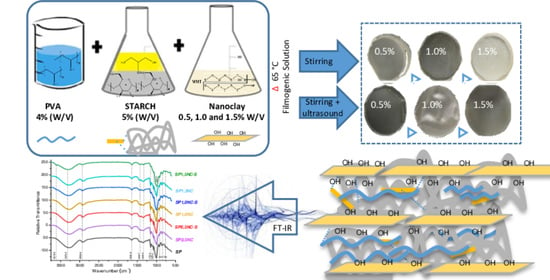
Physical-Mechanical Behavior and Water-Barrier Properties of Biopolymers-Clay Nanocomposites
Abstract
:1. Introduction
2. Results
2.1. Surface Morphology by Light Optical Microscopy
2.2. Surface Morphology by Scanning Electron Microscopy (SEM)
2.3. Opacity
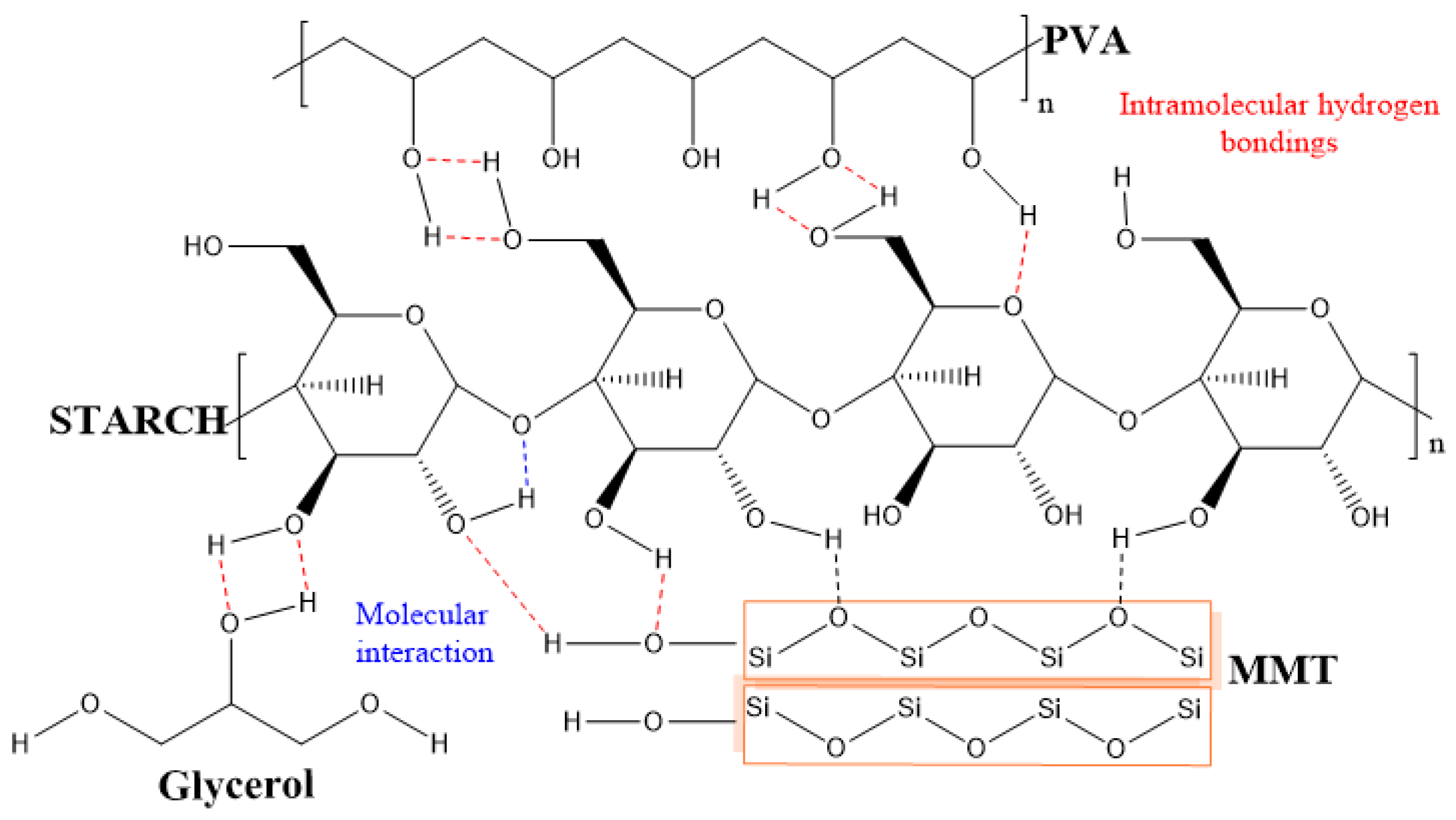
2.4. FTIR-ATR Analysis
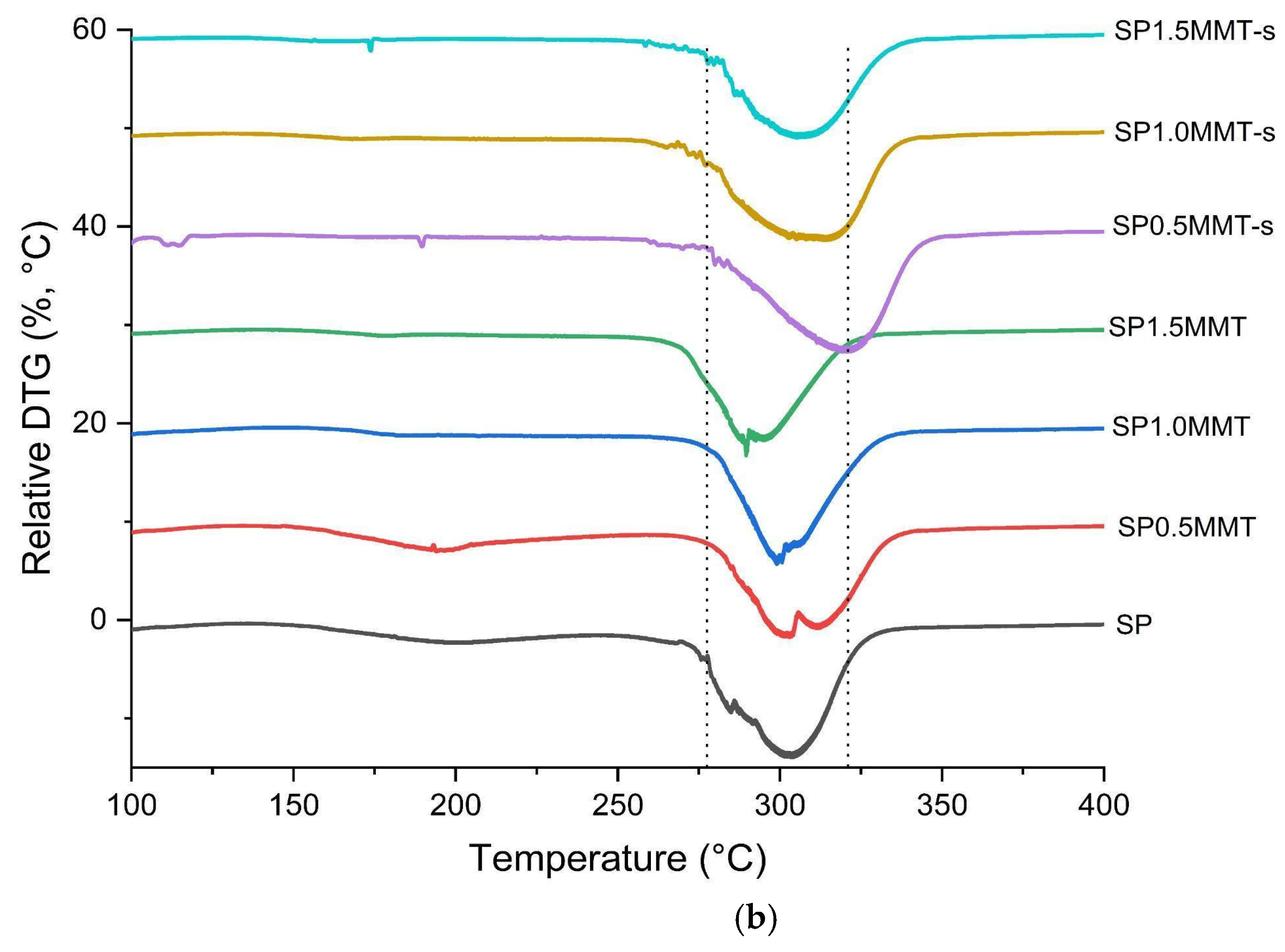
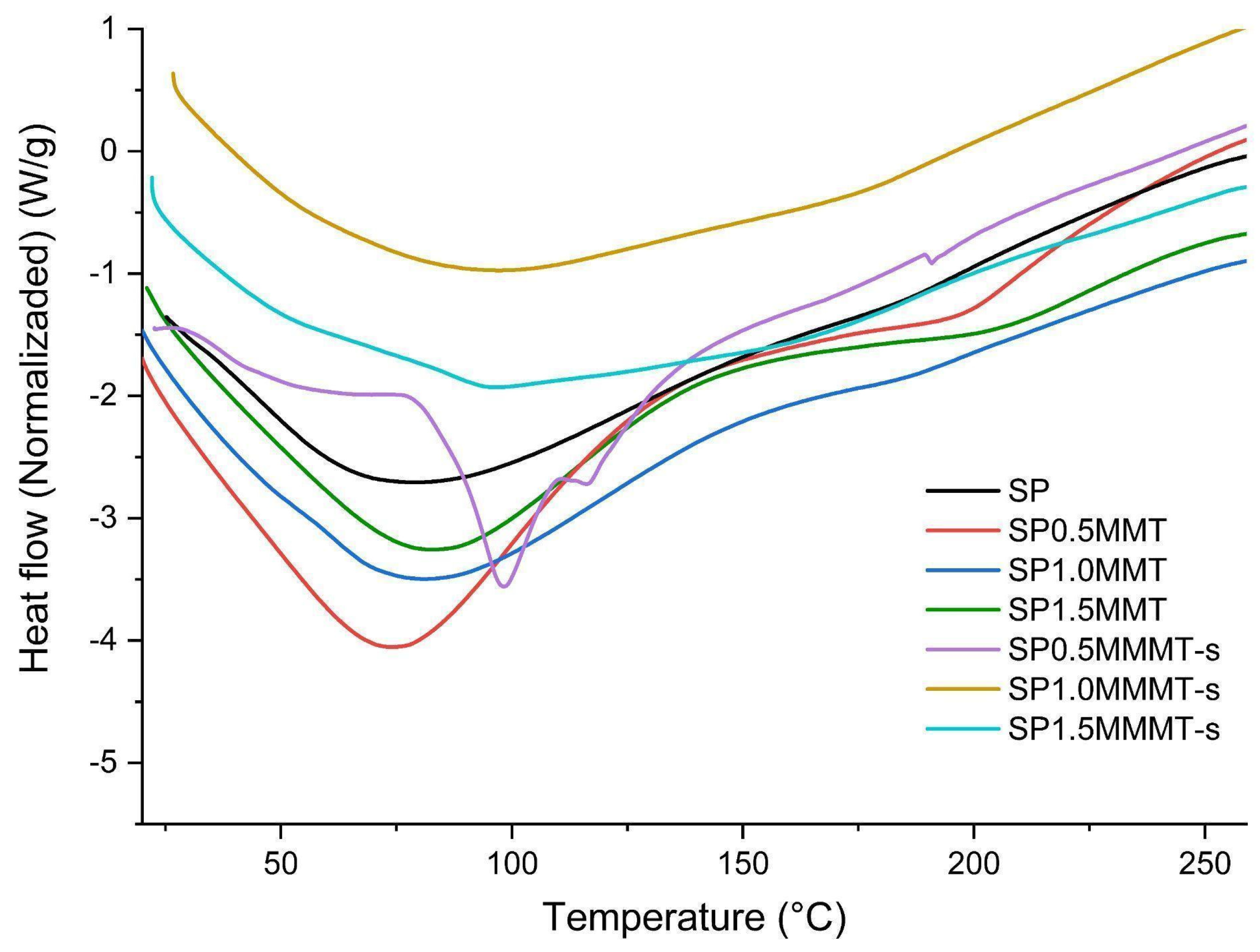
2.5. Thermal Analysis
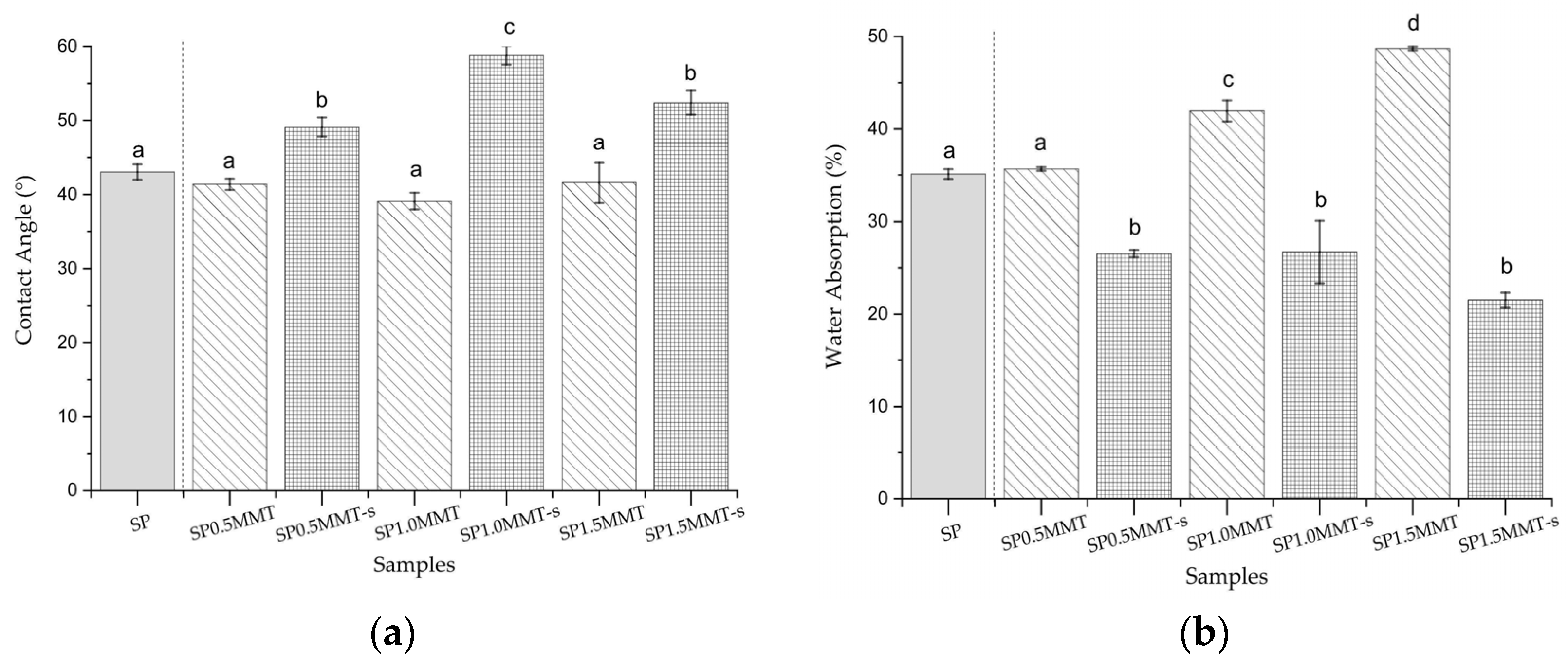
2.6. Contact Angle and Water Absorption
2.7. Water Vapor Permeability (WVP)
2.8. Mechanical Properties
3. Materials and Methods
3.1. Materials
3.2. Film Preparation
3.3. Characterization
3.3.1. Optical Microscopy
3.3.2. Scanning Electron Microscopy
3.3.3. Opacity
3.3.4. Fourier Transformed Infrared Spectroscopy (FT-IR)
3.3.5. Thermal Properties
3.3.6. Contact Angle
3.3.7. Water Absorption
3.3.8. Water Vapor Transmission Rate (WVTR) and Water Vapor Permeability (WVP)
3.3.9. Tensile Test
3.4. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Palai, B.; Biswal, M.; Mohanty, S.; Nayak, S.K. In situ reactive compatibilization of polylactic acid (PLA) and thermoplastic starch (TPS) blends; synthesis and evaluation of extrusion blown films thereof. Ind. Crop. Prod. 2019, 141, 111748. [Google Scholar] [CrossRef]
- Przybytek, A.; Sienkiewicz, M.; Kucińska-Lipka, J.; Janik, H. Preparation and characterization of biodegradable and compostable PLA/TPS/ESO compositions. Ind. Crop. Prod. 2018, 122, 375–383. [Google Scholar] [CrossRef]
- Ranganathan, S.; Dutta, S.; Moses, J.; Anandharamakrishnan, C. Utilization of food waste streams for the production of biopolymers. Heliyon 2020, 6, e04891. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.M.; Laurindo, J.B.; Yamashita, F. Composites of thermoplastic starch and nanoclays produced by extrusion and thermopressing. Carbohydr. Polym. 2012, 89, 504–510. [Google Scholar] [CrossRef] [Green Version]
- Ayana, B.; Suin, S.; Khatua, B. Highly exfoliated eco-friendly thermoplastic starch (TPS)/poly (lactic acid) (PLA)/clay nanocomposites using unmodified nanoclay. Carbohydr. Polym. 2014, 110, 430–439. [Google Scholar]
- Caicedo, C.; Pulgarin, H.L.C. Study of the Physical and Mechanical Properties of Thermoplastic Starch/Poly (Lactic Acid) Blends Modified with Acid Agents. Processes 2021, 9, 578. [Google Scholar] [CrossRef]
- Gómez-Aldapa, C.A.; Velazquez, G.; Gutierrez, M.C.; Rangel-Vargas, E.; Castro-Rosas, J.; Aguirre-Loredo, R.Y. Effect of polyvinyl alcohol on the physicochemical properties of biodegradable starch films. Mater. Chem. Phys. 2020, 239, 122027. [Google Scholar] [CrossRef]
- Cano, A.I.; Cháfer, M.; Chiralt, A.; González-Martínez, C. Physical and microstructural properties of biodegradable films based on pea starch and PVA. J. Food Eng. 2015, 167, 59–64. [Google Scholar] [CrossRef]
- Butron, A.; Llorente, O.; Fernandez, J.; Meaurio, E.; Sarasua, J.-R. Morphology and mechanical properties of poly (ethylene brassylate)/cellulose nanocrystal composites. Carbohydr. Polym. 2019, 221, 137–145. [Google Scholar] [CrossRef]
- Ren, J.; Dang, K.M.; Pollet, E.; Avérous, L. Preparation and characterization of thermoplastic potato starch/halloysite nano-biocomposites: Effect of plasticizer nature and nanoclay content. Polymers 2018, 10, 808. [Google Scholar] [CrossRef] [Green Version]
- Kwaśniewska, A.; Świetlicki, M.; Prószyński, A.; Gładyszewski, G. The quantitative nanomechanical mapping of starch/kaolin film surfaces by peak force AFM. Polymers 2021, 13, 244. [Google Scholar] [CrossRef]
- Kwaśniewska, A.; Chocyk, D.; Gładyszewski, G.; Borc, J.; Świetlicki, M.; Gładyszewska, B. The influence of kaolin clay on the mechanical properties and structure of thermoplastic starch films. Polymers 2020, 12, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaaz, T.S.; Sulong, A.B.; Akhtar, M.N.; Kadhum, A.A.H.; Mohamad, A.B.; Al-Amiery, A.A. Properties and applications of polyvinyl alcohol, halloysite nanotubes and their nanocomposites. Molecules 2015, 20, 22833–22847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müller, C.M.; Laurindo, J.B.; Yamashita, F. Effect of nanoclay incorporation method on mechanical and water vapor barrier properties of starch-based films. Ind. Crop. Prod. 2011, 33, 605–610. [Google Scholar] [CrossRef]
- Majdzadeh-Ardakani, K.; Nazari, B. Improving the mechanical properties of thermoplastic starch/poly (vinyl alcohol)/clay nanocomposites. Compos. Sci. Technol. 2010, 70, 1557–1563. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, H.; Jia, R.; Dai, Y.; Dong, H.; Hou, H.; Guo, Q. High performance extrusion blown starch/polyvinyl alcohol/clay nanocomposite films. Food Hydrocoll. 2018, 79, 534–543. [Google Scholar] [CrossRef]
- Dean, K.M.; Do, M.D.; Petinakis, E.; Yu, L. Key interactions in biodegradable thermoplastic starch/poly (vinyl alcohol)/montmorillonite micro-and nanocomposites. Compos. Sci. Technol. 2008, 68, 1453–1462. [Google Scholar] [CrossRef]
- Gao, W.; Dong, H.; Hou, H.; Zhang, H. Effects of clays with various hydrophilicities on properties of starch–clay nanocomposites by film blowing. Carbohydr. Polym. 2012, 88, 321–328. [Google Scholar] [CrossRef]
- Sorrentino, A.; Gorrasi, G.; Vittoria, V. Potential perspectives of bio-nanocomposites for food packaging applications. Trends Food Sci. Technol. 2007, 18, 84–95. [Google Scholar] [CrossRef]
- Wilhelm, H.-M.; Sierakowski, M.-R.; Souza, G.; Wypych, F. Starch films reinforced with mineral clay. Carbohydrate Polymers 2003, 52, 101–110. [Google Scholar] [CrossRef]
- Romero-Bastida, C.A.; Chávez Gutiérrez, M.; Bello-Pérez, L.A.; Abarca-Ramírez, E.; Velazquez, G.; Mendez-Montealvo, G. Rheological properties of nanocomposite-forming solutions and film based on montmorillonite and corn starch with different amylose content. Carbohydr. Polym. 2018, 188, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Chivrac, F.; Pollet, E.; Dole, P.; Avérous, L. Starch-based nano-biocomposites: Plasticizer impact on the montmorillonite exfoliation process. Carbohydr. Polym. 2010, 79, 941–947. [Google Scholar] [CrossRef]
- Schlemmer, D.; Angélica, R.S.; Sales, M.J.A. Morphological and thermomechanical characterization of thermoplastic starch/montmorillonite nanocomposites. Compos. Struct. 2010, 92, 2066–2070. [Google Scholar] [CrossRef]
- Kampeerapappun, P.; Aht-ong, D.; Pentrakoon, D.; Srikulkit, K. Preparation of cassava starch/montmorillonite composite film. Carbohydr. Polym. 2007, 67, 155–163. [Google Scholar] [CrossRef]
- Müller, P.; Kapin, É.; Fekete, E. Effects of preparation methods on the structure and mechanical properties of wet conditioned starch/montmorillonite nanocomposite films. Carbohydr. Polym. 2014, 113, 569–576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shayan, M.; Azizi, H.; Ghasemi, I.A.; Karrabi, M. Effect of modified starch and nanoclay particles on biodegradability and mechanical properties of cross-linked poly lactic acid. Carbohydr. Polym. 2015, 124, 237–244. [Google Scholar] [CrossRef]
- Guimarães, M., Jr.; Botaro, V.R.; Novack, K.M.; Teixeira, F.G.; Tonoli, G.H.D. Starch/PVA-based nanocomposites reinforced with bamboo nanofibrils. Ind. Crop. Prod. 2015, 70, 72–83. [Google Scholar] [CrossRef]
- Sin, L.T.; Rahman, W.; Rahmat, A.; Khan, M. Detection of synergistic interactions of polyvinyl alcohol–cassava starch blends through DSC. Carbohydr. Polym. 2010, 79, 224–226. [Google Scholar] [CrossRef]
- Majdzadeh-Ardakani, K.; Navarchian, A.H.; Sadeghi, F. Optimization of mechanical properties of thermoplastic starch/clay nanocomposites. Carbohydr. Polym. 2010, 79, 547–554. [Google Scholar] [CrossRef]
- Liu, G.; Song, Y.; Wang, J.; Zhuang, H.; Ma, L.; Li, C.; Liu, Y.; Zhang, J. Effects of nanoclay type on the physical and antimicrobial properties of PVOH-based nanocomposite films. LWT-Food Sci. Technol. 2014, 57, 562–568. [Google Scholar] [CrossRef]
- Kizil, R.; Irudayaraj, J.; Seetharaman, K. Characterization of irradiated starches by using FT-Raman and FTIR spectroscopy. J. Agric. Food Chem. 2002, 50, 3912–3918. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, J. Mechanical and thermal characteristics of pea starch films plasticized with monosaccharides and polyols. J. Food Sci. 2006, 71, E109–E118. [Google Scholar] [CrossRef]
- Liu, H.; Chaudhary, D.; Yusa, S.-I.; Tadé, M.O. Glycerol/starch/Na+-montmorillonite nanocomposites: A XRD, FTIR, DSC and 1H NMR study. Carbohydr. Polym. 2011, 83, 1591–1597. [Google Scholar] [CrossRef]
- Lam, C.K.; Lau, K.T.; Cheung, H.Y.; Ling, H.Y. Effect of ultrasound sonication in nanoclay clusters of nanoclay/epoxy composites. Mater. Lett. 2005, 59, 1369–1372. [Google Scholar] [CrossRef]
- Yoonessi, M.; Toghiani, H.; Kingery, W.L.; Pittman, C.U. Preparation, characterization, and properties of exfoliated/delaminated organically modified clay/dicyclopentadiene resin nanocomposites. Macromolecules 2004, 37, 2511–2518. [Google Scholar] [CrossRef]
- Yilmaz, O.; Cheaburu, C.; Durraccio, D.; Gulumser, G.; Vasile, C. Preparation of stable acrylate/montmorillonite nanocomposite latex via in situ batch emulsion polymerization: Effect of clay types. Appl. Clay Sci. 2010, 49, 288–297. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, Y.; Yi, H.; Chen, T.; Kang, S.; Li, H.; Song, S. Preparation and characterization of self-assembly hydrogels with exfoliated montmorillonite nanosheets and chitosan. Nanotechnology 2017, 29, 025605. [Google Scholar] [CrossRef] [PubMed]
- Alshabanat, M.; Al-Arrash, A.; Mekhamer, W. Polystyrene/montmorillonite nanocomposites: Study of the morphology and effects of sonication time on thermal stability. J. Nanomater. 2013, 2013, 9. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Wang, S.; Lan, W. Fabrication of antibacterial chitosan-PVA blended film using electrospray technique for food packaging applications. Int. J. Biol. Macromol. 2018, 107, 848–854. [Google Scholar] [CrossRef]
- García-López, D.; Fernández, J.F.; Merino, J.C.; Santarén, J.; Pastor, J.M. Effect of organic modification of sepiolite for PA 6 polymer/organoclay nanocomposites. Compos. Sci. Technol. 2010, 70, 1429–1436. [Google Scholar] [CrossRef] [Green Version]
- Sanchez-Olivares, G.; Calderas, F.; Sanchez-Solis, A.; Medina-Torres, L.; Moreno, L.R.; Manero, O. Assessment of extrusion-sonication process on flame retardant polypropylene by rheological characterization. AIMS Mater. Sci. 2016, 3, 620–633. [Google Scholar] [CrossRef]
- Sessini, V.; Arrieta, M.P.; Raquez, J.-M.; Dubois, P.; Kenny, J.M.; Peponi, L. Thermal and composting degradation of EVA/Thermoplastic starch blends and their nanocomposites. Polym. Degrad. Stab. 2019, 159, 184–198. [Google Scholar] [CrossRef]
- Giannakas, A.; Vlacha, M.; Salmas, C.; Leontiou, A.; Katapodis, P.; Stamatis, H.; Barkoula, N.-M.; Ladavos, A. Preparation, characterization, mechanical, barrier and antimicrobial properties of chitosan/PVOH/clay nanocomposites. Carbohydr. Polym. 2016, 140, 408–415. [Google Scholar] [CrossRef]
- Abdullah, Z.W.; Dong, Y. Biodegradable and water resistant poly (vinyl) alcohol (PVA)/starch (ST)/glycerol (GL)/halloysite nanotube (HNT) nanocomposite films for sustainable food packaging. Front. Mater. 2019, 6, 58. [Google Scholar] [CrossRef] [Green Version]
- Pandey, J.K.; Singh, R.P. Green nanocomposites from renewable resources: Effect of plasticizer on the structure and material properties of clay-filled starch. Starch-Stärke 2005, 57, 8–15. [Google Scholar] [CrossRef]
- Ojogbo, E.; Jardin, J.; Mekonnen, T.H. Robust and sustainable starch ester nanocomposite films for packaging applications. Ind. Crop. Prod. 2021, 160, 113153. [Google Scholar] [CrossRef]
- Tian, H.; Wang, K.; Liu, D.; Yan, J.; Xiang, A.; Rajulu, A.V. Enhanced mechanical and thermal properties of poly (vinyl alcohol)/corn starch blends by nanoclay intercalation. Int. J. Biol. Macromol. 2017, 101, 314–320. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E.; García, M.A.; Martino, M.N.; Zaritzky, N.E. Barrier, mechanical and optical properties of plasticized yam starch films. Carbohydr. Polym. 2004, 56, 129–135. [Google Scholar] [CrossRef]





| Sample | Opacity (AU × nm) | Pictures | Sample | Opacity (AU × nm) | Pictures |
|---|---|---|---|---|---|
| SP | 12.51 ± 0.02 a (at 400 nm) * |  | |||
| SP0.5MMT | 12.96 ± 0.09 a (at 510 nm) * |  | SP0.5MMT-s | 12.55 ± 0.09 a (at 510 nm) * |  |
| SP1.0MMT | 23.28 ± 0.12 b (at 590 nm) * |  | SP1.0MMT-s | 15.26 ± 0.08 c (at 590 nm) * |  |
| SP1.5MMT | 28.87 ± 0.17 b (at 590 nm) * |  | SP1.5MMT-s | 17.12 ± 0.11 c (at 590 nm) * |  |
| Sample | T10 | Ti | Td | Tf | Tgel | Tm (SP) | Tg (PVA) |
|---|---|---|---|---|---|---|---|
| (°C) | |||||||
| SP | 105.0 | 264.4 | 297.3 | 329.5 | 77.8 | -- | -- |
| SP0.5MMT | 85.3 | 271.7 | 300.3 | 336.1 | 74.5 | -- | 196.6 |
| SP1.0MMT | 97.2 | 268.8 | 295.9 | 335.5 | 81.1 | -- | 184.8 |
| SP1.5MMT | 122.5 | 268.1 | 292.3 | 334.7 | 82.6 | -- | 203.0 |
| SP0.5MMT-s | 140.6 | 269.5 | 306.2 | 342.8 | 98.1 | 116.4 | 191.3 |
| SP1.0MMT-s | 113.0 | 271.2 | 327.7 | 353.7 | 96.0 | -- | 182.8 |
| SP1.5MMT-s | 156.8 | 279.0 | 312.7 | 346.4 | 96.1 | -- | 170.9 |
| Film Sample | WVTR g·mm/(m2·day) | WVP g/(m2·day) |
|---|---|---|
| SP | 1073.2170 | 4658.290 |
| SP0.5MMT-s | 331.3667 | 3122.959 |
| SP1.0MMT-s | 736.2055 | 3675.283 |
| SP1.5MMT-s | 884.4438 | 4836.468 |
| Sample | TS (MPa) | ε (%) | YM (MPa) |
|---|---|---|---|
| SP | 3.73 ± 0.30 a | 39.45 ± 10.9 a | 0.179 ± 0.01 a |
| SP0.5MMT-s | 3.87 ± 0.32 a | 61.39 ± 9.6 b | 0.138 ± 0.02 a |
| SP1.0MMT-s | 4.89 ± 0.37 b | 33.16 ± 7.3 a | 0.360 ± 0.05 b |
| SP1.5MMT-s | 4.75 ± 0.25 b | 34.54 ± 4.2 a | 0.328 ± 0.03 b |
| Sample * | Starch (%) | PVA (%) | Nanoclay (%) | Mixing Method |
|---|---|---|---|---|
| SP (control) | 60 | 40 | 0 | Stir |
| SP0.5MMT | 60 | 40 | 0.5 | Stir |
| SP1.0MMT | 60 | 40 | 1.0 | Stir |
| SP1.5MMT | 60 | 40 | 1.5 | Stir |
| SP0.5MMT-s | 60 | 40 | 0.5 | Stir and sonication |
| SP1.0MMT-s | 60 | 40 | 1.0 | Stir and sonication |
| SP1.5MMT-s | 60 | 40 | 1.5 | Stir and sonication |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calambas, H.L.; Fonseca, A.; Adames, D.; Aguirre-Loredo, Y.; Caicedo, C. Physical-Mechanical Behavior and Water-Barrier Properties of Biopolymers-Clay Nanocomposites. Molecules 2021, 26, 6734. https://doi.org/10.3390/molecules26216734
Calambas HL, Fonseca A, Adames D, Aguirre-Loredo Y, Caicedo C. Physical-Mechanical Behavior and Water-Barrier Properties of Biopolymers-Clay Nanocomposites. Molecules. 2021; 26(21):6734. https://doi.org/10.3390/molecules26216734
Chicago/Turabian StyleCalambas, Heidy Lorena, Abril Fonseca, Dayana Adames, Yaneli Aguirre-Loredo, and Carolina Caicedo. 2021. "Physical-Mechanical Behavior and Water-Barrier Properties of Biopolymers-Clay Nanocomposites" Molecules 26, no. 21: 6734. https://doi.org/10.3390/molecules26216734
APA StyleCalambas, H. L., Fonseca, A., Adames, D., Aguirre-Loredo, Y., & Caicedo, C. (2021). Physical-Mechanical Behavior and Water-Barrier Properties of Biopolymers-Clay Nanocomposites. Molecules, 26(21), 6734. https://doi.org/10.3390/molecules26216734