Comparison of the Antihypertensive Activity of Phenolic Acids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. ACE Inhibitory Activity Assay
2.3. Cell Culture and Sample Treatment
2.4. Measurement of Intracellular ROS, GSH, Malondialdehyde, and NO Levels
2.5. Western Blot Analysis
2.6. Statistical Analysis
3. Results and Discussion
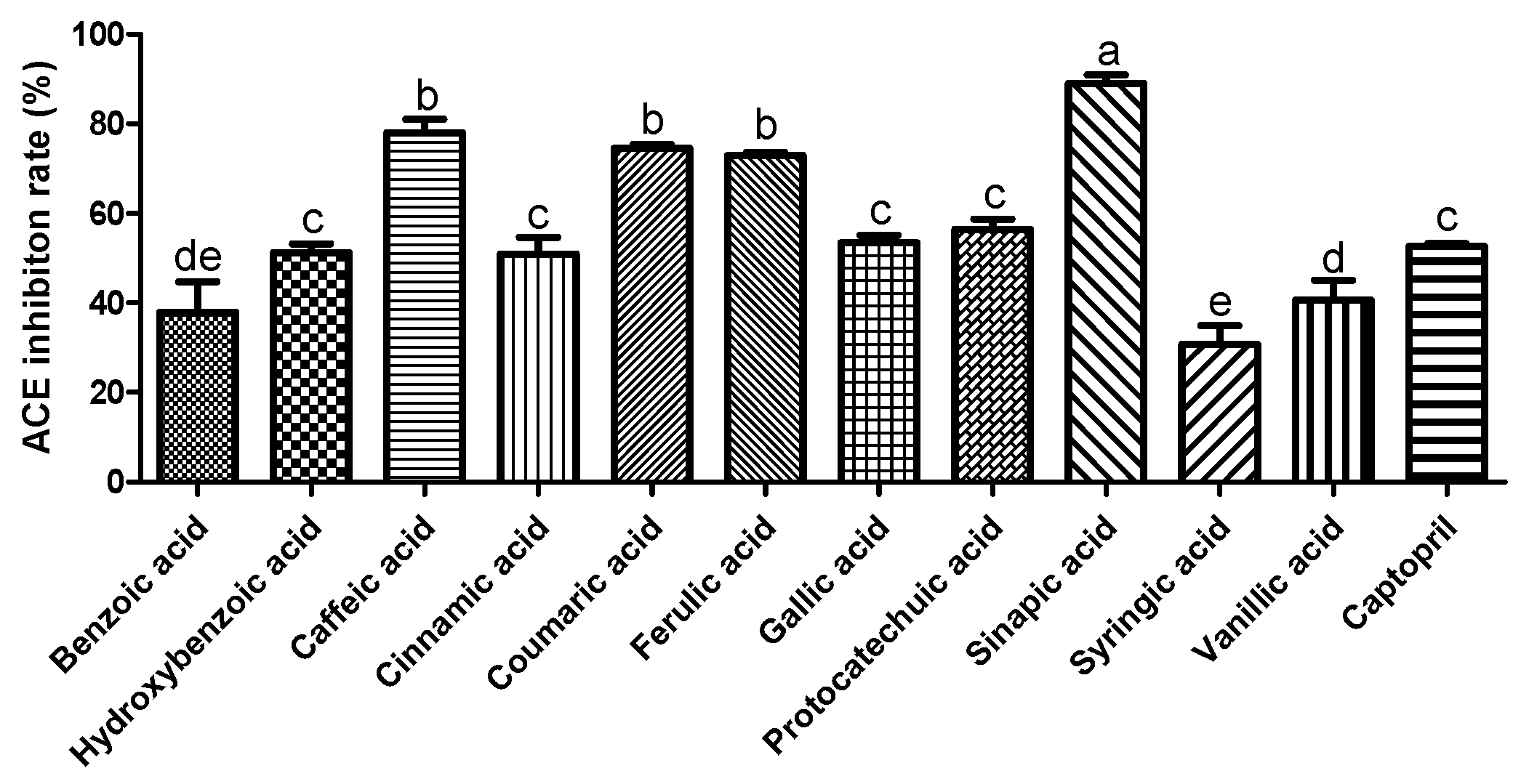
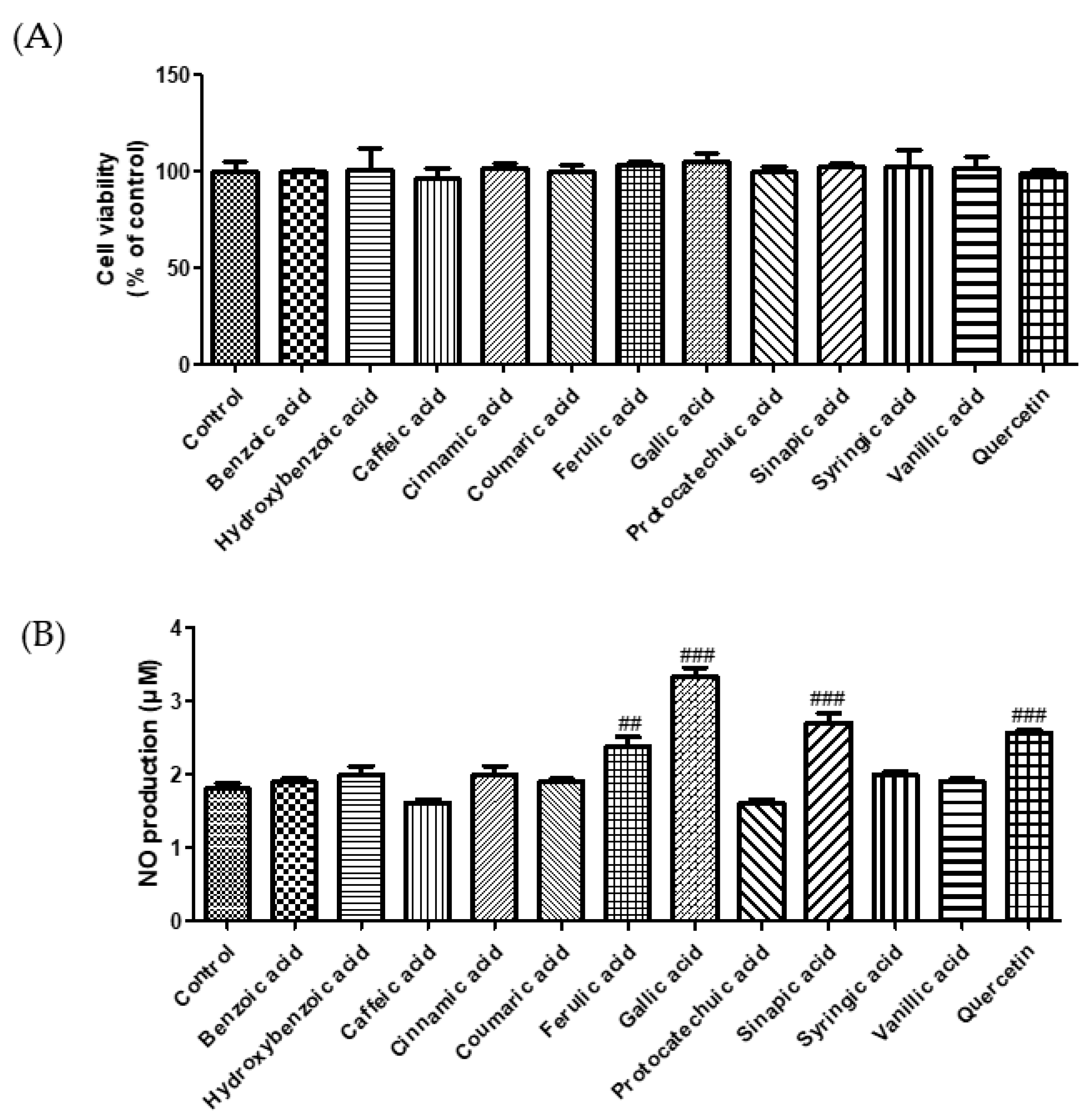
3.1. Effect of Phenolic Acids on ACE Inhibition and NO Production
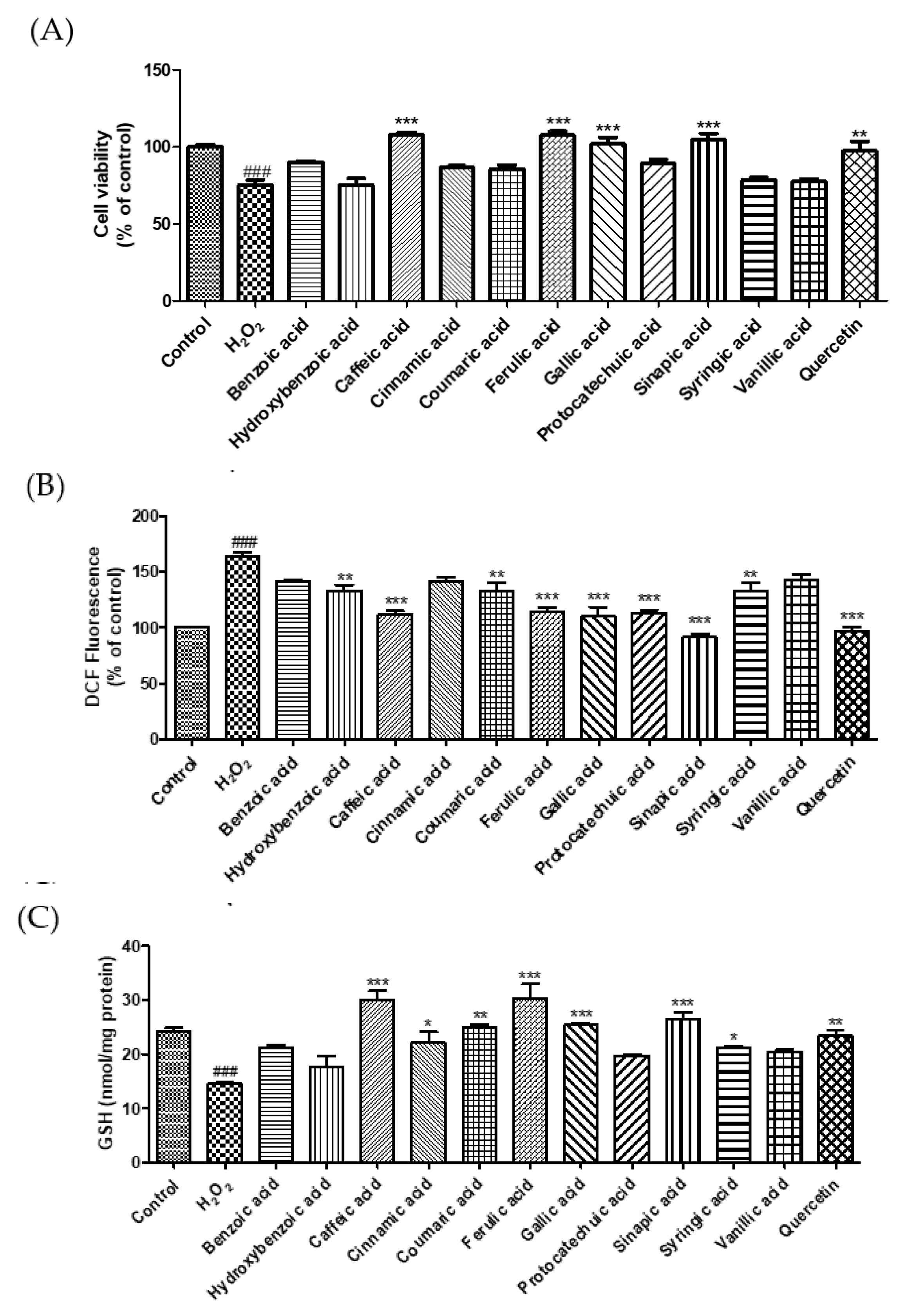
3.2. Cytoprotective Effect of Phenolic Acids against Hydrogen Peroxide
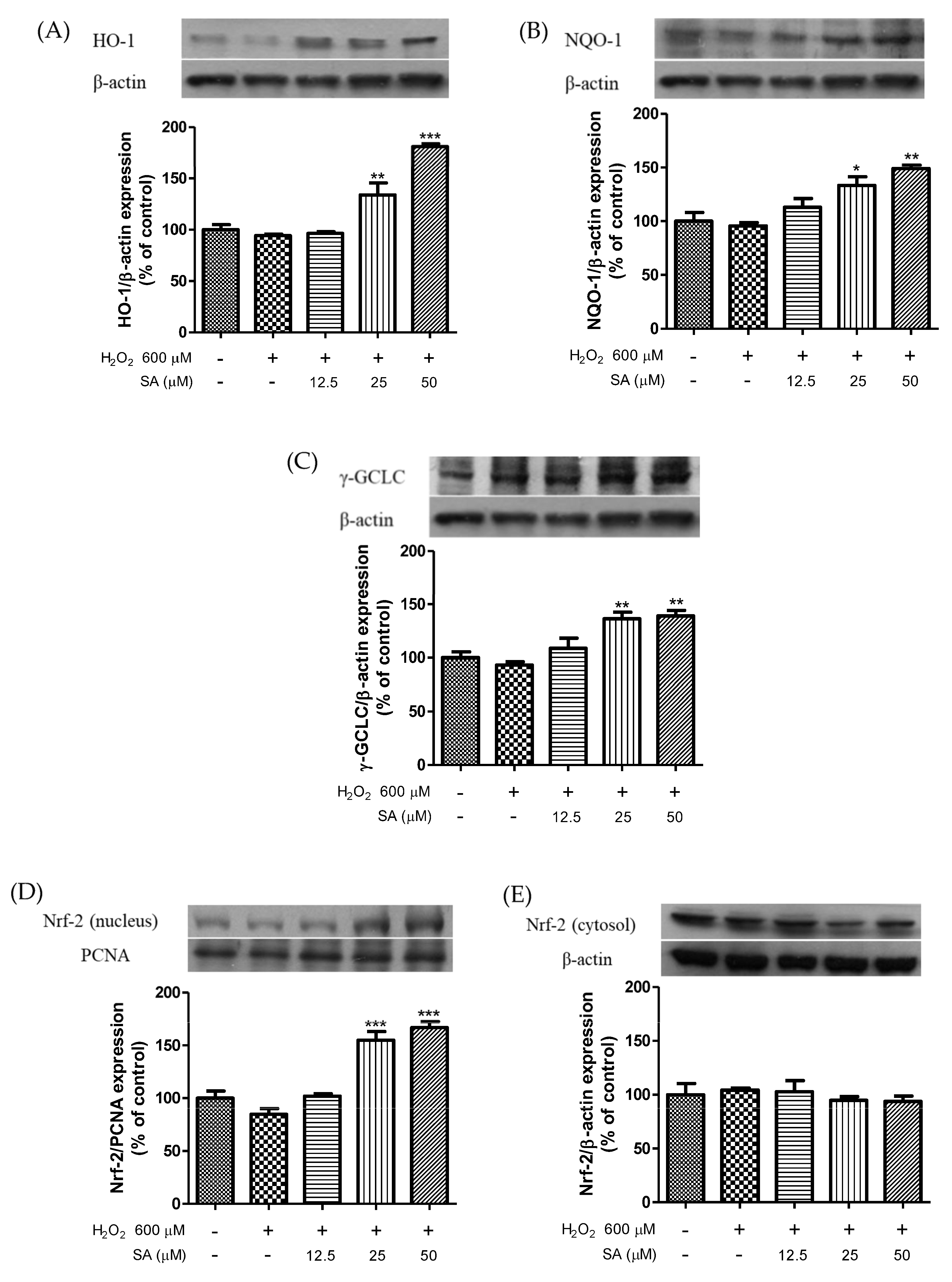
3.3. Effects of Sinapic Acid on the Expression of Phase II Enzymes and the Activation of Nrf2
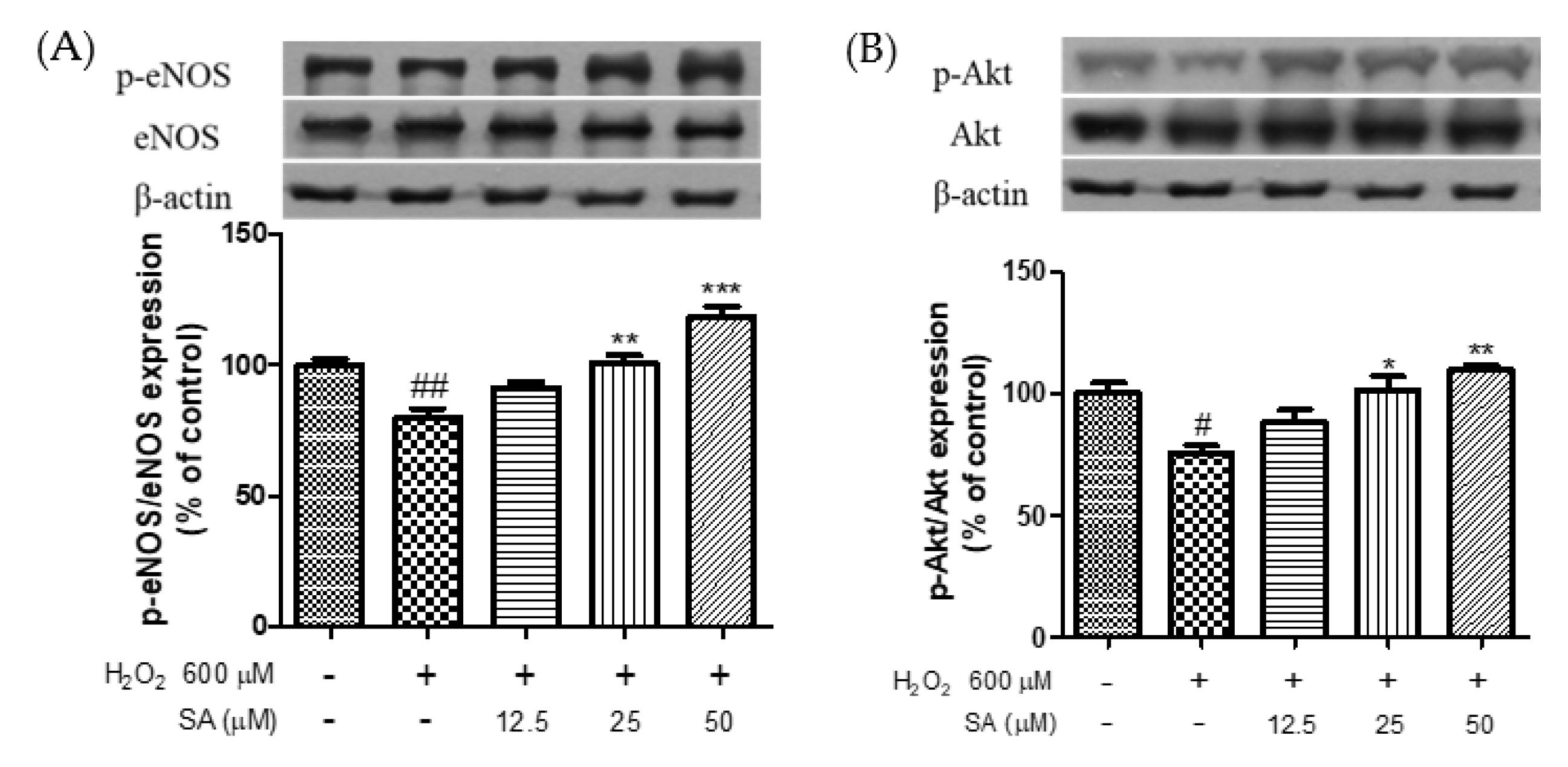
3.4. Effects of Phenolic Acids on Endothelial Dysfunction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Wu, L.; Ashraf, M.H.N.; Facci, M.; Wang, R.; Paterson, P.G.; Ferrie, A.; Juurlink, B.H.J. Dietary Approach to Attenuate Oxidative Stress, Hypertension, and Inflammation in the Cardiovascular System. Proc. Natl. Acad. Sci. USA 2004, 101, 7094–7099. [Google Scholar] [CrossRef]
- Loperena, R.; Harrison, D.G. Oxidative Stress and Hypertensive Diseases. Med. Clin. N. Am. 2017, 101, 169–193. [Google Scholar] [CrossRef]
- Zhang, C.; Hein, T.W.; Wang, W.; Miller, M.W.; Fossum, T.W.; McDonald, M.M.; Humphrey, J.D.; Kuo, L. Upregulation of Vascular Arginase in Hypertension Decreases Nitric Oxide-Mediated Dilation of Coronary Arterioles. Hypertension 2004, 44, 935–943. [Google Scholar] [CrossRef]
- Folkow, B.; Grimby, G.; Thulesius, O. Adaptive Structural Changes of the Vascular Walls in Hypertension and Their Relation to the Control of the Peripheral Resistance. Acta Physiol. Scand. 1958, 44, 255–272. [Google Scholar] [CrossRef]
- Lu, X.; Zhang, L.; Sun, Q.; Song, G.; Huang, J. Extraction, Identification and Structure-Activity Relationship of Antioxidant Peptides from Sesame (Sesamum indicum L.) Protein Hydrolysate. Food Res. Int. 2019, 116, 707–716. [Google Scholar] [CrossRef]
- Howden, R. Nrf2 and Cardiovascular Defense. Oxidative Med. Cell. Longev. 2013, 2013, e104308. [Google Scholar] [CrossRef]
- Zhang, Y.; Guan, L.; Wang, X.; Wen, T.; Xing, J.; Zhao, J. Protection of Chlorophyllin against Oxidative Damage by Inducing HO-1 and NQO1 Expression Mediated by PI3K/Akt and Nrf2. Free Radic. Res. 2008, 42, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Wang, Y.-C.; Zhao, T.-Z.; Zhang, S.; Du, T.-Y.; Yang, C.-B.; Li, Y.-H.; Sun, X.-Q. Effects of Simulated Microgravity on Human Umbilical Vein Endothelial Cell Angiogenesis and Role of the PI3K-Akt-ENOS Signal Pathway. PLoS ONE 2012, 7, e40365. [Google Scholar] [CrossRef]
- Iaccarino, G.; Ciccarelli, M.; Sorriento, D.; Cipolletta, E.; Cerullo, V.; Iovino, G.L.; Paudice, A.; Elia, A.; Santulli, G.; Campanile, A.; et al. AKT Participates in Endothelial Dysfunction in Hypertension. Circulation 2004, 109, 2587–2593. [Google Scholar] [CrossRef]
- Sun, J.; Chu, Y.-F.; Wu, X.; Liu, R.H. Antioxidant and Antiproliferative Activities of Common Fruits. J. Agric. Food Chem. 2002, 50, 7449–7454. [Google Scholar] [CrossRef]
- Saibabu, V.; Fatima, Z.; Khan, L.A.; Hameed, S. Therapeutic Potential of Dietary Phenolic Acids. Adv. Pharmacol. Sci. 2015, 2015, e823539. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, M.; Sudheer, A.R.; Menon, V.P. Ferulic Acid: Therapeutic Potential Through Its Antioxidant Property. J. Clin. Biochem. Nutr. 2007, 40, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.; Lee, J.-H.; Lee, W.; Ko, J.-Y.; Kim, E.-A.; Kim, J.-S.; Heu, M.-S.; Kim, G.H.; Jeon, Y.-J. Gallic Acid Isolated from Spirogyra Sp. Improves Cardiovascular Disease through a Vasorelaxant and Antihypertensive Effect. Environ. Toxicol. Pharmacol. 2015, 39, 764–772. [Google Scholar] [CrossRef]
- Jin, L.; Lin, M.Q.; Piao, Z.H.; Cho, J.Y.; Kim, G.R.; Choi, S.Y.; Ryu, Y.; Sun, S.; Kee, H.J.; Jeong, M.H. Gallic Acid Attenuates Hypertension, Cardiac Remodeling, and Fibrosis in Mice with NG-Nitro-L-Arginine Methyl Ester-Induced Hypertension via Regulation of Histone Deacetylase 1 or Histone Deacetylase 2. J. Hypertens. 2017, 35, 1502–1512. [Google Scholar] [CrossRef]
- Alam, M.A. Anti-Hypertensive Effect of Cereal Antioxidant Ferulic Acid and Its Mechanism of Action. Front. Nutr. 2019, 6, 121. [Google Scholar] [CrossRef]
- Agunloye, O.M.; Oboh, G.; Ademiluyi, A.O.; Ademosun, A.O.; Akindahunsi, A.A.; Oyagbemi, A.A.; Omobowale, T.O.; Ajibade, T.O.; Adedapo, A.A. Cardio-Protective and Antioxidant Properties of Caffeic Acid and Chlorogenic Acid: Mechanistic Role of Angiotensin Converting Enzyme, Cholinesterase and Arginase Activities in Cyclosporine Induced Hypertensive Rats. Biomed. Pharmacother. 2019, 109, 450–458. [Google Scholar] [CrossRef]
- Cushman, D.W.; Cheung, H.S. Spectrophotometric Assay and Properties of the Angiotensin-Converting Enzyme of Rabbit Lung. Biochem. Pharmacol. 1971, 20, 1637–1648. [Google Scholar] [CrossRef]
- Nasution, S.A. The Use of ACE Inhibitor in Cardiovascular Disease. Acta Med. Indones. 2006, 38, 60–64. [Google Scholar]
- Ojeda, D.; Jiménez-Ferrer, E.; Zamilpa, A.; Herrera-Arellano, A.; Tortoriello, J.; Alvarez, L. Inhibition of Angiotensin Convertin Enzyme (ACE) Activity by the Anthocyanins Delphinidin- and Cyanidin-3-O-Sambubiosides from Hibiscus Sabdariffa. J. Ethnopharmacol. 2010, 127, 7–10. [Google Scholar] [CrossRef]
- Zhang, Y.; Pechan, T.; Chang, S.K.C. Antioxidant and Angiotensin-I Converting Enzyme Inhibitory Activities of Phenolic Extracts and Fractions Derived from Three Phenolic-Rich Legume Varieties. J. Funct. Foods 2018, 42, 289–297. [Google Scholar] [CrossRef]
- Joannides, R.; Haefeli, W.E.; Linder, L.; Richard, V.; Bakkali, E.H.; Thuillez, C.; Lüscher, T.F. Nitric Oxide Is Responsible for Flow-Dependent Dilatation of Human Peripheral Conduit Arteries in Vivo. Circulation 1995, 91, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Y.; Fang, Z.; Huang, D.; Yang, Y.; Zhao, D.; Hang, M.; Wang, J. The Effect of Malus Doumeri Leaf Flavonoids on Oxidative Stress Injury Induced by Hydrogen Peroxide (H2O2) in Human Embryonic Kidney 293 T Cells. BMC Complement. Med. Ther. 2020, 20, 276. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhang, C.; Zhang, J.; Zhao, X. Wu Shan Shen Cha (Malus Asiatica Nakai. Leaves)-Derived Flavonoids Alleviate Alcohol-Induced Gastric Injury in Mice via an Anti-Oxidative Mechanism. Biomolecules 2019, 9, 169. [Google Scholar] [CrossRef]
- Xiang, Y.; Ye, W.; Huang, C.; Lou, B.; Zhang, J.; Yu, D.; Huang, X.; Chen, B.; Zhou, M. Brusatol Inhibits Growth and Induces Apoptosis in Pancreatic Cancer Cells via JNK/P38 MAPK/NF-Κb/Stat3/Bcl-2 Signaling Pathway. Biochem. Biophys. Res. Commun. 2017, 487, 820–826. [Google Scholar] [CrossRef]
- Lee, H.; Lee, J. Anti-Diabetic Effect of Hydroxybenzoic Acid Derivatives in Free Fatty Acid-Induced HepG2 Cells via MiR-1271/IRS1/PI3K/AKT/FOXO1 Pathway. J. Food Biochem. 2021, 45, e13993. [Google Scholar] [CrossRef]
- Jadeja, R.N.; Upadhyay, K.K.; Devkar, R.V.; Khurana, S. Naturally Occurring Nrf2 Activators: Potential in Treatment of Liver Injury. Oxid. Med. Cell Longev. 2016, 2016, 3453926. [Google Scholar] [CrossRef]
- Yang, S.-Y.; Pyo, M.C.; Nam, M.-H.; Lee, K.-W. ERK/Nrf2 Pathway Activation by Caffeic Acid in HepG2 Cells Alleviates Its Hepatocellular Damage Caused by t-Butylhydroperoxide-Induced Oxidative Stress. BMC Complement. Altern. Med. 2019, 19, 139. [Google Scholar] [CrossRef]
- Luo, W.; Wang, Y.; Yang, H.; Dai, C.; Hong, H.; Li, J.; Liu, Z.; Guo, Z.; Chen, X.; He, P.; et al. Heme Oxygenase-1 Ameliorates Oxidative Stress-Induced Endothelial Senescence via Regulating Endothelial Nitric Oxide Synthase Activation and Coupling. Aging 2018, 10, 1722–1744. [Google Scholar] [CrossRef]
- Wang, X.; Chen, L.; Wang, T.; Jiang, X.; Zhang, H.; Li, P.; Lv, B.; Gao, X. Ginsenoside Rg3 Antagonizes Adriamycin-Induced Cardiotoxicity by Improving Endothelial Dysfunction from Oxidative Stress via Upregulating the Nrf2-ARE Pathway through the Activation of Akt. Phytomedicine 2015, 22, 875–884. [Google Scholar] [CrossRef]
- Unsal, V.; Kolukcu, E.; Firat, F.; Gevrek, F. The Protective Effects of Sinapic Acid on Acute Renal Ischemia/Reperfusion Injury. Turk. J. Biochem. 2021, 46, 563–571. [Google Scholar] [CrossRef]
- Lee, J.H.; Parveen, A.; Do, M.H.; Lim, Y.; Shim, S.H.; Kim, S.Y. Lespedeza Cuneata Protects the Endothelial Dysfunction via ENOS Phosphorylation of PI3K/Akt Signaling Pathway in HUVECs. Phytomedicine 2018, 48, 1–9. [Google Scholar] [CrossRef]
- Förstermann, U.; Münzel, T. Endothelial Nitric Oxide Synthase in Vascular Disease: From Marvel to Menace. Circulation 2006, 113, 1708–1714. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, K.; Smith, R.S., Jr.; Hsieh, C.M.; Sun, J.; Chao, J.; Liao, J.K. Activation of the phosphatidylinositol 3-kinase/protein kinase Akt pathway mediates nitric oxide-induced endothelial cell migration and angiogenesis. Mol. Cell. Biol. 2003, 23, 5726–5737. [Google Scholar] [CrossRef]
- Anwar, M.A.; Samaha, A.A.; Ballan, S.; Saleh, A.I.; Iratni, R.; Eid, A.H. Salvia fruticosa induces Vasorelaxation in Rat Isolated Thoracic Aorta: Role of the PI3K/Akt/eNOS/NO/cGMP Signaling Pathway. Sci. Rep. 2017, 7, 686. [Google Scholar] [CrossRef]
- Chen, X.; Yao, F.; Song, J.; Fu, B.; Sun, G.; Song, X.; Fu, C.; Jiang, R.; Sun, L. Protective Effects of Phenolic Acid Extract from Ginseng on Vascular Endothelial Cell Injury Induced by Palmitate via Activation of PI3K/Akt/ENOS Pathway. J. Food Sci. 2020, 85, 576–581. [Google Scholar] [CrossRef]
- Yan, X.; Zhang, Q.-Y.; Zhang, Y.-L.; Han, X.; Guo, S.-B.; Li, H.-H. Gallic Acid Attenuates Angiotensin II-Induced Hypertension and Vascular Dysfunction by Inhibiting the Degradation of Endothelial Nitric Oxide Synthase. Front. Pharmacol. 2020, 11, 1121. [Google Scholar] [CrossRef]

| Name | Structure | Name | Structure |
|---|---|---|---|
| Benzoic acid | 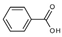 | Gallic acid | 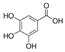 |
| Hydroxybenzoic acid | 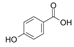 | Protocatechuic acid | 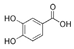 |
| Caffeic acid | 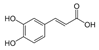 | Sinapic acid |  |
| Cinnamic acid | 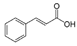 | Syringic acid | 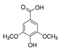 |
| Coumaric acid |  | Vanillic acid | 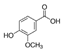 |
| Ferulic acid | 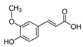 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, M.; Kim, H.J.; Heo, H.; Kim, M.; Jeon, Y.; Lee, H.; Lee, J. Comparison of the Antihypertensive Activity of Phenolic Acids. Molecules 2022, 27, 6185. https://doi.org/10.3390/molecules27196185
Yu M, Kim HJ, Heo H, Kim M, Jeon Y, Lee H, Lee J. Comparison of the Antihypertensive Activity of Phenolic Acids. Molecules. 2022; 27(19):6185. https://doi.org/10.3390/molecules27196185
Chicago/Turabian StyleYu, Myeongnam, Hyun Joo Kim, Huijin Heo, Minjun Kim, Yesol Jeon, Hana Lee, and Junsoo Lee. 2022. "Comparison of the Antihypertensive Activity of Phenolic Acids" Molecules 27, no. 19: 6185. https://doi.org/10.3390/molecules27196185
APA StyleYu, M., Kim, H. J., Heo, H., Kim, M., Jeon, Y., Lee, H., & Lee, J. (2022). Comparison of the Antihypertensive Activity of Phenolic Acids. Molecules, 27(19), 6185. https://doi.org/10.3390/molecules27196185







