Algae-Derived Bioactive Molecules for the Potential Treatment of SARS-CoV-2
Abstract
1. Introduction—Pandemic Problem and Opportunities
2. The Antiviral Activities of Algae-Derived Molecules
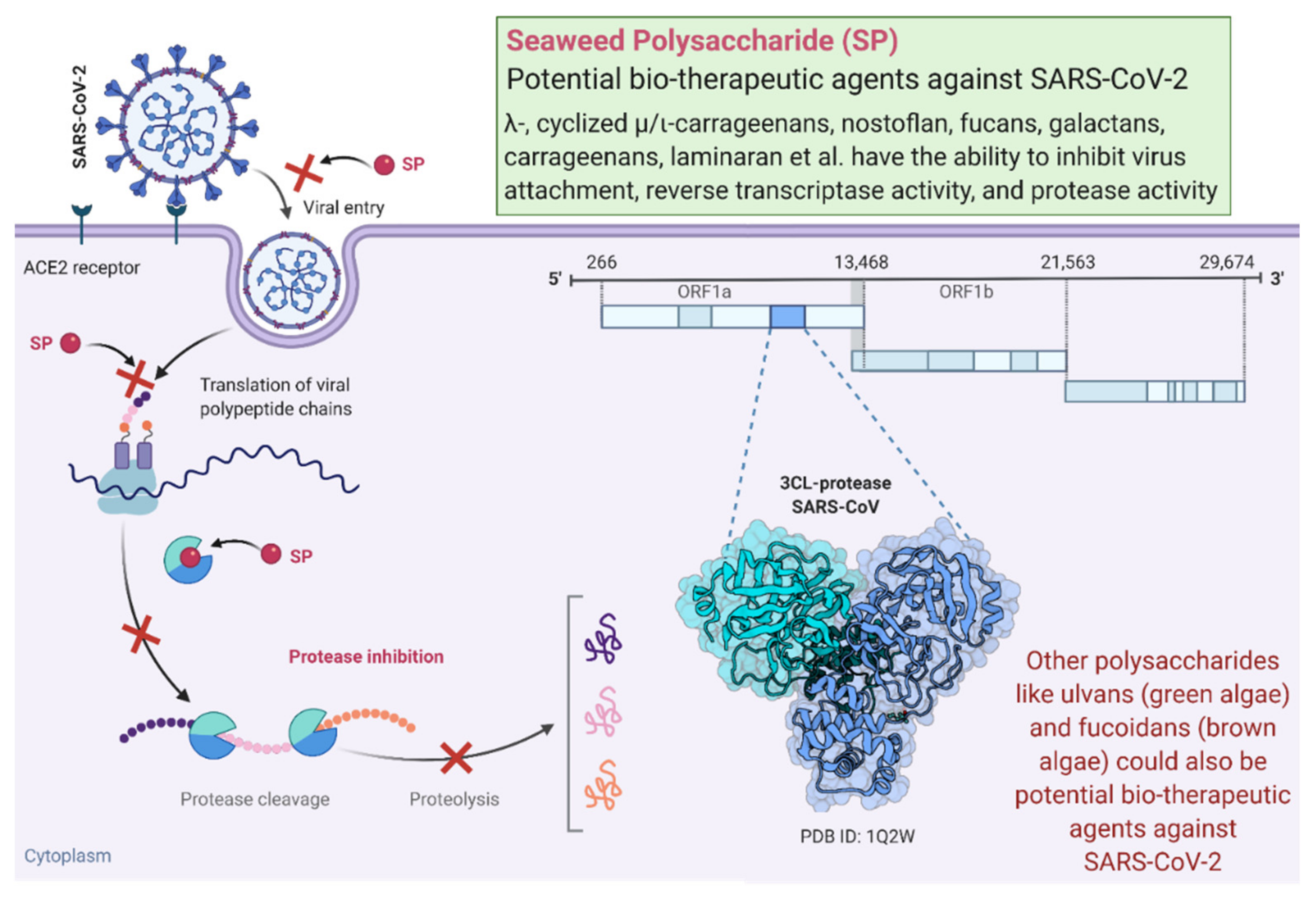
3. Algae-Derived Antiviral Polysaccharides and Mechanisms of Action
4. Potential of Algae-Derived Antiviral Metabolites against SARS-CoV-2
5. Algae-Derived Metabolites for Microbiota-Based Therapy and Immunomodulatory Activity against SARS-CoV-2
6. Algae-Derived Glycan Therapeutics against SARS-CoV-2
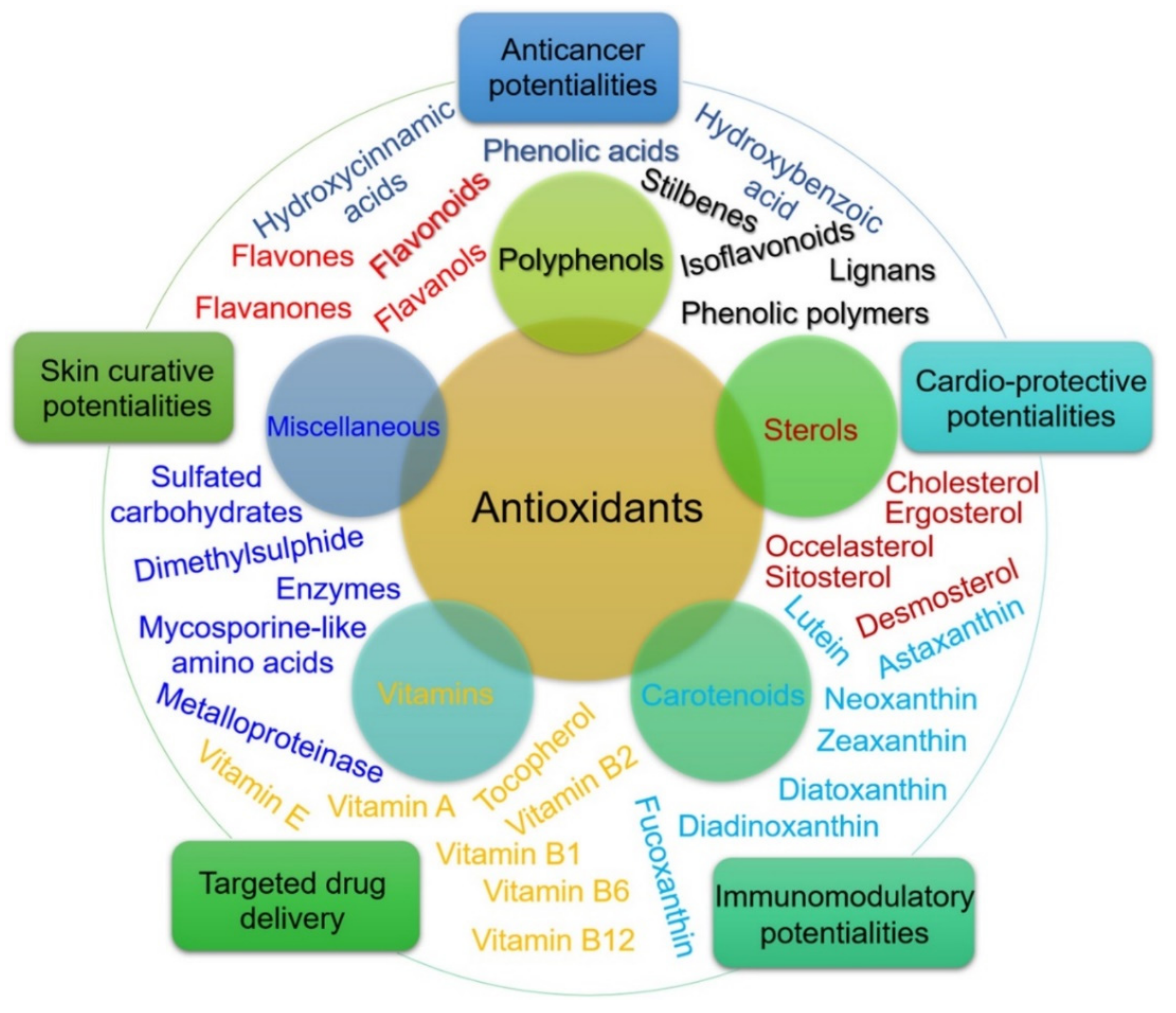
7. Algae-Derived Antioxidants against SARS-CoV-2
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- El Gamal, A.A. Biological importance of marine algae. Saudi Pharm. J. 2010, 18, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.A.; Xu, J.L.; Wang, Z. Microalgae Biotechnology for Food, Health and High Value Products; Springer: Singapore, 2020. [Google Scholar] [CrossRef]
- Zanella, L.; Alam, M.A. Extracts and Bioactives from Microalgae (Sensu Stricto): Opportunities and Challenges for a New Generation of Cosmetics. In Microalgae Biotechnology for Food, Health and High Value Products; Springer: Singapore, 2020; pp. 295–349. [Google Scholar]
- Galasso, C.; Gentile, A.; Orefice, I.; Ianora, A.; Bruno, A.; Noonan, D.M.; Sansone, C.; Albini, A.; Brunet, C. Microalgal Derivatives as Potential Nutraceutical and Food Supplements for Human Health: A Focus on Cancer Prevention and Interception. Nutrients 2019, 11, 1226. [Google Scholar] [CrossRef]
- Pereira, L.; Critchley, A.T. The COVID 19 novel coronavirus pandemic 2020: Seaweeds to the rescue? Why does substantial, supporting research about the antiviral properties of seaweed polysaccharides seem to go unrecognized by the pharmaceutical community in these desperate times? J. Appl. Phycol. 2020, 32, 1875–1877. [Google Scholar] [CrossRef] [PubMed]
- Torres-Tiji, Y.; Fields, F.J.; Mayfield, S.P. Microalgae as a future food source. Biotechnol. Adv. 2020, 41, 107536. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Alam, M.A.; Liu, S.; Xu, J.L.; Parra Saldivar, R. Critical processes and variables in microalgae biomass production coupled with bioremediation of nutrients and CO2 from livestock farms: A review. Sci. Total Environ. 2020, 716, 135247. [Google Scholar] [CrossRef] [PubMed]
- Nova, P.; Pimenta-Martins, A.; Laranjeira Silva, J.; Silva, A.M.; Gomes, A.M.; Freitas, A.C. Health benefits and bioavailability of marine resources components that contribute to health–what’s new? Crit. Rev. Food Sci. Nutr. 2020, 60, 3680–3692. [Google Scholar] [CrossRef] [PubMed]
- Bhowmick, S.; Mazumdar, A.; Moulick, A.; Adam, V. Algal metabolites: An inevitable substitute for antibiotics. Biotechnol. Adv. 2020, 43, 107571. [Google Scholar] [CrossRef] [PubMed]
- Rhimou, B.H.R. Nathalie B, Antiviral activity of the extracts of Rhodophyceae from Morocco. Afr. J. Biotechnol. 2010, 9, 7968–7975. [Google Scholar]
- de Jesus Raposo, M.F.; de Morais, R.M.S.C.; de Morais, A.M.M.B. Health applications of bioactive compounds from marine microalgae. Life Sci. 2013, 93, 479–486. [Google Scholar] [CrossRef]
- Dewi, I.C.; Falaise, C.; Hellio, C.; Bourgougnon, N.; Mouget, J.-L. Anticancer, antiviral, antibacterial, and antifungal properties in microalgae. In Microalgae in Health and Disease Prevention; Elsevier: Cambridge, MA, USA, 2018; pp. 235–261. [Google Scholar]
- Mayer, A.; Guerrero, A.J.; Rodríguez, A.D.; Taglialatela-Scafati, O.; Nakamura, F.; Fusetani, N. Marine pharmacology in 2014–2015: Marine compounds with antibacterial, antidiabetic, antifungal, antiinflammatory, antiprotozoal, antituberculosis, antiviral, and anthelmintic activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2020, 18, 5. [Google Scholar]
- Soares, A.R.; Robaina, M.C.S.; Mendes, G.S.; Silva, T.S.L.; Gestinari, L.M.S.; Pamplona, O.S.; Yoneshigue-Valentin, Y.; Kaiser, C.R.; Romanos, M.T.V. Antiviral activity of extracts from Brazilian seaweeds against herpes simplex virus. Rev. Bras. Farmacogn. 2012, 22, 714–723. [Google Scholar] [CrossRef]
- Pérez-Riverol, A.; Piñón Ramos, A.; Morier Díaz, L.F.; López, Y.T.; Mendoza Llanes, D.; del Barrio Alonso, G. Antiviral activity of an aqueous extract from the red alga Laurencia obtusa against influenza A and B viruses. Rev. Cuba. Med. Trop. 2014, 66, 273–285. [Google Scholar]
- Zaid, S.A.A.-L.; Hamed, N.N.E.-D.; Abdel-Wahab, K.S.E.-D.; Abo El-Magd, E.K.; Salah El-Din, R.A.-L. Antiviral activities and phytochemical constituents of Egyptian marine seaweeds (Cystoseiramyrica (SG Gmelin) C. Agardh and Ulva lactuca Linnaeus) aqueous extract. Egypt. J. Hosp. Med. 2016, 64, 422–429. [Google Scholar] [CrossRef]
- El-Sheekh, M.M.; Shabaan, M.T.; Hassan, L.; Morsi, H.H. Antiviral activity of algae biosynthesized silver and gold nanoparticles against Herps Simplex (HSV-1) virus in vitro using cell-line culture technique. Int. J. Environ. Health Res. 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Luescher-Mattli, M. Algae, a possible source for new drugs in the treatment of HIV and other viral diseases. Curr. Med. Chem. Anti-Infect. Agents 2003, 2, 219–225. [Google Scholar] [CrossRef]
- Moghadamtousi, S.Z.; Nikzad, S.; Kadir, H.A.; Abubakar, S.; Zandi, K. Potential antiviral agents from marine fungi: An overview. Mar. Drugs 2015, 13, 4520–4538. [Google Scholar] [CrossRef]
- Pinto, A.M.V.; Leite, J.P.G.; Ferreira, W.J.; Cavalcanti, D.N.; Villaça, R.C.; Giongo, V.; Teixeira, V.L.; Paixão, I.C.N.d.P. Marine natural seaweed products as potential antiviral drugs against bovine viral diarrhea virus. Rev. Bras. Farmacogn. 2012, 22, 813–817. [Google Scholar] [CrossRef]
- Raveh, A.; Delekta, P.C.; Dobry, C.J.; Peng, W.; Schultz, P.J.; Blakely, P.K.; Tai, A.W.; Matainaho, T.; Irani, D.N.; Sherman, D.H. Discovery of potent broad spectrum antivirals derived from marine actinobacteria. PLoS ONE 2013, 8, e82318. [Google Scholar] [CrossRef]
- Sagar, S.; Kaur, M.; Minneman, K.P. Antiviral lead compounds from marine sponges. Mar. Drugs 2010, 8, 2619–2638. [Google Scholar] [CrossRef]
- Bajpai, A.; Davuluri, S.; Haridas, H.; Kasliwal, G.; Deepti, H.; Sreelakshmi, K.; Chandrashekar, D.; Bora, P.; Farouk, M.; Chitturi, N. In search of the right literature search engine (s). Nat. Preced. 2011, 1. [Google Scholar] [CrossRef]
- Grewal, A.; Kataria, H.; Dhawan, I. Literature search for research planning and identification of research problem. Indian J. Anaesth. 2016, 60, 635. [Google Scholar] [CrossRef]
- Capillo, G.; Savoca, S.; Costa, R.; Sanfilippo, M.; Rizzo, C.; Lo Giudice, A.; Albergamo, A.; Rando, R.; Bartolomeo, G.; Spanò, N. New insights into the culture method and antibacterial potential of Gracilaria gracilis. Mar. Drugs 2018, 16, 492. [Google Scholar] [CrossRef]
- Hoseinifar, S.H.; Yousefi, S.; Capillo, G.; Paknejad, H.; Khalili, M.; Tabarraei, A.; Van Doan, H.; Spanò, N.; Faggio, C. Mucosal immune parameters, immune and antioxidant defence related genes expression and growth performance of zebrafish (Danio rerio) fed on Gracilaria gracilis powder. Fish Shellfish Immunol. 2018, 83, 232–237. [Google Scholar] [CrossRef]
- Elaya Perumal, U.; Sundararaj, R. Algae: A potential source to prevent and cure the novel coronavirus–A review. Int. J. Emerg. Technol. 2020, 11, 479–483. [Google Scholar]
- Buck, C.B.; Thompson, C.D.; Roberts, J.N.; Müller, M.; Lowy, D.R.; Schiller, J.T. Carrageenan is a potent inhibitor of papillomavirus infection. PLoS Pathog. 2006, 2, e69. [Google Scholar] [CrossRef]
- Talarico, L.; Pujol, C.; Zibetti, R.; Faria, P.; Noseda, M.; Duarte, M.; Damonte, E. The antiviral activity of sulfated polysaccharides against dengue virus is dependent on virus serotype and host cell. Antivir. Res. 2005, 66, 103–110. [Google Scholar] [CrossRef]
- Carthew, P. Safety of carrageenan in foods. Environ. Health Perspect. 2002, 110, A176–A177. [Google Scholar] [CrossRef] [PubMed]
- Sanniyasi, E.; Venkatasubramanian, G.; Anbalagan, M.M.; Raj, P.P.; Gopal, R.K. In vitro anti-HIV-1 activity of the bioactive compound extracted and purified from two different marine macroalgae (seaweeds)(Dictyota bartayesiana JV Lamouroux and Turbinaria decurrens Bory). Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, A.; Zorofchian Moghadamtousi, S.; Abubakar, S.; Zandi, K. Antiviral potential of algae polysaccharides isolated from marine sources: A review. Biomed. Res. Int. 2015, 2015, 825203. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, S.-X.; Guan, H.-S. The antiviral activities and mechanisms of marine polysaccharides: An overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar] [CrossRef]
- Park, J.-Y.; Kim, J.H.; Kwon, J.M.; Kwon, H.-J.; Jeong, H.J.; Kim, Y.M.; Kim, D.; Lee, W.S.; Ryu, Y.B. Dieckol, a SARS-CoV 3CLpro inhibitor, isolated from the edible brown algae Ecklonia cava. Bioorganic Med. Chem. 2013, 21, 3730. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Han, W.; Wang, G.; Zhao, X. Application prospect of polysaccharides in the development of anti-novel coronavirus drugs and vaccines. Int. J. Biol. Macromol. 2020, 164, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Koehn, F.E.; Sarath, G.P.; Neil, D.N.; Cross, S.S. Halitunal, an unusual diterpene aldehyde from the marine alga Halimeda tuna. Tetrahedron Lett. 1991, 32, 169–172. [Google Scholar] [CrossRef]
- O’Keefe, B.R.; Giomarelli, B.; Barnard, D.L.; Shenoy, S.R.; Chan, P.K.; McMahon, J.B.; Palmer, K.E.; Barnett, B.W.; Meyerholz, D.K.; Wohlford-Lenane, C.L. Broad-spectrum in vitro activity and in vivo efficacy of the antiviral protein griffithsin against emerging viruses of the family Coronaviridae. J. Virol. 2010, 84, 2511–2521. [Google Scholar] [CrossRef]
- Millet, J.K.; Séron, K.; Labitt, R.N.; Danneels, A.; Palmer, K.E.; Whittaker, G.R.; Dubuisson, J.; Belouzard, S. Middle East respiratory syndrome coronavirus infection is inhibited by griffithsin. Antivir. Res. 2016, 133, 1–8. [Google Scholar] [CrossRef]
- Zumla, A.; Chan, J.F.; Azhar, E.I.; Hui, D.S.; Yuen, K.-Y. Coronaviruses—drug discovery and therapeutic options. Nat. Rev. Drug Discov. 2016, 15, 327–347. [Google Scholar] [CrossRef]
- Lee, C. Griffithsin, a highly potent broad-spectrum antiviral lectin from red algae: From discovery to clinical application. Mar. Drugs 2019, 17, 567. [Google Scholar] [CrossRef]
- Petit, L.; Vernes, L.; Cadoret, J.-P. Docking and in silico toxicity assessment of Arthrospira compounds as potential antiviral agents against SARS-CoV-2. Res. Sq. 2020, 1–24. [Google Scholar] [CrossRef]
- Nikunj, P.N.P. Lensi P, Richa P, haresh P, Red algae derived phycocompounds as an inhibitors of SARS-CoV-2. J. Interdiscip. Cycle Res. 2020, 12, 662–681. [Google Scholar]
- Joseph, J.; Thankamani, K.; Ajay, A.; Das, V.R.A.; Raj, V.S. Green tea and Spirulina extracts inhibit SARS, MERS, and SARS-2 spike pseudotyped virus entry in vitro. bioRxiv 2020. [Google Scholar] [CrossRef]
- Nagle, V.; Gaikwad, M.; Pawar, Y.; Dasgupta, S. Marine red alga Porphyridium sp. as a source of sulfated polysaccharides (SPs) for combating against COVID-19. Preprints 2020, 2020040168. [Google Scholar]
- Talukdar, J.; Dasgupta, S.; Nagle, V.; Bhadra, B. COVID-19: Potential of microalgae derived natural astaxanthin as adjunctive supplement in alleviating cytokine storm. SSRN 2020, 3579738. [Google Scholar] [CrossRef]
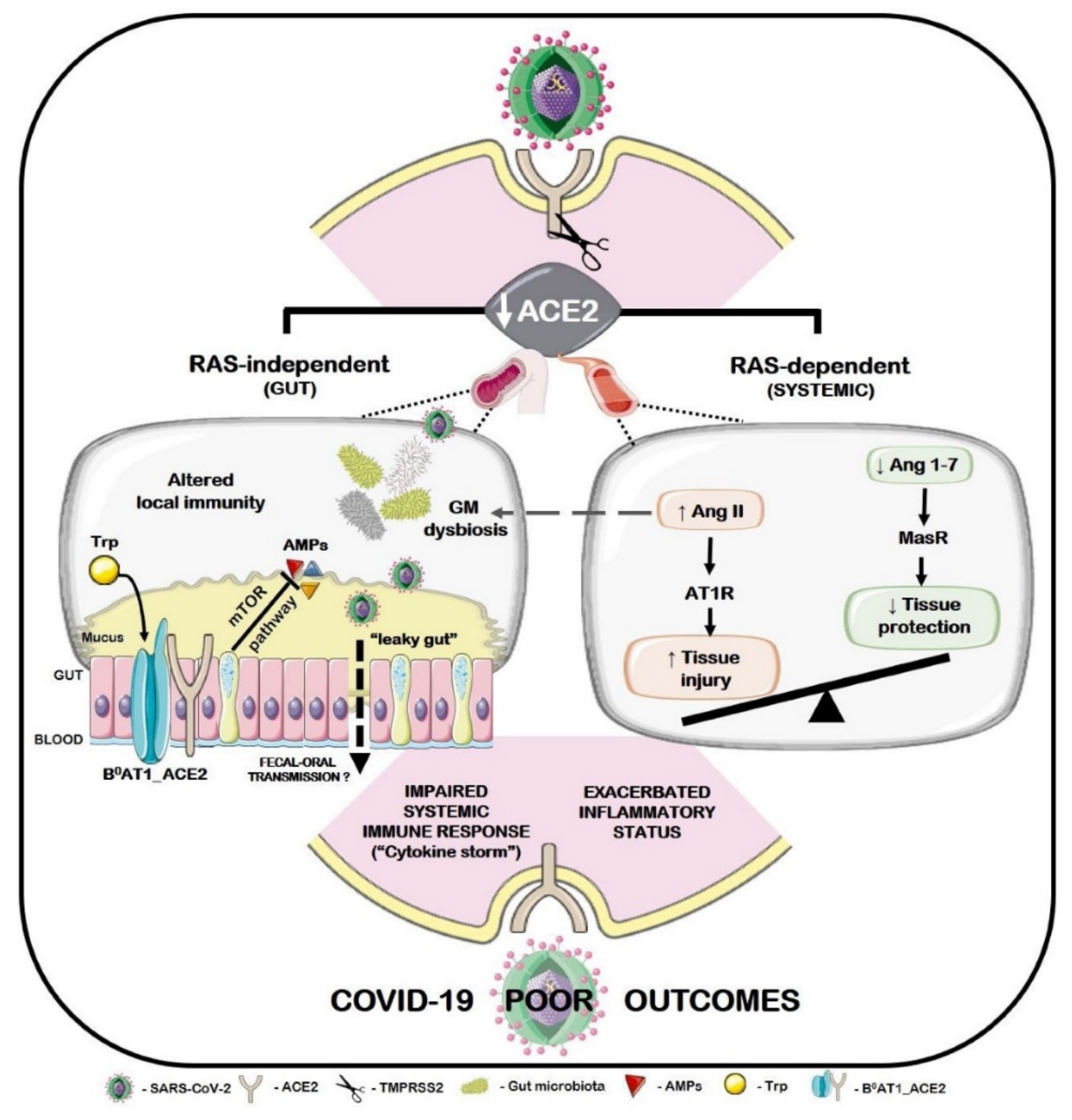
- Cheung, K.S.; Hung, I.F.; Chan, P.P.; Lung, K.; Tso, E.; Liu, R.; Ng, Y.; Chu, M.Y.; Chung, T.W.; Tam, A.R. Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from the Hong Kong cohort and systematic review and meta-analysis. Gastroenterology 2020, 159, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Jiang, X.; Zhang, Z.; Huang, S.; Zhang, Z.; Fang, Z.; Gu, Z.; Gao, L.; Shi, H.; Mai, L. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut 2020, 69, 997–1001. [Google Scholar] [CrossRef] [PubMed]
- Effenberger, M.; Grabherr, F.; Mayr, L.; Schwaerzler, J.; Nairz, M.; Seifert, M.; Hilbe, R.; Seiwald, S.; Scholl-Buergi, S.; Fritsche, G. Faecal calprotectin indicates intestinal inflammation in COVID-19. Gut 2020, 69, 1543–1544. [Google Scholar] [CrossRef] [PubMed]
- Zuo, T.; Zhang, F.; Lui, G.C.; Yeoh, Y.K.; Li, A.Y.; Zhan, H.; Wan, Y.; Chung, A.; Cheung, C.P.; Chen, N. Alterations in Gut Microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology 2020, 159, 944–955.e8. [Google Scholar] [CrossRef] [PubMed]
- Bird, L. Gut bacteria support antiviral immunity. Nat. Rev. Immunol. 2020, 20, 520–521. [Google Scholar] [CrossRef]
- Dhar, D.; Mohanty, A. Gut microbiota and Covid-19-possible link and implications. Virus Res. 2020, 285, 198018. [Google Scholar] [CrossRef]
- Bastiaanssen, T.F.; Cussotto, S.; Claesson, M.J.; Clarke, G.; Dinan, T.G.; Cryan, J.F. Gutted! Unraveling the role of the microbiome in major depressive disorder. Harv. Rev. Psychiatry 2020, 28, 26. [Google Scholar] [CrossRef] [PubMed]
- Amabebe, E.; Robert, F.O.; Agbalalah, T.; Orubu, E.S. Microbial dysbiosis-induced obesity: Role of gut microbiota in homoeostasis of energy metabolism. Br. J. Nutr. 2020, 123, 1127–1137. [Google Scholar] [CrossRef]
- Wang, L.; Alammar, N.; Singh, R.; Nanavati, J.; Song, Y.; Chaudhary, R.; Mullin, G.E. Gut microbial dysbiosis in the irritable bowel syndrome: A systematic review and meta-analysis of case-control studies. J. Acad. Nutr. Diet. 2020, 120, 565–586. [Google Scholar] [CrossRef]
- Gurung, M.; Li, Z.; You, H.; Rodrigues, R.; Jump, D.B.; Morgun, A.; Shulzhenko, N. Role of gut microbiota in type 2 diabetes pathophysiology. EBioMedicine 2020, 51, 102590. [Google Scholar] [CrossRef]
- Viana, S.D.; Nunes, S.; Reis, F. ACE2 imbalance as a key player for the poor outcomes in COVID-19 patients with age-related comorbidities–Role of gut microbiota dysbiosis. Ageing Res. Rev. 2020, 62, 101123. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, J.; Li, F.; Shi, Y. Main clinical features of COVID-19 and potential prognostic and therapeutic value of the microbiota in SARS-CoV-2 infections. Front. Microbiol. 2020, 11, 1302. [Google Scholar] [CrossRef] [PubMed]
- You, L.; Gong, Y.; Li, L.; Hu, X.; Brennan, C.; Kulikouskaya, V. Beneficial effects of three brown seaweed polysaccharides on gut microbiota and their structural characteristics: An overview. Int. J. Food Sci. Technol. 2020, 55, 1199–1206. [Google Scholar] [CrossRef]
- Lopez-Santamarina, A.; Miranda, J.M.; Mondragon, A.d.C.; Lamas, A.; Cardelle-Cobas, A.; Franco, C.M.; Cepeda, A. Potential use of marine seaweeds as prebiotics: A review. Molecules 2020, 25, 1004. [Google Scholar] [CrossRef] [PubMed]
- Jovic, T.H.; Ali, S.R.; Ibrahim, N.; Jessop, Z.M.; Tarassoli, S.P.; Dobbs, T.D.; Holford, P.; Thornton, C.A.; Whitaker, I.S. Could Vitamins Help in the Fight Against COVID-19? Nutrients 2020, 12, 2550. [Google Scholar] [CrossRef]
- Caroprese, M.; Ciliberti, M.G.; Albenzio, M. Immunological Activity of Marine Microalgae Extracts. Mar. Algae Extr. Process. Prod. Appl. 2015, 395–410. [Google Scholar]
- Fields, F.J.; Lejzerowicz, F.; Schroeder, D.; Ngoi, S.M.; Tran, M.; McDonald, D.; Jiang, L.; Chang, J.T.; Knight, R.; Mayfield, S. Effects of the microalgae Chlamydomonas on gastrointestinal health. J. Funct. Foods 2020, 65, 103738. [Google Scholar] [CrossRef]
- Neyrinck, A.M.; Taminiau, B.; Walgrave, H.; Daube, G.; Cani, P.D.; Bindels, L.B.; Delzenne, N.M. Spirulina protects against hepatic inflammation in aging: An effect related to the modulation of the gut microbiota? Nutrients 2017, 9, 633. [Google Scholar] [CrossRef]
- Chandrarathna, H.; Liyanage, T.; Edirisinghe, S.; Dananjaya, S.; Thulshan, E.; Nikapitiya, C.; Oh, C.; Kang, D.-H.; De Zoysa, M. Marine Microalgae, Spirulina maxima-derived modified pectin and modified pectin nanoparticles modulate the gut microbiota and trigger immune responses in mice. Mar. Drugs 2020, 18, 175. [Google Scholar] [CrossRef]
- Kim, K.; Ehrlich, A.; Perng, V.; Chase, J.A.; Raybould, H.; Li, X.; Atwill, E.R.; Whelan, R.; Sokale, A.; Liu, Y. Algae-derived β-glucan enhanced gut health and immune responses of weaned pigs experimentally infected with a pathogenic E. coli. Anim. Feed Sci. Technol. 2019, 248, 114–125. [Google Scholar] [CrossRef]
- Chen, L.; Xu, W.; Chen, D.; Chen, G.; Liu, J.; Zeng, X.; Shao, R.; Zhu, H. Digestibility of sulfated polysaccharide from the brown seaweed Ascophyllum nodosum and its effect on the human gut microbiota in vitro. Int. J. Biol. Macromol. 2018, 112, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Shang, Q.; Li, G.; Wang, X.; Yu, G. Degradation of marine algae-derived carbohydrates by Bacteroidetes isolated from human gut microbiota. Mar. Drugs 2017, 15, 92. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.; Walton, G.; Sousa, S.; Rocha-Santos, T.A.; Duarte, A.C.; Freitas, A.C.; Gomes, A.M. In vitro fermentation and prebiotic potential of selected extracts from seaweeds and mushrooms. LWT 2016, 73, 131–139. [Google Scholar] [CrossRef]
- He, L.-H.; Ren, L.-F.; Li, J.-F.; Wu, Y.-N.; Li, X.; Zhang, L. Intestinal flora as a potential strategy to fight SARS-CoV-2 infection. Front. Microbiol. 2020, 11, 1388. [Google Scholar] [CrossRef] [PubMed]
- Ferraces-Casais, P.; Lage-Yusty, M.; De Quirós, A.R.-B.; López-Hernández, J. Evaluation of bioactive compounds in fresh edible seaweeds. Food Anal. Methods 2012, 5, 828–834. [Google Scholar] [CrossRef]
- Hernández-Carmona, G.; Carrillo-Domínguez, S.; Arvizu-Higuera, D.L.; Rodríguez-Montesinos, Y.E.; Murillo-Álvarez, J.I.; Muñoz-Ochoa, M.; Castillo-Domínguez, R.M. Monthly variation in the chemical composition of Eisenia arborea JE Areschoug. J. Appl. Phycol. 2009, 21, 607–616. [Google Scholar] [CrossRef]
- Norziah, M.H.; Ching, C.Y. Nutritional composition of edible seaweed Gracilaria changgi. Food Chem. 2000, 68, 69–76. [Google Scholar] [CrossRef]
- Fabregas, J.; Herrero, C. Vitamin content of four marine microalgae. Potential use as source of vitamins in nutrition. J. Ind. Microbiol. 1990, 5, 259–263. [Google Scholar] [CrossRef]
- McDermid, K.J.; Stuercke, B. Nutritional composition of edible Hawaiian seaweeds. J. Appl. Phycol. 2003, 15, 513–524. [Google Scholar] [CrossRef]
- Ortiz, J.; Uquiche, E.; Robert, P.; Romero, N.; Quitral, V.; Llantén, C. Functional and nutritional value of the Chilean seaweeds Codium fragile, Gracilaria chilensis and Macrocystis pyrifera. Eur. J. Lipid Sci. Technol. 2009, 111, 320–327. [Google Scholar] [CrossRef]
- Martínez-Francés, E.; Escudero-Oñate, C. Cyanobacteria and microalgae in the production of valuable bioactive compounds. Microalgal Biotechnol. 2018, 6, 104–128. [Google Scholar]
- Lodermeyer, V.; Ssebyatika, G.; Passos, V.; Ponnurangam, A.; Malassa, A.; Ewald, E.; Stürzel, C.M.; Kirchhoff, F.; Rotger, M.; Falk, C.S. The antiviral activity of the cellular glycoprotein LGALS3BP/90K is species specific. J. Virol. 2018, 92, e00226-18. [Google Scholar] [CrossRef] [PubMed]
- Bedoux, G.; Caamal-Fuentes, E.; Boulho, R.; Marty, C.; Bourgougnon, N.; Freile-Pelegrín, Y.; Robledo, D. Antiviral and cytotoxic activities of polysaccharides extracted from four tropical seaweed species. Nat. Prod. Commun. 2017, 12, 1934578X1701200602. [Google Scholar] [CrossRef]
- Bagdonaite, I.; Wandall, H.H. Global aspects of viral glycosylation. Glycobiology 2018, 28, 443–467. [Google Scholar] [CrossRef]
- Ritchie, G.; Harvey, D.J.; Feldmann, F.; Stroeher, U.; Feldmann, H.; Royle, L.; Dwek, R.A.; Rudd, P.M. Identification of N-linked carbohydrates from severe acute respiratory syndrome (SARS) spike glycoprotein. Virology 2010, 399, 257–269. [Google Scholar] [CrossRef]
- Watanabe, Y.; Allen, J.D.; Wrapp, D.; McLellan, J.S.; Crispin, M. Site-specific glycan analysis of the SARS-CoV-2 spike. Science 2020, 369, 330–333. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef]
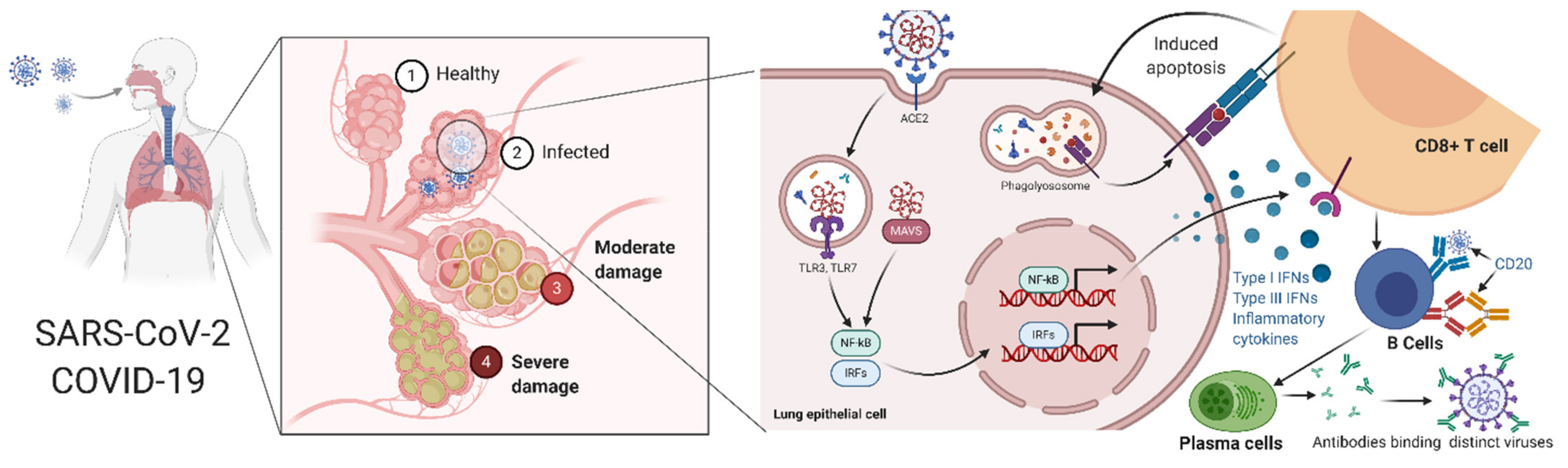
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Hivrale, A.; Ingale, A. Plant as a plenteous reserve of lectin. Plant Signal. Behav. 2013, 8, e26595. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.S.; Walia, A.K. Lectins from red algae and their biomedical potential. J. Appl. Phycol. 2018, 30, 1833–1858. [Google Scholar] [CrossRef] [PubMed]
- Van der Meer, F.; de Haan, C.; Schuurman, N.; Haijema, B.; Verheije, M.; Bosch, B.; Balzarini, J.; Egberink, H. The carbohydrate-binding plant lectins and the non-peptidic antibiotic pradimicin A target the glycans of the coronavirus envelope glycoproteins. J. Antimicrob. Chemother. 2007, 60, 741–749. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhou, Y.; Lu, K.; Pfefferle, S.; Bertram, S.; Glowacka, I.; Drosten, C.; Pöhlmann, S.; Simmons, G. A single asparagine-linked glycosylation site of the severe acute respiratory syndrome coronavirus spike glycoprotein facilitates inhibition by mannose-binding lectin through multiple mechanisms. J. Virol. 2010, 84, 8753–8764. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Deniz, F.S.S. Natural products as potential leads against coronaviruses: Could they be encouraging structural models against SARS-CoV-2? Nat. Prod. Bioprospect. 2020, 10, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Meuleman, P.; Albecka, A.; Belouzard, S.; Vercauteren, K.; Verhoye, L.; Wychowski, C.; Leroux-Roels, G.; Palmer, K.E.; Dubuisson, J. Griffithsin has antiviral activity against hepatitis C virus. Antimicrob. Agents Chemother. 2011, 55, 5159–5167. [Google Scholar] [CrossRef]
- Micewicz, E.D.; Cole, A.L.; Jung, C.-L.; Luong, H.; Phillips, M.L.; Pratikhya, P.; Sharma, S.; Waring, A.J.; Cole, A.M.; Ruchala, P. Grifonin-1: A small HIV-1 entry inhibitor derived from the algal lectin, Griffithsin. PLoS ONE 2010, 5, e14360. [Google Scholar] [CrossRef]
- Huskens, D.; Férir, G.; Vermeire, K.; Kehr, J.-C.; Balzarini, J.; Dittmann, E.; Schols, D. Microvirin, a novel α (1, 2)-mannose-specific lectin isolated from Microcystis aeruginosa, has anti-HIV-1 activity comparable with that of cyanovirin-N but a much higher safety profile. J. Biol. Chem. 2010, 285, 24845–24854. [Google Scholar] [CrossRef]
- Keeffe, J.R.; Gnanapragasam, P.N.; Gillespie, S.K.; Yong, J.; Bjorkman, P.J.; Mayo, S.L. Designed oligomers of cyanovirin-N show enhanced HIV neutralization. Proc. Natl. Acad. Sci. USA 2011, 108, 14079–14084. [Google Scholar] [CrossRef]
- Gondim, A.C.; da Silva, S.R.; Mathys, L.; Noppen, S.; Liekens, S.; Sampaio, A.H.; Nagano, C.S.; Rocha, C.R.C.; Nascimento, K.S.; Cavada, B.S. Potent antiviral activity of carbohydrate-specific algal and leguminous lectins from the Brazilian biodiversity. MedChemComm 2019, 10, 390–398. [Google Scholar] [CrossRef]
- Sato, Y.; Morimoto, K.; Kubo, T.; Sakaguchi, T.; Nishizono, A.; Hirayama, M.; Hori, K. Entry inhibition of influenza viruses with high mannose binding lectin ESA-2 from the red alga Eucheuma serra through the recognition of viral hemagglutinin. Mar. Drugs 2015, 13, 3454–3465. [Google Scholar] [CrossRef]
- Sato, Y.; Morimoto, K.; Hirayama, M.; Hori, K. High mannose-specific lectin (KAA-2) from the red alga Kappaphycus alvarezii potently inhibits influenza virus infection in a strain-independent manner. Biochem. Biophys. Res. Commun. 2011, 405, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Hirayama, M.; Morimoto, K.; Yamamoto, N.; Okuyama, S.; Hori, K. High mannose-binding lectin with preference for the cluster of α1–2-mannose from the green alga Boodlea coacta is a potent entry inhibitor of HIV-1 and influenza viruses. J. Biol. Chem. 2011, 286, 19446–19458. [Google Scholar] [CrossRef]
- Mu, J.; Hirayama, M.; Sato, Y.; Morimoto, K.; Hori, K. A novel high-mannose specific lectin from the green alga Halimeda renschii exhibits a potent anti-influenza virus activity through high-affinity binding to the viral hemagglutinin. Mar. Drugs 2017, 15, 255. [Google Scholar] [CrossRef] [PubMed]
- Bewley, C.A.; Cai, M.; Ray, S.; Ghirlando, R.; Yamaguchi, M.; Muramoto, K. New carbohydrate specificity and HIV-1 fusion blocking activity of the cyanobacterial protein MVL: NMR, ITC and sedimentation equilibrium studies. J. Mol. Biol. 2004, 339, 901–914. [Google Scholar] [CrossRef]
- Takebe, Y.; Saucedo, C.J.; Lund, G.; Uenishi, R.; Hase, S.; Tsuchiura, T.; Kneteman, N.; Ramessar, K.; Tyrrell, D.L.J.; Shirakura, M. Antiviral lectins from red and blue-green algae show potent in vitro and in vivo activity against hepatitis C virus. PLoS ONE 2013, 8, e64449. [Google Scholar] [CrossRef] [PubMed]
- Garrison, A.R.; Giomarelli, B.G.; Lear-Rooney, C.M.; Saucedo, C.J.; Yellayi, S.; Krumpe, L.R.; Rose, M.; Paragas, J.; Bray, M.; Olinger, G.G., Jr. The cyanobacterial lectin scytovirin displays potent in vitro and in vivo activity against Zaire Ebola virus. Antivir. Res. 2014, 112, 1–7. [Google Scholar] [CrossRef]
- Bilal, M.; Iqbal, H.M. Biologically active macromolecules: Extraction strategies, therapeutic potential and biomedical perspective. Int. J. Biol. Macromol. 2020, 151, 1–18. [Google Scholar] [CrossRef]
- Peterhans, E. Oxidants and antioxidants in viral diseases: Disease mechanisms and metabolic regulation. J. Nutr. 1997, 127, 962S–965S. [Google Scholar] [CrossRef]
- Fedoreyev, S.A.; Krylova, N.V.; Mishchenko, N.P.; Vasileva, E.A.; Pislyagin, E.A.; Iunikhina, O.V.; Lavrov, V.F.; Svitich, O.A.; Ebralidze, L.K.; Leonova, G.N. Antiviral and antioxidant properties of echinochrome A. Mar. Drugs 2018, 16, 509. [Google Scholar] [CrossRef]
- Kesarwani, P.; Murali, A.K.; Al-Khami, A.A.; Mehrotra, S. Redox regulation of T-cell function: From molecular mechanisms to significance in human health and disease. Antioxid. Redox Signal. 2013, 18, 1497–1534. [Google Scholar] [CrossRef]
- Silva, T.; Salomon, P.S.; Hamerski, L.; Walter, J.; Menezes, R.B.; Siqueira, J.E.; Santos, A.; Santos, J.A.M.; Ferme, N.; Guimarães, T. Inhibitory effect of microalgae and cyanobacteria extracts on influenza virus replication and neuraminidase activity. PeerJ 2018, 6, e5716. [Google Scholar] [CrossRef]
- Khomich, O.A.; Kochetkov, S.N.; Bartosch, B.; Ivanov, A.V. Redox biology of respiratory viral infections. Viruses 2018, 10, 392. [Google Scholar] [CrossRef]
- Delgado-Roche, L.; Mesta, F. Oxidative stress as key player in severe acute respiratory syndrome coronavirus (SARS-CoV) infection. Arch. Med Res. 2020, 51, 384–387. [Google Scholar] [CrossRef]
- Cecchini, R.; Cecchini, A.L. SARS-CoV-2 infection pathogenesis is related to oxidative stress as a response to aggression. Med. Hypotheses 2020, 143, 110102. [Google Scholar] [CrossRef]
- Derouiche, S. Oxidative stress associated with SARS-Cov-2 (COVID-19) increases the severity of the lung disease-a systematic review. J. Infect. Dis. Epidemiol. 2020, 6, 121. [Google Scholar]
- Sansone, C.; Brunet, C.; Noonan, D.M.; Albini, A. Marine algal antioxidants as potential vectors for controlling viral diseases. Antioxidants 2020, 9, 392. [Google Scholar] [CrossRef] [PubMed]
- Khalid, S.; Abbas, M.; Saeed, F.; Bader-Ul-Ain, H.; Suleria, H.A.R. Therapeutic potential of seaweed bioactive compounds. Seaweed Biomater. 2018, 1, 7–26. [Google Scholar]
- Kelman, D.; Posner, E.K.; McDermid, K.J.; Tabandera, N.K.; Wright, P.R.; Wright, A.D. Antioxidant activity of Hawaiian marine algae. Mar. Drugs 2012, 10, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as sources of high added-value compounds—A brief review of recent work. Biotechnol. Prog. 2011, 27, 597–613. [Google Scholar] [CrossRef] [PubMed]
- Lohrmann, N.L.; Logan, B.A.; Johnson, A.S. Seasonal acclimatization of antioxidants and photosynthesis in Chondrus crispus and Mastocarpus stellatus, two co-occurring red algae with differing stress tolerances. Biol. Bull. 2004, 207, 225–232. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.A.; Khalili, M.; Dehpour, A.A. Antioxidant activity of ethyl acetate and methanolic extracts of two marine algae, Nannochloropsis oculata and Gracilaria gracilis-an in vitro assay. Braz. J. Pharm. Sci. 2018, 54, e17280. [Google Scholar] [CrossRef]
- Cornish, M.L.; Garbary, D.J. Antioxidants from macroalgae: Potential applications in human health and nutrition. Algae 2010, 25, 155–171. [Google Scholar] [CrossRef]
| Algae Spp. | Antiviral Bioactive Compounds | Structures | Active against Viruses | Mechanism of Actions |
|---|---|---|---|---|
| Gigartina skottsbergii | Carrageenan | 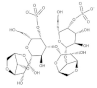 | Influenza virus, DENV, HSV-1, HSV-2, HPV, HRV, HIV | Inhibition of the binding or the internalization of viruses into host cells (Stage I, II, III) * |
| Callophyllis variegata, Agardhiella tenera, Schizymenia binderi, Cryptonemia crenulata | Galactan | 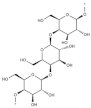 | HSV-1, HSV-2, HIV-1, HIV-2, DENV, HAV | Blockage of virus adhesion and replication into host cells (Stage I, III) |
| Laminaria hyperborea, Laminaria digitata, Laminaria japonica, | Alginate | 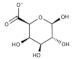 | HIV IAV, HBV | Inhibition of the inverse transcriptase in the RNA virus (Stage III) |
| Adenocytis utricularis, Undaria pinnatifida, Stoechospermum marginatum, Cystoseira indica, Cladosiphon okamuranus, Fucus vesiculosus | Fucan | 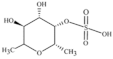 | HSV-1, HSV-2, HCMV, VSV, Sindbis virus, HIV-1 | Inhibition of cell adhesion (Stage I); blockage of reverse transcriptase (Stage III) |
| Fucus vesiculosus, Saccharina longicruris, Ascophyllum nodosum | Laminaran | 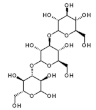 | HIV | Blockage of reverse transcriptase (Stage III) |
| Chlorella vulgaris,Cochlodinium polykrikoides, Porphyridium sp. | Sulfated polysaccharides (e.g., Agar) | 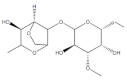 | Influenza A and B viruses, RSV-A, RSV-B, parainfluenza-2 | Inhibition of the cytopathic effect (Stage II); inhibition of PMN migration toward chemoattractant molecules; partial blocking of the adhesion to endothelial cells |
| Dunaliella primolecta | α- and β-Pheophorbide | 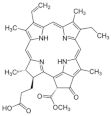 | HSV-1 | Inhibition of adsorption and invasion (Stage I, II) |
| Name of Algae-Derived Metabolites | Source Organisms |
|---|---|
| Griffithsin | Red algae, Griffithsia |
| Ulvans | Green algae, Ulva sp. |
| Fucoidans | Brown algae |
| Phycocyanobilin, phycoerythrobilin, and folic acid | Arthrospira |
| Sulfated polysaccharides | Red algae Porphyridium sp. |
| Astaxanthin (nASX) | Haematococcus pluvialis |
| Spirulina | Arthrospira platensis |
| Halitunal | Halimeda tuna |
| Dieckol | Ecklonia cava |
| n-Hexadecanoic acid, hexadecenoic acid, methyl ester, n-decanoic acid, and 9-dodecenoic acid | Laurencia papillosa, Gracilaria corticata, and Grateloupia filicina |
| Vitamins | Source | Reference |
|---|---|---|
| Vitamin C | Porphyra umbilicalis, Himanthalia elongata, Laminaria spp., Gracilaria changii, Palmaria palmata Eisenia arborea J.E. Areschoug | [70,71,72] |
| Vitamin B1 | Tetraselmis suecica, Isochrysis galbana, Dunaliella tertiolecta, Chlorella stigmatophora, Chondrus ocellatus | [73,74] |
| Vitamin B2 | Tetraselmis suecica, Isochrysis galbana, Dunaliella tertiolecta, Chlorella stigmatophora | [73] |
| Vitamin B12 | Tetraselmis suecica, Isochrysis galbana, Dunaliella tertiolecta, Chlorella stigmatophora | [73] |
| Vitamin E | Laminaria spp., Porphyra umbilicalis, Himanthalia elongata, Palmaria palmata, Eisenia arborea J.E. Areschoug, Tetraselmis suecica, Isochrysis galbana, Dunaliella tertiolecta, Chlorella stigmatophora | [70,71,73] |
| α-tocopherol (vitamin E) | Macrocystis pyrifera, Gracilaria chilensis, Codium fragile | [75] |
| β-carotene | Gracilaria changgi, Laminaria spp., Porphyra umbilicalis, Gracilaria chilensis, Codium fragile, Tetraselmis suecica, Isochrysis galbana, Dunaliella tertiolecta, Chlorella stigmatophora | [70,72,73,75] |
| Lectin Designated | Marine Algae Source | Virus | References |
|---|---|---|---|
| GRFT | Griffithsia Sp. | SARS-CoV, HIV, HCV | [37,89,90] |
| Microvirin | Microcystis aeruginosa | HIV-1 | [91] |
| Cyanovirin | Nostoc ellipsosporum | HIV | [92] |
| AML, BSL, HML, MEL, Sfl | Amansia multifida, Bryothamnion seaforthii, Hypnea musciformis, Meristiella echinocarpa and Solieria filiformis | HIV and influenza | [93] |
| ESA-2 | Eucheuma serrai | Influenza | [94] |
| KAA-2 | Kappaphycus alvarezii | Influenza | [95] |
| BCA | Boodlea coacta | Influenza, HIV | [96] |
| HRL40 | Halimeda renschii | Influenza | [97] |
| MVL | Microcystis viridis | HIV-1 | [98] |
| Scytovirin | Scytonema varium | HCV, HIV, Ebola | [99,100] |
| Antioxidant Compound | Microalgae Source | References |
|---|---|---|
| Chlorophyll-a derivatives, pheophorbidea | Enteromorpha prolifera | |
| Fucoxanthin | Undaria pinnatifida, Turbinaria ornata | [111,112] |
| Phycoerythrobilin | Porphyra sp. | |
| β-carotene | Dunaliella salina, Chondrus crispus, Mastocarpus stellatus | |
| Astaxanthin, canthaxanthin, lutein | Haematococcuspluvialis | |
| Lutein, violaxanthin | Chlorella pyrenoidosa | [113,114] |
| Canthaxanthin, astaxanthin | Chlorella vulgaris | |
| Phenolic and flavonoids | Nannochloropsis oculata, Gracilaria gracilis | [115] |
| Phycoerythrin, phycocyanin | Red algae | |
| Terpenoids | Cystoseira sp. | |
| Sulfated polysaccharides (Fucoidan, alginic acid, laminaran, sulfated galactans, galactans, sulfated glycosaminoglycan, porphyran | Turbinaria conoides, Laminaria japonica, red algae, Sargassum wightii, Porphyra sp. | |
| Stypodiol, taondiol, isoepitaondiol | Taonia atomaria | [116] |
| Vitamins | Ascorbate (vitamin C) | |
| Tocopherol (vitamin E) | α-, γ-, δ- tocopherol | |
| Carotenoids | α-carotene and β-carotene | |
| Fucoxanthin and astaxanthin | ||
| Polyphenols | Phlorotannin - Brown algae polyphenol | Fucol Phlorethol Fucophlorethol Fuhalol Isofuhalol Eckol |
| Catechin | Catechin (3-hydroxyflavone)/catechin gallate Epicatechin/epicatechin gallate Epigallocatechin/epigallocatechin gallate | |
| Phenolic acid | ||
| Flavonoids | Anthocyanins Flavonols Flavanols Flavanones Flavones Isoflavones | |
| Tannins | ||
| Lignans | ||
| Mycosporine-like amino acids | Mycosporine–glycine |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, M.A.; Parra-Saldivar, R.; Bilal, M.; Afroze, C.A.; Ahmed, M.N.; Iqbal, H.M.N.; Xu, J. Algae-Derived Bioactive Molecules for the Potential Treatment of SARS-CoV-2. Molecules 2021, 26, 2134. https://doi.org/10.3390/molecules26082134
Alam MA, Parra-Saldivar R, Bilal M, Afroze CA, Ahmed MN, Iqbal HMN, Xu J. Algae-Derived Bioactive Molecules for the Potential Treatment of SARS-CoV-2. Molecules. 2021; 26(8):2134. https://doi.org/10.3390/molecules26082134
Chicago/Turabian StyleAlam, Md. Asraful, Roberto Parra-Saldivar, Muhammad Bilal, Chowdhury Alfi Afroze, Md. Nasir Ahmed, Hafiz M.N. Iqbal, and Jingliang Xu. 2021. "Algae-Derived Bioactive Molecules for the Potential Treatment of SARS-CoV-2" Molecules 26, no. 8: 2134. https://doi.org/10.3390/molecules26082134
APA StyleAlam, M. A., Parra-Saldivar, R., Bilal, M., Afroze, C. A., Ahmed, M. N., Iqbal, H. M. N., & Xu, J. (2021). Algae-Derived Bioactive Molecules for the Potential Treatment of SARS-CoV-2. Molecules, 26(8), 2134. https://doi.org/10.3390/molecules26082134