Volcanic Rock Materials for Defluoridation of Water in Fixed-Bed Column Systems
Abstract
1. Introduction
2. Results and Discussions
2.1. Characterization of Adsorbents
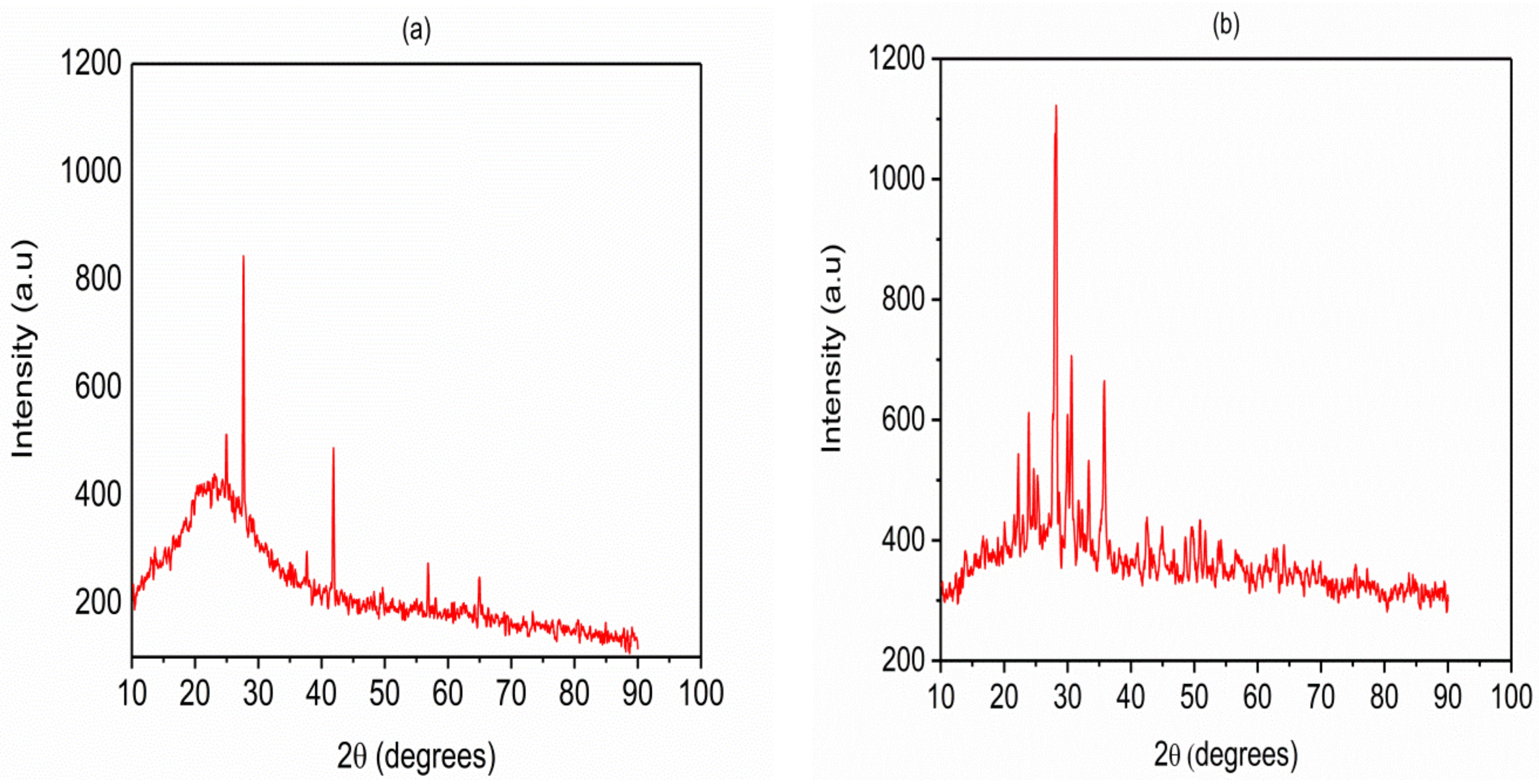
2.1.1. Crystalline Structures and Material Properties and Experimental Conditions
2.1.2. Chemical Composition
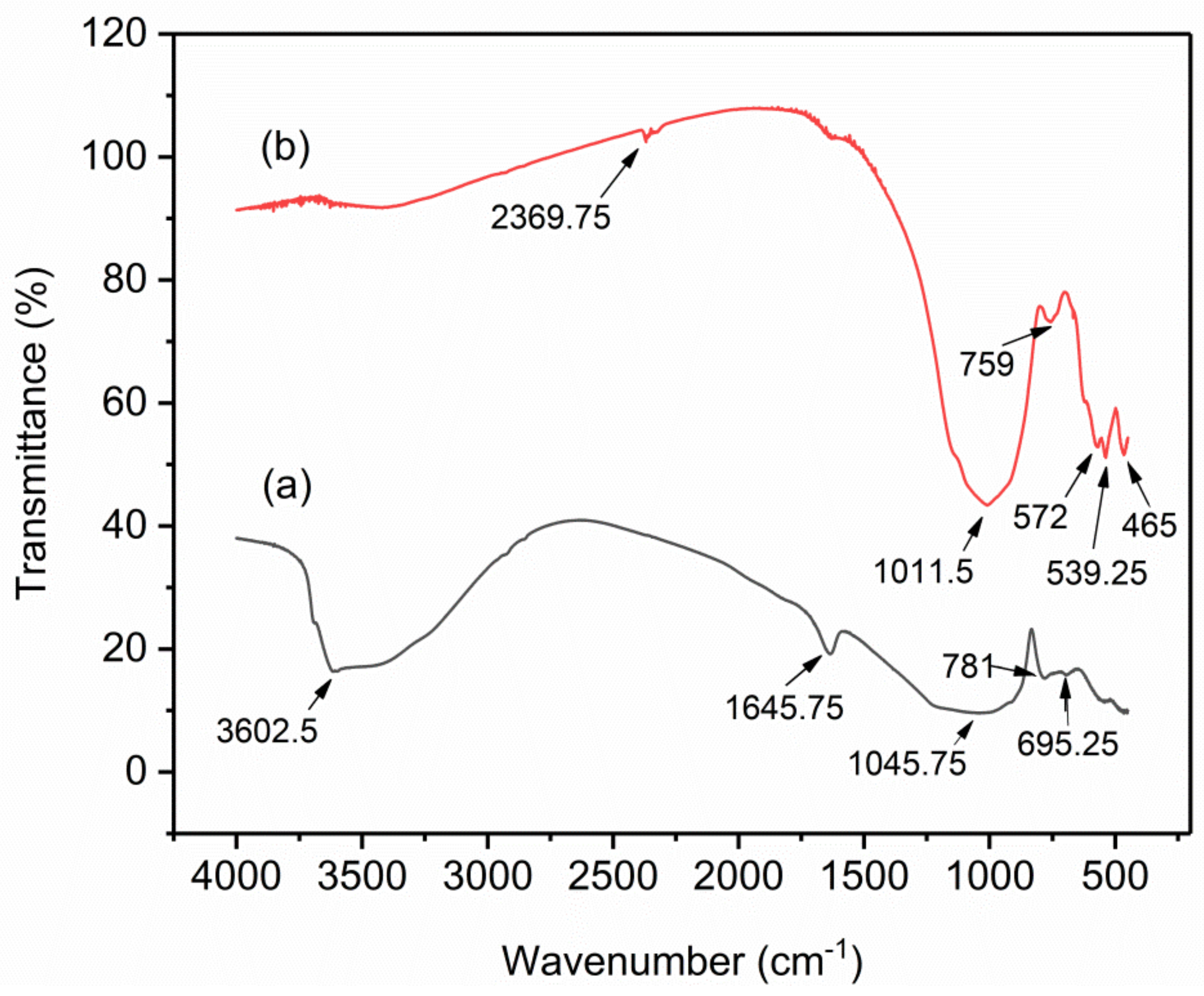
2.1.3. Fourier Transform Infrared (FTIR) Analysis
2.1.4. Scanning Electron Microscope (SEM) Analysis
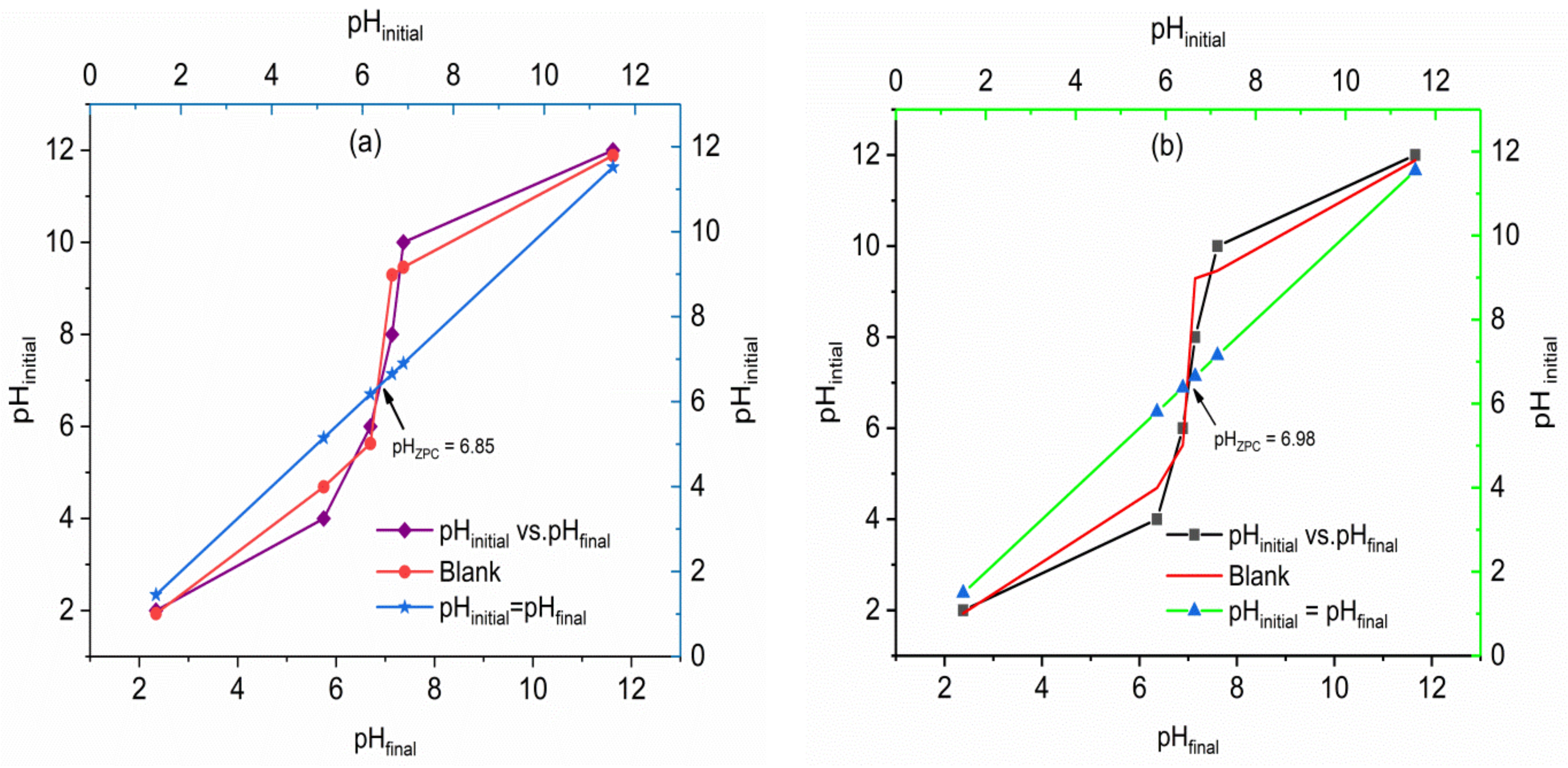
2.1.5. pH and Point of Zero Charges (pHPZC)
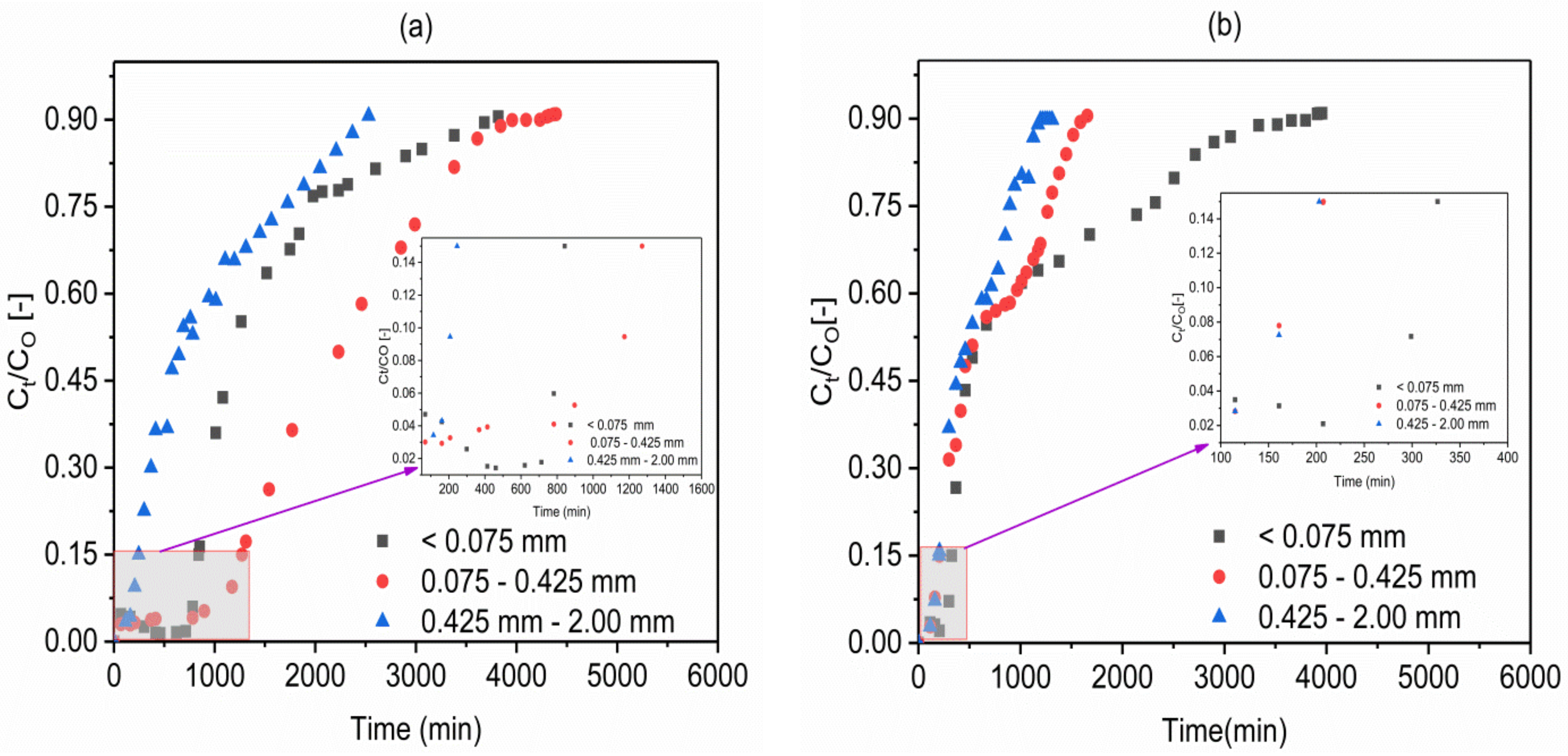
2.2. Effect of Adsorbents Particle Size
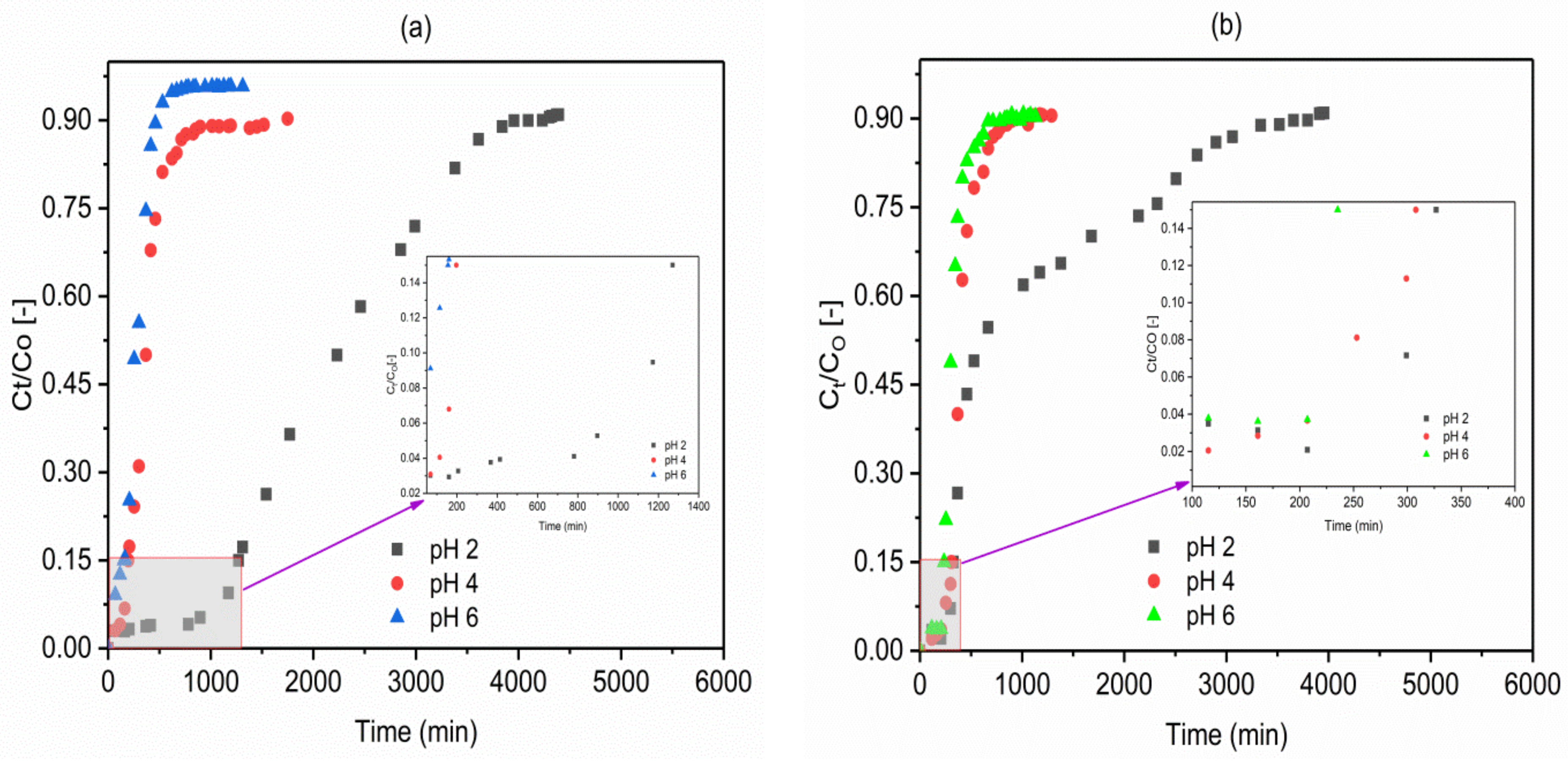
2.3. Effect of Influent pH
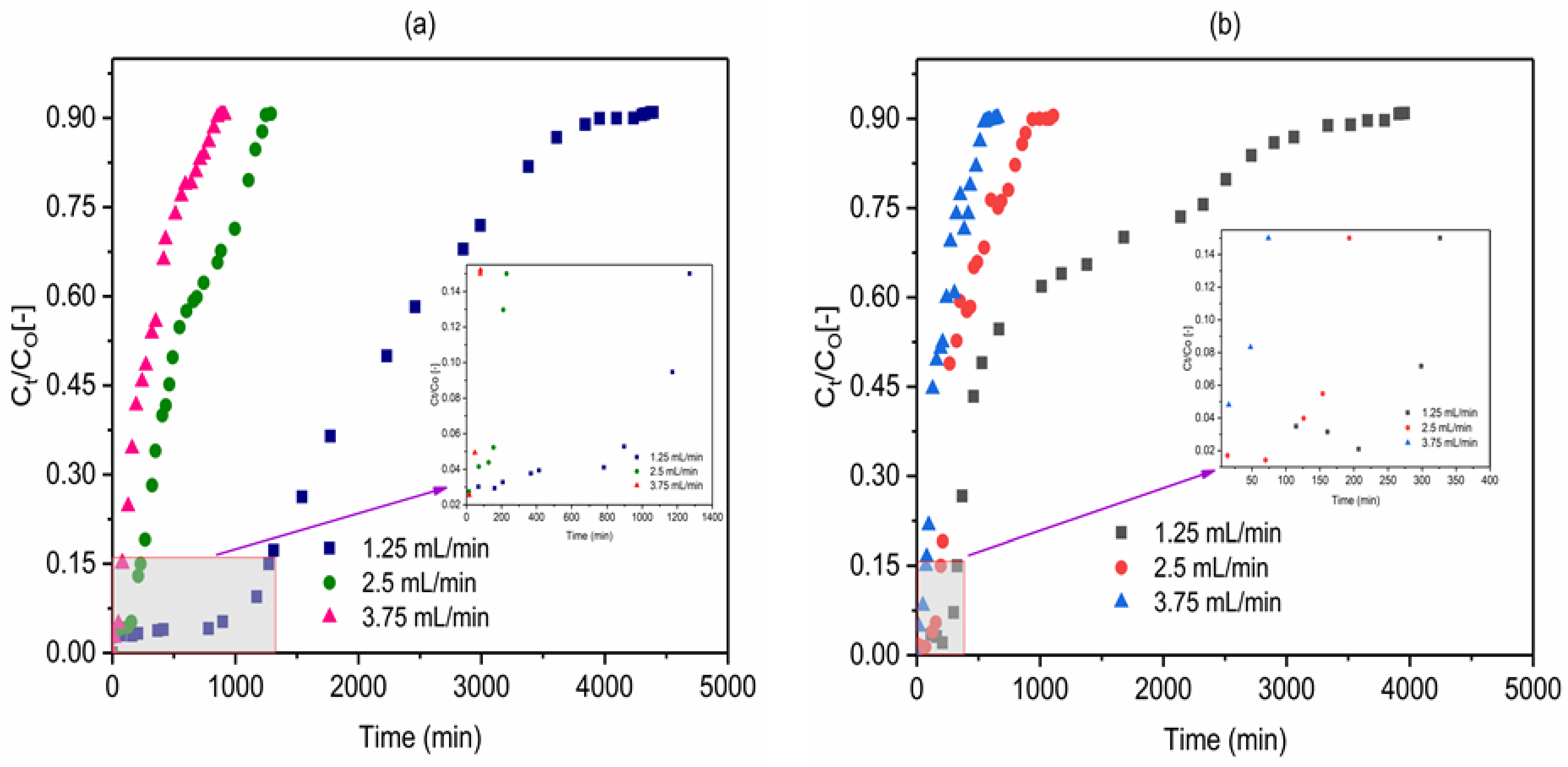
2.4. Effect of Flow Rate
2.5. Application of the Thomas Model
2.6. Application of the Adams–Bohart Model
2.7. Comparison of Different Adsorbents on Fluoride Removal
3. Materials and Methods
3.1. Materials
3.2. Preparations of Adsorbents
3.3. Preparations of Adsorbate
3.4. Adsorbent Characterizations
3.4.1. Crystalline Structures
3.4.2. Chemical Composition
3.4.3. Fourier Transform Infrared (FTIR) Analysis
3.4.4. Scanning Electron Microscope (SEM) Analysis
3.4.5. Determination of pH and Point of Zero Charges (pHPZC)
3.4.6. BET Analysis
3.5. Column Adsorption Experimental Set-Up and Procedures
3.6. Modeling and Analysis of Fixed-Bed Column Data
3.7. Fixed–Bed Column Breakthrough Curve Modeling
3.7.1. Thomas Model
3.7.2. Adams–Bohart Model
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Kumari, U.; Mishra, A.; Siddiqi, H.; Meikap, B.C. Effective Defluoridation of Industrial Wastewater by Using Acid Modified Alumina in Fixed-Bed Adsorption Column: Experimental and Breakthrough Curves Analysis. J. Clean. Prod. 2021, 279, 123645:1–123645:17. [Google Scholar] [CrossRef]
- Kumari, U.; Behera, S.K.; Siddiqi, H.; Meikap, B.C. Facile Method to Synthesize Efficient Adsorbent from Alumina by Nitric Acid Activation: Batch Scale Defluoridation, Kinetics, Isotherm Studies and Implementation on Industrial Wastewater Treatment. J. Hazard. Mater. 2020, 381, 120917. [Google Scholar] [CrossRef]
- Ye, Y.; Yang, J.; Jiang, W.; Kang, J.; Hu, Y.; Ngo, H.H.; Guo, W.; Liu, Y. Fluoride Removal from Water Using a Magnesia-Pullulan Composite in a Continuous Fixed-Bed Column. J. Environ. Manag. 2018, 206, 929–937. [Google Scholar] [CrossRef] [PubMed]
- WHO World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; WHO World Health Organization: Geneva, Swizerland, 2011; ISBN 978-92-4-154815-1. [Google Scholar]
- Rango, T.; Vengosh, A.; Jeuland, M.; Whitford, G.M.; Tekle-haimanot, R. Biomarkers of Chronic Fluoride Exposure in Groundwater in a Highly Exposed Population. Sci. Total Environ. 2017, 596–597, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fufa, F.; Alemayehu, E.; Lennartz, B. Defluoridation of Groundwater Using Termite Mound. Water. Air. Soil Pollut. 2013, 224, 1552:1–1552:15. [Google Scholar] [CrossRef]
- Rango, T.; Bianchini, G.; Beccaluva, L.; Tassinari, R. Geochemistry and Water Quality Assessment of Central Main Ethiopian Rift Natural Waters with Emphasis on Source and Occurrence of Fluoride and Arsenic. J. Afr. Earth Sci. 2010, 57, 479–491. [Google Scholar] [CrossRef]
- Demelash, H.; Beyene, A.; Abebe, Z.; Melese, A. Fluoride Concentration in Ground Water and Prevalence of Dental Fluorosis in Ethiopian Rift Valley: Systematic Review and Meta-Analysis. BMC Public Health 2019, 19, 1298. [Google Scholar] [CrossRef]
- Cai, J.; Zhang, Y.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q. Efficient Defluoridation of Water Using Reusable Nanocrystalline Layered Double Hydroxides Impregnated Polystyrene Anion Exchanger. Water Res. 2016, 102, 109–116. [Google Scholar] [CrossRef]
- CSAE Central Statistical Agency of Ethiopia. Drinking Water Quality in Ethiopia—Results from the 2016 Ethiopia Socioeconomic Survey; CSAE Central Statistical Agency of Ethiopia: Addis Ababa, Ethiopia, 2017.
- Kut, K.M.K.; Sarswat, A.; Srivastava, A.; Pittman, C.U., Jr.; Mohan, D. A Review of Fluoride in African Groundwater and Local Remediation Methods. Groundw. Sustain. Dev. 2016, 2–3, 190–212. [Google Scholar] [CrossRef]
- Shen, J.; Schäfer, A.I. Factors Affecting Fluoride and Natural Organic Matter (NOM) Removal from Natural Waters in Tanzania by Nano Filtration/Reverse Osmosis. Sci. Total Environ. 2015, 527–528, 520–529. [Google Scholar] [CrossRef]
- Smedley, P.L.; Nkotagu, H.; Pelig-Ba, K.; MacDonald, A.M.; TylerWhittle, R.; Whitehead, E.J.; Kinniburgh, D.G. Fluoride in Groundwater from High-Fluoride Areas of Ghana and Tanzania; Commissioned Report CR/02/316; British Geological Survey Groundwater Systems and Water Quality Programme: Nottingham, UK, 2002. [Google Scholar]
- Mohan, S.; Kumar, V.; Singh, D.K.; Hasan, S.H. Synthesis and Characterization of RGO/ZrO2 Nanocomposite for Enhanced Removal of Fluoride from Water: Kinetics, Isotherm, and Thermodynamic Modeling and Its Adsorption Mechanism. RSC Adv. 2016, 6, 87523–87538. [Google Scholar] [CrossRef]
- Araga, R.; Kali, S.; Sharma, C.S. Coconut-Shell-Derived Carbon/Carbon Nanotube Composite for Fluoride Adsorption from Aqueous Solution. Clean Soil Air Water 2019, 47, 1–9. [Google Scholar] [CrossRef]
- Waghmare, S.S.; Arfin, T. Fluoride Removal from Water by Various Techniques: Review. Int. J. Innov. Sci. Eng. Technol. 2015, 2, 560–571. [Google Scholar]
- Manna, S.; Saha, P.; Roy, D.; Adhikari, B.; Das, P. Fixed Bed Column Study for Water Defluoridation Using Neem Oil-Phenolic Resin Treated Plant Bio-Sorbent. J. Environ. Manag. 2018, 212, 424–432. [Google Scholar] [CrossRef]
- Gill, T.; Tiwari, S.; Kumar, P.A. A Review on Feasibility of Conventional Fluoride Removal Techniques in Urban Areas. Int. J. Environ. Res. Dev. 2014, 4, 179–182. [Google Scholar]
- Liu, J.; Yue, X.; Lu, X.; Guo, Y. Uptake Fluoride from Water by Starch Stabilized Layered Double Hydroxides. Water 2018, 10, 745. [Google Scholar] [CrossRef]
- Yadav, K.K.; Gupta, N.; Kumar, V.; Khan, S.A.; Kumar, A. A Review of Emerging Adsorbents and Current Demand for Defluoridation of Water: Bright Future in Water Sustainability. Environ. Int. 2018, 111, 80–108. [Google Scholar] [CrossRef]
- Su, T.; Song, Y.; Lan, X.; Gao, W. Optimization for Removal e Ffi Ciency of Fl Uoride Using La ( III )–Al ( III )-Activated Carbon Modi Fi Ed by Chemical Route. Green Process. Synth. 2020, 9, 405–415. [Google Scholar] [CrossRef]
- Nehra, S.; Raghav, S.; Kumar, D. Biomaterial Functionalized Cerium Nanocomposite for Removal of Fluoride Using Central Composite Design Optimization Study. Environ. Pollut. 2020, 113773:1–113773:32. [Google Scholar] [CrossRef]
- Abu Bakar, A.H.; Abdullah, L.C.; Mohd Zahri, N.A.; Alkhatib, M. Column Efficiency of Fluoride Removal Using Quaternized Palm Kernel Shell (QPKS). Int. J. Chem. Eng. 2019, 5743590:1–5743590:13. [Google Scholar] [CrossRef]
- Kennedy, A.M.; Arias-Paic, M. Fixed-Bed Adsorption Comparisons of Bone Char and Activated Alumina for the Removal of Fluoride from Drinking Water. J. Environ. Eng. 2020, 146, 04019099. [Google Scholar] [CrossRef]
- Alkurdi, S.S.A.; Al-Juboori, R.A.; Bundschuh, J.; Hamawand, I. Bone Char as a Green Sorbent for Removing Health Threatening Fluoride from Drinking Water. Environ. Int. 2019, 127, 704–719. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, S.; Murugesh, S.; Sivasankar, V.; Darchen, A.; Msagati, T.A.M.; Chaabane, T. Low-Cost Fluoride Adsorbents Prepared from a Renewable Biowaste: Syntheses, Characterization and Modeling Studies. Arab. J. Chem. 2019, 12, 3004–3017. [Google Scholar] [CrossRef]
- Ghanbarian, M.; Ghanbarian, M.; Mahvi, A.H.; Tabatabaie, T. Enhanced Fluoride Removal over MgFe2O4–Chitosan–CaAl Nanohybrid: Response Surface Optimization, Kinetic and Isotherm Study. Int. J. Biol. Macromol. 2020, 148, 574–590. [Google Scholar] [CrossRef]
- Alemayehu, E.; Lennartz, B. Virgin Volcanic Rocks: Kinetics and Equilibrium Studies for the Adsorption of Cadmium from Water. J. Hazard. Mater. 2009, 169, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Aregu, M.B.; Asfaw, S.L.; Khan, M.M. Identification of Two Low—Cost and Locally Available Filter Media (Pumice and Scoria) for Removal of Hazardous Pollutants from Tannery Wastewater. Environ. Syst. Res. 2018, 7, 10:1–10:14. [Google Scholar] [CrossRef]
- Asere, T.G.; Mincke, S.; De Clercq, J.; Verbeken, K.; Tessema, D.A.; Fufa, F.; Stevens, C.V.; Du Laing, G. Removal of Arsenic (V) from Aqueous Solutions Using Chitosan–Red Scoria and Chitosan–Pumice Blends. Int. J. Environ. Res. Public Health 2017, 14, 895. [Google Scholar] [CrossRef]
- Asere, T.G.; Verbeken, K.; Tessema, D.A.; Fufa, F.; Stevens, C.V.; Du Laing, G. Adsorption of As(III) versus As(V) from Aqueous Solutions by Cerium-Loaded Volcanic Rocks. Environ. Sci. Pollut. Res. 2017, 24, 20446–20458. [Google Scholar] [CrossRef]
- Birhane, M.; Abebe, A.; Alemayehu, E.; Mengistie, E. Efficiency of Locally Available Filter Media on Fluoride and Phosphate Removal for Household Water Treatment System. Chin. J. Popul. Resour. Environ. 2014, 12, 110–115. [Google Scholar] [CrossRef]
- Viswanathan, N.; Meenakshi, S. Enriched Fluoride Sorption Using Alumina/Chitosan Composite. J. Hazard. Mater. 2010, 178, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Monash, P.; Pugazhenthi, G. Removal of Crystal Violet Dye from Aqueous Solution Using Calcined and Uncalcined Mixed Clay Adsorbents. Sep. Sci. Technol. 2010, 45, 94–104. [Google Scholar] [CrossRef]
- Patel, H. Fixed—Bed Column Adsorption Study: A Comprehensive Review. Appl. Water Sci. 2019, 9, 45:1–45:17. [Google Scholar] [CrossRef]
- US EPA United States Environmental Agency. Control of Organic Substances in Water and Wastewater; Document No.: U.S. EPA-600/8-83-011; U.S. Environmental Protection Agency: Washington, DC, USA, 1983.
- Rowe, M.C.; Brewer, B.J. AMORPH: A Statistical Program for Characterizing Amorphous Materials by X-Ray Diffraction 1. Comput. Geosci. 2018, 120, 24–31. [Google Scholar] [CrossRef]
- Li, X.; Yang, W.; Zou, Q.; Zuo, Y. Investigation on Microstructure, Compostion, and Cytocompatibility of Natural Pumice for Potential Biomedical Application. Tissue Eng. Part C 2010, 16, 427–434. [Google Scholar] [CrossRef]
- Sepehr, M.N.; Amrane, A.; Karimaian, K.A.; Zarrabi, M.; Ghaffari, H.R. Potential of Waste Pumice and Surface Modified Pumice for Hexavalent Chromium Removal: Characterization, Equilibrium, Thermodynamic and Kinetic Study. J. Taiwan Inst. Chem. Eng. 2014, 45, 635–647. [Google Scholar] [CrossRef]
- Pirsaheb, M.; Mohammadi, H.; Sharafi, K.; Asadi, A. Fluoride and Nitrate Adsorption from Water by Fe(III)-Doped Scoria: Optimizing Using Response Surface Modeling, Kinetic and Equilibrium Study. Water Sci. Technol. Water Supply 2017, 18, 1117–1132. [Google Scholar] [CrossRef]
- Djobo, J.N.Y.; Tchadjié, L.N.; Tchakoute, H.K.; Kenne, B.B.D.; Elimbi, A. Synthesis of Geopolymer Composites from a Mixture of Volcanic Scoria and Metakaolin. J. Asian Ceram. Socities 2014, 2, 387–398. [Google Scholar] [CrossRef]
- Panias, D.; Giannopoulou, I.P.; Perraki, T. Effect of Synthesis Parameters on the Mechanical Properties of Fly Ash-Based Geopolymers. Colloids Surfaces A Physicochem. Eng. Asp. 2007, 301, 246–254. [Google Scholar] [CrossRef]
- Asgari, G.; Roshani, B.; Ghanizadeh, G. The Investigation of Kinetic and Isotherm of Fluoride Adsorption onto Functionalize Pumice Stone. J. Hazard. Mater. 2012, 217–218, 123–132. [Google Scholar] [CrossRef]
- Moradi, M.; Mansouri, A.M.; Azizi, N.; Amini, J. Adsorptive Removal of Phenol from Aqueous Solutions by Copper (Cu)—Modified Scoria Powder: Process Modeling and Kinetic Evaluation. Desalin. Water Treat. 2015, 57, 1–15. [Google Scholar] [CrossRef]
- Souza, R.F.; Brandão, P.R.G.; Paulo, J.B.A. Effect of Chemical Composition on the Zeta -Potential of Chromite. Miner. Eng. 2012, 36–38, 65–74. [Google Scholar] [CrossRef]
- Ayoob, S.; Gupta, A.K. Performance Evaluation of Alumina Cement Granules in Removing Fluoride from Natural and Synthetic Waters. Chem. Eng. J. 2009, 150, 485–491. [Google Scholar] [CrossRef]
- Salifu, A.; Petrusevski, B.; Ghebremichael, K.; Modestus, L.; Buamah, R.; Aubry, C.; Amy, G.L. Aluminum (Hydr) Oxide Coated Pumice for Fluoride Removal from Drinking Water: Synthesis, Equilibrium, Kinetics and Mechanism. Chem. Eng. J. 2013, 228, 63–74. [Google Scholar] [CrossRef]
- Alemayehu, E.; Thiele-Bruhn, S.; Lennartz, B. Adsorption Behaviour of Cr (VI) onto Macro and Micro-Vesicular Volcanic Rocks from Water. Sep. Purif. Technol. 2011, 78, 55–61. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, Z.; Feng, C.; Li, M.; Chen, R.; Sugiura, N. Investigations on the Batch and Fixed-Bed Column Performance of Fluoride Adsorption by Kanuma Mud. Desalination 2011, 268, 76–82. [Google Scholar] [CrossRef]
- Alemayehu, E.; Lennartz, B. Adsorptive Removal of Nickel from Water Using Volcanic Rocks. Appl. Geochem. 2010, 25, 1596–1602. [Google Scholar] [CrossRef]
- Obijole, O.A.; Gitari, M.W.; Ndungu, P.G. Mechanochemically Activated Aluminosilicate Clay Soils and Their Application for Defluoridation and Pathogen Removal from Groundwater. Int. J. Environ. Res. Public Health 2019, 16, 654. [Google Scholar] [CrossRef]
- Sepehr, M.N.; Zarrabi, M.; Kazemian, H.; Amrane, A.; Yaghmaian, K.; Ghaffari, H.R. Removal of Hardness Agents, Calcium and Magnesium, by Natural and Alkaline Modified Pumice Stones in Single and Binary Systems. Appl. Surf. Sci. 2013, 274, 295–305. [Google Scholar] [CrossRef]
- Aksu, Z.; Çaǧatay, S.S.; Gönen, F. Continuous Fixed Bed Biosorption of Reactive Dyes by Dried Rhizopus Arrhizus: Determination of Column Capacity. J. Hazard. Mater. 2007, 143, 362–371. [Google Scholar] [CrossRef]
- Golie, W.M.; Upadhyayula, S. Continuous Fixed-Bed Column Study for the Removal of Nitrate from Water Using Chitosan/Alumina Composite. J. Water Process Eng. 2016, 12, 58–65. [Google Scholar] [CrossRef]
- Paudyal, H.; Pangeni, B.; Inoue, K.; Kawakita, H.; Ohto, K.; Alam, S. Adsorptive Removal of Fluoride from Aqueous Medium Using a Fixed Bed Column Packed with Zr (IV) Loaded Dried Orange Juice Residue. Bioresour. Technol. 2013, 146, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Ghorai, S.; Pant, K.K. Equilibrium, Kinetics and Breakthrough Studies for Adsorption of Fluoride on Activated Alumina. Sep. Purif. Technol. 2005, 42, 265–271. [Google Scholar] [CrossRef]
- Salifu, A. Laboratory-Scale Column Filter Studies for Fluoride Removal with Aluminium (Hydr) Oxide Coated Pumice, Regeneration and Disposal. In Fluoride Removal from Groundwater by Adsorption Technology: The Occurrence, Adsorbent Synthesis, Regeneration and Disposal; CRC Press/Balkema: Leiden, The Netherlands, 2017; pp. 127–129. ISBN 9780815392071. [Google Scholar]
- Han, R.; Wang, Y.; Yu, W.; Zou, W.; Shi, J.; Liu, H. Biosorption of Methylene Blue from Aqueous Solution by Rice Husk in a Fixed-Bed Column. J. Hazard. Mater. 2007, 141, 713–718. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Fixed-Bed Dynamic Column Adsorption Study of Methylene Blue (MB) onto Pine Cone. Desalin. Water Treat. 2015, 55, 1026–1039. [Google Scholar] [CrossRef]
- Quintelas, C.; Pereira, R.; Kaplan, E.; Tavares, T. Removal of Ni (II) from Aqueous Solutions by an Arthrobacter Viscosus Biofilm Supported on Zeolite: From Laboratory to Pilot Scale. Bioresour. Technol. 2013, 142, 368–374. [Google Scholar] [CrossRef]
- Kang, W.H.; Kim, E.I.; Park, J.Y. Fluoride Removal Capacity of Cement Paste. Desalination 2007, 202, 38–44. [Google Scholar] [CrossRef]
- García-sánchez, J.J.; Solache-ríos, M.; Martínez-miranda, V.; Morelos, C.S. Removal of Fluoride Ions from Drinking Water and Fluoride Solutions by Aluminum Modified Iron Oxides in a Column System. J. Colloid Interface Sci. 2013, 407, 410–415. [Google Scholar] [CrossRef]
- Ma, Y.; Shi, F.; Zheng, X.; Ma, J.; Gao, C. Removal of Fluoride from Aqueous Solution Using Granular Acid-Treated Bentonite (GHB): Batch and Column Studies. J. Hazard. Mater. 2011, 185, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Sivasankar, V.; Ramachandramoorthy, T.; Chandramohan, A. Fluoride Removal from Water Using Activated and MnO2-Coated Tamarind Fruit (Tamarindus Indica) Shell: Batch and Column Studies. J. Hazard. Mater. 2010, 177, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Ghorai, S.; Pant, K.K. Investigations on the Column Performance of Fluoride Adsorption by Activated Alumina in a Fixed-Bed. Chem. Eng. J. 2004, 98, 165–173. [Google Scholar] [CrossRef]
- Alemayehu, E.; Melak, F.; Sharma, S.K.; Van Der Bruggen, B.; Lennartz, B. Use of Porous Volcanic Rocks for the Adsorptive Removal of Copper. Water Environ. J. 2017, 31, 4–11. [Google Scholar] [CrossRef]
- Kwon, J.-S.; Yun, S.-T.; Lee, J.-H.; Kim, S.-O.; Jo, H.Y. Removal of Divalent Heavy Metals (Cd, Cu, Pb, and Zn) and Arsenic (III) from Aqueous Solutions Using Scoria: Kinetics and Equilibria of Sorption. J. Hazard. Mater. 2010, 174, 307–313. [Google Scholar] [CrossRef]
- Liu, C.; Evett, J.B. Soil Properties-Testing, Measurament, and Evalution; Banta Book Company: Upper Saddle River, NJ, USA, 2003; ISBN 0-13-093005-9. [Google Scholar]
- Chen, I.-P.; Kan, C.-C.; Futalan, C.; Calagui, M.J.; Lin, S.-S.; Tsai, W.-C.; Wan, M.-W. Batch and Fixed Bed Studies: Removal of Copper (II) Using Chitosan-Coated Kaolinite Beads from Aqueous Solution. Sustain. Environ. Res. 2015, 25, 73–81. [Google Scholar]
- De Gennaro, B.; Aprea, P.; Liguori, B.; Galzerano, B.; Peluso, A.; Caputo, D. Zeolite-Rich Composite Materials for Environmental Remediation: Arsenic Removal from Water. Appl. Sci. 2020, 10, 6939. [Google Scholar] [CrossRef]
- Chen, S.; Yue, Q.; Gao, B.; Li, Q.; Xu, X.; Fu, K. Adsorption of Hexavalent Chromium from Aqueous Solution by Modified Corn Stalk: A Fixed-Bed Column Study. Bioresour. Technol. 2012, 113, 114–120. [Google Scholar] [CrossRef]
- Worch, E. Adsorption Technology in Water Treatment: Fundamentals, Processes, and Modeling; Walter de Gruyter and Co.KG: Berlin, Germany, 2012; ISBN 9783110240221. [Google Scholar]
- Mohan, S.; Singh, D.K.; Kumar, V.; Hasan, S.H. Modelling of Fixed Bed Column Containing Graphene Oxide Decorated by MgO Nanocubes as Adsorbent for Lead ( II ) Removal from Water. J. Water Process Eng. 2017, 17, 216–228. [Google Scholar] [CrossRef]
- Thomas, H.C. Heterogeneous Ion Exchange in a Flowing System. J. Am. Chem. Soc. 1944, 66, 1664–1666. [Google Scholar] [CrossRef]
- Singh, T.P.; Ghosh, S.; Cb, M. Adsorption of Fluoride from Industrial Wastewater in Fixed Bed Column Using Java Plum (Syzygium Cumini). Asian J. Pharm. Clin. Res. 2016, 9. [Google Scholar] [CrossRef]
- Bohart, G.S.; Adams, E.Q. Some Aspects of the Behavior of Charcoal with Respect to Chlorine. J. Am. Chem. Society 1920, 42, 523–544. [Google Scholar] [CrossRef]
- Chu, K.H. Breakthrough Curve Analysis by Simplistic Models of Fixed Bed Adsorption: In Defense of the Century-Old Bohart-Adams Model. Chem. Eng. J. 2020, 380, 122513:1–122513:8. [Google Scholar] [CrossRef]
- Han, R.; Zou, L.; Zhao, X.; Xu, Y.; Xu, F.; Li, Y.; Wang, Y. Characterization and Properties of Iron Oxide-Coated Zeolite as Adsorbent for Removal of Copper (II) from Solution in Fixed Bed Column. Chem. Eng. J. 2009, 149, 123–131. [Google Scholar] [CrossRef]

| Parameters | Virgin Scoria (VSco) | Virgin Pumice (VPum) | ||||
|---|---|---|---|---|---|---|
| Particle Size (mm) | <0.075 | 0.075–0.425 | 0.425–2.00 | <0.075 | 0.075–0.425 | 0.425–2.00 |
| Mass of adsorbents, mads (gm) | 737.90 | 763.90 | 680.50 | 376.40 | 265.90 | 186.40 |
| Bulk density, ρb (gm cm−3) | 1.43 | 1.48 | 1.32 | 0.73 | 0.52 | 0.36 |
| Particle density, ρs (gm cm−3) | 2.37 | 2.33 | 1.94 | 1.61 | 1.31 | 0.64 |
| Void volume, Vv (cm3) | 203.70 | 187.20 | 164.70 | 281.10 | 311.80 | 222.30 |
| Total porosity, ε | 0.40 | 0.36 | 0.32 | 0.55 | 0.61 | 0.43 |
| Flow rate, Q (cm3 min−1) | 1.25 | 2.50 | 3.75 | 1.25 | 2.50 | 3.75 |
| Empty Bed Contact Time, EBCT (min) | 412.00 | 206.00 | 137.33 | 412.00 | 206.00 | 137.33 |
| Filter (Superficial) velocity, Vf (cm min−1) | 0.02 | 0.05 | 0.07 | 0.02 | 0.05 | 0.07 |
| Effective (Interstitial) velocity, VI (cm min−1) | 0.06 | 0.13 | 0.23 | 0.05 | 0.08 | 0.17 |
| VPum. | H (cm) | CO (mg/L) | QO (mL/min) | pH | Particles size (Psize) (mm) | tb (min) | te (min) | Vb (mL) | Ve (mL) | MTZ (cm) | EBCT (min) | qtot (mg) | qe (mg/kg) |
| 10 | 10 | 1.25 | 2.00 | <0.075 | 816 | 1623 | 1019.64 | 2033.41 | 4.99 | 412 | 20.28 | 59.6 | |
| 10 | 10 | 1.25 | 2.00 | 0.075–0.425 | 1206 | 2339 | 1507.50 | 2923.70 | 4.84 | 412 | 29.24 | 109.9 | |
| 10 | 10 | 1.25 | 2.00 | 0.425–2.00 | 235 | 1013 | 293.23 | 1265.89 | 7.68 | 412 | 12.67 | 67.9 | |
| 10 | 10 | 1.25 | 4.00 | 0.075–0.425 | 278 | 500 | 347.50 | 625 | 4.44 | 412 | 6.25 | 23.51 | |
| 10 | 10 | 1.25 | 6.00 | 0.075–0.425 | 135 | 315 | 168.75 | 393.75 | 5.71 | 412 | 3.94 | 14.81 | |
| 10 | 10 | 2.50 | 2.00 | 0.075–0.425 | 215 | 634 | 538.47 | 1585.16 | 6.60 | 206 | 7.93 | 29.8 | |
| 10 | 10 | 3.75 | 2.00 | 0.075–0.425 | 75 | 359 | 282.69 | 1346.42 | 7.90 | 137 | 4.49 | 16.89 | |
| VSco | H (cm) | CO (mg/L) | QO (mL/min) | pH | Particles size (Psize)(mm) | tb (min) | te (min) | Vb (mL) | Ve (mL) | MTZ (cm) | EBCT (min) | qtot (mg) | qe (mg/kg) |
| 10 | 10 | 1.25 | 2.00 | <0.075 | 415 | 1286 | 518.03 | 1607.60 | 6.77 | 412 | 16.08 | 22 | |
| 10 | 10 | 1.25 | 2.00 | 0.075–0.425 | 199 | 760 | 248.99 | 849.80 | 7.38 | 412 | 9.50 | 12.4 | |
| 10 | 10 | 1.25 | 2.00 | 0.425–2.00 | 231 | 591 | 288.17 | 739.12 | 6.10 | 412 | 7.39 | 10.9 | |
| 10 | 10 | 1.25 | 4.00 | <0.075 | 296 | 487 | 370 | 608.75 | 3.92 | 412 | 6.09 | 8.2 | |
| 10 | 10 | 1.25 | 6.00 | <0.075 | 227 | 393 | 283.75 | 491.25 | 4.22 | 412 | 4.91 | 6.7 | |
| 10 | 10 | 2.50 | 2.00 | <0.075 | 185 | 445 | 462.95 | 1113.19 | 5.84 | 206 | 5.57 | 7.5 | |
| 10 | 10 | 3.75 | 2.00 | <0.075 | 69 | 249 | 256.82 | 931.87 | 7.24 | 137 | 3.10 | 4.2 |
| VPum | H (cm) | Co (mg/L) | Q (mL/min) | pH | Psize (mm) | KT (L/min.mg) (×104) | q0(cal.) (mg/kg) | qe(exp.) (mg/kg) | R2 |
| 10 | 10 | 1.25 | 2.00 | <0.075 | 2.199 | 48.6 | 59.6 | 0.950 | |
| 10 | 10 | 1.25 | 2.00 | 0.075–0.425 | 1.440 | 109.9 | 109.9 | 0.993 | |
| 10 | 10 | 1.25 | 2.00 | 0.425–2.00 | 1.840 | 57.9 | 67.9 | 0.897 | |
| 10 | 10 | 1.25 | 4.00 | 0.075–0.425 | 8.289 | 17.83 | 23.51 | 0.977 | |
| 10 | 10 | 1.25 | 6.00 | 0.075–0.425 | 12.099 | 13.08 | 14.81 | 0.995 | |
| 10 | 10 | 2.50 | 2.00 | 0.075–0.425 | 3.565 | 55.81 | 29.8 | 0.953 | |
| 10 | 10 | 3.75 | 2.00 | 0.075–0.425 | 5 | 45.82 | 16.9 | 0.962 | |
| VSco | H (cm) | Co (mg/L) | Q (mL/min) | pH | Psize (mm) | KT (L/min.mg) (×104) | q0(cal.) (mg/kg) | qe(exp.) (mg/kg) | R2 |
| 10 | 10 | 1.25 | 2.00 | <0.075 | 1.23 | 19 | 22 | 0.901 | |
| 10 | 10 | 1.25 | 2.00 | 0.075–0.425 | 2.213 | 13.4 | 12.4 | 0.931 | |
| 10 | 10 | 1.25 | 2.00 | 0.425–2.00 | 3.310 | 10.1 | 10.9 | 0.956 | |
| 10 | 10 | 1.25 | 4.00 | <0.075 | 10.140 | 7.06 | 8.2 | 0.973 | |
| 10 | 10 | 1.25 | 6.00 | <0.075 | 15.520 | 5.42 | 6.7 | 0.965 | |
| 10 | 10 | 2.50 | 2.00 | <0.075 | 5.263 | 13.3 | 7.5 | 0.929 | |
| 10 | 10 | 3.75 | 2.00 | <0.075 | 7.220 | 11.2 | 4.2 | 0.944 |
| VPum | H (cm) | Co (mg/L) | Q (mL/min) | pH | Psize (mm) | KAB (L/min.mg) (×104) | N0 (mg/L) | R2 |
| 10 | 10 | 1.25 | 2.00 | <0.075 | 2.187 | 35.55 | 0.950 | |
| 10 | 10 | 1.25 | 2.00 | 0.075–0.425 | 1.439 | 56.85 | 0.993 | |
| 10 | 10 | 1.25 | 2.00 | 0.425–2.00 | 1.741 | 20.77 | 0.911 | |
| 10 | 10 | 1.25 | 4.00 | 0.075–0.425 | 8. 289 | 9.22 | 0.995 | |
| 10 | 10 | 1.25 | 6.00 | 0.075–0.425 | 12. 099 | 6.76 | 0.953 | |
| 10 | 10 | 2.50 | 2.00 | 0.075–0.425 | 3.565 | 29.68 | 0. 962 | |
| 10 | 10 | 3.75 | 2.00 | 0.075–0.425 | 5.000 | 22.74 | 0.995 | |
| VSco | H(cm) | Co (mg/L) | Q (mL/min) | pH | Psize (mm) | KAB (L/min.mg) (×104) | No (mg/L) | R2 |
| 10 | 10 | 1.25 | 2.00 | <0.075 | 1.233 | 27.27 | 0.886 | |
| 10 | 10 | 1.25 | 2.00 | 0.075–0.425 | 2.213 | 17.68 | 0.886 | |
| 10 | 10 | 1.25 | 2.00 | 0.425–2.00 | 3.310 | 13.41 | 0.956 | |
| 10 | 10 | 1.25 | 4.00 | < 0.075 | 10.145 | 10.13 | 0.969 | |
| 10 | 10 | 1.25 | 6.00 | < 0.075 | 15.518 | 7.77 | 0.980 | |
| 10 | 10 | 2.50 | 2.00 | < 0.075 | 5.263 | 19.72 | 0.929 | |
| 10 | 10 | 3.75 | 2.00 | < 0.075 | 7.221 | 15.36 | 0.944 |
| Adsorbents | Surface Area (m2g−1) | Bed Depth (cm) | Fluoride in (mgL−1) | Adsorption Capacity (mg F- g−1) | Adsorption Capacity per Surface Area (mg.m−2) | References |
|---|---|---|---|---|---|---|
| Cement paste | NA* | 20 | 15 | 0.149 | - | [61] |
| aluminum modified iron oxide | NA | 10.5 | 4 | 0.139 | - | [62] |
| Acid-treated bentonite (GHB) | 24.5 | 28 | 2.85 | 0.169 | 0.0069 | [63] |
| MnO2-coated Tamarind Fruit Shell | NA | 6 | 2 | 0.883 | - | [64] |
| kanuma mud | 144.01 | 10 | 20 | 1.560 | 0.0108 | [49] |
| Activated alumina (Grade OA-25) | 250 | 10 | 5 | 0.74 | 0.0029 | [65] |
| VPum | 3.5 | 10 | 10 | 0.110 | 0.0314 | This study |
| VSco | 2.49 | 10 | 10 | 0.022 | 0.0088 | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geleta, W.S.; Alemayehu, E.; Lennartz, B. Volcanic Rock Materials for Defluoridation of Water in Fixed-Bed Column Systems. Molecules 2021, 26, 977. https://doi.org/10.3390/molecules26040977
Geleta WS, Alemayehu E, Lennartz B. Volcanic Rock Materials for Defluoridation of Water in Fixed-Bed Column Systems. Molecules. 2021; 26(4):977. https://doi.org/10.3390/molecules26040977
Chicago/Turabian StyleGeleta, Wondwosen Sime, Esayas Alemayehu, and Bernd Lennartz. 2021. "Volcanic Rock Materials for Defluoridation of Water in Fixed-Bed Column Systems" Molecules 26, no. 4: 977. https://doi.org/10.3390/molecules26040977
APA StyleGeleta, W. S., Alemayehu, E., & Lennartz, B. (2021). Volcanic Rock Materials for Defluoridation of Water in Fixed-Bed Column Systems. Molecules, 26(4), 977. https://doi.org/10.3390/molecules26040977






