High Bio-Content Thermoplastic Polyurethanes from Azelaic Acid
Abstract
:1. Introduction
2. Results and Discussions
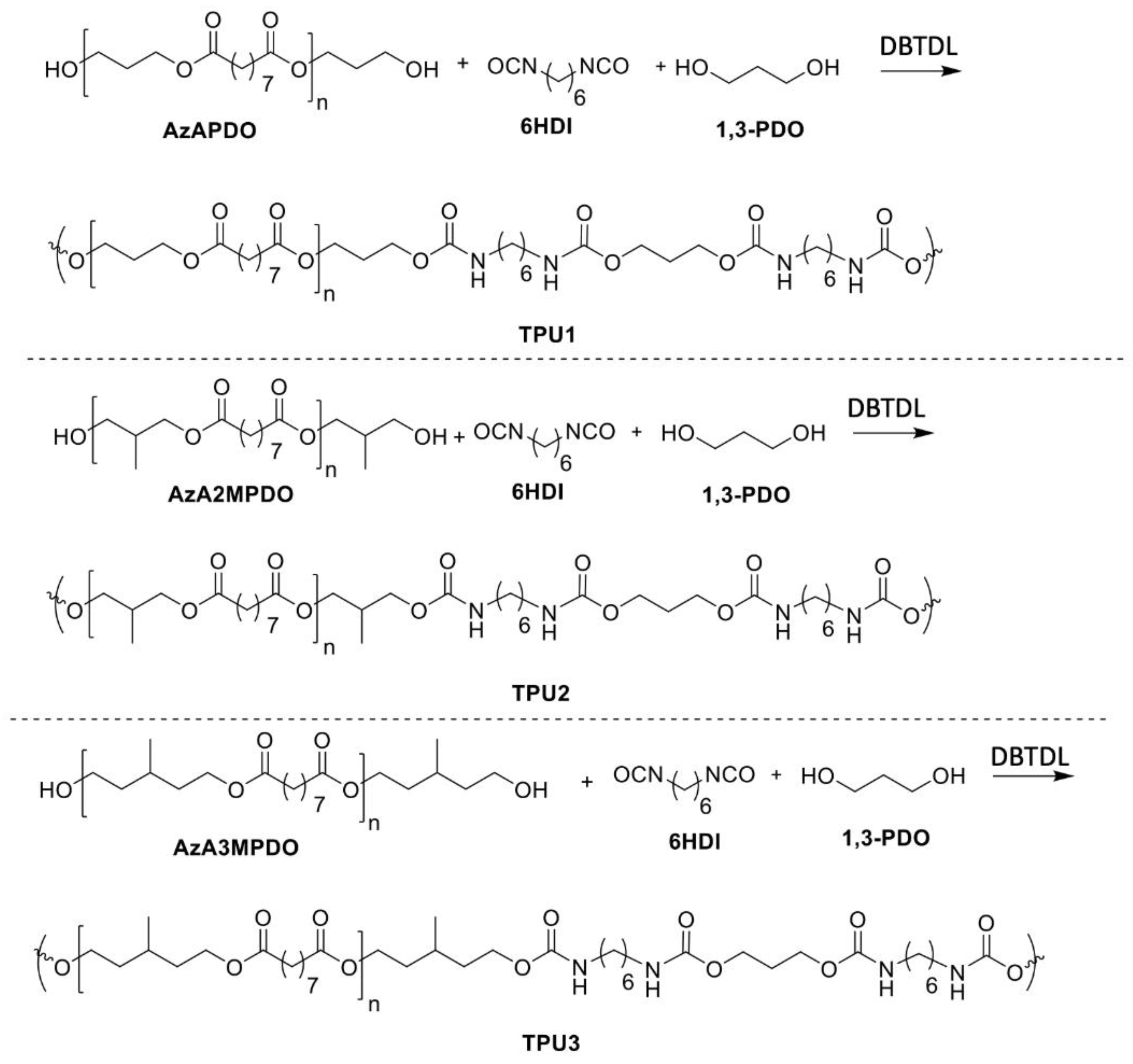
2.1. Polyester Polyol Synthesis
2.2. Synthesis of Thermoplastic Polyurethanes
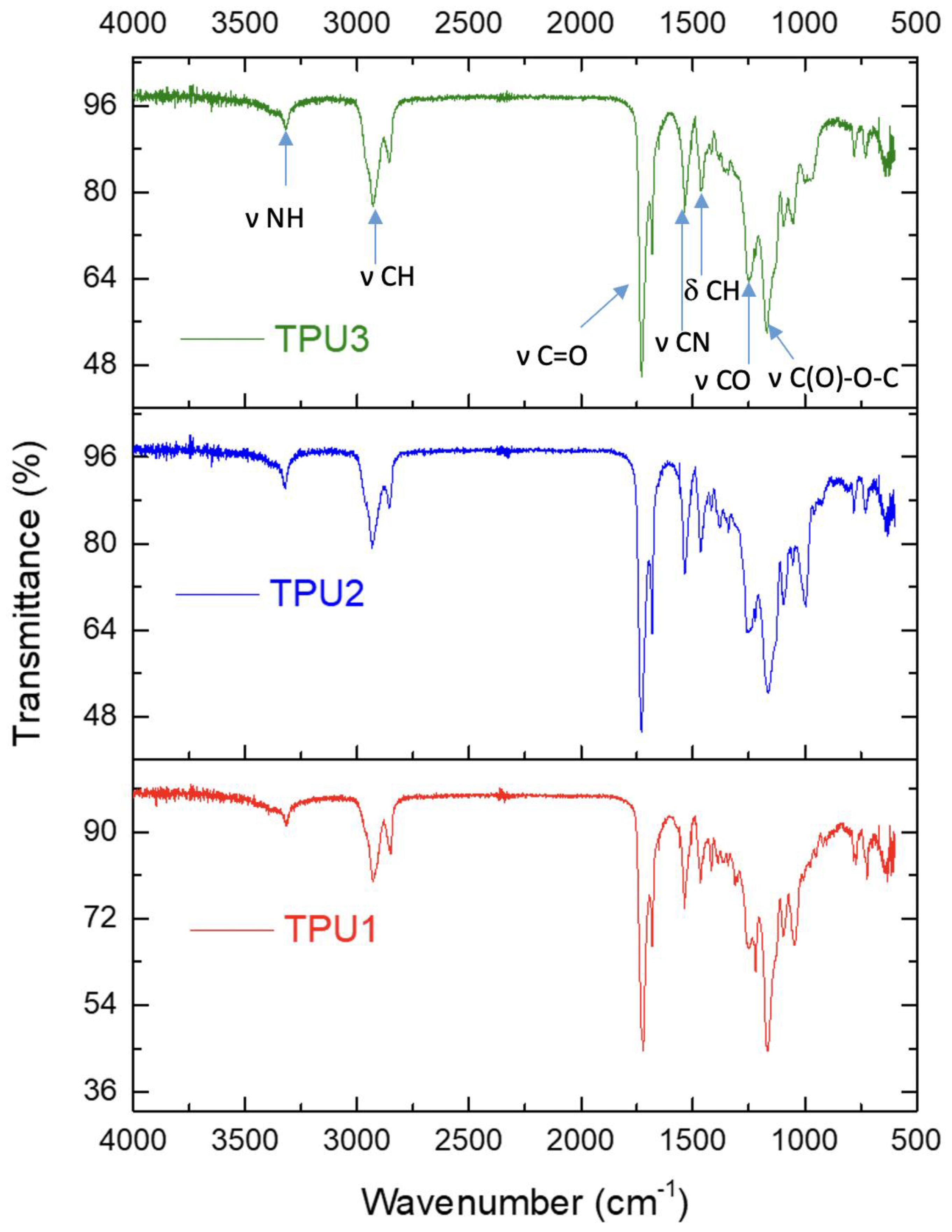
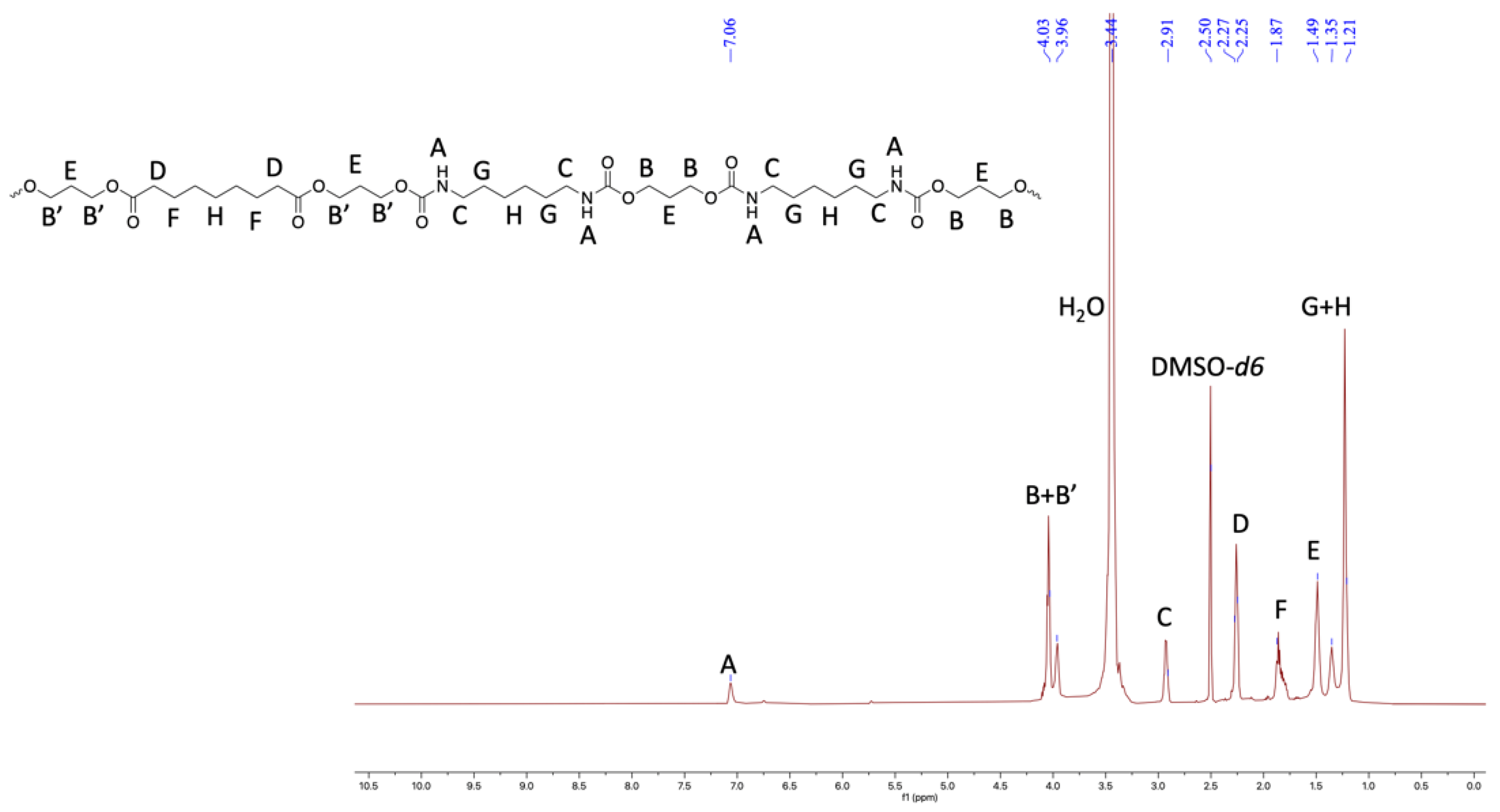
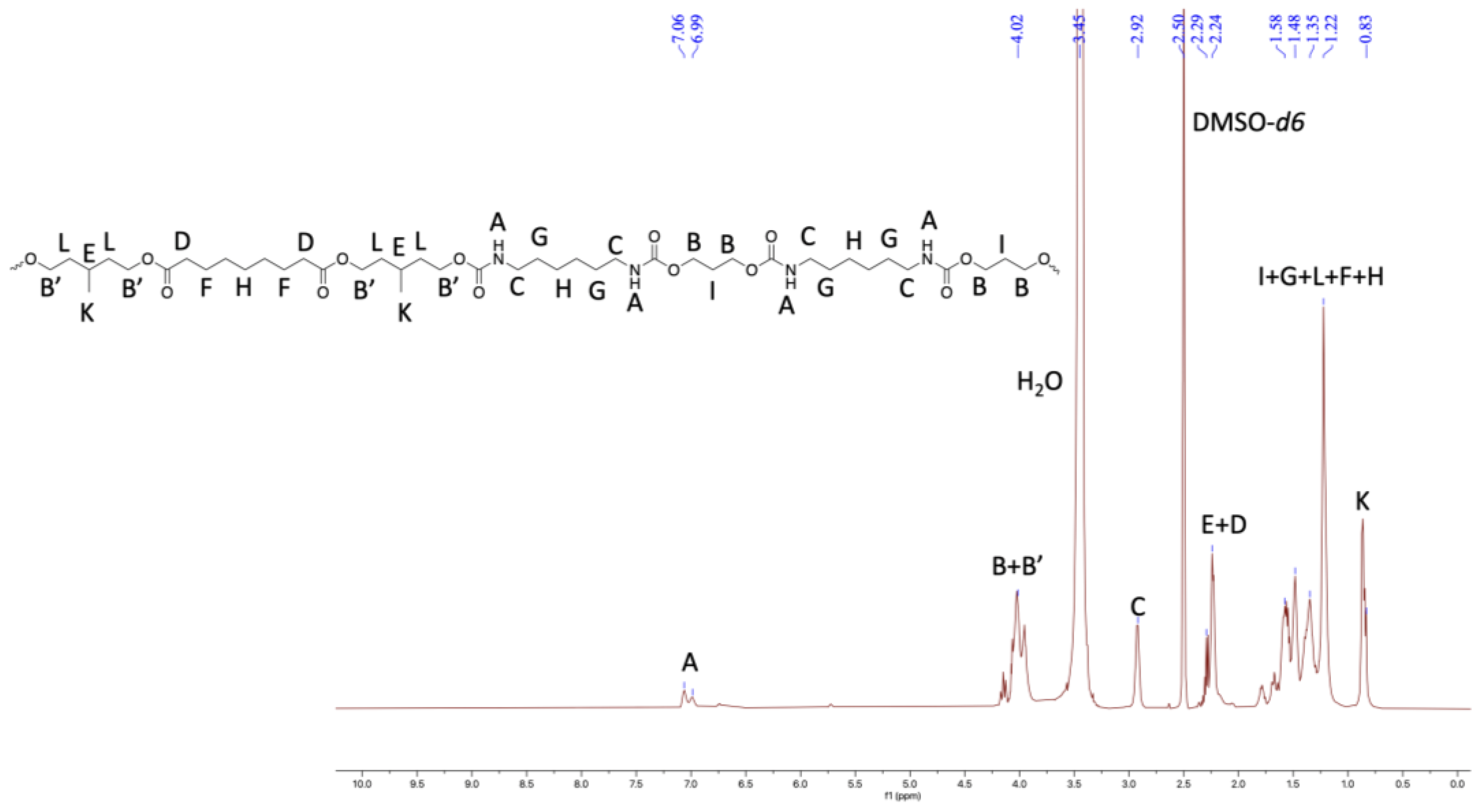
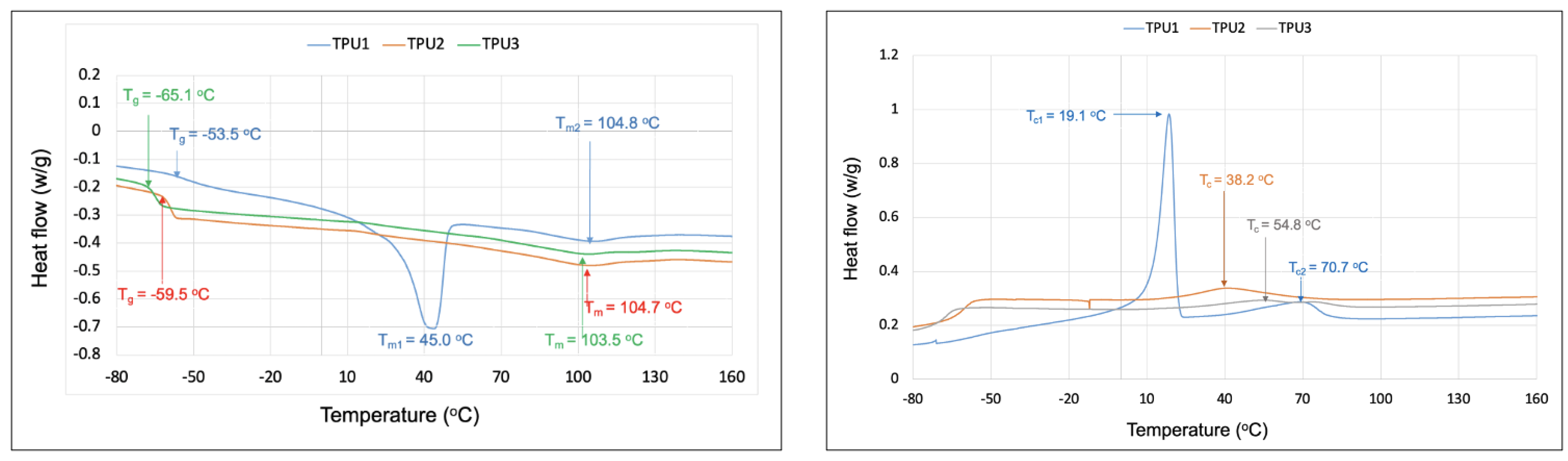
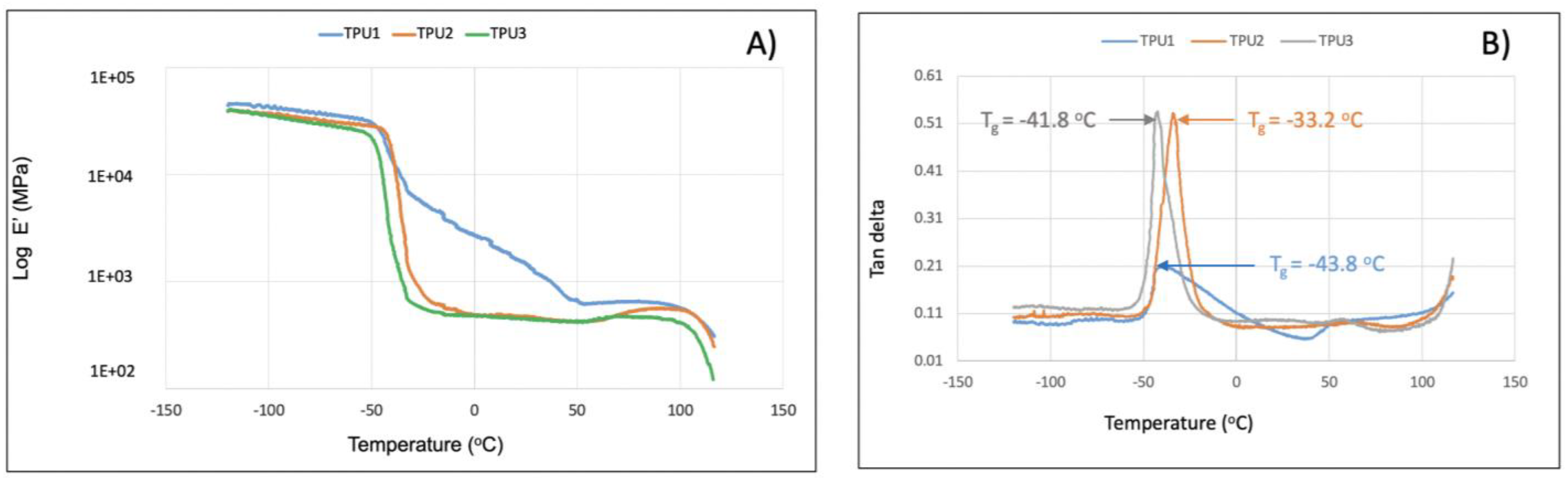
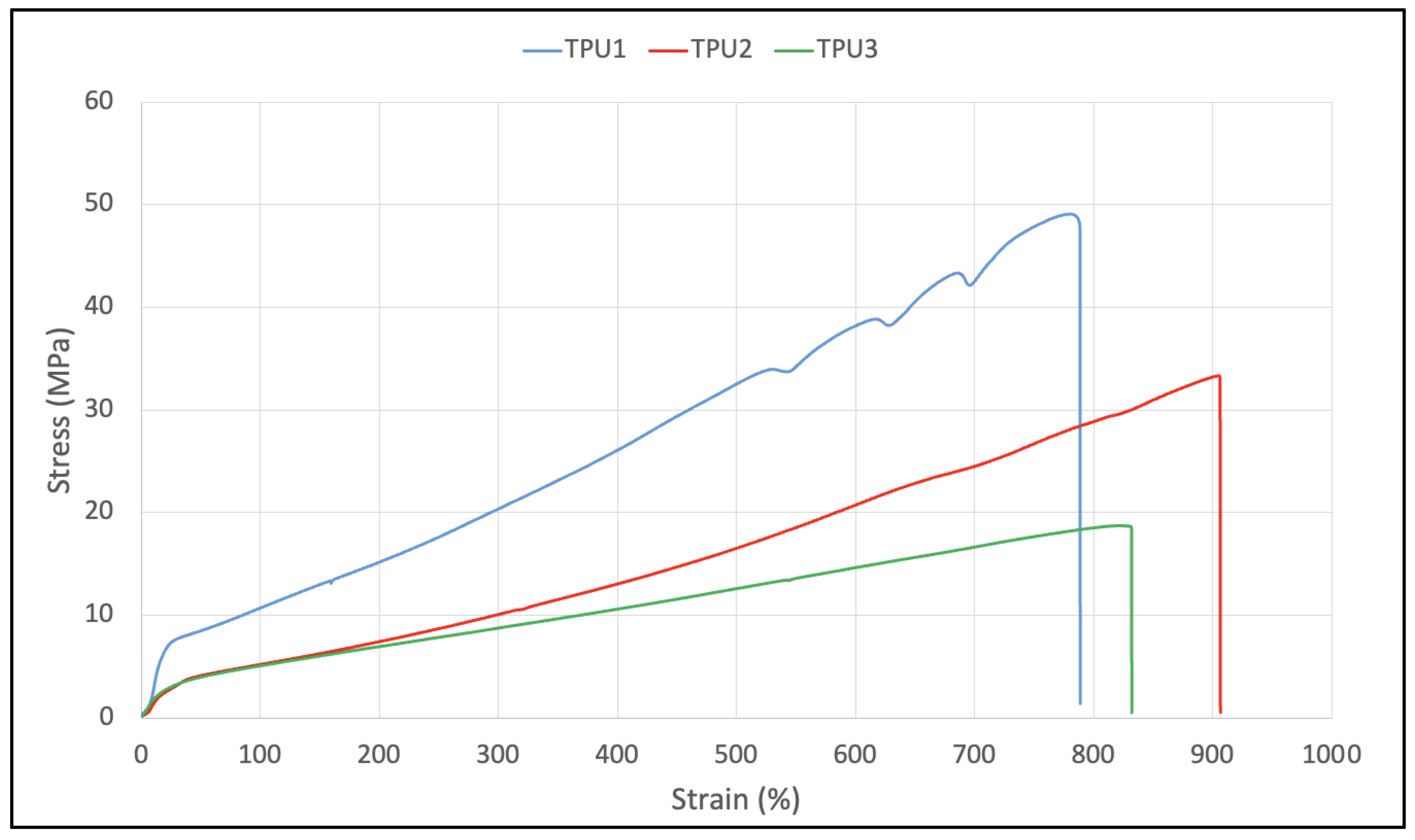
2.3. Properties of Thermoplastic Polyurethanes
3. Materials and Methods
3.1. Materials
3.2. Measurements
3.3. Synthesis Method for Polyester Polyols
3.4. Synthesis Method for TPUs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Tschan, M.J.L.; Brulé, E.; Haquette, P.; Thomas, C.M. Synthesis of Biodegradable Polymers from Renewable Resources. Polym. Chem. 2012, 3, 836–851. [Google Scholar] [CrossRef]
- Sawpan, M.A. Polyurethanes from Vegetable Oils and Applications: A Review. J. Polym. Res. 2018, 25, 184. [Google Scholar] [CrossRef]
- Maisonneuve, L.; Chollet, G.; Grau, E.; Cramail, H. Vegetable Oils: A Source of Polyols for Polyurethane Materials. OCL 2016, 23, D508. [Google Scholar] [CrossRef] [Green Version]
- Rajput, B.S.; Gaikwad, S.R.; Menon, S.K.; Chikkali, S.H. Sustainable Polyacetals from Isohexides. Green Chem. 2014, 16, 3810. [Google Scholar] [CrossRef]
- Rajput, B.S.; Chander, U.; Arole, K.; Stempfle, F.; Menon, S.; Mecking, S.; Chikkali, S.H. Synthesis of Renewable Copolyacetals with Tunable Degradation. Macromol. Chem. Phys. 2016, 217, 1396–1410. [Google Scholar] [CrossRef] [Green Version]
- Rajput, B.S.; Ram, F.; Menon, S.K.; Shanmuganathan, K.; Chikkali, S.H. Cross-Metathesis of Biorenewable Dioxalates and Diols to Film-Forming Degradable Polyoxalates. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 1584–1592. [Google Scholar] [CrossRef]
- Hai, T.A.P.; Tessman, M.; Neelakantan, N.; Samoylov, A.A.; Ito, Y.; Rajput, B.S.; Pourahmady, N.; Burkart, M.D. Renewable Polyurethanes from Sustainable Biological Precursors. Biomacromolecules 2021, 22, 1770–1794. [Google Scholar] [CrossRef]
- Pandey, S.; Rajput, B.S.; Chikkali, S.H. Refining Plant Oils and Sugars to Platform Chemicals, Monomers, and Polymers. Green Chem. 2021, 23, 4255–4295. [Google Scholar] [CrossRef]
- Rosenboom, J.G.; Langer, R.; Traverso, G. Bioplastics for a Circular Economy. Nat. Rev. Mater. 2022, 7, 117–137. [Google Scholar] [CrossRef] [PubMed]
- Gandini, A.; Lacerda, T.M.; Carvalho, A.J.F.; Trovatti, E. Progress of Polymers from Renewable Resources: Furans, Vegetable Oils, and Polysaccharides. Chem. Rev. 2016, 116, 1637–1669. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.M.F.; Rossi, L.M.; Debecker, D.P.; Leonard, K.C.; Li, Z.; Makhubela, B.C.E.; Zhao, C.; Kleij, A. Can CO2and Renewable Carbon Be Primary Resources for Sustainable Fuels and Chemicals? ACS Sustain. Chem. Eng. 2021, 9, 12427–12430. [Google Scholar] [CrossRef]
- Datta, J.; Kasprzyk, P. Thermoplastic Polyurethanes Derived from Petrochemical or Renewable Resources: A Comprehensive Review. Polym. Eng. Sci. 2018, 58, E14–E35. [Google Scholar] [CrossRef] [Green Version]
- Tharcis, M.; Badel, T.; Jéol, S.; Fleury, E.; Méchin, F. High Elongation Thermoplastic Polyester-Urethanes Based on Widely Available Diacid Intermediates. J. Appl. Polym. Sci. 2016, 133, 1–15. [Google Scholar] [CrossRef]
- Wan, Y.; Lee, J.M. Toward Value-Added Dicarboxylic Acids from Biomass Derivatives via Thermocatalytic Conversion. ACS Catal. 2021, 11, 2524–2560. [Google Scholar] [CrossRef]
- Sonnenschein, M.F.; Guillaudeu, S.J.; Landes, B.G.; Wendt, B.L. Comparison of Adipate and Succinate Polyesters in Thermoplastic Polyurethanes. Polymer 2010, 51, 3685–3692. [Google Scholar] [CrossRef]
- Mohd Noor, N.; Sendijarevic, A.; Sendijarevic, V.; Sendijarevic, I.; Tuan Ismail, T.N.M.; Mohd Noor, M.A.; Shoot Kian, Y.; Abu Hassan, H. Comparison of Adipic Versus Renewable Azelaic Acid Polyester Polyols as Building Blocks in Soft Thermoplastic Polyurethanes. JAOCS, J. Am. Oil Chem. Soc. 2016, 93, 1529–1540. [Google Scholar] [CrossRef]
- Tuan Ismail, T.N.M.; Ibrahim, N.A.; Sendijarevic, V.; Sendijarevic, I.; Schiffman, C.M.; Hoong, S.S.; Mohd Noor, M.A.; Poo Palam, K.D.; Yeong, S.K.; Idris, Z.; et al. Thermal and Mechanical Properties of Thermoplastic Urethanes Made from Crystalline and Amorphous Azelate Polyols. J. Appl. Polym. Sci. 2019, 136, 1–11. [Google Scholar] [CrossRef]
- Phung Hai, T.A.; Neelakantan, N.; Tessman, M.; Sherman, S.D.; Griffin, G.; Pomeroy, R.; Mayfield, S.P.; Burkart, M.D. Flexible Polyurethanes, Renewable Fuels, and Flavorings from a Microalgae Oil Waste Stream. Green Chem. 2020, 22, 3088–3094. [Google Scholar] [CrossRef]
- Tuan Ismail, T.N.M.; Azowa Ibrahim, N.; Sendijarevic, A.; Sendijarevic, I.; Schiffman, C.M.; Hoong, S.S.; Mohd Noor, M.A.; Poo Palam, K.D.; Yeong, S.K.; Idris, Z.; et al. Oscillatory Structure-Property Correlation in Azelate Polyols and Thermoplastic Polyurethanes. J. Appl. Polym. Sci. 2018, 135, 1–10. [Google Scholar] [CrossRef]
- Tuan Ismail, T.N.M.; Ibrahim, N.A.; Poo Palam, K.D.; Sendijarevic, A.; Sendijarevic, I.; Schiffman, C.M.; Hoong, S.S.; Mohd Noor, M.A.; Yeong, S.K.; Abd. Malek, E.; et al. Improved Dynamic Properties of Thermoplastic Polyurethanes Made from Co-Monomeric Polyester Polyol Soft Segments Based on Azelaic Acid. J. Appl. Polym. Sci. 2021, 138, 1–14. [Google Scholar] [CrossRef]
- Saralegi, A.; Rueda, L.; Fernández-D’Arlas, B.; Mondragon, I.; Eceiza, A.; Corcuera, M.A. Thermoplastic Polyurethanes from Renewable Resources: Effect of Soft Segment Chemical Structure and Molecular Weight on Morphology and Final Properties. Polym. Int. 2013, 62, 106–115. [Google Scholar] [CrossRef]
- Parcheta, P.; Głowińska, E.; Datta, J. Effect of Bio-Based Components on the Chemical Structure, Thermal Stability and Mechanical Properties of Green Thermoplastic Polyurethane Elastomers. Eur. Polym. J. 2020, 123, 109422. [Google Scholar] [CrossRef]
- Siyanbola, T.O.; Sasidhar, K.; Rao, B.V.S.K.; Narayan, R.; Olaofe, O.; Akintayo, E.T.; Raju, K.V.S.N. Development of Functional Polyurethane-ZnO Hybrid Nanocomposite Coatings from Thevetia Peruviana Seed Oil. J. Am. Oil Chem. Soc. 2015, 92, 267–275. [Google Scholar] [CrossRef]
- Kojio, K.; Furukawa, M.; Nonaka, Y.; Nakamura, S. Control of Mechanical Properties of Thermoplastic Polyurethane Elastomers by Restriction of Crystallization of Soft Segment. Materials 2010, 3, 5097–5110. [Google Scholar] [CrossRef] [PubMed]




| Polyols | Azelaic Acid (mole) | 1,3-PDO (mole) | 2MPDO (mole) | 3MPDO (mole) | (d) Catalyst (mole%) | |
|---|---|---|---|---|---|---|
| 01 | (a) AzAPDO | 2.11 | 2.36 | - | - | 0.019 |
| 02 | (b) AzA2MDO | 1.96 | - | 2.23 | - | 0.020 |
| 03 | (c) AzA3MPDO | 1.73 | - | - | 2.01 | 0.022 |
| Run | Polyols | Acid Value (mg KOH/g) | Hydroxyl Value (mg KOH/g) | (a) Molecular Weight (by OH Number) | (b) Viscosity (70 °C, cP) | Physical State of Polyols at (25 °C) |
|---|---|---|---|---|---|---|
| 01 | AzAPDO | 1.4 | 56 | 2000 | 683 | Solid |
| 02 | AzA2MDO | 1.7 | 53 | 2100 | 781 | Liquid |
| 03 | AzA3MPDO | 1.0 | 57 | 2000 | 504 | Liquid |
| TPUs | Chemical Compositions | Molar Ratio (Polyol:PDO:6HDI) | Hard Segment Concentration (wt %) | Bio-Carbon Content (%) |
|---|---|---|---|---|
| TPU1 | AzAPDO, PDO, 6HDI | 1:1:2.1 | 18.1 | ~85 |
| TPU2 | AzA2MPDO, PDO, 6HDI | 1:1:2.1 | 17.4 | ~65 |
| TPU3 | AzA3MPDO, PDO, 6HDI | 1:1:2.1 | 18.2 | ~85 |
| Properties | TPU1 | TPU2 | TPU3 |
|---|---|---|---|
| GPC Mn (g/mol) | 55 × 103 | 41 × 103 | 63 × 103 |
| GPC Mw (g/mol) | 171 × 103 | 138 × 103 | 179 × 103 |
| GPC Mw/Mn | 3.11 | 3.36 | 2.82 |
| (a) Tensile strength at RT, MPa | 48 | 31 | 17 |
| (a) Elongation at break at RT, % | 765 | 881 | 809 |
| Shore A hardness at RT | 96 | 85 | 82 |
| Thermal transitions (DSC, °C) | Tg = −53.5 °C | Tg = −59.5 °C | Tg = −65.1 °C |
| Tm1 = 45 °C | Tm = 104.7 °C | Tm = 103.5 °C | |
| Tm2 = 104.8 °C |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajput, B.S.; Hai, T.A.P.; Burkart, M.D. High Bio-Content Thermoplastic Polyurethanes from Azelaic Acid. Molecules 2022, 27, 4885. https://doi.org/10.3390/molecules27154885
Rajput BS, Hai TAP, Burkart MD. High Bio-Content Thermoplastic Polyurethanes from Azelaic Acid. Molecules. 2022; 27(15):4885. https://doi.org/10.3390/molecules27154885
Chicago/Turabian StyleRajput, Bhausaheb S., Thien An Phung Hai, and Michael D. Burkart. 2022. "High Bio-Content Thermoplastic Polyurethanes from Azelaic Acid" Molecules 27, no. 15: 4885. https://doi.org/10.3390/molecules27154885
APA StyleRajput, B. S., Hai, T. A. P., & Burkart, M. D. (2022). High Bio-Content Thermoplastic Polyurethanes from Azelaic Acid. Molecules, 27(15), 4885. https://doi.org/10.3390/molecules27154885






