Modulation of Renal Function in a Metabolic Syndrome Rat Model by Antioxidants in Hibiscus sabdariffa L.
Abstract
1. Introduction
2. Results
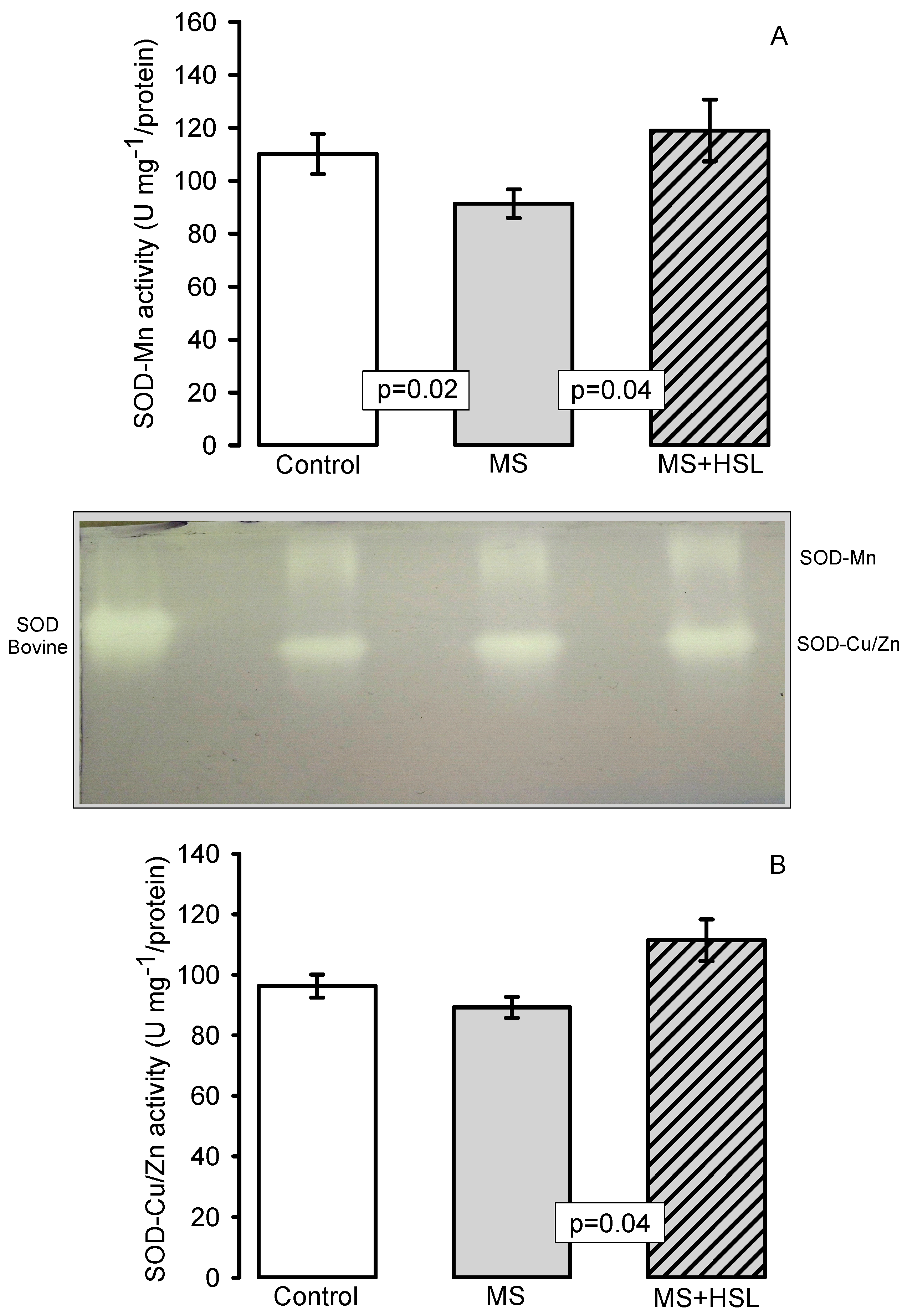
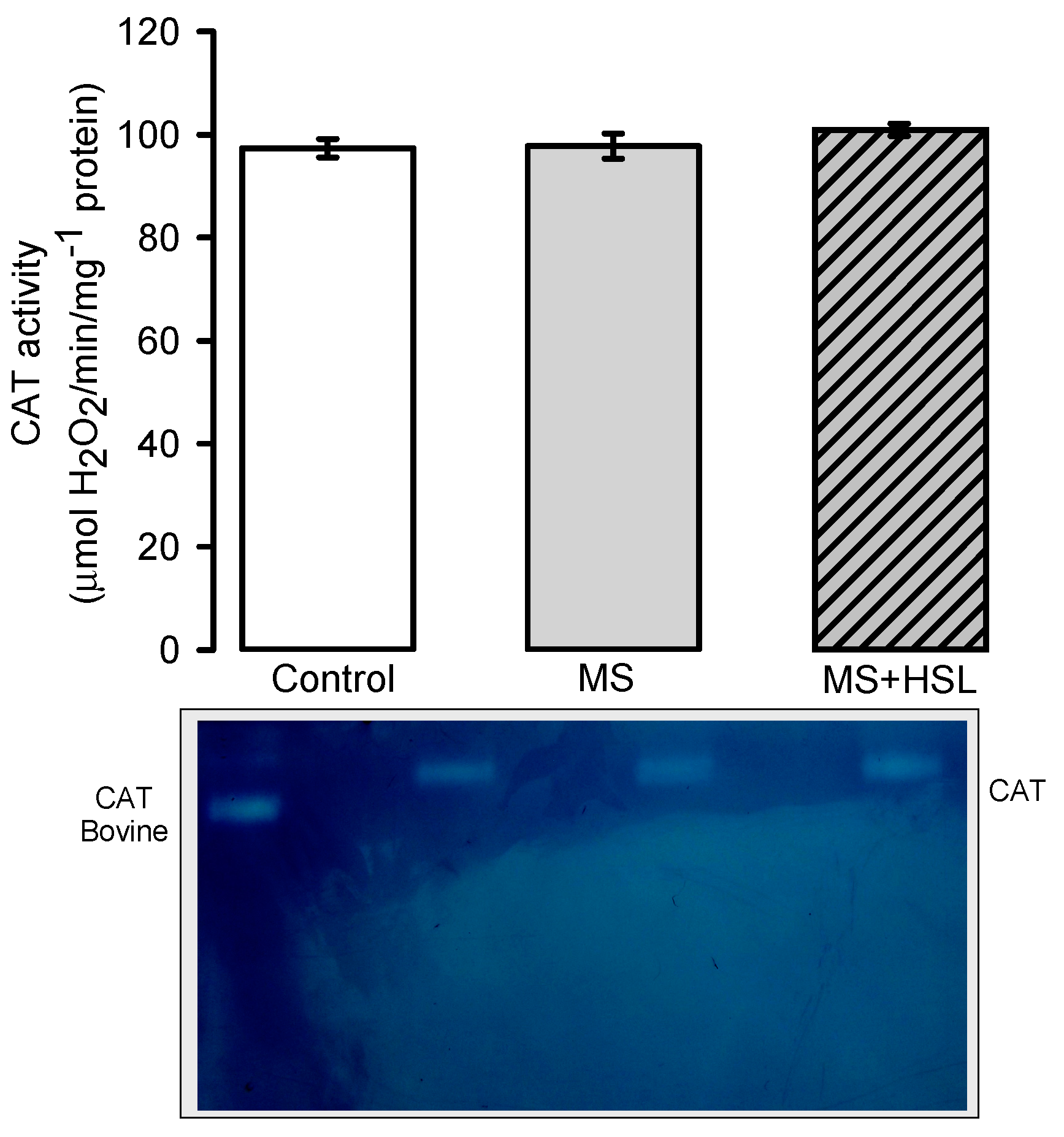
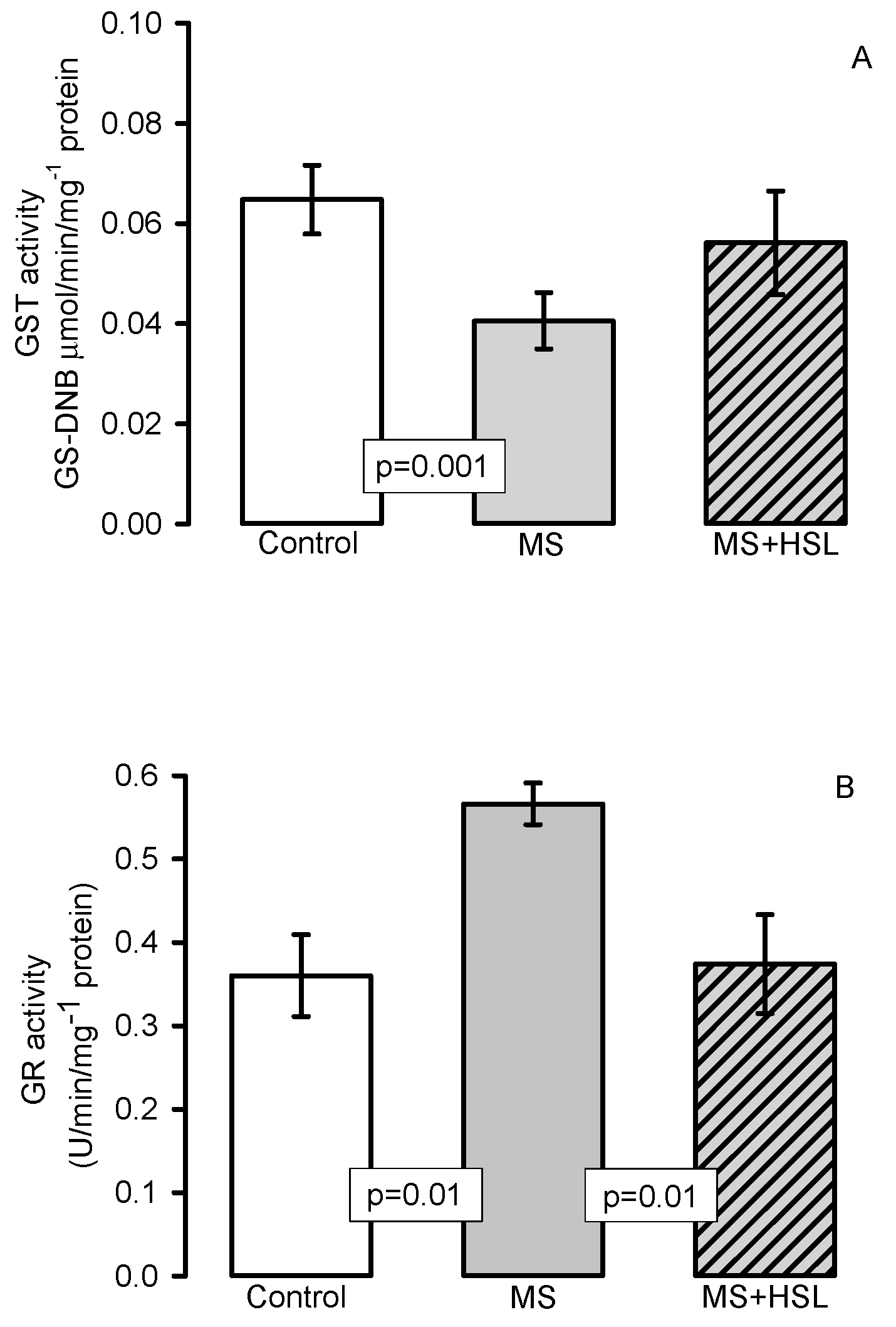
Antioxidant Enzymes
3. Discussion
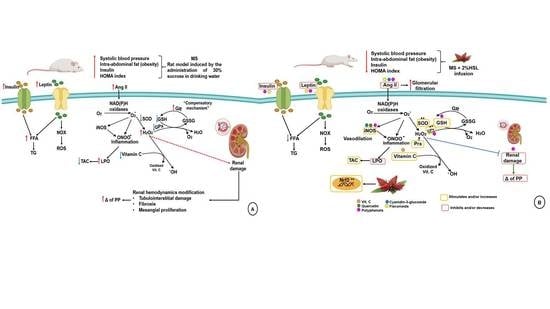
3.1. Physiological Characteristics of the MS Model
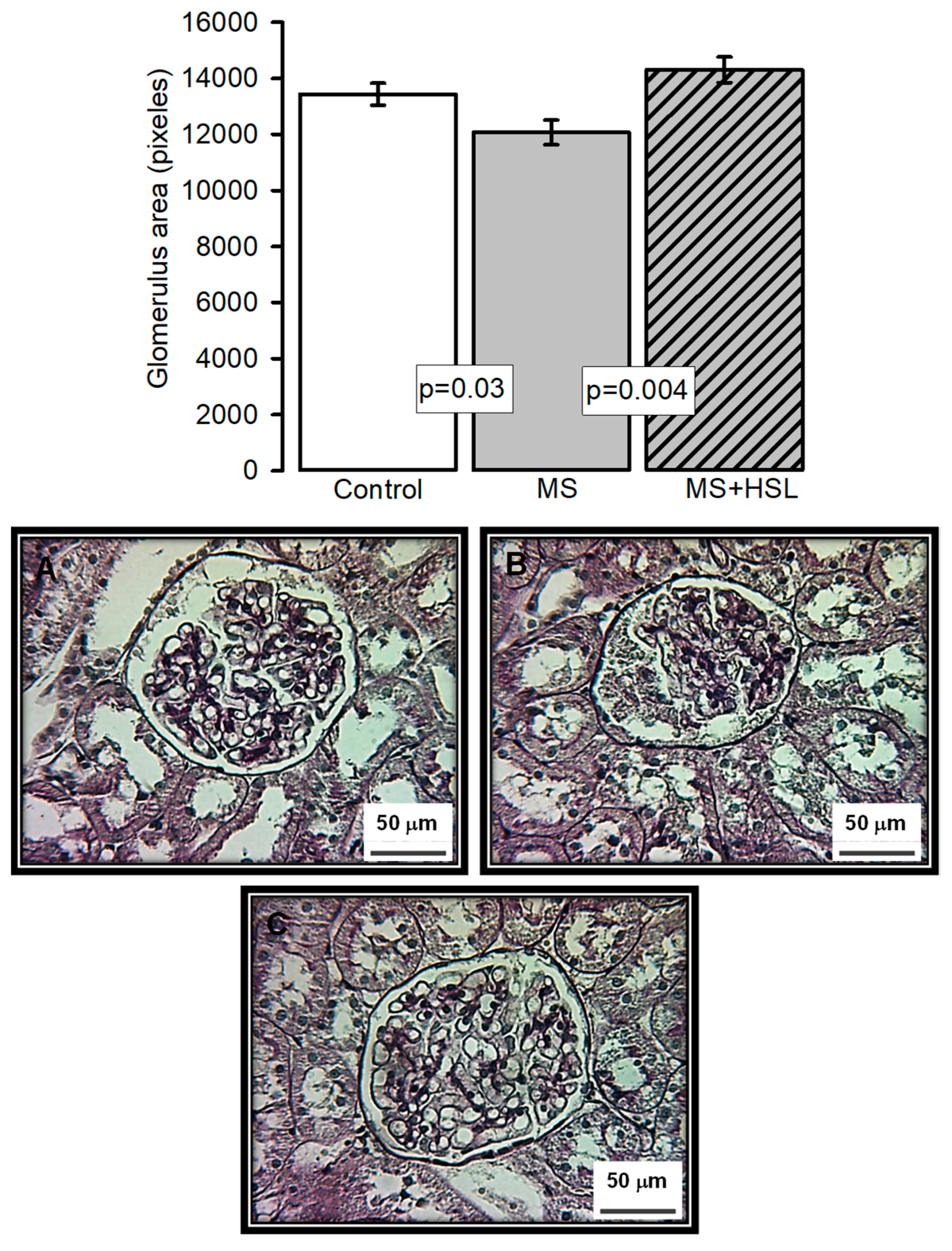
3.2. Effect of a 2% HSL Infusion on the Kidney Function in MS
3.3. Renal Vascular Resistance
3.4. Oxidant/Antioxidant Effect of the HSL Infusion
4. Material and Methods
4.1. Animals
4.2. HSL Infusion
4.3. Albuminuria and Creatinine Depuration
4.4. Isolated Perfused Kidney
4.5. Serum Sample
4.6. Biochemical Variables
4.7. Kidney Homogenate
4.8. Lipid Peroxidation
4.9. Evaluation of Total Antioxidant Capacity
4.10. Vitamin C
4.11. Carbonylation
4.12. GSH Concentration
4.13. NO3−/NO2− Ratio
4.14. Superoxide Dismutase, Peroxidase and Catalase Activities in Native Gels
4.15. Enzymes That Use Glutathione and/or Oxidized Glutathione
4.16. Histopathological Analysis
4.17. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Pérez-Torres, I.; Guarner, V.; El Hafidi, M.; Baños, G. Sex hormones, metabolic syndrome and kidney. Curr. Top. Med. Chem. 2011, 11, 1694–1705. [Google Scholar] [PubMed]
- Nashar, K.; Egan, B.M. Relationship between chronic kidney disease and metabolic syndrome: Current perspectives. Diabetes. Metab. Syndr. Obes. 2014, 7, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Zuo, L.; Zhou, T.; Panell, B.K.; Ziegler, A.C.; Best, T.M. Biological and physiological role of reactive oxygen species–the good, the bad and the ugly. Acta Physiol. 2015, 214, 329–348. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.K.; Sindhu, K.K. Oxidative stress and metabolic syndrome. Life Sci. 2009, 84, 705–712. [Google Scholar] [CrossRef]
- Krata, N.; Zagozdzon, R.; Foroncewicz, B.; Mucha, K. Oxidative Stress in Kidney Diseases: The Cause of the Consequence? Arch. Immunol. Ther. Ex. 2018, 66, 211–220. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line antioxidants-superoxide dismutase (SOD), catalase (CAT) and gluthione peroxidase (GPX): Their fundamental role in the entire antioxidant defense grid. Alexandria. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Bretón-Romero, R.; Lamas, S. Hydrogen peroxide signaling in vascular endothelial cells. Redox. Biol. 2014, 2, 529–534. [Google Scholar] [CrossRef]
- Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox. Biol. 2017, 11, 613–619. [Google Scholar] [CrossRef]
- Goyal, M.M.; Basak, A. Human catalase: Looking for complete identity. Protein. Cell 2010, 10, 888–897. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Guarner-Lans, V.; Rubio-Ruiz, M.E. Reductive stress in inflammation-associated diseases and the pro-oxidant effect of antioxidant agents. Int. J. Mol. Sci. 2017, 18, 2098. [Google Scholar] [CrossRef]
- Samsam-Shariat, S.Z.; Bolhasani, M.; Sarrafzadegan, N.; Najafi, S.; Asgary, S. Relationship between blood peroxidases activity and visfatin levels in metabolic syndrome patients. ARYA Atheroscler. 2014, 10, 218–226. [Google Scholar]
- Raza, H. Dual localization of glutathione S-transferasa in the cytosol and mitochondria: Implications in oxidative stress, toxicity and disease. FEBS Lett. 2011, 278, 4243–4251. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Gupta, S.; Singh, K.; Mehndiratta, M.; Gautam, A.; Kalra, O.P.; Shukla, R.; Gambhir, J.K. Association of glutathione-S-transferasas with patients of type 2 diabetes mellitus with and without nephronpathy. Diabetes Metab. Syndr. 2016, 10, 194–197. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Ruiz, M.E.; Guarner-Lans, V.; Cano-Martínez, A.; Díaz-Díaz, E.; Manzano-Pech, L.; Gamas-Magaña, A.; Castrejón-Tellez, V.; Tapia-Cortina, C.; Pérez-Torres, I. Resveratrol and quercetin administration improves antioxidant DEFENSES and reduces fatty liver in metabolic syndrome rats. Molecules 2019, 24, 1297. [Google Scholar] [CrossRef] [PubMed]
- Bajic, V.P.; VanNeste, C.; Obradovic, M.; Zafirovic, S.; Radak, D.; Bajic, V.B.; Essack, M.; Isenovic, E.R. Glutathione “Redox Homeostasis” and Its Relation to Cardiovascular Disease. Oxid. Med. Cell Longev. 2019, 2019, 5028181. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Torres, I.; Ruíz-Ramírez, A.; Baños, G.; El-Hafidi, M. Hibiscus sabdariffa linnaeus (Malvaceae), curcumin and resveratrol as alternative medicinal agents against metabolic syndrome. Cardiovasc. Hematol. Agents Med. Chem. 2013, 11, 25–37. [Google Scholar] [CrossRef]
- Wen-Chin, L.; Chau-Jong, W.; Yu-Hsin, C.; Jen-Dong, H.; Su-Ya, C.; Hong-Chen, C.; Huei-Jane, L. Polyphenols Extracts from Hibiscus sabdariffa Linnaeus Attenuate Nephropathy in experimental type 1 diabetes. J. Agric. Food Chem. 2009, 57, 2206–2210. [Google Scholar]
- Shun-Ting, C.; Hsin-Yi, L.; Chia-Cheng, L.; Lu-Chen, C.; Pei-Chi, C.; Yu-Chen, L.; Tin-Yun, H.; Chien-Yun, H. Exploring the effect and mechanism of Hibiscus sabdariffa on urinary tract infection and experimental renal inflammation. J. Ethnopharmacol. 2016, 194, 617–625. [Google Scholar]
- Pérez-Torres, I.; Torres-Narváez, J.C.; Guarner-Lans, V.; Díaz-Díaz, E.; Perezpeña-Díazconti, M.; Romero-Palacios, A.; Manzano-Pech, L. Myocardial protection from ischemia reperfusion damage by the antioxidant effect of Hibiscus sabdariffa linnaeus on metabolic syndrome rats. Oxid. Med. Cell Longev. 2019, 2019, 1724194. [Google Scholar] [CrossRef]
- Ojulari, O.V.; Gi, L.S.; Nam, J.O. Beneficial effects of natural bioactive compounds from Hibiscus Sabdariffa L on obesity. Molecules 2019, 24, 210. [Google Scholar] [CrossRef]
- Zuñiga-Muñoz, A.M.; Guarner-Lans, V.; Díaz-Cruz, A.; Diaz-Diaz, E.; Nava-Cuellar, C.; Beltrán-Rodríguez, U.; Pérez-Torres, I. Modulation of oxidative stress in fatty liver of rat with metabolic syndrome by Hibiscus Sabdariffa. Immun. Endoc. Agen. Med. Chem. 2013, 13, 1–10. [Google Scholar]
- Ghadge, A.A.; Khaire, A.A. Leptin as a predictive marker for metabolic syndrome. Cytokine 2019, 121, 154735. [Google Scholar] [CrossRef]
- Diez-Echave, P.; Vezza, T.; Rodríguez-Nogales, A.; Ruiz-Malagón, A.J.; Hidalgo-García, L.; Garrido-Mesa, J.; Molina-Tijeras, J.A.; Romero, M.; Robles-Vera, I.; Pimentel-Moral, S.; et al. The prebiotic properties of Hibiscus sabdariffa extract contribute to the beneficial effects in diet-induced obesity in mice. Food Res. Int. 2020, 127, 108722. [Google Scholar] [CrossRef]
- Hashimoto, S.; Maoka, T.; Kawata, T.; Mochizuki, T.; Koike, T.; Shigematsu, T. Roles of insulin receptor substrates (IRS) in renal function and renal hemodynamics. PLoS ONE 2020, 15, e0242332. [Google Scholar] [CrossRef]
- Sasaki, R.; Nishimura, N.; Hoshino, H.; Isa, Y.; Kadowaki, M.; Ichi, T.; Tanaka, A.; Shin, N.; Fukuda, I.; Ashida, H.; et al. Cyanidin 3-glucoside ameliorates hyperglycemia and insulin sensitivity due to downregulation of retinol binding protein 4 expression in diabetic mice. Biochem. Pharmacol. 2007, 74, 1619–1627. [Google Scholar] [CrossRef] [PubMed]
- Riaz, G.; Chopra, R. A review on phytochemistry and therapeutic uses of Hibiscus sabdariffa L. Biomed. Pharmacother. 2018, 102, 575–586. [Google Scholar] [CrossRef]
- Joven, J.; March, I.; Espinel, E.; Fernández-Arroyo, S.; Rodríguez-Gallego, E.; Aragonès, G.; Beltrán-Debon, R.; Alonso-Villaverde, C.; Rios, L.; Martin-Paredero, V.; et al. Hibiscus sabdariffa extract lowers blood pressure and improves endothelial function. Mol. Nutr. Food Res. 2014, 58, 1374–1378. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.C.; Budin, S.B.; Othman, F.; Latip, J.; Zainalabidin, S. Roselle polyphenols exert potent negative inotropic effects via modulation of intracellular calcium regulatory channels in isolated rat heart. Cardiovasc. Toxicol. 2017, 17, 251–259. [Google Scholar] [CrossRef]
- Lin, C.Y.; Huang, C.S.; Huang, C.Y.; Yin, M.C. Anticoagulatory, antiinflammatory, and antioxidative effects of protocat- echuic acid in diabetic mice. J. Agric. Food Chem. 2009, 57, 6661–6667. [Google Scholar] [CrossRef] [PubMed]
- Locatelli, F.; Pozzoni, P.; Del Vecchio, L. Renal manifestations in the metabolic sydrome. J. Am. Soc. Nephrol. 2006, 17, S81–S85. [Google Scholar] [CrossRef] [PubMed]
- Yi-Sun, Y.; Chau-Jong, W.; Chien-Ning, H.; Mu-Lin, C.; Ming-Jinn, C.; Chiung-Huei, P. Polyphenols of Hibiscus sabdariffa improved diabetic nephropathy via attenuating renal epithelial mesenchymal transition. J. Agric. Food Chem. 2013, 61, 7545–7551. [Google Scholar]
- Ali, B.H.; Cahliková, L.; Opletal, L.; Karaca, T.; Manoj, P.; Ramkumar, A.; Al Suleimani, Y.M.; Al za’abi, M.; Nemmar, A.; Chocholousova-Havlikova, L.; et al. Effect of aqueous extract and anthocyanins of calyces of Hibiscus sabdariffa (Malvaceae) in rats with adenine-induced chronic kidney disease. J. Pharm. Pharmacol. 2017, 69, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Nwachukwu, D.C.; Aneke, E.I.; Nwachukwu, N.Z.; Azubike, N.; Obika, L.F. Does consumption of an aqueous extract of Hibscus sabdariffa affect renal function in subjects with mild to moderate hypertension? J. Physiol. Sci. 2016, 67, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Melchert, A.; Canuto, R.A.; Genari, V.; Salvador-Gonzalez, F.M.; Prandini de la Costa, R.R.; Guimaraes-Okamoto, P.T.C.; Amaro, J.L.; Canavese, R.S. Effect of Hibiscus sabdariffa supplementation on renal function and lipid profile in obese rats. Asian J. Anim. Vet. Adv. 2016, 11, 693–700. [Google Scholar] [CrossRef]
- Imig, J.D.; Cook, K.A.; Inscho, W.E. Postglomerular vasoconstriction to angiotensin II and norepinephrine depends on intracellular calcium release. Gen. Phamacol. 2000, 34, 409–415. [Google Scholar] [CrossRef]
- Correia, A.G.; Madden, A.C.; Bergtröm, G.; Evan, R.G. Effects of renal medullary and intravenous norepinephrine on renal antihypertensive function. Hypertension 2000, 35, 965–970. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rafikova, O.; Salah, E.M.; Tofovic, S.P. Renal and metabolic effects of tempol in obese ZSF1 rats--distinct role for superoxide and hydrogen peroxide in diabetic renal injury. Metabolism 2008, 57, 1434–1444. [Google Scholar] [CrossRef] [PubMed]
- Hatoum, O.A.; Binion, D.G.; Miura, H.; Telford, G.; Otterson, M.F.; Gutterman, D.D. Role of hydrogen peroxide in ACh-induced dilation of human submucosal intestinal microvessels. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H48–H54. [Google Scholar] [CrossRef][Green Version]
- Park, S.W.; Noh, H.J.; Sung, D.J.; Kim, J.G.; Kim, J.M.; Ryu, S.Y.; Kang, K.J.; Kim, B.; Bae, Y.M.; Cho, H. Hydrogen peroxide induces vasorelaxation by enhancing 4-aminopyridine-sensitive Kv currents through S-glutathionylation. Eur. J. Physiol. 2015, 467, 285–297. [Google Scholar] [CrossRef]
- Cseko, C.; Bagi, Z.; Koller, A. Biphasic effect of hydrogen peroxide on skeletal muscle arteriolar tone via activation of endothelial and smooth muscle signaling pathways. J. Appl. Physiol. 2004, 97, 1130–1137. [Google Scholar] [CrossRef]
- Kanner, J. Polyphenols by generating H2O2, affect cell redox signaling, inhibit PTPs and activate Nrf2 axis for adaptation and cell surviving: In vitro, In vivo and human health. Antioxidants 2020, 9, 797. [Google Scholar] [CrossRef] [PubMed]
- Erdei, N.; Bagi, Z.; Edes, I.; Kaley, G.; Koller, A. H2O2 increases production of constrictor prostaglandins in smooth muscle leading to enhanced arteriolar tone in type 2 diabetic mice. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H649–H656. [Google Scholar] [CrossRef][Green Version]
- Makino, A.; Skelton, M.M.; Zou, A.P.; Cowley, A.W. Increased medullary H2O2 leads to hypertension. Hypertnesion 2003, 42, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Ozbek, E. Induction of Oxidative Stress in Kidney. Int. J. Nephrol. 2012, 2012, 465897. [Google Scholar] [CrossRef] [PubMed]
- Azadmanesh, J.; Borgstahl, G.E.O. A Review of the catalytic mechanism of human manganese superoxide dismutase. Antioxidants 2018, 25, 25. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, C.; Lv, L.; Li, P.; Ma, C.; He, S.; Zeng, J.; Ping, F.; Huabing, Z.; Li, W. Relationship between Decreased Serum Superoxide Dismutase Activity and Metabolic Syndrome Mediating Role of Insulin Resistance and B-Cell Dysfunction. Oxid. Med. Cell Longev. 2020, 2020, 5384909. [Google Scholar] [CrossRef]
- Jung, O.S.; Marklund, L.; Geiger, H.; Pedrazzini, T.; Busse, R.; Brandes, R.P. Extracellular superoxide dismutase is a major determinant of nitric oxide bioavailability: In vivo and ex vivo evidence from ecSOD-deficient mice. Cir. Res. 2003, 93, 622–629. [Google Scholar] [CrossRef]
- Adler, S.; Huang, H. Oxidant stress in kidneys of spontaneously hypertensive rats involves both oxidase overexpression and loss of extracellular superoxide dismutase. Am. J. Physiol. Renal. Physiol. 2004, 287, F907–F913. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Castrejón-Téllez, V.; Soto, M.E.; Rubio-Ruiz, M.E.; Manzano-Pech, L.; Guarner-Lans, V. Oxidative stress, plant natural antioxidants, and obesity. Int. J. Mol. Sci. 2021, 22, 1786. [Google Scholar] [CrossRef]
- Soto, M.E.; Zuñiga-Muñoz, A.; Guarner-Lans, V.; Duran-Hernández, E.J.; Pérez-Torres, I. Infusion of Hibiscus sabdariffa L. Modulates oxidative stress in patients with marfan syndrome. Mediators. Inflamm. 2016, 2016, 8625203. [Google Scholar] [CrossRef]
- Prakash, P.A.; Yogeswaran, U.; Chen, S.M. A review on direct electrochemistry of catalase for electrochemical sensors. Sensors 2009, 9, 1821–1844. [Google Scholar] [CrossRef] [PubMed]
- Mirochnitchenko, O.; Weisbrot-Lefkowitz, M.; Reuhl, K.; Chen, L.; Yang, C.; Inouye, M. Acetaminophen toxicity. Opposite effects of two forms of glutathione peroxidase. J. Biol. Chem. 1999, 274, 10349–10355. [Google Scholar] [CrossRef] [PubMed]
- Whitin, J.C.; Bhamre, S.; Tham, D.M.; Harvey, J.C. Extracellular glutathione peroxidase is secreted basolaterally by cells of the human renal proximal tubule. J. Physiol. Renal. Physiol. 2002, 283, F20–F28. [Google Scholar] [CrossRef]
- Shu, W.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar]
- Curtis, J.M.; Grimsrud, P.A.; Wright, W.S.; Xu, X.; Foncea, R.E.; Graham, D.W.; Brestoff, J.R.; Wiczer, B.M.; Ilkayeva, O.; Cianflone, K.; et al. Downregulation of adipose glutathione S-transferase A4 leads to increased protein carbonylation, oxidative stress, and mito-chondrial dysfunction. Diabetes 2010, 59, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- Zúñiga-Muñoz, A.M.; Pérez-Torres, I.; Guarner-Lans, V.; Núñez-Garrido, E.; Velázquez-Espejel, R.; Huesca-Gómez, C.; Gamboa-Ávila, R.; Soto, M.E. Glutathione system participation in thoracic aneurysms from patients with Marfan syndrome. Vasa 2017, 46, 177–186. [Google Scholar] [CrossRef]
- Rybka, J.; Kupczyk, D.; Kędziora-Kornatowska, K.; Motyl, J.; Czuczejko, J.; Szewczyk-Golec, K.; Kozakiewicz, M.; Pawluk, H.; Carvalho, L.A.; Kędziora, J. Glutathione- related antioxidant defense system in elderly patients treated for hypertension. Cardiovasc. Toxicol. 2011, 11, 1–9. [Google Scholar] [CrossRef]
- Vávrová, L.; Kodydková, J.; Zeman, M.; Dusejovska, M.; Macasek, J.; Stankova, B.; Tvrzicka, E.; Zak, A. Altered activities of antioxidant enzymes in patients with metabolic syndrome. Obes. Facts 2013, 6, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Aisa-Alvarez, A.; Soto, M.E.; Guarner-Lans, V.; Camarena-Alejo, G.; Franco-Granillo, J.; Martínez-Rodríguez, E.A.; Gamboa-Ávila, R.; Manzano-Pech, L.; Pérez-Torres, I. Usefulness of antioxidants as adjuvant therapy for septic shock: A randomized clinical trial. Medicina (Kaunas) 2020, 56, 619. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Zúñiga-Muñoz, A.; Beltrán-Rodríguez, U.; Díaz-Díaz, E.; Martínez-Memije, R.; Guarner-Lans, V. Modification of the liver fatty acids by Hibiscus sabdariffa Linnaeus (Malvaceae) infusion, its possible effect on vascular reactivity in a metabolic syndrome model. Clin. Exp. Hypertens. 2014, 36, 123–131. [Google Scholar] [CrossRef]
- Jagota, S.K.; Dani, H.M. A new colorimetric technique for the estimation of vitamin C using Folin phenol reagent. Anal. Biochem. 1982, 127, 178–182. [Google Scholar] [CrossRef]
- Jia, Z.S.; Tang, M.C.; Wu, J.M. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative Study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- BenGershôm, E. Screening for albuminuria: A case for estimation of albumin in urine. Clin. Chem. 1975, 21, 1795–1798. [Google Scholar] [CrossRef]
- Jaffe, M. Ueber den niederschlag welchen pikrinsäure in normalen harn erzeugt und über eine neue reaction des kreatinins. Z. Physiol. Chem. 1886, 10, 391–400. (In Polish) [Google Scholar]
- Pérez-Torres, I.; El Hafidi, M.; Pavón, N.; Infante, O.; Avila-Casado, M.C.; Baños, G. Effect of gonadectomy on the metabolism of arachidonic acid in isolated kidney of a rat model of metabolic syndrome. Metabolism 2010, 59, 414–423. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Roque, P.; El Hafidi, M.; Diaz-Diaz, E.; Baños, G. Association of renal damage and oxidative stress in a rat model of metabolic syndrome. Influence of gender. Free Rad. Res. 2009, 43, 761–771. [Google Scholar] [CrossRef]



| Variables | Control | MS | MS + HSL |
|---|---|---|---|
| Glucose (mmol/L) | 114.7 ± 6.8 | 113.5 ± 5.0 | 118.5 ± 4.9 |
| Insulin (μU/mL) | 5.3 ± 0.5 | 17.6 ± 1.2 ** | 11.6 ± 1.1 * |
| HOMA index | 2.0 ± 0.1 | 9.9 ± 1.8 ** | 4.2 ± 0.3 * |
| Triglycerides (mg/dL) | 52.5 ± 4.6 | 139.5 ± 25.6 ** | 74.1 ± 5.9 * |
| Cholesterol (mg/dL) | 46.1 ± 2.1 | 51.5 ± 1.7 | 51.2 ± 0.8 |
| Intra-abdominal fat (g) | 3.1 ± 0.3 | 11.6 ± 1.3 ** | 6.1 ± 0.4 * |
| Systolic blood pressure (mmHg) | 119.1 ± 4.9 | 140.1 ± 6.9 ** | 132.2 ± 4.4 * |
| Leptin (ng/mL) | 1.9 ± 0.5 | 13.5 ± 7.8 ** | 3.6 ± 0.8 * |
| Body mass (g) | 397.8 ± 26.6 | 556.8 ± 14.1 * | 425.0 ± 27.6 * |
| Variables | Control | MS | MS + HSL |
|---|---|---|---|
| Drinking wáter (mL/24 h) | 18.12 ± 2.30 | 58.75 ± 5.06 ** | 35.00 ± 5.08 § |
| Weight of the right kidney (g) | 1.27 ± 0.03 | 1.51 ± 0.08 † | 1.30 ± 0.04 * |
| Urine (mL/24 h) | 13.62 ± 2.53 | 24.00 ± 4.20 * | 27.00 ± 3.33 |
| UCr (mg/dL) | 12.32 ± 1.29 | 3.95 ± 0.36 ** | 6.76 ± 1.19 |
| SCr (mg/dL) | 0.41 ± 0.02 | 0.67 ± 0.03 ** | 0.40 ± 0.02 § |
| CCr (mL/min) | 2.51 ± 0.35 | 1.45 ± 0.15 ** | 2.79 ± 0.55 * |
| Albuminuria (mg/mL) | 32.28 ± 5.54 | 74.67 ± 8.57 ** | 43.08 ± 7.54 § |
| Variables (mg of Protein) | Control | MS | MS + HSL |
|---|---|---|---|
| LPO (MDA nmol) | 0.34 ± 0.05 | 0.56 ± 0.03 *** | 0.45 ± 0.05 †† |
| Carbonylation (ng carbonyls) | 0.07 ± 0.00 | 0.11 ± 0.01 *** | 0.08 ± 0.01 |
| NO3− and NO2− (nM) | 0.09 ± 0.00 | 0.06 ± 0.01 † | 0.10 ± 0.00 § |
| GSH (μmol) | 0.0014 ± 0.0001 | 0.0009 ± 0.0001 † | 0.0013 ± 0.0000 ** |
| TAC (Trolox nmol) | 0.61 ± 0.01 | 0.57 ± 0.01 † | 0.60 ± 0.01 |
| Vit C (μM) | 0.021 ± 0.002 | 0.015 ± 0.001 * | 0.018 ± 0.000 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Fierros, F.L.; Guarner-Lans, V.; Soto, M.E.; Manzano-Pech, L.; Díaz-Díaz, E.; Soria-Castro, E.; Rubio-Ruiz, M.E.; Jiménez-Trejo, F.; Pérez-Torres, I. Modulation of Renal Function in a Metabolic Syndrome Rat Model by Antioxidants in Hibiscus sabdariffa L. Molecules 2021, 26, 2074. https://doi.org/10.3390/molecules26072074
Rodríguez-Fierros FL, Guarner-Lans V, Soto ME, Manzano-Pech L, Díaz-Díaz E, Soria-Castro E, Rubio-Ruiz ME, Jiménez-Trejo F, Pérez-Torres I. Modulation of Renal Function in a Metabolic Syndrome Rat Model by Antioxidants in Hibiscus sabdariffa L. Molecules. 2021; 26(7):2074. https://doi.org/10.3390/molecules26072074
Chicago/Turabian StyleRodríguez-Fierros, Félix Leao, Verónica Guarner-Lans, María Elena Soto, Linaloe Manzano-Pech, Eulises Díaz-Díaz, Elizabeth Soria-Castro, María Esther Rubio-Ruiz, Francisco Jiménez-Trejo, and Israel Pérez-Torres. 2021. "Modulation of Renal Function in a Metabolic Syndrome Rat Model by Antioxidants in Hibiscus sabdariffa L." Molecules 26, no. 7: 2074. https://doi.org/10.3390/molecules26072074
APA StyleRodríguez-Fierros, F. L., Guarner-Lans, V., Soto, M. E., Manzano-Pech, L., Díaz-Díaz, E., Soria-Castro, E., Rubio-Ruiz, M. E., Jiménez-Trejo, F., & Pérez-Torres, I. (2021). Modulation of Renal Function in a Metabolic Syndrome Rat Model by Antioxidants in Hibiscus sabdariffa L. Molecules, 26(7), 2074. https://doi.org/10.3390/molecules26072074