivo (Mice) | Decreases lung neutrophil extracellular traps, decreases circulating free-DNA following peritonitis-induced sepsis. In vitamin C deficient neutrophils, Upregulates endoplasmic reticulum stress associated gene expression, Induces autophagy signaling and increases PAD4 mRNA | [123] | |
| 162 ± 8 mg vitamin C per 100 g fruit | In vitro (venous blood) | No effect on control (non-stimulated) neutrophil migration, increases neutrophil oxidative burst, improvement of important neutrophil functions | [124] | |
| 57 mg/kg/day | In vivo (Mice IRI) | Lowers the severity of tubular injury and renal arterial resistance | [125] | |
| 100 nM | In vitro (peripheral blood mononuclear cells) | Increases oxidative stress, lowers proinflammatory mediator, ROS, TNF-α,and IL-6, LPS-Induced Autophagy in MH-S Cell Lines | [126] | |
| 200 mg/kg | In vivo (Mice) | Prevents sepsis-mediated disassembly of the Na+-K+-ATPase pump, causes cytoskeletal rearrangements, changes in viscoelastic properties, increases epithelial ion channel and transporter expression, and increases alveolar fluid clearance | [127] | |
| 3.3 g/L | In vivo (Mice) | Increases type I interferons (IFNs), increases IFN-α and -β, increases infiltration of inflammatory cells (IL-1α/β and TNF-α) into the lung | [128] | |
| Vitamin D | 1 to 100 nM | In vitro (peripheral B cell line) | Increases IgE production by 63.9 ± 5.9%., decreases CD19+ CD27high CD38+ B cell population, and decreases AID expression | [129] |
| 0.1 μM, 1.0 μM, 10 μM | In vitro (human PBMCs) | Lowers T helper (Th) cell population-specific cytokine expression of interferon γ (Th1), interleukins IL-17A (Th17) and IL-22 (Th17/Th22), ↑IL-4 (Th2) levels | [130] | |
| (6 µg/kg/day in PBS/0.03% ethanol) | In vivo (Mice) | Lowers the IgE response in a type I allergy mouse model | [129] | |
| n.m | In vitro (PBMCs) | Causes inhibition of IgE production by calcitriol is mediated by its transrepressive activity through the VDR-corepressor complex | [131] | |
| 0.05–50 mg/kg 25(OH)D | In vivo (mice) | Decreases the allergic airway inflammation, Th2 cytokine expression in the lungs. And the humoral immune reaction | [132] | |
| 0–50 nM | In vivo (Mice) and human PBMCs | Decreases CD8 and CD4 T cells proliferation, and IL-2, ↓IFN-γ and ↓IL-17 production, increases IL-4 and IL-10 production and human Treg development | [133] | |
| 25(OH)cholecalciferol (40 ng/mL) or 1,25(OH)2cho- lecalciferol (20 ng/mL) | In vitro (peripheral blood mononuclear cells) | Lowers interleukin (IL)-9, IL-5, and IL-8. Increases IL-13þ cells, downregulates transcription factors PU.1 and interferon regulatory factor 4 | [134] | |
| 10−9 M, 10−8 M or 10−7 M | In vitro (Primary bronchial epithelial cells) | Causes suppression of viral replication, and increases LL-37 expression | [135] | |
| 5(OH)D(<50 nmol/L, 50–75 nmol/L ≥75 nmol/L) | In vitro (peripheral blood mononuclear cells) | Reduces systemic inflammation | [136] | |
| 100, 10 or 1 ng/mL of vitamin D3 | In vitro (human monocytes) | Upregulates the expression of NK cytotoxicity receptors NKp30 and NKp44, as well as NKG2D, downregulates the expression of the killer inhibitory receptor CD158, and downregulates the expression of CCR6 on the surface of iDCs | [137] | |
| 10 nM 1,25D3 | In vitro (peripheral blood mononuclear cells) | inhibits the cytokine response of CD40L-stimulated macrophages, decreases CD40L-induced expression, decreases tumor necrosis factor (TNF)-α production, decreases interleukin (IL)-1β production, and (IFN)- γ proliferation, and increases IL-10 production | [138] | |
| Vitamin D3 (cholecalciferol) at >2000 IU/kg | In vivo (Mice) | 1,25(OH)2D3/VDR-dependent induction of IL-10 production can contribute to the mast cell’s ability to suppress inflammation and skin pathology at sites of chronic UVB irradiation | [139] | |
| four fortnightly doses of 2.5 mg vitamin D3 | In vitro | Increases chemokines (AMP MMP-9) and antigen-stimulated Th1 cytokine suppression, decreases IL-4, CCL5, and IFN-α secretion, accelerates sputum smear conversion, and enhances treatment-induced resolution of lymphopenia, monocytosis, hypercytokinemia, and hyperchemokinemia | [140] | |
| 10−8 M | In vivo (Mice) | Restrains the inflammatory response of NOD and C57BL/6 BM-derived DCs, decreases secretion of CCL3, CCL4, CCL5, and CXCL10, increases secretion of CCL2 and CCL7, decreases T cell stimulatory capacity, and increases migration-competent tolerogenic DCs | [141] | |
| 1 μg/kg VD | In vivo (Mice) | Upregulates p53 acetylation-mediated apoptosis in MH7A cells, promotes Sirt1 translocation and apoptosis of FLSs | [142] | |
| 100/30 nM | In vitro (Human alveolar epithelial cell line) | Decreases autophagy, enhances apoptosis, decreases H1N1-induced TNF-α level, IFN-b (interferon-beta), and IFN-stimulated gene-15. Downregulates IL-8 as well as IL-6 RNA levels and suppresses the H1N1- induced transcription | [143] | |
| 10 or 100 nM | In vitro (peripheral blood mononuclear cells) | Causes suppression of NK cytotoxicity and downregulation of CD107a expression in NK cells, increases CD158a and CD158b expression, decreases the expression of NKp30 and NKp44 on CD56+CD3− NK cells, and lowers CD56+/IFN- γ+ and CD56+/TNF-α+ | [144] | |
| 100 nmol | In vitro (bronchial epithelial cell line) | Decreases rhinovirus replication and release, and increases rhinovirus-induced interferon stimulated genes and cathelicidins | [145] | |
| 100,000 IU of cholecalciferol per week for 4 weeks, followed by 100,000 IU of cholecalciferol per month for 6 months | In vitro (SLE patients) | Increases naïve CD4+ T cells and regulatory T cells, decreases effector Th1 and Th17 cells, and decreases memory B cells and anti-DNA antibodies | [146] | |
| 5 ng/mL | In vitro (peripheral blood mononuclear cells) | Increases Foxp3+ and IL-10+ CD4+T cells | [147] | |
| Vitamin E | TRF supplements (200 mg each capsule) 400 mg per day | In vitro (human blood leukocytes) | Enhances production of IFN-y, f IL-4, IL-6, and TNF-α | [148] |
| 50 mg/d | In vivo (Human) | Decreases incidence of pneumonia by 69% | [149] | |
| 1 kg of T3 supplemented diet (1 g Tocomin 50% +39 g vitamin E- stripped soybean oil + 0.96 kg basal die) | In vivo (Mice) | Increases splenocyte IL-1b production, Lymphocyte proliferation, tumor necrosis factor-a, and interferon-ꭚ | [150] | |
| 50–200 μg /mL | In vitro (human PBMCs) | Increases cAMP production, IL-2 production, IL-17, IL-8, and RANTES | [151] | |
| 30 or 500 ppm of vitamin E (RRR-a-Tocopheryl acetate) | In vivo (Mice) | Gamma-T was more effective but less specific than alpha-T in the presence of vitamin E; CD40L is strongly upregulated by alpha-T, but down-regulated by gamma-T, gamma-T appears to better prevent e induction of gene expression upon T cell stimulation, e.g. of some cytokines (interleukin 3 and 10) and chemokines (chemokine ligand 9, 10, and 11) | [152] | |
| 250 to 500 mg D-α-tocopherol/kg diet | In vivo (Mice) | Decreases lung CD11b+ dendritic cell subsets, lung mRNA expression of IL-4, IL-33, TSLP, CCL11, and CCL24 | [153] | |
| 30 (control) or 500 (supplemented) ppm of vitamin E | In vivo (Mice) | Increases the expression of genes (Ccnb2, Cdc2, Cdc6) in old T cells. Increases upregulation of IL-2 expression, decreases upregulation of IL-4, has impact on signal transduction, transcriptional regulation, and apoptosis pathways in T cells | [154] | |
| 46 mmol/L of vitamin E | In vitro (Mice spleen cells) | Vitamin E eliminates the age-related differences in LAT phosphorylation in both T cell subsets, and difference in the tyrosine phosphorylation of LAT | [155] | |
| Vitamin K | vitamin K (1–5 μM) | In vitro (Bone marrow-derived macrophages) | Causes inhibitors of the NLRP3 inflammasome to block the interaction between NLRP3 and ASC, which attenuates the severity of inflammation | [156] |
| MK-4 (0.5–20 μmol/L) | In vitro (microglial cell line (BV2)) | Suppresses the upregulation in the expression of iNOS and COX-2 in the cells and the production of TNF-α and IL-1β. Inhibits ROS production, p38 activation, and rotenone-induced nuclear translocation of NF-κB in BV2 cells | [157] | |
| 20 mM menadione | In vitro (Mice Splenocytes) | Increases ROS levels, thus suppressing production in lymphocytes and CD4 + T cells, and activation of ERK, JNK and NF- κB | [158] | |
| Vitamin K2 | In vitro (human bladder cancer cell lines) | Induces apoptosis in bladder cancer cells, generates reactive oxy- gen species (ROS), phosphorylating of c-Jun N-terminal kinase (JNK) and p38 MAPK | [159] | |
| 0.1–100 μM | In vitro (PBMCs) | Suppressing the mitogen-activated proliferation, inhibiting the production of tumor necrosis factor (TNF) α, interleukin (IL)-4, -6, and -10, and increases Treg cells | [160] | |
| Vitamin B1 | (0 µg/mL, 0.125 µg/mL, 0.25 µg/mL, 0.5 µg/mL, 1 µg/mL, and 2 µg/mL) | In vitro (breast epithelial cells from non-cancer origin and metastatic site) | Reduces extracellular lactate levels, increases cellular pyruvate dehydrogenase (PDH) activities, decreases non-glycolytic acidification, glycolysis, and glycolytic capacity, and reduces cell proliferation in MCF7 | [161] |
| Not mentioned | In vitro (Mice encephalitogenic cells) | TD aggravated the development of EAE, causing microglial activation, increases leukocyte infiltration in the spinal cord, Th1, and Th17 cells, and upregulates expression of CCL2 | [162] | |
| complete chow (303.3 ± 42.6 nmol/L) thiamine deficiency | In vivo (Mice) | Thiamine deficiency increases TNF-α and MCP-1 concentrations, decreases blood IL-1β level, and increases KC, IL-1 β, and IL-6 | [163] | |
| Vitamin B2 | low (3·1 nM), physiological (10·4 nM) or high (300 and 531 nM) | In vivo (mouse monocyte/macrophage cell line) | Low riboflavin content decreases the proliferation rate and increases apoptotic cell death, completely inhibits the respiratory burst and slightly impairs phagocytosis, and impairs cell adhesion | [164] |
| 3.1 nM to 10.4 nM | In vivo (Mycoplasma-free mouse preadipocytes) | Riboflavin deprivation Induces adipocyte death, increases lipolysis and free fatty acid release, ROS Production, NF-κB Phosphorylation, and pro-inflammatory TNFα and IL-6 | [165] | |
| 1 µg/mL, 0.5 µg/mL, 0.25 µg/mL, 0.125 µg/mL, 0.62 µg/mL, 0.31 µg/mL, 0.15 µg/mL and untreated control 0 µg/mL for vitamin B2 | In vitro (U937 cell line) | Inhibits cell migration of pro-Monocytic cells, decreases the expression of PD-L1, increases secretion of IL-8 and IL-10, and increases GM-CSF | [166] | |
| Vitamin B3 | 100 mg/kg/d niacin | In vivo (guinea pigs) | Downregulates inflammatory factors (IL-6 and TNF-α), suppresses protein expression of CD68 and NF-κB p65, attenuates oxidative stress | [167] |
| 10−3–10−6 M Nicotinic acid | In vitro (preadipocytes cells) | attenuating expression of fractalkine, MCP-1, RANTES, iNOS, and macrophage chemotaxis | [168] | |
| Niacin (0–1 mM) | In vitro (human aortic endothelial cells) | Inhibits production of ROS, LDL oxidation, TNF-α, NF-κB activation, and vascular cell adhesion molecule-1 (VCAM-1) | [169] | |
| Nicotinic acid (0.1–3 mM) | In vitro (human monoblastic leukemia cell line) | Induces macrophage PGD2 secretion | [170] | |
| 80 and 320 mg/kg | In vivo (rats) | Increases colonic MPO activity and TNF-α level, and decreases cytokine IL-10 | [171] | |
| 100 mg and 250 mg niacin | In vivo | Causes macrophage polarization from M1 (pro-inflammatory) to M2 (counter-inflammatory), boosting anti-inflammatory processes, thus suppressing inflammation | [172] | |
| niacin (1, 10, 100 μmol/l) | In vitro (Murine alveolar macrophages) | reduces the levels of TNF-α, IL-6 and IL-1β in LPS-challenged alveolar macrophages, inhibits NF-κB activation, attenuates the LPS-induced pro-inflammatory cytokines | [173] | |
| niacin (0.25–1 mM) | In vitro (umbilical vein endothelial cells) | Decreases IL-6 and TNF-α secretion and inhibits NF-κB p65 and notch1 protein expression | [167] | |
| Vitamin B5 | 10 µM | In vivo (C57BL/6J mice) | significantly inhibits the growth of Mycobacterium tuberculosis by regulating innate immunity and adaptive immunity | [62] |
| Vitamin B6 | (1000 µg/mL, 500 µg/mL, 250 µg/mL, 125 µg/mL, 62.5 µg/mL, 31.25 µg/mL, 15.6 µg/mL | In vitro (U937 cell line) | Inhibit cell migration and proliferation of pro-monocytic cells, decreases the expression of PD-L1 and IL-1β, and increases secretion of IL-8 and IL-10 | [166] |
| 0, 12 mg, and 120 mg per kg diets | In vivo (male BALB/c mice) | Vitamin B6 deficiency decreases growth rate, lymphocyte proliferation, and CD4 T Lymphocyte Differentiation | [66] | |
| 5, 10, 20, and 40 mg/kg | In vivo (Rex rabbits) | Increases IL-2, IFN-γ, M cell number, weight of thymus and spleen, cell division, prolifferation, and maturation | [68] | |
| 100 nM PMA, B6 vitamer (500 µM) | In vivo (Mice) | Vitamin B6 inhibits LPS-induced NF-κB, JNK activation, and gene expression, suppresses NLRP3 inflammasome activation, noncanonical IL-1β secretion and pyroptosis, and inhibits signal 1 and signal 2 for the IL-1β production | [69] | |
| Vitamin B7 | 0.8 mg d-biotin per kg | In vivo (mice) | Biotin deficiency upregulates TNF-α production; however, no differences were detected in NF-κB activity | [174] |
| basal diet (0.8 mg/kg of d-biotin) biotin deficient (biotin free) | In vivo (Mice) | Improves Ni allergy, increases PUFA in rat liver, decreases IL-1b production and proliferation, and decreases TNF-α production | [175] | |
| Control (d-biotin 0.2 mg/L) BD (biotin free) | In vitro (murine macrophage cell line) | Inhibits augmentation of IL-1b production | [175] | |
| 0, 10 and 100 µM biotin | In vitro (Human dendritic cells) | BD enhancing TNF-α, IL-12p40, IL-23, and IL-1β secretion, and IFN-γ and IL-17 induction | [176] | |
| Vitamin B9 | (2 mg/kg diet) | In vivo (Mice) | Decreases total T cells and NK cell, increases TNFα immunoreactive protein, and increases liver Abca1 mRNA | [177] |
| 0.2 to 25 ppb | In vivo (Mice) | In vitro, vit-B9 reduces condition differentiated Treg cells from naïve cells but fails to survive. In vivo, depletion of vit B9 results in reduction of Treg cells in small intestine | [178] | |
| (1000 µg/mL, 500 µg/mL, 250 µg/mL, 125 µg/mL, 62.5 µg/mL, 31.25 µg/mL, 15.6 µg/mL and | In vitro (U937 cell line) | Exerts an anti-tumorigenic effect, inhibits cell migration, proliferation of Pro-Monocytic Cells, lowers the expression of PD-L1, and increases secretion of IL-8 and IL-10 | [166] | |
| 5 mg Folic Acid | In vivo | Increases UMFA concentrations and decreases the number and cytotoxicity of NK cells | [179] | |
| control medium (100 ng/mL) low folate medium (10 ng/mL) and no folate medium | In vitro (mouse monocyte cell line) | Folate deficiency enhances the pro-inflammatory cytokine increase in pro-inflammatory marker expression, blunts the generation of nitric oxide in response to an LPS challenge | [180] | |
| Vitamin B12 | 0.3 μM | In vivo (Mice) | B12 lessens kidney IRI, decreases kidney inflammation and fibrosis, DNA damage response (DDR) and apoptosis, and hypoxia/reperfusion (H/R) injury, and modulates H/R induced chemokine gene expression | [181] |
| 2–64 μM | In vitro | Exerts robust protection against 30 μM concentrations of the pro-oxidants homocysteine and hydrogen peroxide, inhibits intracellular peroxide production, and prevents apoptotic and necrotic cell death | [182] | |
| low (<250 pg/mL) and normal (>250 pg/mL) vitamin B12 | In vitro (PBMCs) | Low vitamin-12 level increases production of IL-6, IFN-γ | [183] | |
| 10–50 nM | In vitro (Primary HAEC) | Prevents homocysteine-induced increases in O2•− and cell death, homocysteine and rotenone-induced mitochondrial O2•− production | [184] | |
| Not mention | In vivo (Mice) | Increases CD8+ T cells, decreases CD4/CD8 ratio, and decreases T reg in control groups than pre and post treatment groups. | [185] | |
| Not mention | Cross- sectional study | Negatively associated with TNF-α, HOMA-IR, serum resistin (children) and positively associated with pro-inflammatory cytokines and biochemical markers | [186] | |
| 300% of the daily intake | In vivo (rats) | Decreased negative impact of protein malnutrition facilitates the production of T lymphocytes | [187] | |
| 1000 µg/day intramuscularly | In vivo | Restoring increased CD4/CD8 ratio and depressed NK cell activity, increases C3, C4, and immunoglobulins | [75] |
| Minerals | Dose/Concentration | Study Model | Findings | References |
|---|---|---|---|---|
| Zinc | 91 mg/kg | In vivo (mice) | Increases Treg cells, decreases severity of EAE, and decreases Th17 RORγT+ cells | [224] |
| 6.77 μM | Pro-inflammatory TNF-α and IL-6 | Causes zinc depletion, decreases production of TNF-α and IL-6, and increases phagocytosis and oxidative burst | [225] | |
| 10 mg/day for 10 days | In vitro (PBMCs) | Decreases Th1-cytokine production and proliferation in MLC, prolongs Foxp3 expression, causes Sirt-1 inhibition and induces regulatory T cells in MLC | [226] | |
| 10 μM, 20 μM, or 45 μM zinc sulfate/pyrithione (50 μM) | In vitro (PBMCs) | Suppresses activation of IκB kinase β (IKKβ) and NF-κB TNF-α release and subsequent TNF-α production. Contributes to anti-inflammatory action of PDE inhibitors | [227] | |
| 2 mM Zn2+ and 2 mM PT | In vitro (Vero-E6 cells) | Inhibits the RNA-synthesizing activity of the RTCs and blocks the initiation step of EAV RNA synthesis | [228] | |
| 1 μM + 10 μM pyrithione | In vitro (PBMCs) | Reduces mRNA expression of proinflammatory cytokines, and inhibits LPS-mediated toxicity | [229] | |
| zinc-deficient diet (0.5–1.5 ppm zinc) or a matched control diet (50.5–51.5 ppm zinc) | In vivo (mice) | Zinc-deficient dietary intake causes excessive inflammation to polymicrobial sepsis in conjunction, ZIP8 is a potent negative regulator of NF-kB activation | [230] | |
| Iron | 10, 50 or 100 μM | In vivo (mice) | Dietary iron loading lowers inflammatory responses such as Il-1β expression, decreases M1 marker, CD86, and I-A/I-E expression, increases IL-4, ↓NF- κB p65 nuclear translocation, and decreases iNos and pro-inflammatory cytokines expression | [231] |
| 250 μM | In vivo (diabetic mice) | Dietary iron overload causes hepatic oxidative stress and NLRP3 inflammasome activation, increase in hepatic inflammatory mediators and immune cell activation, upregulation of chemokine, cytokine, and antioxidant mediators such as iNos, TNF-α, Mcp1, hepcidin (gene name Hamp), Hmox1, and Tlr4 | [232] | |
| (0.2–10 mg iron/kg; 0.2 mL/mouse) | In vivo (mice) | Decreases CD3+ and F4/80+ cells, decreases DTH reactions, and IFN-γ production, increases IL-4 production, and decreases splenic CD11b+ cells | [233] | |
| ferumoxytol (2.73 mg Fe mL−1, 8.37 mg Fe mL−1 | In vivo (mice model) | Inhibits growth of subcutaneous adenocarcinomas Increases the presence of pro-inflammatory M1 macrophages in the tumor tissues | [234] | |
| ferumoxytol (0–30 mg mL−1) | In vitro (mammary tumor cells) | Iincreases caspase-3 activity, increases mRNA associated with pro-inflammatory Th1-type responses, Increases production of tumor-necrosis factor-α (TNF-α) | [234] | |
| FAC (0∼400 μg/mL) | In vitro (RAW264.7 macro-phage cell line) | Decreases mRNA levels of IL-6, IL-1, TNF-α, and decreases iNOS production | [235] | |
| Selenium | 0.08 to 1.00 mg/kg | In vivo (mice) | Increases CD4+ T cell responses and differentiation, increases Ca2+ mobilization, oxidative burst, and NFAT translocation, increases IL-2 transcription, IL-2 receptor expression, and proliferation | [236] |
| with or without 0.2 ppm Se | In vivo (mice) | Mice fed a Se-deficient diet have more adult worms than Se-sufficient diet. Se-sufficient diet increased Il4, Il13, and Il13ra2 expression and decreased Il4 and Il13 expression. Restores anti-fecundity response | [237] | |
| 100 nmol/L Se | In vitro (BMDM culture) | Increases Arg-I, Fizz1, and Mrc-1 expression, decreases TNF-α and IL-1β expression, produces endogenous activators to mediate the PPARg-dependent switch from M1 to M2 phenotype and participate in wound healing and inflammation | [238] | |
| MSA (2.41 μg/mL, 1.5 μg/mL Se) and MSC (4.15 μg/mL, 1.5 μg/mL Se) | In vitro (S. aureus Culture) | Displays a greater defense against uterine inflammatory damage, decreases necrosis factor alpha (TNF-α) and increases interleukin-6 (IL-6), ↓phosphorylation of IκBα and ↓NF-κB p65 | [239] | |
| MSA (0 to 30 μM) | In vitro (DLBCL cell lines) | Inhibits HDAC activity, acetylation of histone H3 | [240] | |
| 0.03 to 1.5 mg/kg | In vivo (mice) | Se-deficient mice increase TNF-α, IL-1β, and IL-6 production, increase mRNA and protein expressions of toll-like receptor 2 (TLR2) | [241] | |
| 100 μg/day | In vivo (mice) | Increases IFN-γ and IL-12 production, higher survival rate, and DTH response | [242] | |
| 0.1 ppm | In vivo (mice) | ↑interferon-γ, ↑interleukin IL-6 | [243] | |
| 100 μg/kg | In vivo (mice) | ↑CD4+ CD25+ Foxp3+ T cells, ↑Foxp3 mRNA expression | [244] | |
| 200 μg of Se | In vitro (PBMCs) | ↑Interleukin (IL-2, IL-4, IL-5, IL-13, and IL-22) | [245] | |
| Magnesium | 60 mg/Lor 2.5 mM | In vitro (PBMCs) | ↓production of TNF-α and IL-6 in maternal and neonatal, ↓cytokine production, ↓NF-kB activation, ↑constitutive IkBa level | [219] |
| 5 nM to 20 nM | In vivo (murine) | Inhibits activation of macrophage, ↓percentage of CCR7-positive cells, ↓cytokines (IL-1β, IL-6 and IL-10), ↓nuclear translocation and phosphorylation of nuclear factor-κB (NF-κB), ↑chondrogenic differentiation of hBMSCs | [246] | |
| Mg2+ (0, 1, 3 and 5 mM) | In vitro (murine MSCs cells) | ↑proliferation rates of MSCs, ↓IL-1β and IL-6, ↑IL-10 and PGE2, ↓pNF-κB/NF-κB, ↑pSTAT-3/STAT-3, modulating the production of IL-1β and IL-6 | [218] | |
| concentrations of Mg- supplemented (0.8, 5, 10, 15, and 20 mmol/L) | In vitro (asthmatic CD4+T cells) | ↓IL-5 and IL-13 secretion, ↑IFN-y secretion, modulating the immune responses of acute asthmatic CD4* T cells | [247] | |
| 499 mg/kg and 44 mg/kg in the control diet and the low-Mg diet, respectively | In vivo (rat) | Mg deficiency increased the levels of mRNA known to be expressed by mast cells in the liver; mast cells were locally distributed around portal triads | [248] | |
| 32 and 950 mg/kg respectively for deficient and control diets | In vivo (rats) | In Mg deficient diet ↑interleukin-6 (IL-6) level, higher u2-macroglobulin and u1-acid glycoprotein, ↑plasma fibrinogen and ↓ albumin concentration | [249] | |
| Copper | 1 µm Cu group, 200 mg/kg CuCl2 group, 200 mg/kg nano-Cu low group, 50 mg/kg nano-Cu medium group, 100 mg/kg nano-Cu nano-Cu high group, 200 mg/kg | In vivo (rat) | Decreased antibody production (IgA, IgG, IgM) altered lymphocyte subpopulation in the spleen, altered the number of blood cells, induces oxidative stress | [250] |
| 20–1000 mg/kg DW nCu- treated seeds (nCu-1000); 2–1000 mg nCu/l-treated mice | In vivo (mice) | Administration of mice with 1000 mg/l nCu leading to inflammatory responses, upregulated expression of serum biochemical indicators of liver and kidney, increased infiltration and activation of splenic immune cells | [251] | |
| 163 mM/L copper sulfate | In vitro (stromal cell) | Copper disrupted the endometrial receptivity signature of dHESCs, decreases IGFBP1 levels, does not increase the apoptosis level | [252] | |
| 163 mM/L copper sulfate | In vitro (endometrial cells) | Changes in the gene expression (42 up- and 9 downregulated), does not increase the apoptosis level induced by the decidualization treatment | [252] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitra, S.; Paul, S.; Roy, S.; Sutradhar, H.; Bin Emran, T.; Nainu, F.; Khandaker, M.U.; Almalki, M.; Wilairatana, P.; Mubarak, M.S. Exploring the Immune-Boosting Functions of Vitamins and Minerals as Nutritional Food Bioactive Compounds: A Comprehensive Review. Molecules 2022, 27, 555. https://doi.org/10.3390/molecules27020555
Mitra S, Paul S, Roy S, Sutradhar H, Bin Emran T, Nainu F, Khandaker MU, Almalki M, Wilairatana P, Mubarak MS. Exploring the Immune-Boosting Functions of Vitamins and Minerals as Nutritional Food Bioactive Compounds: A Comprehensive Review. Molecules. 2022; 27(2):555. https://doi.org/10.3390/molecules27020555
Chicago/Turabian StyleMitra, Saikat, Shyamjit Paul, Sumon Roy, Hriday Sutradhar, Talha Bin Emran, Firzan Nainu, Mayeen Uddin Khandaker, Mohammed Almalki, Polrat Wilairatana, and Mohammad S. Mubarak. 2022. "Exploring the Immune-Boosting Functions of Vitamins and Minerals as Nutritional Food Bioactive Compounds: A Comprehensive Review" Molecules 27, no. 2: 555. https://doi.org/10.3390/molecules27020555
APA StyleMitra, S., Paul, S., Roy, S., Sutradhar, H., Bin Emran, T., Nainu, F., Khandaker, M. U., Almalki, M., Wilairatana, P., & Mubarak, M. S. (2022). Exploring the Immune-Boosting Functions of Vitamins and Minerals as Nutritional Food Bioactive Compounds: A Comprehensive Review. Molecules, 27(2), 555. https://doi.org/10.3390/molecules27020555










