13C-NMR Chemical Shifts in 1,3-Benzazoles as a Tautomeric Ratio Criterion
Abstract
1. Introduction
2. Electronic Effects of Heteroatoms on Aromatic Rings
2.1. Influence of Benzene Ring Substituents in the 13C NMR Chemical Shifts
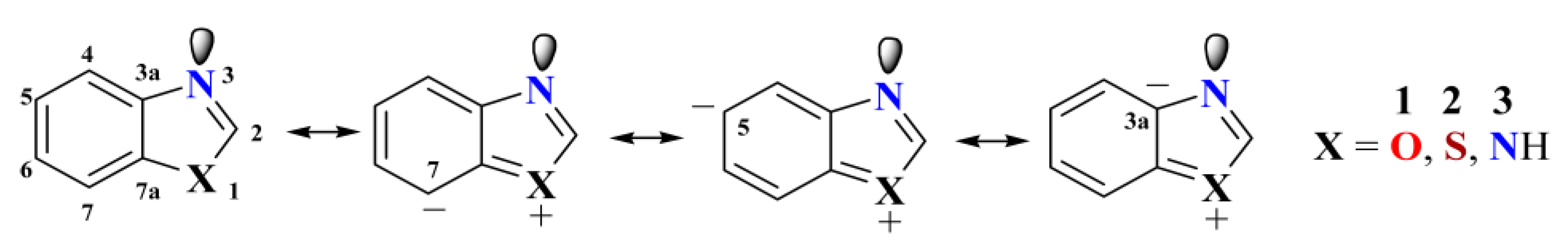
2.2. Effect of Heteroatoms on 13C-NMR of Benzazoles
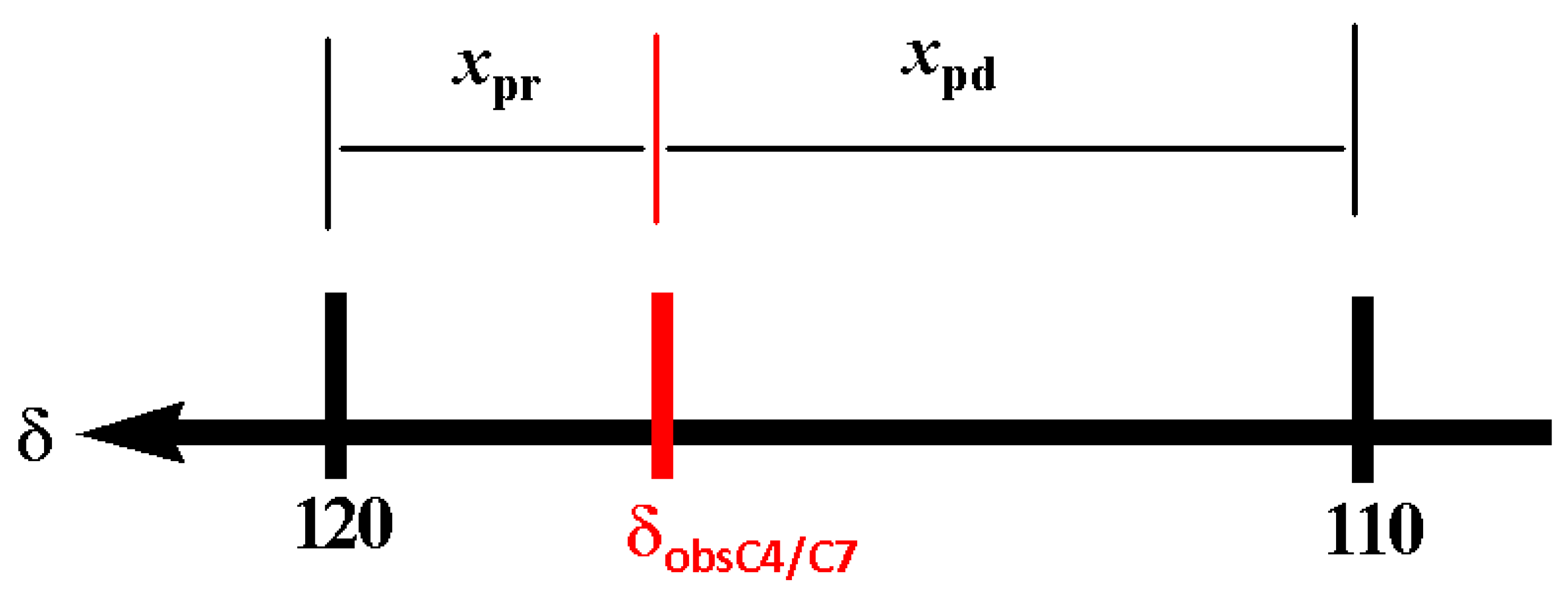
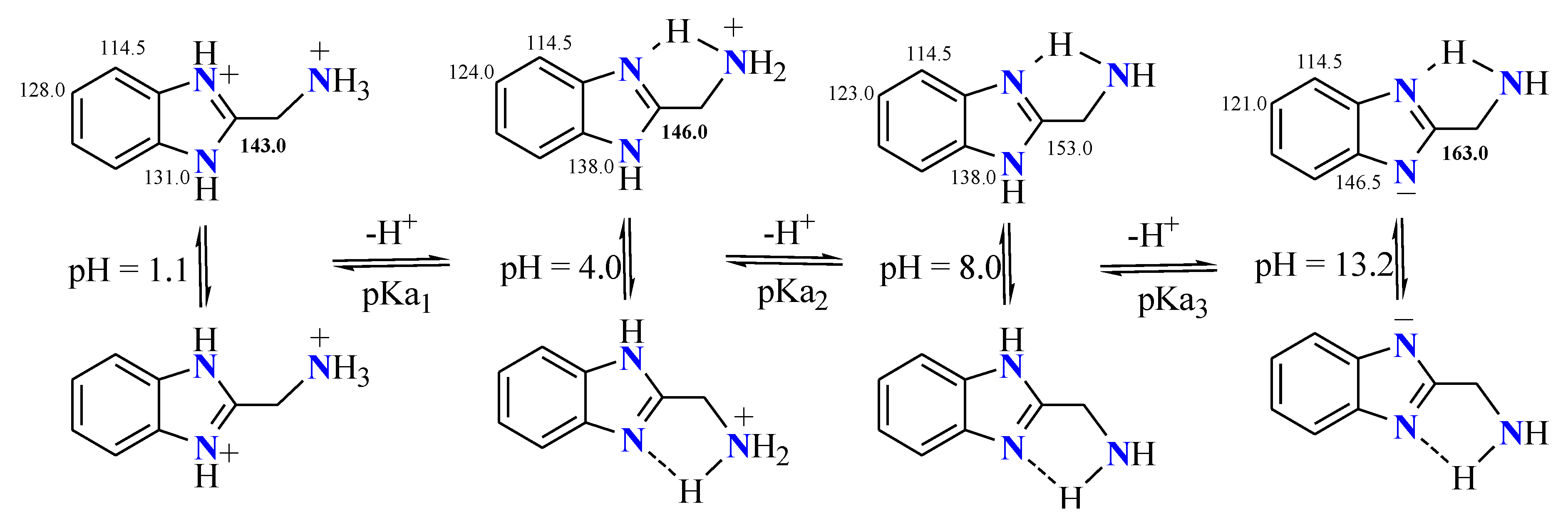
3. Estimation of the Anular Tautomerism on Benzimidazol
4. Tautomers and Mesomers in 1- and 2-Substituted Benzimidazoles
4.1. Effects of the Contribution of Mesomeric Structures in 1-Substituted Benzimidazoles
4.2. Effects of the Contribution of Tautomers in 2-Substituted Benzimidazoles
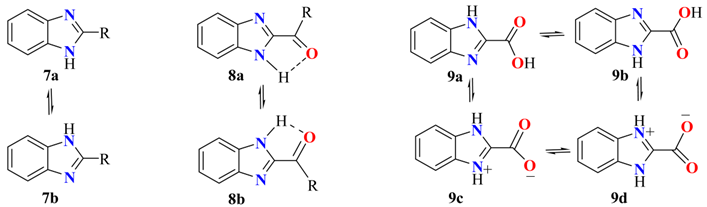
| Compound | C2 | C3a | C4 | C5 | C6 | C7 | C7a | Solvent | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 7 (R = Me) | 151.3 151.2 | 138.3 143.5 | 114.4 117.8 | 122.2 120.7 | 122.2 121.2 | 114.4 110.5 | 138.3 134.3 | CDCl3 DMSOd6 | [38] [33] |
| 7 (R = Et) | 157.4 | 139.4 | 114.7 | 122.7 | 122.7 | 114.7 | 139.4 | DMSOd6 | [33] |
| 7 (R = nPr) | 155.6 | 138.7 | 114.6 | 122.0 | 122.0 | 114.6 | 138.7 | CDCl3 | [33,39] |
| 7 (R = iPr) | 142.3 | 135.4 | 118.9 | 120.9 | 121.7 | 110.8 | 134.4 | DMSOd6 | [33] |
| 7 (R = Bn) | 151.8 | 133.2 | 114.0 | 125.8 | 125.8 | 114.0 | 133.2 | CDCl3 | [40] |
| 7 (R = Ph) | 151.8 | 144.4 | 119.4 | 123.0 | 122.2 | 111.8 | 135.5 | DMSOd6 | [31] |
| 8 (R = Me) | 147.7 | 143.5 | 122.1 | 123.8 | 122.1 | 112.1 | 126.6 | CDCl3 | [41] |
| 8 (R = Ph) | 147.7 | 143.9 | 122.3 | 126.5 | 123.8 | 112.0 | 133.9 | CDCl3 | [39] |
| 9 | 144.9 | 136.6 | 116.3 | 124.8 | 124.8 | 116.3 | 136.6 | DMSOd6 |
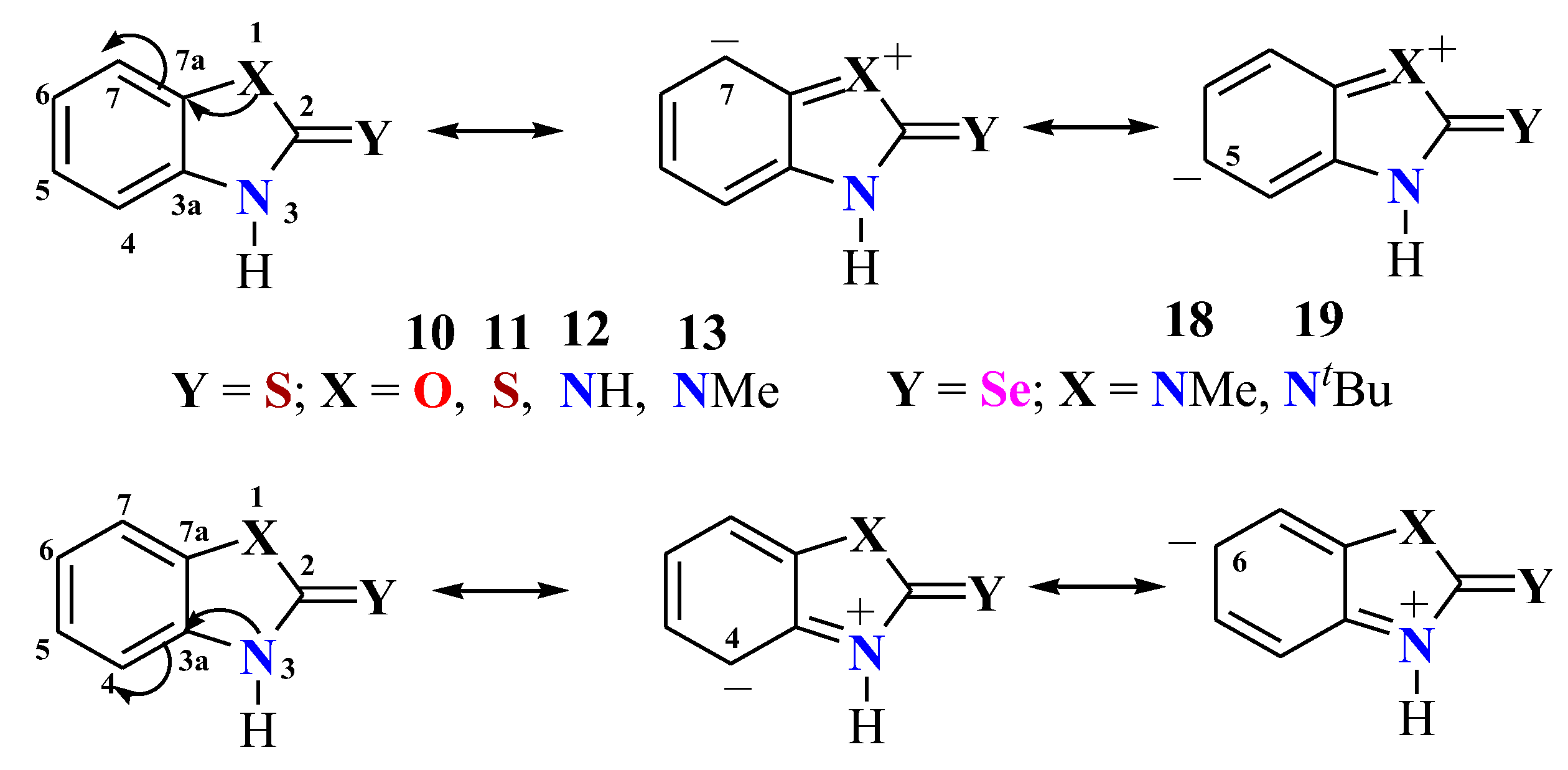
5. Iminothiol-Thioamide Tautomerism and Mesomers in 1,3-Benzoheterazolidine-2-thiones
5.1. Tautomers and Mesomers in Benzothiazolidine Tione
5.2. C4/C7 13C NMR Chemical Shifts of 1,3-Benzoheterazolidine-2-tiones in Iminothiol-Thioamide Tautomerism and Benzimidazolidine-2-selone Mesomerism
5.3. Mesomerism in N1-acyl-1,3-benzoheterazolidine-2-thiones
5.4. Tautomeric Equilibrium in Omeprazole
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elguero, J. Una Aproximación a la Tautomería de los Heterociclos Aromáticos. Rev. Soc. Quíim. Perú. 2020, 86, 452–476. [Google Scholar] [CrossRef]
- Guasch, L.; Yapamudiyansel, W.; Peach, M.L.; Kelley, J.A.; Barchi, J.J.; Nicklaus, M.C. Experimental and Chemoinformatics Study of Tautomerism in a Database of Commercially Available Screening Samples. J. Chem. Inf. Model. 2016, 56, 2149–2161. [Google Scholar] [CrossRef]
- Proj, M.; Sosič, I.; Gobec, S. Synthesis and NMR spectroscopic assignment of chlorinated benzimidazole-2-thione derivatives. Tetrahedron Lett. 2019, 60, 151078. [Google Scholar] [CrossRef]
- Nieto, C.I.; Cabildo, P.; García, M.A.; Claramunt, R.M.; Alkorta, I.; Elguero, J. An experimental and theoretical NMR study of NH-benzimidazoles in solution and in the solid state: Proton transfer and tautomerism. Beilstein J. Org. Chem. 2014, 10, 1620–1629. [Google Scholar] [CrossRef] [PubMed]
- García, M.A.; Claramunt, R.M.; Solcân, T.; Milata, V.; Alkorta, I.; Elguero, J. 13C and 15N NMR spectra of aminobenzimidazoles in solution and in the solid state. Magn. Reson. Chem. 2009, 47, 100–104. [Google Scholar] [CrossRef]
- Claramunt, R.M.; López, C.; Alkorta, I.; Elguero, J.; Yang, R.; Schulman, S. The tautomerism of Omeprazole in solution: A 1H and 13C NMR study. Magn. Reson Chem. 2004, 42, 712–714. [Google Scholar] [CrossRef]
- Larina, L.I. Chapter Five, Tautomerism and Structure of Azoles: Nuclear Magnetic Resonance Spectroscopy. In Advances in Heterocyclic Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; pp. 233–321. [Google Scholar]
- Form, G.R.; Raper, E.S.; Downie, T.C. The Crystal and Molecular Structure of 2-Mercaptobenzimidazole. Acta Crystallogr. 1976, B32, 345–348. [Google Scholar] [CrossRef]
- Khan, H.; Badshah, A.; Shaheen, F.; Gieck, C.; Qureshi, R.A. 1-Methyl-1H-benzimidazole-2(3H)-thione. Acta Crystallogr. Sect. E. Struct. Rep. Online 2008, 6, o1141. [Google Scholar] [CrossRef]
- Palomo-Molina, J.; García-Báez, E.V.; Contreras, R.; Pineda-Urbina, K.; Ramos-Organillo, A. Aminosilanes derived from 1H-benzimidazole-2(3H)-thione. Acta Cryst. Struct. Chem. 2015, C71, 788–792. [Google Scholar] [CrossRef]
- Balestrero, R.S.; Forkey, D.M.; Russell, J.G. Iminol-Thioamide Tautomerism of 2-mercaptobenzazoles and 1-methyl-2-mercaptoimidazoles. Magn. Reson. Chem. 1986, 24, 651–655. [Google Scholar] [CrossRef]
- Pandey, M.; Muthu, S.; Gowda, N.N.M. Quantum mechanical and spectroscopic (FT-IR, FT-Raman,1H,13C NMR, UV-Vis) studies, NBO, NLO, HOMO, LUMO and Fukui function analysis of 5-Methoxy-1H-benzo[d]imidazole-2(3H)-thione by DFT studies. J. Mol. Struct. 2017, 1130, 511–521. [Google Scholar] [CrossRef]
- Zhang, P.; Terefenko, E.; Kern, J.; Fensome, A.; Trybulski, E.; Unwalla, R.; Wrobel, J.; Lockhead, S.; Zhu, Y.; Cohen, J.; et al. 5-(3-Cyclopentyl-2-thioxo-2,3-dihydro-1H-benzimidazol-5-yl)-1-methyl-1H-pyrrole-2-carbonitrile: A novel, highly potent, selective, and orally active non-steroidal progesterone receptor agonist. Bioorg. Med. Chem. 2007, 15, 6556–6564. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Tatsuta, M.; Kataoka, M.; Yasoshima, K.; Shogase, Y.; Shimazaki, M.; Yura, T.; Li, Y.; Yamamoto, N.; Gupta, J.B.; et al. Benzimidazole derivatives as novel nonpeptide luteinizing hormone-releasing hormone (LHRH) antagonists. Part 1: Benzimidazole-5-sulfonamides. Bioorg. Med. Chem. Lett. 2005, 15, 799–803. [Google Scholar] [CrossRef] [PubMed]
- Tatsuta, M.; Kataoka, M.; Yasoshima, K.; Sakakibara, S.; Shogase, Y.; Shimazaki, M.; Yura, T.; Li, Y.; Yamamoto, N.; Gupta, J.; et al. Benzimidazoles as non-peptide luteinizing hormone-releasing hormone (LHRH) antagonists. Part 3: Discovery of 1-(1H-benzimidazol-5-yl)-3-tert-butylurea derivatives. Bioorg. Med. Chem. Lett. 2005, 15, 2265–2269. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, J.M.; Loyns, C.R. Synthesis of novel 1-, 1,4- and 1,7-substituted 2-mercapto- and 2-methylmercapto- benzimidazoles: Acyclic analogues of the HIV-1 RT inhibitor, TIBO. Tetrahedron 1995, 51, 11515–11530. [Google Scholar] [CrossRef]
- Hwu, J.R.; Singha, R.; Hong, S.C.; Chang, Y.H.; Das, A.R.; Vliegen, I.; De Clercq, E.; Neyts, J. Synthesis of new benzimidazole–coumarin conjugates as anti-hepatitis C virus agents. Antivir. Res. 2008, 77, 157–162. [Google Scholar] [CrossRef]
- Mavrova, A.T.; Vuchev, K.; Anichina, K.; Vassilev, N. Synthesis, antitrichinnellosis and antiprotozoal activity of some novel thieno [2,3-d]pyrimidin-4(3H)-ones containing benzimidazole ring. Eur. J. Med. Chem. 2010, 45, 5856–5861. [Google Scholar] [CrossRef]
- Pérez-Villanueva, J.; Hernández-Campos, A.; Yépez-Mulia, L.; Méndez-Cuesta, C.; Méndez-Lucio, O.; Hernández-Luis, F.; Castillo, R. Synthesis and antiprotozoal activity of novel 2-{[2-(1H-imidazol-1-yl)ethyl]sulfanyl}-1H-benzimidazole derivatives. Bioorg. Med. Chem. Lett. 2013, 23, 4221–4224. [Google Scholar] [CrossRef]
- Desai, K.G.; Desai, K.R. Green route for the heterocyclization of 2-mercaptobenzimidazole into β-lactum segment derivatives containing –CONH– bridge with benzimidazole: Screening in vitro antimicrobial activity with various microorganisms. Bioorg. Med. Chem. 2006, 14, 8271–8279. [Google Scholar] [CrossRef]
- Márquez-Navarro, A.; Nogueda-Torres, B.; Hernández-Campos, A.; Soria-Arteche, O.; Castillo, R.; Rodríguez-Morales, S.; Yépez-Mulia, L.; Hernández-Luis, F. Anthelmintic activity of benzimidazole derivatives against Toxocara canis second-stage larvae and Hymenolepis nana adults. Acta Trop. 2009, 109, 232–235. [Google Scholar] [CrossRef]
- Kazimierczuk, Z.; Andrzejewska, M.; Kaustova, J.; Klimešova, V. Synthesis and antimycobacterial activity of 2-substituted halogenobenzimidazoles. Eur. J. Med. Chem. 2005, 40, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Kankala, S.; Kankala, R.K.; Gundepaka, P.; Thota, N.; Nerella, S.; Gangula, M.R.; Guguloth, H.; Kagga, M.; Vadde, R.; Vasam, C.S. Regioselective synthesis of isoxazole–mercaptobenzimidazole hybrids and their in vivo analgesic and anti-inflammatory activity studies. Bioorg. Med. Chem. Lett. 2013, 23, 1306–1309. [Google Scholar] [CrossRef] [PubMed]
- Shingalapur, R.V.; Hosamani, K.M.; Keri, R.S.; Hugar, M.H. Derivatives of benzimidazole pharmacophore: Synthesis, anticonvulsant, antidiabetic and DNA cleavage studies. Eur. J. Med. Chem. 2010, 45, 1753–1759. [Google Scholar] [CrossRef] [PubMed]
- Pretsch, E.; Clerc, T.; Seibl, J.; Simon, W. Tablas Para la Determinación Estructural por Métodos Espectroscópicos; Herrera-Fernández, A., Martínez-Alvarez, R., Eds.; Springer-Verlag Ibérica: Barcelona, Spain, 1998; pp. 83–84, 93–94, 96. [Google Scholar]
- Takahiro, I.; Toshiaki, M. A Novel Practical Synthesis of Benzothiazoles via Pd-Catalyzed Thiol Cross-Coupling. Org. Lett. 2007, 9, 3687–3689. [Google Scholar]
- Khaksar, S.; Heydari, A.; Tajbakhsh, M.; Vahdat, S.M. Lewis acid catalyst free synthesis of benzimidazoles and formamidines in 1,1,1,3,3,3-hexafluoro-2-propanol. J. Fluor. Chem. 2010, 131, 1377–1381. [Google Scholar] [CrossRef]
- Alkorta, I.; Sánchez-Sanz, G.; Trujillo, T.; Elguero, J.; Claramunt, R.M. A theoretical study of the parent NH-benzazoles (benzimidazoles, indazoles and benzotriazoles): Geometries, energies, acidity and basicity, NMR properties and molecular electrostatic potentials. ARKIVOC 2012, 2, 85–106. [Google Scholar] [CrossRef]
- Graham, T.H.; Liu, W.; Shen, D.M. A Method for the Reductive Scission of Heterocyclic Thioethers. Org. Lett. 2011, 13, 6232–6235. [Google Scholar] [CrossRef]
- Ceniceros-Gómez, A.E.; Ramos-Organillo, A.; Hernández-Díaz, J.; Nieto-Martínez, J.; Contreras, R.; Castillo-Blum, S.E. NMR Study of the Coordinating Behavior of 2,6-bis(Benzimidazol-2’-yl)pyridine. Heteroat. Chem. 2000, 11, 392–398. [Google Scholar] [CrossRef]
- Sierra-Zenteno, A.; Galán-Vidal, C.A.; Tapia-Benavides, R. Acid-base equilibrium studies of 2-(aminomethyl)benzimidazole in aqueous solution. Rev. Soc. Quím. Méx. 2002, 46, 125–130. [Google Scholar]
- Koseki, D.; Aoto, E.; Shoji, T.; Watanabe, K.; In, Y.; Kita, Y.; Doh, T. Efficient N-arylation of azole compounds utilizing selective aryl-transfer TMP-iodonium(III) reagents. Tetrahedron Lett. 2019, 60, 1281–1286. [Google Scholar] [CrossRef]
- Saha, M.; Das, A.R. I2/TBHP promoted oxidative C–N bond formation at room temperature: Divergent access of 2-substituted benzimidazoles involving ring distortion. Tetrahedron Lett. 2018, 59, 2520–2525. [Google Scholar] [CrossRef]
- Chen, Z.Z.; Tang, Y.; Zuo, L.; Tang, D.Y.; Zhang, J.; Xu, Z.G. Facile One-Pot Synthesis of Benzimidazole and Quinoxalin-2(1H)-one Scaffolds via Two-Component Coupling Reaction, Deprotection and Intermolecular cyclization. Synlett 2014, 25, 2518–2520. [Google Scholar] [CrossRef]
- She, J.; Jiang, Z.; Wang, Y. One-Pot Synthesis of Functionalized Benzimidazoles and 1H-Pyrimidines via Cascade Reactions of o-Aminoanilines or Naphthalene-1,8-diamine with Alkynes and p-Tolylsulfonyl Azide. Synlet 2009, 12, 2023–2027. [Google Scholar] [CrossRef]
- Krawzyk, S.; Gdaniec, M.; Saczewski, F. 1H-Benzimidazol-3-ium-2-carboxylate dehydrate. Acta Cryst. 2005, E61, o4185–o4187. [Google Scholar]
- Yao, X.J.; Qian Yuan, Q. 1H-Benzimidazol-3-ium-2-carboxylate dihydrate. Acta Cryst. 2011, E67, o1399. [Google Scholar] [CrossRef]
- Mahajabeena, P.; Chadha, A. A novel green route for the synthesis of N-phenylacetamides, benzimidazoles and acridinediones using Candida parapsilosis ATCC 7330. RSC Adv. 2013, 3, 21972–21980. [Google Scholar] [CrossRef]
- Majumdar, S.; Chakraborty, A.; Bhattacharjee, S.; Debnath, S.; Maiti, D.K. Silica-ferric chloride (SiO2–FeCl3) catalyzed selective synthesis of 2-substituted benzimidazole through Csp2Csp3 bond cleavage of β-ketoester/amide. Tetrahedron Lett. 2016, 57, 4595–4598. [Google Scholar] [CrossRef]
- Dos Santos, A.; El Kaim, L.; Grimaud, L. Metal-free aerobic oxidation of benzazole derivatives. Org. Biomol. Chem. 2013, 11, 3282–3287. [Google Scholar] [CrossRef]
- Marko, J.A.; Durham, A.; Bretz, S.L.; Liu, W. Electrochemical benzylic oxidation of C–H bonds. Chem. Commun. 2019, 55, 937–940. [Google Scholar] [CrossRef]
- Rao, C.N.R.; Venkataraghavan, R.; Kasturi, T.R. Contribution to the Infrared Spectra of Organosulphur Compounds. Can. J. Chem. 1964, 42, 36–42. [Google Scholar] [CrossRef]
- Elguero, J.; Marzin, C.; Katritsky, A.R.; Linda, P. The Tautomerism of Heterocycles; Academic Press Inc.: New York, NY, USA, 1976; p. 397. [Google Scholar]
- Groth, P. Crystal Structure of Some Benzoxazoline Derivatives. Acta Chem. Scand. 1973, 27, 945–969. [Google Scholar] [CrossRef][Green Version]
- Chesick, J.P.; Donohue, J. The molecular and crystal structure of 2-mercaptobenzothiazole. Acta Crystallogr. 1971, B27, 1441–1444. [Google Scholar] [CrossRef]
- Prusiner, P.; Sundaralingam, M. Crystal and Molecular Structure of 2-thio-1-(b-D-ribofuranosyl)-3H-benzimidazole. Acta Crystallogr. 1973, B29, 2328–2334. [Google Scholar] [CrossRef]
- Faure, R.; Vincent, E.J.; Assef, G.; Kister, J. Effets de Substituants en Série Diazolique et Diazinique-1,3—Etude par Résonance Magnétique Nucléaire du Carbone-13. Metzg. J. Magn. Reson. Chem. 1977, 9, 688–694. [Google Scholar] [CrossRef]
- Faure, R.; Elguero, J.; Vincent, E.J.; Lazaro, R. Etudes d’Hétérocycles Pentagonaux Polyhétéroatomiques par RMN du 13C. Effets de Substituant, de la Benzocondensation et Etude de la Tautomèrie Prototropique en Série Benzothiazolique. Magn. Reson. Chem. 1978, 11, 617–627. [Google Scholar]
- Cumper, C.W.N.; Pickering, G.D. Electric dipole moments of some imidazolin-2(3H)-ones, benzimidazol-2(3H)-ones, and analogous thiones. J. Chem. Soc. Perkin Trans. 1972, 2, 2045–2048. [Google Scholar] [CrossRef]
- Vasava, M.S.; Rathwa, S.K.; Jethava, D.J.; Acharya, P.T.; Patela, D.B.; Patel, H.D. Benzimidazole: A milestone in the field of medicinal chemistry. Mini-Rev. Med. Chem. 2020, 20, 532–565. [Google Scholar] [CrossRef]
- Ziemkowska, W.; Skałkowska, J.; Ochal, Z.; Cyranski, M.K.; Dobrzycki, L.; Madura, I.; Zachara, J. Role of Lewis bases in reactions of aluminum and gallium trialkyls with 2-mercaptobenzoxazole. J. Organomet. Chem. 2015, 776, 1–6. [Google Scholar] [CrossRef]
- Bethge, L.; Venkatrao, D.; Seitz, O. New cyanine dyes as base surrogates in PNA: Forced intercalation probes (FIT-probes) for homogeneous SNP detection. Bioorg. Med. Chem. 2008, 16, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.O.A.; Mojica, S.J.P.; Valcárcel, G.J.A.; Obledo, B.F.; Macías, H.C.E.; Sumaya, M.M.T.; Martínez, M.F.J.; Gómez, S.Z.; Ramos-Organillo, A. Synthesis of novel amides with antiradical capacity from 2-mercaptobenzimidazole and cinnamic acids: Evaluation through donor-acceptor maps and QSAR. J. Mol. Struct. 2021, 1223, 128917. [Google Scholar]
- Geng, X.; Liu, S.; Wang, W.; Qu, J.; Wang, B. tert-Amino Effect-Promoted Rearrangement of Aryl Isothiocyanate: A Versatile Approach to Benzimidazothiazepines and Benzimidazothioethers. J. Org. Chem. 2020, 85, 12635–12643. [Google Scholar] [CrossRef] [PubMed]
- Colacio, E.; Romerosa, A.; Ruiz, J.; Román, P.; Gutíerrez-Zorrilla, J.M.; Vegas, A.; Martínez-Ripoll, M. Gold(I) phosphine complexes: Mercaptooxopurine base interactions. Molecular and crystal structure of (8-mercaptotheophyllinato-S)(triphenylphosphine)gold(I). Inorg. Chem. 1991, 30, 3743–3749. [Google Scholar] [CrossRef]
- Vicente, J.; Chicote, M.T.; González-Herrero, P.; Jones, P.G. Complexes with S-donor ligands. Part 2. Synthesis of anionic bis(thiolato)gold(I) complexes. Crystal structure of [N(PPh3)2][Au(SR)2](R = benzoxazol-2-yl). J. Chem. Soc. Dalton Trans. 1994, 21, 3183–3187. [Google Scholar] [CrossRef]
- Aguilar, C.H. Síntesis y Reactividad de Benzoderivados-2-tiona y Benzoderivados-2-methylmercapto con el BH3-THF. Bachelor’s Thesis, Universidad Veracruzana, Posa Rica, Veracruz, México, 1996. [Google Scholar]
- Palmer, J.H.; Parkin, G. 2-Seleno-1-alkylbenzimidazoles and their diselenides: Synthesis and structural characterization of a 2-seleno-1-methylbenzimidazole complex of mercury. Polyhedron 2013, 52, 658–668. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Braun, S.; Botzki, A.; Salmen, S.; Textor, C.; Bernhardt, G.; Dove, S.; Buschauer, A. Design of benzimidazole- and benzoxazole-2-thione derivatives as inhibitors of bacterial hyaluronan lyase. Eur. J. Med. Chem. 2011, 46, 4419–4429. [Google Scholar] [CrossRef] [PubMed]
- El Ashry, E.S.H.; El Kilany, Y.; Nariman, M.; Nahas, N.M.; Barakat, A.; Al-Qurashi, N.; Hazem, A.; Ghabbour, H.A.; Fun, H.K. Synthesis and Crystal Structures of Benzimidazole-2-thione Derivatives by Alkylation Reactions. Molecules 2016, 21, 12. [Google Scholar] [CrossRef] [PubMed]
- Obledo-Benicio, F.; Magaña-Vergara, N.E.; Pineda-Urbina, K.; Perez, D.J.; Romero-Chavez, M.M.; Mojica-Sanchez, J.P.; Ramos-Rodríguez, O.R.; Ramos-Organillo, A. Structural analysis of intramolecular 1,5-type O/OandS/Ointeractions in diethyl 2-oxo and diethyl 2-thioxo-1H benzo[d]imidazole-1,3(2H)-dicarboxylate: Experimental and theoretical study. J. Mol. Struct. 2020, 1209, 127929. [Google Scholar] [CrossRef]
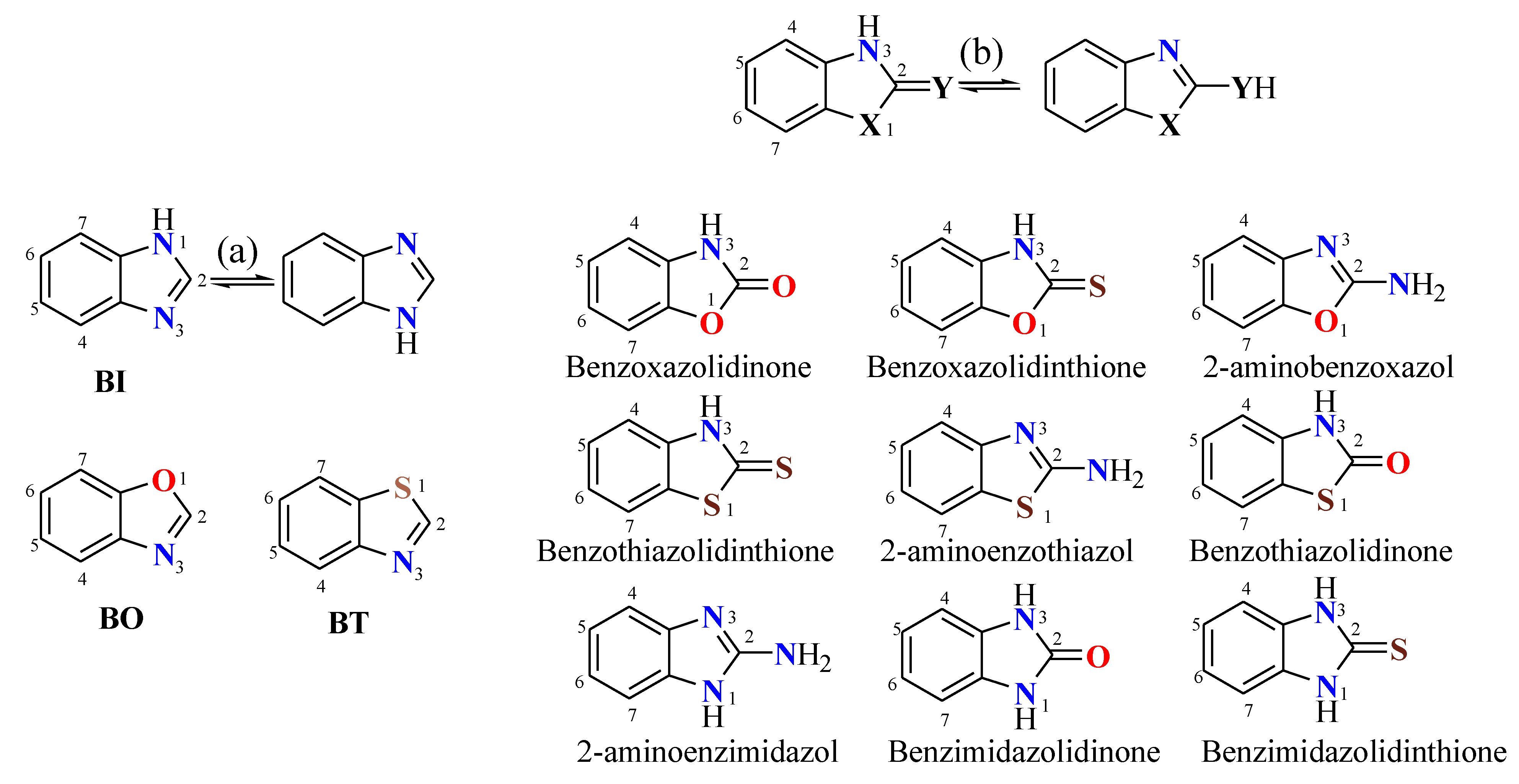
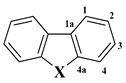
| δ-13C | ||||||
|---|---|---|---|---|---|---|
| Comp. | C1a | C1 | C2 | C3 | C4 | C4a |
| 1 (X = O) | 124.2 | 120.6 | 122.6 | 127.0 | 111.6 | 156.2 |
| 2 (X = S) | 134.9 | 121.9 | 124.6 | 127.0 | 122.9 | 138.5 |
| 3 (X = NH) | 122.6 | 120.0 | 118.4 | 125.4 | 110.8 | 139.6 |
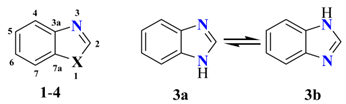
| Comp. | C2 | C3a | C4 | C5 | C6 | C7 | C7a | Solv. | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 1 (X = O) | 152.6 | 140.1 | 120.5 | 125.4 | 124.4 | 110.8 | 150.5 | CDCl3 | [25] |
| 2 (X = S) | 153.8 | 153.2 | 121.8 | 126.1 | 125.4 | 123.6 | 133.6 | CDCl3 | [25,26] |
| 140.5 | 137.1 | 115.3 | 121.3 | 121.3 | 115.3 | 137.1 | CDCl3 | [25,27] | |
| 141.1 | 137.4 | 114.8 | 122.7 | 122.1 | 114.8 | 137.4 | CD3OD | [28] | |
| 3 (X = NH) | 141.9 | 138.4 | 115.3 | 121.7 | 121.7 | 115.3 | 138.4 | DMSOd6 | [4] |
| 141.6 | 143.4 | 118.7 | 121.2 | 120.1 | 110.9 | 133.7 | HMPAd18 | [4] | |
| 142.6 | 143.0 | 119.9 | 122.1 | 123.0 | 112.2 | 136.1 | Solid | [4] | |
| 4 (X = NMe) | 143.9 | 143.7 | 120.4 | 123.1 | 122.2 | 109.5 | 134.7 | CDCl3 | [29] |
| 144.5 | 143.3 | 119.2 | 122.3 | 121.1 | 110.1 | 134.5 | DMSOd6 | [4] |
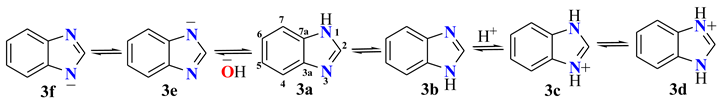
| Comp. | C2 | C3a | C4 | C5 | C6 | C7 | C7a |
|---|---|---|---|---|---|---|---|
| 3a/3b | 142.5 | 139.0 | 116.3 | 122.3 | 122.3 | 116.3 | 139.0 |
| [3c/3d]+ | 143.6 | 133.8 | 118.3 | 130.0 | 130.0 | 118.3 | 133.8 |
| [3d/3f]− Na+ | 153.1 | 146.9 | 116.4 | 116.7 | 116.7 | 116.4 | 146.9 |
| [3d/3f]− Li+ | 153.5 | 147.0 | 116.8 | 117.0 | 117.0 | 116.8 | 147.0 |
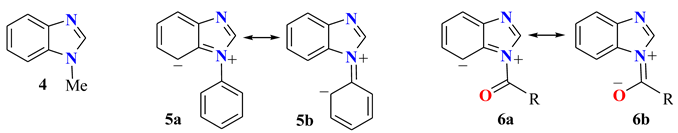
| Comp. | C2 | C3a | C4 | C5 | C6 | C7 | C7a | Solvent | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 4 | 143.9 144.5 | 143.7 143.3 | 120.4 119.2 | 123.1 122.1 | 122.2 121.3 | 109.5 110.1 | 134.7 134.5 | CDCl3 DMSOd6 | [29] [4] |
| 5 | 144.0 151.6 | 142.2 143.8 | 120.6 119.0 | 123.6 122.1 | 122.7 121.2 | 110.4 116.5 | 133.7 134.8 | CDCl3 DMSOd6 | [32] [33] |
| 6 | 143.9 | 141.4 | 120.6 | 126.0 | 125.1 | 115.5 | 131.4 | CDCl3 | [34] |
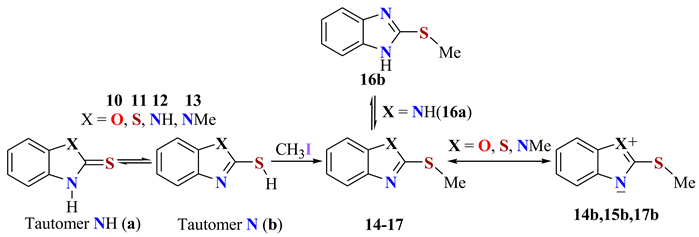
| Comp. | C2 | C4 | C5 | C6 | C7 | C7a | C3a | Solv. | Ref. | % Npr |
|---|---|---|---|---|---|---|---|---|---|---|
| 10 | 180.8 181.8 | 110.2 113.2 | 124.5 119.2 | 125.4 121.6 | 110.5 106.7 | 148.8 151.0 | 130.3 145.4 | CDCl3 DMSOd6 | [51] | 93.7 68.0 |
| 11 | 189.7 | 112.3 | 124.0 | 127.0 | 121.6 | 141.2 | 129.3 | DMSOd6 | 77.0 | |
| 12 | 167.9 | 109.6 | 122.3 | 122.3 | 109.6 | 132.7 | 132.7 | DMSOd6 | 100 | |
| 13 | 168.3 | 118.3 | 123.4 | 123.4 | 110.1 | 132.6 | 130.8 | DMSOd6 | 8.4 | |
| 14 | 165.7 165.2 | 118.3 118.1 | 123.7 124.0 | 124.2 124.5 | 109.8 110.0 | 151.9 151.4 | 141.9 141.4 | CDCl3 DMSOd6 | [52] | 8.4 10.5 |
| 15 | 170.7 167.9 | 122.0 121.7 | 125.4 124.3 | 127.4 126.3 | 123.3 120.9 | 136.1 134.5 | 154.4 152.8 | CD3OD DMSOd6 | [52] | 0.0 2.4 |
| 16 | 151.9 151.2 152.3 | 113.7 113.6 117.8 | 121.7 121.2 121.9 | 121.7 121.2 121.9 | 113.7 113.6 110.9 | 139.5 139.7 136.4 | 139.5 139.7 144.5 | CDCl3 DMSOd6 (27 °C) DMF-d7 (−65 °C) | [53] | 56.8 57.9 13.7 |
| 17 | 153.2 152.6 | 118.0 117.4 | 121.8 121.2 | 121.8 121.2 | 108.3 109.2 | 136.8 136.9 | 143.3 142.9 | CDCl3 DMSOd6 | [54] | 11.6 17.9 |
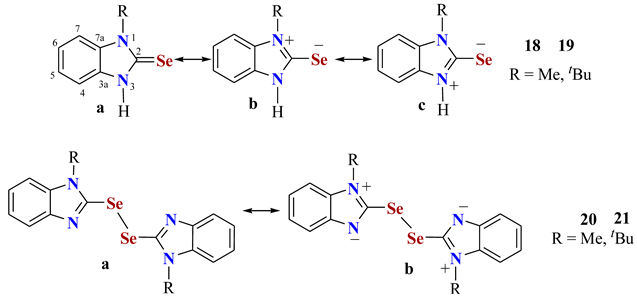
| Comp. | C2 | C3a | C4 | C5 | C6 | C7 | C7a | Solvent | Ref. | N4pr% |
|---|---|---|---|---|---|---|---|---|---|---|
| 18 | 162.8 | 132.1 | 110.0 | 123.3 | 122.6 | 110.0 | 133.7 | DMSOd6 | [58] | 95.8 |
| 19 | 160.3 | 133.0 | 114.4 | 121.1 | 122.8 | 109.9 | 133.8 | DMSOd6 | [58] | 50.5 |
| 20 | 142.6 | 144.3 | 110.5 | 123.0 | 124.4 | 120.4 | 137.3 | CD2Cl2 | [58] | 90.5 |
| 21 | 145.6 | 145.3 | 114.1 | 121.8 | 121.9 | 118.8 | 136.8 | CD2Cl2 | [58] | 52.7 |
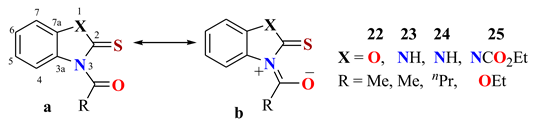
| Comp. | C2 | C3a | C4 | C5 | C6 | C7 | C7a | Solvent | Npr% | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| 22 | 178.8 | 129.5 | 115.8 | 125.5 | 126.0 | 109.7 | 145.9 | DMSOd6 | 34.7 | [58] |
| 23 | 168.9 | 129.8 | 114.3 | 122.3 | 124.3 | 108.5 | 130.1 | DMSOd6 | 50.5 | [60] |
| 24 | 169.4 | 130.9 | 115.0 | 123.3 | 125.2 | 109.4 | 131.0 | DMSOd6 | 43.1 | [60] |
| 25 | 168.1 | 129.5 | 113.2 | 125.1 | 125.1 | 113.2 | 129.5 | CDCl3 | 62.1 | [61] |
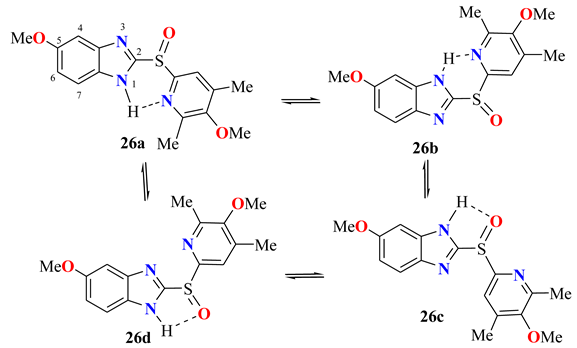
| Claramunt et al. | C2 | C3a | C4 | C5 | C6 | C7 | C7a | Solvent | Npr% |
|---|---|---|---|---|---|---|---|---|---|
| 26a = 26b | 155.4 | 146.0 | 101.1 | 157.4 | 115.3 | 113.1 | 129.9 | THF-d8 | 63 |
| 26c = 26d | 153.5 | 140.2 | 121.6 | 114.2 | 158.2 | 94.0 | 136.5 | THF-d8 | 37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Báez, E.V.; Padilla-Martínez, I.I.; Cruz, A.; Rosales-Hernández, M.C. 13C-NMR Chemical Shifts in 1,3-Benzazoles as a Tautomeric Ratio Criterion. Molecules 2022, 27, 6268. https://doi.org/10.3390/molecules27196268
García-Báez EV, Padilla-Martínez II, Cruz A, Rosales-Hernández MC. 13C-NMR Chemical Shifts in 1,3-Benzazoles as a Tautomeric Ratio Criterion. Molecules. 2022; 27(19):6268. https://doi.org/10.3390/molecules27196268
Chicago/Turabian StyleGarcía-Báez, Efrén V., Itzia I. Padilla-Martínez, Alejandro Cruz, and Martha C. Rosales-Hernández. 2022. "13C-NMR Chemical Shifts in 1,3-Benzazoles as a Tautomeric Ratio Criterion" Molecules 27, no. 19: 6268. https://doi.org/10.3390/molecules27196268
APA StyleGarcía-Báez, E. V., Padilla-Martínez, I. I., Cruz, A., & Rosales-Hernández, M. C. (2022). 13C-NMR Chemical Shifts in 1,3-Benzazoles as a Tautomeric Ratio Criterion. Molecules, 27(19), 6268. https://doi.org/10.3390/molecules27196268







