Application of UHPLC Fingerprints Combined with Chemical Pattern Recognition Analysis in the Differentiation of Six Rhodiola Species
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Sample Preparation
2.2. Optimization of the Chromatographic Conditions
2.3. Methodology Validation
2.4. UHPLC Fingerprint Analysis and Similarity Evaluation
2.5. Chemical Pattern Recognition Analysis
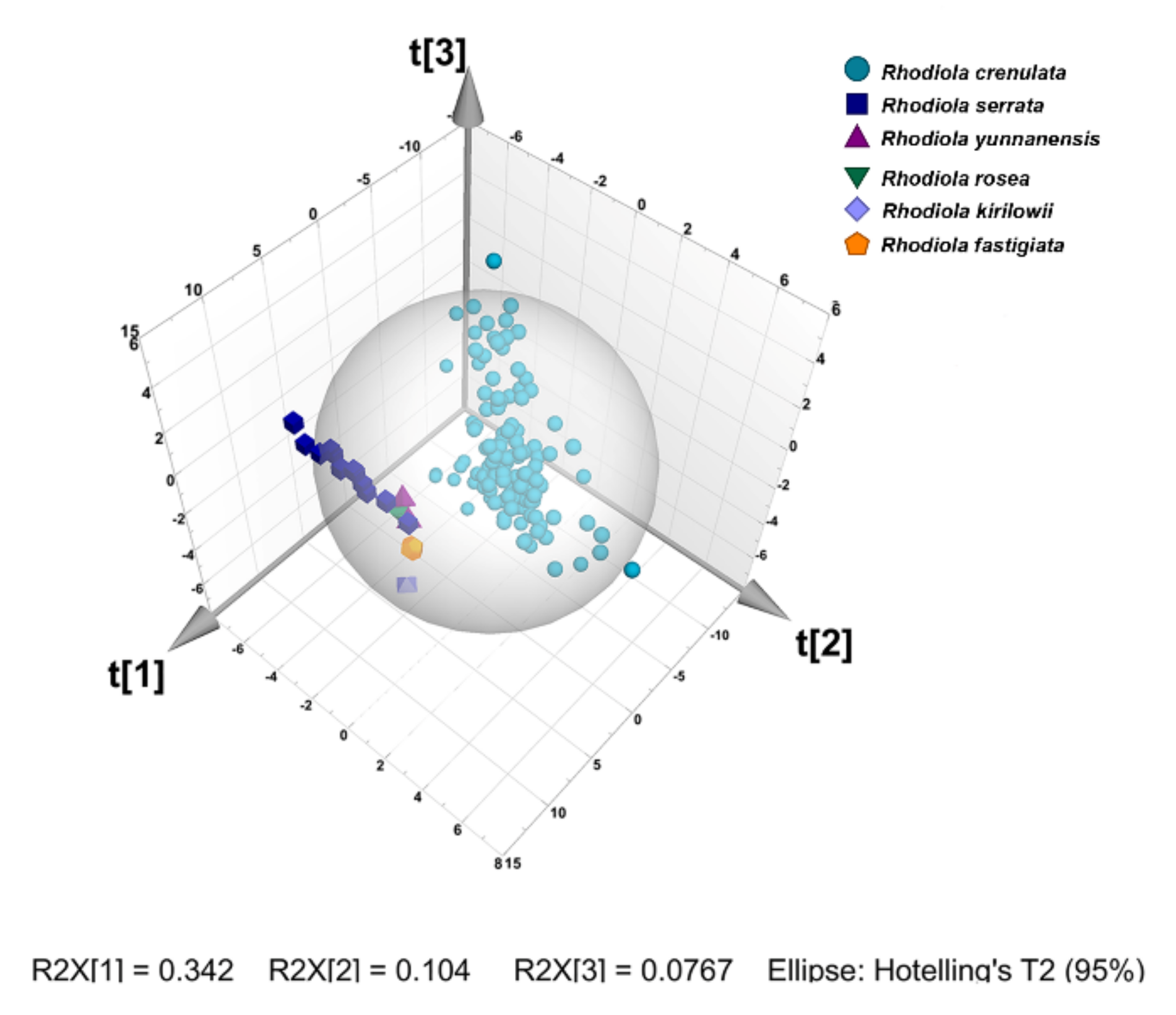
2.5.1. Principal Component Analysis
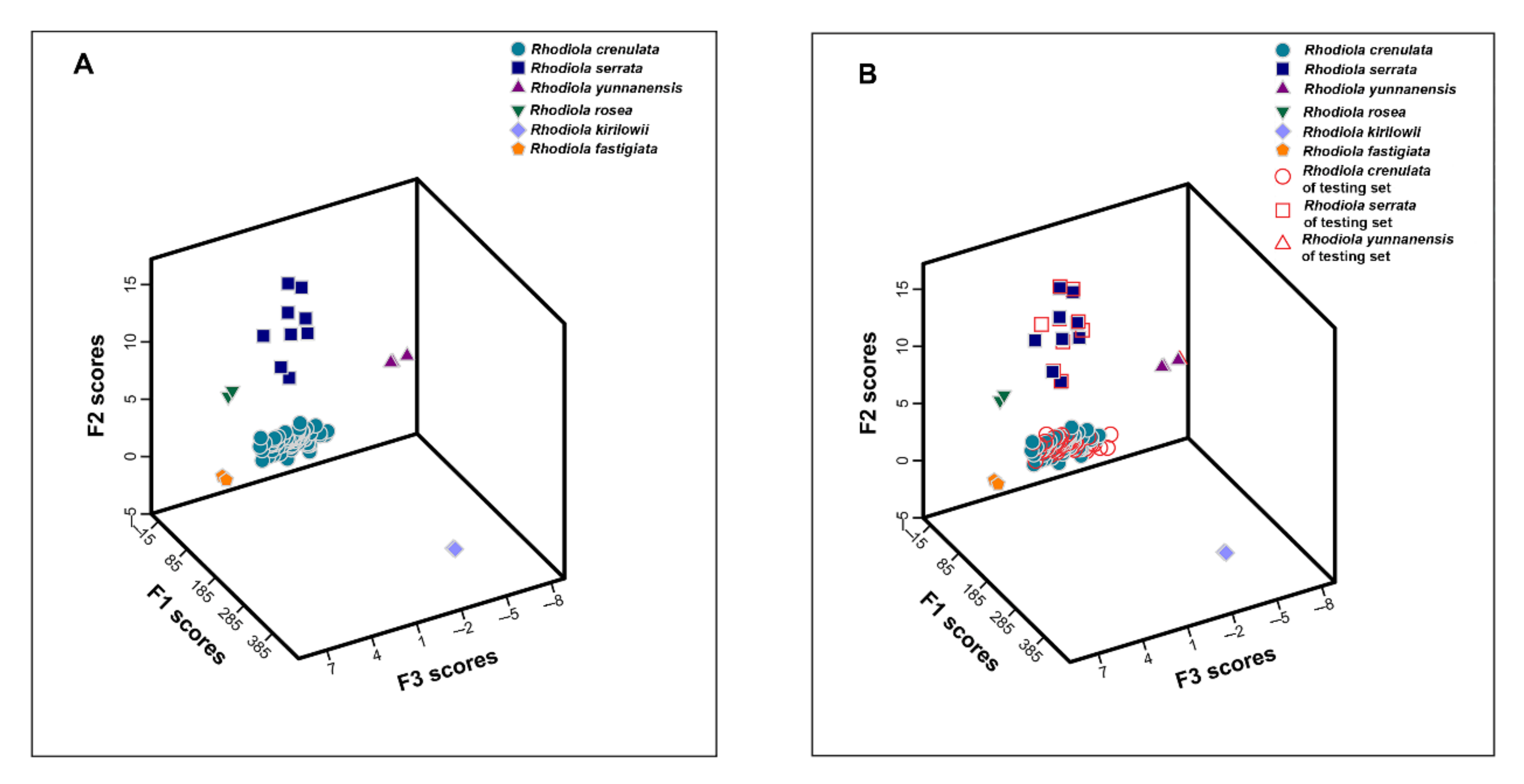
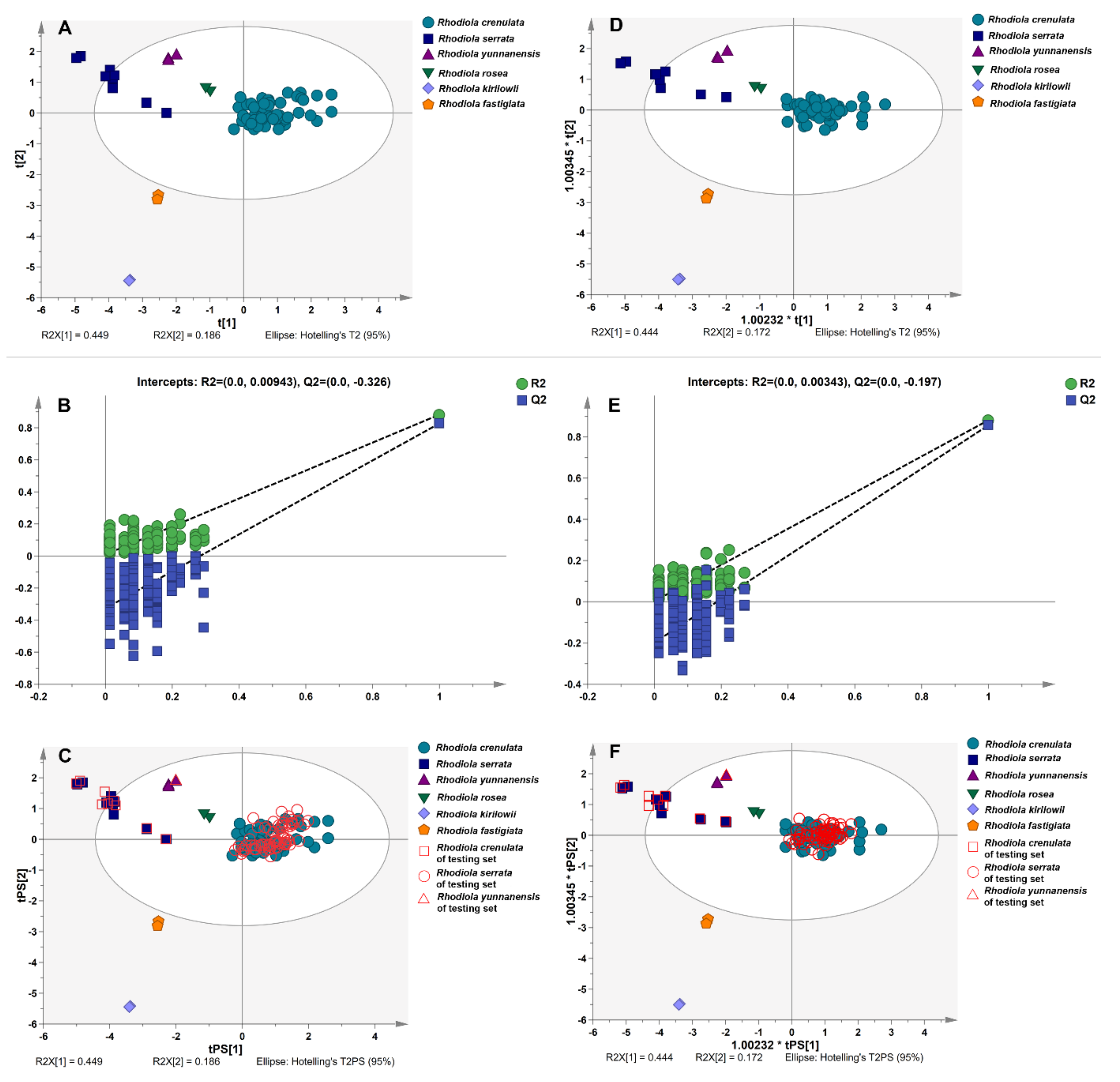
2.5.2. Linear Discriminant Analysis
2.5.3. Verification of Distinguishing Ability of Characteristic Variables
2.6. Identification of the Characteristic Peaks
3. Materials and Methods
3.1. Materials and Reagents
3.2. Apparatus and Conditions
3.3. Preparation of Sample Solutions
3.4. Data Analysis
3.4.1. Similarity Analysis
3.4.2. Chemical Pattern Recognition Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Tao, H.; Wu, X.; Cao, J.; Peng, Y.; Wang, A.; Pei, J.; Xiao, J.; Wang, S.; Wang, Y. Rhodiola species: A comprehensive review of traditional use, phytochemistry, pharmacology, toxicity, and clinical study. Med. Res. Rev. 2019, 39, 1779–1850. [Google Scholar] [CrossRef]
- The State Pharmacopoeia Commission of the PRC. Pharmacopoeia of the People’s Republic of China; The State Pharmacopoeia Commission of the PRC: Beijing, China, 2020; Volume I, pp. 161–162. [Google Scholar]
- Liao, Y.-H.; Chao, Y.-C.; Sim, B.Y.-Q.; Lin, H.-M.; Chen, M.-T.; Chen, C.-Y. Rhodiola/Cordyceps-Based Herbal Supplement Promotes Endurance Training-Improved Body Composition but Not Oxidative Stress and Metabolic Biomarkers: A Preliminary Randomized Controlled Study. Nutrients 2019, 11, 2357. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Pham, V.; Bui, M.; Song, L.; Wu, C.; Walia, A.; Uchio, E.; Smith-Liu, F.; Zi, X. Rhodiola rosea L.: An Herb with Anti-Stress, Anti-Aging, and Immunostimulating Properties for Cancer Chemoprevention. Curr. Pharmacol. Rep. 2017, 3, 384–395. [Google Scholar] [CrossRef]
- Panossian, A. Evidence-Based Efficacy of Adaptogens in Fatigue, and Molecular Mechanisms Related to their Stress-Protective Activity. Curr. Clin. Pharmacol. 2009, 4, 198–219. [Google Scholar] [CrossRef]
- Gansu Institute of Drug Control. Standardization of Traditional Chinese Medicine in Gansu Province; Gansu Institute of Drug Control: Gansu, China, 2009; pp. 103–106. [Google Scholar]
- Li, T.; Zhang, H. Application of microscopy in authentication of traditional Tibetan medicinal plants of fiveRhodiola (Crassulaceae) alpine species by comparative anatomy and micromorphology. Microsc. Res. Tech. 2008, 71, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, A.; Li, H.; Luo, P.; Zhao, W.; Long, X.; Brinckmann, J. There “ain’t no mountain high enough”? The drivers, diversity and sustainability of China’s Rhodiola trade. J. Ethnopharmacol. 2020, 252, 112379. [Google Scholar] [CrossRef] [PubMed]
- Xin, T.; Li, X.; Yao, H.; Lin, Y.; Ma, X.; Cheng, R.; Song, J.; Ni, L.; Fan, C.; Chen, S. Survey of commercial Rhodiola products revealed species diversity and potential safety issues. Sci. Rep. 2015, 5, 8337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; Liu, Y.; Liu, C.; Song, Z.; Li, Q.; Zha, Q.; Lu, C.; Wang, C.; Ning, Z.; Zhang, Y.; et al. The chemotaxonomic classification of Rhodiola plants and its correlation with morphological characteristics and genetic taxonomy. Chem. Cent. J. 2013, 7, 118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cui, S.; Hu, X.; Chen, X.; Hu, Z. Determination of p -tyrosol and salidroside in three samples of Rhodiola crenulata and one of Rhodiola kirilowii by capillary zone electrophoresis. Anal. Bioanal. Chem. 2003, 377, 370–374. [Google Scholar] [CrossRef]
- Linh, P.T.; Kim, Y.H.; Hong, S.P.; Jian, J.J.; Kang, J.S. Quantitative determination of salidroside and tyrosol from the underground part of Rhodiola rosea by high performance liquid chromatography. Arch. Pharmacal Res. 2000, 23, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Chen, W.; Wu, L.; Li, S.; Qi, Q.; Cui, Y.; Liang, L.; Ye, T.; Zhang, L. Quality Evaluation and Chemical Markers Screening of Salvia miltiorrhiza Bge. (Danshen) Based on HPLC Fingerprints and HPLC-MSn Coupled with Chemometrics. Molecules 2017, 22, 478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Wang, T.; Yin, G.; Wang, J.; Jiang, K.; Tu, J. High-performance liquid chromatography–based fingerprint analysis with chemical pattern recognition for evaluation of Mahonia Bealei (Fort.) Carr. J. Sep. Sci. 2020, 43, 3625–3635. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, K.; Wang, L.; Han, D.; Yin, G.; Wang, J.; Qin, B.; Li, S.; Wang, T. Identification of Salvia species using high-performance liquid chromatography combined with chemical pattern recognition analysis. J. Sep. Sci. 2018, 41, 609–617. [Google Scholar] [CrossRef]
- Meng, Z.; Huang, Y.; Wang, L.; Jiang, K.; Guo, L.; Wang, J.; Yin, G.; Wang, T. Quality evaluation of Panax Notoginseng using high-performance liquid chromatography with chemical pattern recognition. Sep. Sci. PLUS 2020, 3, 200–206. [Google Scholar] [CrossRef]
- Wei, W.; Wang, X.; Hou, J.; Yao, C.; Feng, Z.; Zhang, J.; Han, S.; Deng, Y.; Huang, Y.; Wu, W.; et al. Implementation of a Single Quadrupole Mass Spectrometer for Fingerprint Analysis: Venenum bufonis as a Case Study. Molecules 2018, 23, 3020. [Google Scholar] [CrossRef] [Green Version]
- Esteki, M.; Farajmand, B.; Amanifar, S.; Barkhordari, R.; Ahadiyan, Z.; Dashtaki, E.; Mohammadlou, M.; Heyden, Y.V. Classification and authentication of Iranian walnuts according to their geographical origin based on gas chromatographic fatty acid fingerprint analysis using pattern recognition methods. Chemom. Intell. Lab. Syst. 2017, 171, 251–258. [Google Scholar] [CrossRef]
- Cao, X.; Sun, L.; Li, D.; You, G.; Wang, M.; Ren, X. Quality Evaluation of Phellodendri Chinensis Cortex by Fingerprint–Chemical Pattern Recognition. Molecules 2018, 23, 2307. [Google Scholar] [CrossRef] [Green Version]
- Cheng, X.-M.; Zhao, T.; Yang, T.; Wang, C.-H.; Bligh, S.W.A.; Wang, Z.-T. HPLC fingerprints combined with principal component analysis, hierarchical cluster analysis and linear discriminant analysis for the classification and differentiation of Peganumsp. indigenous to China. Phytochem. Anal. 2010, 21, 279–289. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Hong, D.; Zeng, S.; Su, J.; Fan, G.; Zhang, Y. Metabolic Discrimination of Different Rhodiola Species Using 1H-NMR and GEP Combinational Chemometrics. Chem. Pharm. Bull. 2019, 67, 81–87. [Google Scholar] [CrossRef]
- Li, T.; Su, C. Authenticity identification and classification of Rhodiola species in traditional Tibetan medicine based on Fourier transform near-infrared spectroscopy and chemometrics analysis. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 204, 131–140. [Google Scholar] [CrossRef]
- Wu, X.-D.; Chen, H.-G.; Zhou, X.; Huang, Y.; Hu, E.-M.; Jiang, Z.-M.; Zhao, C.; Gong, X.-J.; Deng, Q.-F. Studies on Chromatographic Fingerprint and Fingerprinting Profile-Efficacy Relationship of Saxifraga stolonifera Meerb. Molecules 2015, 20, 22781–22798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.; Yin, D.; Li, N.; Hou, X.; Wang, D.; Li, D.; Liu, J. Influence of Environmental Factors on the Active Substance Production and Antioxidant Activity in Potentilla fruticosa L. and Its Quality Assessment. Sci. Rep. 2016, 6, 28591. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Liu, J.; Ma, Z.; Duan, F.; Guo, Z.; Xiao, H. Rapid identification of chemical constituents of Rhodiola Crenulata using liquid chromatography-mass spectrometry pseudotargeted analysis. J. Sep. Sci. 2021, 44, 3747–3776. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zeng, Q.; Contreras, M.D.M.; Wang, L. Profiling and quantification of phenolic compounds in Camellia seed oils: Natural tea polyphenols in vegetable oil. Food Res. Int. 2017, 102, 184–194. [Google Scholar] [CrossRef]
- Yang, L.-M.; Fu, H.-Z. Chemical constituents of Rhodiola kirilowii Maxim. J. Chin. Pharm. Sci. 2011, 20, 56–60. [Google Scholar] [CrossRef]
- Turrini, F.; Zunin, P.; Boggia, R. Potentialities of Rapid Analytical Strategies for the Identification of the Botanical Species of Several “Specialty” or “Gourmet” Oils. Foods 2021, 10, 183. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, C.-X.; Shao, C.-F.; Li, C.-W.; Liu, S.-H.; Peng, X.-P.; Xu, Y.-Q. Chemical Fingerprint Analysis for the Quality Evaluation of Deepure Instant Pu-erh Tea by HPLC Combined with Chemometrics. Food Anal. Methods 2016, 9, 3298–3309. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, H.; Wei, J.; Chen, X.; Sun, M.; Ouyang, H.; Hao, J.; Chang, Y.; Dou, Z.; He, J. Comparison of the Active Compositions between Raw and Processed Epimedium from Different Species. Molecules 2018, 23, 1656. [Google Scholar] [CrossRef] [Green Version]

| [M-H]- | ||||||
|---|---|---|---|---|---|---|
| Peak No. | Observed Mass (Da) | Error (ppm) | Formula | MS/MS | Identification | Type |
| 2 | 279.1091 | 0.9 | C11H20O8 | 117.0566[M-H-C6H10O5]−, 101.0242[M-H-C6H10O6]− | 1-(2-Hydroxy-2-methylbutanoate) β-D-glucopyranose | Acyclic acid glycoside |
| 4 | 325.0926 | −1 | C15H18O8 | 119.0500[M-H-Glu-CO2]−, 163.0399[M-H-Glu]− | 4-O-glucosyl-p-coumaric acid | Phenylpropanoid |
| 5 a | 299.1134 | −0.8 | C14H20O7 | 299.1155 [M-H]−, 179.0553 [Glu-H]−, 119.0498 [M-H-Glu-H2O]− | Salidroside | The phenethyl glycosides |
| 7 | 305.0665 | −0.3 | C15H14O7 | 221.0470[M-H-2C2H2O]−, 203.0331[M-H-2C2H2O-H2O]−, 179.0348[M-H-C6H6O3]−, 165.0348[M-H-C7H8O3]−, 137.0244[M-H-C8H8O4]−, 125.0247[M-H-C9H8O4]− | Epigallocatechin | Flavonoids |
| 13 | 457.0773 | −1.0 | C22H18O11 | 305.0667[M-H-C7H5O4]−, 287.0568[M-H-C7H5O4-H2O]−, 169.0133[M-H-C15H12O6]−, 125.0238 [M-H-C7H5O4-C9H8O4]− | Epigallocatechin gallate | Flavonoids |
| 36 a | 939.1112 | 0.3 | C41H32O26 | 939.1085[M-H]−, 769.0884[M-H-C7H6O5]−, 617.0785[M-H-C7H6O5-C7H4O4]−, 447.0578[M-H-2C7H6O5-C7H4O4]−, 169.0146[Galloy]− | 1,2,3,4,6-Pentagalloyglucose | Gallic acid derivative |
| 37 | 521.2028 | −0.1 | C26H34O11 | 491.1942[M-HCHO-H]−, 503.1883[M-H2O-H]−, 341.1383[M-Glu-H2O]− | (+)-isolarisiresinol-4′-O-β-D-glucopy ranoside or (+)-isolarisiresinol-4-O-β-D-glucopyranoside) | Phenylpropanoid |
| Sample No. | Species | Origin | Specifications |
|---|---|---|---|
| 1–47/68–115 | Rhodiola crenulata | Tibet | Processed drugs |
| 48–53/116–122 | Rhodiola crenulata | Sichuan | Processed drugs |
| 54–57/123/124 | Rhodiola crenulata | Xinjiang | Processed drugs |
| 58–61/125/126 | Rhodiola crenulata | Jilin | Processed drugs |
| 62–65/127/128 | Rhodiola crenulata | Qinghai | Processed drugs |
| 66 | Rhodiola crenulata | Inner Mongolia | Processed drugs |
| 67 | Rhodiola crenulata | Gansu | Processed drugs |
| 129 | Rhodiola crenulata | Yunnan | Processed drugs |
| 130 | Rhodiola crenulata | Guangxi | Processed drugs |
| 131 | Rhodiola crenulata | Liaoning | Processed drugs |
| 132/141 | Rhodiola serrata | Tibet | Processed drugs |
| 133/142 | Rhodiola serrata | Hunan | Processed drugs |
| 134/143 | Rhodiola serrata | Sichuan | Crude drugs |
| 135–140/144–149 | Rhodiola serrata | / | Crude drugs |
| 150–153 | Rhodiola yunnanensis | Tibet | Processed drugs |
| 154/155 | Rhodiola rosea | / | Crude drugs |
| 156/157 | Rhodiola kirilowii | / | Crude drugs |
| 158/159 | Rhodiola fastigiata | Tibet | Processed drugs |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, D.; Wang, L.; Jin, Y.; Gu, L.; Yu, X.; Xie, X.; Yin, G.; Wang, J.; Bi, K.; Lu, Y.; et al. Application of UHPLC Fingerprints Combined with Chemical Pattern Recognition Analysis in the Differentiation of Six Rhodiola Species. Molecules 2021, 26, 6855. https://doi.org/10.3390/molecules26226855
Ma D, Wang L, Jin Y, Gu L, Yu X, Xie X, Yin G, Wang J, Bi K, Lu Y, et al. Application of UHPLC Fingerprints Combined with Chemical Pattern Recognition Analysis in the Differentiation of Six Rhodiola Species. Molecules. 2021; 26(22):6855. https://doi.org/10.3390/molecules26226855
Chicago/Turabian StyleMa, Didi, Lijun Wang, Yibao Jin, Lifei Gu, Xiean Yu, Xueqing Xie, Guo Yin, Jue Wang, Kaishun Bi, Yi Lu, and et al. 2021. "Application of UHPLC Fingerprints Combined with Chemical Pattern Recognition Analysis in the Differentiation of Six Rhodiola Species" Molecules 26, no. 22: 6855. https://doi.org/10.3390/molecules26226855
APA StyleMa, D., Wang, L., Jin, Y., Gu, L., Yu, X., Xie, X., Yin, G., Wang, J., Bi, K., Lu, Y., & Wang, T. (2021). Application of UHPLC Fingerprints Combined with Chemical Pattern Recognition Analysis in the Differentiation of Six Rhodiola Species. Molecules, 26(22), 6855. https://doi.org/10.3390/molecules26226855






