Cherries and Blueberries-Based Beverages: Functional Foods with Antidiabetic and Immune Booster Properties
Abstract
1. Introduction
2. Functional Foods Definition
| Reference | Definition |
|---|---|
| FOSHAN [19] | Foods for specified health use. The FOSHU can be foods that exhibit health effect, used as foods in a diet, and are in the form of foods, not as supplements |
| Health Canada, Ontario, Canada [20] | A functional food to be similar in appearance to conventional food, to be consumed as part of the usual diet, to demonstrate physiologic benefits, and/or to reduce the risk of chronic disease beyond basic nutritional functions. |
| International Food Information Council, Washington, USA [21] | Foods or dietary components may provide a health benefit beyond basic nutrition. |
| International Life Sciences Institute of North America (ILSI North America) [22] | Foods that by physiologically active food components provide health benefits beyond basic nutrition. |
| Regulation (EC) No 1924/2006 [18] | Functional food is a food with certain beneficial effects on one or more target functions in the body beyond the basic nutritional effects with a result of the improved health state and well-being or reduction of risk of diseases. It is consumed as a part of a normal diet and is not used in the form of a pill or capsule or any other form of dietary supplement. |
| [7] | A food product can be made functional by using any of the five approaches listed below: (1) Eliminating a component known to cause or identified as causing a deleterious effect when consumed (for example, an allergenic protein). (2) Increasing the concentration of a component naturally present in food to a point at which it will induce predicted effects (for example, fortification with a micronutrient to reach a daily intake higher than the recommended daily intake). (3) Adding a component that is not normally present in most foods and is not necessarily a macronutrient or a micronutrient, but for which beneficial effects have been shown (for example, non-vitamin antioxidant or prebiotic fructans). (4) Replacing a component, usually a macronutrient (for example, fats), intake of which is usually excessive and replacing it with a component for which beneficial effects have been shown (for example, modified starch). (5) Increasing bioavailability or stability of a component known to produce a functional effect or to reduce the disease-risk potential of the food. |
| Functional Food Center (FFC) [10] | Natural or processed foods that contain known or unknown biologically-active compounds; which, in defined, effective non-toxic amounts, provide a clinically proven and documented health benefit for the prevention, management, or treatment of chronic disease. In this definition, first functional foods can be natural or processed. Second, bioactive compounds, which are considered to be the source of the functionality of the foods, are secondary metabolites that occur in food usually in small amounts that act synergistically to benefit health. Specifically, bioactive compounds may exert antioxidant, cardio-protective and chemo-preventive effects. |
| Food and Nutrition Board (FNB) of the National Academy of Sciences, Washington, USA) [23] | Functional food is one that encompasses potentially healthful products, including any modified food or food ingredient that may provide a health benefit beyond that of the traditional nutrient it contains. |
| Bioactive Compounds: The Office of Dietary Supplements at the NIH has defined bioactive compounds as constituents in foods or dietary supplements, other than those needed to meet basic human nutritional needs, which are responsible for changes in health status [24]. |
| Dietary Supplements: Dietary supplements mean foodstuffs, the purpose of which is to supplement the normal diet, and which are concentrated sources of nutrients or other substances with a nutritional or physiological effect, alone or in combination, marketed in dose form, namely forms such as capsules, pastilles, tablets, pills and other similar forms, and sachets of powder, ampoules of liquids, drop dispensing bottles, and other similar forms of liquids and powders designed to be taken in measured small unit quantities [25]. |
| Functional Ingredients: Functional ingredients are a diverse group of compounds; health benefits have been attributed, for example, to allyl compounds found in garlic, carotenoids, and flavonoids, found in fruits and vegetables, glucosinolates, found in cruciferous vegetables, hypericin and pseudohypericin found in St. John’s wort, peptides such as epidermal growth factor, opioid peptides, and lactoferrin, found in milk, and arachidonic and docosahexaenoic acids, found in human milk and derived for use in infant formulas from various algal, bacteria, and fish sources. Functional ingredients can be marketed as part of dietary supplements, food additives, or generally recognized as safe (GRAS) ingredients included in functional foods [23]. |
| Medical Foods: A Medical Food is a food that is “formulated to be consumed or administered under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements are established by medical evaluation [26]. |
| Natural health products: Natural health products (NHPs) include homoeopathic preparations, substances used in traditional medicine, a mineral or trace element, a vitamin, an amino acid, an essential fatty acid, or other botanical-, animal-, or microorganism-derived substance [27]. |
| Nutraceutical: The term nutraceutical is a substance that may be considered a food or part of a food that provides medical or health benefits, encompassing prevention and treatment of disease. Products as diverse as isolated nutrients, dietary supplements, and diets, to genetically engineered “designer” foods, herbal products, and processed foods (cereals, soups, beverages) may be included under the umbrella of nutraceuticals [28]. |
3. Fruits and Beverages as Functional Foods
4. An Overview Regarding Cherry and Blueberry Phenolic Compounds
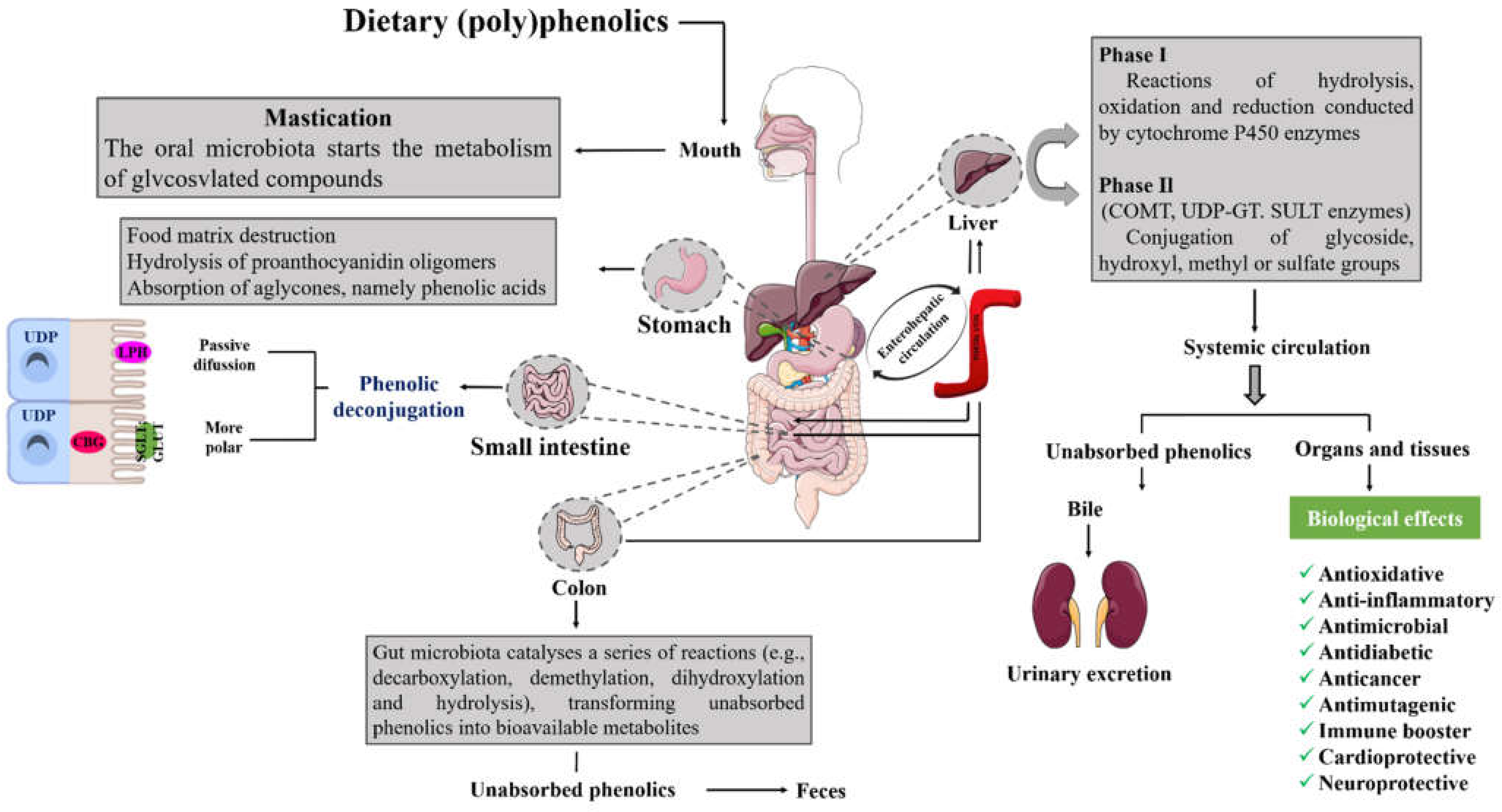
5. Absorption, Digestion, Metabolism, and Bioavailability of Phenolic Compounds
6. Functional Properties of Cherries and Blueberries—Focus on Antidiabetic and Anti-Inflammatory Potential of Phenolic Compounds
6.1. Enzymes’ Inhibition
6.2. Pancreatic β-Cells Protection
6.3. Insulin Release and Regulation
6.4. Anti-Inflammatory Properties
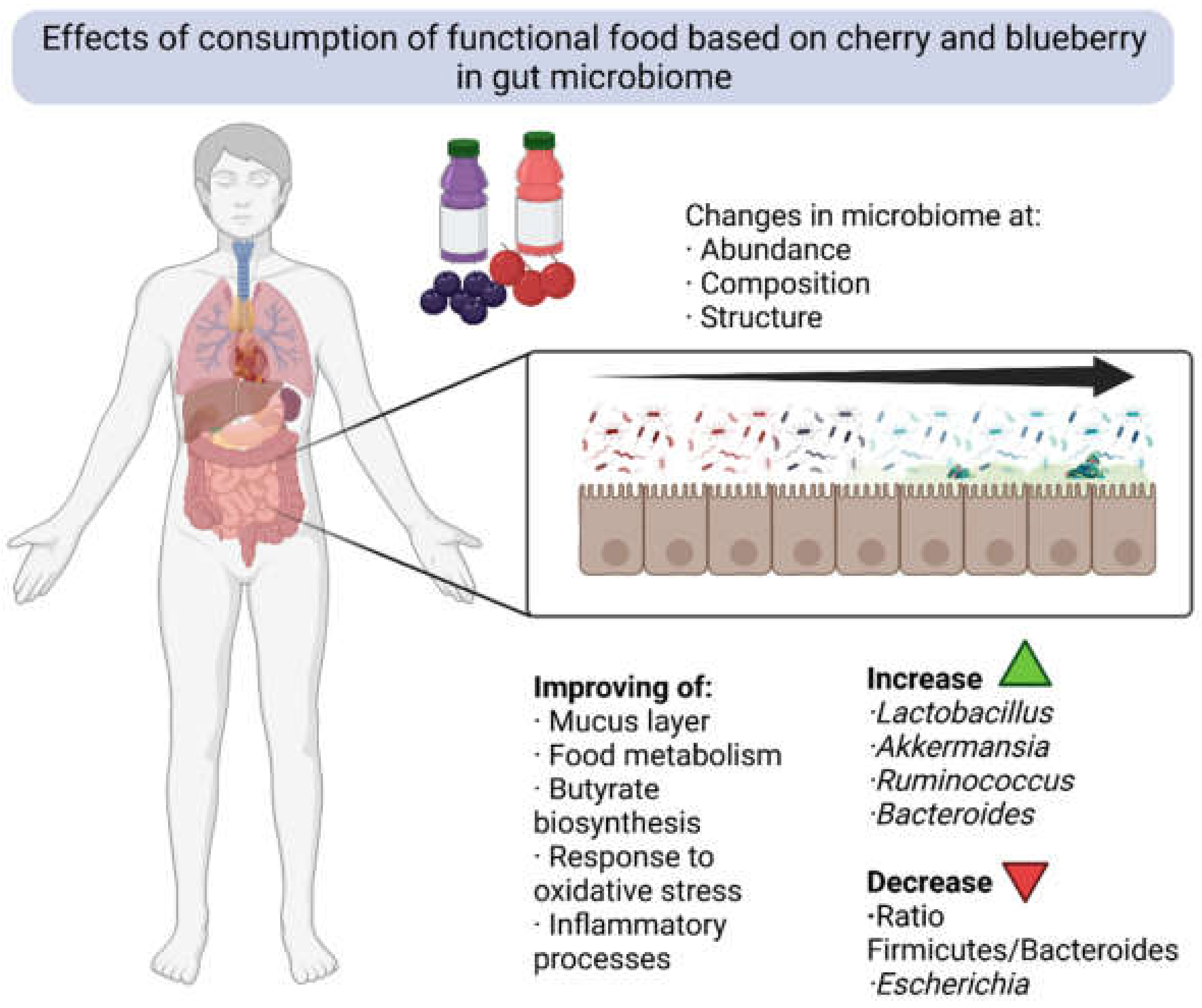
7. Impact on Gut Microbiome
8. Recent Advances and Future Perspectives and Opportunities for Functional Beverages
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Creating a Sustainable Food System; Science Advice for Policy by European Academies (SAPEA): Berlin, Germany, 2020.
- World Health Organization. Diet, Nutrition and the Prevention. Available online: http://apps.who.int/iris/bitstream/handle/10665/42665/WHO_TRS_916.pdf?sequence=1 (accessed on 8 February 2022).
- Ozen, A.E.; Pons, A.; Tur, J.A. Worldwide consumption of functional foods: A systematic review. Nutr. Rev. 2012, 70, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Sloan, A.E. Top 10 functional food trends. Food Technol. 2020, 64, 22–41. [Google Scholar]
- Dimmito, M.P.; Stefanucci, A.; Della Valle, A.; Scioli, G.; Cichelli, A.; Mollica, A. An overview on plants cannabinoids endorsed with cardiovascular effects. Biomed. Pharmacother. 2021, 142, 111963. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Das, M. Functional foods: An overview. Food Sci. Biotechnol. 2011, 20, 861–875. [Google Scholar] [CrossRef]
- Henry, C.J. Functional foods. Eur. J. Clin. Nutr. 2010, 64, 657–659. [Google Scholar] [CrossRef]
- Ohama, H.; Ikeda, H.; Moriyama, H. Health foods and foods with health claims in Japan. Toxicology 2006, 221, 95–111. [Google Scholar] [CrossRef]
- Arai, S. Studies on functional foods in Japan-state of the art. Biosci. Biotechnol. Biochem. 1996, 60, 9–15. [Google Scholar] [CrossRef]
- Martirosyan, D.M.; Singh, J. A new definition of functional food by FFC: What makes a new definition unique? Funct. Foods Health Dis. 2015, 5, 209–223. [Google Scholar] [CrossRef]
- Swinbanks, D.; O’Brien, J. Japan explores the boundary between food and medicine. Nature 1993, 364, 180. [Google Scholar] [CrossRef]
- López-Varela, S.; González-Gross, M.; Marcos, A. Functional foods and the immune system: A review. Eur. J. Clin. Nutr. 2002, 56, S29–S33. [Google Scholar] [CrossRef]
- Diplock, A.T.; Aggett, P.J.; Ashwell, M.; Bornet, F.; Fern, E.B.; Roberfroid, M.B. Scientific concepts of functional foods in Europe: Consensus document. Br. J. Nutr. 1999, 81, S1–S27. [Google Scholar]
- WHO. Europe Promoting Fruit and Vegetable Consumption. Available online: https://www.euro.who.int/en/health-topics/disease-prevention/nutrition/activities/technical-support-to-member-states/promoting-fruit-and-vegetable-consumption (accessed on 17 February 2022).
- Roberfroid, M.B. Prebiotics and probiotics: Are they functional foods? Am. J. Clin. Nutr. 2000, 71, 1682–1687. [Google Scholar] [CrossRef]
- Scioli, G.; Della, A.; Zengin, G.; Locatelli, M.; Tartaglia, A.; Cichelli, A.; Stefanucci, A.; Mollica, A. Artisanal fortified beers: Brewing, enrichment, HPLC-DAD analysis and preliminary screening of antioxidant and enzymatic inhibitory activities. Food Biosci. 2022, 48, 101721. [Google Scholar] [CrossRef]
- Government of Canada. Therapeutic Products Programme and the Food Directorate from the Health Protection Branch, Nutraceuticals/Functional Foods and Health Claims on Foods. Available online: https://www.canada.ca/en/health-canada/corporate/about-health-canada/branches-agencies/health-products-food-branch/therapeutic-products-directorate.html (accessed on 17 February 2022).
- Turck, D.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Kearney, J.; Knutsen, H.K.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Affron® and increase in positive mood: Evaluation of a health claim pursuant to Article 13 (5) of Regulation (EC) No 1924/2006. EFSA J. 2021, 19, e06660. [Google Scholar] [CrossRef]
- Shimizu, T. Health claims on functional foods: The Japanese regulations and an international comparison. Nutr. Res. Rev. 2003, 16, 241–252. [Google Scholar] [CrossRef]
- Health Canada. Standards of Evidence for Evaluating Foods with Health Claims. Available online: https://www.canada.ca/content/dam/hc-sc/migration/hc-sc/fnan/alt_formats/hpfbdgpsa/pdf/labeletiquet/consultation_doc-eng.pdf (accessed on 4 February 2022).
- Bagchi, D. Nutraceutical and Functional Food Regulations in the United States and around the World, 3rd ed.; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar] [CrossRef]
- Wildman, R.E.C.; Wildman, R.; Wallace, T.C. Handbook of Nutraceuticals and Functional Foods, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar] [CrossRef]
- Kruger, C.L.; Mann, S.W. Safety evaluation of functional ingredients. Food Chem. Toxicol. 2003, 41, 793–805. [Google Scholar] [CrossRef]
- Office of Public Health and Science; Office of the Secretary; HHS. Solicitation of written comments on proposed definition of bioactive food components. Fed. Regist. 2004, 69, 55821–55822. [Google Scholar]
- European Commission Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002. Off. J. Eur. Communities 2002, 1, 51–57.
- Steele, E.A.; Breen, C.; Campbell, E.; Martin, R. Food regulations and enforcement in the USA. Ref. Modul. Food Sci. 2016. [Google Scholar] [CrossRef]
- Fitzpatrick, K. Regulatory issues related to functional foods and natural health products in Canada. Regul. Funct. Foods Nutraceuticals A Glob. Perspect. 2007, 79, 213–226. [Google Scholar] [CrossRef]
- Dudeja, P.; Gupta, R.K. Nutraceuticals. In Food Safety 21st Century Public Health Perspective; Academic Press: Cambridge, MA, USA, 2017; pp. 491–496. [Google Scholar] [CrossRef]
- Wootton-Beard, P.C.; Ryan, L. Improving public health?: The role of antioxidant-rich fruit and vegetable beverages. Food Res. Int. 2011, 44, 3135–3148. [Google Scholar] [CrossRef]
- Pap, N.; Fidelis, M.; Azevedo, L.; Carmo, M.A.V.; Wang, D.; Mocan, A.; Pereira, E.P.R.; Xavier-Santos, D.; Sant’Ana, A.S.; Yang, B.; et al. Berry polyphenols and human health: Evidence of antioxidant, anti-inflammatory, microbiota modulation, and cell-protecting effects. Curr. Opin. Food Sci. 2021, 42, 167–186. [Google Scholar] [CrossRef]
- Bashir, K.M.I.; Choi, J.S. Molecular sciences clinical and physiological perspectives of β-glucans: The past, present, and future. Int. J. Mol. Sci. 2017, 18, 1906. [Google Scholar] [CrossRef]
- Neves, M.F.; Trombin, V.G.; Marques, V.N.; Martinez, L.F. Global orange juice market: A 16-year summary and opportunities for creating value. Trop. Plant Pathol. 2020, 45, 166–174. [Google Scholar] [CrossRef]
- Wilkinson, J.; Hall, M. Novel food approvals in Europe: Routes to obtaining regulatory approval for nutraceuticals in the EU. Nutraceutical Bus. Technol. 2008, 4, 12–14. [Google Scholar] [CrossRef]
- European Parliament and of the Council. Regulation 258/97. Off. J. Eur. Communities 1997, 14, 1–9. [Google Scholar]
- Serafini, M.; Stanzione, A.; Foddai, S. Functional foods: Traditional use and European legislation. Int. J. Food Sci. Nutr. 2012, 63, 7–9. [Google Scholar] [CrossRef]
- Lenssen, K.G.M.; Bast, A.; de Boer, A. Clarifying the health claim assessment procedure of EFSA will benefit functional food innovation. J. Funct. Foods 2018, 47, 386–396. [Google Scholar] [CrossRef]
- Corbo, M.R.; Bevilacqua, A.; Petruzzi, L.; Casanova, F.P.; Sinigaglia, M. Functional beverages: The emerging side of functional foods commercial trends, research, and health implications. Comp. Rev. Food Sci. Food Saf. 2014, 13, 1192–1206. [Google Scholar] [CrossRef]
- Pico, J.; Yan, Y.; Gerbrandt, E.M.; Castellarin, S.D. Determination of free and bound phenolics in northern highbush blueberries by a validated HPLC/QTOF methodology. J. Food Compos. Anal. 2022, 108, 104412. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Bento, C.; Silva, B.; Simões, M.; Silva, L.R. Nutrients, bioactive compounds and bioactivity: The health benefits of sweet cherries (Prunus avium L.). Curr. Nutr. Food Sci. 2019, 15, 208–227. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Campos, G.; Alves, G.; Garcia-Viguera, C.; Moreno, D.A.; Silva, L.R. Physical and phytochemical composition of 23 Portuguese sweet cherries as conditioned by variety (or genotype). Food Chem. 2021, 335, 127637. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.R.; Queiroz, M. Bioactive compounds of red grapes from Dão region (Portugal): Evaluation of phenolic and organic profile. Asian Pac. J. Trop. Biomed. 2016, 6, 315–321. [Google Scholar] [CrossRef]
- Randenkovs, V.; Pussa, T.; Juhnevica-Radenkova, K.; Kviesis, J.; Slar, F.J.; Moreno, D.A.; Drudze, I. Wild apple (Malus spp.) by-products as a source of phenolic compounds and vitamin C for food applications. Food Biosci. 2020, 38, 100744. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Bento, C.; Silva, B.M.; Silva, L.R. Sweet cherries from Fundão possess antidiabetic potential and protect human erythrocytes against oxidative damage. Food Res. Int. 2017, 95, 91–100. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Bento, C.; Jesus, F.; Alves, G.; Silva, L.R. Sweet cherry phenolic compounds: Identification, characterization, and health benefits. In Studies in Natural Products Chemistry; Atta-ur-Rahman, F., Ed.; Science Publishers: Amsterdam, The Netherlands, 2018; pp. 31–78. ISBN 9780444641793. [Google Scholar]
- Zhang, Y.; Liu, W.; Wei, Z.; Yin, B.; Man, C.; Jiang, Y. Enhancement of functional characteristics of blueberry juice fermented by Lactobacillus plantarum. LWT 2021, 139, 110590. [Google Scholar] [CrossRef]
- Giovanelli, G.; Buratti, S. Comparison of polyphenolic composition and antioxidant activity of wild Italian blueberries and some cultivated varieties. Food Chem. 2009, 112, 903–908. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, C.Y.; Wang, S.Y.; Zheng, W. Effect of high-oxygen atmospheres on blueberry phenolics, anthocyanins, and antioxidant capacity. J. Agric. Food Chem. 2003, 51, 7162–7169. [Google Scholar] [CrossRef]
- Nunes, S.; Viana, S.D.; Preguiça, I.; Alves, A.; Fernandes, R.; Teodoro, J.S.; Matos, P.; Figueirinha, A.; Salgueiro, L.; André, A.; et al. Blueberry counteracts prediabetes in a hypercaloric diet-induced rat model and rescues hepatic mitochondrial bioenergetics. Nutrients 2021, 13, 4192. [Google Scholar] [CrossRef]
- Nunes, S.; Viana, S.; Rolo, A.; Palmeira, C.; André, A.; Cavadas, C.; Pintado, M.; Reis, F. Blueberry juice as a nutraceutical approach to prevent prediabetes progression in an animal model: Focus on hepatic steatosis. Eur. J. Public Health 2019, 29, ckz034-011. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Antonio, A.L.; Oliveira, M.B.P.P.; Ferreira, I.C.F.R. A comparative study between natural and synthetic antioxidants: Evaluation of their performance after incorporation into biscuits. Food Chem. 2017, 216, 342–346. [Google Scholar] [CrossRef]
- Becker Pertuzatti, P.; Teixeira Barcia, M.; Gómez-Alonso, S.; Teixeira Godoy, H.; Hermosin-Gutierrez, I. Phenolics profiling by HPLC-DAD-ESI-MSn aided by principal component analysis to classify Rabbiteye and Highbush blueberries. Food Chem. 2021, 340, 127958. [Google Scholar] [CrossRef]
- Das, P.R.; Darwish, A.G.; Ismail, A.; Haikal, A.M.; Gajjar, P.; Balasubramani, S.P.; Sheikh, M.B.; Tsolova, V.; Soliman, K.F.A.; Sherif, S.M.; et al. Diversity in blueberry genotypes and developmental stages enables discrepancy in the bioactive compounds, metabolites, and cytotoxicity. Food Chem. 2022, 374, 131632. [Google Scholar] [CrossRef]
- Picariello, G.; De Vito, V.; Ferranti, P.; Paolucci, M.; Volpe, M.G. Species- and cultivar-dependent traits of Prunus avium and Prunus cerasus polyphenols. J. Food Compos. Anal. 2016, 45, 50–57. [Google Scholar] [CrossRef]
- Kim, D.O.; Ho, J.H.; Young, J.K.; Hyun, S.Y.; Lee, C.Y. Sweet and sour cherry phenolics and their protective effects on neuronal cells. J. Agric. Food Chem. 2005, 53, 9921–9927. [Google Scholar] [CrossRef]
- Pissard, A.; Lateur, M.; Baeten, V.; Magein, H.; Dupont, P.; Tabart, J.; Pincemail, J.; Kevers, C. Determination of total phenolic compound content and antioxidant activity in cherry species and cultivars. J. Berry Res. 2016, 6, 81–91. [Google Scholar] [CrossRef]
- Okur, İ.; Baltacıoğlu, C.; Ağçam, E.; Baltacıoğlu, H.; Alpas, H. Evaluation of the effect of different extraction techniques on sour cherry pomace phenolic content and antioxidant activity and determination of phenolic compounds by FTIR and HPLC. Waste Biomass Valoriz. 2019, 10, 3545–3555. [Google Scholar] [CrossRef]
- Ceccarelli, D.; Talento, C.; Favale, S.; Caboni, E.; Cecchini, F. Phenolic compound profile characterization by Q-TOF LC/MS in 12 Italian ancient sweet cherry cultivars. Plant Biosyst. 2018, 152, 1346–1353. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Rodrigues, M.; Flores-Félix, J.D.; Nunes, A.R.; Ribeiro, A.B.; Alves, G. Sweet cherry phenolics revealed to be promising agents in inhibiting P-glycoprotein activity and increasing cellular viability under oxidative stress conditions: In vitro and in silico study. J. Food Sci. 2021, 87, 450–465. [Google Scholar] [CrossRef]
- Bunea, A.; Ruginǎ, D.O.; Pintea, A.M.; Sconţa, Z.; Bunea, C.I.; Socaciu, C. Comparative polyphenolic content and antioxidant activities of some wild and cultivated blueberries from Romania. Not. Bot. Horti Agrobot. Cluj-Napoca 2011, 39, 70–76. [Google Scholar] [CrossRef]
- Guofang, X.; Xiaoyan, X.; Xiaoli, Z.; Yongling, L.; Zhibing, Z. Changes in phenolic profiles and antioxidant activity in rabbiteye blueberries during ripening. Int. J. Food Prop. 2019, 22, 320–329. [Google Scholar] [CrossRef]
- Schüller, E.; Halbwirth, H.; Mikulic-Petkovsek, M.; Slatnar, A.; Veberic, R.; Forneck, A.; Stich, K.; Spornberger, A. High concentrations of anthocyanins in genuine cherry-juice of old local Austrian Prunus avium varieties. Food Chem. 2015, 173, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Ocampo, E.; Torrejón-Valqui, L.; Muñóz-Astecker, L.D.; Medina-Mendoza, M.; Mori-Mestanza, D.; Castro-Alayo, E.M. Antioxidant capacity, total phenolic content and phenolic compounds of pulp and bagasse of four Peruvian berries. Heliyon 2021, 7, e07787. [Google Scholar] [CrossRef] [PubMed]
- Subbiah, V.; Zhong, B.; Nawaz, M.A.; Barrow, C.J.; Dunshea, F.R.; Suleria, H.A.R. Screening of phenolic compounds in Australian grown berries by LC-ESI-QTOF-MS/MS and determination of their antioxidant potential. Antioxidants 2021, 10, 26. [Google Scholar] [CrossRef]
- Sellappan, S.; Akoh, C.C.; Krewer, G. Phenolic compounds and antioxidant capacity of Georgia-grown blueberries and blackberries. J. Agric. Food Chem. 2002, 50, 2432–2438. [Google Scholar] [CrossRef]
- Picariello, G.; Ferranti, P.; De Cunzo, F.; Sacco, E.; Volpe, M.G. Polyphenol patterns to trace sweet (Prunus avium) and tart (Prunus cerasus) varieties in cherry jam. J. Food Sci. Technol. 2017, 54, 2316–2323. [Google Scholar] [CrossRef]
- Martín-Gómez, J.; Varo, M.Á.; Mérida, J.; Serratosa, M.P. Influence of drying processes on anthocyanin profiles, total phenolic compounds and antioxidant activities of blueberry (Vaccinium corymbosum). LWT 2020, 120, 108931. [Google Scholar] [CrossRef]
- Bento, C.; Gonçalves, A.C.; Jesus, F.; Simões, M.; Silva, L.R. Phenolic compounds: Sources, properties and applications. In Bioactive Compounds: Sources, Properties and Applications; Porter, R., Parker, N., Eds.; Nova Publishers: New York, NY, USA, 2017; pp. 271–299. ISBN 6312317269. [Google Scholar]
- Homoki, J.R.; Nemes, A.; Fazekas, E.; Gyémánt, G.; Balogh, P.; Gál, F.; Al-Asri, J.; Mortier, J.; Wolber, G.; Babinszky, L.; et al. Anthocyanin composition, antioxidant efficiency, and α-amylase inhibitor activity of different Hungarian sour cherry varieties (Prunus cerasus L.). Food Chem. 2016, 194, 222–229. [Google Scholar] [CrossRef]
- Kim, J.G.; Kim, H.L.; Kim, S.J.; Park, K.S. Fruit quality, anthocyanin and total phenolic contents, and antioxidant activities of 45 blueberry cultivars grown in Suwon, Korea. J. Zhejiang Univ. Sci. B 2013, 14, 793–799. [Google Scholar] [CrossRef]
- Dóka, O.; Ficzek, G.; Bicanic, D.; Spruijt, R.; Luterotti, S.; Tóth, M.; Buijnsters, J.G.; Végvári, G. Direct photothermal techniques for rapid quantification of total anthocyanin content in sour cherry cultivars. Talanta 2011, 84, 341–346. [Google Scholar] [CrossRef]
- Rodrigues, E.; Poerner, N.; Rockenbach, I.I.; Gonzaga, L.V.; Mendes, C.R.; Fett, R. Phenolic compounds and antioxidant activity of blueberry cultivars grown in Brazil. Food Sci. Technol. 2011, 31, 911–917. [Google Scholar] [CrossRef]
- Kirakosyan, A.; Seymour, E.M.; Llanes, D.E.U.; Kaufman, P.B.; Bolling, S.F. Chemical profile and antioxidant capacities of tart cherry products. Food Chem. 2009, 115, 20–25. [Google Scholar] [CrossRef]
- Blando, F.; Gerardi, C.; Nicoletti, I. Sour cherry (Prunus cerasus L) anthocyanins as ingredients for functional foods. J. Biomed. Biotechnol. 2004, 2004, 253–258. [Google Scholar] [CrossRef]
- Sokół-Łe̜towska, A.; Kucharska, A.Z.; Hodun, G.; Gołba, M. Chemical composition of 21 cultivars of sour cherry (Prunus cerasus) fruit cultivated in Poland. Molecules 2020, 25, 4587. [Google Scholar] [CrossRef]
- Ficzek, G.; Végvári, G.; Sándor, G.; Stéger-Máté, M.; Kállay, E.; Szügyi, S.; Tóth, M. HPLC evaluation of anthocyanin components in the fruits of Hungarian sour cherry cultivars during ripening. J. Food, Agric. Environ. 2011, 9, 30–35. [Google Scholar]
- Martini, S.; Conte, A.; Tagliazucchi, D. Phenolic compounds profile and antioxidant properties of six sweet cherry (Prunus avium) cultivars. Food Res. Int. 2017, 97, 15–26. [Google Scholar] [CrossRef]
- Gu, C.; Howell, K.; Dunshea, F.R.; Suleria, H.A.R. LC-ESI-QTOF/MS characterisation of phenolic acids and flavonoids in polyphenol-rich fruits and vegetables and their potential antioxidant activities. Antioxidants 2019, 8, 405. [Google Scholar] [CrossRef]
- Hayaloglu, A.A.; Demir, N. Phenolic compounds, volatiles, and sensory characteristics of twelve sweet cherry (Prunus avium L.) cultivars grown in Turkey. J. Food Sci. 2016, 81, C7–C18. [Google Scholar] [CrossRef]
- González-Gómez, D.; Lozano, M.; Fernández-León, M.F.; Bernalte, M.J.; Ayuso, M.C.; Rodríguez, A.B. Sweet cherry phytochemicals: Identification and characterization by HPLC-DAD/ESI-MS in six sweet-cherry cultivars grown in Valle del Jerte (Spain). J. Food Compos. Anal. 2010, 23, 533–539. [Google Scholar] [CrossRef]
- Kader, F.; Rovel, B.; Girardin, M.; Metche, M. Fractionation and identification of the phenolic compounds of Highbush blueberries (Vaccinium corymbosum, L.). Food Chem. 1996, 55, 35–40. [Google Scholar] [CrossRef]
- Gavrilova, V.; Kajdžanoska, M.; Gjamovski, V.; Stefova, M. Separation, characterization and quantification of phenolic compounds in blueberries and red and black currants by HPLC-DAD-ESI-MSn. J. Agric. Food Chem. 2011, 59, 4009–4018. [Google Scholar] [CrossRef]
- Brito, A.; Areche, C.; Sepúlveda, B.; Kennelly, E.J.; Simirgiotis, M.J. Anthocyanin characterization, total phenolic quantification and antioxidant features of some chilean edible berry extracts. Molecules 2014, 19, 10936–10955. [Google Scholar] [CrossRef]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef]
- Lohachoompol, V.; Mulholland, M.; Srzednicki, G.; Craske, J. Determination of anthocyanins in various cultivars of highbush and rabbiteye blueberries. Food Chem. 2008, 111, 249–254. [Google Scholar] [CrossRef]
- Xu, W.; Zhou, Q.; Yao, Y.; Li, X.; Zhang, J.L.; Su, G.; Deng, A.P. Inhibitory effect of Gardenblue blueberry (Vaccinium ashei Reade) anthocyanin extracts on lipopolysaccharide-stimulated inflammatory response in RAW 264.7 cells. J. Zhejiang Univ. Sci. B 2016, 17, 425–436. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture. USDA. Available online: https://fdc.nal.usda.gov/ (accessed on 20 March 2022).
- Damar, I.; Ekşi, A. Antioxidant capacity and anthocyanin profile of sour cherry (Prunus cerasus L.) juice. Food Chem. 2012, 135, 2910–2914. [Google Scholar] [CrossRef]
- Wojdyło, A.; Nowicka, P.; Teleszko, M. Degradation kinetics of anthocyanins in sour cherry cloudy juices at different storage temperature. Processes 2019, 7, 367. [Google Scholar] [CrossRef]
- Pataro, G.; Carullo, D.; Bobinaite, R.; Donsì, G.; Ferrari, G. Improving the extraction yield of juice and bioactive compounds from sweet cherries and their by-products by pulsed electric fields. Chem. Eng. Trans. 2017, 57, 1717–1722. [Google Scholar] [CrossRef]
- Jia, C.; Waterhouse, G.I.N.; Sun-Waterhouse, D.; Sun, Y.G.; Wu, P. Variety–compound–quality relationship of 12 sweet cherry varieties by HPLC-chemometric analysis. Int. J. Food Sci. Technol. 2019, 54, 2897–2914. [Google Scholar] [CrossRef]
- Müller, D.; Schantz, M.; Richling, E. High performance liquid chromatography analysis of anthocyanins in bilberries (Vaccinium myrtillus L.), blueberries (Vaccinium corymbosum L.), and corresponding juices. J. Food Sci. 2012, 77, C340–C345. [Google Scholar] [CrossRef]
- Reque, P.M.; Steffens, R.S.; Jablonski, A.; Flôres, S.H.; Rios, A.D.O.; Jong, E.V. Cold storage of blueberry (Vaccinium spp.) fruits and juice: Anthocyanin stability and antioxidant activity. J. Food Compos. Anal. 2014, 33, 111–116. [Google Scholar] [CrossRef]
- Prior, R.L.; Wilkes, S.E.; ROgers, T.; Khanal, R.C.; Wu, X.; Howard, L.R. Purified blueberry anthocyanins and blueberry juice alter development of obesity in mice fed an obesogenic high-fat diet. J. Agric. Food Chem. 2010, 58, 3970–3976. [Google Scholar] [CrossRef] [PubMed]
- Akšić, M.F.; Zagorac, D.D.; Sredojević, M.; Milivojević, J.; Gašić, U.; Meland, M.; Natić, M. Chemometric characterization of strawberries and blueberries according to their phenolic profile: Combined effect of cultivar and cultivation system. Molecules 2019, 24, 4310. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Impact of juice processing on blueberry anthocyanins and polyphenolics: Comparison of two pretreatments. J. Food Sci. 2002, 67, 1660–1667. [Google Scholar] [CrossRef]
- Ockun, M.A.; Gercek, Y.C.; Demirsoy, H.; Demirsoy, L.; Macit, I.; Oz, G.C. Comparative evaluation of phenolic profile and antioxidant activity of new sweet cherry (Prunus avium L.) genotypes in Turkey. Phytochem. Anal. 2022. Epub ahead of print. [Google Scholar] [CrossRef]
- Budak, N.H. Bioactive components of Prunus avium L. black gold (red cherry) and Prunus avium L. stark gold (white cherry) juices, wines and vinegars. J. Food Sci. Technol. 2017, 54, 62–70. [Google Scholar] [CrossRef]
- Gonçalves, J.; Ramos, R.; Rosado, T.; Gallardo, E.; Duarte, A.P. Development and validation of a HPLC–DAD method for quantification of phenolic compounds in different sweet cherry cultivars. SN Appl. Sci. 2019, 1, 954. [Google Scholar] [CrossRef]
- Levaj, B.; Dragović-Uzelac, V.; Delonga, K.; Ganić, K.K.; Banović, M.; Kovačević, D.B. Polyphenols and volatiles in fruits of two sour cherry cultivars, some berry fruits and their jams. Food Technol. Biotechnol. 2010, 48, 538–547. [Google Scholar]
- Will, F.; Hilsendegen, P.; Bonerz, D.; Patz, C.D.; Dietrich, H. Analytical composition of fruit juices from different sour cherry cultivars. J. Appl. Bot. Food Qual. 2005, 79, 12–16. [Google Scholar]
- Pimpão, R.C.; Dew, T.; Oliveira, P.B.; Williamson, G.; Ferreira, R.B.; Santos, C.N. Analysis of phenolic compounds in portuguese wild and commercial berries after multienzyme hydrolysis. J. Agric. Food Chem. 2013, 61, 4053–4062. [Google Scholar] [CrossRef]
- De Leo, M.; Iannuzzi, A.M.; Germanò, M.P.; D’Angelo, V.; Camangi, F.; Sevi, F.; Diretto, G.; De Tommasi, N.; Braca, A. Comparative chemical analysis of six ancient italian sweet cherry (Prunus avium L.) varieties showing antiangiogenic activity. Food Chem. 2021, 360, 129999. [Google Scholar] [CrossRef]
- Ciric, A.; Jelikic-Stankov, M.; Cvijovic, M.; Djurdjevic, P. Statistical optimization of an RP-HPLC method for the determination of selected flavonoids in berry juices and evaluation of their antioxidant activities. Biomed. Chromatogr. 2018, 32, e4150. [Google Scholar] [CrossRef]
- Kang, J.Y.; Park, S.K.; Guo, T.J.; Ha, J.S.; Lee, D.S.; Kim, J.M.; Lee, U.; Kim, D.O.; Heo, H.J. Reversal of trimethyltin-induced learning and memory deficits by 3,5-dicaffeoylquinic acid. Oxid. Med. Cell. Longev. 2016, 2016, 6981595. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, Q.; Chen, X.-Y.; Li, X.; Wang, Y.; Zhang, J.L. Comparison and screening of bioactive phenolic compounds in different blueberry cultivars: Evaluation of anti-oxidation and α-glucosidase inhibition effect. Food Res. Int. 2017, 100, 312–324. [Google Scholar] [CrossRef]
- Hu, T.; Subbiah, V.; Wu, H.; BK, A.; Rauf, A.; Alhumaydhi, F.A.; Suleria, H.A.R. Determination and characterization of phenolic compounds from Australia-grown sweet cherries (Prunus avium L.) and their potential antioxidant properties. ACS Omega 2021, 6, 34687–34699. [Google Scholar] [CrossRef]
- Zhu, N.; Zhu, Y.; Yu, N.; Wei, Y.; Zhang, J.; Hou, Y.; Sun, A.D. Evaluation of microbial, physicochemical parameters and flavor of blueberry juice after microchip-pulsed electric field. Food Chem. 2019, 274, 146–155. [Google Scholar] [CrossRef]
- Reque, P.M.; Steffens, R.S.; Da Silva, A.M.; Jablonski, A.; Flôres, S.H.; Rios, A.D.O.; Jong, E.D.V. Characterization of blueberry fruits (Vaccinium spp.) and derived products. Food Sci. Technol. 2015, 34, 773–779. [Google Scholar] [CrossRef]
- Özen, M.; Özdemir, N.; Ertekin Filiz, B.; Budak, N.H.; Kök-Taş, T. Sour cherry (Prunus cerasus L.) vinegars produced from fresh fruit or juice concentrate: Bioactive compounds, volatile aroma compounds and antioxidant capacities. Food Chem. 2020, 309, 125664. [Google Scholar] [CrossRef]
- Tokatlı, K.; Demirdöven, A. Influences of chitosan coatings on functional compounds of sweet cherries. J. Food Sci. Technol. 2021, 58, 1808–1818. [Google Scholar] [CrossRef]
- Filannino, P.; Bai, Y.; Di Cagno, R.; Gobbetti, M.; Gänzle, M.G. Metabolism of phenolic compounds by Lactobacillus spp. during fermentation of cherry juice and broccoli puree. Food Microbiol. 2015, 46, 272–279. [Google Scholar] [CrossRef]
- Ricci, A.; Cirlini, M.; Maoloni, A.; Del Rio, D.; Calani, L.; Bernini, V.; Galaverna, G.; Neviani, E.; Lazzi, C. Use of dairy and plant-derived lactobacilli as starters for cherry juice fermentation. Nutrients 2019, 11, 213. [Google Scholar] [CrossRef]
- Manquián-Cerda, K.; Cruces, E.; Escudey, M.; Zúñiga, G.; Calderón, R. Interactive effects of aluminum and cadmium on phenolic compounds, antioxidant enzyme activity and oxidative stress in blueberry (Vaccinium corymbosum L.) plantlets cultivated in vitro. Ecotoxicol. Environ. Saf. 2018, 150, 320–326. [Google Scholar] [CrossRef]
- Howard, L.R.; Brownmiller, C.; Mauromoustakos, A.; Prior, R.L. Improved stability of blueberry juice anthocyanins by acidification and refrigeration. J. Berry Res. 2016, 6, 189–201. [Google Scholar] [CrossRef]
- Martín-Gómez, J.; García-Martínez, T.; Varo, M.Á.; Mérida, J.; Serratosa, M.P. Phenolic compounds, antioxidant activity and color in the fermentation of mixed blueberry and grape juice with different yeasts. LWT 2021, 146, 111661. [Google Scholar] [CrossRef]
- Xu, F.; Liu, S.; Liu, Y.; Wang, S. Effect of mechanical vibration on postharvest quality and volatile compounds of blueberry fruit. Food Chem. 2021, 349, 129216. [Google Scholar] [CrossRef]
- Wu, Y.; Han, Y.; Tao, Y.; Li, D.; Xie, G.; Show, P.L.; Lee, S.Y. In vitro gastrointestinal digestion and fecal fermentation reveal the effect of different encapsulation materials on the release, degradation and modulation of gut microbiota of blueberry anthocyanin extract. Food Res. Int. 2020, 132, 109098. [Google Scholar] [CrossRef]
- Bresciani, L.; Martini, D.; Mena, P.; Tassotti, M.; Calani, L.; Brigati, G.; Brighenti, F.; Holasek, S.; Malliga, D.E.; Lamprecht, M.; et al. Absorption profile of (poly)phenolic compounds after consumption of three food supplements containing 36 different fruits, vegetables, and berries. Nutrients 2017, 9, 194. [Google Scholar] [CrossRef]
- Serra, D.; Henriques, J.F.; Serra, T.; Silva, A.B.; Bronze, M.R.; Dinis, T.C.P.; Almeida, L.M. An anthocyanin-rich extract obtained from portuguese blueberries maintains its efficacy in reducing microglia-driven neuroinflammation after simulated digestion. Nutrients 2020, 12, 3670. [Google Scholar] [CrossRef]
- Zhong, S.; Sandhu, A.; Edirisinghe, I.; Burton-Freeman, B. Characterization of wild blueberry polyphenols bioavailability and kinetic profile in plasma over 24-h period in human subjects. Mol. Nutr. Food Res. 2017, 61, 1700405. [Google Scholar] [CrossRef]
- Hollman, P.C.H.; Cassidy, A.; Comte, B.; Heinonen, M.; Richelle, M.; Richling, E.; Serafini, M.; Scalbert, A.; Sies, H.; Vidry, S. The biological relevance of direct antioxidant effects of polyphenols for cardiovascular health in humans is not established. J. Nutr. 2011, 141, 989–1009. [Google Scholar] [CrossRef]
- Martini, S.; Conte, A.; Tagliazucchi, D. Bioactivity and cell metabolism of in vitro digested sweet cherry (Prunus avium) phenolic compounds. Int. J. Food Sci. Nutr. 2019, 70, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A. Bioavailability of dietary anthocyanins and hydroxycinnamic acids. In Polyphenols in Human Health and Disease; Watson, R.R., Preedy, V.R., Zibadi, S.B.T.-P., Eds.; Academic Press: San Diego, CA, USA, 2014; pp. 561–576. ISBN 978-0-12-398456-2. [Google Scholar]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.C.; Nunes, A.R.; Falcão, A.; Alves, G.; Silva, L.R. Dietary effects of anthocyanins in human health: A comprehensive review. Pharmaceuticals 2021, 14, 690. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T. Dietary factors affecting polyphenol bioavailability. Nutr. Rev. 2014, 72, 429–452. [Google Scholar] [CrossRef]
- Oksuz, T.; Tacer-Caba, Z.; Nilufer-Erdil, D.; Boyacioglu, D. Changes in bioavailability of sour cherry (Prunus cerasus L.) phenolics and anthocyanins when consumed with dairy food matrices. J. Food Sci. Technol. 2019, 56, 4177–4188. [Google Scholar] [CrossRef]
- Dudonné, S.; Dal-Pan, A.; Dubé, P.; Varin, T.V.; Calon, F.; Desjardins, Y. Potentiation of the bioavailability of blueberry phenolic compounds by co-ingested grape phenolic compounds in mice, revealed by targeted metabolomic profiling in plasma and feces. Food Funct. 2016, 7, 3421–3430. [Google Scholar] [CrossRef]
- Carbonell-Capella, J.M.; Buniowska, M.; Barba, F.J.; Esteve, M.J.; Frígola, A. Analytical methods for determining bioavailability and bioaccessibility of bioactive compounds from fruits and vegetables: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 155–171. [Google Scholar] [CrossRef]
- Palafox-Carlos, H.; Ayala-Zavala, J.F.; González-Aguilar, G.A. The role of dietary fiber in the bioaccessibility and bioavailability of fruit and vegetable antioxidants. J. Food Sci. 2011, 76, R6–R15. [Google Scholar] [CrossRef]
- Mueller, D.; Jung, K.; Winter, M.; Rogoll, D.; Melcher, R.; Richling, E. Human intervention study to investigate the intestinal accessibility and bioavailability of anthocyanins from bilberries. Food Chem. 2017, 231, 275–286. [Google Scholar] [CrossRef]
- Cosme, P.; Rodríguez, A.B.; Espino, J.; Garrido, M. Plant phenolics: Bioavailability as a key determinant of their potential health-promoting applications. Antioxidants 2020, 9, 1263. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Fazzari, M.; Fukumoto, L.; Mazza, G.; Livrea, M.A.; Tesoriere, L.; Di Marco, L. In vitro bioavailability of phenolic compounds from five cultivars of frozen sweet cherries (Prunus avium L.). J. Agric. Food Chem. 2008, 56, 3561–3568. [Google Scholar] [CrossRef]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The reciprocal interactions between polyphenols and gut microbiota and effects on bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef]
- Ou, K.; Gu, L. Absorption and metabolism of proanthocyanidins. J. Funct. Foods 2014, 7, 43–53. [Google Scholar] [CrossRef]
- Feliciano, R.P.; Istas, G.; Heiss, C.; Rodriguez-Mateos, A. Plasma and urinary phenolic profiles after acute and repetitive intake of wild blueberry. Molecules 2016, 21, 14–16. [Google Scholar] [CrossRef]
- Caleja, C.; Ribeiro, A.; Barreiro, M.F.; Ferreira, I.C.F.R. Phenolic compounds as nutraceuticals or functional food ingredients. Curr. Pharm. Des. 2017, 23, 2787–2806. [Google Scholar] [CrossRef]
- Mihaylova, D.; Desseva, I.; Stoyanova, M.; Petkova, N.; Terzyiska, M.; Lante, A. Impact of in vitro gastrointestinal digestion on the bioaccessibility of phytochemical compounds from eight fruit juices. Molecules 2021, 26, 1187. [Google Scholar] [CrossRef]
- Frank, T.; Netzel, M.; Strass, G.; Bitsch, R.; Bitsch, I. Bioavailability of anthocyanidin-3-glucosides following consumption of red wine and red grape juice. Can. J. Physiol. Pharmacol. 2003, 81, 423–435. [Google Scholar] [CrossRef]
- Zang, Z.; Chou, S.; Si, X.; Cui, H.; Tan, H.; Ding, Y.; Liu, Z.; Wang, H.; Lang, Y.; Tang, S.; et al. Effect of bovine serum albumin on the stability and antioxidant activity of blueberry anthocyanins during processing and in vitro simulated digestion. Food Chem. 2022, 373, 131496. [Google Scholar] [CrossRef]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019, 157, 107843. [Google Scholar] [CrossRef]
- De Paulo Farias, D.; de Araújo, F.F.; Neri-Numa, I.A.; Pastore, G.M. Antidiabetic potential of dietary polyphenols: A mechanistic review. Food Res. Int. 2021, 145, 110383. [Google Scholar] [CrossRef]
- Halim, M.; Halim, A. The effects of inflammation, aging and oxidative stress on the pathogenesis of diabetes mellitus (type 2 diabetes). Diabetes Metab. Syndr. 2019, 13, 1165–1172. [Google Scholar] [CrossRef]
- Ghorbani, A.; Rashidi, R.; Shafiee-Nick, R. Flavonoids for preserving pancreatic beta cell survival and function: A mechanistic review. Biomed. Pharmacother. 2019, 111, 947–957. [Google Scholar] [CrossRef]
- Nunes, A.R.; Gonçalves, A.C.; Falcão, A.; Alves, G.; Silva, L.R. Prunus avium L. (sweet cherry) by-Ppoducts: A source of phenolic compounds with antioxidant and anti-hyperglycemic properties—A review. Appl. Sci. 2021, 11, 8516. [Google Scholar] [CrossRef]
- Cao, H.; Ou, J.; Chen, L.; Zhang, Y.; Szkudelski, T.; Delmas, D.; Daglia, M.; Xiao, J. Dietary polyphenols and type 2 diabetes: Human study and clinical trials. Crit. Rev. Food Sci. Nutr. 2019, 59, 3371–3379. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional Foods and lifestyle approaches for diabetes prevention and management. Nutrients 2017, 9, 1310. [Google Scholar] [CrossRef] [PubMed]
- Maya-Cano, D.A.; Arango-Varela, S.; Santa-Gonzalez, G.A. Phenolic compounds of blueberries (Vaccinium spp) as a protective strategy against skin cell damage induced by ROS: A review of antioxidant potential and antiproliferative capacity. Heliyon 2021, 7, e06297. [Google Scholar] [CrossRef]
- Michalska, A.; Łysiak, G. Bioactive compounds of blueberries: Post-harvest factors influencing the nutritional value of products. Int. J. Mol. Sci. 2015, 16, 18642–18663. [Google Scholar] [CrossRef]
- Serrano, M.; Díaz-Mula, H.M.; Zapata, P.J.; Castillo, S.; Guillén, F.; Martínez-Romero, D.; Valverde, J.M.; Valero, D. Maturity stage at harvest determines the fruit quality and antioxidant potential after storage of sweet cherry cultivars. J. Agric. Food Chem. 2009, 57, 3240–3246. [Google Scholar] [CrossRef]
- Bastos, C.; Barros, L.; Duenas, M.; Calhelha, R.C.; Queiroz, M.J.R.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Chemical characterisation and bioactive properties of Prunus avium L.: The widely studied fruits and the unexplored stems. Food Chem. 2015, 173, 1045–1053. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, H.; Liu, W.; Li, C. Survey of antioxidant capacity and phenolic composition of blueberry, blackberry, and strawberry in Nanjing. J. Zhejiang Univ. Sci. B 2012, 13, 94–102. [Google Scholar] [CrossRef]
- Dias, T.R.; Bernardino, R.L.; Meneses, M.J.; Sousa, M.; Sa, R.; Alves, M.G.; Silva, B.M.; Oliveira, P.F. Emerging potential of natural products as an alternative strategy to pharmacological agents used against metabolic disorders. Curr. Drug Metab. 2016, 17, 582–597. [Google Scholar] [CrossRef]
- Chen, L.; Gnanaraj, C.; Arulselvan, P.; El-Seedi, H.; Teng, H. A review on advanced microencapsulation technology to enhance bioavailability of phenolic compounds: Based on its activity in the treatment of type 2 diabetes. Trends Food Sci. Technol. 2019, 85, 149–162. [Google Scholar] [CrossRef]
- Ji, Y.; Liu, D.; Jin, Y.; Zhao, J.; Zhao, J.; Li, H.; Li, L.; Zhang, H.; Wang, H. In vitro and in vivo inhibitory effect of anthocyanin-rich bilberry extract on α-glucosidase and α-amylase. LWT 2021, 145, 111484. [Google Scholar] [CrossRef]
- Johnson, M.H.; Lucius, A.; Meyer, T.; de Mejia, E.G. Cultivar evaluation and effect of fermentation on antioxidant capacity and in vitro inhibition of α-amylase and α-glucosidase by highbush blueberry (Vaccinium corombosum). J. Agric. Food Chem. 2011, 59, 8923–8930. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Rodrigues, M.; Santos, A.O.; Alves, G.; Silva, L.R. Antioxidant status, antidiabetic properties and effects on Caco-2 cells of colored and non-colored enriched extracts of sweet cherry fruits. Nutrients 2018, 10, 1688. [Google Scholar] [CrossRef]
- Jesus, F.; Gonçalves, A.C.; Alves, G.; Silva, L.R. Exploring the phenolic profile, antioxidant, antidiabetic and anti-hemolytic potential of Prunus avium vegetal parts. Food Res. Int. 2018, 116, 600–610. [Google Scholar] [CrossRef]
- Fan, J.; Johnson, M.H.; Lila, M.A.; Yousef, G.; Mejia, E.G. Berry and citrus phenolic compounds inhibit dipeptidyl peptidase IV: Implications in diabetes management. Evid.-Based Complement. Altern. Med. 2013, 2013, 479505. [Google Scholar] [CrossRef]
- Rouse, M.; Younès, A.; Egan, J.M. Resveratrol and curcumin enhance pancreatic β-cell function by inhibiting phosphodiesterase activity. J. Endocrinol. 2014, 223, 107–117. [Google Scholar] [CrossRef]
- Lachin, T.; Reza, H. Anti diabetic effect of cherries in alloxan induced diabetic rats. Recent Pat. Endocr. Metab. Immune Drug Discov. 2012, 6, 67–72. [Google Scholar] [CrossRef]
- Li, H.; Park, H.M.; Ji, H.S.; Han, J.; Kim, S.K.; Park, H.Y.; Jeong, T.S. Phenolic-enriched blueberry-leaf extract attenuates glucose homeostasis, pancreatic β-cell function, and insulin sensitivity in high-fat diet-induced diabetic mice. Nutr. Res. 2020, 73, 83–96. [Google Scholar] [CrossRef]
- Li, D.; Zhang, Y.; Liu, Y.; Sun, R.; Xia, M. Purified anthocyanin supplementation reduces dyslipidemia, enhances antioxidant capacity, and prevents insulin resistance in diabetic patients. J. Nutr. 2015, 145, 742–748. [Google Scholar] [CrossRef]
- Cao, J.; Li, X.; Liu, Y.; Leng, F.; Li, X.; Sun, C.; Chen, K. Bioassay-based isolation and identification of phenolics from sweet cherry that promote active glucose consumption by HepG2 cells. J. Food Sci. 2015, 80, C234–C240. [Google Scholar] [CrossRef]
- Liu, W.; Mao, Y.; Schoenborn, J.; Wang, Z.; Tang, G.; Tang, X. Whole blueberry protects pancreatic beta-cells in diet-induced obese mouse. Nutr. Metab. 2019, 16, 34. [Google Scholar] [CrossRef]
- Stull, A.J.; Cash, K.C.; Johnson, W.D.; Champagne, C.M.; Cefalu, W.T. Bioactives in blueberries improve insulin sensitivity in obese, insulin-resistant men and women. J. Nutr. 2010, 140, 1764–1768. [Google Scholar] [CrossRef]
- Luo, G.; Xiao, L.; Wang, D.; Wang, N.; Luo, C.; Yang, X.; Hao, L. Resveratrol protects against ethanol-induced impairment of insulin secretion in INS-1 cells through SIRT1-UCP2 axis. Toxicol. In Vitro 2020, 65, 104808. [Google Scholar] [CrossRef]
- Lou, H.; Yao, J.; Sun, Y.; Sun, H.; Song, Z.; Li, H.; Wang, X.; Liu, K.; Liu, X.; Li, Z. Role of blueberry anthocyanin extract in the expression of SIRT1 and NF-κB in rat lens epithelial cells in experimentally induced DM. Curr. Eye Res. 2021, 46, 45–51. [Google Scholar] [CrossRef]
- Johnson, M.H.; de Mejia, E.G. Phenolic compounds from fermented berry beverages modulated gene and protein expression to increase insulin secretion from pancreatic β-cells in vitro. J. Agric. Food Chem. 2016, 64, 2569–2581. [Google Scholar] [CrossRef]
- Bibi Sadeer, N.; Llorent-Martínez, E.J.; Bene, K.; Fawzi Mahomoodally, M.; Mollica, A.; Ibrahime Sinan, K.; Stefanucci, A.; Ruiz-Riaguas, A.; Fernández-de Córdova, M.L.; Zengin, G. Chemical profiling, antioxidant, enzyme inhibitory and molecular modelling studies on the leaves and stem bark extracts of three African medicinal plants. J. Pharm. Biomed. Anal. 2019, 174, 19–33. [Google Scholar] [CrossRef]
- Akkarachiyasit, S.; Yibchok-Anun, S.; Wacharasindhu, S.; Adisakwattana, S. In vitro inhibitory effects of cyanidin-3-rutinoside on pancreatic α-amylase and its combined effect with acarbose. Molecules 2011, 16, 2075–2083. [Google Scholar] [CrossRef]
- Kahn, S.E. The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 2003, 46, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Semwal, D.K.; Kumar, A.; Aswal, S.; Chauhan, A.; Semwal, R.B. Protective and therapeutic effects of natural products against diabetes mellitus via regenerating pancreatic β-cells and restoring their dysfunction. Phytother. Res. 2021, 35, 1218–1229. [Google Scholar] [CrossRef] [PubMed]
- Copps, K.D.; White, M.F. Regulation of insulin sensitivity by serine/threonine phosphorylation of insulin receptor substrate proteins IRS1 and IRS2. Diabetologia 2012, 55, 2565–2582. [Google Scholar] [CrossRef] [PubMed]
- Faienza, M.F.; Corbo, F.; Carocci, A.; Catalano, A.; Clodoveo, M.L.; Grano, M.; Wang, D.Q.-H.; D’Amato, G.; Muraglia, M.; Franchini, C.; et al. Novel insights in health-promoting properties of sweet cherries. J. Funct. Foods 2020, 69, 103945. [Google Scholar] [CrossRef] [PubMed]
- Ambriz-Pérez, D.L.; Leyva-López, N.; Gutierrez-Grijalva, E.P.; Heredia, J.B. Phenolic compounds: Natural alternative in inflammation treatment. A Review. Cogent Food Agric. 2016, 2, 1131412. [Google Scholar] [CrossRef]
- Kelley, D.S.; Rasooly, R.; Jacob, R.A.; Kader, A.A.; Mackey, B.E. Consumption of Bing sweet cherries lowers circulating concentrations of inflammation markers in healthy men and women. J. Nutr. 2006, 136, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Delgado, J.; del Pilar Terrón, M.; Garrido, M.; Barriga, C.; Espino, J.; Paredes, S.D.; Rodríguez, A.B. Jerte Valley cherry-based product modulates serum inflammatory markers in rats and ringdoves. J. Appl. Biomed. 2012, 10, 41–50. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Costa, A.R.; Flores-Félix, J.D.; Alves, G.; Silva, L.R. Anti-inflammatory and antiproliferative properties of sweet cherry phenolic-rich extracts. Molecules 2022, 27, 268. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Momin, R.A.; Nair, M.G.; Bourquin, L.D. Cyclooxygenase inhibitory and antioxidant cyanidin glycosides in cherries and berries. Phytomedicine 2001, 8, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Cutler, B.R.; Gholami, S.; Chua, J.S.; Kuberan, B.; Anandh Babu, P.V. Blueberry metabolites restore cell surface glycosaminoglycans and attenuate endothelial inflammation in diabetic human aortic endothelial cells. Int. J. Cardiol. 2018, 261, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Esposito, D.; Chen, A.; Grace, M.H.; Komarnytsky, S.; Lila, M.A. Inhibitory effects of wild blueberry anthocyanins and other flavonoids on biomarkers of acute and chronic inflammation in vitro. J. Agric. Food Chem. 2014, 62, 7022–7028. [Google Scholar] [CrossRef]
- Li, J.; Deng, R.; Hua, X.; Zhang, L.; Lu, F.; Coursey, T.G.; Pflugfelder, S.C.; Li, D.-Q. Blueberry component pterostilbene protects corneal epithelial cells from inflammation via anti-oxidative Pathway. Sci. Rep. 2016, 6, 19408. [Google Scholar] [CrossRef]
- Basu, A.; Du, M.; Leyva, M.J.; Sanchez, K.; Betts, N.M.; Wu, M.; Aston, C.E.; Lyons, T.J. Blueberries decrease cardiovascular risk factors in obese men and women with metabolic syndrome. J. Nutr. 2010, 140, 1582–1587. [Google Scholar] [CrossRef]
- McLeay, Y.; Barnes, M.J.; Mundel, T.; Hurst, S.M.; Hurst, R.D.; Stannard, S.R. Effect of New Zealand blueberry consumption on recovery from eccentric exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2012, 9, 19. [Google Scholar] [CrossRef]
- Casertano, M.; Fogliano, V.; Ercolini, D. Psychobiotics, gut microbiota and fermented foods can help preserving mental health. Food Res. Int. 2022, 152, 110892. [Google Scholar] [CrossRef]
- Tosti, V.; Bertozzi, B.; Fontana, L. Health benefits of the mediterranean diet: Metabolic and molecular mechanisms. J. Gerontol. Ser. A 2018, 73, 318–326. [Google Scholar] [CrossRef]
- Terpou, A.; Papadaki, A.; Lappa, I.K.; Kachrimanidou, V.; Bosnea, L.A.; Kopsahelis, N. Probiotics in food systems: Significance and emerging strategies towards improved viability and delivery of enhanced beneficial value. Nutrients 2019, 11, 1591. [Google Scholar] [CrossRef]
- Khairudin, M.A.S.; Jalil, A.M.M.; Hussin, N. Effects of polyphenols in tea (Camellia sinensis sp.) on the modulation of gut microbiota in human trials and animal studies. Gastroenterol. Insights 2021, 12, 202–216. [Google Scholar] [CrossRef]
- Thilakarathna, W.W.; Langille, M.G.; Rupasinghe, H.V. Polyphenol-based prebiotics and synbiotics: Potential for cancer chemoprevention. Curr. Opin. Food Sci. 2018, 20, 51–57. [Google Scholar] [CrossRef]
- Burillo, S.P.; Ángel, J.; Henares, R.; Ignacio Martínez-Montoro, J.; Quesada-Molina, M.; Gutiérrez-Repiso, C.; Ruiz-Limón, P.; Subiri-Verdugo, A.; Tinahones, F.J.; Moreno-Indias, I. Effect of moderate consumption of different phenolic-content beers on the human gut microbiota composition: A randomized crossover trial. Antioxidants 2022, 11, 696. [Google Scholar] [CrossRef]
- Nash, V.; Ranadheera, C.S.; Georgousopoulou, E.N.; Mellor, D.D.; Panagiotakos, D.B.; McKune, A.J.; Kellett, J.; Naumovski, N. The effects of grape and red wine polyphenols on gut microbiota—A systematic review. Food Res. Int. 2018, 113, 277–287. [Google Scholar] [CrossRef]
- Mayta-Apaza, A.C.; Pottgen, E.; De Bodt, J.; Papp, N.; Marasini, D.; Howard, L.; Abranko, L.; Van de Wiele, T.; Lee, S.O.; Carbonero, F. Impact of tart cherries polyphenols on the human gut microbiota and phenolic metabolites in vitro and in vivo. J. Nutr. Biochem. 2018, 59, 160–172. [Google Scholar] [CrossRef]
- Groh, I.A.M.; Riva, A.; Braun, D.; Sutherland, H.G.; Williams, O.; Bakuradze, T.; Pahlke, G.; Richling, E.; Haupt, L.M.; Griffiths, L.R.; et al. Long-term consumption of anthocyanin-rich fruit juice: Impact on gut microbiota and antioxidant markers in lymphocytes of healthy males. Antioxidants 2021, 10, 27. [Google Scholar] [CrossRef]
- Al Othaim, A.; Marasini, D.; Carbonero, F. Impact of increasing concentration of tart and sweet cherries juices concentrates on healthy mice gut microbiota. Food Front. 2020, 1, 224–233. [Google Scholar] [CrossRef]
- Barroso, E.; Muñoz-González, I.; Jiménez, E.; Bartolomé, B.; Moreno-Arribas, M.V.; Peláez, C.; del Carmen Martínez-Cuesta, M.; Requena, T. Phylogenetic profile of gut microbiota in healthy adults after moderate intake of red wine. Mol. Nutr. Food Res. 2017, 61, 1600620. [Google Scholar] [CrossRef]
- Quesada-Molina, M.; Muñoz-Garach, A.; Tinahones, F.J.; Moreno-Indias, I. A New Perspective on the health benefits of moderate beer consumption: Involvement of the gut microbiota. Metabolites 2019, 9, 272. [Google Scholar] [CrossRef]
- Vendrame, S.; Guglielmetti, S.; Riso, P.; Arioli, S.; Klimis-Zacas, D.; Porrini, M. Six-week consumption of a wild blueberry powder drink increases Bifidobacteria in the human gut. J. Agric. Food Chem. 2011, 59, 12815–12820. [Google Scholar] [CrossRef]
- Valero-Cases, E.; Cerdá-Bernad, D.; Pastor, J.J.; Frutos, M.J. Non-dairy fermented beverages as potential carriers to ensure probiotics, prebiotics, and bioactive compounds arrival to the gut and their health benefits. Nutrients 2020, 12, 1666. [Google Scholar] [CrossRef]
- Hu, N.; Lei, M.; Zhao, X.; Zhang, Z.; Gu, Y.; Zhang, Y.; Wang, S. Analysis of the microbial diversity and characteristics of fermented blueberry beverages from different regions. Foods 2020, 9, 1656. [Google Scholar] [CrossRef]
- Silva, V.L.M.; Silva, A.C.O.; Costa-Lima, B.R.C.; Carneiro, C.S.; Conte-Junior, C.A. Stability of polyphenols from blueberry (Vaccinium corymbosum L.) in fermented dairy beverage. J. Food Process. Preserv. 2017, 41, e13305. [Google Scholar] [CrossRef]
- Mauro, C.S.I.; Guergoletto, K.B.; Garcia, S. Development of blueberry and carrot juice blend fermented by Lactobacillus reuteri LR92. Beverages 2016, 2, 37. [Google Scholar] [CrossRef]
- Cheng, Y.; Wu, T.; Chu, X.; Tang, S.; Cao, W.; Liang, F.; Fang, Y.; Pan, S.; Xu, X. Fermented blueberry pomace with antioxidant properties improves fecal microbiota community structure and short chain fatty acids production in an in vitro mode. LWT 2020, 125, 109260. [Google Scholar] [CrossRef]
- Kung, S.; Hintze, K.; Ward, R. Effect of a high flavonoid supplement on intestinal inflammation, short chain fatty acids, and the gut microbiome. Curr. Dev. Nutr. 2020, 4, 420. [Google Scholar] [CrossRef]
- Zhong, H.; Deng, L.; Zhao, M.; Tang, J.; Liu, T.; Zhang, H.; Feng, F. Probiotic-fermented blueberry juice prevents obesity and hyperglycemia in high fat diet-fed mice in association with modulating the gut microbiota. Food Funct. 2020, 11, 9192–9207. [Google Scholar] [CrossRef]
- Vergari, F.; Tibuzzi, A.; Basile, G. An overview of the functional food market: From marketing issues and commercial players to future demand from life in space. Adv. Exp. Med. Biol. 2010, 698, 308–321. [Google Scholar] [CrossRef]
- Granato, D.; Branco, G.F.; Nazzaro, F.; Cruz, A.G.; Faria, J.A.F. Functional foods and nondairy probiotic food development: Trends, concepts, and products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 292–302. [Google Scholar] [CrossRef]
- Wallace, T.C.; Bailey, R.L.; Blumberg, J.B.; Burton-Freeman, B.; Chen, C.-Y.O.; Crowe-White, K.M.; Drewnowski, A.; Hooshmand, S.; Johnson, E.; Lewis, R.; et al. Fruits, vegetables, and health: A comprehensive narrative, umbrella review of the science and recommendations for enhanced public policy to improve intake. Crit. Rev. Food Sci. Nutr. 2020, 60, 2174–2211. [Google Scholar] [CrossRef]
- Nazir, M.; Arif, S.; Khan, R.S.; Nazir, W.; Khalid, N. Opportunities and challenges for functional and medicinal beverages: Current and future trends. Trends Food Sci. Technol. 2019, 88, 513–526. [Google Scholar] [CrossRef]
- Sun-Waterhouse, D. The development of fruit-based functional foods targeting the health and wellness market: A review. Int. J. Food Sci. Technol. 2011, 46, 899–920. [Google Scholar] [CrossRef]
- Nayak, S.N.; Aravind, B.; Malavalli, S.S.; Sukanth, B.S.; Poornima, R.; Bharati, P.; Hefferon, K.; Kole, C.; Puppala, N. Omics Technologies to enhance plant based functional foods: An overview. Front. Genet. 2021, 12, 742095. [Google Scholar] [CrossRef]
- Richardson, D.P.; Affertsholt, T.; Asp, N.-G.; Bruce, A.; Grossklaus, R.; Howlett, J.; Pannemans, D.; Ross, R.; Verhagen, H.; Viechtbauer, V. PASSCLAIM—Synthesis and review of existing processes. Eur. J. Nutr. 2003, 42 (Suppl. S1), I96–I111. [Google Scholar] [CrossRef]
- Annunziata, A.; Vecchio, R. Consumer perception of functional foods: A conjoint analysis with probiotics. Food Qual. Prefer. 2013, 28, 348–355. [Google Scholar] [CrossRef]
- Skąpska, S.; Marszałek, K.; Woźniak, L.; Szczepańska, J.; Danielczuk, J.; Zawada2, K. The development and consumer acceptance of functional fruit-herbal beverages. Foods 2020, 9, 1819. [Google Scholar] [CrossRef]
- Paquin, P. (Ed.) Functional and Speciality Beverage Technology, 1st ed.; Woodhead Publishing Limited: Cambridge, UK, 2009; ISBN 978-1-84569-342-8. [Google Scholar]
- Bigliardi, B.; Galati, F. Innovation trends in the food industry: The case of functional foods. Trends Food Sci. Technol. 2013, 31, 118–129. [Google Scholar] [CrossRef]
- Bagchi, D.; Nair, S. Developing New Functional Food and Nutraceutical Products, 1st ed.; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; da Silva Pinto, M. Bioavailability of bioactive food compounds: A challenging journey to bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602. [Google Scholar] [CrossRef]
- Galanakis, C.M. Functionality of food components and emerging technologies. Foods 2021, 10, 128. [Google Scholar] [CrossRef]
- Ofori, J.A.; Peggy, Y.H. Novel technologies for the production of functional foods. In Bio-Nanotechnology: A Revolution in Food, Biomedical and Health Sciences, 1st ed.; Wiley-Blackwell: Chichester, UK, 2013; pp. 141–162. [Google Scholar]
- Kumar, A.; Mosa, K.A.; Ji, L.; Kage, U.; Dhokane, D.; Karre, S.; Madalageri, D.; Pathania, N. Metabolomics-assisted biotechnological interventions for developing plant-based functional foods and nutraceuticals. Crit. Rev. Food Sci. Nutr. 2018, 58, 1791–1807. [Google Scholar] [CrossRef]
- Gomes, J.; Barbosa, J.; Teixeira, P. Natural antimicrobial agents as an alternative to chemical antimicrobials in the safety and preservation of food products. Curr. Chem. Biol. 2019, 13, 25–37. [Google Scholar] [CrossRef]
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Pérez-Gregorio, R. Phenolic compounds and functional beverages. Beverages 2021, 7, 71. [Google Scholar] [CrossRef]
- Maatallah, S.; Dabbou, S.; Castagna, A.; Guizani, M.; Hajlaoui, H.; Ranieri, A.M.; Flamini, G. Prunus persica by-products: A source of minerals, phenols and volatile compounds. Sci. Hortic. 2020, 261, 109016. [Google Scholar] [CrossRef]
- Nunes, A.R.; Gonçalves, A.C.; Alves, G.; Falcão, A.; García-Viguera, C.; Moreno, D.A.; Silva, L.R. Valorisation of Prunus avium L. by-products: Phenolic composition and effect on Caco-2 cells viability. Foods 2021, 10, 1185. [Google Scholar] [CrossRef]
- Iriondo-DeHond, M.; Miguel, E.; Del Castillo, M.D. Food byproducts as sustainable ingredients for innovative and healthy dairy foods. Nutrients 2018, 10, 1358. [Google Scholar] [CrossRef]
|
| Functional foods are: |
|
| Category | Example |
|---|---|
| Basic food | Carrots (containing the antioxidant β-carotene); Turmeric (containing curcumin); Grapes (containing resveratrol) |
| Processed foods | Oat bran cereal |
| Processed foods with added ingredients | Calcium-enriched fruit juice; margarine enriched in phytosterols; Beverages enriched with vitamins and minerals |
| Food enhanced to have more of a functional component | Tomatoes with a higher level of lycopene |
| Isolated, purified preparations of active food ingredients (dosage form) | Isoflavones from soy β-Glucan from oat bran Anthocyanins from red fruits |
| Fruits | Juices | |||||||
|---|---|---|---|---|---|---|---|---|
| Tart Cherries | Sweet Cherries | Blueberries | Tart Cherries | Sweet Cherries | Blueberries | References | ||
| Basic chemical composition | ||||||||
| Water (g per 100 g) | 86.1 | 82.2 | 84.2 | 85.2 | 85.0 | 89.7 | [86] | |
| Energy (kcal per 100 g) | 50.0 | 63.0 | 57.0 | 59.0 | 54.0 | 37.0 | ||
| Energy (kJ per 100 g) | 209.0 | 263.0 | 240.0 | 248.0 | 226.0 | - | ||
| Macronutrients | ||||||||
| Total protein (g per 100 g) | 1.0 | 1.1 | 0.74 | 0.31 | 0.91 | 0.48 | [86] | |
| Betaine (mg per 100 g) | - | - | 0.20 | - | - | - | ||
| Tryptophan (mg per 100 g) | - | 0.009 | 0.030 | - | - | - | ||
| Threonine (mg per 100 g) | - | 0.022 | 0.020 | - | - | - | ||
| Isoleucine (mg per 100 g) | - | 0.020 | 0.023 | - | - | - | ||
| Leucine (mg per 100 g) | - | 0.030 | 0.044 | - | - | - | ||
| Lysine (mg per 100 g) | - | 0.032 | 0.013 | - | - | - | ||
| Methionine (mg per 100 g) | - | 0.010 | 0.012 | - | - | - | ||
| Cystine (mg per 100 g) | - | 0.010 | 0.008 | - | - | - | ||
| Phenylalanine (mg per 100 g) | - | 0.024 | 0.026 | - | - | - | ||
| Tyrosine (mg per 100 g) | - | 0.014 | 0.009 | - | - | - | ||
| Valine (mg per 100 g) | - | 0.024 | 0.031 | - | - | - | ||
| Arginine (mg per 100 g) | - | 0.018 | 0.037 | - | - | - | ||
| Histidine (mg per 100 g) | - | 0.015 | 0.011 | - | - | - | ||
| Alanine (mg per 100 g) | - | 0.026 | 0.031 | - | - | - | ||
| Aspartic acid (mg per 100 g) | - | 0.569 | 0.057 | - | - | - | ||
| Glutamic acid (mg per 100 g) | - | 0.083 | 0.091 | - | - | - | ||
| Glycine (mg per 100 g) | - | 0.023 | 0.031 | - | - | - | ||
| Proline (mg per 100 g) | - | 0.039 | 0.028 | - | - | - | ||
| Serine (mg per 100 g) | - | 0.03 | 0.022 | - | - | - | ||
| Total lipids (g per 100 g) | 0.3 | 0.2 | 0.33 | 0.54 | 0.02 | 0.21 | ||
| Fatty acids, total saturated (g per 100 g) | 0.068 | 0.038 | 0.028 | - | 0.004 | 0.018 | ||
| SFA 14:0 (g per 100 g) | 0.002 | 0.001 | - | - | - | - | ||
| SFA 16:0 (g per 100 g) | 0.048 | 0.027 | 0.017 | - | 0.003 | 0.011 | ||
| SFA 18:0 (g per 100 g) | 0.016 | 0.009 | 0.005 | - | 0.001 | 0.003 | ||
| Fatty acids, total monounsaturated (g per 100 g) | 0.082 | 0.047 | 0.047 | - | 0.005 | 0.031 | ||
| MUFA 16:1 (g per 100 g) | 0.001 | 0.001 | 0.002 | - | - | 0.001 | ||
| MUFA 18:1 (g per 100 g) | 0.081 | 0.047 | 0.047 | - | 0.005 | 0.031 | ||
| Fatty acids, total polyunsaturated (g per 100 g) | 0.09 | 0.052 | 0.146 | - | 0.006 | 0.095 | ||
| PUFA 18:2 (g per 100 g) | 0.046 | 0.027 | 0.088 | - | 0.003 | 0.057 | ||
| PUFA 18:3 (g per 100 g) | 0.044 | 0.026 | 0.058 | - | 0.003 | 0.038 | ||
| Carbohydrates (g per 100 g) (by difference) | 12.2 | 16 | 14.5 | 13.7 | 13.8 | 9.42 | ||
| Total ash (g per 100 g) | 0.4 | 0.48 | 0.24 | 0.28 | 0.031 | - | ||
| Total dietary fiber (g per 100 g) | 1.6 | 2.1 | 2.4 | - | 1.5 | 1.6 | ||
| Total sugars (g per 100 g) | 8.49 | 12.8 | 9.96 | 12.2 | 12.3 | 6.47 | ||
| Fructose (g per 100 g) | 3.51 | 5.37 | 4.97 | 4.95 | - | - | ||
| Glucose (g per 100 g) | 4.18 | 6.59 | 4.88 | 7.26 | - | - | ||
| Sucrose (g per 100 g) | 0.8 | 0.15 | 0.11 | - | - | - | ||
| Lactose (g per 100 g) | - | - | - | - | - | - | ||
| Maltose (g per 100 g) | - | 0.12 | - | - | - | - | ||
| Galactose (g per 100 g) | - | 0.59 | - | - | - | - | ||
| Starch (g per 100 g) | - | 0 | 0.03 | - | - | - | ||
| Micronutrients | ||||||||
| Minerals | ||||||||
| Calcium (mg per 100 g) | 16.0 | 13.0 | 6.0 | 13.0 | 14.0 | 5.0 | [86] | |
| Iron (mg per 100 g) | 0.32 | 0.36 | 0.28 | 0.42 | 0.58 | 0.18 | ||
| Magnesium (mg per 100 g) | 9.0 | 11.0 | 6.0 | 11.0 | 12.0 | 4.0 | ||
| Phosphorus (mg per 100 g) | 15.0 | 21.0 | 12.0 | 17.0 | 22.0 | 8.0 | ||
| Potassium (mg per 100 g) | 173.0 | 222.0 | 77.0 | 161 | 131.0 | 50.0 | ||
| Sodium (mg per 100 g) | 3.0 | - | 1.0 | 4.0 | 3.0 | 2.0 | ||
| Zinc (mg per 100 g) | 0.1 | 0.07 | 0.16 | 0.03 | 0.1 | 0.1 | ||
| Cooper (mg per 100 g) | 0.104 | 0.06 | 0.057 | 0.042 | 0.073 | 0.04 | ||
| Manganese (mg per 100 g) | 0.112 | 0.07 | 0.336 | 0.06 | 0.061 | |||
| Fluoride (μg per 100 g) | - | 2.0 | - | - | - | - | ||
| Selenium (μg per 100 g) | - | - | 0.1 | - | 0 | 0.1 | ||
| Vitamins | [86] | |||||||
| Vitamin C (mg per 100 g) | 10.0 | 7.0 | 9.7 | - | 2.5 | 6.3 | ||
| Thiamin (mg per 100 g) | 0.03 | 0.027 | 0.037 | 0.06 | 0.018 | 0.024 | ||
| Riboflavin (mg per 100 g) | 0.04 | 0.033 | 0.041 | - | 0.024 | 0.027 | ||
| Niacin (mg per 100 g) | 0.4 | 0.154 | 0.418 | - | 0.406 | 0.272 | ||
| Pantothenic acid (mg per 100 g) | 0.143 | 0.199 | 0.124 | - | 0.127 | |||
| Vitamin B6 (mg per 100 g) | 0.044 | 0.049 | 0.052 | 0.037 | 0.03 | 0.034 | ||
| Folate, total (μg per 100 g) | 8.0 | 4.0 | 6.0 | - | 4.0 | 4.0 | ||
| Folate, DFE (μg per 100 g) | 8.0 | 4.0 | 6.0 | - | 4.0 | 4.0 | ||
| Folate, food (μg per 100 g) | 8.0 | 4.0 | 6.0 | - | 4.0 | 4.0 | ||
| Choline (mg per 100 g) | 6.1 | 6.1 | 6.0 | - | 4.7 | 3.9 | ||
| Vitamin A, RAE (μg per 100 g) | 64.0 | 3.0 | 3.0 | - | 6.0 | 2.0 | ||
| Vitamin A, IU (IU per 100 g) | 1280.0 | 64.0 | 54.0 | - | 125 | - | ||
| Vitamin D (D2 + D3) IU (IU per 100 g) | - | 64.0 | - | - | - | - | ||
| β-Carotene (μg per 100 g) | 770.0 | 38.0 | 32.0 | - | 75.0 | 21.0 | ||
| Lutein + zeaxanthin (μg per 100 g) | 85.0 | 85.0 | 80.0 | - | 57.0 | 52.0 | ||
| Vitamin E (mg per 100 g) | 0.07 | 0.07 | 0.57 | - | 0.23 | - | ||
| β-Tocopherol (mg per 100 g) | - | 0.01 | 0.01 | - | - | - | ||
| γ-Tocopherol (mg per 100 g) | - | 0.04 | 0.36 | - | - | - | ||
| Δ-Tocopherol (mg per 100 g) | - | - | 0.03 | - | - | - | ||
| γ-Tocotrienol (mg per 100 g) | - | 0.04 | 0.07 | - | - | - | ||
| Vitamin K (μg per 100 g) | 2.1 | 2.1 | 19.3 | - | 1.4 | 12.5 | ||
| Phenolic Profile | ||||||||
| Cherries | Blueberries | Juices | ||||||
| Tart Cherries | Sweet Cherries | Highbush | Rabbiteye | Tart Cherries | Sweet Cherries | Blueberries | ||
| TPC (mg GAE per 100 g fw) | 275.3–652.27 | 28.3–493.6 | 2.7–585.3 | 390.0–2625.0 | 1510.0–2550.0 a | 582.7–4757.9 a | 1.65 | [53,55,56,57,58,59,60,61,62,63,64,65,66,87] |
| TAC (mg C3G per 100 g fw) | 15.5–295.0 | 3.7–98.4 | 34.5–552.2 | 69.97–378.31 | 553.0 a | 85.1–1095.9 a | 29.00–32.73 a | [53,54,64,68,69,70,71] |
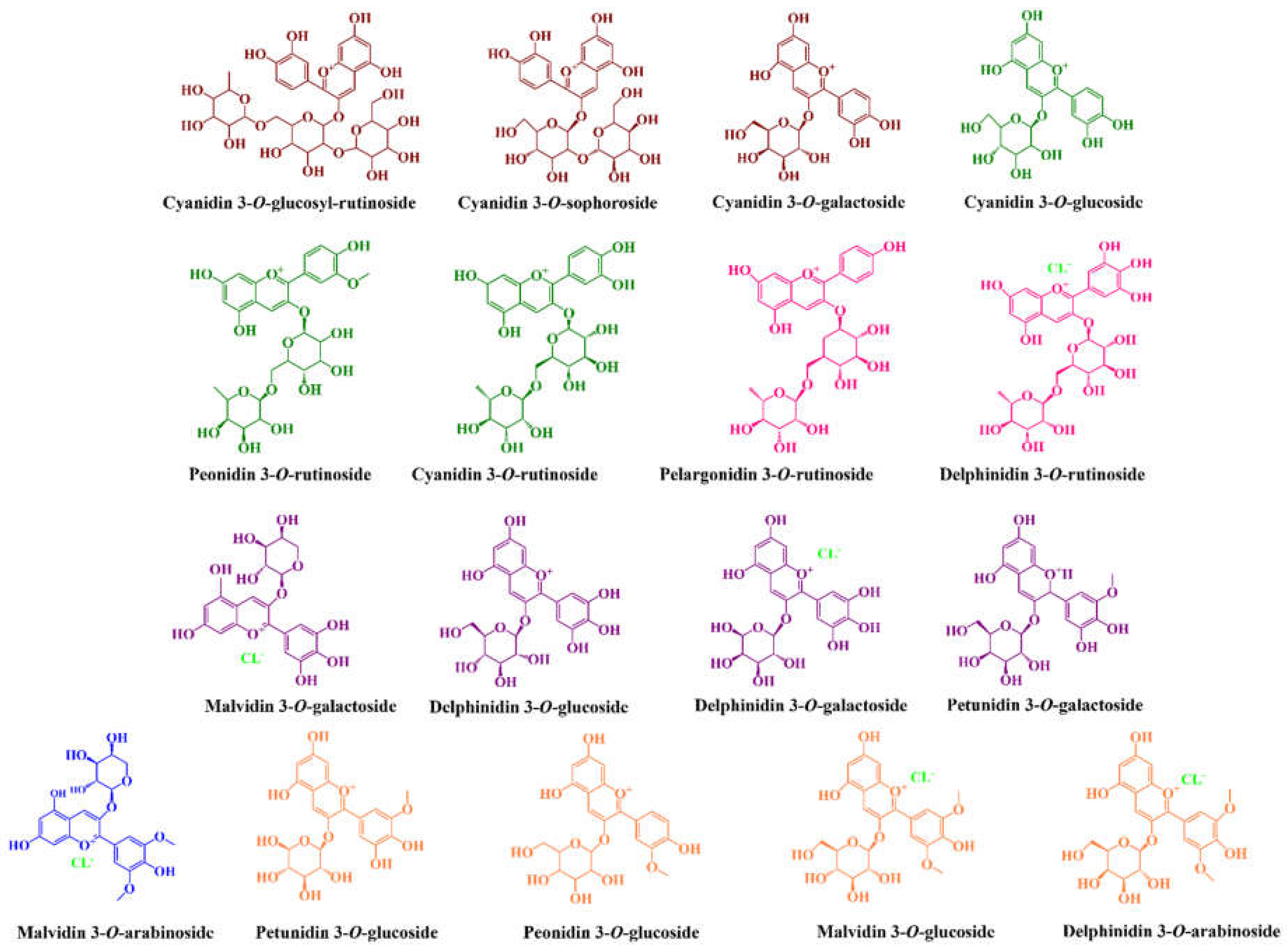
| Anthocyanins | ||||||||
| Cyanidin 3-O-glucosyl-rutinoside | 89.0–227.66 | - | - | - | 92.86–441.11 | - | - | [54,72,73,74,87,88] |
| Cyanidin 3-O-rutinoside | 1.76–74.7 | 0.20–389.9 | - | - | 0.38–85.5 | 104.0–210.0 | - | [40,43,54,72,73,76,77,87,88,89,90] |
| Cyanidin 3-O-sophoroside | 0.13–10.44 | t.r. | - | - | 1.62–292.21 | - | - | [54,72,73,74,76,87,88] |
| Cyanidin 3-O-glucoside | 0.01–142.03 | 0.0–142.03 | 0.11–3.09 | t.r.–8.20 c | 2.0–9.9 | 22.0–37.0 | 0.26–89.0 | [40,43,54,59,66,72,73,76,77,81,84,85,87,89,90,91] |
| Cyanidin 3-O-xylosylrutinoside | t.r. | - | - | - | - | - | - | [53] |
| Cyanidin 3-coumaroyl-diglucoside | - | 0.001–0.44 | - | - | - | - | - | [76] |
| Cyanidin 3-5-diglucoside | - | 0.16–1.05 | - | - | - | - | 0.0–28.5 | [76,92] |
| Cyanidin 3-O-hexoside | - | - | 19.23 | - | - | - | - | [82] |
| Cyanidin 3-O-galactoside | 0.0–2.63 | t.r. | 0.80–9.96 | 5.40–8.90 c | - | - | 13–59 | [59,74,75,81,84,85,91] |
| cyanidin 3-(6″-acetyl-glucoside) | - | - | - | - | - | - | 20.0 | [93] |
| Cyanidin 3-O-sambubioside | - | 0.09–0.16 | - | - | - | - | - | [76] |
| Cyanidin 3-O-arabinoside | - | 0.25–0.40 | 0.42–1.09 | 2.62 c | - | - | 1.40–160 | [66,76,81,85,91] |
| Petunidin 3-O-galactoside | - | - | 2.57–28.54 | 6.94 c | - | - | 0.34–125 | [59,66,81,85,91] |
| Petunidin 3-O-glucoside | - | - | 0.67–25.14 | t.r.–9.93 c | - | - | 7.70–365.0 | [59,81,82,84,85,91,93] |
| Petunidin 3-O-arabinoside | - | - | 1.82–12.70 | 3.5.–4.30 c | - | - | 0.53–59.0 | [59,66,81,84,91] |
| Petunidin 3-(6″-acetyl)glucoside | - | - | - | - | - | - | 57.0 | [93] |
| Peonidin 3-O-glucoside | t.r. | 0.0–0.38 | 12.00–54.37 | 17.6–30.3 c | - | - | 0.63–91.0 | [59,66,76,78,84,90,91,93] |
| Peonidin 3-O-rutinoside | - | 0.0–6.7 | - | - | - | 29.0–36.0 | - | [54,78,89,90] |
| Peonidin 3-O-pentose | - | - | 0.52–0.69 | - | - | - | - | [81] |
| Peonidin 3-O-galactoside | - | - | 0.77–125.79 | 2.90–3.80 c | - | - | 0.54–19.0 | [59,66,81,84,85,91] |
| Peonidin 3-O-arabinoside | - | - | - | 2.4–13.4 c | - | - | <1–2 | [84,85,91] |
| Peonidin 3-(6″-acetyl)galactoside | - | - | - | - | - | - | 6.0 | [93] |
| Peonidin 3-(6″ -acetyl)glucoside | - | - | - | - | - | - | 40.0 | [93] |
| Pelargonidin 3-O-rutinoside | - | 0.0–7.97 | - | - | 0.11–131.42 | 7.0–9.0 | - | [76,78,88,89,90] |
| Pelargonidin 3-O-glucoside | - | 0.22–0.71 | - | - | - | - | 10.1–35.6 | [76,92] |
| Malvidin 3-O-glucoside | - | 0.08–0.45 | 0.68–34.75 | 21.53 c | - | - | 6.25–271.0 | [59,66,78,81,85,91,93] |
| Malvidin 3-O-galactoside | - | - | 12.11–67.45 | 19.57 c | - | - | 6.0–160 | [59,66,80,81,85,91,93] |
| Malvidin 3-O-arabinoside | - | - | 6.77–9.41 | 4.64–17.80 c | - | - | 4.60–73.0 | [66,81,84,85,91,93] |
| Malvidin-3-(6″-acetyl-galactoside) | - | - | 0.99–1.74 | - | - | - | 34.0 | [81,93] |
| Malvidin 3-O-xyloside | - | - | 0.56 | - | - | - | - | [81] |
| Malvidin-3-(6″-acetyl) glucoside | - | - | 1.63 | - | - | - | 131.0 | [81,93] |
| Malvidin 3-O-glucoside-acetaldehyde | - | 0.08–0.11 | - | - | - | - | - | [76] |
| Delphinidin 3-O-rutinoside | t.r. | t.r. | - | - | - | - | - | [53] |
| Delphinidin 3-O-glucoside | - | - | 1.21–53.62 | 0.2–8.08 c | - | - | 7.70–365.0 | [59,81,84,85,91,92] |
| Delphinidin 3-O-galactoside | - | - | 2.29–53.29 | 7.97–16.3 c | - | - | 0.14–223.0 | [59,66,81,84,85,91,93] |
| Delphinidin 3-O-arabinoside | - | - | 1.66–41.07 | 4.67–5.6 c | - | - | 0.67–134.0 | [59,66,81,84,91] |
| Delphinidin 3-(6″-acetyl)glucoside | - | - | - | - | - | - | 2.0 | [93] |
| Delphinidin 3-(malonyl)glucoside | - | - | - | - | - | - | 86.0 | [93] |
| Delphinidin | 0.01–0.52 | - | 8.51–141.1 | - | - | - | 5.40–25.7 | [70,72,82,83,92] |
| Malvidin | 0.27–8.31 | 0.04–0.06 | 131.3–154.6 | - | - | - | 0.37 | [66,70,72,79,83] |
| Peonidin | 0.01–0.19 | 0.11–3.93 | 14.28–36.9 | - | - | - | - | [70,72,79,82,83] |
| Cyanidin | 3.41–6.64 | 0.04–0.18 | 21.17–66.3 | - | - | - | 0.09 | [66,70,72,79,82,83] |
| Pelargonidin | 1.35–64.36 | - | - | - | - | - | - | [70,72] |
| Petunidin | - | - | 1.78–87.6 | - | - | - | - | [82,83] |
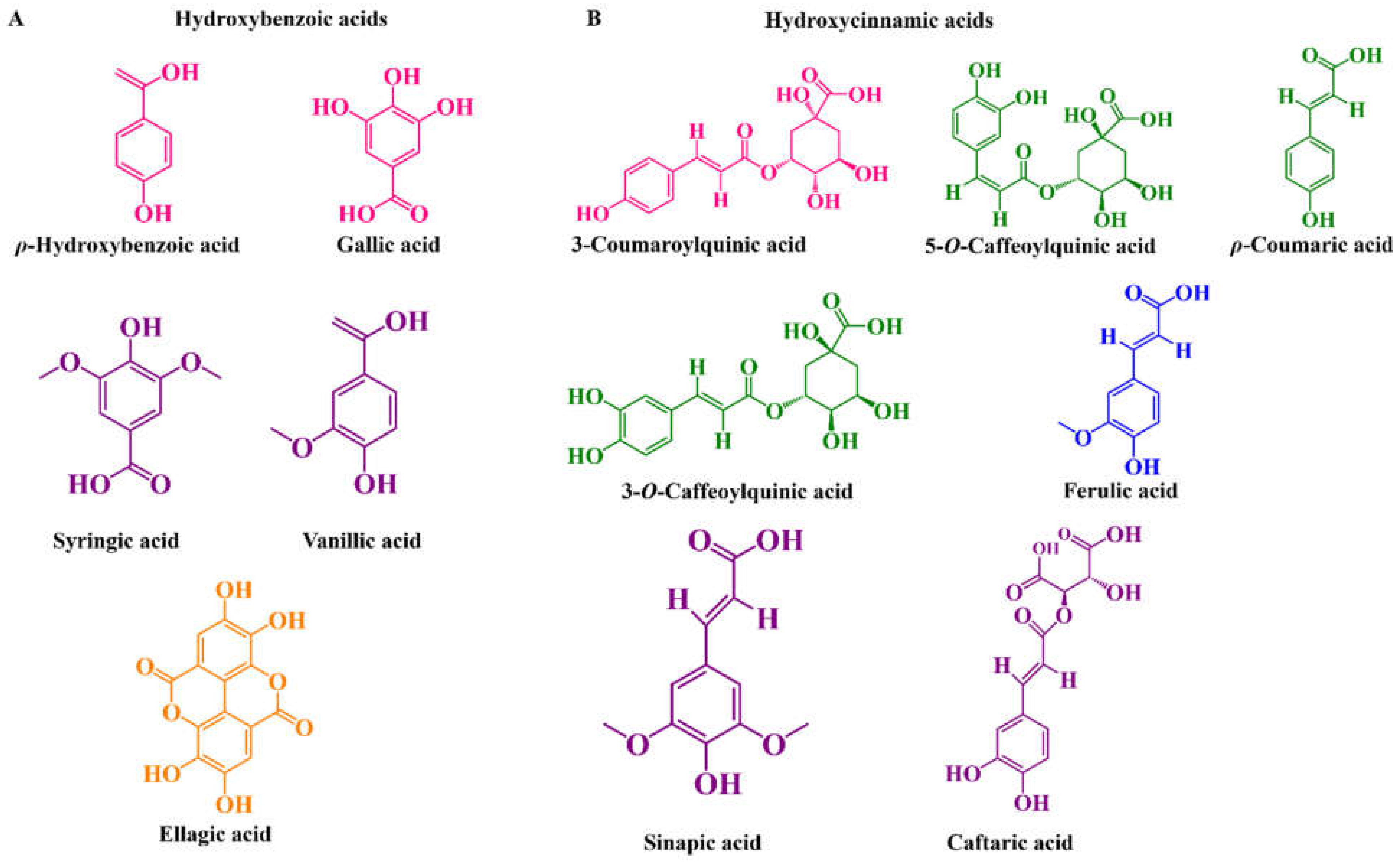
| Hydroxybenzoic acids | ||||||||
| ρ-Hydroxybenzoic acid | - | 10.3–19.1 | 0.054–59.89 | 0.0–103.67 | - | - | t.r. | [63,64,77,90,94,95] |
| Protocatechuic acid | - | 0.054–3.28 | 5.22–41.45 | - | - | - | - | [63,76,77,96] |
| Hydroxybenzoic acid-glycoside | - | 0.15–0.32 | - | - | - | - | - | [76] |
| Hydroxybenzoyl hexose | - | 0.14–0.70 | - | - | - | - | - | [76] |
| Vanillic acid-glycoside | - | 0.76–3.05 | - | - | - | - | - | [76] |
| Vanillic acid | - | - | 0.011–0.027 | - | - | - | t.r. | [94,95] |
| Syringic acid | - | 0.0–0.071 | 0.034–9.95 | - | - | 6.64–14.46 | t.r. | [63,77,94,95,96,97] |
| Gallic acid | - | 0.0018–10.64 | 0.02–5.68 | 1.53–258.9 | - | 0.0–6.55 | - | [60,63,64,77,78,96,97,98] |
| Ethyl gallate | - | 0.0003–0.0014 | - | - | - | - | - | [96] |
| Propyl gallate | - | 0.0005–0.0099 | - | - | - | - | - | [96] |
| Ellagic acid | - | - | 0.75–6.65 | 0.0–19.25 | - | - | - | [60,64] |
| 2,5-Dihydroxybenzoic acid | - | 0.0–1.50 | - | - | - | - | - | [78,96] |
| Hydroxycinnamic acids | ||||||||
| Salicylic acid | - | 0.0037–1.31 | - | - | - | - | - | [90,96] |
| Cinnamic acid | - | 7.8–11.1 | 0.003–0.07 | - | - | - | - | [90,94] |
| Ferulic acid | - | 0–5.7 | 0.018–4.16 | 0.0–16.97 | 1.14–1.27 | 1.01–6.35 | t.r. | [60,64,77,90,94,95,96,97,99] |
| 3-Caffeoylquinic acid | 5.24–27.79 | 38.0–187.0 | 0.46–7.12 | 0.039–2.46 | 82.0–183.0 | 24.77–37.78 | - | [54,60,74,76,77,81,90,97,100] |
| 4-Caffeoylquinic acid | - | 2.6–29.2 | - | - | - | - | - | [90] |
| 5-Caffeoylquinic acid | 0.58–60.33 | 0.21–120.8 | 13.52–65.24 | - | 28.30–995 | - | t.r. | [54,63,74,76,90,95,99,100,101] |
| 3-Coumaroylquinic acid | - | 37.0–452.52 | - | - | 91.0–555.0 | - | - | [76,90,100] |
| 4-Coumaroylquinic acid cis | - | 0.74–18.58 | - | - | - | - | - | [76] |
| 4-Coumaroylquinic acid trans | - | 4.92–19.46 | - | - | - | - | - | [76] |
| 5-Coumaroylquinic acid cis | - | 0.38–0.96 | - | - | 12.0–81.0 | - | - | [76,100] |
| 5-Coumaroylquinic acid trans | - | 0.53–1.53 | - | - | - | - | - | [76] |
| 3-Feruloylquinic acid cis | - | 0.64–2.30 | - | - | - | - | - | [76] |
| 3-Feruloylquinic acid trans | - | 0.72–5.86 | - | - | - | - | - | [76] |
| 4-Feruloylquinic acid cis | - | 0.18–0.49 | - | - | - | - | - | [76] |
| 5-Feruloylquinic acid trans | - | 0.04–0.25 | - | - | - | - | - | [76] |
| 5-Feruloylquinic acid cis | - | 0.11–2.92 | - | - | - | - | - | [76] |
| Caffeoylquinic acid glycoside | - | 0.11–1.71 | - | - | - | - | - | [76] |
| 3,5-diCaffeoylquinic acid | - | 0.26–2.87 | - | - | - | - | - | [76] |
| 4,5-diCaffeoylquinic acid | - | 0.09–0.78 | - | - | - | - | - | [76] |
| Caffeoylshikimic acid | - | 0.26–0.56 | - | - | - | - | - | [76] |
| 3- and 4-Caffeoylquinic lactone | - | 0.39–2.26 | - | - | - | - | - | [76] |
| Caftaric acid | - | - | 4.71 | - | - | - | - | [77] |
| 3-Coumaroylquinic lactone | - | 0.39–0.99 | - | - | - | - | - | [76] |
| 4-Coumaroylquinic lactone | - | 0.11–2.02 | - | - | - | - | - | [76] |
| 3-Coumaroyl-5-caffeoylquinicacid | - | 0.03–0.76 | - | - | - | - | - | [76] |
| 3-Caffeoyl-4-coumaroylquinic acid | - | 0.02–0.46 | - | - | - | - | - | [76] |
| Coumaroyl hexose | - | 2.95 | - | - | - | - | - | [76] |
| Caffeoyl hexose | - | 0.32–2.02 | 0.19–0.22 | - | - | - | - | [76,81] |
| Caffeic acid | - | 0.0–0.83 | 0.042–32.3 | - | 5.39–15.50 | 3.74–4.00 | t.r. | [64,77,94,95,96,97,99] |
| Caffeic acid glycoside | - | 0.52–8.79 | - | - | - | - | - | [76] |
| Caffeoyl alcohol 3/4-O-hexoside | - | 0.7–0.78 | - | - | - | - | - | [76] |
| ρ-Coumaric acid | 0.89–50.69 | 0.11–70.45 | 2.40–25.49 | 0.0–15.78 | 11.30–12.10 | 0.0–0.15 | - | [54,60,63,64,74,77,78,96,97,99] |
| Feruloyl hexose | - | 0.33–0.39 | 0.91–1.63 | - | - | - | - | [76,81] |
| Sinapoyl hexose | - | 0.20–0.50 | - | - | - | - | - | [76] |
| Sinapic acid | - | - | 0.005–0.11 | - | - | - | - | [63,94] |
| 3-O-Coumaroyl quinic acid | - | 0.35–1.7 | - | - | - | - | - | [102] |
| 3-O-Coumaroyl quinic acid II | - | 3.1–15 | - | - | - | - | - | [102] |
| 5-O-Feruloyl quinic acid | - | 0.34 | - | - | - | - | - | [102] |
| 3-O-Feruloyl quinic acid | - | 0.038–0.44 | - | - | - | - | - | [102] |
| Chlorogenic acid isomer II | - | 0.24–0.63 | - | - | - | - | - | [102] |
| 4-O-Coumaroyl quinic acid I | - | 0.12–0.61 | - | - | - | - | - | [102] |
| 4-O-Coumaroyl quinic acid II | - | 0.50–3.0 | - | - | - | - | - | [102] |
| Malonyl-dicaffeoylquinic acid | - | - | 0.76 | - | - | - | - | [81] |
| Malonyl-caffeoylquinic acid | - | - | 9.32 | - | - | - | - | [81] |
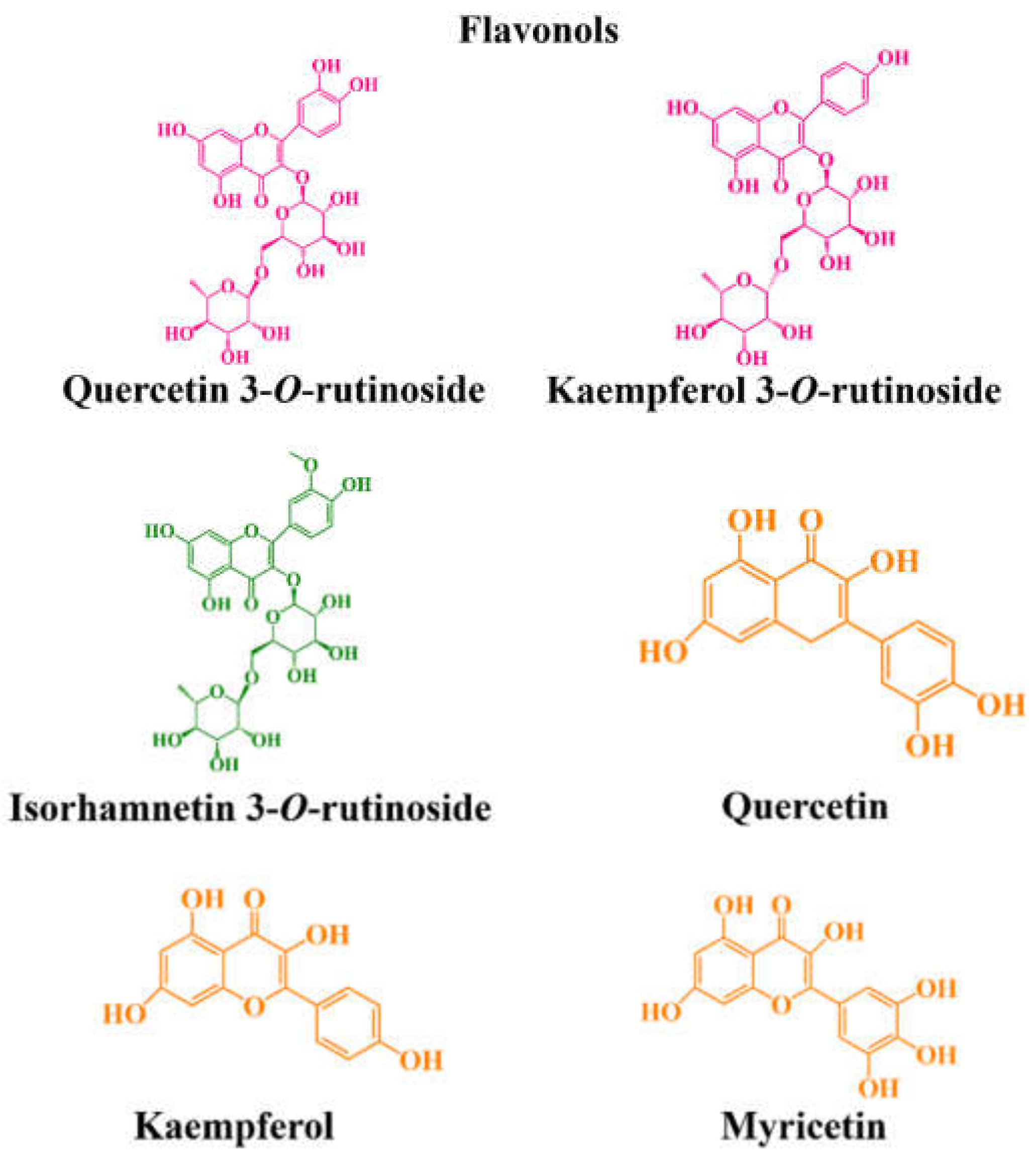
| Flavonols | ||||||||
| Myricetin | - | 0.0005–0.014 | 6.72–6.98 | 0.0–8.62 | - | - | - | [64,96] |
| Myricetin 3-O-glucuronide | - | - | - | 91.0–482.0 b | - | - | - | [51] |
| Myricetin 3-O-galactoside | - | - | - | 44.0–564.0 b | - | - | - | [51] |
| Myricetin-3-(6″-rhamnosyl)galactoside | - | - | - | 0.0–1.1 b | - | - | - | [51] |
| Myricetin 3-O-glucoside | - | - | - | 66.0–121.0 b | - | - | - | [51] |
| Myricetin-3-(6″-rhamnosyl)glucoside | - | - | - | 0.0–210.0 b | - | - | - | [51] |
| Isomers of myricetin 3-O-pentoside | - | - | - | 0.0–110.0 b | - | - | - | [51] |
| Myricetin 3-O-rhamnoside | - | - | - | 0.0–971.0 b | - | - | - | [51] |
| Quercetin | - | 0.0–2.51 | 0.29–21.48 | 0.046–9.97 | 184.0–739.0 b | - | 51.2 | [51,60,64,77,90,94,96,103] |
| Quercetin 3-O-galactoside | - | - | 0.19–31 | 269.0–1174.0 b | 0.0–4.0 | - | - | [51,77,94,100] |
| Quercetin 3-O-glucuronide | - | - | 0.06–1.76 | 475.0–3353.0 b | - | - | - | [51,63,77] |
| Quercetin 3-O-hexoside | - | 0.99–1.39 | - | - | - | - | - | [76] |
| Quercetin 3-O-arabinoside | - | - | 0.58 | - | 0.0–16.0 | - | - | [81,100] |
| Quercetin 3-O-glucoside | 0.21–0.44 | 0.0–26.55 | 0.9–34.64 | 81.0–203.0 b | - | - | t.r. | [51,54,63,76,77,81,95] |
| Quercetin-3-O-[4″-(3-hydroxy-3-methylglutaroyl)]-α-rhamnoside syringetin-3-rhamnoside | - | - | - | 0.0–1719.0 b | - | - | - | [51] |
| Isomers of quercetin 3-O-pentoside | - | - | - | 0.0–1005.0 b | - | - | - | [51] |
| Quercetin 3-O-rhamnoside | - | - | 26.0 | 88.10–4292.0 b | 18.0–45.0 | - | - | [63,100] |
| Quercetin 3-O-rutinoside | 0.84–7.63 | 0.78–51.97 | 0.008–0.056 | 0.044–6.74 | 4.10–53.80 | 0.0–4.74 | 65.0 | [54,60,74,76,94,97,99,102,103] |
| Quercetin 7-O-glucoside-3-O-rutinoside | - | 0.08–5.56 | - | - | - | - | - | [76,102] |
| Quercetin O-glucoside-O-rutinoside II | - | 3.67–132.7 | - | - | - | - | - | [98] |
| Kaempferol | - | 0.0028–10 | 0.061–19.65 | 0.0–3.72 | 5.60 | - | 12.1 | [63,64,77,94,96,103] |
| Kaempferol 3-O-glucoside | - | 0.024–1.36 | 0.008–6.01 | - | 3.40 | - | 0.30 | [51,63,76,77,94,102,103] |
| Kaempferol 3-O-rutinoside | 0.30–1.29 | 0.9–8.13 | - | - | - | - | - | [54,76,90,102] |
| Kaempferol rutinoside-hexoside | - | 0.13–1.08 | - | - | - | - | - | [76] |
| Laricitrin | - | - | - | 0.0–65.0 b | - | - | - | [51] |
| Laricitrin 3-O-galactoside | - | - | - | 41.0–710.0 b | - | - | - | [51] |
| Laricitrin-3-O-glucuronide | - | - | - | 151.0–640.0 b | - | - | - | [51] |
| Laricitrin 3-O-glucoside | - | - | 0.61–0.65 | - | - | - | - | [81] |
| Isorhamnetin | - | 0.0004–0.0024 | - | - | - | - | - | [96] |
| Isorhamnetin 3-O-rutinoside | 0.0–5.37 | 0.08–0.13 | - | - | - | - | - | [54,74] |
| Isorhamnetin 3-O-glucoside | - | - | - | 0.0–76.0 b | - | - | - | [51] |
| Syringetin | - | - | - | 0.0–119.0 b | - | - | - | [51] |
| Syringetin 3-O-galactoside | - | - | - | 70.0–742.0 b | - | - | - | [51] |
| Syringetin 3-O-glucoside | - | - | 0.77–0.97 | 85.0–594.0 b | - | - | - | [51,81] |
| Syringetin 3-O-glucuronide | - | - | - | 53.0–594.0 b | - | - | - | [51] |
| Syringetin 3-O-rhamnoside | - | - | - | 0.0–447.0 b | - | - | - | [51] |
| Syringetin 3-O-pentoside | - | - | - | 0.0–109.0 b | - | - | - | [51] |
| Flavan-3-ols | ||||||||
| (+)-Catechin | - | 0.13–84.34 | 0.067–81.8 | 0.13–387.48 | 4.0–77.0 | 0.38–1.44 | - | [57,60,64,76,77,81,94,96,97,99,100] |
| (−)-Epicatechin | 0.0–28.22 | 0.23–397.19 | 0.0014–20.70 | 0.0–129.51 | 13.60–369.0 | 1.17–1.54 | - | [60,63,64,76,77,94,97,99,100,104] |
| Epigallocatechin | - | - | 0.21–0.40 | - | 0.0–17.20 | - | - | [94,99] |
| Epicatechin 3-gallate | - | 0.29–3.12 | 0.48–19.27 | - | - | - | - | [63,76] |
| Catechin glucoside | - | 2.03–1.16 | - | - | - | - | - | [76] |
| Procyanidin tetramer B type 1 | - | 0.33–1.01 | - | - | - | - | - | [76] |
| Procyanidin tetramer B type 2 | - | 0.62–2.95 | - | - | - | - | - | [76] |
| Procyanidin dimer B type 1 | - | 2.24–6.99 | - | - | - | - | - | [76] |
| Procyanidin dimer B type 2 | - | 1.59–26.47 | - | - | - | - | - | [76] |
| Procyanidin dimer B type 3 | - | 1.28–3.59 | - | - | - | - | - | [76] |
| Procyanidin dimer B type 4 | - | 0.92–3.54 | - | - | - | - | - | [76] |
| Procyanidin dimer B type 5 | - | 4.80–15.26 | - | - | - | - | - | [76] |
| Propelargonidin dimer | - | 0.29–0.77 | - | - | - | - | - | [76] |
| Procyanidin pentamer B type | - | 0.18–1.68 | - | - | - | - | - | [76] |
| Dimer B2 | - | - | 0.40–1.51 | - | - | - | - | [81] |
| Procyanidin B1 | 0.0–27.69 | 0.55–7.29 | - | - | 12.0–92.0 | - | - | [57,74,100] |
| Procyanidin C1 | 0.0–8.6 | - | - | - | - | - | - | [74] |
| Flavanones | ||||||||
| Naringenin | - | - | 0.024–0.028 | - | - | - | - | [94] |
| Naringenin hexoside | - | 0.38–3.41 | - | - | - | - | - | [76] |
| Flavanonols | ||||||||
| Taxifolin 3-O-rutinoside | - | 0.43–100.52 | - | - | - | - | - | [76] |
| Taxifolin O-deoxyhexosylhexoside | - | 0.0–21.1 | - | - | - | - | - | [90] |
| Flavones | ||||||||
| Luteolin | - | 0.0027–0.020 | - | - | - | - | - | [96] |
| Luteolin 7-O-glucoside | - | - | - | - | 56.0 | - | 102.0 | [103] |
| Apigenin 8-C-glucoside | - | - | 0.057–0.14 | - | - | - | - | [94] |
| Coumarins | ||||||||
| Aesculin | - | - | 0.003–0.0011 | - | - | - | - | [94] |
| Chalcones | ||||||||
| Phlorizin | - | - | 0.041–0.57 | - | - | - | - | [96] |
| Plant | Part Used/ Compound | Model | Description | Effects | References |
|---|---|---|---|---|---|
| Enzymes inhibition studies | |||||
| Vaccinium myrtillus | Fruits | In vitro | Evaluation of the inhibitory activity of anthocyanin-rich bilberry extract (BE) on α-glucosidase and α-amylase | ↑ α-glycosidase and α-amylase inhibition in a mixed competitive manner. | [156] |
| Vaccinium myrtillus | Fruits | In vivo | Evaluation of the effect of BE on the digestive properties of carbohydrates in eight-week-old SPF-grade C57BL/6 J male mice | ↓ Postprandial glucose | [156] |
| Vaccinium corymbosum | Fruits | In vitro | Evaluation of the highbush blueberries in the inhibition of α-glycosidase and α-amylase enzymes | ↑ α-glycosidase and α-amylase inhibition | [157] |
| Prunus avium | Fruits | In vitro | Evaluation of the antidiabetic potential of hydroethanolic extract of sweet cherry | ↑ α-glycosidase inhibition in a dose-dependent manner | [43,158] |
| Prunus avium | Stem, leaf, flower | In vitro | Evaluation of the antidiabetic potential of hydroethanolic extract and aqueous infusion of sweet cherry by-products | ↑ α-glycosidase inhibition in a dose-dependent manner | [159] |
| Vaccinium corymbosum | Fruits | In vitro | Analysis of the inhibitory effect of phenolic compounds commonly present in berry on dipeptidyl-peptidase IV (DPP-IV) | Resveratrol and flavone are competitive inhibitors to (DPP-IV) Luteolin and apigenin bond to DPP-IV in a noncompetitive manner | [160] |
| Pancreatic β-cells protection studies | |||||
| n.a. | Resveratrol | In vitro | Evaluation of the effects of resveratrol on pancreatic β-cell function in mouse β-Min6 cells and human islets | ↑ intracellular cAMP levels ↑ insulin secretion ↑ pancreatic β-cell function | [161] |
| Prunus avium | Fruits | In vivo | Evaluation of the effects of ethanolic extract on aloxan-induced diabetic rats | ↓ blood glucose ↓ urinary microalbumin ↑ ctreatinine secretion level in urea | [162] |
| Vaccinium myrtillus | Leaf | In vivo | Analysis of the glucose homeostasis, pancreatic β-cell function, and insulin sensitivity in high-fat diet–induced in diabetic male C57BL/6J mice | ↓ plasma glucose ↓ glycated hemoglobin ↓ insulin resistance ↑ mRNA levels of pancreatic β-cell ↑ pancreatic insulin signaling ↓ transcriptional expression of the β-cellimproved insulin sensitivity ↑ insulin signaling | [163] |
| Vaccinium myrtillus | Fruits | Clinical trial | Evaluation of the effects of purified anthocyanins on dyslipidemia, oxidative status, and insulin sensitivity in patients with type 2 diabetes | improved dyslipidemia ↑ antioxidant status ↓ plasma glucose ↓ insulin resistance ↓ LDL cholesterol and triglycerides ↑ HDL | [164] |
| Insulin release and regulation | |||||
| Prunus avium | Fruits | In vitro | Evaluation of three different phenolic fractions (anthocyanins-rich fraction (ARF), hydroxycinnamic acids-rich fraction (HRF) and flavonols-rich fraction (FRF)) in glucose consumption by HepG2 cells | ↑ glucose consumption ↑ insulin sensitivity inhibited excessive gluconeogenesis | [165] |
| Vaccinium myrtillus | Fruits | In vivo | Analysis of blueberry effects glucose metabolism and pancreatic β-cell proliferation in high fat diet (HFD)-induced obese mice. | ↑ insulin sensitivity ↑ glucose tolerance | [166] |
| Vaccinium ashei Vaccinium corymbosum | Fruits | Clinical trial | Evaluation of the effect of daily dietary supplementation with bioactives from blueberries on whole-body insulin sensitivity in men and women | ↑ insulin sensitivity | [167] |
| n.a. | Resveratrol | In vitro | Evaluation of the protective effects of resveratrol in β-cell dysfunction in INS-1 cells | ↑ glucose-stimulated insulin secretion ↑ SIRT1 expression restored the function of INS-1 cell | [168] |
| Vaccinium myrtillus | Fruits | In vivo | Analysis of the otective effects of blueberry anthocyanin extract (BAE) against oxidative stress and the roles of SIRT1 and NF-κB | ↑ SIRT1 expression ↑ SOD and GSH activity | [169] |
| Vaccinium myrtillus | Fruits | In vitro | Evaluation of the role of berry phenolic compounds to modulate incretin-cleaving DPP-IV and its substrate glucagon-like peptide-1 (GLP-1), insulin secretion | ↑ insulin secretion Upregulated expression of mRNA of insulin-receptor | [170] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, A.C.; Nunes, A.R.; Flores-Félix, J.D.; Alves, G.; Silva, L.R. Cherries and Blueberries-Based Beverages: Functional Foods with Antidiabetic and Immune Booster Properties. Molecules 2022, 27, 3294. https://doi.org/10.3390/molecules27103294
Gonçalves AC, Nunes AR, Flores-Félix JD, Alves G, Silva LR. Cherries and Blueberries-Based Beverages: Functional Foods with Antidiabetic and Immune Booster Properties. Molecules. 2022; 27(10):3294. https://doi.org/10.3390/molecules27103294
Chicago/Turabian StyleGonçalves, Ana C., Ana R. Nunes, José D. Flores-Félix, Gilberto Alves, and Luís R. Silva. 2022. "Cherries and Blueberries-Based Beverages: Functional Foods with Antidiabetic and Immune Booster Properties" Molecules 27, no. 10: 3294. https://doi.org/10.3390/molecules27103294
APA StyleGonçalves, A. C., Nunes, A. R., Flores-Félix, J. D., Alves, G., & Silva, L. R. (2022). Cherries and Blueberries-Based Beverages: Functional Foods with Antidiabetic and Immune Booster Properties. Molecules, 27(10), 3294. https://doi.org/10.3390/molecules27103294










