A Convenient, Rapid, Conventional Heating Route to MIDA Boronates
Abstract
1. Introduction
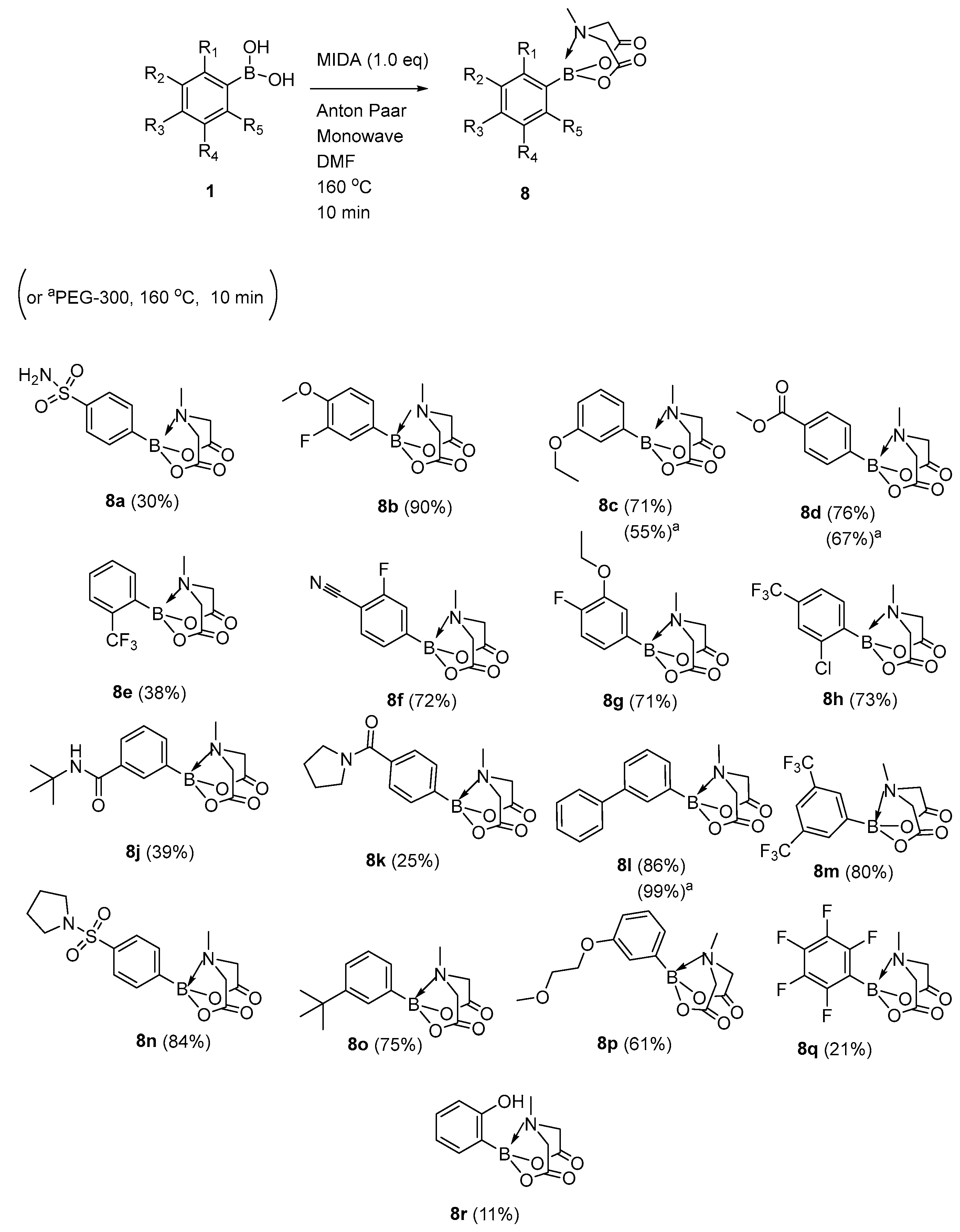
2. Results and Discussion
3. Materials and Methods
3.1. General Conditions
3.2. Experimental Procedures
MIDA Synthesis in DMF as Solvent
3.3. Molecules Synthesised
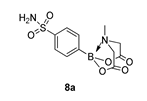



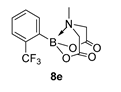

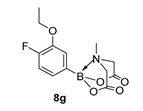
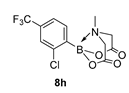
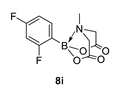
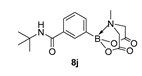
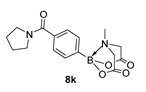

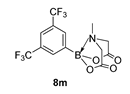


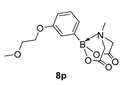
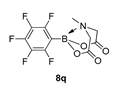
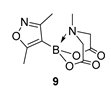

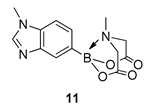
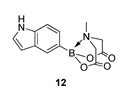
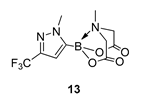
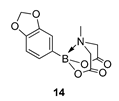

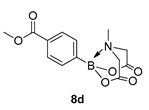
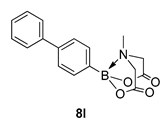
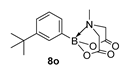




4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Byrne, F.P.; Jin, S.; Paggiola, G.; Petchey, T.H.M.; Clark, J.H.; Farmer, T.J.; Hunt, A.J.; Mcelroy, C.R.; Sherwood, J. Tools and techniques for solvent selection: Green solvent selection guides. Sustain. Chem. Processes 2016, 4, 7. [Google Scholar] [CrossRef]
- Coby, C.J.; Tu, W.-C.; Levers, O.; Bröhl, A.; Hallett, J.P. Green and Sustainable Solvents in Chemical Processes. Chem. Rev. 2018, 118, 747–800. [Google Scholar] [CrossRef]
- Guariento, S.; Biagetti1, M.; Ronchi, P. Non-regioselective functionalization: An underestimate chemical diversity generator in medicinal chemistry. Future Med. Chem. 2021, 13, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Boonseng, S.; Kemmitt, P.D.; Felix, R.; Coles, S.J.; Tizzard, G.J.; Williams, G.; Simmonds, O.; Harvey, J.-L.; Atack, J.; et al. Combining Sanford Arylations on Benzodiazepines with the Nuisance Effect. Adv. Synth. Catal. 2017, 359, 3261–3269. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.; Rathnam, R.P.; Chowdhry, B.Z. 1,4-benzodiazepin-2-ones in medicinal chemistry. Future Med. Chem. 2010, 2, 1441–1449. [Google Scholar] [CrossRef]
- Spencer, J.; Rathnam, R.P.; Harvey, A.L.; Clements, C.J.; Clark, R.L.; Barrett, M.P.; Wong, P.E.; Male, L.; Coles, S.J.; MacKay, S.P. Synthesis and biological evaluation of 1,4-benzodiazepin-2-ones with antitrypanosomal activity. Bioorg. Med. Chem. 2011, 19, 1802–1815. [Google Scholar] [CrossRef] [PubMed]
- Khan Tareque, R.; Hassell-Hart, S.; Krojer, T.; Bradley, A.; Velupillai, S.; Talon, R.; Fairhead, M.; Day, I.J.; Bala, K.; Felix, R.; et al. Deliberately Losing Control of C−H Activation Processes in the Design of Small-Molecule-Fragment Arrays Targeting Peroxisomal Metabolism. ChemMedChem 2020, 15, 2513–2520. [Google Scholar] [CrossRef]
- Suk, J.L.; Gray, K.C.; Paek, J.S.; Burke, M.D. Simple, efficient, and modular syntheses of polyene natural products via iterative cross-coupling. J. Am. Chem. Soc. 2008, 130, 466–468. [Google Scholar] [CrossRef]
- Blair, D.J.; Chitti, S.; Trobe, M.; Kostyra, D.M.; Haley, H.M.S.; Hansen, R.L.; Ballmer, S.G.; Woods, T.J.; Wang, W.; Mubayi, V.; et al. Automated iterative Csp3–C bond formation. Nature 2022, 604, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Lehman, J.W.; Blair, D.J.; Burke, M.D. Burke mIDA review nat rev.pdf. Nat. Rev. Chem. 2018, 2, 0115. [Google Scholar] [CrossRef]
- Yang, L.; Tan, D.H.; Fan, W.X.; Liu, X.G.; Wu, J.Q.; Huang, Z.S.; Li, Q.; Wang, H. Photochemical Radical C–H Halogenation of Benzyl N-Methyliminodiacetyl (MIDA) Boronates: Synthesis of α-Functionalized Alkyl Boronates. Angew. Chem. Int. Ed. 2021, 60, 3454–3458. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.J.; Lee, C.Y.; Cheon, C.H. General methods for synthesis of N-methyliminodiacetic acid boronates from unstable ortho-phenolboronic acids. Adv. Synth. Catal. 2014, 356, 1767–1772. [Google Scholar] [CrossRef]
- Khanizeman, R.N.; Barde, E.; Bates, R.W.; Guérinot, A.; Cossy, J. Modular access to triarylethylene units from arylvinyl MIDA boronates using a regioselective heck coupling. Org. Lett. 2017, 19, 5046–5049. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.B.; Lee, I.H.; Lee, J.; Choi, I.; Choi, T.L. A Rational Design of Highly Controlled Suzuki-Miyaura Catalyst-Transfer Polycondensation for Precision Synthesis of Polythiophenes and Their Block Copolymers: Marriage of Palladacycle Precatalysts with MIDA-Boronates. J. Am. Chem. Soc. 2018, 140, 4335–4343. [Google Scholar] [CrossRef] [PubMed]
- St. Denis, J.D.; Scully, C.C.G.; Lee, C.F.; Yudin, A.K. Development of the direct Suzuki-Miyaura cross-coupling of primary B-alkyl MIDA-boronates and aryl bromides. Org. Lett. 2014, 16, 1338–1341. [Google Scholar] [CrossRef]
- Isley, N.A.; Gallou, F.; Lipshutz, B.H. Transforming Suzuki-Miyaura cross-couplings of MIDA boronates into a green technology: No organic solvents. J. Am. Chem. Soc. 2013, 135, 17707–17710. [Google Scholar] [CrossRef] [PubMed]
- Close, A.J.; Kemmitt, P.; Mark Roe, S.; Spencer, J. Regioselective routes to orthogonally-substituted aromatic MIDA boronates. Org. Biomol. Chem. 2016, 14, 6751–6756. [Google Scholar] [CrossRef] [PubMed]
- Hyodo, K.; Suetsugu, M.; Nishihara, Y. Diborylation of alkynyl MIDA boronates and sequential chemoselective suzuki-miyaura couplings: A formal carboborylation of alkynes. Org. Lett. 2014, 16, 440–443. [Google Scholar] [CrossRef]
- McLaughlin, M.G.; McAdam, C.A.; Cook, M.J. midas SM couplings.pdf. Org. Lett. 2015, 17, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Grob, J.E.; Dechantsreiter, M.A.; Tichkule, R.B.; Connolly, M.K.; Honda, A.; Tomlinson, R.C.; Hamann, L.G. One-pot C-N/C-C cross-coupling of methyliminodiacetic acid boronyl arenes enabled by protective enolization. Org. Lett. 2012, 14, 5578–5581. [Google Scholar] [CrossRef] [PubMed]
- Muir, C.W.; Vantourout, J.C.; Isidro-Llobet, A.; Macdonald, S.J.F.; Watson, A.J.B. One-Pot Homologation of Boronic Acids: A Platform for Diversity-Oriented Synthesis. Org. Lett. 2015, 17, 6030–6033. [Google Scholar] [CrossRef] [PubMed]
- Castro-Godoy, W.D.; Schmidt, L.C.; Argüello, J.E. A Green Alternative for the Conversion of Arylboronic Acids/Esters into Phenols Promoted by a Reducing Agent, Sodium Sulfite. Eur. J. Org. Chem. 2019, 2019, 3035–3039. [Google Scholar] [CrossRef]
- Ivon, Y.M.; Mazurenko, I.V.; Kuchkovska, Y.O.; Voitenko, Z.V.; Grygorenko, O.O. Formyl MIDA Boronate: C1 Building Block Enables Straightforward Access to α-Functionalized Organoboron Derivatives. Angew. Chem. Int. Ed. 2020, 59, 18016–18022. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Wang, L.; Aminoleslami, N.; Lao, Y.; Yagel, C.; Sharma, A. A modular and concise approach to MIDA acylboronates: Via chemoselective oxidation of unsymmetrical geminal diborylalkanes: Unlocking access to a novel class of acylborons. Chem. Sci. 2019, 10, 4684–4691. [Google Scholar] [CrossRef]
- Colgin, N.; Flinn, T.; Cobb, S.L. Synthesis and properties of MIDA boronate containing aromatic amino acids: New peptide building blocks. Org. Biomol. Chem. 2011, 9, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Grob, J.E.; Nunez, J.; Dechantsreiter, M.A.; Hamann, L.G. One-pot reductive amination and Suzuki-Miyaura cross-coupling of formyl aryl and heteroaryl MIDA boronates in array format. J. Org. Chem. 2011, 76, 4930–4940. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, A.F.; North, R.; Eisenbeis, S. Trace Level Quantification of Derivatized Boronic Acids by LC/MS/MS. Org. Process Res. Dev. 2019, 23, 88–92. [Google Scholar] [CrossRef]
- Boureghda, C.; Macé, A.; Berrée, F.; Roisnel, T.; Debache, A.; Carboni, B. Ene reactions of 2-borylated α-methylstyrenes: A practical route to 4-methylenechromanes and derivatives. Org. Biomol. Chem. 2019, 17, 5789–5800. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.M.; Chen, P.J.; Klubnick, J.; Blair, D.J.; Burke, M.D. A Mild Method for Making MIDA Boronates. Org. Lett. 2020, 22, 9408–9414. [Google Scholar] [CrossRef] [PubMed]
- Close, A.J.; Kemmitt, P.; Emmerson, M.K.; Spencer, J. Microwave-mediated synthesis of N-methyliminodiacetic acid (MIDA) boronates. Tetrahedron 2014, 70, 9125–9131. [Google Scholar] [CrossRef][Green Version]
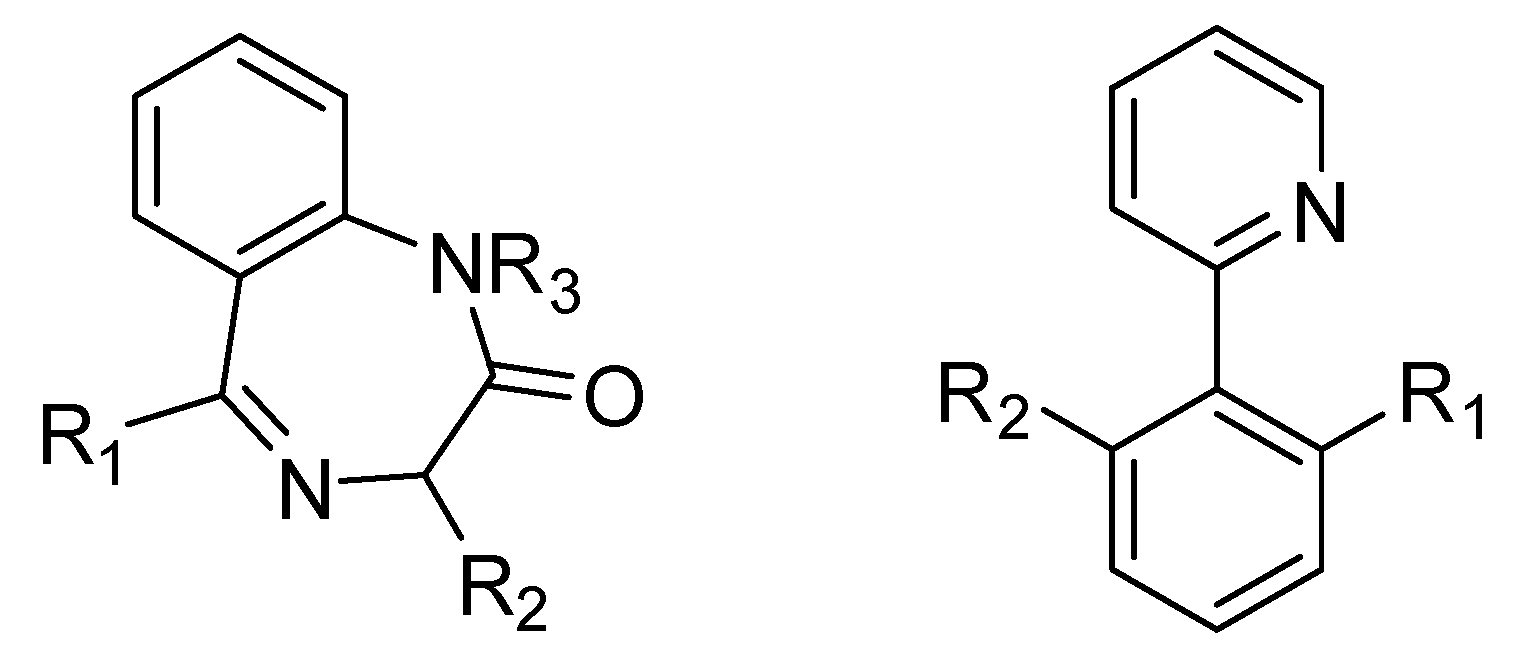
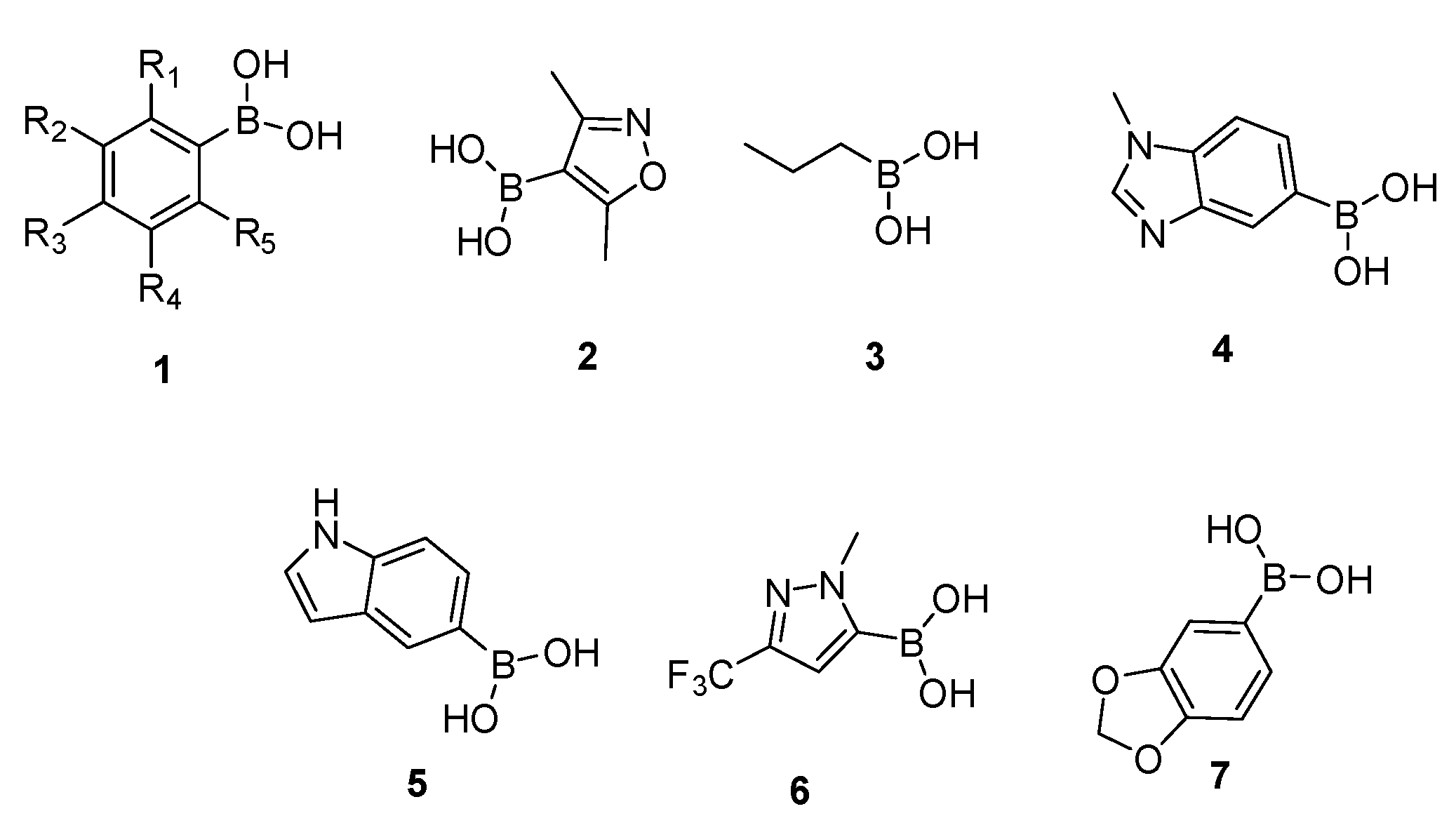

| Compound | Mw | Eq | Mmol | Mg | ρ | µL |
|---|---|---|---|---|---|---|
| Boronic acid | - | 1.0 | 1.0 | - | - | - |
| Methyliminodiacetic acid (MIDA) | 147.13 | 1.0 | 1.0 | - | - | - |
| PEG-300 | - | - | - | - | - | 1000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McGown, A.; Edmonds, A.K.; Guest, D.; Holmes, V.L.; Dadswell, C.; González-Méndez, R.; Goodall, C.A.I.; Bagley, M.C.; Greenland, B.W.; Spencer, J. A Convenient, Rapid, Conventional Heating Route to MIDA Boronates. Molecules 2022, 27, 5052. https://doi.org/10.3390/molecules27165052
McGown A, Edmonds AK, Guest D, Holmes VL, Dadswell C, González-Méndez R, Goodall CAI, Bagley MC, Greenland BW, Spencer J. A Convenient, Rapid, Conventional Heating Route to MIDA Boronates. Molecules. 2022; 27(16):5052. https://doi.org/10.3390/molecules27165052
Chicago/Turabian StyleMcGown, Andrew, Anthony K. Edmonds, Daniel Guest, Verity L. Holmes, Chris Dadswell, Ramón González-Méndez, Charles A. I. Goodall, Mark C. Bagley, Barnaby W. Greenland, and John Spencer. 2022. "A Convenient, Rapid, Conventional Heating Route to MIDA Boronates" Molecules 27, no. 16: 5052. https://doi.org/10.3390/molecules27165052
APA StyleMcGown, A., Edmonds, A. K., Guest, D., Holmes, V. L., Dadswell, C., González-Méndez, R., Goodall, C. A. I., Bagley, M. C., Greenland, B. W., & Spencer, J. (2022). A Convenient, Rapid, Conventional Heating Route to MIDA Boronates. Molecules, 27(16), 5052. https://doi.org/10.3390/molecules27165052







