Characterization of Different Salt Forms of Chitooligosaccharides and Their Effects on Nitric Oxide Secretion by Macrophages
Abstract
1. Introduction
2. Results and Discussion
2.1. Morphology of Chitooligosaccharide and Its Salts
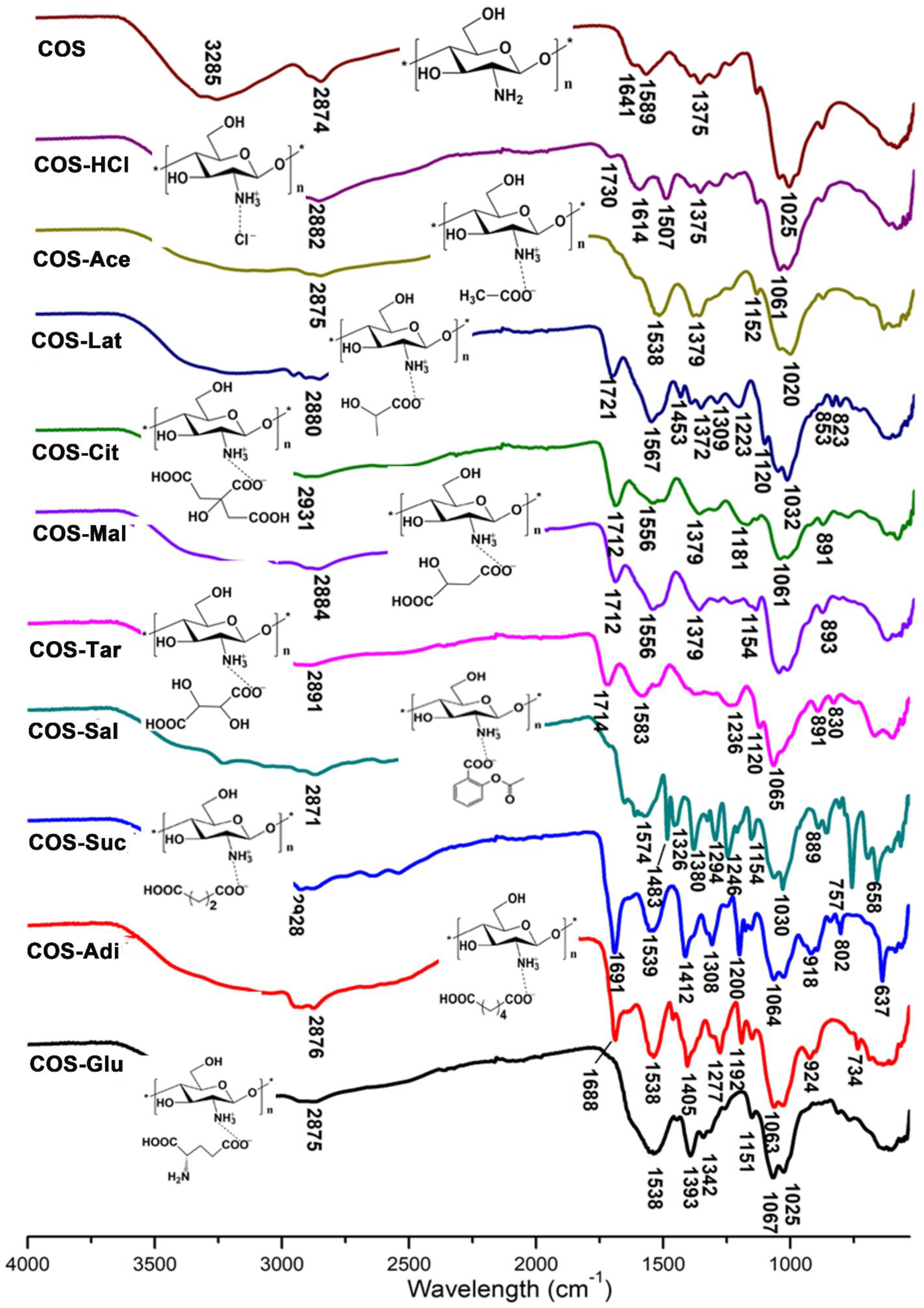
2.2. FTIR Analysis of Chitooligosaccharide and Its Salts
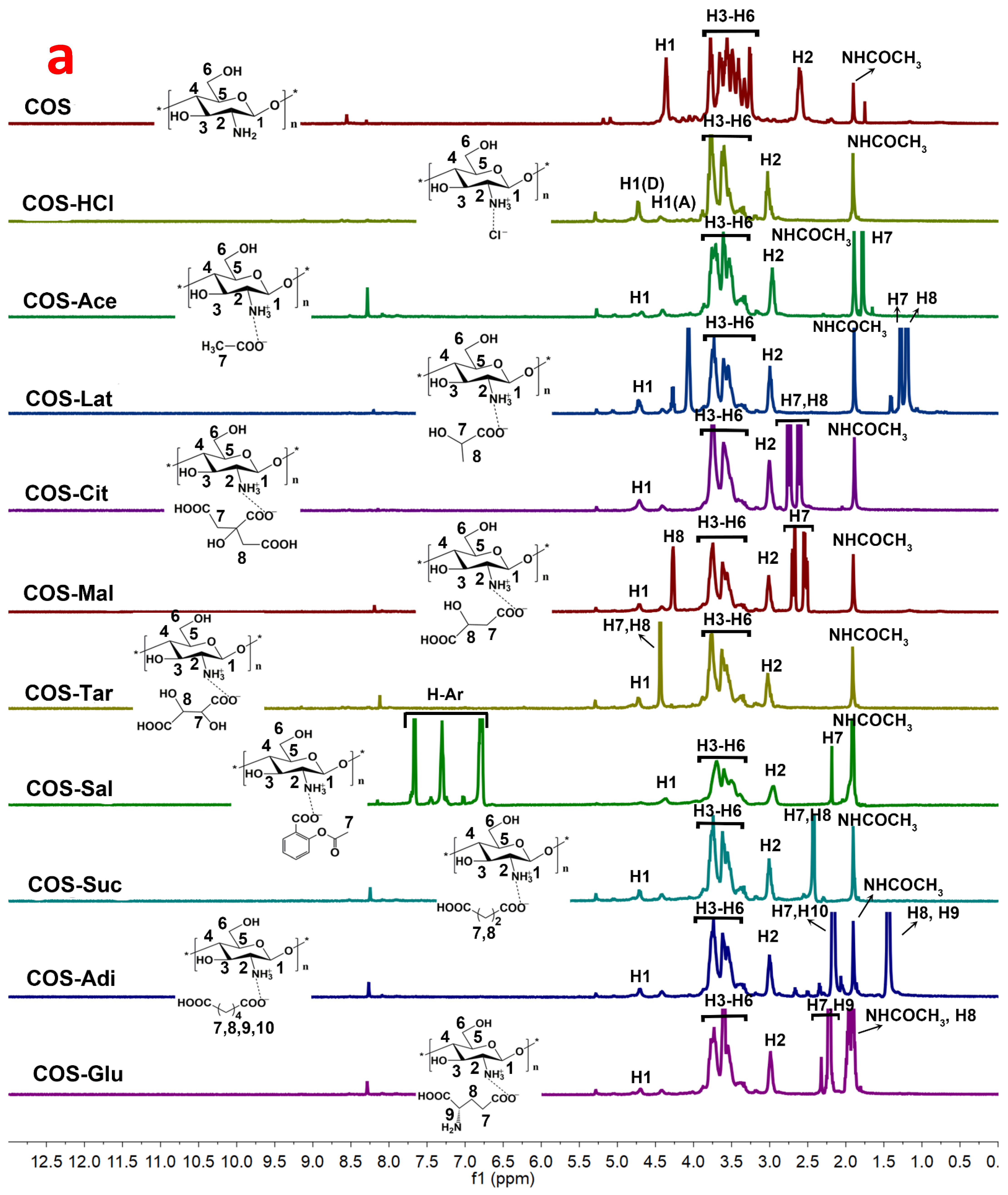
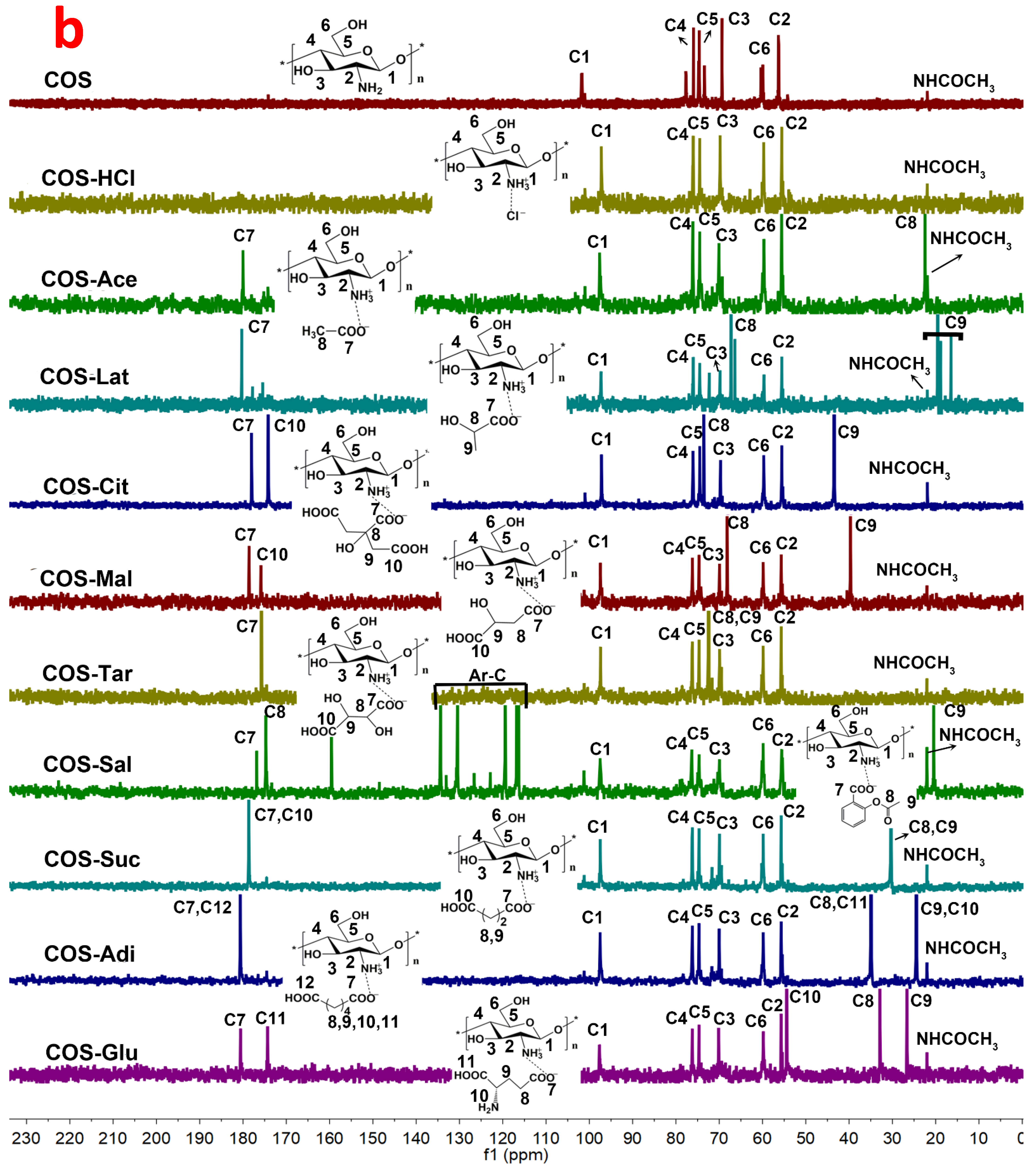
2.3. 1HNMR and 13CNMR Spectroscopy Analysis of Chitooligosaccharide and Its Salts
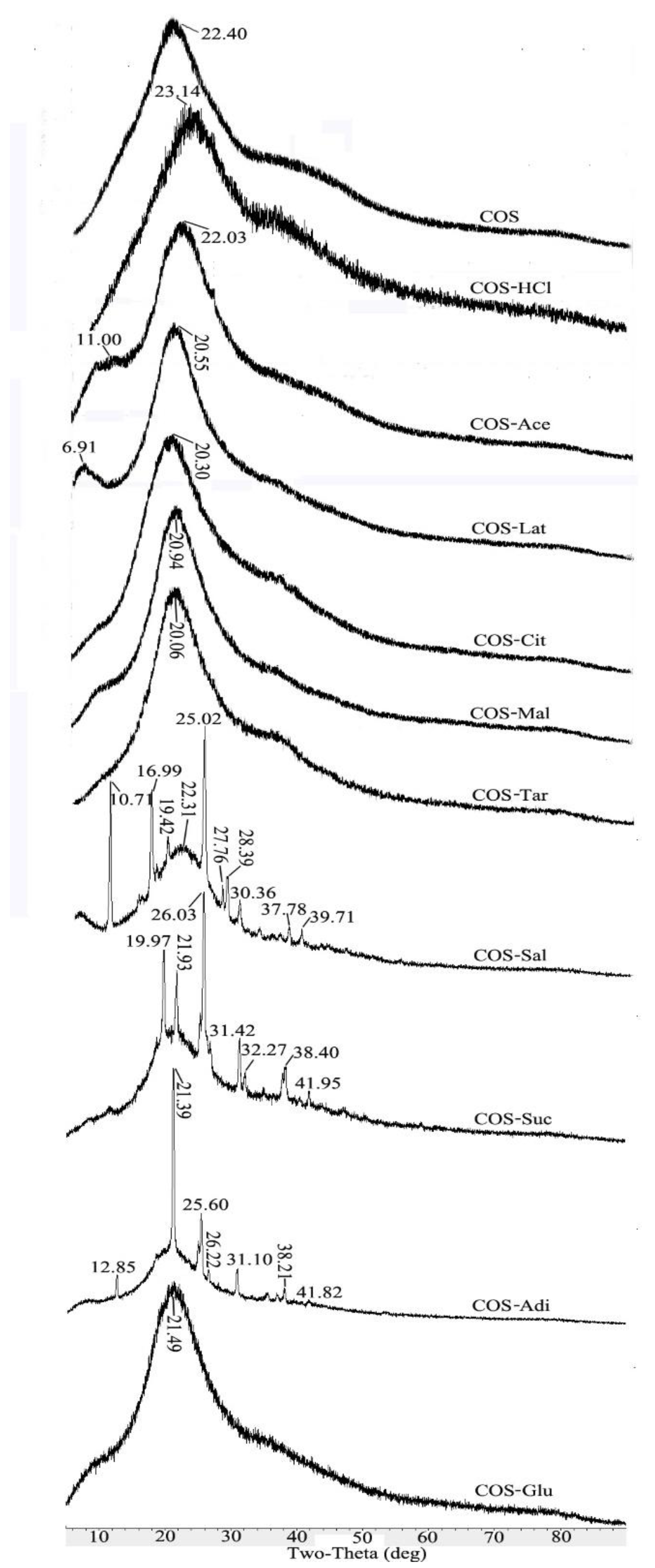
2.4. XRD Analysis of Chitooligosaccharide and Its Salts
2.5. Thermal Behavior Analysis of Chitooligosaccharide and Its Salts
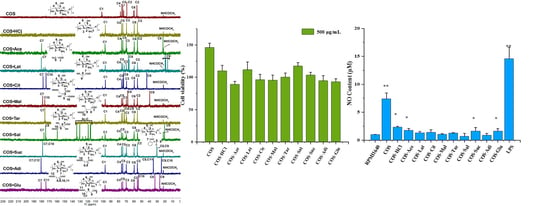
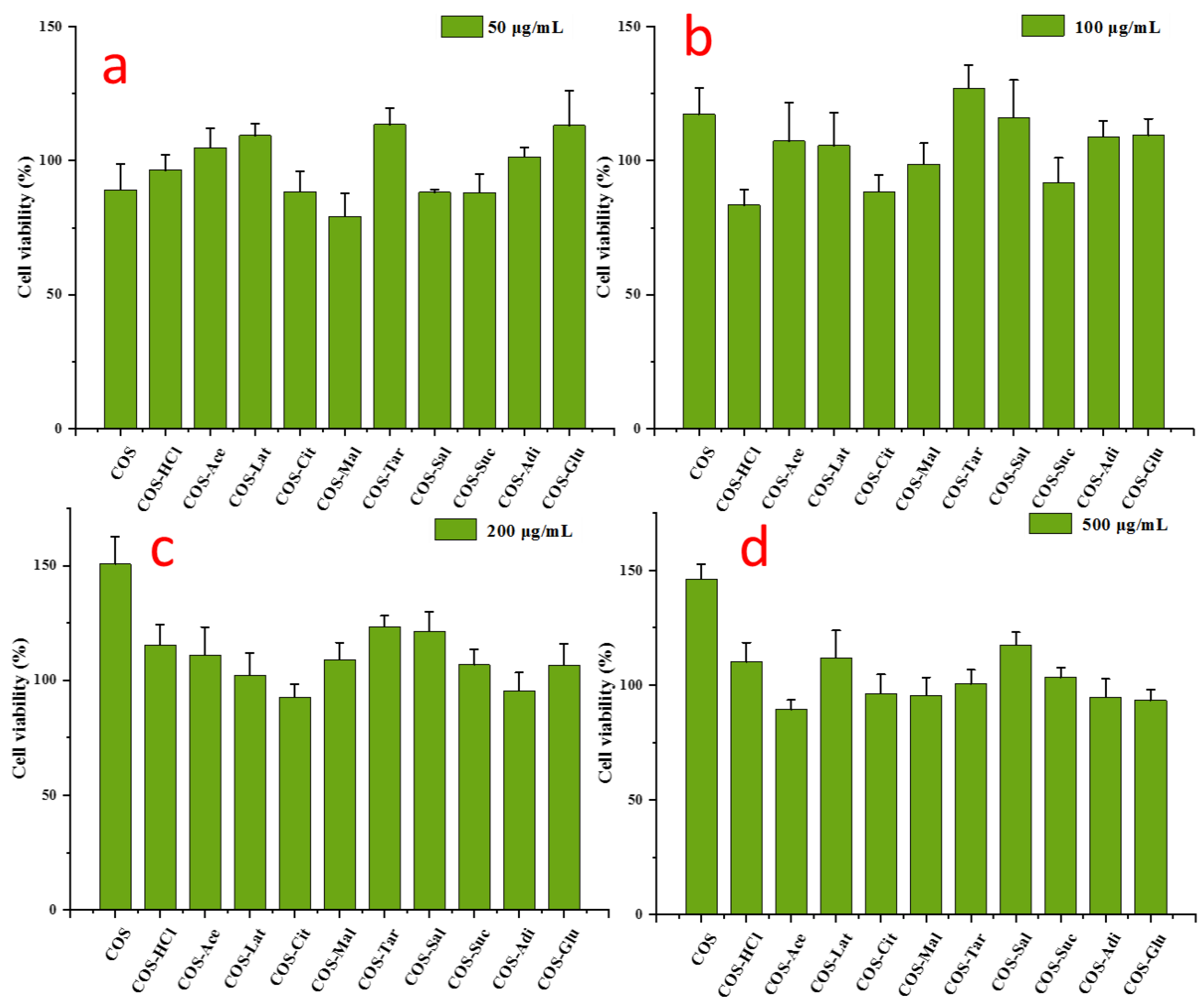
2.6. Cell Viability
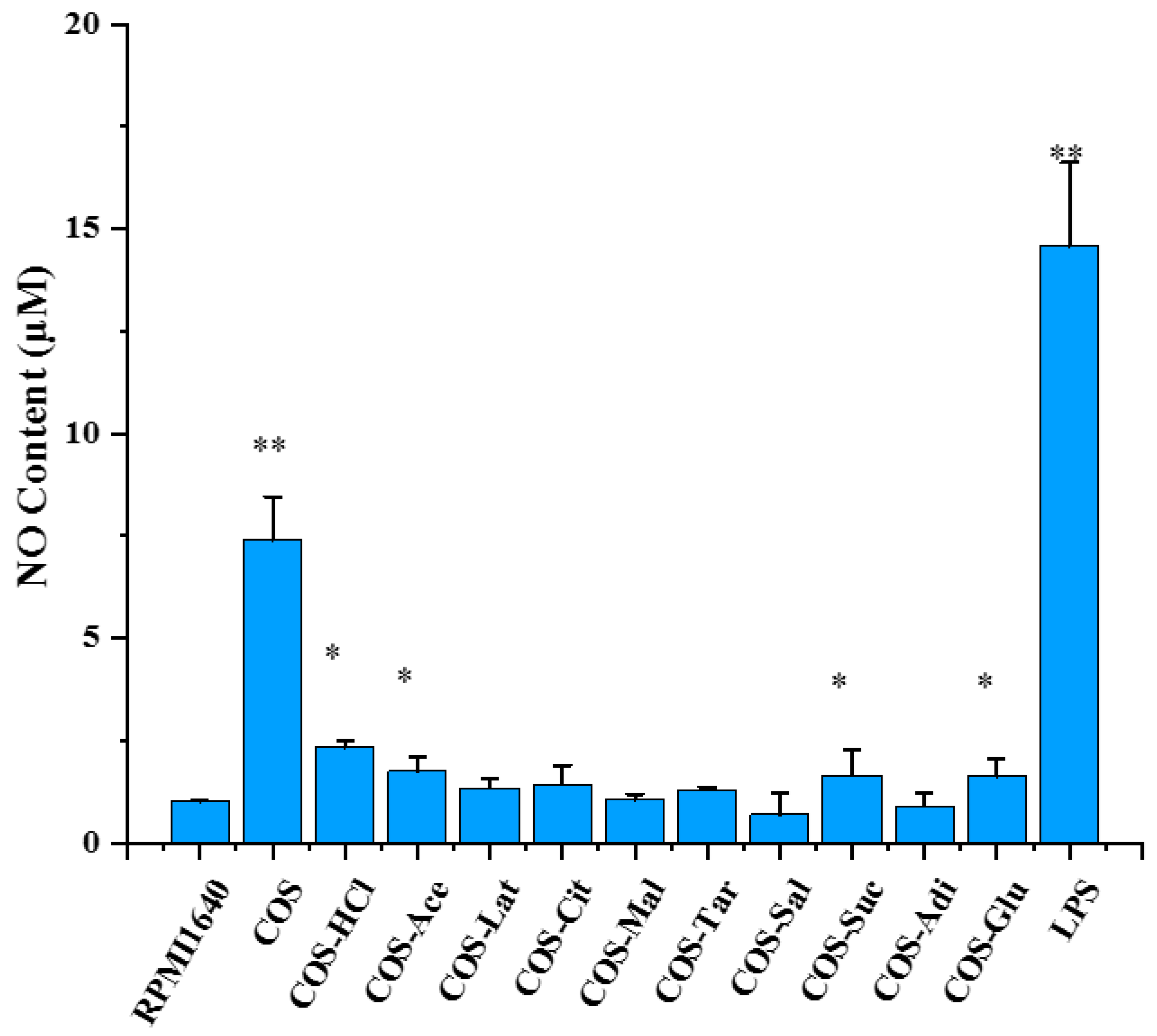
2.7. The Effects of Chitooligosaccharide and Its Salts on Nitric Oxide (NO) Secretion
3. Materials and Methods
3.1. Materials
3.2. Methods
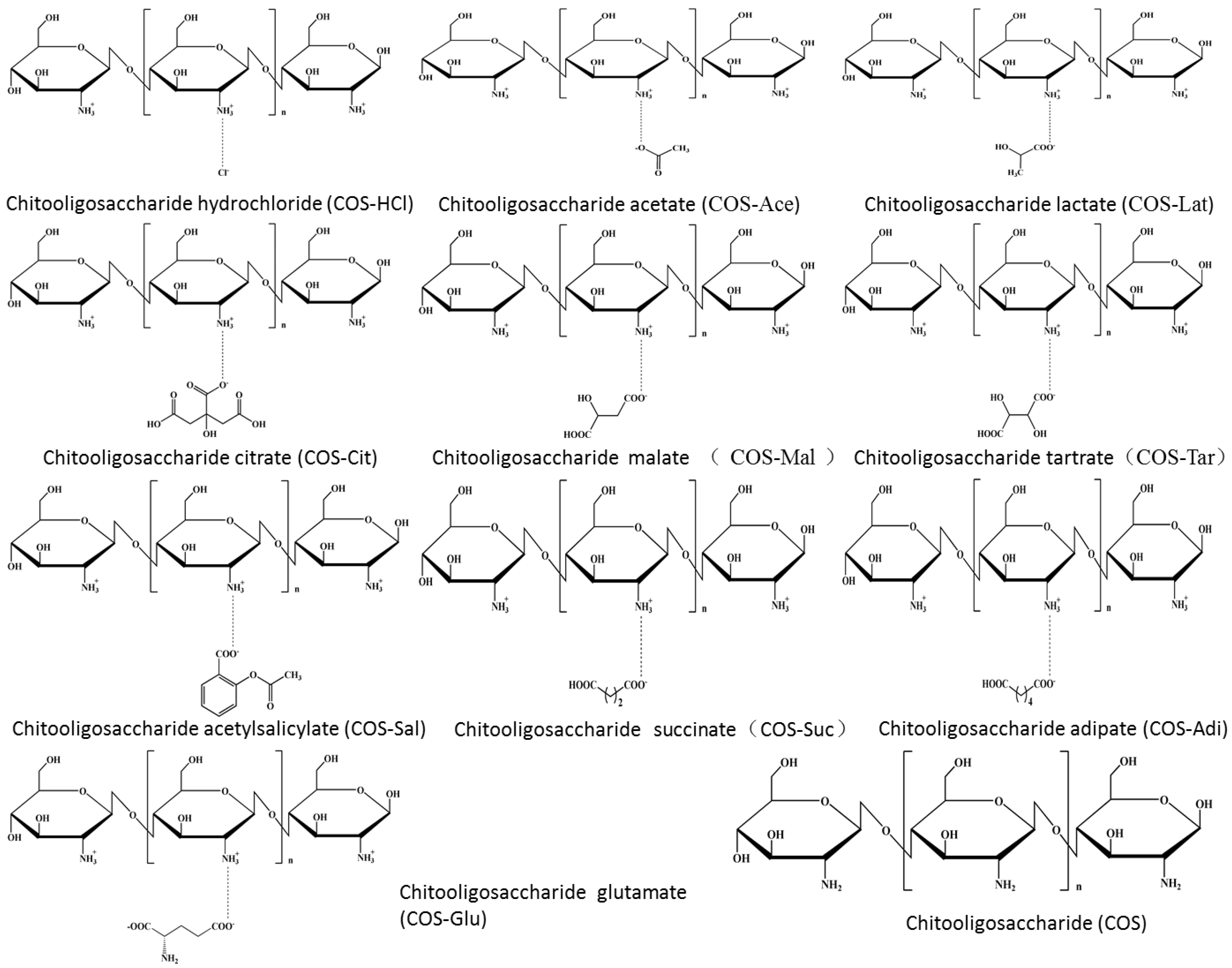
3.2.1. Preparation of Chitooligosaccharides with Different Salt Forms
3.2.2. Characterization Methods
3.3. Cell Culture
3.4. Cell Viability Assay
3.5. Determination of Nitric Oxide (NO) Production in RAW 264.7 Cells
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Hamedi, H.; Moradi, S.; Hudson, S.M.; Tonelli, A.E. Chitosan based hydrogels and their applications for drug delivery in wound dressings: A review. Carbohydr. Polym. 2018, 199, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Rijal, N.P.; Adhikari, U.; Bhattarai, N. Production of electrospun chitosan for biomedical applications. In Chitosan Based Biomaterials. Volume 1: Fundamentals; Amber Jennings, J., Bumgardner, J.D., Eds.; Woodhead Publishing Series in Biomaterials; Part III; Elsevier: Kidlington, UK, 2017; Volume 122, Chapter 9; pp. 211–238. [Google Scholar]
- Badawy, M.E.I.; Rabea, E.I. Chitosan and its derivatives as active ingredients against plant pests and diseases. In Chitosan in the Preservation of Agricultural Commodities; Elsevier: Kidlington, UK, 2016; Chapter 7; pp. 179–216. [Google Scholar]
- Sharif, R.; Mujtaba, M.; Rahman, M.U.; Shalmani, A.; Ahmad, H.; Anwar, T.; Deng, T.C.; Wang, X.P. The multifunctional role of chitosan in horticultural crops: A review. Molecules 2018, 23, 872. [Google Scholar] [CrossRef]
- Gutiérrez, T.J. Chitosan applications in textile and food industry. In Chitosan—Derivatives, Composites and Applications; Ahmed, S., Ikram, S., Eds.; Scrivener Publishing LLC, Wiley: New York, NY, USA, 2017; pp. 185–232. [Google Scholar]
- Han, J.W.; Ruiz-Garcia, L.; Qian, J.P.; Yang, X.T. Food packaging: A comprehensive review and future trends. Compr. Rev. Food Sci. Food Saf. 2018, 17, 860–877. [Google Scholar] [CrossRef]
- Samyn, P.; Barhoum, A.; Ohlund, T.; Dufresne, A. Review: Nanoparticles and nanostructured materials in papermaking. J. Mater. Sci. 2018, 53, 146–184. [Google Scholar] [CrossRef]
- Song, Z.; Li, G.D.; Guan, F.X.; Liu, W.X. Application of chitin/chitosan and their derivatives in the papermaking industry. Polymers 2018, 10, 389. [Google Scholar] [CrossRef] [PubMed]
- Desbrières, J.; Guibal, E. Chitosan for wastewater treatment. Polym. Int. 2018, 67, 7–14. [Google Scholar] [CrossRef]
- Pakdel, P.R.; Peighambardoust, S.J. Review on recent progress in chitosan-based hydrogels for wastewater treatment application. Carbohydr. Polym. 2018, 201, 264–279. [Google Scholar] [CrossRef]
- Huanbutta, K.; Cheewatanakornkool, K.; Terada, K.; Nunthanid, J.; Sriamornsak, P. Impact of salt form and molecular weight of chitosan on swelling and drug release from chitosan matrix tablets. Carbohydr. Polym. 2013, 97, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Opanasopit, P.; Aumklad, P.; Kowapradit, J.; Ngawhiranpat, T.; Apirakaramwong, A.; Rojanarata, T. Effect of Salt Forms and Molecular Weight of Chitosans on In Vitro Permeability Enhancement in Intestinal Epithelial Cells (Caco-2). Pharm. Dev. Technol. 2007, 12, 447–455. [Google Scholar] [CrossRef]
- Tengamnuay, P.; Sahamethapat, A.; Sailasuta, A.; Mitra, A.K. Chitosans as nasal absorption enhancers of peptides: Comparison between free amine chitosans and soluble salts. Int. J. Pharm. 2000, 197, 53–67. [Google Scholar] [CrossRef]
- Kotzé, A.F.; Lueßen, H.L.; de Leeuw, B.J.; de Boer, A.G.; Verhoef, J.C.; Junginger, H.E. Comparison of the effect of different chitosan salts and N-trimethyl chitosan chloride on the permeability of intestinal epithelial cells (Caco-2). J. Control. Release 1998, 51, 35–46. [Google Scholar] [CrossRef]
- Cho, S.Y.; Lee, J.H.; Song, M.J.; Park, P.J.; Shin, E.S.; Sohn, J.H.; Seo, D.-B.; Lim, K.M.; Kim, W.G.; Lee, S.-J. Effects of Chitooligosaccharide Lactate Salt on Sleep Deprivation-Induced Fatigue in Mice. Biol. Pharm. Bull. 2010, 33, 1128–1132. [Google Scholar] [CrossRef] [PubMed]
- Hai, N.T.T.; Thu, L.H.; Nga, N.T.T.; Hoa, T.T.; Tuan, L.N.A.; Phu, D.V.; Hien, N.Q. Preparation of Chitooligosaccharide by Hydrogen Peroxide Degradation of Chitosan and Its Effect on Soybean Seed Germination. J. Polym. Environ. 2019, 27, 2098–2104. [Google Scholar] [CrossRef]
- Li, M.; Han, J.; Xue, Y.; Dai, Y.F.; Liu, J.; Gan, L.H.; Xie, R.S.; Long, M.N. Hydrogen peroxide pretreatment efficiently assisting enzymatic hydrolysis of chitosan at high concentration for chitooligosaccharides. Polym. Degrad. Stab. 2019, 164, 177–186. [Google Scholar] [CrossRef]
- Qin, C.Q.; Dua, Y.M.; Xiao, L. Effect of hydrogen peroxide treatment on the molecular weight and structure of chitosan. Polym. Degrad. Stab. 2002, 76, 211–218. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, W.; Peng, Y.; Han, B.; Yang, Y. Toll like receptor 4 (TLR4) mediates the stimulating activities of chitosan oligosaccharide on macrophages. Int. Immunopharmacol. 2014, 23, 254–261. [Google Scholar] [CrossRef]
- Zheng, B.; Wen, Z.S.; Huang, Y.J.; Xia, M.S.; Xiang, X.W.; Qu, Y.L. Molecular weight-dependent immunostimulative activity of low molecular weight chitosan via regulating NF-κB and AP-1 signaling pathways in RAW264. 7 macrophages. Marine Drugs 2016, 14, 169. [Google Scholar] [CrossRef]
- Peniche, C.; Arguelles-Monal, W.; Davidenko, N.; Sastre, R.; Gallardo, A.; San Roman, J. Self-curing membranes of chitosan/PAA IPNs obtained by radical polymerization: Preparation, characterization and interpolymer complexation. Biomaterials 1999, 20, 1869–1878. [Google Scholar] [CrossRef]
- Vasilieva, T.; Sigarev, A.; Kosyakov, D.; Ul’yanovskii, N.; Anikeenko, E.; Chuhchin, D.; Ladesov, A.; Hein, A.M.; Miasnikov, V. Formation of low molecular weight oligomers from chitin and chitosan stimulated by plasma-assisted processes. Carbohydr. Polym. 2017, 163, 54–61. [Google Scholar] [CrossRef]
- Zajac, A.; Hanuza, J.; Wandas, M.; Dyminska, L. Determination of N-acetylation degree in chitosan using Raman spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 134, 114–120. [Google Scholar] [CrossRef]
- Orienti, I.; Cerchiara, T.; Luppi, B.; Bigucci, F.; Zuccari, G.; Zecchi, V. Influence of different chitosan salts on the release of sodium diclofenac in colon-specific delivery. Int. J. Pharm. 2002, 238, 51–59. [Google Scholar] [CrossRef]
- Nunthanid, J.; Wanchana, S.; Sriamornsak, P.; Limmattavapirat, S.; Luangtana-anan, M.; Puttipipatkhachorn, S. Effect of heat on characteristics of chitosan films coated on theophylline tablets. Drug Dev. Ind. Pharm. 2002, 28, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Liu, Y.; Hu, K.; Zhao, B.Y. The depolymerization mechanism of chitosan by hydrogen peroxide. J. Mater. Sci. 2003, 38, 4709–4712. [Google Scholar] [CrossRef]
- Wu, C.H.; Tian, J.H.; Lia, S.; Wu, T.T.; Hu, Y.Q.; Chen, S.G.; Sugawara, T.; Ye, X.Q. Structural properties of films and rheology of film-forming solutions of chitosan gallate for food packaging. Carbohydr. Polym. 2016, 146, 10–19. [Google Scholar] [CrossRef]
- Wu, T.T.; Wu, C.H.; Xiang, Y.C.; Huang, J.Q.; Luan, L.L.; Chen, S.G.; Hu, Y.Q. Kinetics and mechanism of degradation of chitosan by combining sonolysis with H2O2/ascorbic acid. Rsc Adv. 2016, 6, 76280–76287. [Google Scholar] [CrossRef]
- Prashanth, K.V.H.; Kittur, F.S.; Tharanathan, R.N. Solid state structure of chitosan prepared under different N-deacetylating conditions. Carbohydr. Polym. 2002, 50, 27–33. [Google Scholar] [CrossRef]
- Tantiplapol, T.; Singsawat, Y.; Narongsil, N.; Damrongsakkul, S.; Saito, N.; Prasertsung, I. Influences of solution plasma conditions on degradation rate and properties of chitosan. Innov. Food Sci. Emerg. Technol. 2015, 32, 116–120. [Google Scholar] [CrossRef]
- Huang, Q.Z.; Wang, S.M.; Huang, J.F.; Zhuo, L.H.; Guo, Y.C. Study on the heterogeneous degradation of chitosan with hydrogen peroxide under the catalysis of phosphotungstic acid. Carbohydr. Polym. 2007, 68, 761–765. [Google Scholar] [CrossRef]
- Khalil, E.S.; Saad, B.; Negim, E.S.M.; Saleh, M.I. Novel water-soluble chitosan derivative prepared by graft polymerization of dicyandiamide: Synthesis, characterisation, and its antibacterial property. J. Polym. Res. 2015, 22, 1–12. [Google Scholar] [CrossRef]
- Yoksan, R.; Akashi, M. Low molecular weight chitosan-g-L-phenylalanine: Preparation, characterization, and complex formation with DNA. Carbohydr. Polym. 2009, 75, 95–103. [Google Scholar] [CrossRef]
- Fang, Y.; Xu, Z.; Shi, Y.; Pei, F.; Yang, W.J.; Ma, N.; Kimatu, B.M.; Liu, K.L.; Qiu, W.F.; Hu, Q.H. Protection mechanism of Se-containing protein hydrolysates from Se-enriched rice on Pb2+-induced apoptosis in PC12 and RAW264.7 cells. Food Chem. 2017, 219, 391–398. [Google Scholar] [CrossRef]
- Hartley, J.W.; Evans, L.H.; Green, K.Y.; Naghashfar, Z.; Macias, A.R.; Zerfas, P.M.; Ward, J.M. Expression of infectious murine leukemia viruses by RAW264.7 cells, a potential complication for studies with a widely used mouse macrophage cell line. Retrovirology 2008, 5, 1–6. [Google Scholar] [CrossRef]
- Yu, Y.; Shen, M.Y.; Wang, Z.J.; Wang, Y.X.; Xie, M.Y.; Xie, J.H. Sulfated polysaccharide from Cyclocarya paliurus enhances the immunomodulatory activity of macrophages. Carbohydr. Polym. 2017, 174, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Schepetkin, I.A.; Xie, G.; Kirpotina, L.N.; Klein, R.A.; Jutila, M.A.; Quinn, M.T. Macrophage immunomodulatory activity of polysaccharides isolated from Opuntia polyacantha. Int. Immunopharmacol. 2008, 8, 1455–1466. [Google Scholar] [CrossRef]
- Xu, X.F.; Yan, H.D.; Zhang, X.W. Structure and immuno-stimulating activities of a new heteropolysaccharide from Lentinula edodes. J. Agric. Food Chem. 2012, 60, 11560–11566. [Google Scholar] [CrossRef] [PubMed]
- Volman, J.J.; Helsper, J.P.; Wei, S.; Baars, J.J.; van Griensven, L.J.; Sonnenberg, A.S.; Mensink, R.P.; Plat, J. Effects of mushroom-derived β-glucan-rich polysaccharide extracts on nitric oxide production by bone marrow-derived macrophages and nuclear factor-κB transactivation in Caco-2 reporter cells: Can effects be explained by structure? Mol. Nutr. Food Res. 2010, 54, 268–276. [Google Scholar] [CrossRef] [PubMed]


| Number | Content of Chitosan (g) | Acid Species | Acid Concentration | Yield (%) | Molecular Weight (Da) |
|---|---|---|---|---|---|
| 1 | 5 | acetic acid | 2% | 110 | 981 |
| 2 | 5 | hydrochloric acid | 2% | 104 | 1230 |
| 3 | 5 | citric acid | 5% | 182 | 3039 |
| 4 | 5 | malic acid | 3% | 136 | 867 |
| 5 | 5 | tartaric acid | 4% | 148 | 1780 |
| 6 | 5 | adipic acid | 2% | 100 | 1089 |
| 7 | 5 | succinic acid | 2% | 140 | 1030 |
| 8 | 5 | acetylsalicylic acid | 5% | 162 | 1799 |
| 9 | 5 | lactic acid | 4% | 114 | 1137 |
| 10 | 5 | glutamic acid | 3% | 146 | 1020 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xing, R.; Xu, C.; Gao, K.; Yang, H.; Liu, Y.; Fan, Z.; Liu, S.; Qin, Y.; Yu, H.; Li, P. Characterization of Different Salt Forms of Chitooligosaccharides and Their Effects on Nitric Oxide Secretion by Macrophages. Molecules 2021, 26, 2563. https://doi.org/10.3390/molecules26092563
Xing R, Xu C, Gao K, Yang H, Liu Y, Fan Z, Liu S, Qin Y, Yu H, Li P. Characterization of Different Salt Forms of Chitooligosaccharides and Their Effects on Nitric Oxide Secretion by Macrophages. Molecules. 2021; 26(9):2563. https://doi.org/10.3390/molecules26092563
Chicago/Turabian StyleXing, Ronge, Chaojie Xu, Kun Gao, Haoyue Yang, Yongliang Liu, Zhaoqian Fan, Song Liu, Yukun Qin, Huahua Yu, and Pengcheng Li. 2021. "Characterization of Different Salt Forms of Chitooligosaccharides and Their Effects on Nitric Oxide Secretion by Macrophages" Molecules 26, no. 9: 2563. https://doi.org/10.3390/molecules26092563
APA StyleXing, R., Xu, C., Gao, K., Yang, H., Liu, Y., Fan, Z., Liu, S., Qin, Y., Yu, H., & Li, P. (2021). Characterization of Different Salt Forms of Chitooligosaccharides and Their Effects on Nitric Oxide Secretion by Macrophages. Molecules, 26(9), 2563. https://doi.org/10.3390/molecules26092563