Cranberry Ingestion Modulated Drug Transporters and Metabolizing Enzymes: Gefitinib Used as a Probe Substrate in Rats
Abstract
:1. Introduction
2. Results
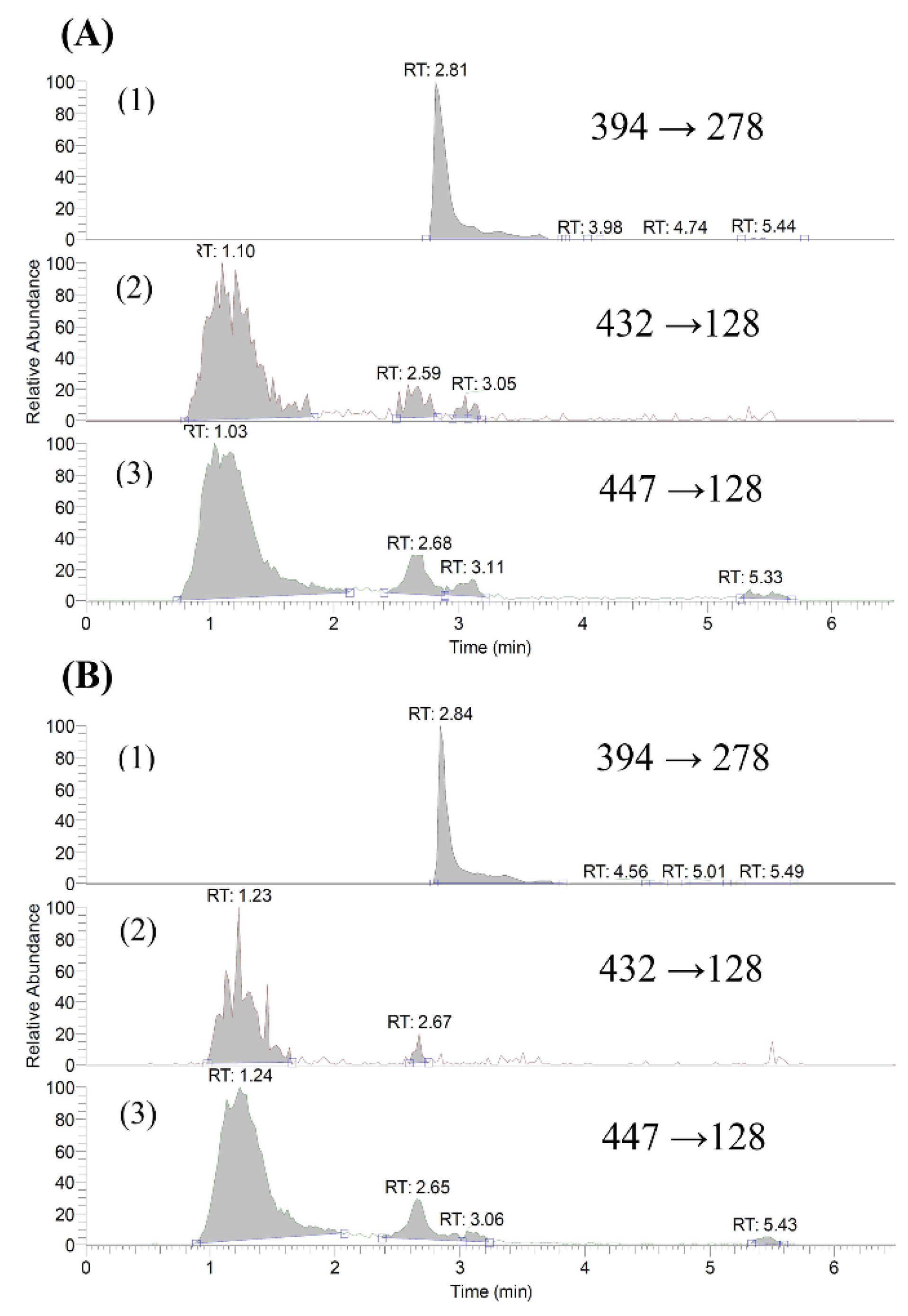
2.1. Quantitation of Gefitinib and O-Desmethyl Gefitinib in Serum
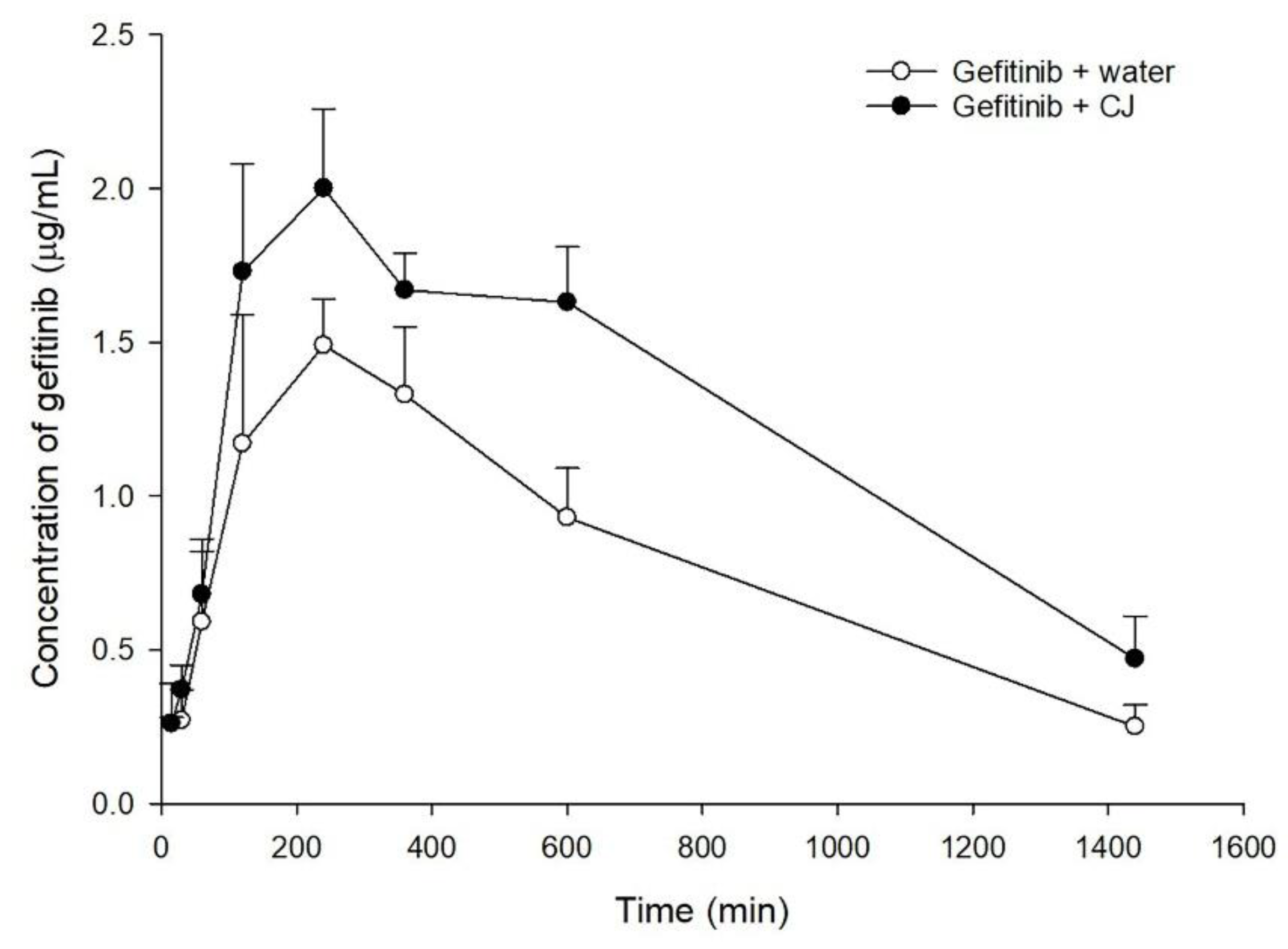
2.2. Effect of CJ Intake on Gefitinib Pharmacokinetics in Rats
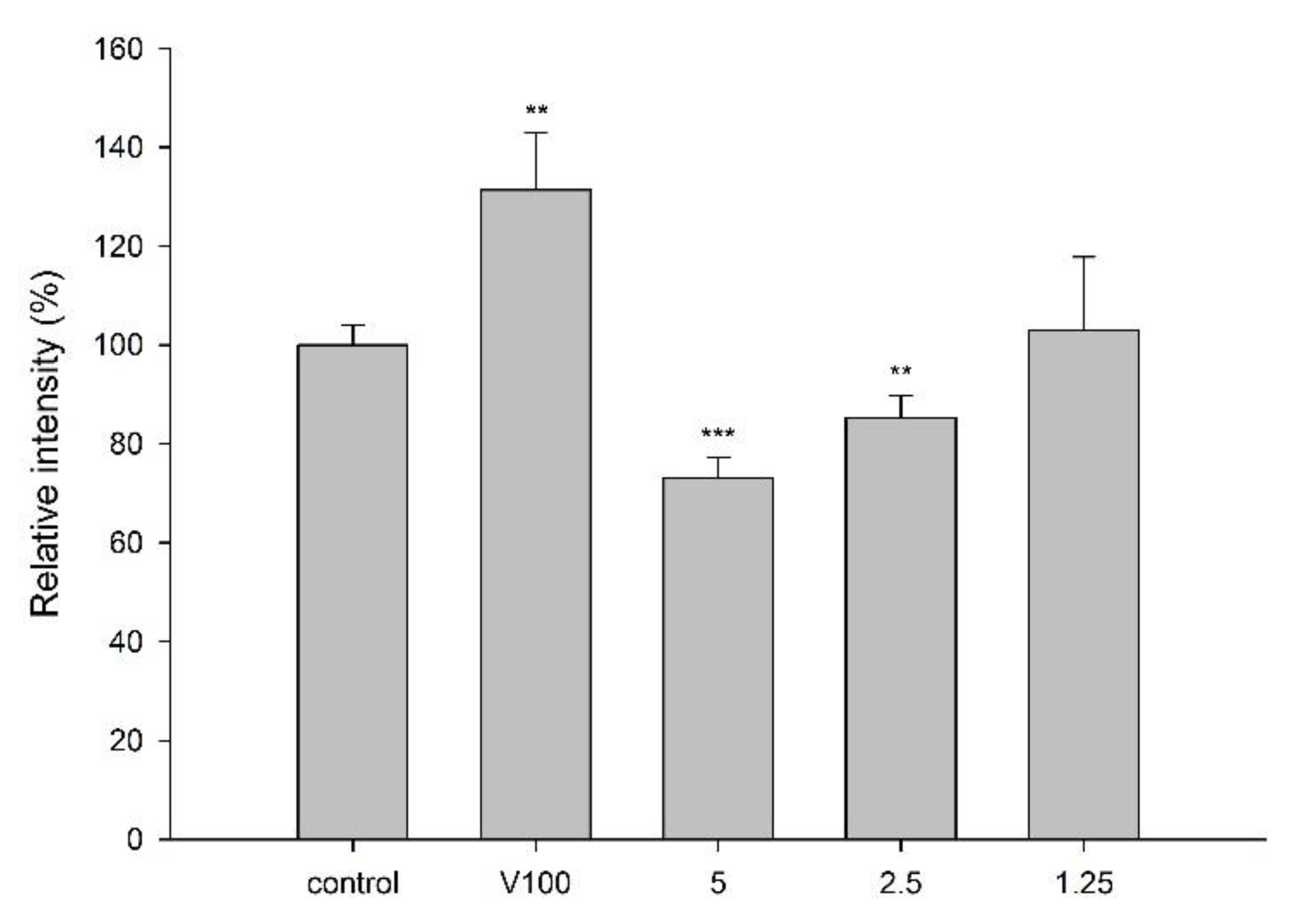
2.3. Effect of CJ on P-gp Activity
2.4. Effect of CM on CYP2D6 Activity
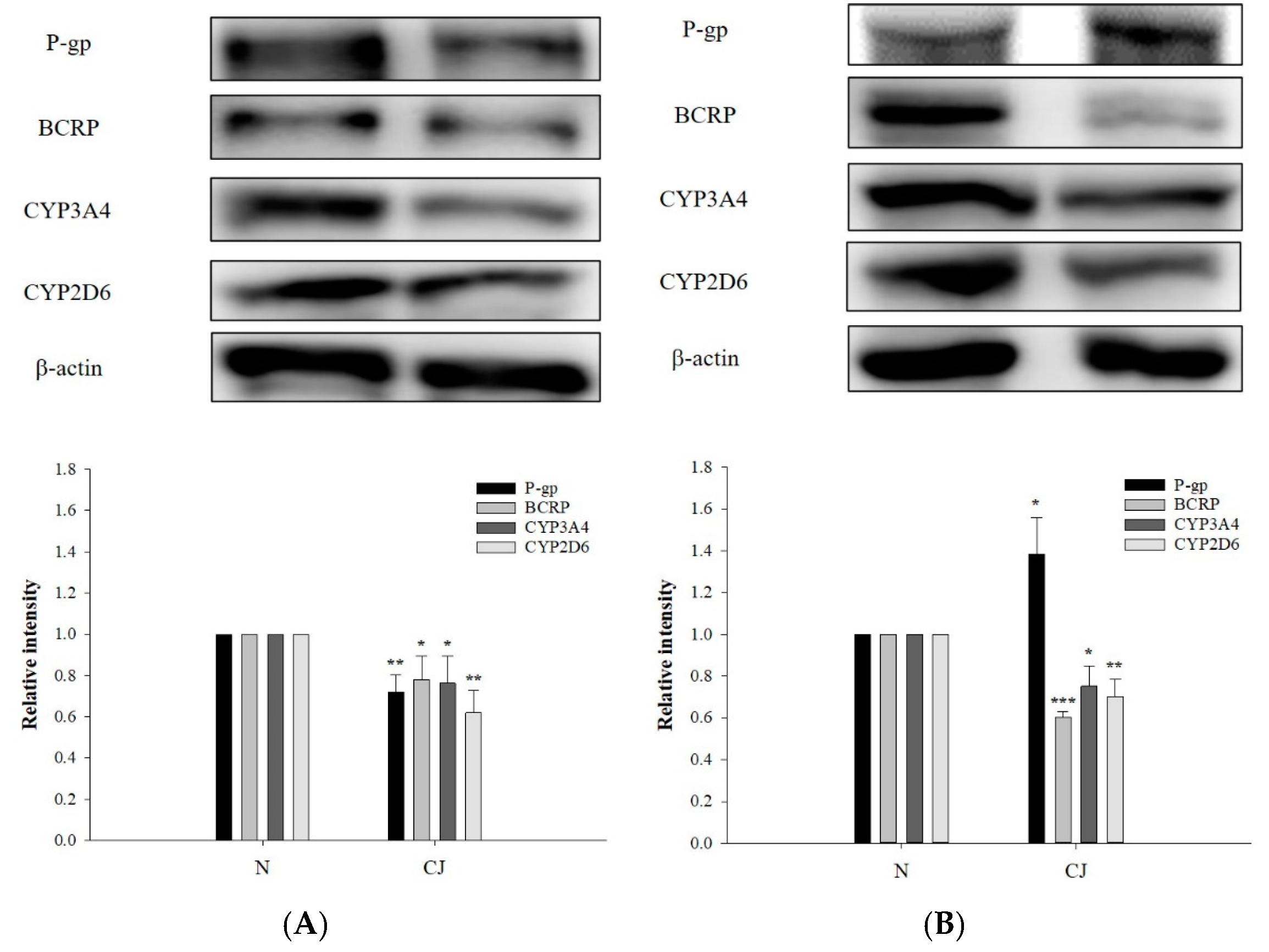
2.5. Effect of CJ Ingestion on the Protein Levels of P-gp, BCRP, CYP3A4 and CYP2D6
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Characterization of Polyphenols in CJ
4.3. Animals, Drug Administration and Blood Collection
4.4. Quantitation of Gefitinib and O-Desmethyl Gefitinib in Serum and Method Validation
4.5. Cell Line and Culture Conditions
4.6. Cell Viability Assay
4.7. Effect of CJ on P-gp Activity
4.8. Preparation of CM
4.9. Effect of CM on CYP2D6 Activity
4.10. Western Blot Analysis of P-gp, BCRP, CYP3A4 and CYP2D6 in Rat Enterocytes and Hepatocytes
4.11. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
Abbreviations
| ACN | acetonitrile |
| AUC | the area under the serum concentration-time curve |
| BCRP | breast cancer resistance protein |
| CJ | cranberry juice |
| CYP | cytochrome P450 |
| Cmax | peak blood concentration |
| CM | serum metabolites of cranberry |
| DMEM | Dulbecco’s modified Eagle medium |
| DMSO | dimethyl sulfoxide |
| ECL | chemiluminescence |
| FBS | fetal bovine serum |
| HBSS | Hank’s buffered salt solution |
| HPLC | high-performance liquid chromatography |
| MDCKII | Madin-Darby canine kidney type II cells |
| MRT | mean residence time |
| MTT | 3-(4′,5′-dimethylthiazol-2′-yl)-2,5-diphenyltetrazolium bromide |
| NEAA | nonessential amino acid |
| P-gp | P-glycoprotein |
| PSG | penicillin streptomycin and glutamine |
| R123 | rhodamine 123 |
| SDS | sodium dodecyl sulfate |
| TBST | Tris-buffered saline with 0.1% Tween® 20 detergent |
References
- Zhao, S.; Liu, H.; Gu, L. American cranberries and health benefits – An evolving story of 25 years. J. Sci. Food Agric. 2018, 100, 5111–5116. [Google Scholar] [CrossRef] [PubMed]
- Sihra, N.; Goodman, A.; Zakri, R.; Sahai, A.; Malde, S. Nonantibiotic prevention and management of recurrent urinary tract infection. Nat. Rev. Urol. 2018, 15, 750–776. [Google Scholar] [CrossRef] [PubMed]
- Katsargyris, A.; Tampaki, E.-C.; Giaginis, C.; Theocharis, S. Cranberry as promising natural source of potential anticancer agents: Current evidence and future perspectives. Anti-Cancer Agents Med. Chem. 2012, 12, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Neto, C.C.; Amoroso, J.W.; Liberty, A.M. Anticancer activities of cranberry phytochemicals: An update. Mol. Nutr. Food Res. 2008, 52, S18–S27. [Google Scholar] [CrossRef] [PubMed]
- Neto, C.C. Cranberry and blueberry: Evidence for protective effects against cancer and vascular diseases. Mol. Nutr. Food Res. 2007, 51, 652–664. [Google Scholar] [CrossRef] [PubMed]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.; Shenouda, S.M.; Chung, W.B.; Titas, M.; A Kluge, M.; Wang, N.; Palmisano, J.; E Milbury, P.; et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef] [PubMed]
- Vattem, D.A.; Ghaedian, R.; Shetty, K. Enhancing health benefits of berries through phenolic antioxidant enrichment: Focus on cranberry. Asia Pac. J. Clin. Nutr. 2005, 14, 120–130. [Google Scholar] [PubMed]
- Zhao, C.; Han, S.Y.; Li, P.P. Pharmacokinetics of gefitinib: Roles of drug metabolizing enzymes and transporters. Curr. Drug Deliv. 2017, 14, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Swaisland, H.; Laight, A.; Stafford, L.; Jones, H.; Morris, C.; Dane, A.; Yates, R.; Swaisland, M.H. Pharmacokinetics and Tolerability of the Orally Active Selective Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor ZD1839 in Healthy Volunteers. Clin. Pharmacokinet. 2001, 40, 297–306. [Google Scholar] [CrossRef]
- Scheffler, M.; Di Gion, P.; Doroshyenko, O.; Wolf, J.; Fuhr, U. Clinical pharmacokinetics of tyrosine kinase inhibitors: Focus on 4-anilinoquinazolines. Clin. Pharmacokinet. 2011, 50, 371–403. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Sane, R.; Gallardo, J.L.; Ohlfest, J.R.; Elmquist, W.F. Distribution of Gefitinib to the Brain Is Limited by P-glycoprotein (ABCB1) and Breast Cancer Resistance Protein (ABCG2)-Mediated Active Efflux. J. Pharmacol. Exp. Ther. 2010, 334, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, O.; Cascorbi, I. Polymorphisms of the drug transporters ABCB1, ABCG2, ABCC2 and ABCC3 and their impact on drug bioavailability and clinical relevance. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1337–1354. [Google Scholar] [CrossRef] [PubMed]
- Pick, A.; Müller, H.; Mayer, R.; Haenisch, B.; Pajeva, I.K.; Weigt, M.; Bönisch, H.; Müller, C.E.; Wiese, M. Structure–activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorg. Med. Chem. 2010, 19, 2090–2102. [Google Scholar] [CrossRef] [PubMed]
- Cooray, H.C.; Janvilisri, T.; van Veen, H.W.; Hladky, S.B.; Barrand, M.A. Interaction of the breast cancer resistance protein with plant polyphenols. Biochem. Biophys. Res. Commun. 2004, 317, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Zanger, U.M.; Schwab, M. Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther. 2013, 138, 103–141. [Google Scholar] [CrossRef] [PubMed]
- Johansson, I.; Ingelman-Sundberg, M. Genetic Polymorphism and Toxicology—With Emphasis on Cytochrome P450. Toxicol. Sci. 2010, 120, 1–13. [Google Scholar] [CrossRef]
- Ingelman-Sundberg, M. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): Clinical consequences, evolutionary aspects and functional diversity. Pharmacogenom. J. 2004, 5, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Mallhi, T.H.; Sarriff, A.; Adnan, A.S.; Khan, Y.H.; Qadir, M.I.; Hamzah, A.A.; Khan, A.H. Effect of Fruit/Vegetable-Drug Interactions on CYP450, OATP and p-Glycoprotein: A Systematic Review. Trop. J. Pharm. Res. 2015, 14, 1927. [Google Scholar] [CrossRef]
- Dreiseitel, A.; Schreier, P.; Oehme, A.; Locher, S.; Rogler, G.; Piberger, H.; Hajak, G.; Sand, P.G. Anthocyanins and anthocyanidins are poor inhibitors of CYP2D6. Methods Find. Exp. Clin. Pharmacol. 2009, 31, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Luca, S.V.; Macovei, I.; Bujor, A.; Miron, A.; Skalicka-Woźniak, K.; Aprotosoaie, A.C.; Trifan, A. Bioactivity of dietary polyphenols: The role of metabolites. Crit. Rev. Food Sci. Nutr. 2019, 60, 626–659. [Google Scholar] [CrossRef]
- Serreli, G.; Deiana, M. In vivo formed metabolites of polyphenols and their biological efficacy. Food Funct. 2019, 10, 6999–7021. [Google Scholar] [CrossRef] [PubMed]
- McKillop, D.; Hutchison, M.; Partridge, E.; Bushby, N.; Cooper, C.; Clarkson-Jones, J.; Herron, W.; Swaisland, H. Metabolic disposition of gefitinib, an epidermal growth factor receptor tyrosine kinase inhibitor, in rat, dog and man. Xenobiotica 2004, 34, 917–934. [Google Scholar] [CrossRef] [PubMed]
- McKillop, D.; Partridge, E.; Hutchison, M.; Rhead, S.; Parry, A.; Bardsley, J.; Woodman, H.; Swaisland, H. Pharmacokinetics of gefitinib, an epidermal growth factor receptor tyrosine kinase inhibitor, in rat and dog. Xenobiotica 2004, 34, 901–915. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-P.; Yang, M.-S.; Hsu, P.-W.; Lin, S.-P.; Hou, Y.-C. Bidirectional Influences of Cranberry on the Pharmacokinetics and Pharmacodynamics of Warfarin with Mechanism Elucidation. Nutrients 2021, 13, 3219. [Google Scholar] [CrossRef]
- Lee, C.A.; O’Connor, M.A.; Ritchie, T.K.; Galetin, A.; Cook, J.A.; Ragueneau-Majlessi, I.; Ellens, H.; Feng, B.; Taub, M.E.; Paine, M.F.; et al. Breast Cancer Resistance Protein (ABCG2) in Clinical Pharmacokinetics and Drug Interactions: Practical Recommendations for Clinical Victim and Perpetrator Drug-Drug Interaction Study Design. Drug Metab. Dispos. 2015, 43, 490–509. [Google Scholar] [CrossRef] [PubMed]
- Jani, M.; Temesszentandrási-Ambrus, C.; Magnan, R.; Jakab, K.T.; Beéry, E.; Zolnerciks, J.K.; Krajcsi, P. Structure and function of BCRP, a broad specificity transporter of xenobiotics and endobiotics. Arch. Toxicol. 2014, 88, 1205–1248. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, R.; Nascimento, Y.D.A.; Crespo, D. Safety aspects of protease inhibitors for chronic hepatitis C: Adverse events and drug-to-drug interactions. Braz. J. Infect. Dis. 2013, 17, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Chiba, K.; Ito, T.; Koue, T.; Sugiyama, Y. Prediction of interindividual variability in pharmacokinetics for CYP3A4 substrates in humans. Drug Metab. Pharmacokinet. 2010, 25, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Lynch, T.; Price, A. The effect of cytochrome P450 metabolism on drug response, interactions, and adverse effects. Am. Fam. Physician 2007, 76, 391–396. [Google Scholar]
- Gressier, F.; Verstuyft, C.; Hardy, P.; Becquemont, L.; Corruble, E. Response to CYP2D6 substrate antidepressants is predicted by a CYP2D6 composite phenotype based on genotype and comedications with CYP2D6 inhibitors. J. Neural Transm. 2014, 122, 35–42. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Yang, S.-Y.; Tsai, S.-Y.; Hou, Y.-C.; Chao, P.-D.L. Inductive modulation on P-glycoprotein and cytochrome 3A by resveratrol, a constituent of grapes. Food Chem. 2012, 133, 683–688. [Google Scholar] [CrossRef]
- Yang, M.-S.; Yu, C.-P.; Huang, C.-Y.; Chao, P.-D.L.; Lin, S.-P.; Hou, Y.-C. Aloe activated P-glycoprotein and CYP 3A: A study on the serum kinetics of aloe and its interaction with cyclosporine in rats. Food Funct. 2016, 8, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-P.; Huang, C.-Y.; Lin, S.-P.; Hou, Y.-C. Activation of P-glycoprotein and CYP 3A by Coptidis Rhizoma in vivo: Using cyclosporine as a probe substrate in rats. J. Food Drug Anal. 2018, 26, S125–S132. [Google Scholar] [CrossRef] [PubMed]

| Treatments | Gefitinib + Water | Gefitinib + CJ | |
|---|---|---|---|
| Parameters | |||
| Tmax (min) | 264.0 ± 44.9 | 216.0 ± 44.9 | |
| Cmax (μg/mL) | 1.8 ± 0.3 a | 2.3 ± 0.2 b | |
| (+28%) | |||
| AUC0-t (μg⋅min/mL) | 1170.5 ± 129.3 a | 1818.2 ± 144.8 b | |
| (+55%) | |||
| MRT (min) | 511.3 ± 40.1 | 543.0 ± 29.8 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, C.-P.; Tsai, P.-L.; Li, P.-Y.; Hsu, P.-W.; Lin, S.-P.; Lee Chao, P.-D.; Hou, Y.-C. Cranberry Ingestion Modulated Drug Transporters and Metabolizing Enzymes: Gefitinib Used as a Probe Substrate in Rats. Molecules 2022, 27, 5772. https://doi.org/10.3390/molecules27185772
Yu C-P, Tsai P-L, Li P-Y, Hsu P-W, Lin S-P, Lee Chao P-D, Hou Y-C. Cranberry Ingestion Modulated Drug Transporters and Metabolizing Enzymes: Gefitinib Used as a Probe Substrate in Rats. Molecules. 2022; 27(18):5772. https://doi.org/10.3390/molecules27185772
Chicago/Turabian StyleYu, Chung-Ping, Pei-Ling Tsai, Pei-Ying Li, Pei-Wen Hsu, Shiuan-Pey Lin, Pei-Dawn Lee Chao, and Yu-Chi Hou. 2022. "Cranberry Ingestion Modulated Drug Transporters and Metabolizing Enzymes: Gefitinib Used as a Probe Substrate in Rats" Molecules 27, no. 18: 5772. https://doi.org/10.3390/molecules27185772
APA StyleYu, C.-P., Tsai, P.-L., Li, P.-Y., Hsu, P.-W., Lin, S.-P., Lee Chao, P.-D., & Hou, Y.-C. (2022). Cranberry Ingestion Modulated Drug Transporters and Metabolizing Enzymes: Gefitinib Used as a Probe Substrate in Rats. Molecules, 27(18), 5772. https://doi.org/10.3390/molecules27185772





