Antioxidant Activity Evaluation of Oviductus Ranae Protein Hydrolyzed by Different Proteases
Abstract
1. Introduction
2. Results and Discussions
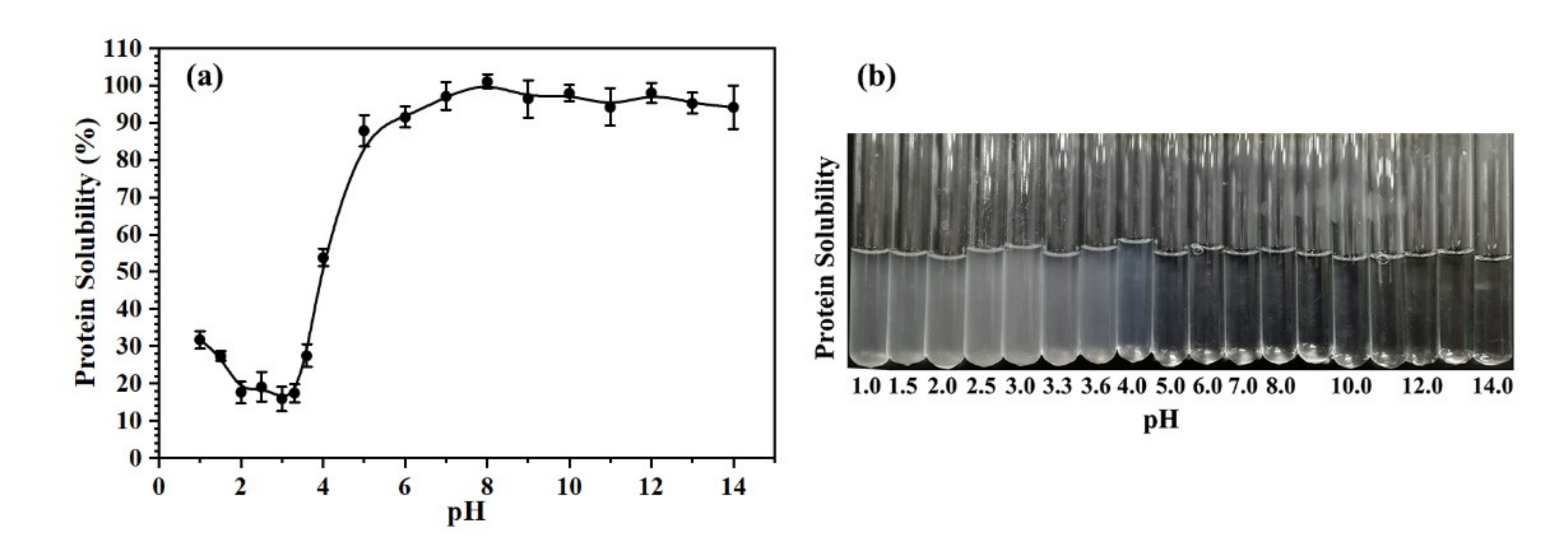
2.1. The Solubility of Oviductus Ranae Protein
2.2. DH of Oviductus Ranae Protein
2.3. 1,1-Diphenyl-2-picrylhydrazyl (DPPH) Radical Scavenging Activity
2.4. Reducing Power
2.5. Hydroxyl Radical Scavenging Activity
2.6. Comprehensive Antioxidant Activity Analysis
2.7. Correlation Analysis
3. Materials and Methods
3.1. Chemicals and Samples
3.2. Oviductus Ranae Protein Extraction
3.3. Determination of Solubility
3.4. Enzymatic Hydrolysis
3.5. Determination of DH
3.6. Determination of Antioxidant Activity
3.6.1. DPPH Radical Scavenging Activity
3.6.2. Determination of Reducing Power
3.6.3. Hydroxyl Radical Scavenging Activity
3.6.4. Comprehensive Analysis of Antioxidant Activity
3.7. Correlation Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. BioMed Res. Int. 2014, 2014, 608979. [Google Scholar] [CrossRef] [PubMed]
- Nemzer, B.; Chang, T.; Xie, Z.; Pietrzkowski, Z.; Reyes, T.; Ou, B. Decrease of free radical concentrations in humans following consumption of a high antioxidant capacity natural product. Food Sci. Nutr. 2014, 2, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, S.; Hashemy, S.I. MicroRNA-mediated redox regulation modulates therapy resistance in cancer cells: Clinical perspectives. Cell. Oncol. 2019, 42, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, D.B.; Barros, J.A.S.; Fernie, A.R.; Araújo, W.L. Eating away at ROS to regulate stomatal opening. Trends Plant Sci. 2020, 25, 220–223. [Google Scholar] [CrossRef]
- Sila, A.; Bougatef, A. Antioxidant peptides from marine by-products: Isolation, identification and application in food systems. A review. J. Funct. Foods 2016, 21, 10–26. [Google Scholar] [CrossRef]
- Harnedy, P.A.; O’Keeffe, M.B.; FitzGerald, R.J. Fractionation and identification of antioxidant peptides from an enzymatically hydrolysed Palmaria palmata protein isolate. Food Res. Int. 2017, 100, 416–422. [Google Scholar] [CrossRef]
- Han, Y.H.; Moon, H.J.; You, B.R.; Park, W.H. Propyl gallate inhibits the growth of calf pulmonary arterial endothelial cells via glutathione depletion. Toxicol. In Vitro 2010, 24, 1183–1189. [Google Scholar] [CrossRef]
- Kamemura, N.; Oyama, K.; Kanemaru, K.; Yokoigawa, K.; Oyama, Y. Diverse cellular actions of tert-butylhydroquinone, a food additive, on rat thymocytes. Toxicol. Res. 2017, 6, 922–929. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Khaneghah, A.M.; Tavakolpour, Y.; Asnaashari, M.; Mehr, H.M. Effects of ultrasound treatment, UV irradiation and Avishan-e-Denaei essential oil on oxidative stability of sunflower oil. J. Essent. Oil Bear. Plants. 2015, 18, 1083–1092. [Google Scholar] [CrossRef]
- Hogan, S.; Zhang, L.; Li, J.; Wang, H.; Zhou, K. Development of antioxidant rich peptides from milk protein by microbial proteases and analysis of their effects on lipid peroxidation in cooked beef. Food Chem. 2009, 117, 438–443. [Google Scholar] [CrossRef]
- Contreras, M.D.M.; Hernández-Ledesma, B.; Amigo, L.; Martín-Álvarez, P.J.; Recio, I. Production of antioxidant hydrolyzates from a whey protein concentrate with thermolysin: Optimization by response surface methodology. LWT Food Sci. Technol. 2011, 44, 9–15. [Google Scholar] [CrossRef]
- Wiriyaphan, C.; Chitsomboon, B.; Yongsawadigul, J. Antioxidant activity of protein hydrolysates derived from threadfin bream surimi byproducts. Food Chem. 2012, 132, 104–111. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, G.; Zhao, Y.; Qiu, Y.; Chi, C.; Wang, B. Identification and active evaluation of antioxidant peptides from protein hydrolysates of skipjack tuna (Katsuwonus pelamis) head. Antioxidants 2019, 8, 318. [Google Scholar] [CrossRef]
- Peñta-Ramos, E.A.; Xiong, Y.L. Antioxidant activity of soy protein hydrolysates in a liposomal system. J. Food Sci. 2002, 67, 2952–2956. [Google Scholar] [CrossRef]
- Xu, S.; Shen, Y.; Li, Y. Antioxidant activities of sorghum kafirin alcalase hydrolysates and membrane/gel filtrated fractions. Antioxidants 2019, 8, 131. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, R.; Meisel, H. Food-derived peptides with biological activity: From research to food applications. Curr. Opin. Biotechnol. 2007, 18, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, S.; Luo, Y.; Wang, Y.; Qu, X. Evaluation of the merits of the new method of Oviductus Ranae by HPLC-DAD. J. Liq. Chromatogr. Rel. Technol. 2015, 38, 1218–1222. [Google Scholar] [CrossRef]
- Gan, Y.; Xiao, Y.; Wang, S.; Guo, H.; Liu, M.; Wang, Z.; Wang, Y. Protein-based fingerprint analysis for the identification of Ranae Oviductus using RP-HPLC. Molecules 2019, 24, 1687. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Ni, S.; Wang, S.; Gan, Y.; Zhou, Y.; Guo, H.; Liu, M.; Wang, Z.; Wang, Y. Environmental influences on quality features of Oviductus Ranae in the Changbai Mountains. RSC Adv. 2019, 9, 36050–36057. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Hu, Y.; Zhang, L.; Wang, Z. Isolation and identification of two steroid compounds from Oviductus Ranae. Nat. Prod. Res. 2010, 24, 1518–1522. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, S.; Luo, Y.; Yang, H.; Hu, X.; Wang, Y.; Qu, X. Separation of steroidal constituents of Oviductus Ranae by one-step method high-speed counter-current chromatography. J. Liq. Chromatogr. Rel. Technol. 2015, 38, 1494–1498. [Google Scholar] [CrossRef]
- Wang, S.; Xu, Y.; Wang, Y.; Yang, H.; Lv, Z.; Jin, X.; Wang, Y. Simultaneous determination of six active components in Oviductus Ranae via quantitative analysis of multicomponents by single marker. J. Anal. Methods Chem. 2017, 2017, 9194847. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Gan, Y.; Liu, M.; Wang, S.; Ni, S.; Zhou, Y.; Xiao, Y.; Wang, Z.; Wang, Y. Quality evaluation of Oviductus Ranae based on PUFAs using HPLC fingerprint techniques combined with chemometric methods. Foods 2019, 8, 322. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Xu, D.; Zhang, J.; Wang, Z.; Wang, S.; Guo, H.; Zhang, K.; Li, Y.; Wang, Y. Rana chensinensis ovum oil based on CO2 supercritical fluid extraction: Response surface methodology optimization and unsaturated fatty acid ingredient analysis. Molecules 2020, 25, 4170. [Google Scholar] [CrossRef]
- Wang, S.; Gan, Y.; Kan, H.; Mao, X.; Wang, Y. Exploitation of HPLC analytical method for simultaneous determination of six principal unsaturated fatty acids in Oviductus Ranae based on quantitative analysis of multi-components by single-marker (QAMS). Molecules 2021, 26, 479. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Zhao, H.; Yu, B.; Cui, B. Comprehensively analysis of components in Oviductus Ranae. Sci. Technol. Food Ind. 2017, 38, 348–352. [Google Scholar]
- Xu, Y.; Wang, F.; Guo, H.; Wang, S.; Ni, S.; Zhou, Y.; Wang, Z.; Bao, H.; Wang, Y. Antitussive and anti-inflammatory dual-active agents developed from natural product lead compound 1-methylhydantoin. Molecules 2019, 24, 2355. [Google Scholar] [CrossRef]
- Lu, H.; Kong, D.; Wu, B.; Wang, S.; Wang, Y. Synthesis and evaluation of anti-inflammatory and antitussive activity of hydantion derivatives. Lett. Drug Des. Discov. 2012, 9, 638–642. [Google Scholar] [CrossRef]
- Klompong, V.; Benjakul, S.; Kantachote, D.; Shahidi, F. Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem. 2007, 102, 1317–1327. [Google Scholar] [CrossRef]
- Zheng, Z.; Li, J.; Li, J.; Sun, H.; Liu, Y. Physicochemical and antioxidative characteristics of black bean protein hydrolysates obtained from different enzymes. Food Hydrocoll. 2019, 97, 105222. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Novel antioxidants in food quality preservation and health promotion. Eur. J. Lipid Sci. Technol. 2010, 112, 930–940. [Google Scholar] [CrossRef]
- Tang, J. Chapter 3—Pepsin A. In Handbook of Proteolytic Enzymes, 3rd ed.; Rawlings, N.D., Salvesen, G., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 27–35. [Google Scholar]
- Storer, A.C.; Ménard, R. Chapter 419—Papain. In Handbook of Proteolytic Enzymes, 3rd ed.; Rawlings, N.D., Salvesen, G., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 1858–1861. [Google Scholar]
- Jamdar, S.N.; Rajalakshmi, V.; Pednekar, M.D.; Juan, F.; Yardi, V.; Sharma, A. Influence of degree of hydrolysis on functional properties, antioxidant activity and ACE inhibitory activity of peanut protein hydrolysate. Food Chem. 2010, 121, 178–184. [Google Scholar] [CrossRef]
- Shu, G.; Huang, J.; Bao, C.; Meng, J.; Chen, H.; Cao, J. Effect of different proteases on the degree of hydrolysis and angiotensin I-converting enzyme-inhibitory activity in goat and cow milk. Biomolecules 2018, 8, 101. [Google Scholar] [CrossRef]
- Thiansilakul, Y.; Benjakul, S.; Shahidi, F. Antioxidative activity of protein hydrolysate from round scad muscle using alcalase and flavourzyme. J. Food Biochem. 2007, 31, 266–287. [Google Scholar] [CrossRef]
- Wu, H.; Chen, H.; Shiau, C. Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasicus). Food Res. Int. 2003, 36, 949–957. [Google Scholar] [CrossRef]
- Ghanbari, R.; Zarei, M.; Ebrahimpour, A.; Abdul-Hamid, A.; Ismail, A.; Saari, N. Angiotensin-I converting enzyme (ACE) inhibitory and anti-oxidant activities of sea cucumber (Actinopyga lecanora) hydrolysates. Int. J. Mol. Sci. 2015, 16, 28870–28885. [Google Scholar] [CrossRef]
- Ambigaipalan, P.; Al-Khalifa, A.S.; Shahidi, F. Antioxidant and angiotensin I converting enzyme (ACE) inhibitory activities of date seed protein hydrolysates prepared using alcalase, flavourzyme and thermolysin. J. Funct. Foods 2015, 18, 1125–1137. [Google Scholar] [CrossRef]
- Bamdad, F.; Wu, J.; Chen, L. Effects of enzymatic hydrolysis on molecular structure and antioxidant activity of barley hordein. J. Cereal Sci. 2011, 54, 20–28. [Google Scholar] [CrossRef]
- Kim, S.S.; Kwon, S.R.; Lim, M.J.; Park, W. The Osteoclast Development in Patients with Rheumatoid Arthritis and the Influence of the Bisphosphonate on Its Development. J. Korean Rheum. Assoc. 2007, 14, 31–42. [Google Scholar] [CrossRef][Green Version]
- Luo, H.; Wang, B.; Li, Z.; Chi, C.; Zhang, Q.; He, G. Preparation and evaluation of antioxidant peptide from papain hydrolysate of Sphyrna lewini muscle protein. LWT Food Sci. Technol. 2013, 51, 281–288. [Google Scholar] [CrossRef]
- Saito, K.; Jin, D.; Ogawa, T.; Muramoto, K.; Hatakeyama, E.; Yasuhara, T.; Nokihara, K. Antioxidative Properties of Tripeptide Libraries Prepared by the Combinatorial Chemistry. J. Agric. Food Chem. 2003, 51, 3668–3674. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, J.; Chang, C.; Chen, J.; Cao, F.; Zhao, J.; Zheng, Y.; Zhu, J. Physicochemical and functional properties of proteins extracted from three microalgal species. Food Hydrocoll. 2019, 96, 510–517. [Google Scholar] [CrossRef]
- Jin, H.; Xu, H.; Li, Y.; Zhang, Q.; Xie, H. Preparation and evaluation of peptides with potential antioxidant activity by microwave assisted enzymatic hydrolysis of collagen from sea cucumber Acaudina mSolpadioides obtained from zhejiang province in China. Mar. Drugs 2019, 17, 169. [Google Scholar] [CrossRef]
- Martín-del-Campo, S.T.; Martínez-Basilio, P.C.; Sepúlveda-Álvarez, J.C.; Gutiérrez-Melchor, S.E.; Galindo-Peña, K.D.; Lara-Domínguez, A.K.; Cardador-Martínez, A. Production of antioxidant and ACEI peptides from cheese whey discarded from Mexican white cheese production. Antioxidants 2019, 8, 158. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.M.; Petersen, D.; Dambmann, C. Improved method for determining food protein degree of hydrolysis. J. Food Sci. 2001, 66, 642–646. [Google Scholar] [CrossRef]
- Xie, L.; Cheng, F.; Yuan, J.; Piao, M. Study on the htot value of foodborne protein. Amino Acids Biot. Resour. 2013, 35, 15–18. [Google Scholar]
- Karadag, A.; Ozcelik, B.; Saner, S. Review of methods to determine antioxidant capacities. Food Anal. Meth. 2009, 2, 41–60. [Google Scholar] [CrossRef]
- Moure, A.; Domínguez, H.; Parajó, J.C. Antioxidant properties of ultrafiltration-recovered soy protein fractions from industrial effluents and their hydrolysates. Process Biochem. 2006, 41, 447–456. [Google Scholar] [CrossRef]
- Pezeshk, S.; Ojagh, S.M.; Rezaei, M.; Shabanpour, B. Fractionation of protein hydrolysates of fish waste using membrane ultrafiltration: Investigation of antibacterial and antioxidant activities. Probiotics Antimicrob. Proteins 2019, 11, 1015–1022. [Google Scholar] [CrossRef]
- You, L.; Zhao, M.; Regenstein, J.M.; Ren, J. Changes in the antioxidant activity of loach (Misgurnus anguillicaudatus) protein hydrolysates during a simulated gastrointestinal digestion. Food Chem. 2010, 120, 810–816. [Google Scholar] [CrossRef]
- Wang, X.; Xing, R.; Chen, Z.; Yu, H.; Li, R.; Li, P. Effect and mechanism of mackerel (Pneumatophorus japonicus) peptides for anti-fatigue. Food Funct. 2014, 5, 2113–2119. [Google Scholar] [CrossRef] [PubMed]
- De Avellar, I.G.J.; Magalhães, M.M.M.; Silva, A.B.; Souza, L.L.; Leitão, A.C.; Hermes-Lima, M. Reevaluating the role of 1,10-phenanthroline in oxidative reactions involving ferrous ions and DNA damage. Biochim. Biophys. Acta Gen. Subj. 2004, 1675, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jiang, B.; Zhang, T.; Mu, W.; Liu, J. Antioxidant and free radical-scavenging activities of chickpea protein hydrolysate (CPH). Food Chem. 2008, 106, 444–450. [Google Scholar] [CrossRef]
- Franzese, M.; Iuliano, A. Correlation analysis. In Encyclopedia of Bioinformatics and Computational Biology; Ranganathan, S., Gribskov, M., Nakai, K., Schönbach, C., Eds.; Academic Press: Oxford, UK, 2019; pp. 706–721. [Google Scholar]



| Protease | Time | DH | DPPH | Reducing Power | OH | CAA | |
|---|---|---|---|---|---|---|---|
| Pepsin | Time | 1.000 | 0.831 ** | 0.864 ** | 0.900 ** | 0.679 * | 0.889 ** |
| DH | 0.831 ** | 1.000 | 0.952 ** | 0.945 ** | 0.934 ** | 0.973 ** | |
| Trypsin | Time | 1.000 | 0.892 ** | 0.717 ** | 0.954 ** | 0.772 ** | 0.903 ** |
| DH | 0.892 ** | 1.000 | 0.902 ** | 0.945 ** | 0.902 ** | 0.965 ** | |
| Papain | Time | 1.000 | 0.916 ** | 0.795 ** | 0.829 ** | 0.563 * | 0.809 ** |
| DH | 0.916 ** | 1.000 | 0.947 ** | 0.945 ** | 0.714 ** | 0.942 ** | |
| Flavourzyme | Time | 1.000 | 0.873 ** | 0.875 ** | 0.918 ** | 0.876 ** | 0.917 ** |
| DH | 0.873 ** | 1.000 | 0.976 ** | 0.902 ** | 0.936 ** | 0.946 ** | |
| Neutral protease | Time | 1.000 | 0.864 ** | 0.745 ** | 0.867 ** | 0.572 * | 0.815 ** |
| DH | 0.864 ** | 1.000 | 0.914 ** | 0.911 ** | 0.711 ** | 0.905 ** | |
| Alcalase | Time | 1.000 | 0.952 ** | 0.689 ** | 0.808 ** | 0.929 ** | 0.839 ** |
| DH | 0.952 ** | 1.000 | 0.854 ** | 0.919 ** | 0.988 ** | 0.949 ** |
| Protease | Origin | Optimal pH | Optimal Temperature (°C) |
|---|---|---|---|
| Pepsin | Porcine gastric mucosa | 2.0 | 37 |
| Trypsin | Porcine pancreas | 8.0 | 37 |
| Papain | Papaya plant | 6.5 | 55 |
| Flavourzyme | Aspergillus oryzae | 6.5 | 50 |
| Neutral protease | Bacillus subtilis | 7.0 | 55 |
| Alcalase | Bacillus licheniformis | 10.0 | 45 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Gan, Y.; Mao, X.; Kan, H.; Li, N.; Zhang, C.; Wang, Z.; Wang, Y. Antioxidant Activity Evaluation of Oviductus Ranae Protein Hydrolyzed by Different Proteases. Molecules 2021, 26, 1625. https://doi.org/10.3390/molecules26061625
Wang S, Gan Y, Mao X, Kan H, Li N, Zhang C, Wang Z, Wang Y. Antioxidant Activity Evaluation of Oviductus Ranae Protein Hydrolyzed by Different Proteases. Molecules. 2021; 26(6):1625. https://doi.org/10.3390/molecules26061625
Chicago/Turabian StyleWang, Shihan, Yuanshuai Gan, Xinxin Mao, Hong Kan, Nan Li, Changli Zhang, Zhihan Wang, and Yongsheng Wang. 2021. "Antioxidant Activity Evaluation of Oviductus Ranae Protein Hydrolyzed by Different Proteases" Molecules 26, no. 6: 1625. https://doi.org/10.3390/molecules26061625
APA StyleWang, S., Gan, Y., Mao, X., Kan, H., Li, N., Zhang, C., Wang, Z., & Wang, Y. (2021). Antioxidant Activity Evaluation of Oviductus Ranae Protein Hydrolyzed by Different Proteases. Molecules, 26(6), 1625. https://doi.org/10.3390/molecules26061625








