Electrochemical Investigation of Iron-Catalyzed Atom Transfer Radical Polymerization
Abstract
1. Introduction
2. Results and Discussion
2.1. Cyclic Voltammetry of FeCl3
2.2. Cyclic Voltammetry of Chloro(2-pyridylamino-N,N-bis(2-methylene-4,6-dichlorophenolate))iron(III), FeIIIL(Cl)
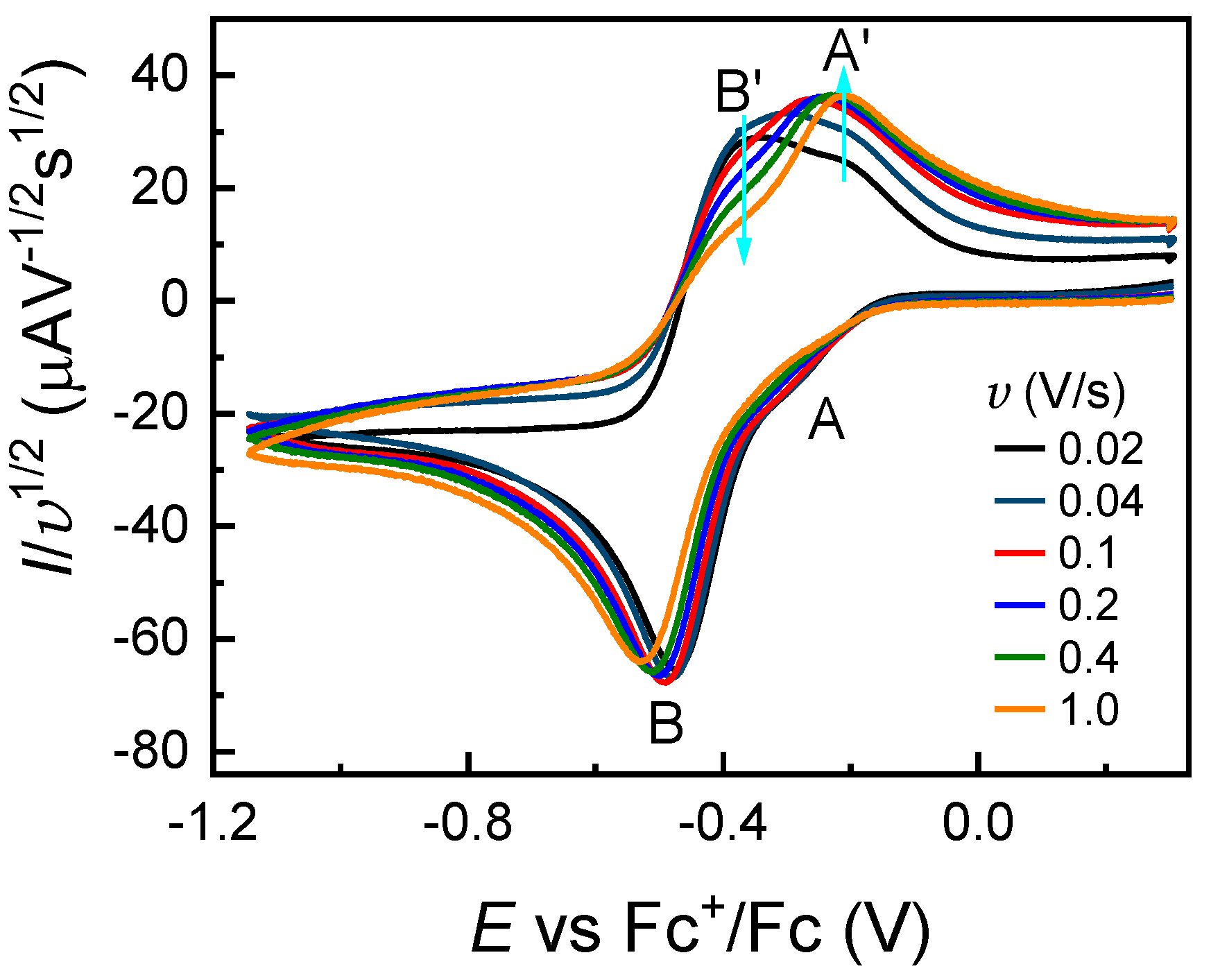
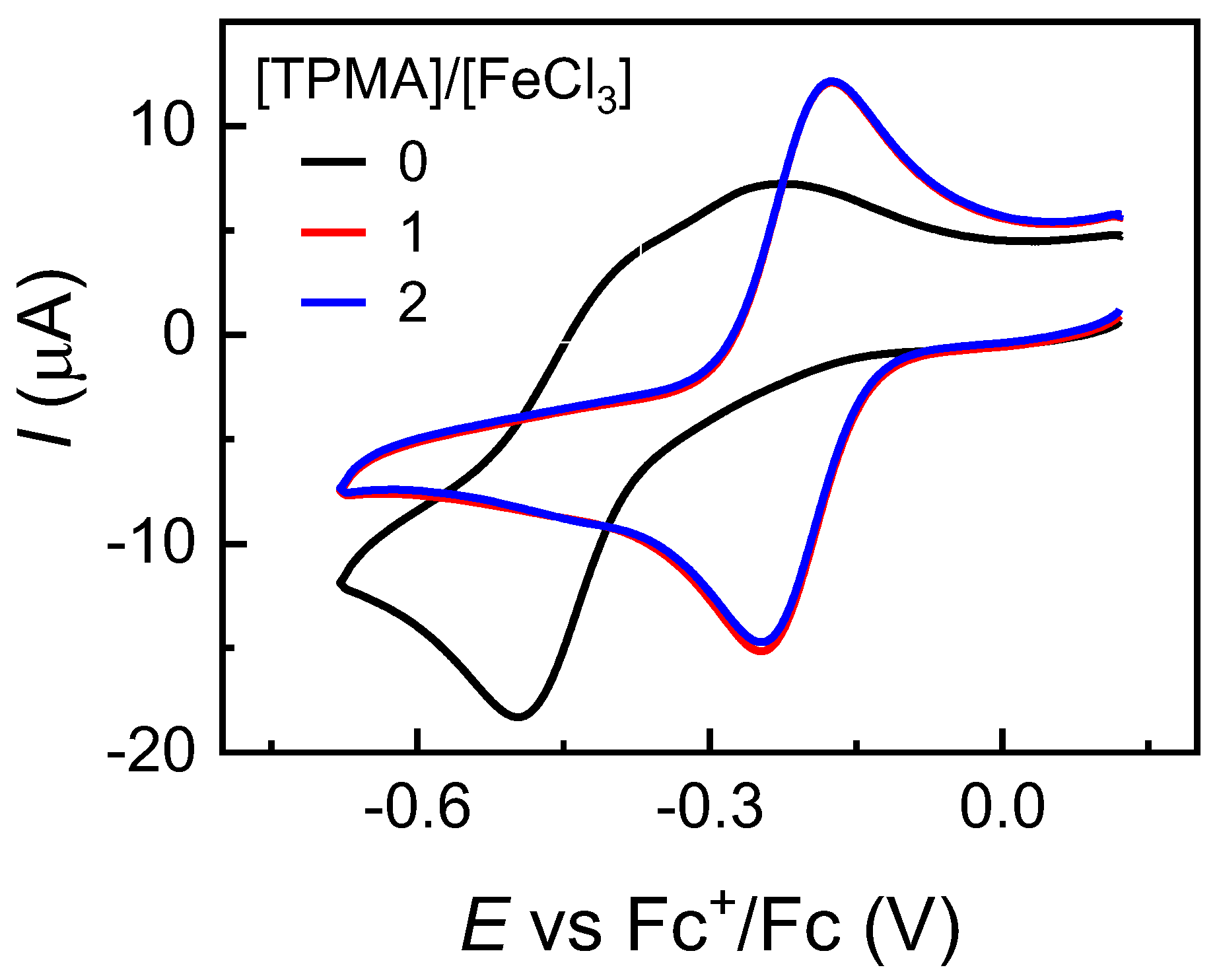
2.3. Cyclic Voltammetry of FeCl3 in the Presence of Other Ligands
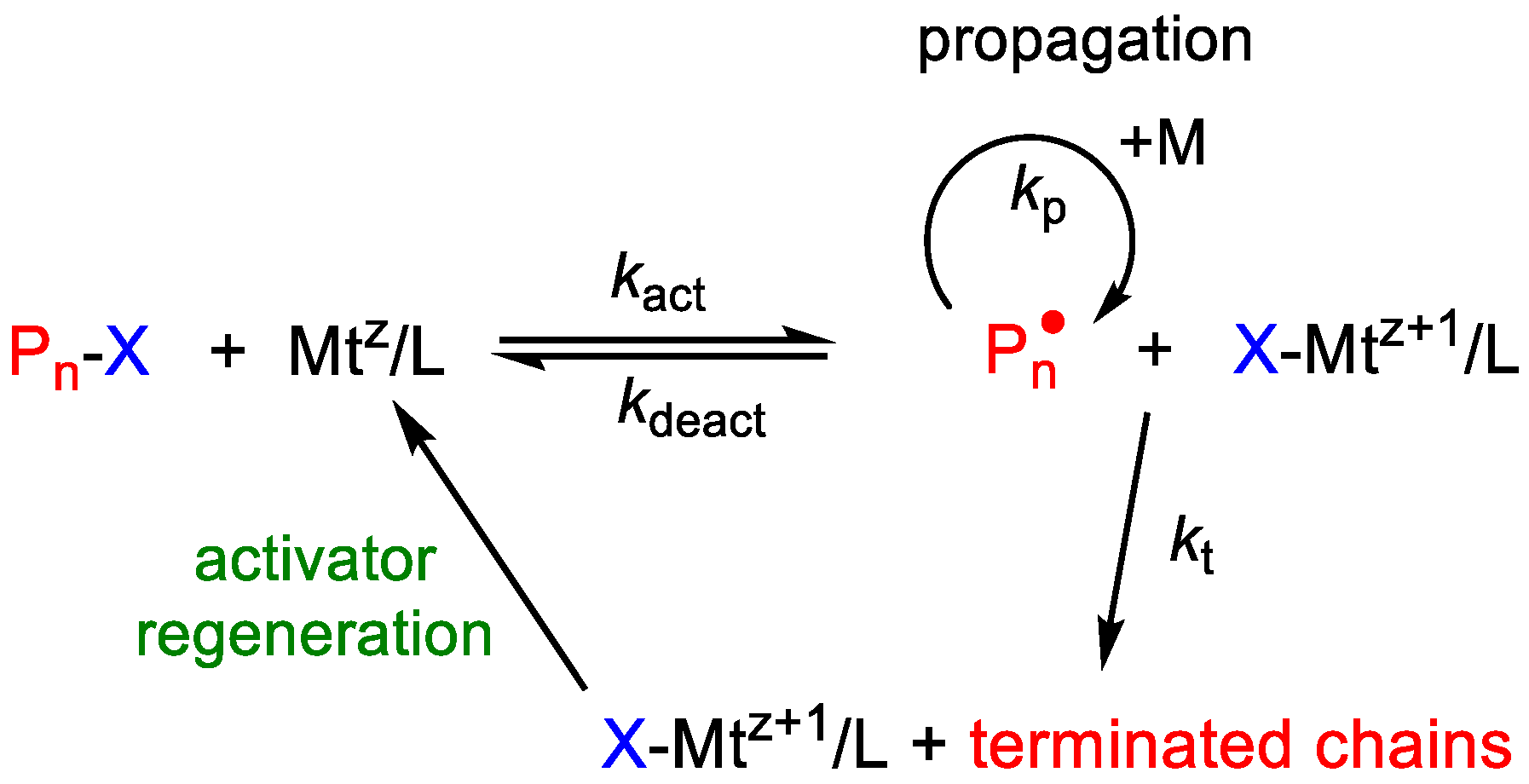
2.4. Electrochemically Mediated ATRP (eATRP)
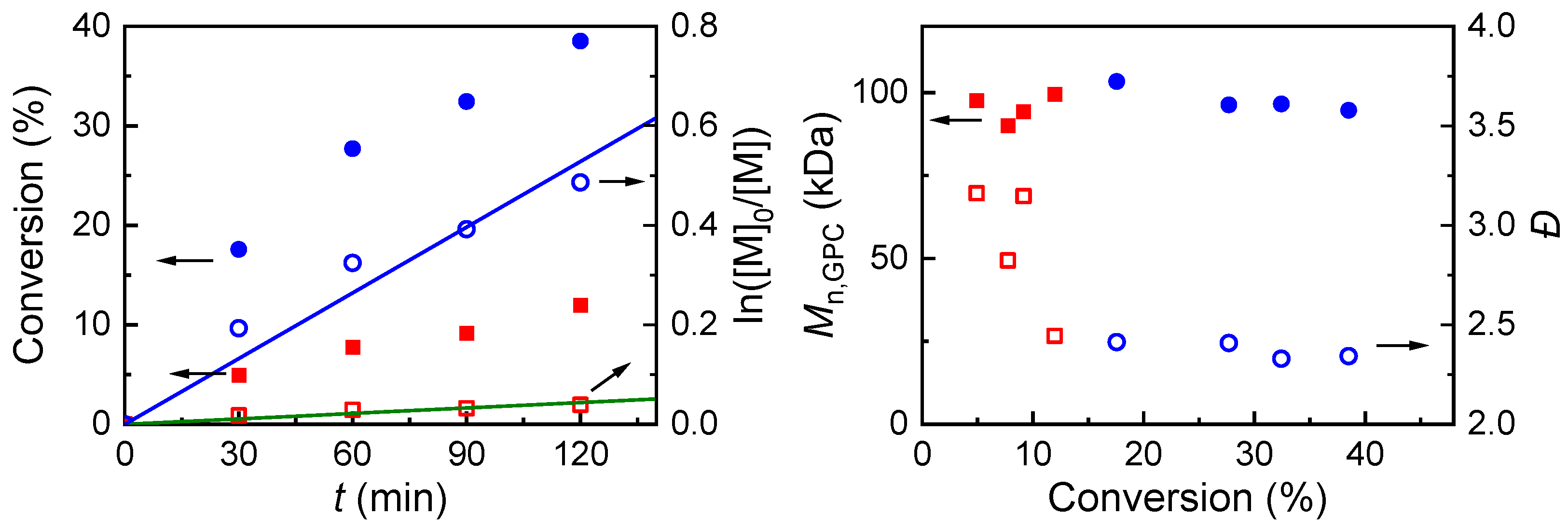
2.4.1. eATRP Mediated by Amine–bis(phenolate) iron(III) Chloride, FeIIIL(Cl)
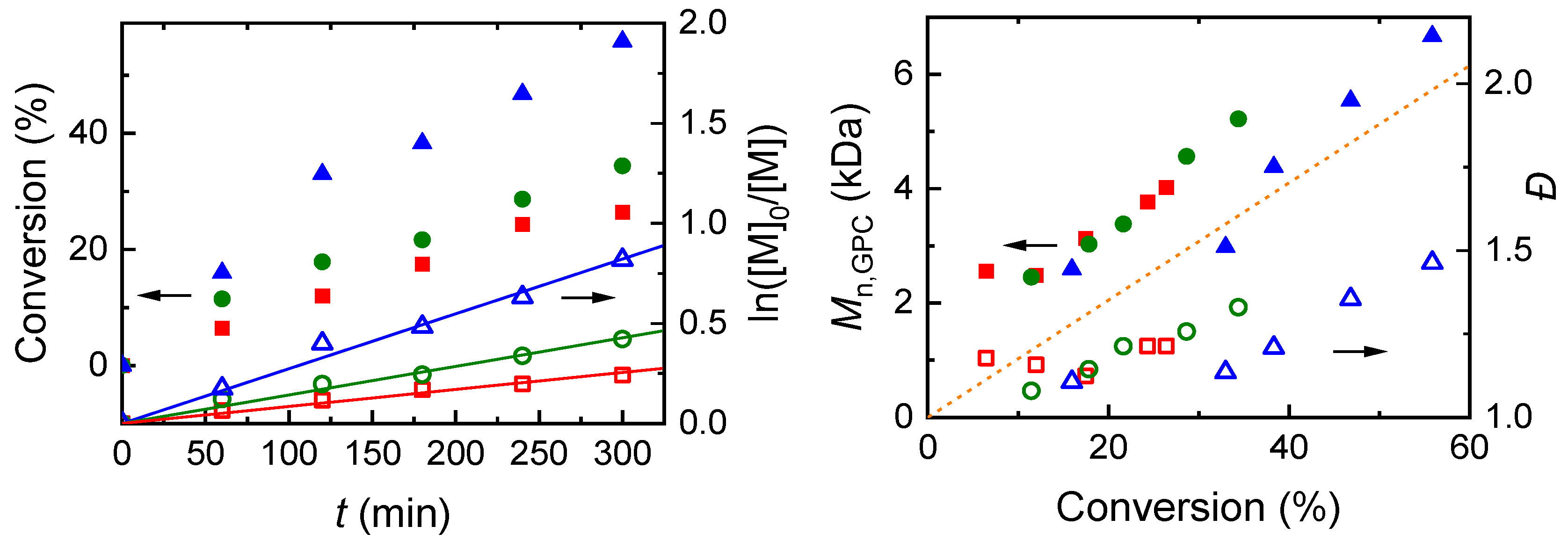
2.4.2. eATRP Mediated by FeCl3
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
3.3. eATRP of Methyl Methacrylate
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Corrigan, N.; Jung, K.; Moad, G.; Hawker, C.J.; Matyjaszewski, K.; Boyer, C. Reversible-Deactivation Radical Polymerization (Controlled/Living Radical Polymerization): From Discovery to Materials Design and Applications. Progress Polym. Sci. 2020, 111, 101311. [Google Scholar] [CrossRef]
- Matyjaszewski, K. Atom Transfer Radical Polymerization (ATRP): Current Status and Future Perspectives. Macromolecules 2012, 45, 4015–4039. [Google Scholar] [CrossRef]
- Ribelli, T.G.; Lorandi, F.; Fantin, M.; Matyjaszewski, K. Atom Transfer Radical Polymerization: Billion Times More Active Catalysts and New Initiation Systems. Macromol. Rapid Commun. 2019, 40, 1800616. [Google Scholar] [CrossRef]
- Lorandi, F.; Fantin, M.; Matyjaszewski, K. Atom Transfer Radical Polymerization: A Mechanistic Perspective. J. Am. Chem. Soc. 2022, 144, 15413–15430. [Google Scholar] [CrossRef]
- Moad, G.; Rizzardo, E.; Thang, S.H. Living Radical Polymerization by the RAFT Process. Aust. J. Chem. 2005, 58, 379. [Google Scholar] [CrossRef]
- Perrier, S. 50th Anniversary Perspective: RAFT Polymerization—A User Guide. Macromolecules 2017, 50, 7433–7447. [Google Scholar] [CrossRef]
- Nicolas, J.; Guillaneuf, Y.; Lefay, C.; Bertin, D.; Gigmes, D.; Charleux, B. Nitroxide-Mediated Polymerization. Progress Polym. Sci. 2013, 38, 63–235. [Google Scholar] [CrossRef]
- Audran, G.; Bagryanskaya, E.G.; Marque, S.R.A.; Postnikov, P. New Variants of Nitroxide Mediated Polymerization. Polymers 2020, 12, 1481. [Google Scholar] [CrossRef]
- Ching, Y.L.; Coote, M.L.; Gennaro, A.; Matyjaszewski, K. Ab Initio Evaluation of the Thermodynamic and Electrochemical Properties of Alkyl Halides and Radicals and Their Mechanistic Implications for Atom Transfer Radical Polymerization. J. Am. Chem. Soc. 2008, 130, 12762–12774. [Google Scholar] [CrossRef]
- Isse, A.A.; Gennaro, A.; Lin, C.Y.; Hodgson, J.L.; Coote, M.L.; Guliashvili, T. Mechanism of Carbon-Halogen Bond Reductive Cleavage in Activated Alkyl Halide Initiators Relevant to Living Radical Polymerization: Theoretical and Experimental Study. J. Am. Chem. Soc. 2011, 133, 6254–6264. [Google Scholar] [CrossRef]
- Isse, A.A.; Bortolamei, N.; de Paoli, P.; Gennaro, A. On the Mechanism of Activation of Copper-Catalyzed Atom Transfer Radical Polymerization. Electrochim. Acta 2013, 110, 655–662. [Google Scholar] [CrossRef]
- Pan, X.; Fantin, M.; Yuan, F.; Matyjaszewski, K. Externally Controlled Atom Transfer Radical Polymerization. Chem. Soc. Rev. 2018, 47, 5457–5490. [Google Scholar] [CrossRef]
- Min, K.; Gao, H.; Matyjaszewski, K. Use of Ascorbic Acid as Reducing Agent for Synthesis of Well-Defined Polymers by ARGET ATRP. Macromolecules 2007, 40, 1789–1791. [Google Scholar] [CrossRef]
- Simakova, A.; Averick, S.E.; Konkolewicz, D.; Matyjaszewski, K. Aqueous ARGET ATRP. Macromolecules 2012, 45, 6371–6379. [Google Scholar] [CrossRef]
- Mendonça, P.V.; Ribeiro, J.P.M.; Abreu, C.M.R.; Guliashvili, T.; Serra, A.C.; Coelho, J.F.J. Thiourea Dioxide As a Green and Affordable Reducing Agent for the ARGET ATRP of Acrylates, Methacrylates, Styrene, Acrylonitrile, and Vinyl Chloride. ACS Macro Lett. 2019, 8, 315–319. [Google Scholar] [CrossRef]
- Percec, V.; Guliashvili, T.; Ladislaw, J.S.; Wistrand, A.; Stjerndahl, A.; Sienkowska, M.J.; Monteiro, M.J.; Sahoo, S. Ultrafast Synthesis of Ultrahigh Molar Mass Polymers by Metal-Catalyzed Living Radical Polymerization of Acrylates, Methacrylates, and Vinyl Chloride Mediated by SET at 25 °C. J. Am. Chem. Soc. 2006, 128, 14156–14165. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Wang, Y.; Matyjaszewski, K. ATRP of Methyl Acrylate with Metallic Zinc, Magnesium, and Iron as Reducing Agents and Supplemental Activators. Macromolecules 2011, 44, 683–685. [Google Scholar] [CrossRef]
- Lorandi, F.; Fantin, M.; Isse, A.A.; Gennaro, A. RDRP in the Presence of Cu0: The Fate of Cu(I) Proves the Inconsistency of SET-LRP Mechanism. Polymer 2015, 72, 238–245. [Google Scholar] [CrossRef]
- Chmielarz, P.; Fantin, M.; Park, S.; Isse, A.A.; Gennaro, A.; Magenau, A.J.D.; Sobkowiak, A.; Matyjaszewski, K. Electrochemically Mediated Atom Transfer Radical Polymerization (EATRP). Progress Polym. Sci. 2017, 69, 47–78. [Google Scholar] [CrossRef]
- Lorandi, F.; Fantin, M.; Isse, A.A.; Gennaro, A. Electrochemical Triggering and Control of Atom Transfer Radical Polymerization. Curr. Opin. Electrochem. 2018, 8, 1–7. [Google Scholar] [CrossRef]
- Tasdelen, M.A.; Uygun, M.; Yagci, Y. Photoinduced Controlled Radical Polymerization. Macromol. Rapid Commun. 2011, 32, 58–62. [Google Scholar] [CrossRef]
- Konkolewicz, D.; Schröder, K.; Buback, J.; Bernhard, S.; Matyjaszewski, K. Visible Light and Sunlight Photoinduced ATRP with Ppm of Cu Catalyst. ACS Macro Lett. 2012, 1, 1219–1223. [Google Scholar] [CrossRef]
- Anastasaki, A.; Nikolaou, V.; Zhang, Q.; Burns, J.; Samanta, S.R.; Waldron, C.; Haddleton, A.J.; McHale, R.; Fox, D.; Percec, V.; et al. Copper(II)/Tertiary Amine Synergy in Photoinduced Living Radical Polymerization: Accelerated Synthesis of ω-Functional and α,ω-Heterofunctional Poly(Acrylates). J. Am. Chem. Soc. 2014, 136, 1141–1149. [Google Scholar] [CrossRef]
- Mohapatra, H.; Kleiman, M.; Esser-Kahn, A.P. Mechanically Controlled Radical Polymerization Initiated by Ultrasound. Nat. Chem. 2017, 9, 135–139. [Google Scholar] [CrossRef]
- Zaborniak, I.; Chmielarz, P. Ultrasound-Mediated Atom Transfer Radical Polymerization (ATRP). Materials 2019, 12, 3600. [Google Scholar] [CrossRef]
- Soly, S.; Mistry, B.; Murthy, C.N. Photo-Mediated Metal-Free Atom Transfer Radical Polymerization: Recent Advances in Organocatalysts and Perfection towards Polymer Synthesis. Polym. Int. 2022, 71, 159–168. [Google Scholar] [CrossRef]
- Pan, X.; Fang, C.; Fantin, M.; Malhotra, N.; So, W.Y.; Peteanu, L.A.; Isse, A.A.; Gennaro, A.; Liu, P.; Matyjaszewski, K. Mechanism of Photoinduced Metal-Free Atom Transfer Radical Polymerization: Experimental and Computational Studies. J. Am. Chem. Soc. 2016, 138, 2411–2425. [Google Scholar] [CrossRef]
- Di Lena, F.; Matyjaszewski, K. Transition Metal Catalysts for Controlled Radical Polymerization. Progress Polym. Sci. 2010, 35, 959–1021. [Google Scholar] [CrossRef]
- Jain, T.K.; Reddy, M.K.; Morales, M.A.; Leslie-Pelecky, D.L.; Labhasetwar, V. Biodistribution, Clearance, and Biocompatibility of Iron Oxide Magnetic Nanoparticles in Rats. Mol. Pharm. 2008, 5, 316–327. [Google Scholar] [CrossRef]
- Oriňaková, R.; Oriňak, A.; Giretová, M.; Medvecký, L.; Kupková, M.; Hrubovčïáková, M.; Maskal’Ová, I.; MacKo, J.; Kal’Avský, F. A Study of Cytocompatibility and Degradation of Iron-Based Biodegradable Materials. J. Biomater. Appl. 2016, 30, 1060–1070. [Google Scholar] [CrossRef]
- Dadashi-Silab, S.; Matyjaszewski, K. Iron Catalysts in Atom Transfer Radical Polymerization. Molecules 2020, 25, 1648. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; He, D.; Xie, X. Iron-Catalyzed Atom Transfer Radical Polymerization. Polym. Chem. 2015, 6, 1660–1687. [Google Scholar] [CrossRef]
- Uchiike, C.; Terashima, T.; Ouchi, M.; Ando, T.; Kamigaito, M.; Sawamoto, M. Evolution of Iron Catalysts for Effective Living Radical Polymerization: Design of Phosphine/Halogen Ligands in FeX2(PR3)2. Macromolecules 2007, 40, 8658–8662. [Google Scholar] [CrossRef]
- Nishizawa, K.; Ouchi, M.; Sawamoto, M. Phosphine–Ligand Decoration toward Active and Robust Iron Catalysts in LRP. Macromolecules 2013, 46, 3342–3349. [Google Scholar] [CrossRef]
- Wang, J.; Han, J.; Xie, X.; Xue, Z.; Fliedel, C.; Poli, R. FeBr2-Catalyzed Bulk ATRP Promoted by Simple Inorganic Salts. Macromolecules 2019, 52, 5366–5376. [Google Scholar] [CrossRef]
- Mukumoto, K.; Wang, Y.; Matyjaszewski, K. Iron-Based ICAR ATRP of Styrene with Ppm Amounts of FeIIIBr 3 and 1,1′-Azobis(Cyclohexanecarbonitrile). ACS Macro Lett. 2012, 1, 599–602. [Google Scholar] [CrossRef]
- Allan, L.E.N.; MacDonald, J.P.; Reckling, A.M.; Kozak, C.M.; Shaver, M.P. Controlled Radical Polymerization Mediated by Amine-Bis(Phenolate) Iron(III) Complexes. Macromol. Rapid Commun. 2012, 33, 414–418. [Google Scholar] [CrossRef]
- Schroeder, H.; Buback, M.; Matyjaszewski, K. Pressure Dependence of Iron-Mediated Methyl Methacrylate ATRP in Different Solvent Environments. Macromol. Chem. Phys. 2014, 215, 44–53. [Google Scholar] [CrossRef]
- Dadashi-Silab, S.; Kim, K.; Lorandi, F.; Schild, D.J.; Fantin, M.; Matyjaszewski, K. Effect of Halogen and Solvent on Iron-Catalyzed Atom Transfer Radical Polymerization. Polym. Chem. 2022, 13, 1059–1066. [Google Scholar] [CrossRef]
- Simakova, A.; Mackenzie, M.; Averick, S.E.; Park, S.; Matyjaszewski, K. Bioinspired Iron-Based Catalyst for Atom Transfer Radical Polymerization. Angew. Chem. Int. Ed. 2013, 52, 12148–12151. [Google Scholar] [CrossRef]
- Yang, D.; He, D.; Liao, Y.; Xue, Z.; Zhou, X.; Xie, X. Iron-Mediated AGET ATRP of Methyl Methacrylate in the Presence of Polar Solvents as Ligands. J. Polym. Sci. A Polym. Chem. 2014, 52, 1020–1027. [Google Scholar] [CrossRef]
- Dadashi-Silab, S.; Pan, X.; Matyjaszewski, K. Photoinduced Iron-Catalyzed Atom Transfer Radical Polymerization with Ppm Levels of Iron Catalyst under Blue Light Irradiation. Macromolecules 2017, 50, 7967–7977. [Google Scholar] [CrossRef]
- Rolland, M.; Truong, N.P.; Whitfield, R.; Anastasaki, A. Tailoring Polymer Dispersity in Photoinduced Iron-Catalyzed ATRP. ACS Macro Lett. 2020, 9, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.K.; Zhou, Y.N.; Luo, Z.H. Kinetic Insights into the Iron-Based Electrochemically Mediated Atom Transfer Radical Polymerization of Methyl Methacrylate. Macromolecules 2016, 49, 4038–4046. [Google Scholar] [CrossRef]
- Guo, J.K.; Zhou, Y.N.; Luo, Z.H. Iron-Based Electrochemically Mediated Atom Transfer Radical Polymerization with Tunable Catalytic Activity. AIChE J. 2018, 64, 961–969. [Google Scholar] [CrossRef]
- Wang, J.; Tian, M.; Li, S.; Wang, R.; Du, F.; Xue, Z. Ligand-Free Iron-Based Electrochemically Mediated Atom Transfer Radical Polymerization of Methyl Methacrylate. Polym. Chem. 2018, 9, 4386–4394. [Google Scholar] [CrossRef]
- Divandari, M.; Pollard, J.; Dehghani, E.; Bruns, N.; Benetti, E.M. Controlling Enzymatic Polymerization from Surfaces with Switchable Bioaffinity. Biomacromolecules 2017, 18, 4261–4270. [Google Scholar] [CrossRef]
- Sigg, S.J.; Seidi, F.; Renggli, K.; Silva, T.B.; Kali, G.; Bruns, N. Horseradish Peroxidase as a Catalyst for Atom Transfer Radical Polymerization. Macromol. Rapid Commun. 2011, 32, 1710–1715. [Google Scholar] [CrossRef]
- Silva, T.B.; Spulber, M.; Kocik, M.K.; Seidi, F.; Charan, H.; Rother, M.; Sigg, S.J.; Renggli, K.; Kali, G.; Bruns, N. Hemoglobin and Red Blood Cells Catalyze Atom Transfer Radical Polymerization. Biomacromolecules 2013, 14, 2703–2712. [Google Scholar] [CrossRef]
- Rifaie-Graham, O.; Pollard, J.; Raccio, S.; Balog, S.; Rusch, S.; Hernández-Castañeda, M.A.; Mantel, P.-Y.; Beck, H.-P.; Bruns, N. Hemozoin-Catalyzed Precipitation Polymerization as an Assay for Malaria Diagnosis. Nat. Commun. 2019, 10, 1369. [Google Scholar] [CrossRef]
- Pollard, J.; Rifaie-Graham, O.; Raccio, S.; Davey, A.; Balog, S.; Bruns, N. Biocatalytically Initiated Precipitation Atom Transfer Radical Polymerization (ATRP) as a Quantitative Method for Hemoglobin Detection in Biological Fluids. Anal. Chem. 2020, 92, 1162–1170. [Google Scholar] [CrossRef] [PubMed]
- Raccio, S.; Pollard, J.; Djuhadi, A.; Balog, S.; Pellizzoni, M.M.; Rodriguez, K.J.; Rifaie-Graham, O.; Bruns, N. Rapid Quantification of the Malaria Biomarker Hemozoin by Improved Biocatalytically Initiated Precipitation Atom Transfer Radical Polymerizations. Analyst 2020, 145, 7741–7751. [Google Scholar] [CrossRef] [PubMed]
- De Bon, F.; Fantin, M.; Isse, A.A.; Gennaro, A. Electrochemically Mediated ATRP in Ionic Liquids: Controlled Polymerization of Methyl Acrylate in [BMIm][OTf]. Polym. Chem. 2018, 9, 646–655. [Google Scholar] [CrossRef]
- Fantin, M.; Isse, A.A.; Venzo, A.; Gennaro, A.; Matyjaszewski, K. Atom Transfer Radical Polymerization of Methacrylic Acid: A Won Challenge. J. Am. Chem. Soc. 2016, 138, 7216–7219. [Google Scholar] [CrossRef] [PubMed]
- Bonometti, V.; Labbé, E.; Buriez, O.; Mussini, P.; Amatore, C. Exploring the First Steps of an Electrochemically-Triggered Controlled Polymerization Sequence: Activation of Alkyl- and Benzyl Halide Initiators by an Electrogenerated FeIISalen Complex. J. Electroanal. Chem. 2009, 633, 99–105. [Google Scholar] [CrossRef]
- Dass, N.N.; George, M.H. IRON(II1)-chloro complexes in n,n-dimethylformamidel styrene systems. Polym. Lett. 1967, 5, 1119–1124. [Google Scholar] [CrossRef]
- Kim, Y.J.; Park, C.R. Analysis of Problematic Complexing Behavior of Ferric Chloride with N,N-Dimethylformamide Using Combined Techniques of FT-IR, XPS, and TGA/DTG. Inorg. Chem. 2002, 41, 6211–6216. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods: Fundamentals and Applications, 2nd ed.; John Wiley & Sons: New York, NY, USA, 2011; ISBN 0-471-04372-9. [Google Scholar]
- Nicholson, R.S. Theory and Application of Cyclic Voltammetry for Measurement of Electrode Reaction Kinetics. Anal. Chem. 1965, 37, 1351–1355. [Google Scholar] [CrossRef]
- Reckling, A.M.; Martin, D.; Dawe, L.N.; Decken, A.; Kozak, C.M. Structure and C–C Cross-Coupling Reactivity of Iron(III) Complexes of Halogenated Amine-Bis(Phenolate) Ligands. J. Organom. Chem. 2011, 696, 787–794. [Google Scholar] [CrossRef]
- David Walker, J.; Poli, R. FeC13-Phosphine Adducts with Trigonal-Bipyramidal Geometry. Influence of the Phosphine on the Spin State; HAL Open Science: Lyon, France, 1989; Volume 28. [Google Scholar]
- Wang, Y.; Kwak, Y.; Matyjaszewski, K. Enhanced Activity of ATRP Fe Catalysts with Phosphines Containing Electron Donating Groups. Macromolecules 2012, 45, 5911–5915. [Google Scholar] [CrossRef]
- Schroeder, H.; Matyjaszewski, K.; Buback, M. Kinetics of Fe-Mediated ATRP with Triarylphosphines. Macromolecules 2015, 48, 4431–4437. [Google Scholar] [CrossRef]
- Kojima, T.; Leising, R.A.; Yan, S.; Que, L. Alkane Functionalization at Nonheme Iron Centers. Stoichiometric Transfer of Metal-Bound Ligands to Alkane. J. Am. Chem. Soc. 1993, 115, 11328–11335. [Google Scholar] [CrossRef]
- Mandon, D.; Machkour, A.; Goetz, S.; Welter, R. Trigonal Bipyramidal Geometry and Tridentate Coordination Mode of the Tripod in FeCl2 Complexes with Tris(2-Pyridylmethyl)Amine Derivatives Bis-α-Substituted with Bulky Groups. Structures and Spectroscopic Comparative Studies. Inorg. Chem. 2002, 41, 5364–5372. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Sastry, N.V. Densities, Excess Molar Volumes at T = (298.15 to 313.15) K, Speeds of Sound, Excess Isentropic Compressibilities, Relative Permittivities, and Deviations in Molar Polarizations at T = (298.15 and 308.15) K for Methyl Methacrylate + 2-Butoxyethanol or Dibutyl Ether + Benzene, Toluene, or p -Xylene. J. Chem. Eng. Data 2004, 49, 1116–1126. [Google Scholar] [CrossRef]
- Wohlfarth, C. Permittivity (Dielectric Constant) of Liquids. In CRC Handbook of Chemistry and Phyiscs; Lide, D.R., Ed.; Taylor and Francis: Boca Raton, FL, USA, 2007. [Google Scholar]
- Allan, L.E.N.; Macdonald, J.P.; Nichol, G.S.; Shaver, M.P. Single Component Iron Catalysts for Atom Transfer and Organometallic Mediated Radical Polymerizations: Mechanistic Studies and Reaction Scope. Macromolecules 2014, 47, 1249–1257. [Google Scholar] [CrossRef]
- Schroeder, H.; Buback, M. SP-PLP-EPR Measurement of Iron-Mediated ATRP Deactivation Rate. Macromolecules 2015, 48, 6108–6113. [Google Scholar] [CrossRef]
- Falciola, L.; Gennaro, A.; Isse, A.A.; Mussini, P.R.; Rossi, M. The Solvent Effect in the Electrocatalytic Reduction of Organic Bromides on Silver. J. Electroanal. Chem. 2006, 593, 47–56. [Google Scholar] [CrossRef]
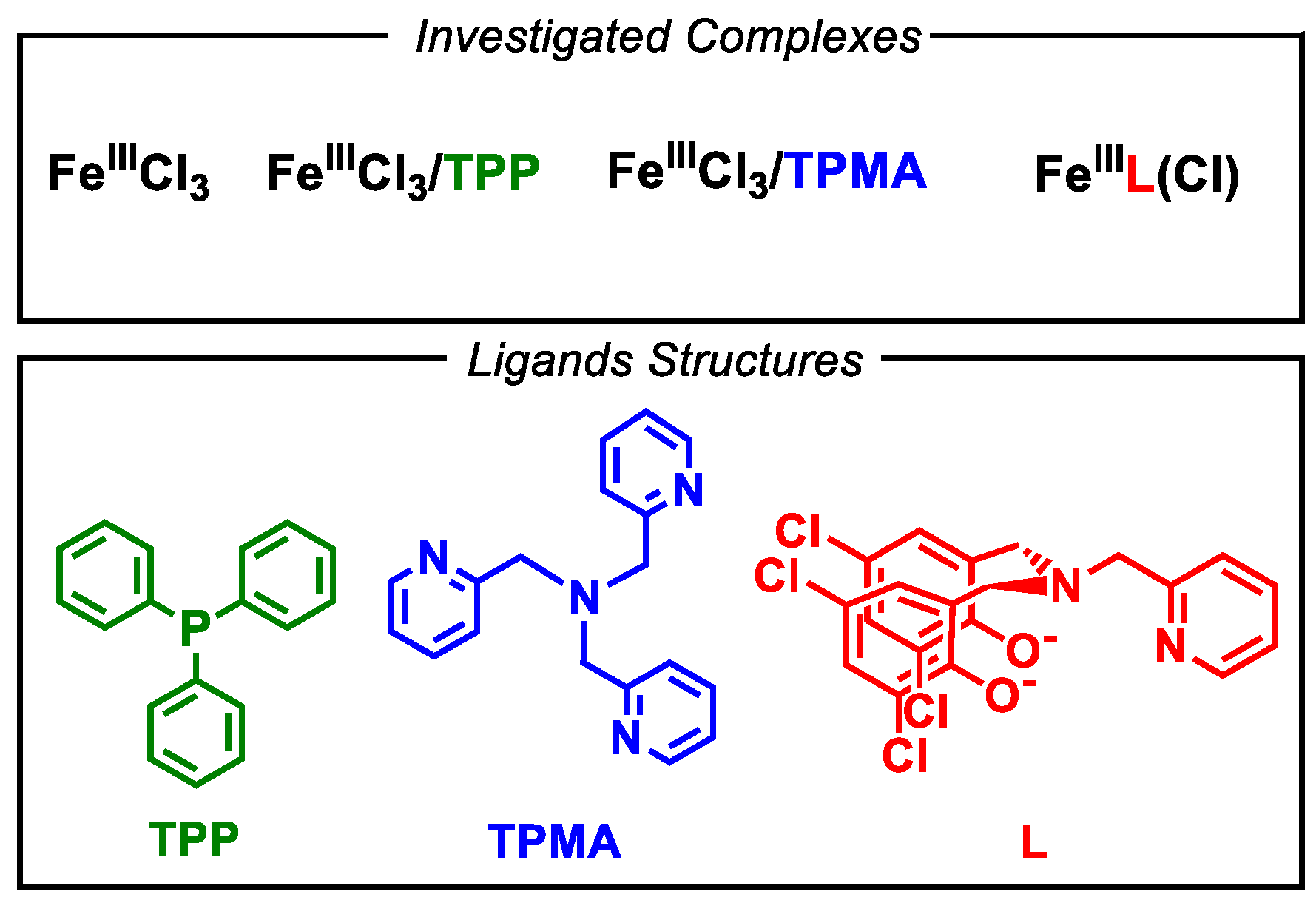

| Ligand | Complex | E° vs. Fc+/Fc (V) |
|---|---|---|
| - | FeCl4− | −0.483 |
| L 1 | FeL(Cl) | −0.853 |
| TPP | FeCl4− | −0.483 |
| TPMA | FeCl2(TPMA)+ | −0.211 |
| Entry | Initiator | Eapp − Epc (V) | T (°C) | t (h) | Q/Qth 2 | Conv. 3 (%) | 103 kapp 4 (min−1) | Mn,GPC5 (kDa) | Mn,th6 (kDa) | Đ5 |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | ECPA | 0 | 70 | 4 | 0.81 | 42.3 | 2.35 | 147.80 | 13.26 | 2.75 |
| 2 7 | ECPA | 0 | 70 | 3 | 0.75 | 55.5 | 3.27 | 157.06 | 17.34 | 2.18 |
| 3 | ECPA | 0 | 50 | 4 | 0.70 | 17.6 | 0.36 | 109.04 | 5.64 | 2.54 |
| 4 | EBPA | 0.06 | 50 | 2 | 0.93 | 38.5 | 4.40 | 94.67 | 12.38 | 2.34 |
| 5 | EBPA | −0.06 | 70 | 2 | 0.59 | 40.0 | 4.63 | 85.52 | 13.04 | 2.52 |
| Entry | [EBPA] (mM) | [FeCl3] (mM) | [M]:[I]:[Fe] 2 | Eapp − E1/2 (V) | T (°C) | Q/Qth 3 (C) | Conv. 4 (%) | 103 kapp 5 (min−1) | Mn,GPC6 (kDa) | Mn,th7 (kDa) | Đ6 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 47.0 | 23.5 | 100:1:0.5 | −0.12 | 70 | 1.08 | 55.9 | 27.4 | 6.67 | 5.84 | 1.46 |
| 2 | 47.0 | 23.5 | 100:1:0.5 | −0.06 | 70 | 0.85 | 34.4 | 14.3 | 5.22 | 3.69 | 1.33 |
| 3 | 47.0 | 23.5 | 100:1:0.5 | 0.0 | 70 | 0.82 | 26.5 | 8.5 | 4.02 | 2.90 | 1.21 |
| 4 | 47.0 | 23.5 | 100:1:0.5 | −0.06 | 55 | 0.93 | 38.3 | 16.8 | 6.37 | 4.33 | 1.36 |
| 5 | 47.0 | 11.75 | 100:1:0.25 | −0.06 | 55 | 1.31 | 43.4 | 19.7 | 6.47 | 4.59 | 1.40 |
| 6 | 23.5 | 11.75 | 100:0.5:0.25 | −0.06 | 55 | 1.23 | 32.6 | 13.5 | 10.24 | 6.77 | 1.55 |
| 7 | 23.5 | 5.88 | 100:0.5:0.126 | −0.06 | 55 | 1.96 | 38.5 | 17.8 | 13.34 | 7.82 | 1.50 |
| 8 | 23.5 | 5.88 | 100:0.5:0.126 | 0.0 | 55 | 1.44 | 23.7 | 9.92 | 9.13 | 5.0 | 1.53 |
| 9 | 23.5 | 5.88 | 100:0.5:0.126 | 0.0 | 70 | 1.66 | 48.3 | 22.7 | 11.66 | 9.92 | 1.53 |
| 10 8 | 23.5 | 5.88 | 100:0.5:0.126 | −0.06 | 55 | 1.66 | 40.6 | 18.2 | 14.75 | 8.22 | 1.50 |
| 11 9 | 23.5 | 5.88 | 100:0.5:0.126 | −0.06 | 55 | 0.50 | 0 | - | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gazzola, G.; Pasinato, S.; Fantin, M.; Braidi, N.; Tubaro, C.; Durante, C.; Isse, A.A. Electrochemical Investigation of Iron-Catalyzed Atom Transfer Radical Polymerization. Molecules 2022, 27, 6312. https://doi.org/10.3390/molecules27196312
Gazzola G, Pasinato S, Fantin M, Braidi N, Tubaro C, Durante C, Isse AA. Electrochemical Investigation of Iron-Catalyzed Atom Transfer Radical Polymerization. Molecules. 2022; 27(19):6312. https://doi.org/10.3390/molecules27196312
Chicago/Turabian StyleGazzola, Gianluca, Sebastiano Pasinato, Marco Fantin, Niccolò Braidi, Cristina Tubaro, Christian Durante, and Abdirisak Ahmed Isse. 2022. "Electrochemical Investigation of Iron-Catalyzed Atom Transfer Radical Polymerization" Molecules 27, no. 19: 6312. https://doi.org/10.3390/molecules27196312
APA StyleGazzola, G., Pasinato, S., Fantin, M., Braidi, N., Tubaro, C., Durante, C., & Isse, A. A. (2022). Electrochemical Investigation of Iron-Catalyzed Atom Transfer Radical Polymerization. Molecules, 27(19), 6312. https://doi.org/10.3390/molecules27196312