Decontamination-Induced Modification of Bioactivity in Essential Oil-Based Plasma Polymer Coatings
Abstract
:1. Introduction
2. Results and Discussion
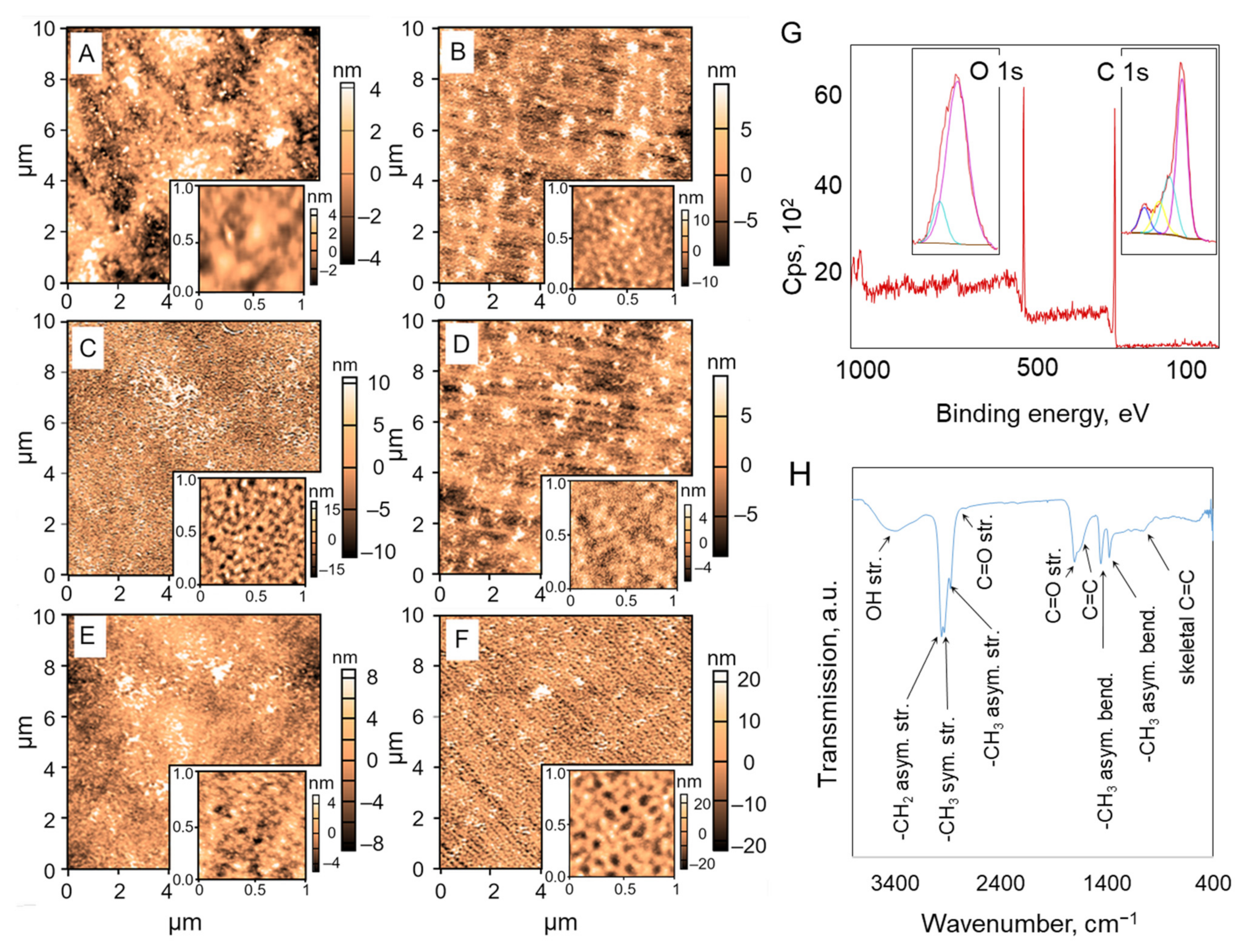
2.1. Surface Properties and Chemical Composition of As-deposited Plasma Polymer Coatings
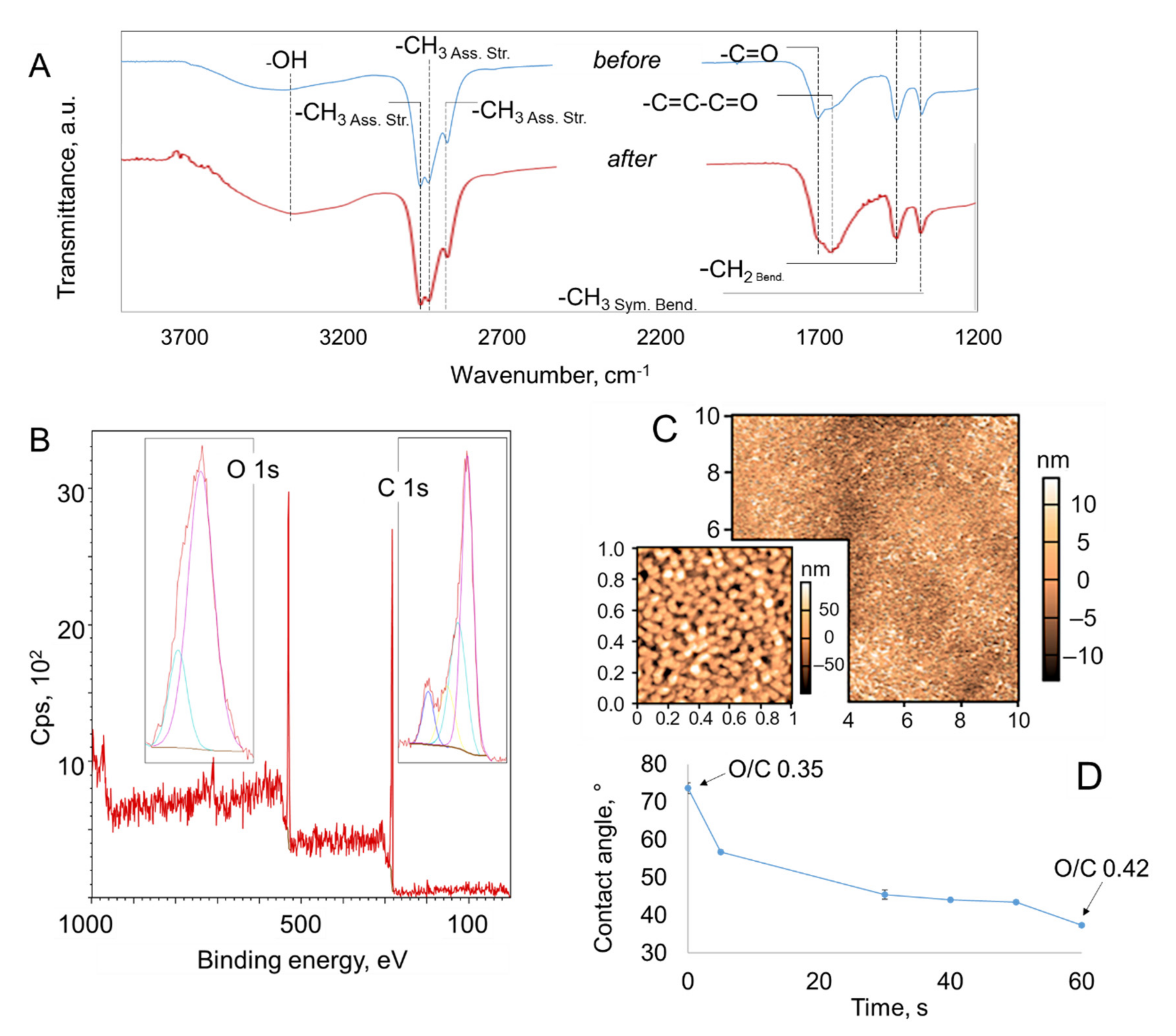
2.2. Effect of Atmospheric Pressure Ar Plasma Treatment on Plasma Polymer Surfaces
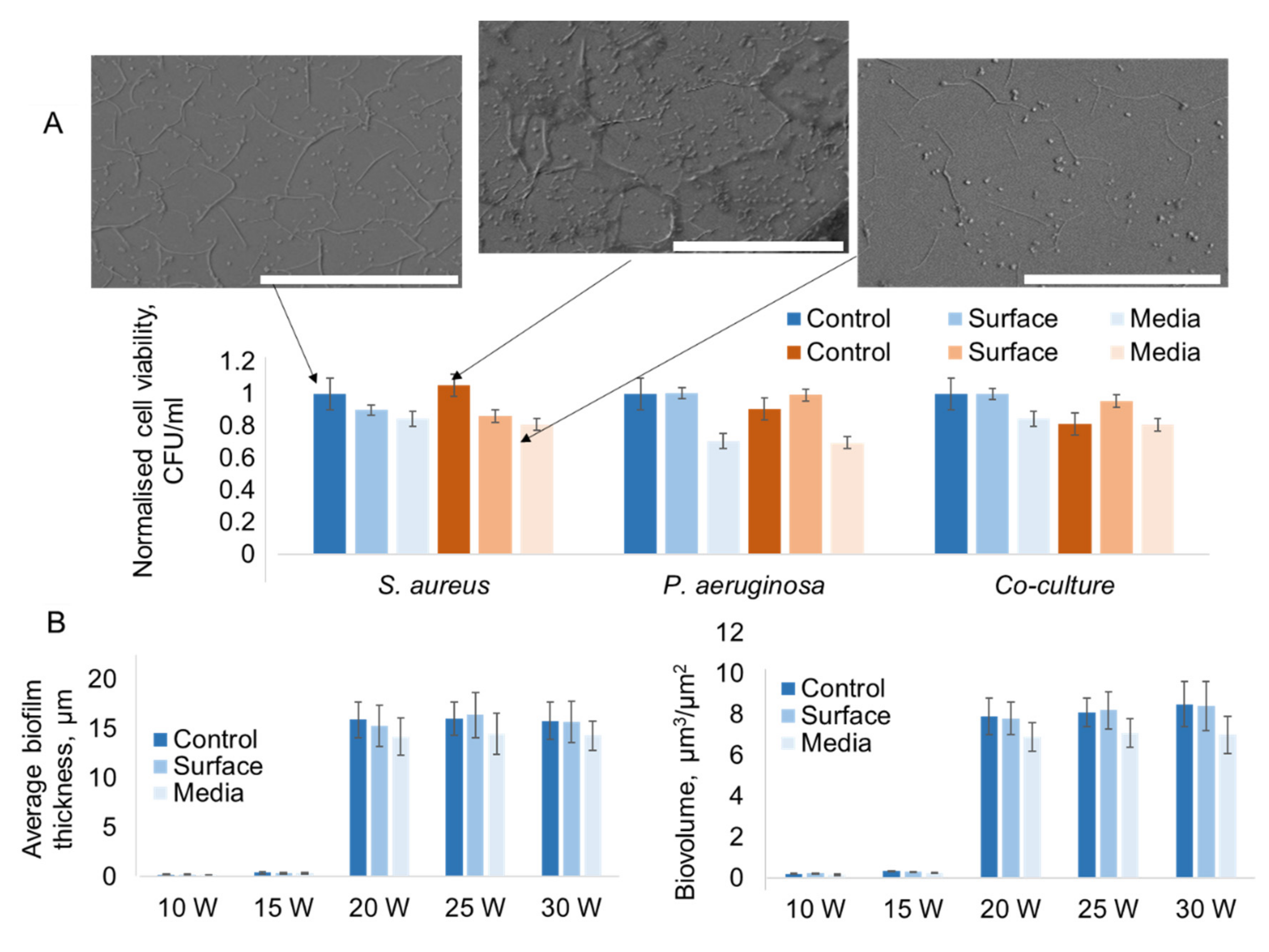
2.3. Effects of Atmospheric Pressure Ar Plasma Treatment on Bacterial Cell Growth on Polymer Coatings
2.3.1. Effect of Substrate Treatment on Attachment of Bacterial Cells
2.3.2. Effect of Plasma-activated Media on Attachment of Bacterial Cells
2.3.3. Effect of Plasma Treatment on Attachment of Mammalian Cells
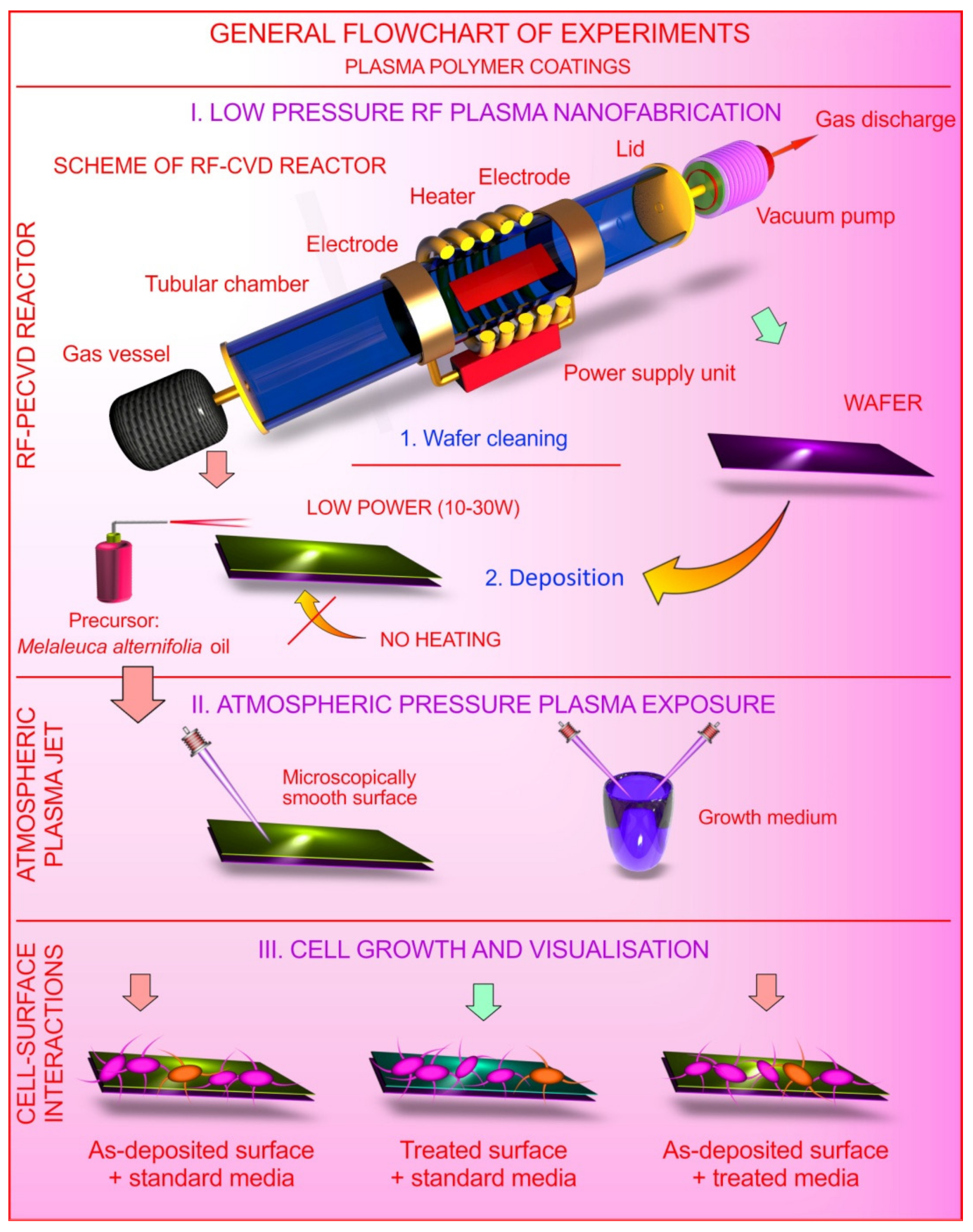
3. Materials and Methods
3.1. Preparation and Characterisation of Polymer Coatings
3.2. Cells, Growth Conditions, Attachment and Viability
3.3. Plasma Treatment of Surfaces and Growth Culture Medium
3.4. Eukaryotic Cell Line, Growth Conditions, Attachment and Viability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Slepička, P.; Rimpelová, S.; Slepičková Kasálková, N.; Fajstavr, D.; Sajdl, P.; Kolská, Z.; Švorčík, V. Antibacterial properties of plasma-activated perfluorinated substrates with silver nanoclusters deposition. Nanomaterials 2021, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- van Hengel, I.A.J.; Tierolf, M.W.A.M.; Fratila-Apachitei, L.E.; Apachitei, I.; Zadpoor, A.A. Antibacterial titanium implants biofunctionalized by plasma electrolytic oxidation with silver, zinc, and copper: A systematic review. Int. J. Mol. Sci. 2021, 22, 3800. [Google Scholar] [CrossRef] [PubMed]
- Griesser, S.S.; Jasieniak, M.; Vasilev, K.; Griesser, H.J. Antimicrobial peptides grafted onto a plasma polymer interlayer platform: Performance upon extended bacterial challenge. Coatings 2021, 11, 68. [Google Scholar] [CrossRef]
- Lee, M.-J.; Kwon, J.-S.; Jiang, H.B.; Choi, E.H.; Park, G.; Kim, K.-M. The antibacterial effect of non-thermal atmospheric pressure plasma treatment of titanium surfaces according to the bacterial wall structure. Sci. Rep. 2019, 9, 1938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benčina, M.; Resnik, M.; Starič, P.; Junkar, I. Use of plasma technologies for antibacterial surface properties of metals. Molecules 2021, 26, 1418. [Google Scholar] [CrossRef]
- Sardella, E.; Palumbo, F.; Camporeale, G.; Favia, P. Non-equilibrium plasma processing for the preparation of antibacterial surfaces. Materials 2016, 9, 515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasilev, K.; Griesser, S.S.; Griesser, H.J. Antibacterial surfaces and coatings produced by plasma techniques. Plasma Process. Polym. 2011, 8, 1010–1023. [Google Scholar] [CrossRef]
- Irfan, M.; Polonskyi, O.; Hinz, A.; Mollea, C.; Bosco, F.; Strunskus, T.; Balagna, C.; Perero, S.; Faupel, F.; Ferraris, M. Antibacterial, highly hydrophobic and semi transparent Ag/plasma polymer nanocomposite coating on cotton fabric obtained by plasma based co-deposition. Cellulose 2019, 26, 8877–8894. [Google Scholar] [CrossRef]
- Akhavan, B.; Bakhshandeh, S.; Najafi-Ashtiani, H.; Fluit, A.C.; Boel, E.; Vogely, C.; van der Wal, B.C.H.; Zadpoor, A.A.; Weinans, H.; Hennink, W.E.; et al. Direct covalent attachment of silver nanoparticles on radical-rich plasma polymer films for antibacterial applications. J. Mater. Chem. B 2018, 6, 5845–5853. [Google Scholar] [CrossRef]
- Dimitrakellis, P.; Ellinas, K.; Kaprou, G.D.; Mastellos, D.C.; Tserepi, A.; Gogolides, E. Bactericidal action of smooth and plasma micro-nanotextured polymeric surfaces with varying wettability, enhanced by incorporation of a biocidal agent. Macromol. Mater. Eng. 2021, 306, 2000694. [Google Scholar] [CrossRef]
- Cavallaro, A.A.; Macgregor-Ramiasa, M.N.; Vasilev, K. Antibiofouling properties of plasma-deposited oxazoline-based thin films. ACS Appl. Mater. Interfaces 2016, 8, 6354–6362. [Google Scholar] [CrossRef] [PubMed]
- Bazaka, O.; Bazaka, K.; Truong, V.K.; Levchenko, I.; Jacob, M.V.; Estrin, Y.; Lapovok, R.; Chichkov, B.; Fadeeva, E.; Kingshott, P.; et al. Effect of titanium surface topography on plasma deposition of antibacterial polymer coatings. Appl. Surf. Sci. 2020, 521, 146375. [Google Scholar] [CrossRef]
- Kumar, A.; Al-Jumaili, A.; Prasad, K.; Bazaka, K.; Mulvey, P.; Warner, J.; Jacob, M.V. Pulse plasma deposition of terpinen-4-ol: An insight into polymerization mechanism and enhanced antibacterial response of developed thin films. Plasma Chem. Plasma Process. 2020, 40, 339–355. [Google Scholar] [CrossRef]
- Lu, X.; Feng, X.; Werber, J.R.; Chu, C.; Zucker, I.; Kim, J.-H.; Osuji, C.O.; Elimelech, M. Enhanced antibacterial activity through the controlled alignment of graphene oxide nanosheets. Proc. Nat. Acad. Sci. USA 2017, 114, E9793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, K.; Bandara, C.D.; Kumar, S.; Singh, G.P.; Brockhoff, B.; Bazaka, K.; Ostrikov, K.K. Effect of precursor on antifouling efficacy of vertically-oriented graphene nanosheets. Nanomaterials 2017, 7, 170. [Google Scholar] [CrossRef] [Green Version]
- Yick, S.; Mai-Prochnow, A.; Levchenko, I.; Fang, J.; Bull, M.K.; Bradbury, M.; Murphy, A.B.; Ostrikov, K. The effects of plasma treatment on bacterial biofilm formation on vertically-aligned carbon nanotube arrays. RSC Adv. 2015, 5, 5142–5148. [Google Scholar] [CrossRef] [Green Version]
- Mai-Prochnow, A.; Clauson, M.; Hong, J.; Murphy, A.B. Gram positive and Gram negative bacteria differ in their sensitivity to cold plasma. Sci. Rep. 2016, 6, 38610. [Google Scholar] [CrossRef] [Green Version]
- Mitra, S.; Veerana, M.; Choi, E.-H.; Park, G. Effects of pre-treatment using plasma on the antibacterial activity of mushroom surfaces. Foods 2021, 10, 1888. [Google Scholar] [CrossRef]
- Grant, D.S.; Ahmed, J.; Whittle, J.D.; Michelmore, A.; Vasilev, K.; Bazaka, K.; Jacob, M.V. Comparative study of natural terpenoid precursors in reactive plasmas for thin film deposition. Molecules 2021, 26, 4762. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Zhou, R.; Prasad, K.; Fang, Z.; Speight, R.; Bazaka, K.; Ostrikov, K. Cold atmospheric plasma activated water as a prospective disinfectant: The crucial role of peroxynitrite. Green Chem. 2018, 20, 5276–5284. [Google Scholar] [CrossRef]
- Bazaka, K.; Jacob, M.V.; Bowden, B.F. Optical and chemical properties of polyterpenol thin films deposited via plasma-enhanced chemical vapor deposition. J. Mater. Res. 2011, 26, 1018–1025. [Google Scholar] [CrossRef] [Green Version]
- Bazaka, K.; Jacob, M.V.; Truong, V.K.; Wang, F.; Pushpamali, W.A.A.; Wang, J.Y.; Ellis, A.V.; Berndt, C.C.; Crawford, R.J.; Ivanova, E.P. Plasma-enhanced synthesis of bioactive polymeric coatings from monoterpene alcohols: A combined experimental and theoretical study. Biomacromolecules 2010, 11, 2016–2026. [Google Scholar] [CrossRef] [PubMed]
- Bazaka, K.; Jacob, M.V.; Truong, V.K.; Crawford, R.J.; Ivanova, E.P. The effect of polyterpenol thin film surfaces on bacterial viability and adhesion. Polymers 2011, 3, 388–404. [Google Scholar] [CrossRef] [Green Version]
- Bazaka, K.; Jacob, M.V. Post-deposition ageing reactions of plasma derived polyterpenol thin films. Polym. Degrad. Stab. 2010, 95, 1123–1128. [Google Scholar] [CrossRef]
- Bazaka, K.; Ketheesan, N.; Jacob, M.V. Polymer encapsulation of magnesium to control biodegradability and biocompatibility. J. Nanosci. Nanotechnol. 2014, 14, 8087–8093. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Al-Jumaili, A.; Bazaka, K.; Mulvey, P.; Warner, J.; Jacob, M.V. In-situ surface modification of terpinen-4-ol plasma polymers for increased antibacterial activity. Materials 2020, 13, 586. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Mills, S.; Bazaka, K.; Bajema, N.; Atkinson, I.; Jacob, M.V. Biodegradable optically transparent terpinen-4-ol thin films for marine antifouling applications. Surf. Coat. Technol. 2018, 349, 426–433. [Google Scholar] [CrossRef]
- Jiang, H.; Grant, J.T.; Enlow, J.; Su, W.; Bunning, T.J. Surface oxygen in plasma polymerized films. J. Mater. Chem. 2009, 19, 2234–2239. [Google Scholar] [CrossRef]
- Kostov, K.G.; Nishime, T.M.C.; Castro, A.H.R.; Toth, A.; Hein, L.R.O. Surface modification of polymeric materials by cold atmospheric plasma jet. Appl. Surf. Sci. 2014, 314, 367–375. [Google Scholar] [CrossRef] [Green Version]
- Bazaka, K.; Ahmad, J.; Oelgemöller, M.; Uddin, A.; Jacob, M.V. Photostability of plasma polymerized γ-terpinene thin films for encapsulation of OPV. Sci. Rep. 2017, 7, 45599. [Google Scholar] [CrossRef] [Green Version]
- Bazaka, K.; Bazaka, O.; Levchenko, I.; Xu, S.; Ivanova, E.P.; Keidar, M.; Ostrikov, K. Plasma-potentiated small molecules—possible alternative to antibiotics? Nano Futures 2017, 1, 025002. [Google Scholar] [CrossRef] [Green Version]
- Zhou, R.; Zhou, R.; Zhuang, J.; Zong, Z.; Zhang, X.; Liu, D.; Bazaka, K.; Ostrikov, K. Interaction of atmospheric-pressure air microplasmas with amino acids as fundamental processes in aqueous solution. PLoS ONE 2016, 11, e0155584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, R.; Zhou, R.; Wang, P.; Luan, B.; Zhang, X.; Fang, Z.; Xian, Y.; Lu, X.; Ostrikov, K.K.; Bazaka, K. Microplasma bubbles: Reactive vehicles for biofilm dispersal. ACS Appl. Mater. Interfaces 2019, 11, 20660–20669. [Google Scholar] [CrossRef]
- Dai, X.; Bazaka, K.; Richard, D.J.; Thompson, E.W.; Ostrikov, K. The emerging role of gas plasma in oncotherapy. Trends Biotechnol. 2018, 36, 1183–1198. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.K.; Chimire, B.; Li, Y.; Adhikari, M.; Veerana, M.; Kaushik, N.; Jha, N.; Adhikari, B.; Lee, S.-J.; Masur, K.; et al. Biological and medical applications of plasma-activated media, water and solutions. Biol. Chem. 2019, 400, 39–62. [Google Scholar] [CrossRef] [PubMed]
- Schuster, D.I.; Baran, P.S.; Hatch, R.K.; Khan, A.U.; Wilson, S.R. The role of singlet oxygen in the photochemical formation of C60O. Chem. Commun. 1998, 2493–2494. [Google Scholar] [CrossRef]
- Tominami, K.; Kanetaka, H.; Sasaki, S.; Mokudai, T.; Kaneko, T.; Niwano, Y. Cold atmospheric plasma enhances osteoblast differentiation. PLoS ONE 2017, 12, e0180507. [Google Scholar] [CrossRef]
- Bremmell, K.E.; Kingshott, P.; Ademovic, Z.; Winther-Jensen, B.; Griesser, H.J. Colloid probe AFM investigation of interactions between fibrinogen and PEG-like plasma polymer surfaces. Langmuir 2006, 22, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Bazaka, K.; Jacob, M.V. Solubility and surface interactions of RF plasma polymerized polyterpenol thin films. Mater. Express 2012, 2, 285–293. [Google Scholar] [CrossRef]

| PET | 10 W | 15 W | 20 W | 25 W | 30 W | |
|---|---|---|---|---|---|---|
| 1 µm × 1 µm | ||||||
| Rq, nm | 1.07 | 3.19 | 5.98 | 2.20 | 1.73 | 8.97 |
| Rmax, nm | 4.31 | 22.49 | 27.12 | 10.87 | 7.64 | 33.75 |
| Rmin, nm | −3.14 | −7.49 | −18.19 | −6.90 | −6.96 | −28.25 |
| ADv, nm | 0.83 | 2.37 | 4.80 | 1.76 | 1.36 | 7.20 |
| Rsk | 0.39 | 1.38 | 0.28 | 0.39 | 0.26 | 0.06 |
| Rku | 0.47 | 3.33 | 0.04 | 0.13 | 0.32 | -0.15 |
| 10 µm × 10 µm | ||||||
| Rq, nm | 2.08 | 4.72 | 5.19 | 4.07 | 3.21 | 9.18 |
| Rmax, nm | 39.73 | 56.82 | 54.84 | 38.02 | 36.58 | 107.03 |
| Rmin, nm | −6.41 | −14.28 | −20.28 | −11.85 | −10.58 | −31.72 |
| ADv, nm | 1.54 | 3.29 | 3.93 | 2.94 | 2.35 | 6.78 |
| Rsk | 1.80 | 2.12 | 0.93 | 1.57 | 1.34 | 1.29 |
| Rku | 14.20 | 10.10 | 2.77 | 5.73 | 5.14 | 7.12 |
| Atomic fraction, % | ||||||
| O 1s (530 eV) | 25.1 | 26.0 | 22.9 | 14.0 | 21.4 | |
| C 1s (285 eV) | 73.0 | 74.0 | 77.1 | 86.1 | 78.7 | |
| N 1s (397 eV) | 1.9 | |||||
| O/C ratio | 0.34 | 0.35 | 0.30 | 0.16 | 0.27 | |
| Water contact angle, ° | 62.1 ± 2.1 | 66.6 ± 1.9 | 70.3 ± 1.5 | 74.2 ± 1.8 | 77.1 ± 0.8 |
| Before | After | |||
|---|---|---|---|---|
| Surface roughness | ||||
| 1 µm × 1 µm | 10 µm × 10 µm | 1 µm × 1 µm | 10 µm × 10 µm | |
| Rq, nm | 5.98 | 5.19 | 7.33 | 5.45 |
| Rmax, nm | 27.12 | 54.84 | 29.76 | 34.29 |
| Rmin, nm | −18.19 | −20.28 | −22.00 | −18.71 |
| ADv, nm | 4.80 | 3.93 | 5.93 | 4.26 |
| Rsk | 0.28 | 0.93 | 0.38 | 0.63 |
| Rku | 0.04 | 2.77 | −0.09 | 0.91 |
| Atomic fraction, % | ||||
| O 1s (530 eV) | 26.0 | 29.6 | ||
| C 1s (285 eV) | 74.0 | 70.4 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bazaka, O.; Prasad, K.; Levchenko, I.; Jacob, M.V.; Bazaka, K.; Kingshott, P.; Crawford, R.J.; Ivanova, E.P. Decontamination-Induced Modification of Bioactivity in Essential Oil-Based Plasma Polymer Coatings. Molecules 2021, 26, 7133. https://doi.org/10.3390/molecules26237133
Bazaka O, Prasad K, Levchenko I, Jacob MV, Bazaka K, Kingshott P, Crawford RJ, Ivanova EP. Decontamination-Induced Modification of Bioactivity in Essential Oil-Based Plasma Polymer Coatings. Molecules. 2021; 26(23):7133. https://doi.org/10.3390/molecules26237133
Chicago/Turabian StyleBazaka, Olha, Karthika Prasad, Igor Levchenko, Mohan V. Jacob, Kateryna Bazaka, Peter Kingshott, Russell J. Crawford, and Elena P. Ivanova. 2021. "Decontamination-Induced Modification of Bioactivity in Essential Oil-Based Plasma Polymer Coatings" Molecules 26, no. 23: 7133. https://doi.org/10.3390/molecules26237133
APA StyleBazaka, O., Prasad, K., Levchenko, I., Jacob, M. V., Bazaka, K., Kingshott, P., Crawford, R. J., & Ivanova, E. P. (2021). Decontamination-Induced Modification of Bioactivity in Essential Oil-Based Plasma Polymer Coatings. Molecules, 26(23), 7133. https://doi.org/10.3390/molecules26237133









