Amygdalin: Toxicity, Anticancer Activity and Analytical Procedures for Its Determination in Plant Seeds
Abstract
1. Introduction
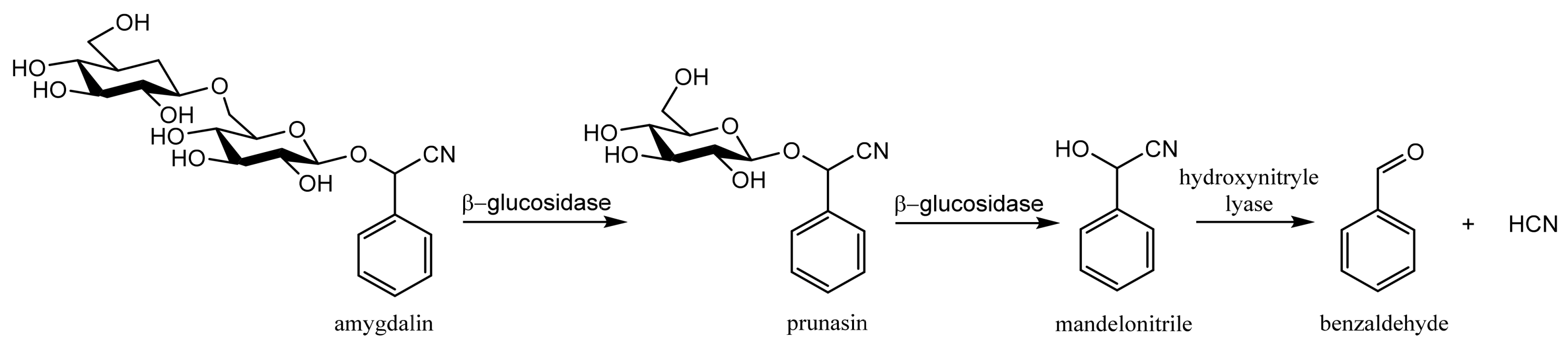
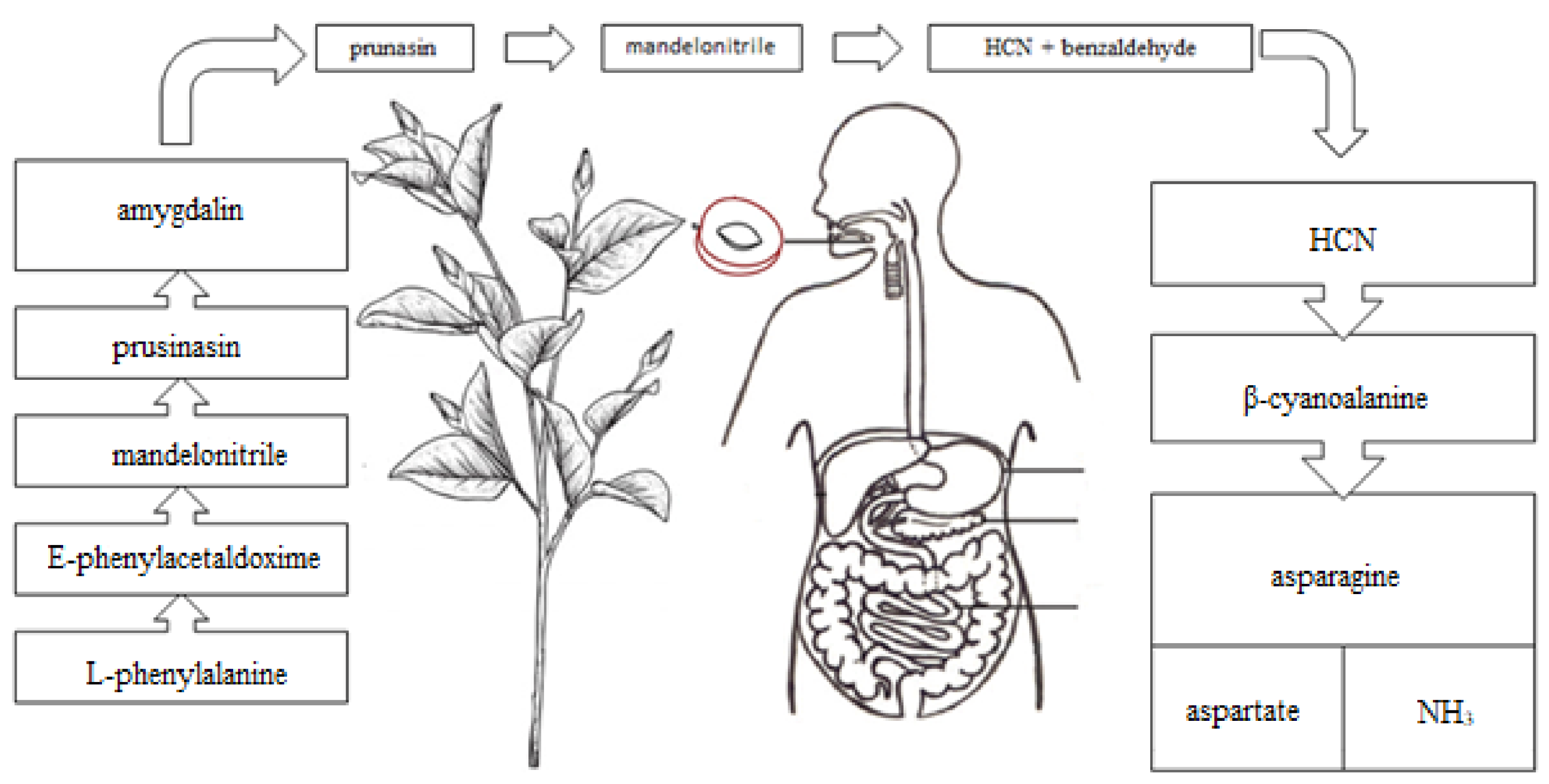
2. Amygdalin as a Member of Cyanogenic Glycosides—Their Sources, Toxicity and Anticancer Activity
2.1. Sources
2.2. Toxicity
2.3. The Anticancer and Other Biological Activities
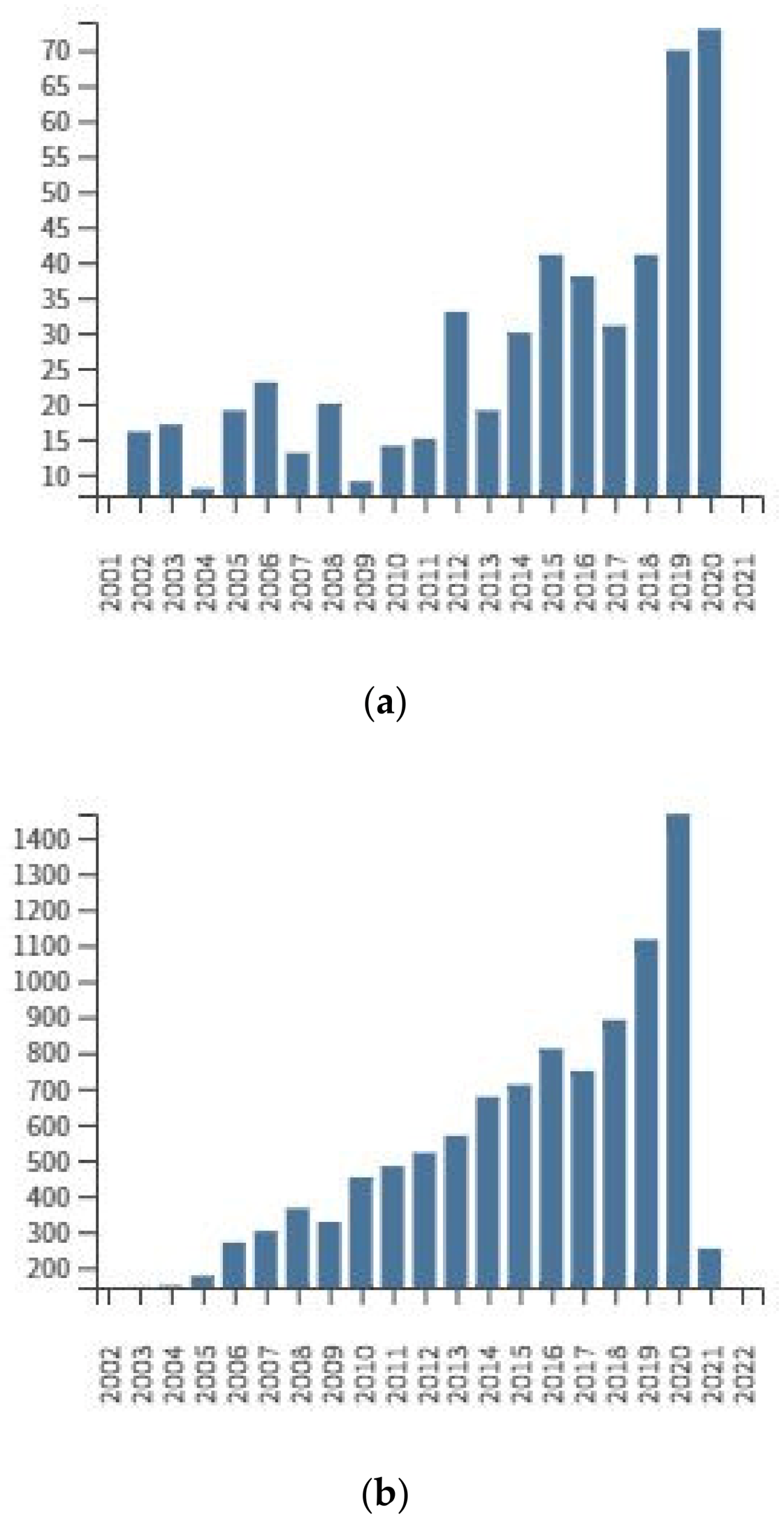
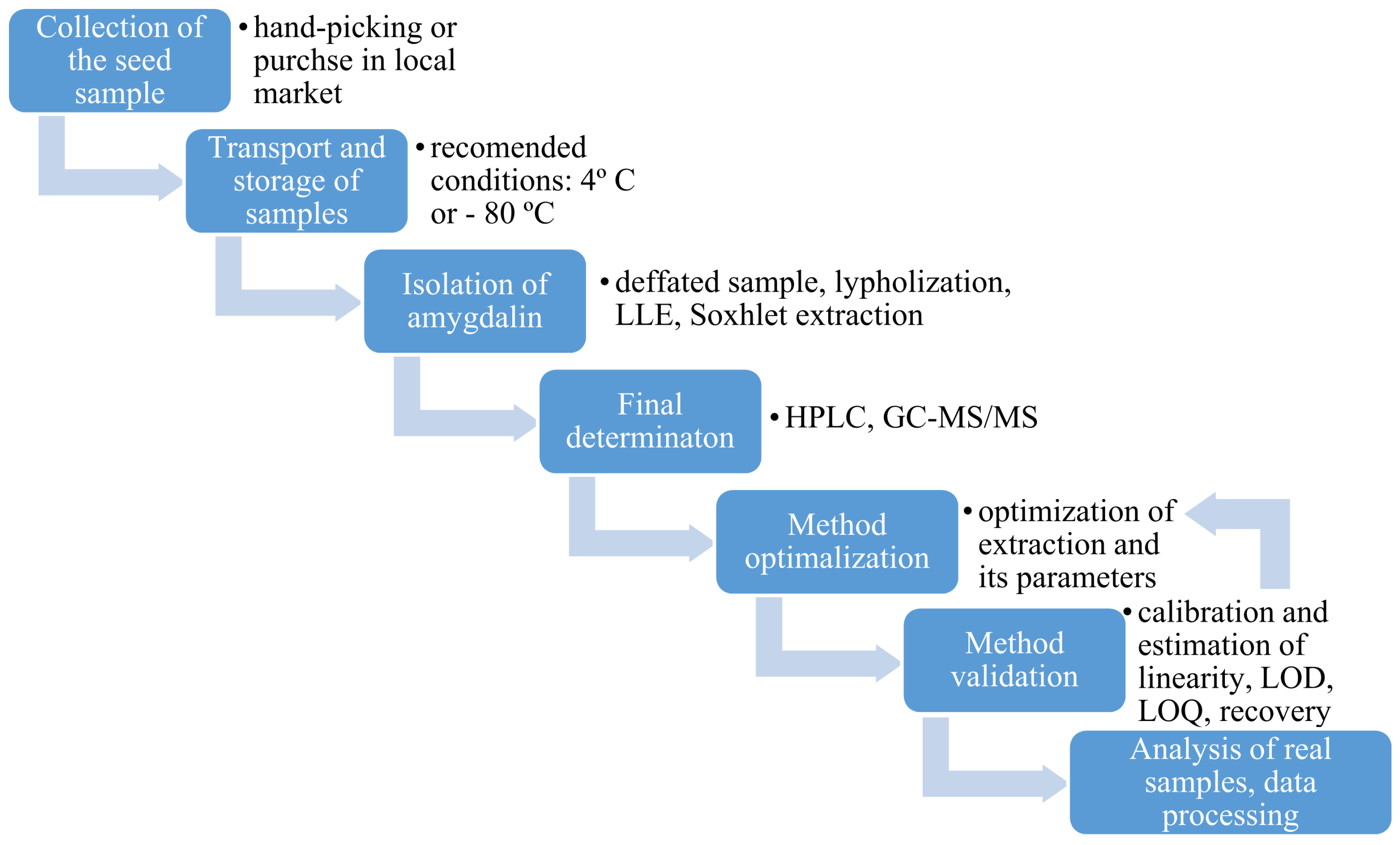
3. Determination of Amygdalin in Plant Seeds
3.1. Collection, Transport and Storage of Plant Seeds
3.2. Sample Preparation
3.3. Analytical Techniques for Plant Seeds Analysis
4. Future Trends and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Flies, E.J.; Mavoa, S.; Zosky, G.R.; Mantzioris, E.; Williams, C.; Eri, R.; Brook, B.W.; Buettel, J.C. Urban-associated diseases: Candidate diseases, environmental risk factors, and a path forward. Environ. Int. 2019, 133, 105187. [Google Scholar] [CrossRef]
- Bray, F.; Ferla, J.; Soerjomatarm, I.; Siegel, R.; Torre, L.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- World Cancer Research Fund/American Institute for Cancer Research Diet. Nutrition, Physical Activity and Cancer: A Global Perspective; International Peace Research Association: Solona, Sweden, 2018; ISBN 9781912259465. [Google Scholar]
- Sirota, H.; Rubovits, D.R.; Cousins, J.H.; Weinberg, A.D.; Laufman, L.; Lane, M. Cancer control: Prevention. Health Values 2007, 12, 33–36. [Google Scholar]
- Wojciechowska, U.; Didkowska, J.; Zatoński, W. Nowotwory złośliwe w Polsce w 2010 roku. Kraj. Rejestr Nowotworów 2010, 2012, 44–45. [Google Scholar] [CrossRef]
- Takayama, Y.; Kwai, S. Study on the prevention of racemization of amygdalin. Chem. Pharm. Bull. 1984, 32, 778–781. [Google Scholar] [CrossRef]
- Santos, S.B.; Sousa Lobo, J.M.; Silva, A.C. Biosimilar medicines used for cancer therapy in Europe: A review. Drug Discov. Today 2018, 24, 293–299. [Google Scholar] [CrossRef]
- Albala, K. Adaptation of Ideas from West to East. Petits Propos Culin. 2009, 88, 19–34. [Google Scholar]
- Chang, H.-K.; Shin, M.-S.; Yang, H.-Y.; Lee, J.-W.; Kim, Y.-S.; Lee, M.-H.; Kim, J.; Kim, K.-H.; Kim, C.-J. Amygdalin induces apoptosis through regulation of Bax and Bcl-2 expressions in human DU145 and LNCaP prostate cancer cells. Biol. Pharm. Bull. 2006, 29, 1597–1602. [Google Scholar] [CrossRef]
- Shim, S.M.; Kwon, H. Metabolites of amygdalin under simulated human digestive fluids. Int. J. Food Sci. Nutr. 2010, 61, 770–779. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Zhang, Y. Catalytic degradation of amygdalin by extracellular enzymes from Aspergillus niger. Process. Biochem. 2012, 47, 195–200. [Google Scholar] [CrossRef]
- Nowak, A.; Zielińska, A. Aktywność przeciwnowotworowa amigdaliny Anticancer activity of amygdalin. Postępy Fitoter. 2016, 17, 282–292. [Google Scholar]
- Arshi, A.; Hosseini, S.M.; Hosseini, F.S.K.; Amiri, Z.Y.; Hosseini, F.S.; Sheikholia Lavasani, M.; Kerdarian, H.; Dehkordi, M.S. The anti-cancer effect of amygdalin on human cancer cell lines. Mol. Biol. Rep. 2019, 46, 2059–2066. [Google Scholar] [CrossRef] [PubMed]
- Sireesha, D.; Reddy, B.S.; Reginald, B.A.; Samatha, M.; Kamal, F. Effect of amygdalin on oral cancer cell line: An in vitro study. J. Oral Maxillofac. Pathol. 2019, 23, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Rosen, G.; Shorr, R. Laetrile: End play around the FDA. A review of legal developments. Ann. Intern. Med. 1979, 90, 418–423. [Google Scholar] [CrossRef]
- Jukes, T.H. Laetrile for cancer. JAMA 1976, 236, 1284–1289. [Google Scholar] [CrossRef]
- Achmatowicz, O.; PTChem. Przewodnik do Nomenklatury Związków Organicznych; PTChem: Warszawa, Poland, 1994; ISBN 8390184427. [Google Scholar]
- Siegień Irena Cyjanogeneza u roslin i jej efektywność w ochronie roślin przed atakiem roślinożerców i patogenów. Kosmos. Probl. Nauk Biol. 2007, 2, 155–166.
- Zagrobelny, M.; Bak, S.; Rasmussen, A.V.; Jørgensen, B.; Naumann, C.M.; Møller, B.L. Cyanogenic glucosides and plant-insect interactions. Phytochemistry 2004, 65, 293–306. [Google Scholar] [CrossRef]
- Lee, S.H.; Oh, A.; Shin, S.H.; Kim, H.N.; Kang, W.W.; Chung, S.K. Amygdalin contents in peaches at different fruit development stages. Prev. Nutr. Food Sci. 2017, 22, 237–240. [Google Scholar] [CrossRef]
- Rezaul Haque, M.; Howard Bradbury, J. Total cyanide determination of plants and foods using the picrate and acid hydrolysis methods. Food Chem. 2002, 77, 107–114. [Google Scholar] [CrossRef]
- Del Cueto, J.; Ionescu, I.A.; Pičmanová, M.; Gericke, O.; Motawia, M.S.; Olsen, C.E.; Campoy, J.A.; Dicenta, F.; Møller, B.L.; Sánchez-Pérez, R. Cyanogenic Glucosides and Derivatives in Almond and Sweet Cherry Flower Buds from Dormancy to Flowering. Front. Plant. Sci. 2017, 8, 1–16. [Google Scholar] [CrossRef]
- Rietjens, I.M.C.M.; Martena, M.J.; Boersma, M.G.; Spiegelenberg, W.; Alink, G.M. Molecular mechanisms of toxicity of important food-borne phytotoxins. Mol. Nutr. Food Res. 2005, 49, 131–158. [Google Scholar] [CrossRef]
- Jaszczak, E.; Polkowska, Ż.; Narkowicz, S.; Namieśnik, J. Cyanides in the environment—analysis—problems and challenges. Environ. Sci. Pollut. Res. 2017, 24, 15929–15948. [Google Scholar] [CrossRef]
- Abraham, K.; Buhrke, T.; Lampen, A. Bioavailability of cyanide after consumption of a single meal of foods containing high levels of cyanogenic glycosides: A crossover study in humans. Arch. Toxicol. 2016, 90, 559–574. [Google Scholar] [CrossRef]
- Simeonova, F.; Fishbein, L. Hydrogen Cyanide and Cyanides: Human Health Aspects. World Health Organ. Geneva 2004. Available online: https://apps.who.int/iris/handle/10665/42942 (accessed on 20 February 2021).
- Kołaciński, Z.; Burda, P.; Łukasik-Głębocka, M.; Sein Anand, J. Postępowanie w ostrych zatruciach cyjankami-stanowisko Sekcji Toksykologii Klinicznej Polskiego Towarzystwa Lekarskiego. Przegl. Lek. 2011, 68, 8–11. [Google Scholar]
- Qadir, M.; Fatima, K. Review on Pharmacological Activity of Amygdalin. Arch. Cancer Res. 2017, 5, 10–12. [Google Scholar] [CrossRef]
- Blaheta, R.A.; Nelson, K.; Haferkamp, A.; Juengel, E. Amygdalin, quackery or cure? Phytomedicine 2016, 23, 367–376. [Google Scholar] [CrossRef]
- Sahin, S.; Kirel, B.; Carman, K. Fatal cyanide poisoning in a child, caused by eating apricot seeds. Am. J. Case Rep. 2011, 12, 70–72. [Google Scholar] [CrossRef]
- Sauer, H.; Wollny, C.; Oster, I.; Tutdibi, E.; Gortner, L.; Gottschling, S.; Meyer, S. Severe cyanide poisoning from an alternative medicine treatment with amygdalin and apricot kernels in a 4-year-old child. Wien. Med. Wochenschr. 2015, 165, 185–188. [Google Scholar] [CrossRef]
- Drankowska, J.; Kos, M.; Kościuk, A.; Tchórz, M. Cyanide poisoning from an alternative medicine treatment with apricot kernels in a 80- year-old female. J. Educ. Heal. Sport 2018, 8, 19–26. [Google Scholar]
- Tatli, M.; Eyüpoğlu, G.; Hocagil, H. Acute cyanide poisoning due to apricot kernel ingestion. J. Acute Dis. 2017, 6, 87–88. [Google Scholar] [CrossRef]
- Unproven methods of cancer management: Laetrile. CA. Cancer J. Clin. 1991, 41, 187–192. [CrossRef]
- Edward, J.C. Possible adverse side effects from treatment with laetrile. Med. Hypotheses 1979, 5, 1045–1049. [Google Scholar]
- Lewis, J.P. Laetrile. West. J. Med. 1977, 127, 55–62. [Google Scholar]
- Moss, R.W. Patient perspectives: Tijuana cancer clinics in post-NATFA era. Integr. Cancer Ther. 2005, 4, 65–86. [Google Scholar] [CrossRef]
- Kamil, A.; Chen, C.Y.O. Health benefits of almonds beyond cholesterol reduction. J. Agric. Food Chem. 2012, 60, 6694–6702. [Google Scholar] [CrossRef]
- Milazzo, S.; Lejeune, S.; Ernst, E. Laetrile for cancer: A systematic review of the clinical evidence. Support. Care Cancer 2007, 15, 583–595. [Google Scholar] [CrossRef]
- Chen, Y.; Ma, J.; Wang, F.; Hu, J.; Cui, A.; Wei, C.; Yang, Q.; Li, F. Amygdalin induces apoptosis in human cervical cancer cell line HeLa cells. Immunopharmacol. Immunotoxicol. 2013, 35, 43–51. [Google Scholar] [CrossRef]
- Makarević, J.; Rutz, J.; Juengel, E.; Kaulfuss, S.; Reiter, M.; Tsaur, I.; Bartsch, G.; Haferkamp, A.; Blaheta, R.A. Amygdalin blocks bladder cancer cell growth in vitro by diminishing cyclin A and cdk2. PLoS ONE 2014, 9, e0105590. [Google Scholar] [CrossRef]
- Al-Bakri, S.A.; Nima, Z.A.; Jabri, R.R.; Ajeel, E.A. Antibacterial activity of apricot kernel extract containing Amygdalin. Iraqi J. Sci. 2010, 51, 571–576. [Google Scholar]
- Savić, I.M.; Nikolić, V.D.; Savić-Gajić, I.M.; Kundaković, T.D.; Stanjković, T.P.; Najman, S.J. Chemical composition and biological activity of agroindustrial residues. Adv. Technol. 2016, 5, 38–45. [Google Scholar] [CrossRef]
- Yashunsky, D.V.; Kulakovskaya, E.V.; Kulakovskaya, T.V.; Zhukova, O.S.; Kiselevskiy, M.V.; Nifantiev, N.E. Synthesis and Biological Evaluation of Cyanogenic Glycosides. J. Carbohydr. Chem. 2015, 34, 460–474. [Google Scholar] [CrossRef]
- Walia, M.; Rawat, K.; Bhushan, S.; Padwad, Y.S.; Singh, B. Fatty acid composition, physicochemical properties, antioxidant and cytotoxic activity of apple seed oil obtained from apple pomace. J. Sci. Food Agric. 2014, 94, 929–934. [Google Scholar] [CrossRef]
- Waszkowiak, K.; Gliszczyńska-Świgło, A.; Barthet, V.; Skręty, J. Effect of Extraction Method on the Phenolic and Cyanogenic Glucoside Profile of Flaxseed Extracts and their Antioxidant Capacity. JAOCS J. Am. Oil Chem. Soc. 2015, 92, 1609–1619. [Google Scholar] [CrossRef]
- Luo, H.; Li, L.; Tang, J.; Zhang, F.; Zhao, F.; Sun, D.; Zheng, F.; Wang, X. Amygdalin inhibits HSC-T6 cell proliferation and fibrosis through the regulation of TGF-β/CTGF. Mol. Cell. Toxicol. 2016, 12, 265–271. [Google Scholar] [CrossRef]
- Guo, J.; Wu, W.; Sheng, M.; Yang, S.; Tan, J. Amygdalin inhibits renal fibrosis in chronic kidney disease. Mol. Med. Rep. 2013, 7, 1453–1457. [Google Scholar] [CrossRef]
- Hwang, H.J.; Lee, H.J.; Kim, C.J.; Shim, I.; Hahm, D.H. Inhibitory effect of amygdalin on lipopolysaccharide-inducible TNF-α and IL-1β mRNA expression and carrageenan-induced rat arthritis. J. Microbiol. Biotechnol. 2008, 18, 1641–1647. [Google Scholar]
- Baroni, A.; Paoletti, I.; Greco, R.; Satriano, R.A.; Ruocco, E.; Tufano, M.A.; Perez, J.J. Immunomodulatory effects of a set of amygdalin analogues on human keratinocyte cells. Exp. Dermatol. 2005, 14, 854–859. [Google Scholar] [CrossRef]
- Yang, C.; Li, X.; Rong, J. Amygdalin isolated from Semen Persicae (Tao Ren) extracts induces the expression of follistatin in HepG2 and C2C12 cell lines. Chin. Med. 2014, 9, 23. [Google Scholar] [CrossRef]
- Heikkila, R.E.; Cabbat, F.S. The prevention of alloxan-induced diabetes by amygdalin. Life Sci. 1980, 27, 659–662. [Google Scholar] [CrossRef]
- Kim, C.S.; Jo, K.; Lee, I.S.; Kim, J. Topical application of apricot kernel extract improves dry eye symptoms in a unilateral exorbital lacrimal gland excision mouse. Nutrients 2016, 8, 750. [Google Scholar] [CrossRef]
- Park, H.J.; Yoon, S.H.; Han, L.S.; Zheng, L.T.; Jung, K.H.; Uhm, Y.K.; Lee, J.H.; Jeong, J.S.; Joo, W.S.; Yim, S.V.; et al. Amygdalin inhibits genes related to cell cycle in SNU-C4 human colon cancer cells. World J. Gastroenterol. 2005, 11, 5156–5161. [Google Scholar] [CrossRef]
- Lee, H.M.; Moon, A. Amygdalin regulates apoptosis and adhesion in Hs578T triple-negative breast cancer cells. Biomol. Ther. 2016, 24, 62–66. [Google Scholar] [CrossRef]
- Moon, J.Y.; Kim, S.W.; Yun, G.M.; Lee, H.S.; Kim, Y.D.; Jeong, G.J.; Ullah, I.; Rho, G.J.; Jeon, B.G. Inhibition of cell growth and down-regulation of telomerase activity by amygdalin in human cancer cell lines. Animal Cells Syst. (Seoul) 2015. [Google Scholar] [CrossRef]
- Yildrim, F.; Askin, M.A. Variability of amygdalin content in seeds of sweet and bitter apricot cultivars in Turkey. African J. Biotechnol. 2010, 9, 6522–6524. [Google Scholar] [CrossRef]
- Wang, T.; Lu, S.; Xia, Q.; Fang, Z.; Johnson, S. Separation and purification of amygdalin from thinned bayberry kernels by macroporous adsorption resins. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 975, 52–58. [Google Scholar] [CrossRef]
- Bolarinwa, I.F.; Olaniyan, S.A.; Olatunde, S.J.; Ayandokun, F.T.; Olaifa, I.A. Effect of processing on amygdalin and cyanide contents of some Nigerian foods. J. Chem. Pharm. Res. 2016, 8, 106–113. [Google Scholar] [CrossRef]
- Savic, I.M.; Nikolic, V.D.; Savic-Gajic, I.M.; Nikolic, L.B.; Ibric, S.R.; Gajic, D.G. Optimization of technological procedure for amygdalin isolation from plum seeds (Pruni domesticae semen). Front. Plant. Sci. 2015, 6, 1–11. [Google Scholar] [CrossRef]
- Cortés, V.; Talens, P.; Barat, J.M.; Lerma-García, M.J. Potential of NIR spectroscopy to predict amygdalin content established by HPLC in intact almonds and classification based on almond bitterness. Food Control. 2018, 91, 68–75. [Google Scholar] [CrossRef]
- Del Cueto, J.; Møller, B.L.; Dicenta, F.; Sánchez-pérez, R. Plant Physiology and Biochemistry β -Glucosidase activity in almond seeds. Plant. Physiol. Biochem. 2018, 126, 163–172. [Google Scholar] [CrossRef]
- Wahab, M.F.; Breitbach, Z.S.; Armstrong, D.W.; Strattan, R.; Berthod, A. Problems and Pitfalls in the Analysis of Amygdalin and Its Epimer. J. Agric. Food Chem. 2015, 63, 8966–8973. [Google Scholar] [CrossRef] [PubMed]
- Burns, A.E.; Bradbury, J.H.; Cavagnaro, T.R.; Gleadow, R.M. Total cyanide content of cassava food products in Australia. J. Food Compos. Anal. 2012, 25, 79–82. [Google Scholar] [CrossRef]
- Lee, J.; Zhang, G.; Wood, E.; Rogel Castillo, C.; Mitchell, A.E.; Wu, H.; Cao, C.; Zhou, C. Quantification of amygdalin in nonbitter, semibitter, and bitter almonds (prunus dulcis) by UHPLC-(ESI)QqQ MS/MS. J. Agric. Food Chem. 2013, 61, 7754–7759. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Xu, X.; Yuan, S.; Liu, H.; Liu, M.; Zhang, Y.; Zhang, H.; Gao, Y.; Lin, R.; Li, X. Identification and analysis of amygdalin, neoamygdalin and amygdalin amide in different processed bitter almonds by HPLC-ESI-MS/MS and HPLC-DAD. Molecules 2017, 22, 1425. [Google Scholar] [CrossRef]
- Bolarinwa, I.F.; Orfila, C.; Morgan, M.R.A. Determination of amygdalin in apple seeds, fresh apples and processed apple juices. Food Chem. 2015, 170, 437–442. [Google Scholar] [CrossRef]
- Bolarinwa, I.F.; Orfila, C.; Morgan, M.R.A. Amygdalin content of seeds, kernels and food products commercially- available in the UK. Food Chem. 2014, 152, 133–139. [Google Scholar] [CrossRef]
- Karsavuran, N.; Charehsaz, M.; Celik, H.; Asma, B.M.; Yakıncı, C.; Aydın, A. Amygdalin in bitter and sweet seeds of apricots. Toxicol. Environ. Chem. 2014, 96, 1564–1570. [Google Scholar] [CrossRef]
- Luo, K.K.; Kim, D.A.; Mitchell-Silbaugh, K.C.; Huang, G.; Mitchell, A.E. Comparison of amygdalin and benzaldehyde levels in California almond (Prunus dulcis) varietals. Acta Hortic. 2018, 1219, 1–7. [Google Scholar] [CrossRef]
- Rogel-Castillo, C.; Luo, K.; Huang, G.; Mitchell, A.E. Effect of Drying Moisture Exposed Almonds on the Development of the Quality Defect Concealed Damage. J. Agric. Food Chem. 2017, 65, 8948–8956. [Google Scholar] [CrossRef]
- Thygesen, L.G.; Løkke, M.M.; Micklander, E.; Engelsen, S.B. Vibrational microspectroscopy of food. Raman vs. FT-IR. Trends Food Sci. Technol. 2003, 14, 50–57. [Google Scholar] [CrossRef]
- Muhammad, S.S.; Abbas, S.M.; Khammas, Z.A.-A. Extraction and Determination of Amygdaline in Iraqi Plant Seeds Using the Combined Simple Extraction Procedure and High-Performance Liquid Chromatography. Baghdad Sci. J. 2013, 10, 350–361. [Google Scholar] [CrossRef]
- Amaya-Salcedo, J.C.; Cárdenas-González, O.E.; Gómez-Castaño, J.A. Solid-to-liquid extraction and HPLC/UV determination of amygdalin of seeds of apple (Malus pumila Mill): Comparison between traditional-solvent and microwave methodologies. Acta Agron. 2018, 67, 381–388. [Google Scholar] [CrossRef]
- Micklander, E.; Brimer, L.; Engelsen, S.B. Noninvasive Assay for Cyanogenic Constituents in Plants by Raman Spectroscopy: Content and Distribution of Amygdalin in Bitter Almond (Prunus amygdalus). Appl. Spectrosc. 2002, 56, 1139–1146. [Google Scholar] [CrossRef]
- Krafft, C.; Cervellati, C.; Paetz, C.; Schneider, B.; Popp, J. Distribution of amygdalin in apricot (Prunus armeniaca) seeds studied by Raman microscopic imaging. Appl. Spectrosc. 2012, 66, 644–649. [Google Scholar] [CrossRef]
- Isozaki, T.; Matano, Y.; Yamamoto, K.; Kosaka, N.; Tani, T. Quantitative determination of amygdalin epimers by cyclodextrin-modified micellar electrokinetic chromatography. J. Chromatogr. A 2001, 923, 249–254. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, Q.-A.; Yao, J.-L.; Zhang, X.-Y. Changes of amygdalin and volatile components of apricot kernels during the ultrasonically-accelerated debitterizing. Ultrason. Sonochem. 2019, 58, 104614. [Google Scholar] [CrossRef]
- Toomey, V.M.; Nickum, E.A.; Flurer, C.L. Cyanide and Amygdalin as Indicators of the Presence of Bitter Almonds in Imported Raw Almonds. J. Forensic Sci. 2012, 57, 1313–1317. [Google Scholar] [CrossRef]
- Wu, H.; Cao, C.; Zhou, C. Determination of amygdalin in the fruit of Eriobotrya japonica Lindl by high performance liquid chromatography. Biomed. Res. 2017, 28, 8827–8831. [Google Scholar]
- Tivana, L.D.; Da Cruz Francisco, J.; Zelder, F.; Bergenståhl, B.; Dejmek, P. Straightforward rapid spectrophotometric quantification of total cyanogenic glycosides in fresh and processed cassava products. Food Chem. 2014, 158, 20–27. [Google Scholar] [CrossRef]
- Savic, I.M.; Nikolic, V.D.; Savic, I.M.; Nikolic, L.B.; Stankovic, M.Z. Development and validation of HPLC method for the determination of amygdalin in the plant extract of plum kernel. Res. J. Chem. Environ. 2012, 16, 80–86. [Google Scholar]
- Senica, M.; Stampar, F.; Veberic, R.; Mikulic-Petkovsek, M. Fruit Seeds of the Rosaceae Family: A Waste, New Life, or a Danger to Human Health? J. Agric. Food Chem. 2017, 65, 10621–10629. [Google Scholar] [CrossRef]
- Senica, M.; Stampar, F.; Veberic, R.; Mikulic-Petkovsek, M. Transition of phenolics and cyanogenic glycosides from apricot and cherry fruit kernels into liqueur. Food Chem. 2016, 203, 483–490. [Google Scholar] [CrossRef]
- García, M.C.; González-García, E.; Vásquez-Villanueva, R.; Marina, M.L. Apricot and other seed stones: Amygdalin content and the potential to obtain antioxidant, angiotensin i converting enzyme inhibitor and hypocholesterolemic peptides. Food Funct. 2016, 7, 4693–4701. [Google Scholar] [CrossRef]
- Koo, J.Y.; Hwang, E.Y.; Cho, S.; Lee, J.H.; Lee, Y.M.; Hong, S.P. Quantitative determination of amygdalin epimers from armeniacae semen by liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 814, 69–73. [Google Scholar] [CrossRef]
- Hosseini, M.; Heydari, R.; Alimoradi, M. Reversed-phase vortex-assisted liquid-liquid microextraction: A new sample preparation method for the determination of amygdalin in oil and kernel samples. J. Sep. Sci. 2015, 38, 663–669. [Google Scholar] [CrossRef]
- Mirzaei, H.; Rezaei, K. Amygdalin Contents of Oil and Meal from Wild Almond: Effect of Different Heat Pretreatment and Extraction Methods. JAOCS J. Am. Oil Chem. Soc. 2019, 96, 1163–1171. [Google Scholar] [CrossRef]
- Pavlović, N.; Vidović, S.; Vladić, J.; Popović, L.; Moslavac, T.; Jakobović, S.; Jokić, S. Recovery of Tocopherols, Amygdalin, and Fatty Acids From Apricot Kernel Oil: Cold Pressing Versus Supercritical Carbon Dioxide. Eur. J. Lipid Sci. Technol. 2018, 120. [Google Scholar] [CrossRef]
- Yildirim, A.N.; Akinci-Yildirim, F.; Polat, M.; Şan, B.; Sesli, Y. Amygdalin content in kernels of several almond cultivars grown in Turkey. HortScience 2014, 49, 1268–1270. [Google Scholar] [CrossRef]
- Lv, W.-F.; Ding, M.-Y.; Zheng, R. Isolation and Quantitation of Amygdalin in Apricot-kernel and Prunus Tomentosa Thunb. by HPLC with Solid -Phase Extraction. J. Chromatogr. Sci. 2005, 43, 383–387. [Google Scholar] [CrossRef]
- Folashade, B.I.; Olaniyan, S.A.; Temilade, A.F.; Ifasegun, O.; Salawu, S. Effect of Processing on Amygdalin and Cyanide Contents of Some Nigerian Foods. In Proceedings of the Fourth International Conference on Engineering and Technology Research, Ogbomoso, Nigeria, 23–25 February 2016. [Google Scholar]
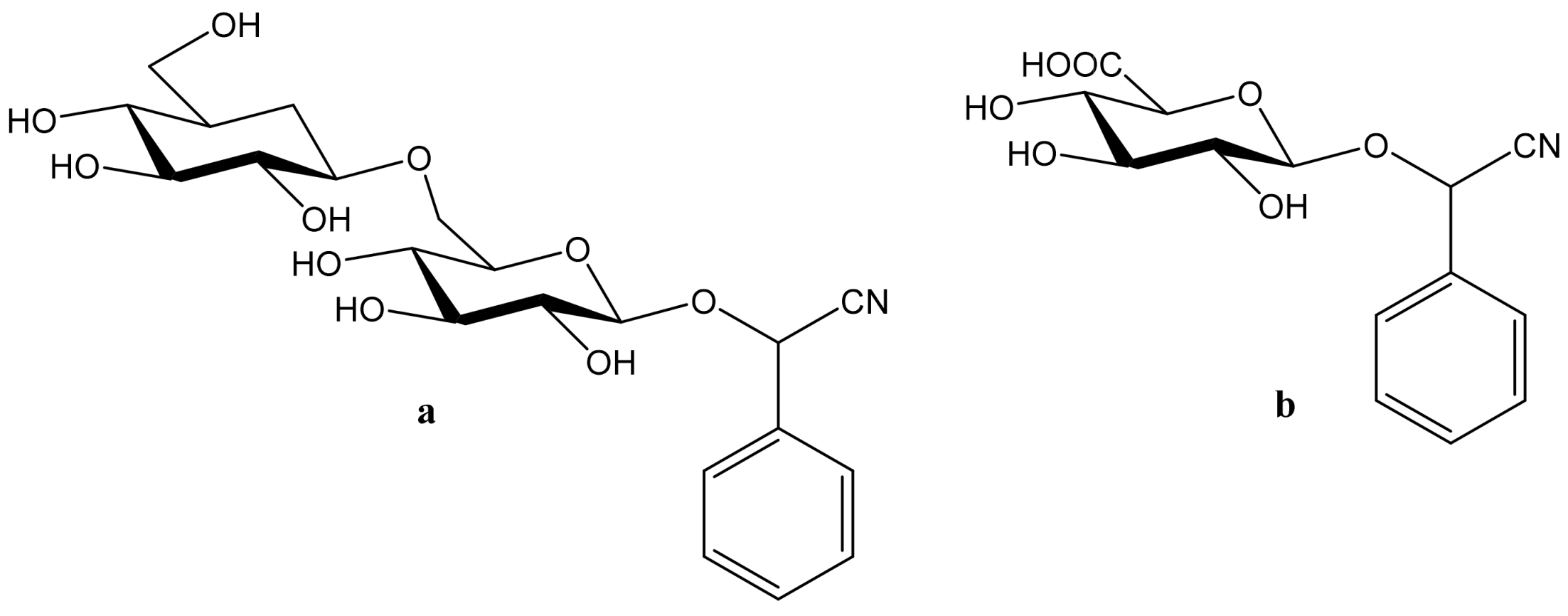
| Cyanogenic Glycoside | Occurrence | Ref | |
|---|---|---|---|
| Amygdalin | 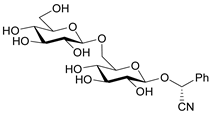 | Amygdalus communis, Cydonia oblonga, Padus | [3] |
| Prunasin | 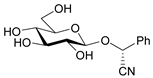 | Prunus | [4] |
| Vicianin | 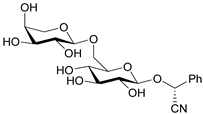 | Papilionaceae | [5] |
| Linmarin | 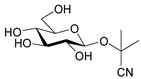 | Linum, Phaseolus | |
| Sambunigryn |  | Sambucus | |
| Plant | Cyanogenic Potential [mg HCN/kg Plant Material] | |
|---|---|---|
| Peach | Kernel | 710 |
| Plum | Kernel | 696 |
| Nectarine | Kernel | 196 |
| Apricot | Kernel | 785 |
| Apple | Seed | 690 |
| Patient | Dose | Effects | Ref |
|---|---|---|---|
| Child (2 years) | 500 mg | vomiting, apathy, diarrhea, accelerated breathing | [30] |
| Child (4 year) | 500 mg | diarrhea, accelerated breath, blood cyanide concentration 163 µg/L | [31] |
| Adult woman (80 years) | 300 mL | dyspnea, vertigo and vomiting. | [32] |
| Adult woman | 9 g | vomiting, dizziness, blood cyanide concentration 143 µg/L | [33] |
| Cell Lines Used for Testing | Amygdalin Concentration [mg mL−1] | Results Observed | Ref. | |
|---|---|---|---|---|
| Bladder cancer | RT 112 UMUC-3 TCCSUP | 1.25–10 | limited proliferative capacity and apoptosis. decrease in cdk4 expression level in RT112 and TCCSUP lines. | [40] |
| Cervical cancer | HeLa | 1.25–20 | initiation of the cell apoptosis, reduction of Bcl-2 expression level, increase of Bax expression level. | [39] |
| Colon cancer | SNU-C4 | 0.25–5 | reduction of the expression level of many genes associated with following cell functions: growth, apoptosis, transmission. | [53] |
| Breast cancer | MDA-MB-231, MCF-7 | 2.5–80 | reduction of proliferative activity of the cells | [54] |
| MDA-MB-231 | 10 | growth rate of cancer cells was inhibited | [55] | |
| Kidney cancer | Caki-1 A498 KTC-26 xds | 10 | -reduced ability collagen and fibronectin. -reduced cell mobility. | [47] |
| Analytical Technique | Sample | Recovery [%] | Intra/Inter-Day Variation [%] | LOD | LOQ | Detected Compounds in Real Samples | Ref |
|---|---|---|---|---|---|---|---|
| LC-DAD | apricot seeds | 91 ± 10 | 0.8/3.8 | 1.2 mg·L−1 | 4.0 mg·L−1 | bitter seeds 26 ± 14 mg·g−1 sweet seeds 0.16 ± 0.09 mg·g−1 | [82] |
| apricot liqueur | - | - | - | - | 38.79 µg·mL−1 | [83] | |
| cherry liqueur | 16.08 µg·mL−1 | ||||||
| HPLC–MS/MS | almonds | - | - | 200 µg·g−1 | - | <LOD | [22] |
| HPLC-UV | plum seeds | - | - | - | - | 25.30 g 100 g−1 | [59] |
| almonds | - | 0.13/0.75 | 2 µg·mL−1 | - | 4.51 ± 0.04% | ||
| loquat fruit kernel | - | - | - | - | 7.58 ± 0.76 mg·g−1 | ||
| almonds | 98.4–102.9 | 0.25/0.31 | 0.02 mg·L−1 | 0.07 mg·L-1 | sweet: <350 mg·kg−1 bitter: 14,700–50,400 mg·kg−1 | [60] | |
| peach seeds | 99.05 | 0.19 | 0.03 mg 100 g−1 | 0.09 mg 100 g−1 | 6.3 ± 0.2 g 100 g−1 | [84] | |
| plum seeds | 0.439 ± 0.001 g 100 g−1 | ||||||
| apricot seeds | 7.9 ± 0.2 g 100 g−1 | ||||||
| peach seeds | - | - | - | - | seed: 12.14 ± 4.80 mg 100g−1 | [19] | |
| citrullus colocynth kernels | 97.34 ± 0.58 | - | 0.88 mg·L−1 | 2.93 mg·L−1 | 0.27 ± 0.03 100 g−1 | [72] | |
| apples | - | 0.095 | 0.0505 mg·g−1 | 0.0548 mg·g−1 | 0.28–1.40 mg·g−1 | [73] | |
| Armeniacae semen | 98.0–102.6 | - | - | - | 45.42 ± 1.21 mg·g−1 | [85] | |
| bitter almond oil | 96.0–102.0 | 4.8/7.2 | 0.07 µg·mL−1 | - | 0.092 ± 0.003 mg·g−1 | [86] | |
| wild almond oil | - | - | - | - | 12.8–12.9 mg/100 mL oil | [87] | |
| sweet apricot kernels | - | - | - | - | 5.0 ± 0.23 mg·g−1 | [88] | |
| HPLC-DAD | apricot seeds | - | - | - | - | 0.861 g·100 g−1 | [56] |
| almonds | - | - | - | - | 0.37–1.46 g·kg−1 | [89] | |
| apple seeds | 98 | - | 0.1 µg·mL−1 | - | 1–3.9 mg·g−1 | [66] | |
| apricots | - | - | 0.1 µg·mL−1 | 0.3 µg·mL- | 14.37 ± 0.28 mg·g−1 | [67] | |
| cherries | 2.68 ± 0.02 mg·g−1 | ||||||
| peaches | 6.81 ± 0.02 mg·g−1 | ||||||
| pears | 1.29 ± 0.04 mg·g−1 | ||||||
| cucumbers | 0.07 ± 0.02 mg·g−1 | ||||||
| courgettes | 0.21 ± 0.13 mg·g−1 | ||||||
| melons | 0.12 ± 0.07 mg·g−1 | ||||||
| apricot kernels | - | - | 0.2 µg·mL−1 | - | - | [69] | |
| apricot kernels | 99.08 | 2.4/3.5 | - | - | 0.217–0.284 mg·mL−1 | [58] | |
| plum seeds | - | - | 1.06 μg·mL−1 | 3.49 μg·mL−1 | 25.30 g 100 g−1 | [65] | |
| bayberry kernels | 77.9 | - | - | - | 129.13–358.68 mg·L−1 | [57] | |
| food suplements | 94.81 | 0.57/1.52 | 0.13 mg·L−1 | 0.40 mg·L−1 | 20.68 ± 1.58 mg·g−1 | [86] | |
| apricot kernels | 91 | 0.8/3.8 | 1.2 mg·L−1 | 4.0 mg·L−1 | 26 ± 14 mg·g−1 | [90] | |
| UHPLC-(ESI)QqQ MS/MS | nonbitter almonds | - | - | 0.1 ng·mL−1 | 0.33 ng·mL−1 | 63.13 ± 57.54 mg·kg−1 | [64] |
| semibitter almonds | 992.24 ± 513.04 mg·kg−1 | ||||||
| bitter almonds | 40,060.34 ± 7855.26 mg·kg−1 | ||||||
| alomnds | - | - | - | - | 1.62–76.50 mg·kg−1 | [67] | |
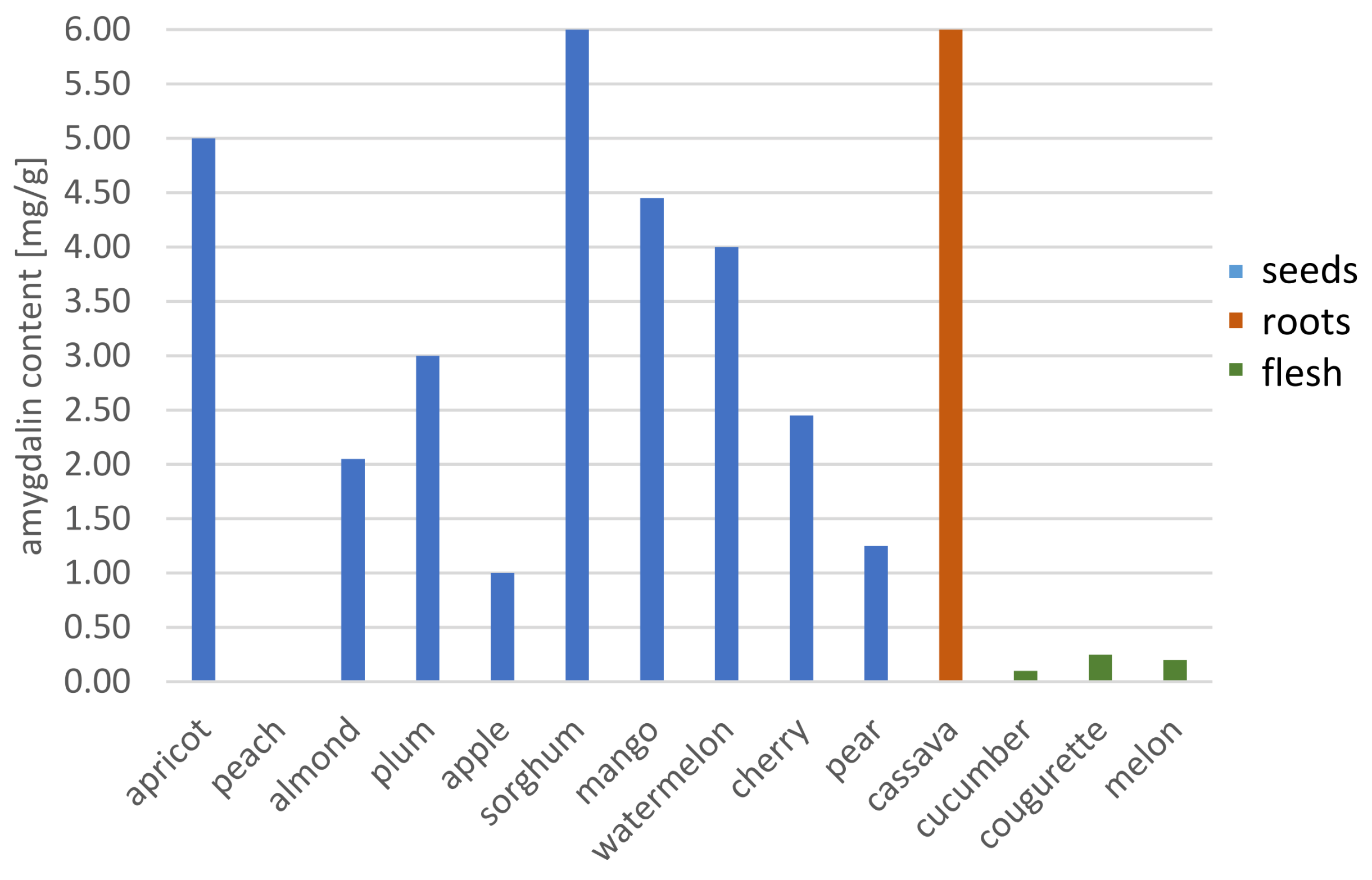
| Spectrophotometric method | cassava root | - | - | - | - | 3.40 mg·L−1 | [80] |
| cassava roots | - | - | - | - | 8.84–48.33 mg·g−1 | [91] | |
| sorghum seeds | 122.31 mg·g−1 | ||||||
| mango seeds | 4.41 mg·g−1 | ||||||
| watermelon seeds | 3.97 mg·g−1 | ||||||
| almond seeds | 3.91 mg·g−1 | ||||||
| ELISA | black cherries | 99 ± 1.2 | - | 200 ± 0.05 pg·mL−1 | - | 2.14 ± 0.15 mg·g−1 | [92] |
| yellow plums | 2.30 ± 0.90 mg·g−1 | ||||||
| peaches | 5.79 ± 0.83 mg·g−1 | ||||||
| black plums | 9.75 ± 1.32 mg·g−1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaszczak-Wilke, E.; Polkowska, Ż.; Koprowski, M.; Owsianik, K.; Mitchell, A.E.; Bałczewski, P. Amygdalin: Toxicity, Anticancer Activity and Analytical Procedures for Its Determination in Plant Seeds. Molecules 2021, 26, 2253. https://doi.org/10.3390/molecules26082253
Jaszczak-Wilke E, Polkowska Ż, Koprowski M, Owsianik K, Mitchell AE, Bałczewski P. Amygdalin: Toxicity, Anticancer Activity and Analytical Procedures for Its Determination in Plant Seeds. Molecules. 2021; 26(8):2253. https://doi.org/10.3390/molecules26082253
Chicago/Turabian StyleJaszczak-Wilke, Ewa, Żaneta Polkowska, Marek Koprowski, Krzysztof Owsianik, Alyson E. Mitchell, and Piotr Bałczewski. 2021. "Amygdalin: Toxicity, Anticancer Activity and Analytical Procedures for Its Determination in Plant Seeds" Molecules 26, no. 8: 2253. https://doi.org/10.3390/molecules26082253
APA StyleJaszczak-Wilke, E., Polkowska, Ż., Koprowski, M., Owsianik, K., Mitchell, A. E., & Bałczewski, P. (2021). Amygdalin: Toxicity, Anticancer Activity and Analytical Procedures for Its Determination in Plant Seeds. Molecules, 26(8), 2253. https://doi.org/10.3390/molecules26082253








