HRMS Characterization, Antioxidant and Cytotoxic Activities of Polyphenols in Malus domestica Cultivars from Costa Rica
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phenolic Yield and Total Phenolic Content
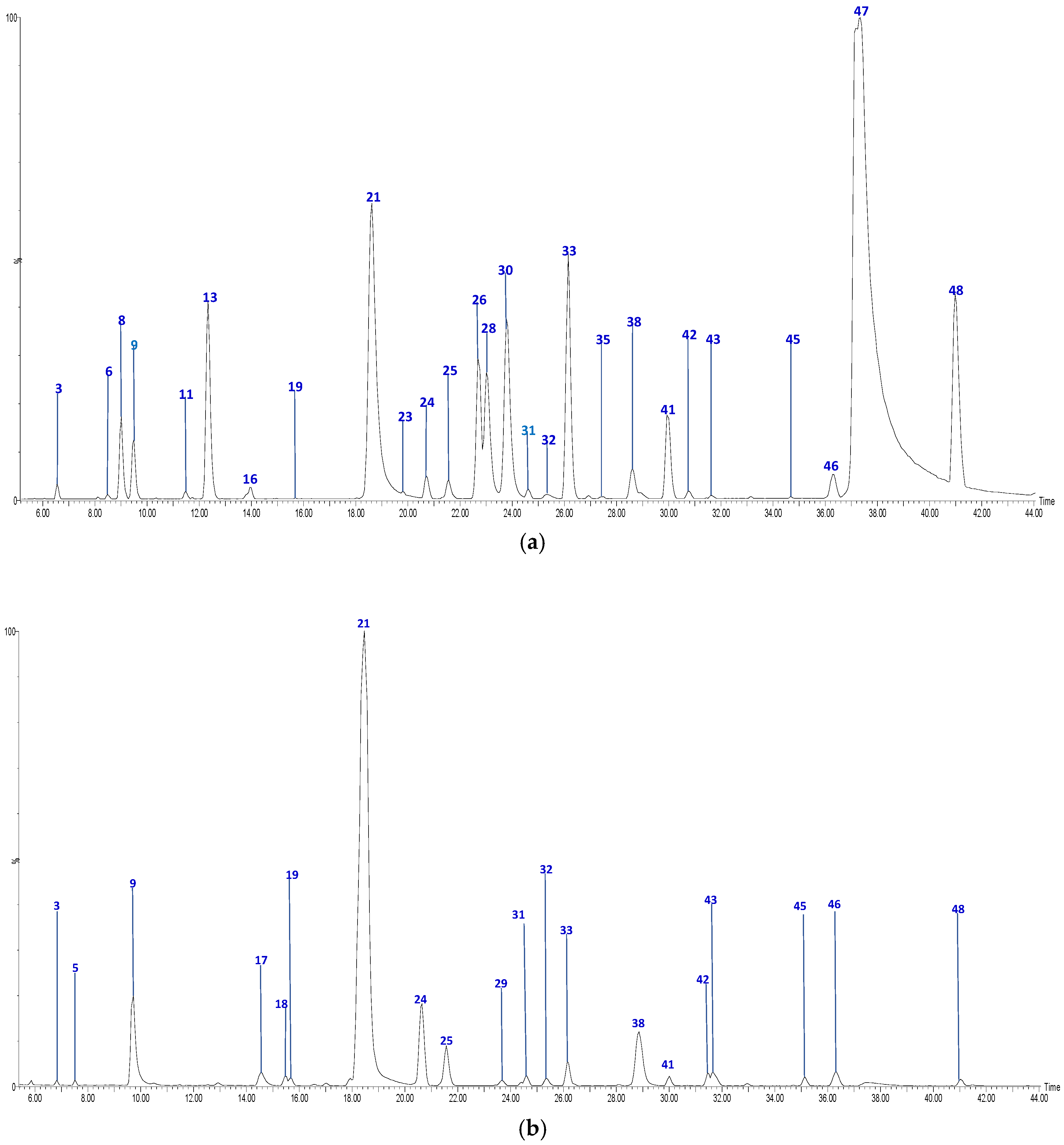
2.2. Profile by UPLC-QTOF-ESI MS Analysis
2.3. Antioxidant Activity
2.4. Cytotoxicity
3. Materials and Methods
3.1. Materials, Reagents and Solvents
3.2. Phenolic Extracts from Malus Domestica Fruits
3.3. Total Phenolic Content
3.4. UPLC-ESI-MS Analysis
3.5. DPPH Radical-Scavenging Activity
3.6. ORAC Antioxidant Activity
3.7. Evaluation of Cytotoxicity of Extracts
3.7.1. Cell Culture
3.7.2. Assessment of Cytotoxicity by MTT Assay
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Ahmad Khan, M.S.; Ahmad, I. Herbal Medicine. In New Look to Phytomedicine; Ahmad Khan, M.S., Ahmad, I., Chattopadhyay, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 3–13. ISBN 9780128146194. [Google Scholar] [CrossRef]
- Boyer, J.; Liu, R.H. Apple Phytochemicals and Their Health Benefits. Nutr. J. 2004, 3, 5. [Google Scholar] [CrossRef] [Green Version]
- Lavie, L.; Lavie, P. Molecular mechanisms of cardiovascular disease in OSAHS: The oxidative stress link. Eur. Respir. J. 2009, 33, 1467–1484. [Google Scholar] [CrossRef]
- Mariani, E.; Polidori, M.C.; Cherubini, A.; Mecocci, P. Oxidative Stress in Brain Aging, Neurodegenerative and Vascular Diseases: An Overview. J. Chromatogr. B 2005, 827, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, F.U.; Chhipa, A.S.; Sagar, N.; Pathak, C. Oxidative Stress and Inflammation Can Fuel Cancer. In Role of Oxidative Stress in Pathophysiology of Diseases; Maurya, P.K., Dua, K., Eds.; Springer: Singapore, 2020; pp. 229–258. ISBN 978-981-15-1568-2. [Google Scholar] [CrossRef]
- Pignatelli, P.; Menichelli, D.; Pastori, D.; Violi, F. Oxidative Stress and Cardiovascular Disease: New Insights. Kardiol. Pol. 2018, 76, 713–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ivanov, A.V.; Bartosch, B.; Isaguliants, M.G. Oxidative Stress in Infection and Consequent Disease. Oxid. Med. Cell. Longev. 2017, 2017, 3496043. [Google Scholar] [CrossRef] [PubMed]
- Koch, W. Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review. Nutrients 2019, 11, 1039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asgary, S.; Rastqar, A.; Keshvari, M. Functional Food and Cardiovascular Disease Prevention and Treatment: A Review. J. Am. Coll. Nutr. 2018, 37, 429–455. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Su, X.; Zubik, L.; Bose, P. Phenol Antioxidant Quantity and Quality in Foods: Fruits. J. Agric. Food Chem. 2011, 49, 5315–5321. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Unusan, N. Proanthocyanidins in Grape Seeds: An Updated Review of Their Health Benefits and Potential Uses in the Food Industry. J. Funct. Foods 2020, 67, 103861. [Google Scholar] [CrossRef]
- Patocka, J.; Bhardwaj, K.; Klimova, B.; Nepovimova, E.; Wu, Q.; Landi, M.; Kuca, K.; Valis, M.; Wu, W. Malus Domestica: A Review on Nutritional Features, Chemical Composition, Traditional and Medicinal Value. Plants 2020, 9, 1408. [Google Scholar] [CrossRef] [PubMed]
- Mariadoss, A.V.A.; Vinyagam, R.; Rajamanickam, V.; Sankaran, V.; Venkatesan, S.; David, E. Pharmacological Aspects and Potential Use of Phloretin: A Systemic Review. Mini-Rev. Med. Chem. 2019, 19, 1060–1067. [Google Scholar] [CrossRef]
- Eberhardt, M.; Lee, C.; Liu, R.H. Antioxidant activity of fresh apples. Nature 2000, 405, 903–904. [Google Scholar] [CrossRef]
- Acquavia, M.; Pascale, R.; Foti, L.; Carlucci, G.; Scrano, L.; Martelli, G.; Brienza, M.; Coviello, D.; Bianco, G.; Lelario, F. Analytical Methods for Extraction and Identification of Primary and Secondary Metabolites of Apple (Malus domestica) Fruits: A Review. Separations 2021, 8, 91. [Google Scholar] [CrossRef]
- Feng, S.; Yi, J.; Li, X.; Wu, X.; Zhao, Y.; Ma, Y.; Bi, J. Systematic Review of Phenolic Compounds in Apple Fruits: Compositions, Distribution, Absorption, Metabolism, and Processing Stability. J. Agric. Food Chem. 2021, 69, 7–27. [Google Scholar] [CrossRef] [PubMed]
- Shehzadi, K.; Rubab, Q.; Asad, L.; Ishfaq, M.; Shafique, B.; Ali Nawaz Ranjha, M.M.; Mahmood, S.; Mueen-Ud-Din, G.; Javaid, T.; Sabtain, B.; et al. A Critical Review on Presence of Polyphenols in Commercial Varieties of Apple Peel, their Extraction and Health Benefits. Op. Acc. J. Bio. Sci. Res. 2020, 6, 18. [Google Scholar] [CrossRef]
- Abdelfattah, A.; Freilich, S.; Bartuv, R.; Zhimo, V.Y.; Kumar, A.; Biasi, A.; Salim, S.; Feygenberg, O.; Burchard, E.; Dardick, C.; et al. Global Analysis of the Apple Fruit Microbiome: Are All Apples the Same? Environ. Microbiol. 2021, 23, 6038–6055. [Google Scholar] [CrossRef] [PubMed]
- Bustos-Segura, C.; Poelman, E.H.; Reichelt, M.; Gershenzon, J.; Gols, R. Intraspecific Chemical Diversity among Neighbouring Plants Correlates Positively with Plant Size and Herbivore Load but Negatively with Herbivore Damage. Ecol. Lett. 2017, 20, 87–97. [Google Scholar] [CrossRef]
- Ramírez, S.M. Informe Comercio Exterior del Sector Agropecuario 2019–2020; SEPSA 2021, 2021–003; Secretaría Ejecutiva de Planificación Sectorial Agropecuaria: San Jose, Costa Rica, 2021. [Google Scholar]
- Navarro, M.; Moreira, I.; Arnaez, E.; Quesada, S.; Azofeifa, G.; Vargas, F.; Alvarado, D.; Chen, P. Polyphenolic characterization and antioxidant activity of Malus domestica and Prunus domestica cultivars from Costa Rica. Foods 2018, 7, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnous, A.; Meyer, A.S. Comparison of methods for compositional characterization of grape (Vitis vinifera L.) and apple (Malus domestica) skins. Food Bioprod. Process. 2008, 86, 79–86. [Google Scholar] [CrossRef]
- Kschonsek, J.; Wolfram, T.; Stöckl, A.; Böhm, V. Polyphenolic Compounds Analysis of Old and New Apple Cultivars and Contribution of Polyphenolic Profile to the In Vitro Antioxidant Capacity. Antioxidants 2018, 7, 20. [Google Scholar] [CrossRef] [Green Version]
- Tsao, R.; Yang, R.; Young, J.C.; Zhu, H. Polyphenolic Profiles in Eight Apple Cultivars Using High-Performance Liquid Chromatography (HPLC). J. Agric. Food Chem. 2003, 51, 6347–6353. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ban, Q.; Shi, J.; Dong, T.; Jiang, C.-Z.; Wang, Q. 1-Methylcyclopropene (1-MCP), Storage Time, and Shelf Life and Temperature Affect Phenolic Compounds and Antioxidant Activity of ‘Jonagold’ Apple. Postharvest Biol. Technol. 2019, 150, 71–79. [Google Scholar] [CrossRef]
- Hilt, P.; Schieber, A.; Yildirim, C.; Arnold, G.; Klaiber, I.; Conrad, J.; Beifuss, U.; Carle, R. Detection of Phloridzin in Strawberries (Fragaria × Ananassa Duch.) by HPLC−PDA−MS/MS and NMR Spectroscopy. J. Agric. Food Chem. 2003, 51, 2896–2899. [Google Scholar] [CrossRef] [PubMed]
- Zupan, A.; Mikulic-Petkovsek, M.; Slatnar, A.; Stampar, F.; Veberic, R. Individual Phenolic Response and Peroxidase Activity in Peel of Differently Sun-Exposed Apples in the Period Favorable for Sunburn Occurrence. J. Plant. Physiol. 2014, 171, 1706–1712. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Bai, N.; He, K.; Ho, C.-T.; Yang, C.S.; Sang, S. Apple Polyphenols, Phloretin and Phloridzin: New Trapping Agents of Reactive Dicarbonyl Species. Chem. Res. Toxicol. 2008, 21, 2042–2050. [Google Scholar] [CrossRef]
- Schwab, W.; Schreier, P. Vomifoliol l-0-β-d-xylopyranosyl-6-o-β-d-glucopyranoside: A disaccharide glycoside from apple fruit. Phytochemistry 2008, 29, 161–164. [Google Scholar] [CrossRef]
- Hamed, A.I.; Al-Ayed, A.S.; Moldoch, J.; Piacente, S.; Oleszek, W.; Stochmal, A. Profiles Analysis of Proanthocyanidins in the Argun Nut (Medemia argun -an Ancient Egyptian Palm) by LC-ESI-MS/MS: Analysis of PACs in M. Argun Nut by LC-ESI-MS/MS. J. Mass Spectrom. 2014, 49, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Rue, E.A.; Rush, M.D.; van Breemen, R.B. Procyanidins: A Comprehensive Review Encompassing Structure Elucidation via Mass Spectrometry. Phytochem. Rev. 2018, 17, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Karonen, M.; Loponen, J.; Ossipov, V.; Pihlaja, K. Analysis of Procyanidins in Pine Bark with Reversed-Phase and Normal-Phase High-Performance Liquid Chromatography–Electrospray Ionization Mass Spectrometry. Anal. Chim. Acta 2004, 522, 105–112. [Google Scholar] [CrossRef]
- Jaiswal, R.; Karakose, H.; Ruhmann, S.; Goldner, K.; Neumuller, M.; Treutter, D.; Kuhnert, N. Identification of phenolic compounds in plum fruits (Prunus salicina L. and Prunus domestica L.) by HPLC/MS and characterization of varieties by quantitative phenolic fingerprints. J. Agric. Food Chem. 2013, 61, 12020–12031. [Google Scholar] [CrossRef] [PubMed]
- Fabre, N.; Rustan, I.; Hoffmann, E.; Quetin-Leclercq, J. Determination of Flavone, Flavonol, and Flavanone Aglycones by Negative Ion Liquid Chromatography Electrospray Ion Trap Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 707–715. [Google Scholar] [CrossRef] [Green Version]
- Ćirić, A.; Prosen, H.; Jelikić-Stankov, M.; Đurđević, P. Evaluation of Matrix Effect in Determination of Some Bioflavonoids in Food Samples by LC–MS/MS Method. Talanta 2012, 99, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, J.; Sánchez-González, C.; Vallverdú-Queralt, A.; Simal-Gándara, J.; Lamuela-Raventós, R.; Izquierdo-Pulido, M. Comprehensive identification of walnut polyphenols by liquid chromatography coupled to linear ion trap–Orbitrap mass spectrometry. Food Chem. 2014, 152, 340–348. [Google Scholar] [CrossRef]
- Lin, L.-Z.; Harnly, J.M. A Screening Method for the Identification of Glycosylated Flavonoids and Other Phenolic Compounds Using a Standard Analytical Approach for All Plant Materials. J. Agric. Food Chem. 2007, 55, 1084–1096. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.-Y.; Zhang, Q.; Li, N.; Wang, Z.-J.; Lu, J.-Q.; Qiao, Y.-J. Diagnostic Fragment-Ion-Based and Extension Strategy Coupled to DFIs Intensity Analysis for Identification of Chlorogenic Acids Isomers in Flos Lonicerae Japonicae by HPLC-ESI-MSn. Talanta 2013, 104, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, K.; Nakamura, S.; Matsumoto, T.; Ohta, T.; Yoshikawa, M.; Ogawa, K.; Kashiwazaki, E.; Matsuda, H. Structures of acylated sucroses from the flower buds of Prunus mume. J. Nat. Med. 2014, 68, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Cádiz-Gurrea, M.D.L.L.; Fernández-Arroyo, S.; Joven, J.; Segura-Carretero, A. Comprehensive Characterization by UHPLC-ESI-Q-TOF-MS from an Eryngium bourgatii Extract and Their Antioxidant and Anti-Inflammatory Activities. Food Res. Int. 2013, 50, 197–204. [Google Scholar] [CrossRef]
- Alberti, A.; Zielinski, A.A.F.; Couto, M.; Judacewski, P.; Mafra, L.I.; Nogueira, A. Distribution of Phenolic Compounds and Antioxidant Capacity in Apples Tissues during Ripening. J. Food Sci. Technol. 2017, 54, 1511–1518. [Google Scholar] [CrossRef] [Green Version]
- Khanizadeh, S.; Tsao, R.; Rekika, D.; Yang, R.; Charles, M.T.; Vasantha Rupasinghe, H.P. Polyphenol Composition and Total Antioxidant Capacity of Selected Apple Genotypes for Processing. J. Food Compost. Anal. 2008, 21, 396–401. [Google Scholar] [CrossRef]
- Łysiak, G.P.; Michalska-Ciechanowska, A.; Wojdyło, A. Postharvest Changes in Phenolic Compounds and Antioxidant Capacity of Apples Cv. Jonagold Growing in Different Locations in Europe. Food Chem. 2020, 310, 125912. [Google Scholar] [CrossRef]
- Lee, J.; Jeong, M.-C.; Ku, K.-H. Chemical, Physical, and Sensory Properties of 1-MCP-Treated Fuji Apple (Malus domestica Borkh.) Fruits after Long-Term Cold Storage. Appl. Biol. Chem. 2017, 60, 363–374. [Google Scholar] [CrossRef]
- Groth, S.; Budke, C.; Neugart, S.; Ackermann, S.; Kappenstein, F.-S.; Daum, D.; Rohn, S. Influence of a Selenium Biofortification on Antioxidant Properties and Phenolic Compounds of Apples (Malus domestica). Antioxidants 2020, 9, 187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hagen, S.F.; Borge, G.I.A.; Bengtsson, G.B.; Bilger, W.; Berge, A.; Haffner, K.; Solhaug, K.A. Phenolic Contents and Other Health and Sensory Related Properties of Apple Fruit (Malus domestica Borkh., Cv. Aroma): Effect of Postharvest UV-B Irradiation. Postharvest Biol. Technol. 2007, 45, 1–10. [Google Scholar] [CrossRef]
- Pires, T.C.S.P.; Dias, M.I.; Barros, L.; Alves, M.J.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Antioxidant and Antimicrobial Properties of Dried Portuguese Apple Variety (Malus Domestica Borkh. Cv Bravo de Esmolfe). Food Chem. 2018, 240, 701–706. [Google Scholar] [CrossRef] [Green Version]
- Xiao, Z.; Zhang, Y.; Chen, X.; Wang, Y.; Chen, W.; Xu, Q.; Li, P.; Ma, F. Extraction, Identification, and Antioxidant and Anticancer Tests of Seven Dihydrochalcones from Malus ‘Red Splendor’ Fruit. Food Chem. 2017, 231, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Hoyos, M.; Arnáez-Serrano, E.; Quesada-Mora, S.; Azofeifa-Cordero, G.; Wilhelm-Romero, K.; Quirós-Fallas, M.I.; Alvarado-Corella, D.; Vargas-Huertas, F.; Sánchez-Kopper, A. Polyphenolic QTOF-ESI MS Characterization and the Antioxidant and Cytotoxic Activities of Prunus domestica Commercial Cultivars from Costa Rica. Molecules 2021, 26, 6493. [Google Scholar] [CrossRef]
- McGhie, T.K.; Hunt, M.; Barnett, L.E. Cultivar and Growing Region Determine the Antioxidant Polyphenolic Concentration and Composition of Apples Grown in New Zealand. J. Agric. Food Chem. 2005, 53, 3065–3070. [Google Scholar] [CrossRef]
- Coll Aráoz, M.V.; Mercado, M.I.; Grau, A.; Catalán, C.A.N. Intraspecific Variation of Sesquiterpene Lactones Associated to a Latitudinal Gradient in Smallanthus macroscyphus (Heliantheae: Asteraceae). Chemoecology 2016, 26, 143–151. [Google Scholar] [CrossRef]
- Vineetha, V.P.; Girija, S.; Soumya, R.S.; Raghu, K.G. Polyphenol-rich apple (Malus domestica L.) peel extract attenuates arsenic trioxide induced cardiotoxicity in H9c2 cells via its antioxidant activity. Food Funct. 2014, 5, 502–511. [Google Scholar] [CrossRef]
- Oszmiański, J.; Lachowicz, S.; Gamsjäger, H. Phytochemical analysis by liquid chromatography of ten old apple varieties grown in Austria and their antioxidative activity. Eur. Food Res. Technol. 2020, 246, 437–448. [Google Scholar] [CrossRef]
- Giomaro, G.; Karioti, A.; Bucchini, A.; Giamperi, L.; Ricci, D.; Fraternale, D. Polyphenols profile and antioxidant activity of skin and pulp of a rare apple from Marche region (Italy). Chem. Central J. 2014, 10, 8–45. [Google Scholar] [CrossRef] [Green Version]
- Yuri, J.A.; Neira, A.; Fuentes, M.; Saez, B.; Razmilic, I. A New Sunburned Apple Category Browning under Conventional and Organic Management: Phenolic Compounds and Antioxidant Capacity in Cold Storage. J. Appl. Bot. Food Qual. 2019, 92, 288–297. [Google Scholar] [CrossRef]
- Lizcano, L.J.; Bakkali, F.; Ruiz-Larrea, B.; Ruiz-Sanz, J.I. Antioxidant activity and polyphenol content of aqueous extracts from Colombia Amazonian plants with medicinal use. Food Chem. 2010, 119, 1566–1570. [Google Scholar] [CrossRef]
- Rodríguez-Ramiro, I.; Ramos, S.; Bravo, L.; Goya, L.; Martín, M.Á. Procyanidin B2 Induces Nrf2 Translocation and Glutathione S-Transferase P1 Expression via ERKs and P38-MAPK Pathways and Protect Human Colonic Cells against Oxidative Stress. Eur. J. Nutr. 2012, 51, 881–892. [Google Scholar] [CrossRef] [Green Version]
- Ohnuma, T.; Matsumoto, T.; Itoi, A.; Kawana, A.; Nishiyama, T.; Ogura, K.; Hiratsuka, A. Enhanced Sensitivity of A549 Cells to the Cytotoxic Action of Anticancer Drugs via Suppression of Nrf2 by Procyanidins from Cinnamomi Cortex Extract. Biochem. Biophys. Res. Commun. 2011, 413, 623–629. [Google Scholar] [CrossRef]
- Turati, F.; Rossi, M.; Pelucchi, C.; Levi, F.; La Vecchia, C. Fruit and vegetables and cancer risk: A review of 722 southern European studies. Br. J. Nutr. 2015, 113, S102–S110. [Google Scholar] [CrossRef] [PubMed]
- La Vecchia, C.; Altieri, A.; Tavani, A. Vegetables, fruit, antioxidants and cancer: A review of Italian studies. Eur. J. Nutr. 2001, 40, 261–267. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Cancer Today-Population fact Sheets-Costa Rica. Available online: https://gco.iarc.fr/today/data/factsheets/populations/188-costa-rica-fact-sheets.pdf (accessed on 25 November 2021).
- Rauf, A.; Imran, M.; Abu-Izneid, T.; Patel, S.; Pan, X.; Naz, S.; Sanches Silva, A.; Saeed, F.; Rasul Suleria, H.A. Proanthocyanidins: A Comprehensive Review. Biomed. Pharmacother. 2019, 116, 108999. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Manach, C. Bioavailability and Bioefficacy of Polyphenols in Humans. II. Review of 93 Intervention Studies. Am. J. Clin. Nutr. 2005, 81, 243S–255S. [Google Scholar] [CrossRef]
- Gosse, F.; Guyot, S.; Roussi, S.; Lobstein, A.; Fischer, B.; Seiler, N.; Raul, F. Chemopreventive properties of apple procyanidins on human colon cancer-derived metastatic SW620 cells and in a rat model of colon carcinogenesis. Carcinogenesis 2005, 26, 1291–1295. [Google Scholar] [CrossRef] [Green Version]
- Pierini, R.; Kroon, P.A.; Guyot, S.; Ivory, K.; Johnson, I.T.; Belshaw, N.J. Procyanidin Effects on Oesophageal Adenocarcinoma Cells Strongly Depend on Flavan-3-Ol Degree of Polymerization. Mol. Nutr. Food Res. 2008, 52, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Butkeviciute, A.; Petrikaite, V.; Jurgaityte, V.; Liaudanskas, M.; Janulis, V. Antioxidant, Anti-Inflammatory, and Cytotoxic Activity of Extracts from Some Commercial Apple Cultivars in Two Colorectal and Glioblastoma Human Cell Lines. Antioxidants 2021, 10, 1098. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.; Kumar, S.; Rana, A.; Padwad, Y.; Bhushan, S. Biological Activity of Phenolics Enriched Extracts from Industrial Apple Pomace. Ind. Crops Prod. 2021, 160, 113158. [Google Scholar] [CrossRef]
- Lokina, S.; Stephen, A.; Kaviyarasan, V.; Arulvasu, C.; Narayanan, V. Cytotoxicity and Antimicrobial Activities of Green Synthesized Silver Nanoparticles. Eur. J. Med. Chem. 2014, 76, 256–263. [Google Scholar] [CrossRef]
- Mariadoss, A.; Ramachandran, V.; Shalini, V.; Agilan, B.; Franklin, J.H.; Sanjay, K.; Alaa, Y.G.; Tawfiq, M.A.; Ernest, D. Green synthesis, characterization and antibacterial activity of silver nanoparticles by Malus domestica and its cytotoxic effect on (MCF-7) cell line. Microb. Pathog. 2019, 135, 103609. [Google Scholar] [CrossRef]
- Li, C.X.; Zhao, X.H.; Zuo, W.F.; Zhang, T.L.; Zhang, Z.Y.; Chen, X.S. Phytochemical Profiles, Antioxidant, and Antiproliferative Activities of Four Red-fleshed Apple Varieties in China. J. Food Sci. 2020, 85, 718–726. [Google Scholar] [CrossRef]
- Han, M.; Li, G.; Liu, X.; Li, A.; Mao, P.; Liu, P.; Li, H. Phenolic Profile, Antioxidant Activity and Anti-Proliferative Activity of Crabapple Fruits. Hortic. Plant. J. 2019, 5, 155–163. [Google Scholar] [CrossRef]
- Liang, X.; Zhu, T.; Yang, S.; Li, X.; Song, B.; Wang, Y.; Lin, Q.; Cao, J. Analysis of Phenolic Components and Related Biological Activities of 35 Apple (Malus pumila Mill.) Cultivars. Molecules 2020, 25, 4153. [Google Scholar] [CrossRef] [PubMed]
- Mahavorasirikul, W.; Viyanant, V.; Chaijaroenkul, W.; Itharat, A.; Na-Bangchang, K. Cytotoxic Activity of Thai Medicinal Plants against Human Cholangiocarcinoma, Laryngeal and Hepatocarcinoma Cells in Vitro. BMC Complementary Altern. Med. 2010, 10, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, L.S.; Phung, S.; Yee, N.; Seeram, N.P.; Li, L.; Chen, S. Blueberry Phytochemicals Inhibit Growth and Metastatic Potential of MDA-MB-231 Breast Cancer Cells through Modulation of the Phosphatidylinositol 3-Kinase Pathway. Cancer Res. 2010, 70, 3594–3605. [Google Scholar] [CrossRef] [Green Version]
- Chatelain, K.; Phippen, S.; McCabe, J.; Teeters, C.A.; O’Malley, S.; Kingsley, K. Cranberry and Grape Seed Extracts Inhibit the Proliferative Phenotype of Oral Squamous Cell Carcinomas. Evid. Based Complementary Altern. Med. 2011, 2011, 467691. [Google Scholar] [CrossRef] [Green Version]
- Kaur, M.; Agarwal, R.; Agarwal, C. Grape Seed Extract Induces Anoikis and Caspase-Mediated Apoptosis in Human Prostate Carcinoma LNCaP Cells: Possible Role of Ataxia Telangiectasia Mutated–P53 Activation. Mol. Cancer Ther. 2006, 5, 1265–1274. [Google Scholar] [CrossRef] [Green Version]
- Kaur, M.; Mandair, R.; Agarwal, R.; Agarwal, C. Grape Seed Extract Induces Cell Cycle Arrest and Apoptosis in Human Colon Carcinoma Cells. Nutr. Cancer 2008, 60, 2–11. [Google Scholar] [CrossRef] [Green Version]
- Dinicola, S.; Cucina, A.; Pasqualato, A.; D’Anselmi, F.; Proietti, S.; Lisi, E.; Pasqua, G.; Antonacci, D.; Bizzarri, M. Antiproliferative and Apoptotic Effects Triggered by Grape Seed Extract (GSE) versus Epigallocatechin and Procyanidins on Colon Cancer Cell Lines. Int. J. Mol. Sci. 2012, 13, 651–664. [Google Scholar] [CrossRef]
- Owczarek, K.; Hrabec, E.; Fichna, J.; Sosnowska, D.; Koziołkiewicz, M.; Szymański, J.; Lewandowska, U. Flavanols from Japanese Quince (Chaenomeles japonica) Fruit Suppress Expression of Cyclooxygenase-2, Metalloproteinase-9, and Nuclear Factor-KappaB in Human Colon Cancer Cells. Acta Biochim. Pol. 2017, 64, 567–576. [Google Scholar] [CrossRef]
- Quesada, M.S.; Azofeifa, G.; Campone, L.; Pagano, I.; Pérez, A.M.; Cortés, C.; Rastrelli, L.; Quesada, S. Bactris guineensis (Arecaceae) Extract: Polyphenol Characterization, Antioxidant Capacity and Cytotoxicity against Cancer Cell Lines. J. Berry Res. 2020, 10, 329–344. [Google Scholar] [CrossRef]
- Minker, C.; Duban, L.; Karas, D.; Järvinen, P.; Lobstein, A.; Muller, C.D. Impact of Procyanidins from Different Berries on Caspase 8 Activation in Colon Cancer. Oxid. Med. Cell. Longev. 2015, 2015, 154164. [Google Scholar] [CrossRef] [PubMed]
- Lizarraga, D.; Lozano, C.; Briedé, J.J.; van Delft, J.H.; Touriño, S.; Centelles, J.J.; Torres, J.L.; Cascante, M. The Importance of Polymerization and Galloylation for the Antiproliferative Properties of Procyanidin-Rich Natural Extracts: Antiproliferative Properties of Natural Extracts. FEBS J. 2007, 274, 4802–4811. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Gao, J.; Hou, R.; Xu, X.; Yang, N.; Huang, S. Grape Seed Proanthocyanidins Inhibit Migration and Invasion of Bladder Cancer Cells by Reversing EMT through Suppression of TGF-β Signaling Pathway. Oxid. Med. Cell. Longev. 2021, 2021, 5564312. [Google Scholar] [CrossRef]
- Liu, W.; Qi, Y.; Liu, L.; Tang, Y.; Wei, J.; Zhou, L. Suppression of Tumor Cell Proliferation by Quinine via the Inhibition of the Tumor Necrosis Factor Receptor-Associated Factor 6-AKT Interaction. Mol. Med. Rep. 2016, 14, 2171–2179. [Google Scholar] [CrossRef] [PubMed]
- Miura, T.; Chiba, M.; Kasai, K.; Nozaka, H.; Nakamura, T.; Shoji, T.; Kanda, T.; Ohtake, Y.; Sato, T. Apple Procyanidins Induce Tumor Cell Apoptosis through Mitochondrial Pathway Activation of Caspase-3. Carcinogenesis 2008, 29, 585–593. [Google Scholar] [CrossRef]
- Navarro, M.; Sanchez, F.; Murillo, R.; Martín, P.; Zamora, W.; Monagas, M.; Bartolomé, B. Phenolic assesment of Uncaria tomentosa L. (Cat’s Claw): Leaves, stem, bark and wood extracts. Molecules 2015, 20, 22703–22717. [Google Scholar] [CrossRef] [Green Version]
- Singleton, V.; Rossi, J. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Navarro, M.; Arnaez, E.; Moreira, I.; Quesada, S.; Azofeifa, G.; Wilhelm, K.; Vargas, F.; Chen, P. Polyphenolic Characterization, Antioxidant, and Cytotoxic Activities of Mangifera indica Cultivars from Costa Rica. Foods 2019, 8, 384. [Google Scholar] [CrossRef] [Green Version]
- Navarro, M.; Arnaez, E.; Moreira, I.; Hurtado, A.; Monge, D.; Monagas, M. Polyphenolic Composition and Antioxidant Activity of Uncaria tomentosa Commercial Bark Products. Antioxidants 2019, 8, 339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scalzo, J.; Politi, A.; Pellegrini, N.; Mezzetti, B.; Battino, M. Plant genotype affects total antioxidant capacity and phenolic contents in fruit. Nutrition 2005, 21, 207–213. [Google Scholar] [CrossRef]
- Monagas, M.; Hernandez, B.; Garrido, I.; Martin-Álvarez, P.J.; Gomez, C.; Bartolomé, B. Quality assessment of commercial dietary antioxidant products from Vitis vinifera L. grape seeds. Nutr. Cancer 2005, 53, 244–254. [Google Scholar] [CrossRef] [PubMed]





| Sample | Lyophilization Yield (%) 1,2 | Extraction Yield (mg Extract/g DM) 2,3 | Total Phenolic Content (mg GAE/g Extract) 2,4 |
|---|---|---|---|
| Anna | |||
| Skin | 22.95 ± 0.21 | 16.47 ± 0.62 | 472.26 a ± 5.5 |
| Flesh | 15.92 ± 0.45 | 10.69 ± 0.34 | 354.46 b ± 8.4 |
| Jonagold | |||
| Skin | 19.94 ± 0.68 | 20.87 ± 0.55 | 417.07 c ± 12.3 |
| Flesh | 14.03 ± 0.53 | 14.18 ± 0.84 | 608.78 d ± 4.4 |
| No | Tentative Identification | Rt (min) | [M-H]− | Formula | MS2 Fragments | Sample 1 |
|---|---|---|---|---|---|---|
| Hydroxycinamic acids | ||||||
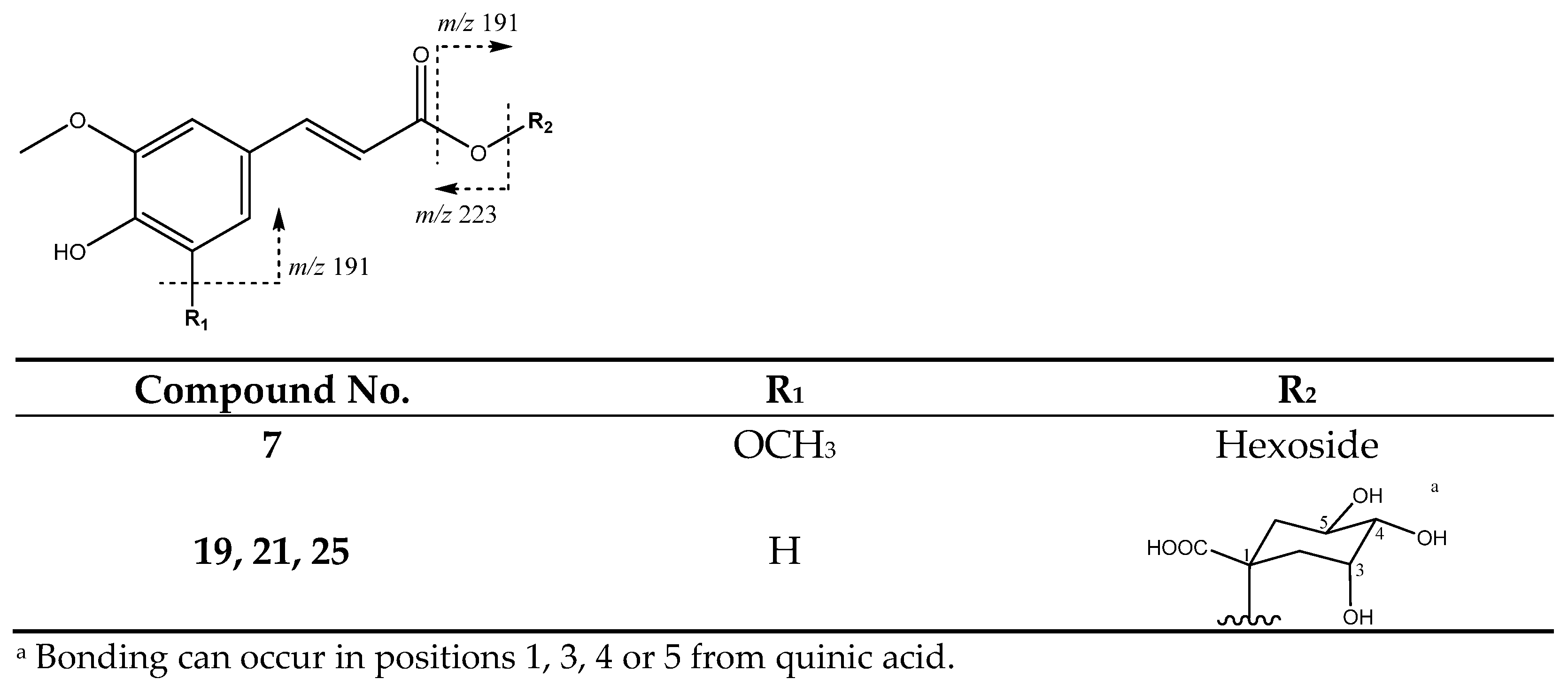
| 7 | Sinapic acid hexoside | 8.91 | 385.1169 | C17H22O10 | [385]: 205, 223 | JF |
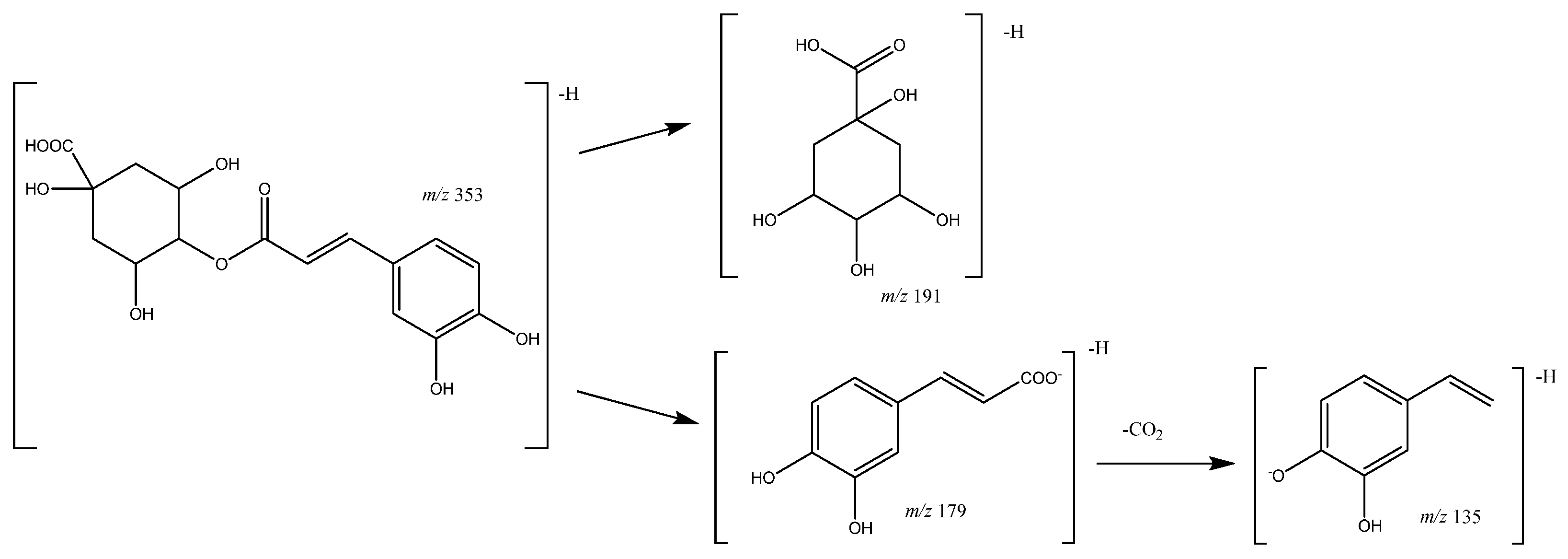
| 10 | Caffeoylquinic acid (I of II) | 10.36 | 353.0871 | C16H17O9 | [353]: 191, 179 | JF |
| 14 | Caffeoylquinic acid (II of II) | 12.45 | 353.0809 | C16H17O9 | [353]: 191, 180 | JF |
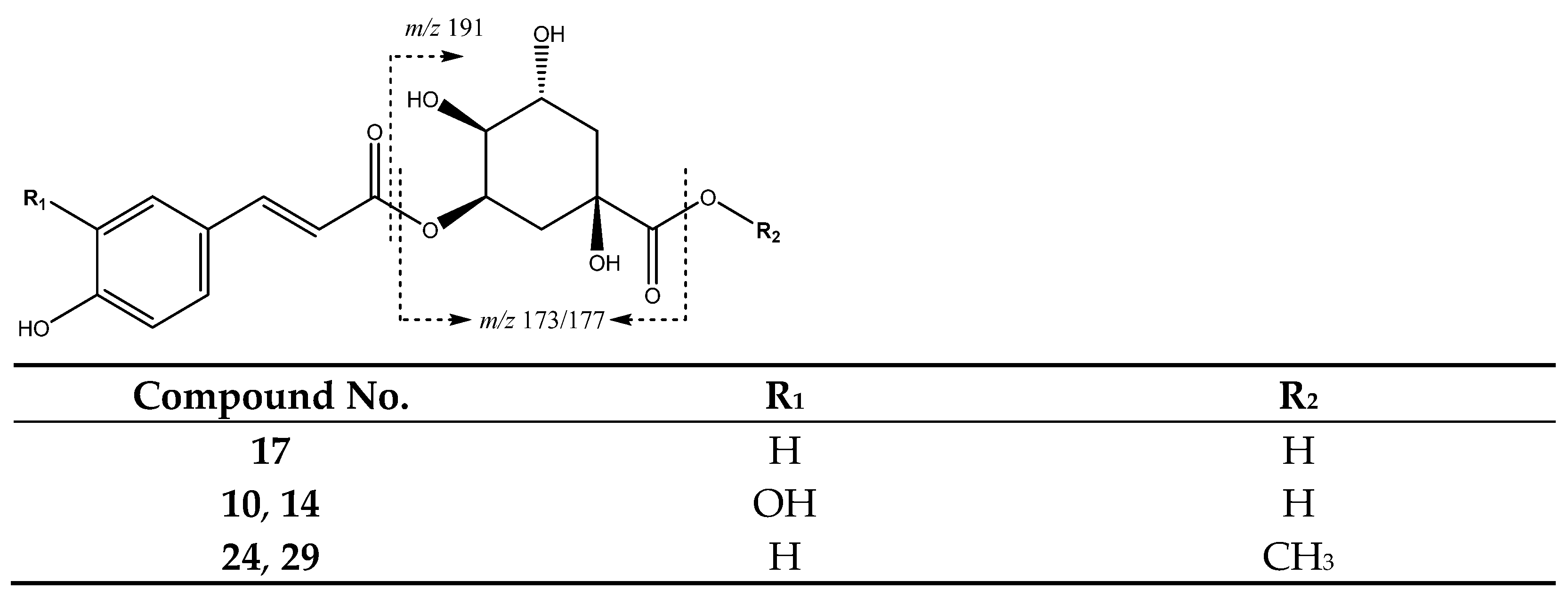
| 17 | p-Coumaroylquinic acid | 14.55 | 337.0912 | C16H18O8 | [337]: 173 | AF, JF |
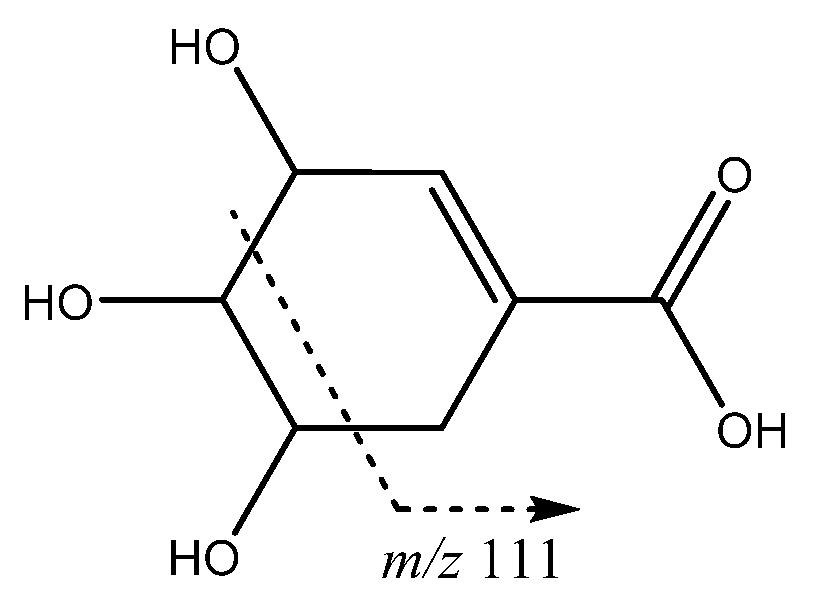
| 18 | Shikimic acid | 15.06 | 173.0447 | C7H9O5 | [173]: 93, 111 | AF |
| 19 | Feruloylquinic acid (I of III) | 15.75 | 367.0983 | C17H20O9 | [367]: 173, 191 | AS, AF |
| 21 | Feruloylquinic acid (II of III) | 18.62 | 367.1012 | C17H20O9 | [367]: 173, 191 | AS, AF, JS, JF |
| 24 | Methyl-p-coumaroylquinic acid (I of II) | 20.82 | 351.1098 | C17H20O9 | [351]: 177 | AS, AF |
| 25 | Feruloylquinic acid (III of III) | 21.60 | 367.0983 | C17H20O9 | [367]: 173, 191 | AS, AF, JF |
| 29 | Methyl-p-coumaroylquinic acid (II of II) | 23.60 | 351.1098 | C17H19O8 | [351]: 177 | AF |
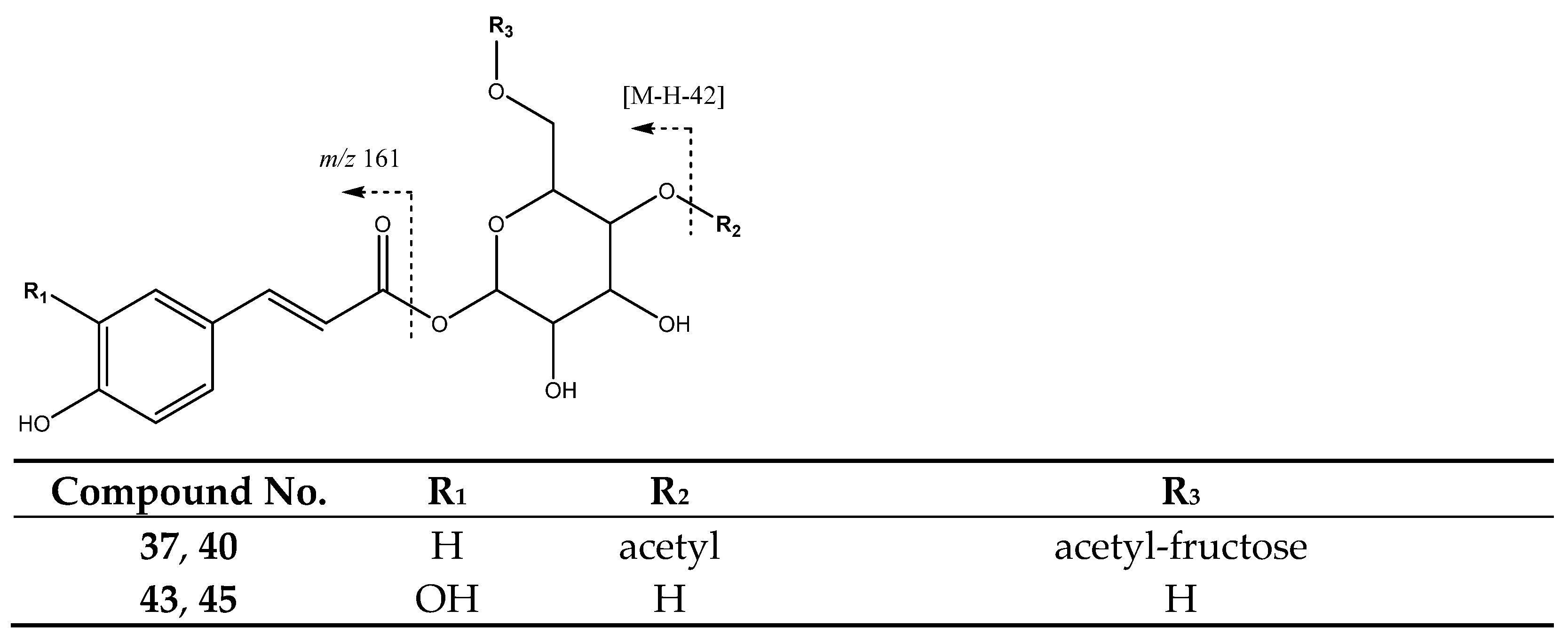
| 37 | Di-O-acetyl-O-p-coumaroylsucrose (I of II) | 28.14 | 571.1675 | C25H31O15 | [571]: 529, 553 | JF |
| 40 | Di-O-acetyl-O-p-coumaroylsucrose (II of II) | 29.88 | 571.1673 | C25H31O15 | [571]: 529, 554 | JS, JF |
| 43 | Caffeoyl hexoside (I of II) | 31.45 | 341.084 | C15H17O9 | [341]: 161, 179 | AS, AF, JS, JF |
| 45 | Caffeoyl hexoside (II of II) | 34.65 | 341.084 | C15H17O9 | [341]: 161, 179 | AS, AF, JS |
| Chalcones | ||||||
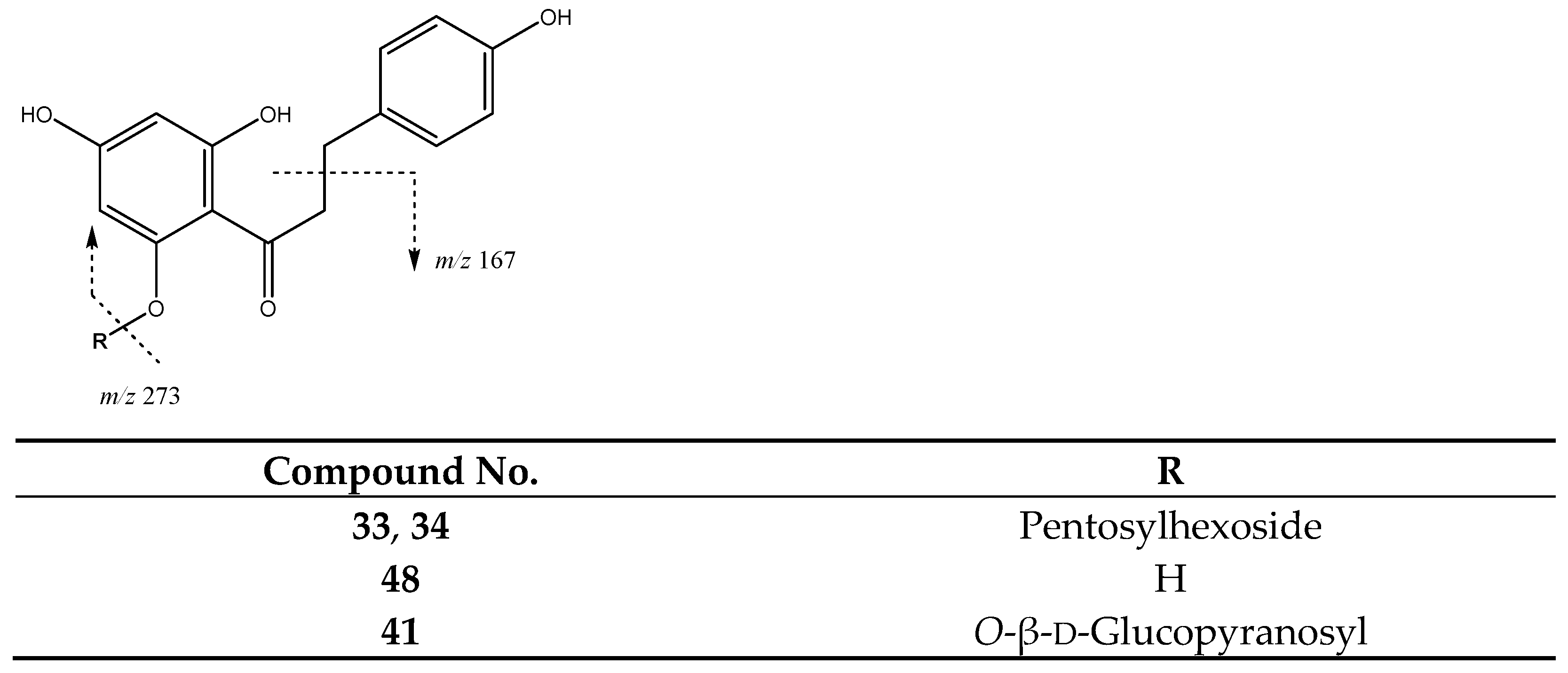
| 33 | Phloretin-pentosilhexoside (I of II) | 26.14 | 567.1725 | C26H31O14 | [567]: 273 | AS, AF, JS, JF |
| 34 | Phloretin-pentosilhexoside (II of II) | 26.95 | 567.1725 | C26H31O14 | [567]: 273 | JF |
| 41 | Phloridzin | 30.00 | 435.1312 | C21H24O10 | [435]: 167, 273 | AS, AF, JS, JF |
| 48 | Phloretin | 40.98 | 273.0757 | C15H13O5 | [273]: 167, 201 | AS, AF, JS |
| Flavonoids | ||||||
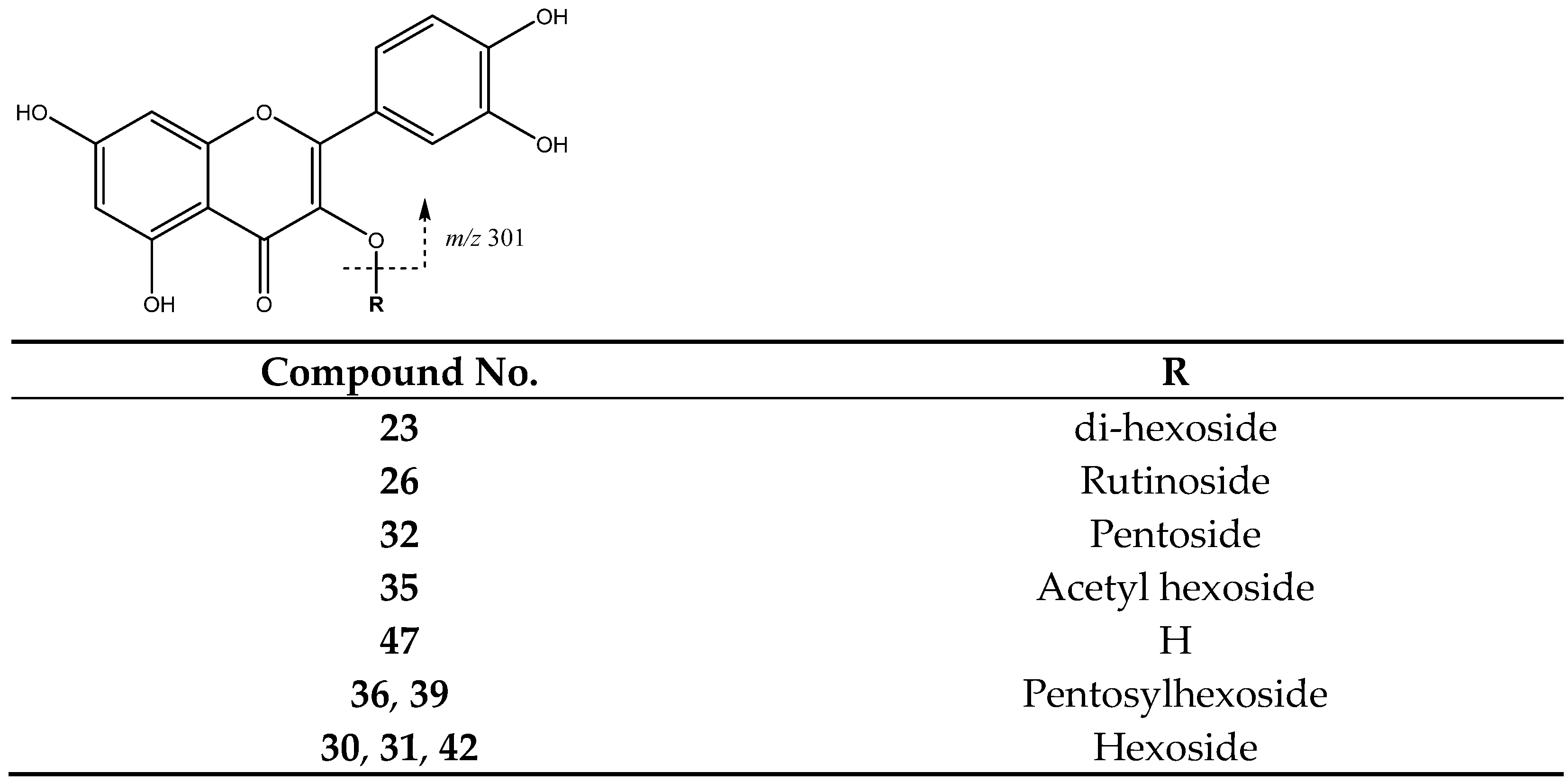
| 23 | Quercetin di-hexoside | 19.82 | 625.1378 | C30H25O13 | [625]: 300, 301 | AS, JS, JF |
| 26 | Quercetin-rutinoside | 22.54 | 609.1459 | C27H29O16 | [609]:300, 301 | AS, JS, JF |
| 30 | Quercetin-hexoside (I of III) | 23.78 | 463.0878 | C21H19O12 | [463]: 300, 301 | AS, JS, JF |
| 31 | Quercetin-hexoside (II of III) | 24.95 | 463.0878 | C21H19O12 | [463]: 300, 301 | AS, AF |
| 32 | Quercetin-pentoside | 25.35 | 433.0732 | C20H17O11 | [433]: 300, 301 | AS, AF, JF |
| 35 | Quercetin acetyl hexoside | 27.16 | 505.1002 | C23H22O13 | [505]: 300, 301 | AS, JS, JF |
| 36 | Quercetin-pentosylhexoside | 27.77 | 595.1245 | C26H27O16 | [595]: 300, 301 | JF |
| 38 | Kaempferol-hexoside | 28.63 | 447.0928 | C21H19O11 | [447]: 284.,285 | AS, AF, JS, JF |
| 39 | Quercetin pentosyl hexoside | 29.24 | 587.1043 | C25H26O15 | [595]: 300, 301 | JS, JF |
| 42 | Quercetin-hexoside (III of III) | 31.25 | 463.0834 | C21H20O12 | [463]: 300, 301 | AS, AF, JS, JF |
| 46 | Kaempferol-hexoside | 36.18 | 477.0944 | C21H20O11 | [447]: 284, 285 | AS, AF, JS |
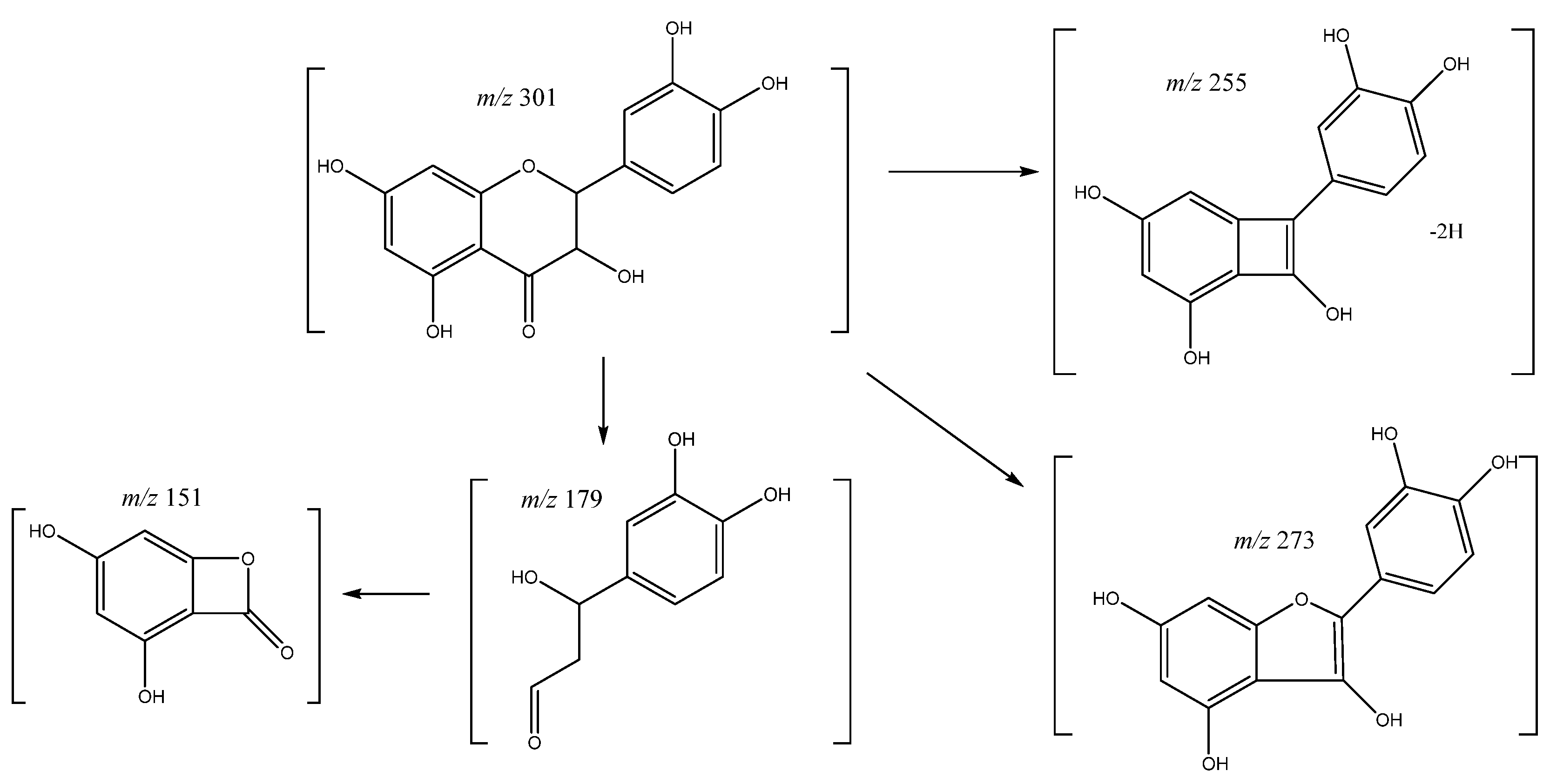
| 47 | Quercetin | 37.19 | 301.0353 | C15H10O7 | [301]: 151, 179, 255, 273, 283 | AS, JS |
| Proantocyanidins | ||||||
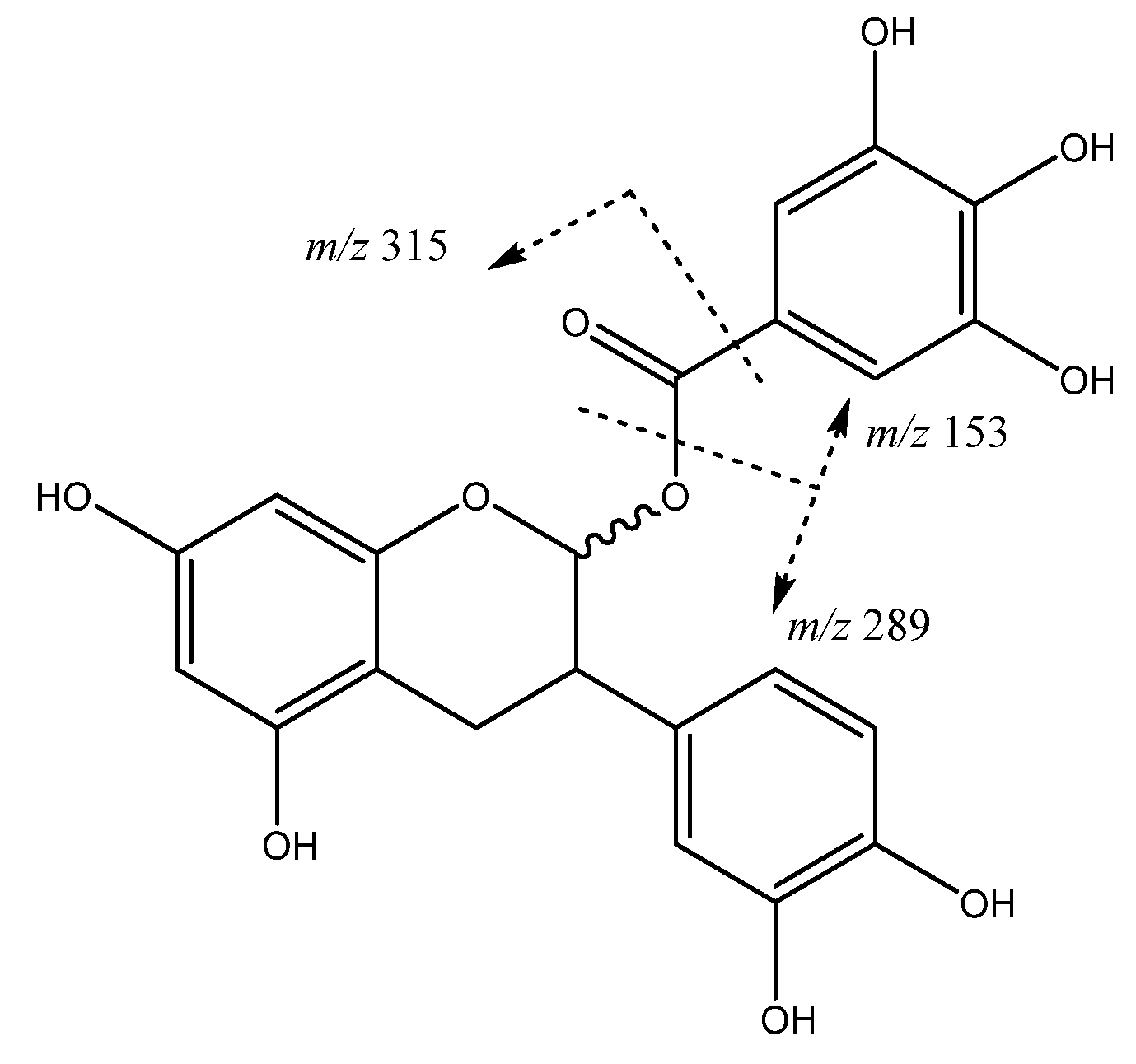
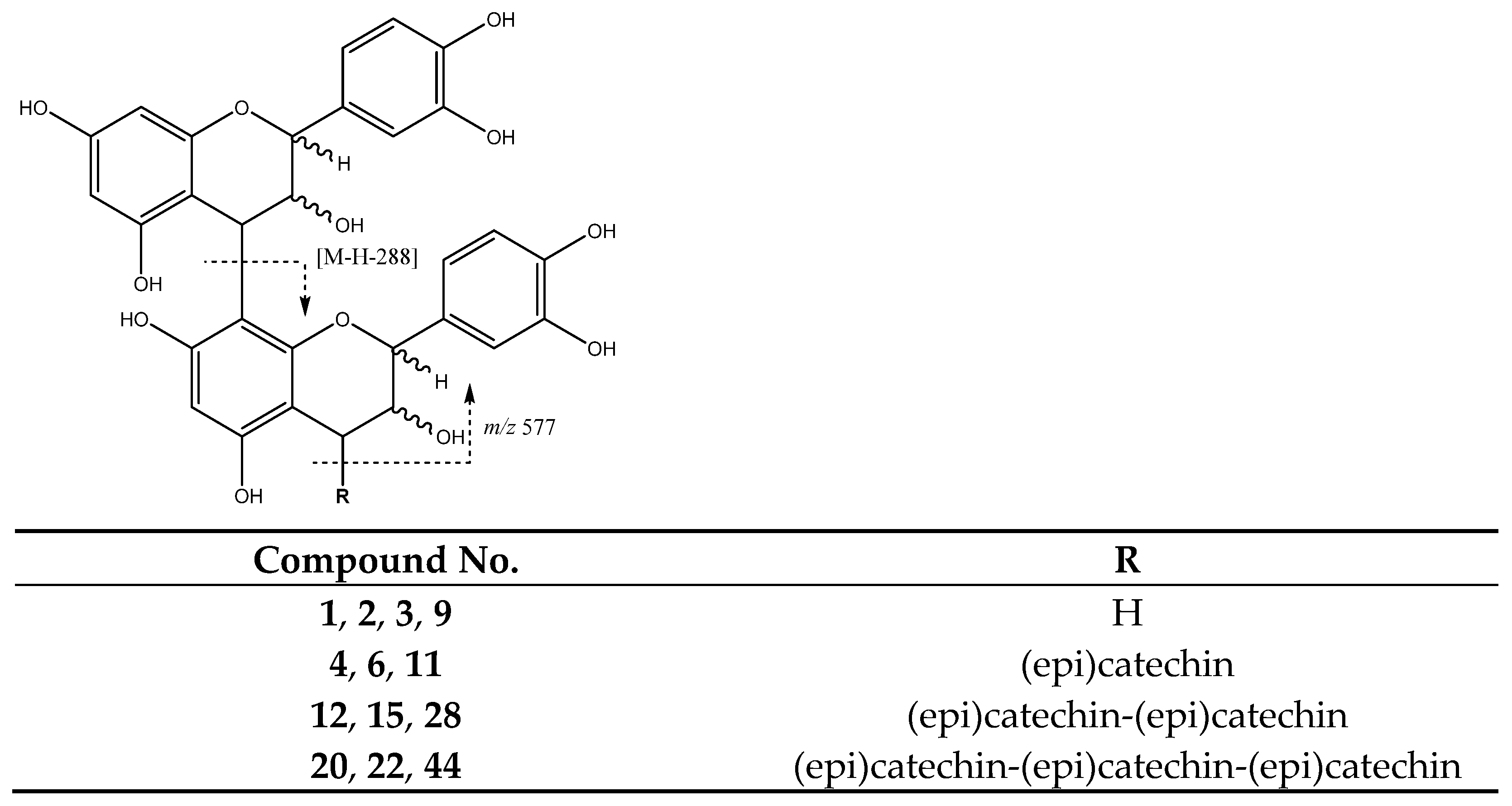
| 1 | Procyanidin B-type dimer (I of IV) | 5.74 | 577.1292 | C30H25O12 | [577]: 287, 289, 407, 425, 451, 559 | JS, JF |
| 2 | Procyanidin B-type dimer (II of IV) | 6.12 | 577.1294 | C30H25O12 | [577]: 287, 289, 407, 425, 451, 559 | JF |
| 3 | Procyanidin B-type dimer (III of IV) | 6.57 | 577.1343 | C30H25O12 | [577]: 287, 289, 407, 425, 451, 559 | AS, AF, JF |
| 4 | Procyanidin B-type trimer (I of III) | 7.31 | 865.2004 | C45H37O18 | [865]: 287, 289, 575, 577, 695, 713, 739 | JF |
| 5 | Procyanidin A-type dimer | 7.67 | 591.1147 | C30H24O13 | [575]: 289, 449 | AF, JS |
| 6 | Procyanidin B-type trimer (II of III) | 8.5 | 865.2004 | C45H37O18 | [865]: 287, 289, 575, 577, 695, 713, 739 | AS, JF |
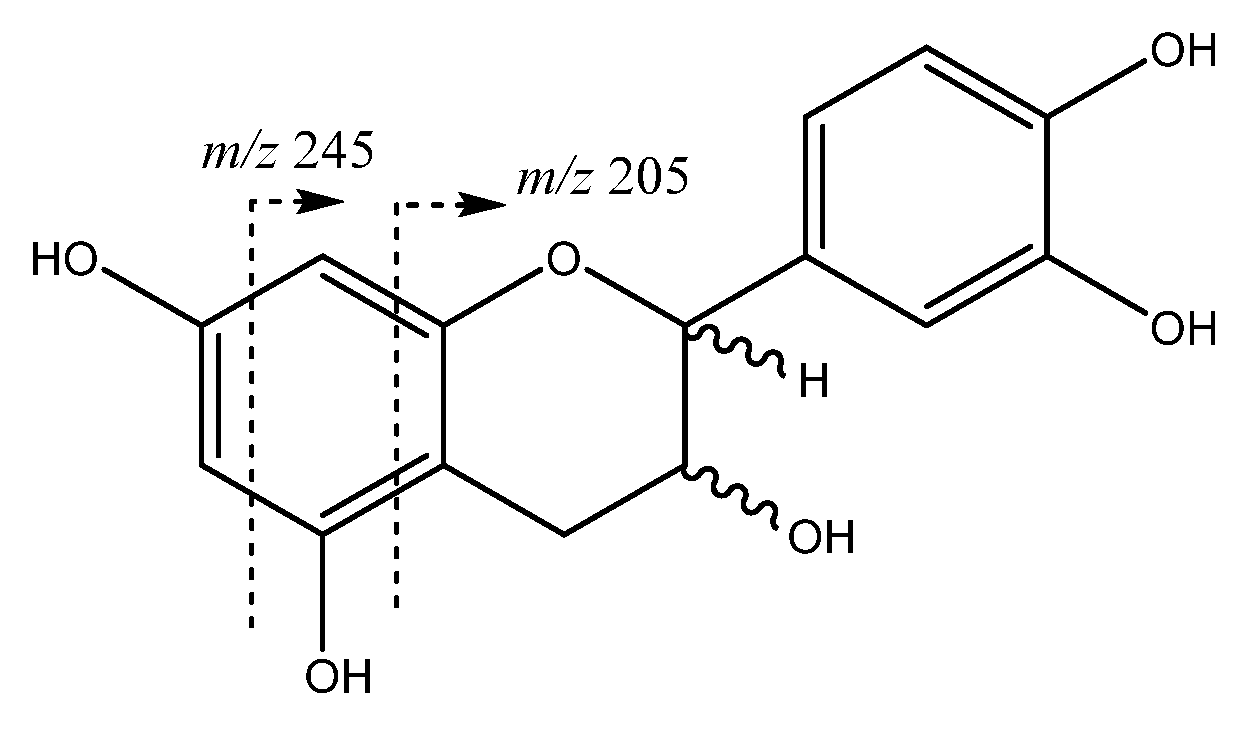
| 8 | Catechin | 9.01 | 289.0708 | C15H14O6 | [289]: 205,245, 271 | AS, JF |
| 9 | Procyanidin B-type dimer (IV of IV) | 9.52 | 577.1392 | C30H25O12 | [577]: 287, 289, 407, 425, 451, 559 | AS, AF, JF |
| 11 | Procyanidin B-type trimer (III of III) | 11.48 | 865.2004 | C45H37O18 | [865]: 287, 289, 575, 577, 695, 713, 739 | AS, JF |
| 12 | Procyanidin tetramer B (I of III) | 11.76 | 1153.2629 | C60H49O24 | [1153]: 287, 289, 575, 577, 863, 865, 983, 1001, 1027, 1135 | JF |
| 13 | Epicatechin | 12.14 | 289.0708 | C15H13O6 | [289]: 205, 245, 271 | AS, JF |
| 15 | Procyanidin B-type tetramer (II of III) | 12.82 | 1153.2704 | C60H49O24 | [1153]: 287, 289, 575, 577, 863, 865, 983, 1001, 1027, 1135 | JF |
| 20 | Procyanidin B-type pentamer (I of III) | 16.55 | 1441.2936 | C75H61O30 | [1441]: 287, 289, 575, 577, 865, 1153, 1315 | JF |
| 22 | Procyanidin B-type pentamer (II of III) | 19.13 | 1441.2939 | C75H61O30 | [1441]: 287, 289, 575, 577, 865, 1153, 1315 | JS, JF |
| 27 | (epi)catechin 3-O-gallate | 22.89 | 609.1459 | C27H29O16 | [441]: 153, 289, 315 | JF |
| 28 | Procyanidin B-type tetramer (III of III) | 23.20 | 1153.2701 | C60H49O24 | [1153]: 287, 289, 575, 577, 863, 865, 983, 1001, 1027, 1135 | AS, JS, JF |
| 44 | Procyanidin B-type pentamer (III of III) | 32.05 | 1441.2931 | C75H61O30 | [1441]: 287, 289, 575, 577, 865, 1153, 1315 | JF |
| Others | ||||||
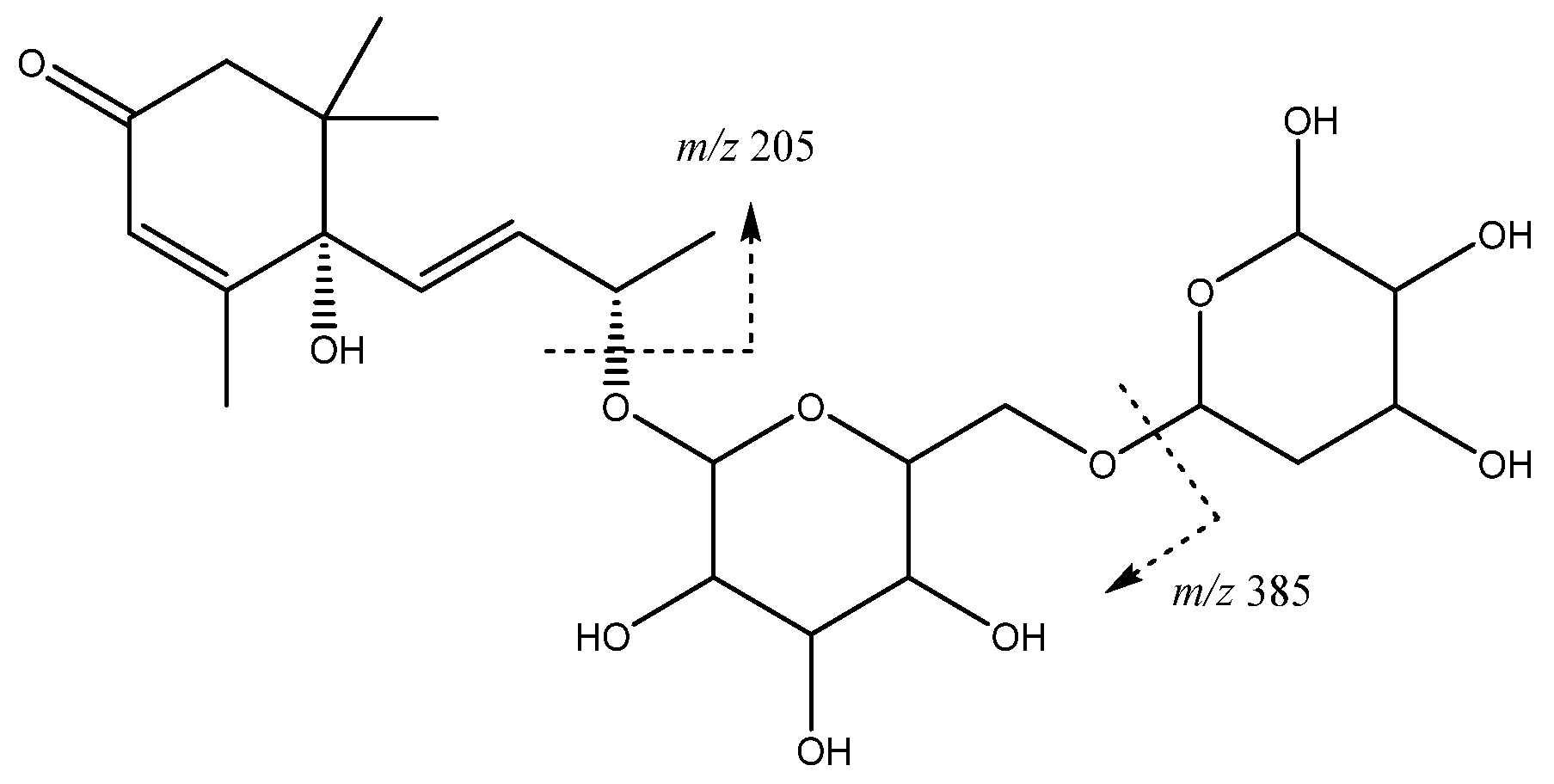
| 16 | Vomifoliol-pentosilhexoside | 13.97 | 517.2293 | C24H37O12 | [517]: 205, 385 | AS, JS, JF |
| Sample | DPPH 1,2 | ORAC 1.2 | |
|---|---|---|---|
| IC50 (μg Extract/mL) | (mmol TE/g Extract) | (mmol TE/g Extract) | |
| Anna | |||
| Skin | 6.90 a ± 0.02 | 3.25 a ± 0.01 | 11.19 a ± 0.25 |
| Flesh | 11.33 b ± 0.05 | 1.98 b ± 0.01 | 5.96 b ± 0.23 |
| Jonagold | |||
| Skin | 9.76 c ± 0.17 | 2.30 c ± 0.04 | 7.44 c ± 0.10 |
| Flesh | 3.96 d ± 0.02 | 5.68 d ± 0.03 | 14.80 d ± 0.26 |
| Sample | IC50 (µg/mL) 1,2 (SI) 3 | ||
|---|---|---|---|
| AGS | SW-620 | Vero | |
| Anna | |||
| Skin | 167.22 a,* ± 10 (3.0) | 295.93 b,# ± 29 (1.7) | > 500 a,≠ |
| Flesh | > 500 b,* | > 500 a,* | > 500 a,* |
| Jonagold | |||
| Skin | 398.44 c,* ± 7 (1.3) | > 500 a,* | 305.72.b,* ± 30 |
| Flesh | 60.03 d,* ± 1.7 (5.1) | 62.41 c,* ± 5.2 (4.9) | > 500 a,# |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro-Hoyos, M.; Arnáez-Serrano, E.; Quesada-Mora, S.; Azofeifa-Cordero, G.; Wilhelm-Romero, K.; Quirós-Fallas, M.I.; Alvarado-Corella, D.; Vargas-Huertas, F.; Sánchez-Kopper, A. HRMS Characterization, Antioxidant and Cytotoxic Activities of Polyphenols in Malus domestica Cultivars from Costa Rica. Molecules 2021, 26, 7367. https://doi.org/10.3390/molecules26237367
Navarro-Hoyos M, Arnáez-Serrano E, Quesada-Mora S, Azofeifa-Cordero G, Wilhelm-Romero K, Quirós-Fallas MI, Alvarado-Corella D, Vargas-Huertas F, Sánchez-Kopper A. HRMS Characterization, Antioxidant and Cytotoxic Activities of Polyphenols in Malus domestica Cultivars from Costa Rica. Molecules. 2021; 26(23):7367. https://doi.org/10.3390/molecules26237367
Chicago/Turabian StyleNavarro-Hoyos, Mirtha, Elizabeth Arnáez-Serrano, Silvia Quesada-Mora, Gabriela Azofeifa-Cordero, Krissia Wilhelm-Romero, Maria Isabel Quirós-Fallas, Diego Alvarado-Corella, Felipe Vargas-Huertas, and Andrés Sánchez-Kopper. 2021. "HRMS Characterization, Antioxidant and Cytotoxic Activities of Polyphenols in Malus domestica Cultivars from Costa Rica" Molecules 26, no. 23: 7367. https://doi.org/10.3390/molecules26237367
APA StyleNavarro-Hoyos, M., Arnáez-Serrano, E., Quesada-Mora, S., Azofeifa-Cordero, G., Wilhelm-Romero, K., Quirós-Fallas, M. I., Alvarado-Corella, D., Vargas-Huertas, F., & Sánchez-Kopper, A. (2021). HRMS Characterization, Antioxidant and Cytotoxic Activities of Polyphenols in Malus domestica Cultivars from Costa Rica. Molecules, 26(23), 7367. https://doi.org/10.3390/molecules26237367







