Caseinolytic Proteins (Clp) in the Genus Klebsiella: Special Focus on ClpK
Abstract
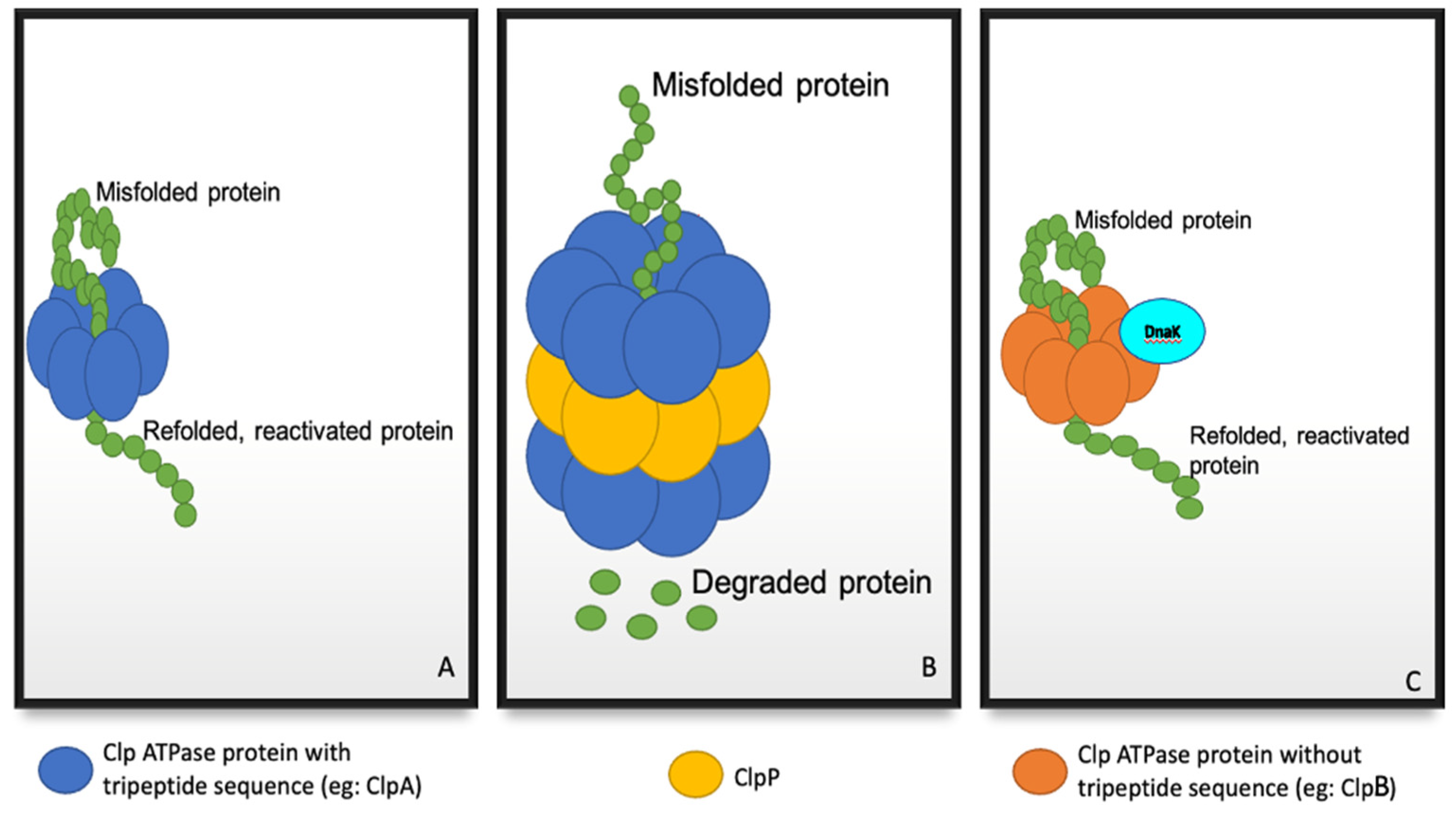
1. Introduction
2. Results and Discussion
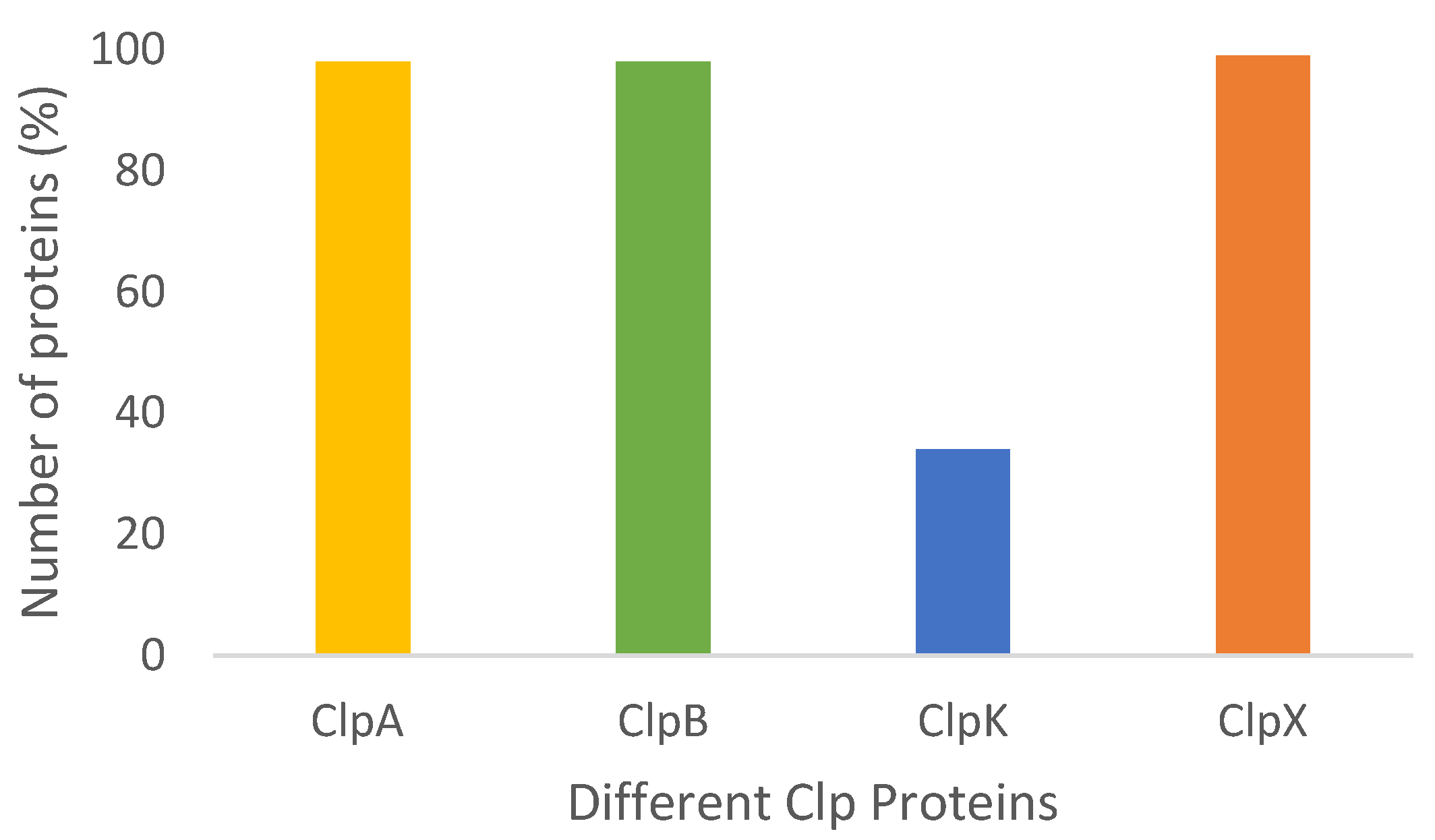
2.1. Caseinolytic ATPase Protein Classification
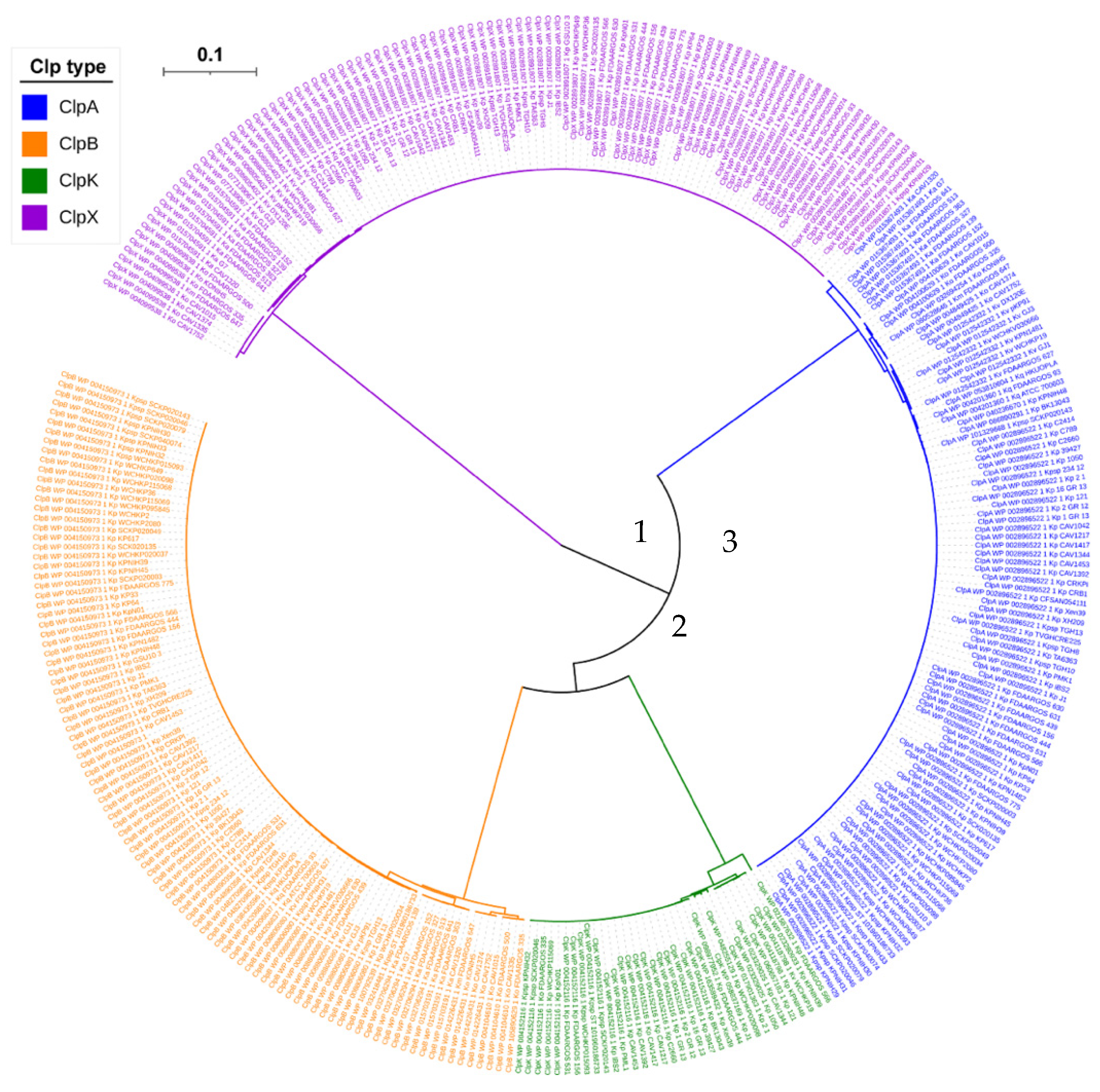
2.2. Phylogenetic Analysis
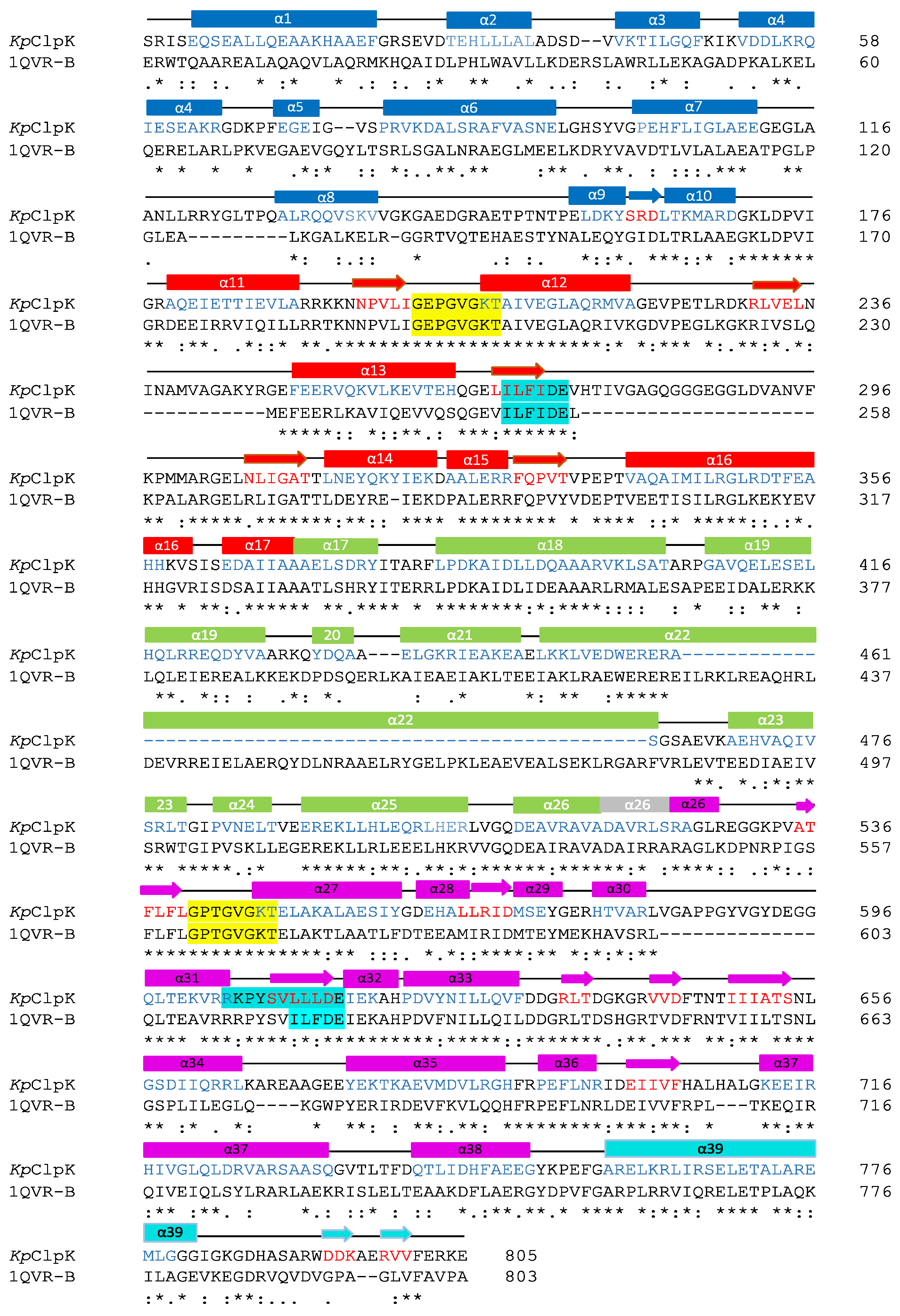
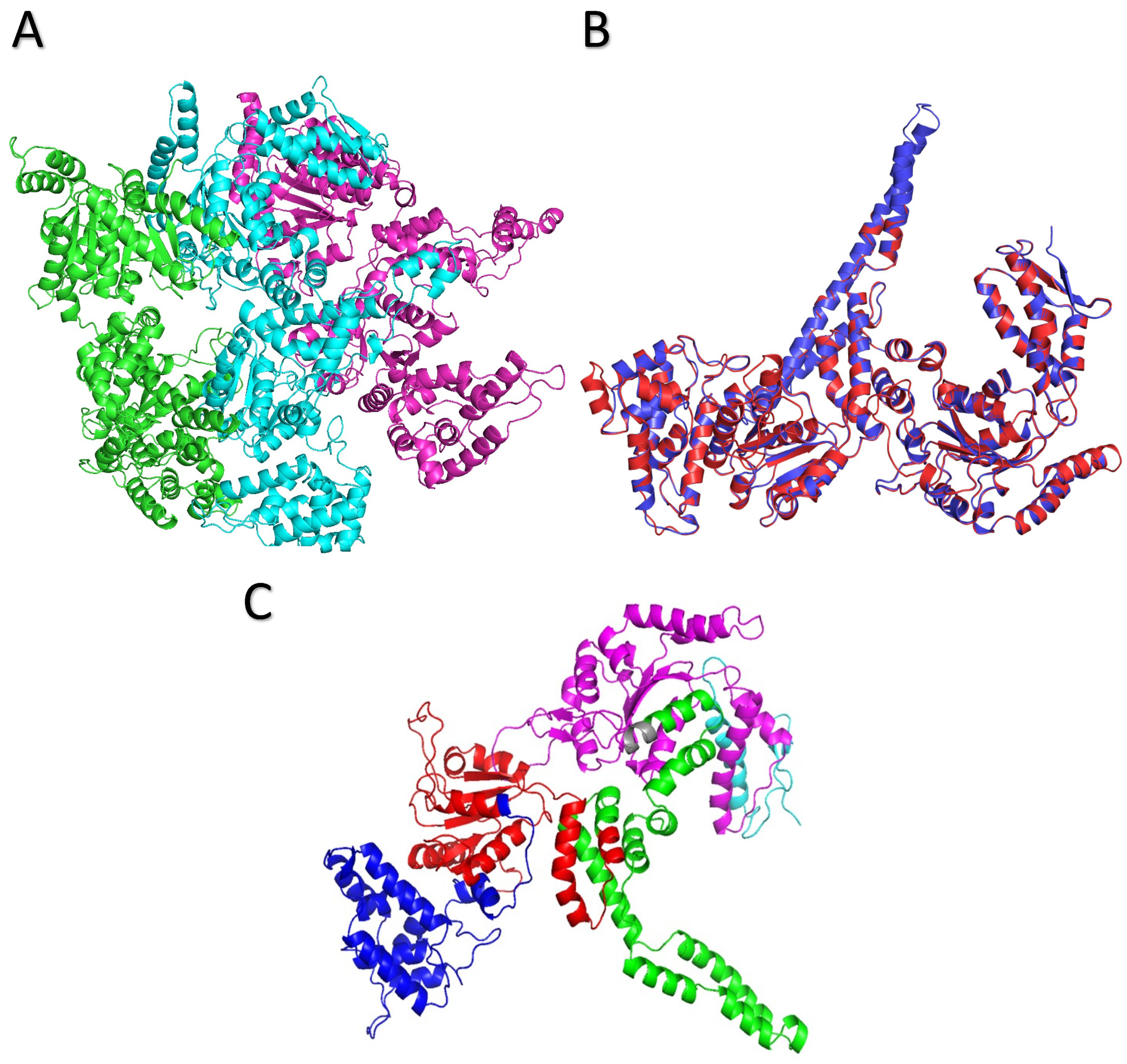
2.3. Hypothetical ClpK Structure
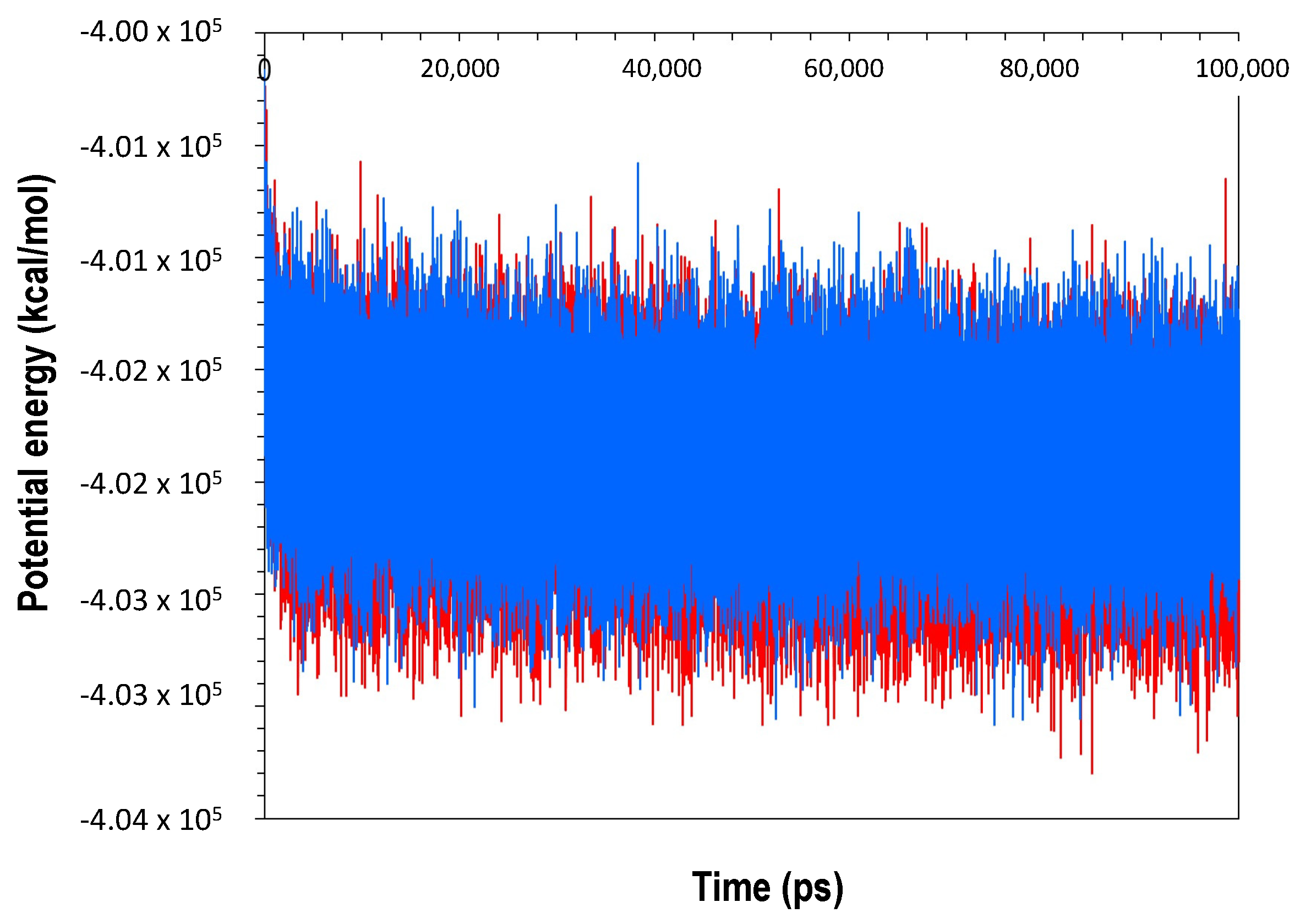
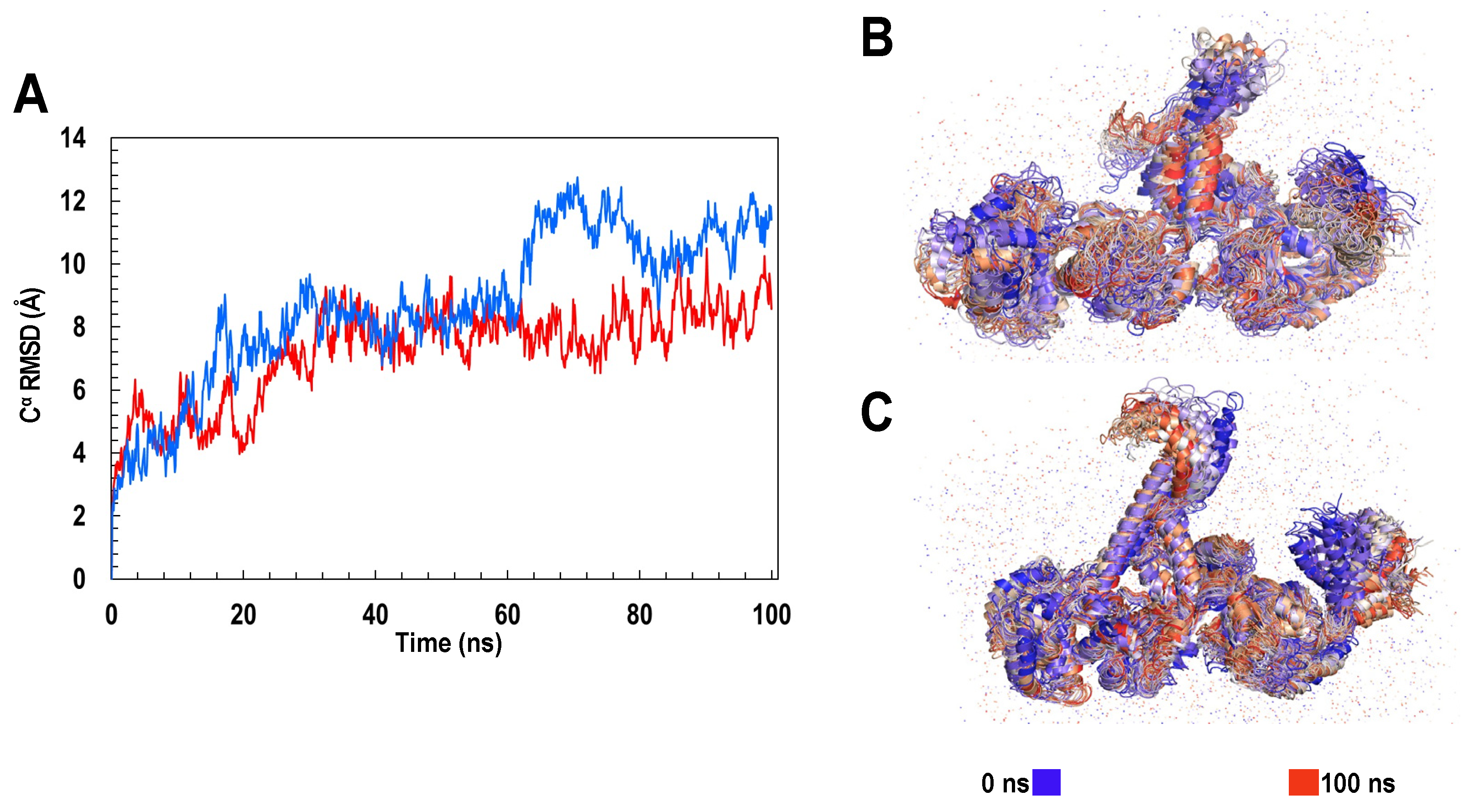
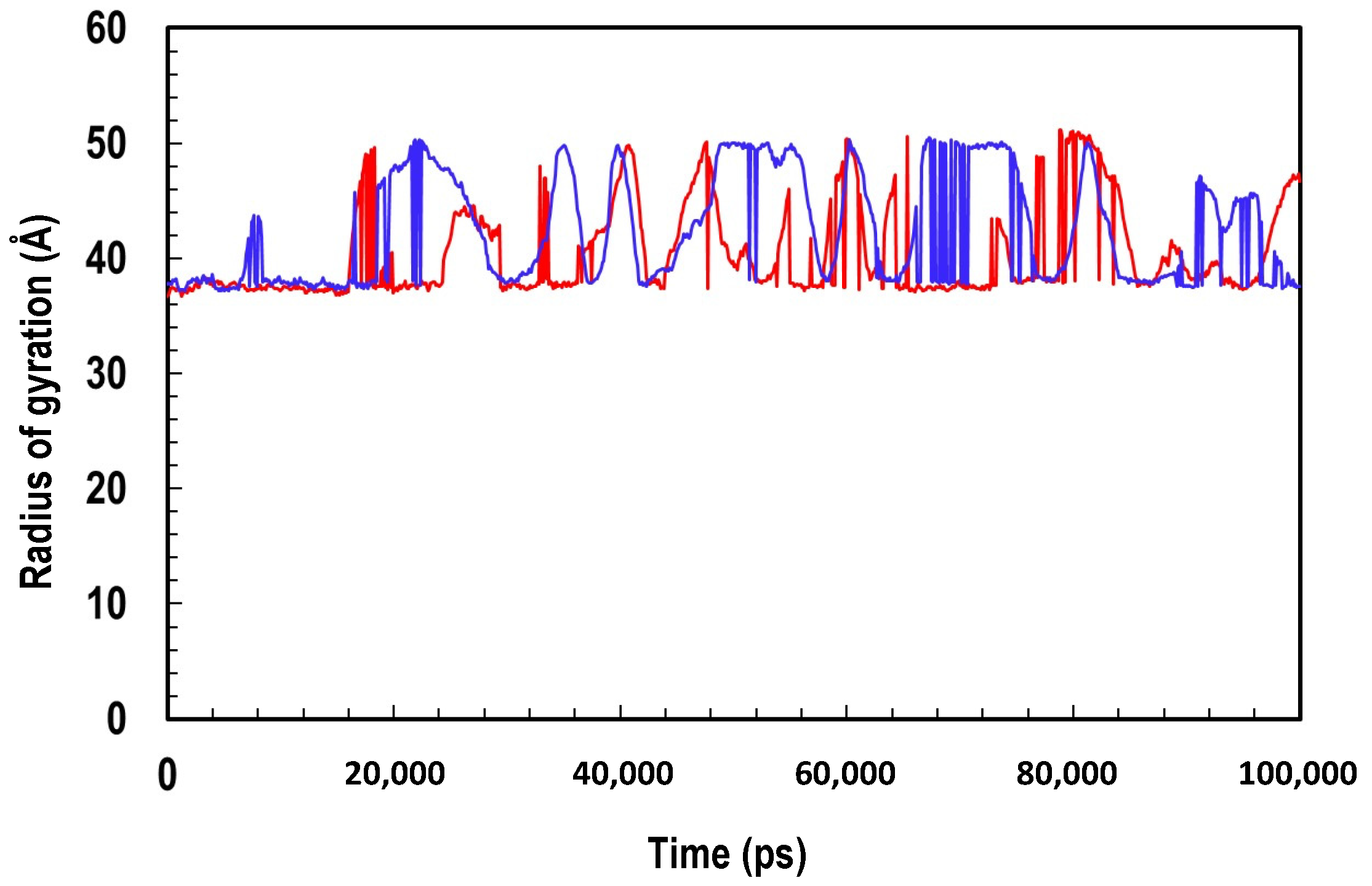
2.4. Molecular Dynamics Simulation
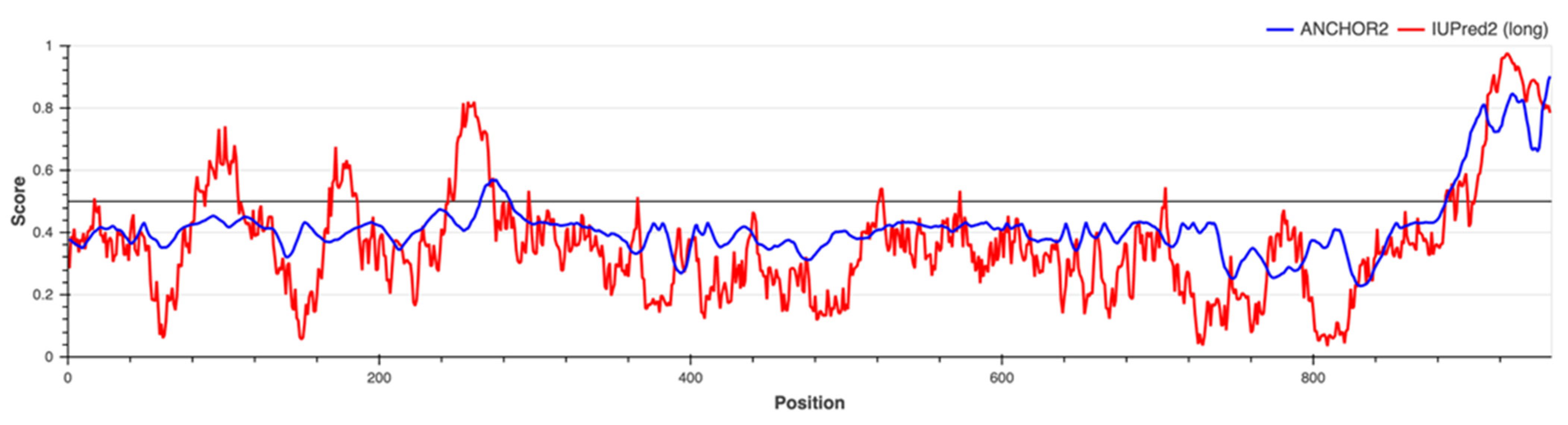
2.5. Protein Disorder Prediction
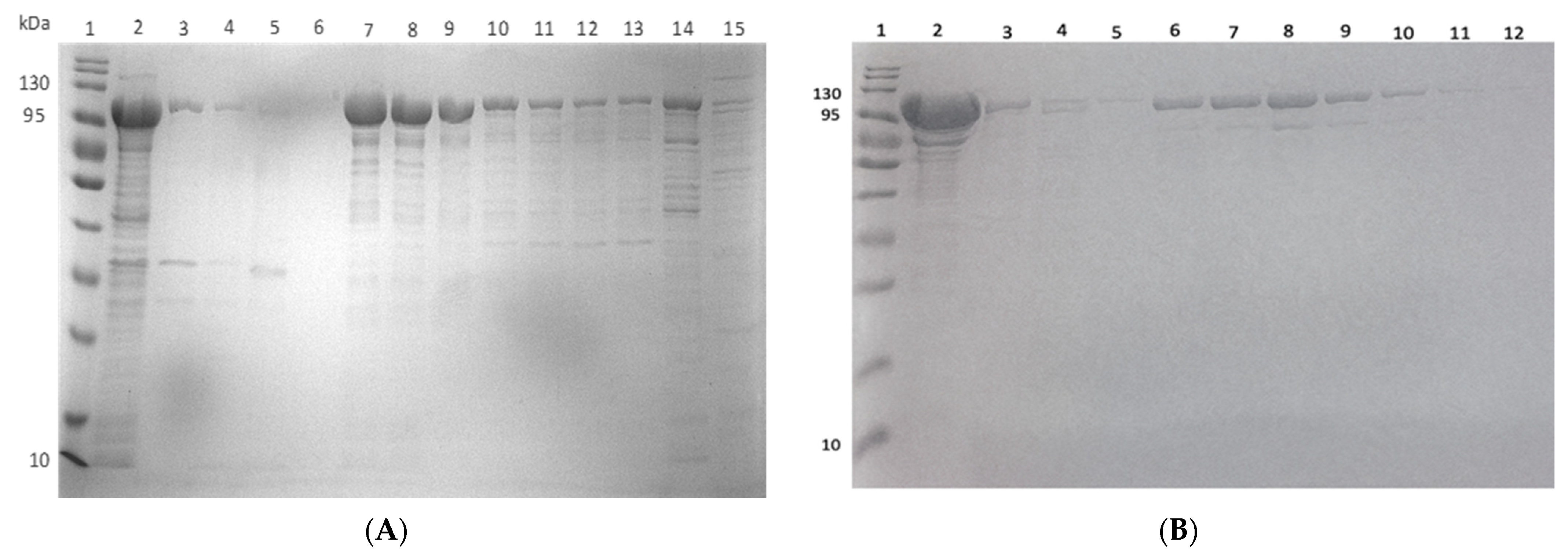
2.6. Expression and Purification of ClpK
2.7. Biophysical Characterisation of ClpK
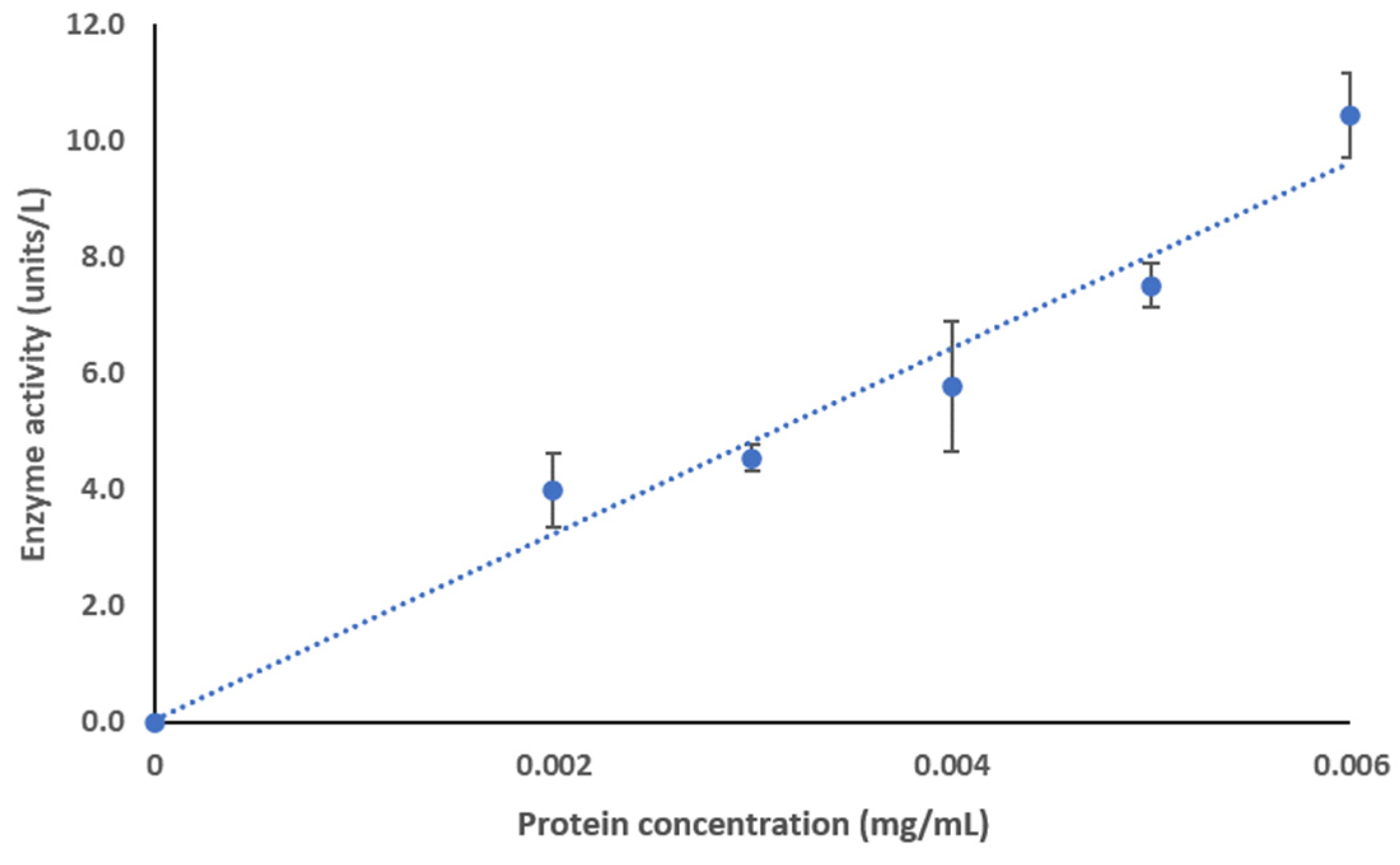
2.7.1. Enzyme Activity Assay
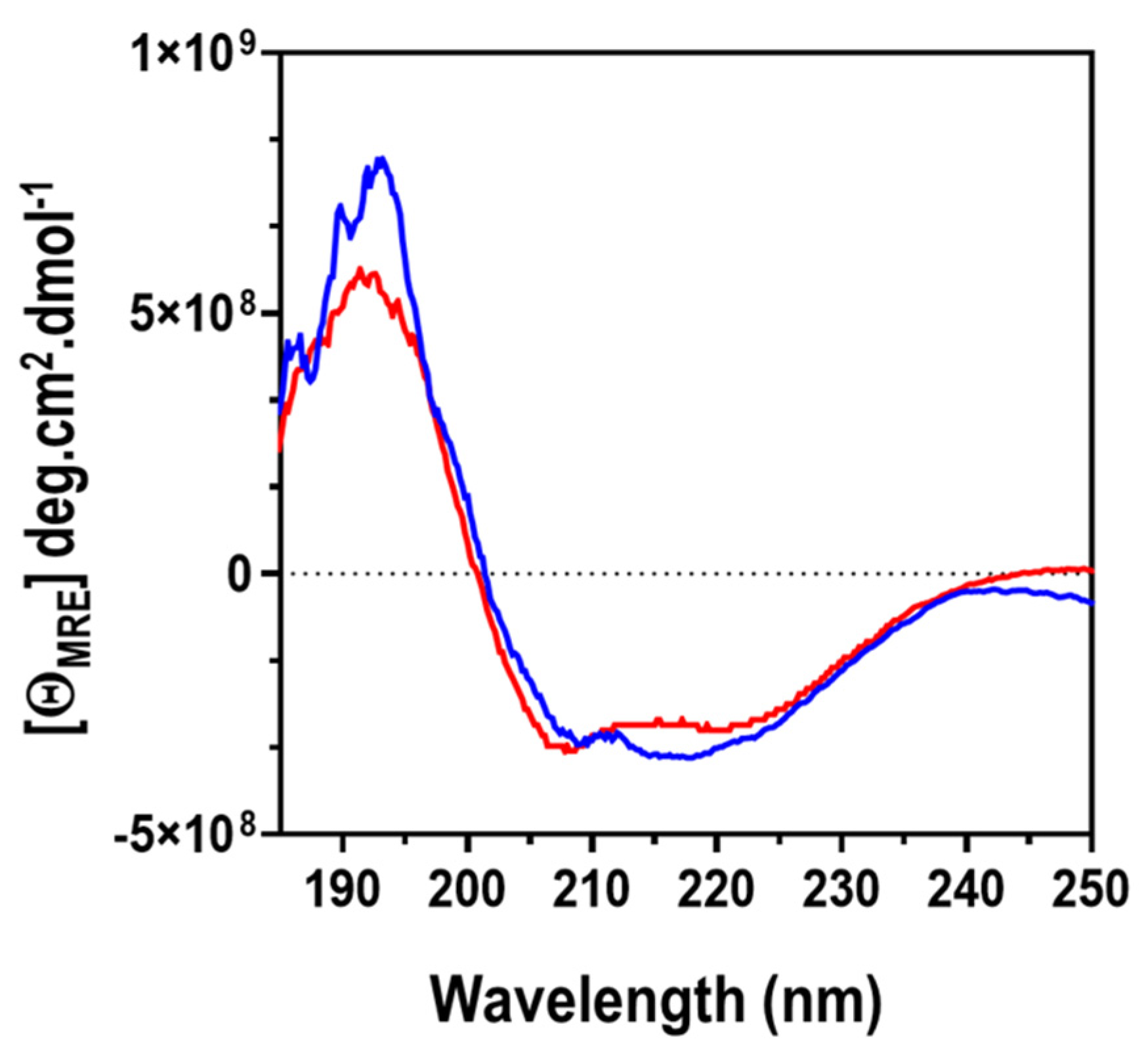
2.7.2. Far UV-CD Spectroscopy
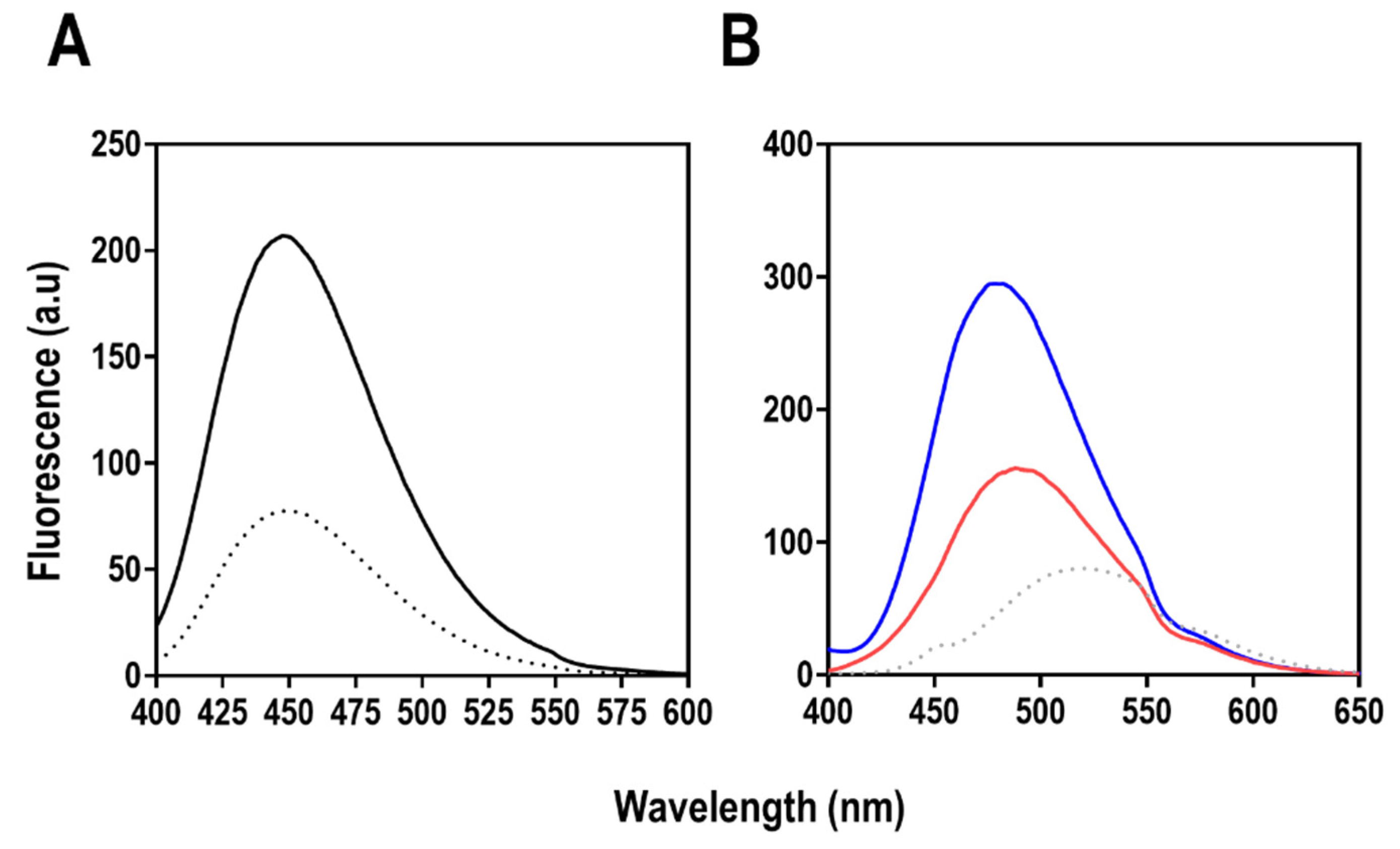
2.7.3. Extrinsic Fluorescence Spectroscopy
3. Materials and Methods
3.1. Species and Databases
3.2. Genome Data Mining and Annotation of Caseinolytic Proteins
3.3. Phylogenetic Analysis of Clp Proteins
3.4. Clp Protein Homology Analysis
3.5. Homology Modelling of ClpK
3.6. Molecular Dynamics Simulation
3.7. Post-Dynamic Analysis
3.8. Protein Disorder and Circular Dichroism Analysis
3.9. ClpK Expression and Purification
3.10. Enzyme Activity Assay
3.11. Far-UV Circular Dichroism Spectroscopy
3.12. Extrinsic Fluorescence Spectroscopy
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Santajit, S.; Indrawattana, N. Mechanisms of Antimicrobial Resistance in ESKAPE Pathogens. BioMed Res. Int. 2016, 2016, 2475067. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.M.; Bachman, M.A. Colonization, infection, and the accessory genome of Klebsiella pneumoniae. Front. Cell. Infect. Microbiol. 2018, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Pendleton, J.N.; Gorman, S.P.; Gilmore, B.F. Clinical relevance of the ESKAPE pathogens. Expert Rev. Anti-Infect. Ther. 2013, 11, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Brisse, S.; Passet, V.; Grimont, P.A. Description of Klebsiella quasipneumoniae sp. nov., isolated from human infections, with two subspecies, Klebsiella quasipneumoniae subsp. quasipneumoniae subsp. nov. and Klebsiella quasipneumoniae subsp. similipneumoniae subsp. nov., and demonstration that Klebsiella singaporensis is a junior heterotypic synonym of Klebsiella variicola. Int. J. Syst. Evol. Microbiol. 2014, 64, 3146–3152. [Google Scholar]
- Chapman, P.; Forde, B.M.; Roberts, L.W.; Bergh, H.; Vesey, D.; Jennison, A.V.; Moss, S.; Paterson, D.L.; Beatson, S.A.; Harris, P.N. Genomic investigation reveals contaminated detergent as the source of an ESBL-producing Klebsiella michiganensis outbreak in a neonatal unit. J. Clin. Microbiol. 2020. [Google Scholar] [CrossRef]
- Doorduijn, D.J.; Rooijakkers, S.H.; van Schaik, W.; Bardoel, B.W. Complement resistance mechanisms of Klebsiella pneumoniae. Immunobiology 2016, 221, 1102–1109. [Google Scholar] [CrossRef]
- Joainig, M.M.; Gorkiewicz, G.; Leitner, E.; Weberhofer, P.; Zollner-Schwetz, I.; Lippe, I.; Feierl, G.; Krause, R.; Hinterleitner, T.; Zechner, E.L. Cytotoxic effects of Klebsiella oxytoca strains isolated from patients with antibiotic-associated hemorrhagic colitis or other diseases caused by infections and from healthy subjects. J. Clin. Microbiol. 2010, 48, 817–824. [Google Scholar] [CrossRef]
- Singh, L.; Cariappa, M.; Kaur, M. Klebsiella oxytoca: An emerging pathogen? Med. J. Armed Forces India 2016, 72, S59–S61. [Google Scholar] [CrossRef]
- Bojer, M.S.; Struve, C.; Ingmer, H.; Hansen, D.S.; Krogfelt, K.A. Heat resistance mediated by a new plasmid encoded Clp ATPase, ClpK, as a possible novel mechanism for nosocomial persistence of Klebsiella pneumoniae. PLoS ONE 2010, 5, e15467. [Google Scholar] [CrossRef]
- AhYoung, A.P.; Koehl, A.; Cascio, D.; Egea, P.F. Structural mapping of the C lp B ATP ases of Plasmodium falciparum: Targeting protein folding and secretion for antimalarial drug design. Protein Sci. 2015, 24, 1508–1520. [Google Scholar] [CrossRef]
- Park, S.S.; Kwon, H.Y.; Tran, T.D.; Choi, M.H.; Jung, S.H.; Lee, S.; Briles, D.E.; Rhee, D.K. ClpL is a chaperone without auxiliary factors. FEBS J. 2015, 282, 1352–1367. [Google Scholar] [CrossRef]
- Zolkiewski, M.; Zhang, T.; Nagy, M. Aggregate reactivation mediated by the Hsp100 chaperones. Arch. Biochem. Biophys. 2012, 520, 1–6. [Google Scholar] [CrossRef]
- Bojer, M.S.; Struve, C.; Ingmer, H.; Krogfelt, K.A. ClpP-dependent and-independent activities encoded by the polycistronic clpK-encoding locus contribute to heat shock survival in Klebsiella pneumoniae. Res. Microbiol. 2013, 164, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Maurizi, M.R.; Xia, D. Protein binding and disruption by Clp/Hsp100 chaperones. Structure 2004, 12, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.M.; Chaudhary, H.; Marsee, J.D. Phylogenetic analysis predicts structural divergence for proteobacterial ClpC proteins. J. Struct. Biol. 2018, 201, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, E.C.; Glover, J.R.; Singer, M.A.; Lindquist, S. HSP100/Clp proteins: A common mechanism explains diverse functions. Trends Biochem. Sci. 1996, 21, 289–296. [Google Scholar] [CrossRef]
- Kress, W.; Maglica, Ž.; Weber-Ban, E. Clp chaperone–proteases: Structure and function. Res. Microbiol. 2009, 160, 618–628. [Google Scholar] [CrossRef]
- Filloux, A.; Hachani, A.; Bleves, S. The bacterial type VI secretion machine: Yet another player for protein transport across membranes. Microbiology 2008, 154, 1570–1583. [Google Scholar] [CrossRef] [PubMed]
- Mfeka, M.S.; Martínez-Oyanedel, J.; Chen, W.; Achilonu, I.; Syed, K.; Khoza, T. Comparative analyses and structural insights of new class glutathione transferases in Cryptosporidium species. Sci. Rep. 2020, 10, 20370. [Google Scholar] [CrossRef]
- Ingmer, H.; Vogensen, F.K.; Hammer, K.; Kilstrup, M. Disruption and analysis of the clpB, clpC, and clpE genes in Lactococcus lactis: ClpE, a new Clp family in gram-positive bacteria. J. Bacteriol. 1999, 181, 2075–2083. [Google Scholar] [CrossRef]
- Lee, S.; Sowa, M.E.; Watanabe, Y.-H.; Sigler, P.B.; Chiu, W.; Yoshida, M.; Tsai, F.T. The structure of ClpB: A molecular chaperone that rescues proteins from an aggregated state. Cell 2003, 115, 229–240. [Google Scholar] [CrossRef]
- Pietrosiuk, A.; Lenherr, E.D.; Falk, S.; Bönemann, G.; Kopp, J.; Zentgraf, H.; Sinning, I.; Mogk, A. Molecular basis for the unique role of the AAA+ chaperone ClpV in type VI protein secretion. J. Biol. Chem. 2011, 286, 30010–30021. [Google Scholar] [CrossRef]
- Zheng, B.; Halperin, T.; Hruskova-Heidingsfeldova, O.; Adam, Z.; Clarke, A.K. Characterization of chloroplast Clp proteins in Arabidopsis: Localization, tissue specificity and stress responses. Physiol. Plant. 2002, 114, 92–101. [Google Scholar] [CrossRef]
- Thibault, G.; Tsitrin, Y.; Davidson, T.; Gribun, A.; Houry, W.A. Large nucleotide-dependent movement of the N-terminal domain of the ClpX chaperone. EMBO J. 2006, 25, 3367–3376. [Google Scholar] [CrossRef]
- Ali, M.S.; Baek, K.-H. Protective Roles of Cytosolic and Plastidal Proteasomes on Abiotic Stress and Pathogen Invasion. Plants 2020, 9, 832. [Google Scholar] [CrossRef]
- Capestany, C.A.; Tribble, G.D.; Maeda, K.; Demuth, D.R.; Lamont, R.J. Role of the Clp system in stress tolerance, biofilm formation, and intracellular invasion in Porphyromonas gingivalis. J. Bacteriol. 2008, 190, 1436–1446. [Google Scholar] [CrossRef]
- Wojtyra, U.A.; Thibault, G.; Tuite, A.; Houry, W.A. The N-terminal zinc binding domain of ClpX is a dimerization domain that modulates the chaperone function. J. Biol. Chem. 2003, 278, 48981–48990. [Google Scholar] [CrossRef]
- Pavlopoulos, G.A.; Soldatos, T.G.; Barbosa-Silva, A.; Schneider, R. A reference guide for tree analysis and visualization. BioData Min. 2010, 3, 1. [Google Scholar] [CrossRef]
- Newell, P.; Fricker, A.; Roco, C.; Chandrangsu, P.; Merkel, S. A Small-Group Activity Introducing the Use and Interpretation of BLAST. J. Microbiol. Biol. Educ. JMBE 2013, 14, 238–243. [Google Scholar] [CrossRef][Green Version]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Section 7.1, Homologs Are Descended from a Common Ancestor. In Biochemistry, 5th ed.; W H Freeman: New York, NY, USA, 2002. [Google Scholar]
- Wlodawer, A.; Minor, W.; Dauter, Z.; Jaskolski, M. Protein crystallography for non-crystallographers, or how to get the best (but not more) from published macromolecular structures. FEBS J. 2008, 275, 1–21. [Google Scholar] [CrossRef]
- Di Tommaso, P.; Moretti, S.; Xenarios, I.; Orobitg, M.; Montanyola, A.; Chang, J.-M.; Taly, J.-F.; Notredame, C. T-Coffee: A web server for the multiple sequence alignment of protein and RNA sequences using structural information and homology extension. Nucleic Acids Res. 2011, 39, W13–W17. [Google Scholar] [CrossRef]
- Carugo, O.; Pongor, S. A normalized root-mean-square distance for comparing protein three-dimensional structures. Protein Sci. 2001, 10, 1470–1473. [Google Scholar] [CrossRef]
- Zhao, Y.; Zeng, C.; Massiah, M.A. Molecular Dynamics Simulation Reveals Insights into the Mechanism of Unfolding by the A130T/V Mutations within the MID1 Zinc-Binding Bbox1 Domain. PLoS ONE 2015, 10, e0124377. [Google Scholar] [CrossRef]
- Ko, J.; Park, H.; Heo, L.; Seok, C. GalaxyWEB server for protein structure prediction and refinement. Nucleic Acids Res. 2012, 40, W294–W297. [Google Scholar] [CrossRef] [PubMed]
- Schrodinger, L. The PyMOL Molecular Graphics System; 2010; Volume 1. [Google Scholar]
- Rosenzweig, R.; Farber, P.; Velyvis, A.; Rennella, E.; Latham, M.P.; Kay, L.E. ClpB N-terminal domain plays a regulatory role in protein disaggregation. Proc. Natl. Acad. Sci. USA 2015, 112, E6872. [Google Scholar] [CrossRef]
- Ye, J.; Osborne, A.R.; Groll, M.; Rapoport, T.A. RecA-like motor ATPases—Lessons from structures. Biochim. Biophys. Acta 2004, 1659, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, G.; Mohanty, D. An In Silico Analysis of the Binding Modes and Binding Affinities of Small Molecule Modulators of PDZ-Peptide Interactions. PLoS ONE 2013, 8, e71340. [Google Scholar] [CrossRef] [PubMed]
- Buchner, J. Molecular chaperones and protein quality control: An introduction to the JBC Reviews thematic series. J. Biol. Chem. 2019, 294, 2074–2075. [Google Scholar] [CrossRef]
- Pathak, R.; Gupta, A.; Shukla, R.; Baunthiyal, M. Identification of new drug-like compounds from millets as Xanthine oxidoreductase inhibitors for treatment of Hyperuricemia: A molecular docking and simulation study. Comput. Biol. Chem. 2018, 76, 32–41. [Google Scholar] [CrossRef]
- Hashemzadeh, H.; Javadi, H.; Darvishi, M. Study of Structural stability and formation mechanisms in DSPC and DPSM liposomes: A coarse-grained molecular dynamics simulation. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Deng, X.; Eickholt, J.; Cheng, J. A comprehensive overview of computational protein disorder prediction methods. Mol. BioSyst. 2012, 8, 114–121. [Google Scholar] [CrossRef]
- Coşkun, O. Separation techniques: Chromatography. North. Clin. Istanb. 2016, 3, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, C.D.; Steers, E.J., Jr.; Weissbach, H. Purification and properties of 5-methyltetrahydropteroyltriglutamate-homocysteine transmethylase. J. Biol. Chem. 1970, 245, 390–401. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Greenfield, N.J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc. 2006, 1, 2876–2890. [Google Scholar] [CrossRef]
- Gasymov, O.K.; Glasgow, B.J. ANS fluorescence: Potential to augment the identification of the external binding sites of proteins. Biochim. Biophys. Acta 2007, 1774, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Aranovich, A.; Gdalevsky, G.; Cohen-Luria, R.; Fishov, I.; Parola, A. Membrane-catalyzed nucleotide exchange on DnaA—Effect of surface molecular crowding. J. Biol. Chem. 2006, 281, 12526–12534. [Google Scholar] [CrossRef]
- Ngcobo, N.S.; Chiliza, Z.E.; Chen, W.; Yu, J.-H.; Nelson, D.R.; Tuszynski, J.A.; Preto, J.; Syed, K. Comparative Analysis, Structural Insights, and Substrate/Drug Interaction of CYP128A1 in Mycobacterium tuberculosis. Int. J. Mol. Sci. 2020, 21, 4816. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef]
- Boc, A.; Diallo, A.B.; Makarenkov, V. T-REX: A web server for inferring, validating and visualizing phylogenetic trees and networks. Nucleic Acids Res. 2012, 40, W573–W579. [Google Scholar] [CrossRef]
- Mullins, J.G. Structural modelling pipelines in next generation sequencing projects. Adv. Protein Chem. Struct. Biol. 2012, 89, 117–167. [Google Scholar]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J.E. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 2015, 10, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yan, R.; Roy, A.; Xu, D.; Poisson, J.; Zhang, Y. The I-TASSER Suite: Protein structure and function prediction. Nat. Methods 2015, 12, 7–8. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Kucukural, A.; Zhang, Y. I-TASSER: A unified platform for automated protein structure and function prediction. Nat. Protoc. 2010, 5, 725–738. [Google Scholar] [CrossRef]
- Zhang, Y. I-TASSER server for protein 3D structure prediction. BMC Bioinform. 2008, 9, 40. [Google Scholar] [CrossRef] [PubMed]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 1–9. [Google Scholar] [CrossRef]
- Laskowski, R.A.; MacArthur, M.W.; Moss, D.S.; Thornton, J.M. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 1993, 26, 283–291. [Google Scholar] [CrossRef]
- Williams, C.J.; Headd, J.J.; Moriarty, N.W.; Prisant, M.G.; Videau, L.L.; Deis, L.N.; Verma, V.; Keedy, D.A.; Hintze, B.J.; Chen, V.B.; et al. MolProbity: More and better reference data for improved all-atom structure validation. Protein Sci. 2018, 27, 293–315. [Google Scholar] [CrossRef]
- Schrödinger. Release 2021-2: Maestro; Schrödinger: New York, NY, USA, 2021. [Google Scholar]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef]
- Schöning-Stierand, K.; Diedrich, K.; Fährrolfes, R.; Flachsenberg, F.; Meyder, A.; Nittinger, E.; Steinegger, R.; Rarey, M. ProteinsPlus: Interactive analysis of protein–ligand binding interfaces. Nucleic Acids Res. 2020, 48, W48–W53. [Google Scholar] [CrossRef] [PubMed]
- Mészáros, B.; Erdős, G.; Dosztányi, Z. IUPred2A: Context-dependent prediction of protein disorder as a function of redox state and protein binding. Nucleic Acids Res. 2018, 46, W329–W337. [Google Scholar] [CrossRef] [PubMed]
- Lobley, A.; Whitmore, L.; Wallace, B.A. DICHROWEB: An interactive website for the analysis of protein secondary structure from circular dichroism spectra. Bioinformatics 2002, 18, 211–212. [Google Scholar] [CrossRef]
- Kruger, N. The Bradford Method for Protein Quantitation. Methods Mol. Biol. 1994, 32, 9–15. [Google Scholar] [CrossRef] [PubMed]




| Class I | |||
| Clp Regulatory Subunits | Species | Functions | References |
| ClpA 1 | Gram-positive Proteobacteria | Protein quality control | [20] |
| ClpB | Prokaryotes, yeast, and plants | Disaggregation of stress-damaged proteins | [21,22,23] |
| ClpC | Gram-positive bacteria (Firmicutes and Actinobacteria) and Cyanobacteria | Protein quality control | [20] |
| ClpD | Chloroplasts of higher plants | Molecular chaperone | [23] |
| ClpE | Firmicutes | Thermotolerance, cell division and virulence | [20] |
| ClpK | K. pneumonia | Thermotolerance | [13] |
| ClpL | Streptococcuss pneumoniae | Nucleotide phosphohydrolase activity, stabilises unfolded proteins, prevents protein aggregation | [11] |
| ClpV | Gram-negative bacteria | Component of the type V1 secretion system | [24] |
| Class II | |||
| Clp Regulatory Subunits | Species | Functions | References |
| ClpM | Mus musculus | Protein quality control | [23,25] |
| ClpN | Pseudomonas aeruginosa | Cell division | [23,25] |
| ClpX | Proteobacteria, Firmicutes and Thermatogae | Protein quality control, cell division and virulence | [20] |
| ClpY | Gram-positive Proteobacteria | Cell division, heat shock response, and capsule transcription | [20] |
| Clp Protein | Identity (%) |
|---|---|
| ClpA | 97.1–100 |
| ClpB | 96.5–100 |
| ClpX | 98.3–100 |
| ClpK | 94.1–100 |
| Steps | Volume (ml) | Protein (mg/mL) b | Total Protein (mg) | Specific Activity (units/mg) c | Total Activity (units) | Yield (%) | Purity (%) |
|---|---|---|---|---|---|---|---|
| Crude extract a | 30 | 0.0345 | 93.10 | 0.0423 | 3.94 | 100 | 0.38 |
| Anion eluant d | 30 | 0.0122 | 32.94 | 0.302 | 9.95 | 35.4 | 2.7 |
| HisTag eluant e | 10 | 0.117 | 1.17 | 11.13 | 13.02 | 1.26 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motiwala, T.; Akumadu, B.O.; Zuma, S.; Mfeka, M.S.; Chen, W.; Achilonu, I.; Syed, K.; Khoza, T. Caseinolytic Proteins (Clp) in the Genus Klebsiella: Special Focus on ClpK. Molecules 2022, 27, 200. https://doi.org/10.3390/molecules27010200
Motiwala T, Akumadu BO, Zuma S, Mfeka MS, Chen W, Achilonu I, Syed K, Khoza T. Caseinolytic Proteins (Clp) in the Genus Klebsiella: Special Focus on ClpK. Molecules. 2022; 27(1):200. https://doi.org/10.3390/molecules27010200
Chicago/Turabian StyleMotiwala, Tehrim, Blessing Oluebube Akumadu, Sbahle Zuma, Mbalenhle Sizamile Mfeka, Wanping Chen, Ikechukwu Achilonu, Khajamohiddin Syed, and Thandeka Khoza. 2022. "Caseinolytic Proteins (Clp) in the Genus Klebsiella: Special Focus on ClpK" Molecules 27, no. 1: 200. https://doi.org/10.3390/molecules27010200
APA StyleMotiwala, T., Akumadu, B. O., Zuma, S., Mfeka, M. S., Chen, W., Achilonu, I., Syed, K., & Khoza, T. (2022). Caseinolytic Proteins (Clp) in the Genus Klebsiella: Special Focus on ClpK. Molecules, 27(1), 200. https://doi.org/10.3390/molecules27010200







