Zero-Biased Photoelectrochemical Detection of Cardiac Biomarker Myoglobin Based on CdSeS/ZnS Quantum Dots and Barium Titanate Perovskite
Abstract
1. Introduction
2. Results and Discussion
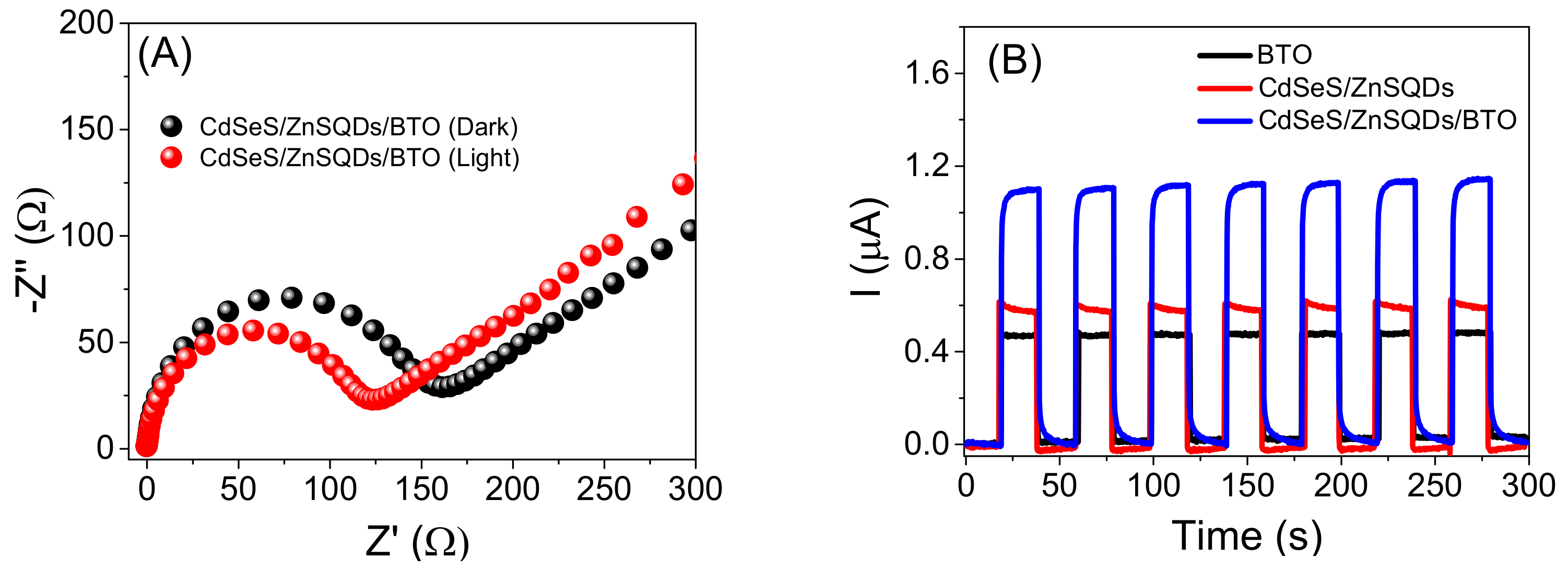
2.1. Characterization of the Materials by SEM, EDX, TEM, XRD and EIS
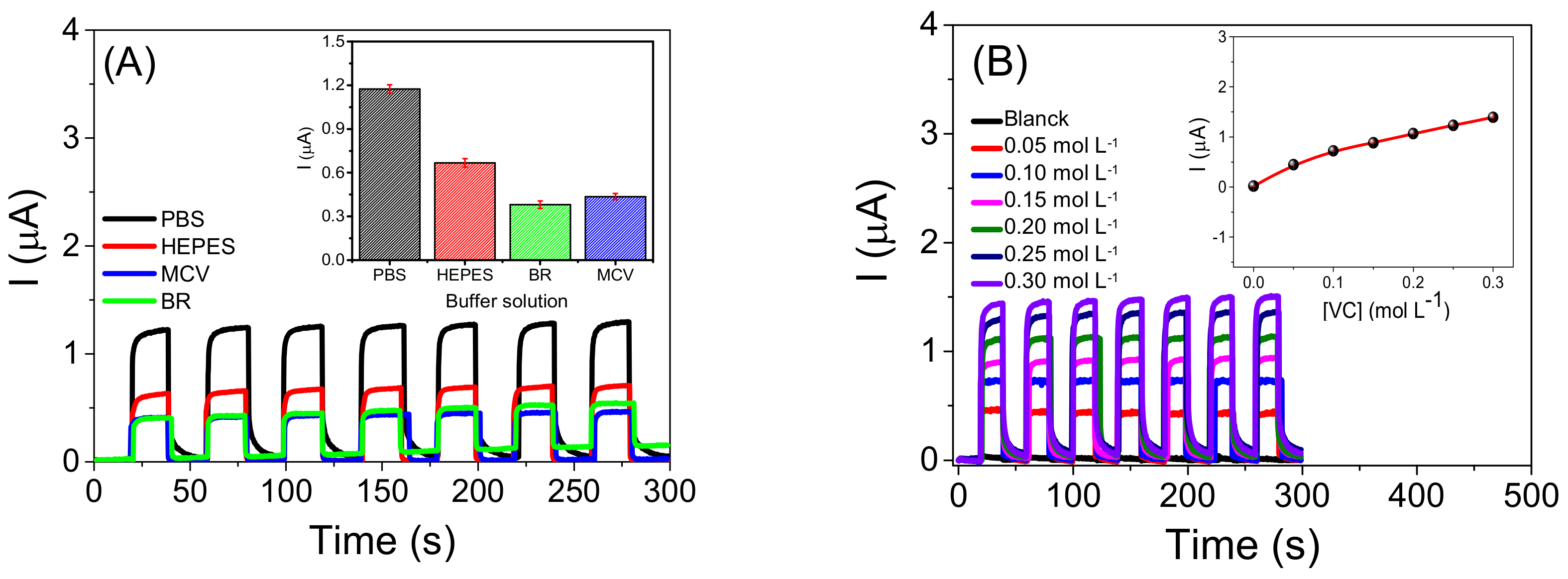
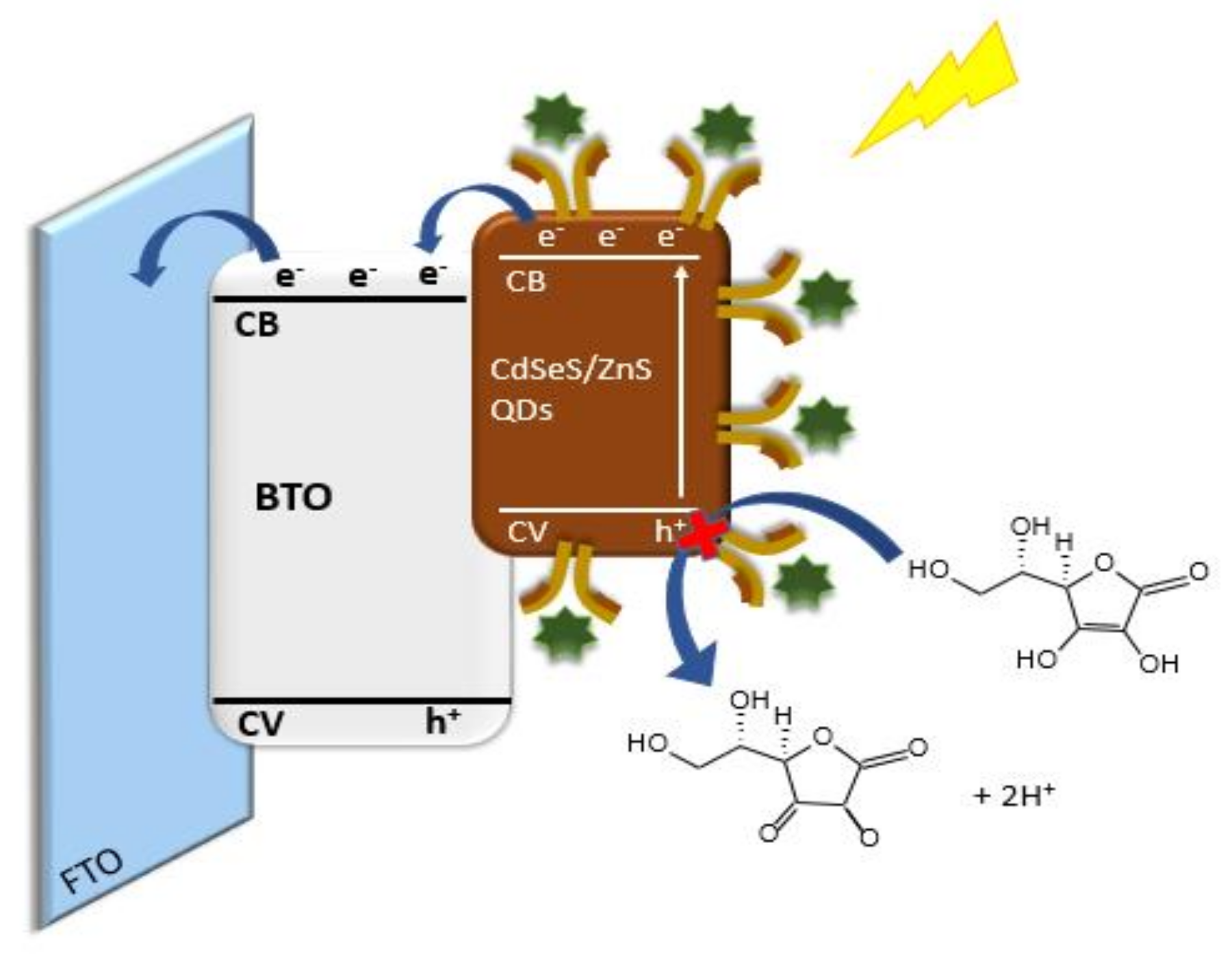
2.2. Characterization of the Photoelectrochemical Platform and Optimization of Experimental Conditions for the CdSeS/ZnSQDs/BTO/FTO Sensor
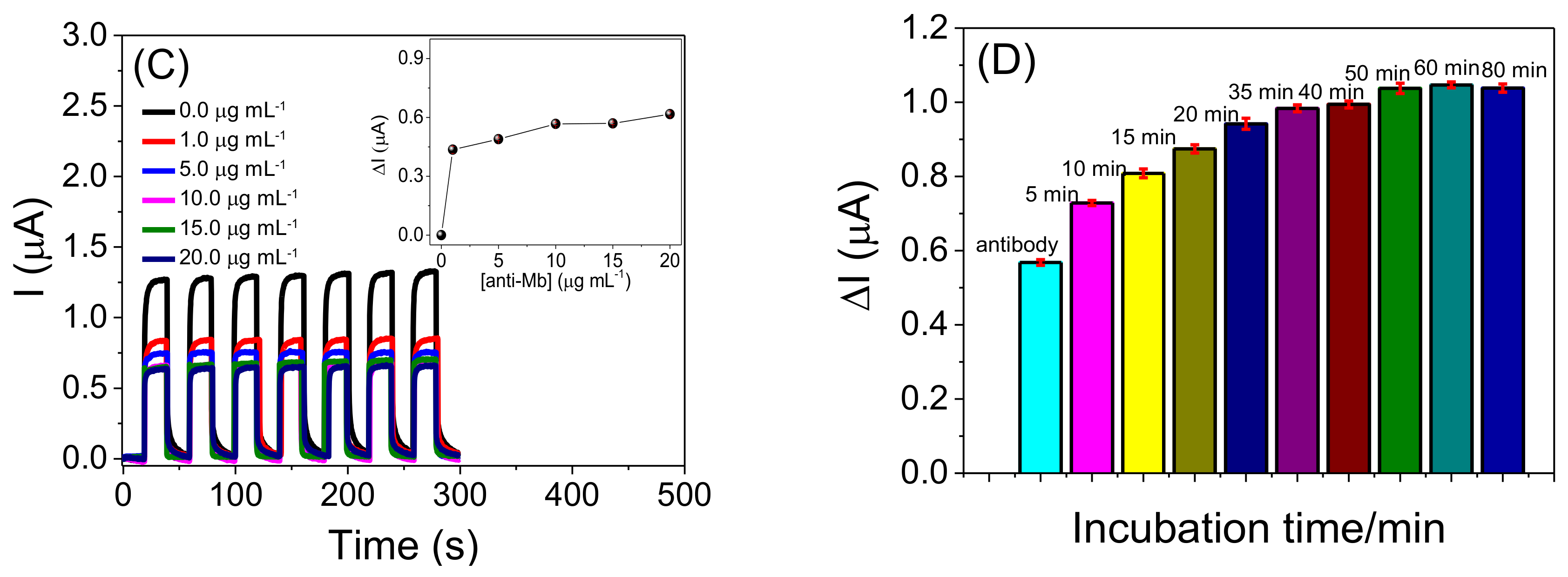
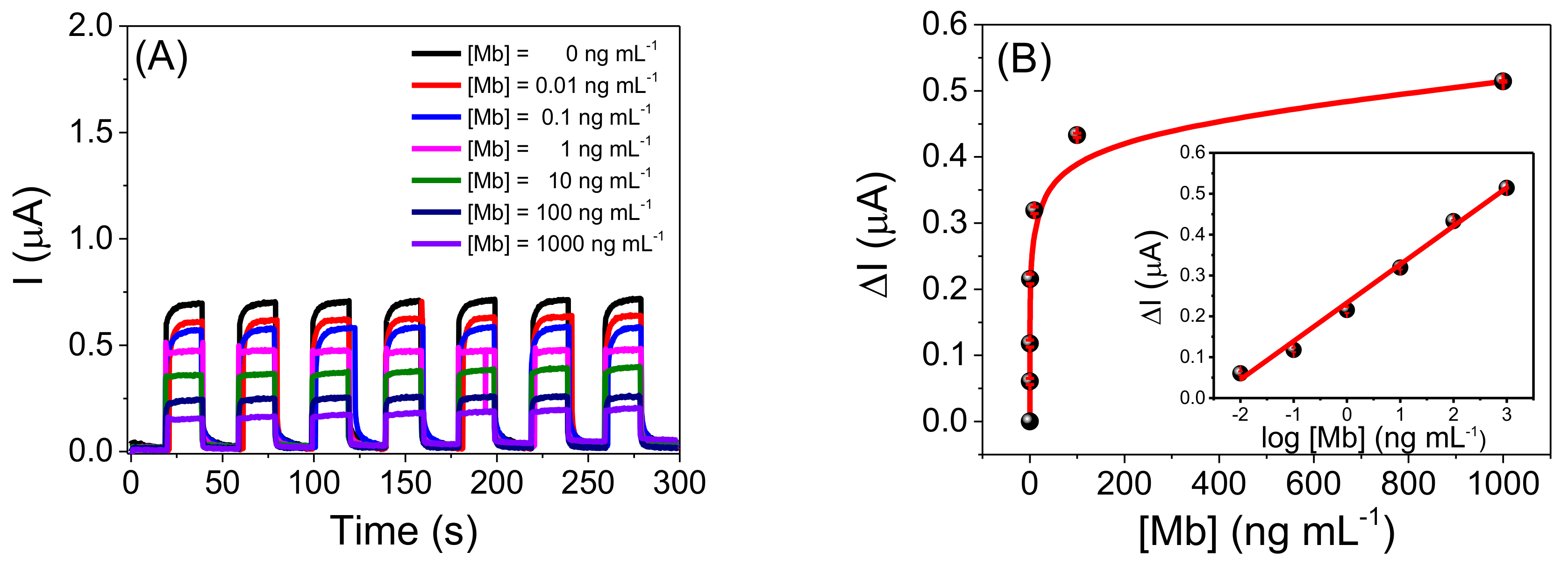
2.3. Analytical Performance of the Photoelectrochemical Immunosensor
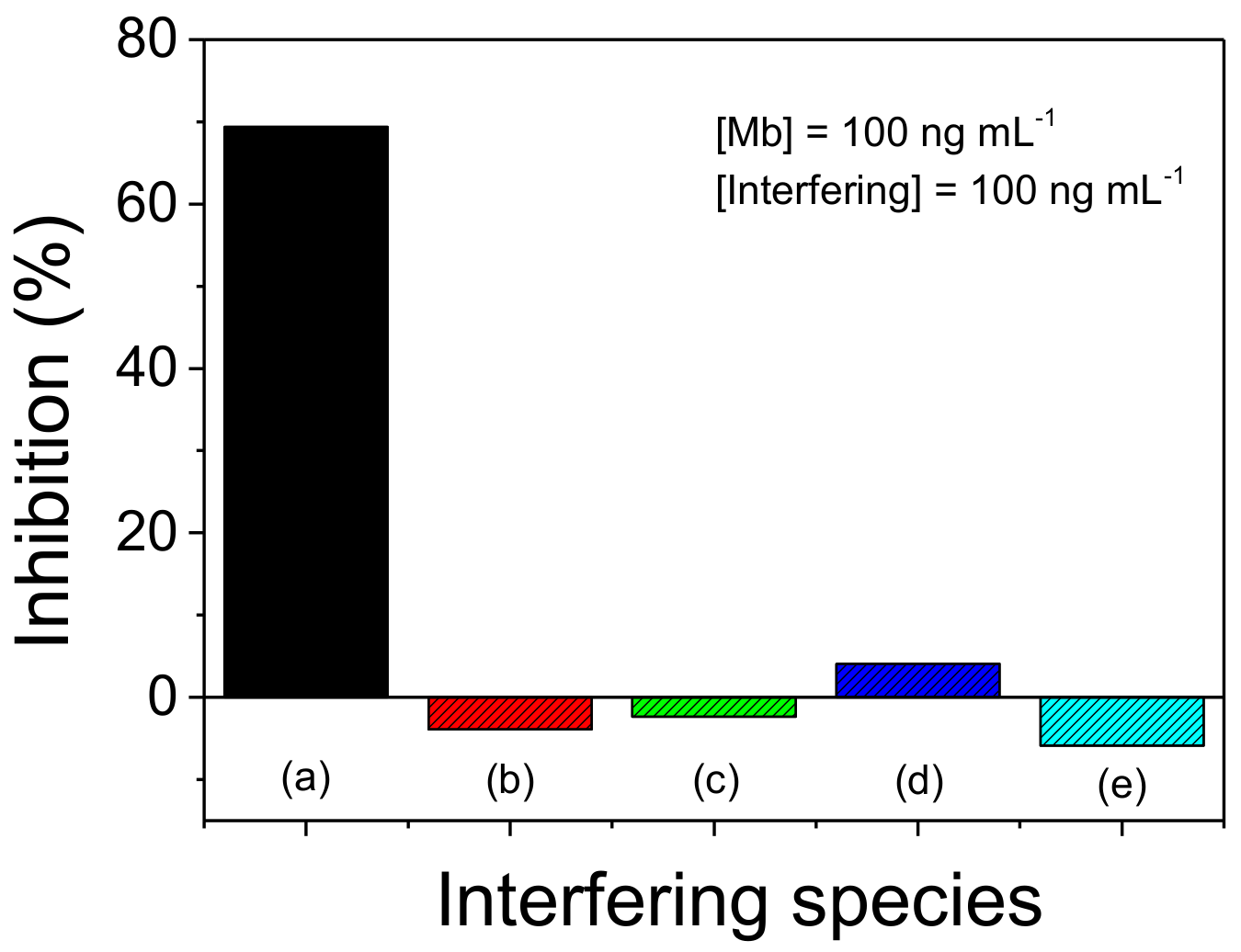
2.4. Application of the Photoelectrochemical Immunosensor in Artificial Samples and Study of Interferents
3. Materials and Methods
3.1. Reagents
3.2. Morphological, XRD, Electrochemical, and Photoelectrochemical Measurements
3.3. Obtention of the Barium Titanate and Modification of the Photoelectrochemical Imunossensor
3.4. Preparation of Artificial Blood Plasma and Urine Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Li, Q.; Liu, H.; Jiang, S.; Zhang, M.; Shan, K.; Ke, W.; Zhao, D.; Nian, Y.; Li, C. The effects of high pressure treatment on the structural and digestive properties of myoglobin. Food Res. Int. 2022, 156, 111193. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Tuteja, S.K.; Sillu, D.; Deep, A.; Suri, C.R. Gold nanoparticles-reduced graphene oxide based electrochemical immunosensor for the cardiac biomarker myoglobin. Microchim. Acta 2016, 183, 1729–1738. [Google Scholar] [CrossRef]
- Zapp, E.; Westphal, E.; Gallardo, H.; Souza, B.; Vieira, I.C. Liquid crystal and gold nanoparticles applied to electrochemical immunosensor for cardiac biomarker. Biosens. Bioelectron. 2014, 59, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.S.; Kim, S.Y.; Kim, K.S.; Hong, S.; Oh, M.J.; Nam, M.G.; Kim, W.-J.; Park, J.; Chung, C.-H.; Choe, W.-S.; et al. Controlling inter-sheet-distance in reduced graphene oxide electrodes for highly sensitive electrochemical impedimetric sensing of myoglobin. Sens. Actuators B Chem. 2020, 305, 127477. [Google Scholar] [CrossRef]
- Sharma, R.V.; Tanwar, V.K.; Mishra, S.K.; Biradar, A.M. Electrochemical impedance immunosensor for the detection of cardiac biomarker Myogobin (Mb) in aqueous solution. Thin Solid Films 2010, 519, 1167–1170. [Google Scholar] [CrossRef]
- Wang, L.; Xu, M.; Huang, R.; Chang, X.; Chen, C.; Li, L.; Zhang, Z.; Han, Y. A Dual-Label Time-Resolved Fluorescence Immunoassay for the Simultaneous Determination of Cardiac Troponin T and Myoglobin. SLAS Technol. 2017, 22, 130–135. [Google Scholar] [CrossRef][Green Version]
- Cho, I.-H.; Paek, E.-H.; Kim, Y.-K.; Kim, J.-H.; Paek, S.-H. Chemiluminometric enzyme-linked immunosorbent assays (ELISA)-on-a-chip biosensor based on cross-flow chromatography. Anal. Chim. Acta 2009, 632, 247–255. [Google Scholar] [CrossRef]
- Yamasaki, T.; Hirakawa, Y.; Momma, K.; Yamaguchi, Y.M.; Kotoura, S.; Miyake, S.; Narita, H. Enzyme-Linked Immunosorbent Assay for Pork Determination in Raw and Heated Meats: Combination of Monoclonal Antibodies to Denatured Porcine Myoglobin and Sodium Dodecyl Sulfate Extraction. ACS Food Sci. Technol. 2022, 2, 136–142. [Google Scholar] [CrossRef]
- Darain, F.; Yager, P.; Gan, K.L.; Tjin, S.C. On-chip detection of myoglobin based on fluorescence. Biosens. Bioelectron. 2009, 24, 1744–1750. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, H.; Li, R.; Ke, P.; Zhu, H.; Guo, H.; Liu, M.; Sun, H. An immunomagnetic separation based fluorescence immunoassay for rapid myoglobin quantification in human blood. Anal. Methods 2016, 8, 7324–7330. [Google Scholar] [CrossRef]
- Osman, B.; Uzun, L.; Beşirli, N.; Denizli, A. Microcontact imprinted surface plasmon resonance sensor for myoglobin detection. Mater. Sci. Eng. C 2013, 33, 3609–3614. [Google Scholar] [CrossRef]
- Piliarik, M.; Bocková, M.; Homola, J. Surface plasmon resonance biosensor for parallelized detection of protein biomarkers in diluted blood plasma. Biosens. Bioelectron. 2010, 26, 1656–1661. [Google Scholar] [CrossRef]
- Naveena, B.M.; Faustman, C.; Tatiyaborworntham, N.; Yin, S.; Ramanathan, R.; Mancini, R.A. Detection of 4-hydroxy-2-nonenal adducts of turkey and chicken myoglobins using mass spectrometry. Food Chem. 2010, 122, 836–840. [Google Scholar] [CrossRef]
- Dosi, R.; Carusone, A.; Chambery, A.; Severino, V.; Parente, A.; Maro, A.D. Rapid primary structure determination of myoglobins by a complementary approach based on mass spectrometry and Edman degradation. Food Chem. 2012, 133, 1646–1652. [Google Scholar] [CrossRef]
- Di Giuseppea, A.M.A.; Giarretta, N.; Lippert, M.; Severino, V.; Di Maro, A. An improved UPLC method for the detection of undeclared horse meat addition by using myoglobin as molecular marker. Food Chem. 2015, 169, 241–245. [Google Scholar] [CrossRef]
- Giaretta, N.; Di Giuseppe, A.M.A.; Lippert, M.; Parente, A.; Di Maro, A. Myoglobin as marker in meat adulteration: A UPLC method for determining the presence of pork meat in raw beef Burger. Food Chem. 2013, 141, 1814–1820. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, F.; Yang, X.; Wang, K.; Wang, H.; Deng, X. Sensitive point-of-care monitoring of cardiac biomarker myoglobin using aptamer and ubiquitous personal glucose meter. Biosens. Bioelectron. 2015, 64, 161–164. [Google Scholar] [CrossRef]
- Bahadır, E.B.; Sezgintürk, M.K. Applications of electrochemical immunosensors for early clinical diagnostics. Talanta 2015, 132, 162–174. [Google Scholar] [CrossRef]
- Moreira, F.T.C.; Dutra, R.A.F.; Noronha, J.P.C.; Sales, M.G.F. Electrochemical biosensor based on biomimetic material for myoglobin detection. Electrochim. Acta 2013, 107, 481–487. [Google Scholar] [CrossRef]
- Sun, X.; Li, C.; Zhu, Q.; Huang, H.; Jing, W.; Chen, Z.; Kong, L.; Han, L.; Wang, J.; Li, Y. A label-free photoelectrochemical immunosensor for detection of the milk allergen β-lactoglobulin based on Ag2S -sensitized spindle-shaped BiVO4/BiOBr heterojunction by an in situ growth method. Anal. Chim. Acta 2020, 1140, 122–131. [Google Scholar] [CrossRef]
- Lima, F.M.R.; Soares, R.-E.P.; Sinfrônio, F.S.M.; Maciel, A.P.; Menezes, A.S.; Pereira, S.R.F.; Damos, F.S.; Luz, R.C.S. Photoelectrochemical Immunosensor for Sensitive Quantification of Prostate Specific Antigen in Human Serum Samples Exploiting BaTiO3−CdS. ChemElectroChem 2020, 7, 3140–3150. [Google Scholar] [CrossRef]
- Huang, Y.; Deng, H.; Zhang, J.; Sun, H.; Li, W.; Li, C.; Zhang, Y.; Sun, D. A photoelectrochemical immunosensor based on ReS2 nanosheets for determination of collagen III related to abdominal aortic aneurysm. Microchem. J. 2021, 168, 106363. [Google Scholar] [CrossRef]
- Rani, D.S.; Meera, M.R. Photoelectrochemical water splitting behavior of CdSeQDs sensitized ferroelectric BaTiO3 perovskite heterostructure. Mater. Today Proc. 2021, 3, 1248–1253. [Google Scholar] [CrossRef]
- Tiwari, D.; Dunn, S. Photochemistry on a polarisable semiconductor: What do we understand today? J. Mater. Sci. 2009, 44, 5063–5079. [Google Scholar] [CrossRef]
- Cui, Y.; Briscoe, J.; Wang, Y.; Tarakina, N.V.; Dunn, S. Enhanced Photocatalytic Activity of Heterostructured Ferroelectric BaTiO3/α-Fe2O3 and the Significance of Interface Morphology Control. ACS Appl. Mater. Interfaces 2017, 9, 24518–24526. [Google Scholar] [CrossRef]
- Sharma, D.; Upadhyay, S.; Satsangi, V.R.; Shrivastav, R.; Waghmare, U.V.; Dass, S. Nanostructured BaTiO3/Cu2O heterojunction with improved photoelectrochemical activity for H2 evolution: Experimental and first-principles analysis. Appl. Catal. B Environ. 2016, 189, 75–85. [Google Scholar] [CrossRef]
- Stoll, C.; Gehring, C.; Schubert, K.; Zanella, M.; Prak, W.J.; Lisdat, F. Photoelectrochemical signal chain based on quantum dots on gold—Sensitive to superoxide radicals in solution. Biosens. Bioelectron. 2008, 24, 260–265. [Google Scholar] [CrossRef]
- Monteiro, T.O.; Tanaka, A.A.; Damos, F.S.; Luz, R.C.S. Photoelectrochemical determination of tert-butylhydroquinone in edible oil samples employing CdSe/ZnS quantum dots and LiTCNE. Food Chem. 2017, 227, 16–21. [Google Scholar] [CrossRef]
- Santos, M.J.L.; Ferreira, J.; Radovanovic, E.; Romano, R.; Alves, O.L.; Girotto, E.M. Enhancement of the photoelectrochemical response of poly(terthiophenes) by CdS(ZnS) core-shell nanoparticles. Thin Solid Film. 2009, 517, 5523–5529. [Google Scholar] [CrossRef]
- Rana, M.; Jain, A.; Rani, V.; Chowdhury, P. Glutathione capped core/shell CdSeS/ZnS quantum dots as a medical imaging tool for cancer cells. Inorg. Chem. Commun. 2020, 112, 107723. [Google Scholar] [CrossRef]
- Aimi, A.; Horiuchi, K.; Yamaguchi, Y.; Ito, S.; Fujimoto, K. Disordered off-center direction of Ti+4 in pseudo-cubic type BaTiO3 prepared by mixed hydroxide process. J. Ceram. Soc. Jpn. 2021, 129, 73–78. [Google Scholar] [CrossRef]
- Fuentes, S.; Zárate, R.A.; Chávez, E.; Muñoz, P.; Ayala, M.; Espinoza-González, R.; Leytone, P. Synthesis and characterization of BaTiO3 nanoparticles in oxygen atmosphere. J. Alloys Compd. 2010, 505, 568–572. [Google Scholar] [CrossRef]
- Kyobe, J.W.; Mubofu, E.B.; Makame, Y.M.M.; Mlowe, S.; Revaprasadu, N. CdSe quantum dots capped with naturally occurring biobased oils. New J. Chem. 2015, 39, 7251. [Google Scholar] [CrossRef]
- Song, Z.; Wang, L.; Hou, S. A study of the chemiluminescence behavior of myoglobin with luminol and its analytical applications. Anal. Bioanal. Chem. 2004, 378, 529–535. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, W.; Xing, Y.; Yang, X.; Wang, K.; Jiang, R.; Wang, P.; Zhao, Q. Screening of DNA Aptamers against Myoglobin Using a Positive and Negative Selection Units Integrated Microfluidic Chip and Its Biosensing Application. Anal. Chem. 2014, 86, 6572–6579. [Google Scholar] [CrossRef]
- Pur, M.R.K.; Hosseini, M.; Faridbod, F.; Ganjali, M.R. Highly sensitive label-free electrochemiluminescence aptasensor for early detection of myoglobin, a biomarker for myocardial infarction. Microchim. Acta 2017, 184, 3529–3537. [Google Scholar] [CrossRef]
- Kumar, V.; Shorie, M.; Ganguli, A.K.; Sabherwal, P. Graphene-CNT nanohybrid aptasensor for label free detection of cardiac biomarker myoglobin. Biosens. Bioelectron. 2015, 72, 56–60. [Google Scholar] [CrossRef]
- Mandal, S.S.; Narayan, K.K.; Bhattacharyya, A.J. Employing denaturation for rapid electrochemical detection of myoglobin using TiO2 nanotubes. J. Mater. Chem. B 2013, 1, 3051–3056. [Google Scholar] [CrossRef]
- Taghdisi, S.M.; Danesh, N.M.; Ramezani, M.; Emranid, A.S.; Abnous, K. A novel electrochemical aptasensor based on Y-shape structure of dual-aptamer-complementary strand conjugate for ultrasensitive detection of myoglobin. Biosens. Bioelectron. 2016, 80, 532–537. [Google Scholar] [CrossRef]
- Pakapongpan, S.; Palangsuntikul, R.; Surareungchai, W. Electrochemical sensors for hemoglobin and myoglobin detection based on methylene blue-multiwalled carbon nanotubes nanohybrid-modified glassy carbon electrode. Electrochim. Acta 2011, 56, 6831–6836. [Google Scholar] [CrossRef]
- Kang, M.; Li, Z.; Hu, M.; Oderinde, O.; Hu, B.; He, L.; Wang, M.; Fu, G.; Zhang, Z.; Du, M. Bimetallic MnCo oxide nanohybrids prepared from Prussian blue analogue for application as impedimetric aptasensor carrier to detect myoglobin. Chem. Eng. J. 2020, 395, 125117. [Google Scholar] [CrossRef]
- Karami, P.; Bagheri, H.; Johari-Ahar, M.; Khoshsafar, H.; Arduini, F.; Afkhami, A. Dual-modality impedimetric immunosensor for early detection of prostatespecific antigen and myoglobin markers based on antibody-molecularly imprinted polymer. Talanta 2019, 202, 111–122. [Google Scholar] [CrossRef]
- Tuteja, S.K.; Chen, R.; Kukkar, M.; Song, C.K.; Mutreja, R.; Singh, S.; Paul, A.K.; Lee, H.; Kim, K.-H.; Deep, A. A label-free electrochemical immunosensor for the detection of cardiac marker using graphene quantum dots (GQDs). Biosens. Bioelectron. 2016, 86, 548–556. [Google Scholar] [CrossRef]
- Suprun, E.V.; Arduini, F.; Moscone, D.; Palleschi, G.; Shumyantseva, V.V.; Archakov, A.I. Direct Electrochemistry of Heme Proteins on Electrodes Modified with Didodecyldimethyl Ammonium Bromide and Carbon Black. Electroanalysis 2012, 24, 1923–1931. [Google Scholar] [CrossRef]
- Adeel, M.; Rahman, M.M.; Lee, J.-J. Label-free aptasensor for the detection of cardiac biomarker myoglobin based on gold nanoparticles decorated boron nitride nanosheets. Biosens. Bioelectron. 2019, 126, 143–150. [Google Scholar] [CrossRef]
- Ma, Y.; Dong, Y.-X.; Wang, B.; Ren, S.-W.; Cao, J.-T.; Liu, Y.-M. CdS:Mn-sensitized 2D/2D heterostructured g-C3N4-MoS2 with excellent photoelectrochemical performance for ultrasensitive immunosensing platform. Talanta 2020, 207, 120288. [Google Scholar] [CrossRef]
- Piloto, A.M.; Ribeiro, D.S.M.; Rodrigues, S.S.M.; Santos, C.; Santos, J.L.M.; Sales, M.G.F. Plastic antibodies tailored on quantum dots for an optical detection of myoglobin down to the femtomolar range. Sci. Rep. 2018, 8, 4944. [Google Scholar] [CrossRef]
- Deng, Y.; Wen, Z.; Cheng, H.; Yan, L.; Shao, B.; Li, G.; Zhang, X.; Sun, W. SnO2 Quantum Dots Functionalized 3D Graphene Composite for Enhanced Performance of Electrochemical Myoglobin Biosensor. SnO2 Quantum Dots Functionalized 3D Graphene Composite for Enhanced Performance of Electrochemical Myoglobin Biosensor. Int. J. Electrochem. Sci. 2020, 15, 10412–10422. [Google Scholar] [CrossRef]
- Song, T.; Wang, Q.; Yu, H.; Gao, W.; Xu, Y.; Lv, Y.; Xing, Y.; Chen, Y.; Yang, M. One-step hydrothermal synthesis of WS2 quantum dots as fluorescent sensor for sensitive and selective recognition of hemoglobin and cardiac biomarker myoglobin. Anal. Bioanal. Chem. 2022, 414, 1623–1630. [Google Scholar] [CrossRef]
- Himiyama, T.; Onoda, A.; Hayashi, T. Photochemical Property of a Myoglobin CdTe Quantum Dot Conjugate Formed by Supramolecular HostGuest Interactions. Chem. Lett. 2014, 43, 1152–1154. [Google Scholar] [CrossRef]
- Fatease, A.A.; Haque, M.; Umar, A.; Ansari, S.G.; Alhamhoom, Y.; Muhsinah, A.B.; Mahnashi, M.H.; Guo, W.; Ansari, Z.A. Label-Free Electrochemical Sensor Based on Manganese Doped Titanium Dioxide Nanoparticles for Myoglobin Detection: Biomarker for Acute Myocardial Infarction. Molecules 2021, 26, 4252. [Google Scholar] [CrossRef]
- Magalhães, R.S.; Wagner, D.M., Jr.; Souza, A.E.; Teixeira, S.R.; Li, M.S.; Longo, E. Synthesis of BaTiO3 and SrTiO3 by microwave assisted hidrotermal method (mah) using anatase as titanium precursor. Quím. Nova 2017, 40, 166–170. [Google Scholar] [CrossRef]
- Liu, L.; Qiu, C.L.; Chen, Q.; Zhang, S.M. Corrosion behavior of Zr-based bulk metallic glasses in different artificial body fluids. J. Alloys Compd. 2006, 425, 268–273. [Google Scholar] [CrossRef]
- Laube, N.; Mohr, B.; Hesse, A. Laser-probe-based investigation of the evolution of particle size distributions of calcium oxalate particles formed in artificial urines. J. Cryst. Growth 2001, 233, 367–374. [Google Scholar] [CrossRef]


| Sample * | Spiked (ng mL−1) | Found (ng mL−1) | Recovery (%) |
|---|---|---|---|
| A1 | 10 | 10.14 (±0.02) | 101.4 |
| A2 | 50 | 49.66 (±0.01) | 99.3 |
| A3 | 100 | 99.31 (±0.03) | 99.3 |
| B1 | 10 | 10.54 (±0.03) | 105.4 |
| B2 | 50 | 55.34 (±0.01) | 110.7 |
| B3 | 100 | 95.72 (±0.04) | 95.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, F.M.R.; de Menezes, A.S.; Maciel, A.P.; Sinfrônio, F.S.M.; Kubota, L.T.; Damos, F.S.; Luz, R.C.S. Zero-Biased Photoelectrochemical Detection of Cardiac Biomarker Myoglobin Based on CdSeS/ZnS Quantum Dots and Barium Titanate Perovskite. Molecules 2022, 27, 4778. https://doi.org/10.3390/molecules27154778
Lima FMR, de Menezes AS, Maciel AP, Sinfrônio FSM, Kubota LT, Damos FS, Luz RCS. Zero-Biased Photoelectrochemical Detection of Cardiac Biomarker Myoglobin Based on CdSeS/ZnS Quantum Dots and Barium Titanate Perovskite. Molecules. 2022; 27(15):4778. https://doi.org/10.3390/molecules27154778
Chicago/Turabian StyleLima, Fernanda M. R., Alan S. de Menezes, Adeilton P. Maciel, Francisco S. M. Sinfrônio, Lauro T. Kubota, Flávio S. Damos, and Rita C. S. Luz. 2022. "Zero-Biased Photoelectrochemical Detection of Cardiac Biomarker Myoglobin Based on CdSeS/ZnS Quantum Dots and Barium Titanate Perovskite" Molecules 27, no. 15: 4778. https://doi.org/10.3390/molecules27154778
APA StyleLima, F. M. R., de Menezes, A. S., Maciel, A. P., Sinfrônio, F. S. M., Kubota, L. T., Damos, F. S., & Luz, R. C. S. (2022). Zero-Biased Photoelectrochemical Detection of Cardiac Biomarker Myoglobin Based on CdSeS/ZnS Quantum Dots and Barium Titanate Perovskite. Molecules, 27(15), 4778. https://doi.org/10.3390/molecules27154778








