Antimicrobial Activity of Green Silver Nanoparticles Synthesized by Different Extracts from the Leaves of Saudi Palm Tree (Phoenix Dactylifera L.)
Abstract
:1. Introduction
2. Results and Discussion
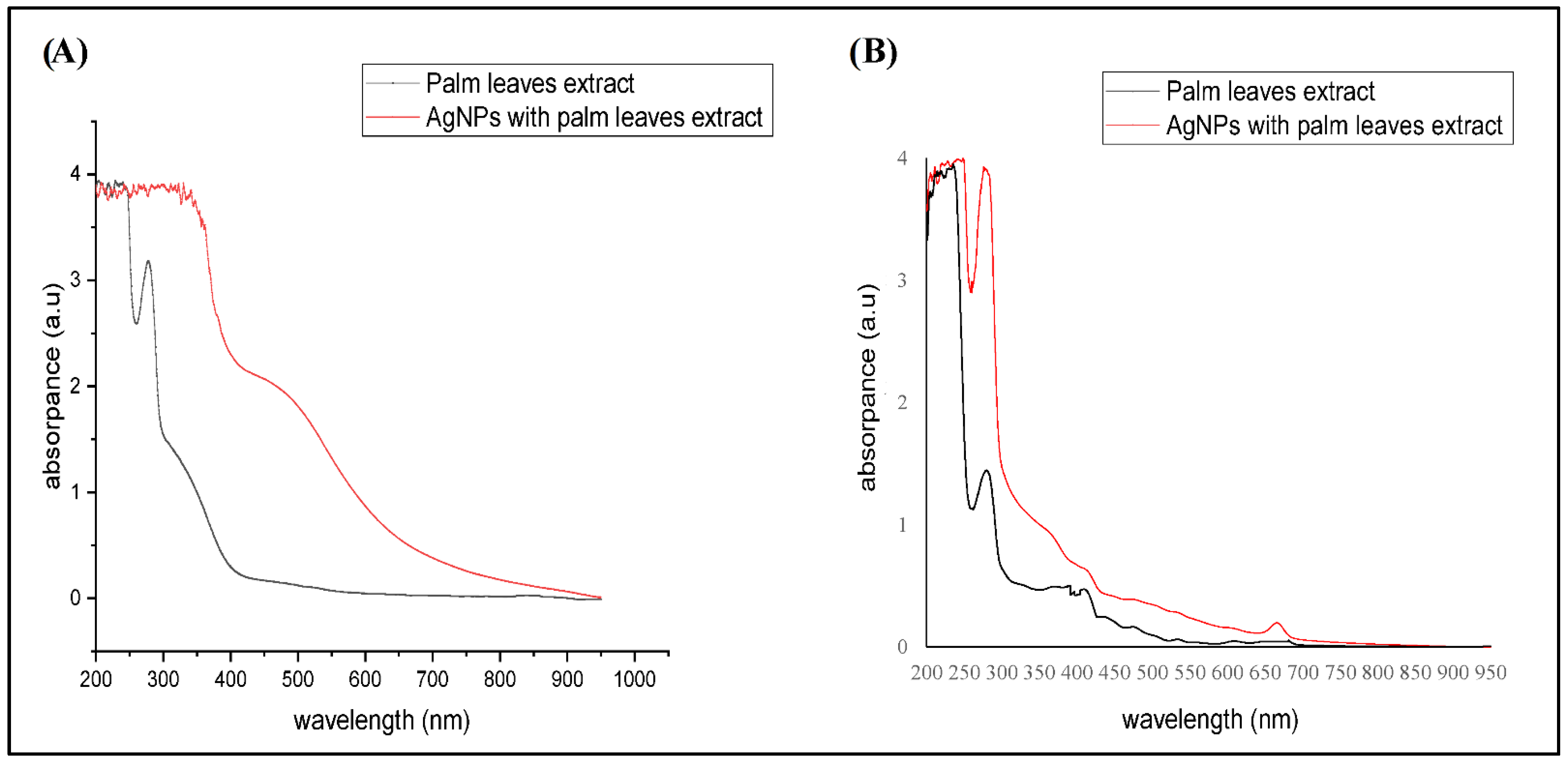
2.1. Structural Characteristics of the AgNPs Synthesized from Different Palm Leaves Extracts
2.2. The Phenolic Constituents of Palm Leave Extracts
2.3. AgNPs Synthesized by Phoenix dactylifera L. Leaves Extract Had Antifungal Activities
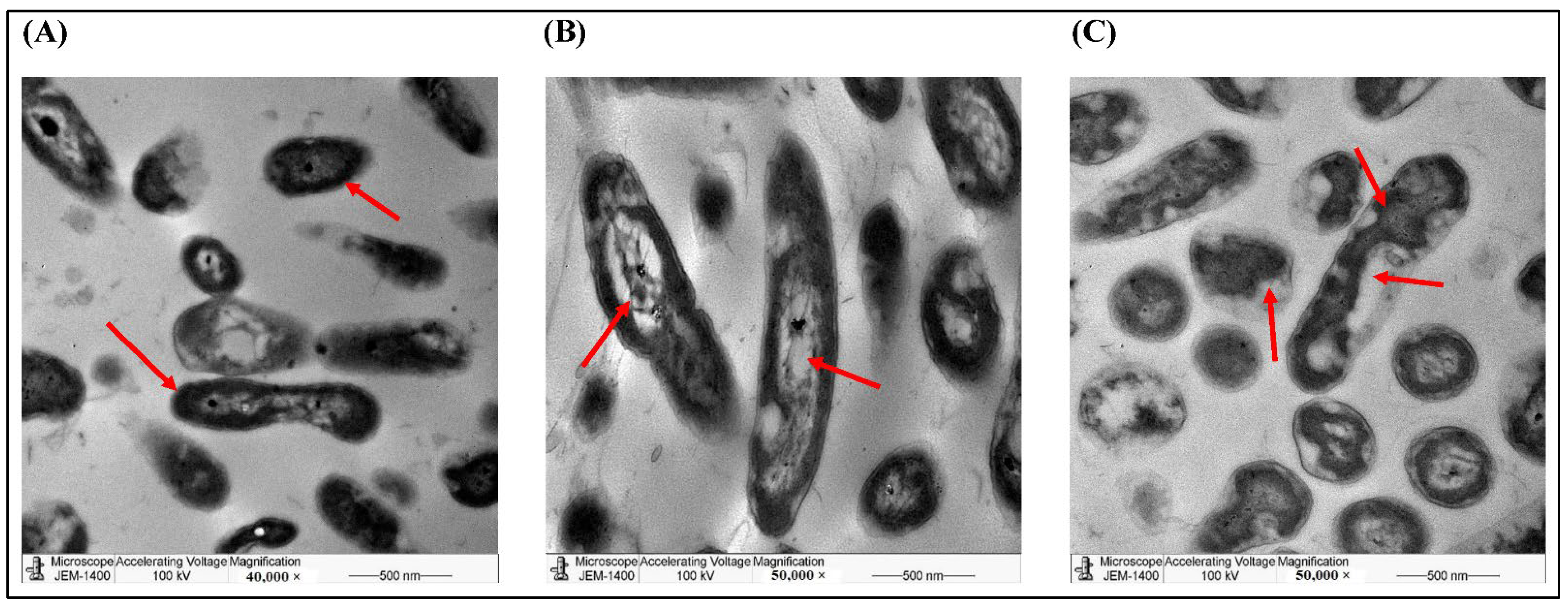
2.4. The Bactericidal Activities of AgNPs of Phoenix dactylifera L. Leave Extracts
3. Materials and Methods
3.1. Plant Material
3.2. Microorganisms
3.3. The Green Synthesis of AgNPs
3.4. Antimicrobial Experiments
3.5. Characterization of Synthesized AgNPs
3.5.1. UV Visible Spectroscopy
3.5.2. Gas Chromatography/Mass Spectrometry Technique (GC/MS)
3.5.3. Transmission Electron Microscopy (TEM)
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Al-Antary, T.; Alkhawaldeh, M.; Ateyyat, M. Economic importance of date palm Phoenix dactylifera L. (Liliopsida: Arecales: Arecaceae) pests in Jordan Valley. Braz. J. Biol. Sci. 2015, 2, 121–134. [Google Scholar]
- Al-Alawi, R.A.; Al-Mashiqri, J.H.; Al-Nadabi, J.; Al-Shihi, B.I.; Baqi, Y. Date Palm Tree (Phoenix dactylifera L.): Natural Products and Therapeutic Options. Front. Plant Sci. 2017, 8, 845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nixon, R.W. The date palm “Tree of Life” in the subtropical deserts. Econ. Bot. 1951, 5, 274–301. [Google Scholar] [CrossRef]
- Bouhlali, E.T.; Alem, C.; Ennassir, J.; Benlyas, M.; Mbark, A.N.; Zegzouti, Y.F. Phytochemical compositions and antioxidant capacity of three date (Phoenix dactylifera L.) seeds varieties grown in the South East Morocco. J. Saudi Soc. Agric. Sci. 2017, 16, 350–357. [Google Scholar] [CrossRef]
- Hittalmani, S.; Mahesh, H.B.; Shirke, M.D.; Biradar, H.; Uday, G.; Aruna, Y.R.; Lohithaswa, H.C.; Mohanrao, A. Genome and Transcriptome sequence of Finger millet (Eleusine coracana (L.) Gaertn.) provides insights into drought tolerance and nutraceutical properties. BMC Genom. 2017, 18, 465. [Google Scholar] [CrossRef]
- Banu, H.; Renuka, N.; Faheem, S.M.; Ismail, R.; Singh, V.; Saadatmand, Z.; Khan, S.S.; Narayanan, K.; Raheem, A.; Premkumar, K.; et al. Gold and Silver Nanoparticles Biomimetically Synthesized Using Date Palm Pollen Extract-Induce Apoptosis and Regulate p53 and Bcl-2 Expression in Human Breast Adenocarcinoma Cells. Biol. Trace Elem. Res. 2018, 186, 122–134. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [Green Version]
- Shameli, K.; Ahmad, M.B.; Zamanian, A.; Sangpour, P.; Shabanzadeh, P.; Abdollahi, Y.; Zargar, M. Green biosynthesis of silver nanoparticles using Curcuma longa tuber powder. Int. J. Nanomed. 2012, 7, 5603–5610. [Google Scholar] [CrossRef] [Green Version]
- Jyoti, K.; Baunthiyal, M.; Singh, A. Characterization of silver nanoparticles synthesized using Urtica dioica Linn. leaves and their synergistic effects with antibiotics. J. Radiat. Res. Appl. Sci. 2016, 9, 217–227. [Google Scholar] [CrossRef] [Green Version]
- Kuppusamy, P.; Yusoff, M.M.; Maniam, G.P.; Govindan, N. Biosynthesis of metallic nanoparticles using plant derivatives and their new avenues in pharmacological applications—An updated report. Saudi Pharm. J. 2016, 24, 473–484. [Google Scholar] [CrossRef]
- Khatami, M.; Pourseyedi, S. Phoenix dactylifera (date palm) pit aqueous extract mediated novel route for synthesis high stable silver nanoparticles with high antifungal and antibacterial activity. IET Nanobiotechnol. 2015, 9, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Aldayel, F.M.; Alsobeg, M.S.; Khalifa, A. In vitro antibacterial activities of silver nanoparticles synthesised using the seed extracts of three varieties of Phoenix dactylifera. Braz. J. Biol. 2021, 82, e242301. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.E.; Al-Qahtani, A.; Al-Mutairi, A.; Al-Shamri, B.; Aabed, K.F. Antibacterial and Cytotoxic Potential of Biosynthesized Silver Nanoparticles by Some Plant Extracts. Nanomaterials 2018, 8, 382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murthy, H.C.A.; Zeleke, T.D.; Ravikumar, C.R.; Kumar, M.R.A.; Nagaswarupa, H.P. Electrochemical properties of biogenic silver nanoparticles synthesized using Hagenia abyssinica (Brace) JF. Gmel. medicinal plant leaf extract. Mater. Res. Express 2020, 7, 55016. [Google Scholar] [CrossRef]
- Kalishwaralal, K.; Deepak, V.; Pandian, S.R.K.; Nellaiah, H.; Sangiliyandi, G. Extracellular biosynthesis of silver nanoparticles by the culture supernatant of Bacillus licheniformis. Mater. Lett. 2008, 62, 4411–4413. [Google Scholar] [CrossRef]
- Rozhin, A.; Batasheva, S.; Kruychkova, M.; Cherednichenko, Y.; Rozhina, E.; Fakhrullin, R. Biogenic Silver Nanoparticles: Synthesis and Application as Antibacterial and Antifungal Agents. Micromachines 2021, 12, 1480. [Google Scholar] [CrossRef]
- Singh, P.; Kim, Y.J.; Zhang, D.; Yang, D.C. Biological Synthesis of Nanoparticles from Plants and Microorganisms. Trends Biotechnol. 2016, 34, 588–599. [Google Scholar] [CrossRef]
- Singh, A.; Gautam, P.K.; Verma, A.; Singh, V.; Shivapriya, P.M.; Shivalkar, S.; Sahoo, A.K.; Samanta, S.K. Green synthesis of metallic nanoparticles as effective alternatives to treat antibiotics resistant bacterial infections: A review. Biotechnol. Rep. 2020, 25, e00427. [Google Scholar] [CrossRef]
- Jeyaraj, M.; Gurunathan, S.; Qasim, M.; Kang, M.-H.; Kim, J.-H. A Comprehensive Review on the Synthesis, Characterization, and Biomedical Application of Platinum Nanoparticles. Nanomaterials 2019, 9, 1719. [Google Scholar] [CrossRef] [Green Version]
- Ponarulselvam, S.; Panneerselvam, C.; Murugan, K.; Aarthi, N.; Kalimuthu, K.; Thangamani, S. Synthesis of silver nanoparticles using leaves of Catharanthus roseus Linn. G. Don and their anti-plasmodial activities. Asian Pac. J. Trop. Biomed. 2012, 2, 574–580. [Google Scholar] [CrossRef] [Green Version]
- Krithiga, N.; Rajalakshmi, A.; Jayachitra, A. Green Synthesis of Silver Nanoparticles Using Leaf Extracts of Clitoria ternatea and Solanum nigrum and Study of Its Antibacterial Effect against Common Nosocomial Pathogens. J. Nanosci. Nanotechnol. 2015, 2015, 928204. [Google Scholar] [CrossRef] [Green Version]
- Charti, I.; Azouzi, A.; Belghiti, A.; Sair, S.; Abboud, Y.; El Bouari, A. Ecofriendly synthesis of stabilized silver nanoparticles and the evaluation of their potential applications. Curr. Res. Green Sustain. Chem. 2021, 4, 100102. [Google Scholar] [CrossRef]
- Ashraf, J.M.; Ansari, M.A.; Khan, H.M.; Alzohairy, M.A.; Choi, I. Green synthesis of silver nanoparticles and characterization of their inhibitory effects on AGEs formation using biophysical techniques. Sci. Rep. 2016, 6, 20414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anandalakshmi, K.; Venugobal, J.; Ramasamy, V. Characterization of silver nanoparticles by green synthesis method using Pedalium murex leaf extract and their antibacterial activity. Appl. Nanosci. 2016, 6, 399–408. [Google Scholar] [CrossRef] [Green Version]
- Mtambo, S.E.; Krishna, S.B.N.; Sershen; Govender, P. Physico-Chemical, antimicrobial and anticancer properties of silver nanoparticles synthesised from organ-specific extracts of Bidens pilosa L. S. Afr. J. Bot. 2019, 126, 196–206. [Google Scholar] [CrossRef]
- El-Hawwary, S.S.; Abd Almaksoud, H.M.; Saber, F.R.; Elimam, H.; Sayed, A.M.; El Raey, M.A.; Abdel-mohsen, U.R. Green-synthesized zinc oxide nanoparticles, anti-Alzheimer potential and the metabolic profiling of Sabal blackburniana grown in Egypt supported by molecular modelling. RSC Adv. 2021, 11, 18009–18025. [Google Scholar] [CrossRef]
- Al-Otibi, F.; Al-Ahaidib, R.A.; Alharbi, R.I.; Al-Otaibi, R.M.; Albasher, G. Antimicrobial Potential of Biosynthesized Silver Nanoparticles by Aaronsohnia factorovskyi Extract. Molecules 2020, 26, 130. [Google Scholar] [CrossRef]
- Mani, M.; Chang, J.H.; Dhanesh Gandhi, A.; Kayal Vizhi, D.; Pavithra, S.; Mohanraj, K.; Mohanbabu, B.; Babu, B.; Balachandran, S.; Kumaresan, S. Environmental and biomedical applications of AgNPs synthesized using the aqueous extract of Solanum surattense leaf. Inorg. Chem. Commun. 2020, 121, 108228. [Google Scholar] [CrossRef]
- Pingale, S.S.; Rupanar, S.V.; Chaskar, M.G. Plant-mediated biosynthesis of silver nanoparticles from Gymnema sylvestre and their use in phtodegradation of Methyl orange dye. J. Water Environ. Nanotechnol. 2018, 3, 106–115. [Google Scholar] [CrossRef]
- Alotibi, F.O.; Ashour, E.H.; Al-Basher, G. Evaluation of the antifungal activity of Rumex vesicarius L. and Ziziphus spina-christi (L) Desf. Aqueous extracts and assessment of the morphological changes induced to certain myco-phytopathogens. Saudi J. Biol. Sci. 2020, 27, 2818–2828. [Google Scholar] [CrossRef]
- Batiha, G.E.-S.; Beshbishy, A.M.; Alkazmi, L.; Adeyemi, O.S.; Nadwa, E.; Rashwan, E.; El-Mleeh, A.; Igarashi, I. Gas chromatography-mass spectrometry analysis, phytochemical screening and antiprotozoal effects of the methanolic Viola tricolor and acetonic Laurus nobilis extracts. BMC Complement. Med. Ther. 2020, 20, 87. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [Green Version]
- Sarker, U.; Oba, S. Phenolic profiles and antioxidant activities in selected drought-tolerant leafy vegetable amaranth. Sci. Rep. 2020, 10, 18287. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Venegas, G.; Gómez-Mora, J.A.; Meraz-Rodríguez, M.A.; Flores-Sánchez, M.A.; Ortiz-Miranda, L.F. Effect of flavonoids on antimicrobial activity of microorganisms present in dental plaque. Heliyon 2019, 5, e03013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arima, H.; Ashida, H.; Danno, G. Rutin-enhanced antibacterial activities of flavonoids against Bacillus cereus and Salmonella enteritidis. Biosci. Biotechnol. Biochem. 2002, 66, 1009–1014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, G.; Guan, Y.; Yi, H.; Lai, S.; Sun, Y.; Cao, S. Antibacterial activity and mechanism of plant flavonoids to gram-positive bacteria predicted from their lipophilicities. Sci. Rep. 2021, 11, 10471. [Google Scholar] [CrossRef]
- Bentrad, N.; Gaceb-terrak, R.; Benmalek, Y.; Rahmania, F. Studies on Chemical Composition and Antimicrobial Activities of Bioactive Molecules from Date Palm (Phoenix dactylifera L.) Pollens and Seeds. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 242–256. [Google Scholar] [CrossRef] [Green Version]
- Belmir, S.; Boucherit, K.; Boucherit-Otmani, Z.; Belhachemi, M.-H. Effect of aqueous extract of date palm fruit (Phoenix dactylifera L.) on therapeutic index of amphotericin B. Pharmacognosie 2015, 14, 97–101. [Google Scholar] [CrossRef]
- Oves, M.; Aslam, M.; Rauf, M.A.; Qayyum, S.; Qari, H.A.; Khan, M.S.; Alam, M.Z.; Tabrez, S.; Pugazhendhi, A.; Ismail, I.M.I. Antimicrobial and anticancer activities of silver nanoparticles synthesized from the root hair extract of Phoenix dactylifera. Mater. Sci. Eng. C 2018, 89, 429–443. [Google Scholar] [CrossRef]
- Sarvat, Z.; Aiman, Z. Biosynthesis and Characterization of Silver Nanoparticles Using Phoenix dactylifera Fruits Extract and their In Vitro Antimicrobial and Cytotoxic Effects. Open Biotechnol. J. 2019, 13, 37–46. [Google Scholar] [CrossRef]
- Rizwana, H.; Fatimah, A.; Alharbi, R.I.; Alqusumi, R. Morphology and Ultrastructure of some pathogenic fungi altered by leaf extracts of Senna alexandrina mill. Pak. J. Agric. Sci. 2021, 58, 389–408. [Google Scholar] [CrossRef]
- Sani, N.M.; Abdulkadir, F.; Sani, M.N. Antimicrobial activity of Phoenix dactylifera (date palm) on some selected members of Enterobacteriaceae. Bayero J. Pure Appl. Sci. 2017, 10, 36–39. [Google Scholar] [CrossRef] [Green Version]
- Ansari, M.A.; Alzohairy, M.A. One-Pot Facile Green Synthesis of Silver Nanoparticles Using Seed Extract of Phoenix dactylifera and Their Bactericidal Potential against MRSA. Evid. Based Complement. Altern. Med. 2018, 2018, 1860280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abusahid, Z.; Kandiah, M. In Vitro Green Synthesis of Phoenix Dactylifera Silver Nanoparticles: Assessing Their Antioxidant and Antimicrobial Properties. Int. J. Nanosci. 2018, 18, 1850031. [Google Scholar] [CrossRef] [Green Version]
- Stojković, D.; Petrović, J.; Soković, M.; Glamočlija, J.; Kukić-Marković, J.; Petrović, S. In situ antioxidant and antimicrobial activities of naturally occurring caffeic acid, p-coumaric acid and rutin using food systems. J. Sci. Food Agric. 2013, 93, 3205–3208. [Google Scholar] [CrossRef]
- Al-zoreky, N.S.; Al–Taher, A.Y. Antibacterial activity of spathe from Phoenix dactylifera L. against some food-borne pathogens. Ind. Crops Prod. 2015, 65, 241–246. [Google Scholar] [CrossRef]
- Alamgir, A.N.M. Herbal Drugs: Their Collection, Preservation, and Preparation, Evaluation, Quality Control and Standardization of Herbal Drugs. In Therapeutic Use of Medicinal Plants and Their Extracts; Alamgir, A.N.M., Ed.; Springer International Publishing: Cham, Switzerland, 2017; Volume 1, pp. 453–495. [Google Scholar] [CrossRef]
- Dropulic, L.K.; Lederman, H.M. Overview of Infections in the Immunocompromised Host. Microbiol. Spectr. 2016, 4, 1–71. [Google Scholar] [CrossRef]





| Phenolic Compounds | Molecular Formula | Molecular Weight g/mol | MS Fragments (m/z) | |
|---|---|---|---|---|
| Aqueous | Ethanol | |||
| Chlorogenic Acid | C16H18O9 | 354.311 | 353.90 | 351.00 |
| Gallic Acid | C6H2(OH)3COOH | 170.12 | 169.20 | 170.10 |
| Vanillic Acid | C8H8O4 | 168.148 | 168.20 | 169.10 |
| Syringic Acid | C9H10O5 | 198.174 | 198.20 | 198.00 |
| Ferulic Acid | C10H10O4 | 194.186 | 193.90 | 195.10 |
| Caffeic Acid | C9H8O4 | 180.159 | 179.10 | 179.10 |
| p-Coumaric Acid | C9H8O | 164.16 | 166.00 | 166.10 |
| Rutin | C27H30O16 | 610.521 | 606.70 | 613.10 |
| Luteolin | C15H10O6 | 286.239 | 286.00 | 286.00 |
| Quercetin | C15H10O7 | 302.238 | 303.10 | 303.00 |
| Species | Crude Extract (10 mg/mL) | AgNO3 (2 mM) | AgNPs (25%) | AgNPs (50%) | AgNPs (100%) | |
|---|---|---|---|---|---|---|
| C. albicans | Mean ± SD | 20.4 ± 0.2 | 24.1 ± 0.1 | 23.3 ± 0.6 | 24.2 ± 0.2 | 26.2 ± 0.2 |
| p value # | ------- | 0.001 | 0.038 | 1.4 × 10−4 | 3.7 × 10−5 | |
| C. tropicalis | Mean ± SD | 16.3 ± 0.2 | 23.4 ± 0.2 | 23.3 ± 0.3 | 25.0 ± 0.1 | 27.1 ± 0.2 |
| p value # | ------- | 3 × 10−6 | 1.1 × 10−4 | 6.4 × 10−5 | 1 × 10−6 | |
| C. parapslosis | Mean ± SD | 20.4 ± 0.3 | 22.1 ± 0.2 | 22.3 ± 0.3 | 24.3 ± 0.1 | 26.2 ± 0.2 |
| p value # | ------- | 0.008 | 0.006 | 0.004 | 8.1 × 10−5 | |
| C. glabrata | Mean ± SD | 10.3 ± 0.1 | 18.0 ± 0.2 | 15.0 ± 0.2 | 17.0 ± 0.1 | 19.3 ± 0.2 |
| p value # | ------- | 8.6 × 10−5 | 3 × 10−4 | 6 × 10−6 | 5.9 × 10−5 | |
| C. krusei | Mean ± SD | 8.0 ± 0.2 | 8.9 ± 0.2 | 10.5 ± 0.5 | 11.1 ± 0.1 | 14.4 ± 0.1 |
| p value # | ------- | 0.022 | 0.034 | 1.6 × 10−4 | 7 × 10−6 | |
| Species | Crude Extract (10 mg/mL) | AgNO3 (2 mM) | AgNPs (25%) | AgNPs (50%) | AgNPs (100%) | |
|---|---|---|---|---|---|---|
| C. albicans | Mean ± SD | 14.7 ± 0.1 | 18.1 ± 0.1 | 18.1 ± 0.1 | 19.3 ± 0.2 | 20.3 ± 0.2 |
| p value # | ------- | 2 × 10−5 | 2 × 10−5 | 2.5 × 10−5 | 1.2 × 10−5 | |
| C. tropicalis | Mean ± SD | 17.1 ± 0.1 | 17.9 ± 0.2 | 18.3 ± 0.1 | 19.0 ± 0.1 | 20.4 ± 0.2 |
| p value # | ------- | 0.018 | 0.001 | 1.3 × 10−4 | 1.4 × 10−4 | |
| C. parapslosis | Mean ± SD | 18.5 ± 0.2 | 21.6 ± 0.3 | 20.2 ± 0.0 | 20.4 ± 0.1 | 20.9 ± 0.2 |
| p value # | ------- | 0.002 | 0.017 | 0.008 | 0.001 | |
| C. glabrata | Mean ± SD | 5.0 ± 0.0 | 18.8 ± 0.2 | 18.0 ± 0.1 | 19.0 ± 0.2 | 20.2 ± 0.2 |
| p value # | ------- | 1.5 × 10−4 | 8.7 × 10−5 | 1.4 × 10−4 | 1.2 × 10−4 | |
| C. krusei | Mean ± SD | 0.0 ± 0.0 | 10.9 ± 0.1 | 9.1 ± 0.1 | 8.9 ± 0.1 | 11.1 ± 0.1 |
| p value # | ------- | 1.1 × 10−4 | 5.8 × 10−5 | 1.5 × 10−4 | 1.1 × 10−4 | |
| Species | Non-Treated | AgNPs (25%) | AgNPs (50%) | AgNPs (100%) | |
|---|---|---|---|---|---|
| E. coli | Mean ± SD | 0.2 ± 0.1 | 13.3 ± 0.3 | 14.5 ± 0.1 | 14.8 ± 0.1 |
| p value # | ------- | 8.9 × 10−5 | 7.2 × 10−8 | 6.6 × 10−8 | |
| B. subtilis | Mean ± SD | 0.1 ± 0.1 | 11.5 ± 0.5 | 12.2 ± 0.2 | 14.7 ± 0.4 |
| p value # | ------- | 0.002 | 6 × 10−6 | 4.5 × 10−4 | |
| P. aeruginosa | Mean ± SD | 0.4 ± 0.2 | 14.0 ± 0.1 | 14.9 ± 0.1 | 15.3 ± 0.1 |
| p value # | ------- | 1.4 × 10−5 | 1.2 × 10−5 | 1.4 × 10−4 | |
| S. aureus | Mean ± SD | 0.2 ± 0.1 | 18.8 ± 0.2 | 18.0 ± 0.1 | 19.0 ± 0.2 |
| p value # | ------- | 3.6 × 10−7 | 1.6 × 10−8 | 3.5 × 10−7 | |
| Species | Non-Treated | AgNPs (25%) | AgNPs (50%) | AgNPs (100%) | |
|---|---|---|---|---|---|
| E. coli | Mean ± SD | 0.2 ± 0.1 | 13.8 ± 0.1 | 14.1 ± 0.1 | 14.4 ± 0.1 |
| p value # | ------- | 8.7 × 10−8 | 6 × 10−7 | 2 × 10−6 | |
| B. subtilis | Mean ± SD | 0.1 ± 0.1 | 12.0 ± 0.5 | 12.1 ± 0.1 | 13.9 ± 1.0 |
| p value # | ------- | 14.1 × 10−5 | 8.1 × 10−8 | 0.01 | |
| P. aeruginosa | Mean ± SD | 0.4 ± 0.2 | 14.7 ± 0.2 | 15.1 ± 0.1 | 16.8 ± 0.2 |
| p value # | ------- | 9.5 × 10−7 | 1.1 × 10−5 | 3.9 × 10−7 | |
| S. aureus | Mean ± SD | 0.2 ± 0.1 | 14.2 ± 0.1 | 14.2 ± 0.1 | 14.1 ± 0.1 |
| p value # | ------- | 7.8 × 10−8 | 7.7 × 10−8 | 7.9 × 10−8 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Mutairi, J.F.; Al-Otibi, F.; Alhajri, H.M.; Alharbi, R.I.; Alarifi, S.; Alterary, S.S. Antimicrobial Activity of Green Silver Nanoparticles Synthesized by Different Extracts from the Leaves of Saudi Palm Tree (Phoenix Dactylifera L.). Molecules 2022, 27, 3113. https://doi.org/10.3390/molecules27103113
Al Mutairi JF, Al-Otibi F, Alhajri HM, Alharbi RI, Alarifi S, Alterary SS. Antimicrobial Activity of Green Silver Nanoparticles Synthesized by Different Extracts from the Leaves of Saudi Palm Tree (Phoenix Dactylifera L.). Molecules. 2022; 27(10):3113. https://doi.org/10.3390/molecules27103113
Chicago/Turabian StyleAl Mutairi, Jihan F., Fatimah Al-Otibi, Hassna M. Alhajri, Raedah I. Alharbi, Saud Alarifi, and Seham S. Alterary. 2022. "Antimicrobial Activity of Green Silver Nanoparticles Synthesized by Different Extracts from the Leaves of Saudi Palm Tree (Phoenix Dactylifera L.)" Molecules 27, no. 10: 3113. https://doi.org/10.3390/molecules27103113
APA StyleAl Mutairi, J. F., Al-Otibi, F., Alhajri, H. M., Alharbi, R. I., Alarifi, S., & Alterary, S. S. (2022). Antimicrobial Activity of Green Silver Nanoparticles Synthesized by Different Extracts from the Leaves of Saudi Palm Tree (Phoenix Dactylifera L.). Molecules, 27(10), 3113. https://doi.org/10.3390/molecules27103113







