Recent Advances in Prodigiosin as a Bioactive Compound in Nanocomposite Applications
Abstract
:1. Introduction
2. Pyrrolic Compounds Produced by Microorganisms: Serratia marcescens
Structure of Prodigiosin
3. Prodigiosin Producing Bacteria
3.1. Prodigiosin Production
3.2. Prodigiosin Separation
4. Biological Activity of Prodigiosin
4.1. Antimicrobial
4.2. Antioxidant
4.3. Antitumoral
| Cancer Type | Cell Line | Mechanism | IC50 | Units | Reference |
|---|---|---|---|---|---|
| Blood/Leukemia | K562 | Increased activity of caspases -3, -8, -9, reactive oxygen species, inhibition of autophagy and apoptosis induction. | >500 | µM | [93] |
| HL-60 | Apoptosis induction | 1.7 | µg/mL | [100] | |
| Wt-p53Molt-4 | Caspase-3-dependent apoptosis | 1.3 | µM | [101] | |
| Brain | GBM8401 | ER stress/autophagy | 7.36 | µM | [102] |
| U87MG | ER stress/autophagy | 12.29 | µM | ||
| Breast | MDA-MB-231 | ER stress; Wnt/β-catenin; JNK/MAPK/RAD51 | 62.52 | nM | [98] |
| MDA-MB-468 | Inhibit Wnt/β-catenin | 261.2 | nM | ||
| MCF-7 | Apoptosis induction | 5.1 | µg/mL | [100] | |
| MCF-7 | Mitogen-activated protein kinase regulators, pH modulators, DNA cleavage agents and cell cycle inhibitors | 0.04 | µg/mL | [94] | |
| MDA-MB-231 | Decreased the levels of mTOR and HSP90α expression and transcription | 2.1 | nM | [99] | |
| Urothelial | RT-112 | Dysregulation of lysosomal function and reduction of cathepsin B and L activity | 675 | nM | [23] |
| Colorectal | DLD1 | c-Jun/ΔNp73/p73/apoptosis; Lysosomal acidification | >1.6 | µM | [103,104] |
| HCT116 | c-Jun/ΔNp73 p73 activation | 4 | µM | ||
| SW480 | c-Jun/ΔNp73 p73 activation | µM | |||
| SW-620 | Apoptosis | 0.273 | µM | ||
| HCT116; SW480 | Blocking autophagosome–lysosome fusion and maturation of lysosomal cathepsin | >0.1 | µM | [92] | |
| WiDr | Mitogen-activated protein kinase regulators, pH modulators, DNA cleavage agents and cell cycle inhibitors | 0.05 | µg/mL | [97] | |
| Liver | HepG2 | Antiproliferative effects | 12.64 | µg/mL | [84] |
| HepG2 | Mitogen-activated protein kinase regulators, pH modulators, DNA cleavage agents and cell cycle inhibitors | 0.04 | µg/mL | [95] | |
| Lung | A549 | PI3K-p85/Akt/mTOR; PKB/SKP2/p27 | 10 | µM | [105] |
| CNE2, NP69 | PKB/SKP2/p27 | 4 and 0.35 | mg/L | [106] | |
| NCHI-292 | Apoptosis induction | 3.6 | µg/mL | [100] | |
| A549 | Mitogen-activated protein kinase regulators, pH modulators, DNA cleavage agents and cell cycle inhibitors | 0.06 | µg/mL | [95] | |
| Prostate | PC3 | Intrinsic apoptosis | >10 | µg/mL | [96] |
| Trophoblast | JEG3 | Intrinsic apoptosis | >10 | µg/mL | |
| Uterus | Hela | Antiproliferative effects | 12.75 | µg/mL | [84] |
| Gingival squamous carcinoma | OECM1 | Cyclin D1 inhibition, arresting cell cycle in G0/G1 phase | 1.59 ± 0.77 | μM | [107] |
| Tongue | SAS | cyclin D1 inhibition, arresting cell cycle in G0/G1 phase | 3.25 ± 0.49 | μM | |
| Uterus | Hela | Intrinsic apoptosis | 0.5–2.1 | μg/mL | [108] |
4.4. Antiprotozoal
4.5. Antiviral
5. Recent Advances and Applications of Prodigiosin Bionanocomposites
6. Concluding Remarks and Futures Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gholamali, I.; Yadollahi, M. Bio-nanocomposite Polymer Hydrogels Containing Nanoparticles for Drug Delivery: A Review. Regen. Eng. Transl. Med. 2021, 7, 129–146. [Google Scholar] [CrossRef]
- Jafarzadeh, S.; Nafchi, A.M.; Salehabadi, A.; Oladzad-Abbasabadi, N.; Jafari, S.M. Application of bio-nanocomposite films and edible coatings for extending the shelf life of fresh fruits and vegetables. Adv. Colloid Interface Sci. 2021, 291, 102405. [Google Scholar] [CrossRef] [PubMed]
- Shahvalizadeh, R.; Ahmadi, R.; Davandeh, I.; Pezeshki, A.; Moslemi, S.A.S.; Karimi, S.; Rahimi, M.; Hamishehkar, H.; Mohammadi, M. Antimicrobial bio-nanocomposite films based on gelatin, tragacanth, and zinc oxide nanoparticles–Microstructural, mechanical, thermo-physical, and barrier properties. Food Chem. 2021, 354, 129492. [Google Scholar] [CrossRef] [PubMed]
- Mallakpour, S.; Radfar, Z.; Hussain, C.M. Current advances on polymer-layered double hydroxides/metal oxides nanocomposites and bionanocomposites: Fabrications and applications in the textile industry and nanofibers. Appl. Clay Sci. 2021, 206, 106054. [Google Scholar] [CrossRef]
- Omran, A.A.B.; Mohammed, A.A.B.A.; Sapuan, S.M.; Ilyas, R.A.; Asyraf, M.R.M.; Koloor, S.S.R.; Petrů, M. Micro- and Nanocellulose in Polymer Composite Materials: A Review. Polymers 2021, 13, 231. [Google Scholar] [CrossRef]
- Camargo, P.H.C.; Satyanarayana, K.G.; Wypych, F. Nanocomposites: Synthesis, structure, properties and new application opportunities. Mater. Res. 2009, 12, 1–39. [Google Scholar] [CrossRef] [Green Version]
- Chikwendu, C. Nanocomposites–An Overview. Int. J. Eng. Res. Dev. 2013, 8, 17–23. [Google Scholar]
- Tariq, A.; Bhawani, S.A.; Nisar, M.; Asaruddin, M.R.; Alotaibi, K.M. 13-Starch-Based Nanocomposites for Gene Delivery. In Woodhead Publishing Series in Biomaterials. Polysaccharide-Based Nanocomposites for Gene Delivery and Tissue Engineering; Bhawani, S.A., Karim, Z., Jawaid, M., Eds.; Woodhead Publishing: Sawston, UK, 2021; pp. 263–277. ISBN 9780128212301. [Google Scholar] [CrossRef]
- Janeni, J.; Adassooriya, N.M. Chapter 3-Nanocellulose Biopolymer-Based Biofilms: Applications and Challenges. In Biopolymer-Based Nano Films; Rai, M., dos Santos, C.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 43–62. ISBN 9780128233818. [Google Scholar] [CrossRef]
- Godbole, R.; Goutam, A.; Mali, A. Microbial Biopolymers: Pharmaceutical, medical, and biotechnological applications. In Microbial Polymers; Vaishnav, A., Choudhary, D.K., Eds.; Springer: Singapore, 2021. [Google Scholar] [CrossRef]
- Udayakumar, G.P.; Muthusamy, S.; Selvaganesh, B.; Sivarajasekar, N.; Rambabu, K.; Banat, F.; Sivamani, S.; Sivakumar, N.; Hosseini-Bandegharaei, A.; Show, P.L. Biopolymers and composites: Properties, characterization and their applications in food, medical and pharmaceutical industries. J. Environ. Chem. Eng. 2021, 9, 105322. [Google Scholar] [CrossRef]
- Singh, T.V.; Shagolsem, L.S. Biopolymer based nano-structured materials and their applications. In Nanostructured Materials and their Applications. Materials Horizons: From Nature to Nanomaterials; Swain, B.P., Ed.; Springer: Singapore, 2021. [Google Scholar] [CrossRef]
- Suresh, R.; Rajendran, S.; Hoang, T.K.; Vo, D.-V.N.; Siddiqui, M.N.; Cornejo-Ponce, L. Recent progress in green and biopolymer based photocatalysts for the abatement of aquatic pollutants. Environ. Res. 2021, 199, 111324. [Google Scholar] [CrossRef]
- Okpala, C.C. The Benefits and Applications of Nanocomposites. Int. J. Adv. Eng. Technol. 2006, 5, 12–18. [Google Scholar]
- Han, R.; Xiang, R.; Li, J.; Wang, F.; Wang, C. High-level production of microbial prodigiosin: A review. J. Basic Microbiol. 2021, 61, 506–523. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Lim, S.; Yoon, K.-H.; Lee, J.I.; Mitchell, R.J. Biotechnological Activities and Applications of Bacterial Pigments Violacein and Prodigiosin. J. Biol. Eng. 2021, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Chilczuk, T.; Monson, R.; Schmieder, P.; Christov, V.; Enke, H.; Salmond, G.; Niedermeyer, T.H.J. Ambigols from the Cyanobacterium Fischerella ambigua Increase Prodigiosin Production in Serratia spp. ACS Chem. Biol. 2020, 15, 2929–2936. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, A.; Sunderrajan, S.; Pennathur, G. Phylogenetic Studies on the Prodigiosin Biosynthetic Operon. Curr. Microbiol. 2019, 76, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Halder, U.; Banerjee, A.; Biswas, R.; Sharma, A.; Pal, S.; Adhikary, A.; Bandopadhyay, R. Production of prodigiosin by a drug-resistant Serratia rubidaea HB01 isolated from sewage. Environ. Sustain. 2020, 3, 279–287. [Google Scholar] [CrossRef]
- Casullo De Araújo, H.W.; Fukushima, K.; Campos Takaki, G.M. Prodigiosin Production by Serratia marcescens UCP 1549 Using Renewable-Resources as a Low Cost Substrate. Molecules 2010, 15, 6931–6940. [Google Scholar] [CrossRef]
- Khanafari, A.; Assadi, M.M.; Fakhr, F.A. Review of Prodigiosin, Pigmentation in Serratia marcescens. Online J. Biol. Sci. 2006, 6, 1–13. [Google Scholar] [CrossRef]
- Chatragadda, R.; Dufossé, L. Ecological and Biotechnological Aspects of Pigmented Microbes: A Way Forward in Development of Food and Pharmaceutical Grade Pigments. Microorganisms 2021, 9, 637. [Google Scholar] [CrossRef]
- Berning, L.; Schlütermann, D.; Friedrich, A.; Berleth, N.; Sun, Y.; Wu, W.; Mendiburo, M.; Deitersen, J.; Brass, H.; Skowron, M.; et al. Prodigiosin Sensitizes Sensitive and Resistant Urothelial Carcinoma Cells to Cisplatin Treatment. Molecules 2021, 26, 1294. [Google Scholar] [CrossRef]
- Metwally, R.A.; El Sikaily, A.; El-Sersy, N.A.; Ghozlan, H.A.; Sabry, S.A. Antimicrobial activity of textile fabrics dyed with prodigiosin pigment extracted from marine Serratia rubidaea RAM_Alex bacteria. Egypt. J. Aquat. Res. 2021, 47, 301–305. [Google Scholar] [CrossRef]
- Darshan, N.; Manonmani, H.K. Prodigiosin and its potential applications. J. Food Sci. Technol. 2015, 52, 5393–5407. [Google Scholar] [CrossRef] [Green Version]
- Stankovic, N.; Senerovic, L.; Ilic-Tomic, T.; Vasiljevic, B.; Nikodinovic-Runic, J. Properties and applications of undecylprodigiosin and other bacterial prodigiosins. Appl. Microbiol. Biotechnol. 2014, 98, 3841–3858. [Google Scholar] [CrossRef]
- Cook, T.B.; Jacobson, T.B.; Venkataraman, M.V.; Hofstetter, H.; Amador-Noguez, D.; Thomas, M.G.; Pfleger, B.F. Stepwise genetic engineering of Pseudomonas putida enables robust heterologous production of prodigiosin and glidobactin A. Metab. Eng. 2021, 67, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Heu, K.; Romoli, O.; Schönbeck, J.C.; Ajenoe, R.; Epelboin, Y.; Kircher, V.; Houël, E.; Estevez, Y.; Gendrin, M. The Effect of Secondary Metabolites Produced by Serratia marcescens on Aedes aegypti and Its Microbiota. Front. Microbiol. 2021, 12, 1650. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, L.; Osire, T.; Fu, W.; Yi, G.; Yang, S.-T.; Yang, T.; Rao, Z. Enhanced Prodigiosin Production in Serratia marcescens JNB5-1 by Introduction of a Polynucleotide Fragment into the pigN 3′ Untranslated Region and Disulfide Bonds into O -Methyl Transferase (PigF). Appl. Environ. Microbiol. 2021, 87, AEM0054321. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C.T.; Garneau-Tsodikova, S.; Howard-Jones, A.R. Biological formation of pyrroles: Nature’s logic and enzymatic machinery. Nat. Prod. Rep. 2006, 23, 517–531. [Google Scholar] [CrossRef] [PubMed]
- Gholap, S.S. Pyrrole: An emerging scaffold for construction of valuable therapeutic agents. Eur. J. Med. Chem. 2016, 110, 13–31. [Google Scholar] [CrossRef] [PubMed]
- Hardjito, L.; Huq, A.; Colwell, R.R. The influence of environmental conditions on the production of pigment bySerratia marcescens. Biotechnol. Bioprocess Eng. 2002, 7, 100–104. [Google Scholar] [CrossRef]
- Wei, Y.-H.; Chen, W.-C. Enhanced production of prodigiosin-like pigment from Serratia marcescens SMΔR by medium improvement and oil-supplementation strategies. J. Biosci. Bioeng. 2005, 99, 616–622. [Google Scholar] [CrossRef]
- Iranshahi, M.; Shahverdi, A.R.; Mirjani, R.; Amin, G.; Shafiee, A. Umbelliprenin from Ferula persica Roots Inhibits the Red Pigment Production in Serratia marcescens. Z. Nat. C 2004, 59, 506–508. [Google Scholar] [CrossRef]
- Habash, S.S.; Brass, H.U.C.; Klein, A.S.; Klebl, D.P.; Weber, T.M.; Classen, T.; Pietruszka, J.; Grundler, F.M.W.; Schleker, A.S.S. Novel Prodiginine Derivatives Demonstrate Bioactivities on Plants, Nematodes, and Fungi. Front. Plant Sci. 2020, 11, 579807. [Google Scholar] [CrossRef] [PubMed]
- Kimata, S.; Izawa, M.; Kawasaki, T.; Hayakawa, Y. Identification of a prodigiosin cyclization gene in the roseophilin producer and production of a new cyclized prodigiosin in a heterologous host. J. Antibiot. 2016, 70, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Williamson, N.R.; Simonsen, H.T.; Ahmed, R.A.A.; Goldet, G.; Slater, H.; Woodley, L.; Leeper, F.J.; Salmond, G.P.C. Biosynthesis of the red antibiotic, prodigiosin, in Serratia: Identification of a novel 2-methyl-3-n-amyl-pyrrole (MAP) assembly pathway, definition of the terminal condensing enzyme, and implications for undecylprodigiosin biosynthesis in Streptomyces. Mol. Microbiol. 2005, 56, 971–989. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.K.P.; Williamson, N.R.; Slater, H.; Cox, A.; Abbasi, S.; Foulds, I.; Simonsen, H.T.; Leeper, F.J.; Salmond, G.P.C. The Serratia gene cluster encoding biosynthesis of the red antibiotic, prodigiosin, shows species- and strain-dependent genome context variation. Microbiology 2004, 150, 3547–3560. [Google Scholar] [CrossRef]
- Lou, X.; Ran, T.; Han, N.; Gao, Y.; He, J.; Tang, L.; Xu, D.; Wang, W. Crystal structure of the catalytic domain of PigE: A transaminase involved in the biosynthesis of 2-methyl-3-n-amyl-pyrrole (MAP) from Serratia sp. FS14. Biochem. Biophys. Res. Commun. 2014, 447, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Hampton, H.G.; McNeil, M.B.; Paterson, T.J.; Ney, B.; Williamson, N.R.; Easingwood, R.A.; Bostina, M.; Salmond, G.P.C.; Fineran, P.C. CRISPR-Cas gene-editing reveals RsmA and RsmC act through FlhDC to repress the SdhE flavinylation factor and control motility and prodigiosin production in Serratia. Microbiology 2016, 162, 1047–1058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kothari, V.; Joshi, C.; Patel, P.; Mehta, M.; Dubey, S.; Mishra, B.; Sarvaiya, N. Influence of a Mono-Frequency Sound on Bacteria can be a Function of the Sound-Level. BioRxiv 2016, 071746. [Google Scholar] [CrossRef]
- Tenconi, E.; Traxler, M.; Hoebreck, C.; Van Wezel, G.P.; Rigali, S. Production of Prodiginines Is Part of a Programmed Cell Death Process in Streptomyces coelicolor. Front. Microbiol. 2018, 9, 1742. [Google Scholar] [CrossRef]
- Haddix, P.L.; Shanks, R.M.Q. Prodigiosin pigment of Serratia marcescens is associated with increased biomass production. Arch Microbiol. 2018, 200, 989–999. [Google Scholar] [CrossRef]
- Kalivoda, E.J.; Stella, N.A.; Aston, M.A.; Fender, J.E.; Thompson, P.P.; Kowalski, R.P.; Shanks, R.M. Cyclic AMP negatively regulates prodigiosin production by Serratia marcescens. Res. Microbiol. 2010, 161, 158–167. [Google Scholar] [CrossRef] [Green Version]
- Xia, Y.; Wang, G.; Lin, X.; Song, X.; Ai, L. Solid-state fermentation with Serratia marcescens Xd-1 enhanced production of prodigiosin by using bagasse as an inertia matrix. Ann. Microbiol. 2016, 66, 1239–1247. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Lee, B.-R.; Sathiyanarayanan, G.; Song, H.-S.; Kim, J.; Jeon, J.-M.; Kim, J.-H.; Park, S.-H.; Yu, J.-H.; Park, K.; et al. Medium engineering for enhanced production of undecylprodigiosin antibiotic in Streptomyces coelicolor using oil palm biomass hydrolysate as a carbon source. Bioresour. Technol. 2016, 217, 141–149. [Google Scholar] [CrossRef]
- Soliev, A.B.; Hosokawa, K.; Enomoto, K. Effects of prodigiosin family compounds from Pseudoalteromonas sp. 1020R on the activities of protein phosphatases and protein kinases. J. Enzym. Inhib. Med. Chem. 2014, 30, 533–538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luti, K.J.K.; Mavituna, F. Streptomyces coelicolor increases the production of undecylprodigiosin when interacted with Bacillus subtilis. Biotechnol. Lett. 2010, 33, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.-F.; Ma, C.-J.; Lu, C.-H.; Tsai, Y.-T.; Wei, Y.-H.; Chang, J.T.; Lai, J.-K.; Chueh, P.J.; Yeh, C.-T.; Tang, P.-C. Undecylprodigiosin selectively induces apoptosis in human breast carcinoma cells independent of p53. Toxicol. Appl. Pharmacol. 2007, 225, 318–328. [Google Scholar] [CrossRef]
- Silva, A.; Guimarães, L.; Ferreira, E.; Torres, M.D.C.; da Silva, A.B.; Branco, P.; Oliveira, F.A.; Silva, G.; Wilke, D.V.; Silveira, E.; et al. Bioprospecting Anticancer Compounds from the Marine-Derived Actinobacteria Actinomadura sp. Collected at the Saint Peter and Saint Paul Archipelago (Brazil). J. Braz. Chem. Soc. 2017, 28, 465–474. [Google Scholar] [CrossRef]
- Srimathi, R.; Priya, R.; Nirmala, M.; Malarvizhi, A. Isolation, identification, optimization of prodigiosin pigment produced by Serratia marcescens and its applications. Int. J. Latest Eng. Manag. Res. 2017, 2, 11–21. Available online: http://www.ijlemr.com/papers/volume2-issue9/23-IJLEMR-22452.pdf (accessed on 28 July 2021).
- Mirzaei, S.A.; Kavishahi, M.S.; Keshavarz, Z.; Elahian, F. Unlike Butylcycloheptylprodigiosin, Isolated Undecylprodigiosin from Streptomyces parvulus Is Not a MDR1 and BCRP Substrate in Multidrug-Resistant Cancers. DNA Cell Biol. 2018, 37, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Tejasvini, S.P.; Ashwini, B.K.; Naiem, H.N. Production, purification, identification of prodigiosin from Serratia sp. and its antimicrobial activity. Life Sci. Inform. Publ. 2016, 1, 1–12. [Google Scholar] [CrossRef]
- Gondil, V.S.; Asif, M.; Bhalla, T.C. Optimization of physicochemical parameters influencing the production of prodigiosin from Serratia nematodiphila RL2 and exploring its antibacterial activity. 3 Biotech 2017, 7, 338. [Google Scholar] [CrossRef]
- Chávez-Castilla, L.; Aguilar, O. Effect of Mineral Supplementation and Type of Starch on the Production of Prodigiosin from a Culture of Serratia marcescens BS303. Revista Mexicana de Ingeniería Química. 2015. Available online: http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1665-27382015000300008 (accessed on 28 July 2021).
- Metwally, R.; El-Sersy, N.A.; El Sikaily, A.; Ghozlan, H.A.; Sabry, S.A. Statistical Optimization and Characterization of Prodigiosin from a Marine Serratia rubidaea RAM_Alex. J. Pure Appl. Microbiol. 2017, 11, 1259–1266. [Google Scholar] [CrossRef]
- Xu, F.; Xia, S.; Yang, Q. Strategy for obtaining inexpensive prodigiosin production by Serratia marcescen. In Proceedings of the 3rd International Conference on Chemical, Biological and Environmental Engineering, Chengdu, China, 23–25 September 2011; Available online: http://ipcbee.com/vol20/6-ICBEE2011E012.pdf (accessed on 28 July 2021).
- Arivizhivendhan, K.V.; Mahesh, M.; Boopathy, R.; Mary, R.; Sekaran, G. A novel method for the extraction of prodigiosin from bacterial fermenter integrated with sequential batch extraction reactor using magnetic iron oxide. Process Biochem. 2016, 51, 1731–1737. [Google Scholar] [CrossRef]
- Isaka, M.; Jaturapat, A.; Kramyu, J.; Tanticharoen, M.; Thebtaranonth, Y. Potent In Vitro Antimalarial Activity of Metacycloprodigiosin Isolated from Streptomyces spectabilis BCC 4785. Antimicrob. Agents Chemother. 2002, 46, 1112–1113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.; Yang, J.; Tian, Y.; Zhou, X.; Wang, S.; Zhu, J.; Sun, D.; Liu, C. An in situ extractive fermentation strategy for enhancing prodigiosin production from Serratia marcescens BWL1001 and its application to inhibiting the growth of Microcystis aeruginosa. Biochem. Eng. J. 2020, 166, 107836. [Google Scholar] [CrossRef]
- Elkenawy, N.M.; Yassin, A.S.; Elhifnawy, H.N.; Amin, M.A. Optimization of prodigiosin production by Serratia marcescens using crude glycerol and enhancing production using gamma radiation. Biotechnol. Rep. 2017, 14, 47–53. [Google Scholar] [CrossRef]
- Bhagwat, A.; Padalia, U. Optimization of prodigiosin biosynthesis by Serratia marcescens using unconventional bioresources. J. Genet. Eng. Biotechnol. 2020, 18, 26. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Veony, E.; Yang, Q. Kitchen Waste as a Novel Available Substrate for Prodigiosin Production by Serratia marcescense. IOP Conf. Ser. Earth Environ. Sci. 2018, 171, 012037. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, L.; Pan, X.; Osire, T.; Fang, H.; Zhang, H.; Yang, S.-T.; Yang, T.; Rao, Z. Improved Prodigiosin Production by Relieving CpxR Temperature-Sensitive Inhibition. Front. Bioeng. Biotechnol. 2020, 8, 344. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Sun, C.; Tang, M.; You, J.; Osire, T.; Zhao, Y.; Xu, M.; Zhang, X.; Shao, M.; Yang, S.; et al. LysR-Type Transcriptional Regulator MetR Controls Prodigiosin Production, Methionine Biosynthesis, Cell Motility, H2O2 Tolerance, Heat Tolerance, and Exopolysaccharide Synthesis in Serratia marcescens. Appl. Environ. Microbiol. 2020, 86, e02241-19. [Google Scholar] [CrossRef]
- Bruna, R.E.; Molino, M.V.; Lazzaro, M.; Mariscotti, J.F.; Véscovi, E.G. CpxR-Dependent Thermoregulation of Serratia marcescens PrtA Metalloprotease Expression and Its Contribution to Bacterial Biofilm Formation. J. Bacteriol. 2018, 200, e00006-18. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.; Wang, L.; Osire, T.; Fu, W.; Yi, G.; Yang, S.-T.; Yang, T.; Rao, Z. Comparative transcriptome analysis reveals metabolic regulation of prodigiosin in Serratia marcescens. Syst. Microbiol. Biomanuf. 2021, 1, 323–335. [Google Scholar] [CrossRef]
- Sun, S.-Q.; Wang, Y.-J.; Xu, W.; Zhu, C.-J.; Liu, X.-X. Optimizing Ultrasound-Assisted Extraction of Prodigiosin by Response Surface Methodology. Prep. Biochem. Biotechnol. 2014, 45, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Paul, T.; Bandyopadhyay, T.K.; Mondal, A.; Tiwari, O.N.; Muthuraj, M.; Bhunia, B. A comprehensive review on recent trends in production, purification, and applications of prodigiosin. Biomass Convers. Biorefinery 2020, 12, 1409–1431. [Google Scholar] [CrossRef]
- Khanam, B.; Chandra, R. Comparative analysis of prodigiosin isolated from endophyte Serratia marcescens. Lett. Appl. Microbiol. 2018, 66, 194–201. [Google Scholar] [CrossRef]
- Othman, M.A.; El-Zamik, F.I.; Hegazy, M.I.; Salama, A.S. Isolation and identification of egyptian strains of Serratia marcescens producing antibacterial and antioxidant prodigiosin pigment. Zagazig J. Agric. Res. 2019, 46, 1573–1582. [Google Scholar] [CrossRef]
- Sreedharan, H.E.; Harilal, C.C.; Pradeep, S. Response surface optimization of prodigiosin production by phthalate degrading Achromobacter denitrificans SP1 and exploring its antibacterial activity. Prep. Biochem. Biotechnol. 2020, 50, 564–571. [Google Scholar] [CrossRef]
- Hage-Hülsmann, J.; Grünberger, A.; Thies, S.; Santiago-Schübel, B.; Klein, A.S.; Pietruszka, J.; Binder, D.; Hilgers, F.; Domröse, A.; Drepper, T.; et al. Natural biocide cocktails: Combinatorial antibiotic effects of prodigiosin and biosurfactants. PLoS ONE 2018, 13, e0200940. [Google Scholar] [CrossRef] [PubMed]
- Yip, C.-H.; Mahalingam, S.; Wan, K.-L.; Nathan, S. Prodigiosin inhibits bacterial growth and virulence factors as a potential physiological response to interspecies competition. PLoS ONE 2021, 16, e0253445. [Google Scholar] [CrossRef]
- Ren, Y.; Gong, J.; Fu, R.; Zhang, J.; Fang, K.; Liu, X. Antibacterial dyeing of silk with prodigiosins suspention produced by liquid fermentation. J. Clean. Prod. 2018, 201, 648–656. [Google Scholar] [CrossRef]
- Woodhams, D.C.; LaBumbard, B.C.; Barnhart, K.L.; Becker, M.H.; Bletz, M.C.; Escobar, L.A.; Flechas, S.V.; Forman, M.E.; Iannetta, A.A.; Joyce, M.D.; et al. Prodigiosin, Violacein, and Volatile Organic Compounds Produced by Widespread Cutaneous Bacteria of Amphibians Can Inhibit Two Batrachochytrium Fungal Pathogens. Microb. Ecol. 2017, 75, 1049–1062. [Google Scholar] [CrossRef]
- Jimtha, C.J.; Jishma, P.; Sreelekha, S.; Chithra, S.; Radhakrishnan, E. Antifungal properties of prodigiosin producing rhizospheric Serratia sp. Rhizosphere 2017, 3, 105–108. [Google Scholar] [CrossRef]
- Alijani, Z.; Amini, J.; Ashengroph, M.; Bahramnejad, B. Antifungal Activity of Serratia rubidaea Mar61-01 Purified Prodigiosin Against Colletotrichum nymphaeae, the Causal Agent of Strawberry Anthracnose. J. Plant Growth Regul. 2021, 41, 585–595. [Google Scholar] [CrossRef]
- John, C.J.; Bibishna, A.; Mallikarjunaswamy, G. Antimycotic effects of a prodigiosin producing Serratia marcescens rhizobacteria. Rhizosphere 2021, 18, 100336. [Google Scholar] [CrossRef]
- Pap, N.; Fidelis, M.; Azevedo, L.; do Carmo, M.A.V.; Wang, D.; Mocan, A.; Pereira, E.P.R.; Xavier-Santos, D.; Sant’Ana, A.S.; Yang, B.; et al. Berry polyphenols and human health: Evidence of antioxidant, anti-inflammatory, microbiota modulation, and cell-protecting effects. Curr. Opin. Food Sci. 2021, 42, 167–186. [Google Scholar] [CrossRef]
- De Carvalho, A.P.A.; Conte-Junior, C.A. Health benefits of phytochemicals from Brazilian native foods and plants: Antioxidant, antimicrobial, anti-cancer, and risk factors of metabolic/endocrine disorders control. Trends Food Sci. Technol. 2021, 111, 534–548. [Google Scholar] [CrossRef]
- Yan, Z.; Zhong, Y.; Duan, Y.; Chen, Q.; Li, F. Antioxidant mechanism of tea polyphenols and its impact on health benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef]
- Nguyen, T.-H.; Wang, S.-L.; Nguyen, D.-N.; Nguyen, A.-D.; Nguyen, T.-H.; Doan, M.-D.; Ngo, V.-A.; Doan, C.-T.; Kuo, Y.-H.; Nguyen, V.-B. Bioprocessing of Marine Chitinous Wastes for the Production of Bioactive Prodigiosin. Molecules 2021, 26, 3138. [Google Scholar] [CrossRef]
- Sajjad, W.; Ahmad, S.; Aziz, I.; Azam, S.S.; Hasan, F.; Shah, A.A. Antiproliferative, antioxidant and binding mechanism analysis of prodigiosin from newly isolated radio-resistant Streptomyces sp. strain WMA-LM31. Mol. Biol. Rep. 2018, 45, 1787–1798. [Google Scholar] [CrossRef]
- Arivizhivendhan, K.V.; Mahesh, M.; Boopathy, R.; Swarnalatha, S.; Refgina Mary, R.; Sekaran, G. Antioxidant and antimicrobial activity of bioactive prodigiosin produces from Serratia marcescens using agricultural waste as a substrate. J. Food Sci. Technol. 2018, 55, 2661–2670. [Google Scholar] [CrossRef]
- World Health Organization. Latest Global Cancer Data: Cancer Burden Rises to 19.3 Million New Cases and 10.0 Million Cancer deaths in 2020. Int. Agency Res. Cancer. 2020, pp. 1–3. Available online: https://www.iarc.who.int/wp-content/uploads/2020/12/pr292_E.pdf (accessed on 28 July 2021).
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Cancer statistics for the year 2020: An overview. Int. J. Cancer 2021, 149, 778–789. [Google Scholar] [CrossRef]
- Soerjomataram, I.; Bray, F. Planning for tomorrow: Global cancer incidence and the role of prevention 2020–2070. Nat. Rev. Clin. Oncol. 2021, 18, 663–672. [Google Scholar] [CrossRef] [PubMed]
- IARC. International Agency for Research on Cancer. Global Cancer Observatory. 2021. Available online: https://gco.iarc.fr/ (accessed on 24 July 2021).
- Mohan, C.D.; Rangappa, S.; Nayak, S.C.; Jadimurthy, R.; Wang, L.; Sethi, G.; Garg, M.; Rangappa, K.S. Bacteria as a treasure house of secondary metabolites with anticancer potential. In Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2021. [Google Scholar] [CrossRef]
- Balasubramaniam, B.; Alexpandi, R.; Darjily, D.R. Exploration of the optimized parameters for bioactive prodigiosin mass production and its biomedical applications in vitro as well as in silico. Biocatal. Agric. Biotechnol. 2019, 22, 101385. [Google Scholar] [CrossRef]
- Zhao, C.; Qiu, S.; He, J.; Peng, Y.; Xu, H.; Feng, Z.; Huang, H.; Du, Y.; Zhou, Y.; Nie, Y. Prodigiosin impairs autophagosome-lysosome fusion that sensitizes colorectal cancer cells to 5-fluorouracil-induced cell death. Cancer Lett. 2020, 481, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.; Sun, R.; Xu, K.; Man, Z.; Ji, J.; Pu, Y.; Yin, L.; Zhang, J.; Pu, Y. Prodigiosin induces apoptosis and inhibits autophagy via the extracellular signal-regulated kinase pathway in K562 cells. Toxicol. Vitr. 2019, 60, 107–115. [Google Scholar] [CrossRef]
- Nguyen, V.B.; Nguyen, D.N.; Nguyen, A.D.; Ngo, V.A.; Ton, T.Q.; Doan, C.T.; Pham, T.P.; Tran, T.P.H.; Wang, S.-L. Utilization of Crab Waste for Cost-Effective Bioproduction of Prodigiosin. Mar. Drugs 2020, 18, 523. [Google Scholar] [CrossRef]
- Nguyen, V.B.; Chen, S.P.; Nguyen, T.H.; Nguyen, M.T.; Tran, T.T.T.; Doan, C.T.; Tran, T.N.; Nguyen, A.D.; Kuo, Y.H.; Wang, S.L. Novel Efficient Bioprocessing of Marine Chitins into Active Anticancer Prodigiosin. Mar. Drugs 2020, 18, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, D.; Liu, J.; Wang, X.; Kong, D.; Du, W.; Li, H.; Hse, C.-Y.; Shupe, T.; Zhou, D.; Zhao, K. Biological Potential and Mechanism of Prodigiosin from Serratia marcescens Subsp. lawsoniana in Human Choriocarcinoma and Prostate Cancer Cell Lines. Int. J. Mol. Sci. 2018, 19, 3465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branco, P.C.; Pontes, C.A.; Rezende-Teixeira, P.; Amengual-Rigo, P.; Alves-Fernandes, D.K.; Maria-Engler, S.S.; da Silva, A.B.; Pessoa, O.D.L.; Jimenez, P.C.; Mollasalehi, N.; et al. Survivin modulation in the antimelanoma activity of prodiginines. Eur. J. Pharmacol. 2020, 888, 173465. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, B.; Zhou, L.; Yu, S.; Su, Z.; Song, J.; Sun, Q.; Sha, O.; Wang, X.; Jiang, W.; et al. Prodigiosin inhibits Wnt/β-catenin signaling and exerts anticancer activity in breast cancer cells. Proc. Natl. Acad. Sci. USA 2016, 113, 13150–13155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anwar, M.M.; Shalaby, M.; Embaby, A.M.; Saeed, H.; Agwa, M.M.; Hussein, A. Prodigiosin/PU-H71 as a novel potential combined therapy for triple negative breast cancer (TNBC): Preclinical insights. Sci. Rep. 2020, 10, 182–195. [Google Scholar] [CrossRef]
- Lapenda, J.C.L.; Alves, V.P.; Adam, M.L.; Rodrigues, M.D.; Nascimento, S.C. Cytotoxic Effect of Prodigiosin, Natural Red Pigment, Isolated from Serratia marcescens UFPEDA 398. Indian J. Microbiol. 2020, 60, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Sam, M.R.; Pourpak, R. Regulation of p53 and survivin by prodigiosin compound derived from Serratia marcescens contribute to caspase-3-dependent apoptosis in acute lymphoblastic leukemia cells. Hum. Exp. Toxicol. 2017, 37, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.-Y.; Chen, N.-F.; Kuo, H.-M.; Yang, S.-N.; Sung, C.-S.; Sung, P.-J.; Wen, Z.-H.; Chen, W.-F. Prodigiosin stimulates endoplasmic reticulum stress and induces autophagic cell death in glioblastoma cells. Apoptosis 2018, 23, 314–328. [Google Scholar] [CrossRef] [PubMed]
- Hassankhani, R.; Sam, M.R.; Esmaeilou, M.; Ahangar, P. Prodigiosin isolated from cell wall of Serratia marcescens alters expression of apoptosis-related genes and increases apoptosis in colorectal cancer cells. Med Oncol. 2014, 32, 366. [Google Scholar] [CrossRef] [PubMed]
- Yenkejeh, R.A.; Sam, M.R.; Esmaeillou, M. Targeting survivin with prodigiosin isolated from cell wall of Serratia marcescens induces apoptosis in hepatocellular carcinoma cells. Hum. Exp. Toxicol. 2017, 36, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Chiu, W.-J.; Lin, S.-R.; Chen, Y.-H.; Tsai, M.-J.; Leong, M.K.; Weng, C.-F. Prodigiosin-Emerged PI3K/Beclin-1-Independent Pathway Elicits Autophagic Cell Death in Doxorubicin-Sensitive and -Resistant Lung Cancer. J. Clin. Med. 2018, 7, 321. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Zhou, H.; Ma, X.; Lin, C.; Lu, L.; Liu, D.; Ma, D.; Gao, X.; Qian, X.Y. Prodigiosin Inhibits Proliferation, Migration, and Invasion of Nasopharyngeal Cancer Cells. Cell. Physiol. Biochem. 2018, 48, 1556–1562. [Google Scholar] [CrossRef]
- Cheng, M.-F.; Lin, C.-S.; Chen, Y.-H.; Sung, P.-J.; Lin, S.-R.; Tong, Y.-W.; Weng, C.-F. Inhibitory Growth of Oral Squamous Cell Carcinoma Cancer via Bacterial Prodigiosin. Mar. Drugs 2017, 15, 224. [Google Scholar] [CrossRef]
- Lin, P.; Shen, J.; Ou, P.; Liu, L.; Chen, Z.; Chu, F.; Wang, J.; Jin, X. Prodigiosin isolated from Serratia marcescens in the Periplaneta americana gut and its apoptosis-inducing activity in HeLa cells. Oncol. Rep. 2019, 41, 3377–3385. [Google Scholar] [CrossRef]
- Harikandei, K.B.; Salehi, P.; Ebrahimi, S.N.; Bararjanian, M.; Kaiser, M.; Al-Harrasi, A. Synthesis, in-vitro antiprotozoal activity and molecular docking study of isothiocyanate derivatives. Bioorg. Med. Chem. 2019, 28, 115185. [Google Scholar] [CrossRef]
- Lindsay, S.W.; Thomas, M.B.; Kleinschmidt, I. Threats to the effectiveness of insecticide-treated bednets for malaria control: Thinking beyond insecticide resistance. Lancet Glob. Health 2021, 9, e1325–e1331. [Google Scholar] [CrossRef]
- Papireddy, K.; Smilkstein, M.; Kelly, J.X.; Shweta; Salem, S.M.; Alhamadsheh, M.; Haynes, S.W.; Challis, G.L.; Reynolds, K.A. Antimalarial Activity of Natural and Synthetic Prodiginines. J. Med. Chem. 2011, 54, 5296–5306. [Google Scholar] [CrossRef] [Green Version]
- Suryawanshi, R.K.; Patil, C.D.; Borase, H.P.; Narkhede, C.P.; Salunke, B.K.; Patil, S.V. Mosquito larvicidal and pupaecidal potential of prodigiosin from Serratia marcescens and understanding its mechanism of action. Pestic. Biochem. Physiol. 2015, 123, 49–55. [Google Scholar] [CrossRef]
- James, C.; Harfouche, M.; Welton, N.J.; Turner, K.M.E.; Abu-Raddad, L.J.; Gottlieb, S.L.; Looker, K.J. Herpes simplex virus: Global infection prevalence and incidence estimates, 2016. Bull. World Health Organ. 2020, 98, 315–329. [Google Scholar] [CrossRef] [PubMed]
- Valerio, G.S.; Lin, C.C. Ocular manifestations of herpes simplex virus. Curr. Opin. Ophthalmol. 2019, 30, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, R.K.; Koujah, L.; Patil, C.D.; Ames, J.M.; Agelidis, A.; Yadavalli, T.; Patil, S.V.; Shukla, D. Bacterial Pigment Prodigiosin Demonstrates a Unique Antiherpesvirus Activity That Is Mediated through Inhibition of Prosurvival Signal Transducers. J. Virol. 2020, 94, e00251-20. [Google Scholar] [CrossRef]
- Zhou, W.; Zeng, C.; Liu, R.; Chen, J.; Li, R.; Wang, X.; Bai, W.; Liu, X.; Xiang, T.; Zhang, L.; et al. Antiviral activity and specific modes of action of bacterial prodigiosin against Bombyx mori nucleopolyhedrovirus in vitro. Appl. Microbiol. Biotechnol. 2015, 100, 3979–3988. [Google Scholar] [CrossRef]
- Peixoto, D.; Pereira, I.; Pereira-Silva, M.; Veiga, F.; Hamblin, M.R.; Lvov, Y.; Liu, M.; Paiva-Santos, A.C. Emerging role of nanoclays in cancer research, diagnosis, and therapy. Co-Ord. Chem. Rev. 2021, 440, 213956. [Google Scholar] [CrossRef]
- Guryanov, I.; Naumenko, E.; Akhatova, F.; Lazzara, G.; Cavallaro, G.; Nigamatzyanova, L.; Fakhrullin, R. Selective Cytotoxic Activity of Prodigiosin@halloysite Nanoformulation. Front. Bioeng. Biotechnol. 2020, 8, 424. [Google Scholar] [CrossRef] [PubMed]
- Azmana, M.; Mahmood, S.; Hilles, A.R.; Rahman, A.; Bin Arifin, M.A.; Ahmed, S. A review on chitosan and chitosan-based bionanocomposites: Promising material for combatting global issues and its applications. Int. J. Biol. Macromol. 2021, 185, 832–848. [Google Scholar] [CrossRef]
- Chen, F.; Chi, C. Development of pullulan/carboxylated cellulose nanocrystal/tea polyphenol bionanocomposite films for active food packaging. Int. J. Biol. Macromol. 2021, 186, 405–413. [Google Scholar] [CrossRef]
- Khairnar, Y.; Hansora, D.; Hazra, C.; Kundu, D.; Tayde, S.; Tonde, S.; Naik, J.; Chatterjee, A. Cellulose bionanocomposites for sustainable planet and people: A global snapshot of preparation, properties, and applications. Carbohydr. Polym. Technol. Appl. 2021, 2, 100065. [Google Scholar] [CrossRef]
- Nguyen, D.N.; Sim, U.; Kim, J.K. Biopolymer-Inspired N-Doped Nanocarbon Using Carbonized Polydopamine: A High-Performance Electrocatalyst for Hydrogen-Evolution Reaction. Polymers 2020, 12, 912. [Google Scholar] [CrossRef] [Green Version]
- Obayemi, J.; Danyuo, Y.; Dozie-Nwachukwu, S.; Odusanya, O.; Anuku, N.; Malatesta, K.; Yu, W.; Uhrich, K.; Soboyejo, W. PLGA-based microparticles loaded with bacterial-synthesized prodigiosin for anticancer drug release: Effects of particle size on drug release kinetics and cell viability. Mater. Sci. Eng. C 2016, 66, 51–65. [Google Scholar] [CrossRef]
- Rastegari, B.; Karbalaei-Heidari, H.R.; Zeinali, S.; Sheardown, H. The enzyme-sensitive release of prodigiosin grafted β-cyclodextrin and chitosan magnetic nanoparticles as an anticancer drug delivery system: Synthesis, characterization and cytotoxicity studies. Colloids Surf. B Biointerfaces 2017, 158, 589–601. [Google Scholar] [CrossRef]
- Akpan, U.; Pellegrini, M.; Obayemi, J.; Ezenwafor, T.; Browl, D.; Ani, C.; Yiporo, D.; Salifu, A.; Dozie-Nwachukwu, S.; Odusanya, S.; et al. Prodigiosin-loaded electrospun nanofibers scaffold for localized treatment of triple negative breast cancer. Mater. Sci. Eng. C 2020, 114, 110976. [Google Scholar] [CrossRef] [PubMed]
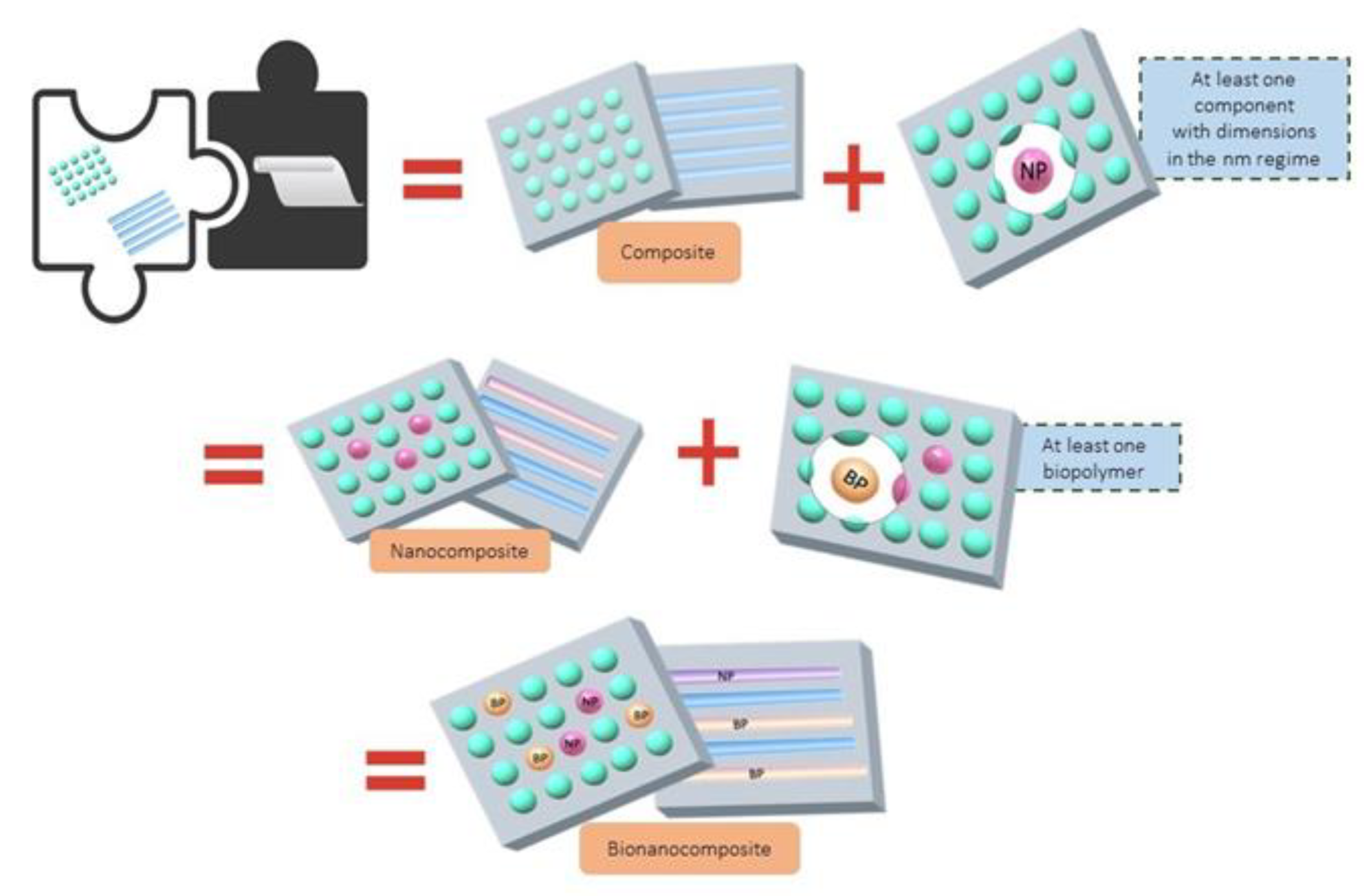
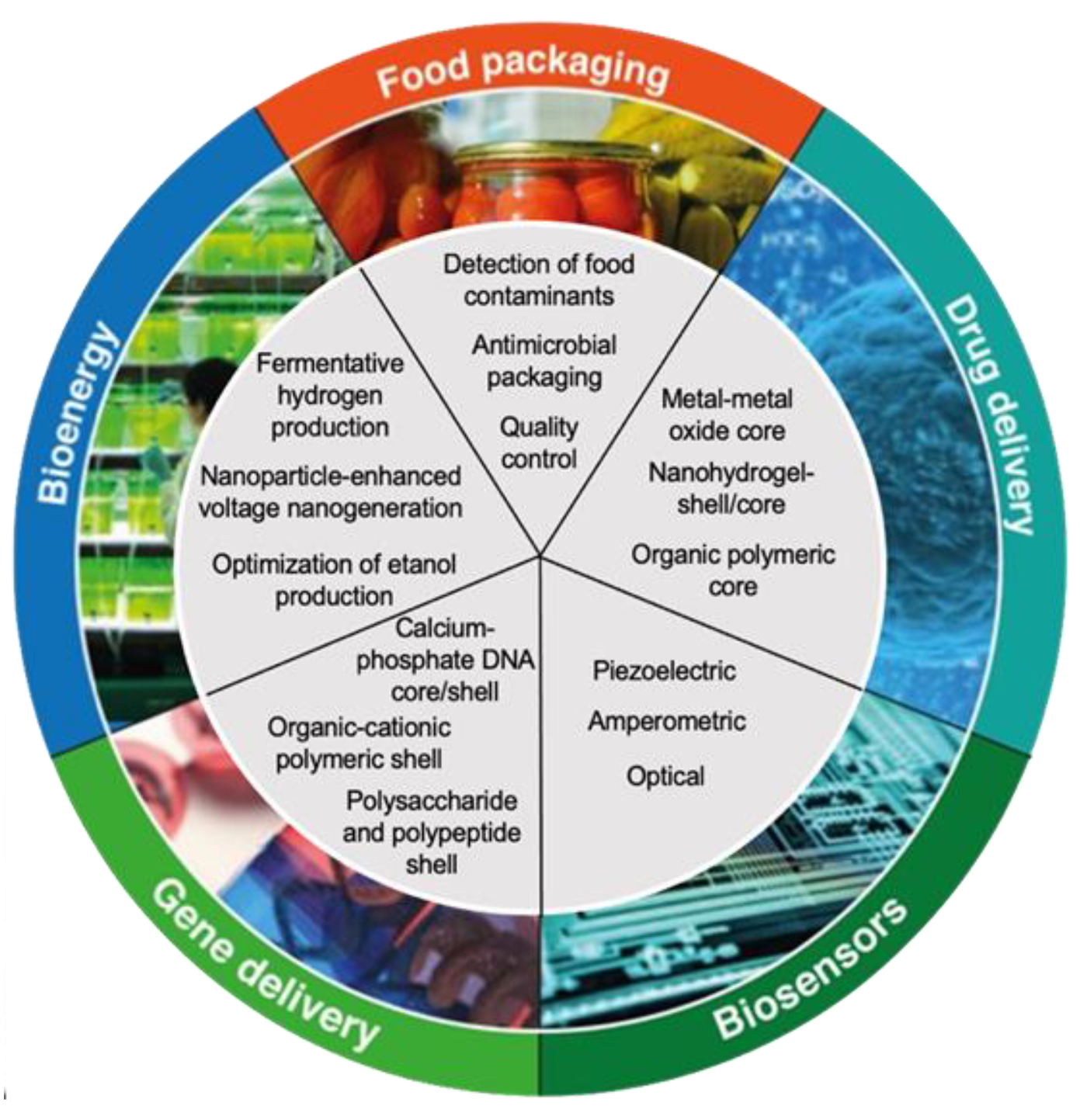
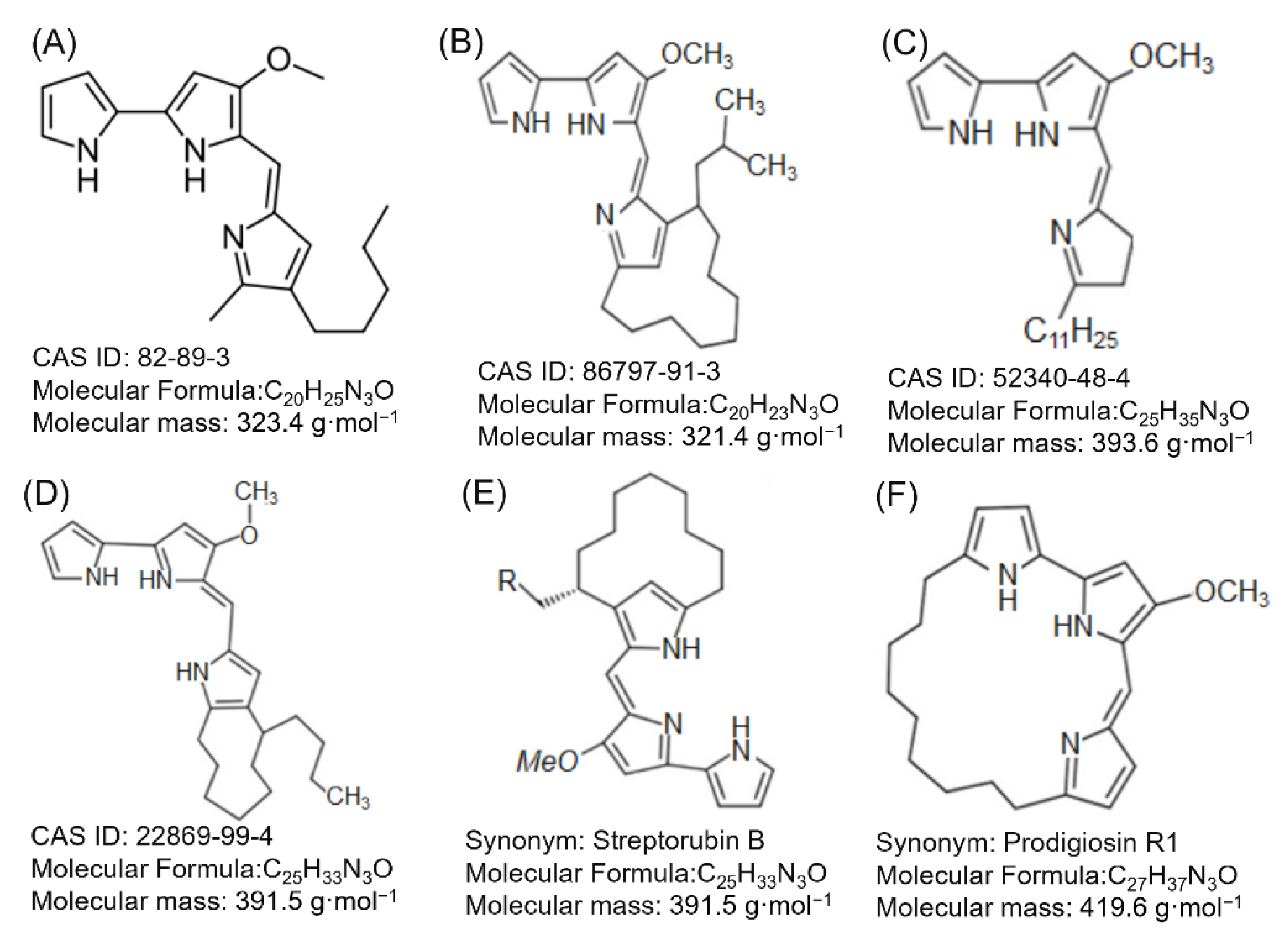
| Microorganism (Reference) | Origin | Prodiginine Type | Microorganism (Reference) | Origin | Prodiginine Type |
|---|---|---|---|---|---|
| Serratia marcescens UCP 1549 [20] | Semi-arid soil around banana trees | Prodigiosin | S. coelicolor M1110 [48] | N/A | Undecylprodigiosin |
| Streptomyces sp. JS520 [26] | Pristine soil sample collected from the cavy area of Miroc mountain in eastern Serbia | Undecylprodigiosin | S. marcescens SMAR [49] | N/A | Undecylprodigiosin |
| S. marcescens Xd-1 [45] | From a mold and tofu sample | Prodigiosin | Actinomadura sp [50] | Sediment samples in Brazil | Nonylprodigiosin Cyclononylprodigiosin Methylcycloctilprodigiosin |
| S. coelicolor M145 [46] | N/A | Undecylprodigiosin | S. marcescens [51] | From rhizospheric soil in different sites of Salem and Namakkal, India | Prodigiosin (possible) |
| S. parvulus [52] | N/A | Butylcycloheptylprodigiosin and Undecylprodigiosin | S. marcescens NCIM 5061 [53] | N/A | Prodigiosin |
| S. nematodiphila RL2 [54] | From Lahul and Spiti region in Himachal Padresh, India | Prodigiosin | S. marcescens ATCC 13880 [55] | N/A | Prodigiosin |
| S. rubiaea RAM Alex [56] | From clam samples collected form Temsah Lake, Egypt | Prodigiosin | S. marcescens [57] | From kitchen waste | Prodigiosin (Possible) |
| S. marcescens [58] | Soil sample from a tannery in Chennai, Tamilnadu, India. | Prodigiosin | S. spectabilis BCC 4785 [59] | From soil sample collected in Thailand | Metacycloprodigiosin |
| Prodigiosin Source | Concentration | Bacteria | Parameter of Bactericide Action | Value | Reference |
|---|---|---|---|---|---|
| Serratia rubidaea RAM_Alex | Chiffon stained by prodigiosin | Escherichia coli | AATCC 100 Bacteria reduction (%) | 95 | [24] |
| Staphylococcus aureus | 97 | ||||
| Satin stained by prodigiosin | Escherichia coli | 91 | |||
| Staphylococcus aureus | 84 | ||||
| Linen stained by prodigiosin | Escherichia coli | 97 | |||
| Staphylococcus aureus | 70 | ||||
| Dacron stained by prodigiosin | Escherichia coli | 90 | |||
| Staphylococcus aureus | 84 | ||||
| Gabardine stained by prodigiosin | Escherichia coli | 19 | |||
| Staphylococcus aureus | 15 | ||||
| Serratia marcescens | Silk pH 2.1 | Staphylococcus aureus | Antibacterial rate | 93.17% | [75] |
| Escherichia coli | 25.12% | ||||
| Silk pH 8.1 | Staphylococcus aureus | 87.80% | |||
| Escherichia coli | 14.70% | ||||
| Serratia marcescens | 25–400 µg/mL | Staphylococcus aureus | IC50 | 51.17 µg/mL | [71] |
| Listeria monocytogenes | 51.54 µg/mL | ||||
| Enterococcus faecium | 26.18 µg/mL | ||||
| Bacillus cereus | 33.61 µg/mL | ||||
| Salmonella enteritidis | 56.56 µg/mL | ||||
| Proteus vulgaris | 50.81 µg/mL | ||||
| Pseudomonas aeruginosa | 69.71 µg/mL | ||||
| klebsiella pneumoniae | 48.63 µg/mL | ||||
| Aeromonas hydrophila | 66.98 µg/mL | ||||
| Escherichia coli | 44.20 µg/mL | ||||
| E. coli O157:H7 | 20.31 µg/mL | ||||
| Achromobacter denitrificans SP1 | 25 µg/mL | Proteus mirabilis | Maximum zone of inhibition (mm) | 17 | [72] |
| Staphylococcus aureus | 15 | ||||
| Serratia marcescens | 250 μg/mL | MRSA | Maximum zone of inhibition (mm) | 20 ± 0.33 | [74] |
| Staphylococcus aureus | 20 ± 0.0 | ||||
| Enterococcus faecalis | 20 ± 0.88 | ||||
| Escherichia coli | 22 ± 0.41 | ||||
| 500 μg/mL | MRSA | 21 ± 0.00 | |||
| Staphylococcus aureus | 22 ± 0.33 | ||||
| Enterococcus faecalis | 20 ± 0.33 | ||||
| Escherichia coli | 27 ± 0.82 | ||||
| P. putida strain pig-r2 | 24.48 μg/mL | Corynebacterium glutamicum | Minimal inhibitory concentration (MIC) | 2.56 μg/mL | [73] |
| Prodigiosin Source | Concentration | Fungi | Parameter of Antifungal Action | Value | Reference |
|---|---|---|---|---|---|
| Serratia plymuthica and S. marcescens | 3.8 µM | Batrachochytrium dendrobatidis (Bd) | IC50 | 3.8 µM | [76] |
| 27.3 μM | B. salamandrivorans (Bsal) | IC50 | 27.3 μM | ||
| 10 μM | Batrachochytrium dendrobatidis (Bd) | Minimum inhibitory concentration (MIC) | 10 μM | ||
| 50 μM | B. salamandrivorans (Bsal) | Minimum inhibitory concentration (MIC) | 50 μM | ||
| Serratia sp. | Crude extract from rhizosphere of Bacopa monnieri (L.) | Pythium myriotylum | Percent inhibition | 71.33 | [77] |
| Rhizoctonia solani | Percent inhibition | 61.33 | |||
| Sclerotium rolfsii | Percent inhibition | 49.33 | |||
| Phytophthora infestans | Percent inhibition | 48.66 | |||
| Fusarium oxysporum | Percent inhibition | 31 | |||
| Serratia rubidaea Mar61-01 | 450 µg/mL | C. nymphaeae | Percent inhibition | 29.27 | [78] |
| 1000 µg/mL | C. nymphaeae | Percent inhibition | 100 | ||
| Serratia spp. isolated from medicinal plant Plumbago indica | - | Pythium myriotylum | Percent inhibition | 40 | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araújo, R.G.; Zavala, N.R.; Castillo-Zacarías, C.; Barocio, M.E.; Hidalgo-Vázquez, E.; Parra-Arroyo, L.; Rodríguez-Hernández, J.A.; Martínez-Prado, M.A.; Sosa-Hernández, J.E.; Martínez-Ruiz, M.; et al. Recent Advances in Prodigiosin as a Bioactive Compound in Nanocomposite Applications. Molecules 2022, 27, 4982. https://doi.org/10.3390/molecules27154982
Araújo RG, Zavala NR, Castillo-Zacarías C, Barocio ME, Hidalgo-Vázquez E, Parra-Arroyo L, Rodríguez-Hernández JA, Martínez-Prado MA, Sosa-Hernández JE, Martínez-Ruiz M, et al. Recent Advances in Prodigiosin as a Bioactive Compound in Nanocomposite Applications. Molecules. 2022; 27(15):4982. https://doi.org/10.3390/molecules27154982
Chicago/Turabian StyleAraújo, Rafael G., Natalia Rodríguez Zavala, Carlos Castillo-Zacarías, Mario E. Barocio, Enrique Hidalgo-Vázquez, Lizeth Parra-Arroyo, Jesús Alfredo Rodríguez-Hernández, María Adriana Martínez-Prado, Juan Eduardo Sosa-Hernández, Manuel Martínez-Ruiz, and et al. 2022. "Recent Advances in Prodigiosin as a Bioactive Compound in Nanocomposite Applications" Molecules 27, no. 15: 4982. https://doi.org/10.3390/molecules27154982
APA StyleAraújo, R. G., Zavala, N. R., Castillo-Zacarías, C., Barocio, M. E., Hidalgo-Vázquez, E., Parra-Arroyo, L., Rodríguez-Hernández, J. A., Martínez-Prado, M. A., Sosa-Hernández, J. E., Martínez-Ruiz, M., Chen, W. N., Barceló, D., Iqbal, H. M. N., & Parra-Saldívar, R. (2022). Recent Advances in Prodigiosin as a Bioactive Compound in Nanocomposite Applications. Molecules, 27(15), 4982. https://doi.org/10.3390/molecules27154982










