Mechanistic Insights into the Pharmacological Significance of Silymarin
Abstract
1. Introduction
2. Pharmacological Aspects of Silymarin
2.1. Hepatoprotective Activity
2.2. Anti-Diabetic Activity
| Study Model | Dose/Concentration Used | Possible Target Site/Mechanism of Action | Reference |
|---|---|---|---|
| Obesity-induced insulin resistance model and HepG 2 cells | 30 mg/kg/day p.o. for one month |
| [86] |
| HFD model | 30 mg/kg/day p.o. for one month |
| [75] |
| HFD-induced insulin resistance | 30 and 60 mg/kg p.o. |
| [87] |
| HFD-induced insulin resistance model and HEK293T cells | 40 μg/mL 50 µM |
| [79] |
| Pancreatectomy model | 200 mg/kg p.o |
| [88] |
| STZ-induced diabetes and INS1 cells | 50 μg/mL 2.5–100 µM |
| [89] |
| STZ- and HFD-induced diabetes | 100 and 300 mg/kg p.o. |
| [90] |
| STZ-induced diabetes | 60 and 120 mg/kg/day p.o. for 2 months |
| [76] |
| STZ-induced diabetes | 80 mg/kg p.o. for 21 days |
| [77] |
2.3. Anti-Cancer Activity
2.4. Neuroprotective Activity
| Pharmacological Activity | Study Model | Dose/Concentration Used | Possible Target Site/Mechanism of Action | References |
|---|---|---|---|---|
| Neuroprotective | Lipopolysaccharide (LPS)-induced neuroinflammatory impairment | 25–100 mg/kg |
| [183] |
| Docetaxel-induced central and peripheral neurotoxicities | 25 and 50 mg/kg |
| [208] | |
| STZ-induced diabetic neuropathy | 30, 60 mg/kg/day p.o. |
| [182] | |
| — | 30–300 g/mL |
| [195] | |
| Manganese-induced neurotoxicity | 100 mg/kg/day i.p. |
| [209] | |
| Acrylamide-induced cerebellar damage | 160 mg/kg |
| [210] | |
| Ischemic surgery | 200 mg/kg |
| [211] | |
| Kainic acid (KA)-induced excitotoxicity | 50–100 mg/kg |
| [188] | |
| Middle cerebral artery occlusion | — |
| [212] | |
| Anti-Alzheimer | Aβ1–42-induced Alzheimer’s | 70 and 140 mg/kg p.o. for 4 weeks |
| [201] |
| APP transgenic mice and PC12 cells | 0–100 µM |
| [202] | |
| APP/PS1 transgenic mice | 2–200 mg/kg/day |
| [200] | |
| Scopolamine-induced dementia | 200–800 mg/kg p.o. for 2 weeks |
| [199] | |
| Aβ1–42-induced Alzheimer | 25–100 mg/kg |
| [206] | |
| Aluminum chloride (AlCl3)-induced Alzheimer’s | 34 mg |
| [198] | |
| Aβ25–35-induced Alzheimer’s | 25–100 mg/kg |
| [181] | |
| Aβ25–35-induce oxidative stress damage in HT-22 cells | — |
| [205] | |
| Anti-Parkinson | Caenorhabditis elegans transgenic model | 24.12 µg/mL |
| [193] |
| MPTP-induced parkinsonism | 40 mg/kg i.p. for 2 weeks |
| [197] | |
| 6-OHDA-induced neurodegeneration and parkinsonism | 100 and 200 mg/kg i.p. |
| [192] | |
| 6-OHDA-induced neurodegeneration and parkinsonism | 100, 200 and 300 mg/kg, i.p. for 5 days |
| [185] | |
| MPTP-induced parkinsonism | 20–400 mg/kg, i.p. |
| [191] | |
| Anti-depression | — | 5–200 mg/kg |
| [213] |
| Olfactory bulbectomized (OBX) technique | 100–200 mg/kg |
| [184] | |
| Reserpine-induced depression | 0–400 mg/kg |
| [214] | |
| Aβ1–42-induced Alzheimer’s | 25–100 mg/kg |
| [215] |
2.5. Cardioprotective and Anti-Hypertensive Activity
2.6. Anti-Viral Activity
| Study Model | Dose/Concentration Used | Finding/Possible Mechanism of Action | Reference |
|---|---|---|---|
| CCl4-induced cardiomyopathy | 200 mg/kg/day p.o. for 21 days |
| [220] |
| Ischemia reperfusion-induced myocardial infarction | 100–500 mg/kg p.o. for one week |
| [221] |
| Acrolein-induced cardio toxicity | 25–100 mg/kg p.o. |
| [222] |
| Isoproterenol-treated rat cardiac myocytes | 0–0.7 mM |
| [254] |
| Perfused adult rat heart model and H9c2 cells | 0.01–10 µM |
| [223] |
| Doxorubicin-induced cardio toxicity and hepatotoxicity | 60 mg/kg p.o. for 12 days |
| [255] |
| DOCA salt-induced hypertension | 300 mg/kg and 500 mg/kg, p. o. for 4 weeks |
| [224] |
| Fructose-induced hypertension | 300 mg/kg and 500 mg/kg, p. o. for 6 weeks |
| [225] |
| Type of Viral Infection | Study Model | Dose/Concentration Used | Possible Target Site/Mechanism of Action | Reference |
|---|---|---|---|---|
| Chikungunya virus | Vero and BHK-21 cells | 50 μg/mL |
| [249] |
| HCV | Huh 7 cells | — |
| [226] |
| HepG2 and Huh 7 cells | — |
| [227] | |
| Huh 7 and PBM cells | 20–200 μg/mL |
| [232] | |
| Huh7.5 cells | 10.4–150 µM |
| [234] | |
| — | 1–1000 µM |
| [256] | |
| uPA+/+/SCID+/+ chimeric mice model | 61.5, 265 and 469 mg/kg i.v. for 14 days |
| [233] | |
| Influenza virus | MDCK cells | 100 μg/mL |
| [242] |
| MDCK, A549 and Vero cells Viral infection of BALB/c mice | 25 mg/kg/day |
| [243] | |
| HIV | PBMC and CEM-T4 cells | 50–500 µM |
| [253] |
| TZM-bl, PBMC and CEM cells | 40–324 µM |
| [252] | |
| Mayaro virus | HepG2 cells | 3.125–1400 μg/mL |
| [246] |
| HBV | HepG2-NTCP-C4 cells | 0–200 μM |
| [240] |
| Herpes virus | Vero cell | 0–125 μg/mL |
| [251] |
| SARS-CoV-2 | Human umbilical vein endothelial cells | 5–25 µM |
| [241] |
| — | 1–100 µM |
| [257] |
2.7. Photoprotection and Dermal Applications
| Pharmacological Activity | Study Model | Dose/Concentration Used | Possible Target Site/Mechanism of Action | Reference |
|---|---|---|---|---|
| Photo protective | UV exposure | 0.1–0.2 mg/mL/kg topically |
| [282] |
| UV exposure | — |
| [274] | |
| UV exposure in HaCaT cells | 75 μm |
| [281] | |
| UVB-induced skin damage in human dermal fibroblasts | 1.6–100 μM |
| [273] | |
| XPA-deficient mice, XPA-deficient and XPA-proficient human fibroblasts and normal human epidermal keratinocytes | 10 and 20 µg/mL |
| [267] | |
| JB6 cells and mouse skin | 100 μm |
| [268] | |
| Human dermal fibroblasts | 100 μm |
| [266] | |
| SKH-1 hairless mouse | 9 mg topically |
| [269] | |
| SKH-1 hairless mice skin | 9 mg topically |
| [272] | |
| Anti-alopecia | Human dermal papilla cells | 0–200 μM |
| [283] |
| Wound healing | Human fibroblast cells | 4.5–36 µg/mL |
| [284] |
| Rat wound model with full-thickness excision | 2% ointment containing 500 mg silymarin |
| [285] | |
| Rat wound model full-thickness cutaneous defect | 6–12 mg/mL |
| [286] | |
| Normal human dermal fibroblasts | — |
| [279] | |
| Anti-aging | — | 0.01–2.5 g/L |
| [287] |
| Disease | No. of Patients | Dose; Duration | Add on Therapy | Study Outcomes | Reference |
|---|---|---|---|---|---|
| Diabetes | 40 | 140 mg tid p.o.; 90 days | — |
| [80] |
| 40 | 140 mg tid p.o.; 45 days | — |
| [288] | |
| 40 | 420 mg tid p.o.; 45 days | — |
| [289] | |
| 85 (diagnosed with Type 1 diabetes) | 105 mg bid p.o.; 6 months | Berberis aristata 588 mg |
| [290] | |
| 69 | 1000 mg/day p.o. | Berberine 210 mg/day |
| [291] | |
| Dyslipidemia | 139 | 105 mg bid p.o.; 6 months | Berberis aristata 500 mg and Monacolin K 10 mg |
| [38] |
| 137 | 105 mg bid p.o.; 6 months | Berberis aristata 588 mg |
| [37] | |
| 105 | 105 mg bid p.o.; 3 months | Berberis aristata 588 mg |
| [36] | |
| Melasma (skin disorder) | 96 | 7 and 14 mg/mL cream bid topically; 4 weeks | — |
| [282] |
| Acne | 20 | 1% seed oil cream bid topically | — |
| [292] |
| 56 | — | N-acetylcysteine and Selenium |
| [293] | |
| Hepatocellular carcinoma | 40 | — | — |
| [172] |
| Cisplatin-induced nephrotoxicity | 60 | 140 mg bid p.o.; 7 days | — |
| [116] |
| 86 | 140 mg tid p.o.; 21 days | — |
| [115] | |
| Capecitabine-induced hand-foot syndrome | 40 (diagnosed with G.I.T. cancer | 1% gel bid topically; 9 weeks | Capecitabine |
| [114] |
| Radiotherapy-induced mucositis | 27 (Diagnosed with head and neck cancer) | 420 mg/ day p.o.; 6 weeks | — |
| [117] |
| Radiation-induced dermatitis | 40 (Diagnosed with breast cancer) | 1% gel bid topically; 5 weeks | — |
| [118] |
| NAFLD | 81 | 280 mg bid p.o.; 90 days | Vitamin C 120 mg, Vitamin E 40 mg, Coenzyme Q10 20 mg and Selenomethionine 83 µg |
| [294] |
| 66 | 140 mg/day p.o. | — |
| [295] | |
| 36 | 540 mg bid. p.o.; 3 months | Vitamin E |
| [296] | |
| 30 | 188 mg p.o.; 6 months | Vitamin E and Phospholipids |
| [297] | |
| 179 | 94 mg bid. p.o.; 12 months | Phosphatidylcholine 194 mg and Vitamin E 89 mg |
| [69] | |
| 150 | 303 mg bid. p.o.; 6 months | Vitamin D 10 mg and Vitamin E 15 mg |
| [61] | |
| 62 | 303 mg bid. p.o.; 6 months | Vitamin D 10 mg and Vitamin E 15 mg |
| [298] | |
| NASH | 64 | 210 mg/day p.o.; 8 weeks | — |
| [299] |
| 100 | 700 mg tid; 48 weeks | — |
| [300] | |
| 116 | 420 and 700 mg tid, p.o.; 48 weeks | — |
| [301] | |
| Multiple sclerosis therapy-induce liver damage | 54 (diagnosed with remitting relapsing multiple sclerosis) | 420 mg, p.o.; 6 months | IFNβ |
| [302] |
| Chronic HCV infection | 64 | 47 mg p.o; 12 months | Ribavirin+Peg–IFN and Vitamin E+ phospholipids |
| [236] |
| 26 | 5, 10, 15, and 20 mg/kg/day i.v.; 7 and 14 days | Ribavirin+ Peg–IFN |
| [229] | |
| 154 | 420 and 700 mg tid p.o.; 24 weeks | — |
| [56] | |
| HIV/HCV coinfection | 16 | 20 mg/kg/day i.v.; 14 days | Ribavirin+ Peg–IFN and Telaprevir for 12 weeks |
| [260] |
| Anti TB drug-induced hepatotoxicity | 55 | 140 mg tid p.o.; 8 weeks | Rifampicin 10 mg/kg/day, Isoniazid 5 mg/kg/day, Ethambutol 15 mg/kg/day or Pyrazinamide 25 mg/kg/day |
| [303] |
| 70 | 140 mg tid p.o.; 2 weeks | Isoniazid 5 mg/kg, Pyrazinamide 20 mg/kg, Ethambutol 15 mg/kg and/or Rifampin 10 mg/kg |
| [304] | |
| 108 | 140 mg bid p.o.; 8 weeks | Isoniazid, Pyrazinamide, Ethambutol and/or Rifampin |
| [305] | |
| Beta thalassemia | 49 | 140 mg tid p.o.; 9 months | Desferrioxamine |
| [306] |
| 25 | 420 mg/ day p.o.; 12 weeks | Desferrioxamine 40 mg/kg/day |
| [307] | |
| 40 | 140 mg tid p.o.; 6 months | Deferasirox |
| [308] | |
| 22 | 420 mg/ day p.o.; 6 months | Desferrioxamine |
| [309] | |
| 80 | 420 mg/ day p.o.; 9 months | Deferiprone |
| [310] |
3. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
List of Abbreviations
| α-SMA | α-Smooth muscle actin |
| γGT | Gamma-glutamyl transferase |
| AChE | Acetylcholinesterase |
| ALP | Alkaline phosphate |
| AP-1 | Activated protein-1 |
| APP | Amyloid precursor protein |
| Aβ | Amyloid β |
| BDNF | brain-derived neurotrophic factors |
| Bid | Two times a day |
| CAT | Catalase |
| CD | Cyclin D |
| CDCA3 | Cell division cycle-associated 3 protein |
| CDK | Cyclin-dependent kinase |
| CK1α | Casein kinase 1α |
| CK-MB | Creatine kinase-MB |
| COL-α1 | Collagen α1 |
| CPDs | Cyclobutane pyrimidine dimers |
| CREB | cAMP response element-binding protein |
| CRP | C-reactive protein |
| DM | Diabetes Mellitus |
| DRP1 | Dynamin-related protein 1 |
| ERK1/2 | Extracellular signal-regulated kinase |
| FADD | Fas-associated death domain |
| FAS | Fatty acid synthase |
| FBS | Fasting blood sugar |
| FoxM1 | Forkhead box M1 |
| G6Pase | Glucose-6-phosphatase |
| GADD45α | Growth arrest and DNA damage-inducible protein α |
| GM-CSF | Granulocyte-macrophage colony stimulating factor |
| GLP-1 | Glucagon-like peptide 1 |
| GSK3β | Glycogen synthase kinase-3β |
| HBV | Hepatitis B virus |
| HCV | Hepatitis C virus |
| HFD | High-fat diet |
| HIF 1α | Hypoxia inducible factor 1α |
| HIV | Human immunodeficiency virus |
| HOMA-IR | Homeostatic model assessment of insulin resistance |
| HSC | Hepatic stellate cells |
| ICAM-1 | Intercellular adhesion molecule-1 |
| IFN-γ | Interferon γ |
| IKK | IkB kinase |
| iNOS | Inducible nitric oxide synthase |
| IRS1 | Insulin receptor substrate 1 |
| JAK | Janus Kinase |
| JNK | c-Jun NH2-terminal kinase |
| KLF 6 | Krueppel-like factor 6 |
| LDH | Lactate dehydrogenase |
| LRP6 | Lipoprotein receptor-related protein 6 |
| MAO-B | Monoamine oxidase-B |
| MAPK | Mitogen-activated protein kinases |
| MCD diet | Methionine–choline deficient diet |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MDA | Malondialdehyde |
| MMP | Matrix metalloproteinases |
| MPO | Myeloperoxidase |
| mTOR | Mammalian target of rapamycin |
| NAFLD | Non-alcoholic fatty liver disease |
| NASH | Non-alcoholic steatohepatitis |
| NF-ĸB | Nuclear factor kappa B |
| NGF | Nerve growth factor |
| NLRP3 | NLR family pyrin domain containing 3 |
| NO | Nitric oxide |
| NPR | Nucleotide excision repair |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| NUSAP1 | Nucleolar and spindle-associated protein 1 |
| PARP | Poly adenosine diphosphate-ribose polymerase |
| PI3K | Phosphatidylinositol 3-kinase |
| PKB/AKt | Protein kinase B |
| ROS | Reactive oxygen species |
| SAPK | Stress-activated protein kinase |
| SARS-CoV2 | Severe acute respiratory syndrome coronavirus 2 |
| SGOT | Serum glutamic-oxalacetic transaminase |
| SGPT | Serum glutamic-pyruvic transaminase |
| SIRT1 | Sirtuin-1 |
| SOD | Superoxide dismutase |
| SREBP-1c | Sterol regulatory element-binding protein 1c |
| STAT | Signal transducers and activators of transcription |
| STZ | Streoptozotocin |
| TBARS | Thiobarbituric acid reactive substances |
| TG | Triglyceride |
| TGF-β | Transforming growth factor β |
| Tid | Three times a day |
| TIMP-1 | Tissue inhibitor of metalloproteinases 1 |
| TNF-α | Tumor necrosis factor α |
| TOPBP1 | Topoisomerase 2 binding protein 1 |
| TRAIL | TNF-related apoptosis inducing ligand |
| TrkB | Tropomyosin receptor kinase B |
| UVB | Ultraviolet-B |
| VEGF | Vascular endothelial growth factor |
References
- Nikam, P.H.; Kareparamban, J.; Jadhav, A.; Kadam, V. Future Trends in Standardization of Herbal Drugs. J. Appl. Pharm. Sci. 2012, 2, 38–44. [Google Scholar] [CrossRef]
- Saller, R.; Melzer, J.; Reichling, J.; Brignoli, R.; Meier, R. An Updated Systematic Review of the Pharmacology of Silymarin. Forsch. Komplement. 2007, 14, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S. Phytotherapeutic Properties of Milk Thistle Seeds: An Overview. J. Adv. Pharm. Educ. Res. 2011, 1, 69–79. [Google Scholar]
- Das, S.K.; Mukherjee, S.; Vasudevan, D.M. Medicinal Properties of Milk Thistle with Special Reference to Silymarin An Overview. Nat. Prod. Radiance 2008, 7, 182–192. [Google Scholar]
- Ghosh, A.; Ghosh, T.; Jain, S. Silymarin—A Review on the Pharmacodynamics and Bioavailability Enhancement Approaches. J. Pharm. Sci. Technol. 2010, 2, 348–355. [Google Scholar]
- Schuppan, D.; Jia, J.I.D.; Brinkhaus, B.; Hahn, E.G. Herbal Products for Liver Diseases: A Therapeutic Challenge for the New Millennium. Hepatology 1999, 30, 1099–1104. [Google Scholar] [CrossRef]
- Ramasamy, K.; Agarwal, R. Multitargeted Therapy of Cancer by Silymarin. Cancer Lett. 2008, 269, 352–362. [Google Scholar] [CrossRef]
- Sharma, A.; Puri, V.; Kakkar, V.; Singh, I. Formulation and Evaluation of Silymarin-Loaded Chitosan-Montmorilloite Microbeads for the Potential Treatment of Gastric Ulcers. J. Funct. Biomater. 2018, 9, 52. [Google Scholar] [CrossRef]
- Chang, L.W.; Hou, M.L.; Tsai, T.H. Silymarin in Liposomes and Ethosomes: Pharmacokinetics and Tissue Distribution in Free-Moving Rats by High-Performance Liquid Chromatography-Tandem Mass Spectrometry. J. Agric. Food Chem. 2014, 62, 11657–11665. [Google Scholar] [CrossRef]
- Flora, K.; Hahn, M.; Rosen, H.; Benner, K. Milk Thistle (Silybum marianum) for the Therapy of Liver Disease. Am. J. Gastroenterol. 1998, 93, 139–143. [Google Scholar] [CrossRef]
- Gazak, R.; Walterova, D.; Kren, V. Silybin and Silymarin-New and Emerging Applications in Medicine. Curr. Med. Chem. 2007, 14, 315–338. [Google Scholar] [CrossRef] [PubMed]
- Neha; Jaggi, A.; Singh, N. Silymarin and Its Role in Chronic Diseases. In Advances in Experimental Medicine and Biology; Springer: Berlin/Hamburg, Germany, 2016; Volume 929, pp. 25–44. [Google Scholar]
- Palit, P.; Mukhopadhyay, A.; Chattopadhyay, D. Phyto-Pharmacological Perspective of Silymarin: A Potential Prophylactic or Therapeutic Agent for COVID-19, Based on Its Promising Immunomodulatory, Anti-Coagulant and Anti-Viral Property. Phyther. Res. 2021, 35, 4246–4257. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Izzo, A.A.; Milić, N.; Cicala, C.; Santini, A.; Capasso, R. Milk Thistle (Silybum marianum): A Concise Overview on Its Chemistry, Pharmacological, and Nutraceutical Uses in Liver Diseases. Phyther. Res. 2018, 32, 2202–2213. [Google Scholar] [CrossRef]
- Lv, Y.; Gao, S.; Xu, S.; Du, G.; Zhou, J.; Chen, J. Spatial Organization of Silybin Biosynthesis in Milk Thistle [Silybum marianum (L.) Gaertn]. Plant J. 2017, 92, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M. Silybin, a Major Bioactive Component of Milk Thistle (Silybum marianum L. Gaernt.)—Chemistry, Bioavailability, and Metabolism. Molecules 2017, 22, 1942. [Google Scholar] [CrossRef]
- Dixit, N.; Baboota, S.; Kohli, K.; Ahmad, S.; Ali, J. Silymarin: A Review of Pharmacological Aspects and Bioavailability Enhancement Approaches. Indian J. Pharmacol. 2007, 39, 172–179. [Google Scholar] [CrossRef]
- Elwekeel, A.; Elfishawy, A.; Abouzid, S. Silymarin Content in Silybum marianum Fruits at Different Maturity Stages. J. Med. Plants Res. 2013, 7, 1665–1669. [Google Scholar] [CrossRef]
- Javed, S.; Kohli, K.; Ali, M. Reassessing Bioavailability of Silymarin. Altern. Med. Rev. 2011, 16, 239–249. [Google Scholar]
- Reddy, K.R. Silymarin for the treatment of chronic liver disease. Gastroenterol. Hepatol. (N. Y.) 2007, 3, 825–826. [Google Scholar]
- Kaur, M.; Agarwal, R. Silymarin and Epithelial Cancer Chemoprevention: How Close We Are to Bedside? Toxicol. Appl. Pharmacol. 2007, 224, 350–359. [Google Scholar] [CrossRef]
- Javed, S.; Ahsan, W.; Kohli, K. Pharmacological Influences of Natural Products as Bioenhancers of Silymarin against Carbon Tetrachloride-Induced Hepatotoxicity in Rats. Clin. Phytoscience 2018, 4, 18. [Google Scholar] [CrossRef]
- Home-Nano Silymarin OIC NEW: Nano Silymarin OIC NEW. Available online: https://nanosilymarin.vn/en/ (accessed on 5 July 2022).
- Zhang, Z.; Li, X.; Sang, S.; McClements, D.J.; Chen, L.; Long, J.; Jiao, A.; Wang, J.; Jin, Z.; Qiu, C. A Review of Nanostructured Delivery Systems for the Encapsulation, Protection, and Delivery of Silymarin: An Emerging Nutraceutical. Food. Res. Int. 2022, 156, 111314. [Google Scholar] [CrossRef]
- Wadhwa, K.; Kadian, V.; Puri, V.; Bhardwaj, B.Y.; Sharma, A.; Pahwa, R.; Rao, R.; Gupta, M.; Singh, I. New Insights into Quercetin Nanoformulations for Topical Delivery. Phytomedicine Plus 2022, 2, 100257. [Google Scholar] [CrossRef]
- Saini, V.; Singh, A.; Shukla, R.; Jain, K.; Yadav, A.K. Silymarin-Encapsulated Xanthan Gum–Stabilized Selenium Nanocarriers for Enhanced Activity against Amyloid Fibril Cytotoxicity. AAPS PharmSciTech 2022, 23, 125. [Google Scholar] [CrossRef]
- Elfaky, M.A.; Sirwi, A.; Ismail, S.H.; Awad, H.H.; Gad, S.S. Hepatoprotective Effect of Silver Nanoparticles at Two Different Particle Sizes: Comparative Study with and without Silymarin. Curr. Issues Mol. Biol. 2022, 44, 2923–2938. [Google Scholar] [CrossRef]
- Abdullah, A.S.; El Sayed, I.E.T.; El-Torgoman, A.M.A.; Kalam, A.; Wageh, S.; Kamel, M.A. Green Synthesis of Silymarin–Chitosan Nanoparticles as a New Nano Formulation with Enhanced Anti-Fibrotic Effects against Liver Fibrosis. Int. J. Mol. Sci. 2022, 23, 5420. [Google Scholar] [CrossRef]
- Patel, P.; Raval, M.; Manvar, A.; Airao, V.; Bhatt, V.; Shah, P. Lung Cancer Targeting Efficiency of Silibinin Loaded Poly Caprolactone/Pluronic F68 Inhalable Nanoparticles: In Vitro and In Vivo Study. PLoS ONE 2022, 17, e0267257. [Google Scholar] [CrossRef]
- Iqbal, J.; Andleeb, A.; Ashraf, H.; Meer, B.; Mehmood, A.; Jan, H.; Zaman, G.; Nadeem, M.; Drouet, S.; Fazal, H.; et al. Potential Antimicrobial, Antidiabetic, Catalytic, Antioxidant and ROS/RNS Inhibitory Activities of Silybum marianum Mediated Biosynthesized Copper Oxide Nanoparticles. RSC Adv. 2022, 12, 14069–14083. [Google Scholar] [CrossRef]
- Staroverov, S.A.; Kozlov, S.V.; Fomin, A.S.; Gabalov, K.P.; Khanadeev, V.A.; Soldatov, D.A.; Domnitsky, I.Y.; Dykman, L.A.; Akchurin, S.V.; Guliy, O.I. Synthesis of Silymarin−selenium Nanoparticle Conjugate and Examination of Its Biological Activity in Vitro. ADMET DMPK 2021, 9, 255–266. [Google Scholar] [CrossRef]
- Silymarin|Side Effects|Dosage|Precautions|Medicine. Available online: https://www.medicoverhospitals.in/medicine/silymarin (accessed on 5 July 2022).
- Han, Y.; Guo, D.; Chen, Y.; Chen, Y.; Tan, Z.R.; Zhou, H.H. Effect of Silymarin on the Pharmacokinetics of Losartan and Its Active Metabolite E-3174 in Healthy Chinese Volunteers. Eur. J. Clin. Pharmacol. 2009, 65, 585–591. [Google Scholar] [CrossRef]
- Moltó, J.; Valle, M.; Miranda, C.; Cedeño, S.; Negredo, E.; Clotet, B. Effect of Milk Thistle on the Pharmacokinetics of Darunavir-Ritonavir in HIV-Infected Patients. Antimicrob. Agents Chemother. 2012, 56, 2837–2841. [Google Scholar] [CrossRef]
- Almazroo, O.A.; Miah, M.K.; Venkataramanan, R. Drug Metabolism in the Liver. Clin. Liver Dis. 2017, 21, 1–20. [Google Scholar] [CrossRef]
- Derosa, G.; Bonaventura, A.; Bianchi, L.; Romano, D.; D’angelo, A.; Fogari, E.; Maffioli, P. Berberis Aristata/Silybum marianum Fixed Combination on Lipid Profile and Insulin Secretion in Dyslipidemic Patients. Expert Opin. Biol. Ther. 2013, 13, 1495–1506. [Google Scholar] [CrossRef]
- Derosa, G.; Romano, D.; D’Angelo, A.; Maffioli, P. Berberis Aristata Combined with Silybum marianum on Lipid Profile in Patients Not Tolerating Statins at High Doses. Atherosclerosis 2015, 239, 87–92. [Google Scholar] [CrossRef]
- Derosa, G.; D’Angelo, A.; Romano, D.; Maffioli, P. Effects of a Combination of Berberis Aristata, Silybum marianum and Monacolin on Lipid Profile in Subjects at Low Cardiovascular Risk; A Double-Blind, Randomized, Placebo-Controlled Trial. Int. J. Mol. Sci. 2017, 18, 343. [Google Scholar] [CrossRef]
- Mengesha, T.; Sekaran, N.G.; Mehare, T. Hepatoprotective Effect of Silymarin on Fructose Induced Nonalcoholic Fatty Liver Disease in Male Albino Wistar Rats. BMC Complement. Med. Ther. 2021, 21, 104. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.M.; Al-Kahtani, M.A.; El-Kersh, M.A.; Al-Omair, M.A. Free Radical-Scavenging, Anti-Inflammatory/Anti-Fibrotic and Hepatoprotective Actions of Taurine and Silymarin against CCl4 Induced Rat Liver Damage. PLoS ONE 2015, 10, e0144509. [Google Scholar] [CrossRef]
- Tsai, J.H.; Liu, J.Y.; Wu, T.T.; Ho, P.C.; Huang, C.Y.; Shyu, J.C.; Hsieh, Y.S.; Tsai, C.C.; Liu, Y.C. Effects of Silymarin on the Resolution of Liver Fibrosis Induced by Carbon Tetrachloride in Rats. J. Viral Hepat. 2008, 15, 508–514. [Google Scholar] [CrossRef]
- Sokar, S.S.; El-Sayad, M.E.S.; Ghoneim, M.E.S.; Shebl, A.M. Combination of Sitagliptin and Silymarin Ameliorates Liver Fibrosis Induced by Carbon Tetrachloride in Rats. Biomed. Pharmacother. 2017, 89, 98–107. [Google Scholar] [CrossRef]
- Keshavarz-Maleki, R.; Shalmani, A.A.; Gholami, M.; Sabzevari, S.; Rahimzadegan, M.; Jeivad, F.; Sabzevari, O. The Ameliorative Effect of Monomethyl Fumarate and Silymarin against Valproic Acid Induced Hepatotoxicity in Rats. Pharm. Chem. J. 2021, 55, 240–245. [Google Scholar] [CrossRef]
- Chen, I.S.; Chen, Y.C.; Chou, C.H.; Chuang, R.F.; Sheen, L.Y.; Chiu, C.H. Hepatoprotection of Silymarin against Thioacetamide-Induced Chronic Liver Fibrosis. J. Sci. Food Agric. 2012, 92, 1441–1447. [Google Scholar] [CrossRef]
- Heidarian, E.; Nouri, A. Hepatoprotective Effects of Silymarin against Diclofenac-Induced Liver Toxicity in Male Rats Based on Biochemical Parameters and Histological Study. Arch. Physiol. Biochem. 2021, 127, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Mahli, A.; Koch, A.; Czech, B.; Peterburs, P.; Lechner, A.; Haunschild, J.; Müller, M.; Hellerbrand, C. Hepatoprotective Effect of Oral Application of a Silymarin Extract in Carbon Tetrachloride-Induced Hepatotoxicity in Rats. Clin. Phytoscience 2015, 1, 5. [Google Scholar] [CrossRef]
- Shaker, E.; Mahmoud, H.; Mnaa, S. Silymarin, the Antioxidant Component and Silybum marianum Extracts Prevent Liver Damage. Food Chem. Toxicol. 2010, 48, 803–806. [Google Scholar] [CrossRef]
- Freitag, A.F.; Cardia, G.F.E.; Da Rocha, B.A.; Aguiar, R.P.; Silva-Comar, F.M.D.S.; Spironello, R.A.; Grespan, R.; Caparroz-Assef, S.M.; Bersani-Amado, C.A.; Cuman, R.K.N. Hepatoprotective Effect of Silymarin (Silybum marianum) on Hepatotoxicity Induced by Acetaminophen in Spontaneously Hypertensive Rats. Evid.-Based Complement. Altern. Med. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Haddad, P.S.; Haddad, Y.; Vallerand, D.; Brault, A. Antioxidant and Hepatoprotective Effects of Silibinin in a Rat Model of Nonalcoholic Steatohepatitis. Evid.-Based Complement. Altern. Med. 2011, 2011, 1–10. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Jiang, N.; Yang, J.; Tu, J.; Zhou, Y.; Xiao, X.; Dong, Y. Silybum marianum Oil Attenuates Hepatic Steatosis and Oxidative Stress in High Fat Diet-Fed Mice. Biomed. Pharmacother. 2018, 100, 191–197. [Google Scholar] [CrossRef]
- Ou, Q.; Weng, Y.; Wang, S.; Zhao, Y.; Zhang, F.; Zhou, J.; Wu, X. Silybin Alleviates Hepatic Steatosis and Fibrosis in NASH Mice by Inhibiting Oxidative Stress and Involvement with the NF-κB Pathway. Dig. Dis. Sci. 2018, 63, 3398–3408. [Google Scholar] [CrossRef] [PubMed]
- Aghazadeh, S.; Amini, R.; Yazdanparast, R.; Ghaffari, S.H. Anti-Apoptotic and Anti-Inflammatory Effects of Silybum marianum in Treatment of Experimental Steatohepatitis. Exp. Toxicol. Pathol. 2011, 63, 569–574. [Google Scholar] [CrossRef]
- Kim, S.H.; Oh, D.S.; Oh, J.Y.; Son, T.G.; Yuk, D.Y.; Jung, Y.S. Silymarin Prevents Restraint Stress-Induced Acute Liver Injury by Ameliorating Oxidative Stress and Reducing Inflammatory Response. Molecules 2016, 21, 443. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Xu, D.; She, L.; Wang, Z.; Yang, N.; Sun, R.; Zhang, Y.; Yan, C.; Wei, Q.; Aa, J.; et al. Silybin Inhibits NLRP3 Inflammasome Assembly through the NAD+/SIRT2 Pathway in Mice with Nonalcoholic Fatty Liver Disease. FASEB J. 2018, 32, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Alaca, N.; Özbeyli, D.; Uslu, S.; Şahin, H.H.; Yiǧittörk, G.; Kurtel, H.; Öktem, G.; Yeǧen, B.Ç. Treatment with Milk Thistle Extract (Silybum marianum), Ursodeoxycholic Acid, or Their Combination Attenuates Cholestatic Liver Injury in Rats: Role of the Hepatic Stem Cells. Turkish J. Gastroenterol. 2017, 28, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.W.; Navarro, V.J.; Afdhal, N.; Belle, S.H.; Wahed, A.S.; Hawke, R.L.; Doo, E.; Meyers, C.M.; Reddy, K.R. Effect of Silymarin (Milk Thistle) on Liver Disease in Patients with Chronic Hepatitis C Unsuccessfully Treated with Interferon Therapy: A Randomized Controlled Trial. JAMA-J. Am. Med. Assoc. 2012, 308, 274–282. [Google Scholar] [CrossRef]
- Clichici, S.; Olteanu, D.; Nagy, A.L.; Oros, A.; Filip, A.; Mircea, P.A. Silymarin Inhibits the Progression of Fibrosis in the Early Stages of Liver Injury in CCl4-Treated Rats. J. Med. Food 2015, 18, 290–298. [Google Scholar] [CrossRef]
- Raghu, R.; Karthikeyan, S. Zidovudine and Isoniazid Induced Liver Toxicity and Oxidative Stress: Evaluation of Mitigating Properties of Silibinin. Environ. Toxicol. Pharmacol. 2016, 46, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Di Rosa, M.; Nicoletti, F.; Malaguarnera, L. Molecular Mechanisms Involved in NAFLD Progression. J. Mol. Med. 2009, 87, 679–695. [Google Scholar] [CrossRef] [PubMed]
- Shaarawy, S.M.; Tohamy, A.A.; Elgendy, S.M.; Abd Elmageed, Z.Y.; Bahnasy, A.; Mohamed, M.S.; Kandil, E.; Matrougui, K. Protective Effects of Garlic and Silymarin on NDEA-Induced Rats Hepatotoxicity. Int. J. Biol. Sci. 2009, 5, 549–557. [Google Scholar] [CrossRef]
- Federico, A.; Dallio, M.; Masarone, M.; Gravina, A.G.; Di Sarno, R.; Tuccillo, C.; Cossiga, V.; Lama, S.; Stiuso, P.; Morisco, F.; et al. Evaluation of the Effect Derived from Silybin with Vitamin D and Vitamin E Administration on Clinical, Metabolic, Endothelial Dysfunction, Oxidative Stress Parameters, and Serological Worsening Markers in Nonalcoholic Fatty Liver Disease Patients. Oxid. Med. Cell. Longev. 2019, 2019, 1–12. [Google Scholar] [CrossRef]
- Gharagozloo, M.; Jafari, S.; Esmaeil, N.; Javid, E.N.; Bagherpour, B.; Rezaei, A. Immunosuppressive Effect of Silymarin on Mitogen-Activated Protein Kinase Signalling Pathway: The Impact on T Cell Proliferation and Cytokine Production. Basic Clin. Pharmacol. Toxicol. 2013, 113, 209–214. [Google Scholar] [CrossRef]
- Sinha, K.; Das, J.; Pal, P.B.; Sil, P.C. Oxidative Stress: The Mitochondria-Dependent and Mitochondria-Independent Pathways of Apoptosis. Arch. Toxicol. 2013, 87, 1157–1180. [Google Scholar] [CrossRef]
- Kisseleva, T.; Brenner, D. Molecular and Cellular Mechanisms of Liver Fibrosis and Its Regression. Nat. Rev. Gastroenterol. Hepatol. 2020, 18, 151–166. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, E.C.; Weyhenmeyer, R.; Weiner, O.H. Effects of Silibinin and of a Synthetic Analogue on Isolated Rat Hepatic Stellate Cells and Myofibroblasts. Arzneimittelforschung 1997, 47, 1383–1387. [Google Scholar] [PubMed]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte Chemoattractant Protein-1 (MCP-1): An Overview. J. Interf. Cytokine Res. 2009, 29, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Tsuruta, S.; Nakamuta, M.; Enjoji, M.; Kotoh, K.; Hiasa, K.; Egashira, K.; Nawata, H. Anti-Monocyte Chemoattractant Protein-1 Gene Therapy Prevents Dimethylnitrosamine-Induced Hepatic Fibrosis in Rats. Int. J. Mol. Med. 2004, 14, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Hemmann, S.; Graf, J.; Roderfeld, M.; Roeb, E. Expression of MMPs and TIMPs in Liver Fibrosis-a Systematic Review with Special Emphasis on Anti-Fibrotic Strategies. J. Hepatol. 2007, 46, 955–975. [Google Scholar] [CrossRef]
- Loguercio, C.; Andreone, P.; Brisc, C.; Brisc, M.C.; Bugianesi, E.; Chiaramonte, M.; Cursaro, C.; Danila, M.; De Sio, I.; Floreani, A.; et al. Silybin Combined with Phosphatidylcholine and Vitamin e in Patients with Nonalcoholic Fatty Liver Disease: A Randomized Controlled Trial. Free Radic. Biol. Med. 2012, 52, 1658–1665. [Google Scholar] [CrossRef]
- Dehmlow, C.; Erhard, J.; De Groot, H. Inhibition of Kupffer Cell Functions as an Explanation for the Hepatoprotective Properties of Silibinin. Hepatology 1996, 23, 749–754. [Google Scholar] [CrossRef]
- Sonnenbichler, J.; Goldbero, M.; Hane, L.; Madubunyi, I.; Vogl, S.; Zetl, I. Stimulatory Effect of Silibinin on the DNA Synthesis in Partially Hepatectomized Rat Livers: Non-Response in Hepatoma and Other Malign Cell Lines. Biochem. Pharmacol. 1986, 35, 538–541. [Google Scholar] [CrossRef]
- Yormaz, S.; Bulbuloglu, E.; Kurutas, E.B.; Ciralik, H.; Yuzbasioglu, M.F.; Yildiz, H.; Coskuner, I.; Silay, E.; Kantarceken, B.; Goksu, M.; et al. The Comparison of the Effects of Hepatic Regeneration after Partial Hepatectomy, Silybum Marinaum, Propofol, N-Acetylcysteine and Vitamin E on Liver. Bratislava Med. J. 2012, 113, 145–151. [Google Scholar] [CrossRef]
- Thakur, V.; Choudhary, M.; Garg, A.; Choudhary, N.; Jangra, A.; Budhwar, V. Evaluation of a Hydroalcoholic Extract of the Leaves from the Endangered Medicinal Plant Gloriosa Superba Linn. (Colchicaceae) for Its Potential Anti-Diabetic Effect. Arch. Med. 2015, 7, 1–8. [Google Scholar]
- Sharma, S.; Wadhwa, K.; Choudhary, M.; Budhwar, V. Ethnopharmacological Perspectives of Glucokinase Activators in the Treatment of Diabetes Mellitus. Nat. Prod. Res. 2021, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Meng, R.; Huang, B.; Shen, S.; Bi, Y.; Zhu, D. Silymarin Alleviates Hepatic Oxidative Stress and Protects against Metabolic Disorders in High-Fat Diet-Fed Mice. Free Radic. Res. 2016, 50, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, R.; Karimi, J.; Khodadadi, I.; Tayebinia, H.; Kheiripour, N.; Hashemnia, M.; Goli, F. Silymarin Ameliorates Expression of Urotensin II (U-II) and Its Receptor (UTR) and Attenuates Toxic Oxidative Stress in the Heart of Rats with Type 2 Diabetes. Biomed. Pharmacother. 2018, 101, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Alabdan, M.A. Silymarin Ameliorates Metabolic Risk Factors and Protects against Cardiac Apoptosis in Streptozotocin-Induced Diabetic Rats. Biomed. Biotechnol. 2015, 3, 20–27. [Google Scholar] [CrossRef]
- Abu-zaiton, A.S. Evaluating the Effect of Silybum marianum Extract on Blood Glucose, Liver and Kidney Functions in Diabetic Rats. Adv. Stud. Biol. 2013, 5, 447–454. [Google Scholar] [CrossRef]
- Gu, M.; Zhao, P.; Huang, J.; Zhao, Y.; Wang, Y.; Li, Y.; Li, Y.; Fan, S.; Ma, Y.M.; Tong, Q.; et al. Silymarin Ameliorates Metabolic Dysfunction Associated with Diet-Induced Obesity via Activation of Farnesyl X Receptor. Front. Pharmacol. 2016, 7, 345. [Google Scholar] [CrossRef]
- Talaat Elgarf, A.; Maher Mahdy, M.; Sabri, N.A. Effect of Silymarin Supplementation on Glycemic Control, Lipid Profile and Insulin Resistance in Patients with Type 2 Diabetes Mellitus. Int. J. Adv. Res. 2015, 3, 812–821. [Google Scholar]
- Numan, A.T.; Hadi, N.A.; Sh Mohammed, N.; Hussain, S.A. Use of Silymarine as Adjuvant in Type 1 Diabetes Mellitus Patients Poorly Controlled with Insulin. J. Fac. Med. Baghdad 2010, 52, 75–79. [Google Scholar] [CrossRef]
- Velussi, M.; Cernigoi, A.M.; Ariella, D.M.; Dapas, F.; Caffau, C.; Zilli, M. Long-Term (12 Months) Treatment with an Anti-Oxidant Drug (Silymarin) Is Effective on Hyperinsulinemia, Exogenous Insulin Need and Malondialdehyde Levels in Cirrhotic Diabetic Patients. J. Hepatol. 1997, 26, 871–879. [Google Scholar] [CrossRef]
- Huseini, H.F.; Larijani, B.; Heshmat, R.; Fakhrzadeh, H.; Radjabipour, B.; Toliat, T.; Raza, M. The Efficacy of Silybum marianum (L.) Gaertn. (Silymarin) in the Treatment of Type II Diabetes: A Randomized, Double-Blind, Placebo-Controlled, Clinical Trial. Phyther. Res. 2006, 20, 1036–1039. [Google Scholar] [CrossRef]
- Hussain, S.A.R. Silymarin as an Adjunct to Glibenclamide Therapy Improves Long-Term and Postprandial Glycemic Control and Body Mass Index in Type 2 Diabetes. J. Med. Food 2007, 10, 543–547. [Google Scholar] [CrossRef]
- Guigas, B.; Naboulsi, R.; Villanueva, G.R.; Taleux, N.; Lopez-Novoa, J.M.; Leverve, X.M.; El-Mir, M.Y. The Flavonoid Silibinin Decreases Glucose-6-Phosphate Hydrolysis in Perifused Rat Hepatocytes by an Inhibitory Effect on Glucose-6-Phosphatase. Cell. Physiol. Biochem. 2007, 20, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Huang, B.; Jing, Y.; Shen, S.; Feng, W.; Wang, W.; Meng, R.; Zhu, D. Silymarin Ameliorates the Disordered Glucose Metabolism of Mice with Diet-Induced Obesity by Activating the Hepatic SIRT1 Pathway. Cell. Signal. 2021, 84, 110023. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wang, S.; Wang, Y.; Zhu, T. Silymarin Improved Diet-Induced Liver Damage and Insulin Resistance by Decreasing Inflammation in Mice. Pharm. Biol. 2016, 54, 2995–3000. [Google Scholar] [CrossRef] [PubMed]
- Soto, C.; Raya, L.; Juárez, J.; Pérez, J.; González, I. Effect of Silymarin in Pdx-1 Expression and the Proliferation of Pancreatic β-Cells in a Pancreatectomy Model. Phytomedicine 2014, 21, 233–239. [Google Scholar] [CrossRef]
- Qin, N.; Hu, X.; Li, S.; Wang, J.; Li, Z.; Li, D.; Xu, F.; Gao, M.; Hua, H. Hypoglycemic Effect of Silychristin A from Silybum marianum Fruit via Protecting Pancreatic Islet β Cells from Oxidative Damage and Inhibiting α-Glucosidase Activity In Vitro and in Rats with Type 1 Diabetes. J. Funct. Foods 2017, 38, 168–179. [Google Scholar] [CrossRef]
- Xu, F.; Yang, J.; Negishi, H.; Sun, Y.; Li, D.; Zhang, X.; Hayashi, T.; Gao, M.; Ikeda, K.; Ikejima, T. Silibinin Decreases Hepatic Glucose Production through the Activation of Gut–Brain–Liver Axis in Diabetic Rats. Food Funct. 2018, 9, 4926–4935. [Google Scholar] [CrossRef]
- Amniattalab, A.; Malekinejad, H.; Rezabakhsh, A.; Rokhsartalab-Azar, S.; Alizade-Fanalou, S. Silymarin: A Novel Natural Agent to Restore Defective Pancreatic β Cells in Streptozotocin (STZ)-Induced Diabetic Rats. Iran. J. Pharm. Res. 2016, 15, 493–500. [Google Scholar] [CrossRef]
- Malekinejad, H.; Rezabakhsh, A.; Rahmani, F.; Hobbenaghi, R. Silymarin Regulates the Cytochrome P450 3A2 and Glutathione Peroxides in the Liver of Streptozotocin-Induced Diabetic Rats. Phytomedicine 2012, 19, 583–590. [Google Scholar] [CrossRef]
- Kim, E.J.; Kim, J.; Lee, M.Y.; Sudhanva, M.S.; Devakumar, S.; Jeon, Y.J. Silymarin Inhibits Cytokine-Stimulated Pancreatic Beta Cells by Blocking the ERK1/2 Pathway. Biomol. Ther. 2014, 22, 282–287. [Google Scholar] [CrossRef]
- Soto, C.; Raya, L.; Pérez, J.; González, I.; Pérez, S. Silymarin Induces Expression of Pancreatic Nkx6.1 Transcription Factor and β-Cells Neogenesis in a Pancreatectomy Model. Molecules 2014, 19, 4654–4668. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Yang, Y.R.Y.; Mo, Z.J.; Ding, Y.; Jiang, W.J. Silibinin Improves Palmitate-Induced Insulin Resistance in C2C12 Myotubes by Attenuating IRS-1/PI3K/AKt Pathway Inhibition. Braz. J. Med. Biol. Res. 2015, 48, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Sun, Y.; Xu, F.; Liu, W.; Hayashi, T.; Onodera, S.; Tashiro, S.I.; Ikejima, T. Involvement of Estrogen Receptors in Silibinin Protection of Pancreatic β-Cells from TNFα- or IL-1β-Induced Cytotoxicity. Biomed. Pharmacother. 2018, 102, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yang, J.; Liu, W.; Yao, G.; Xu, F.; Hayashi, T.; Onodera, S.; Ikejima, T. Attenuating Effect of Silibinin on Palmitic Acid-Induced Apoptosis and Mitochondrial Dysfunction in Pancreatic β-Cells Is Mediated by Estrogen Receptor Alpha. Mol. Cell. Biochem. 2019, 460, 81–92. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, X.; Zhang, L.; Yan, T.; Wu, B.; Xu, F.; Jia, Y. Silychristin A Activates Nrf2-HO-1/SOD2 Pathway to Reduce Apoptosis and Improve GLP-1 Production through Upregulation of Estrogen Receptor α in GLUTag Cells. Eur. J. Pharmacol. 2020, 881, 173236. [Google Scholar] [CrossRef]
- Stolf, A.M.; Cardoso, C.C.; Acco, A. Effects of Silymarin on Diabetes Mellitus Complications: A Review. Phyther. Res. 2017, 31, 366–374. [Google Scholar] [CrossRef]
- Tuorkey, M.J.; El-Desouki, N.I.; Kamel, R.A. Cytoprotective Effect of Silymarin against Diabetes-Induced Cardiomyocyte Apoptosis in Diabetic Rats. Biomed. Environ. Sci. 2015, 28, 36–43. [Google Scholar] [CrossRef]
- Meng, S.; Yang, F.; Wang, Y.; Qin, Y.; Xian, H.; Che, H.; Wang, L. Silymarin Ameliorates Diabetic Cardiomyopathy via Inhibiting TGF-Β1/Smad Signaling. Cell Biol. Int. 2019, 43, 65–72. [Google Scholar] [CrossRef]
- Sheela, N.; Jose, M.A.; Sathyamurthy, D.; Kumar, B.N. Effect of Silymarin on Streptozotocin-Nicotinamide-Induced Type 2 Diabetic Nephropathy in Rats. Iran. J. Kidney Dis. 2013, 7, 117–123. [Google Scholar]
- Vessal, G.; Akmali, M.; Najafi, P.; Moein, M.R.; Sagheb, M.M. Silymarin and Milk Thistle Extract May Prevent the Progression of Diabetic Nephropathy in Streptozotocin-Induced Diabetic Rats. Ren. Fail. 2010, 32, 733–739. [Google Scholar] [CrossRef]
- Guzel, S.; Sahinogullari, Z.U.; Canacankatan, N.; Antmen, S.E.; Kibar, D.; Coskun Yilmaz, B. Potential Renoprotective Effects of Silymarin against Vancomycin-Induced Nephrotoxicity in Rats. Drug Chem. Toxicol. 2020, 43, 630–636. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, L.; Yang, T. Silymarin Nanoliposomes Attenuate Renal Injury on Diabetic Nephropathy Rats via Co-Suppressing TGF-β/Smad and JAK2/STAT3/SOCS1 Pathway. Life Sci. 2021, 271, 119197. [Google Scholar] [CrossRef]
- Fallahzadeh, M.K.; Dormanesh, B.; Sagheb, M.M.; Roozbeh, J.; Vessal, G.; Pakfetrat, M.; Daneshbod, Y.; Kamali-Sarvestani, E.; Lankarani, K.B. Effect of Addition of Silymarin to Renin-Angiotensin System Inhibitors on Proteinuria in Type 2 Diabetic Patients with Overt Nephropathy: A Randomized, Double-Blind, Placebo-Controlled Trial. Am. J. Kidney Dis. 2012, 60, 896–903. [Google Scholar] [CrossRef]
- Zhang, H.T.; Shi, K.; Baskota, A.; Zhou, F.L.; Chen, Y.X.; Tian, H.M. Silybin Reduces Obliterated Retinal Capillaries in Experimental Diabetic Retinopathy in Rats. Eur. J. Pharmacol. 2014, 740, 233–239. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Mattiuzzi, C.; Lippi, G. Current Cancer Epidemiology. J. Epidemiol. Glob. Health 2019, 9, 217–222. [Google Scholar] [CrossRef]
- Elsayed, E.A.; Sharaf-Eldin, M.A.; Wadaan, M. In Vitro Evaluation of Cytotoxic Activities of Essential Oil from Moringa oleifera Seeds on HeLa, HepG2, MCF-7, CACO-2 and L929 Cell Lines. Asian Pacific J. Cancer Prev. 2015, 16, 4671–4675. [Google Scholar] [CrossRef]
- Liang, C.; Pan, H.; Li, H.; Zhao, Y.; Feng, Y. In Vitro Anticancer Activity and Cytotoxicity Screening of Phytochemical Extracts from Selected Traditional Chinese Medicinal Plants. J. Balk. Union Oncol. 2017, 22, 543–551. [Google Scholar]
- Wang, X.; Zhang, Z.; Wu, S.C. Health Benefits of Silybum marianum: Phytochemistry, Pharmacology, and Applications. J. Agric. Food Chem. 2020, 68, 11644–11664. [Google Scholar] [CrossRef]
- Hosseinabadi, T.; Lorigooini, Z.; Tabarzad, M.; Salehi, B.; Rodrigues, C.F.; Martins, N.; Sharifi-Rad, J. Silymarin Antiproliferative and Apoptotic Effects: Insights into Its Clinical Impact in Various Types of Cancer. Phyther. Res. 2019, 33, 2849–2861. [Google Scholar] [CrossRef]
- Elyasi, S.; Shojaee, F.S.R.; Allahyari, A.; Karimi, G. Topical Silymarin Administration for Prevention of Capecitabine-Induced Hand–Foot Syndrome: A Randomized, Double-Blinded, Placebo-Controlled Clinical Tria. Phyther. Res. 2017, 31, 1323–1329. [Google Scholar] [CrossRef]
- Shahbazi, F.; Sadighi, S.; Dashti-Khavidaki, S.; Shahi, F.; Mirzania, M.; Abdollahi, A.; Ghahremani, M.H. Effect of Silymarin Administration on Cisplatin Nephrotoxicity: Report from a Pilot, Randomized, Double-Blinded, Placebo-Controlled Clinical Trial. Phyther. Res. 2015, 29, 1046–1053. [Google Scholar] [CrossRef]
- Momeni, A.; Hajigholami, A.; Geshnizjani, S.; Kheiri, S. Effect of Silymarin in the Prevention of Cisplatin Nephrotoxicity, a Clinical Trial Study. J. Clin. Diagn. Res. 2015, 9, 11–13. [Google Scholar] [CrossRef]
- Elyasi, S.; Hosseini, S.; Niazi Moghadam, M.R.; Aledavood, S.A.; Karimi, G. Effect of Oral Silymarin Administration on Prevention of Radiotherapy Induced Mucositis: A Randomized, Double-Blinded, Placebo-Controlled Clinical Trial. Phyther. Res. 2016, 30, 1879–1885. [Google Scholar] [CrossRef]
- Karbasforooshan, H.; Hosseini, S.; Elyasi, S.; Fani Pakdel, A.; Karimi, G. Topical Silymarin Administration for Prevention of Acute Radiodermatitis in Breast Cancer Patients: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Phyther. Res. 2019, 33, 379–386. [Google Scholar] [CrossRef]
- Becker-Schiebe, M.; Mengs, U.; Schaefer, M.; Bulitta, M.; Hoffmann, W. Topical Use of a Silymarin-Based Preparation to Prevent Radiodermatitis: Results of a Prospective Study in Breast Cancer Patients. Strahlenther. Onkol. 2011, 187, 485–491. [Google Scholar] [CrossRef]
- Imai-Sumida, M.; Chiyomaru, T.; Majid, S.; Saini, S.; Nip, H.; Dahiya, R.; Tanaka, Y.; Yamamura, S. Silibinin Suppresses Bladder Cancer through Down-Regulation of Actin Cytoskeleton and PI3K/Akt Signaling Pathways. Oncotarget 2017, 8, 92032–92042. [Google Scholar] [CrossRef]
- Kim, S.H.; Choo, G.-S.; Yoo, E.S.; Woo, J.S.; Lee, J.H.; Han, S.H.; Jung, S.H.; Kim, H.J.; Jung, J.Y. Silymarin Inhibits Proliferation of Human Breast Cancer Cells via Regulation of the MAPK Signaling Pathway and Induction of Apoptosis. Oncol. Lett. 2021, 21, 492. [Google Scholar] [CrossRef]
- Permana, M.Y.; Soediro, T.M.; Louisa, M. Silymarin Increases the Sensitivity of Breast Cancer Cells to Doxorubicin in Doxorubicin-Induced MCF-7 Cells by Inhibiting Breast Cancer Resistance Protein Expression. J. Phys. Conf. Ser. 2018, 1073, 032055. [Google Scholar] [CrossRef]
- Forghani, P.; Khorramizadeh, M.R.; Waller, E.K. Silibinin Inhibits Accumulation of Myeloid-Derived Suppressor Cells and Tumor Growth of Murine Breast Cancer. Cancer Med. 2014, 3, 215–224. [Google Scholar] [CrossRef]
- Si, L.; Fu, J.; Liu, W.; Hayashi, T.; Nie, Y.; Mizuno, K.; Hattori, S.; Fujisaki, H.; Onodera, S.; Ikejima, T. Silibinin Inhibits Migration and Invasion of Breast Cancer MDA-MB-231 Cells through Induction of Mitochondrial Fusion. Mol. Cell. Biochem. 2020, 463, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Dastpeyman, M.; Motamed, N.; Azadmanesh, K.; Mostafavi, E.; Kia, V.; Jahanian-Najafabadi, A.; Shokrgozar, M.A. Inhibition of Silibinin on Migration and Adhesion Capacity of Human Highly Metastatic Breast Cancer Cell Line, MDA-MB-231, by Evaluation of Β1-Integrin and Downstream Molecules, Cdc42, Raf-1 and D4GDI. Med. Oncol. 2012, 29, 2512–2518. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.J.; Darvin, P.; Kang, D.Y.; Sp, N.; Joung, Y.H.; Park, J.H.; Kim, S.J.; Yang, Y.M. Silibinin Downregulates MMP2 Expression via Jak2/STAT3 Pathway and Inhibits the Migration and Invasive Potential in MDA-MB-231 Cells. Oncol. Rep. 2017, 37, 3270–3278. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.O.; Jeong, Y.J.; Im, H.G.; Kim, C.H.; Chang, Y.C.; Lee, I.S. Silibinin Suppresses PMA-Induced MMP-9 Expression by Blocking the AP-1 Activation via MAPK Signaling Pathways in MCF-7 Human Breast Carcinoma Cells. Biochem. Biophys. Res. Commun. 2007, 354, 165–171. [Google Scholar] [CrossRef]
- Kim, S.; Choi, J.H.; Lim, H.I.; Lee, S.K.; Kim, W.W.; Kim, J.S.; Kim, J.H.; Choe, J.H.; Yang, J.H.; Nam, S.J.; et al. Silibinin Prevents TPA-Induced MMP-9 Expression and VEGF Secretion by Inactivation of the Raf/MEK/ERK Pathway in MCF-7 Human Breast Cancer Cells. Phytomedicine 2009, 16, 573–580. [Google Scholar] [CrossRef]
- Lu, W.; Lin, C.; King, T.D.; Chen, H.; Reynolds, R.C.; Li, Y. Silibinin Inhibits Wnt/β-Catenin Signaling by Suppressing Wnt Co-Receptor LRP6 Expression in Human Prostate and Breast Cancer Cells. Cell. Signal. 2012, 24, 2291–2296. [Google Scholar] [CrossRef]
- Kauntz, H.; Bousserouel, S.; Gosse, F.; Marescaux, J.; Raul, F. Silibinin, a Natural Flavonoid, Modulates the Early Expression of Chemoprevention Biomarkers in a Preclinical Model of Colon Carcinogenesis. Int. J. Oncol. 2012, 41, 849–854. [Google Scholar] [CrossRef]
- Kauntz, H.; Bousserouel, S.; Gossé, F.; Raul, F. Silibinin Triggers Apoptotic Signaling Pathways and Autophagic Survival Response in Human Colon Adenocarcinoma Cells and Their Derived Metastatic Cells. Apoptosis 2011, 16, 1042–1053. [Google Scholar] [CrossRef]
- Eo, H.J.; Park, G.H.; Song, H.M.; Lee, J.W.; Kim, M.K.; Lee, M.H.; Lee, J.R.; Koo, J.S.; Jeong, J.B. Silymarin Induces Cyclin D1 Proteasomal Degradation via Its Phosphorylation of Threonine-286 in Human Colorectal Cancer Cells. Int. Immunopharmacol. 2015, 24, 1–6. [Google Scholar] [CrossRef]
- Eo, H.J.; Jeong, J.B.; Koo, J.S.; Jeong, H.J. Silymarin-Mediated Degradation of c-Myc Contributes to the Inhibition of Cell Proliferation in Human Colorectal Cancer Cells. Korean J. Plant Resour. 2017, 30, 265–271. [Google Scholar] [CrossRef]
- Wang, J.Y.; Chang, C.C.; Chiang, C.C.; Chen, W.M.; Hung, S.C. Silibinin Suppresses the Maintenance of Colorectal Cancer Stem-like Cells by Inhibiting PP2A/AKT/MTOR Pathways. J. Cell. Biochem. 2012, 113, 1733–1743. [Google Scholar] [CrossRef]
- Kim, S.H.; Choo, G.S.; Yoo, E.S.; Woo, J.S.; Han, S.H.; Lee, J.H.; Jung, J.Y. Silymarin Induces Inhibition of Growth and Apoptosis through Modulation of the MAPK Signaling Pathway in AGS Human Gastric Cancer Cells. Oncol. Rep. 2019, 42, 1904–1914. [Google Scholar] [CrossRef]
- Li, R.; Yu, J.; Wang, C. Silibinin Promotes the Apoptosis of Gastric Cancer BGC823 Cells through Caspase Pathway. J. BUON 2017, 22, 1148–1153. [Google Scholar] [PubMed]
- Wang, Y.X.; Cai, H.; Jiang, G.; Zhou, T.B.; Wu, H. Silibinin Inhibits Proliferation, Induces Apoptosis and Causes Cell Cycle Arrest in Human Gastric Cancer MGC803 Cells via STAT3 Pathway Inhibition. Asian Pacific J. Cancer Prev. 2014, 15, 6791–6798. [Google Scholar] [CrossRef] [PubMed]
- Zappavigna, S.; Vanacore, D.; Lama, S.; Potenza, N.; Russo, A.; Ferranti, P.; Dallio, M.; Federico, A.; Loguercio, C.; Sperlongano, P.; et al. Silybin-Induced Apoptosis Occurs in Parallel to the Increase of Ceramides Synthesis and Mirnas Secretion in Human Hepatocarcinoma Cells. Int. J. Mol. Sci. 2019, 20, 2190. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.R.; Park, S.C.; Choi, S.; Lee, J.C.; Kim, Y.C.; Han, C.J.; Kim, J.; Yang, K.Y.; Kim, Y.J.; Noh, G.Y.; et al. Combined Treatment with Silibinin and Either Sorafenib or Gefitinib Enhances Their Growth-Inhibiting Effects in Hepatocellular Carcinoma Cells. Clin. Mol. Hepatol. 2015, 21, 49–59. [Google Scholar] [CrossRef]
- Bektur Aykanat, N.E.; Kacar, S.; Karakaya, S.; Sahinturk, V. Silymarin Suppresses HepG2 Hepatocarcinoma Cell Progression through Downregulation of Slit-2/Robo-1 Pathway. Pharmacol. Rep. 2020, 72, 199–207. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, Y.; Liang, Z.; Duan, W.; Yang, J.; Yan, J.; Wang, N.; Feng, W.; Ding, M.; Nie, Y.; et al. Silybin-Mediated Inhibition of Notch Signaling Exerts Antitumor Activity in Human Hepatocellular Carcinoma Cells. PLoS ONE 2013, 8, 83699. [Google Scholar] [CrossRef]
- Ramakrishnan, G.; Jagan, S.; Kamaraj, S.; Anandakumar, P.; Devaki, T. Silymarin Attenuated Mast Cell Recruitment Thereby Decreased the Expressions of Matrix Metalloproteinases-2 and 9 in Rat Liver Carcinogenesis. Invest. New Drugs 2009, 27, 233–240. [Google Scholar] [CrossRef]
- Yang, X.; Li, X.; An, L.; Bai, B.; Chen, J. Silibinin Induced the Apoptosis of Hep-2 Cells via Oxidative Stress and down-Regulating Survivin Expression. Eur. Arch. Oto-Rhino-Laryngol. 2013, 270, 2289–2297. [Google Scholar] [CrossRef]
- Faezizadeh, Z.; Mesbah-Namin, S.A.R.; Allameh, A. The Effect of Silymarin on Telomerase Activity in the Human Leukemia Cell Line K562. Planta Med. 2012, 78, 899–902. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Bai, X.; Liu, Y.; Shi, S.; Wang, X.; Zhan, Y.; Kang, X.; Chen, Y.; An, H. Inhibition of TMEM16A by Natural Product Silibinin: Potential Lead Compounds for Treatment of Lung Adenocarcinoma. Front. Pharmacol. 2021, 12, 736. [Google Scholar] [CrossRef] [PubMed]
- Won, D.H.; Kim, L.H.; Jang, B.; Yang, I.H.; Kwon, H.J.; Jin, B.; Oh, S.H.; Kang, J.H.; Hong, S.D.; Shin, J.A.; et al. In Vitro and In Vivo Anti-Cancer Activity of Silymarin on Oral Cancer. Tumor Biol. 2018, 40, 1010428318776170. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.S.; Oh, S.; Jang, B.; Yu, H.J.; Shin, J.A.; Cho, N.P.; Yang, I.H.; Won, D.H.; Kwon, H.J.; Hong, S.D.; et al. Silymarin and Its Active Component Silibinin Act as Novel Therapeutic Alternatives for Salivary Gland Cancer by Targeting the ERK1/2-Bim Signaling Cascade. Cell. Oncol. 2017, 40, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, R.M.; Devaraj, E. Silibinin Triggers the Mitochondrial Pathway of Apoptosis in Human Oral Squamous Carcinoma Cells. Asian Pacific J. Cancer Prev. 2020, 21, 1877–1882. [Google Scholar] [CrossRef]
- Fan, L.; Ma, Y.; Liu, Y.; Zheng, D.; Huang, G. Silymarin Induces Cell Cycle Arrest and Apoptosis in Ovarian Cancer Cells. Eur. J. Pharmacol. 2014, 743, 79–88. [Google Scholar] [CrossRef]
- Kacar, S.; Bektur Aykanat, N.E.; Sahinturk, V. Silymarin Inhibited DU145 Cells by Activating SLIT2 Protein and Suppressing Expression of CXCR4. Med. Oncol. 2020, 37, 18. [Google Scholar] [CrossRef]
- Singh, R.P.; Tyagi, A.K.; Zhao, J.; Agarwal, R. Silymarin Inhibits Growth and Causes Regression of Established Skin Tumors in SENCAR Mice via Modulation of Mitogen-Activated Protein Kinases and Induction of Apoptosis. Carcinogenesis 2002, 23, 499–510. [Google Scholar] [CrossRef]
- Vaid, M.; Prasad, R.; Sun, Q.; Katiyar, S.K. Silymarin Targets B-Catenin Signaling in Blocking Migration/Invasion of Human Melanoma Cells. PLoS ONE 2011, 6, e23000. [Google Scholar] [CrossRef]
- Khan, A.Q.; Khan, R.; Tahir, M.; Rehman, M.U.; Lateef, A.; Ali, F.; Hamiza, O.O.; Hasan, S.K.; Sultana, S. Silibinin Inhibits Tumor Promotional Triggers and Tumorigenesis against Chemically Induced Two-Stage Skin Carcinogenesis in Swiss Albino Mice: Possible Role of Oxidative Stress and Inflammation. Nutr. Cancer 2014, 66, 249–258. [Google Scholar] [CrossRef]
- Vaid, M.; Singh, T.; Prasad, R.; Katiyar, S.K. Silymarin Inhibits Melanoma Cell Growth Both In Vitro and In Vivo by Targeting Cell Cycle Regulators, Angiogenic Biomarkers and Induction of Apoptosis. Mol. Carcinog. 2015, 54, 1328–1339. [Google Scholar] [CrossRef] [PubMed]
- Kalla, P.K.; Chitti, S.; Aghamirzaei, S.T.; Senthilkumar, R.; Arjunan, S. Anti-Cancer Activity of Silymarin on MCF-7 and NCIH-23 Cell Lines. Adv. Biol. Res. 2014, 8, 57–61. [Google Scholar] [CrossRef]
- Vinh, P.Q.; Sugie, S.; Tanaka, T.; Hara, A.; Yamada, Y.; Katayama, M.; Deguchi, T.; Mori, H. Chemopreventive Effects of a Flavonoid Antioxidant Silymarin on N-Butyl-N-(4-Hydroxybutyl)Nitrosamine-Induced Urinary Bladder Carcinogenesis in Male ICR Mice. Jpn. J. Cancer Res. 2002, 93, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Luo, J.; Guo, X. Silybin Suppresses Cell Proliferation and Induces Apoptosis of Multiple Myeloma Cells via the PI3K/Akt/MTOR Signaling Pathway. Mol. Med. Rep. 2016, 13, 3243–3248. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Wu, L.J.; Tashiro, S.I.; Onodera, S.; Li, L.H.; Ikejima, T. Silymarin Augments Human Cervical Cancer HeLa Cell Apoptosis via P38/JNK MAPK Pathways in Serum-Free Medium. J. Asian Nat. Prod. Res. 2005, 7, 701–709. [Google Scholar] [CrossRef]
- Deep, G.; Oberlies, N.H.; Kroll, D.J.; Agarwal, R. Identifying the Differential Effects of Silymarin Constituents on Cell Growth and Cell Cycle Regulatory Molecules in Human Prostate Cancer Cells. Int. J. Cancer 2008, 123, 41–50. [Google Scholar] [CrossRef]
- Bhatia, N.; Agarwal, R. Detrimental Effect of Cancer Preventive Phytochemicals Silymarin, Genistein and Epigallocatechin 3-Gallate on Epigenetic Events in Human Prostate Carcinoma DU145 Cells. Prostate 2001, 46, 98–107. [Google Scholar] [CrossRef]
- Katiyar, S.K.; Roy, A.M.; Baliga, M.S. Silymarin Induces Apoptosis Primarily through a P53-Dependent Pathway Involving Bcl-2/Bax, Cytochrome c Release, and Caspase Activation. Mol. Cancer Ther. 2005, 4, 207–216. [Google Scholar] [CrossRef]
- Zi, X.; Agarwal, R. Modulation of Mitogen-Activated Protein Kinase Activation and Cell Cycle Regulators by the Potent Skin Cancer Preventive Agent Silymarin. Biochem. Biophys. Res. Commun. 1999, 263, 528–536. [Google Scholar] [CrossRef]
- Vaid, M.; Katiyar, S.K. Molecular Mechanisms of Inhibition of Photocarcinogenesis by Silymarin, a Phytochemical from Milk Thistle (Silybum marianum L. Gaertn.) (Review). Int. J. Oncol. 2010, 36, 1053–1060. [Google Scholar] [CrossRef]
- Zhu, Z.; Sun, G. Silymarin Mitigates Lung Impairments in a Rat Model of Acute Respiratory Distress Syndrome. Inflammopharmacology 2018, 26, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, G.; Lo Muzio, L.; Elinos-Báez, C.M.; Jagan, S.; Augustine, T.A.; Kamaraj, S.; Anandakumar, P.; Devaki, T. Silymarin Inhibited Proliferation and Induced Apoptosis in Hepatic Cancer Cells. Cell Prolif. 2009, 42, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Aayadi, H.; Mittal, S.P.K.; Deshpande, A.; Gore, M.; Ghaskadbi, S.S. Protective Effect of Geraniin against Carbon Tetrachloride Induced Acute Hepatotoxicity in Swiss Albino Mice. Biochem. Biophys. Res. Commun. 2017, 487, 62–67. [Google Scholar] [CrossRef]
- Kim, E.K.; Choi, E.J. Pathological Roles of MAPK Signaling Pathways in Human Diseases. Biochim. Biophys. Acta-Mol. Basis Dis. 2010, 1802, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Kauntz, H.; Bousserouel, S.; Gossé, F.; Raul, F. The Flavonolignan Silibinin Potentiates TRAIL-Induced Apoptosis in Human Colon Adenocarcinoma and in Derived TRAIL-Resistant Metastatic Cells. Apoptosis 2012, 17, 797–809. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, Y.; Gao, Y.; Li, S. Silibinin-Induced Glioma Cell Apoptosis by PI3K-Mediated but Akt-Independent Downregulation of FoxM1 Expression. Eur. J. Pharmacol. 2015, 765, 346–354. [Google Scholar] [CrossRef]
- Shapiro, G.I. Cyclin-Dependent Kinase Pathways as Targets for Cancer Treatment. J. Clin. Oncol. 2006, 24, 1770–1783. [Google Scholar] [CrossRef]
- Vijayaraghavan, S.; Moulder, S.; Keyomarsi, K.; Layman, R.M. Inhibiting CDK in Cancer Therapy: Current Evidence and Future Directions. Target. Oncol. 2018, 13, 21–38. [Google Scholar] [CrossRef]
- Cui, H.; Li, T.L.; Guo, H.F.; Wang, J.L.; Xue, P.; Zhang, Y.; Fan, J.H.; Li, Z.P.; Gao, Y.J. Silymarin-Mediated Regulation of the Cell Cycle and DNA Damage Response Exerts Antitumor Activity in Human Hepatocellular Carcinoma. Oncol. Lett. 2018, 15, 885–892. [Google Scholar] [CrossRef]
- Jung, Y.S.; Park, J. Il Wnt Signaling in Cancer: Therapeutic Targeting of Wnt Signaling beyond β-Catenin and the Destruction Complex. Exp. Mol. Med. 2020, 52, 183–191. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X. Targeting the Wnt/β-Catenin Signaling Pathway in Cancer. J. Hematol. Oncol. 2020, 13, 165. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wang, D.S.; Li, Q.J.; Sun, W.; Zhang, Y.; Dou, K.F. The Down-Regulation of Notch1 Inhibits the Invasion and Migration of Hepatocellular Carcinoma Cells by Inactivating the Cyclooxygenase-2/Snail/E- Cadherin Pathway In Vitro. Dig. Dis. Sci. 2013, 58, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- de los Fayos Alons Alonso, I.; Liang, H.C.; Turner, S.D.; Lagger, S.; Merkel, O.; Kenner, L. The Role of Activator Protein-1 (AP-1) Family Members in CD30-Positive Lymphomas. Cancers 2018, 10, 93. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Behnam Azad, B.; Nimmagadda, S. The Intricate Role of CXCR4 in Cancer. In Advances in Cancer Research; Academic Press: Cambridge, MA, USA, 2014; Volume 124, pp. 31–82. [Google Scholar]
- Bosch-Barrera, J.; Queralt, B.; Menendez, J.A. Targeting STAT3 with Silibinin to Improve Cancer Therapeutics. Cancer Treat. Rev. 2017, 58, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Kittur, S.; Wilasrusmee, S.; Pedersen, W.A.; Mattson, M.P.; Straube-West, K.; Wilasrusmee, C.; Jubelt, B.; Kittur, D.S. Neurotrophic and Neuroprotective Effects of Milk Thistle (Silybum marianum) on Neurons in Culture. J. Mol. Neurosci. 2002, 18, 265–269. [Google Scholar] [CrossRef]
- Darvesh, A.S.; Carroll, R.T.; Bishayee, A.; Geldenhuys, W.J.; Van Der Schyf, C.J. Oxidative Stress and Alzheimer’s Disease: Dietary Polyphenols as Potential Therapeutic Agents. Expert Rev. Neurother. 2010, 10, 729–745. [Google Scholar] [CrossRef]
- Song, X.; Zhou, B.; Cui, L.; Lei, D.; Zhang, P.; Yao, G.; Xia, M.; Hayashi, T.; Hattori, S.; Ushiki-Kaku, Y.; et al. Silibinin Ameliorates Aβ25-35-Induced Memory Deficits in Rats by Modulating Autophagy and Attenuating Neuroinflammation as Well as Oxidative Stress. Neurochem. Res. 2017, 42, 1073–1083. [Google Scholar] [CrossRef]
- Al-Enazi, M.M. Neuroprotective Effect of Silymarin by Modulation of Endogenous Biomarkers in Streptozotocin Induced Painful Diabetic Neuropathy. Br. J. Pharmacol. Toxicol. 2013, 4, 110–120. [Google Scholar] [CrossRef]
- Song, X.; Zhou, B.; Zhang, P.; Lei, D.; Wang, Y.; Yao, G.; Hayashi, T.; Xia, M.; Tashiro, S.I.; Onodera, S.; et al. Protective Effect of Silibinin on Learning and Memory Impairment in LPS-Treated Rats via ROS–BDNF–TrkB Pathway. Neurochem. Res. 2016, 41, 1662–1672. [Google Scholar] [CrossRef]
- Thakare, V.N.; Aswar, M.K.; Kulkarni, Y.P.; Patil, R.R.; Patel, B.M. Silymarin Ameliorates Experimentally Induced Depressive like Behavior in Rats: Involvement of Hippocampal BDNF Signaling, Inflammatory Cytokines and Oxidative Stress Response. Physiol. Behav. 2017, 179, 401–410. [Google Scholar] [CrossRef]
- Haddadi, R.; Nayebi, A.M.; Farajniya, S.; Brooshghalan, S.E.; Sharifi, H. Silymarin Improved 6-OHDA-Induced Motor Impairment in Hemi-Parkisonian Rats: Behavioral and Molecular Study. DARU J. Pharm. Sci. 2014, 22, 38. [Google Scholar] [CrossRef] [PubMed]
- Galhardi, F.; Mesquita, K.; Monserrat, J.M.; Barros, D.M. Effect of Silymarin on Biochemical Parameters of Oxidative Stress in Aged and Young Rat Brain. Food Chem. Toxicol. 2009, 47, 2655–2660. [Google Scholar] [CrossRef] [PubMed]
- Nencini, C.; Giorgi, G.; Micheli, L. Protective Effect of Silymarin on Oxidative Stress in Rat Brain. Phytomedicine 2007, 14, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.W.; Lin, T.Y.; Chiu, K.M.; Lee, M.Y.; Huang, J.H.; Wang, S.J. Silymarin Inhibits Glutamate Release and Prevents against Kainic Acid-Induced Excitotoxic Injury in Rats. Biomedicines 2020, 8, 486. [Google Scholar] [CrossRef]
- Ullah, H.; Khan, H. Anti-Parkinson Potential of Silymarin: Mechanistic Insight and Therapeutic Standing. Front. Pharmacol. 2018, 9, 422. [Google Scholar] [CrossRef]
- Thome, A.D.; Harms, A.S.; Volpicelli-Daley, L.A.; Standaert, D.G. MicroRNA-155 Regulates Alpha-Synuclein-Induced Inflammatory Responses in Models of Parkinson Disease. J. Neurosci. 2016, 36, 2383–2390. [Google Scholar] [CrossRef]
- Pérez-H, J.; Carrillo-S, C.; García, E.; Ruiz-Mar, G.; Pérez-Tamayo, R.; Chavarría, A. Neuroprotective Effect of Silymarin in a MPTP Mouse Model of Parkinson’s Disease. Toxicology 2014, 319, 38–43. [Google Scholar] [CrossRef]
- Baluchnejadmojarad, T.; Roghani, M.; Mafakheri, M. Neuroprotective Effect of Silymarin in 6-Hydroxydopamine Hemi-Parkinsonian Rat: Involvement of Estrogen Receptors and Oxidative Stress. Neurosci. Lett. 2010, 480, 206–210. [Google Scholar] [CrossRef]
- Srivastava, S.; Sammi, S.R.; Laxman, T.S.; Pant, A.; Nagar, A.; Trivedi, S.; Bhatta, R.S.; Tandon, S.; Pandey, R. Silymarin Promotes Longevity and Alleviates Parkinson’s Associated Pathologies in Caenorhabditis Elegans. J. Funct. Foods 2017, 31, 32–43. [Google Scholar] [CrossRef]
- Mazzio, E.A.; Harris, N.; Soliman, K.F.A. Food Constituents Attenuate Monoamine Oxidase Activity and Peroxide Levels in C6 Astrocyte Cells. Planta Med. 1998, 64, 603–606. [Google Scholar] [CrossRef]
- de Oliveira, D.R.; Schaffer, L.F.; Busanello, A.; Barbosa, C.P.; Peroza, L.R.; de Freitas, C.M.; Krum, B.N.; Bressan, G.N.; Boligon, A.A.; Athayde, M.L.; et al. Silymarin Has Antioxidant Potential and Changes the Activity of Na+/K+-ATPase and Monoamine Oxidase In Vitro. Ind. Crops Prod. 2015, 70, 347–355. [Google Scholar] [CrossRef]
- Wang, M.J.; Lin, W.W.; Chen, H.L.; Chang, Y.H.; Ou, H.C.; Kuo, J.S.; Hong, J.S.; Jeng, K.C.G. Silymarin Protects Dopaminergic Neurons against Lipopolysaccharide-Induced Neurotoxicity by Inhibiting Microglia Activation. Eur. J. Neurosci. 2002, 16, 2103–2112. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, M.K.; Rasheed, M.S.U.; Mishra, A.K.; Patel, D.K.; Singh, M.P. Silymarin Protects against Impaired Autophagy Associated with 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine-Induced Parkinsonism. J. Mol. Neurosci. 2020, 70, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Aboelwafa, H.R.; El-Kott, A.F.; Abd-Ella, E.M.; Yousef, H.N. The Possible Neuroprotective Effect of Silymarin against Aluminum Chloride-Prompted Alzheimer’s-like Disease in Rats. Brain Sci. 2020, 10, 628. [Google Scholar] [CrossRef]
- El-Marasy, S.A.A.; Abd-Elsalam, R.M.; Ahmed-Farid, O.A. Ameliorative Effect of Silymarin on Scopolamine-Induced Dementia in Rats. Open Access Maced. J. Med. Sci. 2018, 6, 1215–1224. [Google Scholar] [CrossRef]
- Duan, S.; Guan, X.; Lin, R.; Liu, X.; Yan, Y.; Lin, R.; Zhang, T.; Chen, X.; Huang, J.; Sun, X.; et al. Silibinin Inhibits Acetylcholinesterase Activity and Amyloid β Peptide Aggregation: A Dual-Target Drug for the Treatment of Alzheimer’s Disease. Neurobiol. Aging 2015, 36, 1792–1807. [Google Scholar] [CrossRef]
- Yaghmaei, P.; Azarfar, K.; Dezfulian, M.; Ebrahim-Habibi, A. Silymarin Effect on Amyloid-β Plaque Accumulation and Gene Expression of APP in an Alzheimer’s Disease Rat Model. DARU J. Pharm. Sci. 2014, 22, 24. [Google Scholar] [CrossRef]
- Urata, N.M.; Urakami, K.M.; Zawa, Y.O.; Inoshita, N.K.; Rie, K.I.; Shirasawa, T.; Shimizu, T. Silymarin Attenuated the Amyloid β Plaque Burden and Improved Behavioral Abnormalities in an Alzheimer’s Disease Mouse Model. Biosci. Biotechnol. Biochem. 2010, 74, 2299–2306. [Google Scholar] [CrossRef]
- Zheng, H.; Koo, E.H. Biology and Pathophysiology of the Amyloid Precursor Protein. Mol. Neurodegener. 2011, 6, 27. [Google Scholar] [CrossRef]
- Lu, P.; Mamiya, T.; Lu, L.L.; Mouri, A.; Zou, L.B.; Nagai, T.; Hiramatsu, M.; Ikejima, T.; Nabeshima, T. Silibinin Prevents Amyloid b Peptide-Induced Memory Impairment and Oxidative Stress in Mice. Br. J. Pharmacol. 2009, 157, 1270–1277. [Google Scholar] [CrossRef]
- Zhou, J.; Chao, G.; Li, Y.L.; Wu, M.; Zhong, S.Z.; Feng, Z.Y. Activation of NRF2/ARE by Isosilybin Alleviates Aβ25-35-Induced Oxidative Stress Injury in HT-22 Cells. Neurosci. Lett. 2016, 632, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Liu, B.; Cui, L.; Zhou, B.; Liu, L.; Liu, W.; Yao, G.; Xia, M.; Hayashi, T.; Hattori, S.; et al. Estrogen Receptors Are Involved in the Neuroprotective Effect of Silibinin in Aβ1–42-Treated Rats. Neurochem. Res. 2018, 43, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Sun, Y.; Xu, F.; Liu, W.; Hayashi, T.; Hattori, S.; Ushiki-Kaku, Y.; Onodera, S.; Tashiro, S.I.; Ikejima, T. Silibinin Protects Rat Pancreatic β-Cell through up-Regulation of Estrogen Receptors’ Signaling against Amylin- or Aβ1–42-Induced Reactive Oxygen Species/Reactive Nitrogen Species Generation. Phyther. Res. 2019, 33, 998–1009. [Google Scholar] [CrossRef] [PubMed]
- Yardım, A.; Kucukler, S.; Özdemir, S.; Çomaklı, S.; Caglayan, C.; Kandemir, F.M.; Çelik, H. Silymarin Alleviates Docetaxel-Induced Central and Peripheral Neurotoxicity by Reducing Oxidative Stress, Inflammation and Apoptosis in Rats. Gene 2021, 769, 145239. [Google Scholar] [CrossRef]
- Chtourou, Y.; Fetoui, H.; Sefi, M.; Trabelsi, K.; Barkallah, M.; Boudawara, T.; Kallel, H.; Zeghal, N. Silymarin, a Natural Antioxidant, Protects Cerebral Cortex against Manganese-Induced Neurotoxicity in Adult Rats. BioMetals 2010, 23, 985–996. [Google Scholar] [CrossRef]
- Elsawy, H.; Alzahrani, A.M.; Alfwuaires, M.; Sedky, A.; El-Trass, E.E.; Mahmoud, O.; Abdel-Moneim, A.M.; Khalil, M. Analysis of Silymarin-Modulating Effects against Acrylamide-Induced Cerebellar Damage in Male Rats: Biochemical and Pathological Markers. J. Chem. Neuroanat. 2021, 115, 101964. [Google Scholar] [CrossRef]
- Hirayama, K.; Oshima, H.; Yamashita, A.; Sakatani, K.; Yoshino, A.; Katayama, Y. Neuroprotective Effects of Silymarin on Ischemia-Induced Delayed Neuronal Cell Death in Rat Hippocampus. Brain Res. 2016, 1646, 297–303. [Google Scholar] [CrossRef]
- Wang, C.; Wang, Z.; Zhang, X.; Zhang, X.; Dong, L.; Xing, Y.; Li, Y.; Liu, Z.; Chen, L.; Qiao, H.; et al. Protection by Silibinin against Experimental Ischemic Stroke: Up-Regulated PAkt, PmTOR, HIF-1α and Bcl-2, down-Regulated Bax, NF-κB Expression. Neurosci. Lett. 2012, 529, 45–50. [Google Scholar] [CrossRef]
- Khoshnoodi, M.; Fakhraei, N.; Dehpour, A.R. Possible Involvement of Nitric Oxide in Antidepressant-like Effect of Silymarin in Male Mice. Pharm. Biol. 2015, 53, 739–745. [Google Scholar] [CrossRef]
- Li, Y.J.; Li, Y.J.; Yang, L.D.; Zhang, K.; Zheng, K.Y.; Wei, X.M.; Yang, Q.; Niu, W.M.; Zhao, M.G.; Wu, Y.M. Silibinin Exerts Antidepressant Effects by Improving Neurogenesis through BDNF/TrkB Pathway. Behav. Brain Res. 2018, 348, 184–191. [Google Scholar] [CrossRef]
- Song, X.; Liu, B.; Cui, L.; Zhou, B.; Liu, W.; Xu, F.; Hayashi, T.; Hattori, S.; Ushiki-Kaku, Y.; Tashiro, S.-I.; et al. Silibinin Ameliorates Anxiety/Depression-like Behaviors in Amyloid β-Treated Rats by Upregulating BDNF/TrkB Pathway and Attenuating Autophagy in Hippocampus. Physiol. Behav. 2017, 179, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Babu, H.; Ramirez-Rodriguez, G.; Fabel, K.; Bischofberger, J.; Kempermann, G. Synaptic Network Activity Induces Neuronal Differentiation of Adult Hippocampal Precursor Cells through BDNF Signaling. Front. Neurosci. 2009, 1, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Poo, M.M. Neurotrophin Regulation of Neural Circuit Development and Function. Nat. Rev. Neurosci. 2013, 14, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.S.; Khan, M.M.; Ashafaq, M.; Ahmad, A.; Khuwaja, G.; Khan, A.; Siddiqui, M.S.; Safhi, M.M.; Islam, F. Silymarin Protects Neurons from Oxidative Stress Associated Damages in Focal Cerebral Ischemia: A Behavioral, Biochemical and Immunohistological Study in Wistar Rats. J. Neurol. Sci. 2011, 309, 45–54. [Google Scholar] [CrossRef]
- Wang, M.; Li, Y.J.; Ding, Y.; Zhang, H.N.; Sun, T.; Zhang, K.; Yang, L.; Guo, Y.Y.; Liu, S.B.; Zhao, M.G.; et al. Silibinin Prevents Autophagic Cell Death upon Oxidative Stress in Cortical Neurons and Cerebral Ischemia-Reperfusion Injury. Mol. Neurobiol. 2016, 53, 932–943. [Google Scholar] [CrossRef]
- Al-Rasheed, N.M.; Al-Rasheed, N.M.; Faddah, L.M.; Mohamed, A.M.; Mohammad, R.A.; Al-Amin, M. Potential Impact of Silymarin in Combination with Chlorogenic Acid and/or Melatonin in Combating Cardiomyopathy Induced by Carbon Tetrachloride. Saudi J. Biol. Sci. 2014, 21, 265–274. [Google Scholar] [CrossRef]
- Rao, P.R.; Viswanath, R.K. Cardioprotective Activity of Silymarin in Ischemia-Reperfusion-Induced Myocardial Infarction in Albino Rats. Exp. Clin. Cardiol. 2007, 12, 179–187. [Google Scholar]
- Taghiabadi, E.; Imenshahidi, M.; Abnous, K.; Mosafa, F.; Sankian, M.; Memar, B.; Karimi, G. Protective Effect of Silymarin against Acrolein-Induced Cardiotoxicity in Mice. Evid.-Based Complement. Altern. Med. 2012, 2012, 1–14. [Google Scholar] [CrossRef]
- Gabrielová, E.; Zholobenko, A.V.; Bartošíková, L.; Nečas, J.; Modriansky, M. Silymarin Constituent 2,3-Dehydrosilybin Triggers Reserpine-Sensitive Positive Inotropic Effect in Perfused Rat Heart. PLoS ONE 2015, 10, e0139208. [Google Scholar] [CrossRef][Green Version]
- Jadhav, G.B.; Upasani, C.D. Antihypertensive Effect of Silymarin on DOCA Salt Induced Hypertension in Unilateral Nephrectomized Rats. Orient. Pharm. Exp. Med. 2011, 11, 101–106. [Google Scholar] [CrossRef]
- Jadhav, G.B.; Upasani, C.D. Antihypertensive Effect of Silymarin on Fructose Induced Hypertensive Rats. Indian J. Pharm. Educ. Res. 2012, 46, 26. [Google Scholar]
- Ahmed-Belkacem, A.; Ahnou, N.; Barbotte, L.; Wychowski, C.; Pallier, C.; Brillet, R.; Pohl, R.T.; Pawlotsky, J.M. Silibinin and Related Compounds Are Direct Inhibitors of Hepatitis C Virus RNA-Dependent RNA Polymerase. Gastroenterology 2010, 138, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Wagoner, J.; Negash, A.; Kane, O.J.; Martinez, L.E.; Nahmias, Y.; Bourne, N.; Owen, D.M.; Grove, J.; Brimacombe, C.; McKeating, J.A.; et al. Multiple Effects of Silymarin on the Hepatitis C Virus Lifecycle. Hepatology 2010, 51, 1912–1921. [Google Scholar] [CrossRef]
- Anthony, K.; Subramanya, G.; Uprichard, S.; Hammouda, F.; Saleh, M. Antioxidant and Anti-Hepatitis C Viral Activities of Commercial Milk Thistle Food Supplements. Antioxidants 2013, 2, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Ferenci, P.; Scherzer, T.M.; Kerschner, H.; Rutter, K.; Beinhardt, S.; Hofer, H.; Schöniger-Hekele, M.; Holzmann, H.; Steindl-Munda, P. Silibinin Is a Potent Antiviral Agent in Patients with Chronic Hepatitis C Not Responding to Pegylated Interferon/Ribavirin Therapy. Gastroenterology 2008, 135, 1561–1567. [Google Scholar] [CrossRef] [PubMed]
- Sabir, S.; Arshad, M.; Asif, S.; Chaudhari, S.K. An Insight into Medicinal and Therapeutic Potential of Silybum marianum (L.) Gaertn. Int. J. Biosci. 2014, 4, 104–115. [Google Scholar] [CrossRef]
- Saller, R.; Meier, R.; Brignoli, R. The Use of Silymarin in the Treatment of Liver Diseases. Drugs 2001, 61, 2035–2063. [Google Scholar] [CrossRef]
- Polyak, S.J.; Morishima, C.; Shuhart, M.C.; Wang, C.C.; Liu, Y.; Lee, D.Y.W. Inhibition of T-Cell Inflammatory Cytokines, Hepatocyte NF-κB Signaling, and HCV Infection by Standardized Silymarin. Gastroenterology 2007, 132, 1925–1936. [Google Scholar] [CrossRef]
- DebRoy, S.; Hiraga, N.; Imamura, M.; Hayes, C.N.; Akamatsu, S.; Canini, L.; Perelson, A.S.; Pohl, R.T.; Persiani, S.; Uprichard, S.L.; et al. Hepatitis C Virus Dynamics and Cellular Gene Expression in UPA-SCID Chimeric Mice with Humanized Livers during Intravenous Silibinin Monotherapy. J. Viral Hepat. 2016, 23, 708–717. [Google Scholar] [CrossRef]
- Blaising, J.; Lévy, P.L.; Gondeau, C.; Phelip, C.; Varbanov, M.; Teissier, E.; Ruggiero, F.; Polyak, S.J.; Oberlies, N.H.; Ivanovic, T.; et al. Silibinin Inhibits Hepatitis C Virus Entry into Hepatocytes by Hindering Clathrin-Dependent Trafficking. Cell. Microbiol. 2013, 15, 1866–1882. [Google Scholar] [CrossRef]
- Malaguarnera, G.; Bertino, G.; Chisari, G.; Motta, M.; Vecchio, M.; Vacante, M.; Caraci, F.; Greco, C.; Drago, F.; Nunnari, G.; et al. Silybin Supplementation during HCV Therapy with Pegylated Interferon-α plus Ribavirin Reduces Depression and Anxiety and Increases Work Ability. BMC Psychiatry 2016, 16, 398. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Motta, M.; Vacante, M.; Malaguarnera, G.; Caraci, F.; Nunnari, G.; Gagliano, C.; Greco, C.; Chisari, G.; Drago, F.; et al. Silybin-Vitamin E-Phospholipids Complex Reduces Liver Fibrosis in Patients with Chronic Hepatitis C Treated with Pegylated Interferon α and Ribavirin. Am. J. Transl. Res. 2015, 7, 2510–2518. [Google Scholar]
- Biermer, M.; Berg, T. Rapid Suppression of Hepatitis C Viremia Induced by Intravenous Silibinin plus Ribavirin. Gastroenterology 2009, 137, 390–391. [Google Scholar] [CrossRef] [PubMed]
- Mariño, Z.; Crespo, G.; D’Amato, M.; Brambilla, N.; Giacovelli, G.; Rovati, L.; Costa, J.; Navasa, M.; Forns, X. Intravenous Silibinin Monotherapy Shows Significant Antiviral Activity in HCV-Infected Patients in the Peri-Transplantation Period. J. Hepatol. 2013, 58, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Bárcena, R.; Moreno, A.; Rodríguez-Gandía, M.A.; Albillos, A.; Arocena, C.; Blesa, C.; García-Hoz, F.; Graus, J.; Nuño, J.; López-Hervás, P.; et al. Safety and Anti-HCV Effect of Prolonged Intravenous Silibinin in HCV Genotype 1 Subjects in the Immediate Liver Transplant Period. J. Hepatol. 2013, 58, 421–426. [Google Scholar] [CrossRef]
- Umetsu, T.; Inoue, J.; Kogure, T.; Kakazu, E.; Ninomiya, M.; Iwata, T.; Takai, S.; Nakamura, T.; Sano, A.; Shimosegawa, T. Inhibitory Effect of Silibinin on Hepatitis B Virus Entry. Biochem. Biophys. Rep. 2018, 14, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Speciale, A.; Muscarà, C.; Molonia, M.S.; Cimino, F.; Saija, A.; Giofrè, S.V. Silibinin as Potential Tool against SARS-CoV-2: In Silico Spike Receptor-Binding Domain and Main Protease Molecular Docking Analysis, and In Vitro Endothelial Protective Effects. Phyther. Res. 2021, 35, 4616–4625. [Google Scholar] [CrossRef]
- Song, J.H.; Choi, H.J. Silymarin Efficacy against Influenza A Virus Replication. Phytomedicine 2011, 18, 832–835. [Google Scholar] [CrossRef]
- Dai, J.P.; Wu, L.Q.; Li, R.; Zhao, X.F.; Wan, Q.Y.; Chen, X.X.; Li, W.Z.; Wang, G.F.; Li, K.S. Identification of 23-(S)-2-Amino-3-Phenylpropanoyl-Silybin as an Antiviral Agent for Influenza A Virus Infection In Vitro and In Vivo. Antimicrob. Agents Chemother. 2013, 57, 4433–4443. [Google Scholar] [CrossRef]
- Qaddir, I.; Rasool, N.; Hussain, W.; Mahmood, S. Computer-Aided Analysis of Phytochemicals as Potential Dengue Virus Inhibitors Based on Molecular Docking, ADMET and DFT Studies. J. Vector Borne Dis. 2017, 54, 255–262. [Google Scholar] [CrossRef]
- Low, Z.X.; OuYong, B.M.; Hassandarvish, P.; Poh, C.L.; Ramanathan, B. Antiviral Activity of Silymarin and Baicalein against Dengue Virus. Sci. Rep. 2021, 11, 21221. [Google Scholar] [CrossRef] [PubMed]
- Camini, F.C.; da Silva, T.F.; da Silva Caetano, C.C.; Almeida, L.T.; Ferraz, A.C.; Alves Vitoreti, V.M.; de Mello Silva, B.; de Queiroz Silva, S.; de Magalhães, J.C.; de Brito Magalhães, C.L. Antiviral Activity of Silymarin against Mayaro Virus and Protective Effect in Virus-Induced Oxidative Stress. Antivir. Res. 2018, 158, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, A.C.; Almeida, L.T.; da Silva Caetano, C.C.; da Silva Menegatto, M.B.; Souza Lima, R.L.; de Senna, J.P.N.; de Oliveira Cardoso, J.M.; Perucci, L.O.; Talvani, A.; Geraldo de Lima, W.; et al. Hepatoprotective, Antioxidant, Anti-Inflammatory, and Antiviral Activities of Silymarin against Mayaro Virus Infection. Antivir. Res. 2021, 194, 105168. [Google Scholar] [CrossRef] [PubMed]
- Lalani, S.S.; Anasir, M.I.; Poh, C.L. Antiviral Activity of Silymarin in Comparison with Baicalein against EV-A71. BMC Complement. Med. Ther. 2020, 20, 97. [Google Scholar] [CrossRef]
- Lani, R.; Hassandarvish, P.; Chiam, C.W.; Moghaddam, E.; Chu, J.J.H.; Rausalu, K.; Merits, A.; Higgs, S.; Vanlandingham, D.; Abu Bakar, S.; et al. Antiviral Activity of Silymarin against Chikungunya Virus. Sci. Rep. 2015, 5, 11421. [Google Scholar] [CrossRef]
- Lani, R.; Agharbaoui, F.E.; Hassandarvish, P.; Teoh, B.T.; Sam, S.S.; Zandi, K.; Rahman, N.A.; Abubakar, S. In Silico Studies of Fisetin and Silymarin as Novel Chikungunya Virus Nonstructural Proteins Inhibitors. Future Virol. 2021, 16, 167–180. [Google Scholar] [CrossRef]
- Cardile, A.P.; Mbuy, G.K.N. Anti-Herpes Virus Activity of Silibinin, the Primary Active Component of Silybum marianum. J. Herb. Med. 2013, 3, 132–136. [Google Scholar] [CrossRef]
- McClure, J.; Lovelace, E.S.; Elahi, S.; Maurice, N.J.; Wagoner, J.; Dragavon, J.; Mittler, J.E.; Kraft, Z.; Stamatatos, L.; Horton, H.; et al. Silibinin Inhibits HIV-1 Infection by Reducing Cellular Activation and Proliferation. PLoS ONE 2012, 7, 41832. [Google Scholar] [CrossRef]
- McClure, J.; Margineantu, D.H.; Sweet, I.R.; Polyak, S.J. Inhibition of HIV by Legalon-SIL Is Independent of Its Effect on Cellular Metabolism. Virology 2014, 449, 96–103. [Google Scholar] [CrossRef]
- Zhou, B.; Wu, L.J.; Li, N.H.; Tashiro, S.I.; Onodera, S.; Uchiumi, F.; Ikejima, T. Silibinin Protects against Isoproterenol-Induced Rat Cardiac Myocyte Injury through Mitochondrial Pathway after up-Regulation of SIRT1. J. Pharmacol. Sci. 2006, 102, 387–395. [Google Scholar] [CrossRef]
- Rašković, A.; Stilinović, N.; Kolarović, J.; Vasović, V.; Vukmirović, S.; Mikov, M. The Protective Effects of Silymarin against Doxorubicin-Induced Cardiotoxicity and Hepatotoxicity in Rats. Molecules 2011, 16, 8601–8613. [Google Scholar] [CrossRef] [PubMed]
- Esser-Nobis, K.; Romero-Brey, I.; Ganten, T.M.; Gouttenoire, J.; Harak, C.; Klein, R.; Schemmer, P.; Binder, M.; Schnitzler, P.; Moradpour, D.; et al. Analysis of Hepatitis C Virus Resistance to Silibinin In Vitro and In Vivo Points to a Novel Mechanism Involving Nonstructural Protein 4B. Hepatology 2013, 57, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Sardanelli, A.M.; Isgrò, C.; Palese, L.L. SARS-CoV-2 Main Protease Active Site Ligands in the Human Metabolome. Molecules 2021, 26, 1409. [Google Scholar] [CrossRef] [PubMed]
- Payer, B.A.; Reiberger, T.; Rutter, K.; Beinhardt, S.; Staettermayer, A.F.; Peck-Radosavljevic, M.; Ferenci, P. Successful HCV Eradication and Inhibition of HIV Replication by Intravenous Silibinin in an HIV-HCV Coinfected Patient. J. Clin. Virol. 2010, 49, 131–133. [Google Scholar] [CrossRef] [PubMed]
- Braun, D.; Rauch, A.; Durisch, N.; Eberhard, N.; Anagnostopoulos, A.; Ledergerber, B.; Metzner, K.; Böni, J.; Weber, R.; Fehr, J. Efficacy of Lead-in Silibinin and Subsequent Triple Therapy in Difficult-to-Treat HIV/Hepatitis C Virus-Coinfected Patients. HIV Med. 2014, 15, 625–630. [Google Scholar] [CrossRef]
- Braun, D.L.; Rauch, A.; Aouri, M.; Durisch, N.; Eberhard, N.; Anagnostopoulos, A.; Ledergerber, B.; Möllhaupt, B.; Metzner, K.J.; Decosterd, L.; et al. A Lead-in with Silibinin Prior to Triple-Therapy Translates into Favorable Treatment Outcomes in Difficult-to-Treat HIV/Hepatitis C Coinfected Patients. PLoS ONE 2015, 10, e0133028. [Google Scholar] [CrossRef]
- Bosch-Barrera, J.; Martin-Castillo, B.; Buxó, M.; Brunet, J.; Encinar, J.A.; Menendez, J.A. Silibinin and SARS-CoV-2: Dual Targeting of Host Cytokine Storm and Virus Replication Machinery for Clinical Management of COVID-19 Patients. J. Clin. Med. 2020, 9, 1770. [Google Scholar] [CrossRef]
- Gorla, U.S.; Rao, K.; Kulandaivelu, U.S.; Alavala, R.R.; Panda, S.P. Lead Finding from Selected Flavonoids with Antiviral (SARS-CoV-2) Potentials against COVID-19: An In-Silico Evaluation. Comb. Chem. High Throughput Screen. 2020, 24, 879–890. [Google Scholar] [CrossRef]
- Boer, M.; Duchnik, E.; Maleszka, R.; Marchlewicz, M. Structural and Biophysical Characteristics of Human Skin in Maintaining Proper Epidermal Barrier Function. Postep. Dermatol. Alergol. 2016, 33, 1–5. [Google Scholar] [CrossRef]
- Benson, H.A.E.; Grice, J.E.; Mohammed, Y.; Namjoshi, S.; Roberts, M.S. Topical and Transdermal Drug Delivery: From Simple Potions to Smart Technologies. Curr. Drug Deliv. 2019, 16, 444–460. [Google Scholar] [CrossRef]
- Svobodová, A.; Vostálová, J. Solar Radiation Induced Skin Damage: Review of Protective and Preventive Options. Int. J. Radiat. Biol. 2010, 86, 999–1030. [Google Scholar] [CrossRef] [PubMed]
- Guillermo-Lagae, R.; Deep, G.; Ting, H.; Agarwal, C.; Agarwal, R. Silibinin Enhances the Repair of Ultraviolet B-Induced DNA Damage by Activating P53-Dependent Nucleotide Excision Repair Mechanism in Human Dermal Fibroblasts. Oncotarget 2015, 6, 39594–39606. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, S.K.; Mantena, S.K.; Meeran, S.M. Silymarin Protects Epidermal Keratinocytes from Ultraviolet Radiation-Induced Apoptosis and DNA Damage by Nucleotide Excision Repair Mechanism. PLoS ONE 2011, 6, e21410. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Deep, G.; Agarwal, C.; Agarwal, R. Silibinin Prevents Ultraviolet B Radiation-Induced Epidermal Damages in JB6 Cells and Mouse Skin in a P53-GADD45α-Dependent Manner. Carcinogenesis 2012, 33, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Rigby, C.M.; Roy, S.; Deep, G.; Guillermo-Lagae, R.; Jain, A.K.; Dhar, D.; Orlicky, D.J.; Agarwal, C.; Agarwal, R. Role of P53 in Silibinin-Mediated Inhibition of Ultraviolet B Radiation-Induced DNA Damage, Inflammation and Skin Carcinogenesis. Carcinogenesis 2017, 38, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Carrier, F.; Georgel, P.T.; Pourquier, P.; Blake, M.; Kontny, H.U.; Antinore, M.J.; Gariboldi, M.; Myers, T.G.; Weinstein, J.N.; Pommier, Y.; et al. Gadd45, a P53-Responsive Stress Protein, Modifies DNA Accessibility on Damaged Chromatin. Mol. Cell. Biol. 1999, 19, 1673–1685. [Google Scholar] [CrossRef]
- Yang, X.; Zhu, L.; Zhao, W.; He, C.; Li, S.; Xu, C. GADD45α Regulates Cell Proliferation and DNA Repair of BRL-3A Cells That Treated by FZD/UVC via P38, JNK, CDC2/CCNB1, AKT and MTOR Pathways. bioRxiv 2017, 148759. [Google Scholar] [CrossRef]
- Gu, M.; Dhanalakshmi, S.; Mohan, S.; Singh, R.P.; Agarwal, R. Silibinin Inhibits Ultraviolet B Radiation-Induced Mitogenic and Survival Signaling, and Associated Biological Responses in SKH-1 Mouse Skin. Carcinogenesis 2005, 26, 1404–1413. [Google Scholar] [CrossRef]
- Rajnochová Svobodová, A.; Gabrielová, E.; Ulrichová, J.; Zálešák, B.; Biedermann, D.; Vostálová, J. A Pilot Study of the UVA-Photoprotective Potential of Dehydrosilybin, Isosilybin, Silychristin, and Silydianin on Human Dermal Fibroblasts. Arch. Dermatol. Res. 2019, 311, 477–490. [Google Scholar] [CrossRef]
- Rajnochová Svobodová, A.; Gabrielová, E.; Michaelides, L.; Kosina, P.; Ryšavá, A.; Ulrichová, J.; Zálešák, B.; Vostálová, J. UVA-Photoprotective Potential of Silymarin and Silybin. Arch. Dermatol. Res. 2018, 310, 413–424. [Google Scholar] [CrossRef]
- Katiyar, S.K.; Meleth, S.; Sharma, S.D. Silymarin, a Flavonoid from Milk Thistle (Silybum marianum L.), Inhibits UV-Induced Oxidative Stress through Targeting Infiltrating CD11b+ Cells in Mouse Skin. Photochem. Photobiol. 2008, 84, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Svobodová, A.; Zdařilová, A.; Walterová, D.; Vostálová, J. Flavonolignans from Silybum marianum Moderate UVA-Induced Oxidative Damage to HaCaT Keratinocytes. J. Dermatol. Sci. 2007, 48, 213–224. [Google Scholar] [CrossRef]
- Svobodová, A.; Zdařilová, A.; Mališková, J.; Mikulková, H.; Walterová, D.; Vostalová, J. Attenuation of UVA-Induced Damage to Human Keratinocytes by Silymarin. J. Dermatol. Sci. 2007, 46, 21–30. [Google Scholar] [CrossRef]
- Li, L.H.; Wu, L.; Tashiro, S.; Onodera, S.; Uchiumi, F.; Ikejima, T. Activation of the SIRT1 Pathway and Modulation of the Cell Cycle Were Involved in Silymarin’s Protection against UV-Induced A375-S2 Cell Apoptosis. J. Asian Nat. Prod. Res. 2007, 9, 245–252. [Google Scholar] [CrossRef]
- Juráňová, J.; Aury-Landas, J.; Boumediene, K.; Baugé, C.; Biedermann, D.; Ulrichová, J.; Franková, J. Modulation of Skin Inflammatory Response by Active Components of Silymarin. Molecules 2019, 24, 123. [Google Scholar] [CrossRef]
- Li, L.H.; Wu, L.J.; Tashiro, S.I.; Onodera, S.; Uchiumi, F.; Ikejima, T. Silibinin Prevents UV-Induced HaCaT Cell Apoptosis Partly through Inhibition of Caspase-8 Pathway. Biol. Pharm. Bull. 2006, 29, 1096–1101. [Google Scholar] [CrossRef]
- Narayanapillai, S.; Agarwal, C.; Tilley, C.; Agarwal, R. Silibinin Is a Potent Sensitizer of UVA Radiation-Induced Oxidative Stress and Apoptosis in Human Keratinocyte HaCaT Cells. In Proceedings of the Photochemistry and Photobiology; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; Volume 88, pp. 1135–1140. [Google Scholar]
- Altaei, T. The Treatment of Melasma by Silymarin Cream. BMC Dermatol. 2012, 12, 18. [Google Scholar] [CrossRef]
- Cheon, H.I.; Bae, S.; Ahn, K.J. Flavonoid Silibinin Increases Hair-Inductive Property via Akt and Wnt/β-Catenin Signaling Activation in 3-Dimensional-Spheroid Cultured Human Dermal Papilla Cells. J. Microbiol. Biotechnol. 2019, 29, 321–329. [Google Scholar] [CrossRef]
- Sharifi, R.; Pasalar, P.; Kamalinejad, M.; Dehpour, A.R.; Tavangar, S.M.; Paknejad, M.; Mehrabani Natanzi, M.; Nourbakhsh, M.; Ahmadi Ashtiani, H.R.; Akbari, M.; et al. The Effect of Silymarin (Silybum marianum) on Human Skin Fibroblasts in an In Vitro Wound Healing Model. Pharm. Biol. 2013, 51, 298–303. [Google Scholar] [CrossRef]
- Tabari, S.A.; Carpi, S.; Polini, B.; Nieri, P.; Esfahani, M.L.; Moghadamnia, A.A.; Ghorbani, H.; Ranaei, M.; Kazemi, S. Topical Application of Silymarin Enhances Cutaneous Wound Healing in Rats. South African J. Bot. 2019, 124, 494–498. [Google Scholar] [CrossRef]
- Oryan, A.; Tabatabaei Naeini, A.; Moshiri, A.; Mohammadalipour, A.; Tabandeh, M.R. Modulation of Cutaneous Wound Healing by Silymarin in Rats. J. Wound Care 2012, 21, 457–464. [Google Scholar] [CrossRef]
- Vostálová, J.; Tinková, E.; Biedermann, D.; Kosina, P.; Ulrichová, J.; Svobodová, A.R. Skin Protective Activity of Silymarin and Its Flavonolignans. Molecules 2019, 24, 1022. [Google Scholar] [CrossRef]
- Ebrahimpour Koujan, S.; Gargari, B.P.; Mobasseri, M.; Valizadeh, H.; Asghari-Jafarabadi, M. Effects of Silybum marianum (L.) Gaertn. (Silymarin) Extract Supplementation on Antioxidant Status and Hs-CRP in Patients with Type 2 Diabetes Mellitus: A Randomized, Triple-Blind, Placebo-Controlled Clinical Trial. Phytomedicine 2015, 22, 290–296. [Google Scholar] [CrossRef]
- Ebrahimpour-koujan, S.; Gargari, B.P.; Mobasseri, M.; Valizadeh, H.; Asghari-Jafarabadi, M. Lower Glycemic Indices and Lipid Profile among Type 2 Diabetes Mellitus Patients Who Received Novel Dose of Silybum marianum (L.) Gaertn. (Silymarin) Extract Supplement: A Triple-Blinded Randomized Controlled Clinical Trial. Phytomedicine 2018, 44, 39–44. [Google Scholar] [CrossRef]
- Derosa, G.; D’Angelo, A.; Maffioli, P. The Role of a Fixed Berberis Aristata/Silybum marianum Combination in the Treatment of Type 1 Diabetes Mellitus. Clin. Nutr. 2016, 35, 1091–1095. [Google Scholar] [CrossRef]
- Di Pierro, F.; Putignano, P.; Villanova, N.; Montesi, L.; Moscatiello, S.; Marchesini, G. Preliminary Study about the Possible Glycemic Clinical Advantage in Using a Fixed Combination of Berberis Aristata and Silybum marianum Standardized Extracts versus Only Berberis Aristata in Patients with Type 2 Diabetes. Clin. Pharmacol. Adv. Appl. 2013, 5, 167–174. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hahn, H.J.; Jung, H.J.; Schrammek-Drusios, M.C.; Lee, S.N.; Kim, J.H.; Kwon, S.B.; An, I.S.; An, S.; Ahn, K.J. Instrumental Evaluation of Anti-Aging Effects of Cosmetic Formulations Containing Palmitoyl Peptides, Silybum marianum Seed Oil, Vitamin E and Other Functional Ingredients on Aged Human Skin. Exp. Ther. Med. 2016, 12, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- AlAnbari, H.; Sahib, A.; Raghif, A. Effects of Silymarin, N-Acetylcysteine and Selenium in the Treatment of Papulopustular Acne. Oxid. Antioxid. Med. Sci. 2012, 1, 201–207. [Google Scholar] [CrossRef]
- Curcio, A.; Romano, A.; Cuozzo, S.; Di Nicola, A.; Grassi, O.; Schiaroli, D.; Nocera, G.F.; Pironti, M. Silymarin in Combination with Vitamin C, Vitamin E, Coenzyme Q10 and Selenomethionine to Improve Liver Enzymes and Blood Lipid Profile in NAFLD Patients. Medicina 2020, 56, 544. [Google Scholar] [CrossRef] [PubMed]
- Hajiaghamohammadi, A.A.; Ziaee, A.; Oveisi, S.; Masroor, H. Effects of Metformin, Pioglitazone, and Silymarin Treatment on Non-Alcoholic Fatty Liver Disease: A Randomized Controlled Pilot Study. Hepat. Mon. 2012, 12, 6099. [Google Scholar] [CrossRef]
- Aller, R.; Izaola, O.; Gómez, S.; Tafur, C.; González, G.; Berroa, E.; Mora, N.; González, J.M.; De Luis, D.A. Effect of Silymarin plus Vitamin E in Patients with Non-Alcoholic Fatty Liver Disease. A Randomized Clinical Pilot Study. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 3118–3124. [Google Scholar]
- Abenavoli, L.; Greco, M.; Nazionale, I.; Peta, V.; Milic, N.; Accattato, F.; Foti, D.; Gulletta, E.; Luzza, F. Effects of Mediterranean Diet Supplemented with Silybin-Vitamin E-Phospholipid Complex in Overweight Patients with Non-Alcoholic Fatty Liver Disease. Expert Rev. Gastroenterol. Hepatol. 2015, 9, 519–527. [Google Scholar] [CrossRef]
- Federico, A.; Dallio, M.; Gravina, A.G.; Diano, N.; Errico, S.; Masarone, M.; Romeo, M.; Tuccillo, C.; Stiuso, P.; Morisco, F.; et al. The Bisphenol a Induced Oxidative Stress in Non-Alcoholic Fatty Liver Disease Male Patients: A Clinical Strategy to Antagonize the Progression of the Disease. Int. J. Environ. Res. Public Health 2020, 17, 3369. [Google Scholar] [CrossRef] [PubMed]
- Solhi, H.; Ghahremani, R.; Kazemifar, A.M.; Yazdi, Z.H. Silymarin in Treatment of Non-Alcoholic Steatohepatitis: A Randomized Clinical Trial. Casp. J. Intern. Med. 2014, 5, 9–12. [Google Scholar]
- Wah Kheong, C.; Nik Mustapha, N.R.; Mahadeva, S. A Randomized Trial of Silymarin for the Treatment of Nonalcoholic Steatohepatitis. Clin. Gastroenterol. Hepatol. 2017, 15, 1940–1949.e8. [Google Scholar] [CrossRef] [PubMed]
- Navarro, V.J.; Belle, S.H.; D’Amato, M.; Adfhal, N.; Brunt, E.M.; Fried, M.W.; Rajender Reddy, K.; Wahed, A.S.; Harrison, S. Silymarin in Non-Cirrhotics with Non-Alcoholic Steatohepatitis: A Randomized, Double-Blind, Placebo Controlled Trial. PLoS ONE 2019, 14, e0221683. [Google Scholar] [CrossRef]
- Abbasirad, F.; Shaygannejad, V.; Hosseininasab, F.; Mirmosayyeb, O.; Mahaki, B.; Moayedi, B.; Esmaeil, N. Significant Immunomodulatory and Hepatoprotective Impacts of Silymarin in MS Patients: A Double-Blind Placebo-Controlled Clinical Trial. Int. Immunopharmacol. 2021, 97, 107715. [Google Scholar] [CrossRef]
- Luangchosiri, C.; Thakkinstian, A.; Chitphuk, S.; Stitchantrakul, W.; Petraksa, S.; Sobhonslidsuk, A. A Double-Blinded Randomized Controlled Trial of Silymarin for the Prevention of Antituberculosis Drug-Induced Liver Injury. BMC Complement. Altern. Med. 2015, 15, 334. [Google Scholar] [CrossRef]
- Marjani, M.; Baghaei, P.; Dizaji, M.K.; Bayani, P.G.; Fahimi, F.; Tabarsi, P.; Velayati, A.A. Evaluation of Hepatoprotective Effect of Silymarin among under Treatment Tuberculosis Patients: A Randomized Clinical Trial. Iran. J. Pharm. Res. 2016, 15, 247–252. [Google Scholar] [CrossRef]
- Heo, E.; Kim, D.K.; Oh, S.H.; Lee, J.K.; Park, J.H.; Chung, H.S. Effect of Prophylactic Use of Silymarin on Anti-Tuberculosis Drugs Induced Hepatotoxicity. Tuberc. Respir. Dis. 2017, 80, 265–269. [Google Scholar] [CrossRef]
- Moayedi, B.; Gharagozloo, M.; Esmaeil, N.; Maracy, M.R.; Hoorfar, H.; Jalaeikar, M. A Randomized Double-Blind, Placebo-Controlled Study of Therapeutic Effects of Silymarin in β-Thalassemia Major Patients Receiving Desferrioxamine. Eur. J. Haematol. 2013, 90, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Gharagozloo, M.; Karimi, M.; Amirghofran, Z. Immunomodulatory Effects of Silymarin in Patients with β-Thalassemia Major. Int. Immunopharmacol. 2013, 16, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Hagag, A.A.; Elfrargy, M.S.; Gazar, R.A.; El-Lateef, A.E.A. Therapeutic Value of Combined Therapy with Deferasirox and Silymarin on Iron Overload in Children with Beta Thalassemia. Mediterr. J. Hematol. Infect. Dis. 2013, 5, 1–7. [Google Scholar] [CrossRef]
- Balouchi, S.; Gharagozloo, M.; Esmaeil, N.; Mirmoghtadaei, M.; Moayedi, B. Serum Levels of TGFβ, IL-10, IL-17, and IL-23 Cytokines in β-Thalassemia Major Patients: The Impact of Silymarin Therapy. Immunopharmacol. Immunotoxicol. 2014, 36, 271–274. [Google Scholar] [CrossRef]
- Hagag, A.; Elfaragy, M.; Elrifaey, S.; Abd El-Lateef, A. Therapeutic Value of Combined Therapy with Deferiprone and Silymarin as Iron Chelators in Egyptian Children with Beta Thalassemia Major. Infect. Disord.-Drug Targets 2015, 15, 189–195. [Google Scholar] [CrossRef] [PubMed]
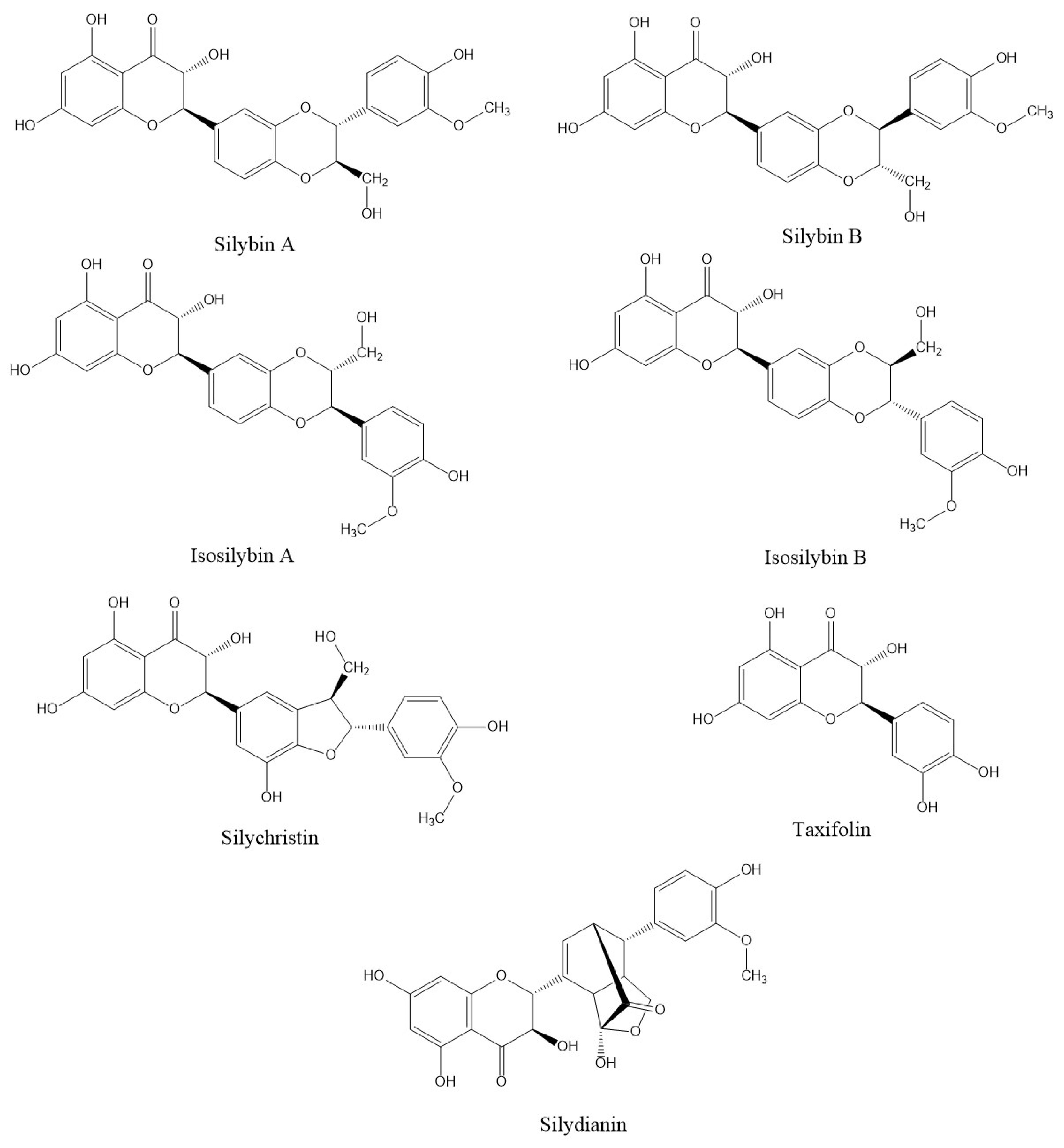
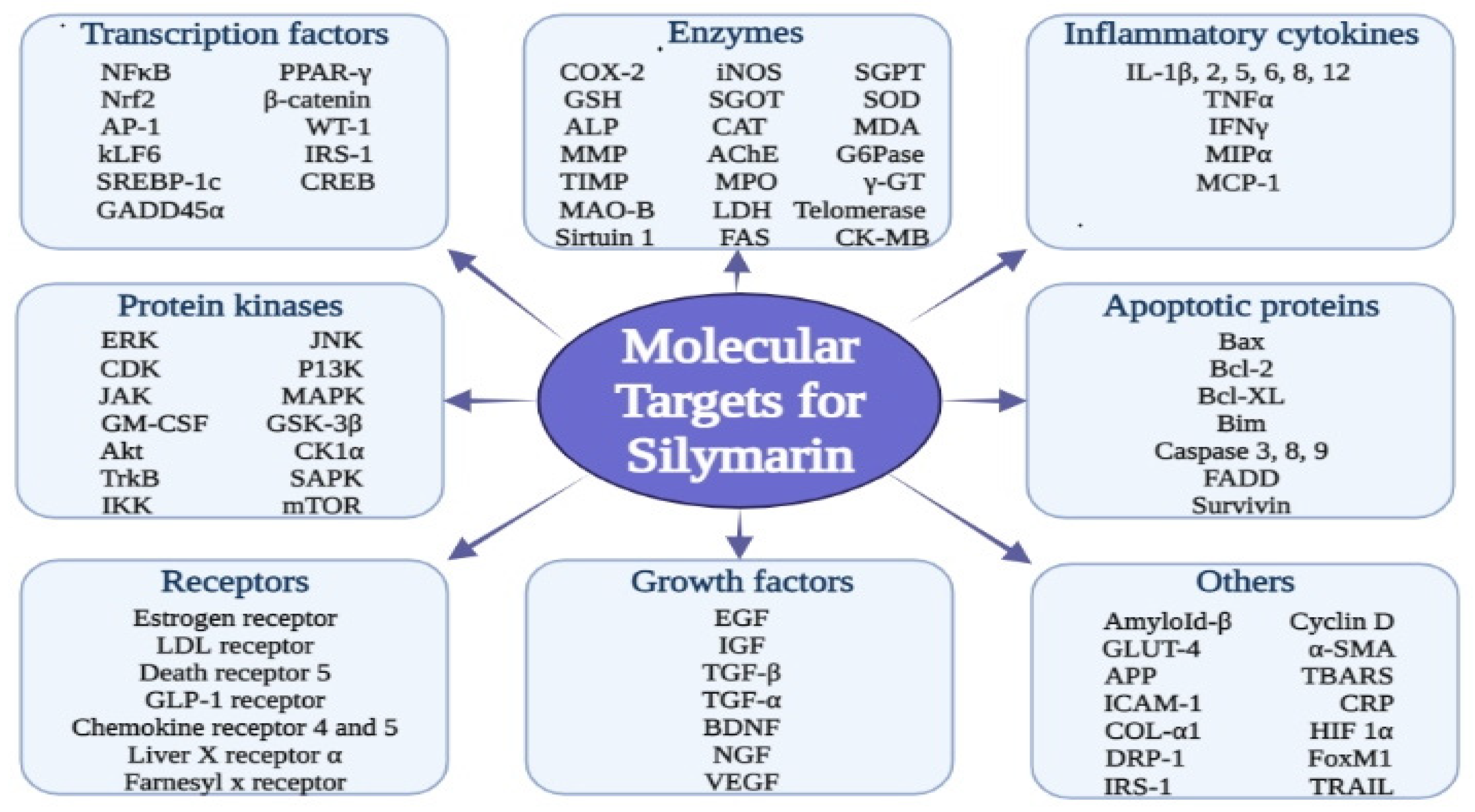
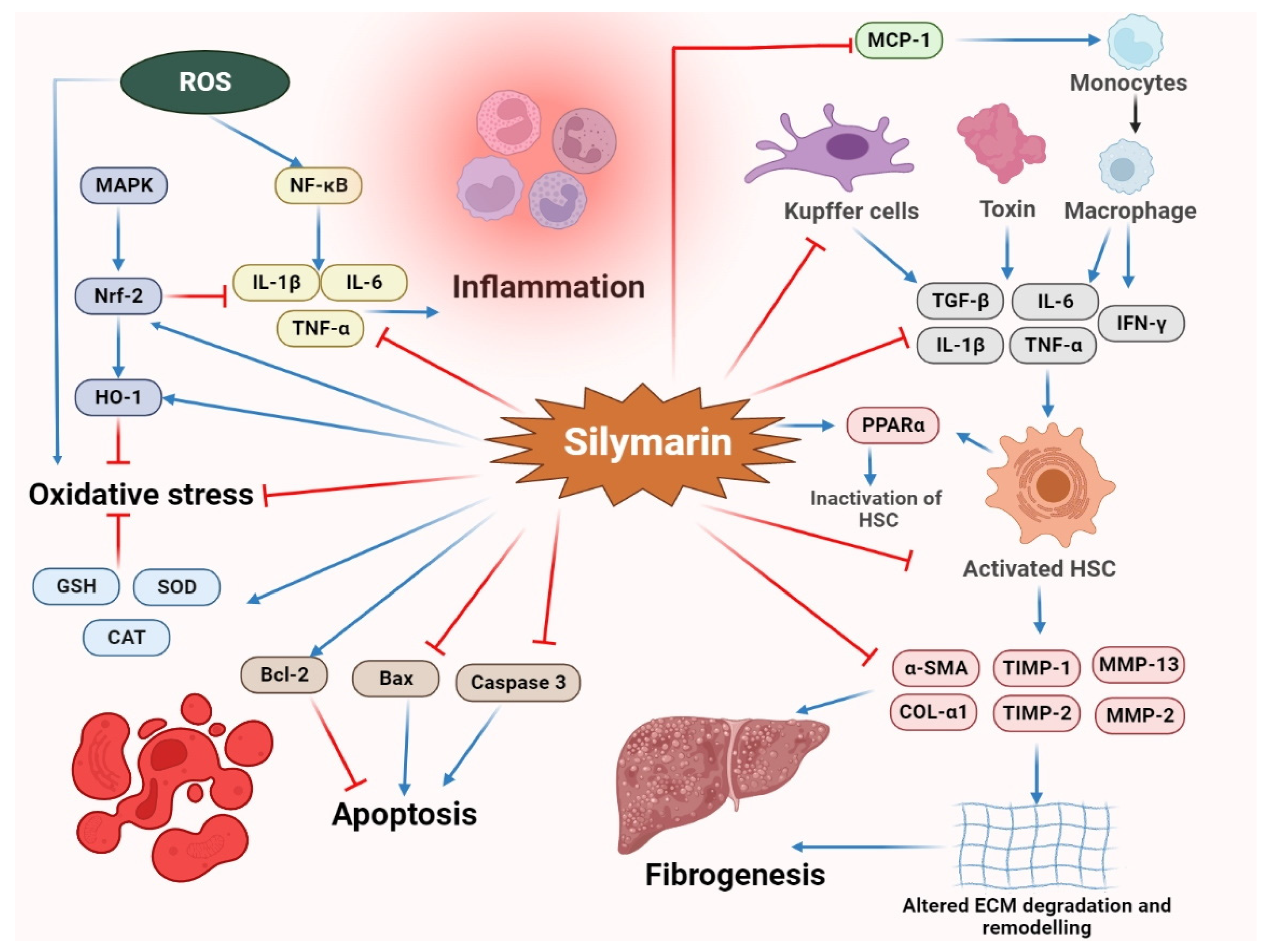
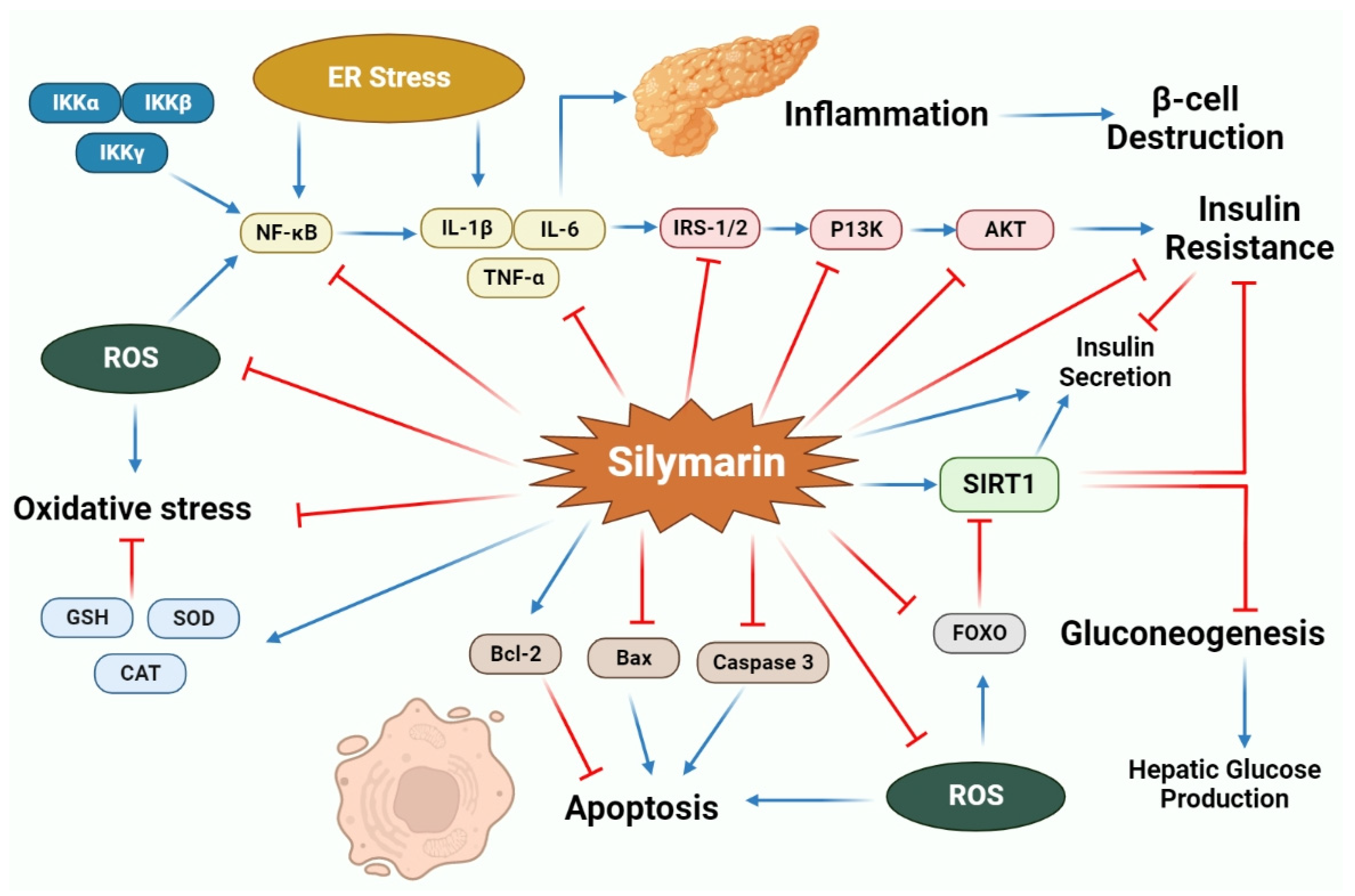
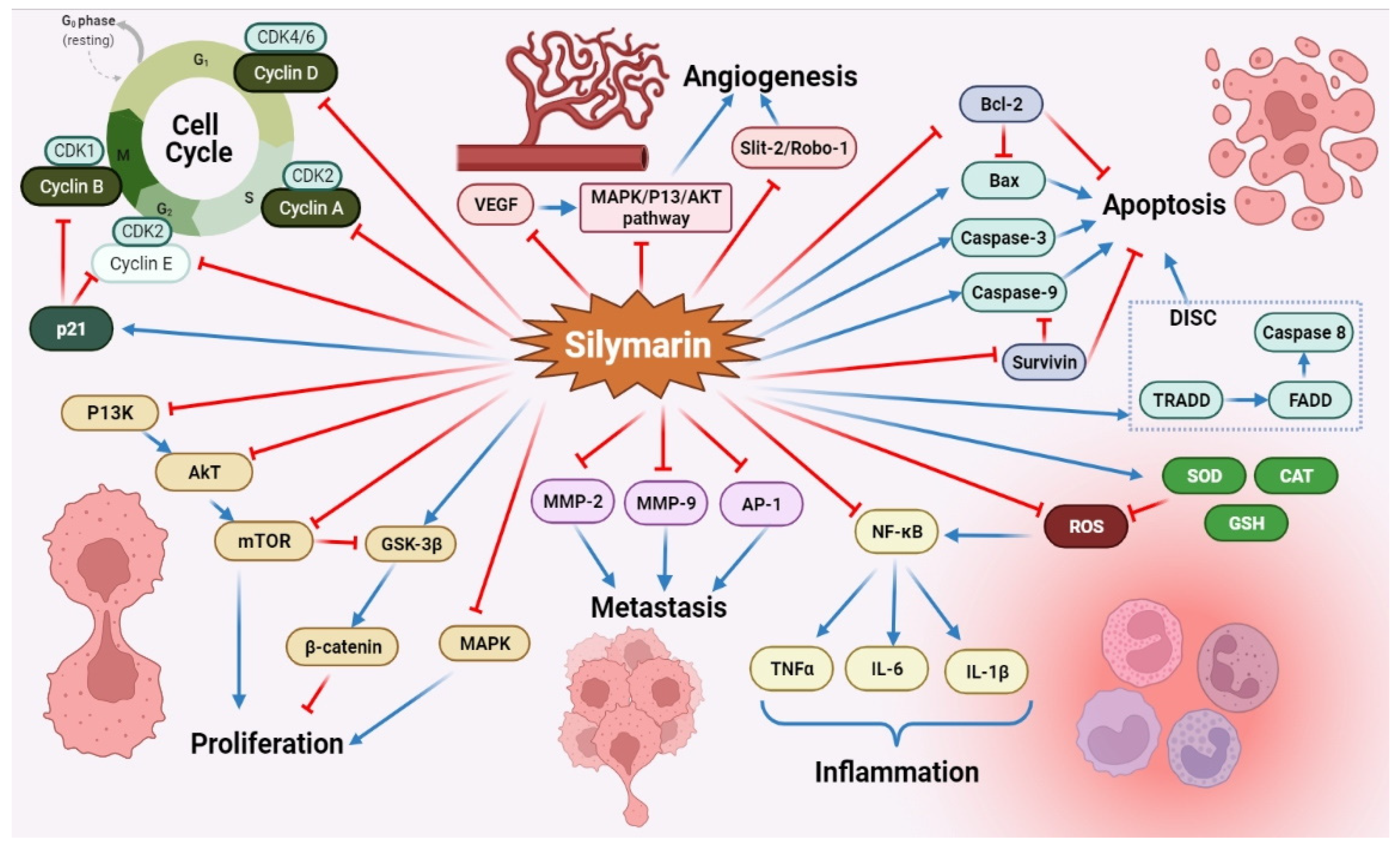
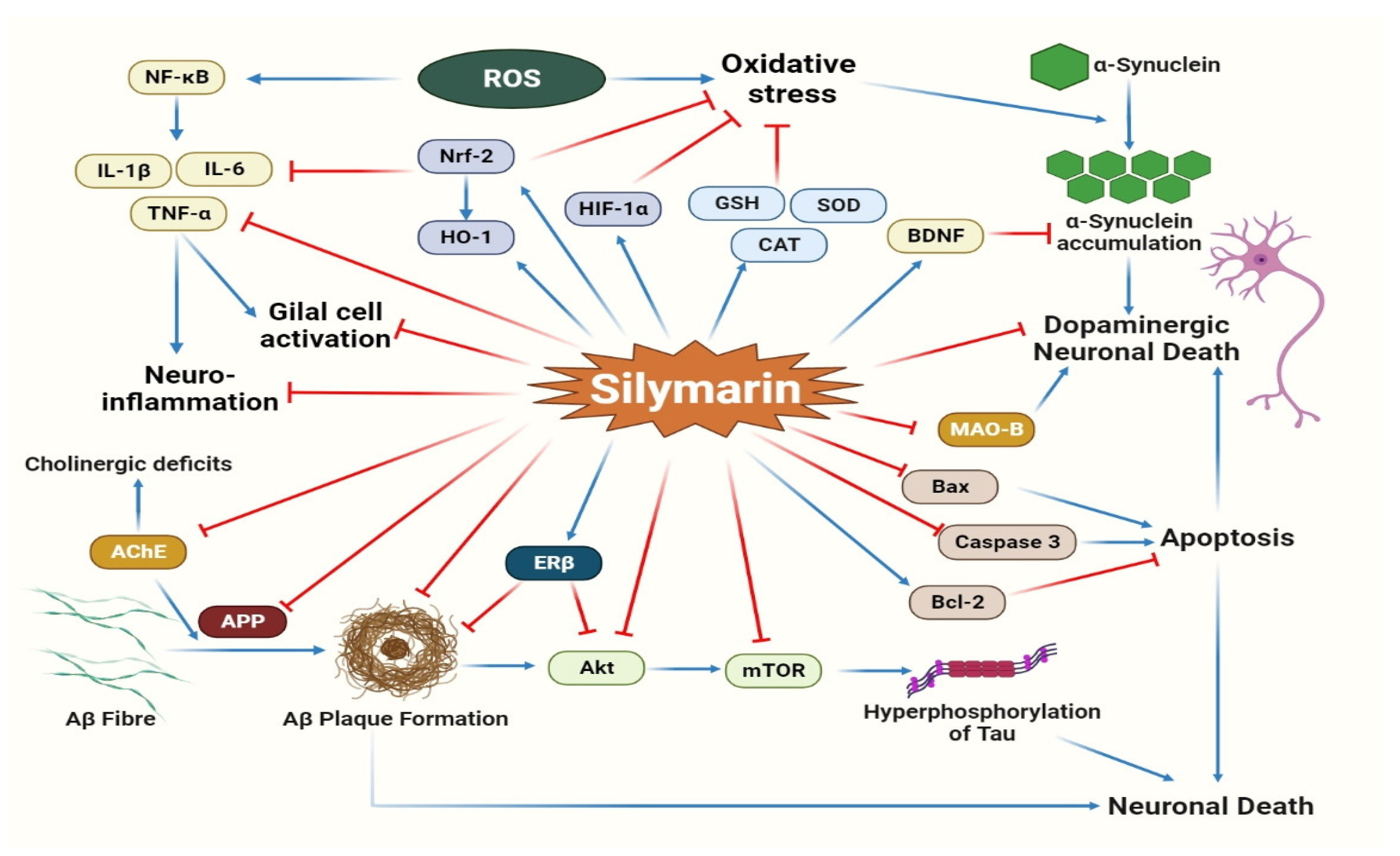
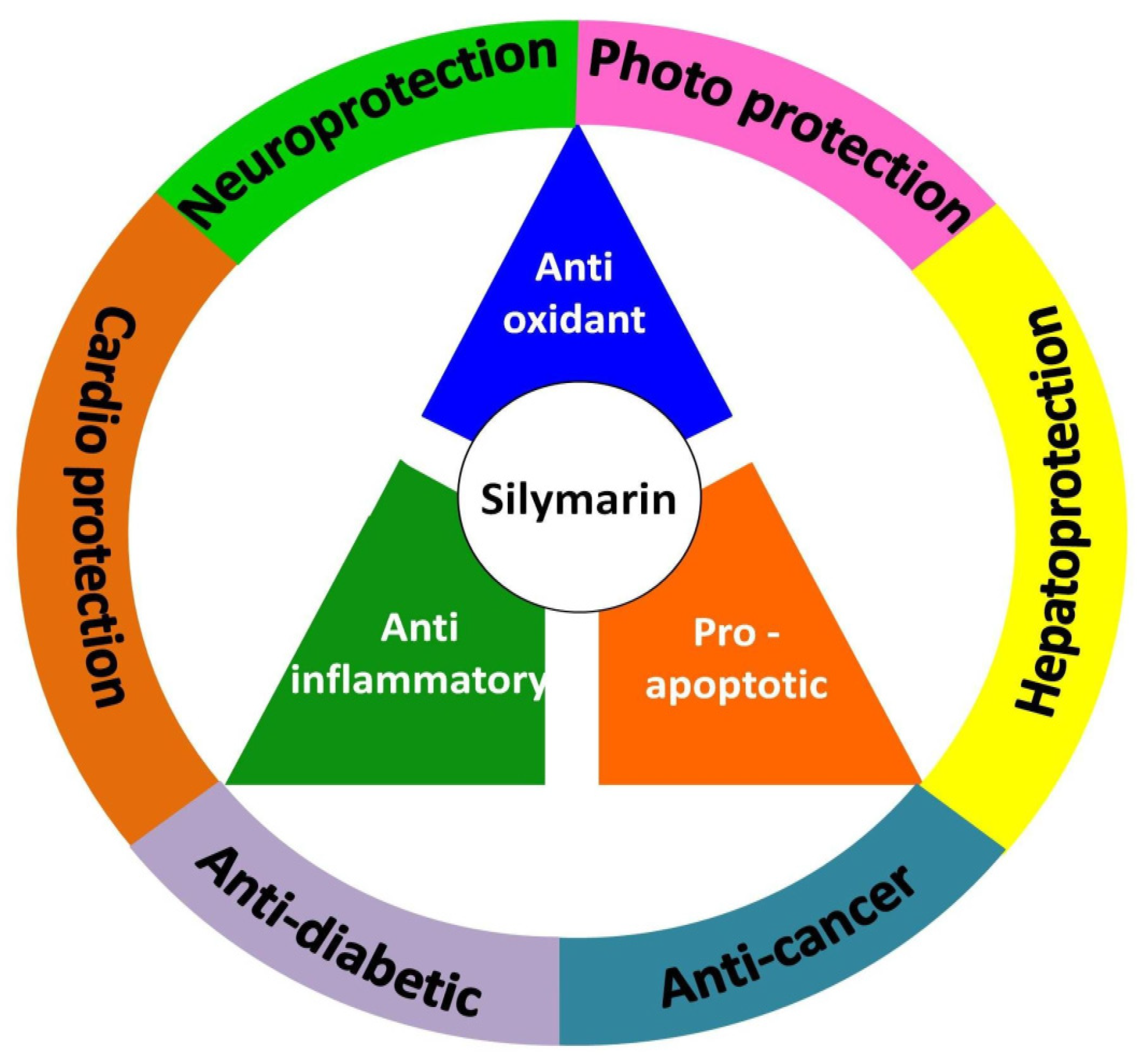
| Study Model | Dose/Concentration Used | Possible Target Site/Mechanism of Action | Reference |
|---|---|---|---|
| CCl4-induced hepatotoxicity | 200 mg/kg p.o |
| [41] |
| CCl4-induced hepatotoxicity | 200 mg/kg p.o |
| [42] |
| Valproic acid-induced hepatotoxicity | 25 and 50 mg/kg |
| [43] |
| Thioacetamide-induced hepatotoxicity | 100 mg/kg p.o |
| [44] |
| Fructose-induced NAFLD | 400 mg/kg/day p.o. |
| [39] |
| Diclofenac-induced hepatotoxicity | 200 mg/kg p.o. |
| [45] |
| CCl4-induced hepatotoxicity and HSC cells | 20 and 100 mg/kg p.o. |
| [46] |
| CCl4-induced hepatotoxicity | 100 mg/kg i.p. |
| [47] |
| CCl4-induced hepatotoxicity | 100 mg/kg i.p. 5 times a week for 4 weeks |
| [40] |
| Acetaminophen-induced hepatotoxicity | 200 mg/kg p.o. |
| [48] |
| NASH rats | 200 mg/kg p.o |
| [49] |
| HFD-induced NAFLD | 5–10 mL/kg p.o. for 8 weeks |
| [50] |
| MCD diet-induced NASH | 105 mg/kg/day p.o. for 8 weeks |
| [51] |
| MCD diet-induced NASH | — |
| [52] |
| Restraint of stress-induced acute liver injury | 100 mg/kg |
| [53] |
| HepG2 cells and HFD-induced liver inflammation | 50 or 100 mg/kg per day |
| [54] |
| — | 600 mg/kg per day p.o. for 10 days |
| [55] |
| HepG2 cells (Benzo[a]pyrene-induced hepatotoxicity) | 0–40 µM |
| [56] |
| CCl4-induced hepatoxicity | 50 and 200 mg/kg |
| [57] |
| Zidovudine and isoniazid-induced liver toxicity | 100 mg/kg |
| [58] |
| Type of Cancer | Study Model | Dose/Concentration Used | Possible Target Site/Mechanism of Action | Reference |
|---|---|---|---|---|
| Bladder cancer | T24 and UM-UC-3 cells | 10 μm |
| [120] |
| Breast cancer | MDA-MB-231 and MCF-7 breast cancer cells in vivo xenograft tumor model | 0–200 25 μg/mL and 50 mg/kg |
| [121] |
| MCF-7 cells | 10–100 μM |
| [122] | |
| 4T1 tumor-bearing BAlB/c mice and myeloid-derived suppressor cells | 150 mg/kg |
| [123] | |
| MCH-7 and MDA-MB-231 cells | 30–90 μM 150–250 μM |
| [124] | |
| MDA-MB-231 cells | 0–400 μM |
| [125] | |
| MDA-MB-231 cells | 50–350 μM |
| [126] | |
| MCH-7 cells | — |
| [127] | |
| MCF-7 cells | — |
| [128] | |
| MDA-MB-231 and T-47D | — |
| [129] | |
| Colorectal cancer | Azoxymethane-induced colon carcinogenesis model | 300 mg/kg p.o. for 7 days |
| [130] |
| SW480 and SW620 cells | 300 μM |
| [131] | |
| HCT116 and SW480 cells | 0–200 μg/mL |
| [132] | |
| HCT116, SW480, LoVo and HT-29 cells | — |
| [133] | |
| Xenograft tumor model | — |
| [134] | |
| Gastric cancer | ASG human gastric cancer cells In vivo xenograft tumor model | 20–120 μg/mL and 100 mg/kg |
| [135] |
| BGC-823 cells | 0–200 µM |
| [136] | |
| MGC803 cells | 0–200 µM |
| [137] | |
| Hepatocellular carcinoma | Hep G2 cells | 0–200 μM |
| [138] |
| HCC cells | — |
| [139] | |
| Hep G2 cells | 12.1–482.4 µg/mL |
| [140] | |
| Hep G2 cell model and tumor xenograft model | 50–200 µM |
| [141] | |
| N-nitrosodiethyl-amine-induced liver cancer | 1000 ppm |
| [142] | |
| Laryngeal carcinoma | Hep 2 cells | 60–300 μM |
| [143] |
| Leukemia | K562 cells | 0–100 μg/mL |
| [144] |
| Lung cancer | LA795, NCI-H1299 cells and tumor xenograft models | 100 mg/kg |
| [145] |
| Oral cancer | HSC-4, YD15 and Ca9.22 cells tumor xenograft models | 40–80 μg/mL 200 mg/kg/day p.o. for 5 weeks |
| [146] |
| MC3 and HN22 cells | — |
| [147] | |
| SCC-25 cells | 50 and 100 μM |
| [148] | |
| Ovarian cancer | A2780s and PA-1 cells | 50 and 100 µg/mL |
| [149] |
| Prostate cancer | PC-3 and DU-145 cells | — |
| [129] |
| DU-145 cells | 15.6 to 1000 μM |
| [150] | |
| Skin cancer | DMBA–TPA-induced skin papilloma and A431 cells | 12.5–50 µM |
| [151] |
| A375 and Hs294t cells | 0–40 μg/mL |
| [152] | |
| DMBA-TPA 2-stage skin carcinogenesis | 9 mg topically |
| [153] | |
| A375 and Hs294t cells tumor xenograft models | 0–60 μg/mL and 500 mg/kg |
| [154] | |
| — | MCF-7 and NCIH-23 cell lines | 12.5–200 µg/mL |
| [155] |
| N-Butyl-N-(4-hydroxybutyl) nitrosamine-induced carcinogenesis | 1000 ppm |
| [156] | |
| U266 MM cell | 50–200 μM |
| [157] |
| Type of Cancer | Cellular Pathway Modulated | References |
|---|---|---|
| Bladder cancer | ↓ PI3K-PKB/Akt signaling pathway | [120] |
| Cervical/ovarian cancer | ↓ MAPK/ERK1/2 and MAPK/p38 signaling pathway ↓ Bcl-2-mediated anti-apoptosis | [158] |
| Prostate cancer | ↓ CDK, MAPK/ERK1/2, and Wnt/β-catenin signaling pathway | [129,159,160] |
| Skin cancer | ↑ p53-mediated apoptosis and MAPK/p38 signaling pathway ↓ MAPK/ERK1/2, MAPK/JNK1 and Wnt/β-catenin signaling pathway | [151,161,162,163] |
| Lung cancer | ↑ Multiple MAPK signaling pathways ↓ CDK signaling pathway | [145,164] |
| Liver cancer (Hepatocellular carcinoma) | ↓ Bcl2-mediated anti-apoptosis ↑ p53, Bax and APAF-1-mediated apoptosis ↓ Slit-2/Robo-1 pathway and Notch pathway | [140,141,165] |
| Breast cancer | ↓ MEK/ERK and Wnt/β-catenin signaling pathway ↓ Bcl-2-mediated anti-apoptosis | [121,128,129] |
| Oral cancer | ↑ Bim-mediated apoptosis ↓ MAPK/ERK1/2 signaling pathway | [147] |
| Colorectal cancer | ↓ PP2A/AKT/mTOR, MAPK/ERK1/2 and MAPK/p38 signaling pathway ↓ Bcl-2-mediated anti-apoptosis ↑ Bax-mediated apoptosis | [130,133,134] |
| Gastric cancer | ↓ MAPK/ERK signaling pathway ↑ MAPK/p38 signaling pathway | [128,135] |
| Peripheral blood cancer | ↓ PI3K-PKB/Akt signaling pathway ↑ Caspase-3-mediated apoptosis | [166] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wadhwa, K.; Pahwa, R.; Kumar, M.; Kumar, S.; Sharma, P.C.; Singh, G.; Verma, R.; Mittal, V.; Singh, I.; Kaushik, D.; et al. Mechanistic Insights into the Pharmacological Significance of Silymarin. Molecules 2022, 27, 5327. https://doi.org/10.3390/molecules27165327
Wadhwa K, Pahwa R, Kumar M, Kumar S, Sharma PC, Singh G, Verma R, Mittal V, Singh I, Kaushik D, et al. Mechanistic Insights into the Pharmacological Significance of Silymarin. Molecules. 2022; 27(16):5327. https://doi.org/10.3390/molecules27165327
Chicago/Turabian StyleWadhwa, Karan, Rakesh Pahwa, Manish Kumar, Shobhit Kumar, Prabodh Chander Sharma, Govind Singh, Ravinder Verma, Vineet Mittal, Inderbir Singh, Deepak Kaushik, and et al. 2022. "Mechanistic Insights into the Pharmacological Significance of Silymarin" Molecules 27, no. 16: 5327. https://doi.org/10.3390/molecules27165327
APA StyleWadhwa, K., Pahwa, R., Kumar, M., Kumar, S., Sharma, P. C., Singh, G., Verma, R., Mittal, V., Singh, I., Kaushik, D., & Jeandet, P. (2022). Mechanistic Insights into the Pharmacological Significance of Silymarin. Molecules, 27(16), 5327. https://doi.org/10.3390/molecules27165327








