Fluorescent Nitrogen-Doped Carbon Dots for Label Live Elder Blood-Stage Plasmodium falciparum through New Permeability Pathways
Abstract
:1. Introduction
2. Results and Discussion
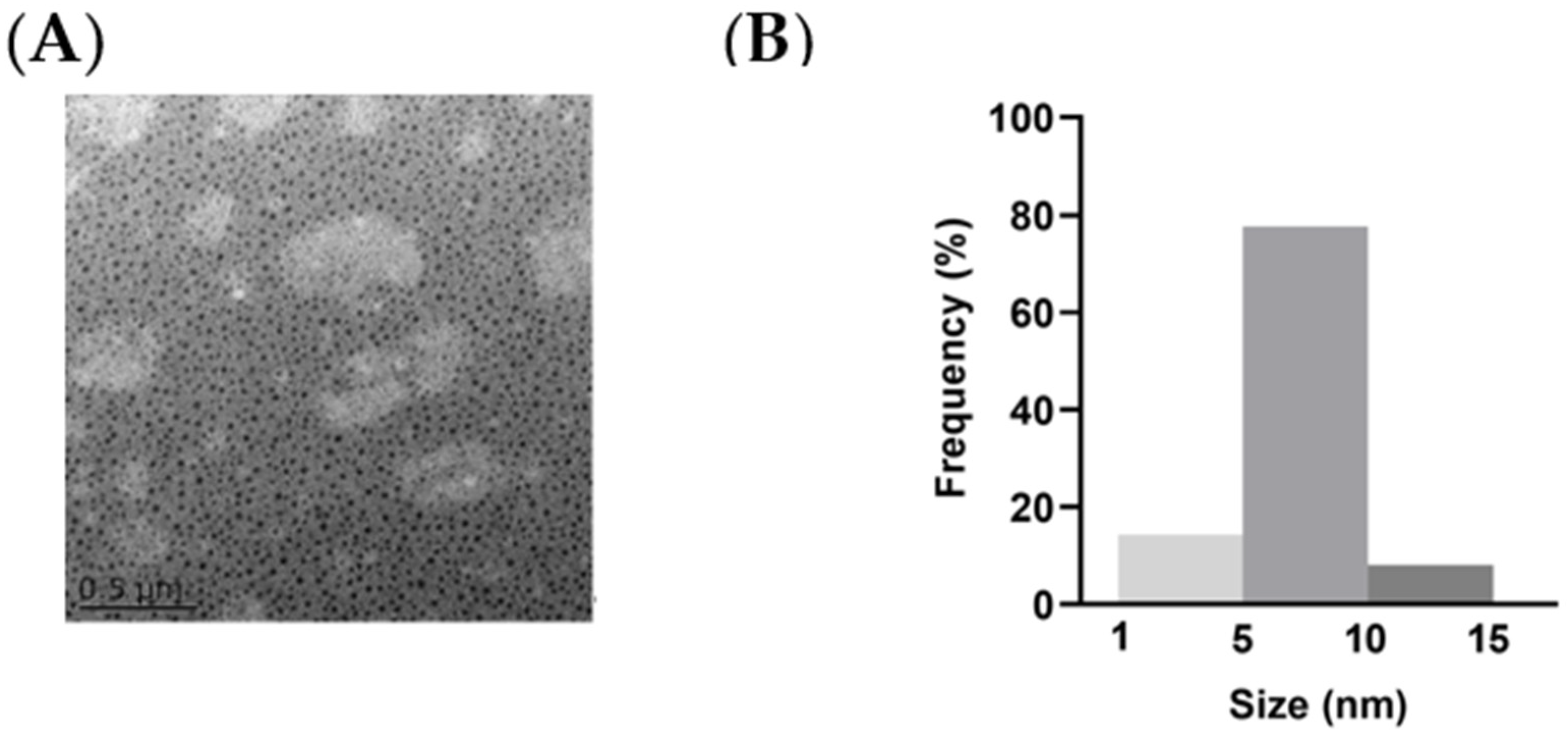
2.1. Synthesis and Purification of NCDs
2.2. Detection of P. falciparum-Infected Erythrocytes by Flow Cytometry with TO and HE
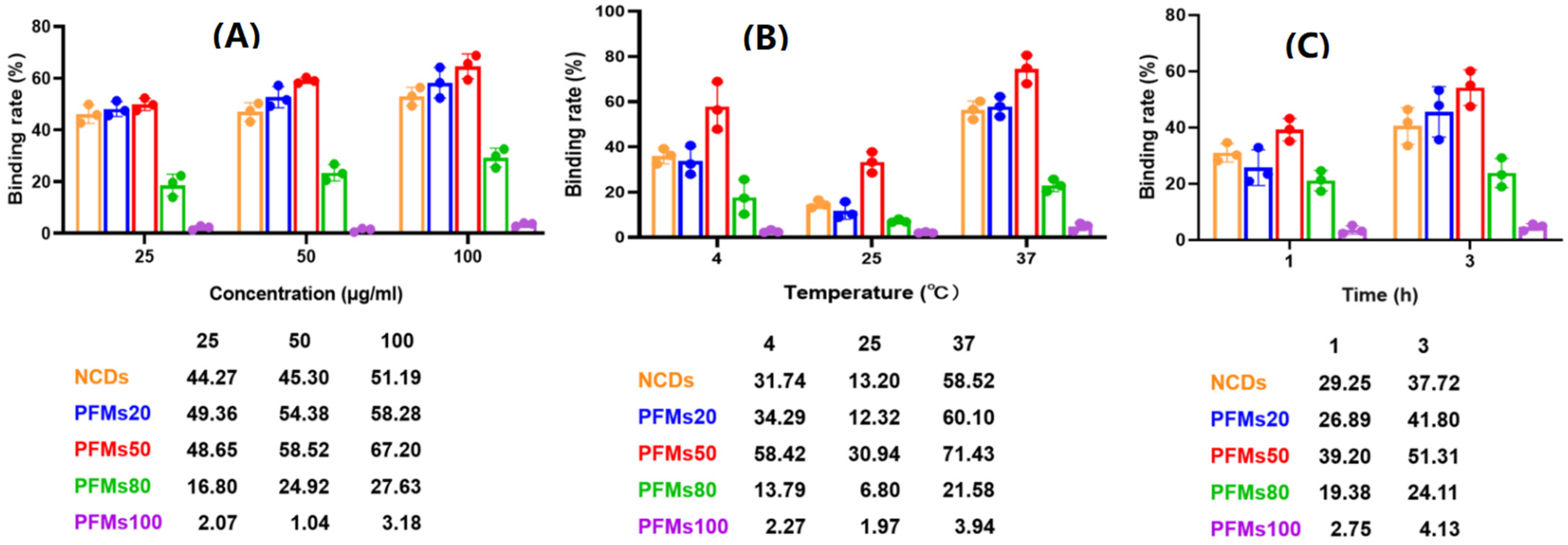
2.3. Detection of P. falciparum-Infected Erythrocytes by Flow Cytometry with Nanomaterials
2.4. NCDs Show No Toxicity to P. falciparum-Infected Erythrocytes
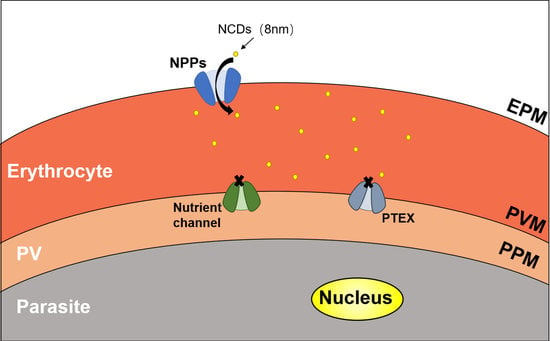
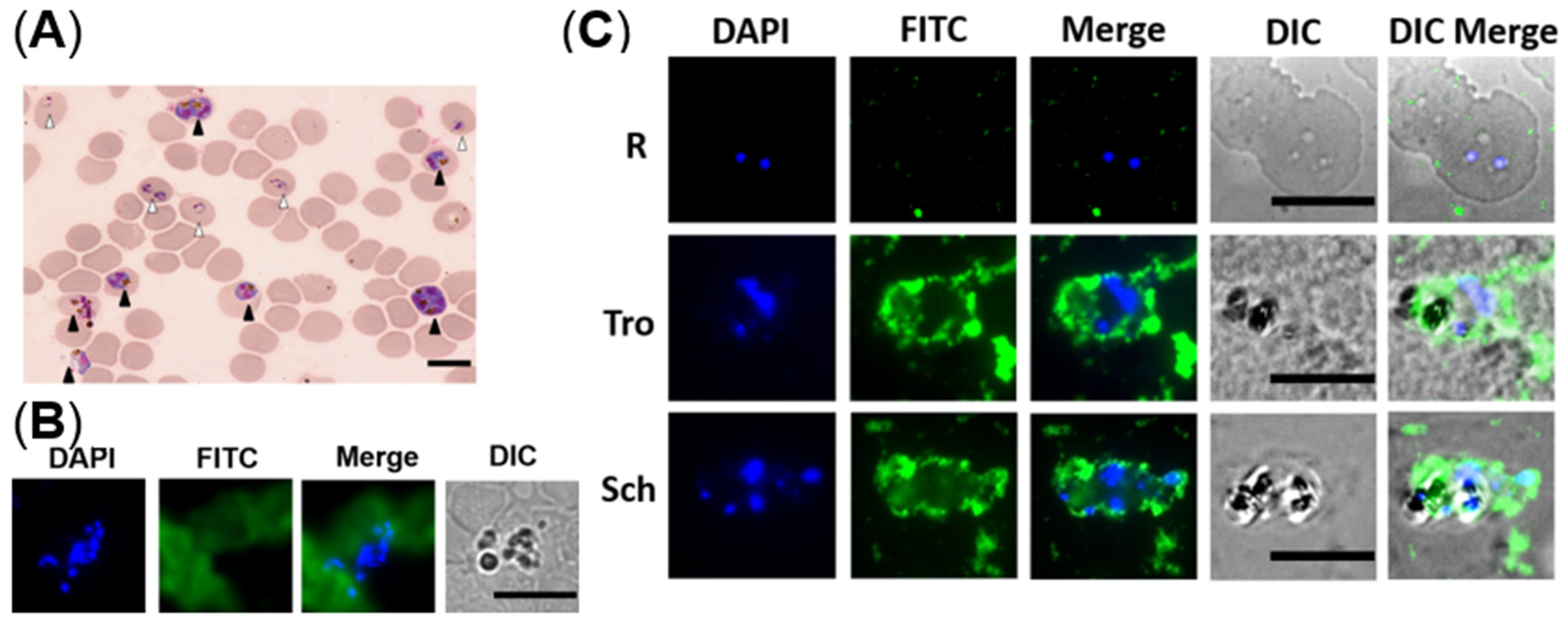
2.5. Determination the Distribution of NCDs in P. falciparum-Infected Erythrocytes
3. Materials and Methods
3.1. Sources of Nanomaterials
3.2. Parasite Cultivation and Synchronization
3.3. Flow Cytometry Analysis
3.4. SYBR Green I-Based Drug Sensitivity Assay
3.5. Fluorescence Microscopic Imaging
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- WHO. World Malaria Report 2021. Available online: https://www.who.int/publications/i/item/9789240040496 (accessed on 6 December 2021).
- Caraballo, H.; King, K. Emergency department management of mosquito-borne illness: Malaria, dengue, and West Nile virus. Emerg. Med. Pract. 2014, 16, 1–23. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention (CDC). Travel-associated Dengue surveillance—United States, 2006–2008. MMWR Morb. Mortal. Wkly. Rep. 2010, 59, 715–719. [Google Scholar]
- Ginsburg, H.; Stein, W.D. New permeability pathways induced by the malarial parasite in the membrane of its host erythrocyte Potential routes for targeting of drugs into infected cells. Biosci. Rep. 1987, 7, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Merckx, A.; Bouyer, G.; Thomas, S.L.; Langsley, G.; Egee, S. Anion channels in Plasmodium-falciparum-infected erythrocytes and protein kinase A. Trends Parasitol. 2009, 25, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Counihan, N.A.; Modak, J.K.; de Koning-Ward, T.F. How Malaria Parasites Acquire Nutrients From Their Host. Front. Cell Dev. Biol. 2021, 9, 649184. [Google Scholar] [CrossRef] [PubMed]
- Mesén-Ramírez, P.; Bergmann, B.; Elhabiri, M.; Zhu, L.; von Thien, H.; Castro-Peña, C.; Gilberger, T.W.; Davioud-Charvet, E.; Bozdech, Z.; Bachmann, A.; et al. The parasitophorous vacuole nutrient channel is critical for drug access in malaria parasites and modulates the artemisinin resistance fitness cost. Cell Host Microbe 2021, 29, 1774–1787.e9. [Google Scholar] [CrossRef]
- Lisk, G.; Desai, S.A. The plasmodial surface anion channel is functionally conserved in divergent malaria parasites. Eukaryot. Cell 2005, 4, 2153–2159. [Google Scholar] [CrossRef] [Green Version]
- Goodyer, I.D.; Pouvelle, B.; Schneider, T.G.; Trelka, D.P.; Taraschi, T.F. Characterization of macromolecular transport pathways in malaria-infected erythrocytes. Mol. Biochem. Parasitol. 1997, 87, 13–28. [Google Scholar] [CrossRef]
- Krugliak, M.; Ginsburg, H. The evolution of the new permeability pathways in Plasmodium falciparum--infected erythrocytes--a kinetic analysis. Exp. Parasitol. 2006, 114, 253–258. [Google Scholar] [CrossRef]
- Barman, M.K.; Patra, A. Current status and prospects on chemical structure driven photoluminescence behaviour of carbon dots. J. Photochem. Photobiol. C Photochem. Rev. 2018, 37, 1–22. [Google Scholar] [CrossRef]
- Wang, J.; Guo, Y.; Geng, X.; Hu, J.; Yan, M.; Sun, Y.; Zhang, K.; Qu, L.; Li, Z. Quantitative Structure-Activity Relationship Enables the Rational Design of Lipid Droplet-Targeting Carbon Dots for Visualizing Bisphenol A-Induced Nonalcoholic Fatty Liver Disease-like Changes. ACS Appl. Mater. Interfaces 2021, 13, 44086–44095. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Zhang, X.; Fu, K.; Zhang, X.; Shang, L.; Su, Z. Highly fluorescent carbon dots as novel theranostic agents for biomedical applications. Nanoscale 2021, 13, 17236–17253. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Ghosh, S.; Jana, N.R. Fluorescent carbon dots as intracellular imaging probes. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1617. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Kim, Y.; Chang, H.; Won, S.; Kim, H.; Kwon, W. Biocompatible nitrogen-doped carbon dots: Synthesis, characterization, and application. J. Mater. Chem. B 2020, 8, 8935–8951. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, B.; Xu, F.; Shi, X.; Feng, D.; Wei, D.; Li, Y.; Feng, Y.; Wang, Y.; Jia, D.; et al. High-yield synthesis of strong photoluminescent N-doped carbon nanodots derived from hydrosoluble chitosan for mercury ion sensing via smartphone APP. Biosens. Bioelectron. 2016, 79, 1–8. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Shi, Y.; Sun, C.; Zhou, N.; Wen, H. The Synthesis and Functional Study of Multicolor Nitrogen-Doped Carbon Dots for Live Cell Nuclear Imaging. Molecules 2020, 25, 306. [Google Scholar] [CrossRef] [Green Version]
- Xie, S.C.; Dogovski, C.; Kenny, S.; Tilley, L.; Klonis, N. Optimal assay design for determining the in vitro sensitivity of ring stage Plasmodium falciparum to artemisinins. Int. J. Parasitol. 2014, 44, 893–899. [Google Scholar] [CrossRef]
- Ullah, I.; Sharma, R.; Biagini, G.A.; Horrocks, P. A validated bioluminescence-based assay for the rapid determination of the initial rate of kill for discovery antimalarials. J. Antimicrob. Chemother. 2017, 72, 717–726. [Google Scholar] [CrossRef]
- Kulkeaw, K. Progress and challenges in the use of fluorescence-based flow cytometric assays for anti-malarial drug susceptibility tests. Malar. J. 2021, 20, 57. [Google Scholar] [CrossRef]
- Hasenkamp, S.; Sidaway, A.; Devine, O.; Roye, R.; Horrocks, P. Evaluation of bioluminescence-based assays of anti-malarial drug activity. Malar J 2013, 12, 58. [Google Scholar] [CrossRef] [Green Version]
- Davis, M.I.; Patrick, S.L.; Blanding, W.M.; Dwivedi, V.; Suryadi, J.; Golden, J.E.; Coussens, N.P.; Lee, O.W.; Shen, M.; Boxer, M.B.; et al. Identification of Novel Plasmodium falciparum Hexokinase Inhibitors with Antiparasitic Activity. Antimicrob. Agents Chemother. 2016, 60, 6023–6033. [Google Scholar] [CrossRef] [Green Version]
- Azevedo, M.F.; Nie, C.Q.; Elsworth, B.; Charnaud, S.C.; Sanders, P.R.; Crabb, B.S.; Gilson, P.R. Plasmodium falciparum transfected with ultra bright NanoLuc luciferase offers high sensitivity detection for the screening of growth and cellular trafficking inhibitors. PLoS ONE 2014, 9, e112571. [Google Scholar] [CrossRef]
- Grimberg, B.T. Methodology and application of flow cytometry for investigation of human malaria parasites. J. Immunol. Methods 2011, 367, 1–16. [Google Scholar] [CrossRef] [Green Version]
- van der Heyde, H.C.; Elloso, M.M.; vande Waa, J.; Schell, K.; Weidanz, W.P. Use of hydroethidine and flow cytometry to assess the effects of leukocytes on the malarial parasite Plasmodium falciparum. Clin. Diagn. Lab. Immunol. 1995, 2, 417–425. [Google Scholar] [CrossRef] [Green Version]
- Kulkeaw, K.; Ketprasit, N.; Tungtrongchitr, A.; Palasuwan, D. A simple monochromatic flow cytometric assay for assessment of intraerythrocytic development of Plasmodium falciparum. Malar. J. 2020, 19, 74. [Google Scholar] [CrossRef] [Green Version]
- Engelbrecht, D.; Coetzer, T.L. The walking dead: Is hydroethidine a suitable viability dye for intra-erythrocytic Plasmodium falciparum? Parasitol. Int. 2012, 61, 731–734. [Google Scholar] [CrossRef]
- Santos-Magalhaes, N.S.; Mosqueira, V.C. Nanotechnology applied to the treatment of malaria. Adv. Drug Deliv. Rev. 2010, 62, 560–575. [Google Scholar] [CrossRef] [Green Version]
- Biagini, G.A.; Ward, S.A.; Bray, P.G. Malaria parasite transporters as a drug-delivery strategy. Trends Parasitol. 2005, 21, 299–301. [Google Scholar] [CrossRef]
- Baumeister, S.; Winterberg, M.; Przyborski, J.M.; Lingelbach, K. The malaria parasite Plasmodium falciparum: Cell biological peculiarities and nutritional consequences. Protoplasma 2010, 240, 3–12. [Google Scholar] [CrossRef]
- Neves Borgheti-Cardoso, L.; San Anselmo, M.; Lantero, E.; Lancelot, A.; Serrano, J.L.; Hernandez-Ainsa, S.; Fernandez-Busquets, X.; Sierra, T. Promising nanomaterials in the fight against malaria. J. Mater. Chem. B 2020, 8, 9428–9448. [Google Scholar] [CrossRef]
- Naik, V.M.; Bhosale, S.V.; Kolekar, G.B. A brief review on the synthesis, characterisation and analytical applications of nitrogen doped carbon dots. Anal. Methods 2022, 14, 877–891. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; Ma, J.; Shan, X.; Feng, H.; Shao, L.; Chen, J. Highly luminescent N-doped carbon quantum dots as an effective multifunctional fluorescence sensing platform. Chemistry 2014, 20, 2254–2263. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, Y.; Lu, S.; Liu, L.; Yang, L.; Guo, Y.; Yu, S.; Yang, H. Labeled-protein corona-coated Bi2S3 nanorods targeted to lysosomes for bioimaging and efficient photothermal cancer therapy. Colloids Surf. B Biointerfaces 2020, 196, 111291. [Google Scholar] [CrossRef]
- Trager, W.; Jensen, J.B. Human malaria parasites in continuous culture. Science 1976, 193, 673–675. [Google Scholar] [CrossRef]
- Klaus, S.; Binder, P.; Kim, J.; Machado, M.; Funaya, C.; Schaaf, V.; Klaschka, D.; Kudulyte, A.; Cyrklaff, M.; Laketa, V.; et al. Asynchronous nuclear cycles in multinucleated Plasmodium falciparum facilitate rapid proliferation. Sci. Adv. 2022, 8, eabj5362. [Google Scholar] [CrossRef]
- Lambros, C.; Vanderberg, J.P. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J. Parasitol. 1979, 65, 418–420. [Google Scholar] [CrossRef]
- Ngernna, S.; Chim-Ong, A.; Roobsoong, W.; Sattabongkot, J.; Cui, L.; Nguitragool, W. Efficient synchronization of Plasmodium knowlesi in vitro cultures using guanidine hydrochloride. Malar. J. 2019, 18, 148. [Google Scholar] [CrossRef] [Green Version]
- Quashie, N.B.; Duah, N.O.; Abuaku, B.; Quaye, L.; Ayanful-Torgby, R.; Akwoviah, G.A.; Kweku, M.; Johnson, J.D.; Lucchi, N.W.; Udhayakumar, V.; et al. A SYBR Green 1-based in vitro test of susceptibility of Ghanaian Plasmodium falciparum clinical isolates to a panel of anti-malarial drugs. Malar. J. 2013, 12, 450. [Google Scholar] [CrossRef] [Green Version]
- Maier, A.G.; Rug, M. In vitro culturing Plasmodium falciparum erythrocytic stages. Methods Mol. Biol. 2013, 923, 3–15. [Google Scholar] [CrossRef]
- Im, K.; Mareninov, S.; Diaz, M.F.P.; Yong, W.H. An Introduction to Performing Immunofluorescence Staining. Methods Mol. Biol. 2019, 1897, 299–311. [Google Scholar] [CrossRef]
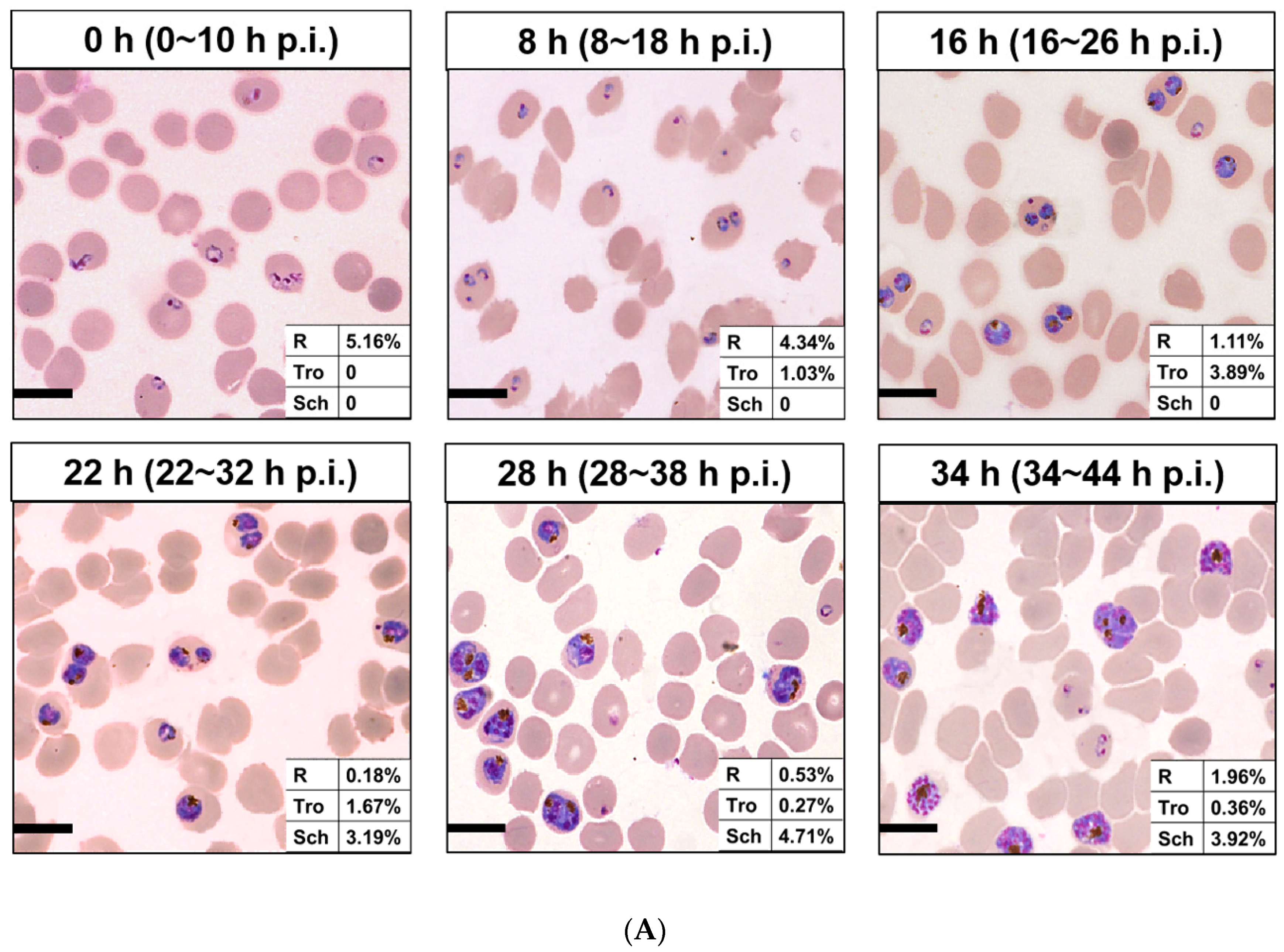
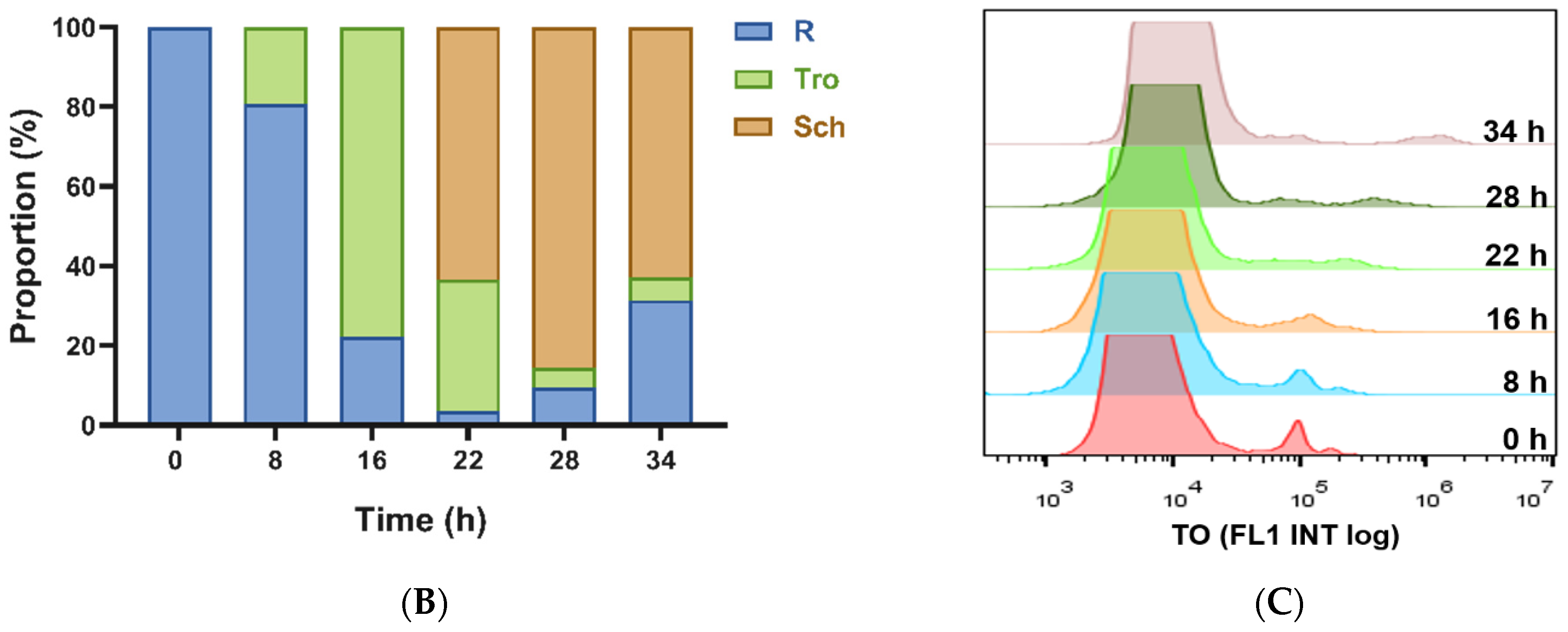
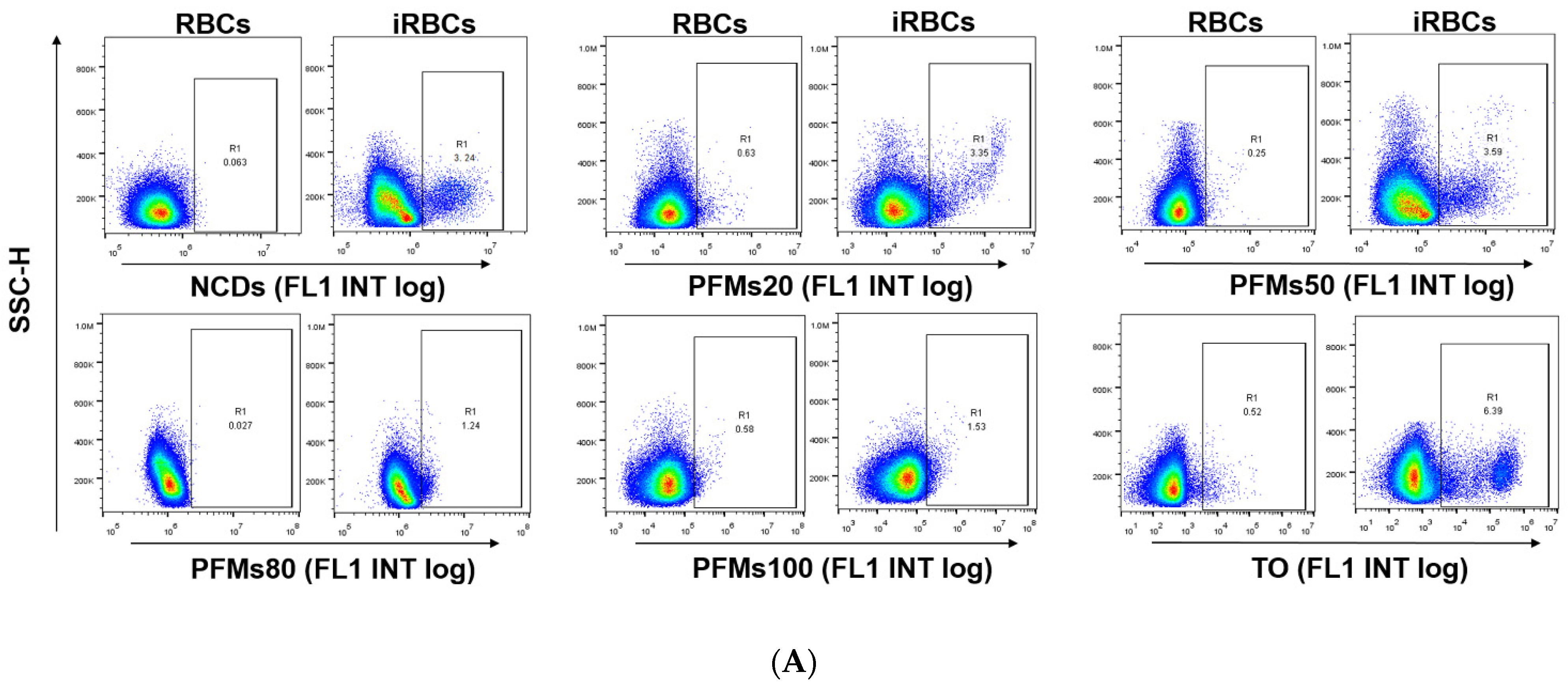
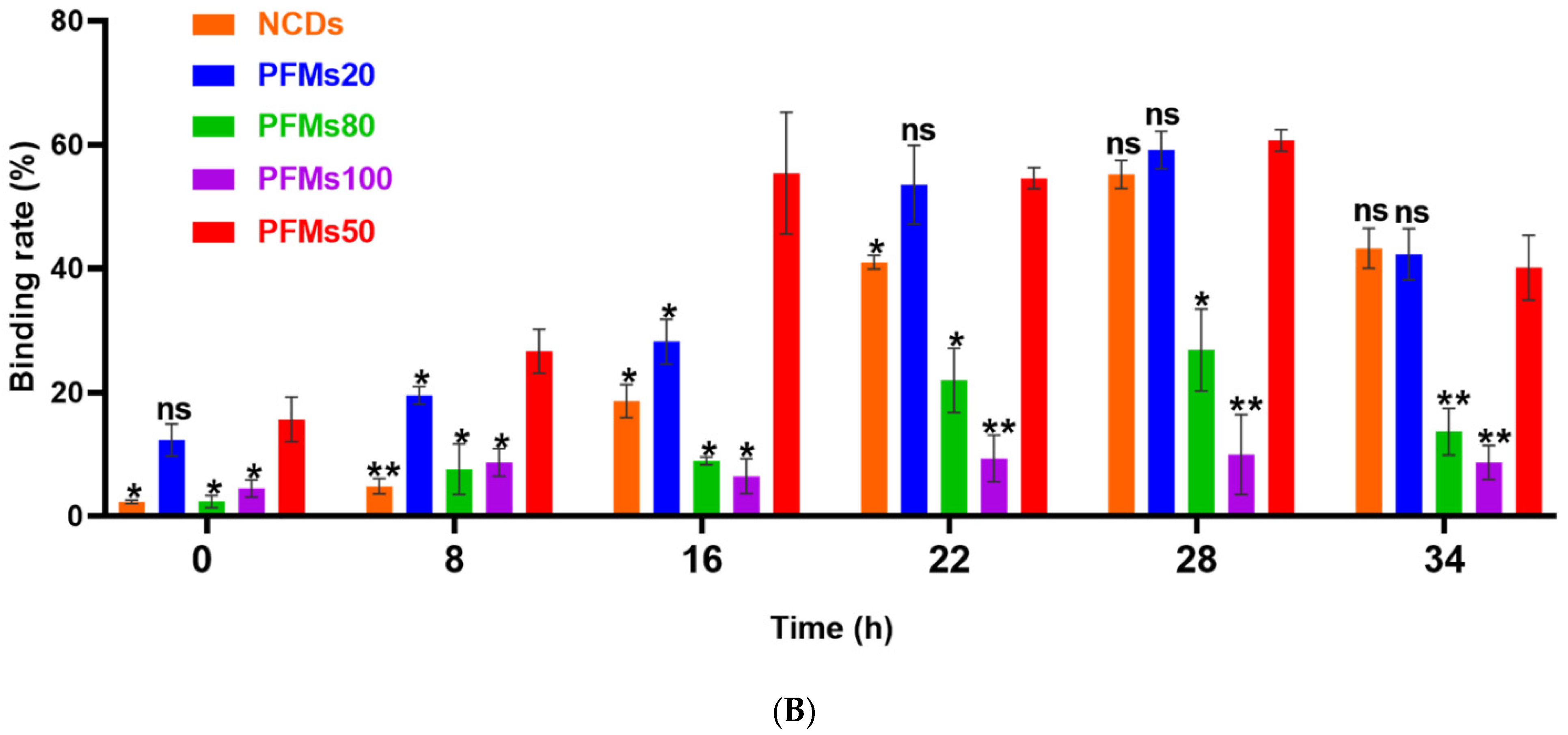
| 0 h | 8 h | 16 h | 22 h | 28 h | 34 h | |
|---|---|---|---|---|---|---|
| NCDs | 2.30 ± 0.00 | 4.84 ± 0.01 | 18.60 ± 0.03 | 41.02 ± 0.01 | 55.18 ± 0.02 | 43.26 ± 0.03 |
| PFMs20 | 12.32 ± 0.04 | 19.53 ± 0.04 | 28.21 ± 0.10 | 53.54 ± 0.02 | 59.17 ± 0.02 | 42.13 ± 0.05 |
| PFMs50 | 15.63 ± 0.01 | 26.64 ± 0.04 | 55.37 ± 0.01 | 54.60 ± 0.05 | 60.08 ± 0.07 | 40.13 ± 0.04 |
| PFMs80 | 2.38 ± 0.01 | 7.61 ± 0.02 | 8.93 ± 0.03 | 21.92 ± 0.04 | 26.83 ± 0.06 | 13.65 ± 0.03 |
| PFMs100 | 4.49 ± 0.04 | 8.67 ± 0.03 | 6.48 ± 0.02 | 9.33 ± 0.04 | 9.95 ± 0.08 | 8.68 ± 0.04 |
| TO | 94.25 ± 0.03 | 97.34 ± 0.01 | 95.13 ± 0.04 | 98.21 ± 0.06 | 96.34 ± 0.03 | 95.35 ± 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, J.; Hu, F.; Li, S.; Bao, J.; Yin, Y.; Ren, Z.; Deng, Y.; Tian, F.; Bao, G.; Liu, J.; et al. Fluorescent Nitrogen-Doped Carbon Dots for Label Live Elder Blood-Stage Plasmodium falciparum through New Permeability Pathways. Molecules 2022, 27, 4163. https://doi.org/10.3390/molecules27134163
Xu J, Hu F, Li S, Bao J, Yin Y, Ren Z, Deng Y, Tian F, Bao G, Liu J, et al. Fluorescent Nitrogen-Doped Carbon Dots for Label Live Elder Blood-Stage Plasmodium falciparum through New Permeability Pathways. Molecules. 2022; 27(13):4163. https://doi.org/10.3390/molecules27134163
Chicago/Turabian StyleXu, Jiahui, Fengyue Hu, Shuang Li, Jiaojiao Bao, Yi Yin, Zhenyu Ren, Ying Deng, Fang Tian, Guangyu Bao, Jian Liu, and et al. 2022. "Fluorescent Nitrogen-Doped Carbon Dots for Label Live Elder Blood-Stage Plasmodium falciparum through New Permeability Pathways" Molecules 27, no. 13: 4163. https://doi.org/10.3390/molecules27134163
APA StyleXu, J., Hu, F., Li, S., Bao, J., Yin, Y., Ren, Z., Deng, Y., Tian, F., Bao, G., Liu, J., Li, Y., He, X., Xi, J., & Lu, F. (2022). Fluorescent Nitrogen-Doped Carbon Dots for Label Live Elder Blood-Stage Plasmodium falciparum through New Permeability Pathways. Molecules, 27(13), 4163. https://doi.org/10.3390/molecules27134163