Thermal and Emission Properties of a Series of Lanthanides Complexes with N-Biphenyl-Alkylated-4-Pyridone Ligands: Crystal Structure of a Terbium Complex with N-Benzyl-4-Pyridone
Abstract
1. Introduction
2. Results and Discussion
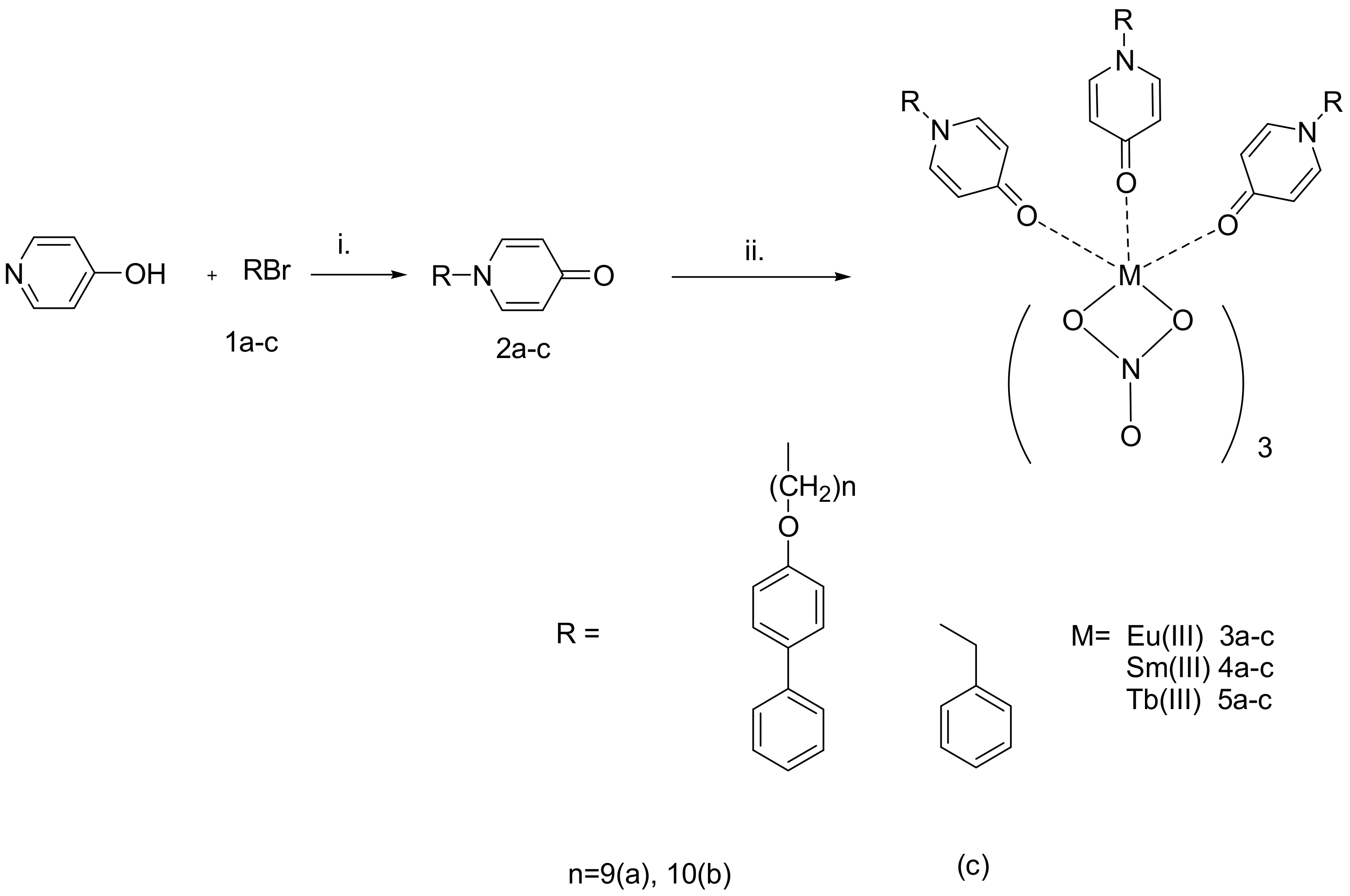
2.1. Synthesis and Characterization of the Organic Ligands and the Lanthanide Complexes
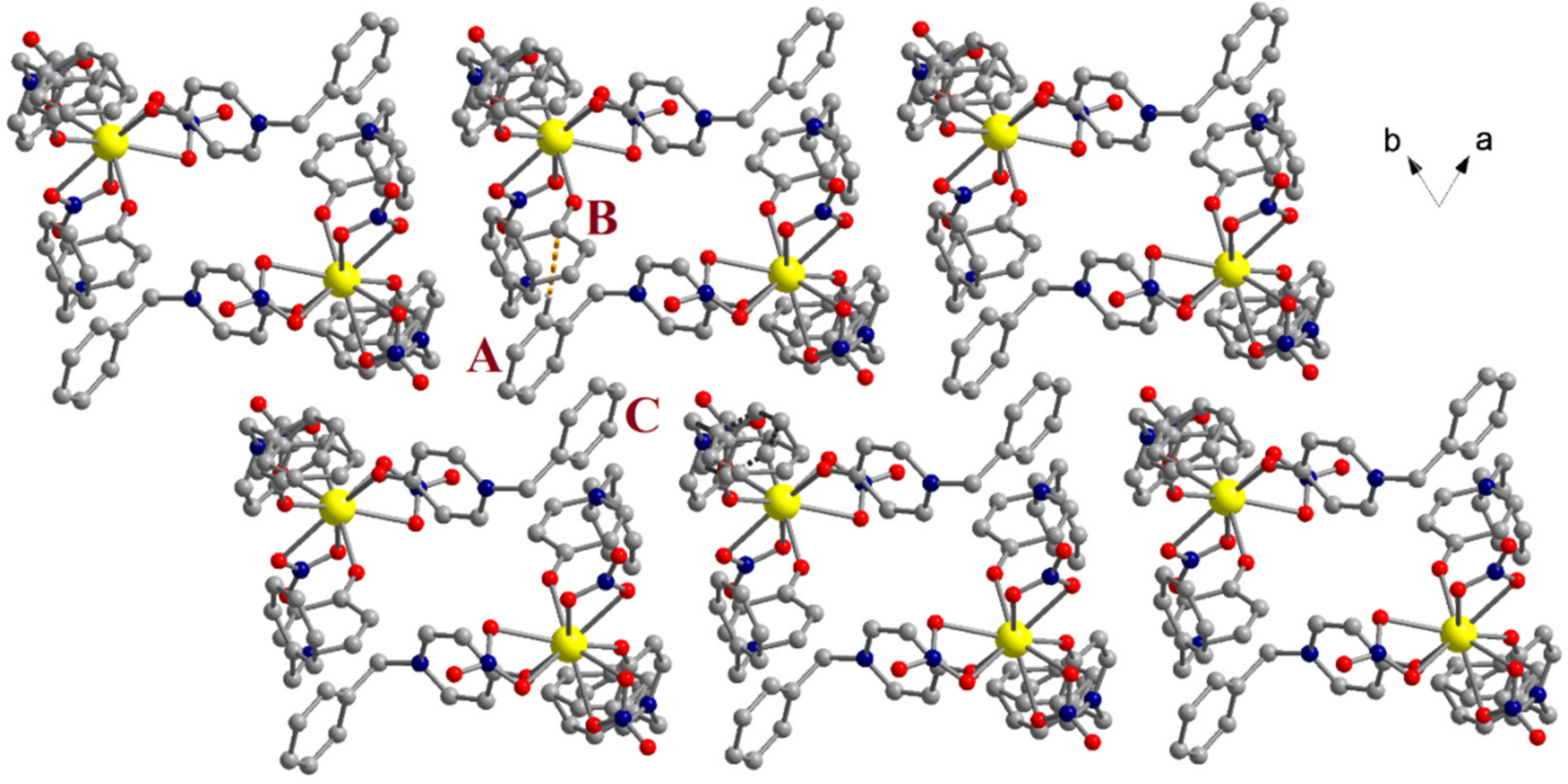
2.2. Crystal Structure of 5c
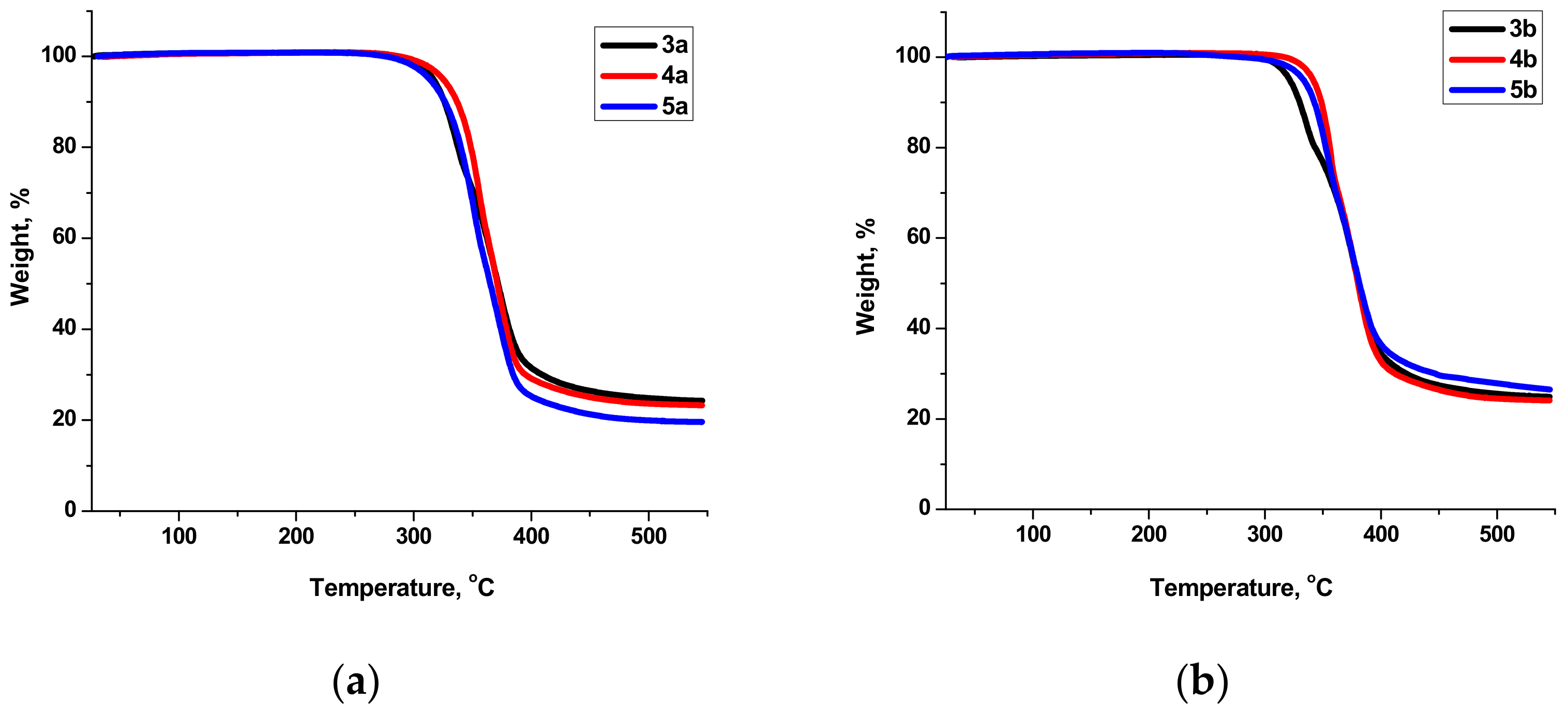
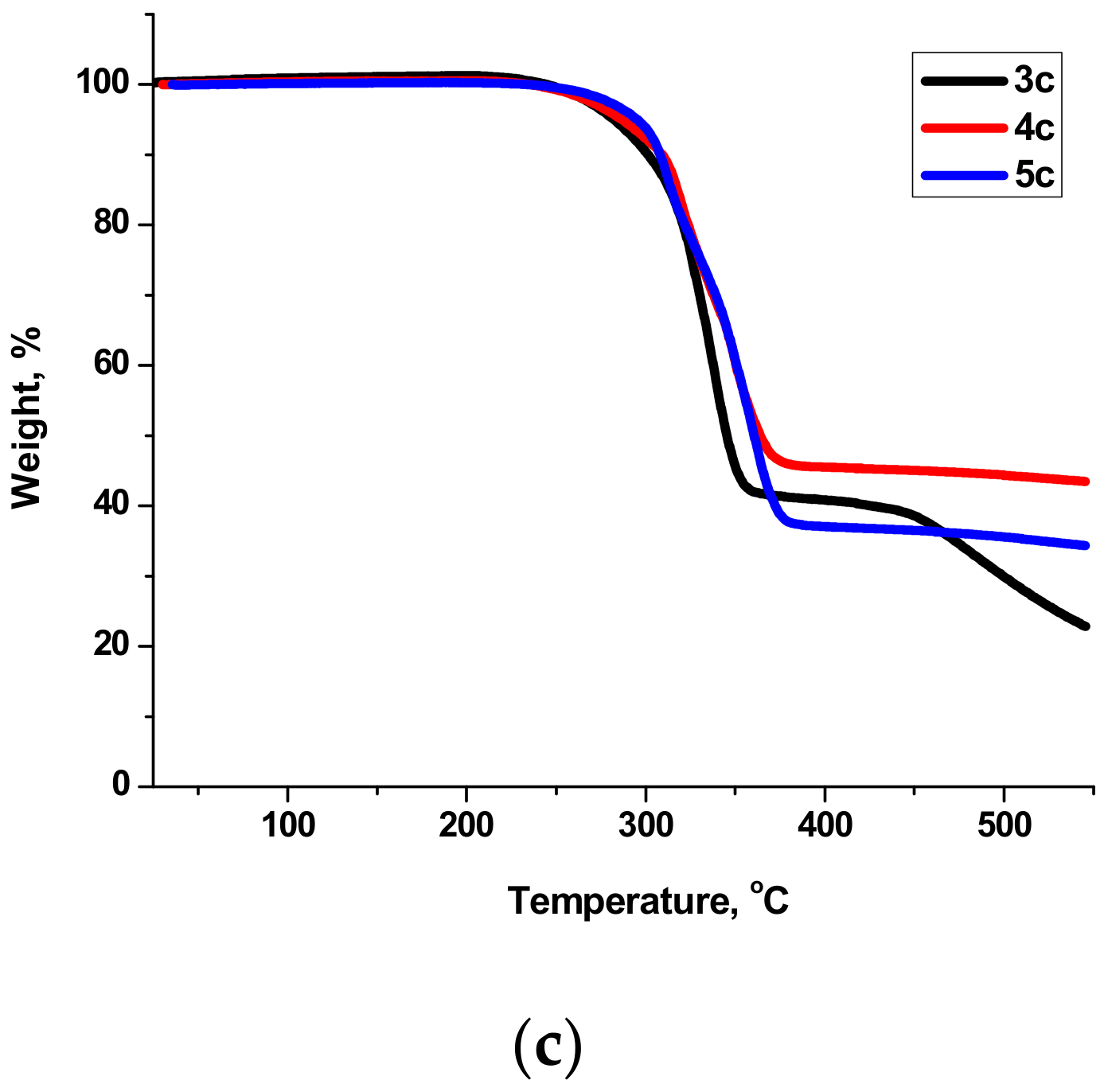
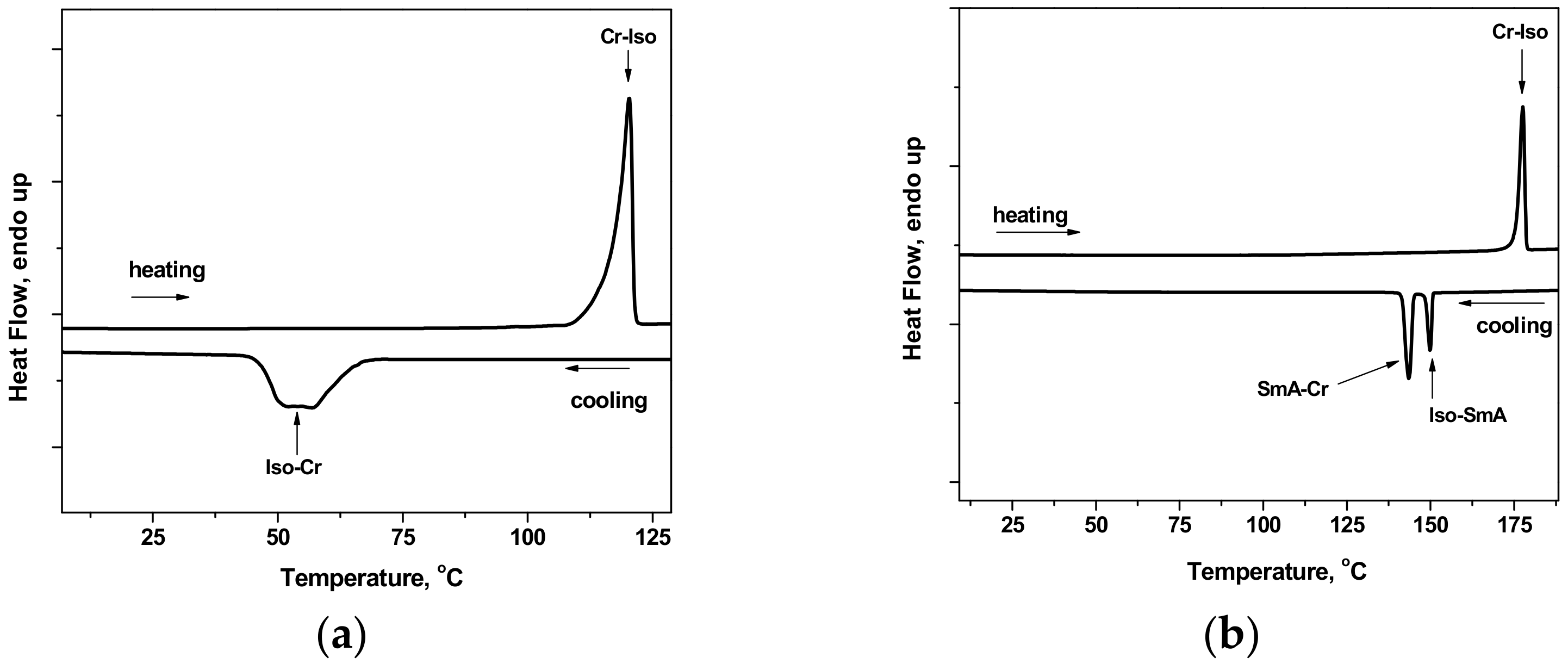
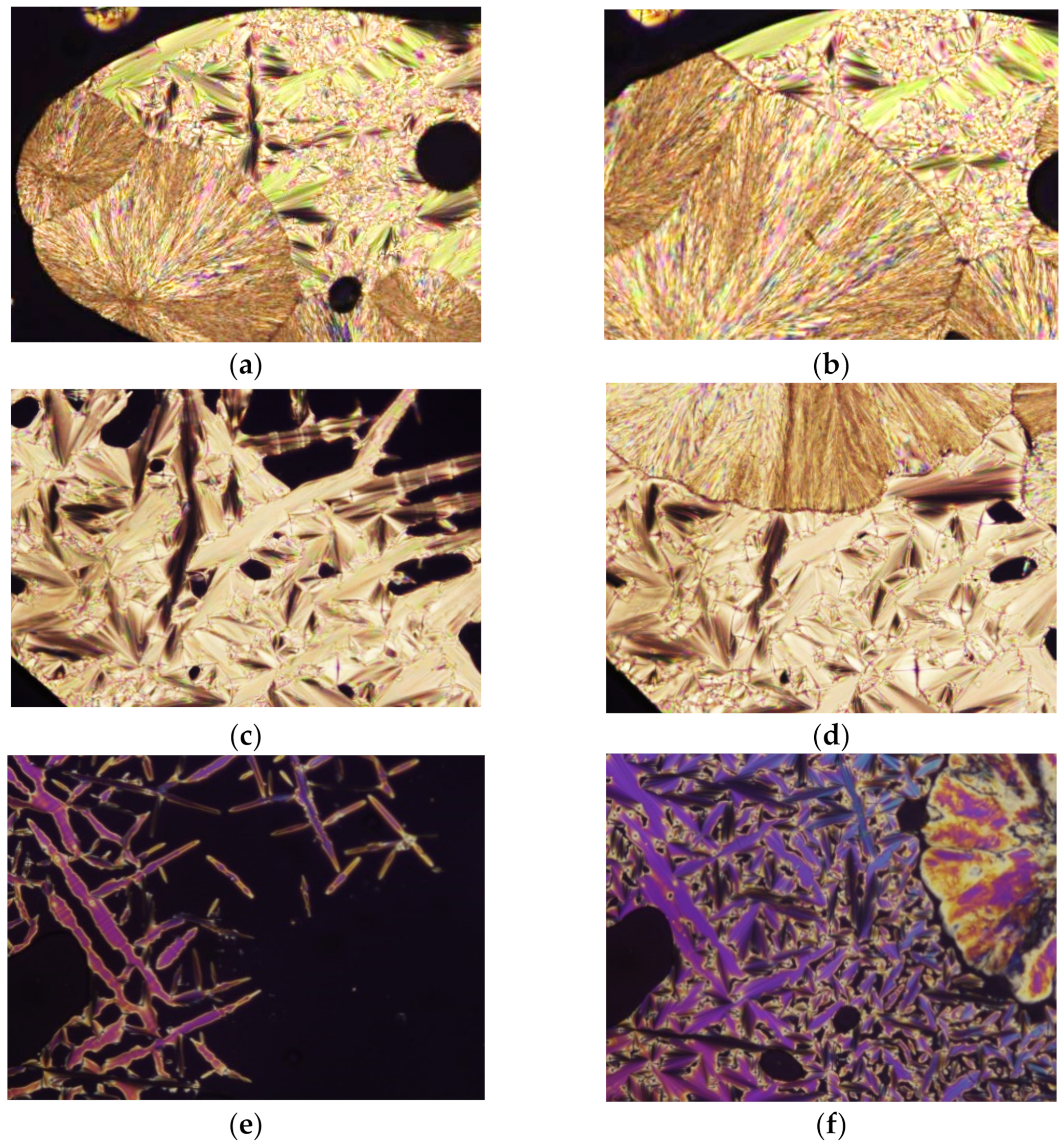
2.3. Thermal Behavior and Liquid Crystals Properties
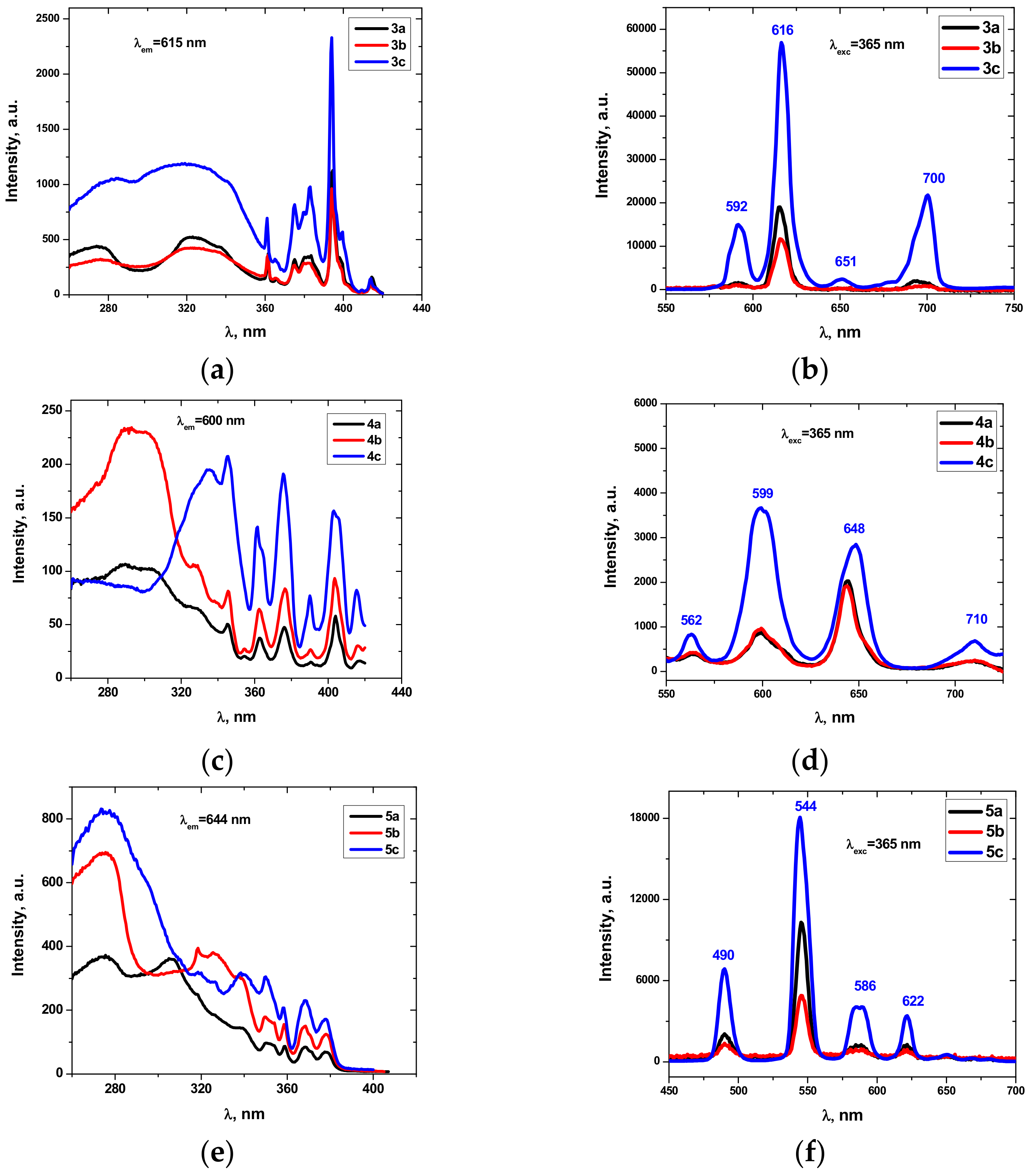
2.4. Emission Properties
3. Materials and Methods
3.1. Synthesis of Intermediate Bromide Derivatives 1a,b
3.2. Synthesis of 4-Pyridone Ligands 2a–c
3.3. Synthesis and Analytical Data of Eu(III) Complexes 3a,b
3.4. Synthesis and Analytical Data of Sm(III) Complexes 4a–c
3.5. Synthesis and Analytical Data of Tb(III) Complexes 5a–c
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Sample Availability
References
- Rajendiran, K.; Yoganandham, S.T.; Arumugam, S.; Arumugam, D.; Thananjeyan, K. An overview of liquid crytalline mesophase transition and photophysical properties of “f block”, “d block” and (SCO) spin-crossover metallomesogens in the optoelectronics. J. Mol. Liq. 2021, 321, 114793. [Google Scholar] [CrossRef]
- Chakrabarty, R.; Dutta, A.; Roy, S.; Das, D.; Ledoux-Rak, I.; Mondal, P.; Prasad, S.K.; Rao, D.S.; Bhattacharjee, C.R. Multifuctional lanthanide complexes: Mesomorphism, photoluminescence and second order NLO property. ChemistrySelect 2018, 3, 8245–8251. [Google Scholar] [CrossRef]
- Pramanik, H.A.R.; Chanda, C.; Paul, P.C.; Bhattacharjee, C.R.; Prasad, S.K.; Rao, D.S. Novel tris-buffer based Schiff base bearing low flexible alkoxy arm and its lanthanide complexes: Mesomorphism and photoluminescence. J. Mol. Struct. 2019, 1180, 472–479. [Google Scholar] [CrossRef]
- Pramanik, H.A.R.; Das, G.; Bhattacharjee, C.R.; Paul, P.C.; Mondal, P.; Prasad, S.K.; Rao, D.S. Tunable emissive lanthanidomesogen derived from a room-temperature liquid-crystalline Schiff-base ligand. Chem. Eur. J. 2013, 19, 13151–13159. [Google Scholar] [CrossRef]
- Rao, N.V.; Choudhury, T.D.; Deb, R.; Paul, M.K.; Rao, T.R.; Francis, T.; Smalyukh, I.I. Fluorescent lanthanide complexes of Schiff base ligands possesing N-aryl moiety: Influence of chain length on crossover (calamitic to discotic) phase behaviour. Liq. Cryst. 2010, 37, 1393–1410. [Google Scholar] [CrossRef]
- Binnemans, K. Lanthanidomesogens. In Handbook on the Physics and Chemistry of Rare Earths; Bünzli, J.-C., Pecharsky, V.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 43, pp. 1–154. [Google Scholar]
- Guerra, S.; Dutronc, T.; Terazzi, E.; KBuchwalder, K.-L.; Guénée, L.; Deschenaux, R.; Eliseeva, S.V.; Petoud, S.; Piguet, C. Taking a last look at lanthanidomesogens? The use of basic thermodynamics from programming the temperature domains of existence of luminescent liquid crystals. Coord. Chem. Rev. 2017, 340, 79–97. [Google Scholar] [CrossRef]
- Knyazev, A.A.; Krupin, A.S.; Heinrich, B.; Donnio, B.; Galyametdinov, Y.G. Controlled polarized luminescence of smectic lanthanide complexes. Dyes Pigm. 2018, 148, 492–500. [Google Scholar] [CrossRef]
- Pană, A.; Chiriac, F.L.; Secu, M.; Pasuk, I.; Ferbinteanu, M.; Micutz, M.; Cîrcu, V. A new class of thermotropic lanthanidomesogensL Eu (III) nitrate complexes with mesogenic 4-pyridone ligands. Dalton Trans. 2015, 44, 14196–14199. [Google Scholar] [CrossRef]
- Chiriac, F.L.; Pasuk, I.; Secu, M.; Micutz, M. Wide-range columnar and lamellar photoluminescent liquid-crystalline lanthanide complexes with mesogenic 4-pyridone derivatives. Chem. Eur. J. 2018, 24, 13512–13522. [Google Scholar] [CrossRef]
- Chiriac, L.F.; Ganea, P.C.; Manaila-Maximean, D.; Pasuk, I.; Cîrcu, V. Synthesis and thermal, emission and dielectric properties of liquid crystalline Eu (III), Sm (III) and Tb (III) complexes based on mesogenic 4-pyridone ligands fuctionalized with cyanobiphenyl groups. J. Mol. Liq. 2019, 290, 111184. [Google Scholar] [CrossRef]
- Chiriac, L.F.; Ilis, M.; Cîrcu, V. Luminescent lanthanides complexes with mesogenic pyridone ligands: Emission and liquid crystals properties. Polyhedron 2020, 190, 114748. [Google Scholar] [CrossRef]
- Dyer, D.J.; Lee, V.Y.; Twieg, R.J. The forgotten liquid crystals of Daniel Vorlander: A new look at mesogenic 4-pyridones. Liq. Cryst. 1997, 23, 551–560. [Google Scholar] [CrossRef]
- You, F.; Twieg, R.J.; Dyer, D.; Lee, V. Synthesis and properties of liquid crystals containing the 4-pyridone and methylene-1,4-dihydropyridine structure unit. Mol. Cryst. Liq. Cryst. 1999, 332, 399–406. [Google Scholar] [CrossRef]
- Dyer, D.J.; Lee, V.Y.; Twieg, R.J. A new class of liquid crystals: Methylene-1,4-dihydripyridines. Liq. Cryst. 1998, 24, 271–281. [Google Scholar] [CrossRef]
- Wolf, J.R.; Dyer, D.J. Hydrogen boded liquid crystalline heterodimers incorporating alkoxystilbazoles and alkoxy-4pyridones. Liq. Cryst. Today 2015, 24, 47–55. [Google Scholar] [CrossRef]
- Yerrasani, R.; Karunakar, M.; Dubey, R.; Singh, A.K.; Nandi, R.; Singh, R.K.; Rao, T.R. Synthesis, characterization and photophysical studies of rare earth metal complexes with a mesogenic Schiff-base. J. Mol. Liq. 2016, 216, 510–515. [Google Scholar] [CrossRef]
- Sáncheza, I.; Cuerva, C.; Marcelo, M.; Oliveira, E.; Santos, H.M.; Campo, J.A.; Lodeiro, C.; Cano, M. Designing Eu-β-diketonate complexes as a support of ionic liquid crystals (ILCs) with additional luminescent properties. Dyes Pigm. 2018, 159, 395–405. [Google Scholar] [CrossRef]
- Li, C.; Robinson, P.D.; Dyer, D.J. A hydrogen-bonded heterodimer of 1-(4-hexyloxyphenyl)pyridin-4(1H)-one with 4-cyanobenzoic acid. Acta Cryst. C 2006, C62, o596–o598. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.D.; Li, C.; Wolf, J.R.; Dyer, D.J. 1-[4-(hexyloxy)pyridin-4(1H)-one monohydrate. Acta Cryst. 2005, E61, o1254–o1256. [Google Scholar] [CrossRef]
- Modec, B. Crystal chemistry of zinc quinaldinate complexes with pyridine-based ligands. Crystals 2018, 8, 52. [Google Scholar] [CrossRef]
- Monroe, J.C.; Turnbull, M.M. Unusual coordination behavior by a hydroxypyridine/pyridone ligand: Synthesis and structure of [(2-bromo4-hydroxypyridine)2(2-bromo-1(H)-4-pyridone)2copper(II)perchlorate • 2(2-bromo-4-hydroxypyridine) • 2(2-bromo-1(H)-4-pyridone). J. Coord. Chem. 2019, 72, 3210–3221. [Google Scholar] [CrossRef]
- Wang, J.; Chorazy, S.; Nakabayashi, K.; Sieklucka, B.; Ohkoshi, S.I. Achieving white light emission and incresed magnetic anisotropy by transition metal substitution in functional materials based on dinuclear DyIII(4-pyridone)[MIII(CN)6]3- (M= Co, Rh) molecules. J. Mater. Chem. C 2018, 6, 473–481. [Google Scholar] [CrossRef]
- Lu, J.T.; Lee, C.K.; Lin, I.J.B. Ionic liquid crystals derived from 4-hydroxypyridine. Soft Matter. 2011, 7, 3491–3501. [Google Scholar] [CrossRef]
- Pană, A.; Iliş, M.; Staicu, T.; Pasuk, I.; Cîrcu, V. Columnar bis (pyridinium) ionic liquid crystals derived from 4-hydroxypyridine: Synthesis, mesomorphism and emission properties. Liq. Cryst. 2016, 43, 381–392. [Google Scholar] [CrossRef]
- Pană, A.; Panait, A.L.; Cîrcu, V. Simple and double pyridinium salts with cyanobiphenyl groups as ionic liquids and ionic liquid crystals: Synthesis and investigation of thermal behavior. Res. Chem. Intermed. 2018, 44, 2025–2038. [Google Scholar] [CrossRef]
- Pană, A.; Pasuk, I.; Micutz, M.; Cîrcu, V. Nematic ionic liquid crystals based o pyridinium salts derived from 4-hydroxypyridine. CrystEngComm 2016, 18, 5066–5069. [Google Scholar] [CrossRef]
- Weng, Q.; Duan, L.; Chen, P.; Shi, D.; Gao, A.; Chen, X.; An, Z. Synthesis and mesomorphic properties of benzoxazole derivatives with lateral multifluoto subtituents. Liq. Cryst. 2019, 46, 59–66. [Google Scholar] [CrossRef]
- Kensuke, K.; Hideaki, H.; Yukiko, T.; Jun-ichi, H. Electronic conduction in biphenyl liquid crystals. Chem. Lett. 2006, 35, 1194–1195. [Google Scholar]
- Thaker, B.T.; Kanojiya, J.B. Synthesis, characterization and mesophase behavior of new liquid crystalline compounds having chalcone as a central linkage. Mol. Cryst. Liq. Cryst. 2011, 542, 84/[606]–98/[620]. [Google Scholar] [CrossRef]
- Jiang, W.; Takayuki, U.; Akira, O.; Jun-ichi, H. Charge carrier transport properties of biphenyl liquid cystals with a dimer structure. Chem. Lett. 2009, 38, 592–593. [Google Scholar]
- Mouquinho, A.; Saavedra, M.; Maiau, A.; Petrova, K.; Barros, M.T.; Figueirinhas, J.L.; Sotomayor, J. Films based on new methacrylate monomers: Synthesis, characterization and electro-optical properties. Mol. Cryst. Liq. Cryst. 2011, 542, 132/[654]–140/[662]. [Google Scholar] [CrossRef]
- Barros, M.T.; Mouquinho, A.; Petrova, K.; Saavedra, M.; Sotomayor, J. Fast synthesis employing a microwave assisted neat protocol of new monomers potentially useful for the preparation of PDLC films. Cent. Eur. J. Chem. 2011, 9, 557–566. [Google Scholar]
- Mocanu, A.S.; Amela-Cortes, M.; Molard, Y.; Cîrcu, V.; Cordier, S. Liquid crystals properties resulting from synergetic effects between non-mesogenic organic molecules and a one nanometre sized octahedral transition metal cluster. Chem. Commun. 2011, 47, 2056–2058. [Google Scholar] [CrossRef] [PubMed]
- Pană, A.; Badea, F.L.; Iliş, M.; Staicu, T.; Micutz, M.; Pasuk, I.; Cîrcu, V. Effect of counterion on the mesomorphic behavior and optical properties of columnar pyridinium ionic liquid crystals derived from 4-hydroxypyridine. J. Mol. Struct. 2015, 1083, 245–251. [Google Scholar] [CrossRef]
- You, F.; Twieg, R.J. Aromatic nucleophilic substitution with 4-hydroxypyridine. Tetrahedron Lett. 1999, 40, 8759–8762. [Google Scholar] [CrossRef]
- Johnson, B.L.; Kitahara, J.; Weakley, T.R.J.; Keana, J.F.W. Intramolecular [2+2] photocycloaddition of juxtaposed 4-pyridone moieties. Tetrahedron Lett. 1993, 34, 5555–5558. [Google Scholar] [CrossRef]
- Itahara, T. Synthesis and liquid crystalline properties of novel pyridine derivatives. J. Heterocycl. Chem. 2008, 45, 913–916. [Google Scholar] [CrossRef]
- Shao, W.; Wang, Y.; Yang, Z.-P.; Zhang, X.; You, S.-L. Efficient synthesis of N-alkylated 4-pyridones by copper-catalyzed intermolecular asymmetric propargylic amination. Chem. Asian J. 2018, 13, 1103–1107. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Jones, A. Infrared absorption of heteroaromatic and benzenoid six-membered monocyclic nuclei. Part X. Pyrirdones and pyridthiones. J. Chem. Soc. 1960, 2947–2953. [Google Scholar] [CrossRef]
- Lothian, A.; Platt, A.W.G. Synthesis and spectroscopy of lanthanide nitrate complexes of thiethyl phosphonoacetate. Polyhedron 1992, 23, 2983–2987. [Google Scholar] [CrossRef]
- Lees, A.M.J.; Kresinski, R.A.; Platt, A.W.G. Complexes of (EtO)2P(O)CH2P(O)(OEt)2 with lanthanide nitrates. New J. Chem. 2004, 28, 1457–1463. [Google Scholar] [CrossRef]
- Xu, X.; Ma, Y.-F.; Liu, W.-S.; Tang, Y.; Tan, M.-Y. Preparation, crystal structures and luminescent properties of terbium and europium complexes with a new amino-alkenone type ligand. J. Fluoresc. 2011, 21, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Law, G.-L.; Wong, K.-L.; Zhou, X.; Wong, W.-T.; Tanner, P.A. Crystal structure and luminescence of lanthanide monodentate complexes [Ln(C(4)N(4)H(6)O)(2)(H(2)O)(6)]Cl(3) and [Ln(C(4)N(4)H(6)O)(2)(H(2)O)(3)(NO(3))(3)] (Ln = Tb or Eu). Inorg. Chem. 2005, 44, 4142–4144. [Google Scholar] [CrossRef] [PubMed]
- Goodgame, D.M.L.; Lalia-Kantouri, M.; Williams, D.J. Crystal and molecular structures of the 4-(1H)-pyridone (= L) lanthanide complexes {[GdL(NO3)3(H2O)]·CH3OH}2 and [DyL4(NO3)2](NO3) and of L2HNO3. J. Crystallogr. Spectrosc. Res. 1993, 23, 373–380. [Google Scholar] [CrossRef]
- Anjaneyulu, O.; Prasad, T.K.; Kumara Swamy, K.C. Tris(4-hydroxypyridinium) nitrato lanthanide complexes [M(4-O-C6H4NH)3(NO3)2(H2O)2]]NO3] {M = La, Ce, Pr, Nd, Eu, Gd}- Synthesis and structural characterization. Inorg. Chim. Acta 2010, 363, 2990–2995. [Google Scholar] [CrossRef]
- Armelao, L.; Quici, S.; Barigelletti, F.; Accorsi, G.; Bottaro, G.; Cavazzini, M.; Tondello, E. Design of luminescent lanthanide complexes: From molecules to highly efficient photo-emitting materials. Coord. Chem. Rev. 2010, 254, 487–505. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, J.; Chen, J.; Zhu, W.; Baranoff, E. Recent progress in luminescent liquid crystal materials: Design, properties and application for linearly polarised emission. J. Mater. Chem. C 2015, 3, 7993–8005. [Google Scholar] [CrossRef]
- Wu, X.; Zhu, M.; Bruce, D.W.; Zhu, W.; Wang, Y. An overview pf phosphorescent matellomesogens based on platinum and iridium. J. Mater. Chem. C 2018, 6, 9848–9860. [Google Scholar] [CrossRef]
- Krupin, A.S.; Knyazev, A.A.; Galyametdinov, Y.G. Lanthanide-containing nematic phase with controlled polarized luminescence at room temperature. Liq. Cryst. Their Appl. 2018, 18, 15–23. [Google Scholar] [CrossRef]
- Knyazev, A.A.; Krupin, A.S.; Haase, W.; Galyametdinov, Y.G. Polarized Luminescence of alighned lanthanide containing nematic mixtures. Liq. Cryst. Their Appl. 2019, 19, 31–39. [Google Scholar] [CrossRef]

| Compound | 5c·2CH3CN |
|---|---|
| Chemical formula | C40H39N8O12Tb |
| M (g mol−1) | 982.71 |
| Temperature, (K) | 200(2) |
| Wavelength, (Å) | 0.71073 |
| Crystal system | Triclinic |
| Space group | P-1 |
| a (Å) | 13.1960(8) |
| b (Å) | 13.2839(8) |
| c (Å) | 14.5787(10) |
| α (°) | 78.722(5) |
| β (°) | 80.321(5) |
| γ (°) | 61.273(4) |
| V (Å3) | 2189.6(3) |
| Z | 2 |
| Dc (g cm-3) | 1.491 |
| µ (mm−1) | 1.684 |
| F(000) | 992 |
| Goodness-of-fit on F2 | 1.042 |
| Final R1, wR2 [I > 2σ(I)] | 0.0411, 0.1039 |
| R1, wR2 (all data) | 0.0461, 0.1067 |
| Largest diff. peak and hole (eÅ−3) | 2.217, −2.174 |
| Tb–O (4-pyridone) | Tb1–O1 = 2.284(3); Tb1–O2 = 2.262(3); Tb1–O3 = 2.272(3) |
| Tb–O (NO3) | Tb1–O4 = 2.557(3); Tb1–O5 = 2.455(3); Tb1–O7 = 2.573(3); Tb1–O8 = 2.465(3); Tb1–O10 = 2.560(3); Tb1–O11 = 2.450(3) |
| Compound | M | n | Transition | T (°C) | ΔH (kJ·mol−1) |
|---|---|---|---|---|---|
| 2a | - | 9 | Cr–Iso | 111 | 3.0 |
| 2b | - | 10 | Cr–Iso | 117 | 45.4 |
| 2c | - | - | Cr–Iso | 115 | 17.5 |
| 3a | Eu | 9 | Cr1–Cr2 | 135 | 34.3 |
| Cr2–Iso | 148 | 46.5 | |||
| 3b | Eu | 10 | Cr–Iso | 176 | 108.2 |
| Iso–SmA | 158 (c) | - | |||
| SmA–Cr | 150 | 104.2 | |||
| 3c | Eu | - | Cr–Iso | 210 | 88.6 |
| 4a | Sm | 9 | Cr1–Cr2 | 134 | 1.6 |
| Cr2–Iso | 145 | 70.6 | |||
| 4b | Sm | 10 | Cr–Iso | 176 | 111.0 |
| Iso–SmA | 151 | 29.5 | |||
| SmA–Cr | 145 | 67.6 | |||
| 4c | Sm | - | Cr–Iso | 217 | 76.7 |
| 5a | Tb | 9 | Cr1–Cr2 | 133 | 37.9 |
| Cr2–Iso | 140 | 42.2 | |||
| 5b | Tb | 10 | Cr–Iso | 173 | 92.5 |
| Iso–SmA | 155 (c) | - | |||
| SmA–Cr | 149 | 95.4 | |||
| 5c | Tb | - | Cr–Iso | 195 | 83.4 |
| Compound | M | τ (ms) |
|---|---|---|
| 3a | 0.716 ± 0.001 | |
| 3b | Eu | 0.719 ± 0.001 |
| 3c | 1.493 ± 0.003 | |
| 4a | 0.055 ± 0.001 | |
| 4b | Sm | 0.055 ± 0.001 |
| 4c | 0.071 ± 0.003 | |
| 5a | 0.807 ± 0.003 | |
| 5b | Tb | 0.624 ± 0.002 |
| 5c | 1.260 ± 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiriac, F.L.; Iliş, M.; Madalan, A.; Manaila-Maximean, D.; Secu, M.; Cîrcu, V. Thermal and Emission Properties of a Series of Lanthanides Complexes with N-Biphenyl-Alkylated-4-Pyridone Ligands: Crystal Structure of a Terbium Complex with N-Benzyl-4-Pyridone. Molecules 2021, 26, 2017. https://doi.org/10.3390/molecules26072017
Chiriac FL, Iliş M, Madalan A, Manaila-Maximean D, Secu M, Cîrcu V. Thermal and Emission Properties of a Series of Lanthanides Complexes with N-Biphenyl-Alkylated-4-Pyridone Ligands: Crystal Structure of a Terbium Complex with N-Benzyl-4-Pyridone. Molecules. 2021; 26(7):2017. https://doi.org/10.3390/molecules26072017
Chicago/Turabian StyleChiriac, Florentina L., Monica Iliş, Augustin Madalan, Doina Manaila-Maximean, Mihail Secu, and Viorel Cîrcu. 2021. "Thermal and Emission Properties of a Series of Lanthanides Complexes with N-Biphenyl-Alkylated-4-Pyridone Ligands: Crystal Structure of a Terbium Complex with N-Benzyl-4-Pyridone" Molecules 26, no. 7: 2017. https://doi.org/10.3390/molecules26072017
APA StyleChiriac, F. L., Iliş, M., Madalan, A., Manaila-Maximean, D., Secu, M., & Cîrcu, V. (2021). Thermal and Emission Properties of a Series of Lanthanides Complexes with N-Biphenyl-Alkylated-4-Pyridone Ligands: Crystal Structure of a Terbium Complex with N-Benzyl-4-Pyridone. Molecules, 26(7), 2017. https://doi.org/10.3390/molecules26072017











