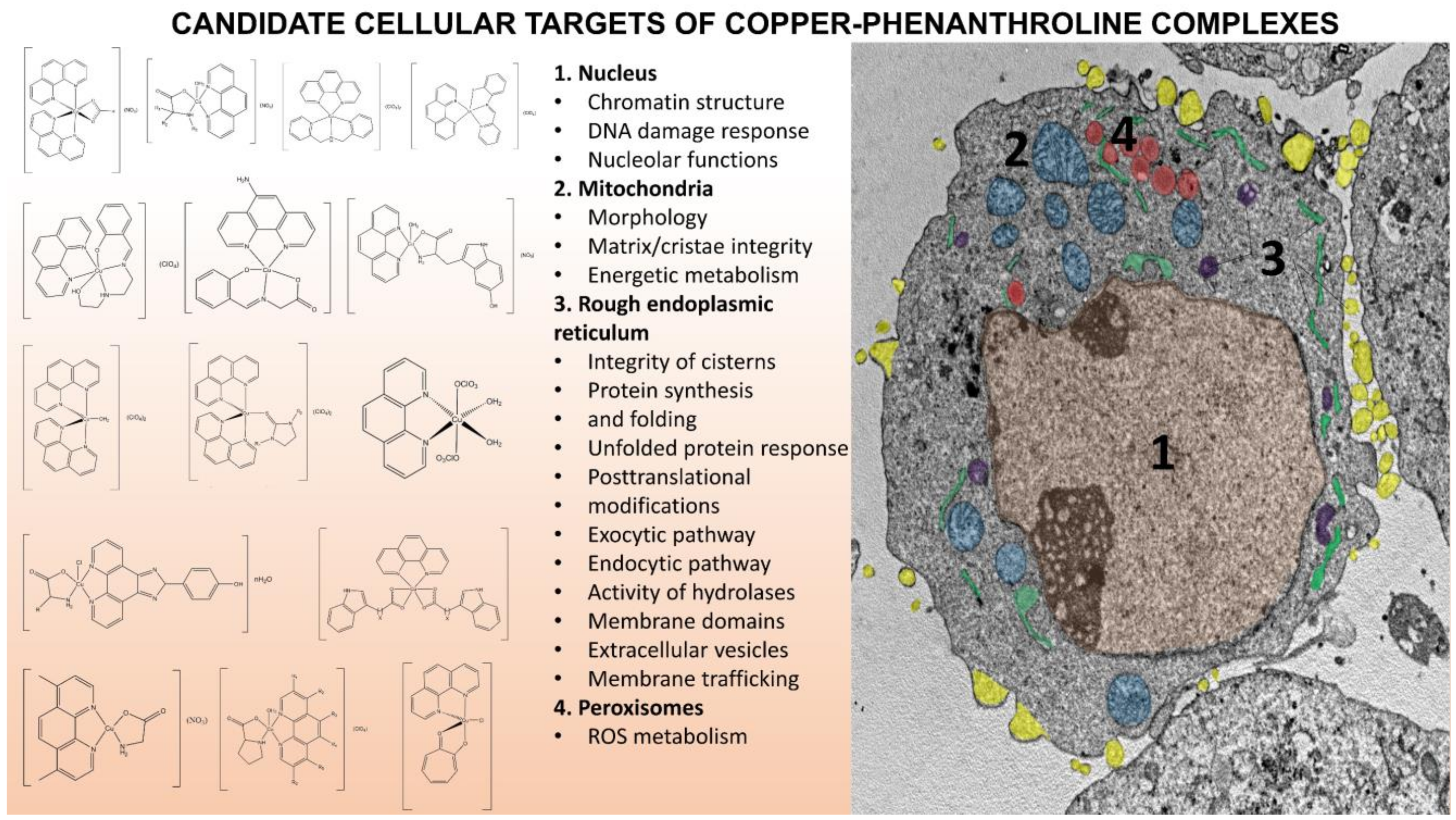
2.1. Inorganic Auxiliary Ligands
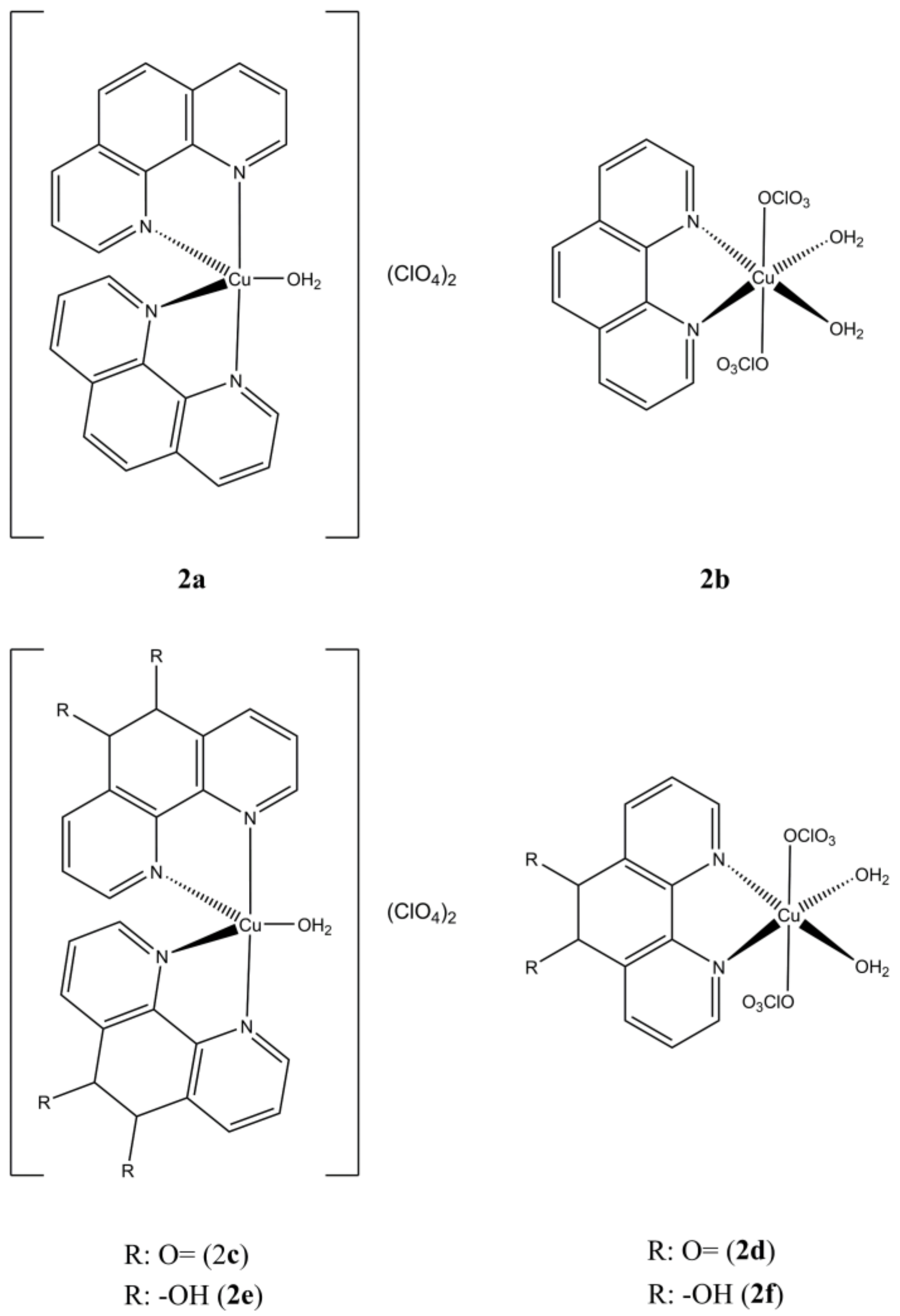
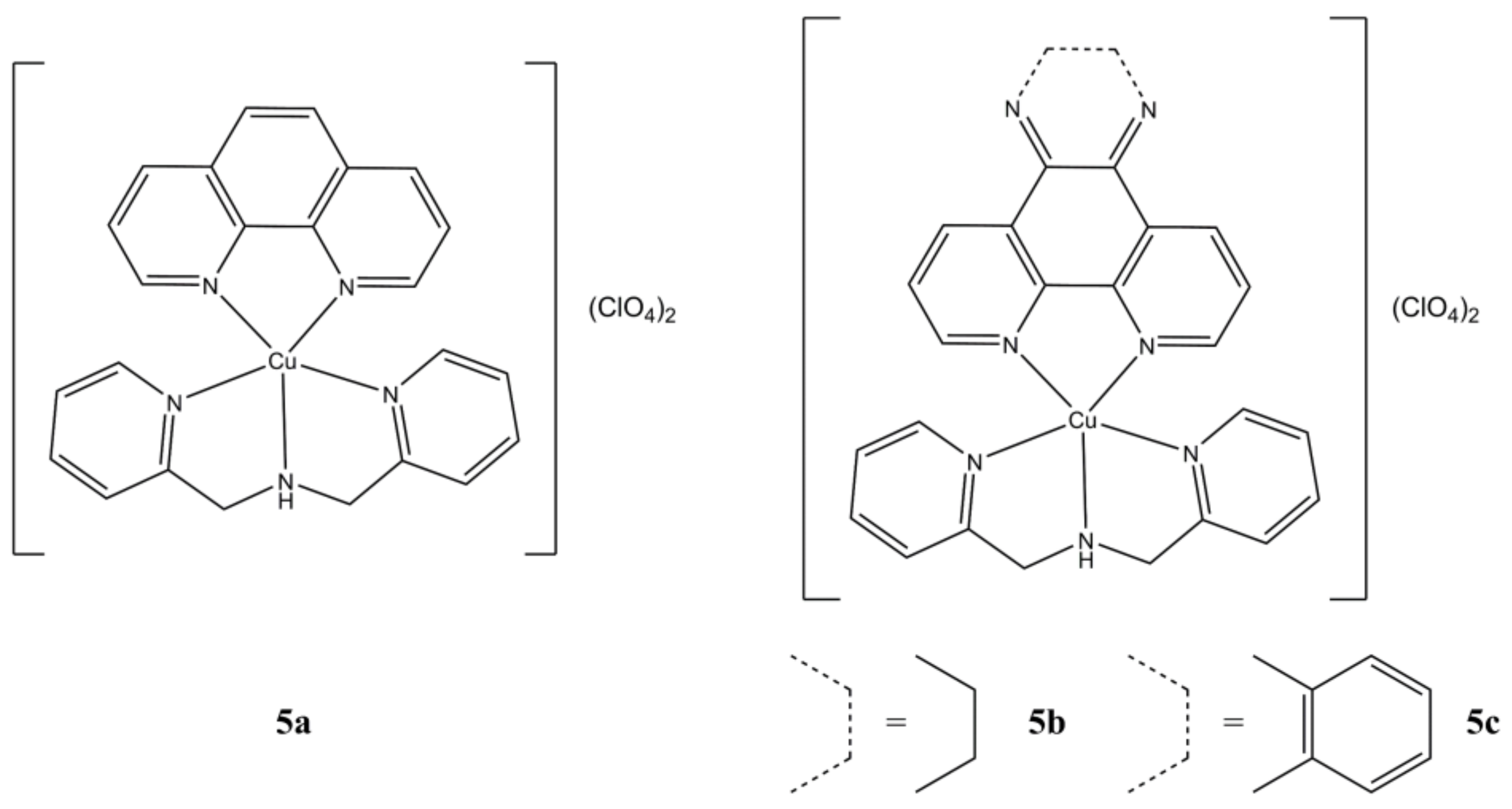
Mixed Cu(II) phenanthroline-based complexes having general formula Cu(N-N
1)
x(OH
2)
y(ClO
4)
z (
Figure 2), where “N-N
1” are phen and some 5,6-disubstituted derivatives, were prepared and tested on a panel of solid and haematological cancer cells [
24,
25,
26].
All the studied complexes show IC
50 in the micro/sub-micromolar range, with complexes having two N-N
1 units that appeared more potent compared to complexes having only one N-N
1 unit. The potential selectivity, calculated taking the normal fibroblasts as reference, varies from a minimum 0.78 to a maximum of 19.2 according to the type and number of the phen-based moieties and the cancer cell line considered. All the studied compounds can interact with DNA, but the inversely correlated relation between DNA binding constants and anticancer potencies, brings to exclude that the biological properties observed would arise from a direct interaction with this target [
25]. Recent results have partially unveiled the biological mechanism in ovarian (A-2780) cells of
2a, which induces Endoplasmic Reticulum (ER) Stress by activating the pro-apoptotic branch of the Unfolded Protein Response (UPR), as observed by overexpression of typical biomarkers, such as PERK, IRE1 and DDIT3, and alleviation of cytotoxicity in co-administration with ER-stress modulator Taursodeoxycholic Acid (TUDCA) [
27].
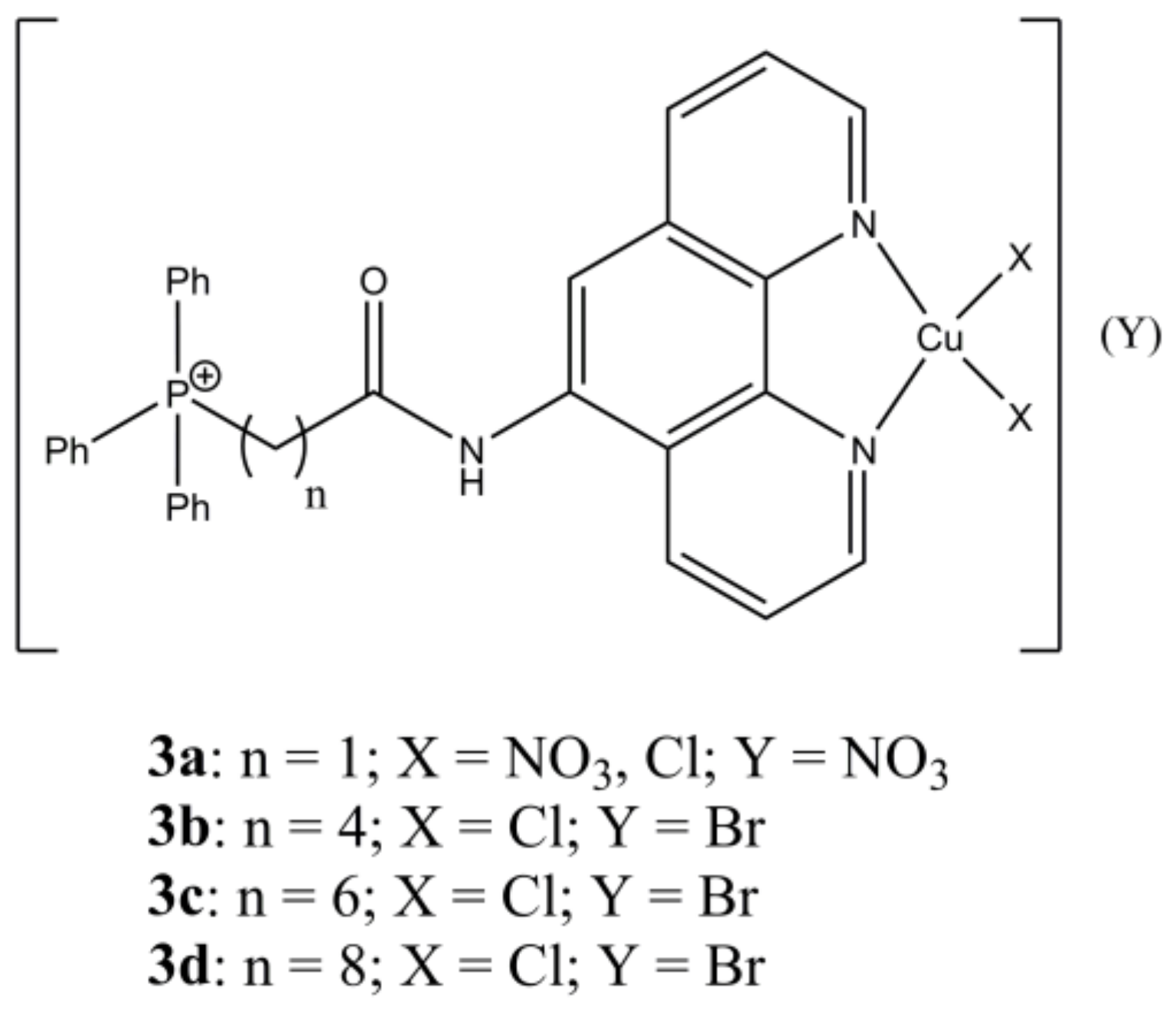
Shi et al. have synthesized a panel of mixed complexes having general formula [Cu(LPTn)
x(X)
y](Y)
z (
Figure 3), where LPTn are phenanthroline derivatives having alkyl chains of different lengths.
Both cellular uptake and activity on cervical (Hela) and ovarian (SKOV-3) cancer cells appear directly correlated with the lipophilicity of the LPTn ligand, while all of them showed mild toxicity towards healthy HK-2 cells. Intracellular ROS production and lipophilicity show a different correlation depending on the cell line, suggesting that the redox properties are not the determinant factors in the cell death induction. The most promising complex of the series (
3d) has shown to possess anti metastatic activity, via inhibition of MMP-2 expression, in SKOV-3 cells. In addition, it also possesses antiangiogenic activity, via downregulation of VEGFR-1 expression, blocking of sprouts and tubes formation, in HUV-EC-C cells [
28].
Table 1 summarizes the IC
50 values reported for the compounds so far discussed.
2.4. Organic-O Donating Auxiliary Ligands
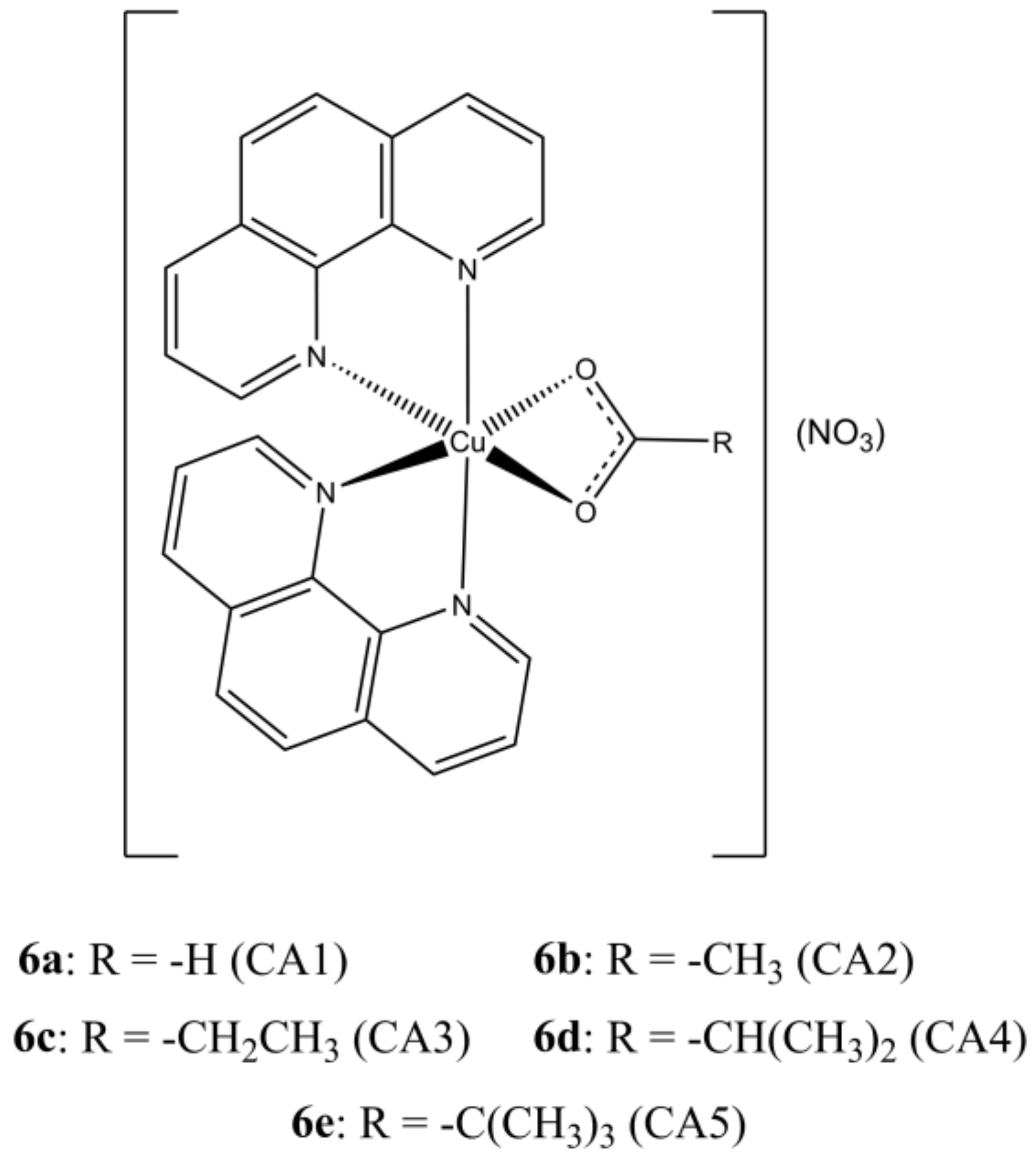
With the aim of enhancing the target specificity of [Cu(phen)
2](NO
3)
2, Prisecaru et al. introduced some simple carboxylic acids in the [Cu(phen)
2]
2+ core obtaining a small library of mixed Cu(II) complexes having general formula [Cu(phen)
2(CAn)](NO
3) (
Figure 6) [
31].
The introduction of an auxiliary carboxylate moiety allowed to increase the affinity towards ct-DNA while reducing that towards albumin. The studied complexes show DNA cleavage activity upon activation by an exogenous reductant (sodium ascorbate) and oxidant (H2O2), which is believed to arise by a combination of a SOD mimetic activity with OH· production by Fenton reaction. DNA damage was also evidenced in SKOV-3 cells by accumulation of phosphorylated γ-H2AX. The studied compounds show IC50 values in the sub-micromolar range.
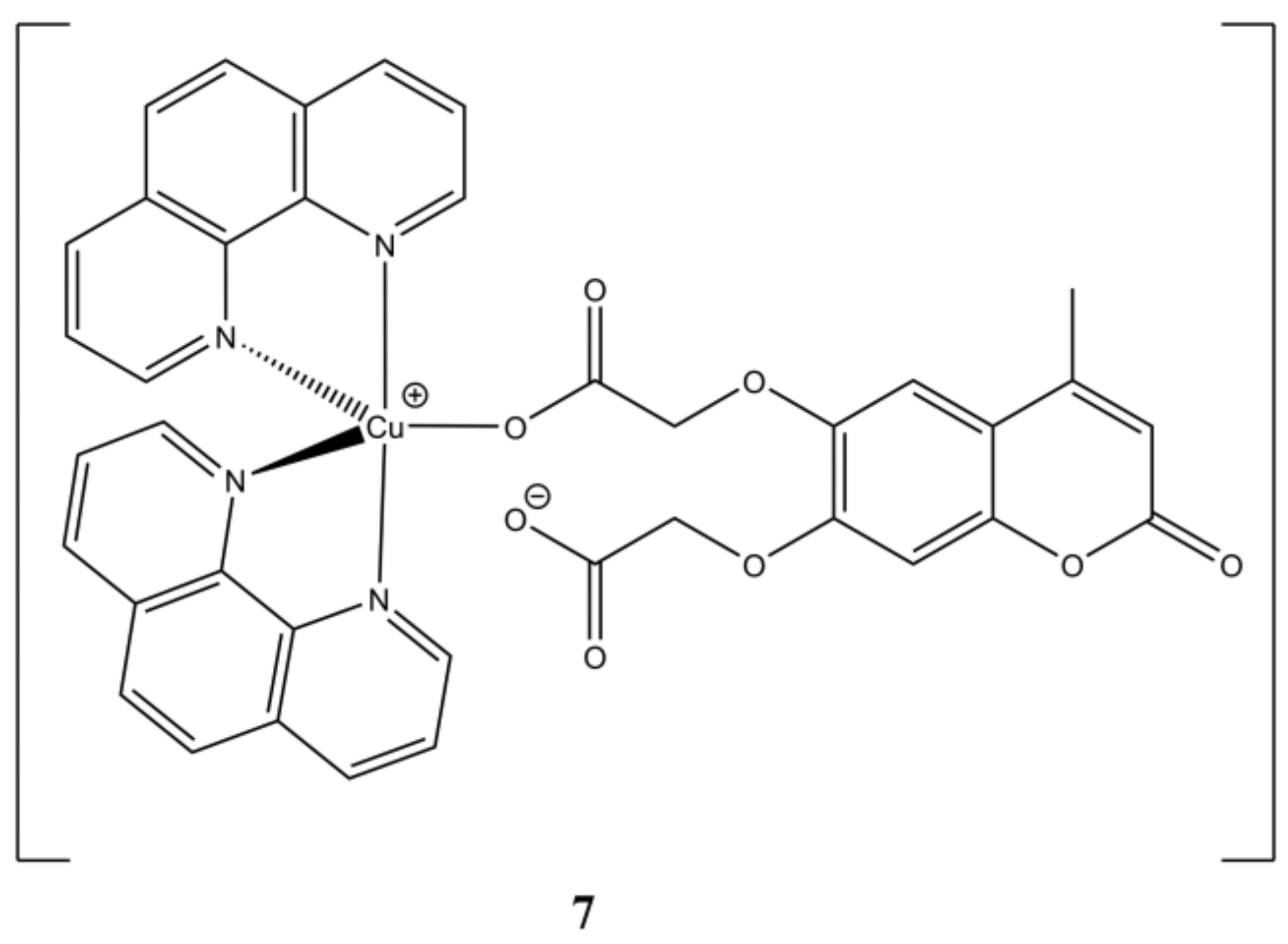
The mixed Cu(II) complex [Cu(phen)
2(4-Mecdoa)] (
Figure 7) is active towards kidney (A-498) and liver (Hep-G2) cancer cells compared to cisplatin, while being less sensitive towards non-cancerous CHANG cells [
32]. However, the same behaviour was not observed on healthy HK-2 lines, this complex appears to interfere with DNA synthesis without any direct interaction with DNA itself. Cell death induction is switchable from apoptosis to necrosis in a dose-dependent manner.
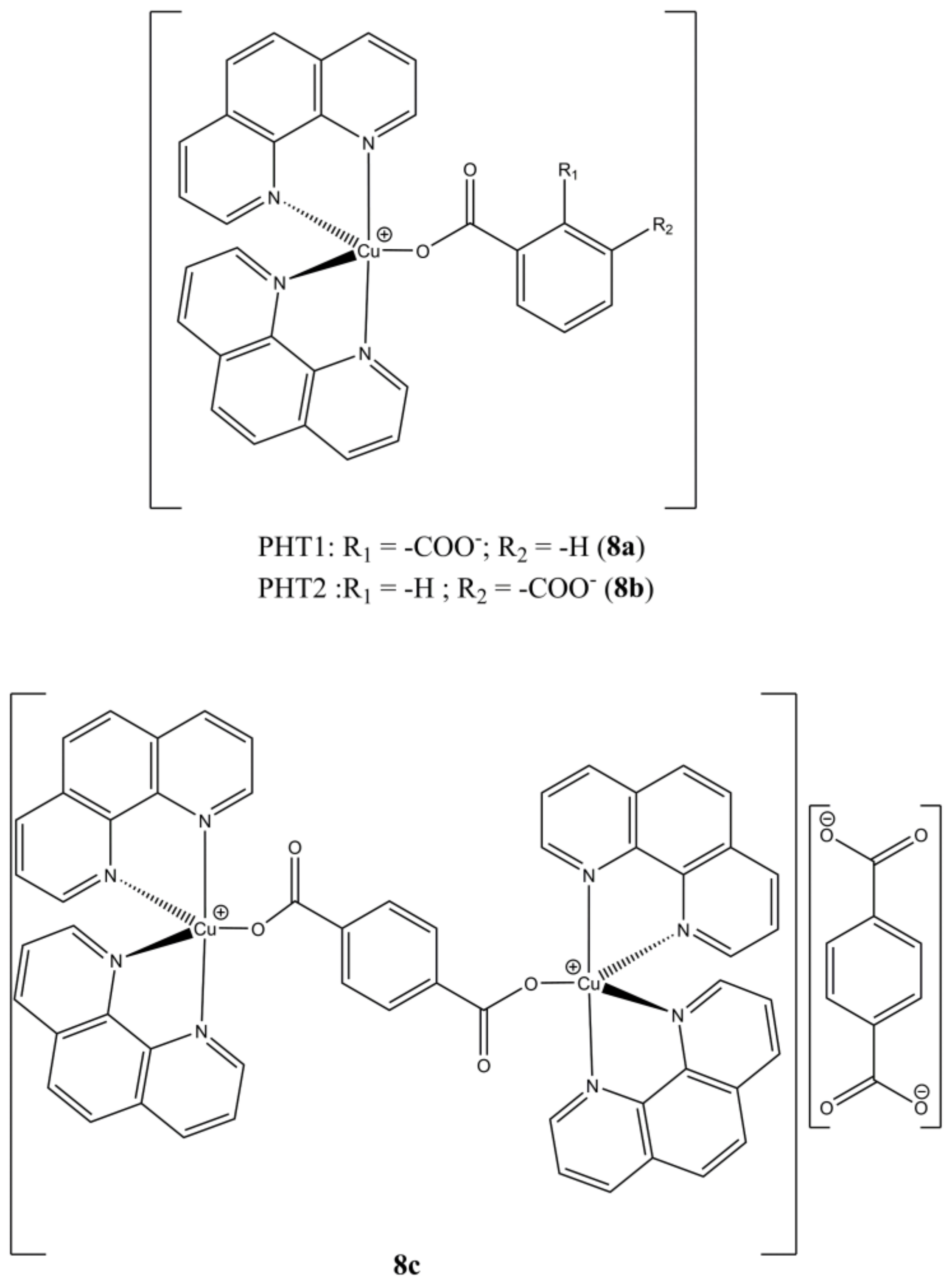
Kellet et al. used ftalic acids to prepare the mixed mononuclear [Cu(phen)
2(PHTn)] (n = 1,2;
Figure 8a,b) and binuclear [Cu
2(phen)
4(µ-PHT3)](PHT3) (
Figure 8c) complexes [
33].
These compounds show higher anticancer potencies (at micromolar level) in breast (MCF-7), prostate (DU145) and colon (HT29) cancer cells compared to the approved drugs cisplatin and mitoxantrone, while the Cu(II) complexes of the same PHTn ligands are devoid of any activity. In addition, they can cleave DNA without the presence of exogenous oxidant or reducing agents.
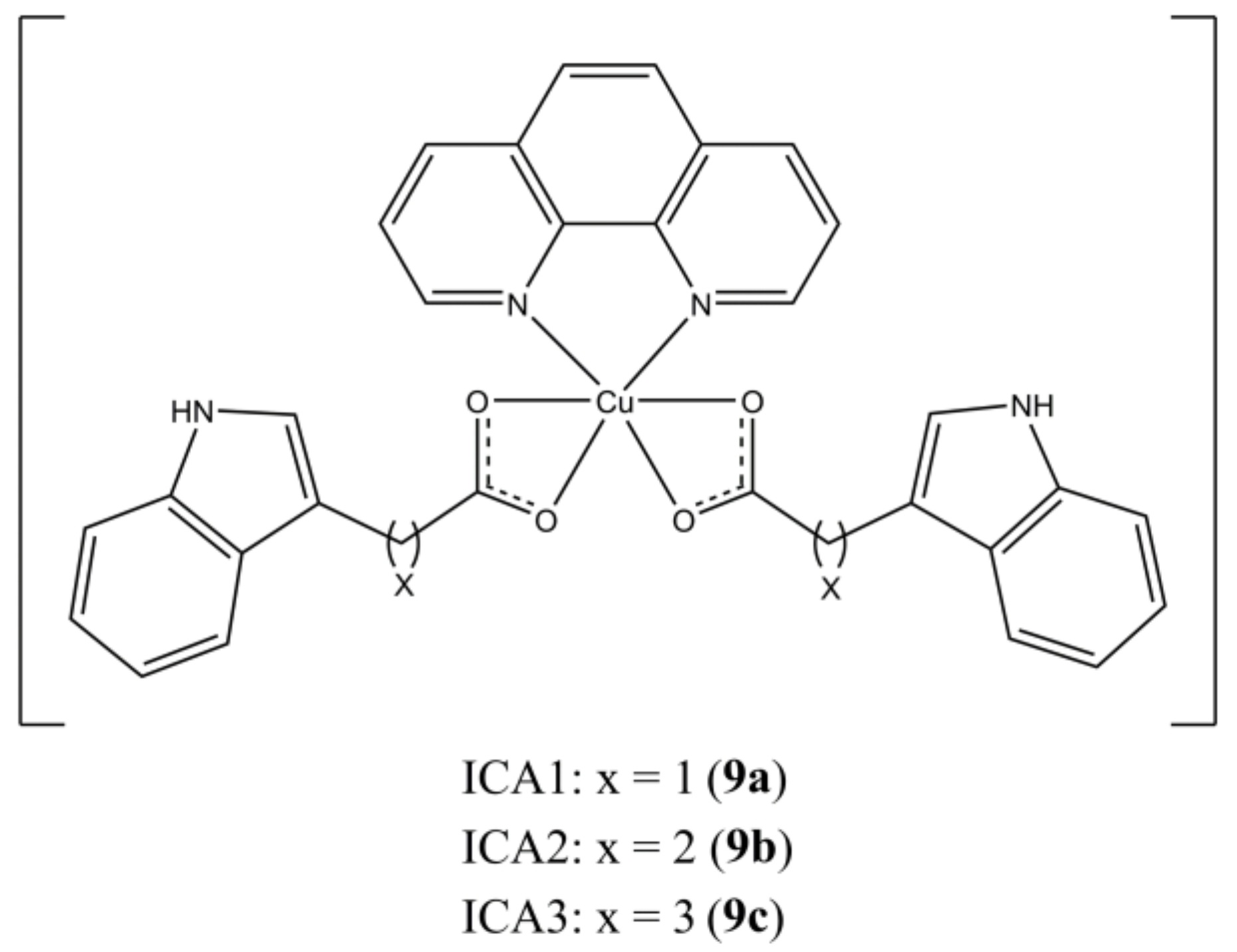
Zhang et al. reported the synthesis and anticancer activity of mixed Cu(II) complexes having general formula [Cu(ICAn)
2(phen)], where ICAn are indole 3-carboxylic acids having different alkyl spacers (
Figure 9) [
34].
These complexes can induce apoptosis in MBA-MDB-231 breast tumours by targeting the Ubiquitin Proteasome Pathway (UPP). It is known that cancer cells are more sensible to UPP inhibition than healthy ones, due to the involvement of this pathway in many carcinogenic processes (e.g., proliferation, apoptosis, and metastasis). In particular, these complexes act at proteasome level by inhibiting the chymotrypsin-like (CT) activity of human 20S proteasome.
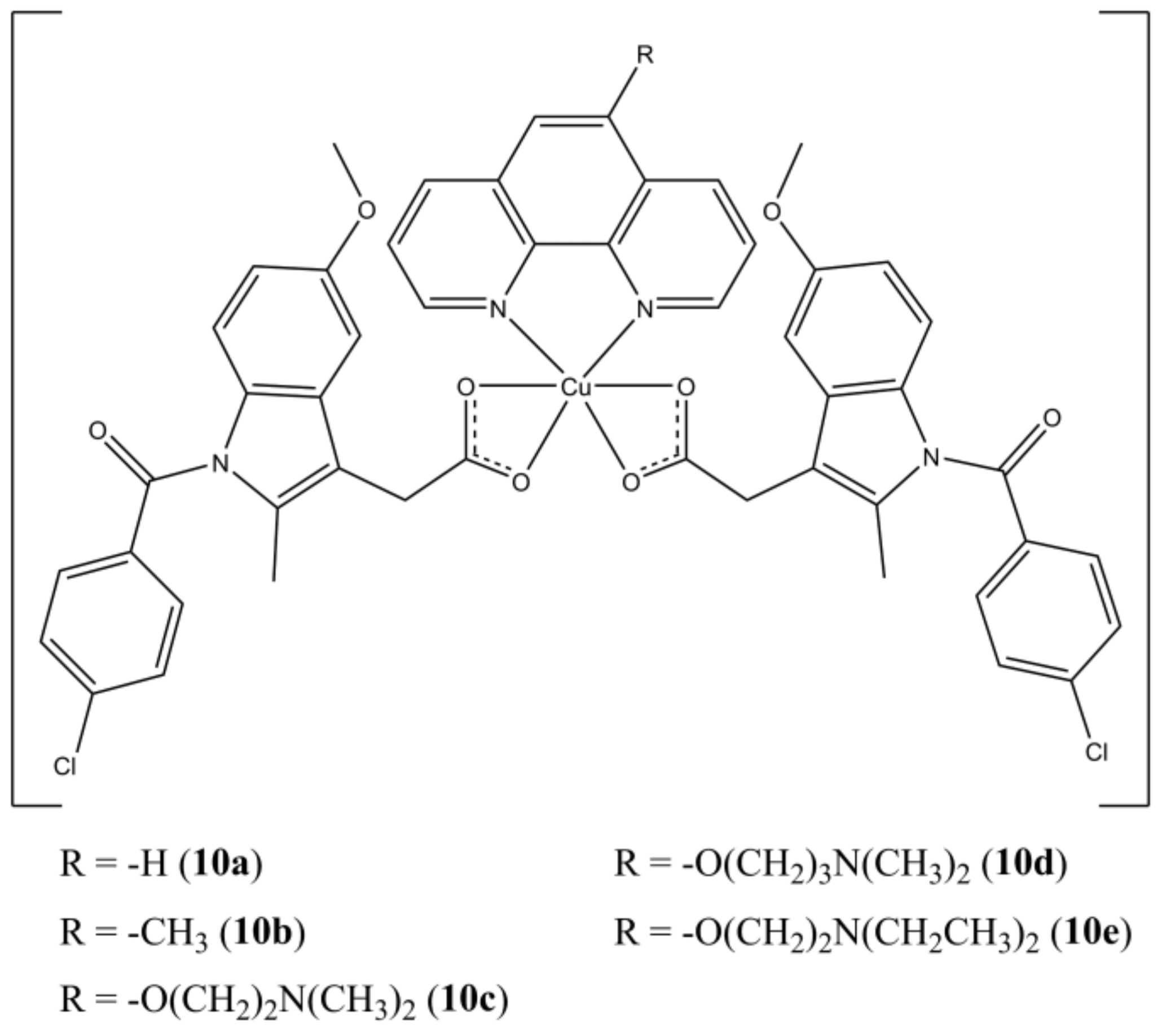
Boodram et al. prepared a panel of mixed complexes [Cu(indomethacin)
2(5-Rphen)], where the Cu(II) centre is coordinated by the NSAID drug Indomethacin and different 5-substituted phenanthrolines (
Figure 10) [
35].
The studied complexes are active towards HMLER breast cancer cells at micromolar level. Notably, complexes 10c,d show selectivity for HMLER-shEcad cells, which possess a higher cancer stem cells (CSCs) content (around 90%) over HLMLER ones (5–8%). The ability of targeting breast CSCs is quite important since these sub-populations of cancer cells are often resistant towards conventional clinical treatments (surgical removal followed by chemo or radiotherapy) and are involved in the resurgence of secondary metastatic breast tumours. The studied compounds can induce DNA damage, as shown by self-activating DNA cleavage activity, cellular ROS production (mainly hydroxyl radicals), and accumulation of γ-H2AX. In addition, they are to target cyclooxygenases by selectively inhibiting COX-2, whose expression is enhanced in several mammary carcinomas.
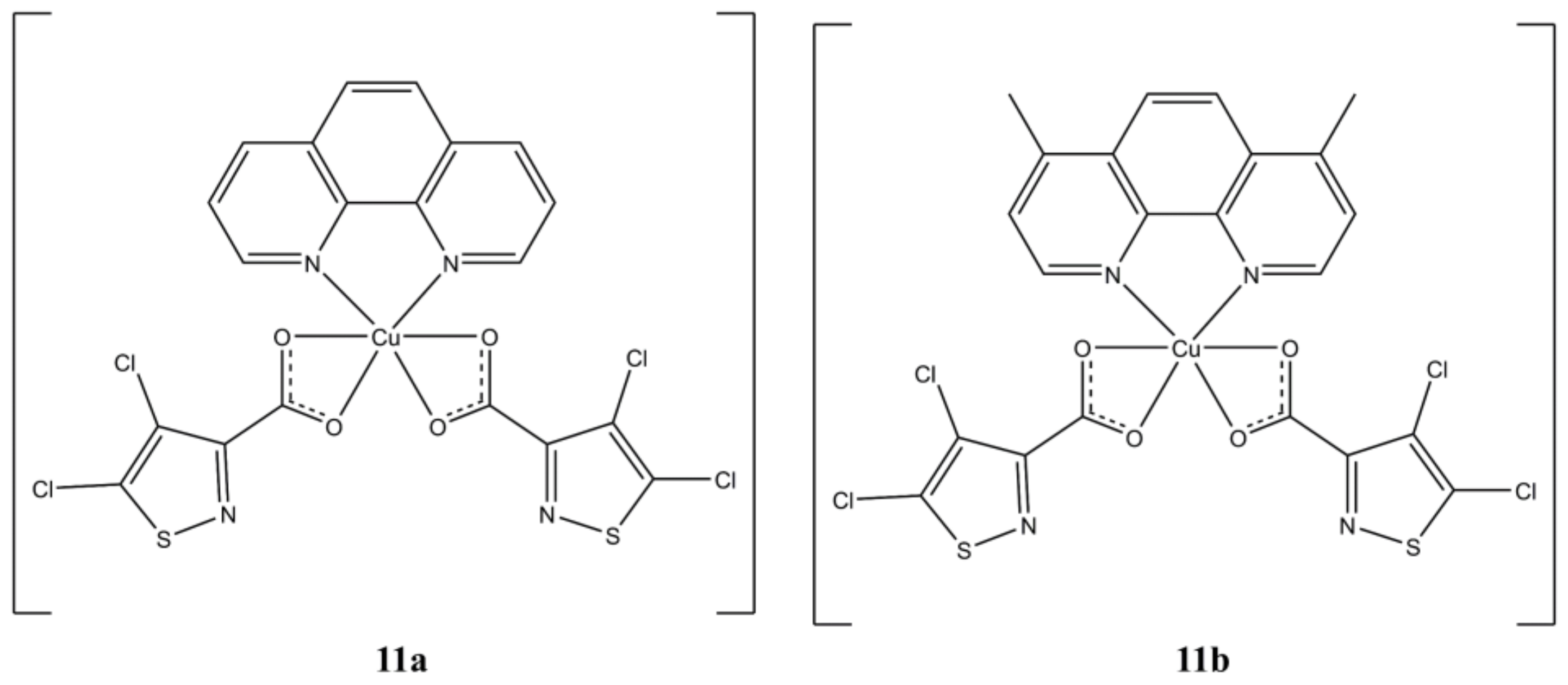
In a series of homo and heteroleptic Cu(II) complexes with 4,5-dichloro-isothiazole 3-carboxylic acid (4,5-dCl-ICA), compounds [Cu(phen)(4,5-dCl-ICA)
2] and [Cu(4,7-diMephen)(4,5-dCl-ICA)
2] (
Figure 11) proved to be the most potent towards Hep-2 (IC
50 of 3.06 and 0.97 µM, respectively) and MCF-7 (IC
50 of 4.2 and 1.8 µM, respectively) cells [
36].
The morphological changes observed in cells after treatment with these compounds are indicative of an apoptotic cell death. These complexes can interact with DNA (with Kb in agreement with cytotoxic data) but are also able to inhibit the activity of different Cytochrome P450 families in a dose-dependent manner.
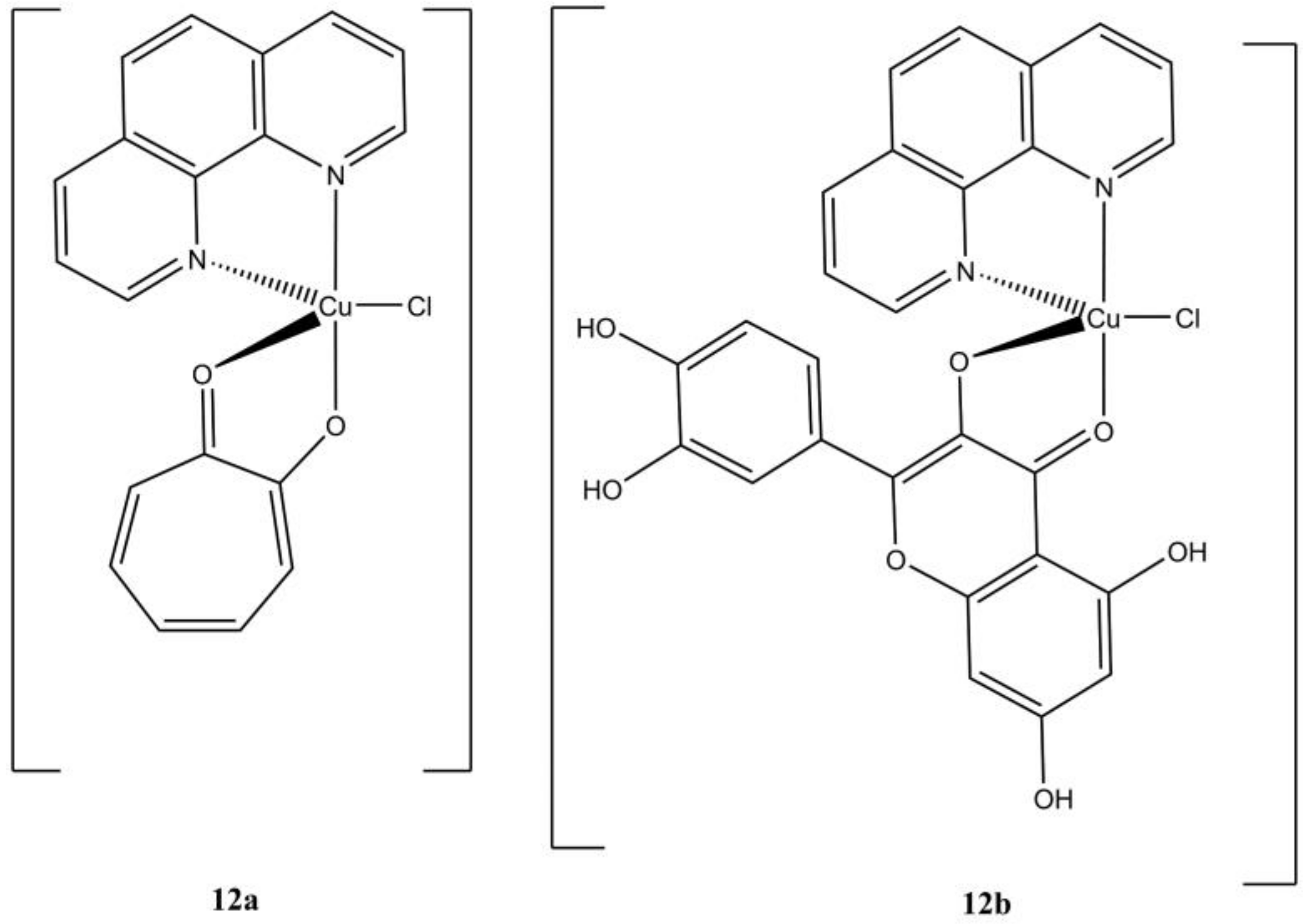
The complex [Cu(phen)(trop)(Cl)], bearing a tropolone molecule as auxiliary ligand (
Figure 12a), showed the highest anticancer potency (at micromolar level) among a series of binary and ternary Cu(II) tropolone-based complexes [
37]. This compound induces cell death in gastric (MGC80-3) cancer cells mainly through a caspase-regulated apoptotic pathway with the induction of ROS production, alteration of the mitochondrial potential, overexpression of many apoptotic signallers (e.g., Bax, cytochrome C, Bak, apaf1). The induction of autophagy in the same cell lines is also observed.
In a screening of novel M(II) (Cu(II), Ni(II), Co(II)) quercetin-based anticancer complexes, Gençkal et al. identified the compound [Cu(phen)(H
4Que)(Cl)] (
Figure 12b) as one of the most promising against MCF-7 and MBA-MDB-231 breast cancers. This compound triggers cell death through caspase-mediated apoptosis, with increasing in cellular ROS content and depolarization of the mitochondrial membrane [
38].
The IC
50 for the compounds discussed in this section are summarized in
Table 4.
2.5. Organic Mixed Donating Auxiliary Ligands
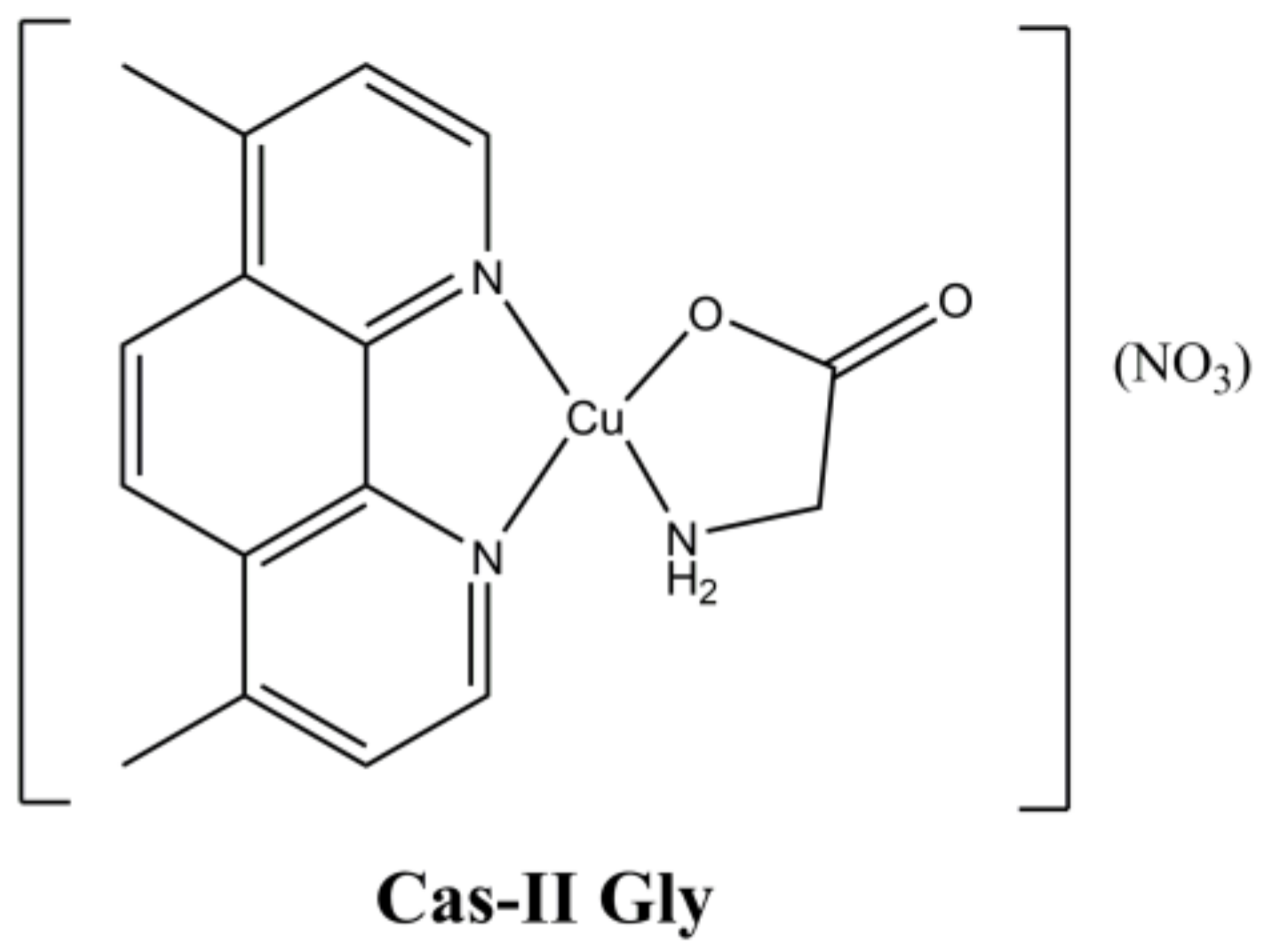
The mixed Cu(II)-phenanthroline complex “Cas-II Gly” [Cu(4,7-diMephen)(Gly)](NO
3) (
Figure 13) belongs to the Casiopeinas
®, a class of complexes having general formula [Cu(N-N)(A-A)](NO
3) (where “N-N” is a neutral di-imine ligand and “A-A” an amino acid) developed by Ruiz-Azuara et al. [
40].
These complexes have been extensively studied on both in vitro and in vivo models, showing that their anticancer activity is exerted through ROS accumulation that causes mitochondrial dysfunction, DNA damage and induces apoptosis [
41,
42,
43,
44,
45,
46,
47]. Some of these derivatives have been selected as candidates for Phase I clinical trials [
48,
49,
50].
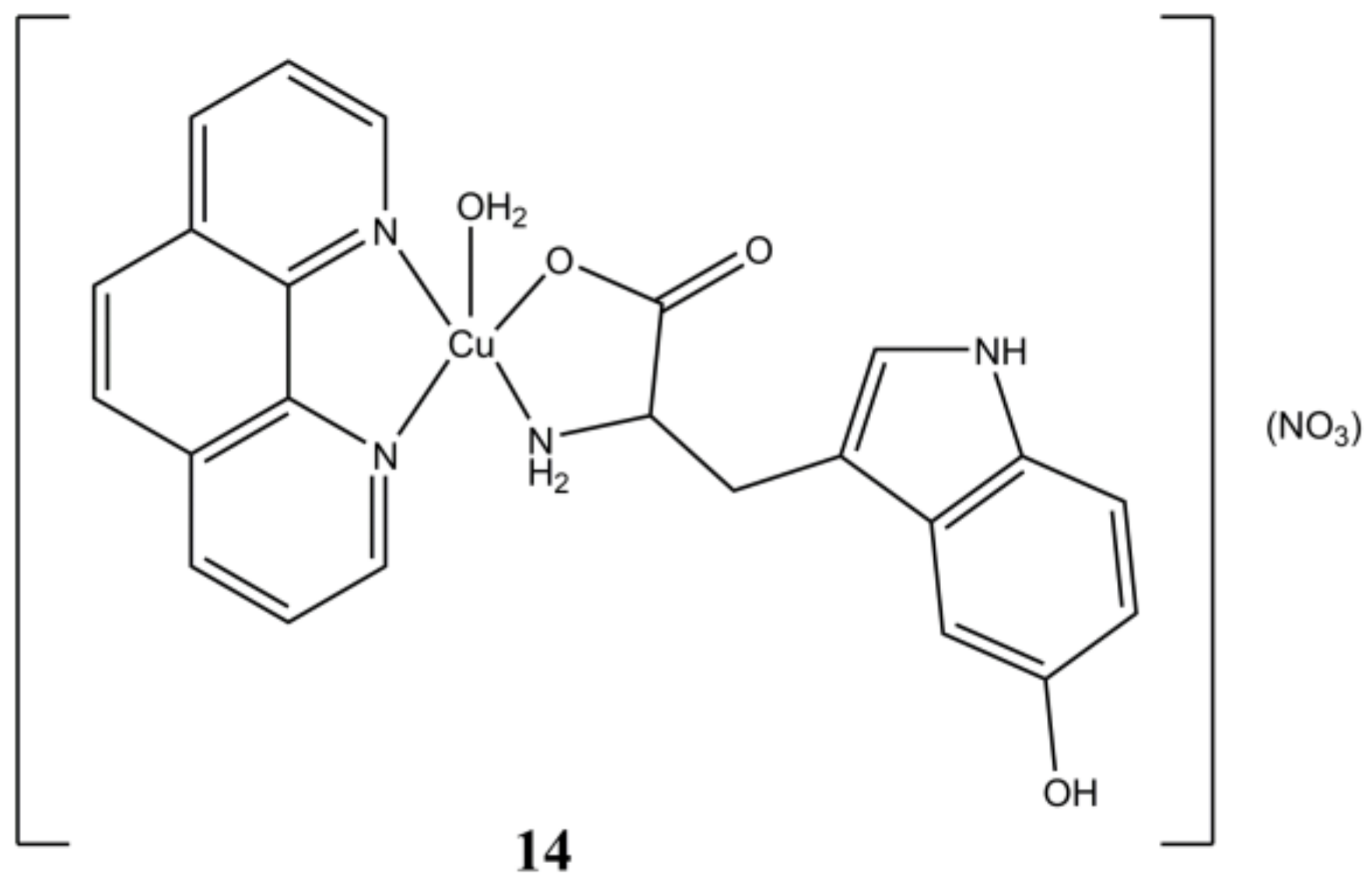
Based on these results, different Cu(II) complexes bearing phen-based ligands and amino acids have been prepared and assayed for their anticancer properties. For instance, the complex [Cu(5HTP)(phen)(H
2O)](NO
3) (5HTP is 5-hydroxytryptophan) reported by Naso et al. (
Figure 14), shows anticancer activity in the micromolar range (IC
50 of 3.6 µM) in A-549 cells, while being devoid of any cytotoxic activity in healthy MRC-5 cells.
The authors demonstrated how the anticancer activity of this compound is exerted through cellular ROS production, GSH depletion and alteration of mitochondrial potential. The complex shows also antimetastatic activity on A-549 cells with inhibition of cells adhesion, migration, and invasion [
51].
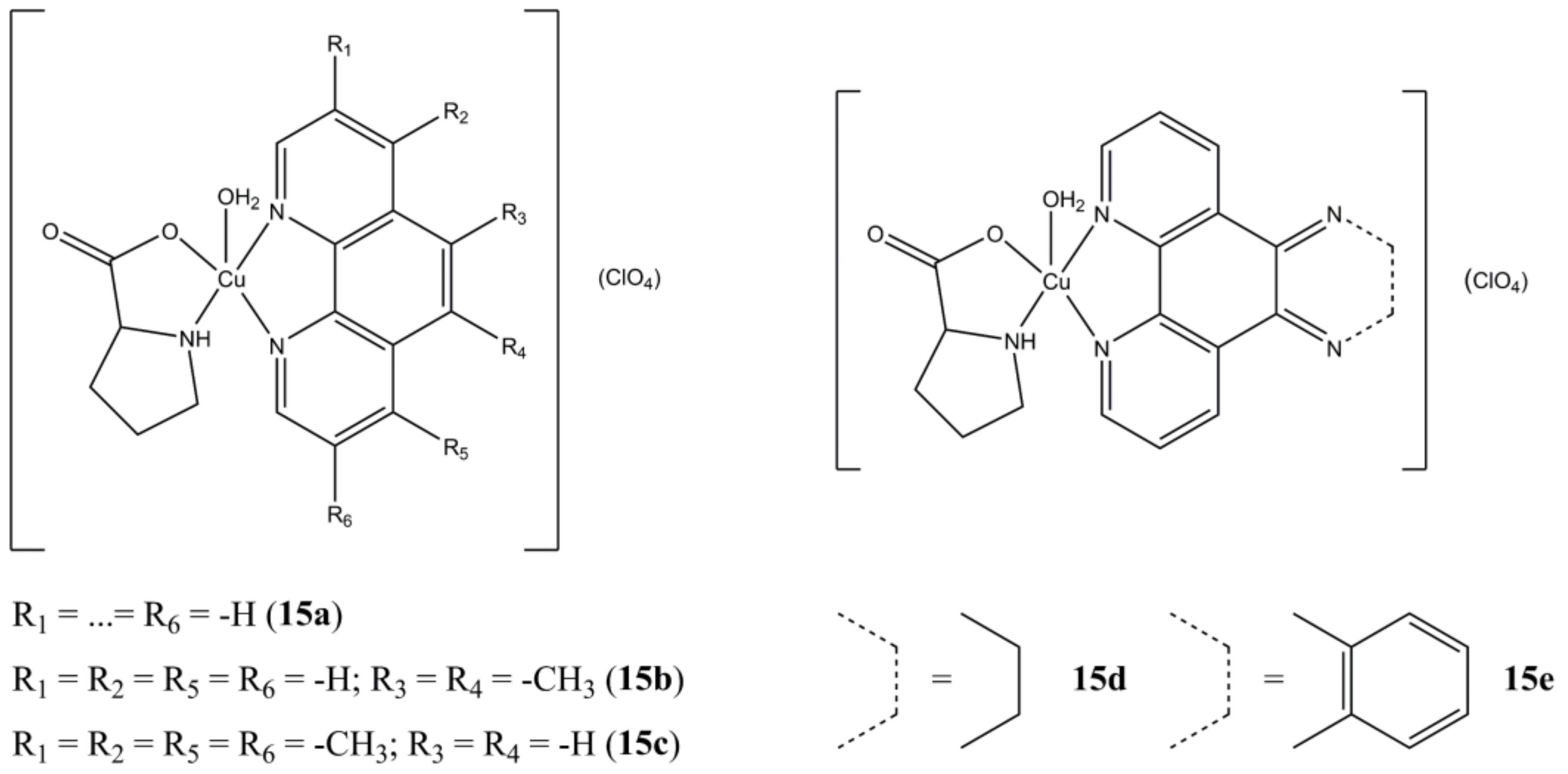
Karpagam et al. prepared a series of Cu(II) Proline-based complexes having general formula [Cu(L-pro)(N-N
3)(H
2O)
n](ClO
4) (n = 0,1) [
52]. The insertion of a phenanthroline-based ligand (
Figure 15) generally increases the anticancer potency (A-549 cell lines) compared to the bipyridine-based analogues. Compounds
15b and
15d proved to be the most promising in terms of potency (IC
50 of 1.4 and 1.3 µM, respectively), showing the importance of the substitution pattern in the 5th and 6th position of the phenanthroline backbone. Cell death induction mainly take place through apoptosis induction, with cellular overexpression of ROS.
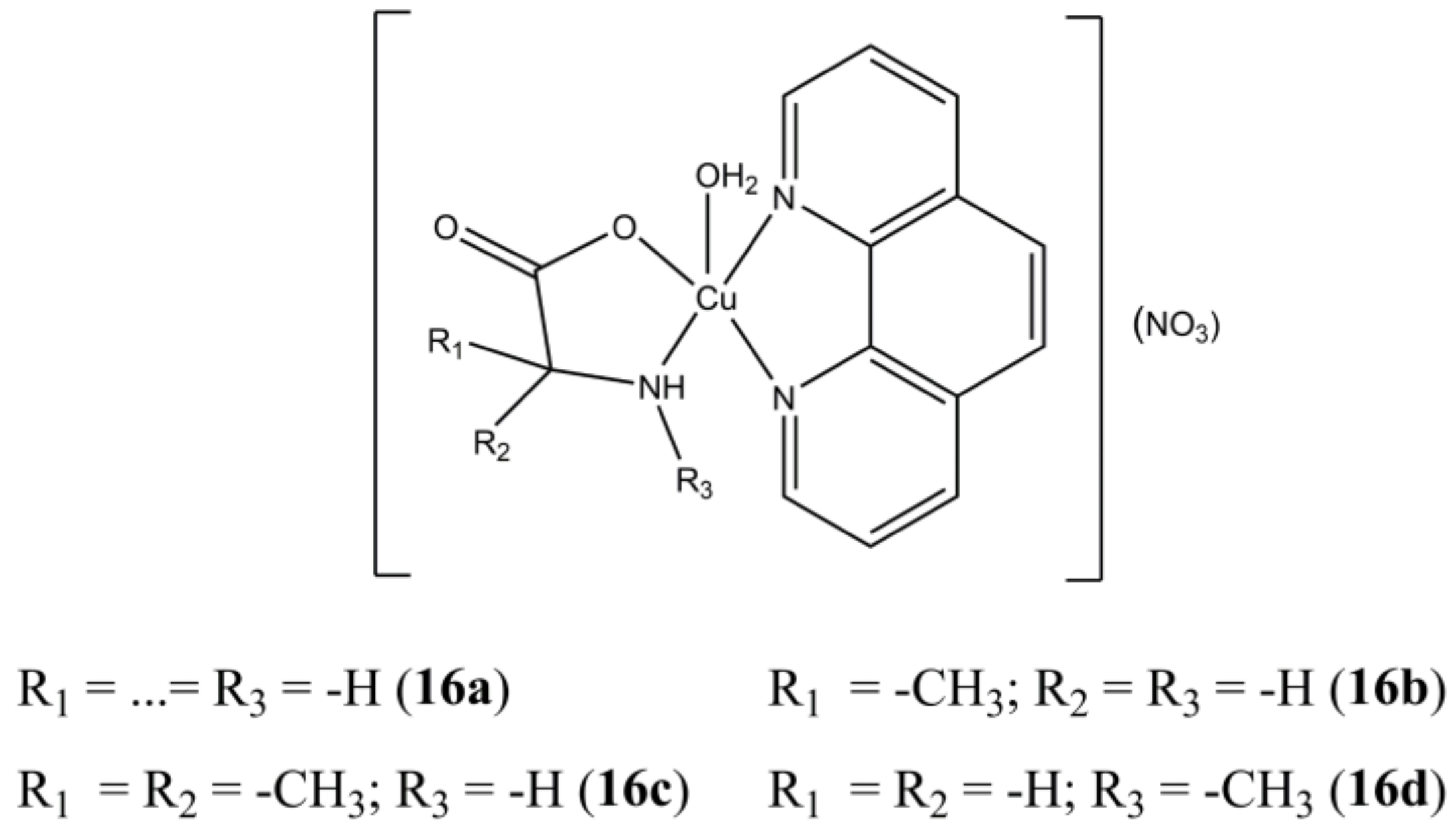
The panel of complexes [Cu(phen)(AAn)(H
2O)](NO
3) (“AAn” are glycine and different methylated glycine derivatives) developed by Seng et al. (
Figure 16) show selectivity towards cancerous nasopharyngeal HK1 cells rather than healthy NP69 ones [
53]. Derivatives
16c and
16d are the most active (IC
50 of 2.2 µM for both) and selective (Selectivity Index SI > 11.4). The studied compounds show moderate affinity towards ct-DNA, with preferential interaction towards G-C sites. Inhibition towards DNA Topo1 in a dose-dependent manner is also evidenced.
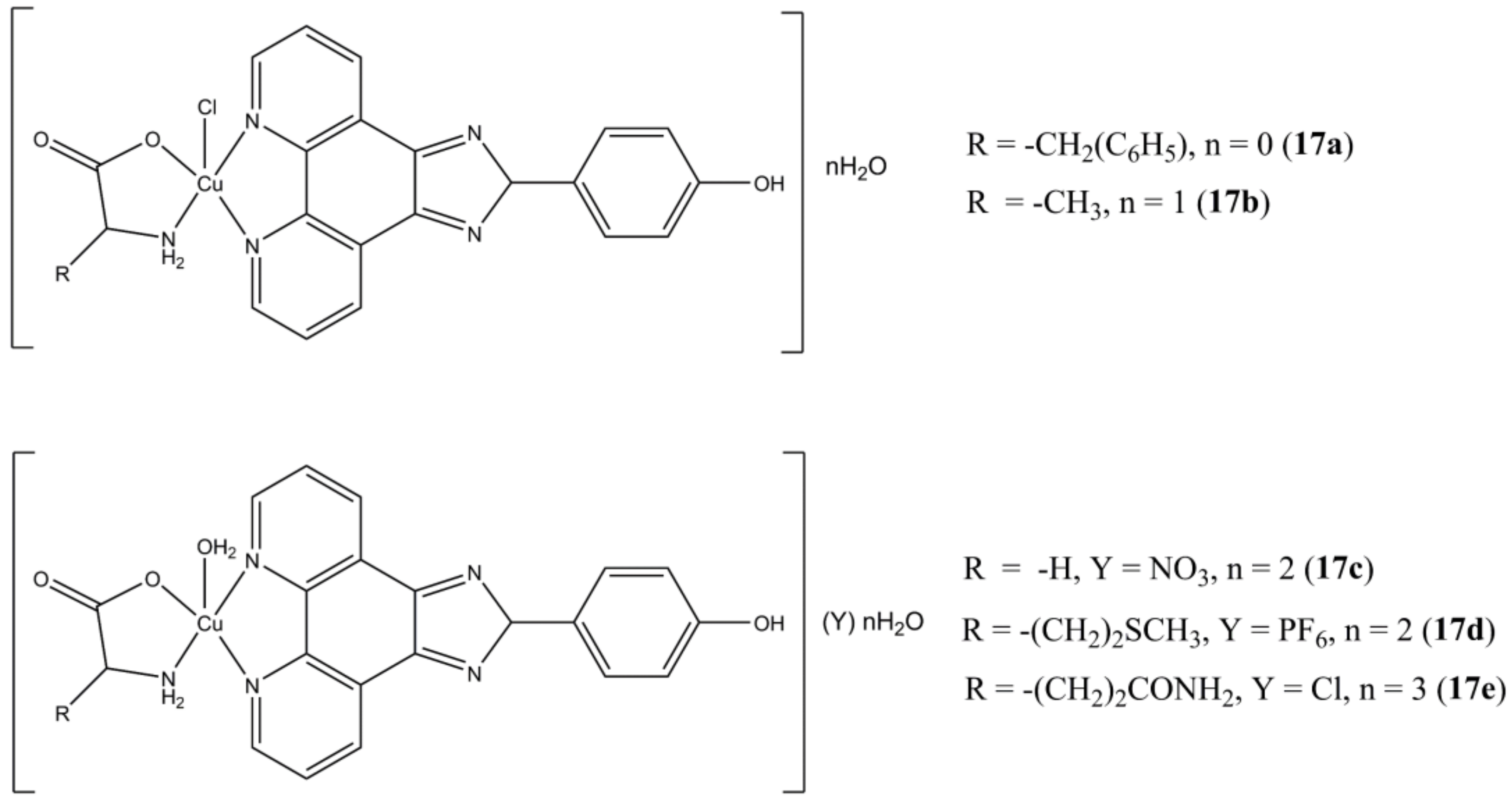
Li et al. prepared a series of heteroleptic Cu(II) complexes having different α-amino acids and the fused phenanthroline derivative OH-PIP (4-(2H-imidazo [4,5-f][1,10]phenanthrolin-2-yl)phenol) as ligands (
Figure 17) [
54].
These compounds show anticancer potency at micromolar/sub-micromolar concentrations against triple negative (MCF-7, MDA-MB-231, CAL-51) breast cancers. The best activity was observed for CAL-51, with IC50 values in the 0.082–0.69 μM range. The complexes here reported show higher potencies compared to clinically approved carboplatin and induces apoptotic cell death by interfering at UPP level (inhibition of CT-like activity of 20S proteasome, PARP cleavage). The most promising complexes of the series (17c and 17e) are also able to significantly reduce the percentage of different triple negative stem cancer sub-populations.
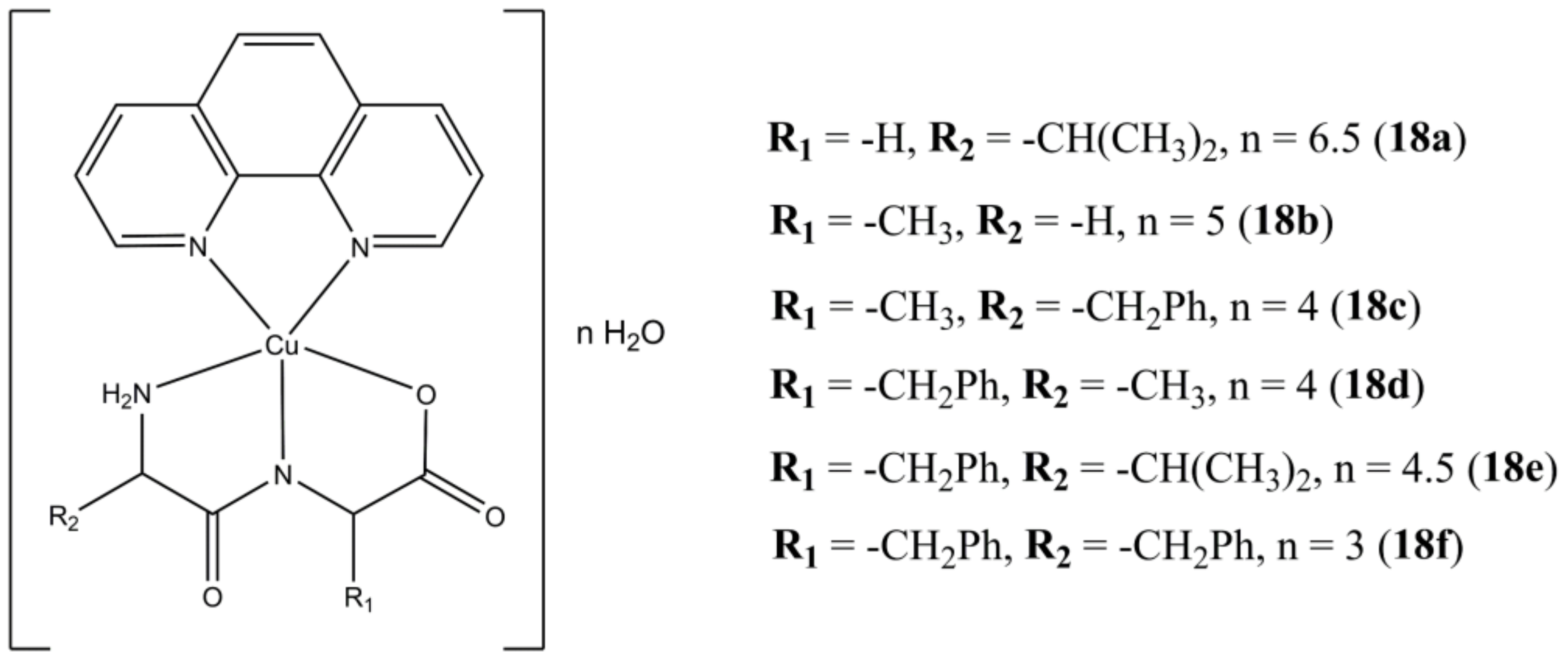
Facchin et al. have been focusing on the design and synthesis of ternary Cu(II) complexes bearing phen-based molecules and L-dipeptides [
55]. For instance, compounds of general formula [Cu(L-dipeptide)(phen)]·nH
2O (
Figure 18) have been screened for anticancer activity in cervical (Hela), breast (MCF-7) and lung (A-549) cancer cells showing anticancer potencies in the micromolar concentration range [
56]. Preliminary studies on Albumin and ct-DNA interaction with the studied molecules show low to moderate affinity towards these biomolecules, with ct-DNA binding constants that appear to not be correlated with neither cytotoxicity nor lipophilicity. Interestingly, the author experimentally evaluated the lipophilicity of these molecules, pointing out how this parameter is influenced not only by the number and types of apolar groups (e.g., methylene, phenyl rings), but also on the spatial arrangement of the complexes, as evidenced for compounds
18c and
18d, where the auxiliary ligands are the Ala-Phe and Phe-Ala L-dipeptides, respectively.
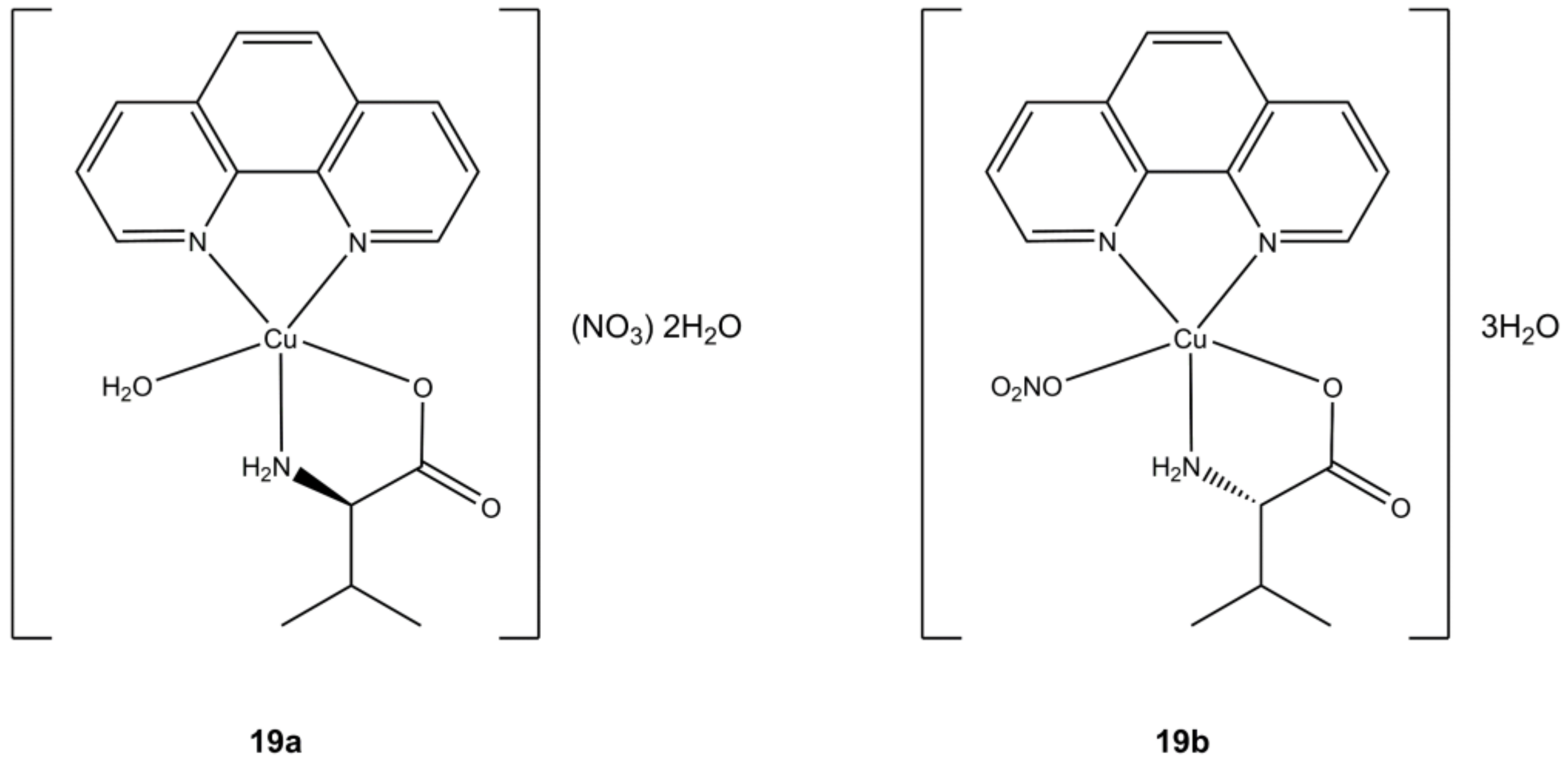
The chiral Cu(II) complexes [Cu(phen)(L-Val)(OH
2)](NO
3)∙2H
2O (
Figure 19a) and [Cu(phen)(D-Val)(ONO
2)]∙3H
2O (
Figure 19b) of Arjmand et al. shows anticancer potency in the micromolar order on breast (MCF-7), pancreatic (BxPC3, AsPC1) and liver (Huh7) cancer cells. Notably, these compounds are able to target G-quadruplex DNA (G4 DNA) by cleaving it at selective sites. In general, compounds that can interfere at G4 DNA level (G4 ligands) are preferred since this interaction will result in telomerase inhibition, whose activity is up regulated in cancer cells while being silent in healthy ones. The inhibition of this enzymatic pathway will result in accumulation of shorter telomers and subsequent induction of apoptosis [
57].
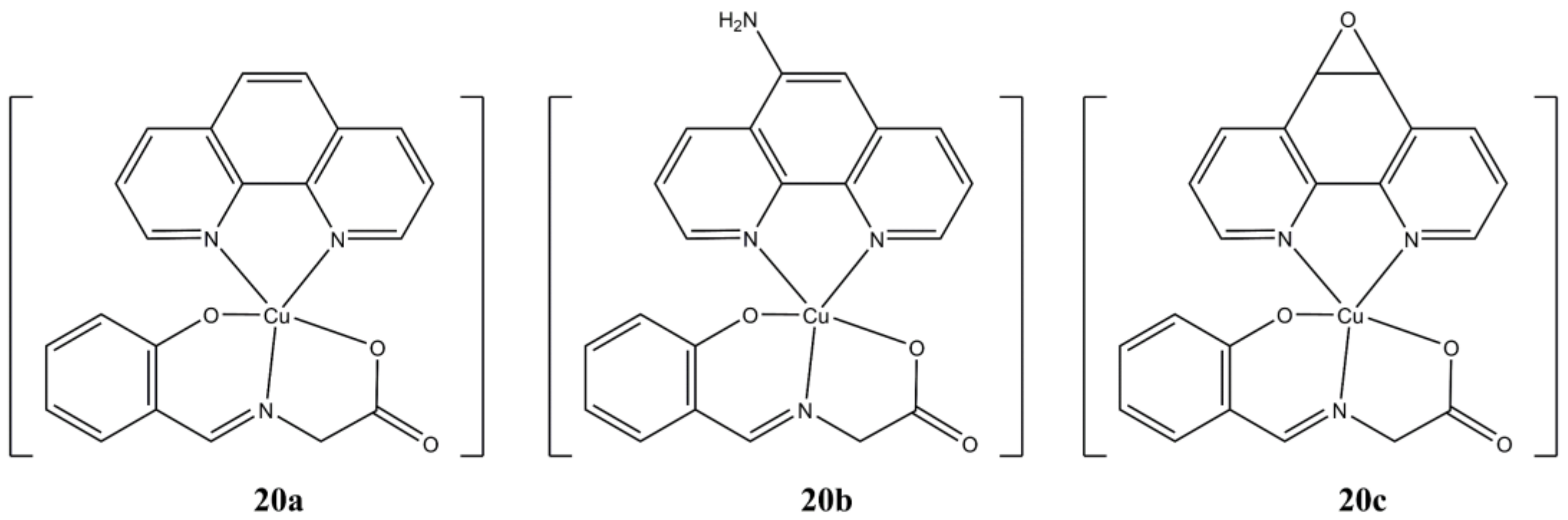
Acilan et al. prepared a series of [Cu(Sal-Gly)(N-N
4)] complexes (
Figure 20), where Sal-Gly is the
O,N,O Schiff Base obtained from Salicylaldehyde and Glycine, while the N-N
4 ligands are phen, 1,10-phenanthroline-5-amine, 5,6-diepoxy-1,10-phenanthroline [
58].
The complexes show dose and time-dependent micromolar cytotoxicity towards cancerous (A-549, HCT-116, HeLa, MBA-MB-231 and SHSY5Y) cells, while generally being less active against healthy HASMC1 and HASMC2 cells. Compound 20b appears to be the most promising in terms of both anticancer potency and selectivity. Rapid apoptotic cell death in HCT-116 and HeLa cells was detected with induction of ROS production, depolarization of mitochondrial membrane, depletion of GSH and cellular DNA damage (γ-H2AX expression). Interestingly, gene-knockdown experiments on HeLa cells have shown that apoptotic cell death doesn’t rely on p53 status, thus supporting the hypothesis that these complexes might be potent also in p53 deficient cancer cell lines.
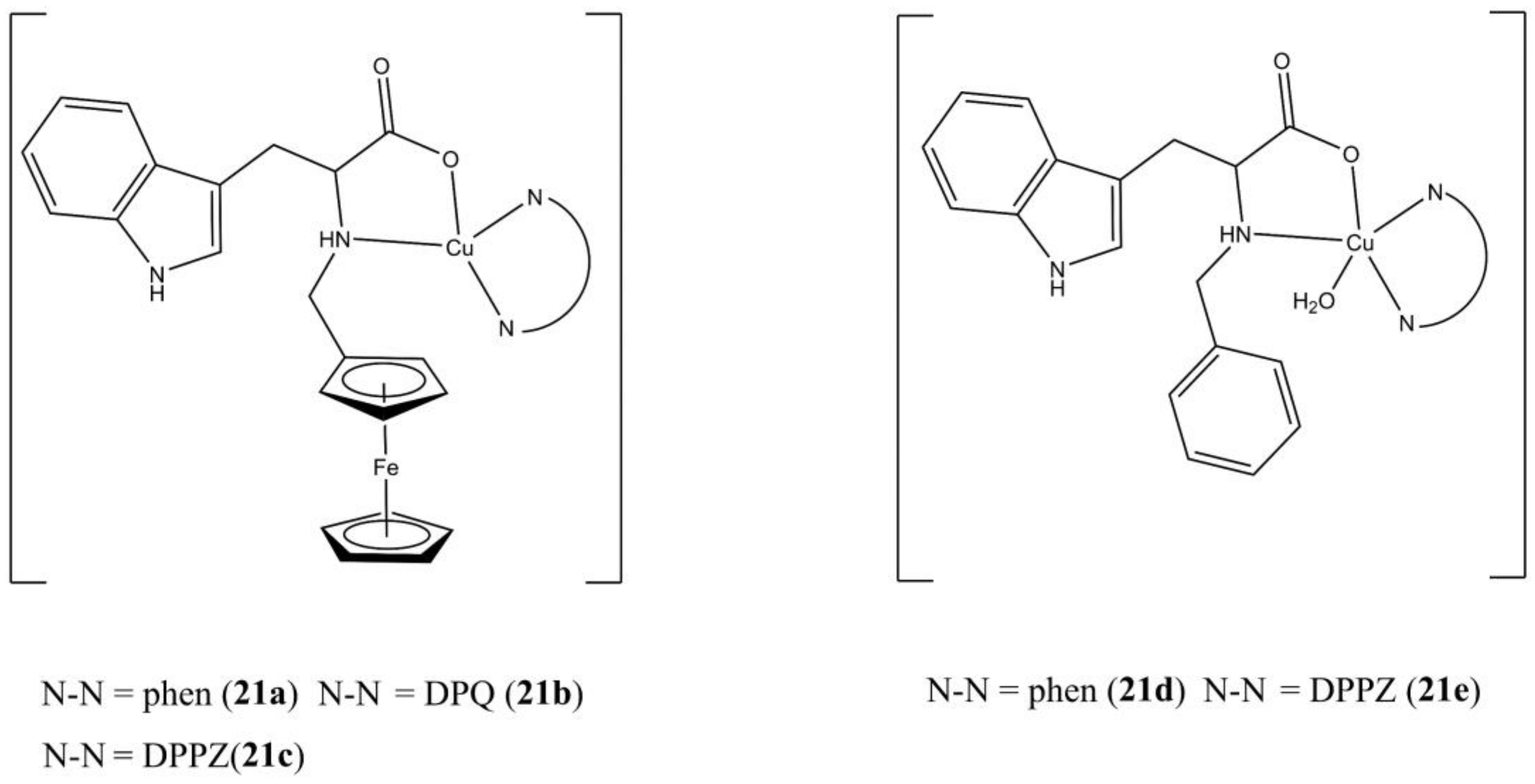
Goswami et al. has prepared and studied the photo-toxicity of a series of mixed Cu(II) complexes having neutral N-N
5 ligands (bipy, phen, DPQ, DPPZ), and tryptophan-based (Fc-Trp and Ph-Trp) ligands (
Figure 21) [
59].
These complexes generally possess enhanced anticancer potencies when exposed to visible light in both He-La (e.g., compound 21c IC50 of 8.95 and 1.29 µM in the dark and under visible light, respectively) and MCF-7 cells (e.g., compound 21c IC50 of 2.99 and 0.65 µM in the dark and under visible light, respectively). Cell death induction takes place through caspase-independent apoptotic mechanism, with higher apoptotic percentage when cells are exposed under visible light. The negligible anticancer activity observed for the [Zn(Fc-Trp)(DPPZ)](ClO4) complex in HeLa cells (IC50 > 80 µM) suggest the importance of the Cu(II) centre in exerting the anticancer activity. The studied compounds interact with ct-DNA as groove binders and can induce DNA cleavage through OH· production.
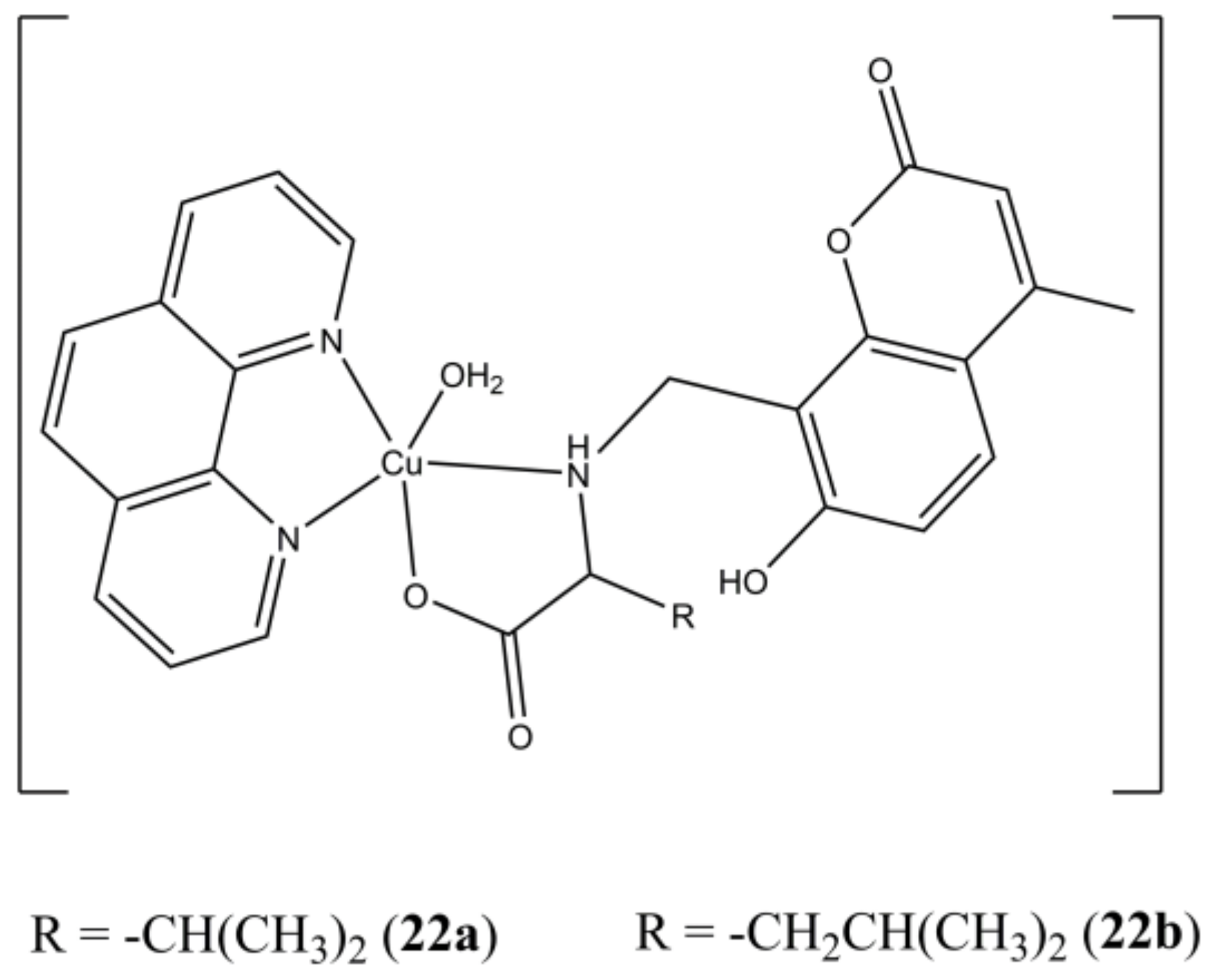
The two [Cu(HMCX)(phen)(OH
2)](ClO
4) complexes (
Figure 22), where the two H
2MCX ligands are obtained by coupling L-Valine (X = V) and L-Leucine (X = L) with 7-hydroxy-4-methylcoumarin, intercalates ct-DNA and possess micromolar anticancer activity against human prostate (PC3), liver (L02) and myeloid leukemia (HL-60) cancer cells [
60].
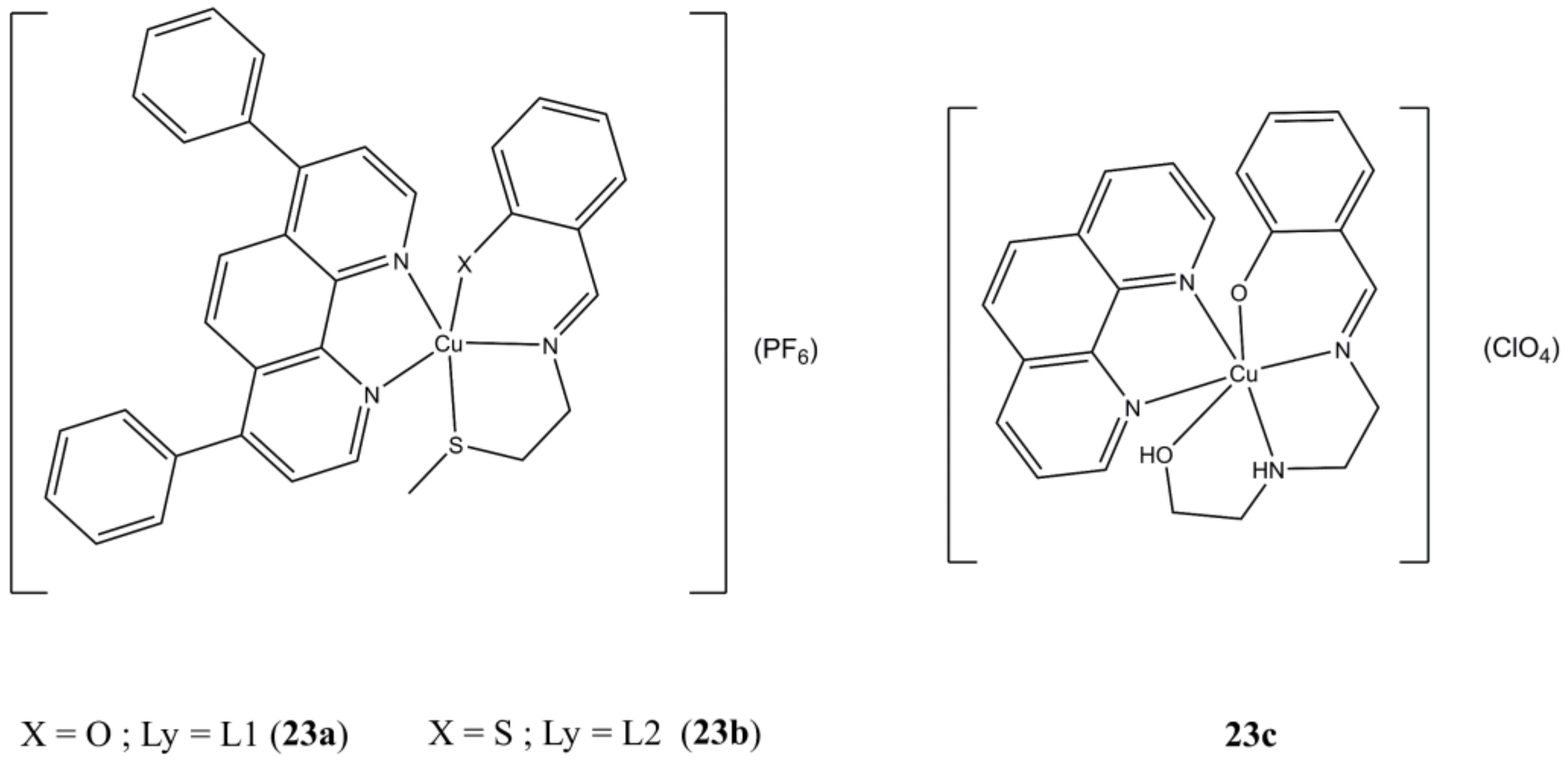
The two [Cu(Ly)(bathophen)](PF
6) (
Figure 23a,b) complexes (Ly are
O/S,N,S Schiff bases, batophen is 4,7-diphenil-1,10-phenanthroline) show sub-micromolar anticancer potencies (IC
50 in the 0.21–0.32 µM range) against both HMLER and HMLER-shEcad cells, thus proving to be active against breast CSC subpopulations too. Both complexes show higher anticancer potencies compared to approved drugs cisplatin and salinomycin. Moreover, they can reduce the number and dimensions of 3D HMLER-shEcad mammospheres [
61].
The Cu(II) complex [Cu(tdp)(phen)](ClO
4) (
Figure 23c), where Htdp is 2-[(2-(2-hydroxyethylamino)-ethylimino)methyl]phenol, shows IC
50 in the micromolar range in both MCF-7 and MDA-MB-231 cancer cells. This compound induces alteration of mitochondrial potential, ROS overexpression, cell-cycle arrest (at S- and G2/M phases) and cellular DNA damage followed by apoptosis, which can turn to necrosis at higher concentrations or longer durations of treatments. Interestingly, the Bax/Bcl-2 expression ratios were differently affected in MCF (p53
+, ER
+) and MDA-MB-231 (p53
−, ER
−), thus suggesting a potential genotype-selective mechanism mediated by the p53 protein, which still need to be clarified though [
62].
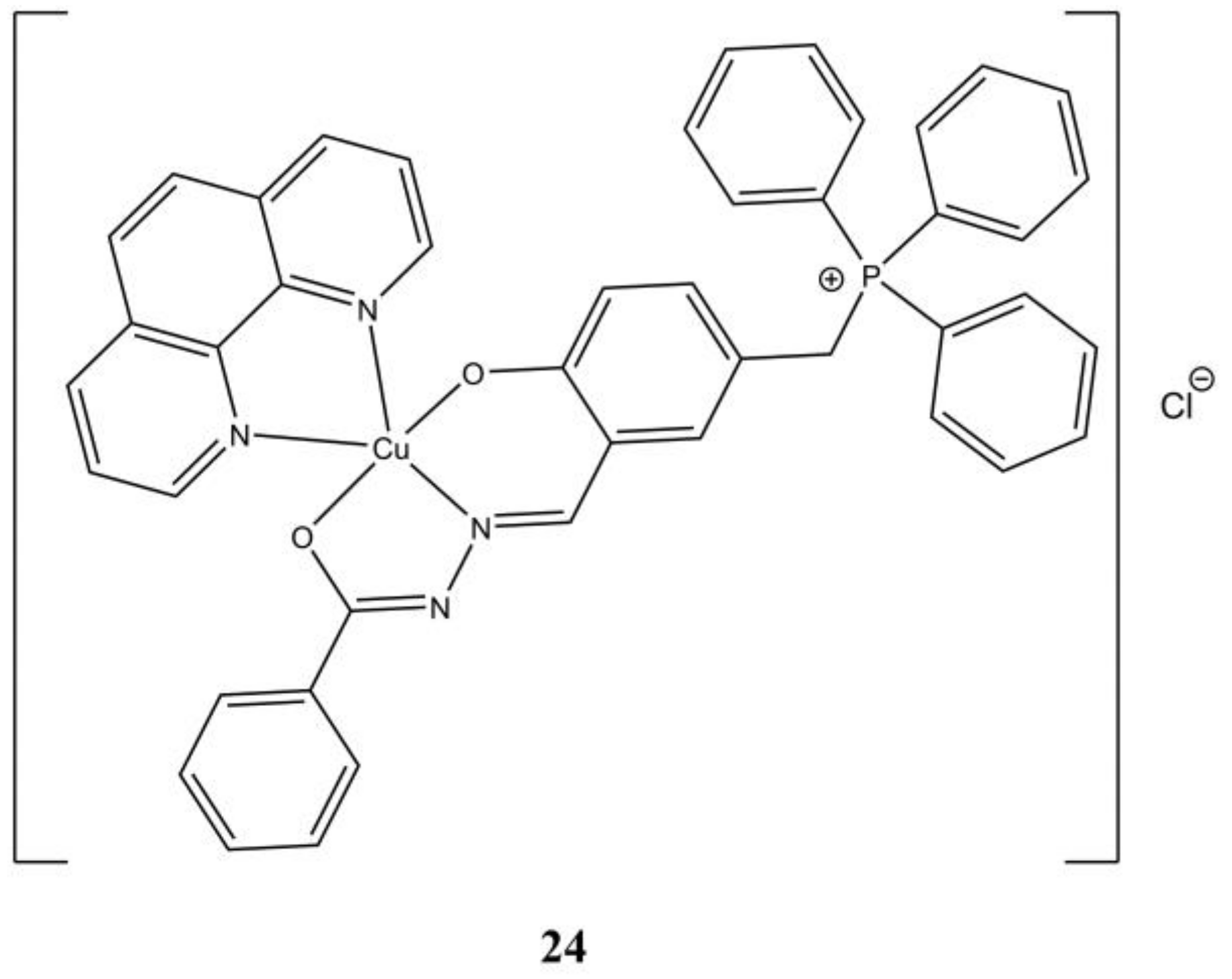
In a series of Cu(II) complexes having 5-(triphenylphosphoniummethyl)-salicylaldehyde benzoylhydrazone] chloride (L3) and
N,
N-diimine ligands, [Cu(phen)(L3)]Cl (
Figure 24) proved to be the most promising against A-549 (IC
50 of 4.2 µM) and PC-3 (IC
50 of 3.2 µM) cancer cells. The studied complexes are able to interfere at DNA level through inhibition of DNA Topoisomerase-I (Topo-I) [
63].
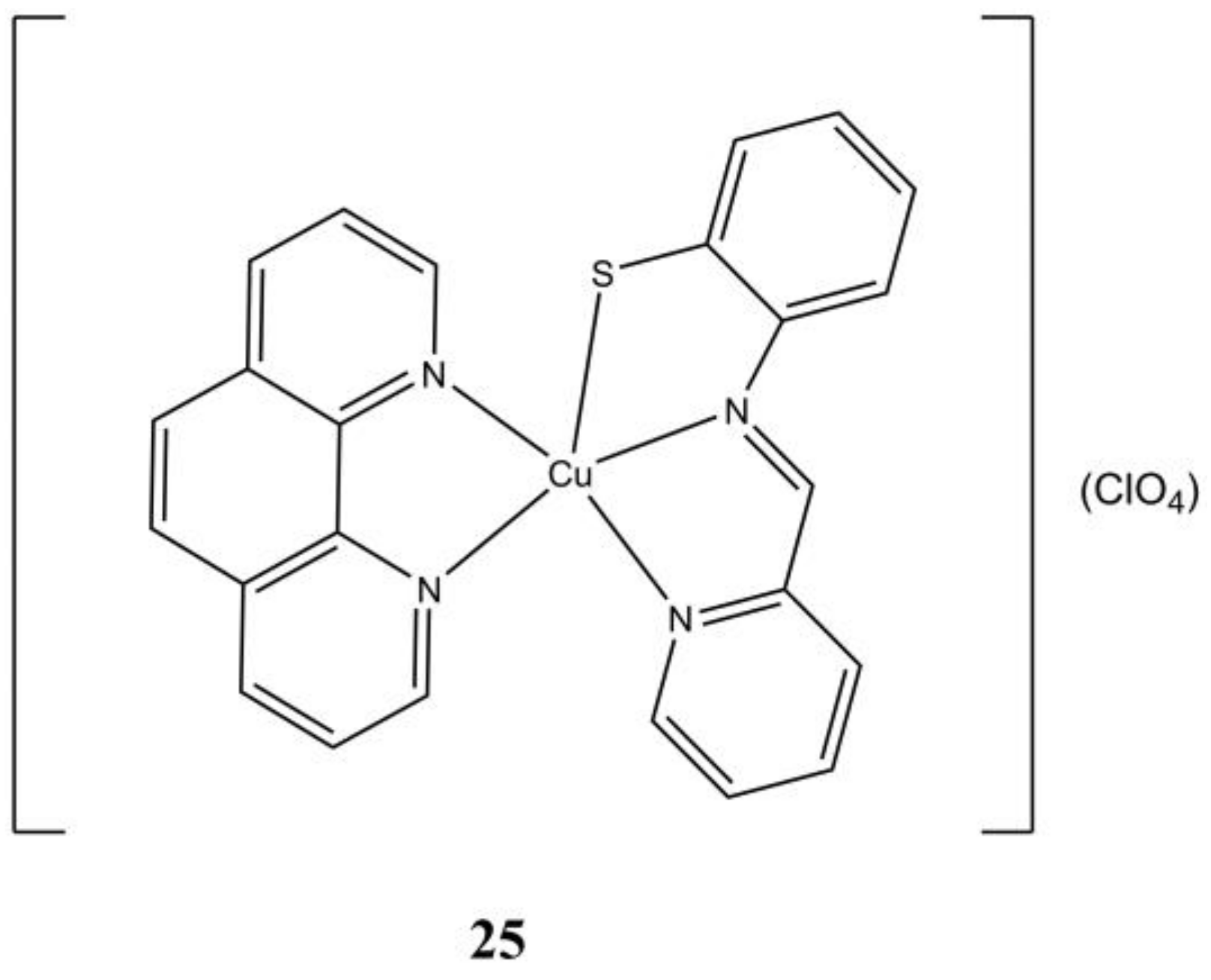
The complex [Cu(pabt)(phen)](ClO
4) (
Figure 25), where Hpabt is
N-(2-mercaptophenyl)-2′-pyridylmethylenimine, shows anticancer activity at micromolar level against A-549 and A-431(IC
50 of 5.26 and 5.41 µM, respectively) cancer cells, while being less cytotoxic against healthy L132 and HaCaT cells (IC
50 of 7.47 and >10 µM, respectively). This compound shows intercalates ct-DNA showing good binding affinity and triggers cell death in A-549 through apoptosis [
64].
IC
50 values for the compounds here reported are shown in
Table 5.
2.6. Polynuclear Complexes
Prisecaru et al. focused their attention on the complex
8c previously synthesized showing that its ability of cleaving DNA takes place oxidatively and in a non-specific manner. The complex shows comparable potency on both Platinum-resistant SKOV-3 (IC
50 of 6.7 µM) cells and healthy HS-832 ones (IC
50 of 4.5 µM) but is also 8 times more active than clinical drug Mitoxantrone on SKOV-3 cells (IC
50 of 54.5 µM). Cellular ROS production in A-549 cells up to nanomolar concentration level was also observed [
39].
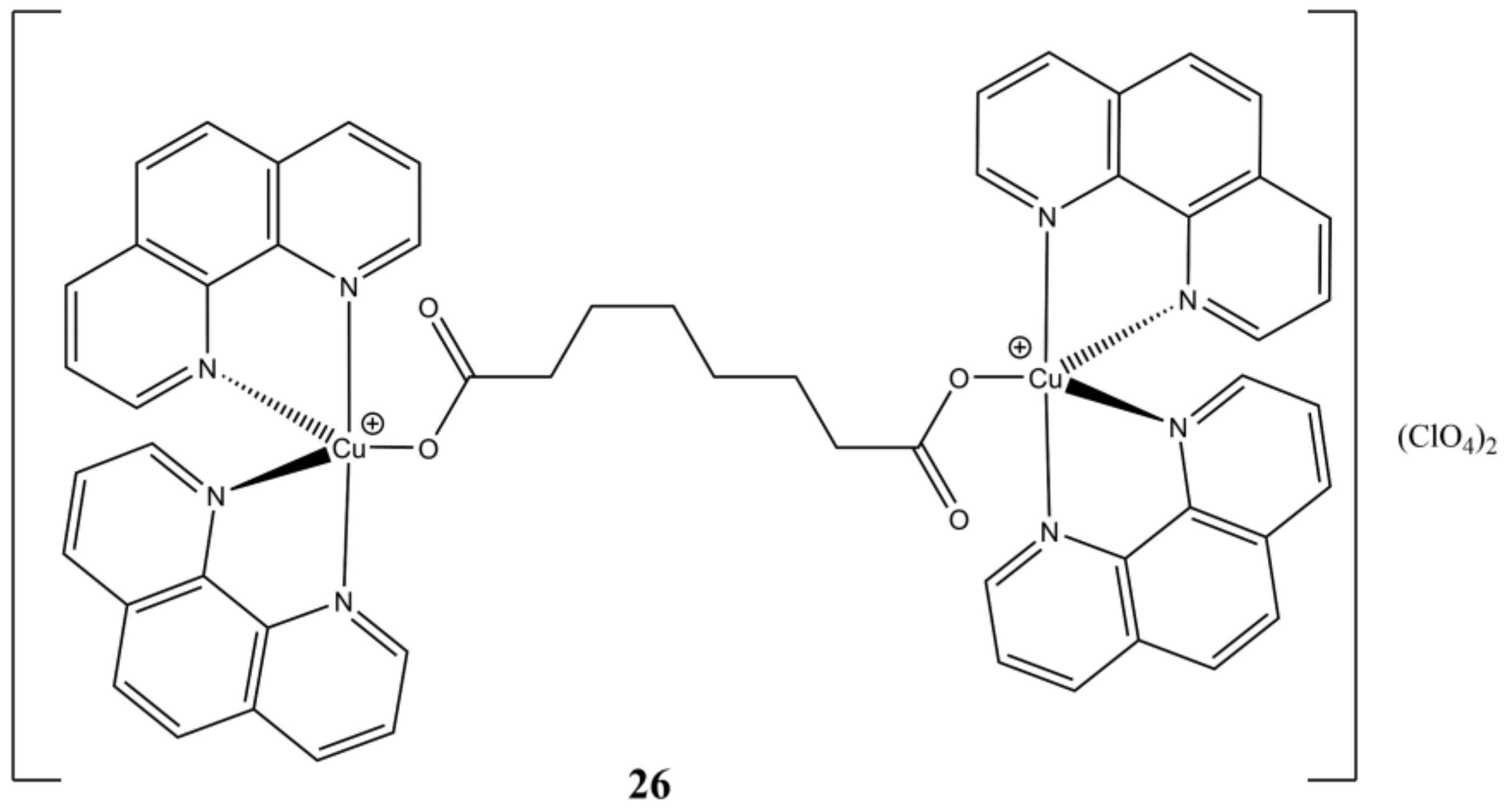
The same authors have evaluated the anticancer properties of [Cu
2(μ-oda)(phen)
4](ClO
4)
2 (
Figure 26), where the bridging ligand “oda” is octanedioic acid.
This complex shows time and concentration dependent (from low micromolar to nanomolar at 24 and 96 h of treatment, respectively) cytotoxic activity towards colorectal HT29, SW480 and SW620 cancer cells. In vivo drug tolerability studies on
G. Melonella larvae shows that the complex is better tolerated than cisplatin in the 200–1000 µg/mL concentration range. The reported compound shows good ct-DNA affinity, self-activating DNA cleavage ability in presence of oxygen, along with artificial SOD activity and cellular ROS induction ability. The authors combined these experimental evidence and proposed a model of DNA cleavage based on the formation of reduced [Cu(phen)
2]
+ complex and a π carboxylate radical, which can oxidatively damage DNA in multiple ways. For instance, the carboxylate radical and/or its alkyl product of decarboxylation might abstract a hydrogen from the DNA phosphodiester backbone, while the [Cu(phen)
2]
+ complex might induce DNA breaks through aerobic-generated oxo and hydroxo complexes [
65].
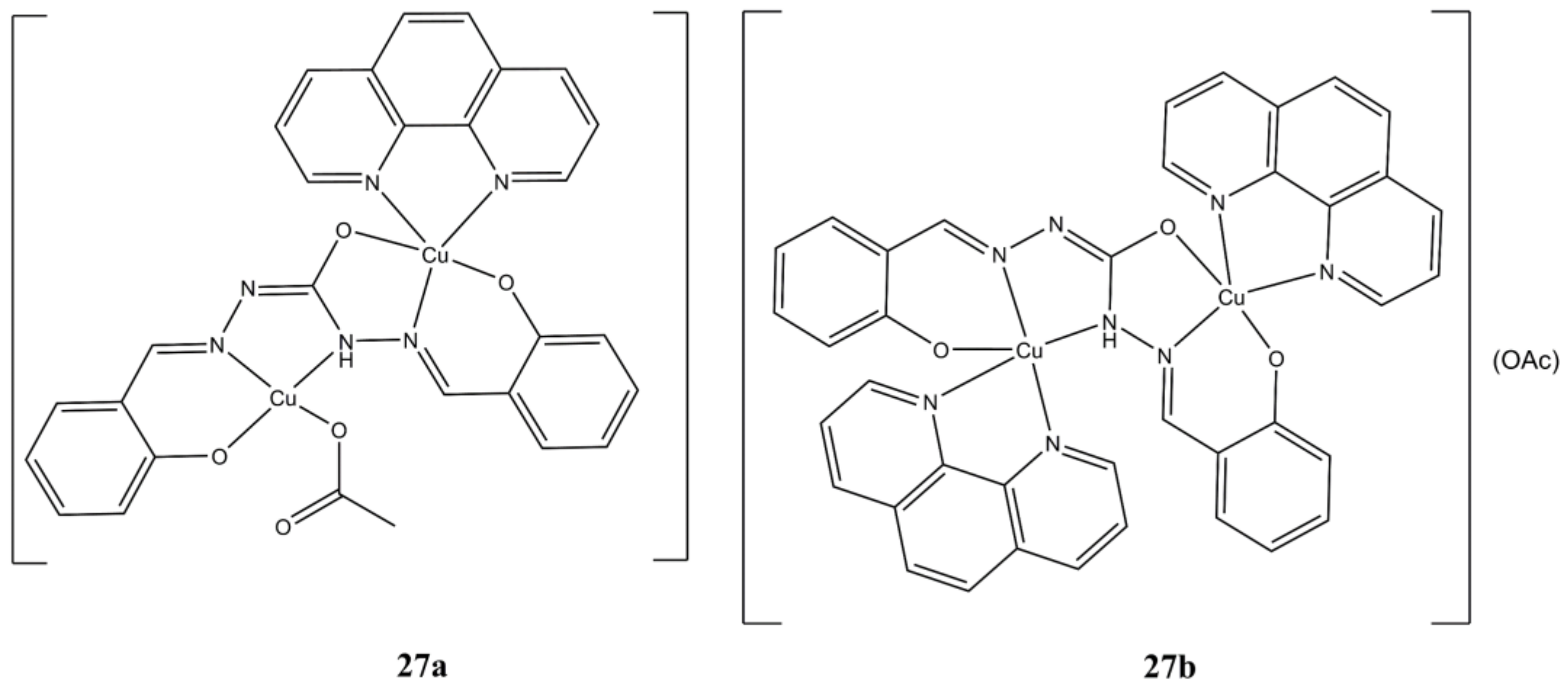
Parsekar et al. obtained the two binuclear Cu(II) complexes [Cu(phen)(SCH)Cu(OAc)] and [Cu
2(SCH)(phen)
2](OAc) by mixing Cu(OAC)
2, phen and the SCH ligand (1,5-bis(salicylidene)carbohydrazide) in different molar ratios (
Figure 27) [
66].
Both complexes are cytotoxic (micromolar level) towards cancerous A-549 and MCF-7 cancer cells and to a lower extent towards healthy HaCaT cells. Cellular growth is arrested mainly at G2/M phase and induction of cell death can switch from early to late apoptosis/necrosis in a dose-dependent manner. Both complexes can intercalate ct-DNA and inducing both hydrolytic and oxidative DNA cleavages. Interestingly, increase in ROS production is evidenced in A-549 while the opposite is observed in MCF-7 lines.
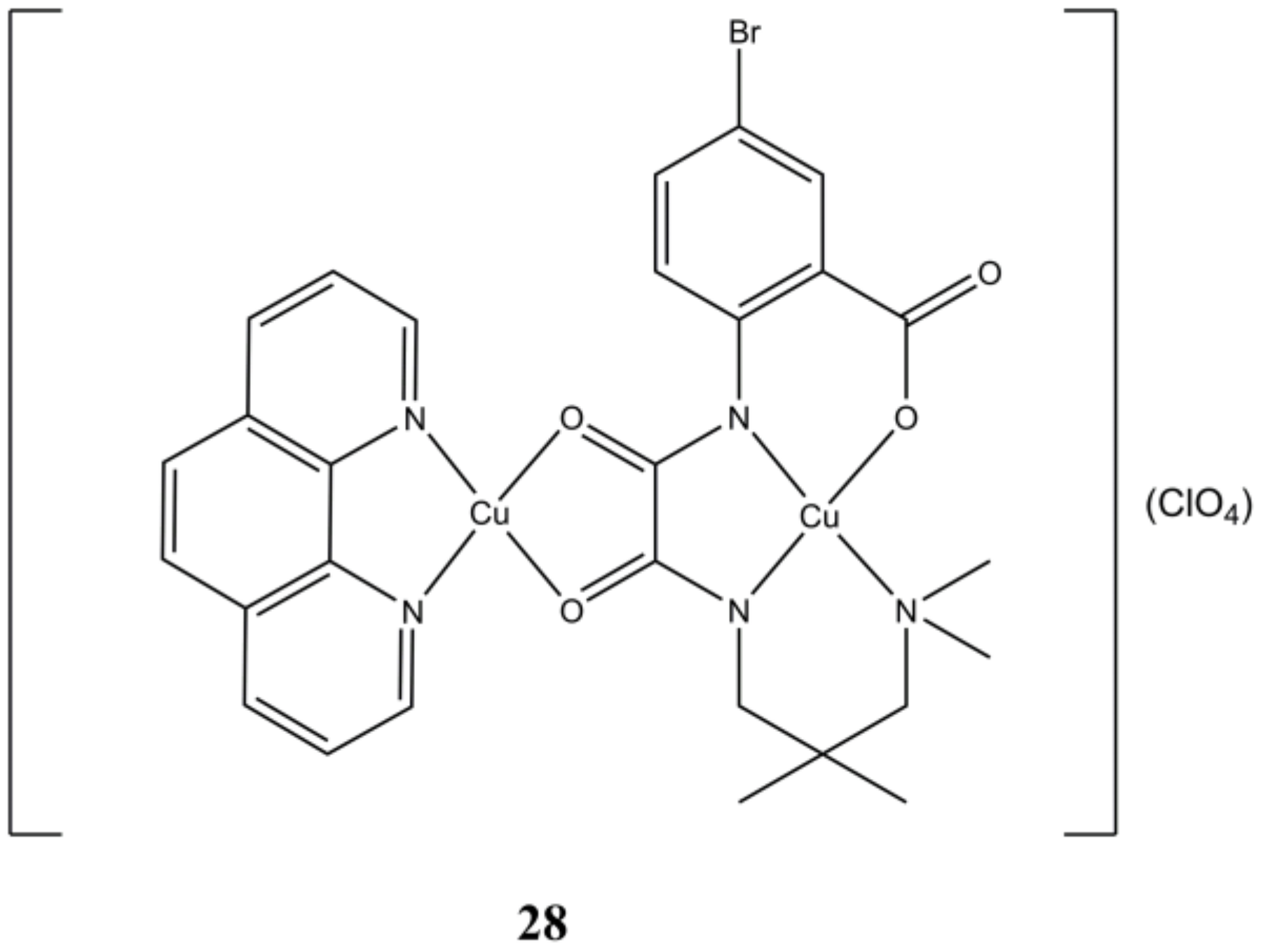
The binuclear [Cu
2(L4)(phen)](ClO
4) complex (
Figure 28) can be reduced by GSH, and in turn catalyze the conversion of H
2O
2 to hydroxy radicals, as demonstrated at both spectroscopic and at cellular level. This complex is cytotoxic at micromolar level towards 4T1, A-549, HepG2 and MCF-7 cancer cells and to a lower extent towards healthy COS-7 cells. This compound arrest cell cycle at G0/G1 phase and induces cell death through apoptosis. The dual ability of
26 in depleting the cellular antioxidant GSH and producing ROS through H
2O
2 (often overexpressed in tumorigenic cells) makes this compound a potential candidate for applications in chemo-dynamic therapy [
67].
IC
50 values for the polynuclear complexes here reported are summarized in
Table 6.