Microalgae Bioactive Compounds to Topical Applications Products—A Review
Abstract
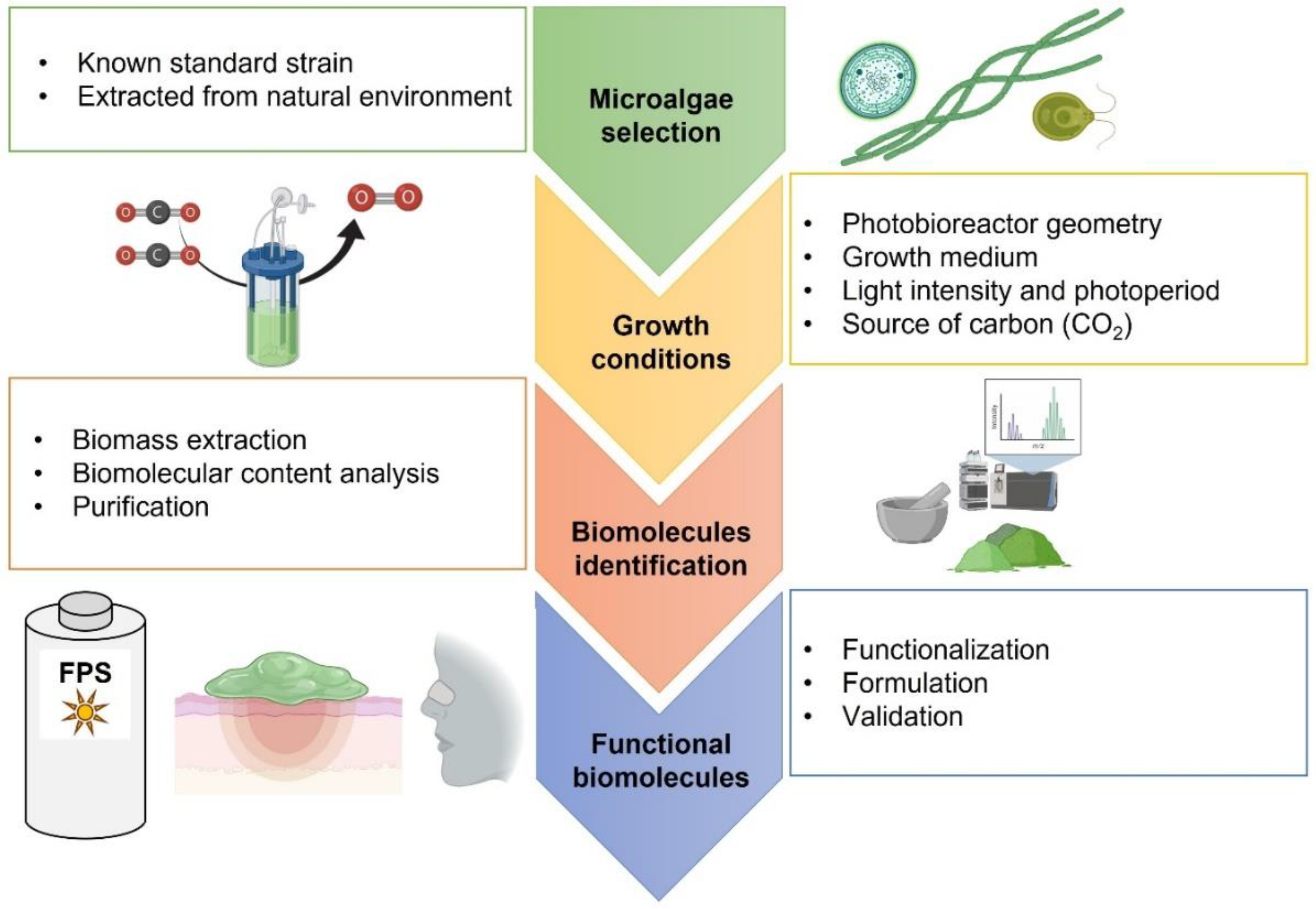
1. Introduction
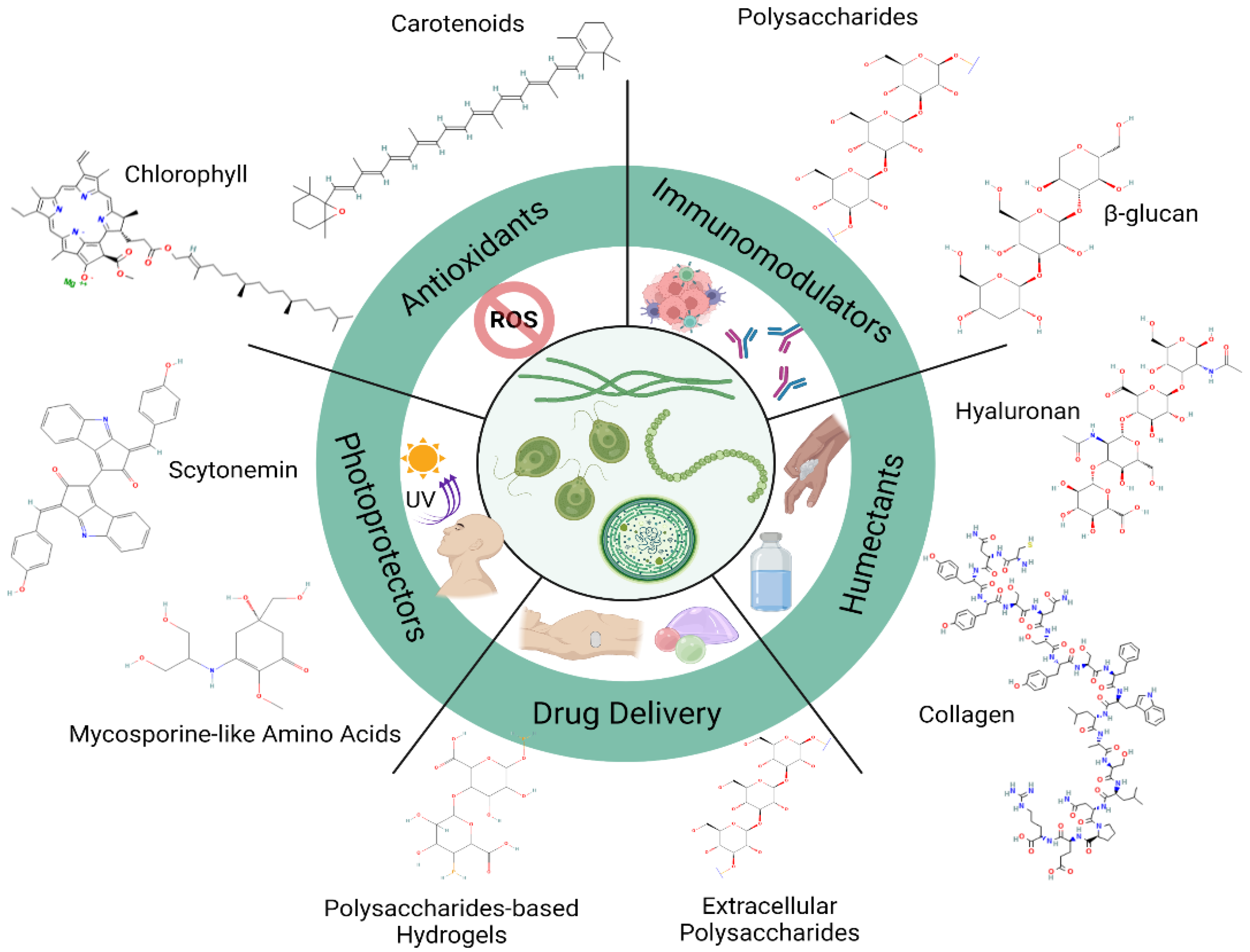
2. Immunomodulatory Activity of Compounds from Microalgae
3. Antioxidants
| Type of Compound | Mechanism of Action | Microalgae Species | Culture Conditions | Extraction | Reference |
|---|---|---|---|---|---|
| Amino acids | ROS attenuation | Chlamydomonas hedleyi (Chlorophyta) | F/2 medium, 22 ± 1 °C, 80 μmol·m−2·s−1 light, 16:8 D:L cycle rotary shaker + bubbled with CO2-enriched (1%) air. | Digestion of dry biomass with aqueous methanol (20% v/v) at 45 °C. | [90] |
| Polysaccharides | Attenuation of free radicals, hydroxyl radicals, and ROS | Arthrospira platensis (Cyanobacteria) | Zarrouk medium, 32 ± 1 °C, 100 μmol·m−2·s−1 light, stirred with CO2-enriched (1%) air. | Tangential ultrafiltration cell—30 Kda membrane | [91] |
| Free radical scavenging | Odontella aurita (Bacillariophyta) | Modified L1 medium (6–18 mM), 25 ± 2 °C, 100 and 300 μmol·m−2·s−1 light, sparging with air + 1% CO2. | Digestion of dry biomass at 60 °C withsulfuric acid | [92] | |
| Sulfated polysaccharides | ROS attenuation | Porphyridium sp. (Rhodophyta) | Seawater medium, 24 ± 3 °C, 150 μmol·m−2·s−1 light, bubbled with air + 1–3% CO2. | Culture centrifugation (17,000× g, 20 min) and supernatant filtrate in dialysis tube (⌀ 2.3 cm—MW cutoff 8000) | [93] |
| Extracellular polysaccharides | Free radical scavenging | Rhodella reticulata (Rhodophyta) | Kock medium, 25 °C, 92 μmol·m−2·s−1 light. | Ethanol extraction | [94] |
| Pigments, peptides and vitamins | ROS attenuation | Skeletonema marinoi (Ochrophyta) | F/2 enriched medium, 20 °C, 150 μmol·m−2·s−1 light, 12:12 D:L cycle. | Mechanical grounding with absolute methanol | [95] |
| Pigments | Free radical scavenging | Dunaliella salina (Chlorophyta) | Johnson medium with artificial seawater (35 g L−1), pH 7.5, 100 μmol·m−2·s−1 light, 12:12 D:L cycle, bubbled with air 2 L min−1. | Sonication with methanol and filtration Fluoropore PTFE 0.2-μm | [96] |
| ROS attenuation | Chlorella vulgaris (Chlorophyta) | BG-11 medium (without and with nitrogen starvation + NaCl addition 30%), 25 °C, 150 and 1000 μmol·m−2·s−1 light, bubbled with air. | Homogenization with acetone and supernatant filtration with Na2SO4 | [97] | |
| Free radical scavenging | Anabaena vaginicola (Cyanobacteria) | BG-11 medium without nitrates, 400 μmol·m−2·s−1 light, 25 ± 2 °C, 12:12 D:L cycle | Freezing of biomass dry at −20 °C with methanol + ultrasonication | [98] |
4. Photoprotectors
5. Moisturizers, Regenerative and Other Activities
| Type of Compound | Mechanism of Action | Microalgae Species | Culture Conditions | Extraction | Reference |
|---|---|---|---|---|---|
| Collagen | Stimulation of collagen synthesis in the skin | C. vulgaris | NR | Methanol extraction | [134] |
| Regeneration by stimulation of collagen synthesis in the skin | S. platensis | Zarrouk’s medium, pH 9.8–10.0, 25 °C, 46 μmol m−2·s−1 light | Raw biomass | [135] | |
| Mycosporine-2-glycine | Collagenase inhibition and glycation products inhibition | Aphanothece halophytica | BG-11 | Methanol extraction and mechanical disruption | [136] |
| Polysaccharides | Moisturizing agents | Pediastrum duplex | MIII medium, pH 7.9 ± 0.1 20 ± 0.5°C, 80 µmol m−2·s−1 light, 12:12 D:L | Methanol extraction | [137] |
| Regulate water distribution in the skin | Codium tomentosum | NR | NR | [138] | |
| Moisturizing agents | Undaria pinnatifa | NR | NR | [138] | |
| Moisturizing agents | Durvillea antarctica | NR | NR | [138] | |
| Moisturizing agents | Cladosiphon okamuranus | NR | NR | [138] | |
| Fat acids | Dermal collagen content rescue | S. rubescens | NR | Hydrophilic hot extrusion | [139] |
| Amino acids | Increases expression of Procollagen C Proteinase | C. hedleyi | F/2, pH 8–9, 22 °C, 80 μmol m−2·s−1 light, 8:16 D:L cycle, 1% CO2 | Raw biomass | [91] |
| TrpA protein | Collagen-like protein | Trichodesmium erythraeum | NR | NR | [11] |
6. Novel Vehicles or Excipient Compounds
7. Perspectives
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mourelle, M.L.; Gómez, C.P.; Legido, J.L. The Potential Use of Marine Microalgae and Cyanobacteria in Cosmetics and Thalassotherapy. Cosmetics 2017, 4, 46. [Google Scholar] [CrossRef]
- Vermeer, B.J.; Gilchrest, B.A. Cosmeceuticals: A proposal for rational definition, evaluation, and regulation. Arch. Dermatol. 1996, 132, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Kligman, A. The future of cosmeceuticals: An interview with Albert Kligman, MD, PhD. Interview by Zoe Diana Draelos. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. 2005, 31, 890–891. [Google Scholar]
- Pandey, A.; Jatana, G.K.; Sonthalia, S. Cosmeceuticals; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- What is the Demand for Natural Ingredients for Cosmetics on the European Market?|CBI. Available online: https://www.cbi.eu/market-information/natural-ingredients-cosmetics/what-demand (accessed on 23 January 2022).
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Chakdar, H.; Jadhav, S.D.; Dhar, D.W.; Pabbi, S. Potential applications of blue green algae. J. Sci. Ind. Res. 2012, 71, 13–20. [Google Scholar]
- Stolz, P.; Obermayer, B. Manufacturing microalgae for skin care. Cosmet. Toilet. 2005, 120, 99–106. [Google Scholar]
- Gauthier, M.R.; Senhorinho, G.N.A.; Scott, J.A. Microalgae under environmental stress as a source of antioxidants. Algal Res. 2020, 52, 102104. [Google Scholar] [CrossRef]
- Yan, N.; Fan, C.; Chen, Y.; Hu, Z. The potential for microalgae as bioreactors to produce pharmaceuticals. Int. J. Mol. Sci. 2016, 17, 962. [Google Scholar] [CrossRef]
- Wu, J.; Gu, X.; Yang, D.; Xu, S.; Wang, S.; Chen, X.; Wang, Z. Bioactive substances and potentiality of marine microalgae. Food Sci. Nutr. 2021, 9, 5279–5292. [Google Scholar] [CrossRef]
- Rath, B. Microalgal Bioremediation:Current Practices and Perspectives. J. Biochem. Technol. 2011, 3, 299–304. [Google Scholar]
- Patel, A.; Matsakas, L.; Rova, U.; Christakopoulos, P. A perspective on biotechnological applications of thermophilic microalgae and cyanobacteria. Bioresour. Technol. 2019, 278, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Sartori, R.B.; Siqueira, S.F.; Maroneze, M.M.; Fagundes, M.B.; Wagner, R.; Zepka, L.Q.; Jacob-Lopes, E. Microalgal secondary metabolites: Effect of climatic variables, seasons, and photocycles on the biogeneration of volatile organic compounds (VOCs). J. Appl. Phycol. 2021, 33, 1457–1472. [Google Scholar] [CrossRef]
- Levasseur, W.; Perré, P.; Pozzobon, V. A review of high value-added molecules production by microalgae in light of the classification. Biotechnol. Adv. 2020, 41, 107545. [Google Scholar] [CrossRef]
- Al-Jabri, H.; Das, P.; Khan, S.; Thaher, M.; AbdulQuadir, M. Treatment of Wastewaters by Microalgae and the Potential Applications of the Produced Biomass—A Review. Water 2021, 13, 27. [Google Scholar] [CrossRef]
- Ummalyma, S.B.; Sahoo, D.; Pandey, A. Resource recovery through bioremediation of wastewaters and waste carbon by microalgae: A circular bioeconomy approach. Environ. Sci. Pollut. Res. 2021, 28, 58837–58856. [Google Scholar] [CrossRef] [PubMed]
- Khanzada, Z.T. Phosphorus removal from landfill leachate by microalgae. Biotechnol. Rep. 2020, 25, e00419. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Raouf, N.; Al-Homaidan, A.A.; Ibraheem, I.B.M. Microalgae and wastewater treatment. Saudi J. Biol. Sci. 2012, 19, 257–275. [Google Scholar] [CrossRef]
- Wang, L.; Min, M.; Li, Y.; Chen, P.; Chen, Y.; Liu, Y.; Wang, Y.; Ruan, R. Cultivation of Green Algae Chlorella sp. in Different Wastewaters from Municipal Wastewater Treatment Plant. Appl. Biochem. Biotechnol. 2010, 162, 1174–1186. [Google Scholar] [CrossRef]
- Martínez-Ruiz, M.; Molina-Vázquez, A.; Santiesteban-Romero, B.; Reyes-Pardo, H.; Villaseñor-Zepeda, K.R.; Meléndez-Sánchez, E.R.; Araújo, R.G.; Sosa-Hernández, J.E.; Bilal, M.; Iqbal, H.M.N.; et al. Micro-algae assisted green bioremediation of water pollutants rich leachate and source products recovery. Environ. Pollut. 2022, 306, 119422. [Google Scholar] [CrossRef]
- Guldhe, A.; Kumari, S.; Ramanna, L.; Ramsundar, P.; Singh, P.; Rawat, I.; Bux, F. Prospects, recent advancements and challenges of different wastewater streams for microalgal cultivation. J. Environ. Manag. 2017, 203, 299–315. [Google Scholar] [CrossRef]
- Cho, S.; Lee, N.; Park, S.; Yu, J.; Luong, T.T.; Oh, Y.-K.; Lee, T. Microalgae cultivation for bioenergy production using wastewaters from a municipal WWTP as nutritional sources. Bioresour. Technol. 2013, 131, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Min, M.; Li, Y.; Hu, B.; Ma, X.; Cheng, Y.; Liu, Y.; Chen, P.; Ruan, R. A hetero-photoautotrophic two-stage cultivation process to improve wastewater nutrient removal and enhance algal lipid accumulation. Bioresour. Technol. 2012, 110, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Mat Aron, N.S.; Khoo, K.S.; Chew, K.W.; Veeramuthu, A.; Chang, J.-S.; Show, P.L. Microalgae cultivation in wastewater and potential processing strategies using solvent and membrane separation technologies. J. Water Process Eng. 2021, 39, 101701. [Google Scholar] [CrossRef]
- Tan, J.S.; Lee, S.Y.; Chew, K.W.; Lam, M.K.; Lim, J.W.; Ho, S.-H.; Show, P.L. A review on microalgae cultivation and harvesting, and their biomass extraction processing using ionic liquids. Bioengineered 2020, 11, 116–129. [Google Scholar] [CrossRef]
- Yarkent, Ç.; Gürlek, C.; Oncel, S.S. Potential of microalgal compounds in trending natural cosmetics: A review. Sustain. Chem. Pharm. 2020, 17. [Google Scholar] [CrossRef]
- Riccio, G.; Lauritano, C. Microalgae with Immunomodulatory Activities. Mar. Drugs 2019, 18, 2. [Google Scholar] [CrossRef]
- Tabarzad, M.; Atabaki, V.; Hosseinabadi, T. Anti-inflammatory Activity of Bioactive Compounds from Microalgae and Cyanobacteria by Focusing on the Mechanisms of Action. Mol. Biol. Rep. 2020, 47, 6193–6205. [Google Scholar] [CrossRef]
- Fu, W.; Nelson, D.R.; Yi, Z.; Xu, M.; Khraiwesh, B.; Jijakli, K.; Chaiboonchoe, A.; Alzahmi, A.; Al-Khairy, D.; Brynjolfsson, S.; et al. Bioactive Compounds From Microalgae: Current Development and Prospects. In Studies in Natural Products Chemistry; Rahman, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 54, pp. 199–225. [Google Scholar] [CrossRef]
- Bellou, S.; Baeshen, M.N.; Elazzazy, A.M.; Aggeli, D.; Sayegh, F.; Aggelis, G. Microalgal lipids biochemistry and biotechnological perspectives. Biotechnol. Adv. 2014, 32, 1476–1493. [Google Scholar] [CrossRef]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef]
- Berthon, J.-Y.; Kappes, R.N.; Bey, M.; Cadoret, J.-P.; Renimel, I.; Filaire, E. Marine algae as attractive source to skin care. Free Radic. Res. 2017, 51, 555–567. [Google Scholar] [CrossRef]
- Majtan, J.; Jesenak, M. β-Glucans: Multi-functional modulator of wound healing. Molecules 2018, 23, 806. [Google Scholar] [CrossRef] [PubMed]
- Raposo, M.F.D.J.; de Morais, R.M.S.C.; de Morais, A.M.M.B. Bioactivity and applications of sulphated polysaccharides from marine microalgae. Mar. Drugs 2013, 11, 233–252. [Google Scholar] [CrossRef] [PubMed]
- Fujitani, N.; Sakaki, S.; Yamaguchi, Y.; Takenaka, H. Inhibitory effects of microalgae on the activation of hyaluronidase. J. Appl. Phycol. 2001, 13, 489–492. [Google Scholar] [CrossRef]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef]
- Guzmán, S.; Gato, A.; Calleja, J.M. Antiinflammatory, analgesic and free radical scavenging activities of the marine microalgae Chlorella stigmatophora and Phaeodactylum tricornutum. Phytother. Res. 2001, 15, 224–230. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Microalgae as sources of pharmaceuticals and other biologically active compounds. J. Appl. Phycol. 1995, 7, 3–15. [Google Scholar] [CrossRef]
- Jovanović, M.; Golušin, Z. Nonsteroidal Topical Immunomodulators in Allergology and Dermatology. Biomed. Res. Int. 2016, 2016, 5185303. [Google Scholar] [CrossRef][Green Version]
- Meykadeh, N.; Hengge, U.R. Topische immunmodulation in der dermatologie. Der Hautarzt 2003, 50, 641–662. [Google Scholar] [CrossRef]
- Hengge, U.R.; Benninghoff, B.; Ruzicka, T.; Goos, M. Topical immunomodulators--progress towards treating inflammation, infection, and cancer. Lancet Infect. Dis. 2001, 1, 189–198. [Google Scholar] [CrossRef]
- Pasparakis, M.; Haase, I.; Nestle, F.O. Mechanisms regulating skin immunity and inflammation. Nat. Rev. Immunol. 2014, 14, 289–301. [Google Scholar] [CrossRef]
- Sauder, D. The Role of Epidermal Cytokines in Inflammatory Skin Diseases. J. Investig. Dermatol. 1990, 95, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Choo, W.-T.; Teoh, M.-L.; Phang, S.-M.; Convey, P.; Yap, W.-H.; Goh, B.-H.; Beardall, J. Microalgae as Potential Anti-Inflammatory Natural Product Against Human Inflammatory Skin Diseases. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Hong, J.T.; Kim, Y.; Han, S.-B. Stimulatory Effect of β-glucans on Immune Cells. Immune Netw. 2011, 11, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D. Dectin-1: A signalling non-TLR pattern-recognition receptor. Nat. Rev. Immunol. 2006, 6, 33–43. [Google Scholar] [CrossRef]
- Huang, L.; Shen, M.; Morris, G.A.; Xie, J. Sulfated polysaccharides: Immunomodulation and signaling mechanisms. Trends Food Sci. Technol. 2019, 92, 1–11. [Google Scholar] [CrossRef]
- Sun, L.; Zhao, Y. The biological role of dectin-1 in immune response. Int. Rev. Immunol. 2007, 26, 349–364. [Google Scholar] [CrossRef]
- Miguel, S.P.; Ribeiro, M.P.; Otero, A.; Coutinho, P. Application of microalgae and microalgal bioactive compounds in skin regeneration. Algal Res. 2021, 58, 102395. [Google Scholar] [CrossRef]
- Matsui, M.S.; Muizzuddin, N.; Arad, S.; Marenus, K. Sulfated polysaccharides from red microalgae have antiinflammatory properties In Vitro and In Vivo. Appl. Biochem. Biotechnol. 2003, 104, 13–22. [Google Scholar] [CrossRef]
- Syarina, P.N.A.; Karthivashan, G.; Abas, F.; Arulselvan, P.; Fakurazi, S. Wound healing potential of Spirulina platensis extracts on human dermal fibroblast cells. EXCLI J. 2015, 14, 385–393. [Google Scholar] [CrossRef]
- Guzmán, F.; Wong, G.; Román, T.; Cárdenas, C.; Alvárez, C.; Schmitt, P.; Albericio, F.; Rojas, V. Identification of Antimicrobial Peptides from the Microalgae Tetraselmis suecica (Kylin) Butcher and Bactericidal Activity Improvement. Mar. Drugs 2019, 17, 453. [Google Scholar] [CrossRef]
- Alsenani, F.; Tupally, K.R.; Chua, E.T.; Eltanahy, E.; Alsufyani, H.; Parekh, H.S.; Schenk, P.M. Evaluation of microalgae and cyanobacteria as potential sources of antimicrobial compounds. Saudi Pharm. J. 2020, 28, 1834–1841. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, C.; Suryawanshi, P.G.; Kalita, M.C.; Deka, D.; Aranda, D.A.G.; Goud, V.V. Polarity-wise successive solvent extraction of Scenedesmus obliquus biomass and characterization of the crude extracts for broad-spectrum antibacterial activity. Biomass Convers. Biorefin. 2022. [Google Scholar] [CrossRef]
- Cepas, V.; Gutiérrez-Del-Río, I.; López, Y.; Redondo-Blanco, S.; Gabasa, Y.; Iglesias, M.J.; Soengas, R.; Fernández-Lorenzo, A.; López-Ibáñez, S.; Villar, C.J.; et al. Microalgae and Cyanobacteria Strains as Producers of Lipids with Antibacterial and Antibiofilm Activity. Mar. Drugs 2021, 19, 675. [Google Scholar] [CrossRef] [PubMed]
- Potocki, L.; Oklejewicz, B.; Kuna, E.; Szpyrka, E.; Duda, M.; Zuczek, J. Application of Green Algal Planktochlorella nurekis Biomasses to Modulate Growth of Selected Microbial Species. Molecules 2021, 26, 4038. [Google Scholar] [CrossRef]
- Marrez, D.A.; Naguib, M.M.; Sultan, Y.; Higazy, A.M. Antimicrobial and anticancer activities of Scenedesmus obliquus metabolites. Heliyon 2019, 5, e01404. [Google Scholar] [CrossRef]
- Chester, K.; Zahiruddin, S.; Ahmad, A.; Khan, W.; Paliwal, S.; Ahmad, S. Bioautography-based identification of antioxidant me-tabolites of Solanum nigrum L. and exploration its hepatoprotective potential against D-galactosamine-induced hepatic fi-brosis in rats. Phcog. Mag. 2019, 15 (Suppl. S1), 104–110. [Google Scholar]
- Lauritano, C.; Andersen, J.H.; Hansen, E.; Albrigtsen, M.; Escalera, L.; Esposito, F.; Helland, K.; Hanssen, K.Ø.; Romano, G.; Ianora, A. Bioactivity Screening of Microalgae for Antioxidant, Anti-Inflammatory, Anticancer, Anti-Diabetes, and Antibacterial Activities. Front. Mar. Sci. 2016, 3, 68. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- de Jager, T.L.; Cockrell, A.E.; Du Plessis, S.S. Ultraviolet Light Induced Generation of Reactive Oxygen Species. In Ultraviolet Light in Human Health, Diseases and Environment; Springer: Cham, Switzerland, 2017; Volume 996, pp. 15–23. [Google Scholar] [CrossRef]
- Sansone, C.; Brunet, C. Promises and Challenges of Microalgal Antioxidant Production. Antioxidants 2019, 8, 199. [Google Scholar] [CrossRef]
- Vechtomova, Y.L.; Telegina, T.A.; Buglak, A.A.; Kritsky, M.S. UV radiation in DNA damage and repair involving DNA-photolyases and cryptochromes. Biomedicines 2021, 9, 1564. [Google Scholar] [CrossRef]
- Goiris, K.; Muylaert, K.; Fraeye, I.; Foubert, I.; De Brabanter, J.; De Cooman, L. Antioxidant potential of microalgae in relation to their phenolic and carotenoid content. J. Appl. Phycol. 2012, 24, 1477–1486. [Google Scholar] [CrossRef]
- Lekshmi, S.; Saramma, A.V. Antioxidant activity of synechococcus sp. Nägeli isolated from cochin estuary, India. Indian J. Geo-Mar. Sci. 2018, 47, 2213–2216. [Google Scholar]
- Foo, S.C.; Yusoff, F.M.; Ismail, M.; Basri, M.; Yau, S.K.; Khong, N.M.H.; Chan, K.W.; Ebrahimi, M. Antioxidant capacities of fucoxanthin-producing algae as influenced by their carotenoid and phenolic contents. J. Biotechnol. 2017, 241, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Jerez-Martel, I.; García-Poza, S.; Rodríguez-Martel, G.; Rico, M.; Afonso-Olivares, C.; Gómez-Pinchetti, J.L. Phenolic Profile and Antioxidant Activity of Crude Extracts from Microalgae and Cyanobacteria Strains. J. Food Qual. 2017, 2017, 2924508. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Gouveia, L.; Batista, A.P.; Sousa, I.; Raymundo, A.; Bandarra, N.M. Microalgae in Novel Food Products; Nova Science Publishers: New York, NY, USA, 2009; ISBN 9781606920084. [Google Scholar]
- Aditi, N.; Jaishini, C.; Raisa, F.; Aamna, K.; Rupali, B. Photosynthetic pigments, lipids and phenolic compounds of three green algae isolated from freshwater ecosystem. J. Algal Biomass Util. 2020, 11, 68–83. [Google Scholar]
- Seth, K.; Kumar, A.; Rastogi, R.P.; Meena, M.; Vinayak, V. Harish Bioprospecting of fucoxanthin from diatoms—Challenges and perspectives. Algal Res. 2021, 60, 102475. [Google Scholar] [CrossRef]
- Su, J.; Guo, K.; Huang, M.; Liu, Y.; Zhang, J.; Sun, L.; Li, D.; Pang, K.-L.; Wang, G.; Chen, L.; et al. Fucoxanthin, a Marine Xanthophyll Isolated From Conticribra weissflogii ND-8: Preventive Anti-Inflammatory Effect in a Mouse Model of Sepsis. Front. Pharmacol. 2019, 10, 906. [Google Scholar] [CrossRef]
- Zarekarizi, A.; Hoffmann, L.; Burritt, D. Approaches for the sustainable production of fucoxanthin, a xanthophyll with potential health benefits. J. Appl. Phycol. 2019, 31, 281–299. [Google Scholar] [CrossRef]
- Vinayak, V. Algae as sustainable food in space missions. In Biomass, Biofuels, Biochemicals; ELSIVIER: Alpharetta, GA, USA, 2021; pp. 517–540. ISBN 9780323898553. [Google Scholar]
- Cycil, L.M.; Hausrath, E.M.; Ming, D.W.; Adcock, C.T.; Raymond, J.; Remias, D.; Ruemmele, W.P. Investigating the Growth of Algae Under Low Atmospheric Pressures for Potential Food and Oxygen Production on Mars. Front. Microbiol. 2021, 12, 199–225. [Google Scholar] [CrossRef]
- Aziz, J.; Shezali, H.; Radzi, Z.; Yahya, N.A.; Abu Kassim, N.H.; Czernuszka, J.; Rahman, M.T. Molecular Mechanisms of Stress-Responsive Changes in Collagen and Elastin Networks in Skin. Ski. Pharmacol. Physiol. 2016, 29, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Becker, E.W. Microalgae for Human and Animal Nutrition. In Handbook of Microalgal Culture: Biotechnology and Applied Phycology; Wiley Online Library: Hoboken, NJ, USA, 2004; pp. 461–503. [Google Scholar]
- Galasso, C.; Gentile, A.; Orefice, I.; Ianora, A.; Bruno, A.; Noonan, D.M.; Sansone, C.; Albini, A.; Brunet, C. Microalgal Derivatives as Potential Nutraceutical and Food Supplements for Human Health: A Focus on Cancer Prevention and Interception. Nutrients 2019, 11, 1226. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef] [PubMed]
- Sansone, C.; Galasso, C.; Orefice, I.; Nuzzo, G.; Luongo, E.; Cutignano, A.; Romano, G.; Brunet, C.; Fontana, A.; Esposito, F.; et al. The green microalga Tetraselmis suecica reduces oxidative stress and induces repairing mechanisms in human cells. Sci. Rep. 2017, 7, 41215. [Google Scholar] [CrossRef]
- Pistelli, L.; Del Mondo, A.; Smerilli, A.; Corato, F.; Piscitelli, C.; Pellone, P.; Carbone, D.A.; Sansone, C.; Brunet, C. Microalgal co-cultivation prospecting to modulate vitamin and bioactive compounds production. Antioxidants 2021, 10, 1360. [Google Scholar] [CrossRef] [PubMed]
- Milito, A.; Orefice, I.; Smerilli, A.; Castellano, I.; Napolitano, A.; Brunet, C.; Palumbo, A. Insights into the light response of skeletonema marinoi: Involvement of ovothiol. Mar. Drugs 2020, 18, 477. [Google Scholar] [CrossRef]
- Shah, M.M.R.; Liang, Y.; Cheng, J.J.; Daroch, M. Astaxanthin-Producing Green Microalga Haematococcus pluvialis: From Single Cell to High Value Commercial Products. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Gaucher, C.; Boudier, A.; Bonetti, J.; Clarot, I.; Leroy, P.; Parent, M. Glutathione: Antioxidant properties dedicated to nanotechnologies. Antioxidants 2018, 7, 62. [Google Scholar] [CrossRef]
- Kageyama, H.; Waditee-Sirisattha, R. Antioxidative, anti-inflammatory, and anti-aging properties of mycosporine-like amino acids: Molecular and cellular mechanisms in the protection of skin-aging. Mar. Drugs 2019, 17, 222. [Google Scholar] [CrossRef]
- Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as sources of high added-value compounds—A brief review of recent work. Biotechnol. Prog. 2011, 27, 597–613. [Google Scholar] [CrossRef]
- Alvares, B.A.; Miola, A.C.; Schimitt, J.V.; Miot, H.A.; Abbade, L.P.F. Efficacy of sunscreen with photolyase or regular sunscreen associated with topical antioxidants in treating advanced photodamage and cutaneous field cancerization: A randomized clinical trial. An. Bras. Dermatol. 2022, 97, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Petruk, G.; Del Giudice, R.; Rigano, M.M.; Monti, D.M. Antioxidants from plants protect against skin photoaging. Oxid. Med. Cell. Longev. 2018, 2018, 1454936. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, L.; Raymundo, A.; Batista, A.P.; Sousa, I.; Empis, J. Chlorella vulgaris and Haematococcus pluvialis biomass as colouring and antioxidant in food emulsions. Eur. Food Res. Technol. 2006, 222, 362–367. [Google Scholar] [CrossRef]
- Suh, S.S.; Hwang, J.; Park, M.; Seo, H.H.; Kim, H.S.; Lee, J.H.; Moh, S.H.; Lee, T.K. Anti-inflammation activities of mycosporine-like amino acids (MAAs) in response to UV radiation suggest potential anti-skin aging activity. Mar. Drugs 2014, 12, 5174–5187. [Google Scholar] [CrossRef] [PubMed]
- Challouf, R.; Trabelsi, L.; Ben Dhieb, R.; El Abed, O.; Yahia, A.; Ghozzi, K.; Ben Ammar, J.; Omran, H.; Ben Ouada, H. Evaluation of cytotoxicity and biological activities in extracellular polysaccharides released by cyanobacterium Arthrospira platensis. Braz. Arch. Biol. Technol. 2011, 54, 831–838. [Google Scholar] [CrossRef]
- Xia, S.; Gao, B.; Li, A.; Xiong, J.; Ao, Z.; Zhang, C. Preliminary Characterization, Antioxidant Properties and Production of Chrysolaminarin from Marine Diatom Odontella aurita. Mar. Drugs 2014, 12, 4883–4897. [Google Scholar] [CrossRef]
- Tannin-Spitz, T.; Bergman, M.; Van-Moppes, D.; Grossman, S.; Arad, S. Antioxidant activity of the polysaccharide of the red microalga Porphyridium sp. J. Appl. Phycol. 2005, 17, 215–222. [Google Scholar] [CrossRef]
- Chen, B.; You, W.; Huang, J.; Yu, Y.; Chen, W. Isolation and antioxidant property of the extracellular polysaccharide from Rhodella reticulata. World J. Microbiol. Biotechnol. 2010, 26, 833–840. [Google Scholar] [CrossRef]
- Smerilli, A.; Balzano, S.; Maselli, M.; Blasio, M.; Orefice, I.; Galasso, C.; Sansone, C.; Brunet, C. Antioxidant and photoprotection networking in the coastal diatom skeletonema marinoi. Antioxidants 2019, 8, 154. [Google Scholar] [CrossRef]
- Colusse, G.A.; Mendes, C.R.B.; Duarte, M.E.R.; de Carvalho, J.C.; Noseda, M.D. Effects of different culture media on physiological features and laboratory scale production cost of Dunaliella salina. Biotechnol. Rep. 2020, 27. [Google Scholar] [CrossRef]
- Hashtroudi, M.S.; Shariatmadari, Z.; Riahi, H.; Ghassempour, A. Analysis of Anabaena vaginicola and Nostoc calcicola from Northern Iran, as rich sources of major carotenoids. Food Chem. 2013, 136, 1148–1153. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, R.P.; Madamwar, D.; Nakamoto, H.; Incharoensakdi, A. Resilience and self-regulation processes of microalgae under UV radiation stress. J. Photochem. Photobiol. C Photochem. Rev. 2020, 43, 100322. [Google Scholar] [CrossRef]
- Sharma, A.; Singh, R.S.; Gupta, G.; Ahmad, T.; Kaur, B. Metabolic engineering of enzyme-regulated bioprocesses. In Biomass, Biofuels, Biochemicals: Advances in Enzyme Catalysis and Technologies; Elsivier: Alpharetta, GA, USA, 2019; pp. 293–323. [Google Scholar] [CrossRef]
- Putu Parwata, I.; Wahyuningrum, D.; Suhandono, S.; Hertadi, R. Production of ectoine by Halomonas elongata BK-AG25 using osmotic shock technique. IOP Conf. Ser. Earth Environ. Sci. 2018, 209. [Google Scholar] [CrossRef]
- Botta, C.; Di Giorgio, C.; Sabatier, A.S.; De Méo, M. Genotoxicity of visible light (400-800 nm) and photoprotection assessment of ectoin, l-ergothioneine and mannitol and four sunscreens. J. Photochem. Photobiol. B Biol. 2008, 91, 24–34. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, P.M.; Javalkote, V.S.; Mazmouz, R.; Pickford, R.; Puranik, P.R.; Neilan, B.A. Comparative profiling and discovery of novel glycosylated mycosporine-like amino acids in two strains of the cyanobacterium Scytonema cf. crispum. Appl. Environ. Microbiol. 2016, 82, 5951–5959. [Google Scholar] [CrossRef] [PubMed]
- Raj, S.; Kuniyil, A.M.; Sreenikethanam, A.; Gugulothu, P.; Jeyakumar, R.B.; Bajhaiya, A.K. Microalgae as a source of mycosporine-like amino acids (Maas); advances and future prospects. Int. J. Environ. Res. Public Health 2021, 18, 12402. [Google Scholar] [CrossRef] [PubMed]
- Gabros, S.; Nessel, T.; Zito, P. Sunscreens and Photoprotection. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537164/ (accessed on 26 January 2022).
- Rai, R.; Shanmuga, S.C.; Srinivas, C. Update on photoprotection. Indian J. Dermatol. 2012, 57, 335–342. [Google Scholar] [CrossRef]
- Núñez-Pons, L.; Avila, C.; Romano, G.; Verde, C.; Giordano, D. UV-Protective Compounds in Marine Organisms from the Southern Ocean. Mar. Drugs 2018, 16, 336. [Google Scholar] [CrossRef]
- White, A.L.; Jahnke, L.S. Contrasting Effects of UV-A and UV-B on Photosynthesis and Photoprotection of β-carotene in two Dunaliella spp. Plant Cell Physiol. 2002, 43, 877–884. [Google Scholar] [CrossRef]
- Joshi, D.; Mohandass, C.; Dhale, M. Effect of UV-B Radiation and Desiccation Stress on Photoprotective Compounds Accumulation in Marine Leptolyngbya sp. Appl. Biochem. Biotechnol. 2018, 184, 35–47. [Google Scholar] [CrossRef]
- Singh, D.K.; Pathak, J.; Pandey, A.; Singh, V.; Ahmed, H.; Kumar, D.; Rajneesh; Sinha, R.P. Response of a rice-field cyanobacterium Anabaena sp. HKAR-7 upon exposure to ultraviolet-B radiation and ammonium chloride. Environ. Sustain. 2021, 4, 95–105. [Google Scholar] [CrossRef]
- Xiong, F.; Komenda, J.; Kopecky, J.; Nedbal, L. Strategies of ultraviolet-B protection in microscopic algae. Physiol. Plant. 1997, 100, 378–388. [Google Scholar] [CrossRef]
- Gao, Y.; Cui, Y.; Xiong, W.; Li, X.; Wu, Q. Effect of UV-C on Algal Evolution and Differences in Growth Rate, Pigmentation and Photosynthesis Between Prokaryotic and Eukaryotic Algae. Photochem. Photobiol. 2009, 85, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, K.; Chidambaram, K.; Janarthanan, R.; Ramasamy, R. Effect of UV-B Radiation on Growth, Photosynthetic Activity andMetabolic Activities of Chlorella vulgaris. Res. Rev. J. Microbiol. Biotechnol. 2017, 6, 53–60. [Google Scholar]
- Hernando, M.; Schlossi, I.; Roy, S.; Ferreyra, G. Symposium-in-Print: UV Effects on Aquatic and Coastal Ecosystems Photoacclimation to Long-Term Ultraviolet Radiation Exposure of Natural Sub-Antarctic Phytoplankton Communities: Fixed Surface Incubations Versus Mixed Mesocosms. Photochem. Photobiol. 2006, 923–935. [Google Scholar] [CrossRef]
- Chen, L.; Deng, S.; De Philippis, R.; Tian, W.; Wu, H.; Wang, J. UV-B resistance as a criterion for the selection of desert microalgae to be utilized for inoculating desert soils. J. Appl. Phycol. 2013, 25, 1009–1015. [Google Scholar] [CrossRef]
- Fouqueray, M.; Mouget, J.-L.; Morant-Manceau, A.; Tremblin, G. Dynamics of short-term acclimation to UV radiation in marine diatoms. J. Photochem. Photobiol. B. 2007, 89, 1–8. [Google Scholar] [CrossRef]
- Helbling, E.W.; Gao, K.; Ai, H.; Ma, Z.; Villafañe, V.E. Differential responses of Nostoc sphaeroides and Arthrospira platensis to solar ultraviolet radiation exposure. J. Appl. Phycol. 2006, 18, 57–66. [Google Scholar] [CrossRef]
- Buma, A.; Wright, S.; Enden, R.; van de Poll, W.; Davidson, A. PAR acclimation and UVBR-induced DNA damage in Antarctic marine microalgae. Mar. Ecol. Prog. Ser. 2006, 315, 33–42. [Google Scholar] [CrossRef]
- Bennett, J.; Soule, T. Expression of Scytonemin Biosynthesis Genes under Alternative Stress Conditions in the Cyanobacterium Nostoc punctiforme. Microorganisms 2022, 10, 427. [Google Scholar] [CrossRef]
- Pathak, J.; Kumar, D.; Singh, D.K.; Ahmed, H.; Kannaujiya, V.K.; Sinha, R.P. Ultraviolet radiation and salinity-induced physiological changes and scytonemin induction in cyanobacteria isolated from diverse habitats. Biointerface Res. Appl. Chem. 2022, 12, 3590–3606. [Google Scholar] [CrossRef]
- Orellana, G.; Gómez-Silva, B.; Urrutia, M.; Galetović, A. UV-A Irradiation Increases Scytonemin Biosynthesis in Cyanobacteria Inhabiting Halites at Salar Grande, Atacama Desert. Microorganisms 2020, 8, 1690. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.P.; Pathak, J.; Rajneesh; Ahmed, H.; Pandey, A.; Singh, P.R.; Mishra, S.; Häder, D.-P. Cyanobacterial photoprotective compounds: Characterization and utilization in human welfare. In Natural Bioactive Compounds; Sinha, R.P., Häder, D.-P., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 187–201. ISBN 9780128206553. [Google Scholar]
- Gao, X.; Jing, X.; Liu, X.; Lindblad, P. Biotechnological Production of the Sunscreen Pigment Scytonemin in Cyanobacteria: Progress and Strategy. Mar. Drugs 2021, 19, 129. [Google Scholar] [CrossRef] [PubMed]
- Sibilla, S.; Godfrey, M.; Brewer, S.; Budh-Raja, A.; Genovese, L. An overview of the beneficial effects of hydrolysed collagen as a nutraceutical on skin properties: Scientific background and clinical studies. Open Nutraceuticals J. 2015, 8, 29–42. [Google Scholar] [CrossRef]
- Perin, G.; Bellan, A.; Bernardi, A.; Bezzo, F.; Morosinotto, T. The potential of quantitative models to improve microalgae photosynthetic efficiency. Physiol. Plant. 2019, 166, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Dixon, C.; Wilken, L.R. Green microalgae biomolecule separations and recovery. Bioresour. Bioprocess. 2018, 5, 14. [Google Scholar] [CrossRef]
- Sheih, I.-C.; Wu, T.-K.; Fang, T.J. Antioxidant properties of a new antioxidative peptide from algae protein waste hydrolysate in different oxidation systems. Bioresour. Technol. 2009, 100, 3419–3425. [Google Scholar] [CrossRef]
- Yaakob, Z.; Ali, E.; Zainal, A.; Mohamad, M.; Takriff, M.S. An overview: Biomolecules from microalgae for animal feed and aquaculture. J. Biol. Res. 2014, 21, 6. [Google Scholar] [CrossRef]
- de la Noue, J.; de Pauw, N. The potential of microalgal biotechnology: A review of production and uses of microalgae. Biotechnol. Adv. 1988, 6, 725–770. [Google Scholar] [CrossRef]
- Odjadjare, E.C.; Mutanda, T.; Olaniran, A.O. Potential biotechnological application of microalgae: A critical review. Crit. Rev. Biotechnol. 2017, 37, 37–52. [Google Scholar] [CrossRef]
- Hamed, I. The Evolution and Versatility of Microalgal Biotechnology: A Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 1104–1123. [Google Scholar] [CrossRef] [PubMed]
- Gold, N.A.; Mirza, T.M.; Avva, U. Alcohol Sanitizer; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Hoang, H.T.; Van Tran, V.; Bui, V.K.H.; Kwon, O.-H.; Moon, J.-Y.; Lee, Y.-C. Novel moisturized and antimicrobial hand gel based on zinc-aminoclay and Opuntia humifusa extract. Sci. Rep. 2021, 11, 17821. [Google Scholar] [CrossRef] [PubMed]
- Bernaerts, T.M.M.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. The potential of microalgae and their biopolymers as structuring ingredients in food: A review. Biotechnol. Adv. 2019, 37, 107419. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; De Philippis, R. Exocellular Polysaccharides in Microalgae and Cyanobacteria: Chemical Features, Role and Enzymes and Genes Involved in Their Biosynthesis. In The Physiology of Microalgae; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 565–590. [Google Scholar]
- Pierre, G.; Delattre, C.; Dubessay, P.; Jubeau, S.; Vialleix, C.; Cadoret, J.-P.; Probert, I.; Michaud, P. What Is in Store for EPS Microalgae in the Next Decade? Molecules 2019, 24, 4296. [Google Scholar] [CrossRef]
- Jain, R.; Raghukumar, S.; Tharanathan, R.; Bhosle, N. Extracellular Polysaccharide Production by Thraustochytrid Protists. Mar. Biotechnol. 2005, 7, 184–192. [Google Scholar] [CrossRef]
- Urbanelli, L.; Buratta, S.; Tancini, B.; Sagini, K.; Delo, F.; Porcellati, S.; Emiliani, C. The Role of Extracellular Vesicles in Viral Infection and Transmission. Vaccines 2019, 7, 102. [Google Scholar] [CrossRef]
- Adamo, G.; Fierli, D.; Romancino, D.P.; Picciotto, S.; Barone, M.E.; Aranyos, A.; Božič, D.; Morsbach, S.; Raccosta, S.; Stanly, C.; et al. Nanoalgosomes: Introducing extracellular vesicles produced by microalgae. J. Extracell. Vesicles 2021, 10, e12081. [Google Scholar] [CrossRef]
- Delalat, B.; Sheppard, V.C.; Rasi Ghaemi, S.; Rao, S.; Prestidge, C.A.; McPhee, G.; Rogers, M.-L.; Donoghue, J.F.; Pillay, V.; Johns, T.G.; et al. Targeted drug delivery using genetically engineered diatom biosilica. Nat. Commun. 2015, 6, 8791. [Google Scholar] [CrossRef]
- Khavari, F.; Saidijam, M.; Taheri, M.; Nouri, F. Microalgae: Therapeutic potentials and applications. Mol. Biol. Rep. 2021, 48, 4757–4765. [Google Scholar] [CrossRef]
- Enamala, M.K.; Enamala, S.; Chavali, M.; Donepudi, J.; Yadavalli, R.; Kolapalli, B.; Aradhyula, T.V.; Velpuri, J.; Kuppam, C. Production of biofuels from microalgae-A review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renew. Sustain. Energy Rev. 2018, 94, 49–68. [Google Scholar] [CrossRef]
- Gunes, S.; Tamburaci, S.; Dalay, M.C.; Gurhan, I.D. In vitro evaluation of spirulina platensis extract incorporated skin cream with its wound healing and antioxidant activities. Pharm. Biol. 2017, 55, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- Tarasuntisuk, S.; Patipong, T.; Hibino, T.; Waditee-Sirisattha, R.; Kageyama, H. Inhibitory effects of mycosporine-2-glycine isolated from a halotolerant cyanobacterium on protein glycation and collagenase activity. Lett. Appl. Microbiol. 2018, 67, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Eigemann, F.; Vanormelingen, P.; Hilt, S. Sensitivity of the Green Alga Pediastrum duplex Meyen to Allelochemicals Is Strain-Specific and Not Related to Co-Occurrence with Allelopathic Macrophytes. PLoS ONE 2013, 8, e78463. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.D.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresour. Technol. 2015, 184, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Campiche, R.; Sandau, P.; Kurth, E.; Massironi, M.; Imfeld, D.; Schuetz, R. Protective effects of an extract of the freshwater microalga Scenedesmus rubescens on UV-irradiated skin cells. Int. J. Cosmet. Sci. 2018, 40, 187–192. [Google Scholar] [CrossRef]
| Type of Compound | Mechanism of Action | Microalgae Species | Culture Conditions | Extraction | Reference |
|---|---|---|---|---|---|
| Peptides | Antimicrobial activity through interaction with negatively charged membranes | T. suecica (Chlorophyta) | F/2 medium, 21 ± 0.5 °C and 40 W Light | Acid extraction and reverse phase column chromatography separation. | [53] |
| Fatty acid methyl esters (FAME), lipids and carotenoids | Interference in biosynthesis of bacterial fatty acids | Various microalgae species | F/2 medium for marine microalgae and Bold’s basal medium (BBM) for freshwater microalgae | Three extraction systems with different solvents | [54] |
| Fatty acids and pigments | Interference in biosynthesis of bacterial fatty acids | S. obliquus | - | Polarity-wise successive solvent extractions | [55] |
| Lipids | Membrane permeabilization | S. brasiliensis, E. acutiformis and Sphaerospermopsis sp. | M7 medium for microalgae, Z8 medium for cyanobacteria | Extraction with different solvents | [56] |
| Fatty acids | Interference in biosynthesis of bacterial fatty acids | P. nurekis | Medium with various macro- and microelements at pH < 6, 30 °C and 16,000 LUX | Extraction with water and ethanol | [57] |
| Fatty acids and phenolic compounds | Interference in biosynthesis of bacterial fatty acids | S. obliquus | BG-11 medium | Extraction with organic solvents and sonication | [58] |
| Pigments | Free radical scavenging | Various microalgae species | Guillard’s F/2 medium, 22 ± 2 °C, 14:10 D:L cycle, 60–65 µ·E·m−2·s−1light for 16 days | Ethanol extraction | [59] |
| Microalgae extract | Various mechanisms tested | 32 microalgae species | Guillard’s F/2 medium, 19 °C, 12:12 D:L cycle, and 100 µmo·m−2·s−1 Light | Sonication and acetone extraction | [60] |
| Microalgae Species | UV Test | Intensity Unit | Resistance Factor | Type of Study | Culture Conditions | Reference |
|---|---|---|---|---|---|---|
| Anabaena spp. (Cyanobacteria) | UVB | 1 W·m−2 for 4 h/day | Scytonemin, MAAs | Exposure to UVB-R | BG-11 medium, NH4Cl as N-source, 28 ± 2 °C | [110] |
| Characium terrestre (Chlorophyta) | UVB | NR | Sporopollenin | Exposure to extreme UVB irradiance | Medium reported by Zachleder et al., 26 °C. | [118] |
| C. protothecoides (Chlorophyta) | UVC | 0.01 to 0.20 W·m−2 | Lutein | Exposure to different UVC irradiances | BG-11 medium, 25 ± 1 °C | [119] |
| C. vulgaris (Chlorophyta) | UVB | 1 to 5 W·m−2 | Sporopollenin, Scytonemin, MAAs | Exposure to different UVB irradiances | Bold Basal medium, 25 ± 1 °C | [111] |
| D.salina (Chlorophyta) | UVA UVB | 110 mmol·m−2·s−1 for UVA | Sporopollenin, Scytonemin, MAAs | Exposure to UVA-R and UVB-R | Medium with NaCl, 26 °C | [108] |
| Eutreptiella sp. (Euglenozoa) | UVB | 280 to 320 nm | Xanthophylls, MAAs | Tested under fixed light | F/2 medium, 10 °C | [120] |
| Nostoc sp. (Cyanobacteria) | UVB | 312 nm | Carotenoids, Scytonemin | Photosynthetic activity essay | BG-11 medium, 25 °C | [112] |
| O. aurita (Bacillariophyta) | UVA UVB | 110 kJ·m−2 | D1 protein, activation of antioxidant enzymes | Exposure to UVA-R and UVB-R | Artificial seawater reported by Harrison et al. | [121] |
| N. sphaeroides (Cyanobacteria) | UVA | 320 nm | Not identified | Exposure to UVA irradiance | BG-11 medium, 23 °C | [122] |
| P. antarctica, P. glacialis (Rhodophyta) | UVB | 280 to 400 nm | MAAs, Xanthophylls | Acclimation to photosynthetically active radiation | GP5 medium, 1.0 ± 0.5 °C | [123] |
| S. platensis (Cyanobacteria) | UVA | 320 nm | Sporopollenin, Scytonemin, MAAs | Exposure to UVA irradiance | BG-11 medium, 23 °C | [122] |
| N. punctiforme | UVA UVC | 100 µm photon m−2s−1 | Scytonemin | Exposure to UVA, UVB and osmotic stress | AA/4 medium | [115] |
| Scytonema sp. | UVA UVB | 6.5 Wm−2 0.56 Wm−2 | MAAs and antioxidant enzymes | Photosynthetically active radiation and salinity | BG-11 medium | [116] |
| Halothece | UVA | 3.6 Wm−2 | Scytonemin | Exposure to UVA-R | Halites | [117] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Ruiz, M.; Martínez-González, C.A.; Kim, D.-H.; Santiesteban-Romero, B.; Reyes-Pardo, H.; Villaseñor-Zepeda, K.R.; Meléndez-Sánchez, E.R.; Ramírez-Gamboa, D.; Díaz-Zamorano, A.L.; Sosa-Hernández, J.E.; et al. Microalgae Bioactive Compounds to Topical Applications Products—A Review. Molecules 2022, 27, 3512. https://doi.org/10.3390/molecules27113512
Martínez-Ruiz M, Martínez-González CA, Kim D-H, Santiesteban-Romero B, Reyes-Pardo H, Villaseñor-Zepeda KR, Meléndez-Sánchez ER, Ramírez-Gamboa D, Díaz-Zamorano AL, Sosa-Hernández JE, et al. Microalgae Bioactive Compounds to Topical Applications Products—A Review. Molecules. 2022; 27(11):3512. https://doi.org/10.3390/molecules27113512
Chicago/Turabian StyleMartínez-Ruiz, Manuel, Carlos Alberto Martínez-González, Dong-Hyun Kim, Berenice Santiesteban-Romero, Humberto Reyes-Pardo, Karen Rocio Villaseñor-Zepeda, Edgar Ricardo Meléndez-Sánchez, Diana Ramírez-Gamboa, Ana Laura Díaz-Zamorano, Juan Eduardo Sosa-Hernández, and et al. 2022. "Microalgae Bioactive Compounds to Topical Applications Products—A Review" Molecules 27, no. 11: 3512. https://doi.org/10.3390/molecules27113512
APA StyleMartínez-Ruiz, M., Martínez-González, C. A., Kim, D.-H., Santiesteban-Romero, B., Reyes-Pardo, H., Villaseñor-Zepeda, K. R., Meléndez-Sánchez, E. R., Ramírez-Gamboa, D., Díaz-Zamorano, A. L., Sosa-Hernández, J. E., Coronado-Apodaca, K. G., Gámez-Méndez, A. M., Iqbal, H. M. N., & Parra-Saldivar, R. (2022). Microalgae Bioactive Compounds to Topical Applications Products—A Review. Molecules, 27(11), 3512. https://doi.org/10.3390/molecules27113512








