Comparing Variants of the Cell-Penetrating Peptide sC18 to Design Peptide-Drug Conjugates
Abstract
1. Introduction
2. Results and Discussion
2.1. Design and Synthesis of Different Novel sC18 Variants
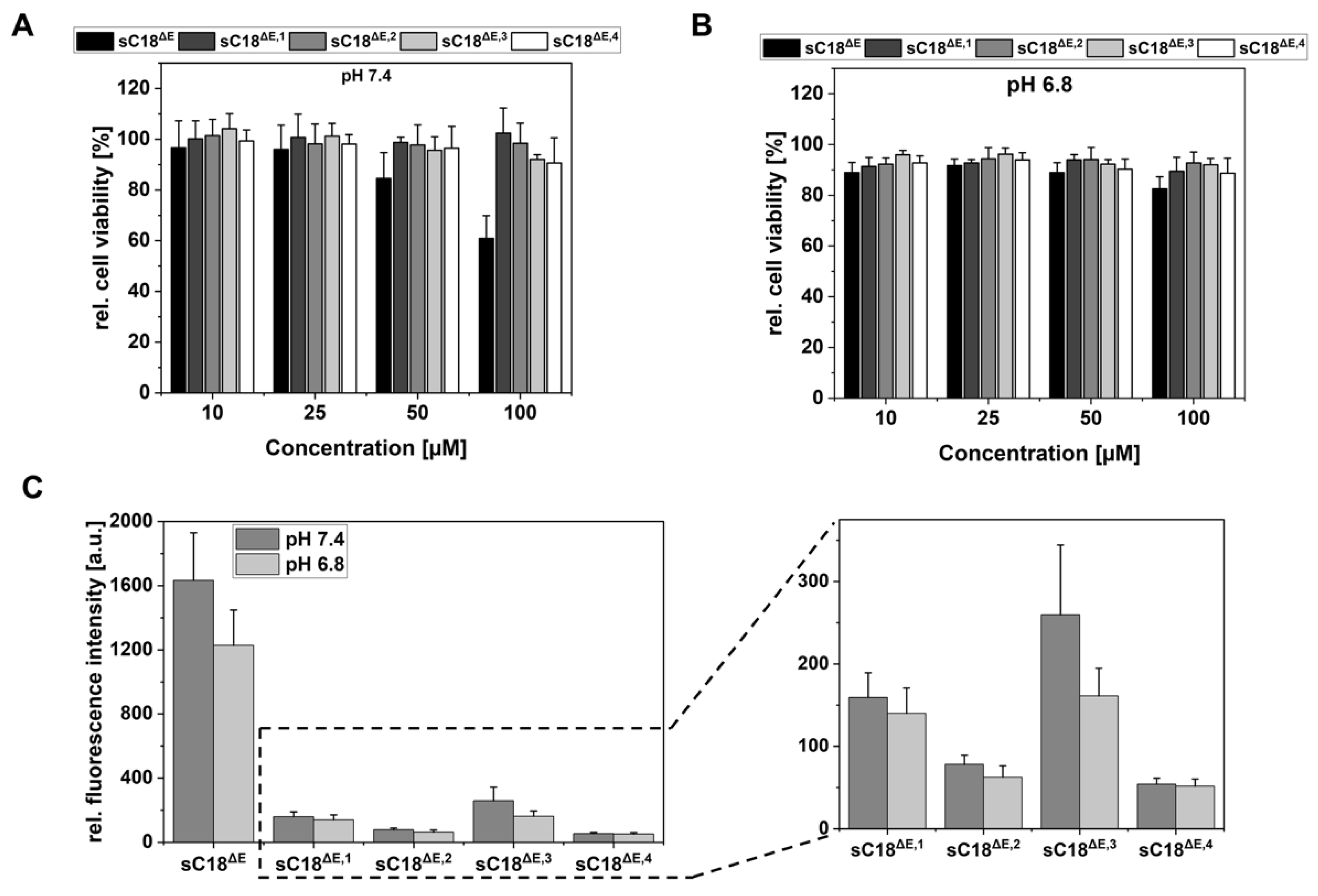
2.2. Analysis of pH-Responsive Histidine Variants
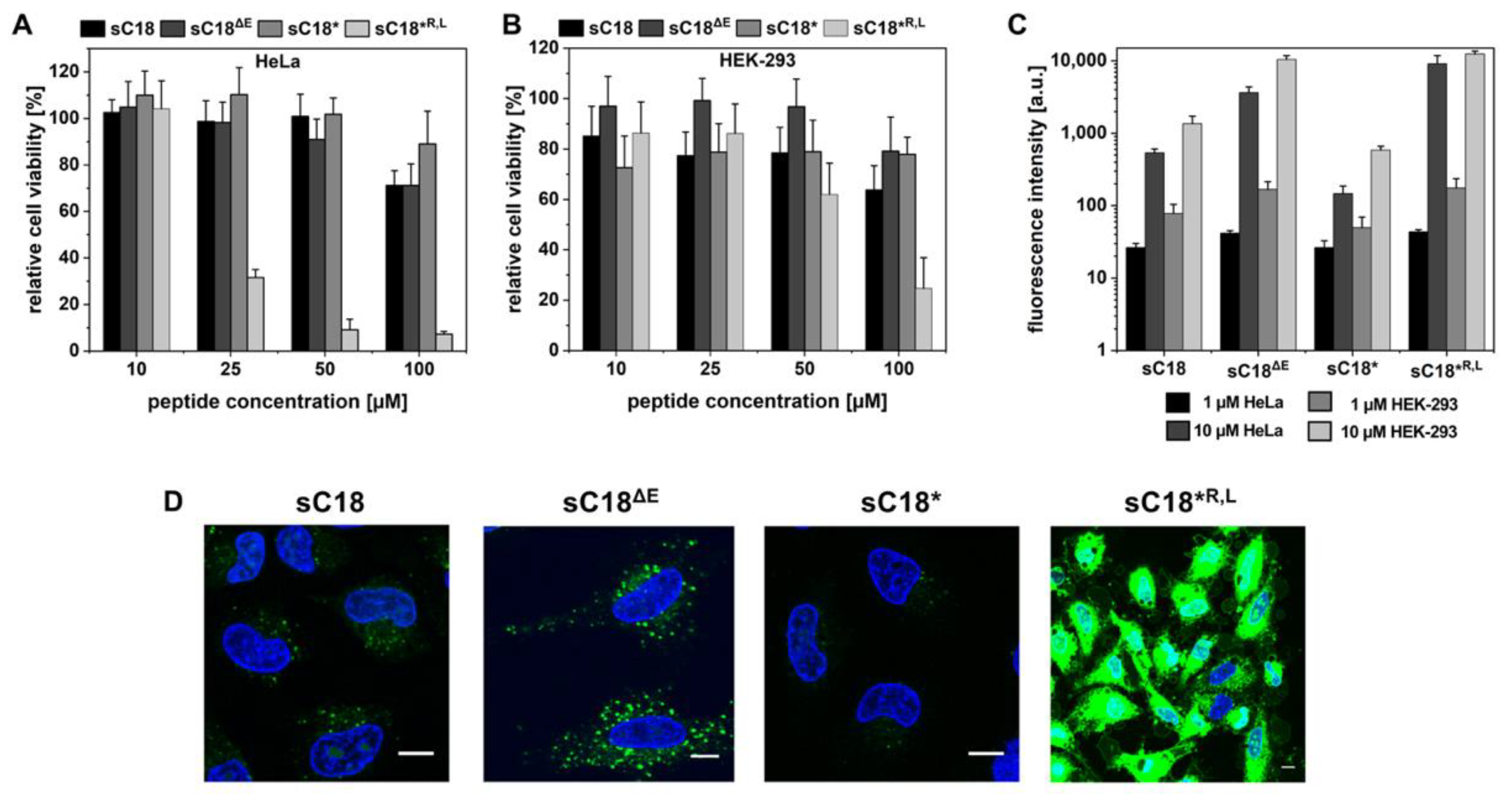
2.3. Evaluation of a Shortened sC18 Variant Comprising a Nearly Perfect Amphipathic Helical Structure
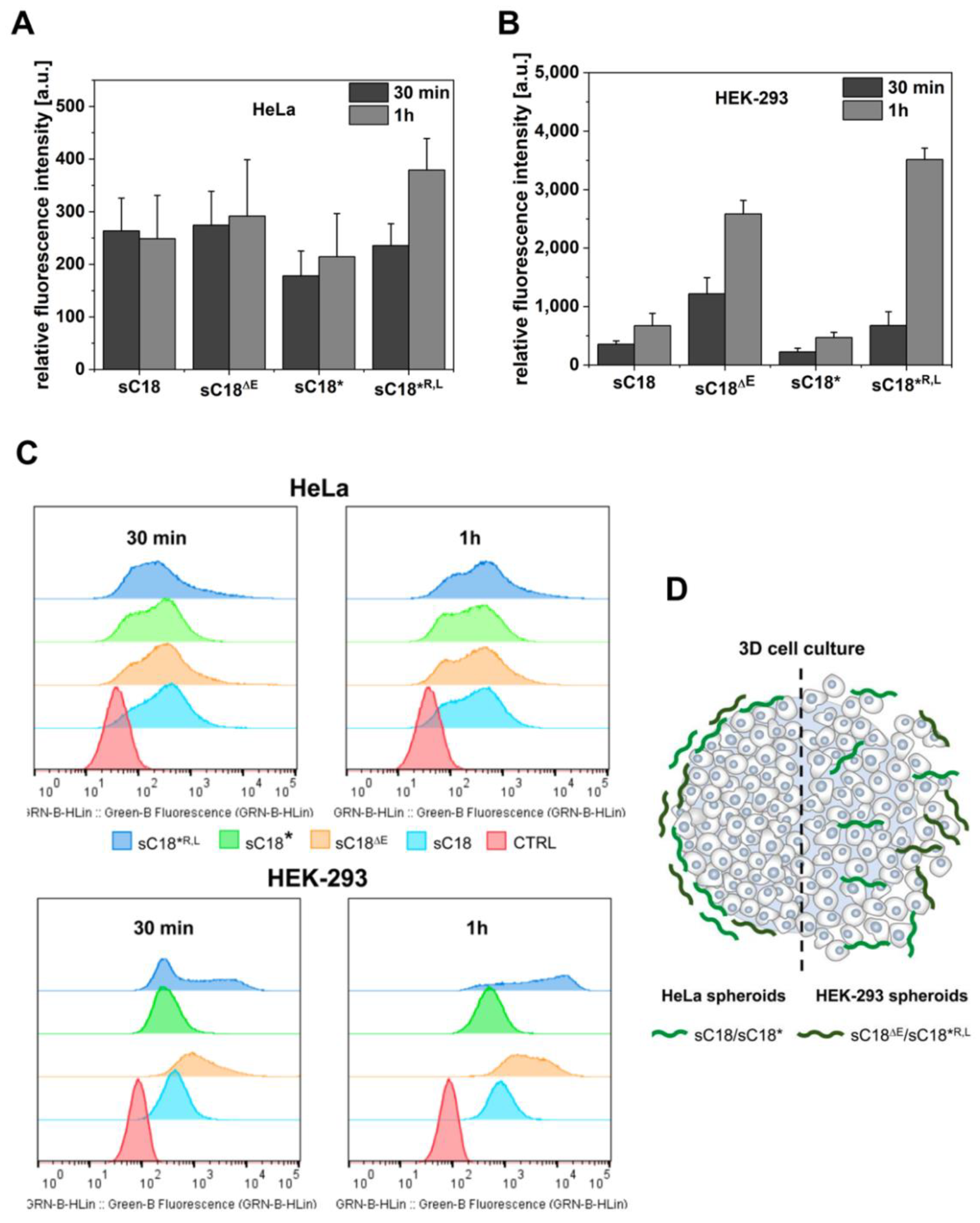
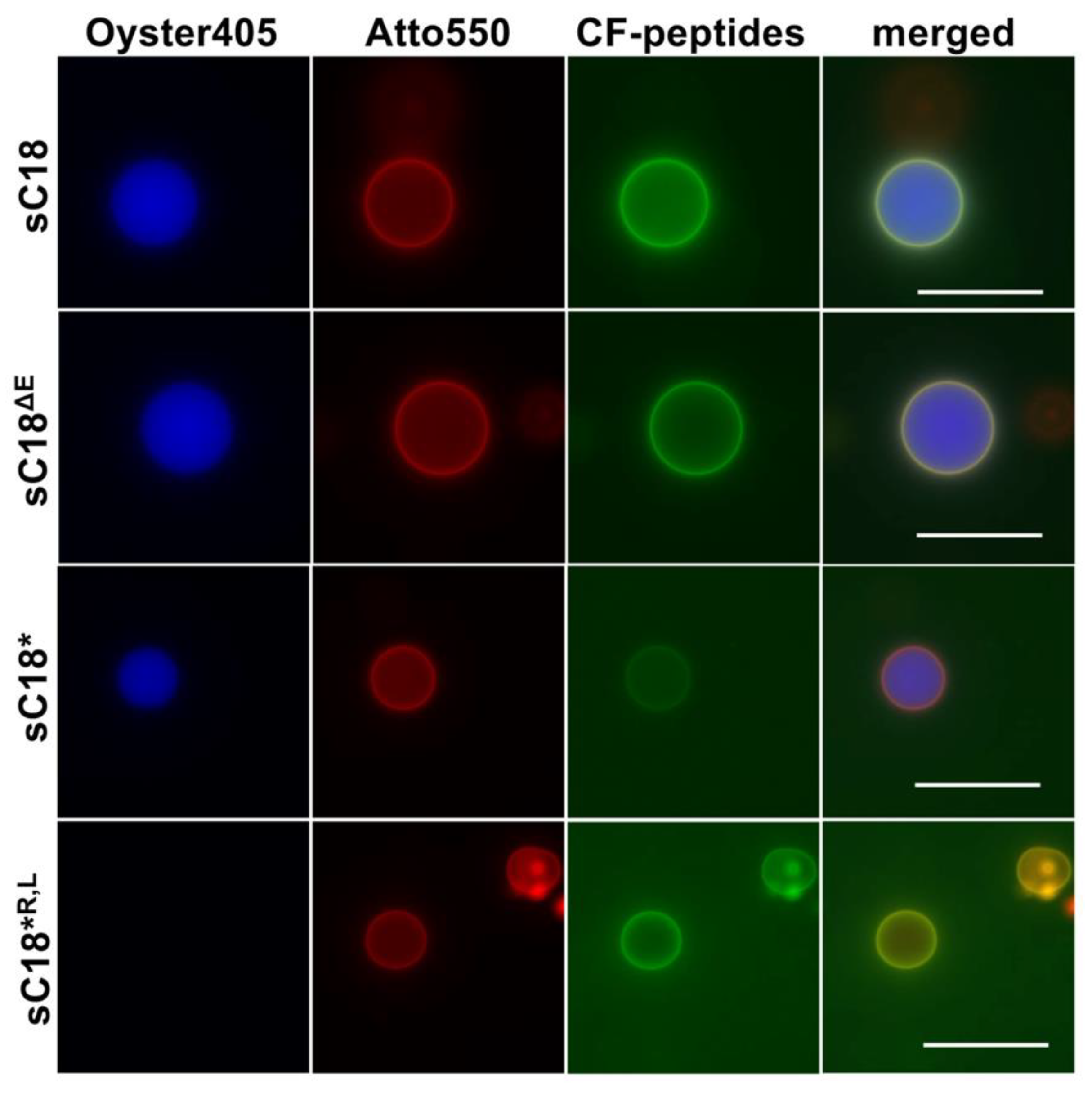
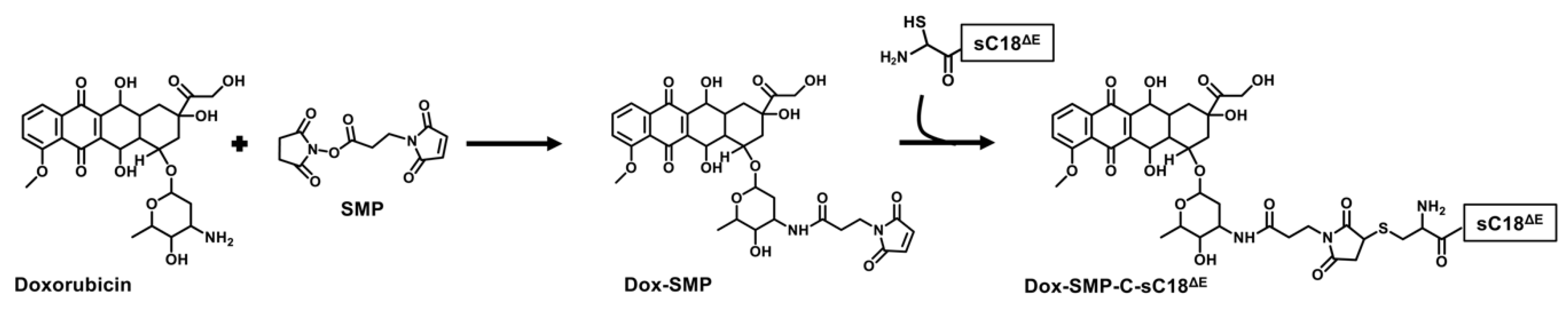
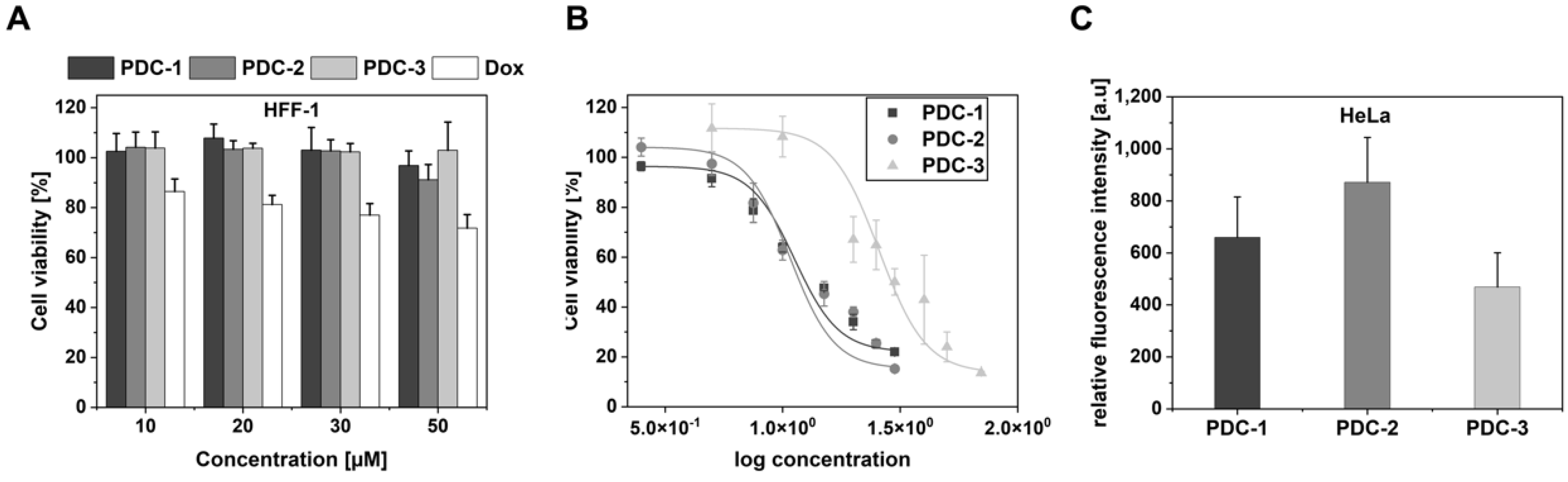
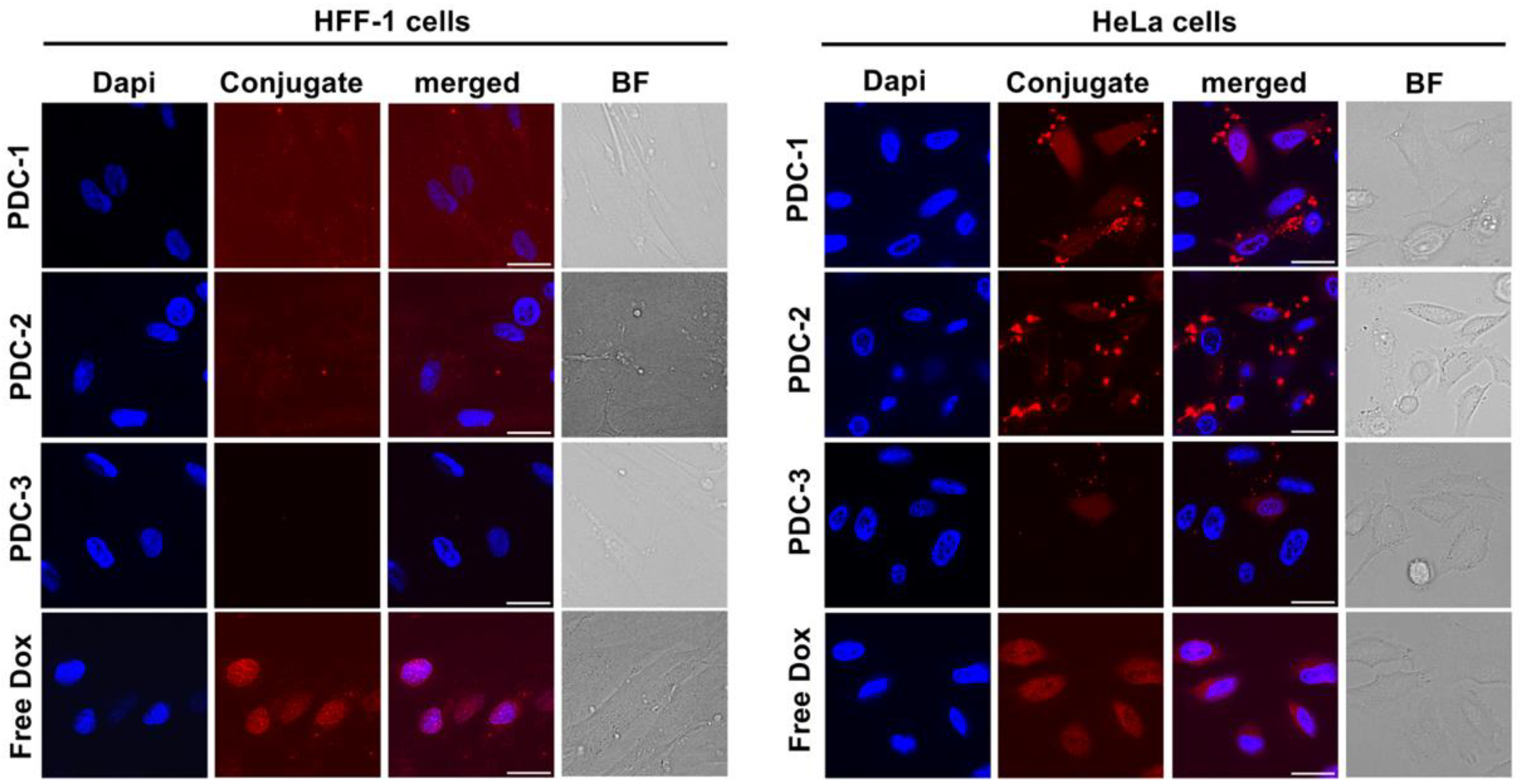
2.4. Synthesis of Peptide-Drug Conjugates (PDCs) and Their Biological Evaluation
3. Materials and Methods
3.1. Materials
3.2. Solid Phase Peptide Synthesis
3.3. PDC Synthesis
3.4. Circular Dichroism (CD) Spectroscopy
3.5. Peptide Interaction with Giant Unilammellar Vesicles (GUVs)
3.6. Cell Culture
3.7. Seeding and Treatment of Tumor Spheroids
3.8. Flow Cytometry Studies
3.9. Microscopy Analysis
3.10. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Derakhshankhah, H.; Jafari, S. Cell penetrating peptides: A concise review with emphasis on biomedical applications. Biomed. Pharmacother. 2018, 108, 1090–1096. [Google Scholar] [CrossRef]
- Reissmann, S. Cell penetration: Scope and limitations by the application of cell-penetrating peptides. J. Pept. Sci. 2014, 20, 760–784. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Bi, Y.; Zhang, H.; Dong, S.; Teng, L.; Lee, R.J.; Yang, Z. Cell-Penetrating Peptides in Diagnosis and Treatment of Human Diseases: From Preclinical Research to Clinical Application. Front. Pharmacol. 2020, 11, 697. [Google Scholar] [CrossRef] [PubMed]
- Gestin, M.; Dowaidar, M.; Langel, Ü. Uptake mechanism of cell-penetrating peptides. Adv. Exp. Med. Biol. 2017, 1030, 255–264. [Google Scholar]
- Sakamoto, K.; Akishiba, M.; Iwata, T.; Arafiles, J.V.V.; Imanishi, M.; Futaki, S. Use of homoarginine to obtain attenuated cationic membrane lytic peptides. Bioorg. Med. Chem. Lett. 2021, 40, 127925. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.I.; Moazzam, M.; Stueber, R.; Park, S.E.; Cho, Y.; Malik, N.A.; Tiwari, R.K. Applications of amphipathic and cationic cyclic cell-penetrating peptides: Significant therapeutic delivery tool. Peptides 2021, 141, 170542. [Google Scholar] [CrossRef] [PubMed]
- Teo, S.L.Y.; Rennick, J.J.; Yuen, D.; Al-Wassiti, H.; Johnston, A.P.R.; Pouton, C.W. Unravelling cytosolic delivery of cell penetrating peptides with a quantitative endosomal escape assay. Nat. Commun. 2021, 12, 1–12. [Google Scholar] [CrossRef]
- Pei, D.; Buyanova, M. Overcoming Endosomal Entrapment in Drug Delivery. Bioconjugate Chem. 2019, 30, 273–283. [Google Scholar] [CrossRef]
- Shoari, A.; Tooyserkani, R.; Tahmasebi, M.; Löwik, D.W.P.M. Delivery of Various Cargos into Cancer Cells and Tissues via Cell-Penetrating Peptides: A Review of the Last Decade. Pharmaceutics 2021, 13, 1391. [Google Scholar] [CrossRef]
- Jauset, T.; Beaulieu, M.E. Bioactive cell penetrating peptides and proteins in cancer: A bright future ahead. Curr. Opin. Pharmacol. 2019, 47, 133–140. [Google Scholar] [CrossRef]
- Neundorf, I.; Rennert, R.; Hoyer, J.; Schramm, F.; Löbner, K.; Kitanovic, I.; Wölfl, S. Fusion of a short HA2-derived peptide sequence to cell-penetrating peptides improves cytosolic uptake, but enhances cytotoxic activity. Pharmaceuticals 2009, 2, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Drexelius, M.; Reinhardt, A.; Grabeck, J.; Cronenberg, T.; Nitsche, F.; Huesgen, P.F.; Maier, B.; Neundorf, I. Multistep optimization of a cell-penetrating peptide towards its antimicrobial activity. Biochem. J. 2021, 478, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Splith, K.; Bergmann, R.; Pietzsch, J.; Neundorf, I. Specific targeting of hypoxic tumor tissue with nitroimidazole-peptide conjugates. ChemMedChem 2012, 7, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Gronewold, A.; Horn, M.; Ranđelović, I.; Tóvári, J.; Vázquez, S.M.; Schomäcker, K.; Neundorf, I. Characterization of a Cell-Penetrating Peptide with Potential Anticancer Activity. ChemMedChem 2017, 12, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Feni, L.; Parente, S.; Robert, C.; Gazzola, S.; Arosio, D.; Piarulli, U.; Neundorf, I. Kiss and Run: Promoting Effective and Targeted Cellular Uptake of a Drug Delivery Vehicle Composed of an Integrin-Targeting Diketopiperazine Peptidomimetic and a Cell-Penetrating Peptide. Bioconjug. Chem. 2019, 30, 2011–2022. [Google Scholar] [CrossRef] [PubMed]
- Horn, M.; Neundorf, I. Design of a novel cell-permeable chimeric peptide to promote wound healing. Sci. Rep. 2018, 8, 16279–16291. [Google Scholar] [CrossRef]
- Negrete-Hurtado, A.; Overhoff, M.; Bera, S.; Bruyckere, E.D.; Schätzmüller, K.; Kye, M.J.; Qin, C.; Lammers, M.; Kondylis, V.; Neundorf, I.; et al. Autophagy lipidation machinery regulates axonal microtubule dynamics but is dispensable for survival of mammalian neurons. Nat. Commun. 2020, 11, 1–19. [Google Scholar] [CrossRef]
- Gessner, I.; Klimpel, A.; Klußmann, M.; Neundorf, I.; Mathur, S. Interdependence of charge and secondary structure on cellular uptake of cell penetrating peptide functionalized silica nanoparticles. Nanoscale Adv. 2020, 2, 453–462. [Google Scholar] [CrossRef]
- Lee, H.J.; Huang, Y.W.; Chiou, S.H.; Aronstam, R.S. Polyhistidine facilitates direct membrane translocation of cell-penetrating peptides into cells. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Tanaka, Y.; Nanasato, Y.; Omura, K.; Endoh, K.; Kawano, T.; Iwasaki, T. Direct protein delivery into intact plant cells using polyhistidine peptides. Biosci. Biotechnol. Biochem. 2021, 85, 1405–1414. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, J.; Wang, F.; Ding, Y.; Wang, T.; Jin, G.; Martz, M.; Gui, Z.; Ouyang, P.; Chen, P. Histidine-Rich Cell-Penetrating Peptide for Cancer Drug Delivery and Its Uptake Mechanism. Langmuir 2019, 35, 3513–3523. [Google Scholar] [CrossRef]
- Liu, Y.; Wan, H.-H.; Tian, D.-M.; Xu, X.-J.; Bi, C.-L.; Zhan, X.-Y.; Huang, B.-H.; Xu, Y.-S.; Yan, L.-P. Development and characterization of high efficacy cell-penetrating peptide via modulation of the histidine and arginine ratio for gene therapy. Materials 2021, 14, 4674. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Ma, Y.; Zhang, W.; Li, L.; Zhang, Y.; Zhang, L.; Liu, H.; Ni, J.; Wang, R. Design of new acid-activated cellpenetrating peptides for tumor drug delivery. PeerJ 2017, 5, e3429. [Google Scholar] [CrossRef] [PubMed]
- Ruseska, I.; Zimmer, A. Internalization mechanisms of cell-penetrating peptides. Beilstein J. Nanotechnol. 2020, 11, 101–123. [Google Scholar] [CrossRef]
- Gronewold, A.; Horn, M.; Neundorf, I. Design and biological characterization of novel cell-penetrating peptides preferentially targeting cell nuclei and subnuclear regions. Beilstein J. Org. Chem. 2018, 14, 1378–1388. [Google Scholar] [CrossRef] [PubMed]
- Regberg, J.; Vasconcelos, L.; Madani, F.; Langel, Ü.; Hällbrink, M. pH-responsive PepFect cell-penetrating peptides. Int. J. Pharm. 2016, 501, 32–38. [Google Scholar] [CrossRef] [PubMed]
- McErlean, E.M.; McCrudden, C.M.; McBride, J.W.; Cole, G.; Kett, V.L.; Robson, T.; Dunne, N.J.; McCarthy, H.O. Rational design and characterisation of an amphipathic cell penetrating peptide for non-viral gene delivery. Int. J. Pharm. 2021, 596, 120223. [Google Scholar] [CrossRef] [PubMed]
- McErlean, E.M.; Ziminska, M.; McCrudden, C.M.; McBride, J.W.; Loughran, S.P.; Cole, G.; Mulholland, E.J.; Kett, V.; Buckley, N.E.; Robson, T.; et al. Rational design and characterisation of a linear cell penetrating peptide for non-viral gene delivery. J. Control. Release 2021, 330, 1288–1299. [Google Scholar] [CrossRef]
- Sun, C.; Shen, W.C.; Tu, J.; Zaro, J.L. Interaction between cell-penetrating peptides and acid-sensitive anionic oligopeptides as a model for the design of targeted drug carriers. Mol. Pharm. 2014, 11, 1583–1590. [Google Scholar] [CrossRef]
- Futaki, S.; Suzuki, T.; Ohashi, W.; Yagami, T.; Tanaka, S.; Ueda, K.; Sugiura, Y. Arginine-rich peptides. An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery. J. Biol. Chem. 2001, 276, 5836–5840. [Google Scholar] [CrossRef] [PubMed]
- Klimpel, A.; Lützenburg, T.; Neundorf, I. Recent advances of anti-cancer therapies including the use of cell-penetrating peptides. Curr. Opin. Pharmacol. 2019, 47, 8–13. [Google Scholar] [CrossRef]
- Oppen, V.; Pille, J.; Stuut, C.; van Stevendaal, M.; van der Vorm, L.N.; Smeitink, J.A.M.; Koopman, W.J.H.; Willems, P.H.G.M.; van Hest, J.C.M.; Brock, R. Octa-arginine boosts the penetration of elastin-like polypeptide nanoparticles in 3D cancer models Citation for published version (APA): Octa-arginine boosts the penetration of elastin-like polypeptide nanoparticles in 3D cancer models. Eur. J. Pharm. Biopharm. 2019, 137, 175–184. [Google Scholar] [CrossRef]
- Goodman, T.T.; Chen, J.; Matveev, K.; Pun, S.H. Spatio-temporal modeling of nanoparticle delivery to multicellular tumor spheroids. Biotechnol. Bioeng. 2008, 101, 388–399. [Google Scholar] [CrossRef]
- Ware, M.J.; Colbert, K.; Keshishian, V.; Ho, J.; Corr, S.J.; Curley, S.A.; Godin, B. Generation of homogenous three-dimensional pancreatic cancer cell spheroids using an improved hanging drop technique. Tissue Eng.-Part C Methods 2016, 22, 312–321. [Google Scholar] [CrossRef]
- van den Brand, D.; Veelken, C.; Massuger, L.; Brock, R. Penetration in 3D tumor spheroids and explants: Adding a further dimension to the structure-activity relationship of cell-penetrating peptides. Biochim. Biophys. Acta-Biomembr. 2018, 1860, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hui, R.; Chen, Y.; Wang, W.; Chen, Y.; Gong, X.; Jin, J. Discovery of novel doxorubicin metabolites in MCF7 doxorubicin-resistant cells. Front. Pharmacol. 2019, 10, 1434. [Google Scholar] [CrossRef] [PubMed]
- Tacar, O.; Sriamornsak, P.; Dass, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2012, 65, 157–170. [Google Scholar] [CrossRef]
| Name | Sequence | MWcalc. [Da] | MWexp. [Da] | µH |
|---|---|---|---|---|
| sC18ΔE | GLRKRLRKFRNKIKK | 1940.44 | 1941.04 | 0.656 |
| sC18ΔE1 | GLRHRLRHFRNHIHH | 1985.27 | 1986.02 | 0.465 |
| sC18ΔE2 | GLHKHLHKFHNKIKK | 1864.25 | 1864.92 | 0.667 |
| sC18ΔE3 | HLRHHLRHFRHHIKH | 2060.38 | 2061.17 | 0.524 |
| sC18ΔE4 | GLHKRLHKFHNKIHK | 1892.26 | 1893.00 | 0.655 |
| sC18* | GLRKRLRKFRNK | 1570.94 | 1571.33 | 0.572 |
| sC18*R,L | RLRKLLRKFLRK | 1626.10 | 1626.46 | 0.790 |
| sC18 | GLRKRLRKFRNKIKEK | 2069.55 | 2069.87 | 0.555 |
| Name | Sequence | MWcalc. [Da] | MWexp. [Da] |
|---|---|---|---|
| PDC-1 | Dox-GLRKRLRKFRNKIKEK | 2868.43 | 2868.03 |
| PDC-2 | Dox-GLRKRLRKFRNKIKK | 2738.31 | 2738.90 |
| PDC-3 | Dox-GLRKRLRKFRNK | 2368.81 | 2368.95 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grabeck, J.; Lützenburg, T.; Frommelt, P.; Neundorf, I. Comparing Variants of the Cell-Penetrating Peptide sC18 to Design Peptide-Drug Conjugates. Molecules 2022, 27, 6656. https://doi.org/10.3390/molecules27196656
Grabeck J, Lützenburg T, Frommelt P, Neundorf I. Comparing Variants of the Cell-Penetrating Peptide sC18 to Design Peptide-Drug Conjugates. Molecules. 2022; 27(19):6656. https://doi.org/10.3390/molecules27196656
Chicago/Turabian StyleGrabeck, Joshua, Tamara Lützenburg, Pia Frommelt, and Ines Neundorf. 2022. "Comparing Variants of the Cell-Penetrating Peptide sC18 to Design Peptide-Drug Conjugates" Molecules 27, no. 19: 6656. https://doi.org/10.3390/molecules27196656
APA StyleGrabeck, J., Lützenburg, T., Frommelt, P., & Neundorf, I. (2022). Comparing Variants of the Cell-Penetrating Peptide sC18 to Design Peptide-Drug Conjugates. Molecules, 27(19), 6656. https://doi.org/10.3390/molecules27196656









