Pinosylvin Shifts Macrophage Polarization to Support Resolution of Inflammation
Abstract
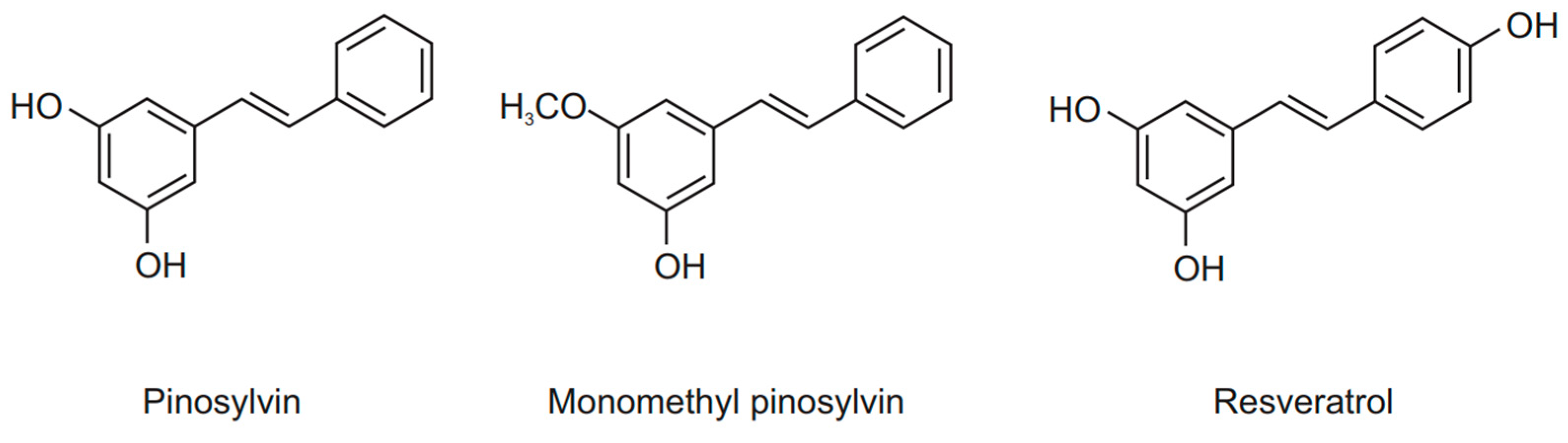
1. Introduction
2. Results
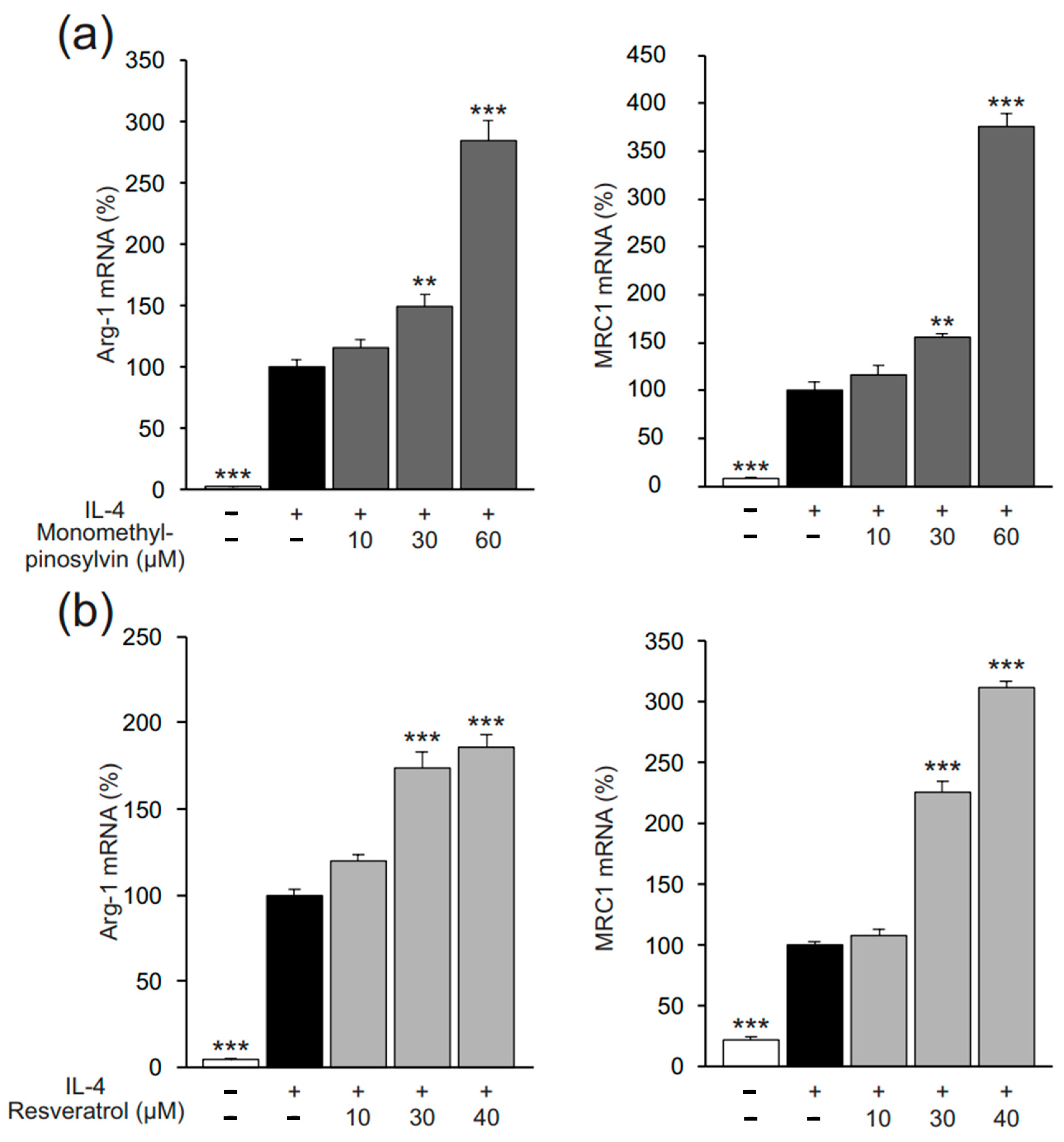
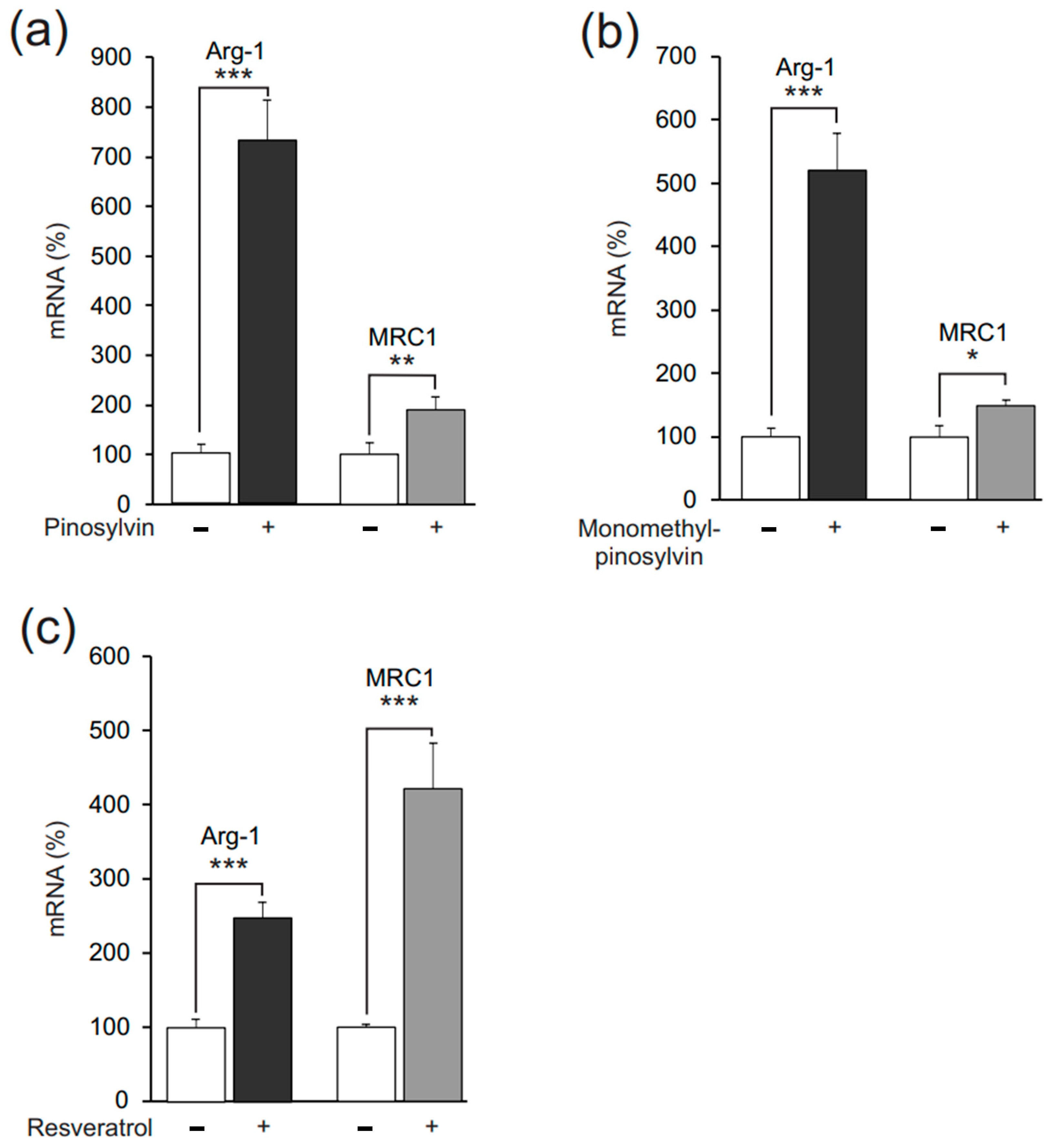
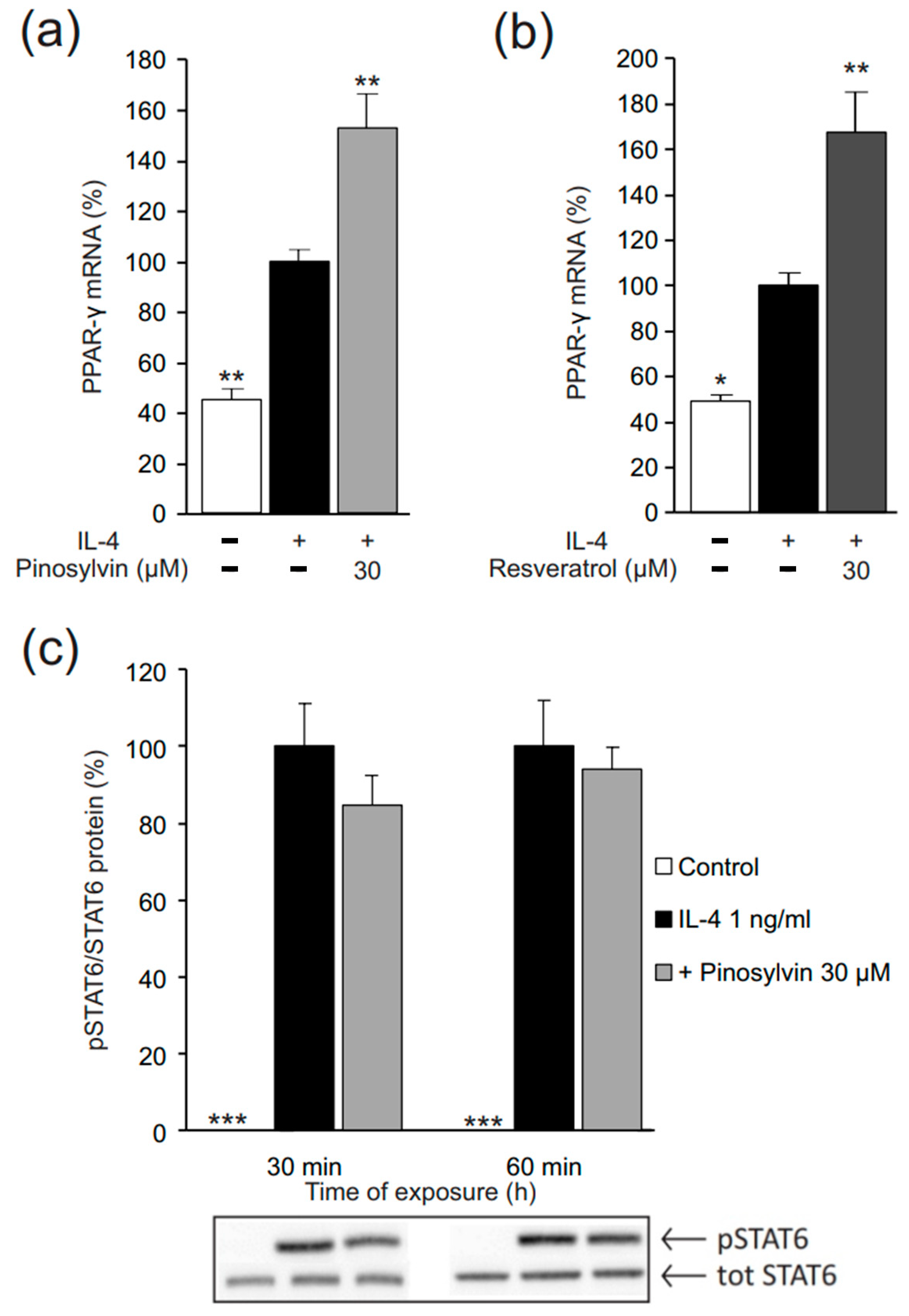
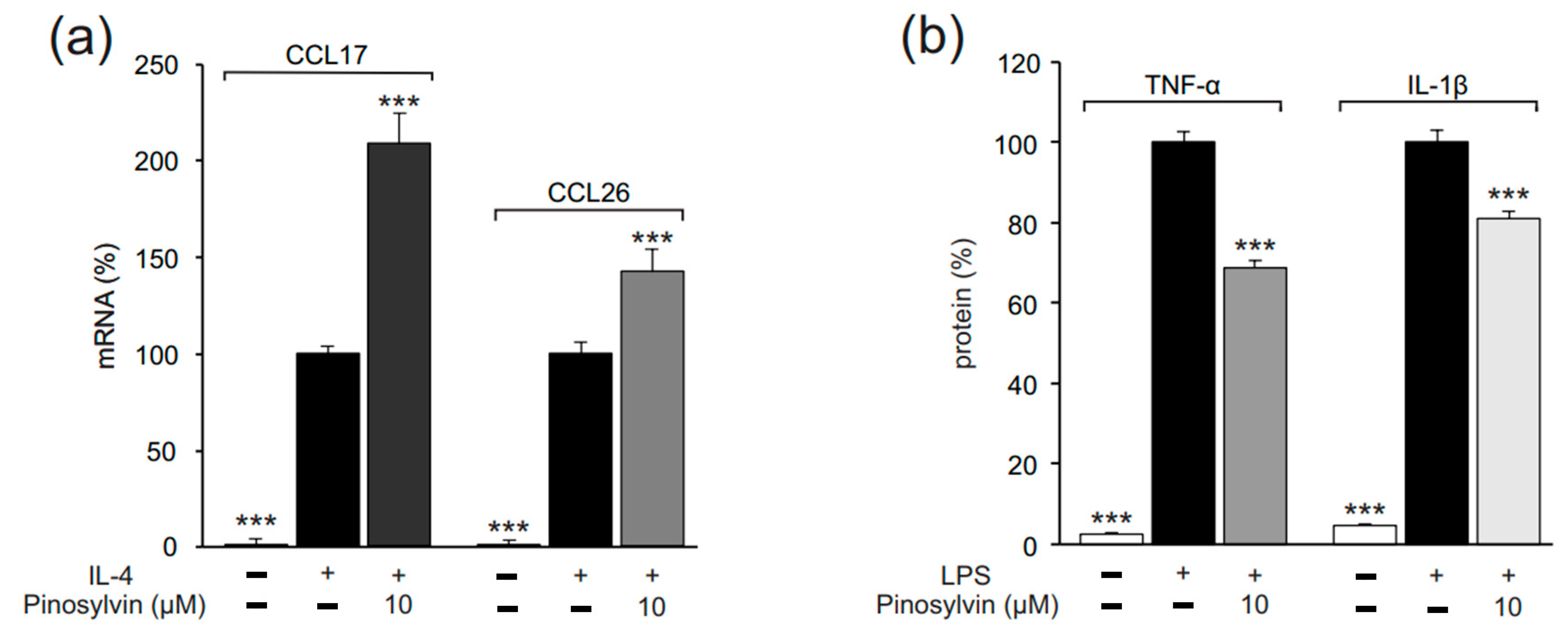
2.1. Pinosylvin Enhances M2-Type Macrophage Activation in Murine J774 Cells
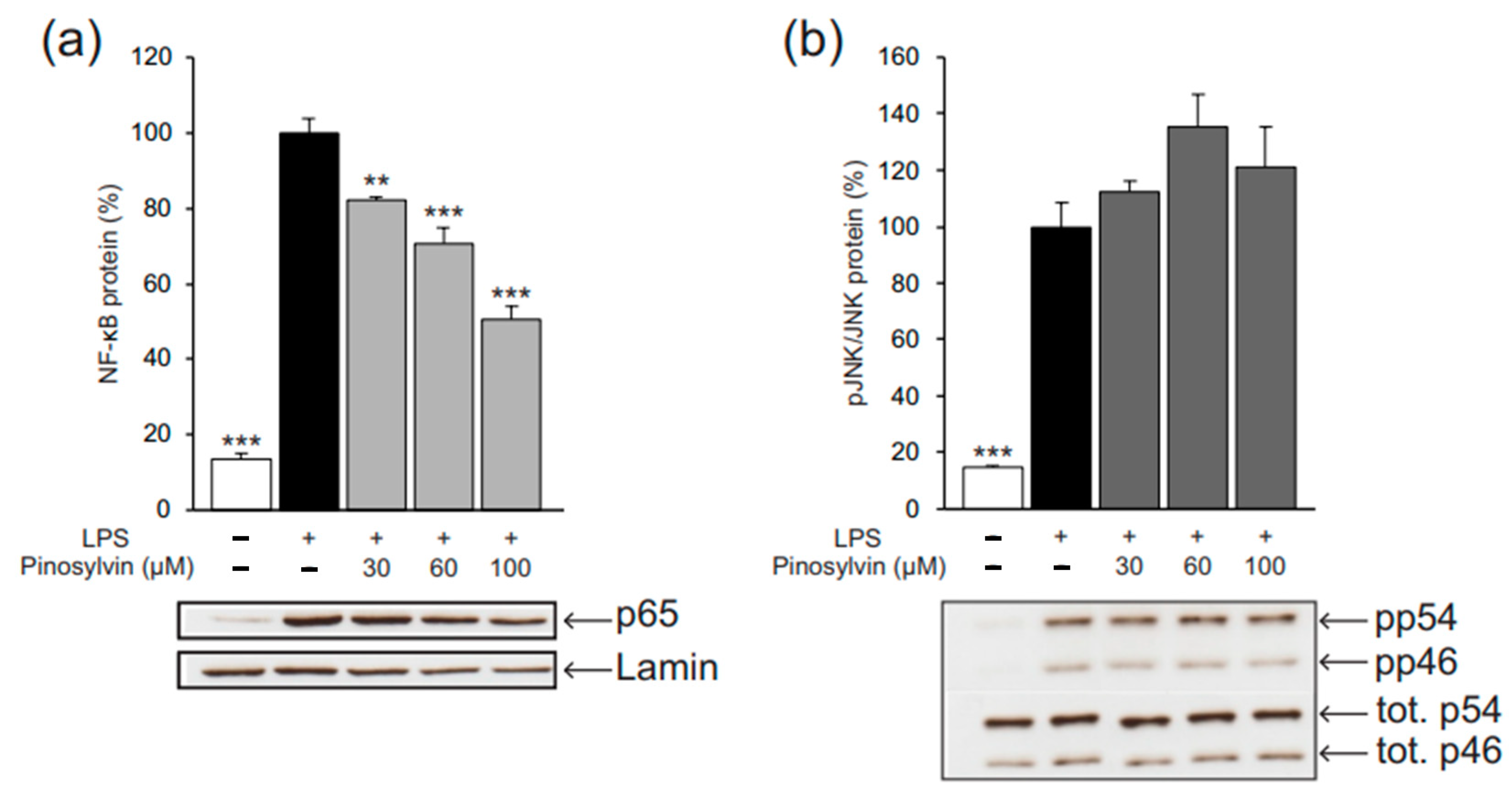
2.2. Pinosylvin Inhibits M1-Type Macrophage Activation in Murine J774 Cells
2.3. Pinosylvin Induces M2 Polarization Also in Human U937 Macrophages
3. Discussion
4. Materials and Methods
4.1. Compounds
4.2. Macrophage Polarization Experiments
4.3. Measurement of mRNA Levels by Quantitative RT-PCR
4.4. Measurement of Protein Levels by Western Blotting
4.5. Cytokine and Nitrite Measurements in Cell Culture Media by ELISA and Griess Method
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Atri, C.; Guerfali, F.Z.; Laouini, D. Role of human macrophage polarization in inflammation during infectious diseases. Int. J. Mol. Sci. 2018, 19, 1801. [Google Scholar] [CrossRef]
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.A.; Mardani, F.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 2018, 233, 6425–6440. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Huang, C.; Lin, Z.; Zhan, S.; Kong, L.; Fang, C.; Li, J. Macrophage polarization and function with emphasis on the evolving roles of coordinated regulation of cellular signaling pathways. Cell. Signal. 2014, 26, 192–197. [Google Scholar] [CrossRef]
- Gordon, S.; Martinez, F.O. Alternative activation of macrophages: Mechanism and functions. Immunity 2010, 32, 593–604. [Google Scholar] [CrossRef] [PubMed]
- Ehrchen, J.M.; Roth, J.; Barczyk-kahlert, K. More than suppression: Glucocorticoid action on monocytes and macrophages. Front. Immunol. 2019, 10, 2028. [Google Scholar] [CrossRef]
- Rivière, C.; Pawlus, A.D.; Mérillon, J.M. Natural stilbenoids: Distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar] [CrossRef] [PubMed]
- Akinwumi, B.C.; Bordun, K.A.M.; Anderson, H.D. Biological activities of stilbenoids. Int. J. Mol. Sci. 2018, 19, 792. [Google Scholar] [CrossRef] [PubMed]
- Pecyna, P.; Wargula, J.; Murias, M.; Kucinska, M. More than resveratrol: New insights into stilbene-based compounds. Biomolecules 2020, 10, 1111. [Google Scholar] [CrossRef]
- Eräsalo, H.; Hämäläinen, M.; Leppänen, T.; Mäki-Opas, I.; Laavola, M.; Haavikko, R.; Yli-Kauhaluoma, J.; Moilanen, E. Natural stilbenoids have anti-inflammatory properties in Vivo and down-regulate the production of inflammatory mediators NO, IL6, and MCP1 possibly in a PI3K/Akt-dependent manner. J. Nat. Prod. 2018, 81, 1131–1142. [Google Scholar] [CrossRef]
- Laavola, M.; Nieminen, R.; Leppänen, T.; Eckerman, C.; Holmbom, B.; Moilanen, E. Pinosylvin and monomethylpinosylvin, constituents of an extract from the knot of Pinus sylvestris, reduce inflammatory gene expression and inflammatory responses in Vivo. J. Agric. Food Chem. 2015, 63, 3445–3453. [Google Scholar] [CrossRef]
- Laavola, M.; Leppänen, T.; Hämäläinen, M.; Vuolteenaho, K.; Moilanen, T.; Nieminen, R.; Moilanen, E. IL-6 in osteoarthritis: Effects of pine stilbenoids. Molecules 2018, 24, 109. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, H.; Wang, L.; Tao, Y.; Du, G.; Guan, W.; Liu, J.; Brennan, C.; Ho, C.T.; Li, S. Effects of selected resveratrol analogues on activation and polarization of lipopolysaccharide-stimulated BV-2 microglial cells. J. Agric. Food Chem. 2020, 68, 3750–3757. [Google Scholar] [CrossRef] [PubMed]
- Hsu, A.T.; Lupancu, T.J.; Lee, M.C.; Fleetwood, A.J.; Cook, A.D.; Hamilton, J.A.; Achuthan, A. Epigenetic and transcriptional regulation of IL4-induced CCL17 production in human monocytes and murine macrophages. J. Biol. Chem. 2018, 293, 11415–11423. [Google Scholar] [CrossRef] [PubMed]
- Stubbs, V.E.L.; Power, C.; Patel, K.D. Regulation of eotaxin-3/CCL26 expression in human monocytic cells. Immunology 2010, 130, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, L. Influence of resveratrol on the immune response. Nutrients 2019, 11, 946. [Google Scholar] [CrossRef]
- Pinheiro, D.M.L.; de Oliveira, A.H.S.; Coutinho, L.G.; Fontes, F.L.; de Medeiros Oliveira, R.K.; Oliveira, T.T.; Faustino, A.L.F.; Lira da Silva, V.; de Melo Campos, J.T.A.; Lajus, T.B.P.; et al. Resveratrol decreases the expression of genes involved in inflammation through transcriptional regulation. Free Radic. Biol. Med. 2019, 130, 8–22. [Google Scholar] [CrossRef]
- Hajizadeh-Sharafabad, F.; Sahebkar, A.; Zabetian-Targhi, F.; Maleki, V. The impact of resveratrol on toxicity and related complications of advanced glycation end products: A systematic review. BioFactors 2019, 45, 651–665. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, L.J.; Hämäläinen, M.; Lehtimäki, L.; Nieminen, R.M.; Muraki, K.; Moilanen, E. Pinosylvin inhibits TRPA1-induced calcium influx in Vitro and TRPA1-mediated acute paw inflammation in Vivo. Basic Clin. Pharmacol. Toxicol. 2016, 118, 238–242. [Google Scholar] [CrossRef]
- Lim, S.J.; Kim, M.; Randy, A.; Nho, C.W. Inhibitory effect of the branches of Hovenia dulcis Thunb. and its constituent pinosylvin on the activities of IgE-mediated mast cells and passive cutaneous anaphylaxis in mice. Food Funct. 2015, 6, 1361–1370. [Google Scholar] [CrossRef]
- Bauerova, K.; Acquaviva, A.; Ponist, S.; Gardi, C.; Vecchio, D.; Drafi, F.; Arezzini, B.; Bezakova, L.; Kuncirova, V.; Mihalova, D.; et al. Markers of inflammation and oxidative stress studied in adjuvant-induced arthritis in the rat on systemic and local level affected by pinosylvin and methotrexate and their combination. Autoimmunity 2015, 48, 46–56. [Google Scholar] [CrossRef]
- Bekhbat, M.; Rowson, S.A.; Neigh, G.N. Checks and balances: The glucocorticoid receptor and NFĸB in good times and bad. Front. Neuroendocrinol. 2017, 46, 15–31. [Google Scholar] [CrossRef]
- Heming, M.; Gran, S.; Jauch, S.L.; Fischer-Riepe, L.; Russo, A.; Klotz, L.; Hermann, S.; Schäfers, M.; Roth, J.; Barczyk-Kahlert, K. Peroxisome proliferator-activated receptor-γ modulates the response of macrophages to lipopolysaccharide and glucocorticoids. Front. Immunol. 2018, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Nummenmaa, E.; Hämäläinen, M.; Moilanen, T.; Vuolteenaho, K.; Moilanen, E. Effects of FGF-2 and FGF receptor antagonists on MMP enzymes, aggrecan, and type II collagen in primary human OA chondrocytes. Scand. J. Rheumatol. 2015, 44, 321–330. [Google Scholar] [CrossRef]
- Salonen, T.; Sareila, O.; Jalonen, U.; Kankaanranta, H.; Tuominen, R.; Moilanen, E. Inhibition of classical PKC isoenzymes downregulates STAT1 activation and iNOS expression in LPS-treated murine J774 macrophages. Br. J. Pharmacol. 2006, 147, 790–799. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Nummenmaa, E.; Hämäläinen, M.; Moilanen, L.J.; Paukkeri, E.-L.; Nieminen, R.M.; Moilanen, T.; Vuolteenaho, K.; Moilanen, E. Transient receptor potential ankyrin 1 (TRPA1) is functionally expressed in primary human osteoarthritic chondrocytes. Osteoarthr. Cartil. 2016, 24, S329. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]


| Primer/Probe | Sequence | |
|---|---|---|
| mArg-1 | forward | 5′-TCCAAGCCAAAGTCCTTAGAGATTAT-3′ |
| reverse | 5′-CGTCATACTCTGTTTCTTTAAGTTTTTCC-3′ | |
| probe | 5′-CGCCTTTCTCAAAAGGACAGCCTCGA-3′ | |
| mYm1 | forward | 5′-AGTGGGTTGGTTATGACAATGTCA-3′ |
| reverse | 5′-GACCACGGCACCTCCTAAATT-3′ | |
| probe | 5′-AGCTTCAAGTTGAAGGCTCAGTGGCTCA-3′ | |
| mGAPDH | forward | 5′-GCATGGCCTTCCGTGTTC-3′ |
| reverse | 5′-GATGTCATCATACTTGGCAGGTTT-3′ | |
| probe | 5′-TCGTGGATCTGACGTGCCGCC-3′ | |
| hGAPDH | forward | 5′-AAGGTCGGAGTCAACGGATTT-3′ |
| reverse | 5′-GCAACAATATCCACTTTACCAGAGTTAA-3′ | |
| probe | 5′-CGCCTGGTCACCAGGGCTGC-3′ | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kivimäki, K.; Leppänen, T.; Hämäläinen, M.; Vuolteenaho, K.; Moilanen, E. Pinosylvin Shifts Macrophage Polarization to Support Resolution of Inflammation. Molecules 2021, 26, 2772. https://doi.org/10.3390/molecules26092772
Kivimäki K, Leppänen T, Hämäläinen M, Vuolteenaho K, Moilanen E. Pinosylvin Shifts Macrophage Polarization to Support Resolution of Inflammation. Molecules. 2021; 26(9):2772. https://doi.org/10.3390/molecules26092772
Chicago/Turabian StyleKivimäki, Konsta, Tiina Leppänen, Mari Hämäläinen, Katriina Vuolteenaho, and Eeva Moilanen. 2021. "Pinosylvin Shifts Macrophage Polarization to Support Resolution of Inflammation" Molecules 26, no. 9: 2772. https://doi.org/10.3390/molecules26092772
APA StyleKivimäki, K., Leppänen, T., Hämäläinen, M., Vuolteenaho, K., & Moilanen, E. (2021). Pinosylvin Shifts Macrophage Polarization to Support Resolution of Inflammation. Molecules, 26(9), 2772. https://doi.org/10.3390/molecules26092772






