Iminosugar Amino Acid idoBR1 Reduces Inflammatory Responses in Microglia
Abstract
:1. Introduction
2. Results
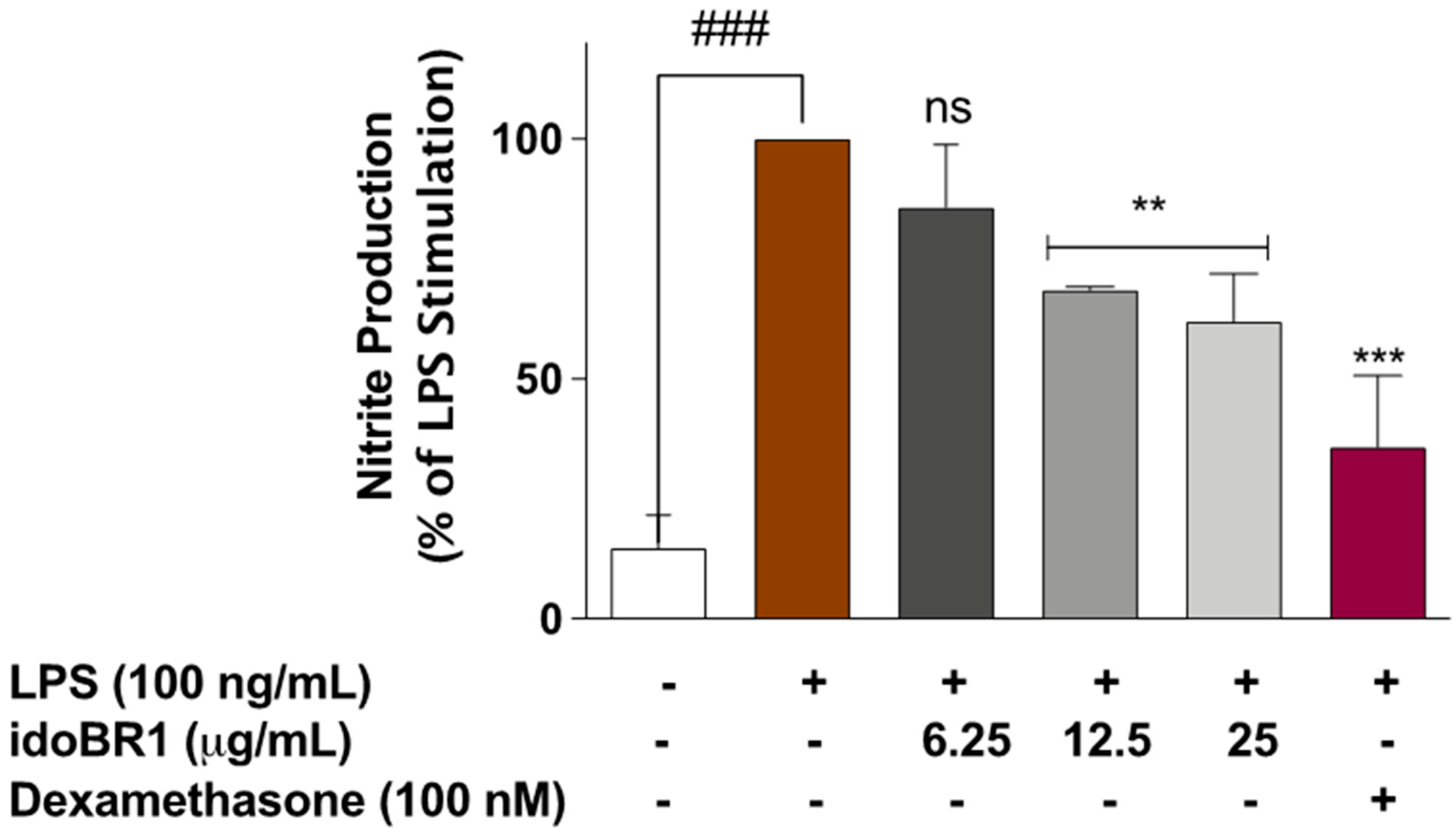
2.1. The Iminosugar idoBR1 Reduced Nitric Oxide Production in LPS-Stimulated BV-2 Microglia
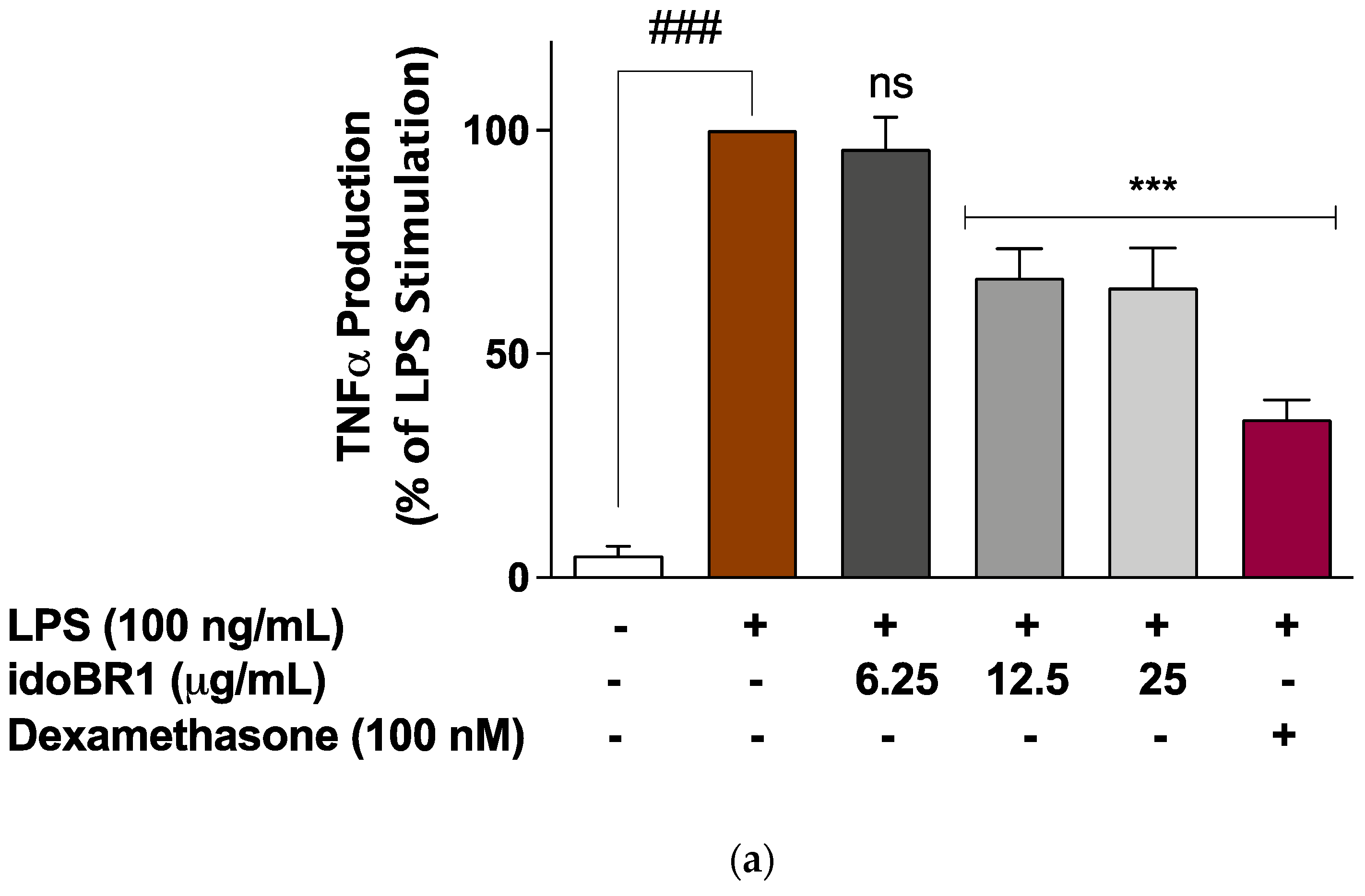
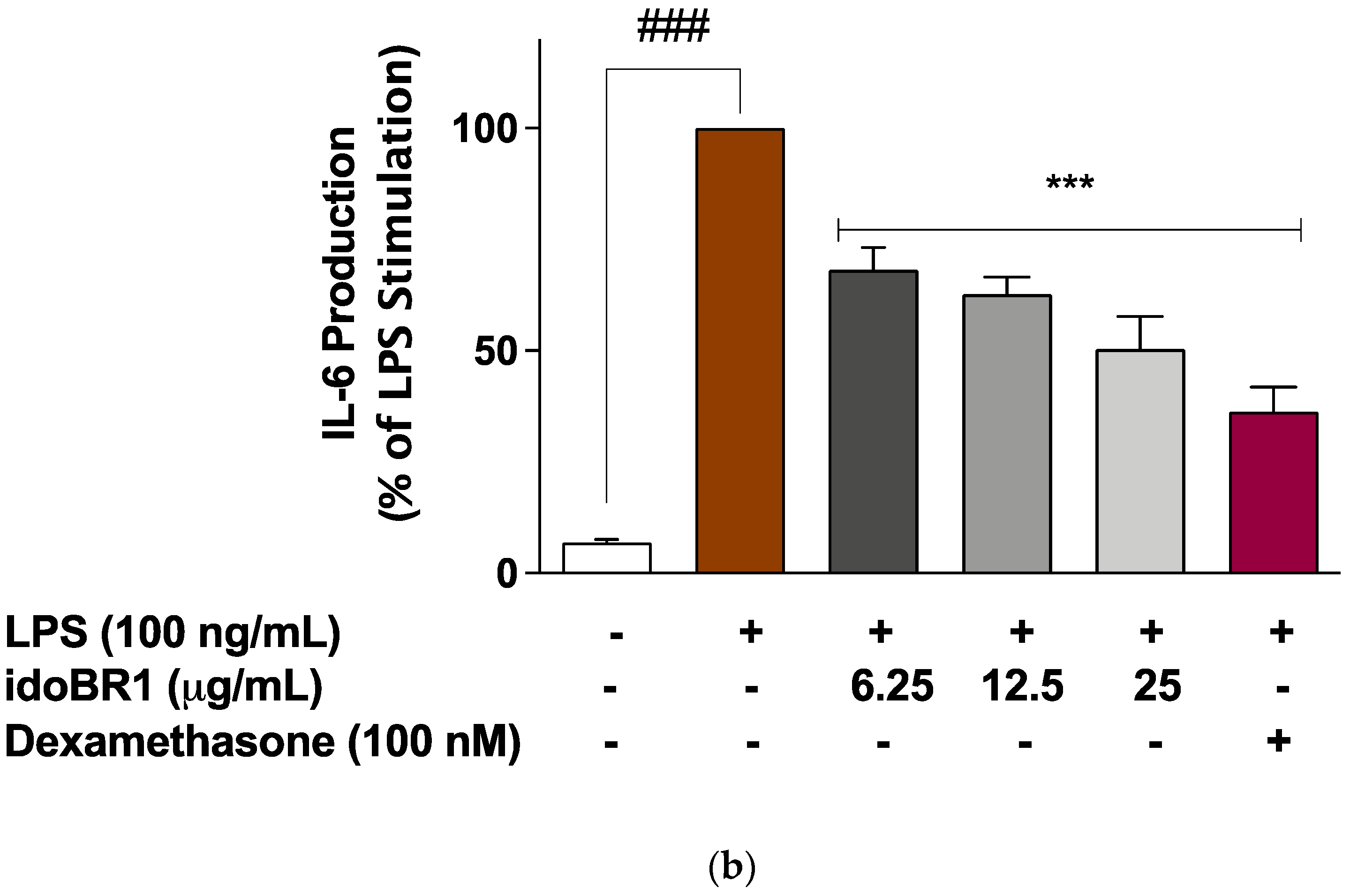
2.2. The Production of TNFα and IL-6 Was Reduced by idoBR1 Treatment
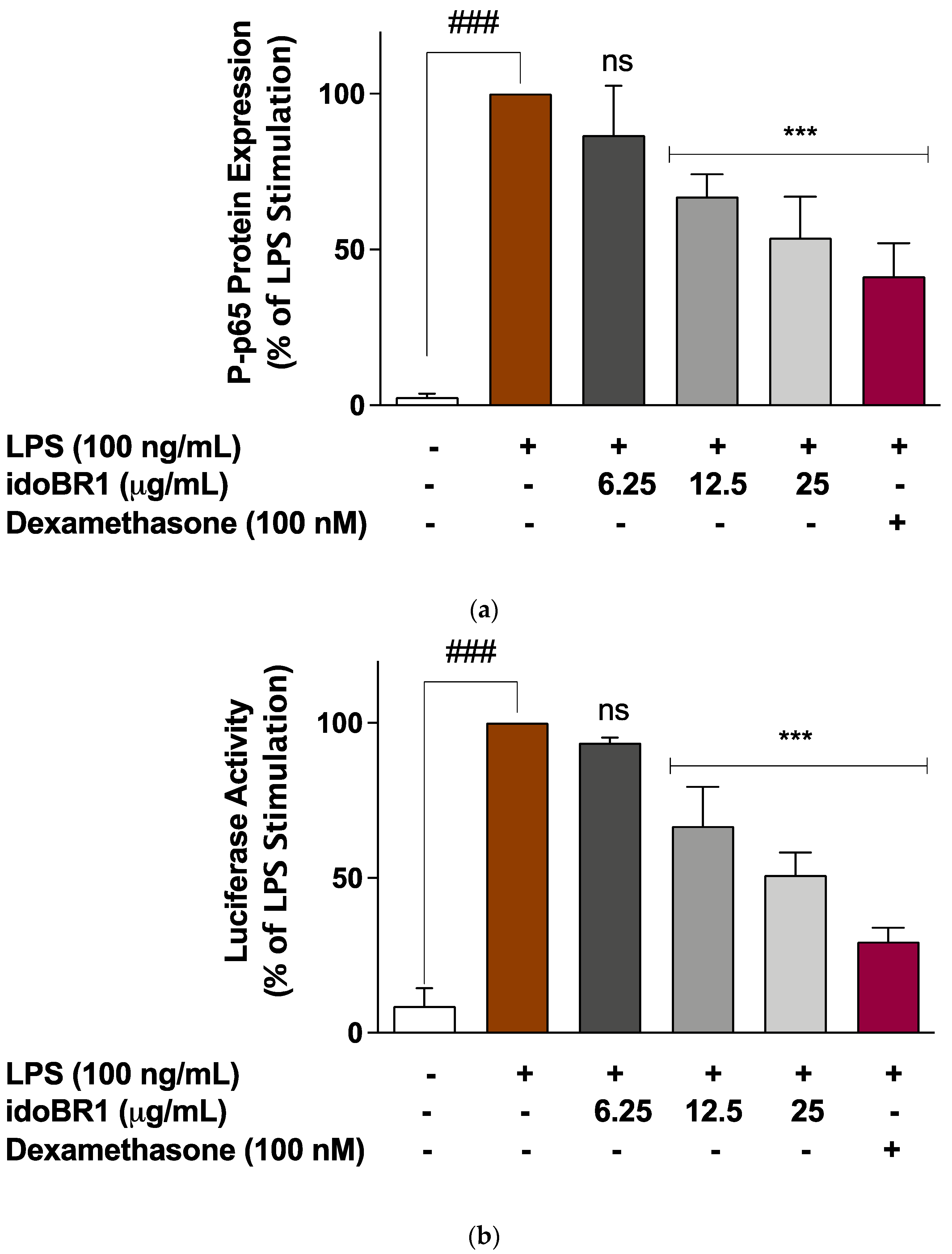
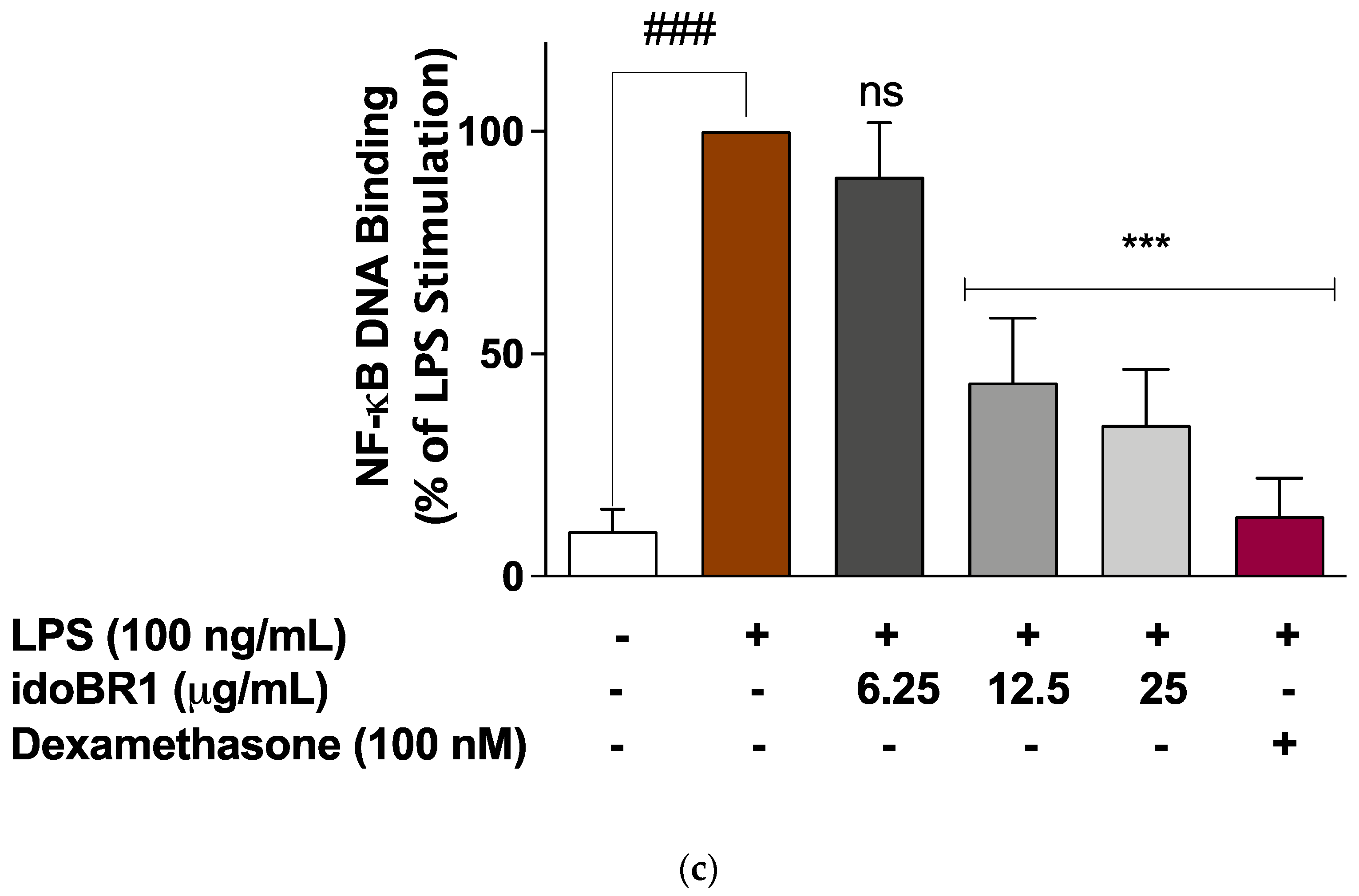
2.3. The Iminosugar idoBR1 Attenuated the Activation of NF-κB in LPS-Stimulated BV-2 Microglia
2.4. The Effects of idoBR1 on the Viability of LPS-Stimulated BV-2 Microglia
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. MTT Cell Viability Assay
4.3. Determination of Nitrite Production
4.4. Determination of TNFα and IL-6 Production
4.5. ELISA for Phospho-NF-κB p65 (Ser536) Sub-Unit
4.6. Transient Transfection and NF-κB Reporter Gene Assay
4.7. NF-κB Transcription Factor Binding Assay
4.8. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Olajide, O.A.; Sarker, S.D. Alzheimer’s disease: Natural products as inhibitors of neuroinflammation. Inflammopharmacology 2020, 28, 1439–1455. [Google Scholar] [CrossRef] [PubMed]
- Walker, K.A. Inflammation and neurodegeneration: Chronicity matters. Aging 2018, 11, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Nema, N.K.; Maity, N.; Sarker, B.K. Phytochemical and therapeutic potential of cucumber. Fitoterapia 2013, 84, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Kimira, M.; Arai, Y.; Shimoi, K.; Watanabe, S. Japanese Intake of Flavonoids and Isoflavonoids from Foods. J. Epidemiol. 1998, 8, 168–175. [Google Scholar] [CrossRef] [Green Version]
- Nash, R.J.; Bartholomew, B.; Penkova, Y.B.; Dino Rotondo, D.; Yamasaka, F.; Stafford, G.P.; Jenkinson, S.F.; Fleet, G.W.J. Iminosugar idoBR1 Isolated from Cucumber Cucumis sativus Reduces Inflammatory Activity. ACS Omega 2020, 5, 16263–16271. [Google Scholar] [CrossRef]
- Griffin, W.S. Perispinal etanercept: Potential as an Alzheimer therapeutic. J. Neuroinflamm. 2008, 5, 3. [Google Scholar] [CrossRef] [Green Version]
- Prinz, M.; Masuda, T.; Wheeler, M.A.; Quintana, F.J. Microglia and Central Nervous System-Associated Macrophages-From Origin to Disease Modulation. Annu. Rev. Immunol. 2021, 39, 251–277. [Google Scholar] [CrossRef]
- Lopes, K.D.P.; Snijders, G.J.L.; Humphrey, J.; Allan, A.; Sneeboer, M.A.M.; Navarro, E.; Schilder, B.M.; Vialle, R.A.; Parks, M.; Missall, R.; et al. Genetic analysis of the human microglial transcriptome across brain regions, aging and disease pathologies. Nat. Genet. 2022, 54, 4–17. [Google Scholar] [CrossRef]
- Webers, A.; Heneka, M.T.; Gleeson, P.A. The role of innate immune responses and neuroinflammation in amyloid accumulation and progression of Alzheimer’s disease. Immunol. Cell Biol. 2020, 98, 28–41. [Google Scholar] [CrossRef]
- Manning, K.S.; Lynn, D.G.; Shabanowitx, J.; Fellows, L.E.; Singh, M. A glucuronidase inhibitor from the seeds of Baphia racemosa: Application of fast atom bombardment coupled with collision activated dissociation in natural product structure assignment. J. Chem. Soc. Chem. Commun. 1985, 127–129. [Google Scholar] [CrossRef]
- Martínez, R.F.; Jenkinson, S.F.; Nakagawa, S.; Kato, A.; Wormald, M.R. Isolation from Stevia rebaudiana of DMDP acetic acid, a novel iminosugar amino acid: Synthesis and glycosidase inhibition profile of glycine and β-alanine pyrrolidine amino acids. Amino Acids 2019, 51, 991–998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleet, G.W.J.; Bashyal, B.P.; Chow, H.-F.; Fellows, L.E. The Synthesis of polyhydroxylated amino acids from glucuronolactone. Tetrahedron 1987, 43, 415–422. [Google Scholar]
- Nash, R.J.; Azantsa, B.K.G.; Sharp, H.; Shanmugham, V. Effectiveness of a Cucumis sativus extract (CSE) versus glucosamine-chondroitin in the management of osteoarthritis: A randomized controlled trial. Clin. Interv. Aging 2018, 13, 2119–2126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kothari, S.; Saravana, M.; Muthusamy, S.; Mozingo, A.; Soni, M. Safety assessment of a standardized cucumber extract (Q-Actin): Oral repeat-dose toxicity and mutagenicity studies. Toxicol. Rep. 2018, 5, 1078–1086. [Google Scholar] [CrossRef] [PubMed]
- Costa, T.; Fernandez-Villalba, E.; Izura, V.; Lucas-Ochoa, A.M.; Menezes-Filho, N.J.; Santana, R.C.; de Oliveira, M.D.; Araújo, F.M.; Estrada, C.; Silva, V.; et al. Combined 1-Deoxynojirimycin and Ibuprofen Treatment Decreases Microglial Activation, Phagocytosis and Dopaminergic Degeneration in MPTP-Treated Mice. J. Neuroimmune Pharmacol. 2021, 16, 390–402. [Google Scholar] [CrossRef] [PubMed]
- Belmatoug, N.; Burlina, A.; Giraldo, P.; Hendriksz, C.J.; Kuter, D.J.; Mengel, E.; Pastores, G.M. Gastrointestinal disturbances and their management in miglustat-treated patients. J. Inherit. Metab. Dis. 2011, 34, 991–1001. [Google Scholar] [CrossRef]
- Ahr, H.J.; Boberg, M.; Brendel, E.; Krause, H.P.; Steinke, W. Pharmacokinetics of miglitol. Absorption, distribution, metabolism, and excretion following administration to rats, dogs, and man. Arzneimittelforschung 1997, 47, 734–745. [Google Scholar]
- Saito, Y.; Miyajima, M.; Yamamoto, S.; Sato, T.; Miura, N.; Fujimiya, M.; Chikenjii, T.S. Accumulation of Senescent Neural Cells in Murine Lupus With Depression-Like Behavior. Front. Immunol. 2021, 12, 692321. [Google Scholar] [CrossRef]
- Alcalde-Estévez, E.; Arroba, A.I.; Sánchez-Fernández, E.M.; Mellet, C.O.; García Fernández, J.M.; Masgrau, L.; Valverde, Á.M. The sp2-iminosugar glycolipid 1-dodecylsulfonyl-5N,6O-oxomethylidenenojirimycin (DSO2-ONJ) as selective anti-inflammatory agent by modulation of hemeoxygenase-1 in Bv.2 microglial cells and retinal explants. Food Chem. Toxicol. 2018, 111, 454–466. [Google Scholar] [CrossRef] [Green Version]
- Miller, J.L.; Hill, M.L.; Brun, J.; Pountain, A.; Sayce, A.C.; Zitzmann, N. Iminosugars counteract the downregulation of the of the interferon gamma receptor by dengue virus. Antivir. Res. 2019, 170, 104551. [Google Scholar] [CrossRef]
- Sayce, A.C.; Martinez, F.O.; Tyrrell, B.E.; Perera, N.; Hill, M.L.; Dwek, R.A.; Miller, J.L.; Zitzmann, N. Pathogen-induced inflammation is attenuated by the iminosugar MON-DNJ via modulation of the unfolded protein response. Immunology 2021, 164, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, W.Y.; Wang, X.; Ip, N.Y. Targeting neuroinflammation as a therapeutic strategy for Alzheimer’s disease: Mechanisms, drug candidates, and new opportunities. ACS Chem. Neurosci. 2018, 10, 872–879. [Google Scholar] [CrossRef]
- Gao, H.; Wang, D.; Jiang, S.; Mao, J.; Yang, X. NF-κB is negatively associated with Nurr1 to reduce the inflammatory response in Parkinson’s disease. Mol. Med. Rep. 2021, 23, 396. [Google Scholar] [CrossRef] [PubMed]
- Bal-Price, A.; Brown, G.C. Inflammatory neurodegeneration mediated by nitric oxide from activated glia-inhibiting neuronal respiration, causing glutamate release and excitotoxicity. J. Neurosci. 2001, 31, 6480–6491. [Google Scholar] [CrossRef]
- Tobinick, E.L.; Gross, H. Rapid cognitive improvement in Alzheimer’s disease following perispinal etanercept administration. J. Neuroinflamm. 2008, 9, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waljee, A.K.; Rogers, M.A.M.; Lin, P.; Singal, A.G.; Stein, J.D. Short term use of oral corticosteroids and related harms among adults in the United States: Population based cohort study. BMJ 2017, 357, j1415. [Google Scholar] [CrossRef] [Green Version]
- Ong, C.K.S.; Lirk, P.; Tan, C.H.; Seymour, R.A. An Evidence-Based Update on Nonsteroidal Anti-Inflammatory Drugs. Clin. Med. Res. 2007, 5, 19–34. [Google Scholar] [CrossRef] [Green Version]
- van Schouwenburg, P.; Rispens, T.; Wolbink, G. Immunogenicity of anti-TNF therapies for rheumatoid therapies for rheumatoid arthritis. Nature reviews. Rheumatology 2013, 9, 164–172. [Google Scholar]
- Kim, E.K.; Choi, E.J. Compromised MAPK signaling in human diseases: An update. Arch. Toxicol. 2015, 89, 867–882. [Google Scholar] [CrossRef]
- Kheiri, G.; Dolatshahi, M.; Rahmani, F.; Rezaei, N. Role of p38/MAPKs in Alzheimer’s disease: Implications for amyloid beta toxicity targeted therapy. Rev. Neurosci. 2018, 30, 9–30. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olajide, O.A.; Iwuanyanwu, V.U.; Banjo, O.W.; Kato, A.; Penkova, Y.B.; Fleet, G.W.J.; Nash, R.J. Iminosugar Amino Acid idoBR1 Reduces Inflammatory Responses in Microglia. Molecules 2022, 27, 3342. https://doi.org/10.3390/molecules27103342
Olajide OA, Iwuanyanwu VU, Banjo OW, Kato A, Penkova YB, Fleet GWJ, Nash RJ. Iminosugar Amino Acid idoBR1 Reduces Inflammatory Responses in Microglia. Molecules. 2022; 27(10):3342. https://doi.org/10.3390/molecules27103342
Chicago/Turabian StyleOlajide, Olumayokun A., Victoria U. Iwuanyanwu, Owolabi W. Banjo, Atsushi Kato, Yana B. Penkova, George W. J. Fleet, and Robert J. Nash. 2022. "Iminosugar Amino Acid idoBR1 Reduces Inflammatory Responses in Microglia" Molecules 27, no. 10: 3342. https://doi.org/10.3390/molecules27103342
APA StyleOlajide, O. A., Iwuanyanwu, V. U., Banjo, O. W., Kato, A., Penkova, Y. B., Fleet, G. W. J., & Nash, R. J. (2022). Iminosugar Amino Acid idoBR1 Reduces Inflammatory Responses in Microglia. Molecules, 27(10), 3342. https://doi.org/10.3390/molecules27103342






